Vitamin D Metabolism and Its Role in Mineral and Bone Disorders in Chronic Kidney Disease in Humans, Dogs and Cats
Abstract
1. Introduction
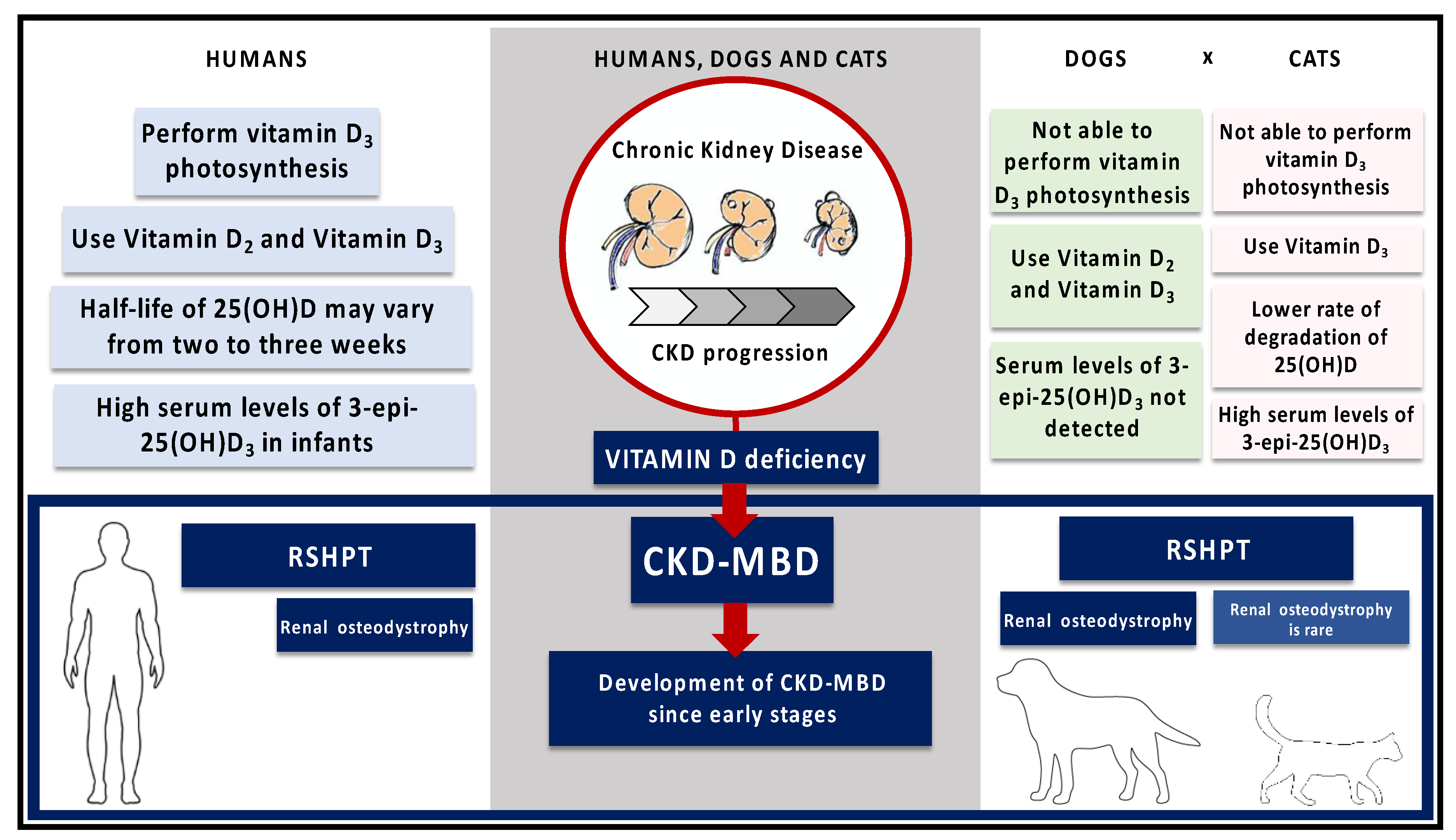
2. Chronic Kidney Disease in Humans, Dogs and Cats
3. Mineral Metabolism
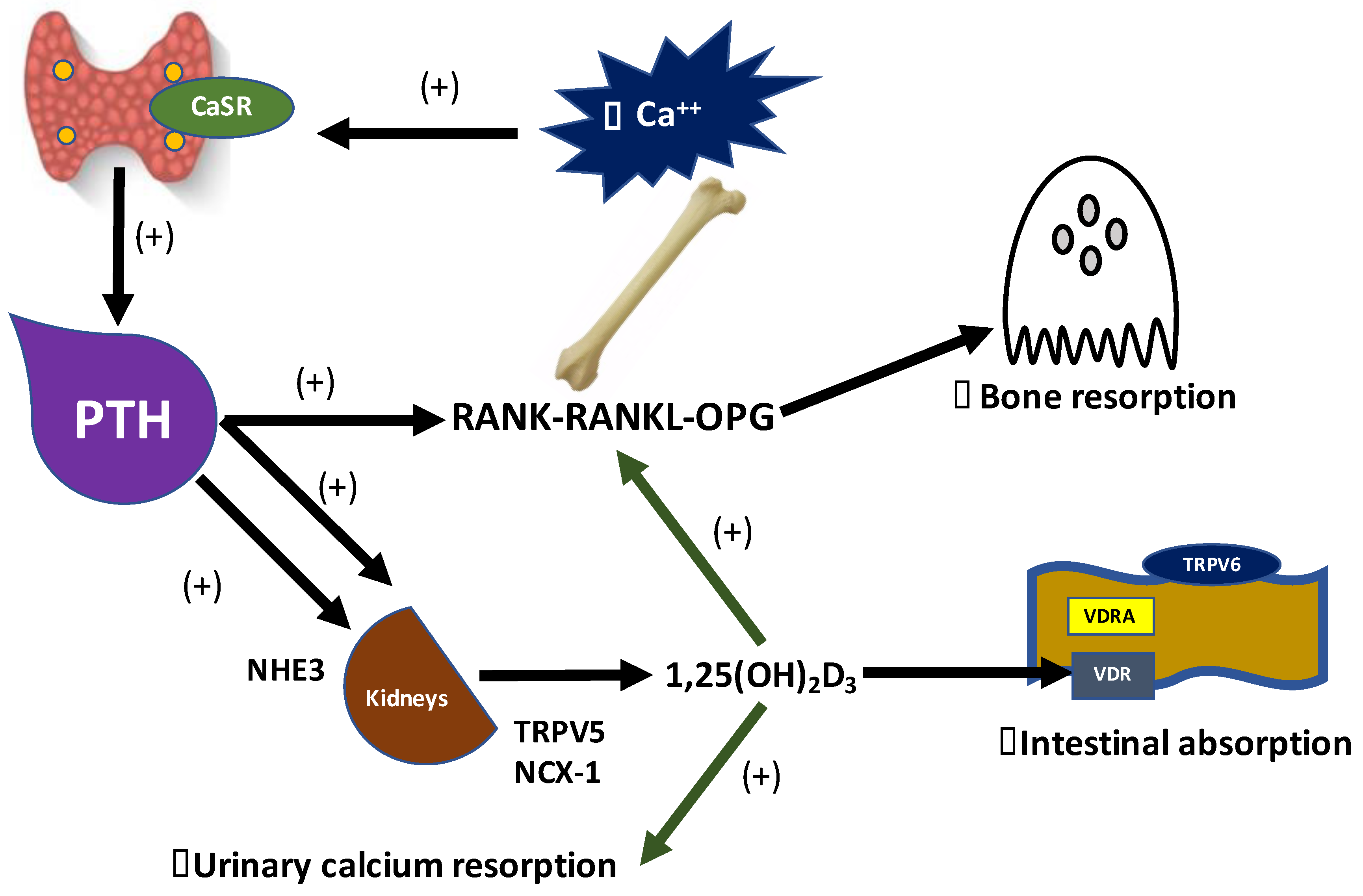
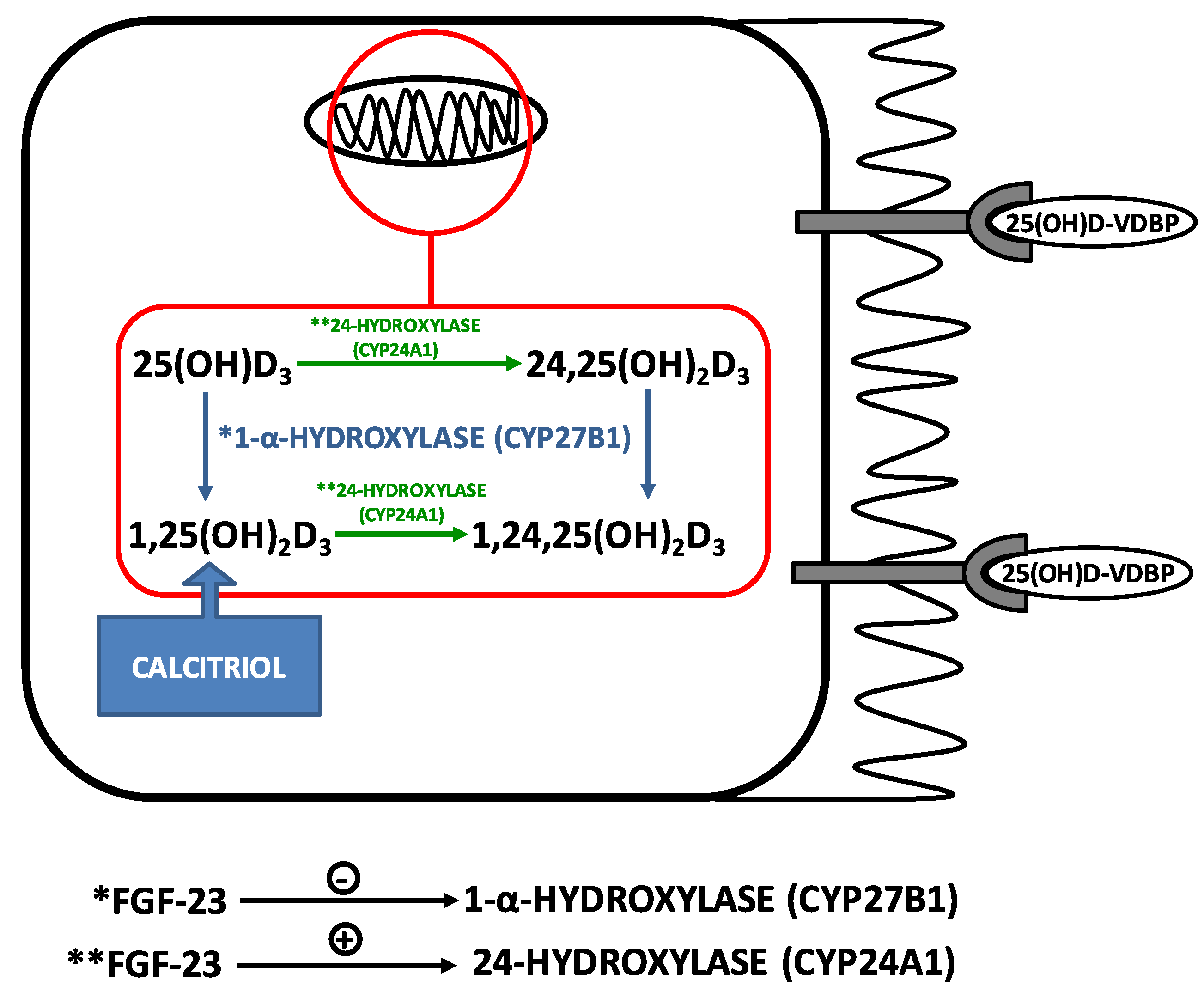
3.1. Vitamin D and PTH: The Main Regulators of Calcium Homeostasis
3.2. Diet and Hormonal Control in Phosphorus Metabolism
4. Mineral and Bone Disorders in Chronic Kidney Disease in Humans, Dogs and Cats—The Role of Vitamin D
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Levey, A.S.; Coresh, J. Chronic kidney disease. Lancet 2012, 379, 165–180. [Google Scholar] [CrossRef]
- Webster, A.C.; Nagler, E.V.; Morton, R.L.; Masson, P. Chronic Kidney Disease. Lancet 2017, 389, 1238–1252. [Google Scholar] [CrossRef]
- Jha, V.; Garcia-Garcia, G.; Iseki, K.; Li, Z.; Naicker, S.; Plattner, B.; Saran, R.; Wang, A.Y.-M.; Yang, C.-W. Chronic kidney disease: Global dimension and perspectives. Lancet 2013, 382, 260–272. [Google Scholar] [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group KDIGO 2017 Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease–Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 2017, 7, 60.
- Bartges, J.W. Chronic Kidney Disease in Dogs and Cats. Vet. Clin. N. Am. Small Anim. Pract. 2012, 42, 669–692. [Google Scholar] [CrossRef]
- Polzin, D.J. Chronic Kidney Disease in Small Animals. Vet. Clin. N. Am. Small Anim. Pract. 2011, 41, 15–30. [Google Scholar] [CrossRef]
- Brown, S.A. Renal pathophysiology: Lessons learned from the canine remnant kidney model: Canine remnant kidney model. J. Vet. Emerg. Crit. Care 2013, 23, 115–121. [Google Scholar] [CrossRef]
- Wen, S.-F.; Stoll, R.W. Renal Phosphate Adaptation in Uraemic Dogs with a Remnant Kidney. Clin. Sci. 1981, 60, 273–282. [Google Scholar] [CrossRef]
- Finco, D.R.; Brown, S.A.; Brown, C.A.; Crowell, W.A.; Cooper, T.A.; Barsanti, J.A. Progression of Chronic Renal Disease in the Dog. J. Vet. Intern. Med. 1999, 13, 516–528. [Google Scholar] [CrossRef]
- Fine, A. Remnant Kidney Metabolism in the Dog. J. Am. Soc. Nephrol. 1991, 2, 70–76. [Google Scholar]
- Brown, S.A.; Brown, C.A. Single-nephron adaptations to partial renal ablation in cats. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 1995, 269, R1002–R1008. [Google Scholar] [CrossRef]
- Watanabe, T.; Mishina, M. Effects of Benazepril Hydrochloride in Cats with Experimentally Induced or Spontaneously Occurring Chronic Renal Failure. J. Vet. Med. Sci. 2007, 69, 1015–1023. [Google Scholar] [CrossRef]
- Mathur, S.; Brown, C.A.; Dietrich, U.M.; Munday, J.S.; Newell, M.A.; Sheldon, S.E.; Cartier, L.M.; Brown, S.A. Evaluation of a technique of inducing hypertensive renal insufficiency in cats. Am. J. Vet. Res. 2004, 65, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Lawson, J.S.; Liu, H.-H.; Syme, H.M.; Purcell, R.; Wheeler-Jones, C.P.D.; Elliott, J. The cat as a naturally occurring model of renal interstitial fibrosis: Characterisation of primary feline proximal tubular epithelial cells and comparative pro-fibrotic effects of TGF-β1. PLoS ONE 2018, 13, e0202577. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, S.; Syme, H.M.; Brown, C.A.; Elliott, J. Histomorphometry of Feline Chronic Kidney Disease and Correlation with Markers of Renal Dysfunction. Vet. Pathol. 2013, 50, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Hill, N.R.; Fatoba, S.T.; Oke, J.L.; Hirst, J.A.; O’Callaghan, C.A.; Lasserson, D.S.; Hobbs, F.D.R. Global Prevalence of Chronic Kidney Disease—A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0158765. [Google Scholar] [CrossRef]
- Zhang, Q.-L.; Rothenbacher, D. Prevalence of chronic kidney disease in population-based studies: Systematic review. BMC Public Health 2008, 8, 117. [Google Scholar] [CrossRef]
- Coresh, J.; Selvin, E.; Stevens, L.A.; Manzi, J.; Kusek, J.W.; Eggers, P.; Levey, A.S. Prevalence of Chronic Kidney Disease in the United States. J. Am. Med. Assoc. 2007, 298, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Prevalence of chronic renal disease in Spain: Results of the EPIRCE study. Nefrología 2010, 30, 78–86.
- Stevens, L.A.; Li, S.; Wang, C.; Huang, C.; Becker, B.N.; Bomback, A.S.; Brown, W.W.; Burrows, N.R.; Jurkovitz, C.T.; McFarlane, S.I.; et al. Prevalence of CKD and Comorbid Illness in Elderly Patients in the United States: Results From the Kidney Early Evaluation Program (KEEP). Am. J. Kidney Dis. 2010, 55, S23–S33. [Google Scholar] [CrossRef]
- Rudinsky, A.J.; Harjes, L.M.; Byron, J.; Chew, D.J.; Toribio, R.E.; Langston, C.; Parker, V.J. Factors associated with survival in dogs with chronic kidney disease. J. Vet. Intern. Med. 2018, 32, 1977–1982. [Google Scholar] [CrossRef] [PubMed]
- Pedrinelli, V.; Lima, D.M.; Duarte, C.N.; Teixeira, F.A.; Porsani, M.; Zarif, C.; Amaral, A.R.; Vendramini, T.H.A.; Kogika, M.M.; Brunetto, M.A. Nutritional and laboratory parameters affect the survival of dogs with chronic kidney disease. PLoS ONE 2020, 15, e0234712. [Google Scholar] [CrossRef] [PubMed]
- Boyd, L.M.; Langston, C.; Thompson, K.; Zivin, K.; Imanishi, M. Survival in Cats with Naturally Occurring Chronic Kidney Disease (2000–2002). J. Vet. Intern. Med. 2008, 22, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- Greene, J.P.; Lefebvre, S.L.; Wang, M.; Yang, M.; Lund, E.M.; Polzin, D.J. Risk factors associated with the development of chronic kidney disease in cats evaluated at primary care veterinary hospitals. J. Am. Vet. Med. Assoc. 2014, 244, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Finch, N.C.; Syme, H.M.; Elliott, J. Risk Factors for Development of Chronic Kidney Disease in Cats. J. Vet. Intern. Med. 2016, 30, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Hokamp, J.A.; Cianciolo, R.E.; Boggess, M.; Lees, G.E.; Benali, S.L.; Kovarsky, M.; Nabity, M.B. Correlation of Urine and Serum Biomarkers with Renal Damage and Survival in Dogs with Naturally Occurring Proteinuric Chronic Kidney Disease. J. Vet. Intern. Med. 2016, 30, 591–601. [Google Scholar] [CrossRef]
- King, J.N.; Tasker, S.; Gunn-Moore, D.A.; Strehlau, G. Prognostic Factors in Cats with Chronic Kidney Disease. J. Vet. Intern. Med. 2007, 21, 906–916. [Google Scholar] [CrossRef]
- Kain, R.; Pagitz, M. Chronic Kidney Failure Affects Humans and Other Mammalians. In Comparative Medicine; Jensen-Jarolim, E., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 27–46. ISBN 978-3-319-47005-4. [Google Scholar]
- Luyckx, V.A.; Tuttle, K.R.; Garcia-Garcia, G.; Gharbi, M.B.; Heerspink, H.J.L.; Johnson, D.W.; Liu, Z.-H.; Massy, Z.A.; Moe, O.; Nelson, R.G.; et al. Reducing major risk factors for chronic kidney disease. Kidney Int. Suppl. 2017, 7, 71–87. [Google Scholar] [CrossRef]
- Hall, J.; Juncos, L.; Wang, Z.; Hall, M.; do Carmo, J.; da Silva, A. Obesity, hypertension, and chronic kidney disease. Int. J. Nephrol. Renov. Dis. 2014, 7, 75–88. [Google Scholar] [CrossRef]
- Parikh, N.I.; Hwang, S.-J.; Larson, M.G.; Meigs, J.B.; Levy, D.; Fox, C.S. Cardiovascular Disease Risk Factors in Chronic Kidney Disease: Overall Burden and Rates of Treatment and Control. Arch. Intern. Med. 2006, 166, 1884–1891. [Google Scholar] [CrossRef]
- Muntner, P.; Judd, S.E.; Gao, L.; Gutiérrez, O.M.; Rizk, D.V.; McClellan, W.; Cushman, M.; Warnock, D.G. Cardiovascular Risk Factors in CKD Associate with Both ESRD and Mortality. J. Am. Soc. Nephrol. 2013, 24, 1159–1165. [Google Scholar] [CrossRef] [PubMed]
- Kazancioğlu, R. Risk factors for chronic kidney disease: An update. Kidney Int. Suppl. 2013, 3, 368–371. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, B.S.; Lefebvre, H.P. Feline CKD: Pathophysiology and risk factors—What do we know? J. Feline Med. Surg. 2013, 15, 3–14. [Google Scholar] [CrossRef]
- Bird, L.; Walker, D. Pathophysiology of chronic kidney disease. Companion Anim. 2015, 20, 15–19. [Google Scholar] [CrossRef]
- Cianciolo, R.E.; Benali, S.L.; Aresu, L. Aging in the Canine Kidney. Vet. Pathol. 2016, 53, 299–308. [Google Scholar] [CrossRef]
- Brown, C.A.; Elliott, J.; Schmiedt, C.W.; Brown, S.A. Chronic Kidney Disease in Aged Cats: Clinical Features, Morphology, and Proposed Pathogeneses. Vet. Pathol. 2016, 53, 309–326. [Google Scholar] [CrossRef]
- Day, M.J. Ageing, Immunosenescence and Inflammageing in the Dog and Cat. J. Comp. Pathol. 2010, 142, S60–S69. [Google Scholar] [CrossRef]
- Kuro-o, M. Klotho and the Aging Process. Korean J. Intern. Med. 2011, 26, 113–122. [Google Scholar] [CrossRef]
- Stenvinkel, P.; Painer, J.; Kuro-o, M.; Lanaspa, M.; Arnold, W.; Ruf, T.; Shiels, P.G.; Johnson, R.J. Novel treatment strategies for chronic kidney disease: Insights from the animal kingdom. Nat. Rev. Nephrol. 2018, 14, 265–284. [Google Scholar] [CrossRef]
- John, G.B.; Cheng, C.-Y.; Kuro-o, M. Role of Klotho in Aging, Phosphate Metabolism, and CKD. Am. J. Kidney Dis. 2011, 58, 127–134. [Google Scholar] [CrossRef]
- Ohara, Y.; Yabuki, A.; Nakamura, R.; Ichii, O.; Mizukawa, H.; Yokoyama, N.; Yamato, O. Renal Infiltration of Macrophages in Canine and Feline Chronic Kidney Disease. J. Comp. Pathol. 2019, 170, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Yabuki, A.; Mitani, S.; Fujiki, M.; Misumi, K.; Endo, Y.; Miyoshi, N.; Yamato, O. Comparative study of chronic kidney disease in dogs and cats: Induction of myofibroblasts. Res. Vet. Sci. 2010, 88, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Yabuki, A.; Mitani, S.; Sawa, M.; Mizukami, K.; Fujiki, M.; Yamato, O. A comparative study of chronic kidney disease in dogs and cats: Induction of cyclooxygenases. Res. Vet. Sci. 2012, 93, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Kuro-o, M. Klotho as a regulator of oxidative stress and senescence. Biol. Chem. 2008, 389, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, S.; Combet, E.; Stenvinkel, P.; Shiels, P.G. Klotho, Aging, and the Failing Kidney. Front. Endocrinol. 2020, 11, 560. [Google Scholar] [CrossRef]
- Kuro-o, M. A potential link between phosphate and aging—Lessons from Klotho-deficient mice. Mech. Ageing Dev. 2010, 131, 270–275. [Google Scholar] [CrossRef]
- Kuro-o, M.; Matsumura, Y.; Aizawa, H.; Kawaguchi, H.; Suga, T.; Utsugi, T.; Ohyama, Y.; Kurabayashi, M.; Kaname, T.; Kume, E.; et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 1997, 390, 45–51. [Google Scholar] [CrossRef]
- Hu, M.C.; Shi, M.; Zhang, J.; Quiñones, H.; Griffith, C.; Kuro-o, M.; Moe, O.W. Klotho Deficiency Causes Vascular Calcification in Chronic Kidney Disease. J. Am. Soc. Nephrol. 2011, 22, 124–136. [Google Scholar] [CrossRef]
- Kuro-o, M. Phosphate and Klotho. Kidney Int. 2011, 79, S20–S23. [Google Scholar] [CrossRef]
- jae Yi, H.; bok Lee, J.; pil Lee, K.; Oh, Y.-I.; ho Song, K.; won Seo, K. Investigation on urinary and serum alpha klotho in dogs with chronic kidney disease. BMC Vet. Res. 2020, 16, 246. [Google Scholar]
- Hu, M.C.; Kuro-o, M.; Moe, O.W. Secreted Klotho and Chronic Kidney Disease. In Endocrine FGFs and Klothos; Kuro-o, M., Ed.; Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2012; Volume 728, pp. 126–157. ISBN 978-1-4614-0886-4. [Google Scholar]
- Imura, A.; Iwano, A.; Tohyama, O.; Tsuji, Y.; Nozaki, K.; Hashimoto, N.; Fujimori, T.; Nabeshima, Y.-I. Secreted Klotho protein in sera and CSF: Implication for post-translational cleavage in release of Klotho protein from cell membrane. FEBS Lett. 2004, 565, 143–147. [Google Scholar] [CrossRef]
- Donate-Correa, J.; Muros-de-Fuentes, M.; Mora-Fernández, C.; Navarro-González, J.F. FGF23/Klotho axis: Phosphorus, mineral metabolism and beyond. Cytokine Growth Factor Rev. 2012, 23, 37–46. [Google Scholar] [CrossRef]
- Kuro-o, M. Klotho, phosphate and FGF-23 in ageing and disturbed mineral metabolism. Nat. Rev. Nephrol. 2013, 9, 650–660. [Google Scholar] [CrossRef]
- Van den Broek, D.H.N.; Lawson, J.S.; Chang, R.; Elliott, J.; Jepson, R.E. Immunohistochemical Staining of Alpha-Klotho Protein in Feline Kidney Tissue. J. Vet. Intern. Med. 2018, 32, 561–562. [Google Scholar]
- Isakova, T.; Wahl, P.; Vargas, G.S.; Gutiérrez, O.M.; Scialla, J.; Xie, H.; Appleby, D.; Nessel, L.; Bellovich, K.; Chen, J.; et al. Fibroblast growth factor 23 is elevated before parathyroid hormone and phosphate in chronic kidney disease. Kidney Int. 2011, 79, 1370–1378. [Google Scholar] [CrossRef]
- Finch, N.C.; Geddes, R.F.; Syme, H.M.; Elliott, J. Fibroblast Growth Factor 23 (FGF-23) Concentrations in Cats with Early Nonazotemic Chronic Kidney Disease (CKD) and in Healthy Geriatric Cats. J. Vet. Intern. Med. 2013, 27, 227–233. [Google Scholar] [CrossRef]
- Parker, V.J.; Harjes, L.M.; Dembek, K.; Young, G.S.; Chew, D.J.; Toribio, R.E. Association of Vitamin D Metabolites with Parathyroid Hormone, Fibroblast Growth Factor-23, Calcium, and Phosphorus in Dogs with Various Stages of Chronic Kidney Disease. J. Vet. Intern. Med. 2017, 31, 791–798. [Google Scholar] [CrossRef]
- Salusky, I.B. Are new vitamin D analogues in renal bone disease superior to calcitriol? Pediatr. Nephrol. 2005, 20, 393–398. [Google Scholar] [CrossRef]
- Kovesdy, C.P. Association of Activated Vitamin D Treatment and Mortality in Chronic Kidney Disease. Arch. Intern. Med. 2008, 168, 397–403. [Google Scholar] [CrossRef]
- Palmer, S.C.; McGregor, D.O.; Macaskill, P.; Craig, J.C.; Elder, G.J.; Strippoli, G.F.M. Meta-analysis: Vitamin D Compounds in Chronic Kidney Disease. Ann. Intern. Med. 2007, 147, 840. [Google Scholar] [CrossRef]
- Cunningham, J. New vitamin D analogues for osteodystrophy in chronic kidney disease. Pediatr. Nephrol. 2004, 19, 705–708. [Google Scholar] [CrossRef]
- Levin, A.; Li, Y.A.N.C. Vitamin D and its analogues: Do they protect against cardiovascular disease in patients with kidney disease? Kidney Int. 2005, 68, 1973–1981. [Google Scholar] [CrossRef]
- Zheng, Z.; Shi, H.; Jia, J.; Li, D.; Lin, S. Vitamin D supplementation and mortality risk in chronic kidney disease: A meta-analysis of 20 observational studies. BMC Nephrol. 2013, 14, 199. [Google Scholar] [CrossRef]
- Bhan, I.; Thadhani, R. Vitamin D Therapy for Chronic Kidney Disease. Semin. Nephrol. 2009, 29, 85–93. [Google Scholar] [CrossRef]
- Nagode, L.A.; Chew, D.J.; Podell, M. Benefits of Calcitriol Therapy and Serum Phosphorus Control in Dogs and Cats with Chronic Renal Failure. Vet. Clin. N. Am. Small Anim. Pract. 1996, 26, 1293–1330. [Google Scholar] [CrossRef]
- Goodman, W.G. Calcium-sensing receptors. Semin. Nephrol. 2004, 24, 17–24. [Google Scholar] [CrossRef]
- Copp, D.H. Endocrine Regulation of Calcium Metabolism. Annu. Rev. Physiol. 1970, 32, 61–86. [Google Scholar] [CrossRef]
- de Brito Galvao, J.F.; Nagode, L.A.; Schenck, P.A.; Chew, D.J. Calcitriol, calcidiol, parathyroid hormone, and fibroblast growth factor-23 interactions in chronic kidney disease: Calcitriol, calcidiol, PTH, and FGF-23 in CKD. J. Vet. Emerg. Crit. Care 2013, 23, 134–162. [Google Scholar] [CrossRef]
- Peacock, M. Calcium Metabolism in Health and Disease. Clin. J. Am. Soc. Nephrol. 2010, 5, S23–S30. [Google Scholar] [CrossRef]
- Christakos, S.; Dhawan, P.; Porta, A.; Mady, L.J.; Seth, T. Vitamin D and intestinal calcium absorption. Mol. Cell. Endocrinol. 2011, 347, 25–29. [Google Scholar] [CrossRef]
- Christakos, S.; Ajibade, D.V.; Dhawan, P.; Fechner, A.J.; Mady, L.J. Vitamin D: Metabolism. Endocrinol. Metab. Clin. N. Am. 2010, 39, 243–253. [Google Scholar] [CrossRef]
- Toka, H.R.; Pollak, M.R.; Houillier, P. Calcium Sensing in the Renal Tubule. Physiology 2015, 30, 317–326. [Google Scholar] [CrossRef]
- Moor, M.B.; Bonny, O. Ways of calcium reabsorption in the kidney. Am. J. Physiol.-Ren. Physiol. 2016, 310, F1337–F1350. [Google Scholar] [CrossRef]
- Bourdeau, J.E.; Burg, M.B. Effect of PTH on calcium transport across the cortical thick ascending limb of Henle’s loop. Am. J. Physiol.-Ren. Physiol. 1980, 239, F121–F126. [Google Scholar] [CrossRef]
- Dusso, A.S.; Brown, A.J.; Slatopolsky, E. Vitamin D. Am. J. Physiol.-Ren. Physiol. 2005, 289, F8–F28. [Google Scholar] [CrossRef]
- Gomes, S.A.; dos Reis, L.M.; Noronha, I.L.; Jorgetti, V.; Heilberg, I.P. RANKL Is a Mediator of Bone Resorption in Idiopathic Hypercalciuria. Clin. J. Am. Soc. Nephrol. 2008, 3, 1446–1452. [Google Scholar] [CrossRef]
- How, K.L.; Hazewinkel, H.A.W.; Mol, J.A. Dietary Vitamin D Dependence of Cat and Dog Due to Inadequate Cutaneous Synthesis of Vitamin D. Gen. Comp. Endocrinol. 1994, 96, 12–18. [Google Scholar] [CrossRef]
- Morris, J.G. Ineffective Vitamin D Synthesis in Cats Is Reversed by an Inhibitor of 7-Dehydrocholestrol-Δ7-Reductase1,2. J. Nutr. 1999, 129, 903–908. [Google Scholar] [CrossRef]
- Corbee, R.J.; Vaandrager, A.B.; Kik, M.J.; Hazewinkel, H.A. Cutaneous Vitamin D Synthesis in Carnivorous Species. J. Vet. Med. Res. 2015, 2, 1031. [Google Scholar]
- Zafalon, R.V.A.; Risolia, L.W.; Pedrinelli, V.; Vendramini, T.H.A.; Rodrigues, R.B.A.; Amaral, A.R.; Kogika, M.M.; Brunetto, M.A. Vitamin D metabolism in dogs and cats and its relation to diseases not associated with bone metabolism. J. Anim. Physiol. Anim. Nutr. 2020, 104, 322–342. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.G. Idiosyncratic nutrient requirements of cats appear to be diet-induced evolutionary adaptations. Nutr. Res. Rev. 2002, 15, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Horst, R.L.; Reinhardt, T.A.; Russel, J.R.; Napoli, J.L. The isolation and identification of vitamin D2 and vitamin D3 from Medicago sativa (alfalfa plant). Arch. Biochem. Biophys. 1984, 231, 67–71. [Google Scholar] [CrossRef]
- Nair, R.; Maseeh, A. Vitamin D: The “sunshine” vitamin. J. Pharmacol. Pharmacother. 2012, 3, 118–126. [Google Scholar] [PubMed]
- Jäpelt, R.B. Vitamin D in plants: A review of occurrence, analysis, and biosynthesis. Front. Plant Sci. 2013, 4, 1–20. [Google Scholar] [CrossRef]
- Oonincx, D.G.A.B.; van Keulen, P.; Finke, M.D.; Baines, F.M.; Vermeulen, M.; Bosch, G. Evidence of vitamin D synthesis in insects exposed to UVb light. Sci. Rep. 2018, 8, 10807. [Google Scholar] [CrossRef]
- Morris, J.G. Cats discriminate between cholecalciferol and ergocalciferol. J. Anim. Physiol. Anim. Nutr. 2002, 86, 229–238. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D Status: Measurement, Interpretation, and Clinical Application. Ann. Epidemiol. 2009, 19, 73–78. [Google Scholar] [CrossRef]
- Sprinkle, M.C.; Hooper, S.E.; Backus, R.C. Previously undescribed vitamin D C-3 epimer occurs in substantial amounts in the blood of cats. J. Feline Med. Surg. 2018, 20, 83–90. [Google Scholar] [CrossRef]
- Pike, J.W.; Meyer, M.B. The Vitamin D Receptor: New Paradigms for the Regulation of Gene Expression by 1,25-Dihydroxyvitamin D3. Endocrinol. Metab. Clin. N. Am. 2010, 39, 255–269. [Google Scholar] [CrossRef]
- Cartwright, J.A.; Gow, A.G.; Milne, E.; Drummond, D.; Smith, S.; Handel, I.; Mellanby, R.J. Vitamin D Receptor Expression in Dogs. J. Vet. Intern. Med. 2018, 32, 764–774. [Google Scholar] [CrossRef]
- Wang, S.; Wang, X.; Wu, J.; Lin, Y.; Chen, H.; Zheng, X.; Zhou, C.; Xie, L. Association of vitamin D receptor gene polymorphism and calcium urolithiasis in the Chinese Han population. Urol. Res. 2012, 40, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Parker, V.J.; Rudinsky, A.J.; Chew, D.J. Vitamin D metabolism in canine and feline medicine. J. Am. Vet. Med. Assoc. 2017, 250, 1259–1269. [Google Scholar] [CrossRef] [PubMed]
- Fukumoto, S. Phosphate metabolism and vitamin D. BoneKEy Rep. 2014, 3, 497. [Google Scholar] [CrossRef] [PubMed]
- Razzaque, M.S. The FGF23–Klotho axis: Endocrine regulation of phosphate homeostasis. Nat. Rev. Endocrinol. 2009, 5, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Penido, M.G.M.G.; Alon, U.S. Phosphate homeostasis and its role in bone health. Pediatr. Nephrol. 2012, 27, 2039–2048. [Google Scholar] [CrossRef]
- Kuro-o, M. The Klotho proteins in health and disease. Nat. Rev. Nephrol. 2019, 15, 27–44. [Google Scholar] [CrossRef]
- Block, G.A.; Ix, J.H.; Ketteler, M.; Martin, K.J.; Thadhani, R.I.; Tonelli, M.; Wolf, M.; Jüppner, H.; Hruska, K.; Wheeler, D.C. Phosphate Homeostasis in CKD: Report of a Scientific Symposium Sponsored by the National Kidney Foundation. Am. J. Kidney Dis. 2013, 62, 457–473. [Google Scholar] [CrossRef]
- Quarles, L.D. Endocrine functions of bone in mineral metabolism regulation. J. Clin. Investig. 2008, 118, 3820–3828. [Google Scholar] [CrossRef]
- Uchihashi, K.; Nakatani, T.; Goetz, R.; Mohammadi, M.; He, X.; Razzaque, M.S. FGF23-Induced Hypophosphatemia Persists in Hyp Mice Deficient in the WNT Coreceptor Lrp6. In Contributions to Nephrology; Razzaque, M.S., Ed.; S. KARGER AG: Basel, Switzerland, 2013; Volume 180, pp. 124–137. ISBN 978-3-318-02369-5. [Google Scholar]
- Meir, T.; Durlacher, K.; Pan, Z.; Amir, G.; Richards, W.G.; Silver, J.; Naveh-Many, T. Parathyroid hormone activates the orphan nuclear receptor Nurr1 to induce FGF23 transcription. Kidney Int. 2014, 86, 1106–1115. [Google Scholar] [CrossRef]
- Covic, A.; Goldsmith, D.; Ureña Torres, P.A. (Eds.) Parathyroid Glands in Chronic Kidney Disease; Springer International Publishing: Cham, Switzerland, 2020; ISBN 978-3-030-43768-8. [Google Scholar]
- Komaba, H.; Fukagawa, M. FGF23–parathyroid interaction: Implications in chronic kidney disease. Kidney Int. 2010, 77, 292–298. [Google Scholar] [CrossRef]
- Martorelli, C.; Kogika, M.; Chacar, F.; Caragelasco, D.; de Campos Fonseca Pinto, A.; Lorigados, C.; Andrade, L. Urinary Fractional Excretion of Phosphorus in Dogs with Spontaneous Chronic Kidney Disease. Vet. Sci. 2017, 4, 67. [Google Scholar] [CrossRef] [PubMed]
- Shipov, A.; Shahar, R.; Sugar, N.; Segev, G. The Influence of Chronic Kidney Disease on the Structural and Mechanical Properties of Canine Bone. J. Vet. Intern. Med. 2018, 32, 280–287. [Google Scholar] [CrossRef]
- Portillo, M.R.; Rodríguez-Ortiz, M.E. Secondary Hyperparthyroidism: Pathogenesis, Diagnosis, Preventive and Therapeutic Strategies. Rev. Endocr. Metab. Disord. 2017, 18, 79–95. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, O.; Isakova, T.; Rhee, E.; Shah, A.; Holmes, J.; Collerone, G.; Jüppner, H.; Wolf, M. Fibroblast Growth Factor-23 Mitigates Hyperphosphatemia but Accentuates Calcitriol Deficiency in Chronic Kidney Disease. J. Am. Soc. Nephrol. 2005, 16, 2205–2215. [Google Scholar] [CrossRef] [PubMed]
- Kogika, M.M.; Chacar, F.; Martorelli, C.; Chew, D.; Toribio, R.; de Brito Galvao, J.F.; Autran, H.; Makowski, A.; Caragelasco, D.; Eleuterio, C.R.; et al. 2018 ACVIM Forum Research Abstract Program: Seattle, Washington, June 14–15, 2018. J. Vet. Intern. Med. 2018, 32, 2144–2309. [Google Scholar]
- Chacar, F.; Kogika, M.M.; Chew, D.J.; de Brito Galvao, J.F.; Autran, H.; Toribio, R.E.; Makowski, A.; Brunetto, M.A.; Reche, A., Jr. 2018 ACVIM Forum Research Abstract Program: Seattle, Washington, June 14–15, 2018. Vitam. D Metab. Chronic Kidney Dis. Cats Nephrolithiasis 2018, 32, 2280–2281. [Google Scholar]
- Geddes, R.F.; Finch, N.C.; Syme, H.M.; Elliott, J. The role of phosphorus in the pathophysiology of chronic kidney disease: Role of phosphorus in the pathophysiology of CKD. J. Vet. Emerg. Crit. Care 2013, 23, 122–133. [Google Scholar] [CrossRef]
- Geddes, R.F.; Finch, N.C.; Elliott, J.; Syme, H.M. Fibroblast Growth Factor 23 in Feline Chronic Kidney Disease. J. Vet. Intern. Med. 2013, 27, 234–241. [Google Scholar] [CrossRef]
- Stubbs, J.R.; Zhang, S.; Friedman, P.A.; Nolin, T.D. Decreased Conversion of 25-hydroxyvitamin D3 to 24,25-dihydroxyvitamin D 3 Following Cholecalciferol Therapy in Patients with CKD. Clin. J. Am. Soc. Nephrol. 2014, 9, 1965–1973. [Google Scholar] [CrossRef]
- Patel, S.R.; Ke, H.Q.; Vanholder, R.; Koenig, R.J.; Hsu, C.H. Inhibition of calcitriol receptor binding to vitamin D response elements by uremic toxins. J. Clin. Investig. 1995, 96, 50–59. [Google Scholar] [CrossRef]
- Michaud, J.; Dubé, P.; Naud, J.; Leblond, F.A.; Desbiens, K.; Bonnardeaux, A.; Pichette, V. Effects of serum from patients with chronic renal failure on rat hepatic cytochrome P450: Human uremic serum downregulates cytochrome P450. Br. J. Pharmacol. 2005, 144, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- Michaud, J.; Naud, J.; Ouimet, D.; Demers, C.; Petit, J.-L.; Leblond, F.A.; Bonnardeaux, A.; Gascon-Barré, M.; Pichette, V. Reduced Hepatic Synthesis of Calcidiol in Uremia. J. Am. Soc. Nephrol. 2010, 21, 1488–1497. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.D. Update on Mineral and Bone Disorders in Chronic Kidney Disease. Vet. Clin. N. Am. Small Anim. Pract. 2016, 46, 1131–1149. [Google Scholar] [CrossRef]
- Segev, G.; Meltzer, H.; Shipov, A. Does Secondary Renal Osteopathy Exist in Companion Animals? Vet. Clin. N. Am. Small Anim. Pract. 2016, 46, 1151–1162. [Google Scholar] [CrossRef] [PubMed]
- Sims, N.A. Coupling the activities of bone formation and resorption: A multitude of signals within the basic multicellular unit. BoneKEy Rep. 2014, 11, 481. [Google Scholar] [CrossRef]
- Gnudi, G.; Bertoni, G.; Luppi, A.; Cantoni, A.M. Unusual Hyperparathyroidism in a Cat. Vet. Radiol. Ultrasound 2001, 42, 250–253. [Google Scholar] [CrossRef]
- Teng, M.; Wolf, M.; Ofsthun, M.N.; Lazarus, J.M.; Hernan, M.A.; Camargo, C.A.; Thadhani, R. Activated Injectable Vitamin D and Hemodialysis Survival: A Historical Cohort Study. J. Am. Soc. Nephrol. 2005, 16, 1115–1125. [Google Scholar] [CrossRef]
- Coyne, D.W.; Goldberg, S.; Faber, M.; Ghossein, C.; Sprague, S.M. A Randomized Multicenter Trial of Paricalcitol versus Calcitriol for Secondary Hyperparathyroidism in Stages 3–4 CKD. Clin. J. Am. Soc. Nephrol. 2014, 9, 1620–1626. [Google Scholar] [CrossRef]
- Polzin, D.J.; Ross, S.; Osborne, C.; Lulich, J.; Swanson, L. Clinical Benefit of Calcitriol in Canine Chronic Kidney Disease. J. Vet. Intern. Med. 2005, 19, 433. [Google Scholar]
- Hostutler, R.A.; DiBartola, S.P.; Chew, D.J.; Nagode, L.A.; Schenck, P.A.; Rajala-Schultz, P.J.; Drost, W.T. Comparison of the effects of daily and intermittent-dose calcitriol on serum parathyroid hormone and ionized calcium concentrations in normal cats and cats with chronic renal failure. J. Vet. Intern. Med. 2006, 20, 1307–1313. [Google Scholar] [CrossRef]
- Ooms, N.; van Daal, H.; Beijers, A.M.; Gerrits, G.P.J.M.; Semmekrot, B.A.; van den Ouweland, J.M.W. Time-course analysis of 3-epi-25-hydroxyvitamin D3 shows markedly elevated levels in early life, particularly from vitamin D supplementation in preterm infants. Pediatr. Res. 2016, 79, 647–653. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chacar, F.C.; Kogika, M.M.; Zafalon, R.V.A.; Brunetto, M.A. Vitamin D Metabolism and Its Role in Mineral and Bone Disorders in Chronic Kidney Disease in Humans, Dogs and Cats. Metabolites 2020, 10, 499. https://doi.org/10.3390/metabo10120499
Chacar FC, Kogika MM, Zafalon RVA, Brunetto MA. Vitamin D Metabolism and Its Role in Mineral and Bone Disorders in Chronic Kidney Disease in Humans, Dogs and Cats. Metabolites. 2020; 10(12):499. https://doi.org/10.3390/metabo10120499
Chicago/Turabian StyleChacar, Fernanda C., Márcia M. Kogika, Rafael V. A. Zafalon, and Marcio A. Brunetto. 2020. "Vitamin D Metabolism and Its Role in Mineral and Bone Disorders in Chronic Kidney Disease in Humans, Dogs and Cats" Metabolites 10, no. 12: 499. https://doi.org/10.3390/metabo10120499
APA StyleChacar, F. C., Kogika, M. M., Zafalon, R. V. A., & Brunetto, M. A. (2020). Vitamin D Metabolism and Its Role in Mineral and Bone Disorders in Chronic Kidney Disease in Humans, Dogs and Cats. Metabolites, 10(12), 499. https://doi.org/10.3390/metabo10120499





