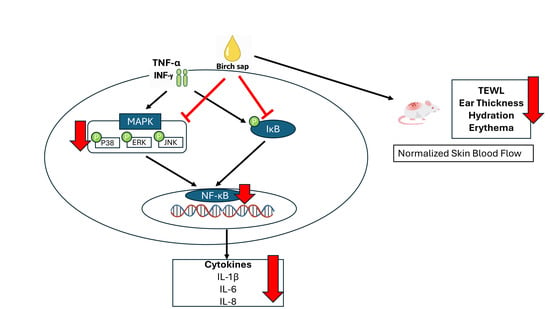
Birch Sap Attenuates Inflammatory Cytokines and Improves Skin Parameters in Cellular and Animal Models of Skin Irritation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. In Vitro Experiments
2.2.1. Cell Lines
2.2.2. Cell Viability Test
2.2.3. Western Blot Assay
2.2.4. Quantitative Polymerase Chain Reaction (qPCR)
2.3. In Vivo Experiments
2.3.1. Experiment Animals
2.3.2. Experiment Groups
2.3.3. Histopathological Analysis
2.4. Data Analysis
3. Results
3.1. Monosaccharides Compositional Analysis in Birch Sap
3.2. Analysis of the Effect of Birch Sap on Keratinocyte Viability
3.3. Effects of Birch Sap on Pro-Inflammatory Cytokines mRNA Expression After HaCaT Cells Stimulation with Tumor Necrosis Factor-α (TNF-α)/Interferon-γ(IFN-γ)
3.4. Effects of Birch Sap on MAPK Phosphorylation After HaCaT Cells Stimulation with TNF-α/IFN-γ
3.5. Effects of Birch Sap on NF-κB Pathway Phosphorylation After HaCaT Cells Stimulation with TNF-α/IFN-γ
3.6. Effects of Birch Sap on Skin Lesions Induced by DNCB
3.7. Effects of Birch Sap on Skin Blood Flow
3.8. Effects of Birch Sap on Skin Physiological Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Nuclear factor kappa B | NF-κB |
| Mitogen-activated protein kinases | MAPK |
| Tumor necrosis factor-α | TNF-α |
| Interferon-γ | INF-γ |
| Dulbecco’s modified eagle medium | DMEM |
| Fetal bovine serum | FBS |
| Dimethyl sulfoxide | DMSO |
| 2,4-Dinitrochlorobenzene | DNCB |
| Hematoxylin and eosin | H&E |
| Standard error of the mean | S.E.M. |
| Quantitative polymerase chain reaction | qPCR |
| Interleukin | IL |
| Laser speckle contrast imaging | LSCI |
| Transepidermal water loss | TEWL |
| Paraformaldehyde | PFA |
| Matrix metalloproteinase | MMP |
| Nuclear factor erythroid 2-related factors 2 | Nrf2 |
References
- Maintz, L.; Bieber, T.; Simpson, H.D.; Demessant-Flavigny, A.-L. From Skin Barrier Dysfunction to Systemic Impact of Atopic Dermatitis: Implications for a Precision Approach in Dermocosmetics and Medicine. J. Pers. Med. 2022, 12, 893. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.; Huang, C.; Radi, R.; Moll, S.B.; Jules, E.; Arbiser, J.L. Skin Barrier Function: The Interplay of Physical, Chemical, and Immunologic Properties. Cells 2023, 12, 2745. [Google Scholar] [CrossRef]
- Chen, B.; Tang, H.; Liu, Z.; Qiao, K.; Chen, X.; Liu, S.; Pan, N.; Chen, T.; Liu, Z.; Chen, B.; et al. Mechanisms of Sensitive Skin and the Soothing Effects of Active Compounds: A Review. Cosmetics 2024, 11, 190. [Google Scholar] [CrossRef]
- Fania, L.; Moretta, G.; Antonelli, F.; Scala, E.; Abeni, D.; Albanesi, C.; Madonna, S. Multiple Roles for Cytokines in Atopic Dermatitis: From Pathogenic Mediators to Endotype-Specific Biomarkers to Therapeutic Targets. Int. J. Mol. Sci. 2022, 23, 2684. [Google Scholar] [CrossRef]
- Shirley, S.N.; Watson, A.E.; Yusuf, N. Pathogenesis of Inflammation in Skin Disease: From Molecular Mechanisms to Pathology. Int. J. Mol. Sci. 2024, 25, 10152. [Google Scholar] [CrossRef]
- Ingrasci, G.; Lipman, Z.M.; Yosipovitch, G. When Topical Therapy of Atopic Dermatitis Fails: A Guide for the Clinician. Expert. Rev. Clin. Immunol. 2021, 17, 1245–1256. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, S.; Pandey, M.M.; Kumar Singh Rawat, A. Medicinal Plants of the Genus Betula—Traditional Uses and a Phytochemical–Pharmacological Review. J. Ethnopharmacol. 2015, 159, 62–83. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Xu, T.; He, J.; Jiang, Y.; Qu, L.; Wang, L.; Ma, J.; Yang, Q.; Wu, W.; Sun, D.; et al. Exploring the Potential of White Birch Sap: A Natural Alternative to Traditional Skin Whitening Agents with Reduced Side Effects. Heliyon 2024, 10, e26715. [Google Scholar] [CrossRef]
- Ge, Z.; Wang, D.; Zhao, W.; Wang, P.; Dai, Y.; Dong, M.; Wang, J.; Zhao, Y.; Zhao, X. Structural and Functional Characterization of Exopolysaccharide from Leuconostoc citreum BH10 Discovered in Birch Sap. Carbohydr. Res. 2024, 535, 108994. [Google Scholar] [CrossRef] [PubMed]
- Mingaila, J.; Čiuldienė, D.; Viškelis, P.; Bartkevičius, E.; Vilimas, V.; Armolaitis, K.; Mingaila, J.; Čiuldienė, D.; Viškelis, P.; Bartkevičius, E.; et al. The Quantity and Biochemical Composition of Sap Collected from Silver Birch (Betula pendula Roth) Trees Growing in Different Soils. Forests 2020, 11, 365. [Google Scholar] [CrossRef]
- Carpintero, M.; Marcet, I.; Zornoza, M.; Rendueles, M.; Díaz, M. Effect of Birch Sap as Solvent and Source of Bioactive Compounds in Casein and Gelatine Films. Membranes 2023, 13, 786. [Google Scholar] [CrossRef]
- Córdova, A. Unlocking the Value of Birch Bark: Betulin as a Sustainable Additive for Advancing Cellulosic Material Performance. ACS Sustain. Resour. Manag. 2025. [Google Scholar] [CrossRef]
- Grabek-Lejko, D.; Kasprzyk, I.; Zaguùa, G.; Puchalski, C. The Bioactive and Mineral Compounds in Birch Sap Collected in Different Types of Habitats. Balt. For. 2017, 23, 394–401. [Google Scholar]
- Softa, M.; Percoco, G.; Lati, E.; Bony, P. Birch Sap (Betula alba) and Chaga Mushroom (Inonotus obliquus) Extracts Show Anti-Oxidant, Anti-Inflammatory and DNA Protection/Repair Activity In Vitro. J. Cosmet. Dermatol. Sci. Appl. 2019, 9, 188–205. [Google Scholar] [CrossRef]
- Huang, C.-F.; Lin, T.-K.; Chang, C.-C.; Lee, M.-Y.; Lu, C.-Y.; Hung, C.-F.; Wang, S.-J. Birch Sap Preserves Memory Function in Rats by Enhancing Cerebral Blood Flow and Modulating the Presynaptic Glutamatergic System in the Hippocampus. Int. J. Mol. Sci. 2025, 26, 5009. [Google Scholar] [CrossRef] [PubMed]
- Pickard, C.; Smith, A.M.; Cooper, H.; Strickland, I.; Jackson, J.; Healy, E.; Friedmann, P.S. Investigation of Mechanisms Underlying the T-Cell Response to the Hapten 2,4-Dinitrochlorobenzene. J. Investig. Dermatol. 2007, 127, 630–637. [Google Scholar] [CrossRef]
- Sung, Y.-Y.; Kim, Y.S.; Kim, H.K. Illicium verum Extract Inhibits TNF-α- and IFN-γ-Induced Expression of Chemokines and Cytokines in Human Keratinocytes. J. Ethnopharmacol. 2012, 144, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Jayasinghe, A.M.K.; Han, E.-J.; Kirindage, K.G.I.S.; Fernando, I.P.S.; Kim, E.-A.; Kim, J.; Jung, K.; Kim, K.-N.; Heo, S.-J.; Ahn, G. 3-Bromo-4,5-Dihydroxybenzaldehyde Isolated from Polysiphonia morrowii Suppresses TNF-α/IFN-γ-Stimulated Inflammation and Deterioration of Skin Barrier in HaCaT Keratinocytes. Mar. Drugs 2022, 20, 563. [Google Scholar] [CrossRef]
- Oh, J.-H.; Kim, S.-H.; Kwon, O.-K.; Kim, J.-H.; Oh, S.-R.; Han, S.-B.; Park, J.-W.; Ahn, K.-S. Purpurin Suppresses Atopic Dermatitis via TNF-α/IFN-γ-Induced Inflammation in HaCaT Cells. Int. J. Immunopathol. Pharmacol. 2022, 36, 03946320221111135. [Google Scholar] [CrossRef] [PubMed]
- Calhelha, R.C.; Haddad, H.; Ribeiro, L.; Heleno, S.A.; Carocho, M.; Barros, L.; Calhelha, R.C.; Haddad, H.; Ribeiro, L.; Heleno, S.A.; et al. Inflammation: What’s There and What’s New? Appl. Sci. 2023, 13, 2312. [Google Scholar] [CrossRef]
- Green, M.; Kashetsky, N.; Feschuk, A.; Maibach, H.I. Transepidermal Water Loss (TEWL): Environment and Pollution—A Systematic Review. Ski. Health Dis. 2022, 2, e104. [Google Scholar] [CrossRef]
- Çetinarslan, T.; Kümper, L.; Fölster-Holst, R. The Immunological and Structural Epidermal Barrier Dysfunction and Skin Microbiome in Atopic Dermatitis-an Update. Front. Mol. Biosci. 2023, 10, 1159404. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Son, S.-H.; Kim, N.-J.; Im, D.-S. P38 MAPK Inhibitor NJK14047 Suppresses CDNB-Induced Atopic Dermatitis-Like Symptoms in BALB/c Mice. Biomol. Ther. 2022, 30, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Andrzejczak, K.; Sternak, A.; Witkowski, W.; Ponikowska, M.; Andrzejczak, K.; Sternak, A.; Witkowski, W.; Ponikowska, M. Inflammation-Driven Molecular Ageing in Chronic Inflammatory Skin Diseases: Is There a Role for Biologic Therapies? Cells 2025, 14, 1442. [Google Scholar] [CrossRef] [PubMed]
- Beck, L.A.; Cork, M.J.; Amagai, M.; De Benedetto, A.; Kabashima, K.; Hamilton, J.D.; Rossi, A.B. Type 2 Inflammation Contributes to Skin Barrier Dysfunction in Atopic Dermatitis. JID Innov. 2022, 2, 100131. [Google Scholar] [CrossRef] [PubMed]
- Al-Dhubaibi, M.S.; Mohammed, G.F.; Bahaj, S.S.; AbdElneam, A.I.; Al-Dhubaibi, A.M.; Atef, L.M. The Role of Keratinocytes in Skin Health and Disease. Dermatol. Rev. 2025, 6, e70028. [Google Scholar] [CrossRef]
- Chen, L.-X.-Y.; Hao, P.-S. The Role of Skin Barrier and Immune Abnormalities in the Pathogenesis of Rosacea. Clin. Exp. Med. 2025, 25, 324. [Google Scholar] [CrossRef]
- Cianciulli, A.; Calvello, R.; Porro, C.; Lofrumento, D.D.; Panaro, M.A. Inflammatory Skin Diseases: Focus on the Role of Suppressors of Cytokine Signaling (SOCS) Proteins. Cells 2024, 13, 505. [Google Scholar] [CrossRef]
- Guo, Y.-J.; Pan, W.-W.; Liu, S.-B.; Shen, Z.-F.; Xu, Y.; Hu, L.-L. ERK/MAPK Signalling Pathway and Tumorigenesis (Review). Exp. Ther. Med. 2020, 19, 1997–2007. [Google Scholar] [CrossRef]
- Yu, H.; Lin, L.; Zhang, Z.; Zhang, H.; Hu, H. Targeting NF-κB Pathway for the Therapy of Diseases: Mechanism and Clinical Study. Signal Transduct. Target. Ther. 2020, 5, 209. [Google Scholar] [CrossRef]
- Guo, Q.; Jin, Y.; Chen, X.; Ye, X.; Shen, X.; Lin, M.; Zeng, C.; Zhou, T.; Zhang, J. NF-κB in Biology and Targeted Therapy: New Insights and Translational Implications. Sig. Transduct. Target. Ther. 2024, 9, 53. [Google Scholar] [CrossRef] [PubMed]
- Bhol, N.K.; Bhanjadeo, M.M.; Singh, A.K.; Dash, U.C.; Ojha, R.R.; Majhi, S.; Duttaroy, A.K.; Jena, A.B. The Interplay between Cytokines, Inflammation, and Antioxidants: Mechanistic Insights and Therapeutic Potentials of Various Antioxidants and Anti-Cytokine Compounds. Biomed. Pharmacother. 2024, 178, 117177. [Google Scholar] [CrossRef]
- Liu, X.; Chen, B.; Liu, X.; Zhang, X.; Wu, J. Interplay between MAPK Signaling Pathway and Autophagy in Skin Aging: Mechanistic Insights and Therapeutic Implications. Front. Cell Dev. Biol. 2025, 13, 1625357. [Google Scholar] [CrossRef]
- Lu, C.; Deng, S.; Liu, Y.; Yang, S.; Qin, D.; Zhang, L.; Wang, R.; Zhang, Y. Inhibition of Macrophage MAPK/NF-κB Pathway and Th2 Axis by Mangiferin Ameliorates MC903-Induced Atopic Dermatitis. Int. Immunopharmacol. 2024, 133, 112038. [Google Scholar] [CrossRef]
- Han, H.-J.; Hyun, C.-G. Anti-Inflammatory Effects and Human Skin Safety of the Eastern Traditional Herb Mosla japonica. Life 2025, 15, 418. [Google Scholar] [CrossRef] [PubMed]
- Xiao, K.; Liu, C.; Tu, Z.; Xu, Q.; Chen, S.; Zhang, Y.; Wang, X.; Zhang, J.; Hu, C.-A.A.; Liu, Y. Activation of the NF-κB and MAPK Signaling Pathways Contributes to the Inflammatory Responses, but Not Cell Injury, in IPEC-1 Cells Challenged with Hydrogen Peroxide. Oxid. Med. Cell Longev. 2020, 2020, 5803639. [Google Scholar] [CrossRef]
- Deng, R.; Wang, F.; Wang, L.; Xiong, L.; Shen, X.; Song, H. Advances in Plant Polysaccharides as Antiaging Agents: Effects and Signaling Mechanisms. J. Agric. Food Chem. 2023, 71, 7175–7191. [Google Scholar] [CrossRef]
- Guo, Q.; Zhang, M.; Mujumdar, A.S. Progress of Plant-Derived Non-Starch Polysaccharides and Their Challenges and Applications in Future Foods. Compr. Rev. Food Sci. Food Saf. 2024, 23, e13361. [Google Scholar] [CrossRef] [PubMed]
- Haiming, C.; Yijing, L.; Bin, T.; Xiaoyu, M.; Hailun, L.; Meiting, D.; Ziqing, L.; Xuwei, Z.; Yujie, Y.; Zuqing, S.; et al. Modulation of the Gut-Skin Axis by Polysaccharides: Mechanisms and Therapeutic Potential in Immune-Related Skin Diseases. Carbohydr. Polym. 2025, 369, 124143. [Google Scholar] [CrossRef]
- Sindhu, R.K.; Goyal, A.; Das, J.; Choden, S.; Kumar, P. Immunomodulatory Potential of Polysaccharides Derived from Plants and Microbes: A Narrative Review. Carbohydr. Polym. Technol. Appl. 2021, 2, 100044. [Google Scholar] [CrossRef]
- Tu, Y.; Li, N.; Liu, H.-Y.; Sun, D.-J.; He, L.; Gu, H. Polysaccharide from Prinsepia Utilis Royle Maintains the Skin Barrier by Mediating Differentiation, Lipid Metabolism and Tight Junction of Keratinocyte. Sci. Rep. 2025, 15, 20470. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, P.B.S.; de Oliveira, W.F.; dos Santos Silva, P.M.; dos Santos Correia, M.T.; Kennedy, J.F.; Coelho, L.C.B.B. Skincare Application of Medicinal Plant Polysaccharides—A Review. Carbohydr. Polym. 2022, 277, 118824. [Google Scholar] [CrossRef] [PubMed]
- Na, K.; Lkhagva-Yondon, E.; Kim, M.; Lim, Y.-R.; Shin, E.; Lee, C.-K.; Jeon, M.-S. Oral Treatment with Aloe Polysaccharide Ameliorates Ovalbumin-Induced Atopic Dermatitis by Restoring Tight Junctions in Skin. Scand. J. Immunol. 2020, 91, e12856. [Google Scholar] [CrossRef] [PubMed]
- Ashigai, H.; Komano, Y.; Wang, G.; Kawachi, Y.; Sunaga, K.; Yamamoto, R.; Takata, R.; Yanai, T. Orally Administered Polysaccharide Derived from Blackcurrants (Ribes nigrum L.) Improves Skin Hydration in Ultraviolet-Irradiated Hairless Mice. J. Nutr. Sci. Vitaminol. 2018, 64, 301–304. [Google Scholar] [CrossRef]
- Li, L.; Huang, T.; Lan, C.; Ding, H.; Yan, C.; Dou, Y. Protective Effect of Polysaccharide from Sophora japonica L. Flower Buds against UVB Radiation in a Human Keratinocyte Cell Line (HaCaT Cells). J. Photochem. Photobiol. Biol. 2019, 191, 135–142. [Google Scholar] [CrossRef]
- Yao, Y.; Xu, B.; Yao, Y.; Xu, B. Skin Health Promoting Effects of Natural Polysaccharides and Their Potential Application in the Cosmetic Industry. Polysaccharides 2022, 3, 818–830. [Google Scholar] [CrossRef]
- Wang, W.; Zou, A.; Yu, Q.; Wang, Z.; Tan, D.; Yang, K.; Cai, C.; Yu, G.; Wang, W.; Zou, A.; et al. Bioactive Polysaccharides from Fermented Dendrobium officinale: Structural Insights and Their Role in Skin Barrier Repair. Molecules 2025, 30, 2875. [Google Scholar] [CrossRef]
- Mourelle, M.L.; Gómez, C.P.; Legido, J.L.; Mourelle, M.L.; Gómez, C.P.; Legido, J.L. Unveiling the Role of Minerals and Trace Elements of Thermal Waters in Skin Health. Appl. Sci. 2024, 14, 6291. [Google Scholar] [CrossRef]
- Shu, X.; Zhao, S.; Huo, W.; Tang, Y.; Zou, L.; Li, Z.; Li, L.; Wang, X. Clinical Study of a Spray Containing Birch Juice for Repairing Sensitive Skin. Arch. Dermatol. Res. 2023, 315, 2271–2281. [Google Scholar] [CrossRef] [PubMed]








| Genes | Primers | Sequence (5′-3′) |
|---|---|---|
| IL-1β | Forward | CTC TCA CCT CTC CTA CTC ACT |
| Reverse | ATC AGA ATG TGG GAG CGA AT | |
| IL-6 | Forward | CGA GCC CAC CGG GAA CGA AA |
| Reverse | GGA CCG AAG GCG CTT GTG GAG | |
| IL-8 | Forward | ACT GAG AGT GAT TGA GAG TGG AC |
| Reverse | AAC CCT CTG CAC CCA GTT TTC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sung, C.-H.; Huang, C.-F.; Hsu, Y.-J.; Pu, C.-M.; Kung, C.-C.; Chu, T.W.; Hung, C.-F. Birch Sap Attenuates Inflammatory Cytokines and Improves Skin Parameters in Cellular and Animal Models of Skin Irritation. Cosmetics 2025, 12, 282. https://doi.org/10.3390/cosmetics12060282
Sung C-H, Huang C-F, Hsu Y-J, Pu C-M, Kung C-C, Chu TW, Hung C-F. Birch Sap Attenuates Inflammatory Cytokines and Improves Skin Parameters in Cellular and Animal Models of Skin Irritation. Cosmetics. 2025; 12(6):282. https://doi.org/10.3390/cosmetics12060282
Chicago/Turabian StyleSung, Chao-Hsien, Chien-Fen Huang, Yu-Jou Hsu, Chi-Ming Pu, Chia-Chi Kung, Thomas W. Chu, and Chi-Feng Hung. 2025. "Birch Sap Attenuates Inflammatory Cytokines and Improves Skin Parameters in Cellular and Animal Models of Skin Irritation" Cosmetics 12, no. 6: 282. https://doi.org/10.3390/cosmetics12060282
APA StyleSung, C.-H., Huang, C.-F., Hsu, Y.-J., Pu, C.-M., Kung, C.-C., Chu, T. W., & Hung, C.-F. (2025). Birch Sap Attenuates Inflammatory Cytokines and Improves Skin Parameters in Cellular and Animal Models of Skin Irritation. Cosmetics, 12(6), 282. https://doi.org/10.3390/cosmetics12060282