Abstract
Hair loss disorders, particularly androgenetic alopecia (AGA), are common conditions that carry significant psychosocial impact. Current standard therapies, including minoxidil, finasteride, and hair transplantation, primarily slow progression or re-distribute existing follicles and do not regenerate lost follicular structures. In recent years, regenerative medicine has been associated with a gradual shift toward approaches that aim to restore follicular function and architecture. Stem cell-derived conditioned media and exosomes have shown the ability to activate Wnt/β-catenin signaling, enhance angiogenesis, modulate inflammation, and promote dermal papilla cell survival, resulting in improved hair density and shaft thickness with favorable safety profiles. Autologous cell-based therapies, including adipose-derived stem cells and dermal sheath cup cells, have demonstrated the potential to rescue miniaturized follicles, although durability and standardization remain challenges. Adjunctive interventions such as microneedling and platelet-rich plasma (PRP) further augment follicular regeneration by inducing controlled micro-injury and releasing growth and neurotrophic factors. In parallel, machine learning-based diagnostic tools and deep hair phenotyping offer improved severity scoring, treatment monitoring, and personalized therapeutic planning, while robotic Follicular Unit Excision (FUE) platforms enhance surgical precision and graft preservation. Advances in tissue engineering and 3D follicle organoid culture suggest progress toward producing transplantable follicle units, though large-scale clinical translation is still in early development. Collectively, these emerging biological and technological strategies indicate movement beyond symptomatic management toward more targeted, multimodal approaches. Future progress will depend on standardized protocols, regulatory clarity, and long-term clinical trials to define which regenerative approaches can reliably achieve sustainable follicle renewal in routine cosmetic dermatology practice.
1. Introduction
For centuries, hair has carried profound cultural, social, and personal meaning. When it is lost, whether gradually through androgenetic alopecia (AGA) or suddenly in alopecia areata (AA), the impact reaches far beyond the scalp. Millions worldwide live with the daily reminder of thinning hair, and multiple studies have demonstrated significant impairments in quality of life, self-esteem, and psychosocial functioning in both men and women with pattern hair loss [1,2].
While FDA-approved treatments exist, they often provide only modest relief. Minoxidil, finasteride, and even surgical transplantation cannot halt the ongoing progression in many patients, nor do they offer the possibility of new follicle creation [3,4,5]. For patients, the disappointment is not only cosmetic; it is psychosocial, economic, and deeply human [6,7].
Recent studies indicate a shift from symptom management to mechanisms that promote follicular regeneration [8,9]. Advances in stem-cell biology, exosome science, and tissue engineering are reshaping what was once thought impossible: the revival, or even rebirth, of the human hair follicle. A systematic review of mesenchymal stem cell-derived exosomes documented consistent increases in hair density (9.5–35 hairs/cm2) and shaft thickness (up to ~13 µm) across early trials, with no serious adverse events reported [10,11] (Figure 1).
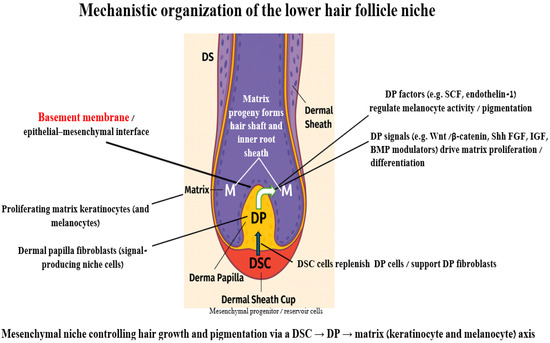
Figure 1.
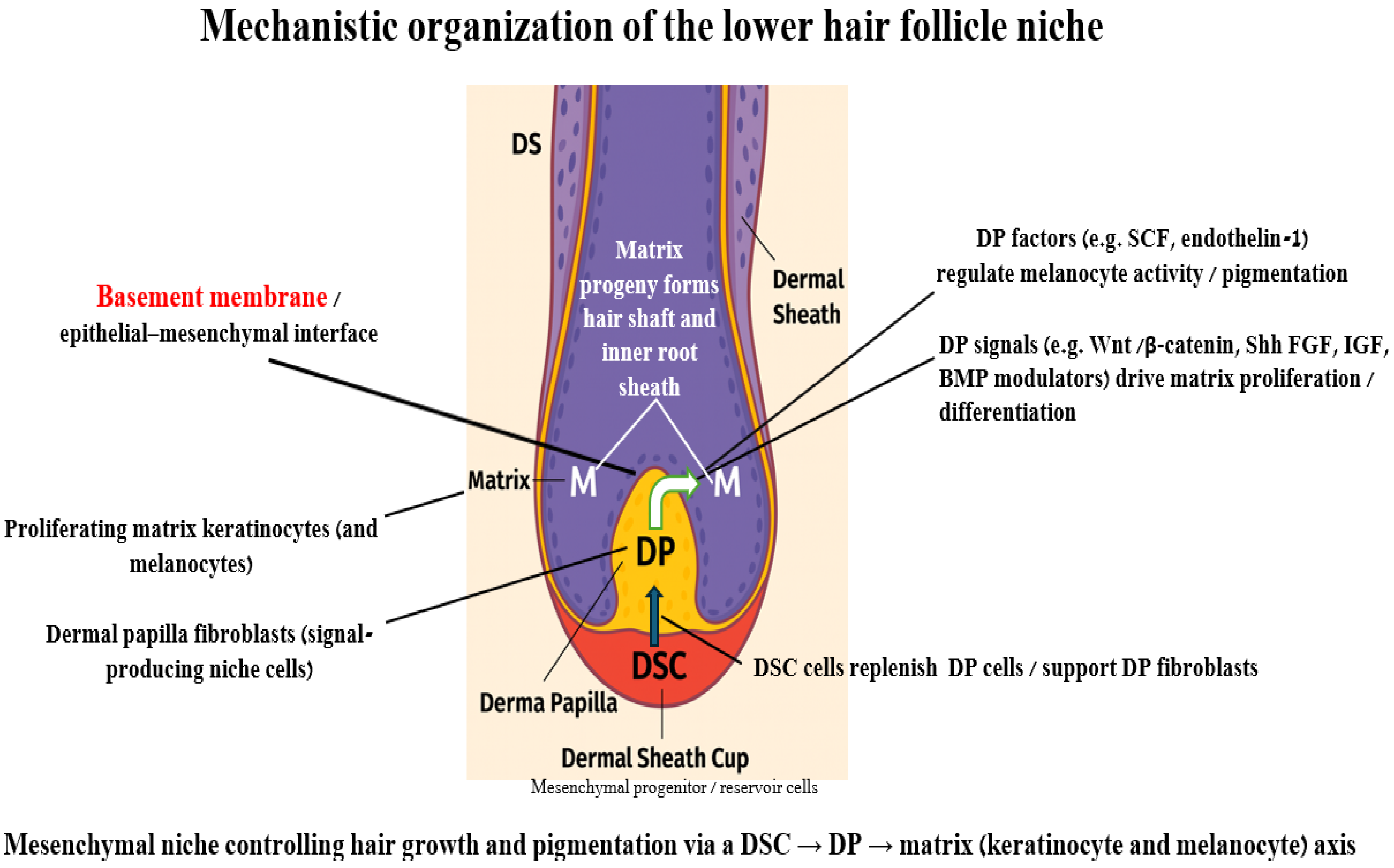
Mechanistic organization of the lower hair-follicle niche. Schematic representation of the human anagen hair-follicle bulb showing the interactions between the dermal sheath cup (DSC), dermal papilla (DP) and matrix (M). The dermal sheath (DS) surrounds the follicle and terminates distally as the dermal sheath cup (DSC), which contains mesenchymal progenitor/reservoir cells. DSC cells can replenish and support dermal papilla (DP) fibroblasts, thereby helping to maintain DP size and function. Across the basement membrane/epithelial–mesenchymal interface, DP fibroblasts secrete key morphogens and growth factors (e.g., Wnt/β-catenin, Shh, FGF, IGF and BMP modulators) that drive proliferation and differentiation of matrix keratinocytes. Progeny of these matrix cells forms the hair shaft and inner root sheath. DP-derived factors such as stem cell factor (SCF) and endothelin-1 regulate matrix melanocyte activity and pigmentation. Together, this DSC → DP → matrix axis defines a mesenchymal niche that controls hair growth and pigmentation.
Mechanistic studies further support these findings: Fu et al. demonstrated that human adipose-derived stem-cell exosomes activate the Wnt/β-catenin pathway by inhibiting GSK-3β, thereby reversing dihydrotestosterone-induced suppression and stimulating dermal papilla cell proliferation [12,13] (Study context: early prospective studies/cohorts with small samples suggest density gains; product characterization and regulation vary—interpret effect sizes cautiously).
Device-assisted strategies are being evaluated as adjuncts; evidence remains limited and heterogenous, and definitive comparative trials are needed. In a prospective clinical trial, exosome therapy combined with microneedling produced mean density gains of approximately 35 hairs/cm2 at 12 months, with high patient satisfaction and only mild, transient side effects [14,15,16].
Reviews of microneedle-based interventions further underscore their synergistic role: by enhancing vascularization, improving local drug and biologic delivery, and promoting follicular survival, microneedling can act as a powerful adjunct to regenerative therapies [17]. (Study context: randomized/evaluator-blinded studies (~12–24 weeks) showed greater hair-count gains when microneedling was added to topical minoxidil vs. minoxidil alone (p < 0.05); protocols vary).
Meanwhile, mechanobiology and follicle stem-cell activation have captured renewed interest. A 2025 review highlighted how controlled mechanical stimulation—stretching, microneedling, or wounding—can awaken quiescent hair follicle stem cells via Wnt and BMP signaling, while macrophage-derived growth factors such as Fand IGF-1 support de novo follicle induction [18]. Experimental work by Chu and colleagues further showed that precisely controlled mechanical stretch activates hair follicle stem cells and promotes telogen-to-anagen transition via M2-like macrophage recruitment and Wnt/β-catenin activation [9] (See Figure 2; Table 1 and Table 2).
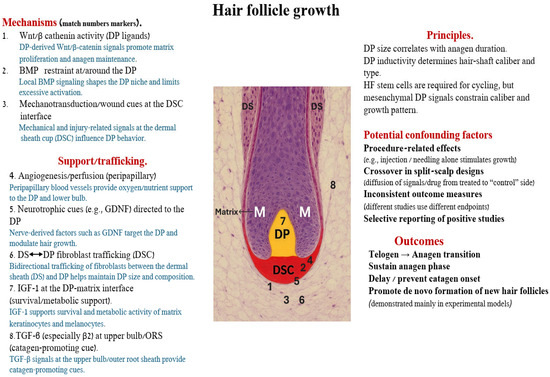
Figure 2.
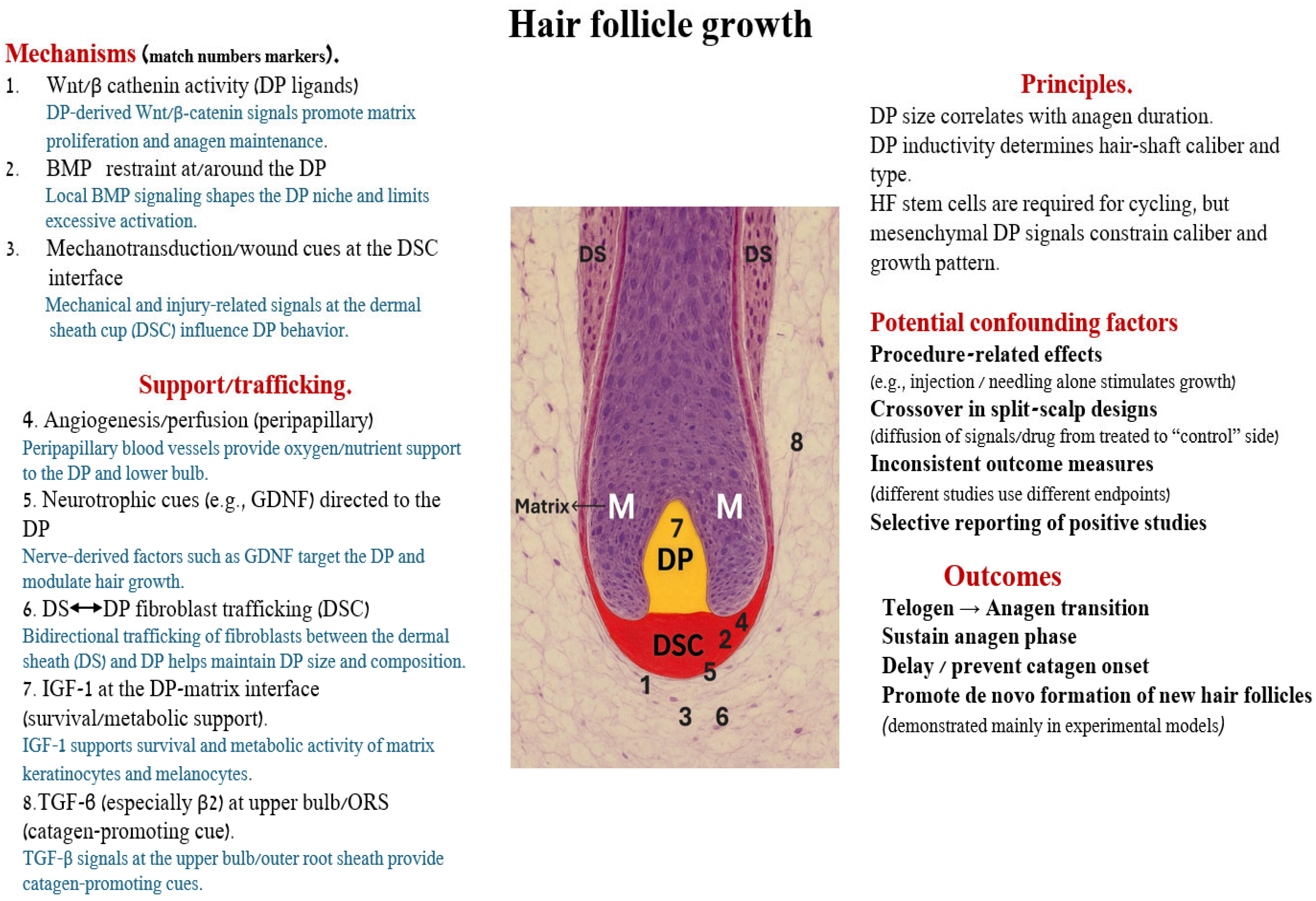
Hair follicle growth: mesenchymal mechanisms, support pathways, confounders and outcomes. Histological image of a human anagen hair follicle bulb. Numbers 1–8 indicate representative mesenchymal mechanisms that influence hair growth. (1) Wnt/β-catenin activity from DP ligands promotes matrix proliferation and anagen maintenance. (2) Local BMP signaling restrains activity at/around the DP and shapes the niche. (3) Mechan transduction and wound-related cues at the DSC interface modulate DP behavior. (4) Peripapillary angiogenesis/perfusion provides oxygen and nutrient support to the DP and lower bulb. (5) Neurotrophic cues (e.g., GDNF) are directed to the DP and modulate growth. (6) Bidirectional trafficking of fibroblasts between DS and DP via the DSC helps maintain DP size and composition. (7) IGF-1 at the DP–matrix interface supports survival and metabolic activity of matrix keratinocytes and melanocytes. (8) TGF-β (especially β2) signals at the upper bulb/outer root sheath provide catagen-promoting cues. Principles on the right summarize how DP size and inductivity govern anagen duration, hair-shaft caliber and type, while potential confounding factors highlight procedural effects, crossover in split-scalp designs, inconsistent outcome measures and selective reporting. Collectively, these pathways influence key outcomes: telogen→anagen transition, maintenance of anagen, delay/prevention of catagen onset and, in experimental models, de novo formation of new hair follicles.

Table 1.
Unified Mechanistic Map.

Table 2.
Pharmachologic Therapies for Androgenic Alopecia.
In addition, novel molecular candidates are entering the pipeline. Liu et al. introduced EX104, a small molecule shown to remodel the follicular microenvironment, promotes angiogenesis, and reactivate miniaturized follicles, outperforming minoxidil in murine AGA models [19]. Related platforms targeting HF stem cells and Wnt/β-catenin signaling further illustrate how small molecules and bioengineered exosomes may complement traditional pharmacologic agents [20,21].
Finally, 3D bioprinting and organoid engineering are at a preclinical stage; clinical feasibility and durability remain unproven. Recent reports of 3D-printed or bioprinted skin constructs containing hair follicles demonstrate de novo follicle-like structures and cycling in animal models, yet translation to humans will require solutions for vascularization, innervation, and scalable manufacturing [22]. A 2025 review of functional follicle regeneration underscores that the central challenge remains replicating embryonic epithelial–mesenchymal interactions and sustaining cyclical follicle activity [9].
Taken together, these developments signal a shift in the therapeutic paradigm: from purely symptomatic management toward mechanism-based attempts at follicular rescue and, eventually, regeneration. For clinicians and researchers, the task now is to convert this expanding body of preclinical and early-phase clinical data into safe, standardized, and accessible treatments [8]. For patients, evidence to date supports modest, incremental benefit, while regenerative endpoints remain under active investigation.
2. Machine Learning in Hair Disorders
2.1. Background and Rationale
Hair is an important evolutionary trait conserved across mammals for its roles in thermoregulation, protection from UV exposure, and environmental sensing [23,24,25,26,27].
Hair loss disorders such as AGA affect a large proportion of men and women worldwide [28]. Beyond its biological function, hair also plays a central role in appearance, identity, and mental well-being [29].
Ongoing research has greatly improved our knowledge of how molecular and cellular processes regulate follicle development and cycling [30,31,32]. Yet, detailed study of the hair fiber itself has been neglected. Fiber-level deviations, like thinning, brittleness, and graying, have been linked to hormonal imbalance, stress, aging, and neurodegenerative disease [33,34,35,36,37].
These changes are often only noticed once advanced, making early intervention difficult [38,39]. For example, AGA is usually diagnosed when visible hair loss is already established [40]. Treatments can slow loss but cannot reverse permanent follicle damage [40].
Monitoring hair fiber phenotypes early could improve outcomes, but fiber-level assessment is technically challenging. Traditional methods are visual, labor-intensive, and lack reproducibility [41,42,43,44,45]. This is where artificial intelligence (AI) and machine learning (ML) provide new possibilities.
2.2. Computer Vision and Deep Phenomics
Advances in computer vision have already improved detection of skin cancers, scars, and other dermatologic conditions [46,47,48,49]. Automated analysis of trichoscopy images has shown promise in assessing scalp health and hair density [50,51]. However, trichoscopy alone cannot capture the structural information within full-length hair fibers. Extracting intact hairs for analysis has been difficult due to overlapping strands, irregular curvature, and varied orientations [52,53].
Deep learning, a branch of machine learning designed to recognize complex and high-dimensional patterns, has transformed image analysis across many fields [54,55]. By enabling the measurement of features that were previously only described qualitatively, it allows researchers to capture subtle biological traits with much greater precision [56,57]. Applications already include identifying seed varieties, counting nematodes, and tracking nuanced biological changes [58,59,60]. Collectively, this approach is known as deep phenotyping, the systematic, high-resolution measurement of biological features using advanced computational tools.
Applied to hair research, this gives rise to deep hair phenotyping: AI-driven segmentation and quantification of individual hair fibers. Using synthetic training data, a sliding-window method, and convolutional neural networks (CNNs), our group has developed a model capable of analyzing hair at single-strand resolution. Early results demonstrate that this approach can detect microstructural changes invisible to the naked eye, changes that are closely linked to endocrinological, developmental, and aging processes.
2.3. Diagnostic, Prognostic, and Therapeutic Applications
Machine learning is now being applied to every stage of hair medicine, from diagnosis and severity assessment to treatment planning and monitoring.
2.4. Diagnosis and Severity Scoring
ML models such as modified Xception networks can analyze trichoscopy, dermoscopy, and OCT images with diagnostic accuracies > 92%, outperforming traditional methods and rivaling expert interpretation [61,62,63]. Algorithms like HairComb, validated in over 400 patients, objectively score alopecia severity with error margins of just 1–9% compared to manual scoring, and correlate strongly with established clinical scales [64,65].
2.5. Prognostic Tools
ML models can predict treatment outcomes and disease progression. One model predicted which AA patients would fail therapy with ~80% accuracy, helping to guide intensity of treatment [66]. Others integrate clinical and genetic data to forecast progression to severe forms of alopecia [66].
2.6. Therapeutic Discovery
AI-guided screening has identified new drug candidates. For example, a manganese-based “nanozyme” with superoxide dismutase–mimetic activity promoted stronger regrowth than minoxidil in mice, even at reduced dosing frequency, by reducing oxidative stress in follicles [67].
2.7. Limitations and Challenges
Despite these advances, challenges remain. Less than 20% of dermatology AI studies report ethnicity or skin type. Models often perform poorly on underrepresented skin tones unless retrained on diverse datasets [68,69]. Preprocessing methods like color normalization can improve accuracy, but diverse image libraries remain essential. AI tools require large volumes of sensitive images, yet transparency around storage and usage is limited [70]. Most dermatology AI systems lack FDA or EMA approval and have limited clinical validation, raising concerns about safety and efficacy [71,72]. Clinicians worry about opaque “black box” algorithms, unclear accountability, and liability. Limited dermatology involvement in AI design adds to skepticism [73,74,75,76,77].
2.8. Conclusions
AI and ML may transform hair medicine. From precise diagnostics and objective severity scoring to predictive models and novel therapies, the opportunities are substantial. Yet adoption depends on overcoming barriers: diversifying training data, safeguarding privacy, ensuring regulatory oversight, and earning clinician trust.
AI will not replace dermatologists but can serve as a powerful co-pilot, bringing objectivity, efficiency, and personalization to the care of hair disorders. If developed responsibly, these technologies could shift the field from subjective observation to precision-driven, regenerative care.
3. Robotic Hair Restoration—ARTAS
AI and ML have already been integrated into mechanical platforms for hair transplant surgery. The ARTAS® robotic follicular unit excision system (FDA cleared in 2011) uses image guidance and ML to identify optimal donor follicles and create recipient sites (Figure 3). In practice, ARTAS can harvest 500–1000 grafts per hour [78] and create approximately 1593 recipient sites per hour, achieving high patient satisfaction scores. Complication rates are low, with postoperative crusting being the most common [79]. Prospective multicenter trials have shown that ARTAS achieves transection (follicle damage) rates around 6.6%, comparable to an experienced surgeon and much better than a beginner operator [80]. In other words, robotic FUE can standardize the most technically demanding steps of surgery, reducing the steep learning curve for new practitioners. In addition to efficiency, robotics minimizes scarring and operator fatigue, which may improve patient safety and outcomes [81,82].
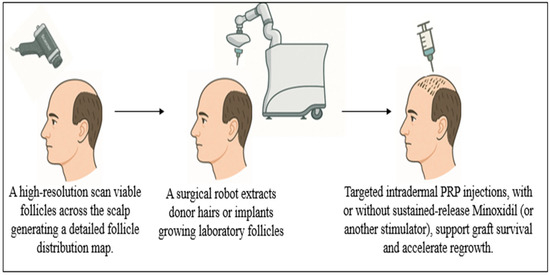
Figure 3.
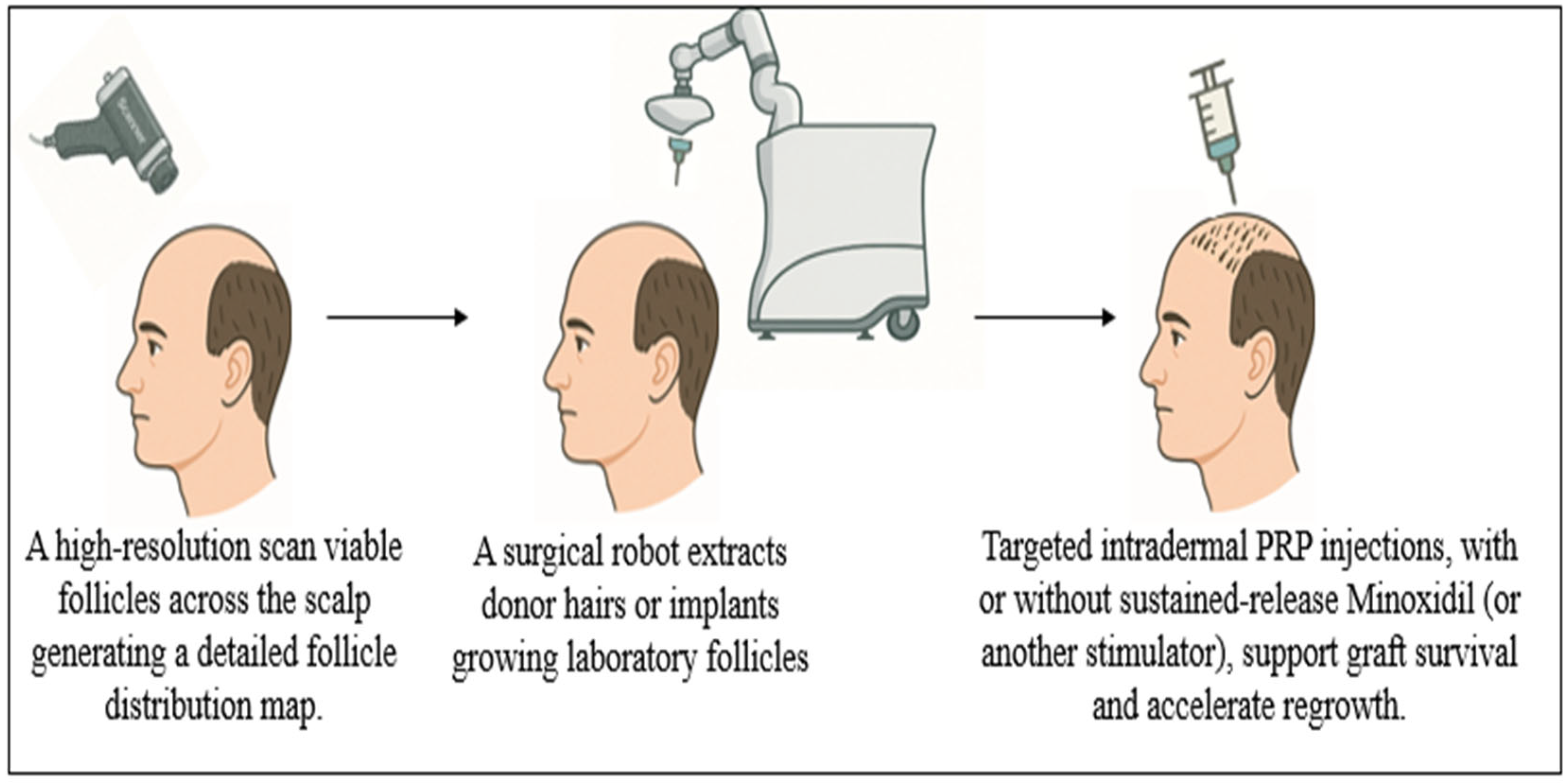
Practical steps toward clinical adoption. Translating AI-guided, cell-based hair regeneration into routine dermatologic practice follows a three-step pathway: (1). A high-resolution scan charts every viable follicle across the scalp, generating a precision density map. (2). A surgical robot either extracts healthy donor hairs or inserts laboratory-grown follicles exactly where coverage is sparse, matching the natural growth angle. (3). Platelet-rich plasma (PRP) or other stimulators support the regrowth of new grafts, reduce inflammation, and accelerate hair regeneration.
The FDA has specifically approved the ARTAS system for use in male patients with brown or black hair. The system incorporates exclusive imaging technology, a computer interface terminal, multiple video cameras, a video display, robotic arm, a suction system for raising harvested grafts, and a specially designed chair for patient positioning. After applying a tensioner, the system calculates the angle between the follicle’s surface exit and its underlying structure. The robotic arm’s dual-bore needle, consisting of a sharp inner tip for initial penetration and a blunt outer needle for deeper dissection, reduces the risk of follicle transection and eases graft removal. The sharp needle includes graduated markings to guide depth. Harvesting is guided by mathematical algorithms that allow randomization and adjustability, reducing human error. Importantly, ARTAS is the only system where the physician operates through a computer interface rather than manually handling the punch tool, reducing ergonomic strain on the operator.
3.1. Advantages Engineered
The advantages of ARTAS include a significantly shorter learning curve compared to manual FUE, the ability to perform repetitive actions without fatigue, and the selection of optimal follicles for harvesting [83]. For early-career practitioners, the robot lowers the barrier to entry into hair restoration by reducing reliance on large teams of assistants, unlike strip harvesting. Typically, one assistant is employed to help control the robot through the interface, while the physician guides harvesting parameters. This streamlined workflow also enhances the robot’s value as a marketing tool for clinics [78].
Robotic follicular unit graft selection algorithms have also been shown to increase the number of hairs per harvest attempt and per graft, improving donor area efficiency [84]. The field continues to evolve, with future directions focusing on more advanced AI integration, improved instrumentation, and potential full automation of both harvesting and implantation [64,85,86].
3.2. Limitations
Despite these advantages, ARTAS has important limitations. The system is expensive, requiring substantial capital investment, maintenance, and consumables, resulting in higher procedural costs than manual FUE [78,87]. Literature to date does not support a clear cost–benefit advantage for routine clinical practice.
Performance challenges also exist. Transection rates are higher in lateral areas such as the supra-auricular region, where hair angles are acute. Variable hair directions, softer tissue at the nape, and regions with scarring or fibrosis from prior strip harvesting can all reduce yield and increase difficulty. The system is also less effective in harvesting curly or tightly coiled hair, particularly in patients of African descent, where angulation and follicle curvature complicate extraction [87,88,89].
In such cases, outcomes may be inferior compared to skilled manual techniques.
3.3. Training and Outcomes
Training challenges are present but not prohibitive. Robotic systems are designed to minimize the learning curve, enabling surgeons of varying experience levels to achieve consistent results. Cosmetic outcomes and patient satisfaction scores are similar across operators, suggesting accessibility of the technology [79]. Nonetheless, optimal use still requires familiarity with device-specific nuances, troubleshooting, and seamless workflow integration.
Patient-reported outcomes are generally favorable, with mean satisfaction of 4.13/5, rapid recovery, and minimal donor scarring [78,79]. Complications such as crusting, itching, and folliculitis are infrequent and comparable to manual FUE. The absence of a linear scar and reduced postoperative discomfort remain highly valued by patients [88].
3.4. Future Directions
Integration with regenerative therapies remains experimental. Robotic systems are being adapted for the implantation of engineered hair follicles derived from stem cells or organoids, potentially enabling true follicle regeneration rather than redistribution [90,91]. Automation and precision may facilitate large-scale, standardized transplantation of bioengineered follicles. However, clinical feasibility has not been established, and translation is constrained by technical and regulatory hurdles.
As these technologies mature, broader adoption will depend on lowering costs and familiarizing surgeons with robotic workflows. Collaborative models, where dermatologists and hair restoration surgeons work together in combined clinics, could further enhance training and patient access.
4. Cellular and Regenerative Therapies
4.1. Moving Beyond Symptom Control
Currently approved drugs such as minoxidil and finasteride remain the cornerstone of androgenetic alopecia treatment, but they provide only partial benefit, primarily slowing or stabilizing hair loss rather than regenerating new follicles (See Table 1 and Table 2). Their effects often plateau, and many patients experience limited cosmetic improvement or treatment fatigue. These limitations underscore the urgent need for next-generation approaches that move beyond symptom control to true regeneration of follicle structures.
With millions affected by alopecia, research is rapidly moving beyond symptom control toward true regeneration of hair follicles. Two major directions stand out: hair follicle neogenesis, once thought impossible in adults but now supported by animal and early translational studies, and precise pathway modulation, especially the Wnt/β-catenin axis that switches follicles from rest to growth [8,92]. In wound-induced hair follicle neogenesis (WIHN), new follicles can form after sufficiently large wounds. Studies in mice, other mammals, and limited human observations show that signals during healing can restart the follicle program. Key immune cells, such as specialized T cells (γδ T cells) and macrophages, play a central role in this process by sending molecular “go” or “stop” signals that influence whether hair stem cells remain dormant or re-enter the growth phase [93,94,95,96]. This discovery has shifted attention to the concept of an “immune niche,” where the immune system does not just fight infection but also acts as a regulator of regeneration, helping stem cells decide when to rest and when to grow.
4.2. Wnt/β-Catenin Pathway Modulation
In parallel, Wnt/β-catenin has become a prime therapeutic target: when it is activated, follicles grow; when it is blocked, they regress [8,97]. Peptide and small-molecule strategies that relieve CXXC5-mediated braking on the pathway, such as PTD-DBM and next-generation activator KY19382, have boosted regrowth and even enhanced WIHN in preclinical models, offering a clearer mechanistic path than classic drugs [98,99,100]. Adjunctive therapies such as low-level laser therapy also influence this pathway, stimulating follicular regrowth [8]. Collectively, these signals act on the dermal papilla–dermal sheath–matrix axis (Figure 1). These Wnt-pathway activators currently have robust preclinical support, but human efficacy and long-term safety remain unknown, and no Phase III trials have yet been completed.
4.3. Cellular Sources: Autologous vs. Allogeneic
Cellular and regenerative therapies for AGA encompass a heterogeneous group of interventions. It is important to distinguish between autologous and allogeneic cell sources, as well as between follicular cells (dermal papilla cells, follicle stem cells) and adipose-derived stem cells (ADSCs). Most clinical studies have focused on autologous sources, particularly ADSCs and follicular stem cells, due to lower risks of immune rejection and ethical issues compared to allogeneic or perinatal sources [101,102,103]. These autologous approaches have shown promising short-term improvements in hair density, shaft thickness, and overall scalp coverage, with relatively favorable safety profiles. However, variability in protocols, small study sizes, and lack of long-term follow-up continue to limit firm conclusions. Overall, these cell-based strategies should still be regarded as preliminary and experimental, based on early-phase cohorts and small randomized trials; Phase III data are not yet available. In contrast, allogeneic and perinatal sources, such as umbilical cord–derived mesenchymal cells, offer the potential for standardized “off-the-shelf” therapies but raise additional challenges, including regulatory hurdles, scalability, and theoretical risks of immune rejection [101,102,103,104].
4.4. Regulatory and Compliance Considerations
At present, no stem cell, exosome/secretome, or cloning/iPSC-derived therapy has approval from the U.S. Food and Drug Administration for the treatment of hair loss. Accordingly, all cell-based interventions (e.g., dermal papilla/dermal sheath cup cells, adipose-derived stem cell/stromal vascular fraction products, iPSC-derived cells, organoids, and 3D bioprinting approaches) and all cell-free secretome/exosome preparations should be regarded as investigational. Clinical use—outside of minimal-manipulation autologous blood products such as platelet-rich plasma—should occur only under appropriate regulatory pathways (e.g., IND/IDE or country-specific equivalents), with GMP-compliant production, product characterization and release testing, donor screening where applicable, and IRB/ethics approval. Regulatory agencies have cautioned against the marketing of unapproved “exosome” products; clinicians and patients should be alert to such claims. In this review, references to these modalities are descriptive and do not constitute endorsement of unapproved clinical use.
4.5. Adipose-Derived Stem Cells and Secretome
ADSCs and their conditioned media (ADSC-CM) are gaining traction because they release growth factors and anti-inflammatory signals that “wake up” follicles. Randomized and controlled studies in AGA show improvements in hair density, shaft thickness, and global impression, alone and when combined with minoxidil, with good tolerability [104,105,106,107]. Standardization of ADSC-derived products as topical or injectable secretome therapies is particularly attractive. (Study context: early prospective studies/cohorts with small samples suggest density gains; product characterization and regulation vary—interpret effect sizes cautiously.)
Systematic reviews and meta-analyses confirm that stem cell-derived conditioned media can significantly increase hair density and thickness in AGA, with pooled mean differences of ~9–15 hairs/cm2 and diameter increases of 2.8–34.6 μm, especially with longer treatment durations [108,109]. A 2024 randomized trial showed that autologous hair follicle mesenchymal stem cell suspensions increased terminal hair proportion and shaft diameter in advanced AGA, especially in follicles <60 μm, with no major adverse events [110]. The available clinical studies of ADSC-derived preparations and follicular stem-cell suspensions are early-phase (typically n < 100 with ≤12-month follow-up) and heterogeneous in protocol; larger, Phase III trials are needed to define durability and long-term safety.
4.6. PRP in Alopecia: Promise, Limits, and Evidence Gaps
Platelet-rich plasma (PRP) has rapidly become one of the most widely used regenerative adjuncts in the treatment of androgenetic alopecia (AGA). Enthusiasm stems from its biologic plausibility and ease of office delivery; however, a balanced appraisal is required to avoid overstating efficacy. Below we synthesize current evidence, explicitly addressing variability in preparation and outcomes, short-term versus durable effects, and randomized controlled data, including trials showing no significant benefit over placebo. (Study context: randomized, double-blind, split-scalp RCT (n = 35) found no significant superiority of PRP over placebo; meta-analyses show heterogeneity and possible publication bias (Table 3).

Table 3.
PRP in androgenetic alopecia: mechanism, sources of variability, and clinical impact.
4.6.1. Mechanistic Rationale
PRP is not a single product but a family of autologous preparations with differing concentrations of platelets, leukocytes, and plasma proteins. Candidate mechanisms relevant to follicle rescue include angiogenic support (e.g., VEGF/PDGF), paracrine stimulation of dermal papilla–matrix crosstalk, and neurotrophic influences. While early work emphasized angiogenesis and mitogenic signaling in dermal papilla cells [104,105], a randomized, blinded pilot identified glial cell line–derived neurotrophic factor (GDNF) as the factor most strongly correlated with density improvement [106], suggesting that patient-specific cytokine ‘signatures’ may determine responsiveness. This composition-dependence provides a biologic basis for heterogeneous clinical outcomes. (Study context: randomized, double-blind, split-scalp RCT (n = 35) found no significant superiority of PRP over placebo; meta-analyses show heterogeneity and possible publication bias.)
4.6.2. Clinical Signals: Meta-Analyses and Cohort Studies
Multiple meta-analyses report that PRP tends to increase hair density at 3–6 months relative to controls, whereas effects on shaft thickness are less consistent; heterogeneity and risk of bias across studies remain high [97,98,99]. Several prospective series describe cosmetic improvement and patient satisfaction, with some reports suggesting stronger density gains in specific subgroups (e.g., male AGA), though sex-related differences remain uncertain [99]. Translational data from a humanized mouse model of AGA further support biological activity closer to human scalp physiology [103]. (Study context: randomized, double-blind, split-scalp RCT (n = 35) found no significant superiority of PRP over placebo; meta-analyses show heterogeneity and possible publication bias).
4.6.3. Randomized, Placebo-Controlled Trials and Procedural Bias
A pivotal split-scalp, double-blind, placebo-controlled RCT reported that both PRP-treated and placebo-injected sites exhibited density gains, with no statistically significant superiority of PRP over placebo [100]. This finding raises two concerns for trial design and interpretation: (i) injection-related micro-injury may induce a regenerative response independent of PRP content; and (ii) diffusion or cross-talk between split-scalp fields may diminish between-arm contrasts. Together with heterogeneous protocols, such procedural effects likely inflate apparent responses and complicate assessment of true PRP-specific benefit. (Study context: randomized, double-blind, split-scalp RCT (n = 35) found no significant superiority of PRP over placebo; meta-analyses show heterogeneity and possible publication bias).
4.6.4. Durability and Maintenance Requirements
Across studies, the clinical signal of PRP most often peaks within ~3–12 months. Sustaining benefit commonly requires maintenance sessions (e.g., every 3–6 months). Long-term durability beyond 12 months without ongoing treatment is rarely documented, and regression after discontinuation is common. These features argue for positioning PRP as a supportive adjunct rather than as a definitive disease-modifying monotherapy in AGA. (Study context: randomized, double-blind, split-scalp RCT (n = 35) found no significant superiority of PRP over placebo; meta-analyses show heterogeneity and possible publication bias).
4.6.5. Variability in Preparation and Reporting
Inter-study variability is substantial. Key parameters—including single versus double spin, activation method (e.g., Ca2+/thrombin), final platelet/leukocyte content, injection schema, dosing intervals, and endpoints—differ widely, limiting reproducibility and the comparability of effect sizes. Meta-analytic signals must therefore be interpreted cautiously, with several analyses noting heterogeneity and potential publication bias [97,98,99,107]. Standardized operating procedures and transparent reporting of cellular/protein yields are necessary to refine the clinical signal and identify true responders.
4.6.6. Safety, Limitations, and Patient Selection
PRP is generally well tolerated; the most frequent issues are injection pain, transient erythema/edema, and procedural inconvenience. However, costs, variable access, and the need for repeated sessions are non-trivial. Importantly, current evidence suggests that PRP primarily augments the function of miniaturized existing follicles rather than inducing true follicle neogenesis; therefore, expectations should be calibrated toward incremental cosmetic gains rather than curative regeneration. Response appears strongest when baseline miniaturization is present but scarring is absent.
4.6.7. Role in Multimodal Care
Given its mechanism and durability profile, PRP is best deployed as an adjunct—paired with disease-stabilizing pharmacotherapy (e.g., Minoxidil, 5-α-reductase inhibitors) and/or procedural strategies (microneedling, transplantation). Combination regimens may leverage complementary pathways (angiogenesis, DP signaling, mechanical cues) while spreading the burden of maintenance across modalities.
4.6.8. Standardization Priorities and Future Directions
To meet the bar set by the reviewer and the field, we outline priorities: (i) harmonized preparation/activation protocols with reporting of platelet and leukocyte yields; (ii) stratification by baseline disease pattern and miniaturization burden; (iii) adoption of objective endpoints (phototrichogram, hair caliber distribution) with pre-registered outcomes; (iv) sham-controlled designs that mitigate injection-related bias; and (v) exploration of biomarker-guided personalization, including GDNF-enriched signatures, to pre-select likely responders. Only with such standards can we resolve conflicting RCT signals and define durable, generalizable benefit.
4.6.9. Conclusions
PRP remains a biologically plausible and clinically accessible adjunct for AGA, with short-term density gains observed in many patients. However, placebo and injection-related effects, protocol heterogeneity, composition-dependent variability, and limited durability—including randomized trials showing no significant superiority over placebo—must be explicitly recognized [97,98,99,100,106,107]. Accordingly, PRP should be framed as an adjunct within multimodal care rather than a stand-alone regenerative therapy. Future studies should prioritize standardized preparation and reporting, sham-controlled designs that minimize procedural bias, and biomarker-guided patient selection to improve reproducibility and long-term outcomes.
4.7. Microneedling: A Regenerative Adjunct with Promising but Temporary Effects
Microneedling has gained attention as a regenerative adjunct in alopecia therapy by inducing controlled micro-injury to the scalp, which activates wound-healing pathways and transiently increases the local release of growth factors such as VEGF, PDGF, and Wnt-related signals. Preclinical work shows that repeated microneedle stimulation upregulates Wnt3a, β-catenin, and VEGF expression, leading to enhanced follicular activity and visible hair regrowth in mice [111]. Clinical data, although limited, are encouraging: randomized and prospective studies demonstrate that microneedling alone, or combined with minoxidil, produces greater improvements in hair density and shaft thickness than minoxidil monotherapy [111], including in both male and female pattern hair loss [112]. (Study context: randomized/evaluator-blinded studies (~12–24 weeks) showed greater hair-count gains when microneedling was added to topical minoxidil vs. minoxidil alone (p < 0.05); protocols vary).
Device-assisted strategies are also emerging: combining microneedling radiofrequency with topical exosome therapy or exosome complexes alone has yielded density gains of ~30–35 hairs/cm2 at 12 months with high patient satisfaction and minimal side effects [16,113]. Still, the effects appear largely temporary, reflecting the short-lived boost in growth factors triggered by wound healing. A 2021 systematic review covering 22 clinical studies and >1000 patients concluded that microneedling is most effective as an adjunct (with minoxidil, PRP, or exosomes), while durability beyond one year remains uncertain [114]. A more recent 2025 meta-analysis confirmed that microneedling plus minoxidil significantly outperformed minoxidil alone, though trial sizes were small and protocols varied [115]. Altogether, microneedling remains a promising but still supplementary strategy in regenerative hair medicine, requiring larger and longer trials to clarify its true role. In particular, microneedling should be viewed as an adjunctive, early-phase intervention: most trials are small of limited duration (≈3–12 months), and use variable or absent sham controls, so reported effect sizes must be interpreted cautiously.
4.8. Broader Stem Cell Strategies
Other studies of autologous follicle micro-grafts, stromal vascular fraction, and cultured dermal-sheath-cup or mesenchymal stromal cells show short-term gains in density and thickness, usually 10–30% above baseline for 3–9 months, but often plateau within a year [102,116]. Combination therapy with ADSC-conditioned media and minoxidil has also demonstrated significant improvements, though differences between concentrated and non-concentrated preparations were not statistically significant [105]. (Study context: pilot RCTs and cohorts report density/diameter improvements vs. controls; preparation methods and cell yields differ widely; longer-term durability remains uncertain.)
4.9. Evidence Gaps and Regulatory Status
Preclinical and early clinical studies are also exploring cell-free secretome therapies and Wnt/β-catenin modulators, but these remain investigational [8,117]. Importantly, no stem-cell product or processing method is approved by the FDA or other major authorities for AGA. In April 2024, the FDA issued a consumer alert clarifying that all marketed stem-cell and exosome treatments for hair loss remain unapproved investigational biologics requiring full regulatory oversight.
Systematic reviews emphasize major gaps: study numbers remain small, protocols vary widely, and outcome measures lack standardization [116,118,119]. Risks such as tumorigenicity and immune rejection are theoretical, especially with allogeneic or manipulated cells, but have not been reported in limited autologous trials [103,104].
4.10. Future Outlook
Looking ahead, biologics and antibody therapies that precisely target key regulators of follicle cycling, along with dermal papilla and stem-cell–based strategies, offer the potential to rebuild follicles “from within,” moving beyond finasteride and minoxidil [92]. The promise lies in combining immune-guided regeneration, Wnt-pathway precision and stem-cell/secretome therapies into a predictable, patient-friendly model. Yet until larger, long-term trials confirm efficacy and safety, stem-cell therapy for pattern hair loss remains experimental and should not be presented as standard care.
5. Biotechnologies: 3D Bioprinting
Beyond administering cells, researchers are actively trying to engineer hair follicles in the laboratory. Three-dimensional bioprinting and organoid culture offer routes to create hair-bearing skin grafts from scratch. In one recent study, scientists printed dermal papilla cell and endothelial cell spheroids directly into a collagen-based dermal matrix [120]. Over time, the tissue self-organized into structures that mimicked native hair follicles, complete with associated epidermal and melanocyte components. In effect, this work produced miniature hair follicles in vitro that could (at least in theory) be implanted as grafts. In a related tissue-engineering advance, researchers used 3D-printed molds to pattern collagen gels: by seeding human dermal papilla cells into hair follicle-shaped microwells, they generated follicle-like structures. When keratinocytes were added on top, the assembled units closely resembled hair follicle stem cell niches with physiological cell organization [120].
Scalable and automated preparation of hair follicle germ-like grafts has been achieved by printing adjacent collagen droplets containing mesenchymal and epithelial cells; these microgel-based constructs contract and enrich cell density, facilitating mesenchymal–epithelial interactions that efficiently regenerate hair follicles and shafts upon transplantation, and guide-inserted microgels have improved hair shaft sprouting through the skin by controlling graft orientation [121]. Recent studies have demonstrated that multilayer composite scaffolds using gelatin/alginate hydrogels can simulate the native hair follicle microenvironment, supporting the formation of self-aggregating dermal papilla spheroids and promoting the expression of hair-inductive genes such as ALP, β-catenin, and α-SMA; these bioprinted constructs, when transplanted into full-thickness wounds in animal models, have shown the ability to regenerate entire hair follicles in vivo with appropriate orientation and physiological organization [120,122].
Three-dimensional bioprinting has also enabled the incorporation of hair follicle-like structures into engineered skin tissues: spheroids of dermal papilla and endothelial cells printed within pre-gelled dermal layers containing fibroblasts matured into follicle-like structures, supported by keratinocyte and melanocyte migration, closely mimicking native skin appendages [123].
Multicellular micropatterning with vascular-cell networks and biomimetic substrates has enhanced both follicular potential and angiogenic capacity, resulting in efficient hair regrowth in animal models [124], and additional advances include the use of skin stem cells and multicomponent hydrogels to fabricate artificial skin via 3D bioprinting, enabling regeneration of epidermis, dermis, hair follicles, blood vessels, and sebaceous glands in wound models [124]. Microenvironmental reprogramming and co-culture systems have further improved the hair-inductive properties of dermal papilla cell aggregates, supporting large-scale production for tissue engineering applications [125,126]. Organoid models and 3D spheroid cultures have demonstrated enhanced extracellular matrix production, improved cell viability, and upregulation of trichogenic signaling pathways, which are critical for folliculogenesis and drug screening [127,128,129,130,131], and these models have shown responsiveness to known hair-growth promoters and inhibitors and can be used for high-throughput screening and mechanistic studies. Despite these advances, technical limitations remain, including scalability issues; producing thousands of functional units for clinical application is not yet feasible at industrial scale [121,126].
High-throughput systems have been proposed, but maintaining cell viability, inductivity, and reproducibility during upscaling remains problematic. Cost is another significant factor. The use of specialized biomaterials (e.g., gelatin/alginate hydrogels, collagen matrices), growth factors, and advanced bioprinting hardware increases production expenses, which may limit accessibility for widespread clinical use [121,122,126]. Additionally, the need for GMP-compliant cell sources and rigorous quality control further elevates costs. Translational challenges include the difficulty of recapitulating the complex epithelial–mesenchymal interactions and microenvironmental cues necessary for long-term follicle function and cycling. While vitro follicle-like structures can be generated and even induce hair shaft formation in animal models, these constructs often fail to sustain normal hair cycling and integration in vivo [9,132,133]. The mismatch between in vitro morphology and in vivo functionality is attributed to incomplete microenvironmental reconstitution, insufficient vascularization, and the lack of proper signaling for hair cycle regulation [120].
The field continues to progress toward functional reconstruction of hair follicles, with ongoing research focused on optimizing cell sources, biomaterials, and microenvironmental cues [91,132,134]. At present, organoids, iPSC-derived follicles, and 3D-bioprinted constructs remain preclinical; human durability, safety, and scalability are unknown, and no Phase III clinical trials have been conducted.
6. Cloning for Alopecia
The hair follicle is a complete mini-organ, making it a powerful model for research in stem cell biology, organ regeneration, and ultimately cloning strategies to restore hair. In this context, “cloning” does not mean genetic duplication, but rather the expansion of follicle-inductive cells outside the body and their use to form new, healthy follicles in areas affected by alopecia (Figure 4). The overarching goal is to create a renewable, reliable source of transplantable follicles, moving beyond the donor limitations of traditional hair transplantation [133].
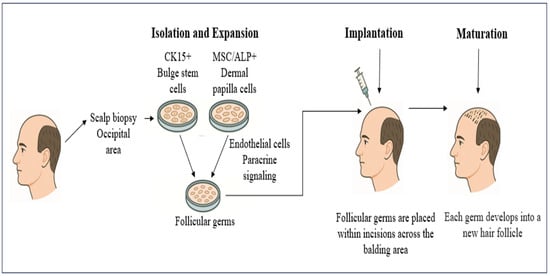
Figure 4.
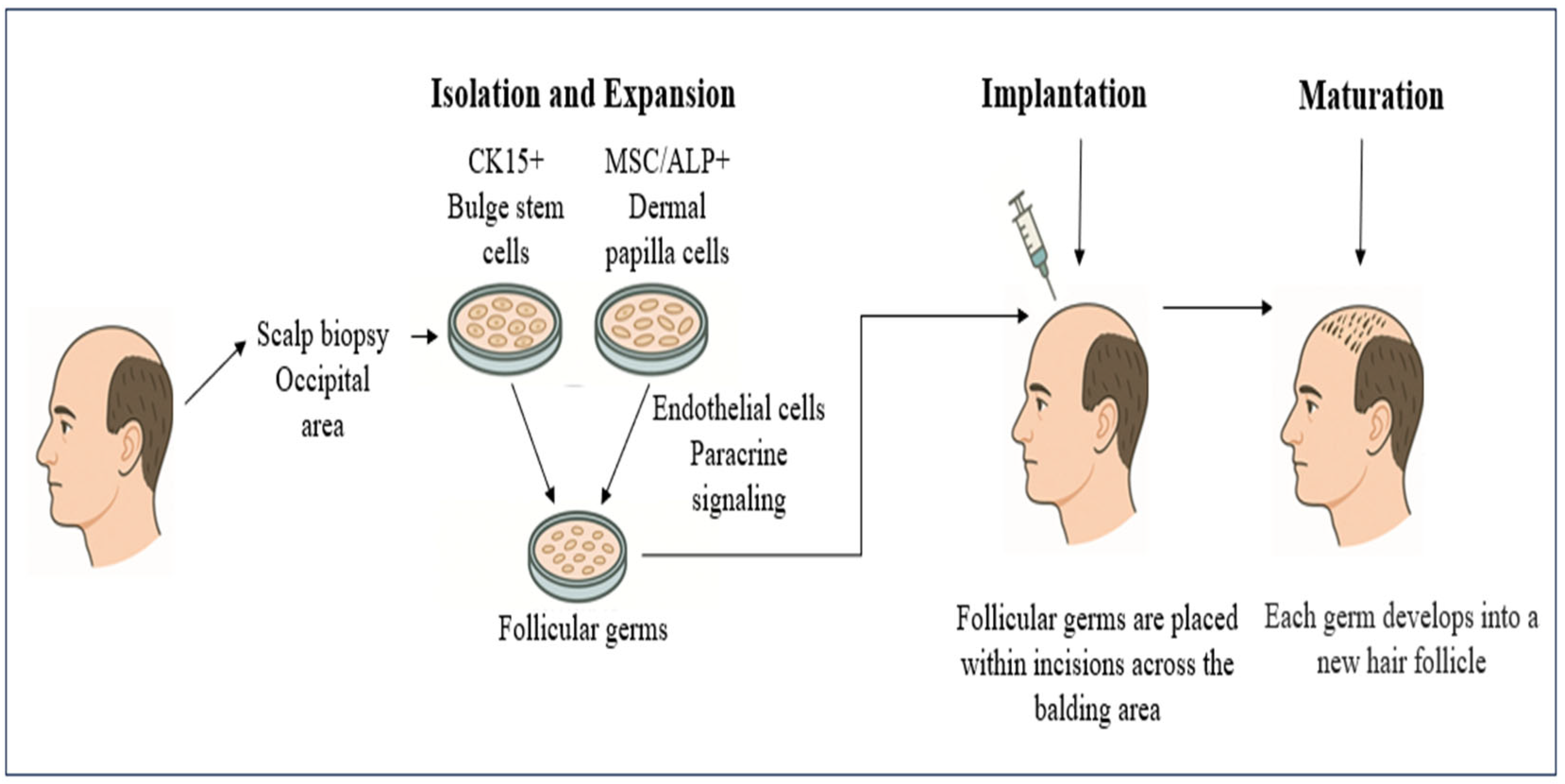
Hair cloning workflow for androgenic alopecia. Hair cloning protocols described in the recent literature generally [135] follow a multi-step process beginning with a small punch biopsy from the permanent occipital scalp to harvest healthy hair follicles. From these follicles, two primary stem cell populations, dermal papilla cells (mesenchymal stem cells, MSCs) and outer root sheath cells (epithelial stem cells), are isolated and expanded separately under conditions that preserve their hair-inductive properties. These stem cells, along with endothelial cells, are then recombined in three-dimensional micro-scaffolds or hanging-drop spheroids to form miniature follicular “germs” that replicate early hair-bud architecture. After in vitro quality control, such as alkaline phosphatase positivity (ALP+) in dermal papilla cells, K15 expression in epithelial cells, and confirmation of cell viability and structural integrity, the germs loaded into fine-gauge needles and implanted via microincisions into balding scalp regions. Upon engraftment, each germ matures into a fully functional hair follicle capable of normal cycling and fiber production, potentially providing an essentially unlimited donor supply. This approach remains at the preclinical or early-phase stage, with limited published data, and is not yet approved for clinical use.
6.1. Experimental Approaches
Early laboratory studies demonstrated that dermal papilla (DP) cells, the key inductive mesenchyme of the follicle, lose their hair-forming power in two-dimensional culture. However, this power can be restored when DP cells are cultured as three-dimensional spheroids, which successfully induced de novo hair follicles in preclinical models [132]. Parallel work with human pluripotent stem cells (hPSCs) has generated skin organoids containing hair follicles, which, when grafted onto immunodeficient mice, formed hair-bearing skin [136]. Another strategy, termed the organ germ method, recombines epithelial and mesenchymal cells to bioengineer follicular “germs” that can develop into innervated, cycling follicles after transplantation [133].
More advanced approaches include assembling dermal papilla, keratinocyte, and endothelial cells into three-cell aggregates, which significantly improved hair yield and activated anagen-associated genes such as LEF1 and VEGF in animal models [135]. In addition, direct reprogramming methods have been developed to convert human fibroblasts into DP-like cells using single small molecules, providing a potentially scalable source of hair-inductive cells [137].
6.2. Clinical Translation
Several early human programs attempted to move hair cloning toward the clinic. Intercytex (ICX-TRC) tested autologous DP cell injections, showing proof-of-concept hair growth but with variable and limited outcomes, preventing approval [138,139]. Aderans Research Institute advanced similar DP-based strategies through multiple clinical trials but eventually discontinued its program, although its innovations continue to inform newer companies [139,140,141,142,143,144]. Taken together, these efforts indicate that hair-follicle “cloning” remains at the preclinical and early-clinical stage, with no approved products and no completed Phase III trials.
A key figure in the development of follicle cell–based therapies is Kevin McElwee, an academic scientist and co-founder of RepliCel Life Sciences. His pioneering work on isolating and expanding dermal sheath cup (DSC) cells laid the foundation for the first-in-human studies of autologous cell injections for AGA [145]. The RCH-01 program, originally developed by McElwee and colleagues at the University of British Columbia and advanced through RepliCel, was later licensed to Shiseido in Japan, enabling a series of early clinical trials. A pivotal milestone came with a randomized, placebo-controlled, double-blind phase II study published in JAAD [146], which evaluated DSC injections in 65 patients with androgenetic alopecia. Small scalp biopsies were harvested, expanded in culture, and injected into four randomized treatment sites per participant. At the 3.0 × 105 cell dose, treated sites demonstrated significant increases in hair density and cumulative hair diameter at 6 and 9 months compared with placebo, while no major adverse events were reported. Importantly, the observed benefits appeared to result from the revitalization of miniaturized follicles rather than true de novo follicle formation, and effects diminished by 12 months, suggesting the need for repeat dosing or combination strategies.
A subsequent multi-center, open-label trial involving 36 participants reported that two rounds of DSC injections led to global photographic improvement in approximately 30% of patients after 12 months [147]. Mechanistic studies suggest that treatment outcomes may depend on the molecular phenotype of the injected cells: DSC subpopulations enriched for integrin α6 (ITGA6) expression demonstrated higher migratory activity and better hair growth outcomes, while those expressing smooth muscle-related genes such as SRF, CALD1, and ACTA2 correlated with reduced efficacy [148]. These findings underscore both the promise and the challenges of DSC-based therapies, showing that cultured follicular support cells can indeed rescue miniaturized follicles in humans, but that durability, consistency, and optimization of cell populations remain major hurdles before widespread clinical adoption [149]. (Study context: randomized, placebo-controlled, double-blind phase II trial (n = 65) of dermal sheath cup cells showed significant gains at 6–9 months vs. placebo; effects diminished by ~12 months.)
At present, Stemson Therapeutics is advancing induced pluripotent stem cell (iPSC)-derived engineered follicular units (EFUs), which have successfully generated human hair follicles in humanized mouse models and are slated for early human trials in 2025–2026 [90]. In parallel, the RIKEN/Organ Tech group in Japan has pioneered organoid-based follicle regeneration platforms, producing robust cycling follicles in mice. Meanwhile, HairClone (UK) has initiated follicle banking, cryopreserving patient follicles for future expansion and reinjection, though this remains preclinical [150].
6.3. Challenges
Despite clear progress, significant barriers remain. (1) DP and dermal sheath cells rapidly lose their hair-inductive signature in standard culture, making 3D culture systems and optimized signaling environments essential [132]. (2) Follicles are complex, requiring epithelial–mesenchymal crosstalk, vascularization, innervation, and correct orientation to yield natural hair growth. Organoid and organ-germ systems attempt to mimic this, but human translation is incomplete [133]. (3) Living, multi-cell constructs must be reproducible, cost-effective, and compliant with Good Manufacturing Practice (GMP) standards, which is a major hurdle. (4) In the United States, the FDA classifies any cultured cell or exosome-based therapy for hair restoration as an unapproved investigational biologic. No such product has received approval to date. Similarly, in Europe, the EMA regulates these products as Advanced Therapy Medicinal Products (ATMPs), requiring rigorous safety and efficacy data before approval [151].
6.4. Current Status and Outlook
At the biological level, we now know that human cells can be guided into forming follicle-like structures using approaches such as DP spheroids, hPSC-derived skin organoids, and organ germ engineering [133,135]. Translationally, first-generation trials by Intercytex and Aderans demonstrated feasibility but lacked durable, consistent results. Newer approaches using iPSCs, direct reprogramming, and organoid-based EFUs offer a clearer roadmap for reproducibility and eventual large-scale clinical use [90,137].
Nevertheless, most evidence remains confined to animal studies and small, short-term human cohorts. Long-term safety data, standardized protocols, and large controlled trials are missing. Regulatory agencies, as well as the International Society of Hair Restoration Surgery (ISHRS), stress that hair cloning remains experimental and investigational, not a proven therapy [152].
If successful, a future workflow might involve taking a small scalp biopsy or generating iPSCs from the patient, expanding the necessary follicular cells in a 3D environment, assembling them into engineered follicular units, and implanting them as grafts. Unlike traditional hair transplantation, this would create new follicles rather than redistribute existing ones, potentially solving the donor limitation once and for all.
7. Discussion
The field of hair restoration is undergoing substantial change, driven by advances in regenerative medicine, biomaterials, and bioengineering. Emerging technologies, ranging from refined transplantation techniques and robotic implantation systems to exosome- and stem-cell-based approaches, are expanding what is clinically feasible. Over the next decade, these modalities may prove safer and more accessible, contingent on adequately powered trials and formal regulatory review. Automation, artificial intelligence, and reproducible regenerative formulations may reduce costs and broaden access, moving therapies from specialized centers toward routine dermatologic practice (Table 4).

Table 4.
Hair Restoration Technologies Overview.
To integrate the signaling pathways discussed, we provide a mechanistic synthesis (Table 1, Figure 1 and Figure 2 and Supplements Tables S1 and S2) that maps each therapy to its dominant molecular lever—Wnt/β-catenin activation, BMP modulation, angiogenesis, mechano-transduction, immune-niche normalization, and dermal papilla signaling—and clarifies how these converge to influence telogen–anagen–catagen transitions. Complementing this framework, a comparative clinical overview (Table 1, Table 2, Table 3, Table 4, Table 5, Table 6 and Table 7) summarizes typical effect sizes, study quality, durability, and key limitations across principal pharmacologic and regenerative therapies. Together, these tables highlight convergent biological pathways while documenting heterogeneity in the strength and durability of clinical evidence among modalities.

Table 5.
Minimally Invasive and Device-Based Therapies.

Table 6.
Cell-based and secretome-based regenerative therapies.

Table 7.
Surgical and Advanced Regenerative Therapies.
A striking example of this evolution is Minoxidil. Once a purely topical treatment for AGA, it is now recognized to act systemically as well. Low-dose oral Minoxidil has proven effective in promoting hair growth and is increasingly used off-label as a systemic therapy [153]. In vivo human scalp studies further show that long-term topical use can rejuvenate aged follicles in AGA skin, upregulating biomarkers such as Lamin B1, SIRT1, and collagen XVIIA, while reducing p16^INK4A and p-S6—indicating anti-aging effects within the follicle itself [169].
This evolution, from topical to systemic use, illustrates how established treatments are moving toward true follicular rejuvenation, not just symptomatic improvement.
At the same time, hair transplantation, the most established surgical approach, has undergone a major transformation. Innovations such as FUE and robot-assisted implantation have dramatically improved precision, graft survival, and esthetic outcomes. Automated follicle harvesting and AI-guided donor–recipient mapping have reduced procedure time and scarring, while bioengineered scaffolds and exosome- or platelet-rich preservation media enhance graft viability. Together, these developments are shifting transplantation from a mechanical procedure to a bio-optimized regenerative intervention, bridging traditional surgery with tissue engineering. In the near future, combining transplantation with stem-cell activation or growth-factor therapy could yield denser, more natural regrowth while lowering costs and broadening accessibility.
Simultaneously, progress in follicle biology is charting the course toward genuine regeneration. Work on the Wnt/β-catenin pathway, notably by Ralf Paus and colleagues, has shown that blocking the Wnt inhibitor SFRP1 enhances hair-shaft production and delays follicle regression ex vivo [170]. This insight originated from studies on cyclosporine A (CsA), a well-known immunosuppressant long observed to promote hair growth. CsA’s stimulatory effect was traced not to immune suppression but to downregulation of SFRP1, thereby releasing Wnt/β-catenin signaling from inhibition. This discovery bridges pharmacology and regenerative biology, transforming incidental drug observations into target-driven follicle regeneration.
In parallel, Janus kinase (JAK) inhibitors have emerged as powerful tools for immune-mediated AA [171]. While their use in AGA remains limited, they exemplify how molecularly targeted therapies can reshape the therapeutic landscape. In time, such agents may merge synergistically with regenerative or gene-based platforms to deliver more comprehensive outcomes.
Beyond molecular modulation, recent years have witnessed groundbreaking bioengineering advances. Techniques in 3D bioprinting, hair-follicle organoid culture, and tissue-engineered skin constructs now enable partial or even complete follicle reformation in the laboratory. Multicellular constructs combining dermal papilla cells, fibroblasts, keratinocytes, and melanocytes within collagen or hydrogel matrices have generated vascularized, pigmented, hair-bearing skin in preclinical models. Developments such as iPSC-derived dermal papilla-like cells, dermal sheath cup (DSC) cell injections, and follicle organ germs mark major steps toward clinical follicle neogenesis. Combined with optimized extracellular matrices and controlled Wnt activation, these techniques could eventually restore full follicular cycling and pigmentation in humans.
The potential of exosome-based therapy is also increasingly evident. Extracellular vesicles from dermal papilla or mesenchymal stem cells can transfer microRNAs and growth factors that reactivate dormant follicles, stimulate angiogenesis, and modulate immune responses, offering a cell-free regenerative option with fewer safety concerns and easier scalability. Exosome and MSC-secretome therapies are therefore best considered experimental, early-phase interventions; published clinical studies involve small pilot RCTs and cohorts with short follow-up, and Phase III data or regulatory approvals for hair loss are not yet available.
Looking further ahead, even the meaning of alopecia may evolve. With ongoing advances in gene editing and molecular repair, permanent prevention of follicular miniaturization may become achievable, potentially rendering AGA and similar disorders obsolete. At the same time, changing social attitudes may redefine the importance of hair itself: as technologies that democratize appearance and identity emerge, the decision to restore or embrace baldness may shift from medical necessity to personal expression.
Ultimately, the convergence of pharmacologic innovation (Minoxidil, Wnt modulators), molecular targeting (JAK inhibitors), and regenerative engineering (organoids, exosomes, iPSC-derived follicles) highlights active preclinical and early clinical investigation, with uncertain clinical effectiveness and durability pending larger, controlled trials.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/cosmetics13010019/s1, Table S1: Mechanistic Integration (Part 1); Table S2: Mechanistic Integration (Part 2).
Author Contributions
Conceptualization, A.G. and R.L.B.; methodology, A.G. and R.L.B.; software, A.G. and R.L.B.; validation, A.G. and R.L.B.; formal analysis, A.G. and R.L.B.; investigation, A.G. and R.L.B.; resources, A.G. and R.L.B.; data curation, A.G. and R.L.B.; writing—original draft preparation, A.G. and R.L.B.; writing—review and editing, A.G. and R.L.B.; visualization, A.G. and R.L.B.; supervision, A.G. and R.L.B.; project administration, A.G. and R.L.B.; funding acquisition, A.G. and R.L.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Cash, T.F. The Psychosocial Consequences of Androgenetic Alopecia: A Review of the Research Literature. Br. J. Dermatol. 1999, 141, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.W.; Ryou, S.; Jeong, J.H.; Lee, J.W.; Lee, K.J.; Lee, S.B.; Shin, H.T.; Byun, J.W.; Shin, J.; Choi, G.S. The Quality of Life and Psychosocial Impact on Female Pattern Hair Loss. Ann. Dermatol. 2024, 36, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Tsuboi, R.; Itami, S.; Inui, S.; Ueki, R.; Katsuoka, K.; Kurata, S.; Kono, T.; Saito, N.; Manabe, M.; Yamazaki, M.; et al. Guidelines for the Management of Androgenetic Alopecia (2010). J. Dermatol. 2012, 39, 113–120. [Google Scholar] [CrossRef]
- Devjani, S.; Ezemma, O.; Kelley, K.J.; Stratton, E.; Senna, M. Androgenetic Alopecia: Therapy Update. Drugs 2023, 83, 701–715. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.; Abdin, R.; Gaumond, S.I.; Issa, N.T.; Jimenez, J.J. Treatment of Androgenetic Alopecia: Current Guidance and Unmet Needs. Clin. Cosmet. Investig. Dermatol. 2023, 16, 1387–1406. [Google Scholar] [CrossRef]
- Gonul, M.; Cemil, B.C.; Ayvaz, H.H.; Cankurtaran, E.; Ergin, C.; Gurel, M.S. Comparison of Quality of Life in Patients with Androgenetic Alopecia and Alopecia Areata. An. Bras. Dermatol. 2018, 93, 651–658. [Google Scholar] [CrossRef]
- Aukerman, E.L.; Jafferany, M. The Psychological Consequences of Androgenetic Alopecia: A Systematic Review. J. Cosmet. Dermatol. 2023, 22, 89–95. [Google Scholar] [CrossRef]
- Mehta, A.; Motavaf, M.; Raza, D.; McLure, A.J.; Osei-Opare, K.D.; Bordone, L.A.; Gru, A.A. Revolutionary Approaches to Hair Regrowth: Follicle Neogenesis, Wnt/β-Catenin Signaling, and Emerging Therapies. Cells 2025, 14, 779. [Google Scholar] [CrossRef]
- Chu, X.; Zhou, Z.; Qian, X.; Shen, H.; Cheng, H.; Zhang, J. Functional Regeneration Strategies of Hair Follicles: Advances and Challenges. Stem Cell Res. Ther. 2025, 16, 77. [Google Scholar] [CrossRef]
- Al Ameer, M.A.; Alnajim, A.T.; Al Ameer, A.; Alsalman, Z.; Al Ameer, G.A.; Alnajim, S.T.; Alghamdi, A.A.; Moideen, R.; Al Hadi, E.M. Exosomes and Hair Regeneration: A Systematic Review of Clinical Evidence Across Alopecia Types and Exosome Sources. Clin. Cosmet. Investig. Dermatol. 2025, 18, 2215. [Google Scholar] [CrossRef]
- Gupta, A.K.; Wang, T.; Rapaport, J.A. Systematic Review of Exosome Treatment in Hair Restoration: Preliminary Evidence, Safety, and Future Directions. J. Cosmet. Dermatol. 2023, 22, 2424–2433. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Han, Y.T.; Xie, J.L.; Liu, R.Q.; Zhao, B.; Zhang, X.L.; Zhang, J.; Zhang, J. Mesenchymal Stem Cell Exosomes Enhance the Development of Hair Follicle to Ameliorate Androgenetic Alopecia. World J. Stem Cells 2025, 17, 102088. [Google Scholar] [CrossRef]
- Poddar, N.; Aratikatla, A.; Gupta, A. Therapeutic Potential of Stem Cell-Derived Exosomes in Hair Regeneration: A Systematic Review. World J. Stem Cells 2025, 17, 108519. [Google Scholar] [CrossRef]
- Wan, J.; Kim, S.B.; Cartier, H.; Garson, S.; Frank, K.; Haykal, D.; Yi, K.H. A Prospective Study of Exosome Therapy for Androgenetic Alopecia. Aesthet. Plast. Surg. 2025, 49, 3151–3156. [Google Scholar] [CrossRef]
- Ersan, M.; Ozer, E.; Akin, O.; Tasli, P.N.; Sahin, F. Effectiveness of Exosome Treatment in Androgenetic Alopecia: Outcomes of a Prospective Study. Aesthet. Plast. Surg. 2024, 48, 4262–4271. [Google Scholar] [CrossRef]
- Koumprentziotis, I.A.; Kroumpouzos, E.; Delavar, S.; Kroumpouzos, G. Microneedling Radiofrequency Followed by Topical Exosome Application Therapy for Pattern Hair Loss: A Scoping Review and Prospective Study. Clin. Dermatol. 2025, 43, 742–751. [Google Scholar] [CrossRef]
- Xiao, L.; Zhang, F.; Chen, Y.; Tang, Y.; Usman, M.; Zhang, J. Microneedle-Mediated Therapies in Hair Loss: Applications, Mechanisms, and Future Directions. Plast. Aesthet. Res. 2025, 12, 25. [Google Scholar] [CrossRef]
- Nam, S.Y.; Jain, S.K.; Kurian, A.G.; Jeong, I.; Park, B.C.; Ban, K.; Knowles, J.C.; Kim, H.W. Hair Regeneration: Mechano-Activation and Related Therapeutic Approaches. J. Tissue Eng. 2025, 16, 20417314251362398. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, Y.; Zhao, J.; Deng, T.; Ben, Y.; Lu, R.; Zhou, X.; Yan, R.; Chen, X.; Zhang, J.V.; et al. Subcutaneous Injection of Genetically Engineered Exosomes for Androgenic Alopecia Treatment. Front. Bioeng. Biotechnol. 2025, 13, 1614090. [Google Scholar] [CrossRef]
- Zhang, J.; Peng, J.; Fan, Z.; Wang, H.; Wen, L.; Miao, Y.; Chai, Y.; Hu, Z.; Chen, R. Therapeutic Potential of Isoproterenol in Androgenetic Alopecia: Activation of Hair Follicle Stem Cells via the PI3K/AKT/β-Catenin Signaling Pathway. Stem Cell Res. Ther. 2025, 16, 306. [Google Scholar] [CrossRef]
- Jin, H.; Zou, Z.; Chang, H.; Shen, Q.; Liu, L.; Xing, D. Photobiomodulation Therapy for Hair Regeneration: A Synergetic Activation of β-Catenin in Hair Follicle Stem Cells by ROS and Paracrine WNTs. Stem Cell Rep. 2021, 16, 1568–1583. [Google Scholar] [CrossRef]
- Ma, X.; Zhu, X.; Lv, S.; Yang, C.; Wang, Z.; Liao, M.; Zhou, B.; Zhang, Y.; Sun, S.; Chen, P.; et al. 3D Bioprinting of Prefabricated Artificial Skin with Multicomponent Hydrogel for Skin and Hair Follicle Regeneration. Theranostics 2025, 15, 2933–2950. [Google Scholar] [CrossRef]
- Makkar, J.; Flores, J.; Matich, M.; Duong, T.T.; Thompson, S.M.; Du, Y.; Busch, I.; Phan, Q.M.; Wang, Q.; Delevich, K.; et al. Deep Hair Phenomics: Implications in Endocrinology, Development, and Aging. J. Investig. Dermatol. 2025, 145, 800–811.e8. [Google Scholar] [CrossRef] [PubMed]
- Agramunt, J.; Parke, B.; Mena, S.; Ubels, V.; Jimenez, F.; Williams, G.; Rhodes, A.D.Y.; Limbu, S.; Hexter, M.; Knight, L.; et al. Mechanical Stimulation of Human Hair Follicle Outer Root Sheath Cultures Activates Adjacent Sensory Neurons. Sci. Adv. 2023, 9, eadh3273. [Google Scholar] [CrossRef] [PubMed]
- Hamann, W. Mammalian Cutaneous Mechanoreceptors. Prog. Biophys. Mol. Biol. 1995, 64, 81–104. [Google Scholar] [CrossRef]
- Lasisi, T.; Smallcombe, J.W.; Kenney, W.L.; Shriver, M.D.; Zydney, B.; Jablonski, N.G.; Havenith, G. Human Scalp Hair as a Thermoregulatory Adaptation. Proc. Natl. Acad. Sci. USA 2023, 120, e2301760120. [Google Scholar] [CrossRef]
- Li, W.Q.; Cho, E.; Han, J.; Weinstock, M.A.; Qureshi, A.A. Male Pattern Baldness and Risk of Incident Skin Cancer in a Cohort of Men. Int. J. Cancer 2016, 139, 2671. [Google Scholar] [CrossRef]
- Ho, C.H.; Sood, T.; Zito, P.M. Androgenetic Alopecia; StatPearls: Tampa, FL, USA, 2024. [Google Scholar]
- Schielein, M.C.; Tizek, L.; Ziehfreund, S.; Sommer, R.; Biedermann, T.; Zink, A. Stigmatization Caused by Hair Loss—A Systematic Literature Review. J. Dtsch. Dermatol. Ges. 2020, 18, 1357–1368. [Google Scholar] [CrossRef]
- Duverger, O.; Morasso, M.I. Epidermal Patterning and Induction of Different Hair Types during Mouse Embryonic Development. Birth Defects Res. C Embryo Today 2009, 87, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Millar, S.E. Molecular Mechanisms Regulating Hair Follicle Development. J. Investig. Dermatol. 2002, 118, 216–225. [Google Scholar] [CrossRef]
- Schmidt-Ullrich, R.; Paus, R. Molecular Principles of Hair Follicle Induction and Morphogenesis. BioEssays 2005, 27, 247–261. [Google Scholar] [CrossRef]
- Grymowicz, M.; Rudnicka, E.; Podfigurna, A.; Napierala, P.; Smolarczyk, R.; Smolarczyk, K. Hormonal Effects on Hair Follicles. Int. J. Mol. Sci. 2020, 21, 5342. [Google Scholar] [CrossRef]
- Hasan, R.; Juma, H.; Eid, F.A.; Alaswad, H.A.; Ali, W.M.; Aladraj, F.J.; Hassan, R.; Mohsen, F.A.; Ali, W. Effects of Hormones and Endocrine Disorders on Hair Growth. Cureus 2022, 14, e32726. [Google Scholar] [CrossRef]
- Mendelsohn, A.R.; Larrick, J.W. The Danger of Being Too Sympathetic: Norepinephrine in Alzheimer’s Disease and Graying of Hair. Rejuvenation Res. 2020, 23, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Neau, J.; Godeneche, G.; Mathis, S.; Godeneche-Mathis, G. Neurodermatology. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2014; Volume 121, pp. 1561–1594. [Google Scholar] [CrossRef]
- Thom, E. Stress and the Hair Growth Cycle: Cortisol-Induced Hair Growth Disruption. J. Drugs Dermatol. 2016, 15, 1001–1004. [Google Scholar] [PubMed]
- Almashali, M.; Alekrish, K.; Shadid, A. The Use of 2D:4D Digit Ratio as a Predictor of Androgenetic Alopecia: A Review. Dermatol. Pract. Concept. 2023, 13, e2023043. [Google Scholar] [CrossRef]
- Liu, F.; Hamer, M.A.; Heilmann, S.; Herold, C.; Moebus, S.; Hofman, A.; Uitterlinden, A.G.; Nöthen, M.M.; Van Duijn, C.M.; Nijsten, T.E.C.; et al. Prediction of Male-Pattern Baldness from Genotypes. Eur. J. Hum. Genet. 2016, 24, 895–902. [Google Scholar] [CrossRef]
- Wolff, H.; Fischer, T.W.; Blume-Peytavi, U. The Diagnosis and Treatment of Hair and Scalp Diseases. Dtsch. Arztebl. Int. 2016, 113, 377–386. [Google Scholar] [CrossRef]
- Chi, W.; Wu, E.; Morgan, B.A. Dermal Papilla Cell Number Specifies Hair Size, Shape, and Cycling and Its Reduction Causes Follicular Decline. Development 2013, 140, 1676–1683. [Google Scholar] [CrossRef]
- Driskell, R.R.; Giangreco, A.; Jensen, K.B.; Mulder, K.W.; Watt, F.M. Sox2-Positive Dermal Papilla Cells Specify Hair Follicle Type in Mammalian Epidermis. Development 2009, 136, 2815–2823. [Google Scholar] [CrossRef] [PubMed]
- Schlake, T. FGF Signals Specifically Regulate the Structure of Hair Shaft Medulla via IGF-Binding Protein 5. Development 2005, 132, 2981–2990. [Google Scholar] [CrossRef]
- Takeo, M.; Toyoshima, K.-E.; Fujimoto, R.; Iga, T.; Takase, M.; Ogawa, M.; Tsuji, T. Cyclical Dermal Micro-Niche Switching Governs the Morphological Infradian Rhythm of Mouse Zigzag Hair. Nat. Commun. 2023, 14, 4371. [Google Scholar] [CrossRef] [PubMed]
- Weger, N.; Schlake, T. Igf-I Signalling Controls the Hair Growth Cycle and the Differentiation of Hair Shafts. J. Investig. Dermatol. 2005, 125, 873–882. [Google Scholar] [CrossRef]
- Jeong, H.; Park, C.; Henao, R.; Kim, M.K. Deep Learning in Dermatology: A Systematic Review of Current Approaches, Outcomes, and Limitations. J. Dermatol. Innov. 2023, 3, 100150. [Google Scholar] [CrossRef]
- Kim, J.; Oh, I.; Lee, Y.N.; Lee, J.H.; Lee, Y.I.; Kim, J.H.; Lee, J.H. Predicting the Severity of Postoperative Scars Using Artificial Intelligence Based on Images and Clinical Data. Sci. Rep. 2023, 13, 13448. [Google Scholar] [CrossRef]
- Tschandl, P.; Rinner, C.; Apalla, Z.; Argenziano, G.; Codella, N.; Halpern, A.; Janda, M.; Lallas, A.; Longo, C.; Malvehy, J.; et al. Human–Computer Collaboration for Skin Cancer Recognition. Nat. Med. 2020, 26, 1220–1226. [Google Scholar] [CrossRef]
- Wei, M.L.; Tada, M.; So, A.; Torres, R. Artificial Intelligence and Skin Cancer. Front. Med. 2024, 11, 1331895. [Google Scholar] [CrossRef]
- Di Fraia, M.; Tieghi, L.; Magri, F.; Caro, G.; Michelini, S.; Pellacani, G.; Rossi, A. A Machine Learning Algorithm Applied to Trichoscopy for Androgenic Alopecia Staging and Severity Assessment. Dermatol. Pract. Concept. 2023, 13, e2023150. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Wang, Y.; Xu, H.; Xu, C.; Yang, X.; Nie, J.; Zhang, Z.; Li, Z.; Hou, W.; Jiang, Y. Deep Learning-Based Trichoscopic Image Analysis and Quantitative Model for Predicting Basic and Specific Classification in Male Androgenetic Alopecia. Acta Derm. Venereol. 2022, 102, 564. [Google Scholar] [CrossRef] [PubMed]
- Arteta, C.; Lempitsky, V.; Noble, J.A.; Zisserman, A. Learning to Detect Partially Overlapping Instances. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Portland, OR, USA, 23–28 June 2013. [Google Scholar] [CrossRef]
- Wang, S.; Yatagawa, T.; Suzuki, H.; Ohtake, Y. Image-Based Measurement of Individual Fiber Lengths for Randomly Oriented Short Fiber Composites. J. Nondestr. Eval. 2022, 41, 45. [Google Scholar] [CrossRef]
- Chai, J.; Zeng, H.; Li, A.; Ngai, E.W.T. Deep Learning in Computer Vision: A Critical Review of Emerging Techniques and Application Scenarios. Mach. Learn. Appl. 2021, 6, 100134. [Google Scholar] [CrossRef]
- Esteva, A.; Chou, K.; Yeung, S.; Naik, N.; Madani, A.; Mottaghi, A.; Liu, Y.; Topol, E.; Dean, J.; Socher, R. Deep Learning-Enabled Medical Computer Vision. NPJ Digit. Med. 2021, 4, 5. [Google Scholar] [CrossRef]
- Archila, J.; Manzanera, A.; Martínez, F. A Multimodal Parkinson Quantification by Fusing Eye and Gait Motion Patterns, Using Covariance Descriptors, from Non-Invasive Computer Vision. Comput. Methods Programs Biomed. 2022, 215, 106607. [Google Scholar] [CrossRef]
- Bae, H.; An, Y.K. Computer Vision-Based Statistical Crack Quantification for Concrete Structures. Measurement 2023, 211, 112628. [Google Scholar] [CrossRef]
- Mori, S.; Tachibana, Y.; Suzuki, M.; Harada, Y. Automatic Worm Detection to Solve Overlapping Problems Using a Convolutional Neural Network. Sci. Rep. 2022, 12, 8521. [Google Scholar] [CrossRef]
- Pun, T.B.; Neupane, A.; Koech, R.; Walsh, K. Detection and Counting of Root-Knot Nematodes Using YOLO Models with Mosaic Augmentation. Biosens. Bioelectron. X 2023, 13, 100283. [Google Scholar] [CrossRef]
- Toda, Y.; Okura, F.; Ito, J.; Okada, S.; Kinoshita, T.; Tsuji, H.; Saisho, D. Training Instance Segmentation Neural Network with Synthetic Datasets for Crop Seed Phenotyping. Commun. Biol. 2020, 3, 94. [Google Scholar] [CrossRef] [PubMed]
- Shakeel, C.S.; Khan, S.J.; Chaudhry, B.; Aijaz, S.F.; Hassan, U. Classification Framework for Healthy Hairs and Alopecia Areata: A Machine Learning (ML) Approach. Comput. Math. Methods Med. 2021, 2021, 1102083. [Google Scholar] [CrossRef]
- Shakeel, C.S.; Khan, S.J. Machine Learning (ML) Techniques as Effective Methods for Evaluating Hair and Skin Assessments: A Systematic Review. Proc. Inst. Mech. Eng. H 2024, 238, 132–148. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, M.S.; Sultan, T.; Jahan, N.; Mridha, M.F.; Safran, M.; Alfarhood, S.; Che, D. Leveraging Deep Neural Networks to Uncover Unprecedented Levels of Precision in the Diagnosis of Hair and Scalp Disorders. Skin Res. Technol. 2024, 30, e13660. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Ivanova, I.A.; Renaud, H.J. How Good Is Artificial Intelligence (AI) at Solving Hairy Problems? A Review of AI Applications in Hair Restoration and Hair Disorders. Dermatol. Ther. 2021, 34, e14811. [Google Scholar] [CrossRef] [PubMed]
- Gudobba, C.; Mane, T.; Bayramova, A.; Rodriguez, N.; Castelo-Soccio, L.; Ogunleye, T.A.; Taylor, S.C.; Cotsarelis, G.; Bernardis, E. Automating Hair Loss Labels for Universally Scoring Alopecia from Images: Rethinking Alopecia Scores. JAMA Dermatol. 2022, 159, 143. [Google Scholar] [CrossRef]
- Du, A.X.; Emam, S.; Gniadecki, R. Review of Machine Learning in Predicting Dermatological Outcomes. Front. Med. 2020, 7, 547105. [Google Scholar] [CrossRef]
- Jartarkar, S.R.; Patil, A.; Waskiel-Burnat, A.; Rudnicka, L.; Starace, M.; Grabbe, S.; Goldust, M. Artificial Intelligence in Hair and Nail Disorders. J. Drugs Dermatol. 2022, 21, 1049–1052. [Google Scholar] [CrossRef]
- Choy, S.P.; Kim, B.J.; Paolino, A.; Tan, W.R.; Lim, S.M.L.; Seo, J.; Tan, S.P.; Francis, L.; Tsakok, T.; Simpson, M.; et al. Systematic Review of Deep Learning Image Analyses for the Diagnosis and Monitoring of Skin Disease. NPJ Digit. Med. 2023, 6, 180. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Koban, K.C.; Schenck, T.L.; Giunta, R.E.; Li, Q.; Sun, Y. Artificial Inelligence in Dermatology Image Analysis: Current Developments and Future Trends. J. Clin. Med. 2022, 11, 6826. [Google Scholar] [CrossRef]
- Wongvibulsin, S.; Yan, M.J.; Pahalyants, V.; Murphy, W.; Daneshjou, R.; Rotemberg, V. Current State of Dermatology Mobile Applications with Artificial Intelligence Features. JAMA Dermatol. 2024, 160, 646–650. [Google Scholar] [CrossRef]
- Nahm, W.J.; Sohail, N.; Burshtein, J.; Goldust, M.; Tsoukas, M. Artificial Intelligence in Dermatology: A Comprehensive Review of Approved Applications, Clinical Implementation, and Future Directions. Int. J. Dermatol. 2025, 64, 1568–1583. [Google Scholar] [CrossRef] [PubMed]
- Rundle, C.W.; Hollingsworth, P.; Dellavalle, R.P. Artificial Intelligence in Dermatology. Clin. Dermatol. 2021, 39, 657–666. [Google Scholar] [CrossRef]
- Chen, M.; Zhou, A.E.; Jain, N.; Gronbeck, C.; Feng, H.; Grant-Kels, J.M. Ethics of Artificial Intelligence in Dermatology. Clin. Dermatol. 2024, 42, 313–316. [Google Scholar] [CrossRef]
- MacKenzie, J.; Coric, C.; Kim, Y.W.; Lee, C.; Grant-Kels, J.M. Current Applications of Artificial Intelligence in Dermatology Offices and Potential Ethical Landmines. Clin. Dermatol. 2025, 43, 732–737. [Google Scholar] [CrossRef]
- Gomolin, A.; Netchiporouk, E.; Gniadecki, R.; Litvinov, I.V. Artificial Intelligence Applications in Dermatology: Where Do We Stand? Front. Med. 2020, 7, 100. [Google Scholar] [CrossRef] [PubMed]
- Young, A.T.; Xiong, M.; Pfau, J.; Keiser, M.J.; Wei, M.L. Artificial Intelligence in Dermatology: A Primer. J. Investig. Dermatol. 2020, 140, 1504–1512. [Google Scholar] [CrossRef]
- Daneshjou, R.; Smith, M.P.; Sun, M.D.; Rotemberg, V.; Zou, J. Lack of Transparency and Potential Bias in Artificial Intelligence Data Sets and Algorithms: A Scoping Review. JAMA Dermatol. 2021, 157, 1362–1369. [Google Scholar] [CrossRef]
- Pereira, J.C.; Pereira Filho, J.C.; Pereira, J.P.C. Megasessions for Robotic Hair Restoration. J. Drugs Dermatol. 2016, 15, 1407–1412. [Google Scholar]
- Kanayama, K.; Kato, H.; Mori, M.; Sakae, Y.; Okazaki, M. Robotically Assisted Recipient Site Preparation in Hair Restoration Surgery: Surgical Safety and Clinical Outcomes in 31 Consecutive Patients. Dermatol. Surg. 2021, 47, 1365–1370. [Google Scholar] [CrossRef]
- Zhu, Y.; Yang, K.; Lin, J.M.; Ni, C.; Zhang, Y.; Li, Z.; Liu, Q.; Zhou, Y.; Lin, J.; Wu, W. A Comparative Study on the Application of Robotic Hair Restoration Technology Versus Traditional Follicular Unit Excision in Male Androgenetic Alopecia. J. Cosmet. Dermatol. 2024, 23, 4213–4222. [Google Scholar] [CrossRef]
- Thuangtong, R.; Suthakorn, J. Design, Proof-of-Concept of Single Robotic Hair Transplant Mechanisms for Both Harvest and Implant of Hair Grafts. Comput. Struct. Biotechnol. J. 2024, 24, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Thuangtong, R.; Anantawilailekha, O.; Prasertsin, P.; Suthakorn, J. Development and Evaluation of an Integrated Image-Guided Robotic System for Hair Transplant Surgery. Comput. Struct. Biotechnol. J. 2025, 28, 80–93. [Google Scholar] [CrossRef]
- Chauhan, K.; Tandon, M.; Kumar, A.; Taneja, N.; Hamid, S.A.T. A Comprehensive Review of Evolution of Advanced Follicular Unit Excision Systems. J. Cutan. Aesthet. Surg. 2025, 18, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, R.M.; Wolfeld, M.B. Robotic Follicular Unit Graft Selection. Dermatol. Surg. 2016, 42, 710–714. [Google Scholar] [CrossRef]
- Queen, D.; Avram, M.R. Hair Transplantation: State of the Art. Dermatol. Surg. 2025, 51, 874–881. [Google Scholar] [CrossRef]
- Moeckel, C.; Chacharidakis, G.; Balasis, S.; Queen, D.; Avram, M.R.; Panagiotopoulou, G. From Scalp Flaps to Follicular Units: A Historical Perspective on Hair Transplantation Techniques. Dermatol. Surg. 2025, 51, 1011–1016. [Google Scholar] [CrossRef]
- Rose, P.T.; Nusbaum, B. Robotic Hair Restoration. Dermatol. Clin. 2014, 32, 97–107. [Google Scholar] [CrossRef]
- Shin, J.W.; Kwon, S.H.; Kim, S.A.; Kim, J.Y.; Na, J.I.; Park, K.C.; Huh, C.H. Characteristics of Robotically Harvested Hair Follicles in Koreans. J. Am. Acad. Dermatol. 2015, 72, 146–150. [Google Scholar] [CrossRef]
- Rogers, N.E.; Callender, V.D. Advances and Challenges in Hair Restoration of Curly Afrocentric Hair. Dermatol. Clin. 2014, 32, 163–171. [Google Scholar] [CrossRef]
- Pinto, A.; Terskikh, A.V. The Rise of Induced Pluripotent Stem Cell Approach to Hair Restoration. Plast. Reconstr. Surg. 2021, 148, 39S–46S. [Google Scholar] [CrossRef]
- Cao, X.; Lu, M.; Li, N.; Cai, L.; Wang, Y.; Zhao, Y. Emerging Biomedical Engineering Strategies for Hair Follicle Regeneration. Bioact. Mater. 2025, 53, 84. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Song, S.Y.; Sung, J.H. Recent Advances in Drug Development for Hair Loss. Int. J. Mol. Sci. 2025, 26, 3461. [Google Scholar] [CrossRef] [PubMed]
- Tao, N.; Sun, Q.; Ying, Y.; Wang, Y.; Gao, J. Dermal T Cell Immunity and Key Regulatory Signaling Pathways: Implications in Immune-Mediated Alopecia and Hair Regeneration. Genes Dis. 2025, 12, 101518. [Google Scholar] [CrossRef] [PubMed]
- Lim, C.; Lim, J.; Choi, S. Wound-Induced Hair Follicle Neogenesis as a Promising Approach for Hair Regeneration. Mol. Cells 2023, 46, 573–582. [Google Scholar] [CrossRef]
- Lee, S.H. Molecular Signaling Pathways in Wound-Induced Hair-Follicle Neogenesis. Cells 2025, 14, 440. [Google Scholar] [CrossRef]
- Oak, A.S.W.; Cotsarelis, G. Wound-Induced Hair Neogenesis: A Portal to the Development of New Therapies for Hair Loss and Wound Regeneration. Cold Spring Harb. Perspect. Med. 2023, 15, a041239. [Google Scholar] [CrossRef]
- Bellani, D.; Patil, R.; Prabhughate, A.; Shahare, R.; Gold, M.; Kapoor, R.; Shome, D. Pathophysiological Mechanisms of Hair Follicle Regeneration and Potential Therapeutic Strategies. Stem Cell Res. Ther. 2025, 16, 302. [Google Scholar] [CrossRef] [PubMed]
- Ryu, Y.C.; Park, J.; Kim, Y.R.; Choi, S.; Kim, G.U.; Kim, E.; Hwang, Y.; Kim, H.; Han, G.; Lee, S.H.; et al. CXXC5 Mediates DHT-Induced Androgenetic Alopecia via PGD2. Cells 2023, 12, 555. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Seo, S.H.; Lee, D.H.; Pi, L.Q.; Lee, W.S.; Choi, K.Y. Targeting of CXXC5 by a Competing Peptide Stimulates Hair Regrowth and Wound-Induced Hair Neogenesis. J. Investig. Dermatol. 2017, 137, 2260–2269. [Google Scholar] [CrossRef]
- Ryu, Y.C.; Lee, D.H.; Shim, J.; Park, J.; Kim, Y.R.; Choi, S.; Bak, S.S.; Sung, Y.K.; Lee, S.H.; Choi, K.Y. KY19382, a Novel Activator of Wnt/β-Catenin Signalling, Promotes Hair Regrowth and Hair Follicle Neogenesis. Br. J. Pharmacol. 2021, 178, 2533–2546. [Google Scholar] [CrossRef]
- Anudeep, T.C.; Jeyaraman, M.; Muthu, S.; Rajendran, R.L.; Gangadaran, P.; Mishra, P.C.; Sharma, S.; Jha, S.K.; Ahn, B.C. Advancing Regenerative Cellular Therapies in Non-Scarring Alopecia. Pharmaceutics 2022, 14, 612. [Google Scholar] [CrossRef]
- Krefft-Trzciniecka, K.; Piętowska, Z.; Nowicka, D.; Szepietowski, J.C. Human Stem Cell Use in Androgenetic Alopecia: A Systematic Review. Cells 2023, 12, 951. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Garcovich, S. Advances in Regenerative Stem Cell Therapy in Androgenic Alopecia and Hair Loss: Wnt Pathway, Growth-Factor, and Mesenchymal Stem Cell Signaling Impact Analysis on Cell Growth and Hair Follicle Development. Cells 2019, 8, 466. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, F.; Hu, Y.; Cong, X. Mechanisms and Clinical Progress of Adipose-Derived Stem Cells and Their Derivatives in the Treatment of Hair Loss. Stem Cell Res. Ther. 2025, 16, 439. [Google Scholar] [CrossRef]
- Legiawati, L.; Suseno, L.S.; Sitohang, I.B.S.; Yusharyahya, S.N.; Pawitan, J.A.; Liem, I.K.; Kurniawati, T.; Ardelia, A.; Paramastri, K. Combination of Adipose-Derived Stem Cell Conditioned Media and Minoxidil for Hair Regrowth in Male Androgenetic Alopecia: A Randomized, Double-Blind Clinical Trial. Stem Cell Res. Ther. 2023, 14, 210. [Google Scholar] [CrossRef]
- Tak, Y.J.; Lee, S.Y.; Cho, A.R.; Kim, Y.S. A Randomized, Double-Blind, Vehicle-Controlled Clinical Study of Hair Regeneration Using Adipose-Derived Stem Cell Constituent Extract in Androgenetic Alopecia. Stem Cells Transl. Med. 2020, 9, 839–849. [Google Scholar] [CrossRef]
- Zari, S. Efficacy of Adipocyte-Derived Stem Cells-Conditioned Media in Telogen Effluvium. Stem Cells Cloning 2023, 16, 77–89. [Google Scholar] [CrossRef] [PubMed]
- Chien, W.Y.; Huang, H.M.; Kang, Y.N.; Chen, K.H.; Chen, C. Stem Cell–Derived Conditioned Medium for Alopecia: A Systematic Review and Meta-Analysis. J. Plast. Reconstr. Aesthet. Surg. 2024, 88, 182–192. [Google Scholar] [CrossRef]
- Legiawati, L.; Suseno, L.S.; Sitohang, I.B.S.; Yusharyahya, S.N.; Fahira, A.; Ramadan, E.T.; Paramastri, K. Adipose-Derived Stem Cell Conditioned Medium for Hair Regeneration Therapy in Alopecia: A Review of Literature. Arch. Dermatol. Res. 2024, 316, 525. [Google Scholar] [CrossRef] [PubMed]
- Gan, Y.; Du, L.; Wang, H.; Li, K.; Fan, Z.; Sun, P.; Zhang, J.; Hu, Z.; Miao, Y. A Clinical Trial of Treating Androgenic Alopecia with Mesenchymal Stem Cell Suspension Derived from Autologous Hair Follicle. Plast. Reconstr. Surg. 2024, 154, 444e–450e. [Google Scholar] [CrossRef]
- Dhurat, R.; Sukesh, M.; Avhad, G.; Dandale, A.; Pal, A.; Pund, P. A Randomized Evaluator Blinded Study of Effect of Microneedling in Androgenetic Alopecia: A Pilot Study. Int. J. Trichol. 2013, 5, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sheng, Y.; Zeng, Y.; Hu, R.; Zhao, J.; Wang, W.; Yang, Q. Randomized Trial of Microneedling Combined with 2% Minoxidil Topical Solution for the Treatment of Female Pattern Hair Loss in a Chinese Population. J. Cosmet. Dermatol. 2022, 21, 6985–6991. [Google Scholar] [CrossRef]
- Ablon, G. An Exosome Regenerative Complex Plus Microneedling Promotes Hair Growth in Subjects with Self-Perceived Thinning Hair. J. Drugs Dermatol. 2025, 24, 988–994. [Google Scholar] [CrossRef]
- English, R.S.; Ruiz, S.; DoAmaral, P. Microneedling and Its Use in Hair Loss Disorders: A Systematic Review. Dermatol. Ther. 2022, 12, 41–60. [Google Scholar] [CrossRef]
- Ahmed, K.M.A.; Kozaa, Y.A.; Abuawwad, M.T.; Al-Najdawi, A.I.; Mahmoud, Y.W.; Ahmed, A.M.; Taha, M.J.J.; Fadhli, T.; Giannopoulou, A. Evaluating the Efficacy and Safety of Combined Microneedling Therapy versus Topical Minoxidil in Androgenetic Alopecia: A Systematic Review and Meta-Analysis. Arch. Dermatol. Res. 2025, 317, 528. [Google Scholar] [CrossRef]
- Mao, Y.; Liu, P.; Wei, J.; Xie, Y.; Zheng, Q.; Li, R.; Yao, J. Cell Therapy for Androgenetic Alopecia: Elixir or Trick? Stem Cell Rev. Rep. 2023, 19, 1785–1803. [Google Scholar] [CrossRef] [PubMed]
- Pozo-Pérez, L.; Tornero-Esteban, P.; López-Bran, E. Clinical and Preclinical Approach in AGA Treatment: A Review of Current and New Therapies in the Regenerative Field. Stem Cell Res. Ther. 2024, 15, 260. [Google Scholar] [CrossRef]
- Kang, B.Y.; Li, A.W.; Lee, M.H.; Wong, C.C.; Naseer, N.; Ibrahim, S.A.; Miller, C.H.; Keimig, E.L.; Poon, E.; Alam, M. The Safety and Efficacy of Autologous Adipose-Derived Stromal Vascular Fraction for Nonscarring Alopecia: A Systematic Review. Arch. Dermatol. Res. 2022, 314, 349–356. [Google Scholar] [CrossRef]
- Gentile, P.; Garcovich, S. Systematic Review of Platelet-Rich Plasma Use in Androgenetic Alopecia Compared with Minoxidil®, Finasteride®, and Adult Stem Cell-Based Therapy. Int. J. Mol. Sci. 2020, 21, 2702. [Google Scholar] [CrossRef]
- Abaci, H.E.; Coffman, A.; Doucet, Y.; Chen, J.; Jacków, J.; Wang, E.; Guo, Z.; Shin, J.U.; Jahoda, C.A.; Christiano, A.M. Tissue Engineering of Human Hair Follicles Using a Biomimetic Developmental Approach. Nat. Commun. 2018, 9, 5301. [Google Scholar] [CrossRef]
- Nanmo, A.; Yan, L.; Asaba, T.; Wan, L.; Kageyama, T.; Fukuda, J. Bioprinting of Hair Follicle Germs for Hair Regenerative Medicine. Acta Biomater. 2023, 165, 50–59. [Google Scholar] [CrossRef]
- Kang, D.; Liu, Z.; Qian, C.; Huang, J.; Zhou, Y.; Mao, X.; Qu, Q.; Liu, B.; Wang, J.; Hu, Z.; et al. 3D Bioprinting of a Gelatin-Alginate Hydrogel for Tissue-Engineered Hair Follicle Regeneration. Acta Biomater. 2023, 165, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Motter Catarino, C.; Cigaran Schuck, D.; Dechiario, L.; Karande, P. Incorporation of Hair Follicles in 3D Bioprinted Models of Human Skin. Sci. Adv. 2023, 9, eadg0297. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Qin, C.; Wu, J.; Zhuang, H.; Du, L.; Xu, J.; Wu, C. 3D Multicellular Micropatterning Biomaterials for Hair Regeneration and Vascularization. Mater. Horiz. 2023, 10, 3773–3784. [Google Scholar] [CrossRef]
- Kageyama, T.; Shimizu, A.; Anakama, R.; Nakajima, R.; Suzuki, K.; Okubo, Y.; Fukuda, J. Reprogramming of Three-Dimensional Microenvironments for In Vitro Hair Follicle Induction. Sci. Adv. 2022, 8, eadd4603. [Google Scholar] [CrossRef]
- Liu, Z.; Huang, J.; Kang, D.; Zhou, Y.; Du, L.; Qu, Q.; Wang, J.; Wen, L.; Fu, D.; Hu, Z.; et al. Microenvironmental Reprogramming of Human Dermal Papilla Cells for Hair Follicle Tissue Engineering. Acta Biomater. 2023, 165, 31–49. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Xu, C.H. Innovative Approaches and Advances for Hair Follicle Regeneration. ACS Biomater. Sci. Eng. 2023, 9, 2251–2276. [Google Scholar] [CrossRef]
- Kageyama, T.; Miyata, H.; Seo, J.; Nanmo, A.; Fukuda, J. In Vitro Hair Follicle Growth Model for Drug Testing. Sci. Rep. 2023, 13, 4847. [Google Scholar] [CrossRef]
- Gupta, A.C.; Chawla, S.; Hegde, A.; Singh, D.; Bandyopadhyay, B.; Lakshmanan, C.C.; Kalsi, G.; Ghosh, S. Establishment of an in Vitro Organoid Model of Dermal Papilla of Human Hair Follicle. J. Cell. Physiol. 2018, 233, 9015–9030. [Google Scholar] [CrossRef]
- Marinho, P.A.; Jeong, G.; Shin, S.H.; Kim, S.N.; Choi, H.; Lee, S.H.; Park, B.C.; Hong, Y.D.; Kim, H.J.; Park, W.S. The Development of an in Vitro Human Hair Follicle Organoid with a Complexity Similar to That In Vivo. Biomed. Mater. 2024, 19, 025041. [Google Scholar] [CrossRef]
- Kalabusheva, E.P.; Vorotelyak, E.A. Generation of Hair Follicle Germs In Vitro Using Human Postnatal Skin Cells. In Methods in Molecular Biology; Springer: New York, NY, USA, 2020; Volume 2154, pp. 153–163. [Google Scholar] [CrossRef]
- Andl, T.; Zhou, L.; Zhang, Y. The Dermal Papilla Dilemma and Potential Breakthroughs in Bioengineering Hair Follicles. Cell Tissue Res. 2022, 391, 221. [Google Scholar] [CrossRef]
- Castro, A.R.; Logarinho, E. Tissue Engineering Strategies for Human Hair Follicle Regeneration: How Far from a Hairy Goal? Stem Cells Transl. Med. 2020, 9, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Jain, P.; Kathuria, H.; Dubey, N. Advances in 3D Bioprinting of Tissues/Organs for Regenerative Medicine and In-Vitro Models. Biomaterials 2022, 287, 121639. [Google Scholar] [CrossRef] [PubMed]
- Kageyama, T.; Chun, Y.S.; Fukuda, J. Hair Follicle Germs Containing Vascular Endothelial Cells for Hair Regenerative Medicine. Sci. Rep. 2021, 11, 624. [Google Scholar] [CrossRef] [PubMed]
- Vatanashevanopakorn, C.; Sartyoungkul, T. IPSC-Based Approach for Human Hair Follicle Regeneration. Front. Cell Dev. Biol. 2023, 11, 1149050. [Google Scholar] [CrossRef]
- Zeng, Q.; Ma, Y.; Cai, R.; Li, X.; Luo, Y.; Zheng, B.; Wang, G.; Xu, X.; Wang, X.; Liu, Z. Direct Reprogramming of Human Fibroblasts into Hair-Inducing Dermal Papilla Cell-Like Cells by a Single Small Molecule. Biochem. Pharmacol. 2025, 233, 116744. [Google Scholar] [CrossRef] [PubMed]
- Intercytex Phase II Cell Therapy Research Trial Completion Data. Available online: https://www.farjo.com/news/intercytex-phase-ii-cell-therapy-research-trial-completion-data/ (accessed on 18 October 2025).
- Egger, A.; Tomic-Canic, M.; Tosti, A.; Frost, P. Advances in Stem Cell-Based Therapy for Hair Loss. CellR4 Repair Replace Regen. Reprogram. 2020, 8, e2894. [Google Scholar]
- Balañá, M.E.; Charreau, H.E.; Leirós, G.J. Epidermal Stem Cells and Skin Tissue Engineering in Hair Follicle Regeneration. World J. Stem Cells 2015, 7, 711–722. [Google Scholar] [CrossRef]
- Ji, S.; Zhu, Z.; Sun, X.; Fu, X. Functional Hair Follicle Regeneration: An Updated Review. Signal Transduct. Target. Ther. 2021, 6, 66. [Google Scholar] [CrossRef]
- A Study to Evaluate and Compare Injections of Autologous Dermal and Epidermal Cells into the Balding Scalp of Subjects with Hair Loss (CA-0002012) (NCT01451047). Available online: https://clinicaltrials.gov/study/NCT01451047 (accessed on 29 February 2012).
- A Study to Evaluate and Compare Injections of Autologous Mixed Population of Dermal Cells into the Balding Scalp of Subjects with Hair Loss (CA-0004542) (NCT01451151). Available online: https://clinicaltrials.gov/study/NCT01451151 (accessed on 11 July 2013).
- A Study to Evaluate and Compare Injections of Autologous Dermal and Epidermal Cells into the Balding Scalp of Subjects with Hair Loss (NCT01451021). Available online: https://clinicaltrials.gov/study/NCT01451021 (accessed on 29 February 2012).
- McElwee, K.J.; Kissling, S.; Wenzel, E.; Huth, A.; Hoffmann, R. Cultured Peribulbar Dermal Sheath Cells Can Induce Hair Follicle Development and Contribute to the Dermal Sheath and Dermal Papilla. J. Investig. Dermatol. 2003, 121, 1267–1275. [Google Scholar] [CrossRef]
- Tsuboi, R.; Niiyama, S.; Irisawa, R.; Harada, K.; Nakazawa, Y.; Kishimoto, J. Autologous Cell-Based Therapy for Male and Female Pattern Hair Loss Using Dermal Sheath Cup Cells: A Randomized Placebo-Controlled Double-Blinded Dose-Finding Clinical Study. J. Am. Acad. Dermatol. 2020, 83, 109–116. [Google Scholar] [CrossRef]
- Harada, K.; Ohyama, M.; Niiyama, S.; Irisawa, R.; Mae, K.; Mori, M.; Wakimoto, H.; Kinoshita-Ise, M.; Fukuyama, M.; Hayakawa, R.; et al. Efficacy of Autologous Dermal Sheath Cup Cell Transplantation in Male and Female Pattern Hair Loss: A Single-Arm, Multi-Center, Phase III Equivalent Clinical Study. J. Dermatol. 2023, 50, 1539–1549. [Google Scholar] [CrossRef]
- Yoshida, Y.; Tsuboi, R.; Kishimoto, J. Dermal Sheath Cells and Hair Follicle Regeneration. In Hair Follicle Regeneration; Springer International Publishing: Cham, Switzerland, 2022; Volume 72, pp. 91–106. [Google Scholar] [CrossRef]
- Liu, D.; Xu, Q.; Meng, X.; Liu, X.; Liu, J. Status of Research on the Development and Regeneration of Hair Follicles. Int. J. Med. Sci. 2024, 21, 80–94. [Google Scholar] [CrossRef]
- Hair Follicle Banking—HairClone. Available online: https://hairclone.me/about-us/hair-follicle-banking/ (accessed on 4 August 2024).
- Correia, M.; Lopes, J.; Lopes, D.; Melero, A.; Makvandi, P.; Veiga, F.; Coelho, J.F.J.; Fonseca, A.C.; Paiva-Santos, A.C. Nanotechnology-Based Techniques for Hair Follicle Regeneration. Biomaterials 2023, 302, 122348. [Google Scholar] [CrossRef]
- ISHRS. Tissue Engineering (Hair Follicle Cloning)—ISHRS. Available online: https://ishrs.org/ (accessed on 3 June 2014).
- Gupta, A.K.; Talukder, M.; Venkataraman, M.; Bamimore, M.A. Minoxidil: A Comprehensive Review. J. Dermatol. Treat. 2022, 33, 1896–1906. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Talukder, M.; Shemar, A.; Piraccini, B.M.; Tosti, A. Low-Dose Oral Minoxidil for Alopecia: A Comprehensive Review. Skin Appendage Disord. 2023, 9, 423–437. [Google Scholar] [CrossRef]
- Mella, J.M.; Perret, M.C.; Manzotti, M.; Catalano, H.N.; Guyatt, G. Efficacy and Safety of Finasteride Therapy for Androgenetic Alopecia: A Systematic Review. Arch. Dermatol. 2010, 146, 1141–1150. [Google Scholar] [CrossRef]
- Sharma, A.; Chouhan, K.; Bhatia, S.; Dashore, S. Platelet-Rich Plasma in Androgenetic Alopecia. Indian Dermatol. Online J. 2021, 12, S31. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Qu, K.; Lei, Q.; Chen, M.; Bian, D. Effectiveness of Platelet-Rich Plasma in the Treatment of Androgenic Alopecia: A Meta-Analysis. Aesthet. Plast. Surg. 2024, 48, 977–984. [Google Scholar] [CrossRef]
- Yan, W.; Liu, J.; Xie, X.; Jin, Q.; Yang, Y.; Pan, Y.; Zhang, Y.; Zhang, F.; Wang, Y.; Liu, J.; et al. Restoration of Follicular β-Catenin Signaling by Mesenchymal Stem Cells Promotes Hair Growth in Mice with Androgenetic Alopecia. Stem Cell Res. Ther. 2024, 15, 439. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Jeong, K.H.; Kim, J.E.; Woo, Y.J.; Kim, B.J.; Kang, H. Repeated Microneedle Stimulation Induces Enhanced Hair Growth in a Murine Model. Ann. Dermatol. 2016, 28, 586–592. [Google Scholar] [CrossRef]
- Plikus, M.V.; Mayer, J.A.; De La Cruz, D.; Baker, R.E.; Maini, P.K.; Maxson, R.; Chuong, C.M. Cyclic Dermal BMP Signalling Regulates Stem Cell Activation during Hair Regeneration. Nature 2008, 451, 340–344. [Google Scholar] [CrossRef]
- Chu, S.Y.; Chou, C.H.; Huang, H.D.; Yen, M.H.; Hong, H.C.; Chao, P.H.; Wang, Y.H.; Chen, P.Y.; Nian, S.X.; Chen, Y.R.; et al. Mechanical Stretch Induces Hair Regeneration through the Alternative Activation of Macrophages. Nat. Commun. 2019, 10, 1524. [Google Scholar] [CrossRef]
- Wu, X.; Tang, S. Advances in the Extracellular Vesicles Treatment of Alopecia. Ann. Med. 2025, 57, 2543517. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Faes, L.; Kale, A.U.; Wagner, S.K.; Fu, D.J.; Bruynseels, A.; Mahendiran, T.; Moraes, G.; Shamdas, M.; Kern, C.; et al. A Comparison of Deep Learning Performance against Health-Care Professionals in Detecting Diseases from Medical Imaging: A Systematic Review and Meta-Analysis. Lancet Digit. Health 2019, 1, e271–e297. [Google Scholar] [CrossRef] [PubMed]
- Adamson, A.S.; Smith, A. Machine Learning and Health Care Disparities in Dermatology. JAMA Dermatol. 2018, 154, 1247–1248. [Google Scholar] [CrossRef]
- Takwale, A.; Arthur, E.; Pearce, J.; Farrant, P.; Holmes, S.; Harries, M. A Practical Guide to the Standardization of Hair Loss Photography for Clinicians. Clin. Exp. Dermatol. 2025, 50, 564–572. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, H.; Lee, C.H.; Lee, W.S. Photographic Assessment Improves Adherence to Recommended Follow-Up in Patients with Androgenetic Alopecia and Alopecia Areata: A Retrospective Cohort Study. Indian J. Dermatol. Venereol. Leprol. 2019, 85, 431–434. [Google Scholar] [CrossRef] [PubMed]
- Ankawa, R.; Fuchs, Y. May the Best Wound WIHN: The Hallmarks of Wound-Induced Hair Neogenesis. Curr. Opin. Genet. Dev. 2022, 72, 53–60. [Google Scholar] [CrossRef]
- Vañó-Galván, S.; Fernández-Crehuet, P.; Garnacho, G.; Gómez-Zubiaur, A.; Hermosa-Gelbard, A.; Moreno-Arrones, O.M.; Saceda-Corralo, D.; Serrano-Falcón, C. Recommendations on the Clinical Management of Androgenetic Alopecia: A Consensus Statement from the Spanish Hair Disorders Group of the Spanish Academy of Dermatology and Venereology (AEDV). Actas Dermosifiliogr. 2024, 115, T347–T355. [Google Scholar] [CrossRef]
- Zeltzer, A.A.; Keren, A.; Paus, R.; Gilhar, A. Topical Minoxidil Rejuvenates Hair Follicles from Men with Androgenetic Alopecia in Vivo. Acta Derm. Venereol. 2024, 104, adv24213. [Google Scholar] [CrossRef]
- Hawkshaw, N.J.; Hardman, J.A.; Haslam, I.S.; Shahmalak, A.; Gilhar, A.; Lim, X.; Paus, R. Identifying Novel Strategies for Treating Human Hair Loss Disorders: Cyclosporine A Suppresses the Wnt Inhibitor, SFRP1, in the Dermal Papilla of Human Scalp Hair Follicles. PLoS Biol. 2018, 16, e2003705. [Google Scholar] [CrossRef]
- Kalil, L.; King, B. Innovation in Alopecia Areata. Dermatol. Clin. 2025, 43, 27–33. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.