Abstract
This study developed a biodegradable dissolvable face mask incorporating liposomal kojic acid (KA) and licochalcone A from licorice extract (LE) to enhance skin delivery and performance. Liposomes were prepared by thin-film hydration method. The film matrix, composed of PVA/PVP/PEG400/HA, was optimized using factorial design to achieve suitable mechanical strength and rapid dissolution. The optimized mask, containing liposomal KA (1% w/v) and licochalcone A (0.025% w/v), was evaluated for antioxidant activity, ex vivo skin deposition, and short-term efficacy (Approval from the Institutional Review Board of Silpakorn University, Thailand; Ethics Approval No. REC 67.1001-146-7726/COA 68.0320-013 Date of registration: 20 March 2025). The optimized liposomes exhibited a mean particle size of 66–72 nm, entrapment efficiency above 65%, and a zeta potential of −12.5 mV (licochalcone A) and −1.67 mV (KA). Liposomal licochalcone A and KA showed potent antioxidant activity compared to their native forms. The optimized film dissolved within approximately 15 min on moist skin and showed favorable handling properties. Ex vivo studies revealed significantly higher skin deposition of both KA and licochalcone A from the liposomal mask compared with free and liposomal dispersions (p < 0.05). In a 7-day clinical evaluation, the mask significantly improved skin hydration and reduced melanin index (p < 0.05). No irritation or adverse reactions were observed, and user satisfaction was high. This liposomal dissolvable mask offered an effective, well-tolerated, and eco-friendly approach to enhancing skin brightness and hydration, supporting its potential as a sustainable cosmeceutical innovation.
1. Introduction
The demand for advanced skincare solutions has intensified globally, driven by a greater understanding of the detrimental effects of environmental impacts and intrinsic aging processes on skin health [1]. Chronic exposure to factors such as urban pollution and ultraviolet (UV) radiation accelerates the manifestation of undesirable conditions, including hyperpigmentation, rhytides, and compromised barrier function. This has spurred significant research and development into innovative products that deliver bioactive compounds with enhanced efficacy and safety. Among these, facial masks have become a dominant product category, valued for their ability to provide an intensive treatment experience [2,3].
Despite their market prevalence, conventional sheet masks exhibit inherent limitations that compromise both their performance and environmental sustainability. The majority are fabricated from single-use, non-biodegradable substrates, contributing to significant landfill waste. The design presented critical formulation challenges, namely the inefficient delivery of active ingredients across the stratum corneum and the potential for skin irritation. The aqueous vehicle in a typical mask necessitates high concentrations of preservatives, and the duration of mask sheet application can elevate the risk of sensitization and dryness [4]. These intersecting drawbacks underscore the need for transformative innovations in mask technology.
In response, research has explored alternatives to traditional formats, including biodegradable and hydrogel masks [5]. While representing incremental improvements, these platforms do not fully resolve the core issues of waste and delivery efficiency. Other attempts have been investigated and commercialized, such as sleeping masks and leave-on masks, but some issues were not eradicated [6]. A more advanced approach is the development of a dissolvable mask. Fabricated from biodegradable, water-soluble polymers, such a system fundamentally alters the product lifecycle by eliminating post-use waste [6]. The mask matrix dissolves upon contact with skin moisture, ensuring intimate and uniform contact between the formulation and the epidermis without requiring physical removal [7]. While sleeping masks and other leave-on formulations offer the advantage of prolonged skin contact time, the dissolvable mask platform represented a substantial technological advancement by addressing their inherent limitations in delivery, stability, and user experience [8,9]. Conventional leave-on products rely on the passive, slow diffusion of actives from a large, undefined volume of an aqueous cream or gel, a process prone to inefficiency and significant product loss through transference to bedding [10]. In contrast, a dissolvable mask functions as a solid-state system, allowing for a precise, pre-measured unit dose of bioactives to be embedded within the polymer matrix [11,12]. Upon contact with skin moisture, the mask dissolves to create a highly concentrated interface that establishes a strong concentration gradient for enhanced dermal penetration. This dynamic delivery mechanism ensures the payload is directed to the target site, maximizing bioavailability [13,14]. Furthermore, the dissolvable format eliminates the unfavorable sensorial profile of sticky or greasy residues common to sleeping masks and overcomes the microbial contamination risks associated with jar packaging by being ideally suited for sterile, single-use applications [15,16]. This paradigm shift from a passive vehicle to an active, precision-dosed delivery system positions the dissolvable mask as a superior platform for high-performance skincare with a substantial leap forward in both user convenience and sustainability.
For targeting hyperpigmentation, the combination of kojic acid (KA) and licorice extract (LE), which contains licochalcone A, offered a compelling combined action for skin brightness. KA is a well-established tyrosinase inhibitor that directly suppresses melanin biosynthesis and is widely used in cosmetic and dermatological products for the management of hyperpigmentation. However, its clinical effectiveness is often limited by poor skin penetration and instability [17]. LE, particularly its marker compound licochalcone A, was selected due to its well-documented anti-inflammatory, antioxidant, and anti-erythematous activities [18,19]. Since hyperpigmentation is closely associated with inflammation- and oxidative stress–mediated melanocyte activation, licochalcone A provides a complementary mechanism to kojic acid by suppressing upstream inflammatory signals rather than directly inhibiting melanin synthesis [20,21,22]. Their clinical potentials are restricted by suboptimal physicochemical properties for dermal penetration [23,24]. To overcome these significant delivery barriers, liposomal encapsulation has emerged as a highly effective strategy. As biocompatible vesicles composed of phospholipid bilayers, liposomes are uniquely capable of encapsulating both hydrophilic and lipophilic compounds. By sequestering hydrophilic KA within their aqueous core and integrating lipophilic licochalcone A into their lipid membrane, liposomes can markedly improve the stability, solubility, and dermal penetration of both actives [25]. This nanocarrier technology effectively shields the compounds from degradation and facilitates their transport into deeper epidermal strata, thereby enhancing their efficacy.
Therefore, the primary objective of this study was to develop a biodegradable, dissolvable facial mask as a sustainable alternative to conventional sheet masks, while simultaneously enhancing dermal delivery of skin-brightening agents. The secondary objectives were to improve the stability and skin deposition of kojic acid and licorice extract through liposomal encapsulation, and evaluate the antioxidant, skin deposition, short-term brightening efficacy, and safety of the developed formulation. As such, this research provided a proposal for sustainable, high-performance cosmeceutical facial masks designed to meet modern consumer expectations for efficacy, convenience, and environmental responsibility.
2. Materials and Methods
2.1. Materials
Kojic acid (purity ≥ 98.5%), niacinamide, glycine, L-arginine, polyvinyl pyrrolidone (PVP), polyvinyl alcohol (PVA), polyethylene glycol (PEG 400), soybean phosphatidylcholine (PC), Tween® 20, and 2,2-diphenyl-1-picrylhydrazyl (DPPH) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Licorice extract (contain 20% (w/w) licochalcone A), phenoxyethanol, and pantothenic acid were bought from Chanjao Longevity Co., Ltd. (Bangkok, Thailand). Hyaluronic acid (HA, MW = 1550 kDa) was procured from P.C. Chemicals Co., Ltd. (Bangkok, Thailand). All other chemical reagents and solvents utilized in this study were of analytical grade.
2.2. Preparation of Liposomes
The liposomal LE and KA were prepared using thin-film hydration method. The stock solution of PC (100 mM) was prepared in organic solvent mixture containing chloroform and methanol (2:1). Then, PC equivalent to 10 mM was placed in a test tube prior to vortex mixing. The solvent was removed by N2 gas flush to form thin films, which were further desiccated for 24 h. The phosphate-buffed solution (pH 7.4) containing 2% Tween® 20 was then added to the thin film, vortex mixed, bath sonicated for 15 min, and ultrasonicated for 45 min to reduce the particle size. The non-entrapped actives and lipid components were removed via centrifugation at 14,000 rpm for 15 min. The supernatant was collected for further analyses. For liposomal LE and KA, LE (equivalent to 0.01–0.05% licochalcone A) was added to the organic mixture, while KA (1–3%) was added to the aqueous phase. All other steps were conducted in the same manner as for the blank liposomes.
2.3. Characterization of Liposomes
2.3.1. Physicochemical Characteristics
The physicochemical properties of the liposomes were characterized by dynamic light scattering technique. A Zetasizer NanoZS (Malvern, Worcestershire, UK) was used to examine the particle size, polydispersity index (PDI), and zeta potential, with measurements conducted at 25 °C and a 90° scattering angle. Each sample was measured in triplicates immediately following its preparation.
2.3.2. Quantification of Active Compounds
The contents of KA and licochalcone A (the primary marker in LE) were quantified using UV–visible spectrophotometry with a VICTOR Nivo™ Multi-Mode Plate Reader. KA content was measured at an absorbance wavelength of 272 nm, while licochalcone A in LE was measured at 365 nm. The encapsulation efficiency (%EE) of each active compound was determined using a method appropriate to its properties. For KA-loaded liposomes, an indirect quantification method was used. The liposomal suspension was centrifuged through a filter unit (14,000 rpm, 15 min, 4 °C) to separate the liposomal KA from the unencapsulated KA in the aqueous phase. The filtrate containing free KA was then diluted with methanol for the analysis. For LE-loaded liposomes, the %EE was determined directly by disrupting the liposomal structure to release the encapsulated licochalcone A. The disrupted liposomal suspension was diluted directly with methanol and centrifuged to collect the release licochalcone A for the quantification. The concentrations of KA and licochalcone A were then calculated using their respective standard calibration curves with an R2 > 0.99.
2.4. Antioxidant Activity
The antioxidant capacity of the samples was quantified using a DPPH free radical scavenging assay. For this analysis, samples including free LE, free KA, and their respective liposomal formulations were prepared at various concentrations in PBS. To aid the dissolution, the free LE and KA solutions were subjected to bath sonication for 2 h. A methanolic DPPH solution was then added to each sample in a 1:1 volume ratio. Ascorbic acid was used to generate a standard curve, serving as a positive control (R2 = 0.999). After being gently mixed, the reaction mixtures were incubated for 30 min in the dark at room temperature. The reduction of DPPH was then determined by measuring the absorbance of each mixture at 515 nm with a microplate spectrophotometer. The percentage of DPPH radical scavenging was calculated using Equation (1).
2.5. Preparation of Dissolving Face Mask
To develop an optimal dissolving face mask, a series of blank face mask formulations was prepared and evaluated. The formulations were based on two primary film-forming polymers, PVA and PVP, combined with HA as a moisturizing agent and PEG 400 as a plasticizer. To investigate the influence of each component on the physical properties of the films (strength, flexibility, and dissolution rate), nine distinct formulations were designed, as detailed in Table 1. A master formula (F1) was established, and subsequent formulations (F2–F9) were created by systematically varying the concentration of a single component while holding the others constant. The face mask formulations were prepared using a casting method. First, the stock solution of PVA (10% w/v) was prepared in deionized water heated to 80 °C on a water bath with continuous stirring. The required amount of PVA for a given formula was gradually dispersed into the specified amounts of PVP, HA, and PEG 400 and stirred until fully dissolved. To remove any entrapped air bubbles, the final mixture was centrifuged at 4000 rpm for 5 min at 25 °C. Subsequently, 2 g of the solution was precisely weighed and poured into each well of a casting mold. The molds were then dried in a hot air oven at 40 °C for 5 h to produce the final solid face mask for further analysis.

Table 1.
Composition of the face mask formulations (% w/w).
2.6. Characterization of Dissolving Mask
2.6.1. Mechanical Strength
The prepared face mask formulations were evaluated for their mechanical properties to determine their suitability and applicability. The film’s strength and elasticity were quantified using a texture analyzer (TA.XT Plus, Stable Micro Systems, Godalming, UK) equipped with a 5 kg load cell. A tensile test was performed to assess the mechanical integrity of the films. Dried films were carefully cut into uniform rectangular strips (50 × 10 mm). Each strip was securely mounted between the tensile grips of the analyzer and stretched at a constant speed (1.0 mm/s) until the film ruptured. Two key parameters were determined, including force at breakage and Young’s modulus. The force at breakage serves as a direct indicator of the film’s strength and resistance to tearing during handling and application. The Young’s modulus or modulus of elasticity was calculated from the initial linear slope of the stress–strain curve.
2.6.2. Face Mask Dissolution
The dissolution rate of the face mask films was determined to assess their intended dissolving characteristic. Uniform film samples were precisely cut into 25 × 25 mm size. Each square was placed into a plate containing 2.0 mL of deionized water, which served as the dissolution medium. It is to be noted that deionized water was used as the dissolution medium to simulate the intended mode of application of the dissolvable face mask. Prior to use, consumers should be instructed to dampen the skin with water to promote film adhesion and facilitate dissolution. The plates were then placed in an orbital shaker incubator set to maintain a constant temperature of 37 °C, simulating physiological conditions. Continuous agitation was applied at a speed of 100 rpm to ensure homogeneous mixing and reproducible dissolution conditions, consistent with previously reported dissolution studies of polymeric films [26]. The time up to 30 min was recorded from the moment of immersion until the film was fully dissolved, defined as the point at which no visible fragments of the film remained in the solution. The experiment was conducted in triplicate for each formulation, and the mean dissolution time was calculated and reported.
2.7. Effect of Active Compounds on the Dissolving Face Mask
Preliminary investigations revealed that the incorporation of some active compounds significantly altered the physicochemical properties of the blank face mask film. Therefore, a Design of Experiments (DoE) approach was employed to systematically optimize the concentrations of these additives. A two-level factorial screening design was utilized to evaluate the influence of four key factors, which were pantothenol (9–10%), niacinamide (2–5%), arginine (1–5%), and glycine (0.5–1%) on four critical quality attributes including tensile strength, Young’s modulus, and dissolution time at 5 min and 10 min. The dissolution at each time point was given as a score between 0 and 6, where 0 refers to no to slightly swollen hydrogel and 6 refers to completely dissolved. The software (DesignExpert® v.11) generated a set of 24 experimental runs, with each factor varied between a high and a low level within its cosmetically recommended concentration range, as detailed in Table 2.

Table 2.
Experimental runs generated by DesignExpert® v.11 software.
The face mask was prepared using the most suitable blank formulation as investigated earlier. Briefly, PVP and HA were weighed and mixed, followed by the addition of PEG 400 and a 5% w/v PVA from a stock solution. The specific amounts of niacinamide, pantothenic acid, arginine, and glycine for each of the 8 experimental runs were then incorporated into this blank mixture. Following fabrication, each formulation was subjected to mechanical and dissolution testing. The experimental response values were then input into the DoE software for analysis. This allowed for the identification of the factors that most significantly impacted the film’s properties, ultimately leading to the generation of a final, optimized formulation with the most desirable balance of mechanical integrity and rapid dissolution.
2.8. Skin Deposition Study
The amount of liposomal KA and LE that accumulated within the skin was evaluated using an ex vivo model. Abdominal skin from a neonatal pig that had died naturally was utilized as the model membrane. The underlying subcutaneous fat was carefully dissected away with a surgical blade, and the prepared full-thickness skin was stored at −20 °C until required. For the experiment, the thawed skin was mounted onto vertical Franz diffusion cells. The dissolvable film formulation, loaded with KA and LE liposomes, was cut into 1 × 1 cm sections and applied directly to the skin surface in the donor compartment. An aqueous solution containing equivalent concentrations of free KA and LE served as the control. After a 24 h incubation period, the skin samples were carefully removed from the cells. To eliminate any residual formulation, each skin section was thoroughly wiped with a cotton swab soaked in PBS (pH 7.4). The effective diffusion area was then excised, minced into small pieces, and immersed in a 1:1 (v/v) mixture of isopropanol and PBS. To extract the accumulated actives, the mixture was vortexed for 10 min and subsequently incubated in an orbital shaker at 37 °C for 24 h. The resulting extract was collected and centrifuged at 12,000 rpm for 15 min at 4 °C. The supernatant was then filtered through a 0.45 µm syringe filter prior to analysis. The quantities of KA and licochalcone A (as the marker compound for LE) deposited in the skin were quantified using UV-visible spectrophotometry. UV–Vis spectrophotometry was employed due to the distinct absorption peaks of kojic acid and licochalcone A and the absence of interfering matrix components. The method provided adequate sensitivity and linearity for comparative deposition analysis (R2 > 0.99). The total amount of each compound accumulated was calculated and expressed as micrograms per unit area of skin (µg/cm2).
2.9. Exploratory Study of Skin Brightening Efficacy and Safety
2.9.1. Study Design and Participants
A single-arm, before-and-after clinical study was conducted to evaluate the efficacy and safety of the dissolvable film formulation on human skin. The study protocol received full approval from the Institutional Review Board of Silpakorn University, Thailand (Ethics Approval No. REC 67.1001-146-7726/COA 68.0320-013 Date of registration: 20 March 2025). A total of 24 healthy volunteers (male and female, aged 20–40 years) were enrolled after providing written informed consent. The sample size was determined using G*Power software v3.1, based on a Wilcoxon signed-rank test with an effect size of 0.8, an alpha of 0.05, and a power of 0.95. Inclusion criteria required participants to be in good general health with no history of allergies to cosmetic ingredients. Individuals were excluded if they had active skin diseases, lesions, or infections on the test area; had used systemic antihistamines or anti-inflammatory medications within 72 h of the study; were pregnant or planning to become pregnant; or had known skin sensitivities. Adherence to the protocol was monitored, and participants with compliance below 80% or who experienced adverse cutaneous reactions were withdrawn from the study.
Participants were provided with seven units of the test film. They were instructed to apply one film daily to a designated site on their right volar forearm for seven consecutive days. The application was timed to occur each evening between 8:00 PM and 11:59 PM. To standardize skin hydration at the point of application and mimic real-world usage on damp skin, participants were given a spray bottle containing deionized water. The protocol required them to spray the test area twice immediately before applying to allow face mask dissolution into the skin. The film was designed to be left on the skin and was not to be rinsed or peeled off. Throughout the 7-day study period, volunteers were strictly instructed to refrain from applying any other topical products to either of their forearms.
2.9.2. Instrumentation and Measurement Conditions
All biophysical skin parameters were measured using a DermaLab® Series skin analysis system (Cortex Technology, Hadsund, Denmark). Prior to any measurements, participants acclimatized to the room conditions (25 ± 2 °C, 50 ± 5% RH) for at least 20 min. They were also instructed to abstain from applying any topical products to the test area for a minimum of 12 h before each assessment.
2.9.3. Skin Irritation Assessment
The potential for skin irritation was evaluated via a 7-day patch test. A baseline reading of the erythema index (skin redness) was taken from a designated site on the volar forearm of each volunteer. Subsequently, the dissolvable film was applied to this same site once daily for seven consecutive days. The site was visually monitored for any signs of irritation (e.g., erythema, edema, or itching), and a final erythema index measurement was taken on Day 8 to quantify any changes from baseline.
2.9.4. Skin Brightening and Hydration Efficacy
The skin brightening and hydrating effects were assessed on the same cohort. Baseline measurements for melanin index, skin hydration, and skin elasticity were recorded at the designated test site on the forearm. Following the baseline assessment, participants applied the dissolvable film to the area once daily every 24 h for a period of seven days. On Day 8, the final melanin index, hydration, and elasticity values were measured. The efficacy of the formulation was determined by comparing the measurements on Day 8 to the baseline values obtained on Day 1.
2.10. Satisfactory Assessment
Following the completion of the final instrumental measurements on Day 8, a subjective evaluation of the product experience was conducted. Each participant was guided to a quiet, private area to complete a self-assessment questionnaire that has been evaluated based on the index of item-objective congruence (IOC) by experts. This controlled setting was designed to prevent external distractions or influence, thereby ensuring the integrity of their responses.
The questionnaire consisted of five distinct statements aimed at evaluating key aspects of product performance and user experience. Participants were asked to rate their level of agreement with each statement using a 5-point Likert scale, ranging from 1 (Strongly Disagree) to 5 (Strongly Agree). The statements assessed the following attributes and questions:
- ‑
- Immediate Hydration: “My skin feels instantly more hydrated after using the product.”
- ‑
- Tolerability: “I did not experience any irritation after using the product.”
- ‑
- Perceived Efficacy: “After using the product for the full test period, my skin appears brighter.”
- ‑
- Convenience: “The product is convenient to use.”
- ‑
- Purchase Intent: “I would like to use this product again.”
2.11. Statistical Analysis
All quantitative data are presented as mean ± standard deviation (SD) based on at least three independent replicates (n = 3). For the in vitro experiments, statistical differences were analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test. For the in vivo study, each parameter was measured in triplicate under the same conditions and at the same time points. The resulting data were analyzed using the Wilcoxon signed-rank test. For all analyses, a p-value of less than 0.05 was considered statistically significant.
3. Results
3.1. Preparation and Characterization of Liposomes
The physicochemical characteristics of the liposomal formulations are summarized in Table 3. The blank liposomes formed nanosized, uniformly distributed vesicles (PDI < 0.3). The near-neutral zeta potential when measured at 7.4 was consistent with the zwitterionic nature of the primary component, which was phosphatidylcholine. The slight size increase upon KA loading (1–3%) suggested successful incorporation without particle aggregation [27]. Similarly, the zeta potential showed only negligible shifts, remaining close to neutral. This lack of change strongly indicated that KA was successfully sequestered within the aqueous core of the vesicles, without adsorbing to the surface or intercalating into the lipid membrane. The incorporation of LE exhibited a profound and highly concentration-dependent influence on the liposomal properties. At a low concentration of 0.01% licochalcone A, the vesicle size increased slightly; thus the particle size grew with higher LE content incorporated, suggesting the successful loading. Also, the surface charge became significantly more negative [28]. The zeta potential values obtained were relatively low, reflecting the use of non-ionic formulation components, PC and Tween® 20. Such compositions typically result in vesicles with near-neutral surface charge. Although this approach is advantageous for cosmetic safety and skin compatibility, limited electrostatic repulsion may increase the risk of vesicle aggregation. Notably, the low zeta potential represents a potential limitation of the formulation. This suggested that amphiphilic and lipophilic components of the extract began to integrate into the lipid bilayer, expanding its structure and imparting a negative charge from compounds.

Table 3.
Characteristics of the liposomal formulations with various drug content.
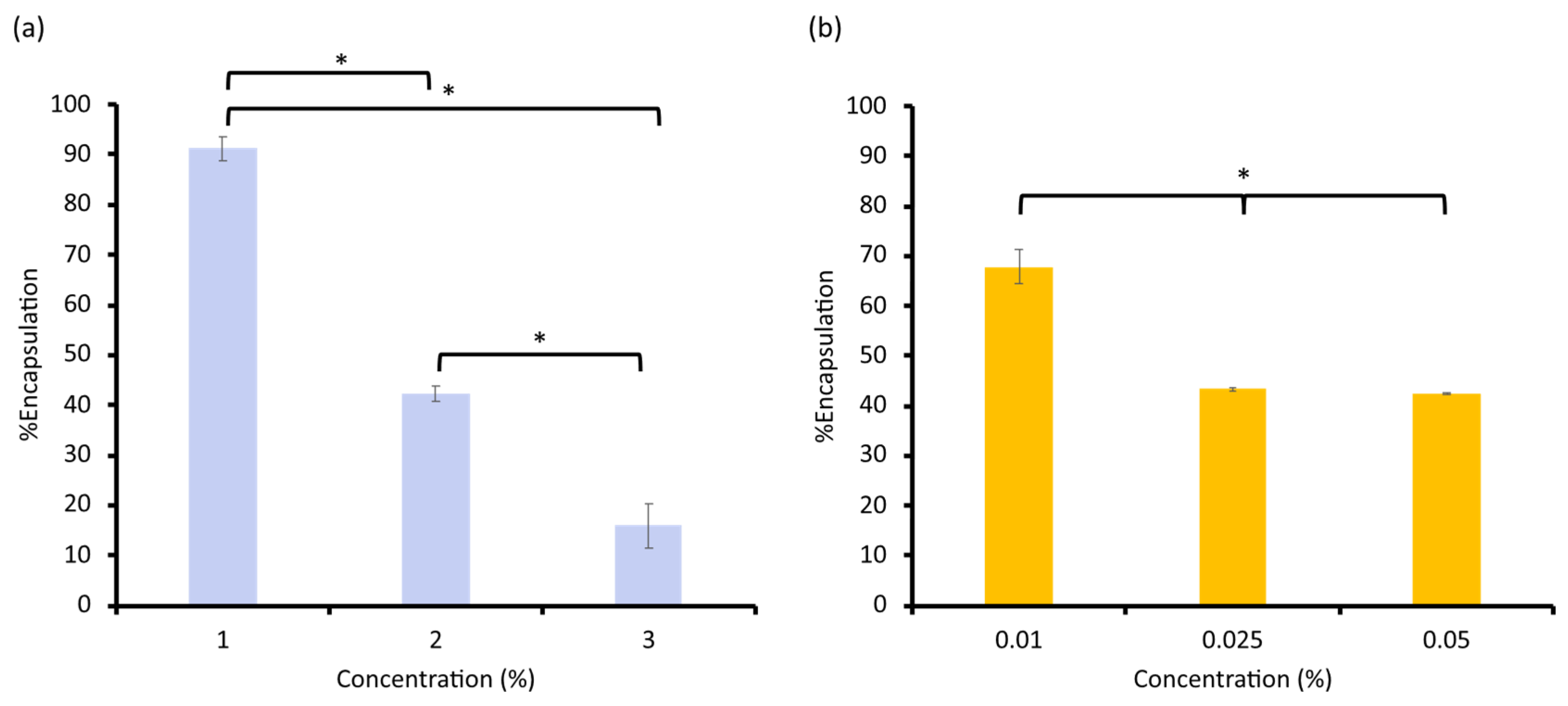
As presented in Figure 1, the %EE decreased at higher loadings, reflecting saturation of the vesicle core and bilayer. The optimal compositions (1% KA and 0.01% LE) balanced loading capacity and colloidal stability and were selected for subsequent formulation studies. The %EE of the active compounds in the liposomal vesicles showed an inverse correlation with the concentrations for both KA and LE. The highest encapsulation was achieved at the lowest tested concentrations of each compound. For KA, a decline in encapsulation at higher initial drug concentration was consistent with the limited aqueous core volume of liposomes. Once saturation is reached, additional drug molecules were less likely to be accommodated within the vesicles, leading to decreased loading efficiency, and potentially increasing the proportion of unencapsulated drug. Similarly, the reduction in %EE with increased concentration of licochalcone A, as a marker for LE, was likely a result of limited bilayer capacity. Since licochalcone A resides in the hydrophobic region of the vesicle, higher concentrations may disturb the membrane structure, thereby reducing loading performance [29,30]. These observations suggested that both KA at 1% and licochalcone A at 0.01% represented optimal formulations, offering a favorable balance between %EE and physicochemical stability. The drug loading at these concentrations was statistically superior to that at higher concentrations and did not compromise vesicle size or stability. These formulations were therefore selected for subsequent studies, including evaluations of release kinetics, skin penetration, and efficacy when incorporated into the dissolvable facial mask platform [31]. Their nanoscale size, uniformity, and high loading capacity collectively support their potential as efficient carriers in skin-brightening cosmeceuticals. These characteristics indicated that both KA- and LE-loaded liposomes were stable carriers suitable for cosmetic skin-delivery applications.
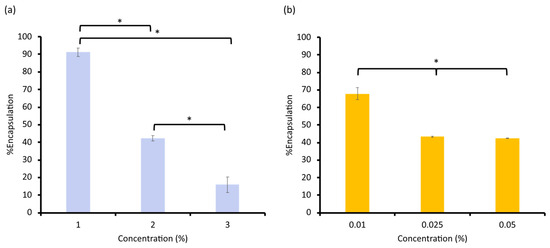
Figure 1.
The percentage of encapsulation efficiency of (a) KA and (b) licochalcone A in the liposomes (* Significant difference, p < 0.05).
3.2. Antioxidant Activity
The antioxidant activities of KA, LE (corresponding to licochalcone A content), and their corresponding liposomal formulations were evaluated using the DPPH radical scavenging assay, and the IC50 values are summarized in Table 4. Ascorbic acid, used as a positive control, exhibited the strongest antioxidant effect with an IC50 of 4.99 ± 0.12 µg/mL. Encapsulation of LE and KA into liposomes notably improved radical-scavenging efficiency, reducing the IC50, especially with liposomal LE (IC50 = 5.40 ± 0.17 µg/mL vs. 11.15 ± 0.20 µg/mL of free LE, p < 0.05), which was comparable to that of ascorbic acid. This improvement can be attributed to the protective effect of the phospholipid bilayer, which stabilized licochalcone A and promoted its interaction with the hydrophobic DPPH radicals [32]. In contrast, liposomal KA exhibited a modest increase in antioxidant activity (IC50 = 118.74 ± 1.58 µg/mL) compared to the free form (IC50 = 196.55 ± 7.07 µg/mL), reflecting partial enhancement of radical scavenging capacity following encapsulation [33]. These results highlighted that liposomal incorporation, particularly for LE, significantly improved the antioxidant potential of the actives. Such enhanced antioxidant potential is cosmetically relevant, as protection against oxidative stress contributes to overall skin radiance and resilience. The strong radical scavenging capacity supported their potential contribution to protecting skin cells from oxidative stress and photo-induced damage.

Table 4.
Characteristics of the dissolvable face mask.
3.3. Preparation and Characterization of Dissolving Face Mask Base
A total of 9 hydrogel film formulations were prepared and evaluated to determine the optimal composition to be used as a dissolvable facial mask capable of incorporating liposomal active compounds. The formulations were based on three primary excipient groups, which were film-forming agents (PVA and PVP), a moisturizing agent (HA), and a plasticizer (PEG 400). The influence of varying the proportions of these components on film appearance, mechanical strength, elasticity, and dissolution time was studied. As shown in Table 4, films with higher concentrations of PVA tended to exhibit improved structural coherence and mechanical strength. As the content of PVA increased, the resulting films became more resistant to tearing, as reflected in higher force at breakage and Young’s modulus values. This behavior was attributable to PVA’s excellent film-forming capacity, where increased polymer entanglement creates a denser, more cohesive matrix [34]. However, films with excessive PVA, particularly when not balanced with enough plasticizer, tended to be more rigid, less elastic, and in some cases, visually brittle or glass-like. These characteristics may hinder conformability to the skin and reduce user comfort [35,36]. In contrast, increasing the proportion of PVP resulted in films that were softer, more pliable, and generally more transparent. PVP contributed hydrophilicity and flexibility, promoting ease of folding and better facial adherence [37,38]. However, films with excessively high or low PVP exhibited limitations. Less PVP content reduced the hydrophilic balance, leading to slower dissolution and a rubbery texture upon hydration. Conversely, at higher levels, films became overly soft and exhibited tackiness, especially when coupled with low PVA or PEG 400 levels. These films occasionally adhered to themselves when folded, complicating handling and application. HA also influenced dissolution performance. At low concentrations, HA contributed marginally to structural properties, but at slightly increased levels, a pronounced enhancement in water uptake and dissolution rate was observed. This can be explained by HA’s hygroscopic nature and high-water retention capacity, which facilitated faster film disintegration upon contact with skin moisture [39,40]. However, its effect on mechanical strength was less significant compared to PVA or PVP. PEG 400 played a critical role in plasticization, modulating the balance between strength and flexibility. When used in appropriate amounts, it improved film elasticity and reduced brittleness. However, reducing PEG 400 content led to slightly more rigid films, which, while sometimes beneficial for mechanical strength, may reduce comfort and usability [41,42]. Importantly, the level of PEG 400 also influenced dissolution time: lower concentrations tended to improve dissolution speed, likely due to a reduced hydrophobic barrier, allowing more efficient water penetration into the polymer matrix [43,44].
The combined effects of these components resulted in a spectrum of film characteristics. While some films demonstrated high mechanical strength, they lacked flexibility or dissolved too slowly. Others dissolved rapidly but were either too soft or lacked structural integrity. Among all, F2 displayed the most favorable overall performance. It offered a visually smooth and uniform appearance, with a mechanical profile that balanced adequate tensile strength with desirable elasticity. More importantly, F2 had one of the shortest dissolution times, reflecting an optimized hydrophilic–hydrophobic balance that supports efficient water interaction without compromising film integrity. These findings suggested the importance of compositional balance when designing dissolvable films for facial application. F2’s performance across all evaluated parameters made it a suitable candidate for further development, particularly for incorporation with liposomal KA and LE in a dissolvable face mask format. This optimized base composition was subsequently used for incorporation of liposomal actives.
3.4. Effect of Active Compounds on the Dissolving Face Mask
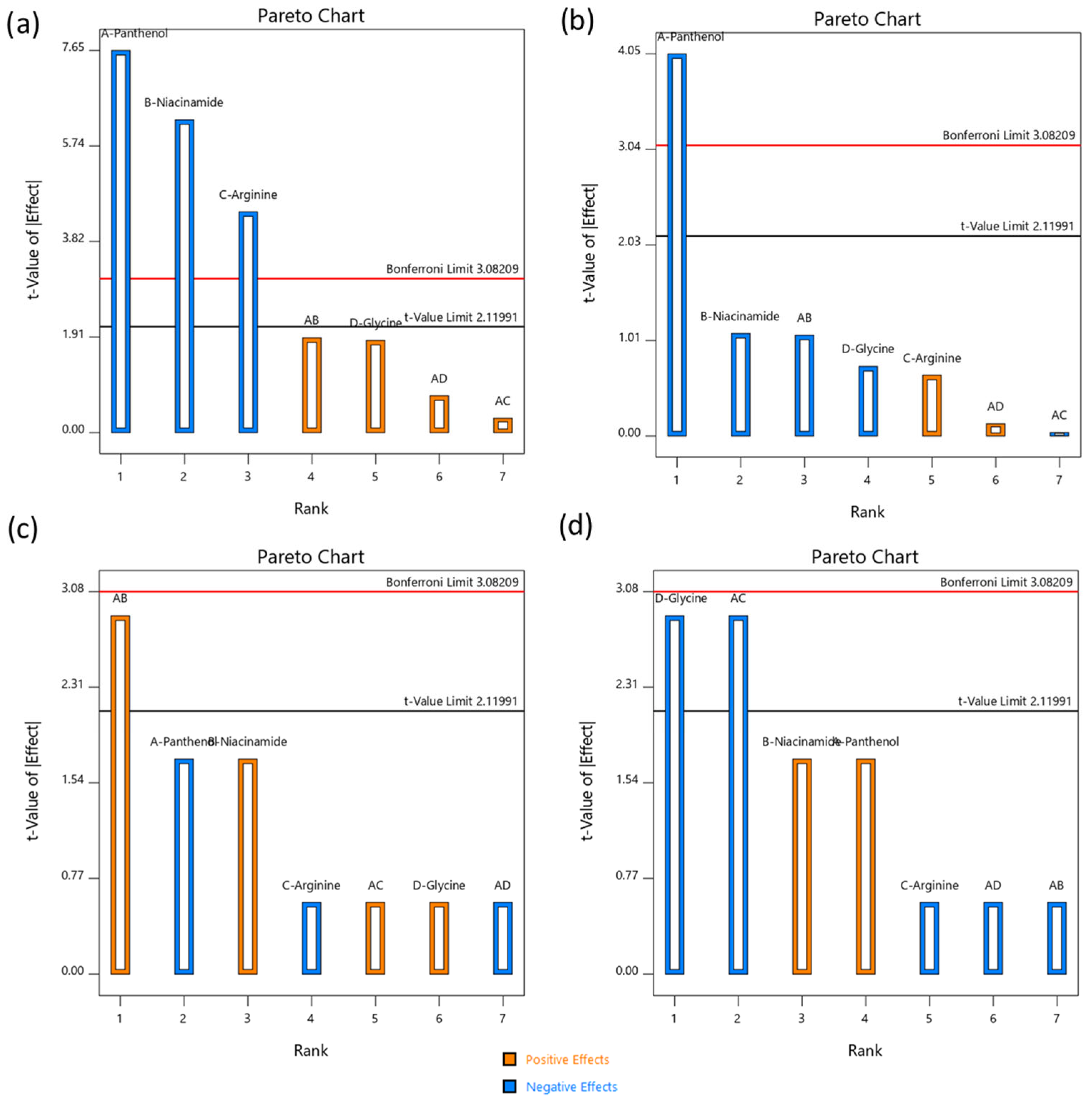
After the incorporation of active compounds into the selected dissolvable face mask base, it was found that some of the added components altered the characteristics of the final face mask significantly. This was found to result from the properties and concentration of the added panthenol, niacinamide, arginine, and glycine. So, to further investigate the influence of each active component on the physical and functional characteristics of the dissolvable face mask, a two-level factorial screening design was employed. The results are interpreted using standardized effect Pareto charts generated from Design-Expert® 11 software. The t-value reflected the standardized effect of each independent variable relative to the experimental error, which represents the ratio of the estimated effect size to its standard deviation. Higher t-values indicated factors exerting a more pronounced influence on the measured response. Factors whose t-values exceeded the statistical significance thresholds (the t-value limit or Bonferroni limit) were considered to have a statistically significant impact on the formulation property at a 95% confidence level (p < 0.05).
As shown in Figure 2a, panthenol (A) was the most significant factor influencing tensile strength, with a t-value exceeding both limits. Niacinamide (B) and arginine (C) also exhibited statistically significant negative effects. Their inclusion appeared to soften the mask matrix, likely due to their humectant and water-attracting nature, which reduces the internal hydrogen bonding in the dry film. Glycine (D) and interaction terms (AB, AD, AC) were less impactful, with t-values below significance thresholds. However, the negative sign of all main effects suggested that increasing these active concentrations may compromise the mechanical strength of the dissolving film, and thus their levels must be balanced to maintain structural integrity.
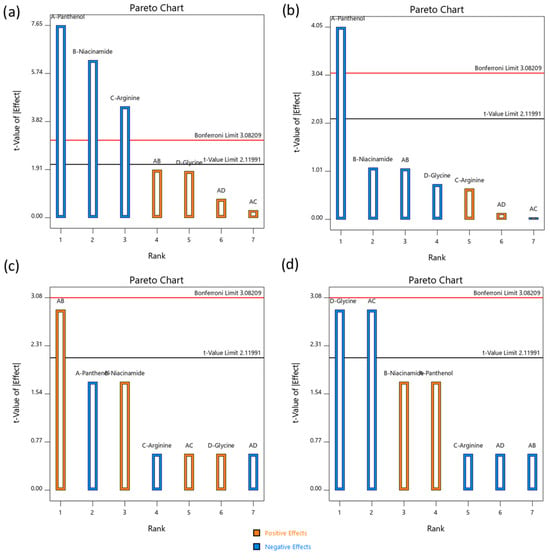
Figure 2.
Pareto charts illustrating the standardized effects of formulation variables on the mechanical and physical properties of the dissolvable face mask. (a) Force at breakage, (b) Young’s modulus, (c) elongation at break, and (d) dissolution time. The red and black lines represented the Bonferroni and t-value significance limits, respectively. Bars above these thresholds indicate statistically significant effects (p < 0.05). Blue bars denoted negative effects, while orange bars denoted positive effects.
The stiffness of the mask, as measured by Young’s modulus (Figure 2b), was primarily influenced by panthenol (A), with a statistically significant negative effect. Its humectant property may increase flexibility and decrease brittleness, desirable traits for user comfort during application. Other variables had minimal and non-significant contributions to modulus values, though all showed a tendency toward further decreasing stiffness. This outcome highlighted panthenol’s dominant role in modulating both tensile strength and elasticity, supporting its dual functional role in formulation as both an active and a plasticizing agent [45].
The rate at which the mask dissolved within 5 min (Figure 2c) was significantly influenced by the interaction between panthenol and niacinamide (AB). This interaction had a strong positive effect, suggesting that the combination accelerated water absorption and fiber disruption. This synergy may stem from their combined hydrophilicity and capacity to disrupt the physical structure of the dry matrix. Other factors had moderate but non-significant effects, suggesting that dissolution behavior in the initial phase was primarily governed by interactive rather than individual effects. At the 10 min mark (Figure 2d), the most influential factors shifted. Both glycine (D) and the AC interaction (panthenol × arginine) had significant negative effects, implying that they slowed dissolution. Glycine, being a zwitterionic amino acid, may reinforce the internal hydrogen-bonded structure, while the AC interaction might create a more cohesive film network. In contrast, panthenol (A) and niacinamide (B) showed moderate positive effects, potentially promoting longer-term disintegration via gradual water uptake. This contrast between 5 and 10 min profiles underscored the complexity of formulation behavior where early dissolution was enhanced by hydrophilic synergy, but extended integrity may be preserved by amino acid network effects.
A confirmation experiment was performed using the predicted optimal concentrations of panthenol, niacinamide, arginine, and glycine. The optimization provided a desirability value of 1.000, which indicated that the predicted formulation simultaneously meets all targeted response criteria at their most desirable levels, achieving the defined optimization goals (Table 5) [46]. The experimental values for all critical quality attributes, including tensile strength, Young’s modulus, and dissolution at 5 and 10 min, were found to be in close agreement with the predicted values. No statistically significant differences were observed (p > 0.05), indicating that the model accurately captured the effects of the selected formulation variables. These results confirmed the reliability of the predictive model and its applicability for guiding formulation development toward a dissolvable face mask with balanced mechanical properties and effective dissolution behavior.

Table 5.
Confirmation of the optimal dissolvable face mask formulation. The desirability of the prediction was 1.000.
A two-level factorial design was used to model the effects of the four variables on the performance of the dissolvable face mask. The regression analysis revealed that each response was significantly influenced by a distinct subset of variables, with p-values < 0.05 indicating statistical significance (Table S1, †ESI). The final predictive models were simplified to include only the significant terms, providing focused guidance for formulation optimization. These predictive models (Equations (2)–(5)) demonstrated good fit and provided interpretable insights into the formulation-performance relationship. The inclusion of interaction terms (AB, AC) highlighted the importance of considering not only individual ingredient effects but also how they behave in combination. These equations offer a valuable tool for rational formulation design by allowing prediction of product performance from composition.
Tensile strength = 1.7297 − 0.5433A − 0.4447B − 0.3139C
Young’s modulus = 0.0858 − 0.0183A
5-min dissolution = 4.7917 + 0.2083AB
10-min dissolution = 5.2917 − 0.2083C − 0.2083AC
3.5. Preparation of Dissolvable Face Mask with Liposomes
The composition of the final dissolvable face mask is shown in Table 6. The formulation incorporated liposomal KA and liposomal LE as primary functional ingredients to enhance skin brightening and anti-inflammatory effects [20]. Liposomal encapsulation not only improved the dermal penetration of these actives but also provides protection from premature degradation [31]. In addition, the inclusion of bioactives, such as panthenol, niacinamide, vitamin C, glycine, and arginine, would support a range of skin benefits, including hydration, antioxidant defense, barrier restoration, and collagen stimulation [47,48]. Each component was selected and optimized to achieve a balanced profile of mechanical performance and rapid disintegration while maintaining compatibility with the skin.

Table 6.
The composition of the final dissolvable face mask formulation.
Importantly, performance testing of the optimized formulation demonstrated desirable tensile strength and flexibility, ensuring the film remains intact during handling and application. Upon contact with skin moisture, the film exhibited complete dissolution within 15 min, ensuring efficient delivery without the need for rinsing or manual removal. This formulation offered several practical and cosmeticeutical advantages over conventional sheet masks. Its dissolvable format eliminated the need for removal and disposal, reducing waste and improving hygiene while enhancing user convenience. By disintegrating directly on moist skin, the mask ensured intimate contact and efficient release of active ingredients without leaving residue. Furthermore, the inclusion of liposomal KA and LE would enhance skin penetration and stability, supporting effective skin brightening and anti-inflammatory activity. Complementary actives provide additional benefits, including hydration, antioxidant protection, and stimulation of collagen synthesis, contributing to an overall improvement in skin tone, texture, and barrier function [20].
3.6. Skin Deposition Study
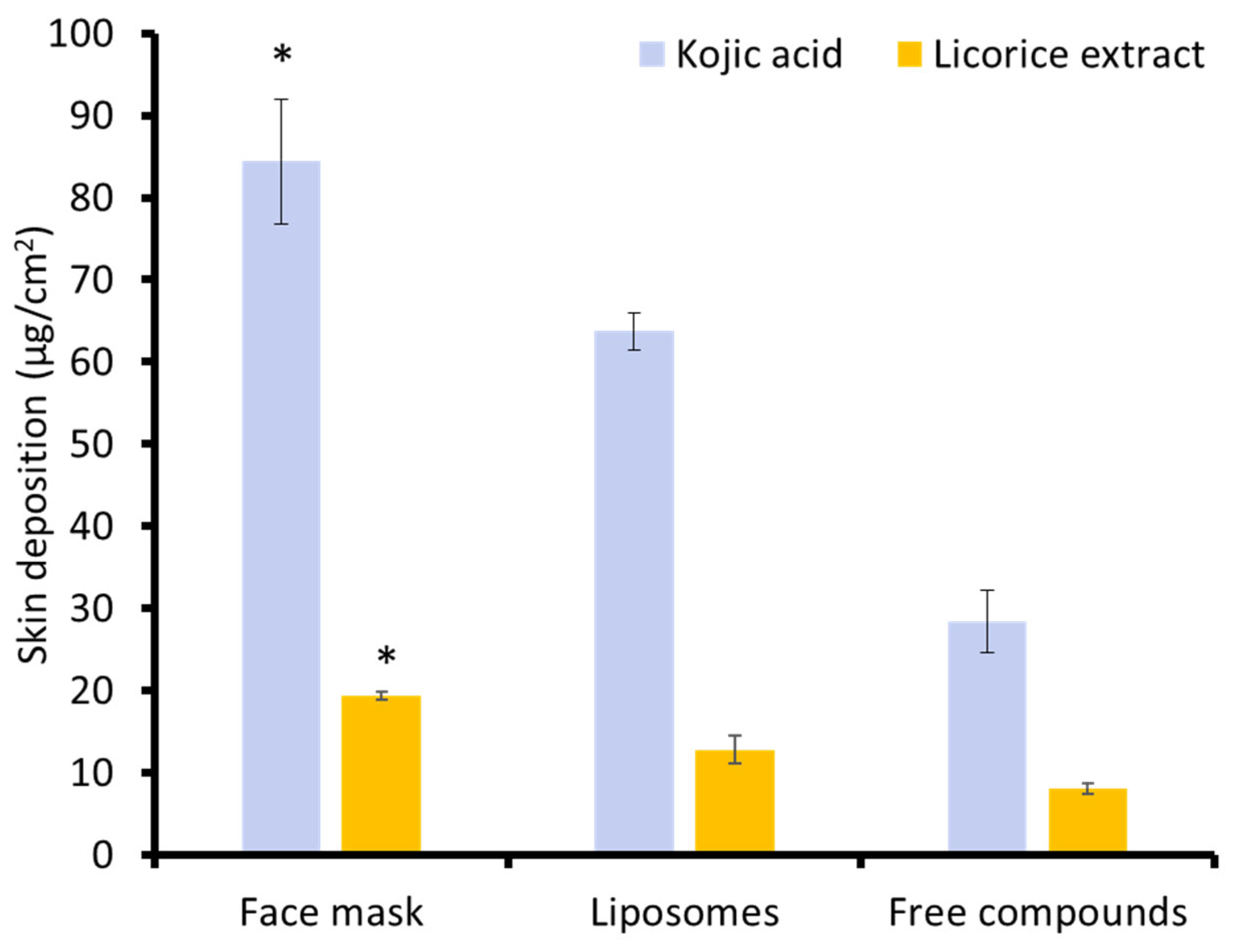
To evaluate the performance of the dissolvable face mask as a skin-delivery platform, a skin deposition study was conducted. As shown in Figure 3, the results demonstrated that the face mask enhanced the skin deposition of both KA and LE. Among the tested systems, the dissolvable face mask achieved the highest deposition of KA. This was greater than that observed for liposomal dispersions and free KA. A similar trend was noted for LE, with deposition levels from the face mask nearly doubling those achieved by liposomes and more than twice that of the free compound (p < 0.05). The superior performance of the face mask can be attributed to multiple formulation-related factors. The film-forming matrix ensured uniform and intimate contact with the skin surface upon application. This close adhesion, coupled with the occlusive nature of the film, promoted localized hydration of the stratum corneum, thereby enhancing skin permeability. Additionally, the presence of humectants such as HA and panthenol might also facilitate water retention within the upper skin layers, further aiding transdermal diffusion [49]. Moreover, the incorporation of KA and LE in liposomal form likely played a crucial role in modulating their release kinetics and improving penetration. Liposomes are known to enhance the bioavailability of encapsulated actives by protecting them from premature degradation and facilitating their passage through the skin barrier [31]. Within the mask matrix, these liposomes were stabilized by hydrophilic polymers and plasticizers, which support a controlled release environment. Possibly, the synergistic interplay of these components collectively contributed to the significantly enhanced skin deposition observed. These results demonstrated that the dissolving mask enhances active retention in the epidermis, supporting its utility for brightening products where localized delivery is desired. These findings not only confirm the mask’s potential as a dermal delivery system for skin-brightening agents but also highlight the importance of rational excipient selection in optimizing performance.
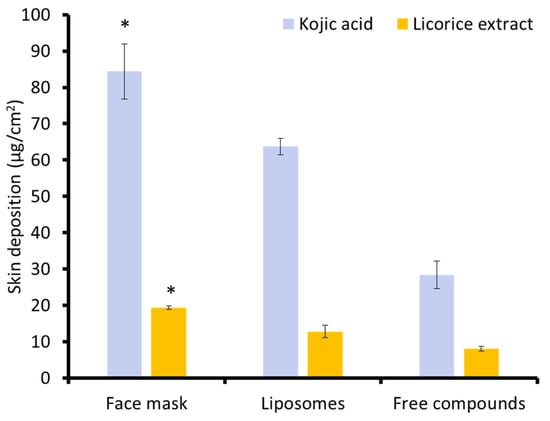
Figure 3.
The skin deposition study of the free KA and LE, their liposomal formulations, and the face mask formulation (* Significant difference, p < 0.05).
It is noted that while this ex vivo deposition study quantified the total retention of actives within the full-thickness skin without differentiating between the stratum corneum and the viable epidermis, the subsequent exploratory study provided functional evidence of penetration depth. As melanogenesis and inflammatory responses occur in the basal layer of the epidermis and the dermis, respectively, the ability of the formulation to elicit biological changes, specifically the reduction in melanin and erythema indices, confirmed that the actives successfully permeated the stratum corneum barrier to reach their therapeutic targets. This deep penetration is likely facilitated by the synergistic effect of the nanometric liposomal carriers (<100 nm) and the occlusion-induced hydration provided by the mask matrix, which drives the concentration gradient across the skin barrier.
3.7. Exploratory Study of Skin Brightening Efficacy and Safety
3.7.1. Demographic Data
A total of 24 healthy volunteers were enrolled and completed the 7-day clinical study designed to evaluate the skin-brightening efficacy and tolerability of the dissolvable face mask. As summarized in Table 7, the study population was predominantly female and represented a range of Fitzpatrick skin types, with the majority classified as type IV. This distribution was particularly relevant, as individuals with Fitzpatrick types III to V are more prone to post-inflammatory hyperpigmentation, making them a suitable population for evaluating the efficacy of brightening agents [50].

Table 7.
Demographic data of the studied population.
Baseline skin measurements further characterized the cohort. Mean skin hydration, melanin index, elasticity, and transepidermal water loss (TEWL) values indicated that participants had healthy skin and no visible irritation or dermatological conditions at the start of the study, providing a stable baseline for assessing product performance. These initial values were essential for quantifying changes post-application and for ensuring homogeneity in skin biophysical parameters across the study group.
3.7.2. Skin Brightening Efficacy and Safety
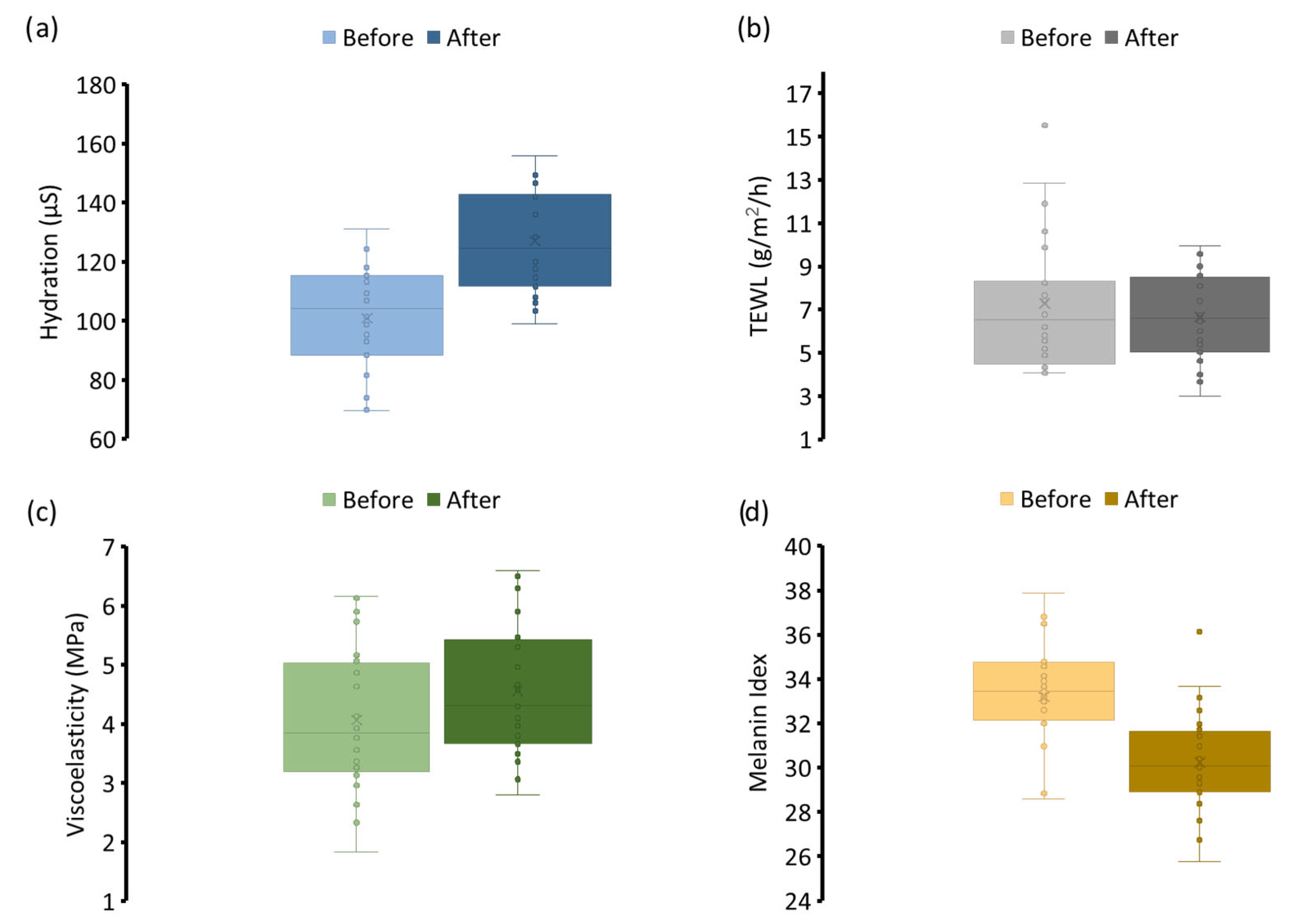
Key biophysical parameters, which were skin hydration, TEWL, elasticity, and melanin index, were monitored to assess short-term cosmetic performance. As illustrated in Figure 4a, a statistically significant increase in skin hydration was observed after treatment. This enhancement was presumably attributed to the synergistic combination of humectants alongside the mask’s hydrogel-based film-forming matrix. The film provided occlusion, which reduced water evaporation and promoted moisture retention, thereby enhancing the stratum corneum’s water-binding capacity [51]. This immediate hydrating effect was also positively noted by participants in the satisfaction survey, reinforcing the clinical findings. TEWL (Figure 4b) measurements showed a downward trend post-application, suggesting improved barrier integrity. Although the reduction was not statistically significant, the observed change indicated that regular use of the formulation may contribute to restoring or maintaining epidermal barrier function. The presence of glycine, arginine, and niacinamide, which are known to support lipid synthesis and skin structure, likely contributed to this effect [52,53]. Viscoelasticity improved modestly following treatment, reflecting increased skin suppleness and elasticity, as presented in Figure 4c. This effect is consistent with enhanced hydration status and may also be linked to the inclusion of collagen-supportive ingredients such as glycine and arginine, which provide structural amino acids involved in extracellular matrix regeneration [54]. Although the change was not dramatic, the upward shift in elastic response supported the long-term benefits of the formulation for skin texture and resilience.
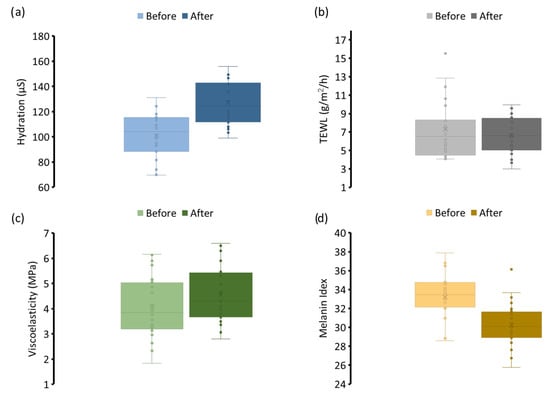
Figure 4.
The 7-day clinical efficacy data pre- and post-application of the dissolvable face mask (a) hydration, (b) TWEL, (c) viscoelasticity, and (d) melanin index. Values are mean ± SD, n = 24.
Most notably, the melanin index (Figure 4d) showed a significant reduction, indicating a measurable cosmetic brightening effect of the mask. This outcome served as in vivo confirmation that the liposomal actives successfully penetrated the stratum corneum to reach the melanocytes in the basal layer. The liposomal encapsulation likely enhanced dermal delivery and stabilized the actives, improving their bioavailability and retention in the target tissue. In addition, niacinamide and vitamin C, which also possess pigment-suppressing and antioxidant properties, may have contributed synergistically to the observed effect [52]. Altogether, the data affirmed that the dissolvable face mask formulation not only delivered aesthetic improvements, but also supported core aspects of skin health. The rapid onset of visible effects within one week underscored the formulation’s potential as a short-course, high-performance cosmetic treatment. It is to be noted that a limitation of this study is its single-group, open-label design. Without a placebo control group, we cannot completely rule out the influence of other confounding factors or the placebo effect. However, the statistically significant changes observed in key biophysical parameters strongly suggested the efficacy of the mask patch. As this was an open-label pilot, results should be interpreted as preliminary and warrant confirmation in controlled studies.
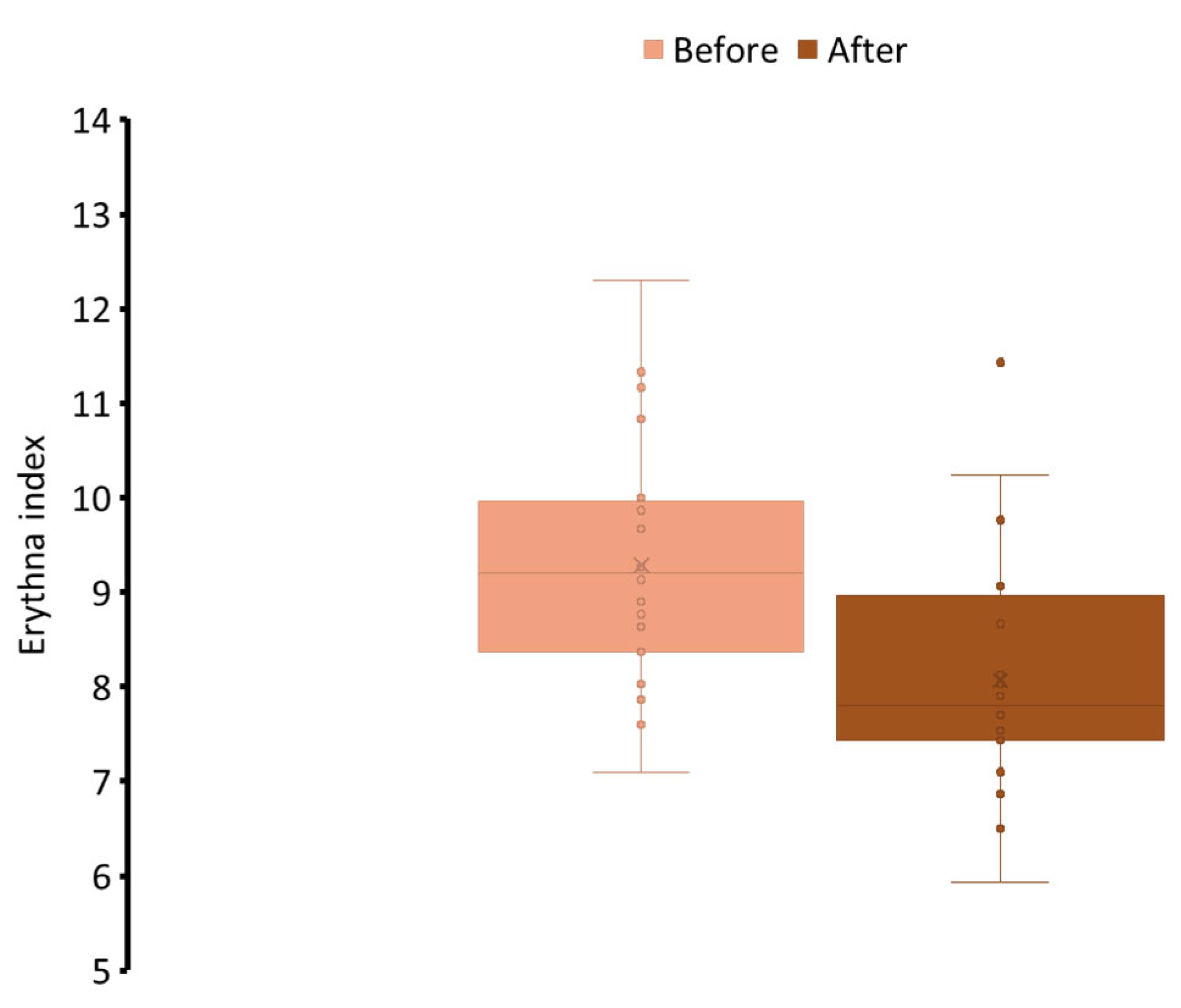
To assess the dermal tolerability of the dissolvable face mask, skin irritation was evaluated using the erythema index as an objective marker of inflammation. As illustrated in Figure 5, erythema values showed a slight but consistent decrease after the 7-day application period, indicating that the product was not only non-irritating but may also have contributed to calming the skin. This outcome can be attributed to the LE containing licochalcone A, which is known to reduce erythema and support recovery of irritated skin, further reinforcing the formulation’s gentle profile [55]. Importantly, no participants reported adverse effects, subjective discomfort, or visible irritation during the study. The reduction in erythema index, though modest, aligns with the intended function of the formulation as a treatment that enhances skin appearance without compromising skin health. These findings confirmed the safety and skin compatibility of the dissolvable face mask, supporting its suitability for regular use across a range of skin types, including those prone to sensitivity. The inclusion of biocompatible excipients, liposome-encapsulated actives, and skin-conditioning agents provides additional assurance of low irritation potential while maintaining high performance in cosmetic efficacy.
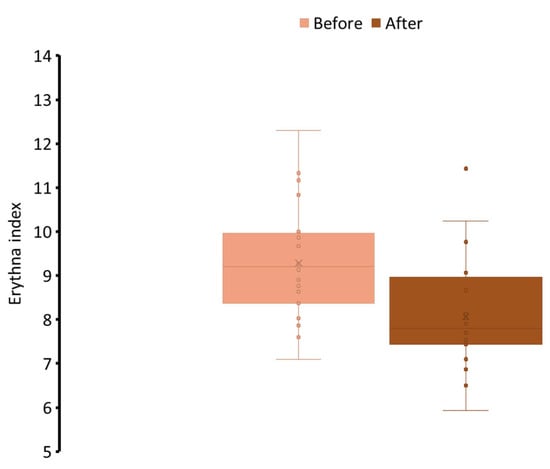
Figure 5.
The participants’ erythema index, which indicates the irritation effect of the face mask after 7-day application.
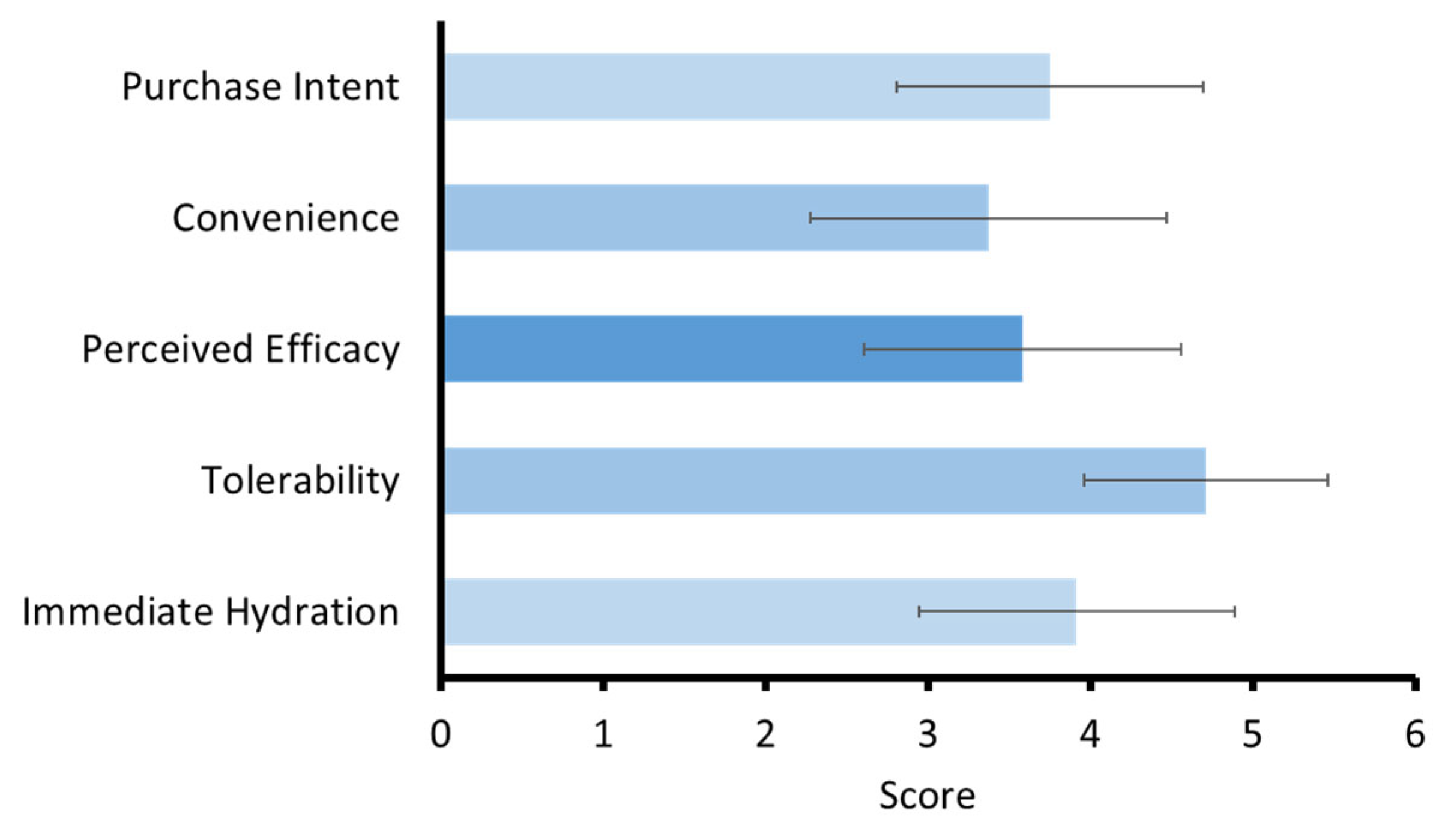
3.8. Satisfactory Assessment
Participant feedback indicated high satisfaction, especially for tolerability and immediate hydration (Figure 6). The evaluation focused on five key dimensions, which were purchase intent, convenience, perceived efficacy, tolerability, and immediate hydration. These metrics provided insight into the practical acceptability and perceived benefits of the formulation beyond objective skin measurements. The highest average rating was observed for tolerability, indicating that participants found the mask gentle, non-irritating, and suitable for repeated application over the study period. Perceived efficacy also received a strong rating, reflecting user recognition of visible skin improvements, which corroborated the clinical improvements in melanin index and moisture levels. Participants also rated immediate hydration favorably, with many users reporting a noticeable increase in skin moisture shortly after application. This aligned with the mask’s formulation, which includes hydrating agents such as HA, panthenol, and niacinamide, as well as the occlusive nature of the dissolving film that promotes moisture retention.
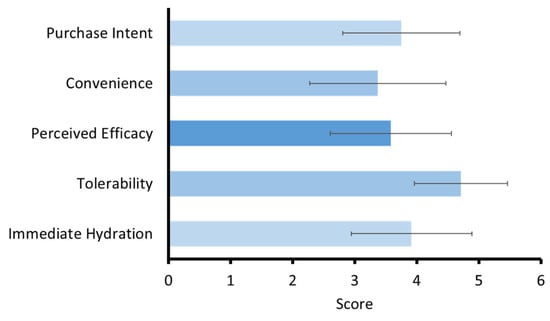
Figure 6.
The user experience and satisfaction score upon using the dissolvable face mask.
The convenience scores were positive, reflecting the practical appeal of a mask that dissolved upon contact with moisture, eliminating the need for removal or cleanup. However, minor suggestions concerned reducing tackiness and improving backing-film separation. Despite these minor critiques, purchase intent scores were encouraging, indicating that most users would consider using or purchasing the product in the future. Also, the participants reported the average complete dissolution time of the patch at 16.17 ± 2.32 min, which was aligned with that observed in vitro. Qualitative feedback further emphasized the innovative and sustainable appeal of the dissolvable format. Participants appreciated the reduced environmental burden compared to conventional sheet masks, highlighting the product’s potential in the growing market for eco-conscious skincare. Taken together, the satisfactory assessment confirmed that the dissolvable face mask was well accepted by users, offering a favorable combination of efficacy, comfort, and sustainability. Overall, user perception aligned with the objective efficacy data, reinforcing consumer acceptance potential.
4. Conclusions
This study presented a novel and sustainable approach to facial skincare through the development of a biodegradable dissolvable mask integrating liposomal KA and LE. Unlike conventional sheet masks, this system combined liposomal nanocarrier technology with a rapidly dissolving polymeric film matrix, achieving superior delivery efficiency, stability, and skin compatibility while eliminating the environmental burden of single-use masks. The liposomal encapsulation effectively enhanced the solubility, stability, and skin penetration of both KA and LE, overcoming a key limitation of conventional brightening formulations. Ex vivo studies demonstrated that the liposomal dissolvable mask significantly increased the skin deposition of both active ingredients compared with free and liposomal dispersions, confirming its enhanced delivery performance. Clinically, the formulation produced measurable improvements in skin hydration and melanin index within only seven days of application, without irritation or adverse effects. User feedback further supported its comfort, ease of use, and perceived efficacy. Overall, this work introduced a novel, eco-friendly dermal delivery platform that bridges the gap between cosmetic appeal and pharmaceutical performance. By integrating biodegradable film technology with liposomal encapsulation, the system enabled efficient transdermal delivery of diverse bioactives while supporting global sustainability goals.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/cosmetics13010021/s1, Table S1: Regression analysis of the predictive models of the dissolvable face mask performances.
Author Contributions
Conceptualization, C.P., P.C. and T.T.; methodology, T.T., C.P. and S.P.; software, P.P. and P.C.; validation, P.P., T.C. and B.P.; formal analysis, T.T., P.C., and C.P.; investigation, T.T., P.C., and C.P.; resources, S.P. and P.P.; data curation, B.P. and P.P.; writing—original draft preparation, T.T. and P.C.; writing—review and editing, C.P. and P.P.; visualization, T.C. and S.P.; supervision, C.P.; project administration, P.P.; funding acquisition, C.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Fundamental Fund under the fiscal year 2568, provided by the Thailand Science Research and Innovation (TSRI) and Silpakorn University.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of Silpakorn University, Thailand (Ethics Approval No. REC 67.1001-146-7726/COA 68.0320-013 Date of registration: 20 March 2025). The evaluated product is a cosmetic formulation, and the study involved short-term assessment of cosmetic effects and skin compatibility without any therapeutic intent. Under the Thai Cosmetic Act B.E. 2558 (2015) and Thai FDA policies, clinical trial registration is mandatory only for drug or medical product trials, not for non-interventional cosmetic product evaluations; therefore, clinical trial was not required for this study.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data is contained within the article or supplementary material.
Acknowledgments
The authors acknowledged the Faculty of Pharmacy, Silpakorn University for facility supports.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Chaudhary, M.; Khan, A.; Gupta, M. Skin Ageing: Pathophysiology and Current Market Treatment Approaches. Curr. Aging Sci. 2020, 13, 22–30. [Google Scholar] [CrossRef]
- Nilforoushzadeh, M.A.; Amirkhani, M.A.; Zarrintaj, P.; Salehi Moghaddam, A.; Mehrabi, T.; Alavi, S.; Mollapour Sisakht, M. Skin care and rejuvenation by cosmeceutical facial mask. J. Cosmet. Dermatol. 2018, 17, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Chen, J.; Chen, T.; Chang, H.; Cui, W.; She, Y.; Li, Z.; Tang, W.; Yuan, D.; Chen, Z.; et al. Investigation of actual exposure to facial sheet mask preceding its risk assessment. Sci. Rep. 2022, 12, 1215. [Google Scholar] [CrossRef]
- Wang, Y.; Cao, Y.; Huang, X.; Zhang, M.; Hu, J.; Li, L.; Xiong, L. Short-term skin reactions and changes in stratum corneum following different ways of facial sheet mask usage. J. Tissue Viability 2024, 33, 831–839. [Google Scholar] [CrossRef]
- Akl, E.M.; Hasanin, M.S.; Dacrory, S. Skin mask hydrogel-based natural sources: Characterization and biological properties evaluations. Bioact. Carbohydr. Diet. Fibre 2023, 29, 100355. [Google Scholar] [CrossRef]
- Bom, S.; Jorge, J.; Ribeiro, H.M.; Marto, J. A step forward on sustainability in the cosmetics industry: A review. J. Clean. Prod. 2019, 225, 270–290. [Google Scholar] [CrossRef]
- Lee, D.H.; Lim, S.; Kwak, S.S.; Kim, J. Advancements in Skin-Mediated Drug Delivery: Mechanisms, Techniques, and Applications. Adv. Healthc. Mater. 2024, 13, e2302375. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, Z.; Tao, H.; He, X.; Hsu, K.; Wang, W.; Fang, X.; Steel, A. Comprehensive assessment of the efficacy and safety of a clay mask in oily and acne skin. Ski. Res. Technol. 2023, 29, e13513. [Google Scholar] [CrossRef]
- Moraes, J.D.D.; Bertolino, S.R.A.; Cuffini, S.L.; Ducart, D.F.; Bretzke, P.E.; Leonardi, G.R. Clay minerals: Properties and applications to dermocosmetic products and perspectives of natural raw materials for therapeutic purposes—A review. Int. J. Pharm. 2017, 534, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Stephane, P. Stability of Cosmetic Formulations Containing UV Filters and Preservatives, Based on Physical and Chemical Parameters. MOJ Toxicol. 2015, 1, 12–21. [Google Scholar] [CrossRef]
- Shen, C.; Shen, B.; Xu, H.; Bai, J.; Dai, L.; Lv, Q.; Han, J.; Yuan, H. Formulation and optimization of a novel oral fast dissolving film containing drug nanoparticles by Box–Behnken design–response surface methodology. Drug Dev. Ind. Pharm. 2014, 40, 649–656. [Google Scholar] [CrossRef]
- Bharti, K.; Mittal, P.; Mishra, B. Formulation and characterization of fast dissolving oral films containing buspirone hydrochloride nanoparticles using design of experiment. J. Drug Deliv. Sci. Technol. 2019, 49, 420–432. [Google Scholar] [CrossRef]
- Kamaly, N.; Yameen, B.; Wu, J.; Farokhzad, O.C. Degradable Controlled-Release Polymers and Polymeric Nanoparticles: Mechanisms of Controlling Drug Release. Chem. Rev. 2016, 116, 2602–2663. [Google Scholar] [CrossRef]
- Zurdo Schroeder, I.; Franke, P.; Schaefer, U.F.; Lehr, C.-M. Development and characterization of film forming polymeric solutions for skin drug delivery. Eur. J. Pharm. Biopharm. 2007, 65, 111–121. [Google Scholar] [CrossRef]
- Oliveira, R.; Almeida, I.F. Patient-Centric Design of Topical Dermatological Medicines. Pharmaceuticals 2023, 16, 617. [Google Scholar] [CrossRef]
- Ali, A.; Skedung, L.; Burleigh, S.; Lavant, E.; Ringstad, L.; Anderson, C.D.; Wahlgren, M.; Engblom, J. Relationship between sensorial and physical characteristics of topical creams: A comparative study on effects of excipients. Int. J. Pharm. 2022, 613, 121370. [Google Scholar] [CrossRef]
- Phasha, V.; Senabe, J.; Ndzotoyi, P.; Okole, B.; Fouche, G.; Chuturgoon, A. Review on the Use of Kojic Acid—A Skin-Lightening Ingredient. Cosmetics 2022, 9, 64. [Google Scholar] [CrossRef]
- Cerulli, A.; Masullo, M.; Montoro, P.; Piacente, S. Licorice (Glycyrrhiza glabra, G. uralensis, and G. inflata) and Their Constituents as Active Cosmeceutical Ingredients. Cosmetics 2022, 9, 7. [Google Scholar] [CrossRef]
- Stiani, S.; Yusransyah, Y.; Septiana, D.; Sumantri, I. Effectivity and Evaluation of Licorice Root (Glycyrrhiza glabra) Extract Serum Formula as a Facial Brightening. Res. J. Pharm. Technol. 2024, 17, 4142–4148. [Google Scholar] [CrossRef]
- Ciganovic, P.; Jakimiuk, K.; Tomczyk, M.; Zovko Koncic, M. Glycerolic Licorice Extracts as Active Cosmeceutical Ingredients: Extraction Optimization, Chemical Characterization, and Biological Activity. Antioxidants 2019, 8, 445. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.H.; Jang, G.Y.; Ji, Y.J.; Lee, J.H.; Choi, S.J.; Hyun, T.K.; Kim, H.D. Antioxidant and Anti-Melanogenic Activities of Heat-Treated Licorice (Wongam, Glycyrrhiza glabra × G. uralensis) Extract. Curr. Issues Mol. Biol. 2021, 43, 1171–1187. [Google Scholar] [CrossRef]
- Liu, D.; Jiang, X.; Yang, F.; Zhou, J.; Li, Y.; Yang, H. Therapeutic potential of Licochalcone A in dermatological diseases: From basic to clinical research. Front. Pharmacol. 2025, 16, 1632006. [Google Scholar] [CrossRef]
- Saeedi, M.; Eslamifar, M.; Khezri, K. Kojic acid applications in cosmetic and pharmaceutical preparations. Biomed. Pharmacother. 2019, 110, 582–593. [Google Scholar] [CrossRef] [PubMed]
- Ramadon, D.; McCrudden, M.T.C.; Courtenay, A.J.; Donnelly, R.F. Enhancement strategies for transdermal drug delivery systems: Current trends and applications. Drug Deliv. Transl. Res. 2022, 12, 758–791. [Google Scholar] [CrossRef]
- Lombardo, D.; Kiselev, M.A. Methods of Liposomes Preparation: Formation and Control Factors of Versatile Nanocarriers for Biomedical and Nanomedicine Application. Pharmaceutics 2022, 14, 543. [Google Scholar] [CrossRef] [PubMed]
- Korelc, K.; Tzanova, M.M.; Larsson, A.; Grassi, M.; Di Cagno, M.P.; Tho, I. A simplified method to interpret the mechanism of drug release from thin polymeric films by drug diffusivity measurements. Int. J. Pharm. 2025, 675, 125491. [Google Scholar] [CrossRef] [PubMed]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M.R. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef]
- Selyutina, O.Y.; Polyakov, N.E. Glycyrrhizic acid as a multifunctional drug carrier—From physicochemical properties to biomedical applications: A modern insight on the ancient drug. Int. J. Pharm. 2019, 559, 271–279. [Google Scholar] [CrossRef]
- Pande, S. Factors affecting response variables with emphasis on drug release and loading for optimization of liposomes. Artif. Cells Nanomed. Biotechnol. 2024, 52, 334–344. [Google Scholar] [CrossRef]
- Liu, J.; Zhu, Z.; Yang, Y.; Adu-Frimpong, M.; Chen, L.; Ji, H.; Toreniyazov, E.; Wang, Q.; Yu, J.; Xu, X. Preparation, characterization, pharmacokinetics, and antirenal injury activity studies of Licochalcone A-loaded liposomes. J. Food Biochem. 2022, 46, e14007. [Google Scholar] [CrossRef]
- Huang, Z.; Meng, H.; Xu, L.; Pei, X.; Xiong, J.; Wang, Y.; Zhan, X.; Li, S.; He, Y. Liposomes in the cosmetics: Present and outlook. J. Liposome Res. 2024, 34, 715–727. [Google Scholar] [CrossRef]
- Liu, M.; Du, Y.; Gao, D. Licochalcone A: A review of its pharmacology activities and molecular mechanisms. Front. Pharmacol. 2024, 15, 1453426. [Google Scholar] [CrossRef]
- Mucha, M.; Skrzydlewska, E.; Gęgotek, A. Natural protection against oxidative stress in human skin melanocytes. Commun. Biol. 2025, 8, 1283. [Google Scholar] [CrossRef]
- Tanwar, R.; Gupta, V.; Kumar, P.; Kumar, A.; Singh, S.; Gaikwad, K.K. Development and characterization of PVA-starch incorporated with coconut shell extract and sepiolite clay as an antioxidant film for active food packaging applications. Int. J. Biol. Macromol. 2021, 185, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.-z.; He, W.; Yao, Y.; Kwok, R.T.K.; Lam, J.W.Y.; Wang, K.; Tang, B.Z.; Fu, Q. Effects of Plasticizers on Structural Transitions and Heterogeneity of Poly(vinyl alcohol) Films as Studied via AIE Materials. Macromolecules 2023, 56, 8641–8650. [Google Scholar] [CrossRef]
- Fu, Z.Z.; Yao, Y.H.; Guo, S.J.; Wang, K.; Zhang, Q.; Fu, Q. Effect of Plasticization on Stretching Stability of Poly(Vinyl Alcohol) Films: A Case Study Using Glycerol and Water. Macromol. Rapid Commun. 2023, 44, e2200296. [Google Scholar] [CrossRef] [PubMed]
- Riccio, B.V.F.; Nascimento, A.; Meneguin, A.B.; Rodero, C.F.; Santos, K.P.; Sabio, R.M.; Annunzio, S.R.; Fontana, C.R.; Barud, H.D.S.; Ferrari, P.C.; et al. Solid Dispersions Incorporated into PVP Films for the Controlled Release of Trans-Resveratrol: Development, Physicochemical and In Vitro Characterizations and In Vivo Cutaneous Anti-Inflammatory Evaluation. Pharmaceutics 2022, 14, 1149. [Google Scholar] [CrossRef]
- Franco, P.; De Marco, I. The Use of Poly(N-vinyl pyrrolidone) in the Delivery of Drugs: A Review. Polymers 2020, 12, 1114. [Google Scholar] [CrossRef]
- Juncan, A.M.; Moisa, D.G.; Santini, A.; Morgovan, C.; Rus, L.L.; Vonica-Tincu, A.L.; Loghin, F. Advantages of Hyaluronic Acid and Its Combination with Other Bioactive Ingredients in Cosmeceuticals. Molecules 2021, 26, 4429. [Google Scholar] [CrossRef]
- Gupta, R.C.; Lall, R.; Srivastava, A.; Sinha, A. Hyaluronic Acid: Molecular Mechanisms and Therapeutic Trajectory. Front. Vet. Sci. 2019, 6, 192. [Google Scholar] [CrossRef]
- Roy, A.; Ghosh, A.; Datta, S.; Das, S.; Mohanraj, P.; Deb, J.; Bhanoji Rao, M.E. Effects of plasticizers and surfactants on the film forming properties of hydroxypropyl methylcellulose for the coating of diclofenac sodium tablets. Saudi Pharm. J. 2009, 17, 233–241. [Google Scholar] [CrossRef]
- Domján, A.; Bajdik, J.; Pintye-Hódi, K. Understanding of the Plasticizing Effects of Glycerol and PEG 400 on Chitosan Films Using Solid-State NMR Spectroscopy. Macromolecules 2009, 42, 4667–4673. [Google Scholar] [CrossRef]
- Jadhav, Y.G.; Galgatte, U.C.; Chaudhari, P.D. Overcoming Poor Solubility of Dimenhydrinate: Development, Optimization and Evaluation of Fast Dissolving Oral Film. Adv. Pharm. Bull. 2018, 8, 721–725. [Google Scholar] [CrossRef]
- Scott, R.A.; Park, K.; Panitch, A. Water soluble polymer films for intravascular drug delivery of antithrombotic biomolecules. Eur. J. Pharm. Biopharm. 2013, 84, 125–131. [Google Scholar] [CrossRef][Green Version]
- Draelos, Z.D. Updates in Medical Skin Care. Adv. Cosmet. Surg. 2018, 1, 211–217. [Google Scholar] [CrossRef]
- Aye, K.C.; Rojanarata, T.; Ngawhirunpat, T.; Opanasopit, P.; Pornpitchanarong, C.; Patrojanasophon, P. Development and characterization of curcumin nanosuspension-embedded genipin-crosslinked chitosan/polyvinylpyrrolidone hydrogel patch for effective wound healing. Int. J. Biol. Macromol. 2024, 274, 133519. [Google Scholar] [CrossRef] [PubMed]
- Michalak, M.; Pierzak, M.; Krecisz, B.; Suliga, E. Bioactive Compounds for Skin Health: A Review. Nutrients 2021, 13, 203. [Google Scholar] [CrossRef]
- Liu, J.K. Natural products in cosmetics. Nat. Prod. Bioprospecting 2022, 12, 40. [Google Scholar] [CrossRef]
- Punnel, L.C.; Lunter, D.J. Film-Forming Systems for Dermal Drug Delivery. Pharmaceutics 2021, 13, 932. [Google Scholar] [CrossRef] [PubMed]
- Markiewicz, E.; Karaman-Jurukovska, N.; Mammone, T.; Idowu, O.C. Post-Inflammatory Hyperpigmentation in Dark Skin: Molecular Mechanism and Skincare Implications. Clin. Cosmet. Investig. Dermatol. 2022, 15, 2555–2565. [Google Scholar] [CrossRef]
- Mawazi, S.M.; Ann, J.; Othman, N.; Khan, J.; Alolayan, S.O.; Al thagfan, S.S.; Kaleemullah, M. A Review of Moisturizers; History, Preparation, Characterization and Applications. Cosmetics 2022, 9, 61. [Google Scholar] [CrossRef]
- Marques, C.; Hadjab, F.; Porcello, A.; Lourenco, K.; Scaletta, C.; Abdel-Sayed, P.; Hirt-Burri, N.; Applegate, L.A.; Laurent, A. Mechanistic Insights into the Multiple Functions of Niacinamide: Therapeutic Implications and Cosmeceutical Applications in Functional Skincare Products. Antioxidants 2024, 13, 425. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Quan, T.; Li, R.; Chen, Y.; Gao, T. Role of Nutritional Elements in Skin Homeostasis: A Review. Biomolecules 2025, 15, 808. [Google Scholar] [CrossRef]
- Murakami, H.; Shimbo, K.; Inoue, Y.; Takino, Y.; Kobayashi, H. Importance of amino acid composition to improve skin collagen protein synthesis rates in UV-irradiated mice. Amino Acids 2012, 42, 2481–2489. [Google Scholar] [CrossRef]
- Kolbe, L.; Immeyer, J.; Batzer, J.; Wensorra, U.; tom Dieck, K.; Mundt, C.; Wolber, R.; Stab, F.; Schonrock, U.; Ceilley, R.I.; et al. Anti-inflammatory efficacy of Licochalcone A: Correlation of clinical potency and in vitro effects. Arch. Dermatol. Res. 2006, 298, 23–30. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.