Abstract
Robot-assisted implant surgery (RAIS) represents the most advanced form of digitally guided implant placement, integrating virtual planning with mechanically constrained execution and real-time control. Although multiple systematic reviews suggest superior accuracy with robotic systems, the magnitude and clinical relevance of these gains remain uncertain at the highest level of evidence. This umbrella review, conducted according to PRISMA 2020 and Joanna Briggs Institute guidelines, aimed to synthesize and critically appraise systematic reviews and meta-analyses evaluating the accuracy of Robot-assisted implant surgery (RAIS) in dental implantology. Search across five major databases identified seven eligible reviews published between 2023 and 2025, including clinical, cadaveric, and in vitro evidence. Across reviews, RAIS consistently demonstrated the highest placement accuracy, with pooled mean coronal deviations of 0.60–0.73 mm, apical deviations of 0.63–0.70 mm, and angular deviations typically between 1.4° and 1.7°. Comparative meta-analyses reported significant reductions in linear (−0.15 to −0.21 mm) and angular deviations (−1.2° to −1.4°) compared with dynamic navigation. Despite these technical advantages, evidence linking improved accuracy to enhanced implant survival, reduced complications, or superior patient-reported outcomes was limited. Robotic workflows were associated with longer setup times, while safety profiles were comparable to other guided techniques. Overall, RAIS provides the highest placement accuracy currently reported; however, further high-quality clinical trials are needed to clarify its impact on long-term clinical outcomes and cost-effectiveness.
1. Introduction
Accurate implant positioning is a fundamental determinant of success in dental implant therapy [1]. Deviations between the planned and the achieved implant position may compromise prosthetic-driven rehabilitation, esthetic integration, biomechanical load distribution, and the long-term stability of peri-implant tissues [2]. Even minor linear or angular inaccuracies can increase the risk of biological complications, prosthetic misfit, and damage to adjacent anatomical structures, particularly in anatomically constrained or esthetically demanding cases [3]. Consequently, improving the precision, predictability, and reproducibility of implant placement has remained a central objective in implant dentistry [4].
Conventional freehand implant surgery relies primarily on the clinician’s experience, visual judgment, and tactile feedback [5]. While acceptable outcomes can be achieved by skilled operators, freehand placement is inherently subject to variability and cumulative human error [6]. Limited visibility, restricted access in posterior regions, patient movement, and operator fatigue may all contribute to deviations from the planned implant trajectory [7]. These limitations have driven the progressive integration of digital technologies-aimed at supporting surgical accuracy and standardization [8].
Computer-assisted implant surgery (CAIS) represented the first major paradigm shift toward digitally guided implant placement [9]. Static guidance systems, based on preoperative virtual planning and patient-specific surgical guides, enabled the transfer of planned implant positions to the clinical setting with improved accuracy compared with freehand techniques [10]. Dynamic navigation systems subsequently introduced real-time intraoperative feedback, allowing continuous visualization of the drill position relative to the planned trajectory without the physical constraints of a surgical guide [11]. Both static and dynamic CAIS have been extensively investigated and are now widely used in clinical practice, demonstrating significant accuracy improvements over freehand surgery [12].
Despite these advances, CAIS technologies remain dependent on the surgeon’s manual execution of osteotomy and implant insertion. Errors related to hand stability, guide positioning, tracking calibration, and cumulative workflow inaccuracies may still occur [13]. Moreover, the degree to which improved geometric accuracy achieved with static or dynamic navigation translates into meaningful clinical benefits remains debated. Implant survival, marginal bone stability, prosthetic success, and patient-reported outcomes are influenced by multiple biological, mechanical, and patient-related factors that extend beyond implant positioning alone [14].
In this context, robot-assisted implant surgery (RAIS) has emerged as the most advanced iteration of digitally guided implant placement [15]. Robotic systems integrate preoperative virtual planning, real-time tracking, and mechanically constrained execution, actively limiting deviations from the planned implant trajectory [16]. By reducing reliance on freehand motor control, RAIS aims to minimize intraoperative variability and achieve a level of precision that surpasses that of conventional navigation systems. From a conceptual standpoint, RAIS represents a shift from guidance-assisted surgery to constraint-assisted surgery [17].
Early investigations of RAIS, primarily conducted in vitro and in cadaveric models, consistently demonstrated sub-millimetric linear deviations and low angular error. More recently, the clinical application of robotic systems in dental implantology has expanded, supported by commercially available platforms designed specifically for oral and maxillofacial procedures [5]. These developments have generated increasing interest due to the potential of RAIS to enhance accuracy and reproducibility, particularly in complex clinical scenarios such as limited bone volume, proximity to vital anatomical structures, and full-arch rehabilitations [18].
Parallel to the growth of primary research, multiple systematic reviews and meta-analyses have been published evaluating robotic and digitally guided implant surgery [19,20,21]. These reviews generally report superior accuracy outcomes for robotic systems compared with static and dynamic navigation [15,22]. However, substantial heterogeneity exists regarding study design, outcome definitions, reference standards, and clinical contexts. Importantly, few reviews critically address whether the observed improvements in accuracy translate into tangible clinical advantages, such as improved survival rates or reduced complications [15,23,24].
The proliferation of systematic reviews has resulted in a fragmented evidence landscape, making it challenging for clinicians and decision-makers to interpret the true value of robotic assistance in implant dentistry. Umbrella reviews, which synthesize evidence from multiple systematic reviews and meta-analyses, provide an appropriate methodological framework to clarify areas of consensus, assess the robustness of findings, and identify gaps between technical performance and clinical relevance.
Therefore, this umbrella review aimed to synthesize and critically appraise systematic reviews and meta-analyses evaluating the accuracy of robot-assisted implant surgery in dental implantology, while secondarily exploring operative efficiency, safety, and clinical relevance. The objective of this umbrella review was to synthesize and critically appraise systematic reviews and meta-analyses evaluating the accuracy of robot-assisted implant surgery, while secondarily exploring operative efficiency, safety, and clinical relevance.
2. Materials and Methods
2.1. Review Design and Reporting
This study is an umbrella review synthesizing evidence from published systematic reviews and meta-analyses evaluating digital, navigated, and robot-assisted systems used to plan and/or guide implant placement in dental implantology. The review was designed and reported in accordance with the PRISMA 2020 [25] statement and conducted following the Joanna Briggs Institute (JBI) guidance for umbrella reviews [26], with a predefined methodology to ensure transparency and reproducibility. The protocol was prospectively registered in PROSPERO (CRD420251240444). Because the review synthesized only published aggregate data, ethics approval and informed consent were not required.
2.2. Information Sources and Search Strategy
A systematic search was conducted in PubMed (MEDLINE), Embase, Scopus, Web of Science Core Collection, and the Cochrane Library from inception to 13 December 2025.
Search strategies combined controlled vocabulary (e.g., MeSH/Emtree where applicable) and free-text terms covering:
- implant/component placement (dental implants),
- navigation/robotics/digital guidance (static and dynamic computer-assisted navigation, surgical navigation, image-guided surgery, augmented reality, accelerometer-based/portable navigation, patient-specific instrumentation, robotic platforms), and
- review design filters (systematic review/meta-analysis).
Database-specific syntaxes were adapted, and full strategies are provided in Supplementary File S1 (Table S1).
2.3. Eligibility Criteria:
The eligibility criteria were defined a priori using a modified PICOS framework, as described below.
Population/Setting:
- Dental: implant placement in partially/fully edentulous jaws, including clinical and experimental accuracy settings (e.g., cadaveric/phantom/resin models) when synthesized within systematic reviews/meta-analyses.
Interventions (digital/robotic guidance):
- Robot-assisted platforms (semi-active/active guidance)
- Computer-assisted navigation (static guides; dynamic navigation)
- Augmented reality-guided navigation
- Accelerometer-/inertial sensor-based navigation (portable systems)
- Patient-specific instrumentation (PSI)
Comparators: Freehand/conventional techniques, alternative navigation modalities, or head-to-head comparisons between guidance systems.
Outcomes:
Primary outcomes were placement accuracy, reported as appropriate to the field:
- Dental: entry/coronal deviation, apical deviation, angular deviation, global/platform deviation, and related 3D accuracy metrics.
Secondary outcomes included intraoperative efficiency (operative time, workflow steps), safety/complications (intra- and postoperative adverse events), and clinical performance (e.g., revision surgery, validated clinical scores where available).
Study design: Only systematic reviews and meta-analyses were included. Narrative reviews, scoping reviews, editorials, conference abstracts, and primary studies were excluded. Where multiple reviews overlapped substantially in the same indication/device category, the most recent and methodologically robust review was prioritized for synthesis, while older reviews were retained only if they contributed unique device categories, outcomes, or populations.
To enhance completeness and reproducibility, supplementary search strategies, including manual screening of reference lists from relevant reviews and citation tracking, were also performed.
2.4. Study Selection
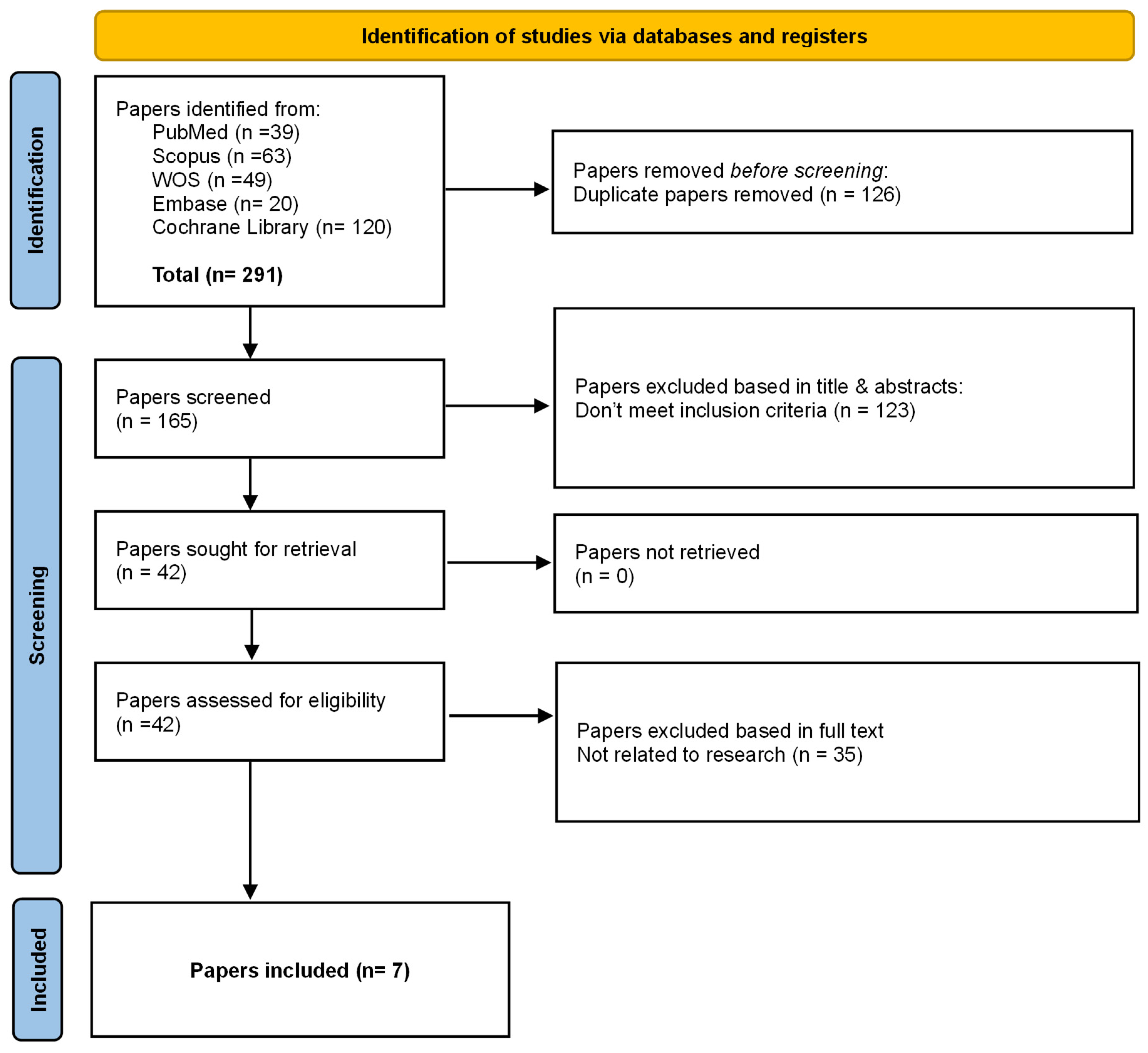
Study selection was performed using Rayyan, a web-based platform designed to facilitate blinded screening and management of records in systematic reviews [27] for deduplication and screening. Two reviewers independently screened titles/abstracts, followed by full-text assessment of potentially eligible reports. Disagreements were resolved by discussion; a third reviewer adjudicated unresolved conflicts. The selection process is summarized in the PRISMA flow diagram (Figure 1).
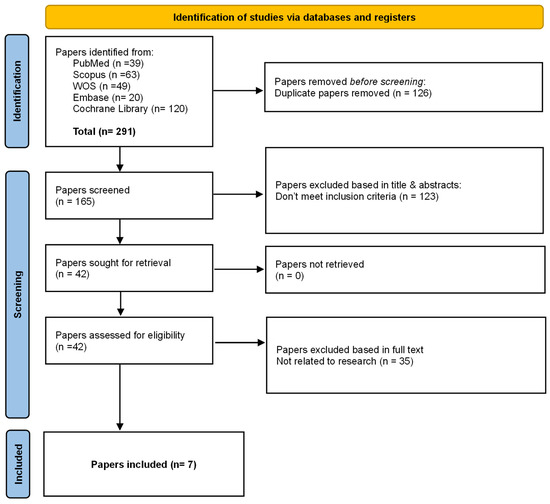
Figure 1.
PRISMA 2020 flow diagram of study selection.
Flow diagram illustrates the identification, screening, eligibility assessment, and inclusion of systematic reviews in this umbrella review. Database searches yielded 291 records (PubMed, Scopus, Web of Science, Embase, and Cochrane Library). After removal of 126 duplicates, 165 records were screened by title and abstract, resulting in the exclusion of 123 records that did not meet the inclusion criteria. Forty-two full-text articles were assessed for eligibility, of which 35 were excluded for not being related to the research question. Seven systematic reviews fulfilled all predefined inclusion criteria and were included in the final qualitative synthesis.
2.5. Data Extraction and Management
A standardized extraction form (Microsoft Excel) was piloted and refined before formal extraction. Two reviewers extracted data independently and cross-checked entries. For each included systematic review/meta-analysis, we extracted:
- Bibliographic details (author, year, journal)
- Review characteristics (protocol registration, search dates/databases, eligibility criteria, synthesis approach)
- Number and design of included primary studies; sample size (implants/procedures/patients)
- Technology category (robotic, static, dynamic, AR, accelerometer/portable, PSI)
- Reference standards for accuracy (CBCT/CT, optical scanning, postoperative imaging, coordinate systems)
- Primary and secondary outcomes (accuracy metrics, time, complications, clinical endpoints)
- Subgroup analyses (jaw, flap/flapless, learning curve, device type, orthopedic approach)
- Authors stated limitations and conclusions
When essential information was unclear, we planned to contact corresponding authors for clarification.
2.6. Methodological Quality Appraisal of Included Reviews
The methodological quality of included systematic reviews/meta-analyses was assessed using AMSTAR 2 [28] by two independent reviewers, with consensus resolution of discrepancies. AMSTAR 2 domain-level judgments and overall confidence ratings (high, moderate, low, critically low) were recorded and incorporated into the interpretation of findings. The overall interpretation of findings in this umbrella review was explicitly informed by the methodological quality of the included systematic reviews as assessed with AMSTAR 2. Evidence derived from reviews rated as low or critically low confidence was not excluded a priori but was interpreted with caution and was not used to support umbrella-level clinical recommendations when higher-quality evidence was available. Greater interpretative weight was assigned to reviews rated as moderate or high confidence, particularly when findings were consistent across multiple reviews.
2.7. Overlap Management and Synthesis Approach
Because umbrella reviews may double-count evidence when primary studies appear in multiple reviews, we planned to map overlap across reviews by creating a citation matrix and estimating overlap (e.g., Corrected Covered Area) when feasible. When overlap was substantial within the same device category and indication, interpretation prioritized the most recent and highest-quality review. Given expected heterogeneity across indications (dental vs. orthopedic), device taxonomies, accuracy definitions, imaging workflows, and comparators, the primary synthesis strategy was a structured narrative synthesis, grouping findings by: (1) robotic platforms, (2) static navigation, (3) dynamic navigation, (4) AR-guided systems, (5) accelerometer-/portable navigation, and (6) PSI.
Where reviews reported pooled estimates, we summarized effect directions, magnitude, and precision as presented by the review authors rather than re-pooling across reviews. Overlap analysis using the corrected covered area (CCA) was conducted specifically for reviews addressing robot-assisted implant accuracy outcomes. Given the expected heterogeneity across indications and study designs, synthesis prioritized the most recent and methodologically robust reviews within overlapping clusters.
2.8. Patient and Public Involvement
No patients or members of the public were involved in the design, conduct, or reporting of this umbrella review.
3. Results
3.1. Selection of Included Studies
The literature search identified a total of 291 records, of which 165 remained after removal of duplicates (n = 126). Following title and abstract screening, 123 records were excluded for not meeting the inclusion criteria. Full-text assessment was conducted for 42 articles, resulting in the exclusion of 35 studies, primarily because they were not systematic reviews or did not report accuracy-related outcomes relevant to robot-assisted implant surgery. Ultimately, seven systematic reviews met the eligibility criteria and were included in the umbrella review. The study selection process is presented in the PRISMA flow diagram (Figure 1).
3.2. Characteristics of the Included Reviews
This umbrella reviews synthesized evidence from seven systematic reviews [29,30,31,32,33,34,35], published between 2023 and 2025, all focused exclusively on dental implant placement using digital or robotic navigation systems. The included reviews evaluated a spectrum of computer-assisted technologies, including static computer-assisted implant surgery (sCAIS), dynamic navigation systems (dCAIS), and robot-assisted implant surgery, either as isolated modalities or in comparative frameworks. An overview of the different digital navigation modalities evaluated in the included reviews and their relative impact on implant placement accuracy is presented in Table 1.

Table 1.
Digital navigation modalities for dental implant placement and their impact on accuracy.
Across all reviews, implant placement accuracy constituted the primary outcome of interest. Accuracy was consistently defined as the deviation between the virtually planned and the postoperatively achieved implant position, and was commonly reported as coronal (platform) deviation, apical deviation, and angular deviation, measured using postoperative cone-beam computed tomography (CBCT). Several reviews additionally explored modifiers of accuracy, such as flap versus flapless approaches, arch location (maxilla vs. mandible), degree of edentulism, and guided protocol (fully, partially, or pilot guided).
Among the reviews included, some focused specifically on robot-assisted implant surgery, reporting pooled accuracy outcomes derived from clinical and preclinical studies, while others provided broader comparisons between robotic, static, and dynamic navigation systems. Reviews incorporating robotic systems generally reported lower mean deviations in coronal, apical, and angular parameters compared with static and dynamic approaches, although conclusions were tempered by the limited number of clinical robotic studies and substantial heterogeneity.
Despite differences in scope and methodology, all seven reviews converged on the conclusion that digitally guided and robotic-assisted implant placement achieves a level of positional accuracy considered clinically acceptable. However, notable heterogeneity was observed in study inclusion criteria, reference standards, outcome definitions, and statistical approaches. Due to this heterogeneity and the overlap of primary studies across reviews, no quantitative pooling was performed at the umbrella-review level, and results were synthesized narratively. The main methodological characteristics, technological scope, and accuracy-related outcomes of the included systematic reviews are summarized in Table 2. A substantial proportion of the available evidence on robot-assisted implant surgery originates from in vitro and cadaveric studies. While these investigations provide valuable insights into technical accuracy under controlled conditions, their findings should not be directly extrapolated to clinical performance without caution.

Table 2.
Characteristics of included systematic reviews and main accuracy outcomes (dental implantology only).
Secondary outcomes, including operative time, learning curve, workflow efficiency, and cost considerations, were inconsistently reported across the included reviews and were frequently derived from early clinical series or experimental settings. Several reviews noted that robot-assisted implant surgery may initially increase operative time due to system setup, registration procedures, and training requirements. However, improvements in workflow efficiency were commonly reported after an initial learning phase, suggesting progressive procedural stabilization with operator experience. Despite these observations, quantitative data on operative time reduction, learning curve thresholds, or cost-effectiveness were sparse, heterogeneous, and insufficient to support firm conclusions. Consequently, the certainty of evidence for these secondary outcomes remains low, and their relevance to routine clinical adoption should be interpreted with caution.
3.3. Accuracy Outcomes of Robot-Assisted Implant Surgery
Across reviews, robot-assisted implant surgery consistently achieved sub-millimetric coronal and apical deviations, with angular deviations generally below 2°. Differences among reviews mainly reflected study design (clinical, cadaveric, or in vitro), synthesis approach (pooled meta-analytic estimates versus descriptive summaries), and comparator modality, rather than conflicting accuracy outcomes.
Yang et al. [29] conducted the most comprehensive systematic review and meta-analysis evaluating the accuracy of robot-assisted implant surgery (RAIS), including 11 in vitro studies (809 implants) and 10 clinical studies (257 implants). In the pooled analysis of in vitro data, the mean global coronal deviation for RAIS was 0.7 mm (95% CI: 0.6–0.8 mm), the mean apical deviation was 0.8 mm (95% CI: 0.6–1.0 mm), and the mean angular deviation was 1.8° (95% CI: 1.2–2.5°). In clinical studies, pooled estimates demonstrated similarly low deviations, with a mean coronal deviation of 0.6 mm (95% CI: 0.5–0.8 mm), mean apical deviation of 0.7 mm (95% CI: 0.6–0.8 mm), and mean angular deviation of 1.6° (95% CI: 1.1–2.0°). When directly compared with computer-assisted implant surgery (CAIS) in in vitro studies, RAIS showed significantly lower coronal deviation (SMD = −0.2; 95% CI: −0.4 to −0.0; p = 0.012), apical deviation (SMD = −0.3; 95% CI: −0.5 to −0.1; p = 0.001), and angular deviation (SMD = −1.3; 95% CI: −1.5 to −1.1; p < 0.001). Heterogeneity was high across outcomes (I2 > 70% for most analyses).
Jain [30] synthesized evidence from comparative in vitro studies evaluating robot-assisted implant placement versus CAIS. In meta-analysis, robot-assisted systems demonstrated significantly greater accuracy than dynamic navigation. The pooled mean difference favored robotics for angular deviation (−1.22°, 95% CI −1.06 to −1.39; p < 0.00001), although heterogeneity was substantial (I2 = 90%). For coronal deviation, three studies showed no heterogeneity (I2 = 0%), with a pooled reduction of −0.15 mm (95% CI −0.24 to −0.07) favoring robotics. Similarly, apical deviation was consistently lower with robotics, and meta-analysis indicated a pooled difference of −0.19 mm (95% CI −0.27 to −0.10; p < 0.00001) with no heterogeneity (I2 = 0%).
Khan et al. [31] evaluated the positional accuracy of robot-assisted computer-aided implant surgery (r-CAIS) through a systematic review and meta-analysis including 13 studies (9 in vitro and 4 clinical), comprising a total of 920 dental implants. In the comparative meta-analysis against dynamic navigation systems, robot-assisted surgery demonstrated significantly greater accuracy across all deviation parameters. The pooled mean difference for coronal deviation favored robotic assistance by −0.17 mm (95% CI: −0.24 to −0.09; p < 0.001), with no detected heterogeneity (I2 = 0%). For apical deviation, the pooled mean difference was −0.21 mm (95% CI: −0.36 to −0.06; p = 0.006), with moderate heterogeneity (I2 = 66%). The largest effect was observed for angular deviation, where robotic systems reduced deviation by −1.41° (95% CI: −1.56 to −1.26; p < 0.001), again with no heterogeneity (I2 = 0%).
When weighted mean deviations were analyzed independently of comparator groups, robot-assisted implant placement achieved mean values of 0.73 ± 0.38 mm (coronal), 0.70 ± 0.39 mm (apical), and 2.20 ± 1.03° (angular) in clinical studies, and 0.78 ± 0.50 mm, 0.92 ± 0.52 mm, and 1.09 ± 0.49°, respectively, in in vitro studies. Comparisons with static guided surgery were limited to a single study and did not allow quantitative synthesis. Overall, the review consistently showed superior geometric accuracy for robot-assisted implant placement compared with dynamic navigation, although the authors emphasized that the evidence base was dominated by experimental studies and that clinical data remained limited.
Ravipati et al. [32] evaluated the accuracy of robot-assisted dental implant placement through a systematic review restricted to in vitro studies, including a total of 13 investigations using phantom and simulated models. Accuracy was assessed primarily through entry (coronal), exit (apical), and angular deviations, reported as means and standard deviations across studies. Pooled accuracy outcomes favored robotic systems, with a mean entry deviation of 0.72 ± 0.68 mm, mean exit deviation of 0.86 ± 0.92 mm, and mean angular deviation of 1.47 ± 1.61°. Across individual studies, coronal deviations commonly range between 0.31 and 1.22 mm, apical deviations between 0.37 and 1.44 mm, and angular deviations between 0.33° and 2.40°, depending on the robotic system configuration and experimental model used.
Comparative analyses reported that robot-assisted implant surgery demonstrated significantly lower linear and angular deviations when compared with freehand implant placement, while differences between robotic systems and dynamic navigation were less pronounced and varied across study designs. In several reviews, statistical comparisons were reported for selected accuracy parameters; however, for some outcomes the level of statistical significance could not be verified due to incomplete reporting in the original sources (p-value not reported).
Pozzi et al. [33] evaluated the in vivo accuracy of autonomous dental implant robotic surgery (Autonomous dental implant robotics, ADIR) through a systematic review and meta-analysis including six clinical studies, comprising 96 patients and 299 dental implants placed in partially and completely edentulous jaws. Accuracy was assessed by superimposing preoperative digital planning with postoperative CBCT images, reporting global platform deviation, global apical deviation, and angular deviation. The meta-analysis demonstrated a mean platform deviation of 0.60 mm (95% CI: 0.51–0.70 mm), a mean apical deviation of 0.63 mm (95% CI: 0.57–0.69 mm), and a mean angular deviation of 1.42° (95% CI: 1.22–1.63°).
Substantial heterogeneity was observed across studies for all accuracy parameters (I2 = 87.1% for platform deviation, 73.3% for apical deviation, and 85.0% for angular deviation), reflecting variability in study design and clinical scenarios. Despite this heterogeneity, accuracy outcomes were consistent across different rehabilitative contexts, including single-tooth gaps, partially edentulous cases, and complete-arch restorations, with low standard deviations reported in individual studies. Across all included investigations, robotic systems demonstrated highly reproducible accuracy outcomes across different clinical scenarios. The certainty of evidence for accuracy outcomes was graded as moderate, primarily limited by heterogeneity and the absence of randomized controlled trials.
Yu et al. [34] synthesized evidence from 8 clinical studies including 109 patients and 242 dental implants, together with 13 preclinical investigations, to evaluate the positional accuracy of robot-assisted dental implant placement. In the pooled analysis of clinical data, robotic systems achieved a mean platform deviation of 0.68 mm (95% CI: 0.57–0.79 mm), a mean apical deviation of 0.67 mm (95% CI: 0.58–0.75 mm), and a mean angular deviation of 1.69° (95% CI: 1.25–2.12°). Considerable heterogeneity was observed across studies for all parameters (I2 > 85%), reflecting variability in study design, patient characteristics, and robotic platforms.
Subgroup analyses demonstrated no significant differences in accuracy between partially and fully edentulous patients, with pooled platform deviations of 0.65 mm and 0.69 mm, apical deviations of 0.66 mm and 0.67 mm, and angular deviations of 1.70° and 1.46°, respectively. In preclinical models, pooled deviations remained within a similar sub-millimetric range, with a mean platform deviation of 0.72 mm (95% CI: 0.58–0.86 mm), mean apical deviation of 0.90 mm (95% CI: 0.74–1.06 mm), and mean angular deviation of 1.46° (95% CI: 1.22–1.70°). Across both clinical and experimental settings, robot-assisted implant placement consistently achieved linear deviations below 1 mm and angular deviations around 1.5–2°, with no reported surgical complications or adverse events.
Khaohoen et al. [35] conducted a large systematic review and meta-analysis evaluating the accuracy of static, dynamic, and robot-assisted implant surgery, including 67 clinical studies and 5673 implants, of which two clinical investigations (44 implants) specifically assessed robot-assisted implant placement. In the subgroup analysis focusing on robotic systems, robot-assisted surgery demonstrated the lowest deviations across all three spatial domains when compared with static and dynamic navigation. The pooled analysis showed a mean coronal deviation of 0.81 mm (95% CI: 0.37–1.25 mm), a mean apical deviation of 0.77 mm (95% CI: 0.43–1.11 mm), and a mean angular deviation of 1.71° (95% CI: 0.04–3.38°).
When contrasted with other digital navigation modalities within the same meta-analysis, robotic systems achieved markedly lower deviations than static navigation (coronal: 1.11 mm; apical: 1.44 mm; angular: 3.58°) and dynamic navigation (coronal: 1.18 mm; apical: 1.36 mm; angular: 3.51°). These findings indicate that robot-assisted implant placement consistently reached sub-millimetric linear accuracy and angular deviations below 2°, representing the highest precision among computerized guidance approaches evaluated. Despite the superior geometric accuracy, the authors emphasized that the robotic subgroup was limited by the small number of clinical studies and implants, and that heterogeneity remained substantial due to differences in robotic platforms, support mechanisms, and registration workflows. Consequently, while robotic systems showed a clear accuracy advantage, the certainty of evidence was considered moderate, and further controlled clinical trials were deemed necessary to confirm the robustness and generalizability of these findings. Table 3 summarizes the primary outcomes, navigation characteristics, and secondary findings reported in the included systematic reviews.

Table 3.
Primary outcomes, robotic navigation characteristics, and secondary findings.
3.4. Secondary Outcomes: Comparison with Non-Robotic Navigation Systems
3.4.1. Static and Dynamic Computer-Assisted Implant Surgery
Static and dynamic CAIS were consistently reported as secondary comparators in reviews primarily focused on robotic systems. Both modalities demonstrated significant improvements in accuracy over freehand surgery, with overlapping ranges of coronal, apical, and angular deviations. However, their performance remained inferior to that of robotic systems, particularly in terms of variability and susceptibility to operator- and workflow-dependent errors.
3.4.2. Augmented Reality-Guided Implant Placement
Augmented reality-based systems were evaluated as exploratory or emerging technologies and were generally reported as secondary outcomes. Accuracy improvements over freehand placement were modest and less consistent than those observed with robotic or conventional navigation systems, reflecting the early developmental stage of AR technologies.
3.4.3. Operative Efficiency and Safety
Robot-assisted workflows were consistently associated with longer setup and operative times, particularly during early adoption phases. Reviews reporting quantitative data described increases in total surgical time ranging from approximately 10 to 25 min compared with static or dynamic navigation, largely attributable to system calibration and registration procedures. No consistent increase in intraoperative or postoperative complication rates was observed for robotic systems, with reported adverse event incidences comparable to those of static and dynamic CAIS. Learning curve effects were repeatedly identified as a determinant of operative efficiency, with reductions in setup time reported after initial case series.
Clinical recommendations derived from the umbrella-level evidence are outlined in Table 4. The specific accuracy domains enhanced by each digital navigation modality are summarized in Table 5.

Table 4.
Clinical recommendations for digital navigation in dental implant place based on systematic review evidence.

Table 5.
Accuracy domains improved by digital navigation modalities in dental implantology.
3.5. Methodological Quality (AMSTAR 2)
The methodological quality of the seven included systematic reviews was assessed using AMSTAR 2 (Table 6). Overall, the quality ranged from low to high, with the majority of reviews achieving moderate methodological confidence. The highest-rated reviews demonstrated prospective protocol registration, comprehensive literature searches, duplicate study selection and data extraction, and appropriate risk-of-bias assessment. Common methodological limitations across lower-rated reviews included the absence of protocol registration, partial reporting of excluded studies, and limited consideration of risk-of-bias findings in the interpretation of results. Despite these limitations, all included reviews met the minimum criteria for inclusion in an umbrella review.

Table 6.
Methodological quality of the included systematic reviews assessed using AMSTAR 2.
3.6. Certainty of Evidence (ConQual Assessment)
The certainty of evidence for the primary outcomes related to robot-assisted implant surgery was evaluated using the JBI ConQual approach (Table 7). Confidence in the evidence was moderate to high for accuracy-related outcomes, including coronal, apical, and angular deviations, reflecting consistent findings across reviews and relatively precise effect estimates. In contrast, the certainty of evidence was low for clinical outcomes such as implant survival and complication rates, owing to methodological limitations, heterogeneity, and imprecision. These findings indicate that while evidence supporting the superior accuracy of robotic systems is robust, evidence linking these accuracy gains to clinical outcomes remains limited.

Table 7.
ConQual assessment of confidence in evidence for primary outcomes (robot-assisted implant surgery).
3.7. Overlap of Primary Studies
Overlap of primary studies across the included systematic reviews was assessed using the Corrected Covered Area (CCA) method (Table 8). The calculated CCA value of 8.4% indicated a moderate degree of overlap among reviews. Several foundational studies on robotic and guided implant surgery were included in multiple reviews; however, the level of overlap was not excessive and did not compromise the independence of the evidence synthesis. Accordingly, the findings of this umbrella review were considered to reflect a balanced integration of overlapping yet methodologically distinct systematic reviews. Given the moderate degree of overlap (CCA = 8.4%), the potential for redundancy was considered limited. To mitigate the impact of overlapping primary studies, narrative synthesis prioritized consistency of effect direction rather than study counts, and conclusions were cross-checked against the highest-quality and most recent reviews within each device category. As such, the observed overlap is unlikely to have materially affected the independence or robustness of the umbrella-level conclusions.

Table 8.
Overlap of primary studies across included systematic reviews (Corrected Covered Area—CCA).
3.8. Summary of Key Findings
In summary, the umbrella-level evidence indicates that robot-assisted implant surgery achieves the highest degree of placement accuracy currently reported in dental implantology. While static and dynamic navigation systems remain effective accuracy-enhancing tools, their performance does not consistently match that of robotic platforms. However, the clinical significance of the superior accuracy achieved with robotic systems remains uncertain, as robust evidence linking these gains to improved long-term outcomes is still lacking.
4. Discussion
This umbrella review synthesized the highest level of evidence currently available on robot-assisted implant surgery (RAIS) in dental implantology, with placement accuracy as the primary outcome and non-robotic navigation modalities considered as secondary comparators. The findings consistently indicate that robotic systems achieve the highest degree of geometric accuracy reported to date, characterized by sub-millimetric coronal and apical deviations and angular deviations frequently below 2°. These results were robust across the included systematic reviews and meta-analyses, positioning RAIS at the top of the accuracy hierarchy among digital guidance technologies.
The superior accuracy observed with RAIS is biologically and mechanically plausible. Unlike static or dynamic computer-assisted implant surgery (CAIS), robotic systems introduce mechanical constraint into the surgical workflow, actively limiting deviations from the planned trajectory [9,36]. By reducing reliance on manual hand control, robotic guidance minimizes involuntary movements, tremor-related errors, and fatigue-induced variability [37]. This mechanical stabilization appears to be the key differentiating factor underlying the consistently lower deviation values reported across reviews [30,33,38].
Moreover, robotic systems integrate real-time positional feedback with preoperative planning in a closed-loop environment, allowing continuous correction of deviations during osteotomy and implant insertion [39]. This contrasts with static guidance, which is susceptible to cumulative errors arising from guide fabrication, seating instability, and limited intraoperative adaptability, and with dynamic navigation, which remains dependent on the surgeon’s manual execution despite visual feedback. Consequently, RAIS represents a conceptual shift from guidance-assisted surgery to constraint-assisted surgery, explaining its superior reproducibility.
Although robot-assisted implant surgery demonstrates superior geometric accuracy, accuracy alone is not sufficient to guarantee improved implant survival or better patient-centered outcomes. The translation of technical precision into clinical benefit appears to be modulated by multiple factors, including anatomical complexity, surgical risk profile, and operator experience. Consequently, the clinical value of RAIS is likely maximized in demanding or high-risk scenarios, rather than as a universal replacement for established static or dynamic digital workflows.
This finding mirrors observations previously reported for static and dynamic CAIS, where substantial improvements in accuracy compared with freehand surgery did not consistently result in superior survival or complication profiles [9,14,36]. RAIS appears to extend this pattern, achieving even greater precision without yet demonstrating proportional clinical gains. These results caution against equating technological advancement with clinical superiority and highlight the importance of outcome hierarchies when evaluating novel surgical technologies [39].
Mehta et al. [40] published an umbrella review focusing on the accuracy of robot-assisted dental implant surgery, primarily synthesizing positional deviation outcomes reported across existing systematic reviews. While their work confirms the superior geometric accuracy of robotic systems, the present umbrella review extends the available evidence in several important ways. In contrast to Mehta et al., our review incorporates a structured overlap analysis using the corrected covered area (CCA), applies a formal certainty-of-evidence appraisal through the ConQual framework, and systematically contrasts robotic systems with static and dynamic navigation approaches within a unified interpretative framework. Furthermore, whereas Mehta et al. [40] largely treat accuracy as an isolated endpoint, the present review explicitly addresses the clinical relevance of accuracy gains, emphasizing the limited and inconsistent evidence linking increased robotic precision to improved implant survival, reduced complication rates, or enhanced patient-reported outcomes. By integrating modality-specific clinical considerations and domain-based accuracy assessment, this umbrella review offers a more clinically contextualized and decision-oriented synthesis of robot-assisted implant surgery.
From a practical perspective, increased operative time may offset accuracy gains in routine clinical practice, particularly in high-volume settings. Moreover, the financial costs associated with robotic systems, including acquisition, maintenance, and training introduce additional barriers to widespread adoption. These considerations underscore the importance of contextualizing accuracy gains within broader cost–benefit and workflow frameworks [22]. Although learning curve effects were not uniformly quantified across reviews, several authors reported that improvements in operative efficiency occurred after initial case series, suggesting that robotic systems require a defined training phase before workflow stabilization. This observation aligns with evidence from dynamic navigation systems, where operator experience has been shown to significantly influence accuracy and efficiency outcomes.
When considered as secondary outcomes, static and dynamic CAIS consistently demonstrated clinically acceptable accuracy improvements over freehand surgery, with overlapping deviation ranges and comparable safety profiles. The absence of a clear accuracy superiority between static and dynamic systems across reviews suggests that these modalities may be interchangeable in many clinical scenarios, with selection driven by workflow preferences and operator experience rather than precision alone.
The conclusions of this umbrella review must be interpreted in light of several methodological limitations inherent to the existing evidence. First, a substantial proportion of robotic accuracy data originates from in vitro or cadaveric studies, which may overestimate clinical performance under controlled conditions. Second, overlap of primary studies across systematic reviews limits independent evidence synthesis and may amplify the influence of a small number of foundational investigations.
Third, heterogeneity in accuracy definitions, reference standards, and outcome reporting complicates cross-study comparisons. While coronal, apical, and angular deviations are commonly reported, thresholds for clinical relevance are rarely predefined, and patient-centered outcomes remain underrepresented. Finally, long-term follow-up data are scarce, limiting assessment of whether robotic accuracy influences outcomes beyond the immediate postoperative period.
From a clinical standpoint, the findings of this umbrella review suggest that RAIS should currently be viewed as a precision-enhancing adjunct rather than a universally superior replacement for established navigation techniques. Its use may be justified in complex cases where minimizing outlier risk is critical, but routine adoption cannot yet be supported by robust evidence of improved clinical outcomes.
Given the substantial heterogeneity in study designs, clinical indications, comparator modalities, and outcome definitions across the included systematic reviews, as well as the moderate overlap of primary studies, a narrative umbrella synthesis was considered the most appropriate approach. Quantitative re-pooling at the umbrella level could be misleading by artificially inflating precision and obscuring methodological dependencies among reviews.
The findings of the present umbrella review should be interpreted within the context of the extensive body of evidence generated over the past decade on computer-assisted implant surgery. Seminal systematic reviews and meta-analyses on static guided surgery consistently demonstrated submillimetric coronal and apical deviations with angular errors generally below 4°, although clinically relevant variability related to guide design, manufacturing accuracy, and workflow execution was also reported [41,42]. Similarly, systematic reviews focusing on dynamic navigation systems reported improved accuracy compared with freehand implant placement and accuracy levels comparable to static guidance, while emphasizing the influence of operator experience, calibration procedures, and real-time tracking stability on final implant positioning [43,44,45,46]. Evidence syntheses addressing broader digital and navigational workflows further highlighted that accuracy gains are highly dependent on the integrity of each step of the digital chain, from image acquisition to surgical execution, particularly in partially edentulous patients and complex anatomical scenarios [21,47]. Additional qualitative and quantitative reviews on specialized applications, such as zygomatic implants and model-based navigation assessments, reinforced the reliability of dynamic navigation while underscoring technical and learning-related constraints [39,48]. Against this established pre-robotic evidence base, the consistently lower linear and angular deviations observed for robot-assisted implant surgery in the present umbrella review appear to reflect a natural technological progression rather than an isolated performance gain. Importantly, classic navigation reviews repeatedly identified learning curve effects and workflow-dependent determinants of accuracy and efficiency, providing a conceptual framework that supports the interpretation of robotic systems as precision-enhancing tools whose clinical benefits are maximized following adequate training and procedural standardization [44].
Beyond dental implantology-specific evidence, recent robotic research highlights broader advances in robotic systems, sensing, and autonomous workflows that are relevant to the interpretation of robot-assisted technologies in surgery. Emerging studies emphasize improvements in robotic control architectures, real-time spatial mapping, and human–robot interaction, underscoring how integration of robotics with advanced imaging and navigation can enhance task precision and workflow standardization [49,50]. Furthermore, developments in robotic mapping and assessment tools demonstrate the feasibility of low-cost, autonomous systems capable of generating accurate spatial representations with minimal operator input, reinforcing the concept that automation and robotics may reduce operator-dependent variability across complex procedural environments [51]. Although these studies are not specific to dental implant placement, they provide a complementary technological framework that supports the observed accuracy gains and workflow stabilization associated with robot-assisted implant surgery.
Based on the available evidence, robot-assisted implant surgery may offer the greatest clinical value in anatomically demanding situations, such as proximity to critical anatomical structures, full-arch rehabilitation, or high-risk cases requiring maximal positional control. Conversely, current evidence does not support the routine replacement of well-established static or dynamic guided implant techniques in standard clinical scenarios. Future research should prioritize well-designed randomized clinical trials comparing RAIS with static and dynamic CAIS in clearly defined clinical scenarios, incorporating standardized accuracy metrics and long-term outcome measures. Additionally, studies assessing cost-effectiveness, learning curves, and patient-reported outcomes are essential to inform evidence-based integration of robotic systems into implant practice.
5. Conclusions
In summary, this umbrella review demonstrates that robot-assisted implant surgery achieves the highest placement accuracy currently reported in dental implantology. However, the translation of these technical gains into meaningful clinical benefits remains insufficiently supported by existing evidence. Bridging the gap between precision and patient-centered outcomes represents the next critical challenge in the evolution of robotic implant surgery.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/app16042159/s1, Table S1. Search Queries.
Author Contributions
Conceptualization, L.D., E.F. and C.B.; methodology, L.D., V.C.-B., C.M.-G., J.B.A. and C.B.; formal analysis, L.D. and E.F.; investigation, L.D., J.B.A., C.M.-G., V.C.-B. and C.B.; data curation, L.D. and E.F.; writing—original draft preparation, L.D.; writing—review and editing, E.F., V.C.-B., C.M.-G., J.B.A. and C.B.; visualization, L.D. and E.F.; supervision, E.F.; project administration, E.F. All authors have read and agreed to the published version of the manuscript.
Funding
This review did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. No external financial support, including sponsorship, material provision, or data access, was provided for the development of this work. The authors conducted the review independently, and no funding body had any role in the design, execution, data collection, analysis, interpretation, or decision to submit the manuscript for publication.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data supporting the findings of this study are contained within the article. No new datasets were generated or analyzed for this review.
Acknowledgments
The authors thank the Faculty of Dentistry, University of Chile, for academic support. No external technical or material assistance was received.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| Abbreviation | Definition |
| RAIS | Robot-Assisted Implant Surgery |
| CAIS | Computer-Assisted Implant Surgery |
| sCAIS | Static Computer-Assisted Implant Surgery |
| dCAIS | Dynamic Computer-Assisted Implant Surgery |
| r-CAIS | Robotic Computer-Assisted Implant Surgery |
| ADIR | Autonomous Dental Implant Robotics |
| CBCT | Cone-Beam Computed Tomography |
| AR | Augmented Reality |
| PSI | Patient-Specific Instrumentation |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| JBI | Joanna Briggs Institute |
| AMSTAR 2 | A Measurement Tool to Assess Systematic Reviews 2 |
| ConQual | Confidence in the Qualitative Synthesis of Evidence |
| CI | Confidence Interval |
| SMD | Standardized Mean Difference |
| CCA | Corrected Covered Area |
References
- Peitsinis, P.R.; Blouchou, A.; Chatzopoulos, G.S.; Vouros, I.D. Optimizing Implant Placement Timing and Loading Protocols for Successful Functional and Esthetic Outcomes: A Narrative Literature Review. J. Clin. Med. 2025, 14, 1442. [Google Scholar] [CrossRef] [PubMed]
- Nagni, M.; D’Orto, B.; De Cunto, R.; Cattoni, F.; Vinci, R. Implant-Prosthetic Rehabilitation with Immediately Loaded Post-Extractive Implants: Retrospective Clinical Cohort Study at 18-Month Follow-Up. Appl. Sci. 2025, 15, 6080. [Google Scholar] [CrossRef]
- Sabău, D.T.; Saitos, P.; Moca, R.T.; Juncar, R.I.; Juncar, M. Mechanical and Biological Complications Two Years After Full-Arch Implant-Supported Prosthetic Rehabilitation: A Retrospective Clinical Study. Clin. Pract. 2025, 15, 134. [Google Scholar] [CrossRef] [PubMed]
- Kafedzhieva, A.; Vlahova, A.; Chuchulska, B. Digital Technologies in Implantology: A Narrative Review. Bioengineering 2025, 12, 927. [Google Scholar] [CrossRef]
- Pisla, D.; Bulbucan, V.; Hedesiu, M.; Vaida, C.; Pusca, A.; Mocan, R.; Tucan, P.; Dinu, C.; Pisla, D. Accuracy of Navigation and Robot-Assisted Systems for Dental Implant Placement: A Systematic Review. Dent. J. 2025, 13, 537. [Google Scholar] [CrossRef]
- Tsokkou, S.; Konstantinidis, I.; Keramas, A.; Kiosis, G.; Skourtsidis, K.; Alexiou, D.; Keskesiadou, G.N.; Karachrysafi, S.; Papamitsou, T.; Chatzistefanou, I. Comparative Analysis of Fully Guided and Free-Hand Orthognathic Surgery: Advancements, Precision, and Clinical Outcomes. Dent. J. 2025, 13, 260. [Google Scholar] [CrossRef]
- Liu, C.; Liu, Y.; Xie, R.; Li, Z.; Bai, S.; Zhao, Y. The evolution of robotics: Research and application progress of dental implant robotic systems. Int. J. Oral Sci. 2024, 16, 28. [Google Scholar] [CrossRef]
- Perez, A.; Lombardi, T. Integration and Innovation in Digital Implantology—Part I: Capabilities and Limitations of Contemporary Workflows: A Narrative Review. Appl. Sci. 2025, 15, 12214. [Google Scholar] [CrossRef]
- Sadilina, S.; Vietor, K.; Doliveux, R.; Siu, A.; Chen, Z.; Al-Nawas, B.; Mattheos, N.; Pozzi, A. Beyond Accuracy: Clinical Outcomes of Computer Assisted Implant Surgery. Clin Exp. Dent. Res. 2025, 11, e70129. [Google Scholar] [CrossRef]
- Marquez Bautista, N.; Meniz-García, C.; López-Carriches, C.; Sánchez-Labrador, L.; Cortés-Bretón Brinkmann, J.; Madrigal Martínez-Pereda, C. Accuracy of Different Systems of Guided Implant Surgery and Methods for Quantification: A Systematic Review. Appl. Sci. 2024, 14, 11479. [Google Scholar] [CrossRef]
- Shusterman, A.; Nashef, R.; Tecco, S.; Mangano, C.; Mangano, F. Implant placement using mixed reality-based dynamic navigation: A proof of concept. J. Dent. 2024, 149, 105256. [Google Scholar] [CrossRef] [PubMed]
- Werny, J.G.; Frank, K.; Fan, S.; Sagheb, K.; Al-Nawas, B.; Narh, C.T.; Schiegnitz, E. Freehand vs. computer-aided implant surgery: A systematic review and meta-analysis—Part 1: Accuracy of planned and placed implant position. Int. J. Implant Dent. 2025, 11, 35. [Google Scholar] [CrossRef] [PubMed]
- Sarhan, M.M.; Ibrahim, E.A.; Ezzelarab, S.; Marei, M.K. Navigating the future of guided dental implantology: A scoping review. Int. J. Med. Robot. Comput. Assist. Surg. 2024, 20, e2627. [Google Scholar] [CrossRef] [PubMed]
- Pimkhaokham, A.; Jiaranuchart, S.; Kaboosaya, B.; Arunjaroeunsuk, S.; Subbalekha, K.; Mattheos, N. Can computer-assisted implant surgery improve clinical outcomes and reduce the frequency and intensity of complications in implant dentistry? A critical review. Periodontol 2000 2022, 90, 197–223. [Google Scholar] [CrossRef]
- Bahrami, R.; Pourhajibagher, M.; Nikparto, N.; Bahador, A. Robot-assisted dental implant surgery procedure: A literature review. J. Dent. Sci. 2024, 19, 1359–1368. [Google Scholar] [CrossRef]
- Tuygunov, N.; Mattheos, N.; Tsoi, J.; Ruzikulova, M.; Osathanon, T.; Samaranayake, L. Contemporary Applications of Robotic Systems in Dental Implantology: A Review. Int. Dent. J. 2026, 76, 109335. [Google Scholar] [CrossRef]
- Webb, M.M.; Bridges, P.; Aruparayil, N.; Chugh, C.; Beacon, T.; Singh, T.; Sawhney, S.S.; Bains, L.; Hall, R.; Jayne, D.; et al. The RAIS Device for Global Surgery: Using a Participatory Design Approach to Navigate the Translational Pathway to Clinical Use. IEEE J. Transl. Eng. Health Med. 2022, 10, 3700212. [Google Scholar] [CrossRef]
- James, J.R.; Kharat, A.; Chinnakutti, S.; Kamble, S.; Mandal, M.; Das, A. The Future of Dental Implants: A Narrative Review of Trends, Technologies, and Patient Considerations. Cureus 2025, 17, e90380. [Google Scholar] [CrossRef]
- Aghaloo, T.; Hadaya, D.; Schoenbaum, T.R.; Pratt, L.; Favagehi, M. Guided and Navigation Implant Surgery: A Systematic Review. Int. J. Oral Maxillofac. Implant. 2023, 38, 7–15. [Google Scholar] [CrossRef]
- Araujo-Corchado, E.; Pardal-Pelaez, B. Computer-Guided Surgery for Dental Implant Placement: A Systematic Review. Prosthesis 2022, 4, 540–553. [Google Scholar] [CrossRef]
- Gargallo-Albiol, J.; Barootchi, S.; Salomó-Coll, O.; Wang, H.L. Advantages and disadvantages of implant navigation surgery. A systematic review. Ann. Anat. 2019, 225, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Rana, M.M.; Akash, M.M.H.; Mridula, A.T.; Al-Mamoon, I.A.; Rahman, Q.B. Robotics, artificial intelligence, and computer vision in dental implant surgery: A systematic review of accuracy, efficiency, and future directions. J. Robot. Surg. 2026, 20, 42. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Debnath, A.; Paul, P.; Banerjee, T.N. Comparison of accuracy in freehand versus computer-Assisted (dynamic and static) dental implant placement: A systematic review and meta-Analysis. J. Indian Prosthodont. Soc. 2025, 25, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Bover-Ramos, F.; Viña-Almunia, J.; Cervera-Ballester, J.; Peñarrocha-Diago, M.; García-Mira, B. Accuracy of Implant Placement with Computer-Guided Surgery: A Systematic Review and Meta-Analysis Comparing Cadaver, Clinical, and In Vitro Studies. Int. J. Oral Maxillofac. Implant. 2018, 33, 101–115. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Hilton, M. JBI Critical appraisal checklist for systematic reviews and research syntheses. J. Can. Health Libr. Assoc. 2024, 45, 180–183. [Google Scholar] [CrossRef]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, 358, j4008. [Google Scholar] [CrossRef]
- Yang, J.; Li, H.N. Accuracy assessment of robot-assisted implant surgery in dentistry: A systematic review and meta-analysis. J. Prosthet. Dent. 2024, 132, 747.e1–747.e15. [Google Scholar] [CrossRef]
- Jain, S.; Sayed, M.E.; Ibraheem, W.I.; Ageeli, A.A.; Gandhi, S.; Jokhadar, H.F.; AlResayes, S.S.; Alqarni, H.; Alshehri, A.H.; Huthan, H.M.; et al. Accuracy Comparison between Robot-Assisted Dental Implant Placement and Static/Dynamic Computer-Assisted Implant Surgery: A Systematic Review and Meta-Analysis of In Vitro Studies. Medicina 2024, 60, 11. [Google Scholar] [CrossRef]
- Khan, M.; Javed, F.; Haji, Z.; Ghafoor, R. Comparison of the positional accuracy of robotic guided dental implant placement with static guided and dynamic navigation systems: A systematic review and meta-analysis. J. Prosthet. Dent. 2024, 132, 746.e1–746.e8. [Google Scholar] [CrossRef]
- Implants, D.; Ravipati, V.; Mathews, J.; Altuhafy, M.; Nagi, R.; Khan, J. Accuracy of Dental Implant Placement using Robotics. A Systematic Review of in vitro Studies. Eur. J. Prosthodont. Restor. Dent. 2024, 32, 287–300. [Google Scholar] [CrossRef]
- Pozzi, A.; Carosi, P.; Lorenzi, C.; Chow, J.; Wang, H.L.; Gallucci, G.O. In Vivo Accuracy of Autonomous Dental Implant Robotic Surgery: Systematic Review and Meta-analysis. Int. J. Oral Maxillofac. Implant. 2025, 40, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.B.; Tao, B.X.; Wang, F.; Wu, Y.Q. Accuracy assessment of dynamic navigation during implant placement: A systematic review and meta-analysis of clinical studies in the last 10 years. J. Dent. 2023, 135, 104567. [Google Scholar] [CrossRef] [PubMed]
- Khaohoen, A.; Powcharoen, W.; Yoda, N.; Rungsiyakull, C.; Rungsiyakull, P. Accuracy in dental implant placement: A systematic review and meta-analysis comparing computer-assisted (static, dynamic, robotics) and noncomputer-assisted (freehand, conventional guide) approaches. J. Prosthet. Dent. 2025, 134, 91.e1–91.e25. [Google Scholar] [CrossRef] [PubMed]
- Reiff, F.S.; Kroeger, A.; Roehling, S.; Reiff, C.; Kebschull, M. Accuracy of Freehand, Static, and Dynamic Computer-Assisted Implant Placement: A Systematic Review and Meta-Analysis. J. Periodontal Res. 2025. [Google Scholar] [CrossRef]
- Traboulsi-Garet, B.; Jorba-García, A.; Bara-Casaus, J.; Camps-Font, O.; Valmaseda-Castellón, E.; Figueiredo, R.; Sánchez-Garcés, M.À. Accuracy of freehand surgery, static and dynamic computer assisted surgery on zygomatic implant placement: A systematic review and meta-analyses. J. Cranio-Maxillofac. Surg. 2025, 53, 301–311. [Google Scholar] [CrossRef]
- Pozzi, A.; Polizzi, G.; Moy, P.K. Guided surgery with tooth-supported templates for single missing teeth: A critical review. Eur. J. Oral Implantol. 2016, 9, 135–153. [Google Scholar]
- Ramezanzade, S.; Keyhan, S.O.; Tuminelli, F.J.; Fallahi, H.R.; Yousefi, P.; Lopez-Lopez, J. Dynamic-Assisted Navigational System in Zygomatic Implant Surgery: A Qualitative and Quantitative Systematic Review of Current Clinical and Cadaver Studies. J. Oral Maxillofac. Surg. 2021, 79, 799–812. [Google Scholar] [CrossRef]
- Mehta, V.; Mathur, A.; Bhadania, M.; Patil, P.G. Accuracy assessment of robot-assisted dental implant surgery: An umbrella review of systematic reviews. J. Prosthet. Dent. 2025, 134, 1630.e1–1630.e12. [Google Scholar] [CrossRef]
- Tahmaseb, A.; Wismeijer, D.; Coucke, W.; Derksen, W. Computer technology applications in surgical implant dentistry: A systematic review. Int. J. Oral Maxillofac. Implant. 2014, 29, 25–42. [Google Scholar] [CrossRef] [PubMed]
- Tahmaseb, A.; Wu, V.; Wismeijer, D.; Coucke, W.; Evans, C. The accuracy of static computer-aided implant surgery: A systematic review and meta-analysis. Clin. Oral Implant. Res. 2018, 29, 416–435. [Google Scholar] [CrossRef] [PubMed]
- Jorba-García, A.; González-Barnadas, A.; Camps, O.; Figueiredo, R.; Valmaseda-Castellõn, E. Accuracy assessment of dynamic computer–aided implant placement: A systematic review and meta-analysis. Clin. Oral Investig. 2021, 25, 2479–2494. [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, G.; Ferri, A.; Del Fabbro, M.; Prati, C.; Gandolfi, M.G.; Marchetti, C. Dynamic Navigation in Implant Dentistry: A Systematic Review and Meta-analysis. Int. J. Oral Maxillofac. Implant. 2021, 36, e121–e140. [Google Scholar] [CrossRef]
- Wei, S.M.; Zhu, Y.; Wei, J.X.; Zhang, C.N.; Shi, J.Y.; Lai, H.C. Accuracy of dynamic navigation in implant surgery: A systematic review and meta-analysis. Clin. Oral Implant. Res. 2021, 32, 383–393. [Google Scholar] [CrossRef]
- Wang, F.; Wang, Q.; Zhang, J. Role of Dynamic Navigation Systems in Enhancing the Accuracy of Implant Placement: A Systematic Review and Meta-Analysis of Clinical Studies. J. Oral Maxillofac. Surg. 2021, 79, 2061–2070. [Google Scholar] [CrossRef]
- Siqueira, R.; Chen, Z.; Galli, M.; Saleh, I.; Wang, H.L.; Chan, H.L. Does a fully digital workflow improve the accuracy of computer-assisted implant surgery in partially edentulous patients? A systematic review of clinical trials. Clin. Implant. Dent. Relat. Res. 2020, 22, 660–671. [Google Scholar] [CrossRef]
- Emery, R.W.; Merritt, S.A.; Lank, K.; Gibbs, J.D. Accuracy of Dynamic Navigation for Dental Implant Placement-Model-Based Evaluation. J. Oral Implantol. 2016, 42, 399–405. [Google Scholar] [CrossRef]
- Li, K.; Li, T.; Guo, G.; Liu, L.; Jiang, Z.; Ma, L.; Li, Y.; Jia, J. Dynamic Navigation vs. Static Navigation in Implant Placement: A Meta-Analysis. J. Dent. 2024, 151, 105395. [Google Scholar] [CrossRef]
- Schmidt, L.; Cree, I.; Campbell, F. Digital Tools to Support the Systematic Review Process: An Introduction. J. Eval. Clin. Pract. 2025, 31, e70100. [Google Scholar] [CrossRef]
- Su, X.; Zamora, D.C.; Froehlich, J.E. RAIS: Towards A Robotic Mapping and Assessment Tool for Indoor Accessibility Using Commodity Hardware. In Proceedings of the 26th International ACM SIGACCESS Conference on Computers and Accessibility, St. John’s, NL, Canada, 27–30 October 2024. Article 86. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.