Abstract
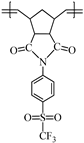
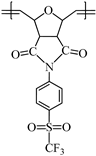
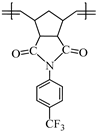
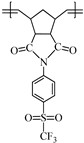
This work reports on the synthesis and ring-opening metathesis polymerization (ROMP) of two novel homologous sulfonyl-containing norbornene dicarboximide monomers, specifically, N-4-(trifluoromethylsulfonyl)phenyl-norbornene-5,6-dicarboximide (1a) and N-4-(trifluoromethylsulfonyl)phenyl-7-oxanorbornene-5,6-dicarboximide (1b) using the Grubbs 2nd generation catalyst (I). The polymers are thoroughly characterized by nuclear magnetic resonance (NMR), Fourier transform infrared spectroscopy (FT-IR), thermomechanical analysis (TMA), thermogravimetric analysis (TGA), atomic force microscopy (AFM), and X-ray diffraction (XRD), among other techniques. A comparative study of gas transport in membranes based on these ROMP-prepared polymers is performed and the gases studied are hydrogen, oxygen, nitrogen, carbon dioxide, methane, ethylene and propylene. It is found that the presence of sulfonyl pendant groups in the polymer backbone increases the gas permselectivity in slight detriment of the gas permeability compared to a polynorbornene dicarboximide lacking sulfonyl groups. The membrane of the sulfonyl-containing polymer with an oxygen heteroatom in the cyclopentane ring, 2b, is also found to have one of the largest permselectivity coefficients reported to date for the separation of H2/C3H6 in glassy polynorbornene dicarboximides.
1. Introduction
Gas separation is a research topic of the utmost importance due to its relevance in industrial applications in several sectors, such as oil and gas, chemistry, petrochemicals, medicine, the environment and the food and beverage industry, among others [1,2]. Some gas separation technologies include cryogenic distillation, liquid or chemical absorption, pressure swing adsorption or vacuum adsorption (PSA, VSA), and adsorption on solid materials. However, some of the above require high energy consumption, maintenance, and complex equipment [3,4,5]. Membrane technology for gas separation is a process in which the gas dissolves in the membrane and diffuses through it at a certain rate, driven by the pressure difference on each side of the membrane [6]. It is considered an exceptional method for gas separation due to its simple operating mechanism, high efficiency, low costs and energy demands, lower environmental impact, and the possibility of chemically adapting the membranes to specific applications [7,8]. It is worth noting that gas separation membranes are mainly manufactured from polymeric materials.
In this sense, polynorbornene derivatives are attractive materials for gas separation membranes due to their remarkable thermo-mechanical and gas transport properties; in addition, they can be obtained by ring-opening metathesis polymerization (ROMP) as well as addition polymerization, resulting in different polymeric materials (polyalkenamers and polyolefins) with, in turn, different gas transport properties [9,10,11]. In particular, norbornene dicarboximide monomers can be easily modified, thereby facilitating the synthesis of homologous series of functionalized polymers, both linear [12,13] and cross-linked [14,15]. Likewise, polynorbornene dicarboximides also exhibit easy modification of their polymer backbones, including post-polymerization reactions, which facilitates obtaining membranes with tailored properties for various purposes [16,17,18].
The introduction of heteroatoms into the polymer main chain is another tool for tailoring physicochemical properties such as hydrophilicity [19,20,21], metal adsorption [22,23,24] and the uptake of other elements such as iodine [25,26,27], increasing electron density [28], tunning the energy gap in conducting polymers [29,30] and, of course, improving gas transport properties, among others [31]. For instance, the introduction of oxygen containing groups to the polynorbornene structure, such as epoxy, has improved selectivity over voluminous gases such as CO2 due to the molecular interactions between carbon dioxide molecules and the present ether bonds [32,33].
The sulfonyl group is the basis of polysulfones, a class of amorphous thermoplastic polymers characterized by their high thermal resistance and superior mechanical strength. This class of polymers has been shown to form membranes that are attractive for gas separation applications [34,35]. Another approach to enhance gas separation performance in polymer membranes involves mixing two polymers with different physicochemical properties and several studies have addressed this issue with a mixed polymer composed of polysulfone and polyimide [36,37]. The outcomes indicate that these mixed polymer membranes can provide higher gas permeability and improved membrane features compared to pure polymers in simple membranes [38,39,40]. Since imide moieties tend to compact polymer chains, the addition of bulky sulfonyl groups decreases intermolecular chain interactions and semicrystalline structure, thereby increasing free volume and improving gas permeability [41,42,43].
This study aims to elucidate the fundamental structure–property relationships governing gas permeability in a novel homologous series of functionalized polynorbornene dicarboximides synthesized via ROMP and comprehensively characterized by Fourier transform infrared spectroscopy, nuclear magnetic resonance, thermomechanical analysis, thermogravimetric analysis, X-ray diffraction, atomic force microscopy, and related techniques. In particular, we examine how the deliberate incorporation of sulfonyl pendant groups and an oxygen heteroatom within the cyclopentane ring modulates the physicochemical properties of the macromolecular architecture. Although polynorbornene-based structures have been addressed in gas separation, the synergistic effect arising from the combination of an electron-withdrawing sulfonyl group with an oxygen-bridging heteroatom in polynorbornene dicarboximides has remained largely unexplored in the membrane literature. This strategy enables the decoupling and assessment of the influence of oxygen heteroatom-induced chain packing efficiency and the strong electron-withdrawing nature of the sulfonyl moiety on membrane gas permeability. To the best of our knowledge, this work represents the first study employing these specific sulfonyl-containing polynorbornene dicarboximides for gas separation membranes, thereby establishing a new framework for the rational design of membranes with enhanced gas permselectivity.
2. Experimental Part
2.1. Characterization Techniques
The FT-IR spectra were recorded using a Thermo Scientific Nicolet iS10 FT-IR spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) equipped with an ATR accessory and a diamond crystal, over the range of 4000–650 cm−1 at a spectral resolution of 4 cm−1. The 1H-NMR, 13C-NMR and 19F-NMR spectra were recorded on a Bruker Avance III HD spectrometer (Bruker AXS GmbH, Karlsruhe, Germany) at 400, 100 and 370 MHz, respectively, using CDCl3 and DMF-d7 as solvents. Tetramethylsilane (TMS) and hexafluorobenzene were employed as internal standards for the NMR analysis. The glass transition temperature (Tg) was measured with a TA Instruments Thermomechanical Analyzer TMA Q400 (TA Instruments, New Castle, DE, USA) using a 10 °C/min heating rate under nitrogen atmosphere between 30 °C and 400 °C. The decomposition temperature (Td) was measured with a TA Instruments Thermogravimetric analyzer TGA 5500 (TA Instruments, New Castle, DE, USA) using a 10 °C/min heating rate under nitrogen atmosphere between 30 °C and 800 °C. A universal mechanical testing machine (Instron 1125-5500R, Instron, Norwood, MA, USA) was used to measure the Young’s modulus (E) and tensile strength (σ), employing a 50 kg load cell and a crosshead speed of 10 mm/min. The density of the polymers was determined by the flotation method, in ethanol at ambient conditions, with a Sartorius analytical balance model Quintix 124-1s (Sartorius AG, Göttingen, Germany). The X-ray diffractograms were obtained in a Bruker diffractogram model D2-Phaser 2nd Generation (Bruker AXS GmbH, Karlsruhe, Germany) using CuKα radiation from 7 to 70° in the 2θ scale, at 30 Kv and 10 mA. The atomic force microscopy images of the polymer films were obtained in a Veeco Multimode SPM AFM (Veeco Instruments, Plainview, NY, USA) using the tapping mode.
2.2. Reagents
Norbornene-5,6-dicarboxylic anhydride (NDA) was prepared via the Diels–Alder cycloaddition reaction between maleic anhydride and cyclopentadiene, following literature procedures [14]. 1,2-Dichloroethane was dried and distilled over CaH2. Acetic anhydride, dichloromethane, chloroform, hexanes, methanol, ethanol, N,N-dimethylformamide (DMF) and anhydrous sodium acetate were used without further purification. 4-(Trifluoromethylsulfonyl)aniline, furan, tricyclohexylphosphine [1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidene][benzylidene] ruthenium dichloride (I), as well as other reagents, were acquired from Merck Sigma-Aldrich (St. Louis, MO, USA) and used as received.
2.3. Synthesis of Monomers
2.3.1. Synthesis of Monomer N-4-(Trifluoromethylsulfonyl)phenyl-norbornene-5,6-dicarboximide (1a)
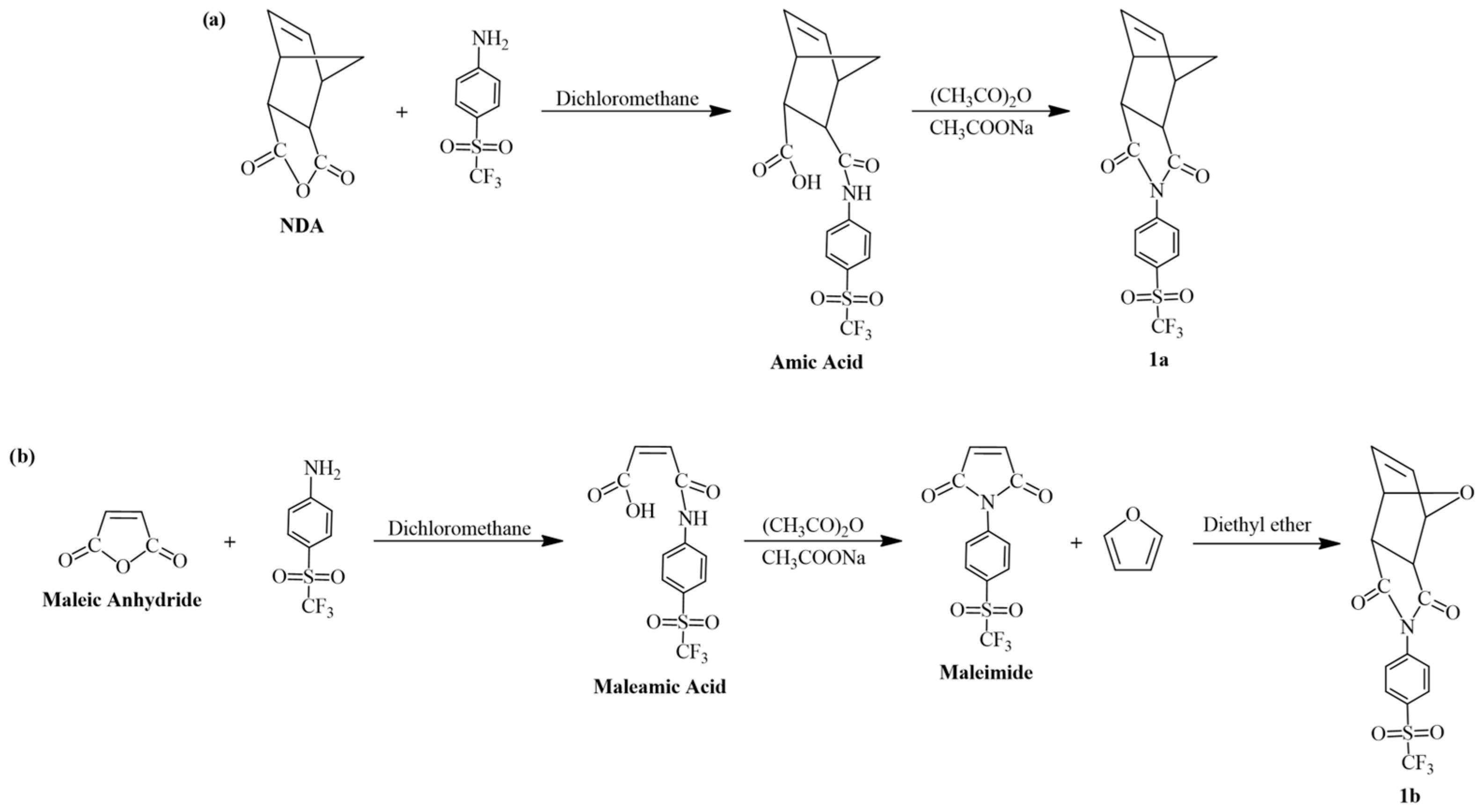
0.2 g (0.88 mmol) of 4-(trifluoromethylsulfonyl)aniline and 0.1457 g (0.88 mmol) of NDA were dissolved in 20 mL of dichloromethane. The solution was maintained under reflux conditions for 24 h and an amic acid was obtained. The product was separated by filtration and dried at room temperature. Then, it was dissolved in 20 mL of acetic anhydride along with 2.0 g of anhydrous sodium acetate. This solution was maintained for 24 h at 65 °C, then allowed to cool to room temperature. The resulting product was precipitated in ice water and washed several times. The obtained monomer 1a was recovered by filtration and recrystallized twice in hexanes (Scheme 1a). Yield: 36%. Melting point: 153–155 °C.
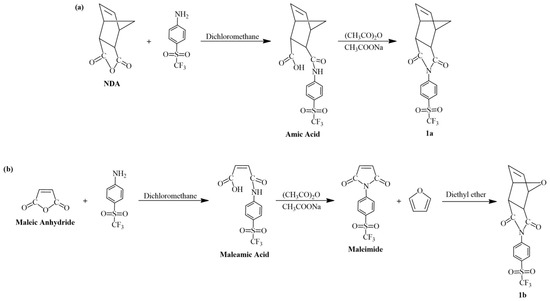
Scheme 1.
Synthesis of the sulfonyl-containing monomers: (a) 1a and (b) 1b, respectively.
FT-IR (powder, cm−1): ν 3100 (C=C–H), 2990 (C–H, asym.), 2891 (C–H, sym.), 1773 (C=O, asym), 1701 (C=O, sym.), 1586 (C=C), 1376 (C–N), 1301 (S=O), 1169 (C–F), 698 (C–S).
1H-NMR (400 MHz, CDCl3, ppm): δ 8.06–7.55 (4H, m), 7.64–7.52 (endo, m), 6.37 (2H, s), 6.26 (endo, s), 3.59–3.32 (2H, m), 3.56–3.47 (endo, m), 2.98–2.87 (2H, m), 1.70–1.58 (1H, m), 1.81 (endo, d), 1.49–1.36 (1H, m).
13C-NMR (100 MHz, CDCl3, ppm): δ 175.99 (C=O), 139.18 (C–S), 138.08 (C=C), 132.19 (C–H, ar.), 130.38 (C–N), 127.89 (C–H, ar.), 124.55 (C–F), 121.31 (C–F), 118.07 (C–F), 114.83 (C–F), 47.96 (C–H), 46.09 (C–H), 43.10 (CH2).
19F-NMR (370 MHz, CDCl3, ppm): δ −78.09.
2.3.2. Synthesis of Monomer N-4-(Trifluoromethylsulfonyl)phenyl-7-oxanorbornene-5,6-dicarboximide (1b)
0.2 g (0.88 mmol) of 4-(trifluoromethylsulfonyl)aniline and 0.0862 g (0.88 mmol) of maleic anhydride were dissolved in 20 mL of dichloromethane. The solution was maintained under reflux conditions for 24 h and a maleamic acid was obtained. The product was separated by filtration and dried at room temperature. Then, it was dissolved in 20 mL of acetic anhydride along with 2.0 g of anhydrous sodium acetate. This solution was maintained for 24 h at 65 °C, then allowed to cool to room temperature. The resulting maleimide was precipitated in ice water and washed several times. The maleimide was recovered by filtration and recrystallized twice in hexanes. Next, the maleimide and furan were dissolved in diethyl ether at a molar ratio of 1:1. The solution was maintained under stirring at room temperature for 5 days, and the precipitation of a white powder was observed. The obtained monomer 1b was collected by filtration and recrystallized twice in ethanol (Scheme 1b). Yield: 31%. Melting point: 137–139 °C.
FT-IR (powder, cm−1): ν 3108 (C=C–H), 3026 (C–H, asym.), 2987 (C–H, sym.), 1777 (C=O, asym), 1712 (C=O, sym.), 1592 (C=C), 1362 (C–N), 1309 (S=O), 1172 (C–F), 1085 (C–O), 693 (C–S).
1H-NMR (400 MHz, CDCl3, ppm): δ 7.88–7.62 (4H, m), 6.44 (2H, s), 5.26 (2H, s), 2.91 (2H, s).
13C-NMR (100 MHz, CDCl3, ppm): δ 174.37 (C=O), 139.00 (C–S), 136.83 (C=C), 131.66 (C–H, ar.), 130.69 (C–N), 127.31 (C–H, ar.), 124.89 (C–F), 121.33 (C–F), 118.09 (C–F), 115.26 (C–F), 81.67 (C–H), 47.70 (C–H).
19F-NMR (370 MHz, CDCl3, ppm): δ −78.09.
2.4. Polymerization of the Sulfonyl-Containing Monomers
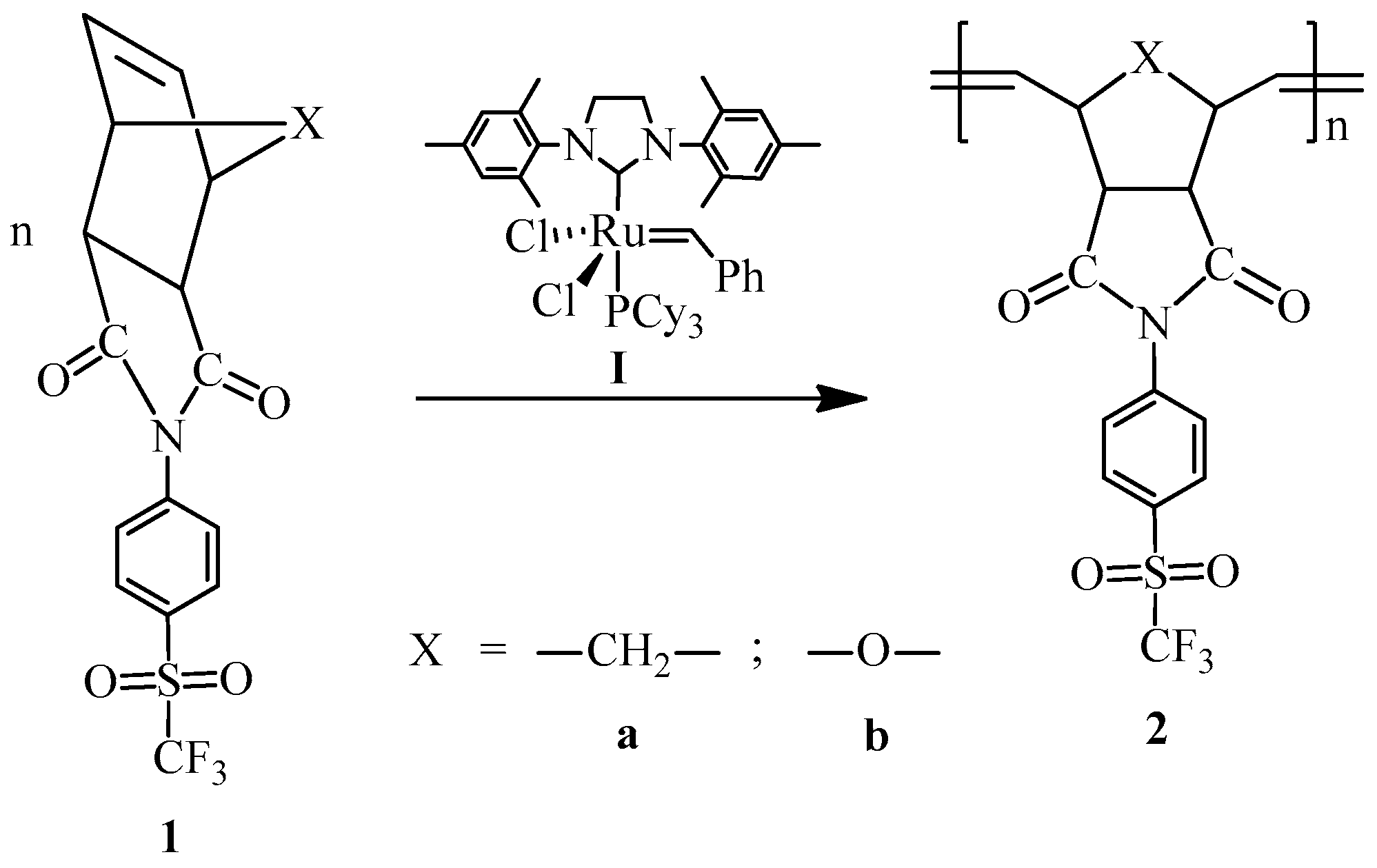
ROMP of the sulfonyl-containing monomers was conducted in 25 mL round bottom flasks under a dry nitrogen atmosphere. Ethyl vinyl ether was used in small amounts to inhibit polymerization. White polymer fibers formed upon pouring the polymer solution into excess acidified methanol under constant stirring at room temperature. The products were recovered by filtration and purified by solubilization in DMF and subsequent precipitation in stirring methanol. The polymers were finally dried under vacuum at 50 °C overnight.
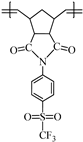
2.4.1. Synthesis of Poly(N-4-(Trifluoromethylsulfonyl)phenyl-norbornene-5,6-dicarboximide) (2a)
Monomer 1a (0.2 g, 0.53 mmol) and catalyst I (0.449 × 10−3 g, 0.00053 mmol) were dissolved in 0.7 mL of 1,2-dichloroethane and stirred at 45 °C for 2 h. The resulting poly(N-4-(trifluoromethylsulfonyl)phenyl-norbornene-5,6-dicarboximide) (2a) was obtained as white fibers and is soluble in DMF (Scheme 2).
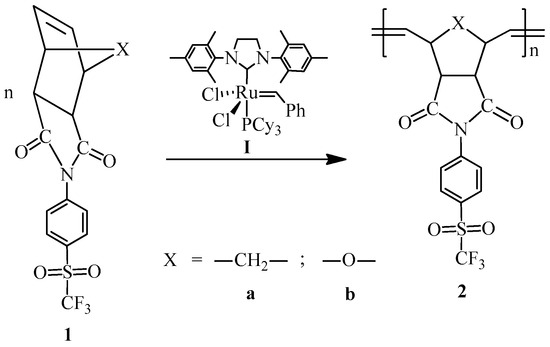
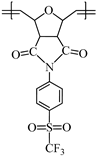
Scheme 2.
Synthesis of the sulfonyl-containing polymers via ROMP.
FT-IR (thin film, cm−1): ν 3101 (C=C–H), 2990 (C–H, asym.), 2896 (C–H, sym.), 1773 (C=O, asym), 1704 (C=O, sym.), 1597 (C=C), 1371 (C–N), 1302 (S=O), 1169 (C–F), 693 (C–S).
1H-NMR (400 MHz, DMF-d7, ppm): δ 8.69–7.97 (4H, m), 6.04 (1H, s, trans), 5.84 (1H, s,cis), 4.05–3.43 (2H, m), 3.42–3.02 (2H, m), 2.36 (1H, s), 1.88 (1H, s).
13C-NMR (100 MHz, DMF-d7, ppm): δ 176.92 (C=O), 141.26 (C–S), 134.44 (C=C), 131.72 (C–H, ar.), 129.41 (C–N), 128.89 (C–H, ar.), 124.94 (C–F), 121.70 (C–F), 118.46 (C–F), 115.23 (C–F), 51.70 (C–H), 45.85 (C–H), 43.30 (CH2).
19F-NMR (370 MHz, DMF-d7, ppm): δ −81.70.
2.4.2. Synthesis of Poly(N-4-(Trifluoromethylsulfonyl)phenyl-7-oxanorbornene-5,6-dicarboximide) (2b)
Monomer 1b (0.2 g, 0.53 mmol) and catalyst I (0.449 × 10−3 g, 0.00053 mmol) were dissolved in 0.7 mL of 1,2-dichloroethane and stirred at 45 °C for 2 h. The resulting poly(N-4-(trifluoromethylsulfonyl)phenyl-7-oxanorbornene-5,6-dicarboximide) (2b) was obtained as white fibers and is soluble in DMF (Scheme 2).
FT-IR (thin film, cm−1): ν 3106 (C=C–H), 3055 (C–H, asym.), 2970 (C–H, sym.), 1788 (C=O, asym), 1716 (C=O, sym.), 1592 (C=C), 1362 (C–N), 1301 (S=O), 1162 (C–F), 662 (C–S).
1H-NMR (400 MHz, DMF-d7, ppm): δ 8.49–7.88 (4H, m), 6.20 (1H, s, trans), 5.91 (1H, m, cis), 5.49–4.74 (2H, m), 3.78 (2H, s).
13C-NMR (100 MHz, DMF-d7, ppm): δ 174.60 (C=O), 140.64 (C–S), 131.74 (C–H, ar.), 132.16 (C=C), 129.31 (C–N), 128.83 (C–H, ar.), 124.77 (C–F), 121.51 (C–F), 118.28 (C–F), 115.03 (C–F), 77.28 (C–H), 53.20 (C–H).
19F-NMR (370 MHz, DMF-d7, ppm): δ −79.41.
2.5. Preparation of Membranes and Gas Permeability Experiments
Membranes were prepared by casting polymer solutions in DMF (~8 wt%) at 45 °C. After filtration, the solution was poured onto a glass plate and the solvent was allowed to evaporate slowly in a controlled DMF atmosphere. Next, the resulting polymer membranes were vacuum-dried at 100 °C overnight.
Gas permeation experiments were performed on a previously described system [14]. Gas permeation experiments were performed in a cell composed of two half-cells divided by the polymer membrane. A vacuum is created on both sides of the half-cells, followed by the addition of a gas at a fixed pressure to the high-pressure half-cell, also called the upstream half-cell. The upstream half-cell is connected to a Gometric pressure transducer. The flow of gas through the membrane into the low-pressure half-cell, also called the downstream half-cell, is monitored using an MKS 628/B pressure transducer. The entire permeation cell was placed in a thermostated water bath to ensure a stable temperature.
Given the downstream volume (V) in cm3, the area (A) of the membrane inside the half-cell in cm2, the membrane thickness (l) in cm, and the system pressures in cm Hg, the permeability coefficient (P) may be calculated according to the following expression:
where T denotes the temperature in K, and p and p0 correspond to the downstream and upstream gas pressures, respectively. Permeability coefficients are expressed in barrer, with 1 barrer defined as 10−10 cm3 (STP) cm/(cm2s cmHg). The diffusion coefficient of the gas (D) is calculated using the following expression [44]:
where θ is the time lag, obtained from the p versus t isotherm plot. The units of the diffusion coefficient are cm2 s−1. The apparent solubility coefficient (S) can be expressed as follows:
The units of the solubility coefficient are cm3(STP)/(cm3 cmHg).
3. Results and Discussion
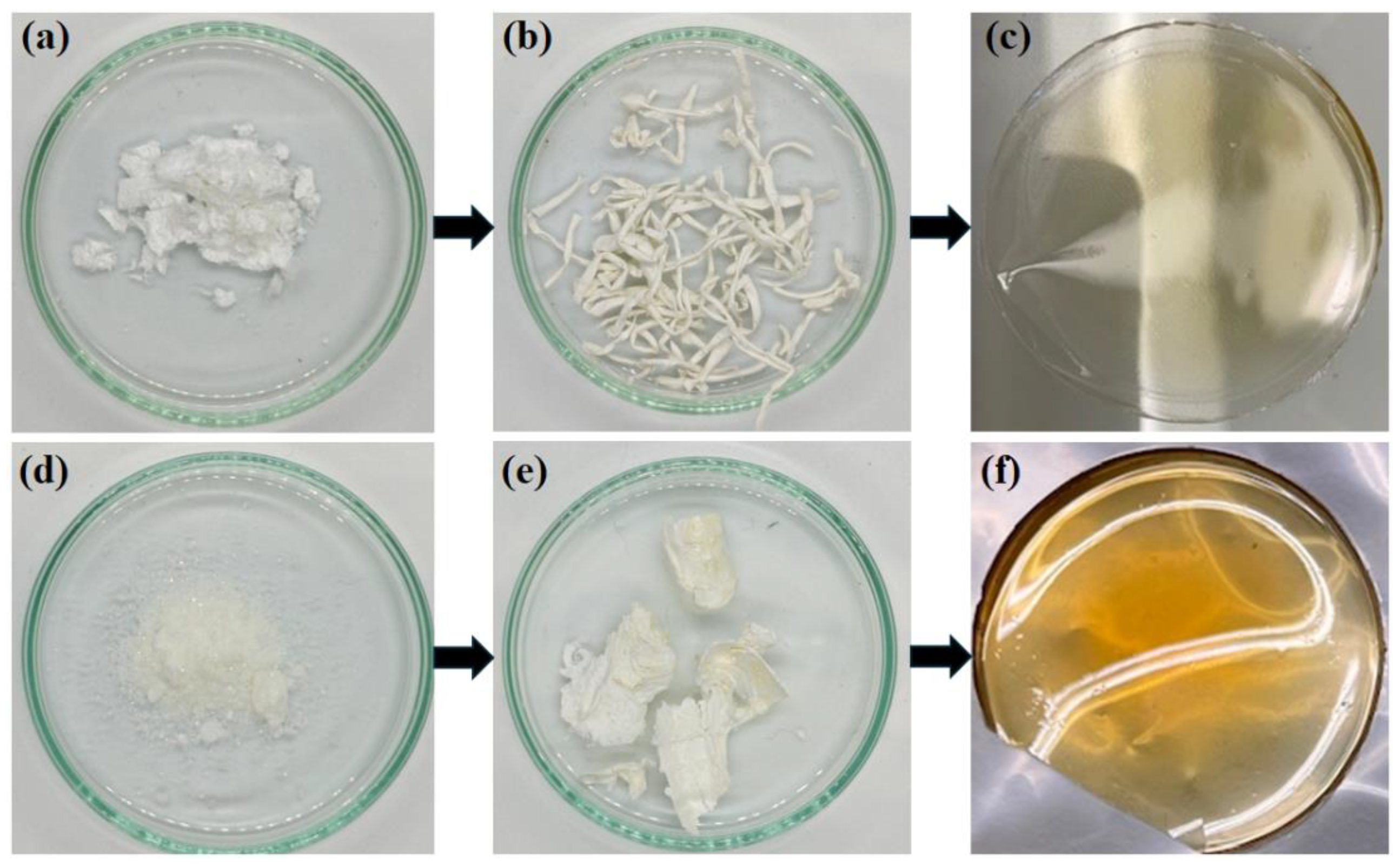
On the one hand, monomer 1a was successfully synthetized by reacting norbornene-5,6-dicarboxylic anhydride (NDA) with 4-(trifluoromethylsulfonyl)aniline to yield an amic acid that was then converted into an imide using acetic anhydride as a dehydrating agent. On the other hand, monomer 1b was successfully synthetized by reacting maleic anhydride with 4-(trifluoromethylsulfonyl)aniline to obtain a maleamic acid, which was subsequently transformed into maleimide using the dehydrating agent mentioned above. The maleimide was then subjected to a Diels-Alder reaction with furan to obtain the norbornene dicarboximide monomer. The melting points of monomers 1a and 1b were found to be in the ranges of 153–155 °C and 137–139 °C, respectively. Next, the monomers were polymerized via ring-opening metathesis polymerization (ROMP) using the Grubbs 2nd generation Catalyst (I). The monomers reacted for 2 h, affording polymers in high yields (>98%). The experimental number average molecular weights (Mn) and the molecular weight distributions (MWD) could not be determined due to the insolubility of the polymers in tetrahydrofuran (THF). However, the high yields obtained suggest the formation of high molecular weight polymers, and the theoretical molecular weight calculated from the monomer/catalyst molar ratio yields values of the order of 105 g/mol. Figure 1 shows photographs of the sulfonyl-containing norbornene dicarboximide monomers prepared in this work, along with their corresponding raw polymers obtained via ROMP and the resulting films. It can be observed that the monomers are white crystals that gave rise to white fibrous polymers capable of forming resistant transparent films with a slightly yellowish coloration. The color change in the films could be related to the effect of charge transfer (CT) interaction formed by π electrons of ring arrangements in polynorbornene dicarboximides. One of these arrangements is preferred layer packing, in which neighboring imide rings are positioned adjacent to one another. Another arrangement is mixed-layer packing, which resembles a sandwich-like arrangement formed between imide groups and adjacent aromatic rings. DMF is a high-boiling-point solvent (153 °C). Therefore, its slow evaporation during membrane casting at 45 °C enhances polymer segment mobility and promotes stronger CT interactions, leading to changes in the molecular ordering arrangement of polynorbornene dicarboximide membranes. This effect has also been observed for linear polyimides [45].
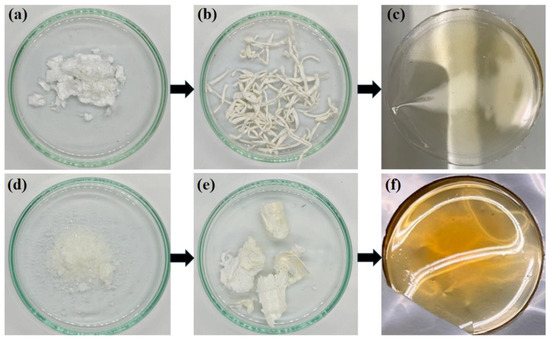
Figure 1.
Photographic images of (a) monomer 1a, (b) raw polymer 2a, (c) polymer film 2a, (d) monomer 1b, (e) raw polymer 2b, (f) polymer film 2b.
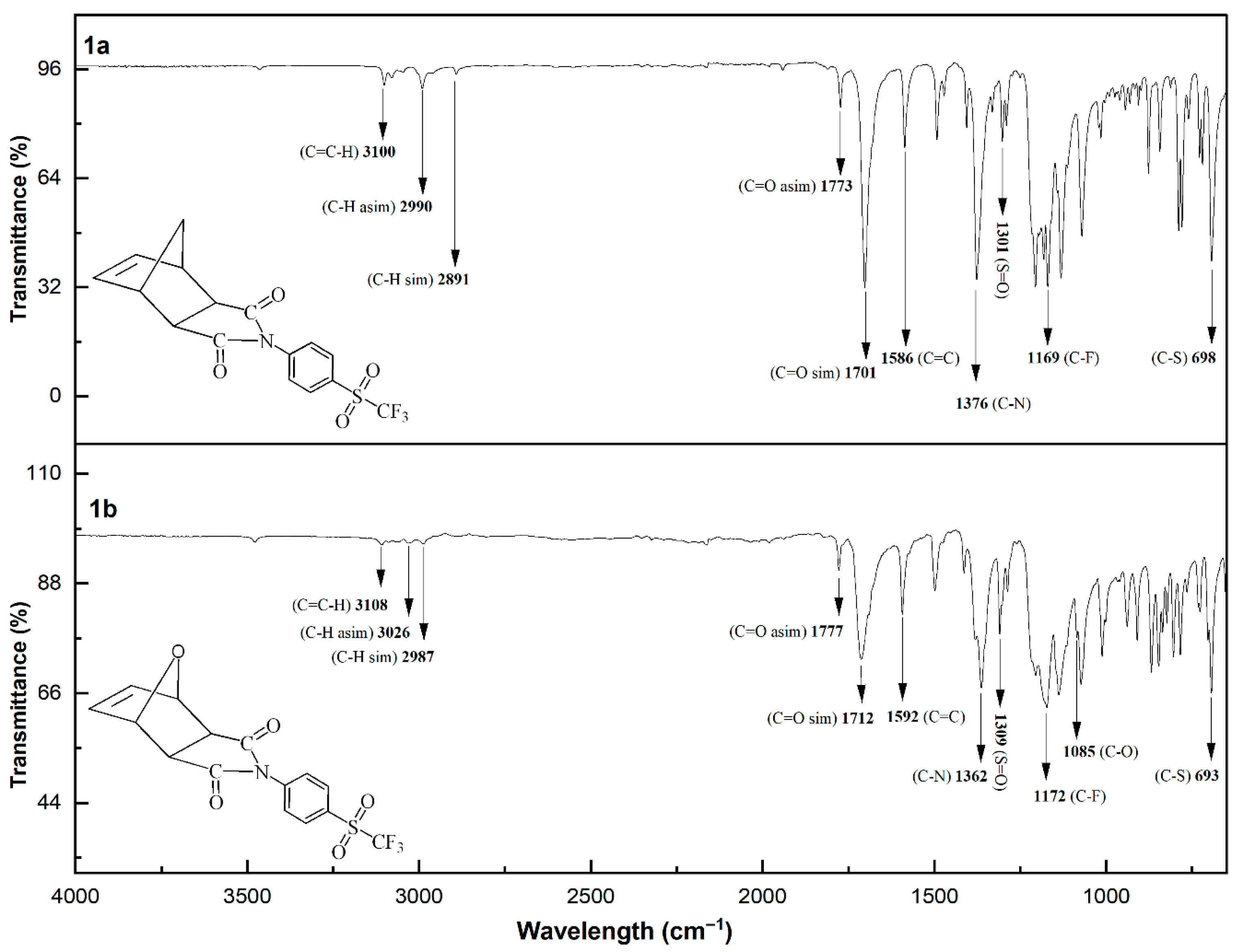
The chemical structures of the sulfonyl-containing norbornene dicarboximide monomers were confirmed by FT-IR spectroscopy, and the corresponding spectra are shown in Figure 2. The spectra are very similar to each other. In general, the following characteristic absorption bands can be observed: the aromatic H–C stretching vibration appears at approximately 3100 cm−1; the antisymmetric and symmetric stretching vibrations of the methylene C–H bonds are observed at 2990 and 2891 cm−1, respectively; the antisymmetric and symmetric stretching vibrations of the C=O bonds are located at 1773 and 1701 cm−1, respectively; the C=C stretching vibration is observed at 1586 cm−1; the C–N stretching vibration appears at around 1376 cm−1; the S=O stretching vibration is detected at approximately 1301 cm−1; the C–F stretching vibration is found near 1169 cm−1; the C–O stretching vibration appears at 1085 cm−1; and the band observed at approximately 698 cm−1 is attributed to the C–S stretching vibration.
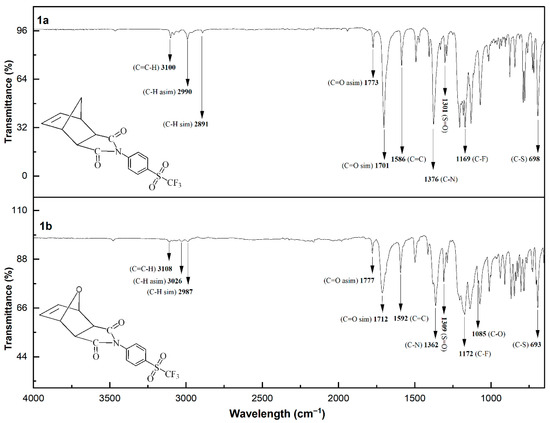
Figure 2.
FT-IR spectra of the sulfonyl-containing norbornene dicarboximide monomers.
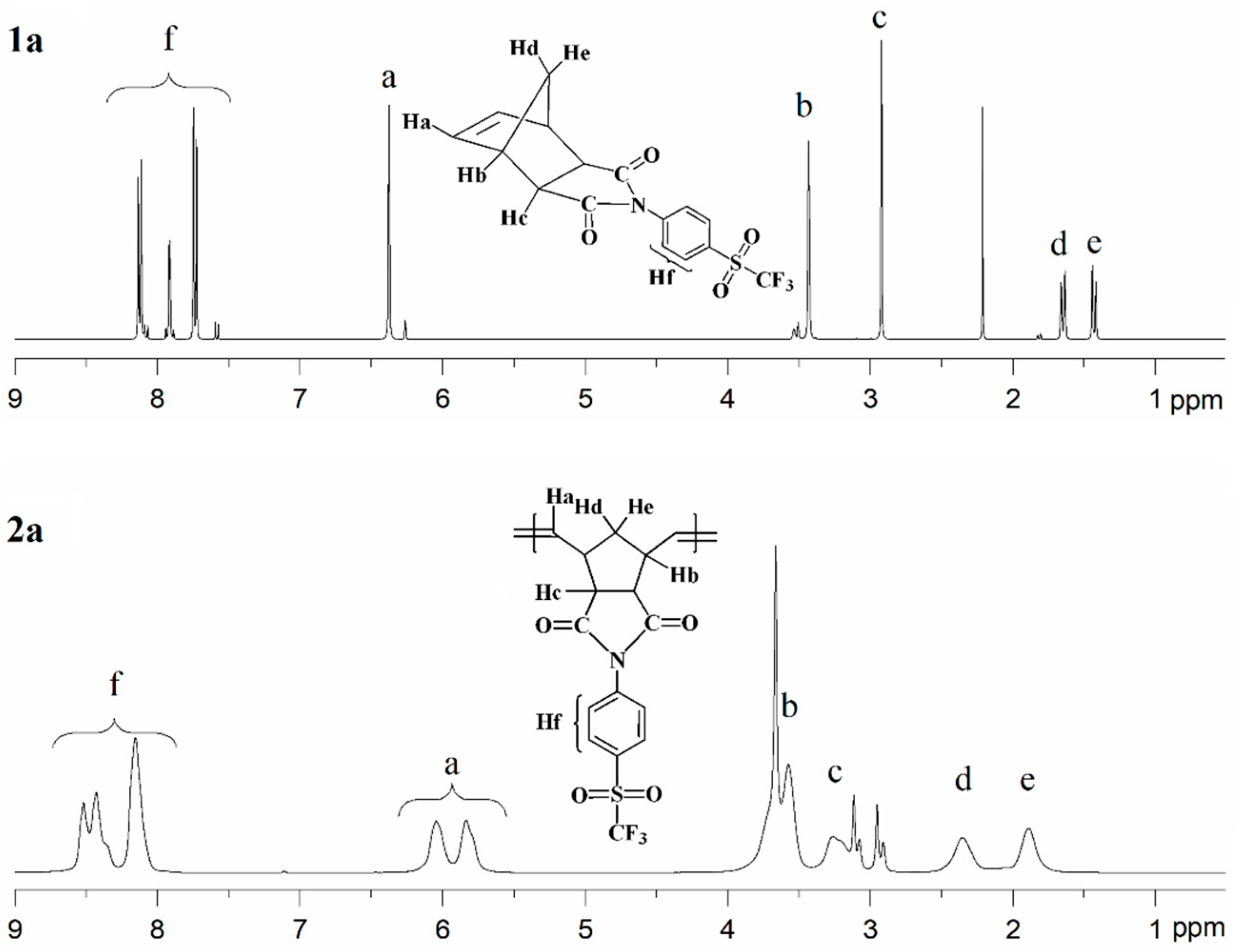
The chemical structures of the monomers and their corresponding polymers were confirmed by 1H, 13C, and 19F-NMR spectroscopy. The 1H-NMR spectra of monomer 1a and its corresponding polymer 2a are presented in Figure 3, while those of monomer 1b and polymer 2b are shown in Figure 4. The 1H-NMR spectrum of monomer 1a (Figure 3, top) exhibits aromatic proton signals (Hf) in the range of 8.06–7.55 ppm. The signal attributed to the olefinic protons (Ha) appears at 6.37 ppm, whereas the resonances assigned to the –CH– protons (Hb and Hc) are observed in the range of 3.59–2.87 ppm. The signals corresponding to the methylene protons (Hd and He) are detected between 1.70 and 1.36 ppm. The 13C-NMR spectrum of monomer 1a exhibits characteristic resonances for the carbonyl (C=O) carbons at approximately 175.9 ppm. Signals corresponding to the C–S carbons are observed at 139.1 ppm, while those attributed to the C=C carbons appear at 138.0 ppm. The aromatic C–H carbons give rise to signals at 132.1 and 127.8 ppm, and the C–N carbons are detected at 130.3 ppm. In addition, the –CF3 carbons display resonances at 124.5, 121.3, 118.0, and 114.8 ppm. The 13C-NMR spectrum of monomer 1a also shows well-resolved aliphatic carbon signals at 47.9 and 46.0 ppm (C–H) and at 43.1 ppm (CH2). The 19F-NMR analysis revealed that the fluorine atoms in the –CF3 group of monomer 1a are magnetically equivalent; accordingly, a single resonance was observed at approximately −78.09 ppm. The 1H-NMR spectrum of polymer 2a is shown in Figure 3 (bottom). The signals assigned to the aromatic protons (Hf) appear in the range of 8.69–7.97 ppm. The olefinic proton signal of the monomer at δ = 6.37 ppm is replaced by two new olefinic proton signals (Ha) at δ = 6.04 ppm and δ = 5.84 ppm, corresponding to the trans and cis double bonds of the polymer main chain, respectively. The resonances attributed to the –CH– protons (Hb and Hc) are observed in the range of 4.05–3.02 ppm, while the signals corresponding to the methylene protons (Hd and He) are detected between 2.36 and 1.88 ppm.
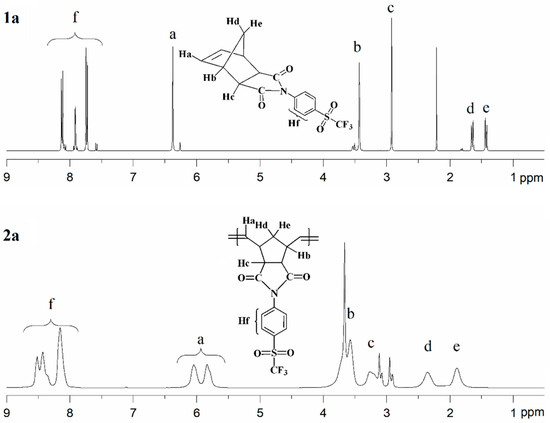
Figure 3.
1H-NMR spectra of monomer 1a (top) and polymer 2a (bottom).
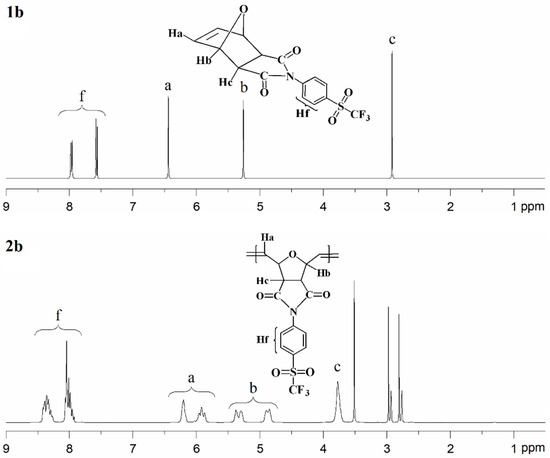
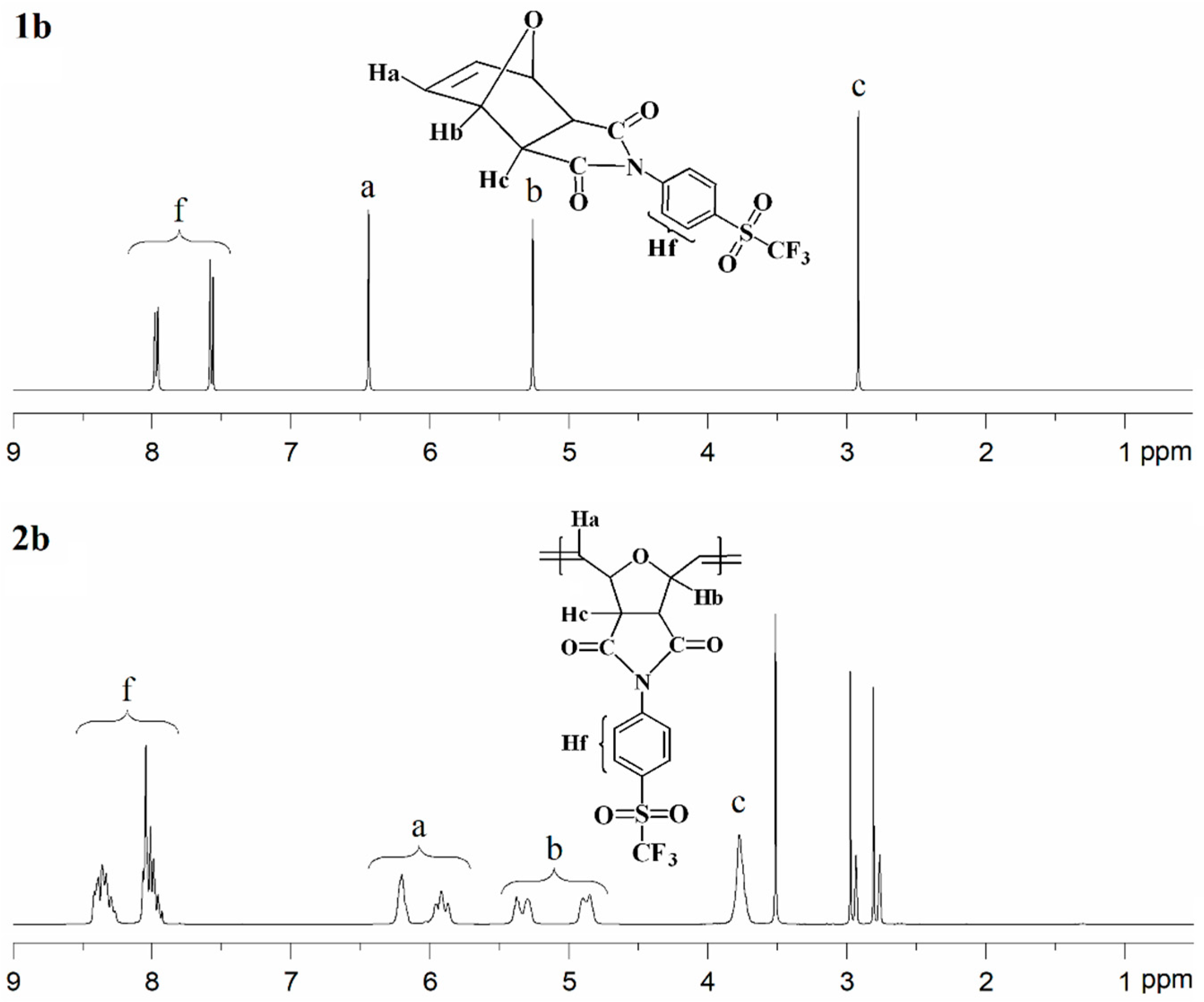
Figure 4.
1H-NMR spectra of monomer 1b (top) and polymer 2b (bottom).
The 1H-NMR spectrum of monomer 1b is shown in Figure 4 (top). The aromatic proton signals (Hf) are observed in the range of 7.88–7.62 ppm. The signal corresponding to the olefinic protons (Ha) appears at 6.44 ppm, while the resonances assigned to the –CH– protons (Hb and Hc) are detected in the range of 5.26–2.91 ppm. The 13C-NMR spectrum of monomer 1b exhibits characteristic resonances for the carbonyl (C=O) carbons at approximately 174.3 ppm. Signals associated with the C–S carbons are observed at 139.0 ppm, while those attributed to the C=C carbons appear at 136.8 ppm. The aromatic C–H carbons give rise to signals at 131.6 and 127.3 ppm, and the C–N carbons are detected at 130.6 ppm. In addition, the –CF3 carbons show resonances at 124.8, 121.3, 118.0, and 115.2 ppm. The 13C-NMR spectrum of monomer 1b also displays well-resolved aliphatic carbon signals at 81.6 and 47.7 ppm, corresponding to C–H carbons. The 19F-NMR analysis indicated that the fluorine atoms in the –CF3 group of monomer 1b are magnetically equivalent; therefore, a single resonance was observed at approximately −78.09 ppm. The 1H-NMR spectrum of polymer 2b is shown in Figure 4 (bottom). The signals attributed to the aromatic protons (Hf) appear in the range of 8.49–7.88 ppm. The olefinic proton signal of the monomer at δ = 6.44 ppm is replaced by two new olefinic proton signals (Ha) at δ = 6.20 ppm and δ = 5.91 ppm, corresponding to the trans and cis double bonds of the polymer main chain, respectively. The resonances assigned to the –CH– protons (Hb) are observed in the range of 5.49–4.74 ppm, while the signal corresponding to the –CH– proton (Hc) is detected at 3.78 ppm.
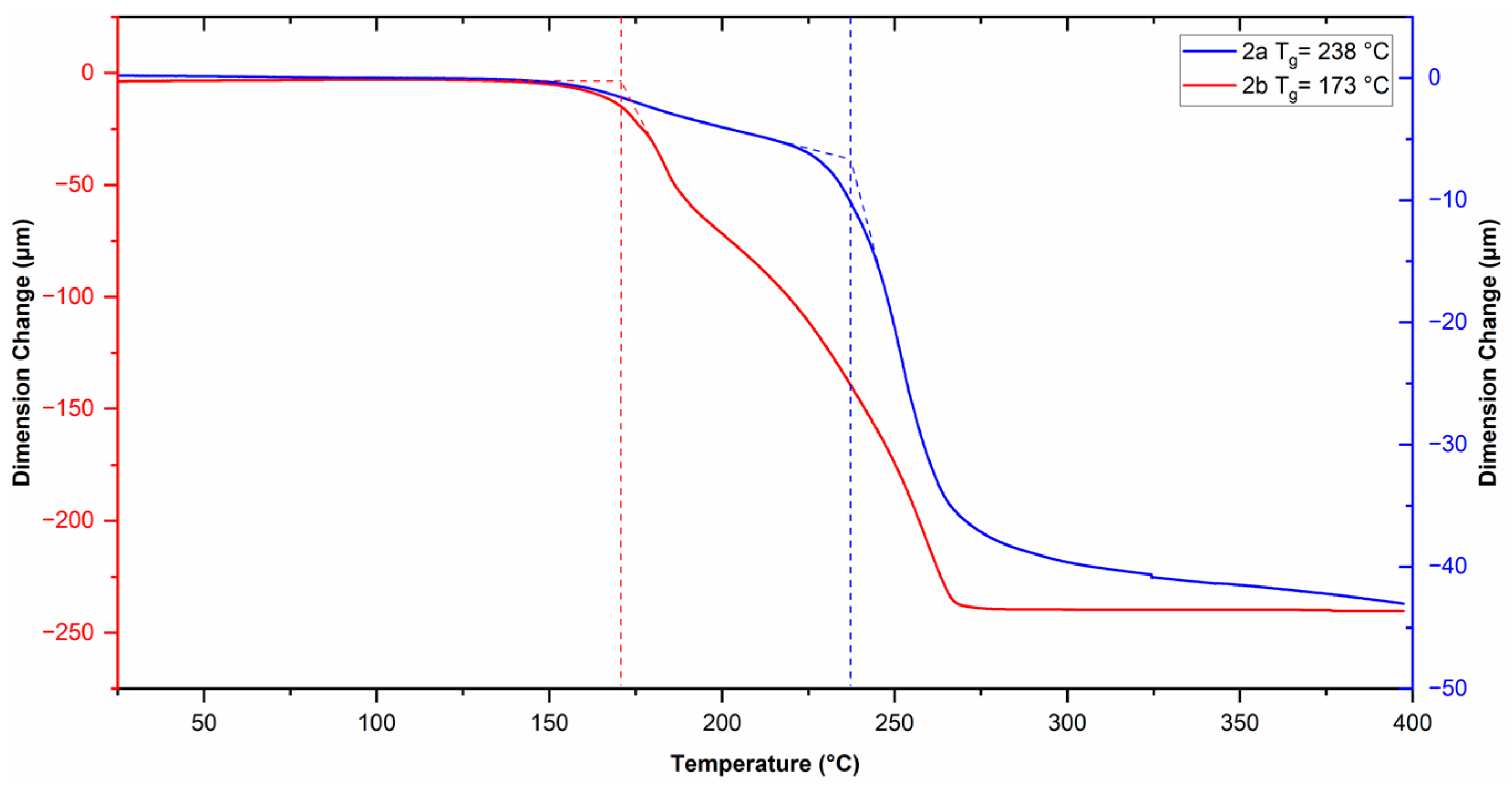
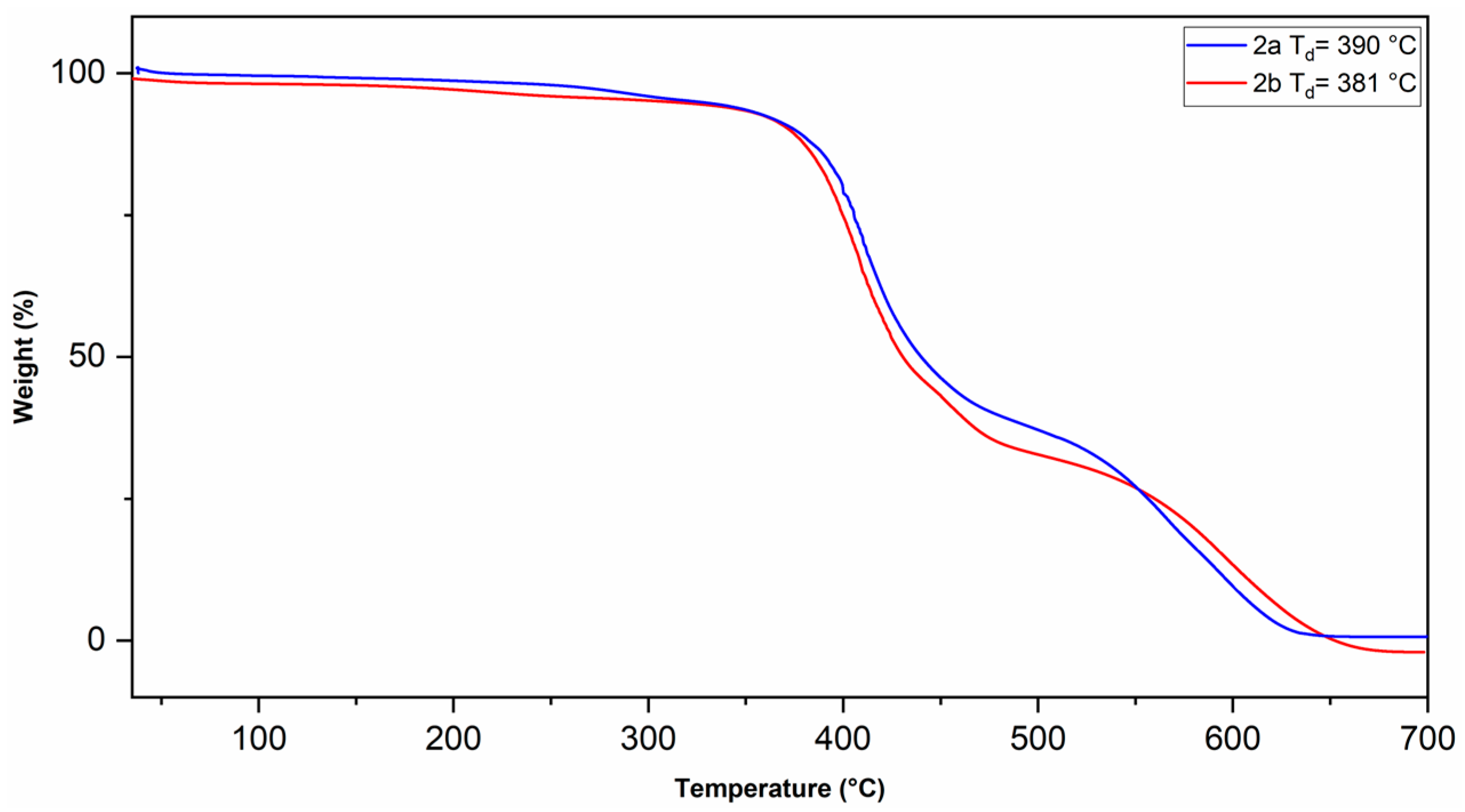
The physical properties of the polymers are summarized in Table 1. The glass transition temperature (Tg) of the polymers was measured by TMA (Figure 5). A Tg value of 238 °C was observed for polymer 2a and 173 °C for polymer 2b. The decrease in the Tg value for polymer 2b compared to that of 2a is ascribed to the presence of the oxygen heteroatom in the cyclopentane ring of the polymer backbone, which increases the conformational mobility of the polymer main chain, thus achieving the relaxation process at a lower temperature. The onset of decomposition temperature (Td) of the polymers was measured by TGA (Figure 6). The thermal profiles shifted 2% relative to each other, starting from the one corresponding to polymer 2a, to clearly follow the thermal decomposition curves of both polymers. A Td value of 390 °C was observed for polymer 2a and 381 °C for polymer 2b, this indicates that these polymers possess relatively high thermal stability. The small difference in the thermal stability of the polymers is attributed to the ether-like bond in polymer 2b, which has a lower bond energy compared to that of the C–H bond [46].

Table 1.
Physical properties of sulfonyl-containing polynorbornene dicarboximides.
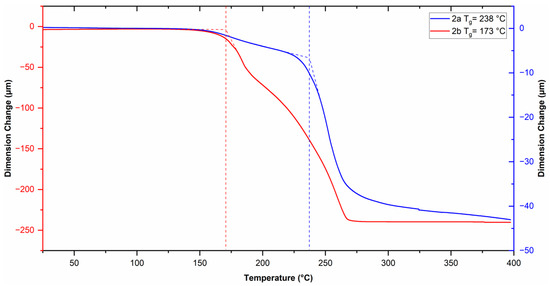
Figure 5.
TMA thermograms of the sulfonyl-containing polynorbornene dicarboximides (Dashed lines indicate Tg values).
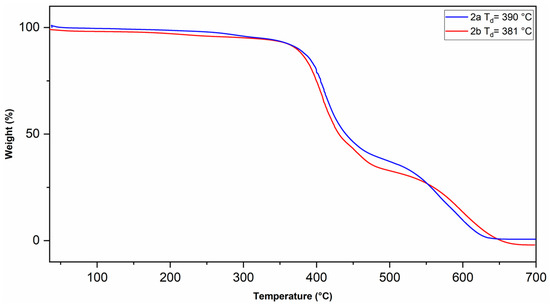
Figure 6.
TGA thermograms of the sulfonyl-containing polynorbornene dicarboximides.
The elastic modulus (E) and the tensile strength (σ) of the polymers were measured by stress–strain tests in tension on the prepared films. According to Table 1, polymer 2a has a higher elastic modulus and tensile strength (E = 2123 MPa and σ = 63.5 MPa) compared to polymer 2b (E = 2078 MPa and σ = 59.3 MPa). This observation indicates that the presence of an oxygen heteroatom in the polymer backbone facilitates chain relaxation by enabling the polymer to adopt different conformations. This is reflected in lower thermomechanical properties compared to the homologous polymer that lacks oxygen heteroatoms.
The density, ρ, of the polymers was measured in film form using the flotation method at room temperature in an ethanol medium. The density values reported in Table 1 indicate that the presence of an oxygen heteroatom in the cyclopentane ring of polymer 2b enhances chain packing efficiency, as reflected by its higher density and lower fractional free volume (FFV) compared to polymer 2a. In contrast, polymer 2a appears to have more restricted backbone mobility, as a result of the presence of the methylene group in the cyclopentane ring of the polymer’s main chain; therefore, this polymer displays the lowest density and the highest FFV among the two polymers examined in this study.
The FFV was determined using the Bondi group contribution method [14] according to the following equation:
where V denotes the specific volume (1/ρ), and V0 represents the specific occupied volume, which can be calculated from the Van der Waals volume Vw as V0 = 1.3 Vw, estimated using van Krevelen’s data [47].
FFV = (V − V0)/V
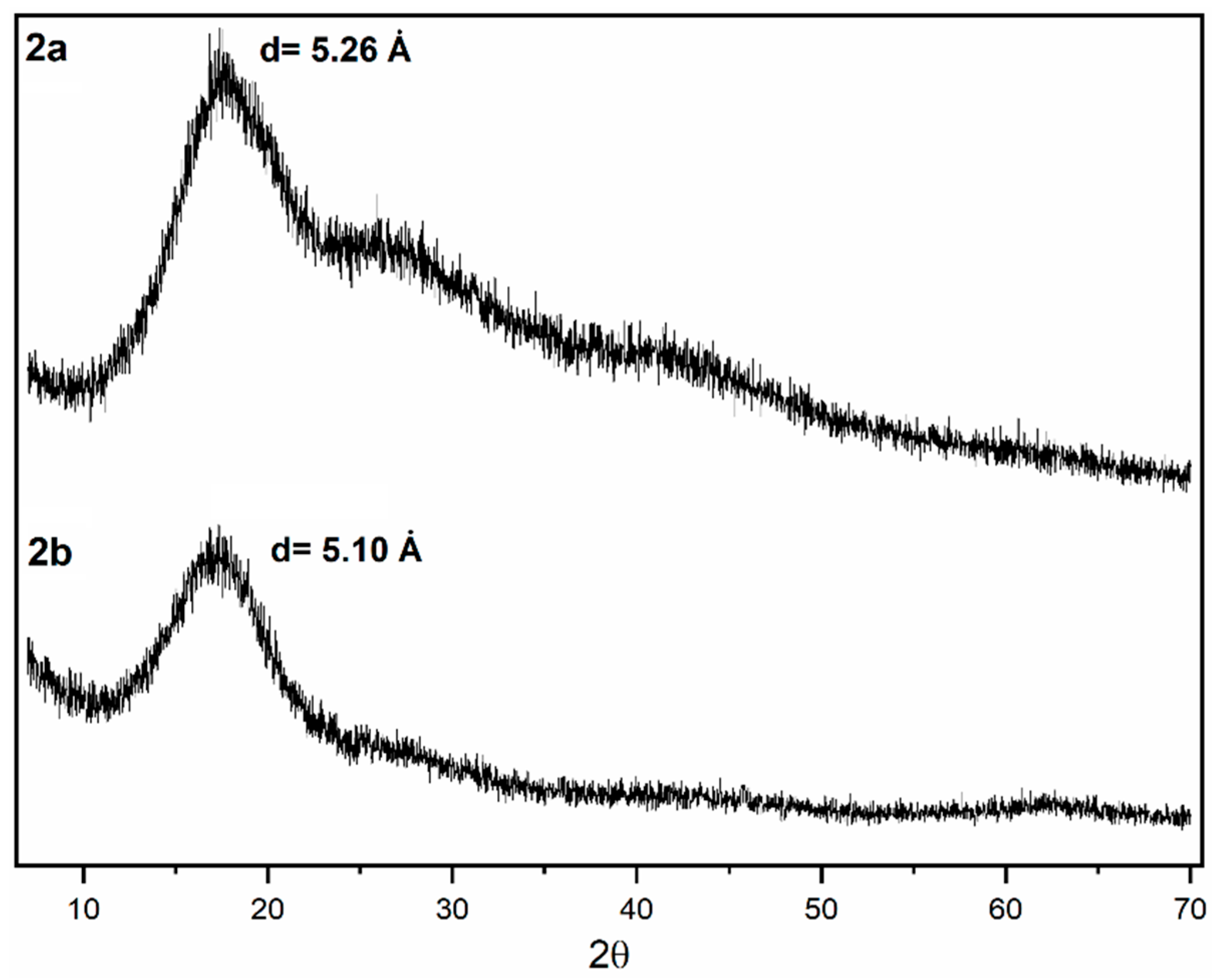
X-ray diffraction (XRD) measurements (Figure 7) performed on sulfonyl-containing polymer films display patterns typical of dense amorphous polymers such as polynorbornene dicarboximides, in which a broad diffraction trace can be clearly identified showing a maximum around 17° (2θ). Two additional diffraction halos can also be observed at approximately 28° and 42° (2θ) and may be attributed to the coexistence of different levels or types of structural order within the material. Each halo corresponds to a distinct characteristic spacing (d-spacing): a low-angle halo is commonly associated with longer-range correlations related to interchain packing, whereas a high-angle halo is associated with short-range distances arising from local segmental interactions. On the one hand, the structural heterogeneity of this class of polymers is likely to result in the appearance of more than one diffraction halo, since polynorbornene dicarboximides contain both an aliphatic, hydrophobic moiety formed by the cyclopentane ring and an aromatic, hydrophilic moiety located in the imide region. Furthermore, the membrane casting process may induce partial orientation of the polymer chains, leading to the separation of halos associated with specific packing directions. Likewise, additional halos may arise from specific correlations, such as the π−π interactions discussed above, which are associated with the charge transfer (CT) interaction formed by π electrons of ring arrangements in polynorbornene dicarboximides. From the XRD patterns, the mean intersegmental distance or chain packing density, d-spacing, of polymers 2a and 2b can be estimated. To accomplish this, the Bragg’s equation is used, nλ = 2dsinθ [48], taking the angle at which the reflective intensity in the amorphous peak is maximum. As shown in Table 1, the average d-spacing and FFV values are well correlated. In other words, a larger d-spacing is expected to result in a higher FFV, and vice versa.
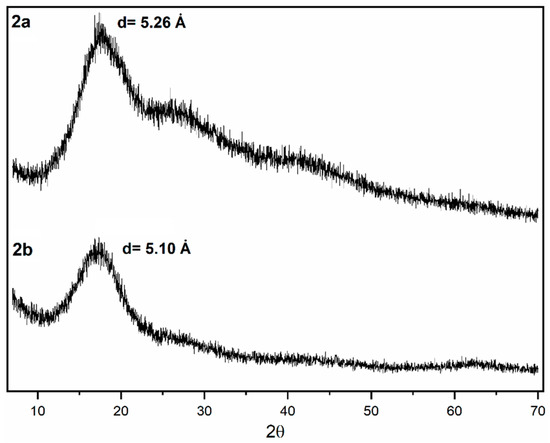
Figure 7.
XRD patterns of the sulfonyl-containing polynorbornene dicarboximides.
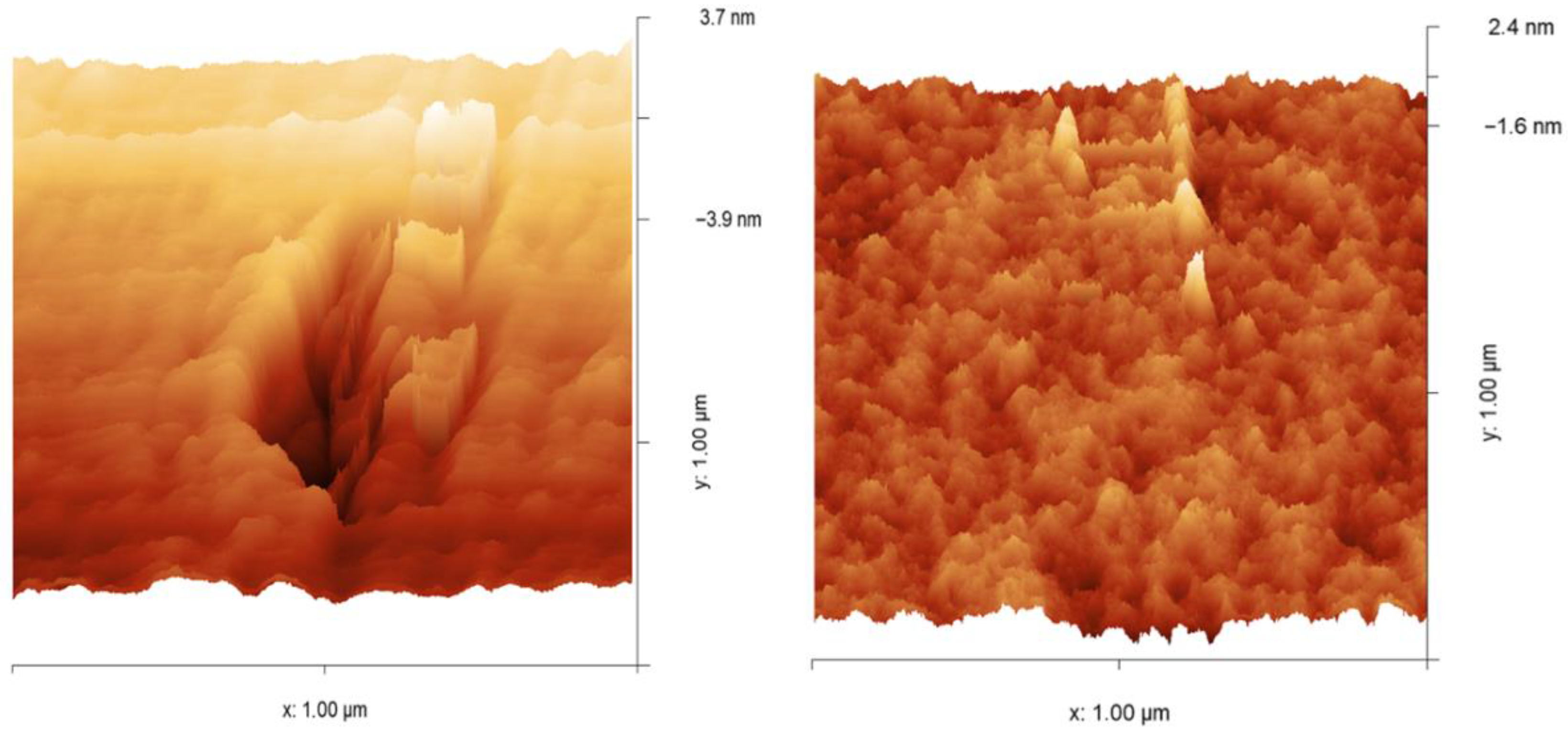
In Figure 8, the surface morphologies of polymer 2a (left) and polymer 2b (right), obtained by the Grubbs 2nd generation catalyst (I), are observed by tapping mode atomic force microscopy in three dimensions. Polymer 2a exhibits an irregular surface morphology characterized by large cavities per unit area, which may be attributed to reduced main-chain mobility arising from steric hindrance of the –CH2– group in the cyclopentane ring of the ROMP-prepared polymer. The latter leads to lower efficiency in the packing of polymer chains, which in turn is reflected in lower density. Polymer 2b shows a disrupted surface morphology that lacks cavities, suggesting that the presence of an oxygen heteroatom in the cyclopentane ring of polymer 2b increases chain mobility, leading to enhanced chain packing ability, which is reflected in a higher density. The absence of cavities also accounts for the lower FFV of this polymer compared to polymer 2a.
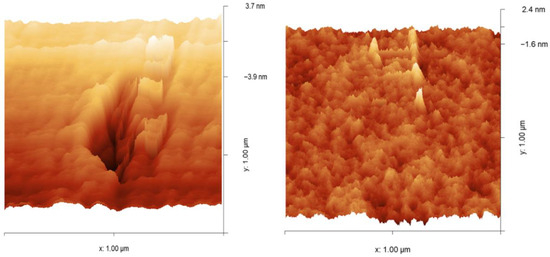
Figure 8.
Three-dimensional AFM micrographs (1 × 1 μm) of polymer 2a (left) and polymer 2b (right).
Table 2 presents the permeability (P), diffusion (D), and solubility (S) values of different gases measured at 30 °C and an upstream pressure of 1 atm for membranes of sulfonyl-containing polynorbornene dicarboximides. In general, the permeability coefficients of sulfonyl-containing polynorbornene dicarboximides are observed to follow the order P(H2) > P(CO2) > P(O2) > P(C2H4) > P(N2) ≥ P(CH4) > P(C3H6). This trend differs from that of the diffusion coefficients, which decrease in the sequence D(H2) > D(O2) > D(N2) ≥ D(CO2) > D(CH4) > D(C2H4) ≥ D(C3H6). Table 2 clearly shows that the presence of the sulfonyl group decreases the gas permeability coefficient of both polymeric membranes studied here as a consequence of the decrease mainly in gas diffusion compared to that of membrane 3c, which corresponds to a polynorbornene dicarboximide that lacks sulfonyl group. The latter suggests that the strong electron-withdrawal exerted by the sulfonyl group leads to greater efficiency in the packing of polymer chains, which in turn is reflected in lower permeabilities, as reveals the fact that the gas permeability coefficients in the sulfonyl-containing 2a membrane have values ranging from 27% (H2) to 65% (C3H6) lower than those of membrane 3c, which lacks sulfonyl group. In addition, it is also observed that the presence of an oxygen heteroatom in the cyclopentane ring of the sulfonyl-containing 2b membrane further decreases gas permeability, showing values between 48% (H2) and 86% (C3H6) lower than those of membrane 3c. On the other hand, when comparing the gas permeability coefficients of membranes 2a and 2b, which contain sulfonyl groups, it is observed that membrane 2b is less permeable. This decrease in gas permeability of membrane 2b is due to both the diffusion process and the gas sorption stage. The lowering in diffusion coefficients is mainly due to a lower FFV associated with the greater chain packing efficiency of polymer 2b compared to polymer 2a, which in turn hinders the diffusion of gas molecules through the polymer. In general, as shown in Table 2, the apparent solubility coefficients for membrane 2b are lower than those found for membrane 2a. It is well known that non-equilibrium excess free volumes are a key feature of glassy polymers, thus, the lower solubility of the gas can be directly attributed to the smaller amount of this additional free volume in which sorption processes can take place. In this regard, Table 1 shows that polymer 2b has a lower FFV than polymer 2a, which correlates quite well with the previous statement. Commonly, the dual-mode model has been employed to interpret gas sorption in glassy polymers. This approach assumes that the glassy state consists of a continuous phase with dispersed microcavities, which explains the excess volume. Solubility in the continuous phase follows Henry’s law, whereas microcavities function as Langmuir sites where processes such as adsorption occur. The sorption at Langmuir sites is often employed to correlate the amount of non-equilibrium excess free volume in the glassy state [49]. According to the dual-mode model, Langmuir sorption in glassy polymer membranes depends on the difference between Tg and T; therefore, as the Tg of the polymer decreases, the nonequilibrium excess free volume also decreases at a given temperature T [50]. Based on the above, the relatively low S values found for membrane 2b used in this research could also be due to the proximity of the glass transition temperature of the sulfonyl-containing polymer to the working temperature. As can be seen from Table 2, carbon dioxide and the hydrocarbon gases of greater condensability exhibit the largest apparent solubility coefficients, S, as determined from the P/D ratio, so that for polymer 2a the following trends are observed: S(C3H6) > S(CO2) > S(C2H4) > S(CH4) > S(O2) > S(N2) > S(H2), while for polymer 2b the trends are: S(CO2) > S(C2H4) > S(CH4) > S(C3H6) > S(O2) ≥ S(N2) > S(H2). These solubility trends suggest that the bulky methylene group in 2a is much more effective for the sorption of C3H6 compared to the oxygen heteroatom in 2b. This fact is related to the strong affinity of olefins for less polar polymer matrices [51]. The large difference in the solubility coefficient of propylene between the two polymers is due both to the greater sorption of this gas in polymer 2a, attributed to polymer-penetrant interactions, and to the greater diffusion of C3H6 in polymer 2b. Further research, including the calculation of solubility activation energy (ES) and diffusion activation energy (ED), is needed to clarify this issue.

Table 2.
Permeability (P), diffusion (D), and solubility (S) coefficient values of several gases measured at 30 °C and an upstream pressure of 1 atm for membranes of sulfonyl-containing polynorbornene dicarboximides.
The effects of the sulfonyl group and the oxygen heteroatom on the performance of polynorbornene dicarboximide membranes were also evaluated using the permselectivity coefficient, or ideal separation factor (α) for gas A over gas B, which serves as an indicator of a polymer membrane’s ability to separate a specific gas mixture and is defined as:
Equation (5), defined as the ratio of the pure-gas permeability coefficients P(A)/P(B), points out that the discriminative effects governing gas transport in membranes may arise from the diffusion stage, the solubility step, or a combination of both. Accordingly, the ideal separation factor can be decomposed into two contributions: a diffusivity selectivity term, αD = D(A)/D(B), and a solubility selectivity term, αS = S(A)/S(B). This approach enables determination of whether αD or αS plays the dominant role in achieving separation of a given gas pair.
Table 3 compares the permselectivities of different gas pairs for membranes 2a and 2b. As shown, both sulfonyl-containing polynorbornene dicarboximide membranes, which exhibit lower gas permeability coefficients, display higher ideal separation factors than membrane 3c, which lacks the sulfonyl group. This behavior reflects the trade-off commonly observed in glassy polymers, whereby lower permeability is accompanied by higher permselectivity. For instance, the permselectivity coefficients α(CO2/CH4) for membranes 2a and 2b increase by approximately 35% and 23%, respectively, at the expense of the CO2 permeability coefficient, which decreases by approximately 27% and 51%, respectively.

Table 3.
Ideal gas separation factors (α) determined at 30 °C for various gas pairs in membranes 2a and 2b.
Interestingly, when the two sulfonyl-containing membranes are compared to each other, it is seen that membrane 2a not only increases the permeability coefficient of O2 by 54%, of N2 by 42%, of CO2 by 51% and of CH4 by 37%, but also augments the permselectivity coefficient α(O2/N2) by almost 7% and α(CO2/CH4) by almost 10%, among others. In addition to exhibiting separation performances for O2/N2, CO2/CH4, and CO2/N2 comparable to those of membrane 2a, membrane 2b also displays the highest permselectivity coefficients for the separation of H2 from low-molecular-weight hydrocarbon gases such as C2H4 and C3H6. In this regard, α(H2/C3H6) is almost double that of membrane 2a and is mainly due to an increase in the solubility selectivity contribution, αS, which in the case of this pair of gases is approximately eight times greater than in membrane 2a.
In this context, it is worth noting that α(H2/C3H6) = 112.4 for membrane 2b represents the highest separation factor reported to date for polynorbornene dicarboximides and the value that most closely approaches this result is only obtained by the CF3-meta substitution of the phenyl group in this type of polymers [52]. Furthermore, it was found that αD = D(H2)/D(C3H6) = 8034 for membrane 2a, since αD ˃˃˃ αS the diffusivity selectivity makes the greatest contribution to achieving the separation of the gas pair. However, the relatively low solubility of hydrogen compared to that of propylene is responsible for the membrane’s moderate discriminatory performance in H2/C3H6 separation. As mentioned above, the contribution of solubility to permselectivity is expressed as αS = S(A)/S(B), where A and B denote the gases to be separated. Examination of the data in Table 3 indicates that solubility is a poor contributor to permselectivity for condensable gases. For example, when propylene is taken as gas A in the most permeable membrane (2a), the αS values are lower than 2 when gas B is ethylene or carbon dioxide. In contrast, the solubility selectivity shows good performance for less condensable gases, with αS values reaching up to 33 and 24 for nitrogen and oxygen, respectively.
4. Conclusions
The synthesis and subsequent ROMP of two new homologous sulfonyl-containing polynorbornene dicarboximide monomers was successfully carried out, yielding materials with high thermal and mechanical properties. The properties of polymers are influenced by the presence of a methylene group or an oxygen heteroatom in the cyclopentane ring of the polymer backbone, primarily due to steric hindrance affecting the main chain’s mobility. The presence of the methylene group increases the thermomechanical properties of the polymer, while its substitution by an oxygen heteroatom has the opposite effect. The gas transport properties of the sulfonyl-containing polynorbornene dicarboximides 2a (with methylene group) and 2b (with oxygen heteroatom) were compared with those of a polynorbornene dicarboximide lacking sulfonyl groups. It is observed that the presence of sulfonyl groups in both polymers decreases the gas permeability of their corresponding membranes as a consequence of the decrease mainly in gas diffusion. Moreover, the presence of the oxygen heteroatom in polymer 2b further decreases gas permeability compared to polymer 2a, generally as a consequence of the decrease in both gas diffusion and gas solubility, attributed mainly to a lower FFV. It was also found that the polymer 2b membrane has one of the highest permselectivity coefficients reported to date for the separation of H2/C3H6 in glassy polynorbornene dicarboximides.
Author Contributions
Conceptualization, A.A.S. and J.V.; data curation, A.O. and L.R.-R.; investigation, A.O., L.R.-R., B.O.M.-M. and M.L.-G.; methodology, A.O., A.A.S. and M.L.-G.; writing—original draft, A.O., A.A.S. and J.V.; writing—review and editing, A.O. and J.V. All authors have read and agreed to the published version of the manuscript.
Funding
Financial support from DGAPA-UNAM PAPIIT through project IN105525 is gratefully acknowledged. We thank SECIHTI for its generous support through the Basic and Frontier Science Project CBF-2025-I-2226. We gratefully acknowledge financial support provided by Ministerio de Ciencia, Innovación y Universidades through the Project PID2019-108552GB-100.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
We are grateful to Gerardo Cedillo Valverde, Karla Eriseth Reyes Morales, and Eliezer Hernández Mecinas for their assistance in the NMR, thermal properties, and mechanical properties, respectively. Financial support from the Secretariat of Science, Humanities, Technology and Innovation (SECIHTI) (PhD Scholarship given to A.O.; number 813793) is gratefully acknowledged.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| 1a | N-4-(trifluoromethylsulfonyl)phenyl-norbornene-5,6-dicarboximide |
| 1b | N-4-(trifluoromethylsulfonyl)phenyl-7-oxanorbornene-5,6-dicarboximide |
| 2a | poly(N-4-(trifluoromethylsulfonyl)phenyl-norbornene-5,6-dicarboximide) |
| 2b | poly(N-4-(trifluoromethylsulfonyl)phenyl-7-oxanorbornene-5,6-dicarboximide) |
| I | Grubbs 2nd generation catalyst |
| NMR | Nuclear magnetic resonance |
| FT-IR | Fourier transform infrared spectroscopy |
| TMA | Thermomechanical analysis |
| TGA | Thermogravimetric analysis |
| AFM | Atomic force microscopy |
| XRD | X-ray diffraction |
| ROMP | Ring-opening metathesis polymerization |
| Tg | Glass transition temperature |
| Td | Onset of decomposition temperature |
| E | Young’s modulus |
| σ | Tensile strength |
| ρ | Density |
| NDA | Norbornene-5,6-dicarboxylic anhydride |
| P | Permeability coefficient |
| D | Diffusion coefficient |
| S | Apparent solubility coefficient |
| FFV | Fractional free volume |
| d-spacing | Mean intersegmental distance |
| α | Permselectivity coefficient |
References
- Arshad, N.; Batool, S.R.; Razzaq, S.; Arshad, M.; Rasheed, A.; Ashraf, M.; Nawab, Y.; Nazeer, M.A. Recent Advancements in Polyurethane-Based Membranes for Gas Separation. Environ. Res. 2024, 252, 118953. [Google Scholar] [CrossRef] [PubMed]
- Stern, S.A. Polymers for Gas Separations: The next Decade. J. Memb. Sci. 1994, 94, 1–65. [Google Scholar] [CrossRef]
- Alkandari, S.H.; Castro-Dominguez, B. Advanced and Sustainable Manufacturing Methods of Polymer-Based Membranes for Gas Separation: A Review. Front. Membr. Sci. Technol. 2024, 3, 1390599. [Google Scholar] [CrossRef]
- Carta, M.; Bernardo, P. Gas Separation by Membrane Operations. In Encyclopedia of Membranes; Springer: Berlin/Heidelberg, Germany, 2015; pp. 855–857. [Google Scholar]
- Saddeeq, A.; Mahmoudi Kouhi, M.; Kammakakam, I. Advances of Ionic-Mediated Polymer Architectures for CO2 Gas Separation Membranes: A Comprehensive Review of Design, Progress, and Future Prospects. ACS Omega 2025, 10, 26266–26292. [Google Scholar] [CrossRef]
- Yampolskii, Y.; Pinnau, I.; Freeman, B. Materials Science of Membranes for Gas and Vapor Separation; Yampolskii, Y., Pinnau, I., Freeman, B., Eds.; Wiley: Hoboken, NJ, USA, 2006; ISBN 9780470853450. [Google Scholar]
- Borah, D.; Hazarika, G.; Gogoi, A.; Goswami, S.; Sawake, S.V.; Yadav, D.; Karki, S.; Gohain, M.B.; Sahu, L.R.; Ingole, P.G. Polymeric Membranes for Sustainable Gas Separation: A Comprehensive Review with Challenges, Innovations and Future Perspectives. Renew. Sustain. Energy Rev. 2025, 219, 115868. [Google Scholar] [CrossRef]
- Yu, S.; Li, C.; Zhao, S.; Chai, M.; Hou, J.; Lin, R. Recent Advances in the Interfacial Engineering of MOF-Based Mixed Matrix Membranes for Gas Separation. Nanoscale 2024, 16, 7716–7733. [Google Scholar] [CrossRef]
- Onchi, A.; Corona-García, C.; Santiago, A.A.; Téllez Arias, M.G.; Alfonso, I.; Vargas, J. Synthesis and Gas Permeation Properties of Functionalized Norbornene-Based Polymers. Curr. Org. Chem. 2024, 28, 1144–1153. [Google Scholar] [CrossRef]
- Khrychikova, A.P.; Medentseva, E.I.; Bermesheva, E.V.; Borisov, I.L.; Wozniak, A.I.; Lezhnin, P.P.; Sadovnikov, K.S.; Ren, X.-K.; Il’in, M.M.; Shantarovich, V.P.; et al. Vinyl-Addition Polynorbornenes Based on Renewable Feedstock: Synthesis and Gas Transport Properties. Sep. Purif. Technol. 2025, 376, 133997. [Google Scholar] [CrossRef]
- Medentseva, E.I.; Khrychikova, A.P.; Bermesheva, E.V.; Borisov, I.L.; Petukhov, D.I.; Karpov, G.O.; Morontsev, A.A.; Nesterova, O.V.; Bermeshev, M.V. CO2-Separation Performance of Vinyl-Addition Polynorbornenes with Ester Functionalities. J. Memb. Sci. 2024, 705, 122916. [Google Scholar] [CrossRef]
- Resendiz-Lara, D.A.; Azhdari, S.; Gojzewski, H.; Gröschel, A.H.; Wurm, F.R. Water-Soluble Polyphosphonate-Based Bottlebrush Copolymers via Aqueous Ring-Opening Metathesis Polymerization. Chem. Sci. 2023, 14, 11273–11282. [Google Scholar] [CrossRef] [PubMed]
- Onchi, A.; Corona-García, C.; Santiago, A.A.; Abatal, M.; Soto, T.E.; Alfonso, I.; Vargas, J. Synthesis and Characterization of Thiol-Functionalized Polynorbornene Dicarboximides for Heavy Metal Adsorption from Aqueous Solution. Polymer 2022, 14, 2344. [Google Scholar] [CrossRef] [PubMed]
- Aranda-Suárez, I.; Corona-García, C.; Santiago, A.A.; López Morales, S.; Abatal, M.; López-González, M.; Vargas, J. Synthesis and Gas Permeability of Chemically Cross-Linked Polynorbornene Dicarboximides Bearing Fluorinated Moieties. Macromol. Chem. Phys. 2019, 220, 1800481. [Google Scholar] [CrossRef]
- Sadovnikov, K.S.; Nazarov, I.V.; Zhigarev, V.A.; Danshina, A.A.; Makarov, I.S.; Bermeshev, M.V. Cross-Linked Metathesis Polynorbornenes Based on Nadimides Bearing Hydrocarbon Substituents: Synthesis and Physicochemical Properties. Polymer 2024, 16, 2671. [Google Scholar] [CrossRef] [PubMed]
- Radwan, M.F.; Abdu, M.E.; Basyouni, M.Z.; Elkady, M.M.; Zohair, M.M.; Shimizu, K.; Spring, A.M. From Monomer Design to Multifunctional Polymers via Controlled ROMP: Novel Indole-Functionalized Norbornene Dicarboximide Copolymers with Enhanced Thermal, Optical, and Antibacterial Properties. Macromolecules 2025, 58, 8007–8031. [Google Scholar] [CrossRef]
- Ruiz, I.; Corona-García, C.; Santiago, A.A.; Abatal, M.; Téllez Arias, M.G.; Alfonso, I.; Vargas, J. Synthesis, Characterization, and Assessment of Novel Sulfonated Polynorbornene Dicarboximides as Adsorbents for the Removal of Heavy Metals from Water. Environ. Sci. Pollut. Res. 2021, 28, 52014–52031. [Google Scholar] [CrossRef]
- Vargas, J.; Santiago, A.A.; Cruz-Morales, J.A.; Tlenkopatchev, M.A.; de Lys, T.; López-González, M.; Riande, E. Gas Transport Properties of Hydrogenated and Fluorinated Polynorbornene Dicarboximides. Macromol. Chem. Phys. 2013, 214, 2607–2615. [Google Scholar] [CrossRef]
- Fang, S.; Su, L.; Sun, D.; Zhang, G. Quaternized Side Chain: A Hydrophilic Strategy for Boosting the Visible-Light Catalytic Hydrogen Evolution of Linear Conjugated Polymers. ACS Appl. Polym. Mater. 2024, 6, 13253–13259. [Google Scholar] [CrossRef]
- Rahman, M.Z.; Wang, X.; Song, L.; Hu, Y. A Novel Sustainable Phosphorus-Containing Heteroatom-Based Coating for Polyamide 6.6 Textiles: Enhanced Flame Retardancy and Hydrophilicity. Ind. Crops Prod. 2023, 205, 117425. [Google Scholar] [CrossRef]
- Sánchez, J.M.; Hidalgo, M.; Valiente, M.; Salvadó, V. New Macroporous Polymers for the Selective Adsorption of Gold (III) and Palladium (II). I. The Synthesis, Characterization, and Effect of Spacers on Metal Adsorption. J. Polym. Sci. Part. A 2000, 38, 269–278. [Google Scholar] [CrossRef]
- Aurangzeb, N.; Zulfiqar, S.; Khosa, M.K.; Slovák, V.; Maamoun, A.A.; Forrester, M.; Sarwar, M.I.; Cochran, E.W. Heteroatom Decorated Polythiourethane Sorbent for Copper (II) Extraction in Wastewater Treatment. Polym. Eng. Sci. 2024, 64, 3109–3119. [Google Scholar] [CrossRef]
- Brirmi, N.E.H.; Chabbah, T.; Chatti, S.; Alimi, K.; Romdhame, H.B.; Mercier, R.; Marestin, C.; Jaffrezic-Renault, N. Progress and Challenges in Heterocyclic Polymers for the Removal of Heavy Metals from Wastewater: A Review. Water Emerg. Contam. Nanoplastics 2024, 3, 23. [Google Scholar] [CrossRef]
- Sowmya, P.; Prakash, S.; Joseph, A. Adsorption of Heavy Metal Ions by Thiophene Containing Mesoporous Polymers: An Experimental and Theoretical Study. J. Solid. State Chem. 2023, 320, 123836. [Google Scholar] [CrossRef]
- Bera, S.; Sau, S.; Banerjee, F.; Kumar, N.; Samanta, S.K. Phosphate-Based, Heteroatom-Rich Porous Organic Polymers for Efficacious Uptake of Iodine in Vapor Phase. Sep. Purif. Technol. 2025, 352, 128123. [Google Scholar] [CrossRef]
- Cetinkaya, A.; Kara, S.S.; Sadak, A.E.; Ayhan, M.M.; Zorlu, Y.; Kahveci, M.U. Multifunctional Benzene-Cored Covalent Organic Polymer via IEDDA Click Chemistry for Iodine Capture and pH-Responsive Drug Delivery. J. Polym. Sci. 2025, 63, 4989–5001. [Google Scholar] [CrossRef]
- Purushothaman, P.; Abesake, K.R.; Santhosh, V.; Karpagam, S. Iodine Adsorption by Thiophene-Based Covalent Organic Polymer: A Study of Structural Influence on Performance. J. Mater. Chem. A Mater. 2025, 13, 32362–32374. [Google Scholar] [CrossRef]
- He, Y.; Feng, W.; Qiao, Y.; Tian, Z.; Tang, B.Z.; Yan, H. Hyperbranched Polyborosiloxanes: Non-traditional Luminescent Polymers with Red Delayed Fluorescence. Angew. Chem. Int. Ed. 2023, 62, e202312571. [Google Scholar] [CrossRef]
- Mariyappan, V.; Gnanasekaran, R. Tuning the Electronic and Structural Properties of Polypyrrole via Heteroatom Doping: Computational Insights for High-Performance Chemical Sensors. Comput. Theor. Chem. 2025, 1252, 115392. [Google Scholar] [CrossRef]
- Zhou, F.; Zheng, S. Heteroatom Engineering of Nonfused Ring Electron Acceptors: Design Strategy for Optoelectronic Enhancement. J. Phys. Chem. A 2025, 129, 9701–9711. [Google Scholar] [CrossRef]
- Santiago, A.A.; Vargas, J.; Gaviño, R.; Cerda, A.M.; Tlenkopatchev, M.A. Synthesis and Ring-Opening Metathesis Polymerization of New Oxanorbornene Dicarboximides with Fluorine Pendant Groups. Macromol. Chem. Phys. 2007, 208, 1085–1092. [Google Scholar] [CrossRef]
- Belov, N.A.; Gringolts, M.L.; Morontsev, A.A.; Starannikova, L.E.; Yampolskii, Y.P.; Finkelstein, E.S. Gas-Transport Properties of Epoxidated Metathesis Polynorbornenes. Polym. Sci. Ser. B 2017, 59, 560–569. [Google Scholar] [CrossRef]
- Morontsev, A.A.; Zhigarev, V.A.; Nikiforov, R.Y.; Belov, N.A.; Gringolts, M.L.; Finkelshtein, E.S.; Yampolskii, Y.P. A New Approach to Improvement of Gas Permeation Properties of Olefin Metathesis Derived Poly(Norbornenes): Gem-Difluorocyclopropanation of Backbone Double Bonds. Eur. Polym. J. 2018, 99, 340–349. [Google Scholar] [CrossRef]
- Abdulabbas, A.A.; Mohammed, T.J.; Al-Hattab, T.A. Parameters Estimation of Fabricated Polysulfone Membrane for CO2/CH4 Separation. Results Eng. 2024, 21, 101929. [Google Scholar] [CrossRef]
- Jasrotia, S.; Puttaiahgowda, Y.M.; Praveen, B.M. Polysulfone-Based Membranes for Industrial CO2 Capture: Advanced Fabrication Strategies, Performance Enhancement, and Commercial Viability—A Review. Process Saf. Environ. Prot. 2025, 203, 107964. [Google Scholar] [CrossRef]
- Sakellaropoulos, G.P.; Kalis, S.P.; Kapantaiakis, G.C.; Dabou, X.S. New Polymer Membranes Prepared from Polysulfone and Polyimide Blends for the Separation of Industrial Gas Mixtures. Patent EP0778077A2, 11 June 1997. [Google Scholar]
- Hamid, M.A.A.; Chung, Y.T.; Rohani, R.; Junaidi, M.U.M. Miscible-Blend Polysulfone/Polyimide Membrane for Hydrogen Purification from Palm Oil Mill Effluent Fermentation. Sep. Purif. Technol. 2019, 209, 598–607. [Google Scholar] [CrossRef]
- Dorosti, F.; Omidkhah, M.R.; Pedram, M.Z.; Moghadam, F. Fabrication and Characterization of Polysulfone/Polyimide–Zeolite Mixed Matrix Membrane for Gas Separation. Chem. Eng. J. 2011, 171, 1469–1476. [Google Scholar] [CrossRef]
- Anbealagan, L.D.; Ng, T.Y.S.; Chew, T.L.; Yeong, Y.F.; Low, S.C.; Ong, Y.T.; Ho, C.-D.; Jawad, Z.A. Modified Zeolite/Polysulfone Mixed Matrix Membrane for Enhanced CO2/CH4 Separation. Membranes 2021, 11, 630. [Google Scholar] [CrossRef]
- Kluge, S.; Kose, T.; Tutuş, M. Tuning the Morphology and Gas Separation Properties of Polysulfone Membranes. Membranes 2022, 12, 654. [Google Scholar] [CrossRef] [PubMed]
- Cetina-Mancilla, E.; Camacho-Zuñiga, C.; González-Díaz, M.O.; Alondra, C.T.; Ruiz-Treviño, A.F.; Vivaldo-Lima, E.; Vera-Graziano, R.; Zolotukhin, M.G.; Sulub-Sulub, R.; Aguilar-Vega, M. Room Temperature Synthesis, Characterization and Enhanced Gas Transport Properties of Novel Poly(Oxindolylidene Arylene)s with Dibenzothiophene, Dibenzothiophene-S-Oxide and Dibenzothiophene-S,S-Dioxide Fragments in the Main Chain. Sep. Purif. Technol. 2024, 341, 126853. [Google Scholar] [CrossRef]
- Constantin, C.-P.; Asandulesa, M.; Varganici, C.; Melinte, V.; Bruma, M.; Jankowski, A.; Wolińska-Grabczyk, A.; Damaceanu, M.-D. Exploring the Potential of Thin Films Made from Poly(Imide-Amide-Sulfone)s for Engineering Applications. Mater. Sci. Eng. B 2021, 270, 115217. [Google Scholar] [CrossRef]
- Xu, Z.; Croft, Z.L.; Guo, D.; Cao, K.; Liu, G. Recent Development of Polyimides: Synthesis, Processing, and Application in Gas Separation. J. Polym. Sci. 2021, 59, 943–962. [Google Scholar] [CrossRef]
- Leszczynski, P., Jr.; Lashkari, S.; Kruczek, B. Revisiting the Effect of the Resistance to Gas Accumulation in Constant Volume Systems on the Membrane Time Lag. Membranes 2024, 14, 167. [Google Scholar] [CrossRef] [PubMed]
- Kanehashi, S.; Sato, S.; Nagai, K. Membrane Color and Gas Permeability of 6FDA-TeMPD Polyimide Membranes Prepared with Various Membrane Preparation Protocols. Polym. Eng. Sci. 2011, 51, 2360–2369. [Google Scholar] [CrossRef]
- Blanksby, S.J.; Ellison, G.B. Bond Dissociation Energies of Organic Molecules. Acc. Chem. Res. 2003, 36, 255–263. [Google Scholar] [CrossRef]
- Van Krevelen, D.W.; Te Nijenhuis, K. Volumetric Properties. In Properties of Polymers; Elsevier: Amsterdam, The Netherlands, 2009; pp. 71–108. [Google Scholar]
- Bragg, W.H.; Bragg Apr, W.L.; H Bragg, B.W. The Reflection of X-Rays by Crystals. Proc. R. Soc. London. Ser. A Contain. Pap. A Math. Phys. Character 1913, 88, 428–438. [Google Scholar] [CrossRef]
- Eguchi, H.; Kim, D.J.; Koros, W.J. Chemically Cross-Linkable Polyimide Membranes for Improved Transport Plasticization Resistance for Natural Gas Separation. Polymer 2015, 58, 121–129. [Google Scholar] [CrossRef]
- Kanehashi, S.; Nagai, K. Analysis of Dual-Mode Model Parameters for Gas Sorption in Glassy Polymers. J. Memb. Sci. 2005, 253, 117–138. [Google Scholar] [CrossRef]
- Sunderrajan, S. Propane and Propylene Sorption in Solid Polymer Electrolytes Based on Poly(Ethylene Oxide) and Silver Salts. J. Memb. Sci. 2001, 182, 1–12. [Google Scholar] [CrossRef]
- Cruz-Morales, J.A.; Vargas, J.; Santiago, A.A.; Vásquez-García, S.R.; Tlenkopatchev, M.A.; de Lys, T.; López-González, M. Synthesis and Gas Transport Properties of New Polynorbornene Dicarboximides Bearing Trifluoromethyl Isomer Moieties. High. Perform. Polym. 2016, 28, 1246–1262. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.