Abstract
Background: The Memorial Sloan–Kettering Cancer Center (MSKCC) prognostic model has been widely used for the prediction of the outcome of metastatic renal cell carcinoma (mRCC) patients treated with systemic therapies, however, data from large studies are limited. This study aimed at the evaluation of the impact of the MSKCC score on the outcomes in mRCC patients treated with first-line sunitinib, with a focus on the intermediate-risk group. Methods: Clinical data from 2390 mRCC patients were analysed retrospectively. Progression-free survival (PFS), overall survival (OS), and objective response rate (ORR) were analysed according to the MSKCC risk score. Results: ORR, median PFS, and OS for patients with one risk factor were 26.7%, 10.1, and 28.2 months versus 18.7%, 6.2, and 16.2 months, respectively, for those with two risk factors (ORR: p = 0.001, PFS: p < 0.001, OS: p < 0.001). ORR, median PFS, and OS were 33.0%, 17.0, and 44.7 months versus 24.1%, 9.0, and 24.1 months versus 13.4%, 4.5, and 9.5 months in the favourable-, intermediate-, and poor-risk groups, respectively (ORR: p < 0.001, PFS: p < 0.001, OS: p < 0.001). Conclusions: The results of the present retrospective study demonstrate the suitability of the MSKCC model in mRCC patients treated with first-line sunitinib and suggest different outcomes between patients with one or two risk factors.
1. Background
Renal cell carcinoma (RCC) represents a common malignancy, with incidence increasing in developed countries [1,2]. The management of metastatic RCC (mRCC) has been markedly changing in recent years with the introduction of targeted therapies and immune checkpoint inhibitors, leading to significant improvements in patient survival. Several prognostic models have been proposed for the prediction of outcome of mRCC patients treated with systemic therapies. The Memorial Sloan–Kettering Cancer Center (MSKCC) prognostic model developed by Motzer et al. has been widely used in clinical trials as well as in the common clinical practice. In this model, patients are classified based on the number of risk factors into three prognostic groups, that is, favourable-, intermediate-, and poor-risk [3].
Antiangiogenic tyrosine kinase inhibitors, such as sunitinib and pazopanib, had been shown to be safe and effective in MSKCC favourable- or intermediate-risk patients, and were established as a standard of care in the first-line systemic therapy [4,5]. Despite its worldwide use, there has been limited number of large studies comparing efficacy with regard to the MSKCC score in mRCC patients treated with targeted agents outside of clinical trials. About 50–60% of all mRCC patients are classified as MSKCC intermediate-risk. This group comprises a heterogeneous population of patients with regard to the type and number of risk factors.
In the present retrospective registry-based study, we analysed the impact of MSKCC classification on the outcomes in a large cohort of mRCC patients treated with first-line sunitinib in the real-life clinical practice, with a focus on the intermediate-risk group.
2. Materials and Methods
2.1. Study Design and Treatment
The present study is a retrospective registry-based analysis of adult mRCC patients treated with first-line sunitinib between May 2006 and January 2018. Patients who had received chemotherapy or cytokines prior to sunitinib were not included. Progression-free survival (PFS), overall survival (OS), and objective response rate (ORR) were analysed in the entire cohort and based on the number of risk factors in the MSKCC intermediate-risk group. Sunitinib (Sutent; Pfizer Inc, New York, USA) was administered orally at the standard approved dosing until disease progression, unacceptable toxicity, or patient refusal. Temporary discontinuation or dose reductions for toxicity followed clinical practice guidelines. Subsequent anticancer therapy after progression was at the discretion of the treating physicians.
2.2. Data Source
The data were obtained from the renal cell carcinoma information system (RENIS) registry that includes data on approximately 95% of mRCC patients treated with targeted therapy in the Czech Republic [6]. The RENIS registry, initiated in 2007, provides retrospective anonymised data on patient baseline clinical characteristics as well as on previous therapies for mRCC, laboratory parameters, treatment course and outcomes, and toxicity that are updated twice a year (http://renis.registry.cz). The RENIS registry and the use of registry data for analysis were approved on 15 May 2013 by the Multicentre Ethics Committee of the Masaryk Memorial Cancer Institute in Brno, Czech Republic. The patients signed informed consent with the inclusion of their data in the registry.
2.3. Outcome Assessment
The clinical status of the patients was assessed continuously during the course of treatment. Physical examination and routine laboratory tests were performed at least every six weeks, and computed tomography (CT) was performed every three to four months during the treatment. The objective tumour response was assessed locally by the attending physician using Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 in terms of: complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD); ORR included patients achieving CR and PR [7].
2.4. Statistical Analysis
Data were described by absolute and relative frequencies for categorical variables and median for quantitative variables. Statistical differences in ORR were calculated using Pearson’s chi-square test; for >2 groups (i.e., good-, intermediate-, and poor-risk), Bonferroni correction for multinomial testing was performed so as to adjust p-values. OS was defined as the time from sunitinib treatment initiation to the date of death due to any cause. PFS was defined as the time from sunitinib treatment initiation to the date of first documented progression or death of any cause. Survival analysis for OS and PFS was conducted using the Kaplan–Meier method complemented by the 95% confidence interval (95% CI) for estimates of probability survival. Statistical significance of differences in survival among subgroups was assessed using the log-rank test. Univariable Cox proportional hazards models were used to evaluate the effect of individual risk factors on the survival measures. Hazard ratios (HRs) were calculated with 95% confidence interval and the statistical significance of hazard ratios was assessed by means of the Wald test. All statistical tests were performed at a significance level of α = 0.05 (two groups) and α = 0.025 (three groups Bonferroni correction) (all tests two-sided). Analysis was performed in the SPSS software (IBM Corp. Released 2016. IBM SPSS Statistics for Windows, Version 24.0.0.1 Armonk, NY: IBM Corp.) and software R version 3.6.0 (www.r-project.org).
3. Results
3.1. Baseline Characteristics
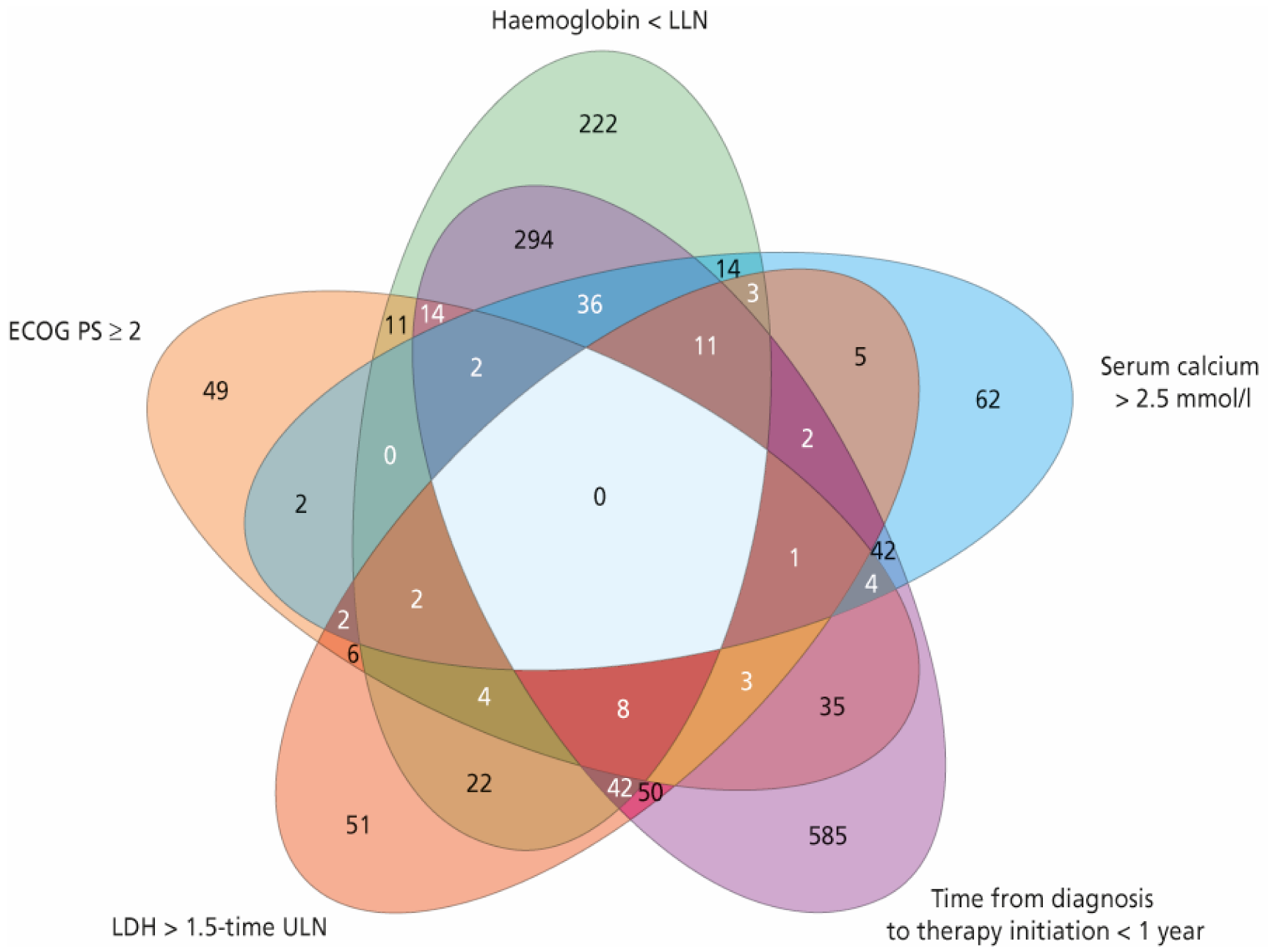
A total of 2390 patients with mRCC were treated with first-line sunitinib, including 806, 1450, and 134 patients in the favourable, intermediate, and poor MSKCC risk group, respectively. In the intermediate-risk group, 969 patients had one risk factor and 481 patients had two risk factors. Patient characteristics, including a detailed distribution of MSKCC score within the respective MSKCC risk group, are summarised in Table 1. A frequency diagram of each combination of risk factors for the MSKCC score is shown in Figure 1.

Table 1.
Patient characteristics based on MSKCC risk groups and details on MSKCC risk factors.
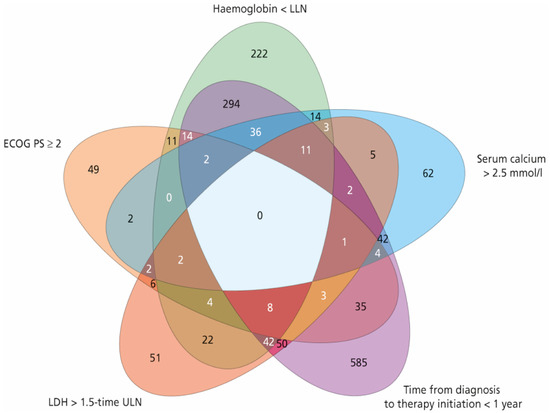
Figure 1.
Frequency diagram of each combination of risk factors for the Memorial Sloan–Kettering Cancer Center (MSKCC) score. ECOG PS = Eastern Cooperative Oncology Group performance status, LLN = lower limit of normal, ULN = upper limit of normal, LDH = lactate dehydrogenase.
3.2. Treatment Outcomes
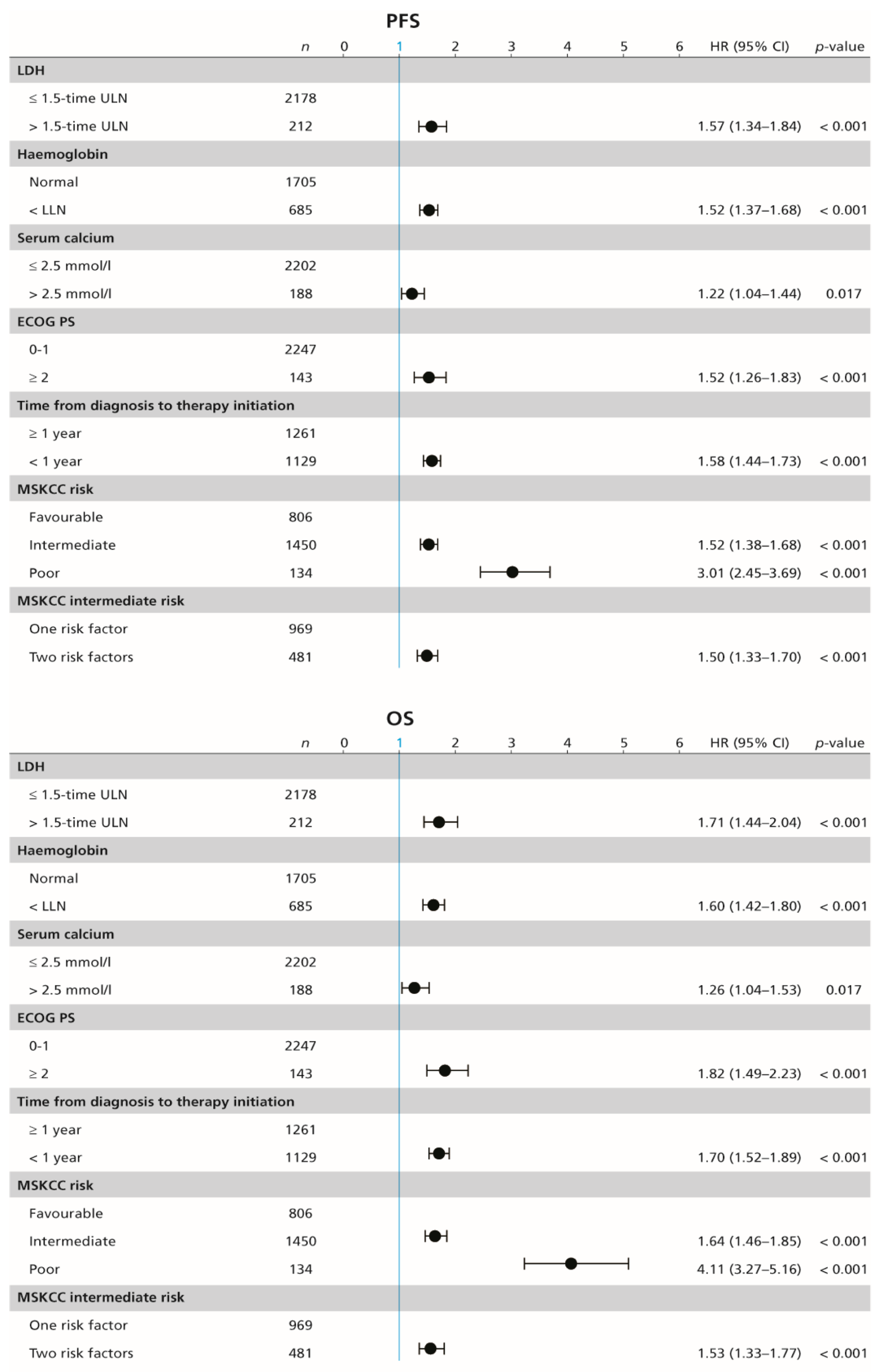
In the whole cohort, median PFS and OS were 10.6 (95% CI 9.9–11.5) months and 28.5 (95% CI 26.3–30.5) months, respectively, with ORR of 26.5%. Each of the individual MSKCC risk factors show significant association with PFS and OS in the univariable Cox proportional-hazards regression; that is, high serum lactate dehydrogenase (LDH) (HR 1.57; 95% CI 1.34–1.84; p < 0.001, and 1.71; 95% CI 1.44–2.04; p < 0.001, respectively), haemoglobin concentration below the lower limit of normal (HR 1.52; 95% CI 1.37–1.68; p < 0.001, and 1.60; 95% CI 1.42–1.80; p < 0.001, respectively), serum calcium concentration above the upper limit of normal (HR 1.22; 95% CI 1.04–1.44; p = 0.017, and 1.26; 95% CI 1.04–1.53; p = 0.017, respectively), ECOG PS ≥ 2 (HR 1.52; 95% CI 1.26–1.83; p < 0.001, and 1.82; 95% CI 1.49–2.23; p < 0.001, respectively), and time from diagnosis to the initiation of systemic therapy of less than one year (HR 1.58; 95% CI 1.44–1.73; p < 0.001, and 1.70; 95% CI 1.52–1.89; p < 0.001, respectively; Figure 2).
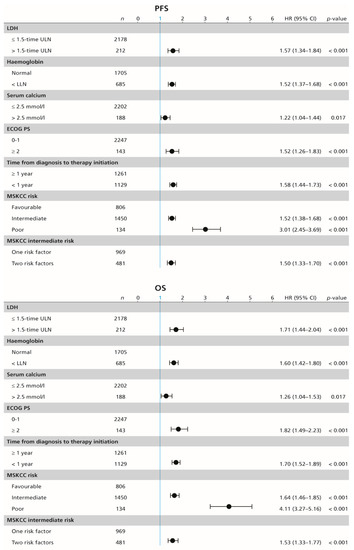
Figure 2.
Forest plot (univariable Cox proportional-hazard regression model) showing the association between survival and MSKCC risk factors. MSKCC = Memorial Sloan–Kettering Cancer Center (MSKCC) score, PFS = progression-free survival, OS = overall survival, n = number of included patients, ECOG PS = Eastern Cooperative Oncology Group performance status, ULN = upper limit of normal, LDH = lactate dehydrogenase, HR = hazard ratio, CI = confidence interval.
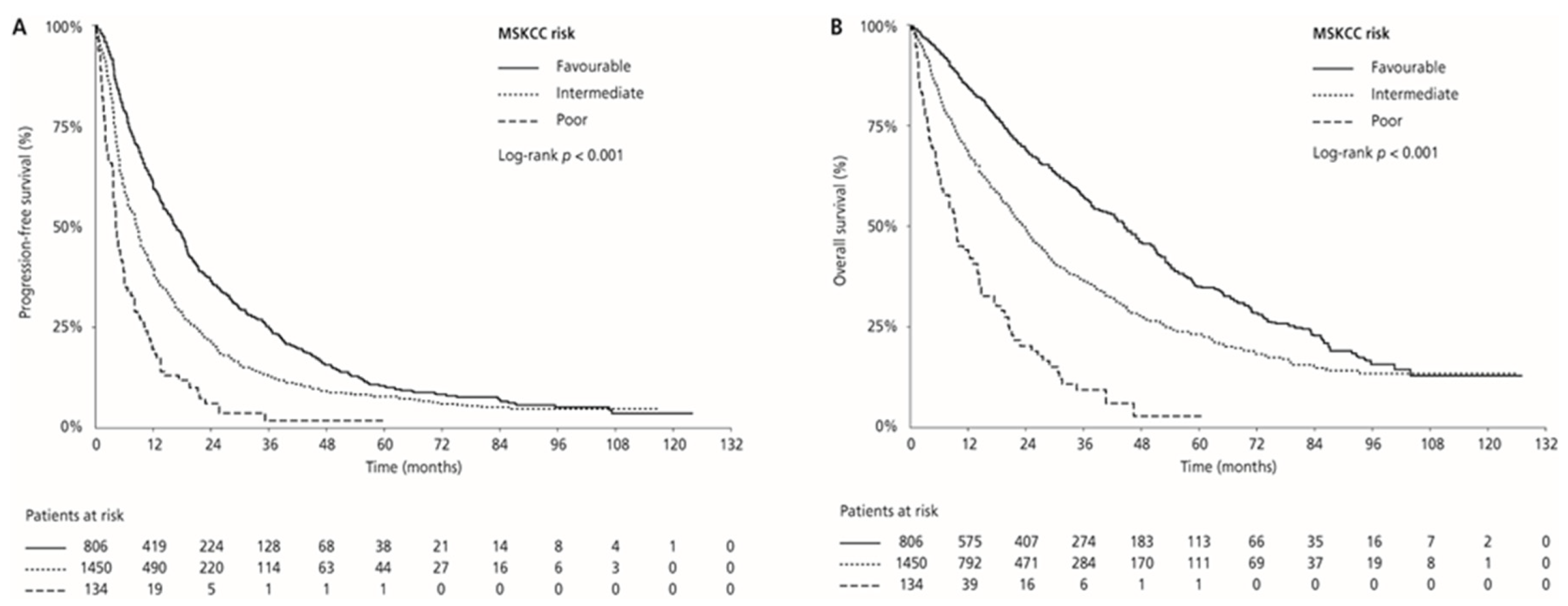
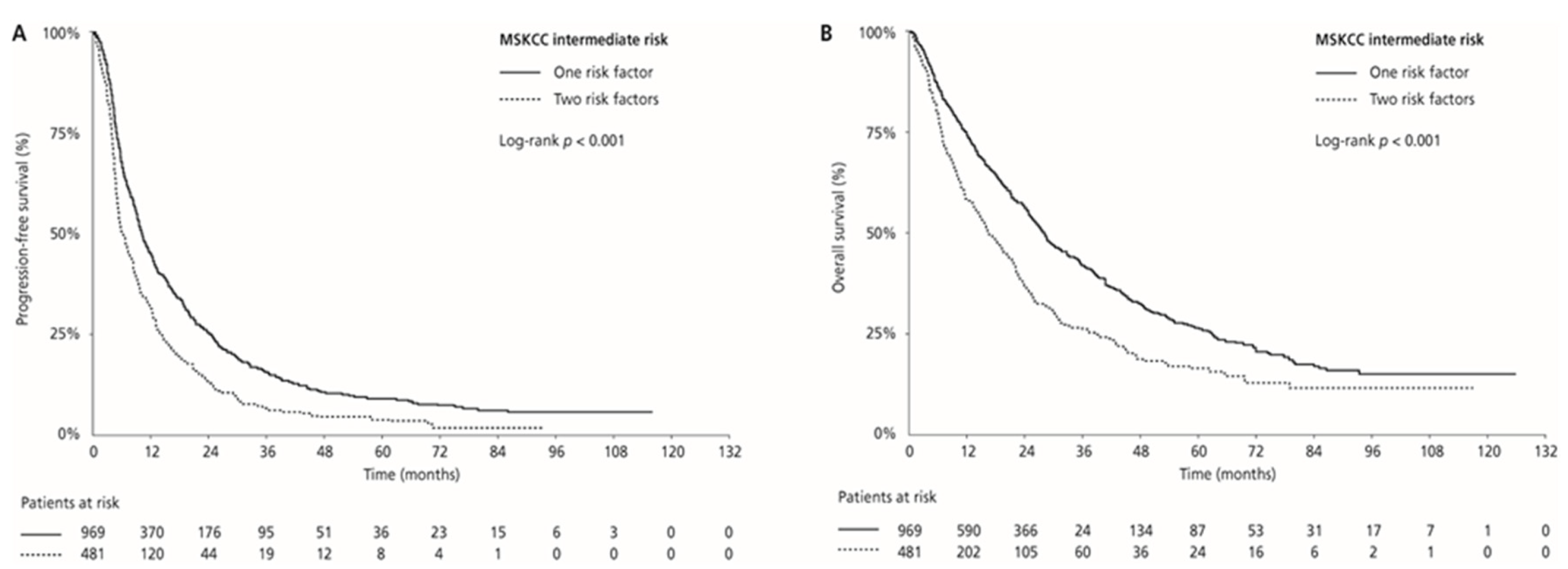
Median PFS and OS were 17.0 (95% CI 15.4–18.8) and 44.7 (95% CI 40.9–50.5) months in the MSKCC favourable-risk group, 9.0 (95% CI 8.3–9.5) and 24.1 (95% CI 21.9–26.0) months in the MSKCC intermediate-risk group, and 4.5 (95% CI 3.9–6.1) and 9.5 (95% CI 7.2–14.1) months in the MSKCC poor-risk group (PFS: p < 0.001 and OS: p < 0.001, respectively), respectively. ORR was 33.0% in the favourable-risk group, 24.1% in the intermediate-risk group, and 13.4% in the poor-risk group (p < 0.001) (Table 2, Figure 3). Median PFS and OS for patients with one risk factor were 10.1 (95% CI 9.4–11.4) months and 28.2 (95% CI 25.9–30.7) months compared with 6.2 (95% CI 5.5–7.5) months and 16.2 (95% CI 14.5–20.2) months in patients with two risk factors (p < 0.001 and p < 0.001, respectively). ORR was 26.7% for patients with one risk factor compared with 18.7% for those with two risk factors (p = 0.001; Table 2; Figure 4).

Table 2.
Overall response rate (ORR), overall survival (OS), and progression-free survival (PFS) results for sunitinib treatment according to MSKCC risk groups.
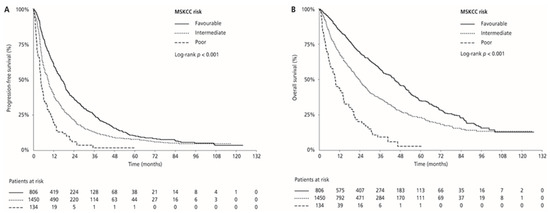
Figure 3.
Kaplan–Meier estimates of progression-free survival (PFS) (A) and overall survival (OS) (B) according to the MSKCC risk category.
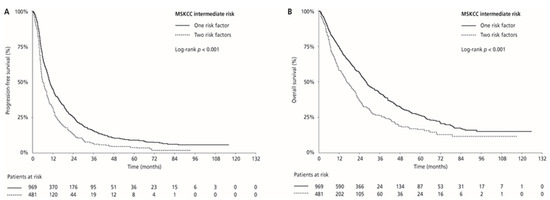
Figure 4.
Kaplan–Meier estimates of progression-free survival (PFS) (A) and overall survival (OS) (B) according to the number of risk factors in the MSKCC intermediate-risk group.
4. Discussion
The results of the present retrospective, registry-based analysis confirm the relevance of the MSKCC prognostic model in a large cohort of mRCC patients treated with first-line sunitinib in real-life clinical practice. Moreover, within the intermediate-risk group, present data suggest a significantly higher ORR as well as longer PFS and OS for patients with one risk factor as compared with patients with two risk factors.
The MSKCC prognostic model is based on five independent risk factors, including low Karnofsky performance status (KPS), high serum LDH, low haemoglobin level, high corrected serum calcium level, and time from diagnosis to the initiation of systemic therapy of less than one year [3]. The favourable-risk group includes patients with no risk factor, the intermediate-risk group comprises patients with one or two risk factors, and the poor-risk group includes patients with three and more risk factors [3]. The MSKCC model had been proposed and validated in mRCC patients treated with cytokines before the advent of targeted agents [8,9]. In addition to the MSKCC prognostic model, the International Metastatic Renal Cell Carcinoma Database Consortium (IMDC) prognostic model that was subsequently proposed and validated by Heng et al. represents another commonly used tool for the prognostic stratification [10]. In contrast to the MSKCC model, IMDC is based on data of mRCC patients treated with targeted therapies and encompasses six risk factors, that is, low KPS, low haemoglobin level, high platelet count, high neutrophil count, high corrected serum calcium level, and time from diagnosis to the initiation of systemic therapy of less than one year [10]. The MSKCC and IMDC models are both widely used to determine the patient risk group in daily clinical practice as well as in the design and interpretation of clinical trials. The MSKCC model has been prospectively used in the principal clinical trials that introduced targeted agents including sunitinib, pazopanib, bevacizumab, and axitinib [5,11,12,13,14], while the IMDC model has been used in the more recent clinical trials evaluating first-line targeted agents including cabozantinib, or combinations with immune checkpoint inhibitors [15,16,17,18,19]. Both MSKCC and IMDC models are highly concordant with the vast majority of patients being classified into the same risk group. Retrospective studies have validated the feasibility and indicated similar outcomes of patients treated with sunitinib within the stratified prognostic groups [20,21]. The present results showing markedly different outcomes for the favourable-, intermediate-, and poor-risk groups confirmed the feasibility of the MSKCC prognostic model in mRCC patients treated with sunitinib in the real clinical practice, in agreement with prior reports [20,21,22,23]. In the present study, each of the individual MSKCC risk factors had significant association with PFS and OS in the univariable Cox proportional-hazards regression. Among the MSKCC prognostic groups, the intermediate-risk group is numerically the largest, comprising more than one half of all mRCC patients. In the present national cohort, 60.7% of patients were classified as intermediate risk, similarly to previously reported data [20,21,22,23,24,25,26]. The intermediate-risk group is rather heterogeneous because it includes patients with one or two risk factors of different weight, and it had to be determined whether further stratification would improve prognostic performance. Significantly longer PFS (10.7 vs. 6.5 months; p < 0.001) and OS (26.3 vs. 14.1 months; p < 0.001) for patients with one risk factor compared with patients with two risk factors was reported by Sella et al. in a retrospective analysis of pooled data from six sunitinib clinical trials of 1059 mRCC patients, including 548 MSKCC intermediate-risk patients [27]. Similar results were obtained in the same cohort with the IMDC prognostic model [27]. A difference in prognosis within the MSKCC intermediate-risk group according to the number of positive risk factors has been also suggested by Tamada et al. and Miyazaki et al. in smaller retrospective studies based on the data of 234 and 217 mRCC patients treated with targeted agents, respectively [28,29]. However, these two studies included a limited number of patients, particularly patients in the intermediate-risk group, and patients were treated in different lines and with different targeted agents, introducing heterogeneity as well as a possible bias. The present results are in agreement with the above mentioned reports. A major strength of the present study is the large cohort of patients treated with first-line sunitinib in conditions of the real-life clinical practice. To the best of our knowledge, this is the largest retrospective study reported so far investigating prognostic stratification based on the MSKCC model. Thus, the present results could be used as a benchmark data for the future analyses. Principal limitations include a retrospective design introducing possible selection bias. In particular, current reimbursement criteria in the Czech Republic result in the exclusion of patients with unfavourable prognosis. Another limitation is that only the MSKCC prognostic model was used as the data for the calculation of the IMDC risk score were not available in all patients. Moreover, the MSKCC criteria were slightly modified and total calcium rather than corrected calcium concentration and ECOG PS rather than KPS were used in the present analysis, although these modifications could hardly affect the results.
Biomarkers play a central role in the management of cancer patients [30]. Both MSKCC and IMDC scores are composite prognostic biomarkers combining laboratory and clinical parameters. The systemic treatment during the past than 10 years and more has been dominated by agents targeting the vascular endothelial growth factor (VEGF) pathway. Combinations of anti-VEGF drugs with other targeted agents did not result in increased efficacy [31], and sequential administration of single agents has been established as the principal treatment strategy. With the advent of immunotherapy based on immune checkpoint blockade, the paradigm of sequential therapy has shifted toward combination regimens with immune checkpoint inhibitors [16,17,18]. Although the MSKCC model has been established in the cytokine therapy era and several studies suggested superiority of the IMDC model in patients treated with targeted agents [10,21], the present data indicate, in agreement with other reports, that the MSKCC risk score is still suitable in the targeted therapy era [27,28,29]. However, with the advent of immunotherapy in the first line mRCC treatment, the use of an optimal prognostic model along with the role of prognostic scores will need to be redefined.
5. Conclusions
The results of the present retrospective study demonstrate the suitability of the MSKCC model for the prognostic stratification of mRCC patients treated with first-line sunitinib. In addition, within the intermediate-risk group, significantly different outcomes are evident between patients with one or two risk factors. These findings suggest that the number of risk factors should be taken into account during the design of future clinical trials as well as in the data interpretation.
Author Contributions
Conceptualization, O.F. and J.F.; Methodology, O.F. and L.C.; Software, L.C.; Formal Analysis, L.C.; Investigation, O.F., J.F., A.P., B.M., J.K., M.Z., K.K., T.M., T.D. and T.B.; Data Curation, L.C.; Writing Original Draft Preparation, O.F.; Writing Review & Editing, B.M. and T.B.; Visualization, T.M. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the National Sustainability Program I (NPU I) Nr. LO1503 provided by the Ministry of Education Youth and Sports of the Czech Republic and by the Charles University Research Fund (Progres Q39), by the European Regional Development Fund-Project ”Application of Modern Technologies in Medicine and Industry” (No. CZ.02.1.01/0.0/0.0/17_048/0007280), by the grant of Ministry of Health of the Czech Republic - AZV NV19-08-00250, and by the European Union’s Horizon 2020 research and innovation programme under grant agreement N-856620 and by Conceptual Development of Research Organization (Faculty Hospital in Pilsen – FNPl, 00669806) provided by the Ministry of Health of the Czech Republic
Acknowledgments
The authors would like to thank all patients voluntarily taking part in the study. We would like to thank the following heads of the comprehensive cancer centres for the permission to use data of patients from their respective regional networks: Martina Chodacka, Chomutov Hospital and Masaryk Hospital in Usti nad Labem; Vaclav Janovsky, Ceske Budejovice Hospital; Jiří Mayer, University Hospital, Brno; Jana Prausova, Motol University Hospital, Prague; David Feltl, University Hospital, Ostrava; Jiri Petera, University Hospital, Hradec Kralove; Lubomir Slavicek, Jihlava Hospital; Jana Katolicka, St Anna University Hospital, Brno; Rostislav Vyzula, Masaryk Memorial Institute of Oncology, Brno; Jiri Bartos, County Hospital, Liberec; Martin Safanda, Na Homolce Hospital, Prague; Renata Soumarova, Novy Jicin Hospital; Lubos Petruzelka, General University Hospital, Prague; Milan Kohoutek, T Bata Memorial Hospital, Zlin. We are also indebted to all physicians who provided data for the RENIS registry.
Conflicts of Interest
O.F. received honoraria from Roche, Janssen, GSK, and Pfizer for consultations and lectures unrelated to this project. J.F. has received honoraria from Astra Zeneca, Roche, and Novartis for consultations and lectures unrelated to this project. A.P. received lecture honoraria from Novartis, Pfizer, Bayer-Schering, and Roche. B.M. received honoraria for lectures and advisory boards from Novartis, Pfizer, Bayer-Schering, Astellas, and Roche. J.K. declare no conflicts of interest that might be relevant to the contents of this manuscript. M.Z. has received honoraria from Astra Zeneca, Roche, Bristol-Myers-Squibb, and Novartis for consultations and lectures unrelated to this project. K.K. received lecture honoraria from Pfizer. T.M. and T.D. work both for iHETA (non-profit scientific organization) and Value Outcomes (consultancy company in the field of health economics/pharmacoeconomics and prepares projects for pharmaceutical industry). L.C. declared no potential conflict of interest. T.B. received lecture honoraria from Novartis, Pfizer, Bayer, Amgen, Servier, Bristol Myers Squibb, Astellas, Janssen, and Roche, and research support from Roche, Servier, and Novartis. R.L. and T.B. received lecture honoraria from Novartis, Pfizer, Bayer-Schering, Astellas, and Roche.
References
- Znaor, A.; Lortet-Tieulent, J.; Laversanne, M.; Jemal, A.; Bray, F. International variations and trends in renal cell carcinoma incidence and mortality. Eur. Urol. 2015, 67, 519. [Google Scholar] [CrossRef]
- Srigley, J.R.; Delahunt, B.; Eble, J.N.; Egevad, L.; Epstein, J.I.; Grignon, D.; Hes, O.; Moch, H.; Montironi, R.; Tickoo, S.K.; et al. The International Society of Urological Pathology (ISUP) Vancouver classification of renal neoplasia. Am. J. Surg. Pathol. 2013, 37, 1469–1489. [Google Scholar] [CrossRef]
- Motzer, R.J.; Bacik, J.; Murphy, B.A.; Russo, P.; Mazumdar, M. Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J. Clin. Oncol. 2002, 20, 289–296. [Google Scholar] [CrossRef]
- Motzer, R.J.; Hutson, T.E.; Tomczak, P.; Michaelson, M.D.; Bukowski, R.M.; Oudard, S.; Negrier, S.; Szczylik, C.; Pili, R.; Bjarnason, G.A.; et al. Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J. Clin. Oncol. 2009, 27, 3584–3590. [Google Scholar] [CrossRef] [PubMed]
- Sternberg, C.N.; Davis, I.D.; Mardiak, J.; Szczylik, C.; Lee, E.; Wagstaff, J.; Barrios, C.H.; Salman, P.; Gladkov, O.A.; Kavina, A.; et al. Pazopanib in locally advanced or metastatic renal cell carcinoma: results of a randomized phase III trial. J. Clin. Oncol. 2010, 28, 1061–1068. [Google Scholar] [CrossRef] [PubMed]
- Buchler, T.; Klapka, R.; Melichar, B.; Brabec, P.; Dusek, L.; Vyzula, R.; Abrahamova, J. Sunitinib followed by sorafenib or vice versa for metastatic renal cell carcinoma – data from the Czech registry. Ann. Oncol. 2012, 23, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Therasse, P.; Arbuck, S.G.; Eisenhauer, E.A.; Wanders, J.; Kaplan, R.S.; Rubinstein, L.; Verweij, J.; Van Glabbeke, M.; van Oosterom, A.T.; Christian, M.C.; et al. New guidelines to evaluate the response to treatment in solid tumours. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J. Natl. Cancer Inst. 2000, 3, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Mazumdar, M.; Bacik, J.; Berg, W.; Amsterdam, A.; Ferrara, J. Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J. Clin. Oncol. 1999, 17, 2530–2540. [Google Scholar] [CrossRef] [PubMed]
- Mekhail, T.M.; Abou-Jawde, R.M.; Boumerhi, G.; Malhi, S.; Wood, L.; Elson, P.; Bukowski, R. Validation and extension of the Memorial Sloan-Kettering prognostic factors model for survival in patients with previously untreated metastatic renal cell carcinoma. J. Clin. Oncol. 2005, 23, 832–841. [Google Scholar] [CrossRef] [PubMed]
- Heng, D.Y.; Xie, W.; Regan, M.M.; Harshman, L.C.; Bjarnason, G.A.; Vaishampayan, U.N.; Mackenzie, M.; Wood, L.; Donskov, F.; Tan, M.H.; et al. External validation and comparison with other models of the International Metastatic Renal-Cell Carcinoma Database Consortium prognostic model: a population-based study. Lancet Oncol. 2013, 14, 141–148. [Google Scholar] [CrossRef]
- Motzer, R.J.; Hutson, T.E.; Tomczak, P.; Michaelson, M.D.; Bukowski, R.M.; Rixe, O.; Oudard, S.; Negrier, S.; Szczylik, C.; Kim, S.T.; et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N. Engl. J. Med. 2007, 356, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Hutson, T.E.; Cella, D.; Reeves, J.; Hawkins, R.; Guo, J.; Nathan, P.; Staehler, M.; de Souza, P.; Merchan, J.R.; et al. Pazopanib versus sunitinib in metastatic renal-cell carcinoma. N. Engl. J. Med. 2013, 369, 722–731. [Google Scholar] [CrossRef] [PubMed]
- Escudier, B.; Pluzanska, A.; Koralewski, P.; Ravaud, A.; Bracarda, S.; Szczylik, C.; Chevreau, C.; Filipek, M.; Melichar, B.; Bajetta, E.; et al. Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial. Lancet 2007, 370, 2103–2111. [Google Scholar] [CrossRef]
- Hutson, T.E.; Lesovoy, V.; Al-Shukri, S.; Stus, V.P.; Lipatov, O.N.; Bair, A.H.; Rosbrook, B.; Chen, C.; Kim, S.; Vogelzang, N.J. Axitinib versus sorafenib as first-line therapy in patients with metastatic renal-cell carcinoma: a randomised open-label phase 3 trial. Lancet Oncol. 2013, 14, 1287–1294. [Google Scholar] [CrossRef]
- Choueiri, T.K.; Halabi, S.; Sanford, B.L.; Hahn, O.; Michaelson, M.D.; Walsh, M.K.; Feldman, D.R.; Olencki, T.; Picus, J.; Small, E.J.; et al. Cabozantinib Versus Sunitinib As Initial Targeted Therapy for Patients With Metastatic Renal Cell Carcinoma of Poor or Intermediate Risk: The Alliance A031203 CABOSUN Trial. J. Clin. Oncol. 2017, 35, 591–597. [Google Scholar] [CrossRef]
- Motzer, R.J.; Tannir, N.M.; McDermott, D.F.; Arén Frontera, O.; Melichar, B.; Choueiri, T.K.; Plimack, E.R.; Barthélémy, P.; Porta, C.; George, S.; et al. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2018, 378, 1277–1290. [Google Scholar] [CrossRef]
- Rini, B.I.; Plimack, E.R.; Stus, V.; Gafanov, R.; Hawkins, R.; Nosov, D.; Pouliot, F.; Alekseev, B.; Soulières, D.; Melichar, B.; et al. Pembrolizumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2019, 380, 1116–1127. [Google Scholar] [CrossRef]
- Rini, B.I.; Powles, T.; Atkins, M.B.; Escudier, B.; McDermott, D.F.; Suarez, C.; Bracarda, S.; Stadler, W.M.; Donskov, F.; Lee, J.L.; et al. Atezolizumab plus bevacizumab versus sunitinib in patients with previously untreated metastatic renal cell carcinoma (IMmotion151): a multicentre, open-label, phase 3, randomised controlled trial. Lancet. 2019, 393, 2404–2415. [Google Scholar] [CrossRef]
- Motzer, R.J.; Penkov, K.; Haanen, J.; Rini, B.; Albiges, L.; Campbell, M.T.; Venugopal, B.; Kollmannsberger, C.; Negrier, S.; Uemura, M.; et al. Avelumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2019, 380, 1103–1115. [Google Scholar] [CrossRef]
- Rini, B.I.; Hutson, T.E.; Figlin, R.A.; Lechuga, M.J.; Valota, O.; Serfass, L.; Rosbrook, B.; Motzer, R.J. Sunitinib in Patients With Metastatic Renal Cell Carcinoma: Clinical Outcome According to International Metastatic Renal Cell Carcinoma Database Consortium Risk Group. Clin. Genitourin Cancer 2018, 16, 298–304. [Google Scholar] [CrossRef]
- Kubackova, K.; Melichar, B.; Bortlicek, Z.; Pavlik, T.; Poprach, A.; Svoboda, M.; Lakomy, R.; Vyzula, R.; Kiss, I.; Dusek, L.; et al. Comparison of Two Prognostic Models in Patients with Metastatic Renal Cancer Treated with Sunitinib: a Retrospective, Registry-Based Study. Target. Oncol. 2015, 10, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Kwon, W.A.; Cho, I.C.; Yu, A.; Nam, B.H.; Joung, J.Y.; Seo, H.K.; Lee, K.H.; Chung, J. Validation of the MSKCC and Heng risk criteria models for predicting survival in patients with metastatic renal cell carcinoma treated with sunitinib. Ann. Surg. Oncol. 2013, 20, 4397–4404. [Google Scholar] [CrossRef] [PubMed]
- Bamias, A.; Karadimou, A.; Lampaki, S.; Lainakis, G.; Malettou, L.; Timotheadou, E.; Papazisis, K.; Andreadis, C.; Kontovinis, L.; Anastasiou, I.; et al. Prognostic stratification of patients with advanced renal cell carcinoma treated with sunitinib: comparison with the Memorial Sloan-Kettering prognostic factors model. BMC Cancer 2010, 10, 45. [Google Scholar] [CrossRef] [PubMed]
- Gore, M.E.; Szczylik, C.; Porta, C.; Bracarda, S.; Bjarnason, G.A.; Oudard, S.; Lee, S.H.; Haanen, J.; Castellano, D.; Vrdoljak, E.; et al. Final results from the large sunitinib global expanded-access trial in metastatic renal cell carcinoma. Br. J. Cancer 2015, 113, 9–12. [Google Scholar] [CrossRef]
- Motzer, R.J.; Nosov, D.; Eisen, T.; Bondarenko, I.; Lesovoy, V.; Lipatov, O.; Tomczak, P.; Lyulko, O.; Alyasova, A.; Harza, M.; et al. Tivozanib versus sorafenib as initial targeted therapy for patients with metastatic renal cell carcinoma: results from a phase III trial. J. Clin. Oncol. 2013, 31, 3791–3799. [Google Scholar] [CrossRef]
- Eichelberg, C.; Vervenne, W.L.; De Santis, M.; Fischer von Weikersthal, L.; Goebell, P.J.; Lerchenmüller, C.; Zimmermann, U.; Bos, M.M.; Freier, W.; Schirrmacher-Memmel, S.; et al. SWITCH: a randomised, sequential, open-label study to evaluate the efficacy and safety of sorafenibsunitinib versus sunitinib-sorafenib in the treatment of metastatic renal cell cancer. Eur. Urol. 2015, 68, 837–847. [Google Scholar] [CrossRef]
- Sella, A.; Michaelson, M.D.; Matczak, E.; Simantov, R.; Lin, X.; Figlin, R.A. Heterogeneity of Patients With Intermediate-Prognosis Metastatic Renal Cell Carcinoma Treated With Sunitinib. Clin Genitourin Cancer 2017, 15, 291–299. [Google Scholar] [CrossRef]
- Tamada, S.; Iguchi, T.; Yasuda, S.; Kato, M.; Yamasaki, T.; Nakatani, T. The difference in the survival rate of patients with metastatic renal cell carcinoma in the intermediate-risk group of the Memorial Sloan Kettering Cancer Center criteria. Oncotarget. 2018, 9, 27752–27759. [Google Scholar] [CrossRef][Green Version]
- Miyazaki, A.; Miyake, H.; Harada, K.I.; Inoue, T.A.; Fujisawa, M. Prognostic outcome in patients treated with tyrosine kinase inhibitors as first-line molecular-targeted therapy for metastatic renal cell carcinoma: Experience in real-world clinical practice in Japan. Mol. Clin. Oncol. 2015, 3, 601–606. [Google Scholar] [CrossRef]
- Melichar, B. Laboratory medicine and medical oncology: the tale of two Cinderellas. Clin. Chem. Lab. Med. 2013, 51, 99–112. [Google Scholar] [CrossRef]
- Ravaud, A.; Barrios, C.H.; Alekseev, B.; Tay, M.H.; Agarwala, S.S.; Yalcin, S.; Lin, C.C.; Roman, L.; Shkolnik, M.; Anak, O.; et al. RECORD-2: phase II randomized study of everolimus and bevacizumab versus interferon α-2a and bevacizumab as first-line therapy in patients with metastatic renal cell carcinoma. Ann. Oncol. 2015, 26, 1378–1384. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).