Abstract
The study evaluates the effects of different types of popular exercises on muscle mass reduction during diet-induced body mass loss in overweight and obese women. Participants were randomly allocated to three exercises (Whole-Body Electromyostimulation (WB-EMS), Pilates, Yoga) combined with nutritional guidance and one nutritional guidance-only group (control). All exercise protocols specified a 2 × 50 min (Pilates, Yoga) or 2 × 20 min (WB-EMS) training, respectively. Weekly applied nutritional guidance focused on −500 kcal/d of energy restriction but increased protein intake. A total 221 overweight/obese women 19–65 years old and largely compliant with the exercise and nutritional guidance protocol were included. We observed significant decreases in body mass in all groups (−3.9 to 6.0 kg, all p < 0.001), with the most pronounced reduction in the nutritional guidance-only group. Apart from WB-EMS (−166 ± 799 g, p = 0.14), all the other groups significantly lost muscle mass (Pilates −343 ± 774 g, p = 0.003; Yoga: −507 ± 801 g, p < 0.001), however, compared to control (−1113 ± 881 g, p < 0.001), all the groups showed significantly more favorable results. WB-EMS, Pilates, and Yoga were associated with a reduced loss of muscle mass during diet-induced weight loss, indicating their potential role as adjunctive interventions in overweight and obese individuals.
1. Introduction
Sedentary life is gradually increasing due to technological developments and longer working hours. This tendency is reflected in the high prevalence of overweight and obesity worldwide [1]. Overweight and obesity are commonly defined according to body mass index (BMI), with values ≥ 25 kg/m2 indicating overweight and ≥30 kg/m2 indicating obesity [2]. Healthy nutrition and physical exercise play a key role in weight management and improvement of body composition [3]. Diet-induced weight loss typically refers to a negative energy balance achieved through caloric restriction, which leads to a reduction in body mass but may also result in an undesired loss of lean tissue [4]. However, very few adults are regularly engaged in sports and physical exercise [5,6]. Modern-age, customized types of exercises going beyond strenuous, tiring, and boring training routines, might increase participation and sustainability of exercise habits.
Whole-body electromyostimulation (WB-EMS) is a novel training technology that applies electrical impulses from an external device to elicit muscle contractions. WB-EMS can stimulate all the main muscle groups simultaneously with dedicated stimulus intensity per electrode; thus, WB-EMS can be considered a high caloric burning activity [5]. By inducing involuntary muscle contractions, WB-EMS may provide a sufficient anabolic stimulus to preserve skeletal muscle mass during periods of reduced energy intake [7]. Further, since adjuvant intense voluntary exercises are not necessary [8], WB-EMS can be considered as time efficient and joint-friendly aspects that attract many people unable or unmotivated to exercise conventionally. A large variety of WB-EMS studies addressed outcomes related to body composition [9,10]. A recent meta-analysis on WB-EMS-induced changes in lean body mass (LBM) and total body fat reported significant positive effects on both outcomes [11]. In parallel, there is some evidence that WB-EMS, be it with or without protein supplementation, reduces or even stops the reduction in LBM during hypocaloric diet [7,12].
Pilates has become one of the most popular organized physical activities, with many women participating in it to improve their physical, psychological, and social state [13]. Pilates offers a “core” musculature and stability workout in which fitness components, such as muscular strength and endurance, flexibility, balance, and cardiorespiratory endurance, are trained with the aim of connecting and conditioning body and mind [14]. Through repeated low- to moderate-intensity muscle activation, Pilates may contribute to the maintenance of muscle mass during weight loss interventions [15]. Pilates includes different modalities, with or without devices. “Reformer Pilates”, for example, involves the use of a machine (the reformer) to perform the exercises with a safe and effective technique [13]. Pilates has been linked to many positive effects on health-related outcomes, such as enhanced psychological well-being [16], improved spinal posture [16], augmented trunk stability, flexibility, dynamic balance [16], as well as favorable effects on body composition parameters [17,18].
Yoga is a holistic, mind–body exercise that improves physical, mental, emotional, and spiritual functions. Lifestyle-based exercises for health and wellness include physical postures (asana), breath work (pranayama), meditation (dhyana), stretching, mind–body awareness, and concentration of attention [19]. Depending on the style and postures applied, it can be considered both an aerobic- and strength-type exercise with a medium caloric burning character [5]. Sustained isometric contractions and weight-bearing postures in Yoga may help attenuate muscle mass loss during caloric restriction [20]. Yoga is recommended for people with chronic pain, stress, and anxiety, particularly those affected by musculoskeletal and mental health problems [21,22], i.e., cohorts with limited access to conventional fitness or exercise training programs. Despite its commonly assumed “soft” character, there is some evidence that Yoga favorably affects body composition in obese people [20,23,24].
In summary, there is evidence that WB-EMS, Pilates, and Yoga exert positive effects on body composition in overweight or obese individuals. However, their effects on body composition during energy restriction-induced weight loss remain unclear. This particularly refers to the reduction in skeletal muscle mass and fat-free or lean body mass occurring during weight loss interventions [25]. Therefore, the present study aimed to evaluate the effects of 12 weeks of WB-EMS, Pilates, and Yoga combined with nutritional guidance, compared with nutritional guidance alone (control group), on body composition during weight loss. We hypothesized that body mass loss would be similar between the exercise groups and the non-exercising control group (secondary hypothesis), whereas muscle mass loss would be attenuated in the exercise groups compared with the non-exercising control group (primary hypothesis).
2. Materials and Methods
2.1. Study Design
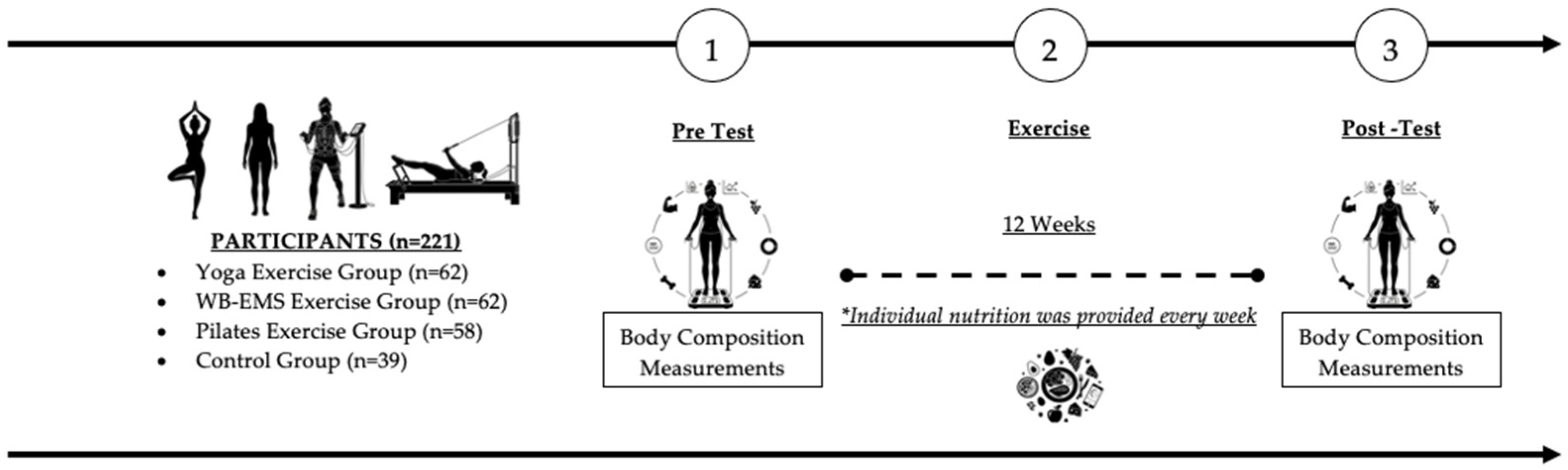
The objective of the present sub-analysis of a large 12-week randomized controlled trial (RCT) was to investigate the impact of three different exercise regimens (WB-EMS, Pilates, Yoga) combined with weekly nutritional guidance versus nutritional guidance alone on diet-induced loss of muscle mass in adult women. Of importance, only females 18–65 years old largely compliant with the exercise and nutritional protocol listed below were included in the present per protocol analysis. The study was planned, initiated, and conducted by the Institute of Sport Science, Karabuk University, Turkey. The University Ethics Committee approved the trial (Nr. 2021/664) that fully complies with the Helsinki Declaration “Ethical Principles for Medical Research Involving Human Subjects” [26]. The study was fully registered under ClinicalTrials.gov (NCT06584578). All participants were fully informed about the risks and benefits of the study and gave written informed consent. Figure 1 shows the design and procedures of the present study.
Figure 1.
Study Design of the present project.
2.2. Participants
All study participants were clients of the “Diyetterapi” Private Health Center (Karabuk, Turkey), closely affiliated with Karabuk University. Participants were recruited between May and June 2024 by personal contact and information and were assessed for eligibility applying the following criteria: (1) women and men, 18 years and older, (2) overweight (BMI: ≥25 kg/m2) or obese (BMI: ≥30 kg/m2), (3) no chronic diseases, (4) no contraindications for WB-EMS [27], (5) non-smokers and non-alcohol consumers, (6) occupations that require little physical activity, and (7) no or low exercise participation (<2 days/week) in slow paced sports (e.g., walking). In cases of doubt, the study physician decided on inclusion after personal interview. In summary, 400 women and men were included and enrolled in the trial (June 2024) and were randomly assigned to the four groups (i.e., Pilates, WB-EMS, Yoga, Control). However due to gender differences in baseline body composition and potentially varying exercise-induced changes in body mass, muscle mass and total body fat rate, the present sub-analysis focuses on (1) non-pregnant women, (2) 18 to a maximum of 65 years old, (3) with a BMI below 40.0 kg/m2, who were (4) largely compliant with the exercise (i.e., at least 22 of 24 exercise sessions) and nutritional guidance protocols (according to the assessment of the dietitian). In summary, 221 women (WB-EMS: n = 62, Pilates: n = 58, Yoga: n = 62, CG: n = 39) were eligible and included in the per-protocol analysis (PPA).
2.3. Randomization and Blinding
Participants allocated themselves to the study groups by drawing lots. Of note, the lots were prepared by a researcher not involved in the trial. Thus, neither participants nor researcher knew the allocation beforehand (“allocation concealment”). We applied a non-balanced allocation with higher sample sizes in the exercise groups (Figure 1). After the randomization procedure, the principal investigator (HDG) registered participants and instructed them in detail about the study specifications. The blinding strategy focused on the dietitian and research/test assistants who were not aware of the group status of the participants and were not allowed to ask.
2.4. Study Procedures
2.4.1. Study Outcomes
Primary study outcome
- changes in skeletal muscle mass (SMM) from baseline to the 12-week follow-up were assessed using bioelectrical impedance analysis (BIA).
Secondary study outcome
- changes in body mass (“body weight”) from baseline to 12-week FU as determined by BIA.
Explanatory outcome
- changes in body fat rate from baseline to 12-week FU as determined by BIA
2.4.2. Assessments
High emphasis was placed on the standardization of the tests. All the participants were requested to refrain from intense physical activity and exercise 48 h before the assessments. Baseline and FU assessments were consistently performed by the same research assistant using the identically calibrated devices, in exactly the same setting and at about the same time of the day (±90 min).
Anthropometric Measurements
Body height was determined by a calibrated stadiometer, body mass and body compositions were measured using direct segmental multifrequency bioimpedance analysis (DSM-BIA, InBody120, Seoul, Republic of Korea). This device measures impedance of the trunk, arms, and legs separately using a tetrapolar eight-point tactile electrode system that applies two frequencies (20 and 100 kHz). Briefly, BIA is a non-invasive technique that estimates body composition based on tissue resistance to a low-level electrical current. Under standardized conditions (e.g., a fasted state) with healthy men and women, multifrequency BIA systems has high methodological reliability and demonstrates stable day-to-day measurements of major body composition components [28].
Waist circumference was determined as the minimum circumference between the distal end of the rib cage and the top of the iliac crest along the midaxillary line.
Questionnaires
Detailed questionnaires asked for demographic parameters, lifestyle including physical activity, exercise, medication, diseases, and other health-related conditions. Follow-up questionnaires focused on changes in parameters with impact on body composition (i.e., new diseases/conditions, relevant changes in medications).
2.4.3. Changes in Trial Outcomes After Trial Commencement
No changes in trial outcomes were made after trial commencement.
2.4.4. Study Interventions
The study interventions were conducted over a period of 12 weeks (Figure 1) starting in June 2024. The training sessions for the Pilates and Yoga groups were set for two non-consecutive days per week, with each session lasting 50 min. Similarly, the training for the WB-EMS group took place two days per week on non-consecutive days, albeit with a duration of only 20 min/session [27]. The exercise sessions for the WB-EMS and Pilates groups were consistently guided and supervised by experienced and certified instructors, while Yoga exercises were conducted once a week in a joint, supervised setting and once a week at home. Participants were instructed to maintain their usual sleep patterns and abstain from changing physical activity and diet outside the study protocol. Participants were also advised to refrain from engaging in intense physical activity prior to the exercise sessions.
Whole-Body Electromyostimulation (WB-EMS) Program: Participants underwent continuously guided WB-EMS (miha bodytec®, Gersthofen, Germany) for 20 min twice a week (Table 1). The application of bipolar current at 85 Hz, 350 µs impulse width, and direct impulse increment was conducted with 10 electrode areas that stimulated both thighs and upper arms, gluteal, abdomen, chest, lower back, the latissimus dorsi region, and the upper back. Impulse phases of 30 s of EMS stimulation were interspersed by 10 s impulse breaks. Stimulus intensity was specified as rate of perceived exertion (RPE) “16”–“17” (“hard+” to “very hard”) on the Borg scale. This stimulus intensity was realized and maintained during the session in close cooperation between trainer and trainee. The participants were instructed to perform a series of fundamental physical exercises, including the plank, squat, lunge, crunch, push-up, high knee, and jumping jack, during the impulse phase. This approach is fully compliant with the updated international guidelines for the safe and effective use of WB-EMS [29].

Table 1.
Design of the Whole-Body Electromyostimulation program.
Reformer Pilates Exercise Program: The Reformer Exercise Program was conducted by a certified reformer trainer on two occasions per week for a period of 12 weeks. The program was based on a slightly modified version of a previously published protocol [30]. Exercises were preceded by Pilates exercises using a Reformer machine (Pen Pilates, Istanbul, Turkey). The participants were provided with comprehensive information regarding the reformer machine and the exercise program by the trainer. Reformer Pilates exercises were performed as general muscle strengthening and flexibility exercises under the supervision of the trainer. The reformer program is outlined in Table 2. Over the course of the 12-week intervention period, all the participants engaged in a supervised training program on a biweekly basis, under the guidance of a Pilates specialist. Each session lasted 50 min, comprising a 10-min warm-up, 30 min of reformer exercises and a 10-min cool-down on the mat. Exercise progression was achieved through the alteration of spring resistance and the introduction of new positions, tailored to the specific exercise. Each exercise was performed at an intensity corresponding to 50–60% of the estimated maximum heart rate (HRmax), with heart rate continuously monitored using a Polar H10 chest strap (Polar Electro Oy, Finland), and a rate of perceived exertion (RPE) of 13–14 was stipulated (Table 2).

Table 2.
Design of the Reformer Pilates program.
Yoga Exercise Program: Yoga exercise was conducted by a certified Yoga instructor twice per week for 12 weeks using a slightly modified previously published protocol [30]. Each exercise session consisted of three phases for a total of 50 min: 10 min of warming up/stretching in standing poses and a sitting pose, 30 min of Yoga involving poses that were performed continuously with no rest or break, and 10 min of relaxation (cool-down) in sitting poses and a corpse pose (Table 2). The sequence of Yoga exercises included isometric contraction (Shivasana), standing at ease (Tadasana) and balancing the weight on both feet (Centering), transitioning from the standing position (Tadasana), bending to the right and left (Ardhakati Cakrasana), and finally, forward bending (Padahastasana). It also involved forward bending (pada Hastasana), backward bending (ardha chakrasana), and supine posture for rest (shavasana). Although the participants sometimes practiced breathing exercises (Bhramari Pranayama) or meditation (Om meditation), they mainly practiced cyclic meditation Yoga. Each exercise was performed at an intensity corresponding to 50–60% of the estimated maximum heart rate (HRmax), with heart rate continuously monitored using a Polar H10 chest strap (Polar Electro Oy, Kempele, Finland), and a rate of perceived exertion (RPE) of 13–14 was stipulated (Table 3)

Table 3.
Design of the Yoga program.
Nutritional Protocol: Participants received nutritional advice from a registered dietitian on a weekly basis throughout the program. High-protein (at least 1.2 g/kg/day) nutritional guidance was given face-to-face at each meeting to also promote weight loss in those who practiced sport. Participants’ diets were planned as recommended in national and international guidelines for the prevention and treatment of obesity [31,32]. Dietary recommendations are based on exchange lists that encourage a reduction of 0.5 kg per week (2 kg per month) by reducing 500 kilocalories (kcal) from total dietary calories [33]. Personalized dietary advice was given based on participants’ current eating habits rather than traditional “one size fits all” dietary guidance.
2.5. Sample Size Calculation
The sample size analysis of the project focused on a specific research issue (differences between the exercise groups) not addressed in the present article. However, an ANOVA based on a priori analysis indicated that in order to determine differences in expected muscle mass changes of −0.25 ± 1.0 kg (WB-EMS), −0.5 ± 1.0 kg (Pilates), −0.75 ± 1.0 (Yoga), and 1.0 ± 1.0 kg (CG) with α = 0.005 and β − 1 = 0.80 at least 36 participants/group were required.
2.6. Statistical Analysis
All the participants completed all baseline and 12-week follow-up tests; thus, we applied a per protocol analysis with the full data set. Based on a statistically (Shapiro–Wilkes Test) and graphically (QQ and box plots) checked normal distribution of the primary and secondary outcome presented here, dependent (“paired”) t-tests were applied to analyze within-group changes between baseline and post-intervention (12 weeks) assessments. ANCOVA adjusted for baseline differences was applied to determine longitudinal differences between the groups. In case of relevant (ANCOVA) differences (p < 0.10), t-test based (post hoc) comparisons of two groups were conducted using emmeans Version 2.0.1. based on the approach of Searle et al. [34]. The Holm–Bonferroni method [35] was used to adjust for multiple testing. All tests were two-tailed; significance was accepted at p < 0.05.
3. Results
All the participants finished the 12 weeks of intervention and received the treatment or control condition as allocated. Due to the retrospective inclusion of only largely compliant overweight and obese female participants 19–65 years in the present analysis, we are unable to report parameters related to adherence, compliance, loss to follow-up, or withdrawal for this cohort. In parallel, relevant adverse effects with impact on withdrawal or reduced compliance cannot be recorded by our study design. Nevertheless, participants included in the present analysis did not report relevant adverse effects except occasional muscular soreness.
3.1. Baseline Group Differences
Table 4 shows the baseline characteristics of the study groups. In summary, we observed significant differences for body height with the lowest height in the CG (p < 0.001 versus WB-EMS and Yoga). In parallel, considerable group differences were determined for BMI, body fat rate, lean body mass, and particular muscle mass with the least favorable results among the CG (Table 4). While a detailed pairwise analysis revealed no significant differences among the exercise groups, we observed a significant difference between the Yoga and the control group (p = 0.046). In summary, these findings confirmed the validity of our approach to adjusting for baseline group differences within the ANCOVA.

Table 4.
Baseline characteristics of the four study groups. p-values refer to differences between the study groups.
3.2. Weight Loss
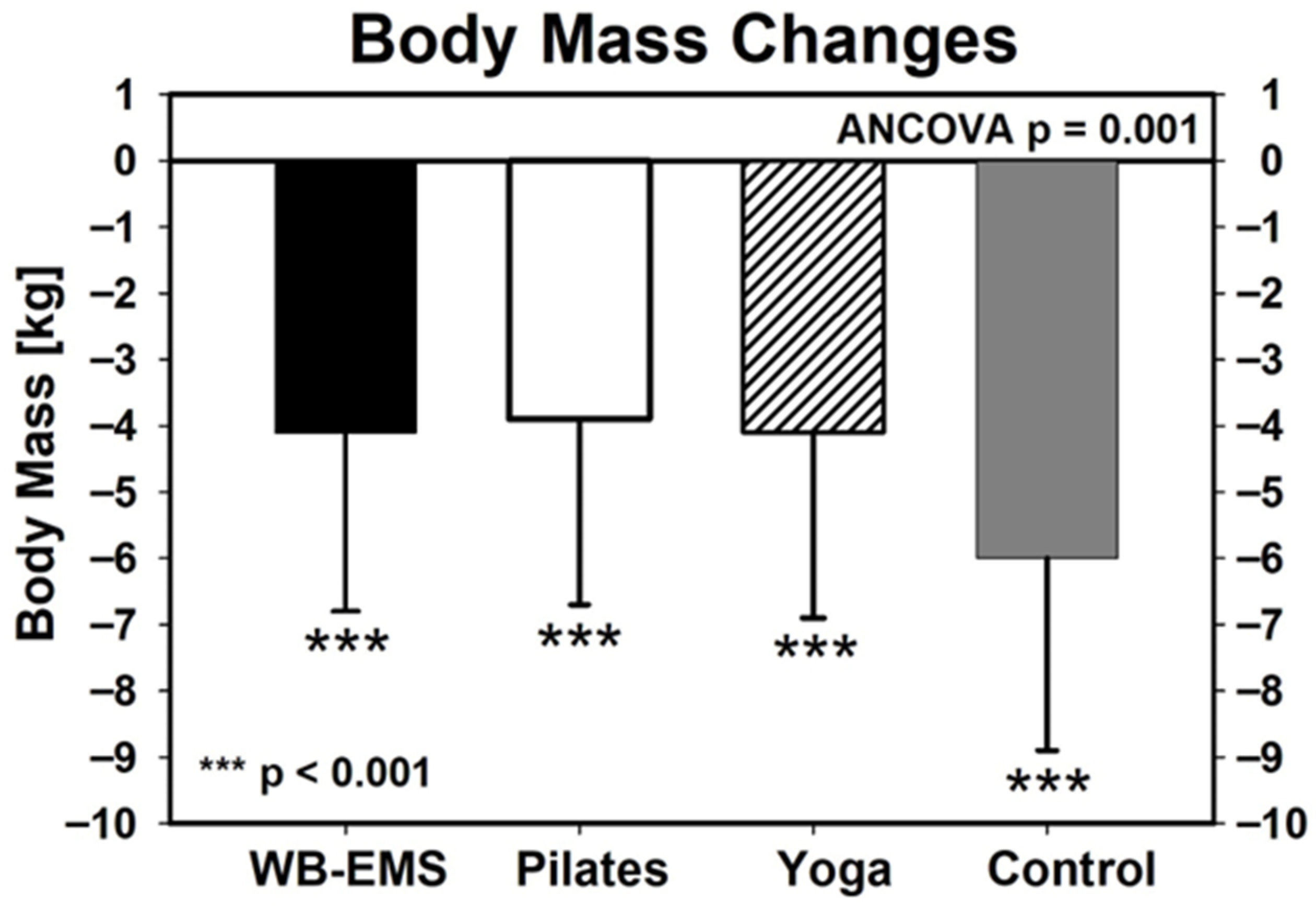
Table 1 shows the baseline body mass data for the exercise and control groups. In summary, all the groups significantly lost body mass (Figure 2). However, ANCOVA (adjusted for baseline group differences) revealed significant differences (p ≤ 0.001) between the groups (Figure 2). Post hoc tests indicated that body mass loss in the control group (CG; −6.0 ± 2.9 kg) was significantly more pronounced (p = 0.003–0.004) compared with the WB-EMS (−4.1 ± 2.7 kg), Pilates (−3.9 ± 2.8 kg), and Yoga (−4.1 ± 2.8 kg) exercise groups, while no differences were observed between the exercise groups. Even after adjustment for multiple testing (Holm–Bonferroni method), the significant differences remained. Accordingly, the secondary hypothesis that body mass loss would be similar across all groups was not supported.
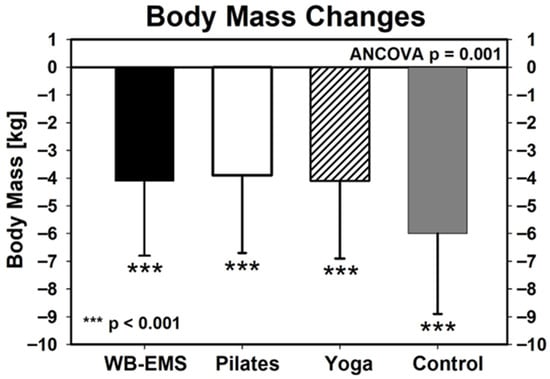
Figure 2.
Changes in body mass in the exercise and control group during the 12-week intervention WB-EMS: n = 62, Pilates: n = 58, Yoga: n = 62, CG: n = 39. Asterisks under the standard deviations indicate significant changes within the group. ANCOVA refer to group differences.
3.3. Muscle Mass Changes
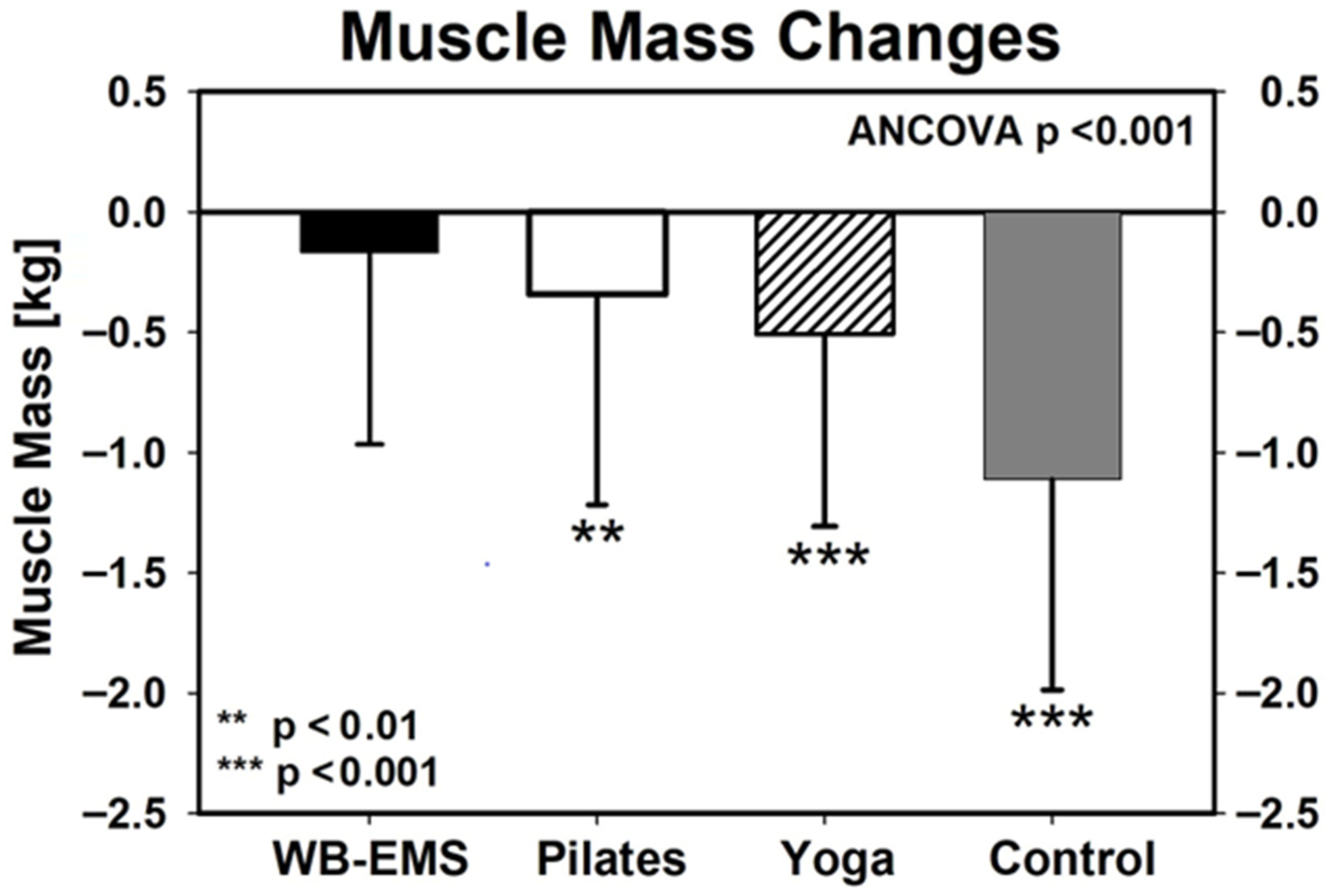
Baseline values for muscle mass were listed in Table 1. Apart from the WB-EMS group (p = 0.14), muscle mass decreased significantly in all the other groups (p ≤ 0.003, Figure 3). ANCOVA revealed significant differences between the groups (Figure 3). Post hoc testing indicates significant differences between WB-EMS (−166 ± 799 g, p < 0.001 vs. CG), Pilates (−343 ± 874 g, p = 0.001 vs. CG), and Yoga (−507 ± 801 g, p = 0.002 vs. CG) compared with control (−1113 ± 881 g), however with no significant differences (p ≥ 0.093) between the exercise groups (Figure 3). Muscle mass reduction was significantly lower in all exercise groups compared with the non-exercising control group. In parallel to body mass, this result remained even after adjusting for multiplicity, i.e., we confirmed our primary hypothesis, that muscle mass was attenuated in the exercise groups compared with the non-exercising control group.
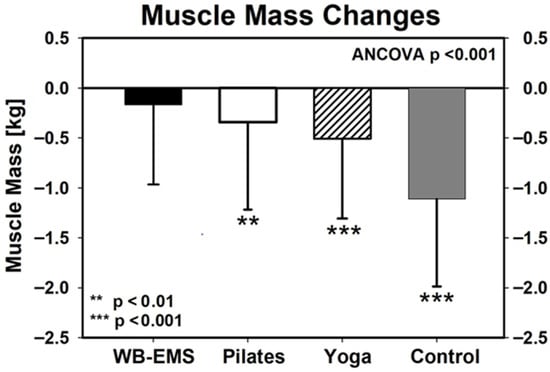
Figure 3.
Changes in muscle mass in the exercise and control group during the 12-week intervention. WB-EMS: n = 62, Pilates: n = 58, Yoga: n = 62, CG: n = 39. Asterisks under the standard deviations indicate significant changes within the group. ANCOVA refer to group differences.
3.4. Body Fat Rate
Baseline values for body fat rate (%) were listed in Table 1. Body fat rate (%) significantly decreased in all groups (p < 0.001) without significant differences between WB-EMS, Pilates, and Yoga as analyzed by ANCOVA (p = 0.447). In detail, the reduction in body fat rate averaged 3.6 ± 2.7% in the WB-EMS (n = 62), 3.1 ± 2.6% in the Pilates (n = 58), 3.4 ± 2.7% in the Yoga (n = 62), and 4.4 ± 2.8% in the non-exercising CG (n = 39).
3.5. Confounding Factors
As stated, only participants who were largely compliant with the exercise and nutritional guidance protocol were included in the study. Changes in physical activity and exercise beside the study exercise program were not recorded. As already mentioned, people with chronic diseases were not included. In addition, newly occurring medical conditions or changes in medication were not reported or observed.
4. Discussion
The present study demonstrates that Yoga, Pilates, and particularly WB-EMS are associated with a reduced loss of muscle mass during diet-induced weight loss in overweight and obese women. In contrast, we do not observe enhanced effects of exercise added to nutritional guidance versus diet alone on body mass or body fat rate. While the significantly higher effect on body mass observed in the non-exercise group can be partially explained by the significantly higher loss of muscle mass, the aspect that the loss of body fat is most pronounced (however: ANCOVA: p = 0.447) in the nutritional guidance-only group is surprising. Considering that the majority of studies provide evidence for favorable effects of Pilates [17], Yoga [36], and particularly WB-EMS [11] on total body fat mass, one would expect higher effects in the exercise compared with the control group. However, reviewing the literature, e.g., no WB-EMS trial that determined changes in body composition during diet-induced energy restriction (≈500–600 kcal/d) in overweight and obese people [7,12,37], reported significant positive effects of WB-EMS vs. control groups on body fat parameters. Although a significant additive effect of exercise and diet on body fat reduction would be welcome, we consider muscle mass prevention during energy-restrictive weight loss to be the more important aspect in the interaction of exercise and nutritional interventions. The rationale for this opinion is the change in resting energy expenditure (REE) after loss of lean body mass that includes not only muscle mass but also other metabolically (very) active organs like liver, heart, pancreas, and kidney [38]. Bosy-Westphal et al. [39] determined a roughly comparable loss of body (10% vs. CG: 8%) and muscle mass (i.e., 923 g vs. CG: 1113 g) after 13 weeks of low-caloric diet in their 22–46 years old overweight and obese female cohort and reported a significant decrease in REE of 7.7% (−0.57 MJ/d) as determined by indirect calorimetry. Of note, REE is by far the most important component of total energy expenditure (TEE) in non-athletic cohorts, explaining up to 70% of TEE [40]. As a consequence, to reach a neutral energy balance, energy intake, energy expenditure or both have to be adjusted to the new equilibrium to prevent a rapid post-diet weight regain (“yoyo effect”) [41]. Another issue not discussed so far is the functional aspect of muscle mass loss. One may argue that due to the more pronounced loss of body weight a decrease in muscle mass would not impair mobility or physical function; however, this view does not necessarily apply to older people at risk for sarcopenia or dynapenia [41].
It has been suggested that higher intakes of protein protect against loss of lean body-/muscle mass, acting either separately or synergistically with exercise [41]. In the present trial we focused on protein intake ≥ 1.2 g/kg body mass/d, i.e., an intake significantly higher compared to most current guidelines of 0.8 g/kg/d (e.g., RDA, DACH) for the healthy adult population. Although we are unable to decide whether the increased protein intake prevents an even more pronounced loss of muscle mass, we conclude that the strategy of increased protein (or ammino acids) alone will not adequately prevent negative side effects of energy restriction on lean body and muscle mass at least in the present cohort.
In summary, the most effective type of exercise, WB-EMS (and dietary recommendation), featured a minor, non-significant loss of muscle mass that accounts for less than 5% of the total weight change in this group. Two studies confirmed the favorable effect of WB-EMS during energy-restrictive weight loss. After 26 weeks of energy-restrictive diet (−600 kcal/d of TEE), Bellia et al. [12] reported comparable weight loss but significant differences in fat-free mass in the diet and WB-EMS (1.1 ± 1.2 kg) vs. the diet only group (−1.5 ± 0.7 kg) in predominately obese women. Willert et al. [7], who compared the effects of 16 weeks of negative energy balance (−650 kcal) realized by either energy restriction and increased activity or by energy restriction alone in overweight and obese premenopausal women, determined more favorable changes in total body fat (−3.3 kg vs. −2.2 kg, p = 0.131) and LBM (387 g vs. −113 g, p = 0.070) in the combined WB-EMS and protein (1.5 g/kg/d) group vs. the isolated protein intervention. The very low LBM loss in the latter control group is, however, in contrast to our finding of high muscle mass loss despite increased protein intake in a similar, (predominately premenopausal) female cohort with comparable physical activity during the intervention and a roughly comparable WB-EMS intervention.
Apart from WB-EMS, both Pilates and Yoga exercise exhibit significantly lower muscle mass loss compared to the control group. This finding indicates that even low intensity exercises (Yoga) performed with a feasible volume of 2 × 50 min/week, potentially practiced at home might be sufficient to halve muscle mass loss during diet. Apart from exercise volume, the narrative review of Rioux et al. [36] suggested (a) longer intervention duration, (b) a yogic dietary component, (c) a residential component, (d) comprehensive inclusion of yogic components, and (e) a home-practice component for therapeutic Yoga programs to be effective in promoting weight loss and/or improvements in body composition.
We would like to point out some specific features and limitations of the study that might aggravate the reliable interpretation of our findings. (1) We focus on overweight to obese women compliant with the exercise and nutritional guidance protocol. Nevertheless, average age (36.2 ± 9.2 years) varied from 19 to 65 years, and BMI averaged between 25.1 and 38.5 kg/m2 in the cohort. We agree that the exclusion of postmenopausal women with their altered energy expenditure and fat distribution pattern [42] might have strengthened our findings. Unfortunately, a reliable analysis of differences in body mass, fat mass/rate, and muscle mass in pre- vs. post-menopausal women failed due to the low proportion of post-menopausal women in the cohort (i.e., n = 4–5 per group). (2) Further, we included only women largely compliant with the nutritional recommendation of 500 kcal energy deficit (to promote a reduction of 0.5 kg per week) and protein intake about 1.2 g/kg body mass/d. Within the exercise protocol, we focus on attendance rates recorded by the instructors and assume reliable reporting by the participants for the non-supervised Yoga session at home. (3) Due to the retrospective analysis, we are unable to report data on withdrawal, adherence and safety of the exercise, and nutritional guidance protocol. However, apart from effectiveness, attractiveness and safety are key aspects of the successful implementation of sustainable exercise protocols. (4) We did not apply standard exercise protocols. This refers particularly to WB-EMS in which usually shorter impulse and rest periods (i.e., 4–6 s impulse and 4 s impulse break [9,43]) with a lower duty cycle or “time under impulse” were applied. (5) We did not use dual-energy X-ray absorptiometry (DXA) as the reference method for muscle mass assessment [44], because our cohort mainly includes premenopausal women of child-bearing age. (6) We focus on muscle mass as the primary outcome. Lean Body Mass (LBM) or Soft Lean Mass is also output by the BIA device; however, we think the focus on muscle mass facilitates the interpretation of the results by the reader. Nevertheless, changes in organ mass, i.e., highly metabolically active tissues, by magnetic resonance imaging, has allowed deeper insights into the protective effect of exercise during energy-restrictive diets. (7) Finally, we applied the Holm–Bonferroni correction [35] to adjust for multiple testing (“multiplicity”), even though this does not relevantly impact our core outcomes in the light of the high significance levels of our results.
5. Conclusions
In summary, whole-body electrical muscle stimulation (WB-EMS), Pilates, and Yoga may be promising options for preventing or reducing muscle mass loss during energy-restrictive diets. WB-EMS in particular can be considered as a time effective and joint-friendly training technology able to prevent muscle mass loss during negative energy balance. Our findings might be transferable to other issues of energy-restrictive weight loss. Considering the application of Glucagon-Like Peptide 1 Receptor agonists (e.g., Semaglutide, Tirzepatid) in the therapy of overweight and obesity with their pronounced weight loss-induced reduction in LBM [45,46] for example, WB-EMS might be a comfortable option to maintain REE in order to prevent immediate weight regain after termination of the pharmacologic therapy.
Author Contributions
Conceptualization, H.D.G., N.A. and C.S.; methodology, H.D.G., N.A. and C.S.; software, M.K. and W.K.; validation, H.D.G., N.A., C.S., M.K. and W.K.; formal analysis, M.K. and W.K.; investigation, H.D.G., N.A., C.S., M.K. and W.K.; resources, H.D.G. and N.A.; data curation, H.D.G., N.A., C.S., M.K. and W.K.; writing—original draft preparation, H.D.G., N.A., C.S., M.K. and W.K.; writing—H.D.G., N.A., C.S. and W.K.; funding acquisition, H.D.G., N.A. and C.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the ethics committee, Karabuk University, Turkey. Approval number 2021/664.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Bluher, M. Obesity: Global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef]
- WHO. Obesity and Overweight. WHO Fact Sheet; WHO: Geneva, Switzerland, 2025. [Google Scholar]
- Eglseer, D.; Traxler, M.; Embacher, S.; Reiter, L.; Schoufour, J.D.; Weijs, P.J.M.; Voortman, T.; Boirie, Y.; Cruz-Jentoft, A.; Bauer, S.; et al. Nutrition and Exercise Interventions to Improve Body Composition for Persons with Overweight or Obesity Near Retirement Age: A Systematic Review and Network Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2023, 14, 516–538. [Google Scholar] [CrossRef]
- Cava, E.; Yeat, N.C.; Mittendorfer, B. Preserving Healthy Muscle during Weight Loss. Adv. Nutr. 2017, 8, 511–519. [Google Scholar] [CrossRef] [PubMed]
- PAC. 2018 Participation Report. The Physical Activity Council’s Annual Study. Tracking Sports, Fitness and Recreation Participation in the USA; Physical Activity Council (PAC): Jupiter, FL, USA, 2018. [Google Scholar]
- Pahmeier, I. Partizipation, Bindung und Dropout im Freizeit-, Breiten-und Gesundheitssport. Anwendungen Der. Sportpsychol. 2008, 2, 425–497. [Google Scholar]
- Willert, S.; Weissenfels, A.; Kohl, M.; von Stengel, S.; Frohlich, M.; Kleinoder, H.; Schone, D.; Teschler, M.; Kemmler, W. Effects of Whole-Body Electromyostimulation on the Energy-Restriction-Induced Reduction of Muscle Mass During Intended Weight Loss. Front. Physiol. 2019, 10, 1012. [Google Scholar] [CrossRef] [PubMed]
- Stephan, H.; Wehmeier, U.F.; Forster, T.; Tomschi, F.; Hilberg, T. Additional Active Movements Are Not Required for Strength Gains in the Untrained during Short-Term Whole-Body Electromyostimulation Training. Healthcare 2023, 11, 741. [Google Scholar] [CrossRef] [PubMed]
- Beier, M.; Schoene, D.; Kohl, M.; von Stengel, S.; Uder, M.; Kemmler, W. Non-athletic cohorts enrolled in longitudinal whole-body electromyostimulation trials—An evidence map. Sensors 2024, 24, 972. [Google Scholar] [CrossRef]
- Le, Y.E.; Schoene, D.; Kohl, M.; von Stengel, S.; Uder, M.; Kemmler, W. Outcomes addressed by longitudinal whole-body electromyostimulation trials in middle aged-older adults—An evidence map. Jpn. J. Med. Sci. 2024, 2, 2024030183. [Google Scholar] [CrossRef]
- Rodrigues-Santana, L.; Hugo, L.; Perez-Gomez, J.; Hernandez-Mocholi, M.A.; Carlos-Vivas, J.; Saldana-Cortes, P.; Contreras-Barraza, N.; Adsuar, J.C. The effects of whole-body muscle stimulation on body composition and strength parameters: A PRISMA systematic review and meta-analysis. Medicine 2023, 102, e32668. [Google Scholar] [CrossRef]
- Bellia, A.; Ruscello, B.; Bolognino, R.; Briotti, G.; Gabrielli, P.R.; Silvestri, A.; Rosazza, C.; Ambruoso, F.; Lombardo, M.; Bernardini, A.; et al. Whole-body Electromyostimulation plus Caloric Restriction in Metabolic Syndrome. Int. J. Sports Med. 2020, 41, 751–758. [Google Scholar] [CrossRef]
- Isacowitz, R. Pilates; Human Kinetics: Champaign, IL, USA, 2022. [Google Scholar]
- Latey, P. The Pilates method: History and philosophy. J. Bodyw. Mov. Ther. 2001, 5, 275–282. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, Z.; Wu, Z.; Ye, X.; Xu, X. Pilates for Overweight or Obesity: A Meta-Analysis. Front. Physiol. 2021, 12, 643455. [Google Scholar] [CrossRef]
- Cruz-Ferreira, A.; Fernandes, J.; Kuo, Y.L.; Bernardo, L.M.; Fernandes, O.; Laranjo, L.; Silva, A. Does pilates-based exercise improve postural alignment in adult women? Women Health 2013, 53, 597–611. [Google Scholar] [CrossRef]
- Silva, L.L.; Mendes da Silva, K.B.; Sousa, L.; Pinhata Rocha, C. Is the Pilates method efficient to cause changes in the body composition of healthy individuals? A systematic review. J. Bodyw. Mov. Ther. 2022, 32, 51–59. [Google Scholar] [CrossRef]
- Savkin, R.; Aslan, U.B. The effect of Pilates exercise on body composition in sedentary overweight and obese women. J. Sports Med. Phys. Fit. 2017, 57, 1464–1470. [Google Scholar] [CrossRef]
- Deshmukh, L.; Janghel, U.; Sonkuwar, A.; Thakur, R. Exploring the Synergy between Yoga and Mindfulness Sport Practices: Enhancing Mental and Physical Well-Being. Indian J. YOGA Exerc. Sport Sci. Phys. Educ. 2024, 9, 26–32. [Google Scholar] [CrossRef]
- Na Nongkhai, M.P.; Yamprasert, R.; Punsawad, C. Effects of Continuous Yoga on Body Composition in Obese Adolescents. Evid. Based Complement. Altern. Med. 2021, 2021, 6702767. [Google Scholar] [CrossRef]
- Grilli Cadieux, E.; Gemme, C.; Dupuis, G. Effects of yoga interventions on psychological health and performance of competitive athletes: A systematic review. J. Sci. Sport Exerc. 2021, 3, 158–166. [Google Scholar] [CrossRef]
- Lohan, A. Elevation of physical and mental strength with sports performance with traditional exercises and Yoga. Acad. Int. Multidiscip. Res. J. 2021, 11, 496–502. [Google Scholar] [CrossRef]
- Cramer, H.; Thoms, M.S.; Anheyer, D.; Lauche, R.; Dobos, G. Yoga in Women With Abdominal Obesity—A Randomized Controlled Trial. Dtsch. Arztebl. Int. 2016, 113, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.A.; Kim, J.W.; Kim, D.Y. Effects of yoga exercise on serum adiponectin and metabolic syndrome factors in obese postmenopausal women. Menopause 2012, 19, 296–301. [Google Scholar] [CrossRef]
- Heymsfield, S.B.; Gonzalez, M.C.; Shen, W.; Redman, L.; Thomas, D. Weight loss composition is one-fourth fat-free mass: A critical review and critique of this widely cited rule. Obes. Rev. 2014, 15, 310–321. [Google Scholar] [CrossRef]
- WHO. World medical association declaration of helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 291–294. [Google Scholar] [CrossRef]
- von Stengel, S.; Frohlich, M.; Ludwig, O.; Eifler, C.; Berger, J.; Kleinoder, H.; Micke, F.; Wegener, B.; Zinner, C.; Mooren, F.C.; et al. Revised contraindications for the use of non-medical WB-electromyostimulation. Evidence-based German consensus recommendations. Front. Sports Act. Living 2024, 6, 1371723. [Google Scholar] [CrossRef]
- Looney, D.P.; Schafer, E.A.; Chapman, C.L.; Pryor, R.R.; Potter, A.W.; Roberts, B.M.; Friedl, K.E. Reliability, biological variability, and accuracy of multi-frequency bioelectrical impedance analysis for measuring body composition components. Front. Nutr. 2024, 11, 1491931. [Google Scholar] [CrossRef]
- Kemmler, W.; Fröhlich, M.; Ludwig, O.; Eifler, C.; von Stengel, S.; Willert, S.; Teschler, M.; Weissenfels, A.; Kleinoder, H.; Micke, F.; et al. Position statement and updated international guideline for safe and effective whole-body electromyostimulation training-the need for common sense in WB-EMS application. Front. Physiol. 2023, 14, 1174103. [Google Scholar] [CrossRef]
- Vaquero-Cristobal, R.; Alacid, F.; Esparza-Ros, F.; Lopez-Plaza, D.; Muyor, J.M.; Lopez-Minarro, P.A. The effects of a reformer Pilates program on body composition and morphological characteristics in active women after a detraining period. Women Health 2016, 56, 784–806. [Google Scholar] [CrossRef]
- Holtzman, B.; Ackerman, K.E. Recommendations and Nutritional Considerations for Female Athletes: Health and Performance. Sports Med. 2021, 51, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Juul-Hindsgaul, N.; Alalwani, Z.; Boylan, A.M.; Hartmann-Boyce, J.; Nunan, D. Defining success in adult obesity management: A systematic review and framework synthesis of clinical practice guidelines. Clin. Obes. 2024, 14, e12631. [Google Scholar] [CrossRef]
- Wheeler, M.L.; Franz, M.; Barrier, P.; Holler, H.; Cronmiller, N.; Delahanty, L.M. Macronutrient and energy database for the 1995 Exchange Lists for Meal Planning: A rationale for clinical practice decisions. J. Am. Diet. Assoc. 1996, 96, 1167–1171. [Google Scholar] [CrossRef] [PubMed]
- Searle, S.R.; Speed, F.M.; Milliken, G.A. Population marginal means in the linear model: An alternative to least squares means. Am. Stat. 1980, 34, 216–221. [Google Scholar] [CrossRef]
- Holm, S. A simple sequentially rejective multiple test procedure. Scand. J. Stat. 1979, 6, 65–70. [Google Scholar]
- Rioux, J.G.; Ritenbaugh, C. Narrative review of yoga intervention clinical trials including weight-related outcomes. Altern. Ther. Health Med. 2013, 19, 32–46. [Google Scholar]
- Reljic, D.; Dieterich, W.; Herrmann, H.J.; Neurath, M.F.; Zopf, Y. “HIIT the Inflammation”: Comparative Effects of Low-Volume Interval Training and Resistance Exercises on Inflammatory Indices in Obese Metabolic Syndrome Patients Undergoing Caloric Restriction. Nutrients 2022, 14, 1996. [Google Scholar] [CrossRef]
- Gallagher, D.; Kelley, D.E.; Thornton, J.; Boxt, L.; Pi-Sunyer, X.; Lipkin, E.; Nyenwe, E.; Janumala, I.; Heshka, S. MRI Ancillary Study Group of the Look AHEAD Research Group. Changes in skeletal muscle and organ size after a weight-loss intervention in overweight and obese type 2 diabetic patients. Am. J. Clin. Nutr. 2017, 105, 78–84. [Google Scholar] [CrossRef]
- Bosy-Westphal, A.; Kossel, E.; Goele, K.; Later, W.; Hitze, B.; Settler, U.; Heller, M.; Gluer, C.C.; Heymsfield, S.B.; Muller, M.J. Contribution of individual organ mass loss to weight loss-associated decline in resting energy expenditure. Am. J. Clin. Nutr. 2009, 90, 993–1001. [Google Scholar] [CrossRef]
- Lam, Y.Y.; Ravussin, E. Analysis of energy metabolism in humans: A review of methodologies. Mol. Metab. 2016, 5, 1057–1071. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, D.; Berg, A. Weight Loss Strategies and the Risk of Skeletal Muscle Mass Loss. Nutrients 2021, 13, 2473. [Google Scholar] [CrossRef]
- Pöhlman, E.; Tchernof, A. Transversing the menopause: Changes in energy expenditure and body composition. Coron. Artery Dis. 1998, 9, 799–803. [Google Scholar] [CrossRef]
- Kemmler, W.; Fröhlich, M.; Eifler, C. Whole-Body Electromyostimulation. Effects, Limitations, Perspectives of an Innovative Training Method; Springer: Cham, Switzerland, 2024. [Google Scholar]
- Buckinx, F.; Landi, F.; Cesari, M.; Fielding, R.A.; Visser, M.; Engelke, K.; Maggi, S.; Dennison, E.; Al-Daghri, N.M.; Allepaerts, S.; et al. Pitfalls in the measurement of muscle mass: A need for a reference standard. J. Cachexia Sarcopenia Muscle 2018, 9, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Jastreboff, A.M.; Aronne, L.J.; Stefanski, A. Tirzepatide Once Weekly for the Treatment of Obesity. Reply. N. Engl. J. Med. 2022, 387, 1434–1435. [Google Scholar] [CrossRef] [PubMed]
- Wilding, J.P.H.; Batterham, R.L.; Calanna, S.; Davies, M.; Van Gaal, L.F.; Lingvay, I.; McGowan, B.M.; Rosenstock, J.; Tran, M.T.D.; Wadden, T.A.; et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N. Engl. J. Med. 2021, 384, 989–1002. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.