Abstract
In this study, hydroxyapatite (HAp) was obtained from fishbones of four species: gilt-head bream (Sparus aurata), salmon (Salmo salar), hake (Merluccius merluccius), and megrim (Lepidorhombus boscii). Batch adsorption experiments were performed with Cr3+, Ni2+, and Zn2+ ions under different pH conditions (natural, 3, and 11) and contact times (6 and 72 h), which is innovative in this study and allows a unified comparison across species and thermal treatment (non-calcined vs. calcined). Results indicated that non-calcinated materials were particularly effective for Ni2+ and Zn2+ removal at natural and acidic pH, whereas calcinated samples were more suitable for Cr3+ adsorption under alkaline conditions. Given the precipitation of its insoluble hydroxide under alkaline conditions, zinc removal was limited to natural and acidic pH. Among the tested precursors, megrim and hake-derived (non-calcined) HAp exhibited the highest performance, achieving up to 99.99% removal efficiency at 6 h of contact time and 20 °C. The analysis of the used adsorbents confirmed metal incorporation into the HAp lattice with minimal crystallographic disruption. These findings demonstrate the potential of fishbone-derived HAp as an efficient and low-cost adsorbent for heavy metal removal from aqueous systems, while simultaneously contributing to the valorization of fishery waste.
1. Introduction
Water contamination by heavy metals is one of the most pressing environmental challenges of the 21st century [1]. This pollutant originates from anthropogenic activities such as industrial discharge, mining, and agriculture, and unlike organic contaminants, they are non-biodegradable and tend to accumulate in ecosystems and living organisms [2]. Their persistence has been directly linked to ecological damage and human health risks, making aquatic environments particularly vulnerable [3]. In response to these environmental and health risks, several treatment strategies have been explored, including chemical, biological, and physicochemical methods [1,4].
Exposure to Cr3+ in contaminated water has been associated with gastrointestinal disturbances and systemic effects such as diarrhea, body-weight changes, reduced activity, tearing, and pupil dilation [5]. Similarly, ingestion of Ni2+ contaminated water may cause symptoms such as dizziness, fatigue, headache, and shortness of breath, in addition to gastrointestinal discomfort [5]. In case of Zn2+, excessive exposure has been linked to acute symptoms such as headache and marked gastrointestinal discomfort (nausea, vomiting, diarrhea, abdominal pain/cramps, and loss of appetite); prolonged high exposure may also be associated with copper-deficiency-related hematological and neurological manifestations (e.g., anemia, leukopenia, myeloneuropathy) [6]. Beyond acute symptoms, chronic heavy metal exposure has been discussed in relation to broader outcomes (e.g., neurological impairment, kidney dysfunction, respiratory issues, reproductive impacts, and carcinogenic risk), reinforcing the need to control metal releases to aquatic systems [7]. Therefore, removing these metals from wastewater is essential to reduce both environmental release and related health risks.
In practice, Ni2+ and Cr3+ are recurrent in industrial wastewater, particularly in electroplating and metal-finishing effluents. Reported concentrations in these effluents span a wide window, from trace amounts to values exceeding 1 g/dm3; examples include 394–526 mg/dm3 for Ni2+ and 44.5–193 mg/dm3 for Cr3+ in highly impacted streams [5].
Conventional techniques such as precipitation, ion exchange, and membrane technologies are widely applied but often face drawbacks such as high costs, sludge generation, and incomplete removal. By contrast, adsorption is considered one of the most promising approaches due to its efficiency, simplicity, and adaptability [4,5].
A wide range of adsorbents have been investigated, including activated carbon, activated alumina, waste-derived materials, and biosorbents. However, these options often present challenges such as high production costs, regeneration requirements, or secondary disposal issues [1,8]. Adsorbents such as hydroxyapatite (HAp, Ca10(PO4)6(OH)2) are particularly attractive due to their ion-exchange capacity, chemical stability, and structural affinity for toxic metals [9,10]. Building on these properties, the preparation of HAp from waste resources such as fishbones offers an environmentally friendly alternative.
Most HAp adsorbents reported to date come from three routes: laboratory-made HAp (typically obtained by precipitation or hydrothermal synthesis), biogenic HAp recovered from calcium-phosphate wastes such as bones and scales, and modified HAp (e.g., doped phases or composites) designed to perform under more demanding water matrices [5]. Across these routes, metal uptake is usually described as a combination of Ca2+ exchange and interactions at phosphate/hydroxyl surface sites, while dissolution–precipitation may also contribute depending on the solution chemistry [11,12]. Reported performance is also sensitive to how HAp is produced: crystallinity and microstructure have been discussed as factors that can affect both adsorption efficiency and metal selectivity [12]. This makes cross-study benchmarking difficult because adsorption outcomes shift with operating conditions, especially pH and competitive multi-ion environments. Consequently, data generated under different experimental windows are rarely comparable [12]. Consistent head-to-head evaluations under aligned conditions have been described as limited, leaving uncertainty about how much variability is truly driven by the precursor itself rather than by mismatched testing conditions [7]. Synthetic HAp offers tighter control over composition and reproducibility, whereas biogenic HAp supports circularity but may introduce precursor-dependent variability; modified HAp can enhance performance, although at the cost of added processing complexity. Moreover, even within fish-derived sources, different fish species have been reported to yield HAp with distinct morphology, porosity, and purity, supporting the relevance of a cross-species assessment under aligned conditions [13].
For fish-derived HAp, this point matters: “biogenic HAp” should not automatically be treated as a single uniform class, since precursor-dependent features may vary and the specific contribution of fish species remains insufficiently resolved when conditions are not standardized [7,12]. Accordingly, a species-to-species comparison under aligned preparation and adsorption windows, including acidic and basic pH, helps test whether fishbone-derived HAp behaves consistently across species or exhibits meaningful differences in Cr3+, Ni2+, and Zn2+ removal.
Within the framework of the circular economy, valorizing waste as raw material for adsorbent production both reduces environmental impact and enhances sustainability [14]. In the fish industry, bones can account for 25–30% of the total mass, and their inadequate disposal contributes significantly to environmental pollution [15]. Fish-processing residues such as skins, scales, and bones are collected by the food and pharmaceutical industries to produce collagen and gelatin. In the industrial sector, fishbones are also used in the production of biomaterials and as biological matrices for catalytic and antibacterial purposes, as well as in the treatment of colored wastewater. However, despite these applications, a large fraction of bones and other solid remains are still discarded or underutilized [15,16].
Fishbones contain approximately 70% of inorganic material, of which about 95% is hydroxyapatite (HAp), making them an abundant and low-cost precursor for adsorbent synthesis [16]. Several studies have demonstrated the efficiency of fishbone-derived HAp in removing heavy metals such as Pb2+, Ni2+, Cu2+, Co2+, and Zn2+, with adsorption capacities varying according to the system and conditions [9,17,18]. Table 1 shows studies that have been conducted using fishbone-derived HAp for heavy metal removal.

Table 1.
Comparison of heavy metal removal using fishbone-derived hydroxyapatite.
To the best of our knowledge, fish-derived hydroxyapatite has not yet been studied for the removal of Cr3+, Ni2+, and Zn2+ ions from aqueous solutions at acidic and basic pH.
The objectives of this paper were (1) to prepare fishbone-derived hydroxyapatite, (2) to evaluate its adsorption performance, and (3) to characterize the material.
2. Materials and Methods
2.1. Preparation and Pretreatment of Fishbones
Four types of fish residues were collected from a local fish shop in Pamplona, Spain: gilt-head bream (Sparus aurata), salmon (Salmo salar), hake tail (Merluccius merluccius), and megrim (Lepidorhombus boscii). These species were chosen because they are among the most frequently consumed and commercially available fish in Northern Spain. The bones were manually separated from the meat scraps to remove most of the tissue. They were immersed in 0.1 M NaOH (Panreac, Barcelona, Spain, 98.0%) solution at 60 °C for 4 h, followed by manual cleaning to eliminate residual flesh. This alkaline washing was selected based on the literature, where NaOH pretreatment has been reported to provide effective cleaning of fishbone raw material prior to adsorption studies [19]. Further treatment was performed by immersing the bones again in 0.1 M NaOH solution at 60 °C for 2 h to ensure complete removal of residual organic matter, as visible remnants persisted after a single cycle during sample handling, after which a final water cleaning step was carried out. Finally, the bones were frozen until further processing.
2.2. Preparation of Hydroxyapatite
For the preparation of HAp, two different procedures were followed (Figure S1):
- The bones were dried at 70 °C for 36 h (batch 1: XF samples) and then calcined at 800 °C for 3 h (batch 2: XFC samples). The oven employed was a Memmert UN30 with natural convection. The fishbones were placed directly into the furnace without pulverization.
- The bones were frozen for 3 h at −80 °C in liquid N2, then lyophilized at 0.1 mbar for 40 h (batch 3: XL) and finally calcined at 800 °C for 3 h (batch 4: XLC). Samples were calcined at 800 °C to eliminate residual organic matter and carbonate species. This temperature was selected because calcium hydroxyapatite is reported to remain thermally stable up to about 800 °C, without evidencing phase transformations [5].where X refers to the fish species used (H: Hake tail, S: Salmon, M: Megrim, G: Gilt-head bream). An electrical furnace (Hobersal MOD 12 PR/300, Hobersal, Barcelona, Spain) and a freezer dryer (Telstar Cryodos-50, Telstar, Barcelona, Spain) were employed for this purpose. Commercial HAp (Sigma-Aldrich, Burlington, MA, USA) was also characterized by comparison.
The nomenclature used to refer to each fishbone sample and treatment received is indicated in Table 2.

Table 2.
Sample coding used in the pretreatment procedures.
The potential availability of fishbone waste volume is enormous. Globally, approximately 970–2700 billion tons of fish have been caught with 450–1000 billion being consumed by humans [20,21]. This work was carried out on a laboratory scale, and estimating the preparation costs for the adsorbents is beyond the scope of this study. However, the reagent costs were relatively low because they were commodity chemicals. On the other hand, it can be assumed that energy and labor costs were necessarily high given the volumes of waste handled in each processed batch. For these reasons, it is risky to provide actual (or even approximate) figures in this manuscript. Future studies should provide a cost evaluation, techno-economic, and lifecycle analysis of waste preparation before practical application.
2.3. Characterization of Fishbones and Hydroxyapatite
FTIR (Fourier Transform Infrared Spectroscopy) was performed using a Shimadzu IRAffinity-1S spectrometer (Shimadzu Corporation, Kyoto, Japan) equipped with a Golden Gate diamond ATR accessory. Thirty-two scans were recorded in the 600–4000 cm−1 range, and spectra were analyzed with OMNIC software (version 6.0).
TGA (thermogravimetry) measurements were carried out on a TA Instruments SDT650 system (TA Instruments, Newcastle, DE, USA). Samples were heated from 35 °C to 1000 °C at the rate of 10 °C min−1 in alumina crucibles under static air atmosphere. The data was processed with TA Instruments TRIOS software (v.5).
The humidity content of the samples was determined using Equation (1), and the mass loss after lyophilization was calculated with Equation (2):
XRD (X-ray diffraction) analyses were performed with a Bruker D8 Advance ECO diffractometer (Bruker Corporation, Karlsruhe, Germany) equipped with a Kristalloflex K760 generator (Bruker Corporation, Cu–Kα1 radiation, λ = 1.54 Å) and a LINXEYE XE-T scintillation detector (Bruker Corporation). The experimental conditions for XRD experiments were as follows: 2θ range from 20° to 60°, 1 s per step, and step size of 0.02°. Measurements were conducted at room temperature, and phase identification was performed using Bruker AXS DIFFRAC.EVA V4.3 software. Crystallinity index (CI) was calculated according to the following:
The area of crystalline peaks corresponds to the reduced area of the diffractogram exclusively assigned to the crystalline part; the total area peak is assigned to the overall area of the diffractogram. Bruker’s EVA software (version 5.1) was used to process the diffractograms and calculate a semi-quantitative crystallinity index, after adjusting the background of the diffractograms [22].
Compositional mappings and punctual elemental chemical analyses of the samples were obtained using a FESEM Zeiss Sigma 300 VP microscope (Carl Zeiss AG, Oberkochen, Germany) equipped with an Oxford energy dispersive X-ray (EDX) analyzer (Oxford Instruments, Concord, MA, USA) at an accelerating voltage of 10 kV.
Elemental analysis was conducted by XRF (X-ray fluorescence) using a Bruker S2 Puma spectrometer (Bruker Corporation) with a Ag anode on a He atmosphere. The samples were analyzed using a 4 μm filter. The detector resolution was 10.8 eV (at 20 kV). Quantification was performed using the Spectra Manager commercial software (SPECTRA.Elements, Measurement Software version 2.3).
Colorimetric measurements were conducted with a Konica Minolta CM-2300d (Konica Minolta, Inc., Tokyo, Japan) portable colorimeter using a D65 xenon lamp (Konica Minolta, Inc., Tokyo, Japan). , , and values were measured, where ranges from 0 to 100 (luminosity), indicates the red–green axis, and the yellow–blue axis. Chroma (), hue angle (), and color difference () were also calculated through the following equations:
2.4. Adsorption Experiments
Adsorption studies were conducted using high-purity analytical-grade reagents: CrCl3·6H2O (Panreac, 95.0%), NiCl2·6H2O (Panreac, 99.0%), ZnCl2 (Panreac, 98.0%), and hydroxyapatite (HAp) (Sigma-Aldrich, 98.0%).
Preliminary experiments were performed to determine optimal adsorption conditions, using commercial HAp as the reference adsorbent. Solutions containing 50 mg/L of Cr3+ were prepared and contacted with HAp under four different conditions: 25 mL of solution with 40 mg HAp, 25 mL of solution with 80 mg HAp, 50 mL of solution with 40 mg HAp, and 50 mL of solution with 80 mg HAp. Adsorption assays were conducted at 20 °C with constant stirring at 160 rpm, and supernatant samples were collected at 6, 17, 24, 48, and 72 h at three pH values (3, 11, and natural).
The same procedure was repeated for Ni2+ and Zn2+ solutions (excepting basic pH for Zn2+ due to precipitation concerns). The concentrations of residual metal ions (supernatant samples) were determined using an atomic absorption spectrometer (PerkinElmer AAnalyst 800, PerkinElmer, Shelton, CT, USA).
The methods used to determine the concentrations of each metal ion (Cr, Ni and Zn) were as follows: Liquid samples of the supernatant in the medium containing the salt solution with Cr, Ni, or Zn in contact with solid hydroxyapatite derived from fishbones were studied by atomic absorption spectroscopy (AAS). Solid samples of fishbones, containing varying amounts of metals, were measured by XRF.
In this work, pH 3 and pH 11 were adjusted using H2SO4 (Panreac, 95–98%) and NaOH, respectively. In contrast, “natural pH” denotes the unadjusted condition: neither acid nor base was added. Under this unadjusted condition, the measured pH values were Cr3+, pH = 3.80; Ni2+, pH = 6.41; and Zn2+, pH = 6.44 (recorded at 20 °C, prior to adsorbent contact). A summary of the experimental conditions used in the adsorption tests is provided in Table 3.

Table 3.
Summary of adsorption experimental conditions.
The main parameters to describe the quality of the calibration curves are listed in Table S1. Six replicates were performed to determine the limits of detection (LOD) and of quantitation (LOQ), as 3 and 10 times the signal-to-noise ratio, respectively.
The removal efficiency of metal ions () was determined by the following:
The adsorption capacity at equilibrium ( mg·g−1) was calculated according to the following:
where and are the initial equilibrium metal concentrations (mg/L) and metal concentration (mg/L) at different times, respectively, is the solution volume (L), and is the mass of the adsorbent (g).
The adsorption isotherm delineates the dynamic equilibrium established between the quantity of solute retained by the solid phase (qe) and its residual concentration in the liquid phase (Ce) at a constant temperature. The Langmuir and Freundlich isotherms are the most frequently employed in adsorption, seen in Equations (9) and (10), respectively.
qe = KF · Ce1/n
Once the optimal conditions were established, the experiments were repeated using XF and XFC samples (batches 1 and 2) instead of HAp. In addition, tests were performed at pH 3 and pH 11 to extend the evaluation of adsorption performance under acidic (pH 3) and alkaline (pH 11) conditions.
In addition, experiments with blanks were carried out. Solutions containing 50 mg/L of Cr3+, Ni2+ and Zn2+ were prepared, adsorption assays were conducted at 20 °C with constant stirring at 160 rpm, and supernatant samples were collected at 6, 17, 24, 48, and 72 h at three pH values (3, 11, and natural) measured by AAS.
3. Results and Discussion
The physicochemical characterization of the materials thus obtained and the derived hydroxyapatite was first carried out prior to adsorption experiments. Different techniques were applied, including FTIR, TGA, XRD, SEM-EDX, XRF, and colorimetry, each providing complementary information on the materials.
3.1. Characterization of Fishbones
3.1.1. FTIR
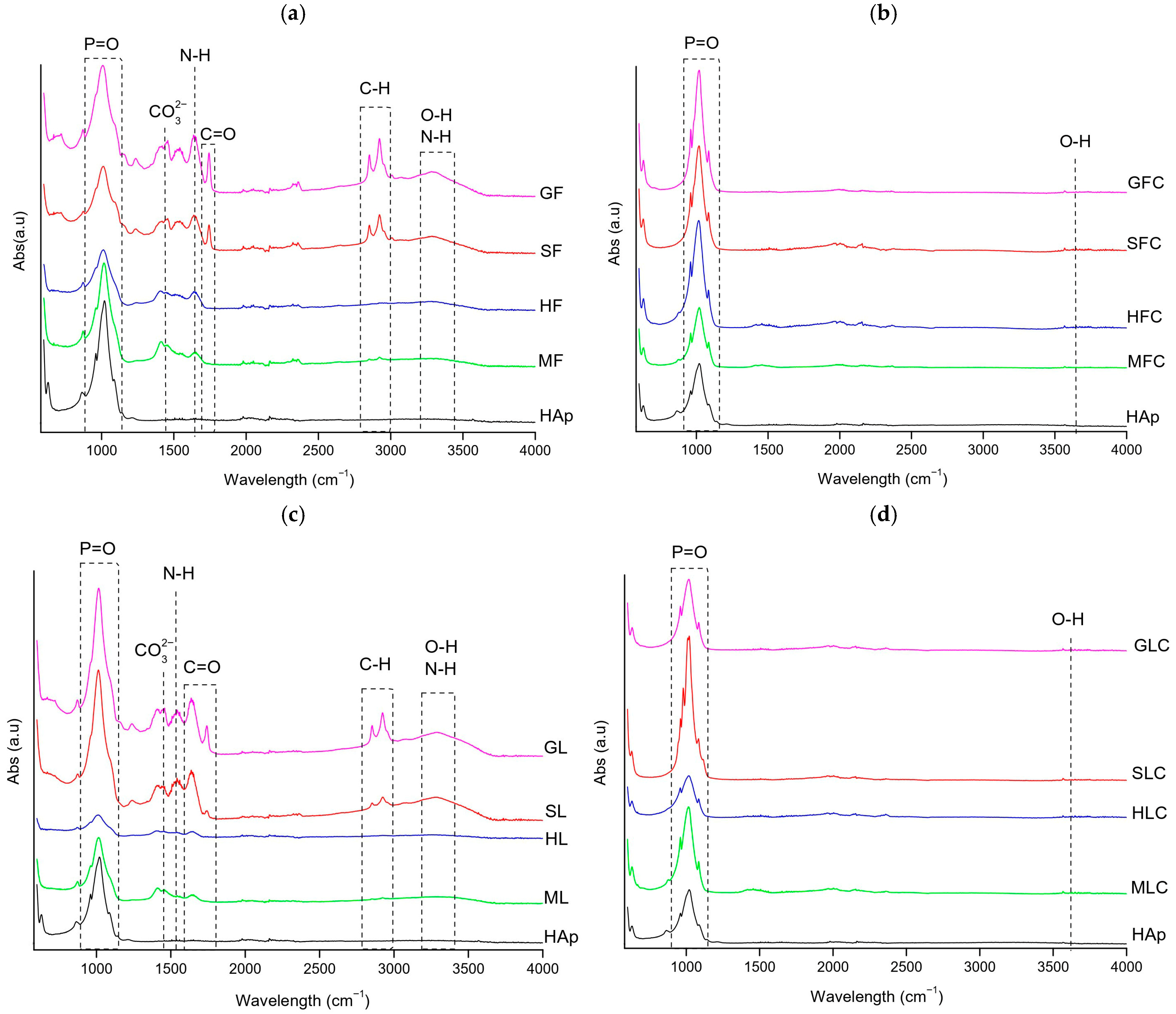
FTIR spectra of the HAp standards and fishbone samples (XF, XFC, XL, XLC) are shown in Figure 1.
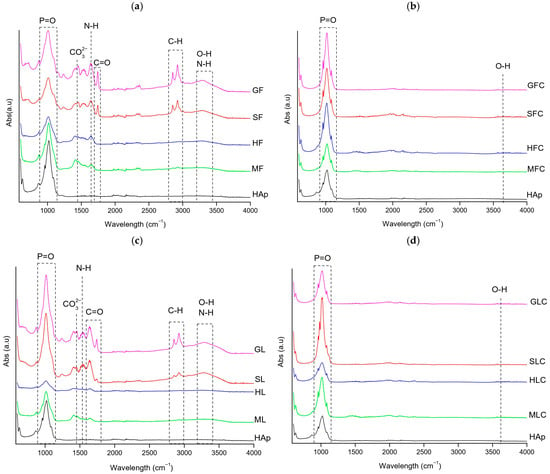
Figure 1.
FT-IR spectra of the clean fishbone samples: (a) XF, (b) XFC, (c) XL, (d) XLC.
In XF samples (Figure 1a), the following main bands were observed: P-O stretching asymmetric vibration (1000–1080 cm−1), C-N stretching of amide II (1200–1300 cm−1), CO32− asymmetric stretching (1400 cm−1), N-H bending of amide II (1500–1600 cm−1), C=O stretching (1650–1690 cm−1), C-H stretching (2800–2900 cm−1), OH stretching (3300 cm−1), and N-H stretching (3300–3400 cm−1) [23,24,25]. Gilt-head bream (GF) and salmon (SF) showed more intense organic-related bands (3300, 2800–2900, and 1750 cm−1) than hake (HF) and megrim (MF), showing higher organic content in the first two samples (Figure 1a).
The phosphate band was broader in XF compared to the HAp standard, suggesting greater amorphicity due to residual organic matter. XL samples contained slightly less organic matter than XF, although the difference was not significant (Figure 1c).
3.1.2. TGA
Thermogravimetric analysis (TGA) was used to evaluate the thermal stability and composition of the fishbone samples before treatment (Figure S2). All of them exhibited an initial weight loss below 200 °C, corresponding to the release of absorbed water; a major mass loss was recorded between 200 °C and 600 °C due to the degradation of organic matter, followed by a smaller decrease between 600 °C and 1000 °C associated with carbonate decomposition. The total mass loss ranged from 21.8% for megrim and 29.3% for hake tail to 39.0% for salmon and 40.0% for gilt-head bream, reflecting the higher fraction of thermally labile components in the latter species (Table 4).

Table 4.
Mass losses (%) at different temperature ranges obtained from TGA.
The same trend in total mass loss was observed across the different treatments. Gilt-head bream and salmon showed the highest mass losses after calcination, with values of 45.6% (GFC) and 43.7% (SFC) for the furnace-calcined samples (XFC) and 43.4% (GLC) and 44.3% (SLC) for the lyophilized calcinated samples (XLC), respectively (Table 4). In contrast, hake tail and megrim exhibited lower mass losses upon calcination, around 31.0% (HFC) and 21.9% (MFC) for XFC and 30.3% (HLC) and 23.6% (MLC) for XLC. A close agreement between XFC and XLC within each species was observed.
3.1.3. XRD
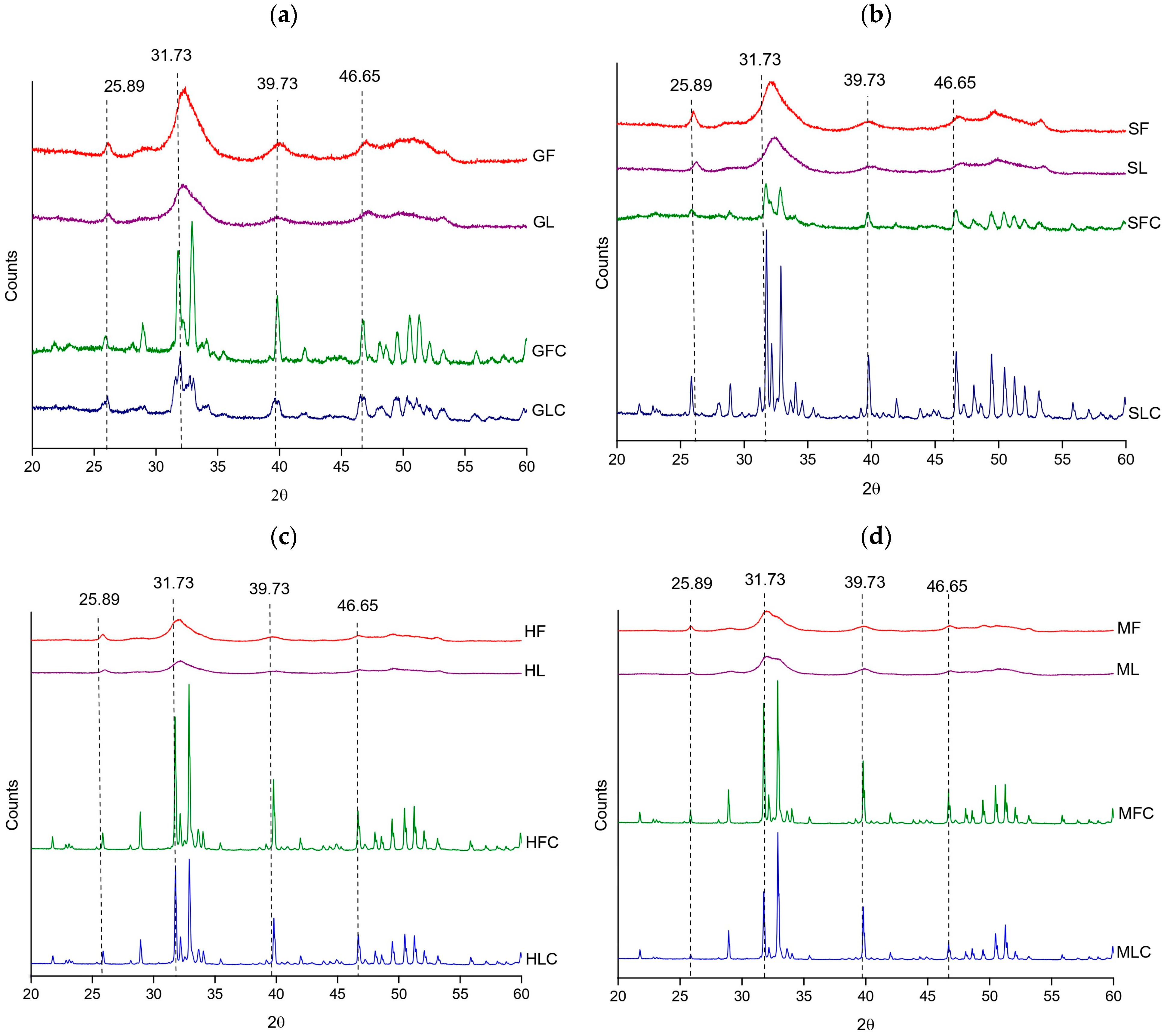
X-ray diffraction (XRD) confirmed hydroxyapatite as the dominant crystalline phase in all samples, with characteristic reflections at 25.89°, 31.73°, 39.73°, and 46.65° (PDF card 54-0022). XF and XL samples showed an average crystallinity of 74%. The broader and less intense peaks observed in these samples reflected the presence of residual organic matter and a partially amorphous character.
Calcination markedly enhanced structural order. Most XFC and XLC samples reached crystallinity values of 90–92%, comparable to that of the HAp reference (91.3%) (Table 5). Two exceptions were observed in gilt-head bream samples, GFC (76.61%) and GLC (84.3%). In all cases, the (300) plane was the most intense reflection, confirming the predominance of the hydroxyapatite phase after thermal treatment.

Table 5.
Crystallinity index (CI) of the fishbone samples *.
As shown in Figure 2, the diffraction patterns of XF and XL were characterized by broader peaks of lower intensity, while XFC and XLC exhibited sharper reflections at the same 2θ positions, differing mainly in intensity and width. This trend is consistent with the FTIR and TGA results, where residual organic matter in XF and XL was associated with greater amorphicity and peak broadening.
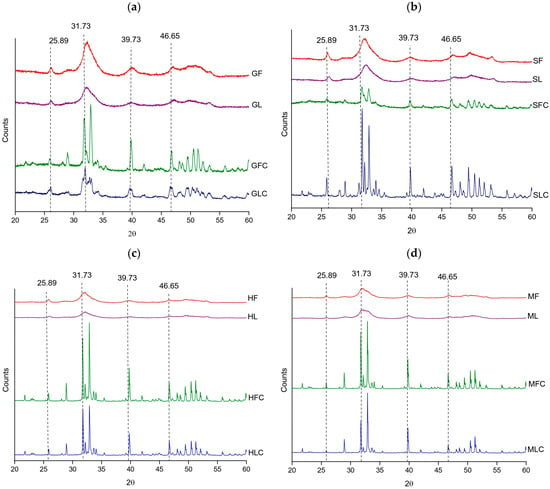
Figure 2.
XRD diffractograms of the treated fishbones: (a) gilt-head bream, (b) salmon, (c) hake, and (d) megrim.
3.1.4. Colorimetry
Colorimetric analysis in the CIE space provided a general overview of the visual appearance of the samples before metal adsorption. As summarized in Table 6 and Table S2, non-calcined groups (XF, XL) exhibited moderate luminosity ( 75–80) and higher chroma values ( 11–14), indicating more saturated yellow tones. Calcined groups (XFC, XLC) were markedly more luminous () and less saturated (), reflecting the removal of organic residues and stabilization of the hydroxyapatite structure.

Table 6.
Average colorimetric parameters (mean ± SD, n = 4 species per group) of fishbone samples before adsorption.
The hue angle () ranged from 88° to 95° in group averages, consistently located in the yellow region of the CIE space. Calcination caused slight shifts toward higher values, corresponding to more orange tones, although the overall appearance remained pale yellow to white.
Figure 3 illustrates the visual transition in megrim samples, chosen as the most representative case, where the difference between non-calcined and calcined materials is clear as an increase in luminosity and loss of color saturation.

Figure 3.
Photographs of samples before and after calcination: (a) G, (b) S, (c) H, (d) M.
3.2. Adsorption
Adsorption tests were carried out under batch conditions using 80 mg of adsorbent in 25 mL of a 50 mg/L metal ion solution at 20 °C and up to 72 h of contact time. Hydroxyapatite samples obtained from fishbones were evaluated both in non-calcined (XF) and calcined (XFC) forms, while commercial HAp served as a benchmark at natural pH.
Although lyophilized samples (XL and XLC) were also synthesized and characterized, their FTIR, XRD, TGA, and colorimetric profiles revealed no significant structural or compositional differences compared with the oven-dried materials (XF and XFC). Since both treatments produced hydroxyapatite phases with comparable structure, crystallinity, thermal behavior, and optical properties, the lyophilization route was considered redundant for adsorption testing. Therefore, subsequent adsorption experiments were limited to the XF and XFC series, as they provide equivalent performance at significantly lower processing cost and operational complexity.
For each pH–metal combination, the results are summarized by reporting the highest-performing case, defined as the sample reaching the best compromise between removal efficiency, adsorption capacity (qe), and contact time, and the non-optimal case, corresponding to the lowest performance observed in the same group. Discussion about the tendencies is also presented for each system. This framework provides a clear overview of both the maximum potential and the variability within each experimental condition. Full adsorption data (qe and removal efficiency) for all fish species, pH values and contact times are provided in Table S3 (HAp natural pH), Table S4 (Cr, natural pH), Table S5 (Ni, natural pH), Table S6 (Zn, natural pH), Table S7 (Cr, pH 3), Table S8 (Ni, pH3), Table S9 (Zn, pH 3), Table S10 (Cr, pH 11) and Table S11 (Ni, pH 11). Results of the blank samples are shown in Table S12 (results of pH) and Figure S3 (concentration of each element in blanks).
Figures S4–S9 show the concentration profiles for the XF and XFC samples from all metals, pH conditions, and fish species. In the following subsections, adsorption performance is discussed by pH window, starting with neutral conditions.
Given the relative similarities in the adsorption performance of the prepared materials, to simplify the results, Table 7 lists the parameters of the isotherm models, as well as the statistical indicators, performed with HAp at natural pH. As observed, the Langmuir model provided the best fit for the adsorption data.

Table 7.
Nonlinear fitting parameters of the isotherm models for adsorption of the heavy metals on HAp adsorbent at natural pH.
3.2.1. Neutral Conditions (Natural pH)
- HAp benchmark
At natural pH (Table 8 and Table S3), commercial HAp showed consistently high adsorption efficiencies across all metals, though with marked differences in kinetics: Zn2+ reached almost complete removal within only 17 h, while Cr3+ needed 48 h and Ni2+ up to 72 h to achieve comparable performance. Despite these variations in time, all metals exhibited final adsorption capacities of around 14 mg·g−1, confirming the suitability of HAp as a benchmark reference for subsequent comparisons.

Table 8.
Adsorption performance of HAp at natural pH (benchmark results) [pH values: Cr3+ 3.80, Ni2+ 6.41, Zn2+ 6.44].
For comparison, adsorption capacities reported in the literature were compiled for hydroxyapatites tested at natural pH. Table 9 summarizes representative qe values for Cr3+, Ni2+ and Zn2+ on commercial, nanocrystalline and modified HAp. These reports show a wide range of adsorption capacities, reflecting differences in surface area, crystallinity and chemical modification across studies.

Table 9.
Adsorption capacities of hydroxyapatites reported in the literature under near-neutral pH conditions.
It should be noted that the literature examples compiled in Table 9 correspond to synthetic nano-HAp and/or chemically modified HAp, which are intentionally engineered to maximize capacity and can therefore yield qe values one order-of-magnitude higher than those typically observed for raw or waste-derived hydroxyapatites. Under the fixed batch setup used throughout (C0 = 50 mg·L−1, V = 25 mL, m = 80 mg), mass balance sets a theoretical upper limit of qe = 15.6 mg·g−1. Thus, qe values in the 13–15 mg·g−1 range (Table 8) indicate that the best-performing samples operate close to the maximum uptake permitted by the experimental design, while removal (%) reflects how rapidly that uptake level is approached at a given contact time. Accordingly, the main contribution of this work is not the pursuit of record capacities but a systematic comparison of four reused fishbone sources (XF/XFC) across pH windows and contact times, highlighting fish-dependent trends and practical kinetic differences associated with calcination.
In the blank samples at natural pH conditions (Table S12), the values of the solutions without adsorbent increase very slightly over time. For Cr, from 3.69 (at time 0 h) to 3.86 (at time 72 h); from 5.60 to 6.64 after two days of treatment for Ni; and from 5.59 to 6.2 after 72 h of treatment for the Zn solution. The influence of pH on the availability of each solubilized metal cation (initial concentration, 50 mg/L) in the aqueous medium is shown in Figure S3. As can be seen, it was of low relevance at natural pH.
- Chromium
At natural pH, XF (Table 10 and Table S4) samples showed strong variability, ranging from almost negligible adsorption (<10%) to almost complete removal (98%). In contrast, XFC samples displayed faster kinetics, with megrim-derived MFC achieving 99% removal within 6 h, while salmon-derived SFC remained below 40% under the same conditions. These results highlight both the influence of fish species and the kinetic advantage provided by calcination in the selected cases.

Table 10.
Summary of Cr3+ adsorption in XF and XFC samples at natural pH (highest and lowest performances).
Under the experimental conditions used, XF samples reached a plateau (apparent equilibrium) at fish-dependent times. GF stabilized by 24 h (qe = 15.05 mg·g−1; removal = 94.51%), whereas HF leveled off by 17 h (qe = 15.55 mg·g−1; removal = 97.89%). MF showed the earliest stabilization, reaching the plateau already at 6 h (qe = 15.35 mg·g−1; removal = 96.39%). In contrast, SF did not exhibit a clear plateau within the tested window and continued increasing up to 72 h (qe = 11.44 mg·g−1; removal = 71.86%); after the plateau times identified for the other XF samples, further changes were marginal.
In the calcined group, a plateau (apparent equilibrium) was generally reached at the earliest sampling time. GFC stabilized at 6 h (qe = 15.37 mg·g−1; removal = 96.43%), and the same early plateau behavior was observed for HFC (6 h: qe = 15.76 mg·g−1; removal = 99.00%) and MFC (6 h: qe = 15.88 mg·g−1; removal = 99.49%). SFC was the only delayed case at early time, but it stabilized by 17 h (qe = 15.05 mg·g−1; removal = 95.21%); beyond these plateau times, the response remained essentially unchanged.
- Nickel
At natural pH (Table 11 and Table S5), Ni2+ removal at early times (6 h) were strongly fish-dependent, ranging in XF from 19.86% (GF; qe = 3.22 mg·g−1) to 89.50% (MF; qe = 14.51 mg·g−1) and in XFC from 54.23% (SFC; qe = 8.75 mg·g−1) to 89.08% (MFC; qe = 14.39 mg·g−1). With increasing contact time, all systems tended to evolve toward a plateau (apparent equilibrium), although the time required to approach stable performance remained sample-dependent.

Table 11.
Summary of Ni2+ adsorption in XF and XFC samples at natural pH (highest and lowest performances).
Within XF, HF and MF behaved as fast-uptake materials, approaching a plateau by 48 h (HF: qe = 15.25 mg·g−1; removal = 94.56%; MF: qe = 15.12 mg·g−1; removal = 93.13%) and remaining stable thereafter. SF showed slower kinetics, increasing from 32.48% (qe = 5.32 mg·g−1) at 6 h to 64.75% (qe = 10.46 mg·g−1) at 48 h). GF was the slowest XF sample, rising from 19.86% (qe = 3.22 mg·g−1) at 6 h to 49.22% (qe = 7.94 mg·g−1) at 72 h, without a clear plateau within the tested window.
In XFC, the plateau was generally reached earlier and at higher removals. MFC stabilized by 17 h (qe = 15.59 mg·g−1; removal = 96.90%) and remained essentially unchanged thereafter, while HFC reached a comparable plateau by 24 h (qe = 15.83 mg·g−1; removal = 97.75%). GFC increased more gradually, approaching stable performance between 48 and 72 h (48 h: 94.91%; qe = 15.24 mg·g−1; 72 h: 96.47%; qe = 15.70 mg·g−1). SFC showed the most delayed early-time response but reached a clear plateau by 48 h (qe = 15.44 mg·g−1; removal = 95.84%) and remained stable at 72 h.
- Zinc
For Zn2+ at natural pH (Table 12 and Table S6), both the highest and lowest performances were observed at 6 h of contact. Megrim-derived samples achieved the best results in both groups, with MF reaching 99.99% removal ( 15.0 mg·g−1) and MFC achieving 95.9% ( 14.3 mg·g−1). In contrast, gilt-head bream exhibited the weakest performance under the same conditions, with efficiencies close to 50% and qe values around 7 mg·g−1. These findings indicate that the sorbent derived from megrim was consistently more effective than that of gilt-head bream in Zn2+ adsorption at natural pH.

Table 12.
Summary of Zn2+ adsorption in XF and XFC samples at natural pH (highest and lowest performances).
Within XF, HF and MF showed fast uptake, achieving high Zn2+ removal already at 6 h (HF: qe = 14.41 mg·g−1; removal = 97.10%; MF: qe = 15.00 mg·g−1; removal = 99.99%) and remaining essentially unchanged thereafter. SF displayed intermediate kinetics, increasing from 58.24% (qe = 8.65 mg·g−1) at 6 h to 94.24% (qe = 14.02 mg·g−1) at 48 h, after which changes were marginal. By contrast, GF approached stable performance later, rising from 50.39% (qe = 7.56 mg·g−1) at 6 h to 87.79% (qe = 13.13 mg·g−1) at 48 h and remaining near-constant thereafter.
In XFC, MFC exhibited rapid uptake, increasing from 95.94% (qe = 14.31 mg·g−1) at 6 h to 99.31% (qe = 14.72 mg·g−1) at 17 h and remaining essentially unchanged thereafter. SFC also increased sharply, reaching 93.19% at 17 h (qe = 13.99 mg·g−1) and approaching a plateau by 48 h (97.32%; qe = 14.43 mg·g−1). In contrast, GFC and HFC approached stable removal more gradually: GFC rose from 53.33% (qe = 7.95 mg·g−1) at 6 h to 93.70% (qe = 13.96 mg·g−1) at 48 h, while HFC increased from 76.01% (qe = 11.37 mg·g−1) at 6 h to 94.16% (qe = 14.01 mg·g−1) at 24 h and reached near-complete removal at the latest sampling point (99.90%; qe = 14.92 mg·g−1).
To put the performances of the fish-derived adsorbents into perspective, adsorption capacities reported for hydroxyapatites obtained from biological wastes were compiled from the literature (Table 13). These studies mainly describe HAp synthesized from distinct types of bones and other biogenic precursors evaluated under conditions comparable to the near-natural pH used in this work and focus on Ni2+ and Zn2+ removal. In contrast, no indexed studies reporting Cr3+ adsorption by biogenic hydroxyapatite under similar conditions were found.

Table 13.
Adsorption capacities of biowaste-derived hydroxyapatites under near-neutral pH conditions (literature comparison).
3.2.2. Acidic Conditions (pH 3)
In the blank samples at acidic conditions, pH evolution was registered (Table S12). The pH values of the solutions without adsorbent decreased to some extent over time. For Cr, from 3.03 (at time 0 h) to 2.66 (at time 72 h); from 3.30 to 2.55 (a decrease of 23%) after 3 days of treatment for Ni; and from 3.14 to 2.17 after 48 h of treatment for the Zn solution. Under acidic conditions (initial pH ≈ 3), the evolution of pH with time differed among the studied metal ions. Although Cr3+ is the strongest Lewis acid, its pH values showed the smallest variation, indicating equilibrium immediately after pH adjustment. In contrast, the weaker Lewis acidity of Ni2+ and Zn2+ results in slower hydrolysis kinetics, allowing continued proton release over time and leading to more pronounced pH decreases.
The influence of pH on the availability of each solubilized metal cation (initial concentration, 50 mg/L) in the aqueous medium is shown in Figure S3. As can be seen, it was of low relevance at acidic pH.
- Chromium
With Cr3+ under acidic conditions (Table 14 and Table S7), highly promising results were obtained for both groups. Once again, megrim-derived XF reached nearly complete Cr3+ removal within the first 6 h (98.5%), whereas gilt-head bream showed the lowest performance among XF species (69.6%), although still relatively high. In contrast, all XFC samples performed exceptionally well, with negligible differences between the highest and lowest cases: SFC-Cr-6-3 achieved 99.99% removal, while HFC-Cr-6-3 reached 98.6% under the same conditions; XF samples can be regarded as almost as effective as their calcined counterparts.

Table 14.
Summary of Cr3+ adsorption in XF and XFC samples at acidic pH (highest and lowest performances).
Within XF, stabilization times were fish-dependent. HF approached steady performance by 17 h (qe = 14.12 mg·g−1; removal = 98.93%), while MF was already near-steady at 6 h (qe = 13.92 mg·g−1; removal = 98.52%); in both cases, subsequent changes were minor. SF reached a stable regime by 48 h (qe = 13.85 mg·g−1; removal = 97.91%) and remained essentially unchanged thereafter. In contrast, GF showed a slower, still time-dependent uptake up to 48 h (qe = 12.11 mg·g−1; removal = 85.88%), consistent with delayed stabilization compared to the other XF samples.
In XFC, performance was effectively stabilized from the earliest sampling time across all fish species (6 h: qe = 14.01–14.34 mg·g−1; removal = 98.64–99.99%), remaining essentially unchanged thereafter.
- Nickel
For Ni2+ under acidic conditions (Table 15 and Table S8), both XF and XFC showed a notable difference between the most and least efficient cases. Once again, the XF megrim-derived sample was the optimal species, requiring 48 h to reach 84.3% removal, while gilt-head bream showed the lowest efficiency with only 15.1% after 6 h. In the XFC group, the same hake accounted for both the highest and lowest performances, starting at 28.26% removal after 6 h and rising to 97.9% after 48 h.

Table 15.
Summary of Ni2+ adsorption in XF and XFC samples at acidic pH (highest and lowest performances).
Within XF, early-time performance was strongly fish-dependent: GF and SF remained low-to-moderate at 6 h (GF: qe = 2.44 mg·g−1; removal = 15.11%; SF: qe = 4.16 mg·g−1; removal = 25.60%), whereas HF and MF already showed high uptake at 6 h (HF: qe = 11.16 mg·g−1; removal = 68.84%; MF: qe = 12.68 mg·g−1; removal = 78.19%). In terms of stabilization, MF and HF approached a near-steady regime by 48 h (MF: qe = 13.74 mg·g−1; removal = 84.34%; HF: qe = 13.89 mg·g−1; removal = 84.51%), with subsequent changes being relatively small. By contrast, GF and SF showed a more delayed response, with uptake still increasing toward later times (e.g., at 72 h: GF qe = 9.24 mg·g−1, 56.96%; SF qe = 10.92 mg·g−1, 66.09%).
In XFC, variability was also pronounced at early time, but two distinct behaviors emerged (Table S7). GFC and SFC increased gradually up to 48 h (GFC: qe = 10.04 mg·g−1; removal = 61.33%; SFC: qe = 9.53 mg·g−1; removal = 59.37%), consistent with slower kinetics. In contrast, HFC and MFC showed a marked rise between 24 and 48 h, reaching near-complete removal by 48 h (HFC: qe = 16.17 mg·g−1; removal = 97.86%; MFC: qe = 15.69 mg·g−1; removal = 96.52%), after which performance remained essentially stable.
- Zinc
At acidic pH (Table 16 and Table S9), Zn2+ removal was highly efficient across both XF and XFC groups, with all cases achieving substantial adsorption after only 6 h. Among the XF samples, megrim showed the highest performance (98.5%), while gilt-head bream recorded the lowest efficiency at 69.6%. In the XFC group, removal efficiency was uniformly high (>98%). These results confirm that under acidic conditions Zn2+ is rapidly adsorbed, with only moderate variability between fish species.

Table 16.
Summary of Zn2+ adsorption in XF and XFC samples at acidic pH (highest and lowest performances).
Within XF, stabilization times were fish-dependent. HF and MF behaved as fast-uptake materials, approaching a steady regime by 17 h (HF: qe = 14.12 mg·g−1; removal = 98.93%; MF: qe = 14.02 mg·g−1; removal = 98.94%), with subsequent changes being minor. SF reached stable performance by 48 h (qe = 13.85 mg·g−1; removal = 97.91%) and remained essentially unchanged thereafter. In contrast, GF showed a slower, still time-dependent increase up to 48 h (qe = 12.11 mg·g−1; removal = 85.88%), consistent with delayed stabilization relative to the other XF samples.
In XFC, an apparent steady regime was reached at the earliest sampling time across all fish species (6 h: qe = 14.01–14.34 mg·g−1; removal = 98.64–99.99%), remaining essentially unchanged thereafter.
To our better knowledge, biogenic hydroxyapatite studies reporting adsorption of Cr3+, Ni2+ or Zn2+ under strongly acidic conditions (pH 3) were not found in indexed literature sources.
3.2.3. Alkaline Conditions (pH 11)
At pH 11, the observed metal removal cannot be interpreted as adsorption alone because alkaline conditions may promote both surface uptake and the formation of sparingly soluble hydroxide phases, which can contribute to removal via precipitation and subsequent settling/retention. In our experiments, however, no visible precipitation was observed for Cr3+ and Ni2+ upon NaOH addition (pH ≈ 11), consistent with the fact that both metals can remain at least partly dissolved in strongly alkaline media when hydroxo-complexation and/or chloride-related complexation reduces the activity of the free metal ion and delays (or suppresses) macroscopic hydroxide nucleation. In particular, Cr3+ behavior at high pH is strongly speciation-dependent, and complexed Cr3+ can require more than simple alkalinization for efficient separation, while Ni2+ hydroxide formation is likewise sensitive to solution chemistry and complexing conditions. By contrast, Zn2+ formed an immediate precipitate upon NaOH addition under our conditions, so Zn removal at pH 11 is dominated by rapid hydroxide formation rather than interfacial adsorption; therefore, Zn results at pH 11 are not directly comparable with Cr3+ and Ni2+ adsorption trends and are discussed separately [32,33,34].
Based on this framework, the following subsections discuss Cr3+ and Ni2+ removal under alkaline conditions in terms of time-dependent uptake (qe) and removal efficiency, while zinc is excluded from adsorption comparisons at pH 11 due to precipitation.
In the blank samples, pH evolution was registered (Table S12). The pH values of the solutions without adsorbent decreased to some extent over time. For Cr, from 11.25 (at time 0 h) to 9.42 (at time 72 h); from 10.75 to 9.24 after 3 days of treatment for Ni; and from 10.95 to 8.81 after 72 h of treatment for the Zn solution. Among the studied systems, Zn2+ showed the largest overall decrease in pH, suggesting a higher tendency to react with OH− in solution, likely through the formation of poorly soluble hydroxide species. Ni2+ and Cr3+ also induced a significant pH decrease, although to a lesser extent and with more stabilized values at longer times. The precipitation of Zn under basic conditions in the presence of the hydroxyapatite is consistent with the pH evolution observed for the salt solutions without adsorbent (blanks), which indicates a higher hydroxide consumption for Zn2+.
The influence of pH on the availability of each solubilized metal cation (initial concentration, 50 mg/L) in the aqueous medium is shown in Figure S3. As can be seen, this was significant at basic pH, especially in the case of zinc. It is well known that the formation of hydroxides of these metals is greatly favored under alkaline conditions.
- Chromium
At basic pH (Table 17 and Table S10), XF samples exhibited the lowest performance observed across the entire study. After 6 h, gilt-head bream showed almost no adsorption (1.7%), and even the best XF case, hake after 72 h, reached only 53.1% removal, which remains modest compared with other conditions. In contrast, XFC samples achieved near-complete Cr3+ removal within 6 h (>98%), underscoring that calcination is decisive for effective uptake under alkaline conditions. Across the XF series, Cr3+ removal increased steadily with contact time, and the highest values were reached at 72 h, with HF showing the largest increase (qe = 7.23 mg·g−1; removal = 53.07%), followed by GF and SF (qe = 5.25 mg·g−1; removal = 38.20% and qe = 5.09 mg·g−1; removal = 37.31%, respectively) and MF (qe = 5.09 mg·g−1; removal = 37.65%). By contrast, the calcined materials reached their stable regime at the earliest sampling time: at 6 h, all XFC samples already showed near-quantitative removal (GFC: qe = 13.63 mg·g−1; removal = 99.47%; SFC: qe = 13.39 mg·g−1; removal = 98.99%; HFC: qe = 13.47 mg·g−1; removal = 99.21%; MFC: qe = 13.36 mg·g−1; removal = 98.75%), with only minor fluctuations thereafter.

Table 17.
Summary of Cr3+ adsorption in XF and XFC samples at basic pH (highest and lowest performances).
- Nickel
At basic pH (Table 18 and Table S11), Ni2+ removal was highly effective for both XF and XFC samples, with all cases exceeding 90% efficiency within 6 h. Among XF, removal ranged from 92.11% to 95.39%, while XFC samples performed slightly better overall (97.53–99.06%) under the same contact time, indicating consistently fast Ni2+ uptake under alkaline conditions with limited dependence on fish species or calcination.

Table 18.
Summary of Ni2+ adsorption in XF and XFC samples at basic pH (highest and lowest performances).
Within XF, the system was already near an apparent plateau at 6 h across all species (GF: qe = 12.91 mg·g−1; removal = 95.39%; SF: qe = 12.85 mg·g−1; removal = 94.28%; HF: qe = 12.86 mg·g−1; removal = 93.18%; MF: qe = 12.57 mg·g−1; removal = 92.11%), and later measurements showed only minor fluctuations. In the calcined group, XFC samples similarly stabilized at 6 h (GFC: qe = 13.48 mg·g−1; removal = 98.76%; SFC: qe = 13.49 mg·g−1; removal = 99.06%; HFC: qe = 13.48 mg·g−1; removal = 98.75%; MFC: qe = 13.38 mg·g−1; removal = 97.53%), remaining essentially unchanged thereafter.
- Zinc
Zn2+ adsorption tests under alkaline conditions (pH 11) were not evaluated because precipitation occurred immediately during pH adjustment: upon adding NaOH to the ZnCl2 solution, a solid phase formed before introducing the adsorbent. Under these conditions, the depletion of dissolved zinc is governed by Zn(II) alkaline speciation and hydroxide-driven precipitation rather than adsorbent-controlled uptake, so removal cannot be interpreted as adsorption capacity (qe) and is not directly comparable to Cr and Ni datasets at the same nominal pH. Therefore, Zn2+ is discussed only under acidic and near-neutral conditions in this work, and alkaline cross-metal comparisons explicitly exclude Zn2+.
No biogenic hydroxyapatite studies reporting adsorption of Cr3+, Ni2+ or Zn2+ under strongly basic conditions (pH 11) were found in indexed literature sources.
3.2.4. Adsorption Data Comparison
Non-calcined samples (XF) generally achieved comparable performance to that of calcined ones (XFC), indicating that energy-intensive calcination is not always necessary. However, calcination provided critical chromium removal at alkaline pH and for nickel under acidic conditions, where efficiencies exceeded 95%.
The influence of species was decisive factors: megrim (Lepidorhombus boscii) and hake (Merluccius merluccius) consistently showed the highest adsorption capacities and removal efficiency. An added novel observation was the structural clustering of gilt-head bream (Sparus aurata) and salmon (Salmo salar), and hake with megrim, despite their lack of phylogenetic proximity. This unexpected grouping suggests that mineral composition and processing response may be stronger determinants of characteristic hydroxyapatite structure than biological lineage. The association between species is proven by detailed elemental chemical analysis performed by XRF (Table 19) and structural information by XRD (Figure 2).

Table 19.
XRF analyses of non-calcined (XF) and calcined species (XFC).
The association between gilt-head bream and salmon, and hake and megrim are evident when analyzing the elemental analysis results obtained by XRF (Table 19). This association is much clearer in the uncalcined samples. GF and SF show higher Ca and lower P content compared to HF and MF. The Ca/P ratios were 3.4–3.8 for the first group (GF and SF) and 2.6–2.8 for the second (HF and MF). In the calcined samples, these differences are not clearly observed. Structurally, the diffractogram profiles of gilt-head bream and salmon, and hake and megrim are very similar; however, in the case of the calcined samples, a clear amorphization of the samples is evident (Figure 2).
Metal-specific behavior further underscored these trends. Zinc was removed with the highest efficiency across conditions, nickel showed intermediate behavior, and chromium was the most challenging metal, requiring calcination at high pH to achieve optimal performance. These patterns are summarized in Table 20, which highlights the optimal adsorption conditions for each metal and pH.

Table 20.
Optimal adsorption conditions for each metal and pH.
Our work has focused on establishing a proof of concept for the viability of fishbone-derived hydroxyapatite as an effective adsorbent for treating water contaminated with metals. About the end-of-life management of sorbents already loaded with heavy metals, recent work agrees that poor handling can lead to secondary contamination; therefore, routes that either immobilize the solid used or give it a controlled second life are recommended. Controlled reuse of spent adsorbents in functional applications (e.g., catalytic or other technical uses) has been proposed as a way to reduce the environmental risk associated with direct disposal [35]. Another widely reported route is stabilization/solidification (S/S) in cementitious matrices (including mortar- or brick-type formulations), where incorporating the spent adsorbent can reduce metal leaching, provided that the residue fraction remains compatible with the mechanical performance of the final product [35,36,37]. In addition, geopolymer encapsulation has been reported as an immobilization approach to retain metals within inorganic matrices, again with mechanical performance discussed as part of feasibility [38]. Based on this landscape, and while the most suitable route for our materials is being defined, the post-adsorption solids will be kept in sealed, clearly labeled containers under institutional control until a final treatment/disposal option is selected to minimize any risk of metal release.
A comparison between lyophilized (XL and XLC) and oven-dried samples (XF and XFC) (Table 2) could provide additional insight into the role of hydroxyapatite pore structure in adsorption performance. The objective of the present study was not to compare drying routes but to evaluate the adsorption behavior of biogenic hydroxyapatite derived from different fishbone sources under consistent preparation and testing conditions. The inclusion of additional adsorption experiments on lyophilized samples with a comprehensive experimental and characterization study, including post-adsorption analyses (e.g., AAS, XRF, XRD, FTIR, SEM–EDX, colorimetry, and mercury intrusion porosimetry), to ensure a rigorous and meaningful comparison would be very time-consuming and beyond the scope of the present work. These experiments should follow the same methodology and employ the same experimental techniques as those used with the previous samples (oven-dried samples, XF and XFC). This limitation has now been explicitly acknowledged in the Conclusions section, where a systematic comparison between lyophilized and oven-dried hydroxyapatite is identified as a relevant and valuable direction for future research.
3.3. Characterization of Used Adsorbents
Post-adsorption characterization was conducted to investigate the structural and compositional modifications in fishbone-derived hydroxyapatite after exposure to heavy metals under different pH conditions and contact times. The used solids were examined using FTIR, XRD, XRF and SEM-EDX to assess phase stability, identify any detectable changes in vibrational and diffraction features, and track possible shifts in bulk composition after metal uptake. This analysis aimed to assess whether metal exposure and retention produced detectable changes in the chemical structure and physicochemical properties of the material, rather than to demonstrate a single uptake pathway.
3.3.1. FTIR
Only calcined samples (XFC) were analyzed by FTIR for clarity reasons. For each pH condition, representative spectra at 6 h and 72 h were selected. The samples with the highest adsorption capacities are shown.
- Chromium
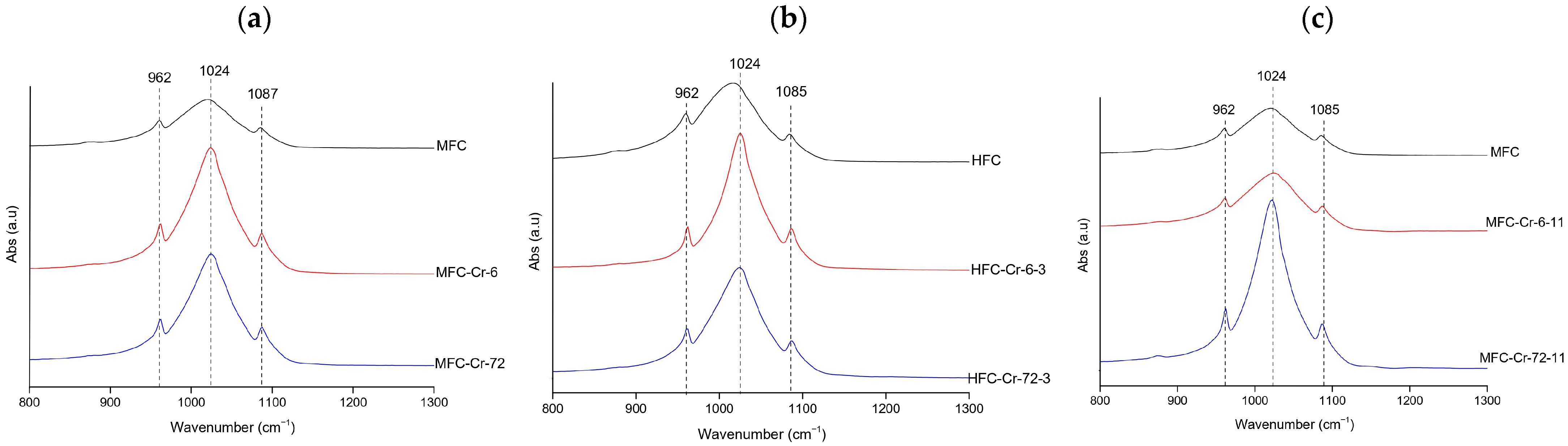
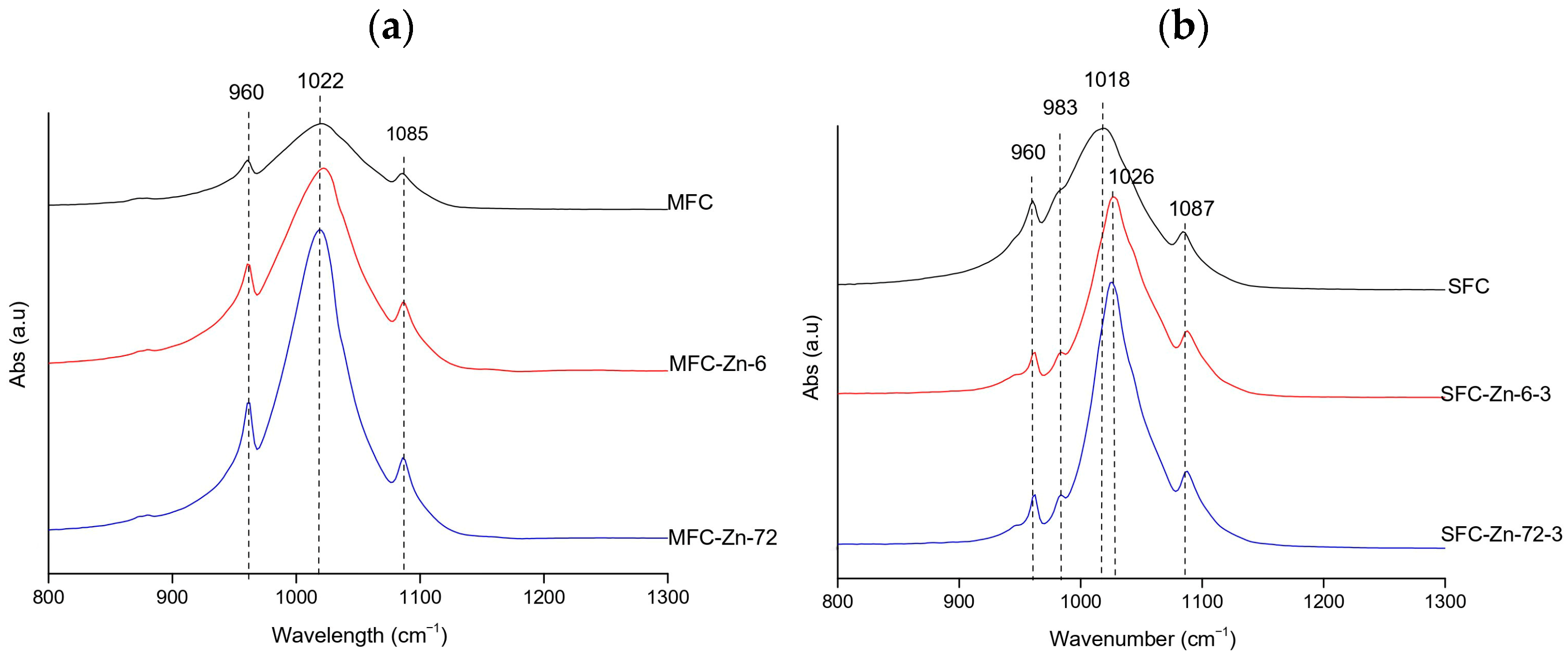
FTIR spectra consistently exhibited the phosphate vibrations bands of hydroxyapatite at 962 ( symmetric stretching vibration), 1022–1024, and 1085–1087 cm−1 ( asymmetric stretching vibration) [39]. At natural pH (megrim, Figure 4a), the Full Width at Half Maximum (FWHM) of the v3(PO43−) band decreased from 85.15 to 58.15 cm−1, suggesting a temporary increase in lattice order. At 72 h, the width increased again to 81.05 cm−1, showing that this effect was not fully preserved. Under acidic conditions (hake, Figure 4b), the broadening and intensity loss were more pronounced, reflecting significant amorphization; FWHM values remained high (81.97–84.06 cm−1 at 0–6 h) and only decreased to 69.04 cm−1 at 72 h, indicating that the structure, although slightly more ordered at longer contact times, remained disordered. In contrast, at alkaline pH (megrim, Figure 4c), the phosphate bands remained sharp and stable, becoming narrower and more intense after 72 h, with the FWHM decreasing from 85.15 cm−1 in the control to 52.29 cm−1 for the Cr-treated sample, evidencing improved crystallinity and a higher degree of structural order [40]. Overall, chromium induced some structural alterations, particularly under acidic conditions. These findings should be corroborated by post-adsorption XRD and SEM-EDX analyses.
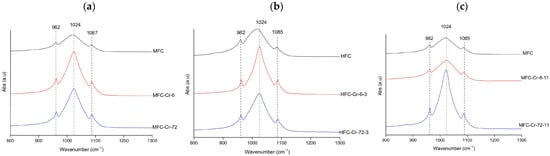
Figure 4.
Representative FTIR spectra of Cr-treated hydroxyapatite (XFC) at (a) natural pH (megrim), (b) pH 3 (hake), and (c) pH 11 (megrim), showing the samples with the highest adsorption capacity at 6 h and 72 h of contact.
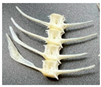
- Nickel
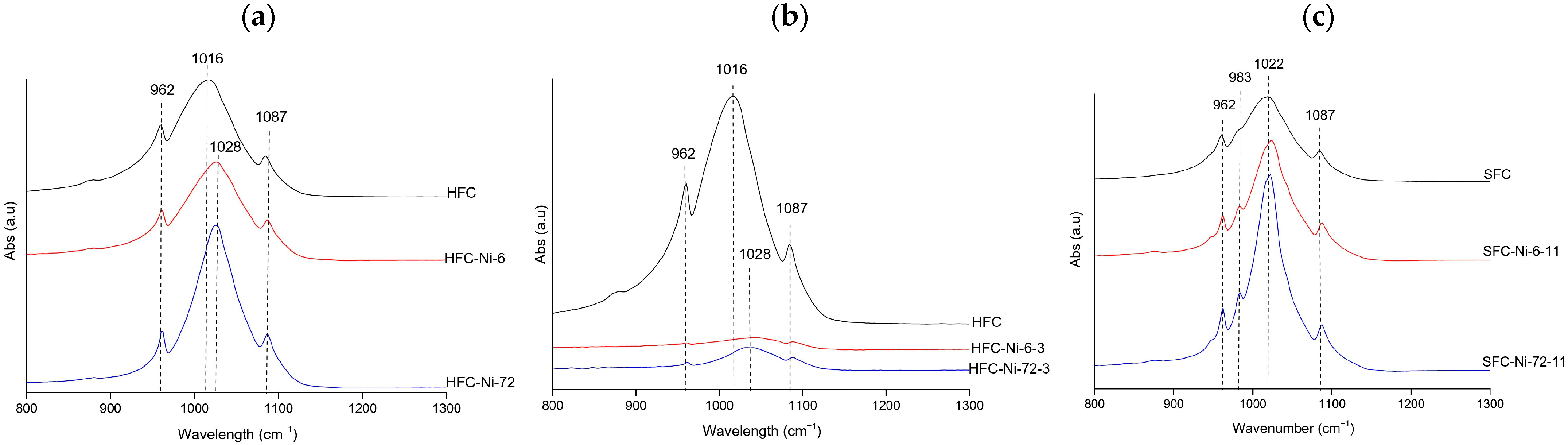
FTIR spectra of Ni samples consistently displayed the phosphate vibrational bands at 962, 983, 1016–1026, and 1085–1087 cm−1. At natural pH (hake, Figure 5a), the spectra showed broader and weaker phosphate bands at 6 h, indicating amorphization; FWHM values were also high (84.51 and 82.96 cm−1 for HFC and HFC–Ni–6, respectively) and increased to 92.09 cm−1 at 72 h, which is consistent with a gradual loss of crystallinity [39,40]. Under acidic conditions (hake, Figure 5b), the phosphate signals were greatly attenuated and poorly defined, with very weak bands, so the material was already disordered at all contact times. In contrast, at alkaline pH (salmon, Figure 5c), the band became sharper and more intense after 72 h, and the FWHM decreased from 79.33 cm−1 in the control to 56.92 cm−1 at 6 h and 44.52 cm−1 at 72 h, which indicates that crystallinity improved under alkaline conditions [39,40]. Additionally, a small band at 983 cm−1 was only observed for the salmon samples; this feature is usually attributed to hydrogen phosphate groups (HPO42−) or carbonate substitution in non-stoichiometric hydroxyapatite [23]. Overall, Ni-treated samples exhibited high stability of the phosphate framework, with acidic environments promoting disorder, while alkaline conditions favored preservation and even improvement of crystallinity.
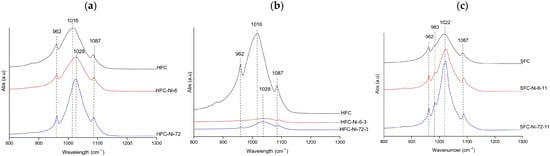
Figure 5.
Representative FTIR spectra of Ni-treated hydroxyapatite (XFC) at (a) natural pH (hake tail), (b) pH 3 (hake tail), and (c) pH 11 (salmon), showing the samples with the highest adsorption capacity at 6 h and 72 h of contact.
- Zinc
FTIR spectra of Zn-treated hydroxyapatite exhibited phosphate vibrational bands at 962, 983, 1020–1026, and 1085–1087 cm−1 [23,39], confirming the persistence of the hydroxyapatite structure across all treatments. At natural pH (gilt-head bream, Figure 6a), the bands were sharp and well-defined at both 6 h and 72 h; the FWHM values decreased from 85.15 cm−1 in the control to 75.44 cm−1 at 6 h and 59.07 cm−1 at 72 h, indicating a gradual improvement in band definition and crystallinity over time.
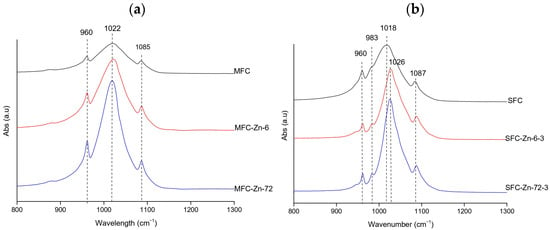
Figure 6.
Representative FTIR spectra of Zn-treated hydroxyapatite (XFC) at (a) natural pH (megrim) and (b) pH 3 (salmon), showing the samples with the highest adsorption capacity at 6 h and 72 h of contact. No spectra were obtained at pH 11 due to precipitation phenomena.
Under acidic conditions (salmon, Figure 6b), the spectra also remained well-defined, and a similar narrowing of the v3(PO43−) band was observed, with FWHM values decreasing from 79.33 cm−1 in the control to 50.50 cm−1 at 6 h and 44.18 cm−1 at 72 h [26,27]. The salmon samples also displayed a small band at 983 cm−1, which is typically attributed to HPO42− or carbonate-related environments in non-stoichiometric hydroxyapatite [23]. No FTIR data was available at pH 11 due to precipitation issues that hindered analysis. Overall, zinc exposure did not substantially disrupt the hydroxyapatite framework; in both pH conditions, the phosphate bands became progressively narrower and more intense, suggesting that Zn2+ favored preservation and, in some cases, slight enhancement of structural order [39,40].
3.3.2. XRD
Compared with the clean samples from fishbone-derived adsorbents (before adsorption assays) (Figure S10) whose crystallinity typically ranged 65–80%, the metal-exposed materials exhibited metal-dependent variability (Table 21). Zinc delivered the highest and most uniform values, from 73% at natural pH (at 6 h) up to 88% at pH 3 (72 h), which could be interpreted as a favorable accommodation of Zn2+ in the Ca/PO4 structural sites or as sorption on the apatite surface without modification in the apatite lattice [41]. Nickel also exhibited high crystallinity, peaking at pH 11 with 93% at 6 h (SD 7%) (see Table 21). By contrast, chromium showed the widest dispersion, with SDs up to 29%, a behavior compatible with the coexistence of multiple oxidation states and ionic-size constraints that could hinder regular lattice incorporation [41].

Table 21.
Crystallinity index (CI) and standard deviation (SD) of hydroxyapatite samples exposed to heavy metals (Cr3+, Ni2+, and Zn2+) at different pH values and two contact times (6 h and 72 h).
Large standard deviations in the crystallinity index (CI) indicate that the post-adsorption solids exhibit substantial variability, which is expected for heterogeneous biogenic apatites and can be amplified by differences in peak broadening, background definition, and signal-to-noise during CI calculation. Accordingly, CI is treated here as a comparative descriptor to support metal- and pH-dependent trends, rather than as an absolute metric where small differences between individual values should be over-interpreted. For this reason, the discussion emphasizes consistent shifts observed across conditions and the preservation (or loss) of the apatite phase evidenced by the XRD patterns, while changes that fall within the reported dispersion are interpreted cautiously.
The role of contact time is condition-dependent, enhancing order in Zn2+ (natural pH) and Cr3+ (acidic pH) but offering no benefit and even a decline- for Ni2+ under strongly alkaline conditions (Table 21).
pH exerted decisive control over the final crystallinity, with metal-specific responses (Table 21). For chromium, acidic media delivered the highest order (79.2%) and a clear time-resolved rinse (72% to 86%, from 6 to 72 h; Table 21), probably because of low pH increasing surface reactivity, while natural pH can induce slight destabilization through slower ion exchange or partial dissolution. Nickel achieved its maximum uptake (85.8%) under alkaline conditions, which is consistent with the improved Ni2+ sorption and precipitation pathways that stabilize a more ordered phase in basic media; at natural and acidic pH the averages remained high but changed little, reflecting limited structural reorganization after early uptake. Zinc maintained consistently high crystallinity across pH, peaking at pH 3 (88% at 72 h); this pattern aligns with the favorable lattice accommodation of Zn2+ or surface deposition [41,42] (Table 21).
More specifically, MFC to MFC-Zn-6 shows a marked decrease in crystallinity (92% to 48%), while GFC to GFC-Ni-6-3 and GFC-Cr-6-11 exhibit pronounced increases (76% to 100% and 76% to 99%, respectively), consistent with the metal–pH responses discussed in the text.
3.3.3. SEM-EDX
A comprehensive comparison of SEM–EDX elemental maps obtained under natural (Figure 7), acidic (Figure 8), and basic conditions (Figure 9) reveals metal-specific differences in immobilization behavior within calcium phosphate systems. While a calcium phosphate matrix consistently formed under all pH conditions, as evidenced by the strong spatial correlation between Ca and P, the extent and robustness of metal incorporation varied among Cr, Zn, and Ni. Mappings analyses were done in the samples with the higher removal capacity: MF-Cr-6, MF-Ni-6, MF-Zn-6 at natural pH (Figure 7); MF-Cr-6-3, MFC-Ni-48-3, MF-Zn-6-3 at acid conditions (Figure 8); and HFC-Cr-6-11, GF-Ni-6-11 at basic conditions (Figure 9).

Figure 7.
SEM-EDX mapping corresponding to samples at natural pH.

Figure 8.
SEM-EDX mapping corresponding to samples at pH 3.
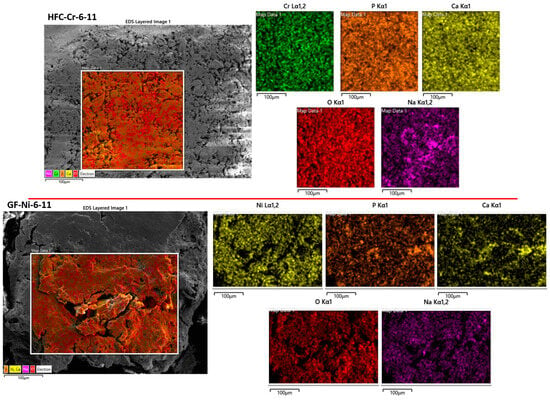
Figure 9.
SEM-EDX mapping corresponding to samples at pH 11.
Chromium exhibited the most stable and pH-independent behavior. Under natural (Figure 7), acidic (Figure 8), and basic (Figure 9) conditions, Cr showed a homogeneous spatial distribution associated with the Ca–P matrix, with no evidence of Cr-rich secondary phases or localized segregation or precipitation. This consistent behavior seems to indicate that chromium immobilization is governed primarily by Cr–phosphate interactions. As a result, Cr stabilization remains highly effective across the entire pH range investigated. Although results from FTIR and XRD could indicate different arrangements depending on pH conditions, the homogeneity shown by SEM-EDX proves a homogeneous distribution of the metal, possibly by adsorption-driven removal.
Zinc displayed intermediate but still robust behavior. Under natural (Figure 7) and acidic (Figure 8) pH conditions, Zn exhibited a homogeneous distribution and strong spatial overlap with the Ca–P phase. At these pH values, no Zn-rich segregated phases were detected, suggesting that Zn–phosphate interactions are sufficiently strong to promote immobilization without external pH adjustment, possibly by adsorption-driven removal. Although Zn behavior under basic conditions was not dominant in the present dataset, the results demonstrate that Zn immobilization is effective at environmentally relevant pH values and closely resembles that of chromium under acidic and natural conditions.
In contrast, nickel showed the strongest dependence on pH and the weakest overall immobilization. These results match with those from the adsorption capacity. Nickel adsorption efficiency was lower (Table 20). Under acidic conditions (Figure 8), Ni exhibited a heterogeneous distribution with partial association to the Ca–P matrix, indicating limited immobilization. At natural pH (Figure 7), Ni behavior improved slightly, with reduced segregation compared to basic conditions. Under basic conditions (Figure 9), Ni displayed pronounced heterogeneity and localized Ni-enriched regions, consistent with segregation and the formation of Ni-rich phases that compete with phosphate-mediated stabilization. In this form, Ni immobilization is strongly controlled by pH-dependent hydrolysis processes rather than by adsorption-driven removal alone.
The comparison between FTIR, XRD and SEM-EDX helped clarify how the structure evolved after adsorption and showed that the relationship between local and long-range order depended on the metal and the pH. This aligns with studies indicating that the balance between crystallinity and amorphous character governs ion exchange and phosphate accessibility in hydroxyapatite [43,44,45]. Clear agreement between FTIR and XRD appeared in cases such as Ni2+ at pH 11, where the strong narrowing of the v3(PO43−) band (79.33 → 44.52 cm−1) coincided with the highest crystallinity measured (up to 92.7%), indicating a consistent improvement in both local and global order. A similar match was observed for Zn2+ under natural and acidic pH, where decreases in FWHM paralleled increases in crystallinity, suggesting good structural compatibility between Zn2+ and the apatite lattice. Cr3+ showed a weaker correspondence between techniques. Under acidic conditions, FTIR recorded broad, attenuated phosphate bands (82–84 cm−1), reflecting strong local disorder, while XRD still detected high crystallinity but with a larger dispersion. This combination has been associated with materials where well-defined crystalline domains coexist with locally distorted regions when multiple surface and structural mechanisms occur simultaneously [43,45]. By SEM-EDX, the interaction between the metals and the hydroxyapatite of fishbones appears to be mainly determined by surface adsorption mechanisms, although some precipitation phenomena could also be observed [11,12], as in the case of nickel at basic pH. This precipitation phenomena also appeared in the zinc samples at basic pH (Section 3.2.3).
3.3.4. XRF
X-ray fluorescence (XRF) quantified the total metal content retained in the hydroxyapatite-based samples after exposure to the three metals under different pH conditions and contact times. As expected for a bulk technique, XRF provides total metal content but does not distinguish whether it is surface-adsorbed, incorporated into the lattice, or present as precipitated phases.
Condition-wise averages confirmed a strong pH dependence of retention, with natural pH consistently outperforming acidic and alkaline treatments. Table 22 shows the average percentage of metal retained under each condition; detailed values for all samples are provided in Tables S13–S15. Under these conditions, zinc showed the highest incorporation (8.40%), followed by chromium (7.34%) and nickel (5.97%). At acidic pH, average retention decreased for all three metals, and alkaline pH yielded the lowest values.

Table 22.
Condition-wise averages of retained metal (%).
Several samples showed higher metal content at 72 h than at 6 h, particularly for Zn in salmon and gilt-head bream and for Cr in hake, whereas Ni exhibited smaller variations with time. When analyzed by species, salmon (S) and gilt-head bream (G) retained the highest metal contents, especially for Zn and Cr at natural pH. Hake (H) and megrim (M) showed intermediate values, following the same pH- and time-dependent trends. Across all species, calcined samples (XFC) retained less metal, particularly under acidic and alkaline conditions, reflecting the more reduced availability of incorporation sites after thermal treatment. Possibly, that one was the reason for the higher order and crystallinity for species at pH 3 and pH 11 than those at pH natural (Section 3.3.1 and Section 3.3.2).
The highest retention was obtained for SS-Zn-6 (16.57%), closely followed by SS-Zn-72 (15.12%) and HS-Cr-6 (13.66%), all under natural pH. In contrast, the lowest value corresponded to HS-Cr-72-11 (1.67%) at pH 11, with other low performers also associated with calcined samples at the extreme pH values (Table 23).

Table 23.
The best (top-5) and worst (bottom-5) samples according to metal retention by XRF.
Although XRF quantifies the total metal associated with the solid and AAS measures the dissolved portion remaining in the aqueous phase [46], the results provided by both techniques matched. Metal retained by XRF and removal efficiency by AAS indicated that fishbones were more effective for retaining Zn, and then Ni and Cr. Precipitation of Zn at basic pH and nickel (observed by SEM-EDX) could indicate that in both metals, adsorption and precipitation mechanisms provoked the highest values of removal efficiency.
Overall, XRF showed that natural pH favored metal association with the solid, that longer contact times increased retention in some systems, that calcination reduced uptake, and that the biological origin of the fishbones influenced how much metal remains in the material.
3.3.5. Colorimetry
Colorimetric evaluation after adsorption focused on chroma () and total color difference (), determined according to (6) and the CIE (Tables S16–S18).
Chromium produced the strongest changes in color characteristics of the samples exposed to individual metal solutions, particularly under acidic and alkaline conditions, as was determined by FTIR and XRD. At natural pH, some samples exhibited marked increases in such as HF-Cr-6 ( 5.05), while others decreased considerably, exemplified by MF-Cr-6 ( −21.99). A similar pattern was observed at pH 3 and 11, with certain samples recording the highest increases in saturation, such as HF-Cr-72-3 ( 8.11) and MFC-Cr-6-11 ( 2.05), and others presenting the most pronounced reductions, including MF-Cr-72-3 ( −16.71) and MF-Cr-6-11 ( −20.28). The values confirmed these contrasts, ranging from minimal differences at neutral conditions to highly significant changes in acidic and alkaline media (Tables S16–S18).
Nickel exposure also generated appreciable variations in color of samples although generally less intense than those induced by chromium. Increased saturation was clear in some uncalcined samples, particularly HF-Ni-6 ( 16.13) and HF-Ni-6-3 ( 18.30), while reductions were more common in calcined ones such as MF-Ni-6 ( −15.97) and MF-Ni-72-3 ( −22.14). The values also reflected this dual behavior, with noticeable differences under alkaline conditions as seen in HF-Ni-6-11 ( 8.52) and MF-Ni-6-11 ( −21.41) (Tables S16–S18).
In contrast, zinc exposure induced only moderate modifications on adsorbent color. Variations in were detected, particularly at natural and acidic pH with increases in samples such as GF-Zn-6 ( 8.54) and HF-Zn-6-3 ( 1.86), while decreases were more notable in SF-Zn-72 ( −14.32) and MF-Zn-72-3 ( −9.88), but they were less pronounced. The values were consistent with this trend, showing minor differences in most cases (Tables S16–S18).
This interpretation was consistent with visual inspection: only chromium-treated samples displayed evident color differences, while nickel- and zinc-treated samples showed changes that were measurable but imperceptible to the naked eye (see Table 24, Table 25 and Table 26).

Table 24.
Photographic record of gilt-head bream samples (GF and GFS) after chromium adsorption at natural, acidic (pH 3), and alkaline (pH 11) conditions, showing visual changes at 0, 6, and 72 h.

Table 25.
Photographic record of gilt-head bream samples (GF and GFS) after nickel adsorption at natural, acidic (pH 3), and alkaline (pH 11) conditions, showing visual changes at 0, 6, and 72 h.

Table 26.
Photographic record of gilt-head bream samples (GF and GFS) after zinc adsorption at natural, acidic (pH 3), conditions, showing visual changes at 0, 6, and 72 h.
The objective of the present study was not to test the reusability and regeneration of hydroxyapatite generated from fishbones. In fact, only a small proportion of articles on adsorption address the reusability of their adsorbents. Desorption experiments would be very useful for evaluating the amount of metal ions that can be recovered from these hydroxyapatite adsorbents. However, these experiments would require several weeks of additional work (clearly out of the standards of the responses time of this journal) and a comprehensive experimental and characterization framework in order to ensure a rigorous and meaningful study. This effort would be costly and is beyond the scope of the present manuscript. The desorption tests should be included in a future article that emphasizes the reusability of fishbones as adsorbent materials, after several adsorption–desorption cycles with harmful metals, as mentioned in Conclusions.
Regenerating hydroxyapatite after adsorbing heavy metals is essential for sustainable wastewater treatment. The process would be focused on chemical desorption to break the bonds between the metal cations and the HAp surface functional groups. One possible approach would involve forming water-soluble complexes using a strong chelating agent (e.g., EDTA) to recover the metals adsorbed onto the HAp.
4. Conclusions
This study demonstrated the feasibility of producing hydroxyapatite-based adsorbents from fishbone waste, confirming their potential as sustainable and low-cost materials for heavy metal water purification. Structural characterization confirmed that hydroxyapatite was the predominant phase and revealed unexpected similarities between fish species pairs without a phylogenetic relationship (megrim with hake, and gilt-head bream with salmon), offering new insights into bone-derived mineral structures.
In adsorption tests, uncalcined samples proved effective, while calcination only improved removal efficiency for chromium at alkaline pH and nickel at acidic pH. For each metal and pH condition, the strong influence of both the fish species and the type of treatment applied to each fishbone was verified. Megrim and hake bones emerged as the most promising candidates to prepare new adsorbents for heavy metal removal in water treatment, particularly under natural and acidic conditions.
Overall, zinc was the most efficiently removed metal from water, followed by nickel and chromium. These findings emphasize the value of fishbone-derived hydroxyapatite as a circular-economy solution, contributing to sustainable water treatment technologies and effective metal removal from contaminated waters.
This is the first paper in the scientific literature to study the adsorption capacity of three heavy metals (Cr, Zn and Ni) on four different types of fishbones and under three different pH conditions, thus providing very interesting data in this field. The interaction between the metals and the hydroxyapatite of fishbones appears to be mainly determined by surface adsorption mechanisms. Looking forward to several directions for future work, the use of mixed-species material could be evaluated to determine potential synergic effects. Competitive adsorption in multi-metal solutions, as well as validation under naturally contaminated waters, are essential to confirm environmental applicability. Finally, strategies for the safe disposal or reuse of spent adsorbents must be developed to ensure sustainable long-term deployment. Desorption studies would be very useful for evaluating the amount of metal ions that can be recovered from the hydroxyapatite adsorbents. In addition, a systematic comparison in adsorption performance between lyophilized and oven-dried hydroxyapatite is identified as a relevant and valuable direction for future research.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/environments13020102/s1, Figure S1: Diagrammatic representation of the processes for the preparation of HAp from fishbones. Figure S2: Thermogravimetry of the clean fishbones samples: (a) gilt-head bream, (b) salmon, (c) hake tail, and (d) megrim. Figure S3: Effect of pH on the concentration of each dissolved metal in blanks samples under acid (pH 3), natural pH and alkaline conditions (pH 11). Figure S4: Cr3+ adsorption at the different pH values: (a) gilt-head bream, (b) salmon, (c) hake tail, and (d) megrim. Figure S5: Ni2+ adsorption at the different pH values: (a) gilt-head bream, (b) salmon, (c) hake tail, and (d) megrim. Figure S6: Zn2+ adsorption at the different pH values: (a) gilt-head bream, (b) salmon, (c) hake tail, and (d) megrim. Figure S7: Cr3+ adsorption at the different pH values: (a) gilt-head bream, (b) salmon, (c) hake tail, and (d) megrim. Figure S8: Ni2+ adsorption at the different pH values: (a) gilt-head bream, (b) salmon, (c) hake tail, and (d) megrim. Figure S9: Zn2+ adsorption at the different pH values: (a) gilt-head bream, (b) salmon, (c) hake tail, and (d) megrim. Figure S10: XRD of fishbone-derived hydroxyapatite; overlapped diffractograms before and after metal adsorption for (a) MFC vs. MFC-Zn-6, (b) GFC vs. GFC-Ni-6-3, and (c) GFC vs. GFC-Cr-6-11. Table S1: Summary of the adsorption experimental conditions used for the preliminary screening (commercial HAp) and the main adsorption tests (fishbone-derived HAp: XF and XFC) with Cr3+, Ni2+, and Zn2+. Table S2: Colorimetric parameters for individual fishbone samples before adsorption. Table S3: Optimal procedure for HAp (80 mg) success removal for 50 mg/L of the metal ion, 20 °C, and natural pH. Table S4: XF and XFC adsorption of Cr3+ (50 mg/L) at 20 °C and natural pH. Table S5: XF and XFC adsorption of Ni2+ (50 mg/L) at 20 °C and natural pH. Table S6: XF and XFC adsorption of Zn2+ (50 ppm) at 20 °C and natural pH. Table S7: XF and XFC adsorption of Cr3+ (50 ppm) at 20 °C and pH 3. Table S8: XF and XFC adsorption of Ni2+ (50 mg/L) at 20 °C and pH 3. Table S9: XF and XFC adsorption of Zn2+ (50 mg/L) at 20 °C and pH 3. Table S10: XF and XFC adsorption of Cr3+ (50 mg/L) at 20 °C and pH 11. Table S11: XF and XFC adsorption of Ni2+ (50 mg/L) at 20 °C and pH 11. Table S12: pH values corresponding to the metal blank samples without adsorbent. Table S13: Metal retained (%) at natural pH for XF and XFC samples. Table S14: Metal retained (%) at acidic pH for XF and XFC samples. Table S15: Metal retained (%) at basic pH for XF and XFC samples. Table S16: Colorimetry data for XF and XFC samples at natural pH. Table S17: Colorimetry data for XF and XFC samples at acidic pH. Table S18: Colorimetry data for XF and XFC samples at basic pH.
Author Contributions
Conceptualization, M.M.C., F.J.P.E. and A.D.B.; methodology, M.M.C., F.J.P.E. and A.D.B.; validation, F.J.P.E. and A.D.B.; formal analysis, M.M.C.; investigation, M.M.C., F.J.P.E. and A.D.B.; resources, F.J.P.E. and A.D.B.; data curation, M.M.C.; writing—original draft preparation, M.M.C.; writing—review and editing, M.M.C., F.J.P.E. and A.D.B.; visualization, M.M.C.; supervision, F.J.P.E. and A.D.B.; project administration, F.J.P.E. and A.D.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The original contributions presented in this study are included in the article/Supplementary Materials. Further inquiries can be directed to the corresponding author.
Acknowledgments
The authors thank Cristina Luzuriaga for technical assistance with XRD and XRF measurements and Marta Yárnoz for support in atomic absorption spectroscopy analyses. SEM-EDX images and spectra were performed by Mikel Domeño in a FE-SEM Zeiss microscope, model Sigma 300 VP. During the preparation of this manuscript, the authors used ChatGPT (OpenAI, version GPT-5) to assist in refining the English writing and improving clarity. The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Wang, Z.; Luo, P.; Zha, X.; Xu, C.; Kang, S.; Zhou, M.; Nover, D.; Wang, Y. Overview Assessment of Risk Evaluation and Treatment Technologies for Heavy Metal Pollution of Water and Soil. J. Clean. Prod. 2022, 379, 134043. [Google Scholar] [CrossRef]
- Hama Aziz, K.H.; Mustafa, F.S.; Omer, K.M.; Hama, S.; Hamarawf, R.F.; Rahman, K.O. Heavy Metal Pollution in the Aquatic Environment: Efficient and Low-Cost Removal Approaches to Eliminate Their Toxicity: A Review. RSC Adv. 2023, 13, 17595–17610. [Google Scholar] [CrossRef]
- Calmuc, V.A.; Calmuc, M.; Arseni, M.; Topa, C.M.; Timofti, M.; Burada, A.; Iticescu, C.; Georgescu, L.P. Assessment of Heavy Metal Pollution Levels in Sediments and of Ecological Risk by Quality Indices, Applying a Case Study: The Lower Danube River, Romania. Water 2021, 13, 1801. [Google Scholar] [CrossRef]
- Saravanan, A.; Senthil Kumar, P.; Jeevanantham, S.; Karishma, S.; Tajsabreen, B.; Yaashikaa, P.R.; Reshma, B. Effective Water/Wastewater Treatment Methodologies for Toxic Pollutants Removal: Processes and Applications towards Sustainable Development. Chemosphere 2021, 280, 130595. [Google Scholar] [CrossRef]
- Sokić, K.; Dikić, J.; Veljović, Đ.; Jelić, I.; Radovanović, D.; Štulović, M.; Jevtić, S. Preparation and Characterization of Hydroxyapatite-Modified Natural Zeolite: Application as Adsorbent for Ni2+ and Cr3+ Ion Removal from Aqueous Solutions. Processes 2025, 13, 818. [Google Scholar] [CrossRef]
- Schoofs, H.; Schmit, J.; Rink, L. Zinc Toxicity: Understanding the Limits. Molecules 2024, 29, 3130. [Google Scholar] [CrossRef] [PubMed]
- Bansal, N.; Mortula, M.M.; Al-Asheh, S. Heavy Metal Removal from Produced Water Using Waste Materials: A Comparative Study. Water 2025, 17, 2789. [Google Scholar] [CrossRef]
- Ungureanu, G.; Santos, S.; Boaventura, R.; Botelho, C. Arsenic and Antimony in Water and Wastewater: Overview of Removal Techniques with Special Reference to Latest Advances in Adsorption. J. Environ. Manag. 2015, 151, 326–342. [Google Scholar] [CrossRef]
- Hernández-Cocoletzi, H.; Salinas, R.A.; Águila-Almanza, E.; Rubio-Rosas, E.; Chai, W.S.; Chew, K.W.; Mariscal-Hernández, C.; Show, P.L. Natural Hydroxyapatite from Fishbone Waste for the Rapid Adsorption of Heavy Metals of Aqueous Effluent. Environ. Technol. Innov. 2020, 20, 101109. [Google Scholar] [CrossRef]
- Gnanasekaran, R.; Yuvaraj, D.; Muthu, C.M.M.; Ashwin, R.; Kaarthikeyan, K.; Kumar, V.V.; Ramalingam, R.J.; Al-Lohedan, H.; Reddy, K. Extraction and Characterization of Biocompatible Hydroxyapatite (Hap) from Red Big Eye Fish Bone: Potential for Biomedical Applications and Reducing Biowastes. Sustain. Chem. Environ. 2024, 7, 100142. [Google Scholar] [CrossRef]
- Biedrzycka, A.; Skwarek, E. Composites of Hydroxyapatite and Their Application in Adsorption, Medicine and as Catalysts. Adv. Colloid Interface Sci. 2024, 334, 103308. [Google Scholar] [CrossRef]
- Salem, A.; Fattahi, B.; Salem, A. Competitive adsorption of lead, cadmium, and zinc ions from wastewater by poorly crystalline hydroxyapatite sustainably fabricated from eggshells via ultrasound-assisted precipitation. J. Mol. Liq. 2025, 436, 128232. [Google Scholar] [CrossRef]
- Nam, P.V.; Hoa, N.V.; Trung, T.S. Properties of Hydroxyapatites Prepared from Different Fish Bones: A Comparative Study. Ceram. Int. 2019, 45, 20141–20147. [Google Scholar] [CrossRef]
- Rangappa, H.S.; Herath, I.; Lin, C.; Subrahmanyam, C. Industrial Waste-Based Adsorbents as a New Trend for Removal of Water-Borne Emerging Contaminants. Environ. Pollut. 2024, 343, 123140. [Google Scholar] [CrossRef] [PubMed]
- Coppola, D.; Lauritano, C.; Palma Esposito, F.; Riccio, G.; Rizzo, C.; de Pascale, D. Fish Waste: From Problem to Valuable Resource. Mar. Drugs 2021, 19, 116. [Google Scholar] [CrossRef]
- Miyah, Y.; Benjelloun, M.; Salim, R.; Nahali, L.; Mejbar, F.; Lahrichi, A.; Iaich, S.; Zerrouq, F. Experimental and DFT Theoretical Study for Understanding the Adsorption Mechanism of Toxic Dye onto Innovative Material Fb-HAp Based on Fishbone Powder. J. Mol. Liq. 2022, 362, 119739. [Google Scholar] [CrossRef]
- El-Nagar, D.A.; Massoud, S.A.; Ismail, S.H. Removal of Some Heavy Metals and Fungicides from Aqueous Solutions Using Nano-Hydroxyapatite, Nano-Bentonite and Nanocomposite. Arab. J. Chem. 2020, 13, 7695–7706. [Google Scholar] [CrossRef]
- Ferri, M.; Campisi, S.; Gervasini, A. Nickel and Cobalt Adsorption on Hydroxyapatite: A Study for the de-Metalation of Electronic Industrial Wastewaters. Adsorption 2019, 25, 649–660. [Google Scholar] [CrossRef]
- Kizilkaya, B.; Tekinay, A.A.; Dilgin, Y. Adsorption and Removal of Cu (II) Ions from Aqueous Solution Using Pretreated Fish Bones. Desalination 2010, 264, 37–47. [Google Scholar] [CrossRef]
- Ahamed, A.F.; Manimohan, M.; Kalaivasan, N. Fabrication of Biologically Active Fish Bone Derived Hydroxyapatite and Montmorillonite Blended Sodium Alginate Composite for In-Vitro Drug Delivery Studies. J. Inorg. Organomet. Polym. Mater. 2022, 32, 3902–3922. [Google Scholar] [CrossRef]
- Modolon, H.B.; Inocente, J.; Bernardin, A.M.; Klegues Montedo, O.R.; Arcaro, S. Nanostructured Biological Hydroxyapatite from Tilapia Bone: A Pathway to Control Crystallite Size and Crystallinity. Ceram. Int. 2021, 47, 27685–27693. [Google Scholar] [CrossRef]
- Bruker AXS GmbH. Diffrac.Suite. User Manual. Diffrac.Evaluation Package. Diffrac.Eva; Bruker AXS GmbH: Karlsruhe, Germany, 2010; pp. 149–150. [Google Scholar]
- Jastrzębski, W.; Sitarz, M.; Rokita, M.; Bułat, K. Infrared Spectroscopy of Different Phosphates Structures. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 79, 722–727. [Google Scholar] [CrossRef]
- Bouilloud, M.; Fray, N.; Bénilan, Y.; Cottin, H.; Gazeau, M.-C.; Jolly, A. Bibliographic Review and New Measurements of the Infrared Band Strengths of Pure Molecules at 25 K: H2O, CO2, CO, CH4, NH3, CH3OH, HCOOH and H2CO. Mon. Not. R. Astron. Soc. 2015, 451, 2145–2160. [Google Scholar] [CrossRef]
- Mokari, A.; Guo, S.; Bocklitz, T. Exploring the Steps of Infrared (IR) Spectral Analysis: Pre-Processing, (Classical) Data Modelling, and Deep Learning. Molecules 2023, 28, 6886. [Google Scholar] [CrossRef]
- Zinicovscaia, I.; Yushin, N.; Grozdov, D.; Humelnicu, I.; Humelnicu, D.; Mitina, T. Removal of Chromium(III) Ions from Aqueous Solutions Using Different Types of Hydroxyapatites. Desalin. Water Treat. 2020, 204, 297–305. [Google Scholar] [CrossRef]
- Mobasherpour, I.; Salahi, E.; Pazouki, M. Removal of Nickel (II) from Aqueous Solutions by Using Nano-Crystalline Calcium Hydroxyapatite. J. Saudi Chem. Soc. 2011, 15, 105–112. [Google Scholar] [CrossRef]
- Sheha, R.R. Sorption Behavior of Zn(II) Ions on Synthesized Hydroxyapatites. J. Colloid Interface Sci. 2007, 310, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Mo, N.; Zhu, Z.; Zhu, Y.; Liu, Y.; Wang, X.; Yang, H.; Zhao, N. Purification Behavior of Zn(II) in Water by Magnesium Hydroxyapatite: Surface Complexation, and Dissolution–Precipitation. Int. J. Environ. Res. Public Health 2020, 17, 3804. [Google Scholar] [CrossRef]
- Ofudje, E.A.; Adedapo, A.E.; Oladeji, O.B.; Sodiya, E.F.; Ibadin, F.H.; Zhang, D. Nano-Rod Hydroxyapatite for the Uptake of Nickel Ions: Effect of Sintering Behaviour on Adsorption Parameters. J. Environ. Chem. Eng. 2021, 9, 105931. [Google Scholar] [CrossRef]
- Lim, H.K.; Teng, T.T.; Ibrahim, M.H.; Ahmad, A.; Chee, H.T. Adsorption and Removal of Zinc (II) from Aqueous Solution Using Powdered Fish Bones. APCBEE Procedia 2012, 1, 96–102. [Google Scholar] [CrossRef]
- Chen, C.; Liu, P.; Li, Y.; Tian, H.; Zhang, Y.; Zheng, X.; Liu, R.; Zhao, M.; Huang, X. Electro-Peroxone Enables Efficient Cr Removal and Recovery from Cr(III) Complexes and Inhibits Intermediate Cr(VI) Generation in Wastewater: Performance and Mechanism. Water Res. 2022, 218, 118502. [Google Scholar] [CrossRef]
- Almendros-Ginestà, O.; Duckworth, S.; Fürst, P.Q.; Missana, T.; Altmaier, M.; Gaona, X. Thermodynamic Description of the Ni(II)-Citrate System in Alkaline, Dilute to Concentrated NaCl Solutions. Formation of Quaternary Complexes with Ca. Appl. Geochem. 2025, 180, 106290. [Google Scholar] [CrossRef]
- Kostrzewa, M.; Staszak, K.; Ginter-Kramarczyk, D.; Kruszelnicka, I.; Góra, W.; Baraniak, M.; Lota, G.; Regel-Rosocka, M. Chromium(III) Removal from Nickel(II)-Containing Waste Solutions as a Pretreatment Step in a Hydrometallurgical Process. Materials 2022, 15, 6217. [Google Scholar] [CrossRef]
- Velempini, T.; Ahamed, M.E.H.; Pillay, K. Heavy-Metal Spent Adsorbents Reuse in Catalytic, Energy and Forensic Applications- a New Approach in Reducing Secondary Pollution Associated with Adsorption. Results Chem. 2023, 5, 100901. [Google Scholar] [CrossRef]
- Zein, R.; Fauzia, S.; Bielan, Z.; Aziz, H.; Dahlan, D.; Konieczka, P.; Cieślik, B.M. Analytical Chemistry in Technical Approaches: Immobilization of Biosorbent Waste Containing Heavy Metals in Cemented Materials. Monatsh. Chem. 2022, 153, 789–800. [Google Scholar] [CrossRef]
- Gwenzi, W.; Marumure, J.; Makuvara, Z.; Simbanegavi, T.T. Recycling and Disposal of Spent Metal (Loid)-Laden Adsorbents. In Remediation of Heavy Metals; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2024; pp. 275–290. [Google Scholar]
- Rożek, P.; Florek, P.; Król, M.; Mozgawa, W.; Rożek, P.; Florek, P.; Król, M.; Mozgawa, W. Immobilization of Heavy Metals in Boroaluminosilicate Geopolymers. Materials 2021, 14, 214. [Google Scholar] [CrossRef] [PubMed]
- Sahadat Hossain, M.; Ahmed, S. FTIR Spectrum Analysis to Predict the Crystalline and Amorphous Phases of Hydroxyapatite: A Comparison of Vibrational Motion to Reflection. RSC Adv. 2023, 13, 14625–14630. [Google Scholar] [CrossRef]
- Dal Sasso, G.; Asscher, Y.; Angelini, I.; Nodari, L.; Artioli, G. A Universal Curve of Apatite Crystallinity for the Assessment of Bone Integrity and Preservation. Sci. Rep. 2018, 8, 12025. [Google Scholar] [CrossRef]
- Tang, Y.; Chappell, H.F.; Dove, M.T.; Reeder, R.J.; Lee, Y.J. Zinc Incorporation into Hydroxylapatite. Biomaterials 2009, 30, 2864–2872. [Google Scholar] [CrossRef]
- Sallam, S.M.; Tohami, K.M.; Sallam, A.M.; Abo Salem, L.I.; Adel Mohamed, F. Synthesis and Characterization of Hydroxyapatite Contain Chromium. J. Biophys. Chem. 2012, 3, 278–282. [Google Scholar] [CrossRef]
- He, M.; Zhang, Z.; Wang, M.; Liang, C.; Wang, H.; Cheng, C.; Li, Y.; Wang, Y.; Zhang, Z. A Review of Hydroxyapatite Synthesis for Heavy Metal Adsorption Assisted by Machine Learning. J. Hazard. Mater. 2025, 481, 136525. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wu, W.; Pan, D.; Wu, H. Study on the Behaviors and Mechanism of Ni(II) Adsorption at the Hydroxyapatite-Water Interface: Effect of Particle Size. Adsorpt. Sci. Technol. 2022, 2022, 3838766. [Google Scholar] [CrossRef]
- Jaffar, F.H.; Othman, M.H.D.; Ismail, N.J.; Puteh, M.H.; Kurniawan, T.A.; Abu Bakar, S.; Abdullah, H. Hydroxyapatite-Based Materials for Adsorption, and Adsorptive Membrane Process for Heavy Metal Removal from Wastewater: Recent Progress, Bottleneck and Opportunities. J. Taiwan Inst. Chem. Eng. 2024, 164, 105668. [Google Scholar] [CrossRef]
- Spearman, S.; Bartrem, C.; Sharshenova, A.A.; Salymbekova, K.S.; Isirailov, M.B.; Gaynazarov, S.A.; Gilmanov, R.; von Lindern, I.H.; von Braun, M.; Möller, G. Comparison of X-Ray Fluorescence (XRF) and Atomic Absorption Spectrometry (AAS) Results for an Environmental Assessment at a Mercury Site in Kyrgyzstan. Appl. Sci. 2022, 12, 1943. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.