Effects of Ethanolic and Aqueous Extracts of Garcinia gardneriana Leaves in an In Vivo Experimental Model Induced by a Hyperlipidic Diet
Abstract
1. Introduction
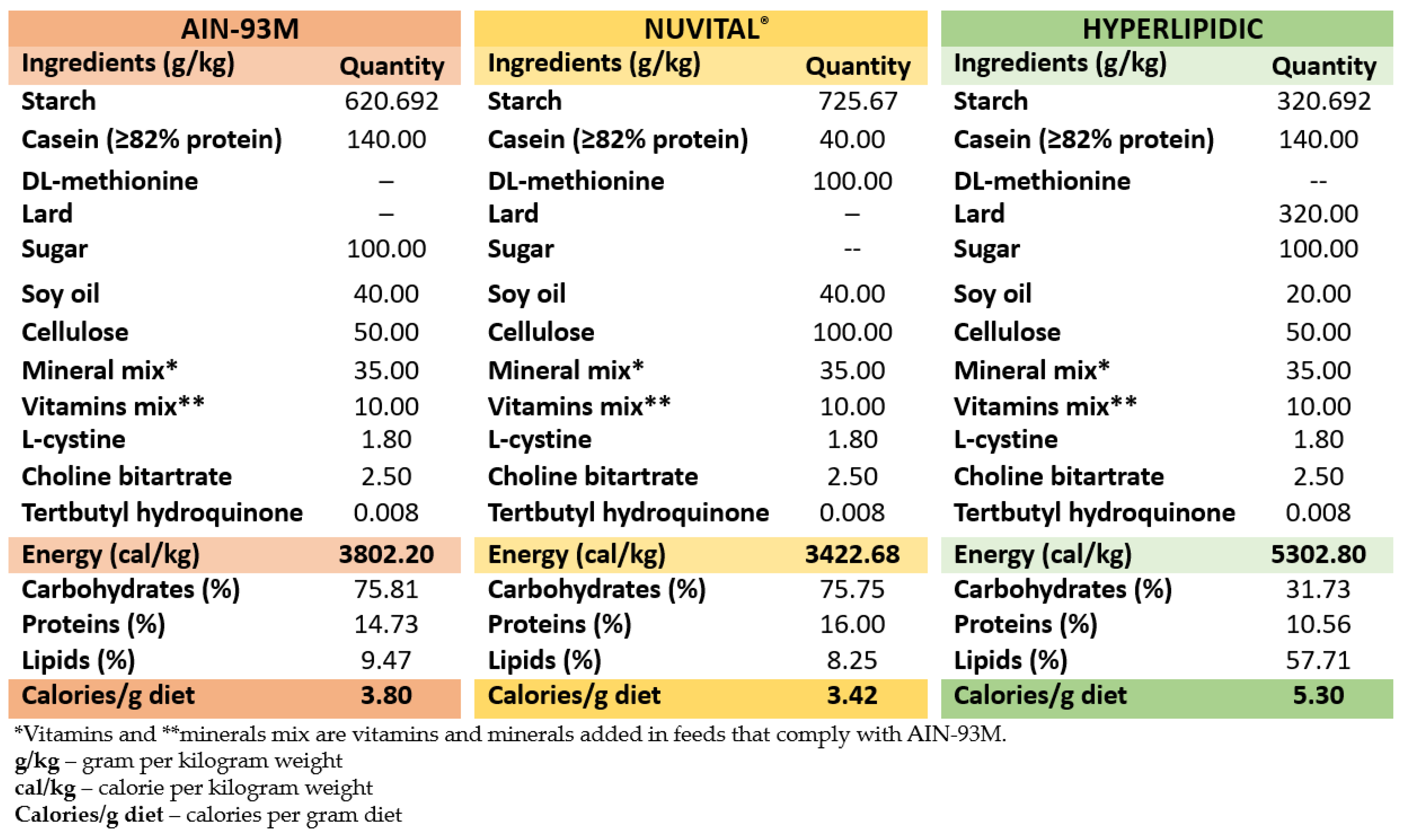
2. Materials and Methods
2.1. Leaf Collection
2.2. Extract Preparation
2.3. Animals
2.4. Experimental Design
2.5. Metabolic Changes in Serum
2.6. Concentration of Adipokines: IL-10 and MCP-1
2.7. Statistical Analysis
3. Results
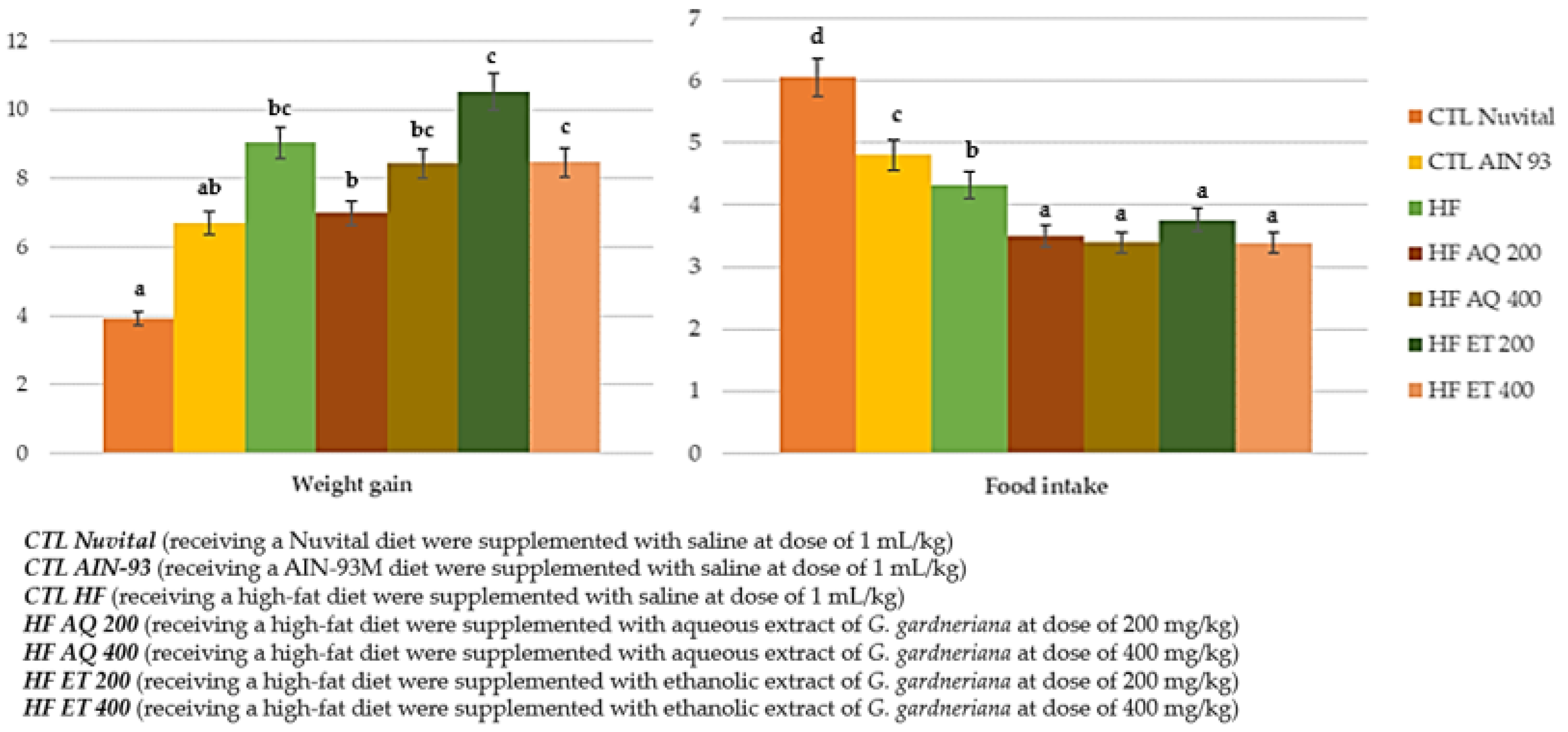
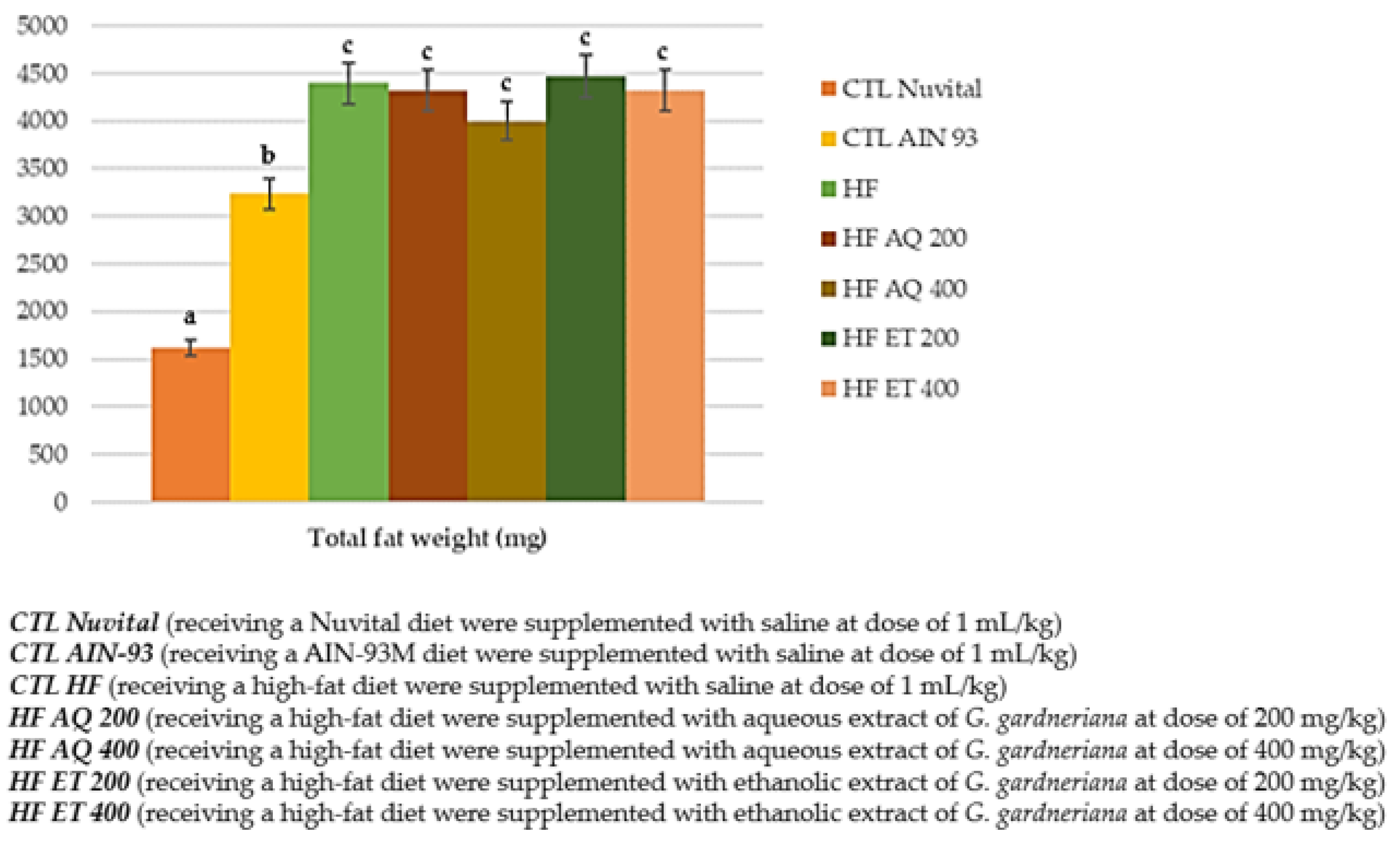
3.1. Food Intake, Weigh Again, Body Fat Percentage and Adipocyte Area
3.2. Serum Metabolic Changes
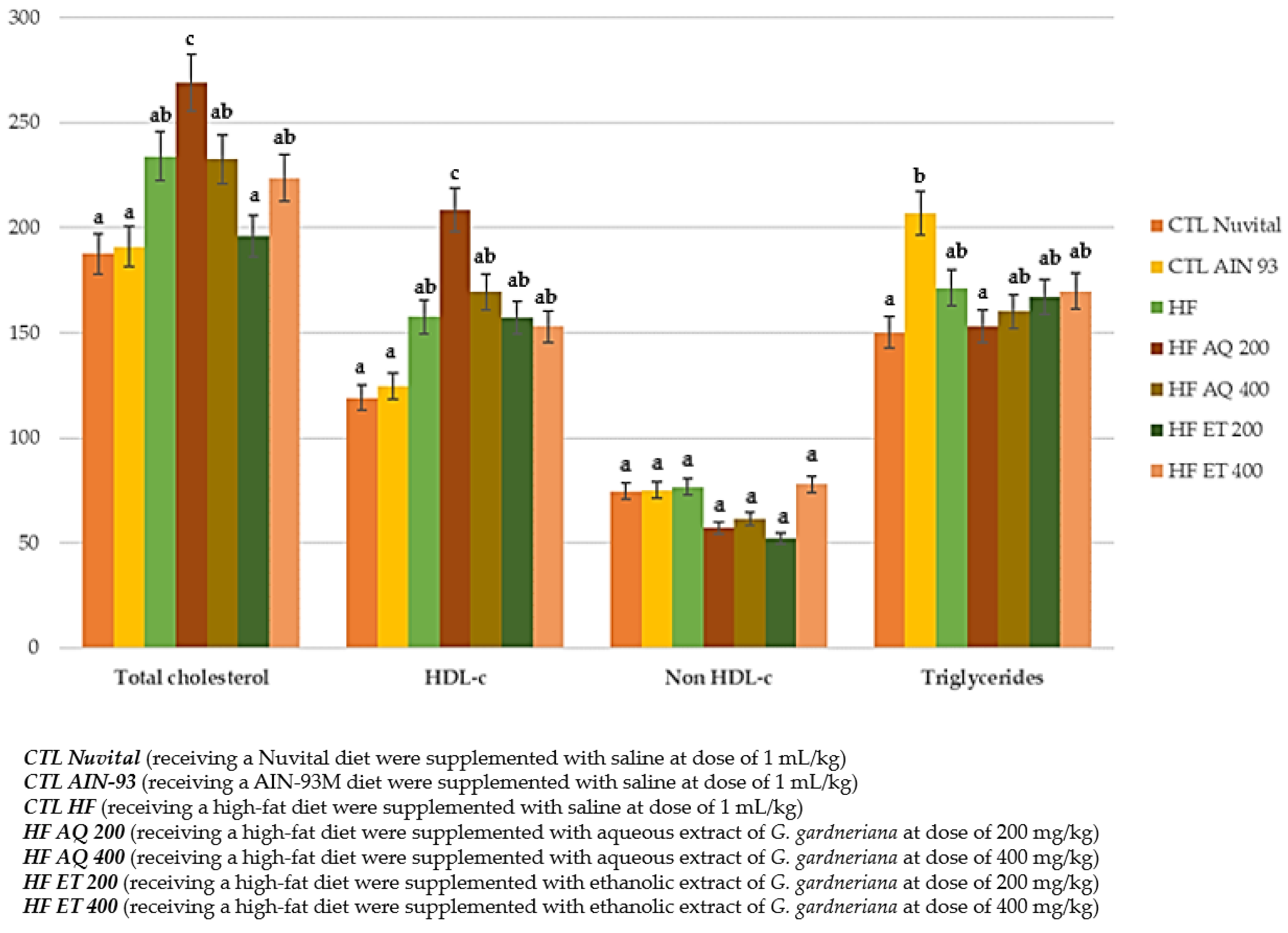
3.2.1. Triglycerides and Cholesterol (Total and Fractions) in Serum
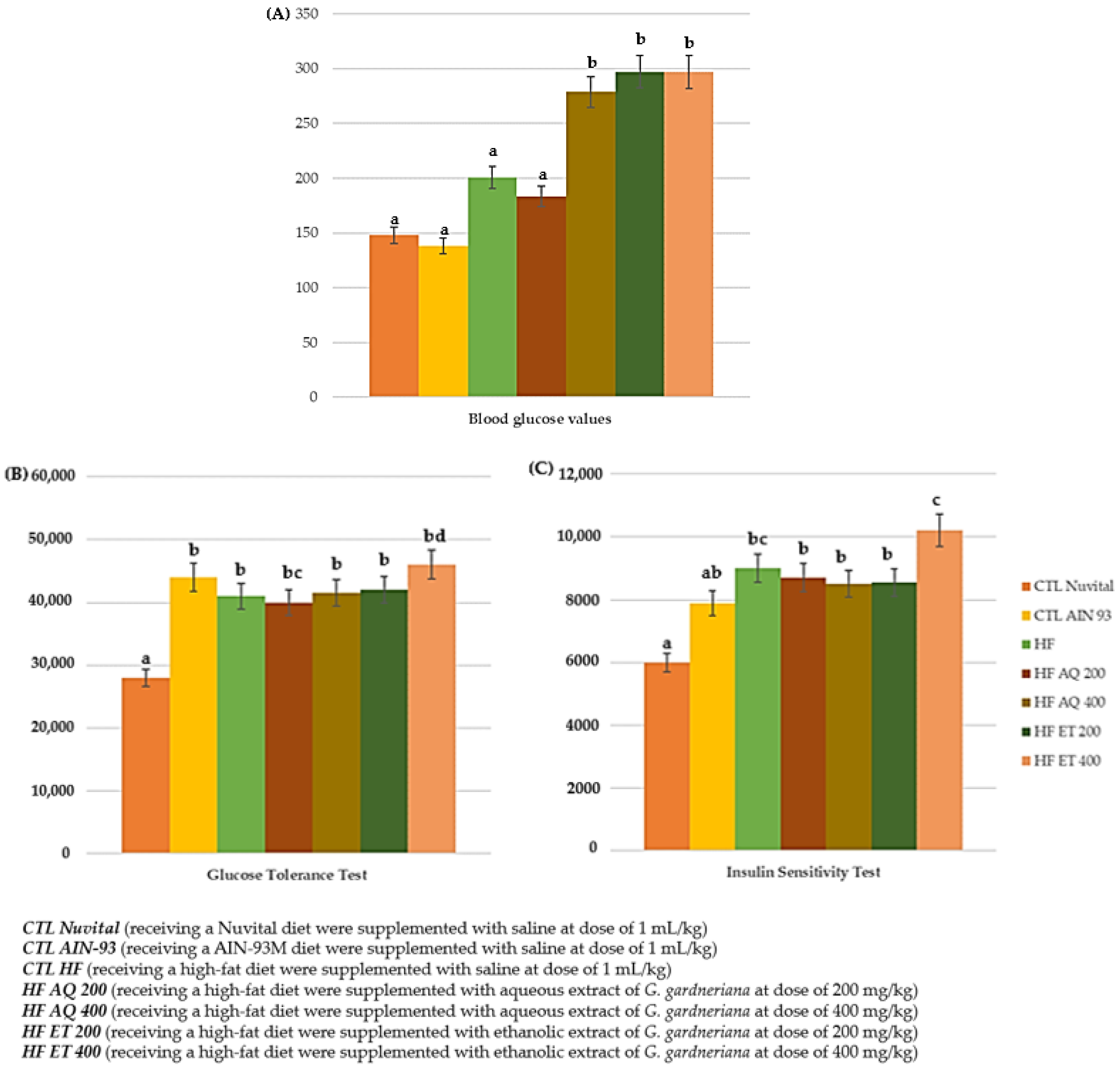
3.2.2. Glycemic Profile: Fasting Blood Glucose, Oral Glucose Tolerance and Insulin Sensitivity Tests
3.2.3. Adipokine Concentration: Cytokines IL-10 and MCP-1
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Almeida, L.S.B.; Murata, R.M.; Yatsuda, R.; Dos Santos, M.H.; Nagem, T.J.; Alencar, S.M.; Koo, H.; Rosalen, P.L. Antimicrobial activity of Rheedia brasiliensis and 7-epiclusianone against Streptococcus Mutans. Phytomed 2008, 15, 886–891. [Google Scholar] [CrossRef] [PubMed]
- Panthong, A.; Norkaew, P.; Kanjanapothi, D.; Taesotikul, T.; Anantachoke, N.; Reutakul, V. Anti-inflammatory, analgesic, and antipyretic activies of the extract of gamboge from Garcinia hanburyi Hook. J. Ethnophamacol. 2007, 111, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Hu, W.; Cai, Z.; Liu, Y.; Li, S.; Tao, W. New medicinal properties of mangostins: Analgesic activity and pharmacological characterization of active ingredients from the fruit hull of Garcinia mangostana. Pharm. Biochem. Behav. 2010, 95, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Sordat-Diserens, I.; Rogers, B.S.C.; Hostettmann, K. Prenylated xanthones from Garcinia livingstonei. Phytochem 1992, 31, 313–316. [Google Scholar] [CrossRef]
- Khanum, S.A.; Shashikanth, S.; Deepak, A.V. Synthesis and anti-inflammatory activity of benzophenone analogues. Bioorg. Chem. 2004, 32, 211–222. [Google Scholar] [CrossRef]
- Diaz-Carballo, D.; Seeber, S.; Strumberg, D.; Hilger, R.A. Novel antitumoral compound isolated from Clusiarosea. Int. J. Clin. Pharm. 2003, 41, 622–623. [Google Scholar] [CrossRef]
- Merza, J.; Aumond, M.C.; Rondeau, D.; Dumontet, V.; Le Ray, A.M.; Seraphin, D.; Richomme, P. Prenylated xanthones and tocotrienols from Garcinia Virgata. Phytochem. 2004, 65, 2915–2920. [Google Scholar] [CrossRef]
- Gustafson, K.R.; Blunt, J.W.; Munro, M.H.G.; Fuller, R.W.; McKee, T.C.; Cardellina, J.H.; McMahon, J.B.; Cragg, G.M.; Boyd, M.R. The guttiferones, HIV-inhibitory benzophenones from Symphonia globulifera, Garcinia livingstonei, Garcinia ovalifolia and Clusia rosea. Tetrahedron 1992, 48, 10093–10102. [Google Scholar] [CrossRef]
- Hay, A.E.A.; Mallet, M.C.; Dumontet, S.; Litaudon, V.; Rondeau, M.; Richomme, D. Antioxidant xanthones from Garcinia vieillardii. J. Nat. Prod. 2004, 67, 707–709. [Google Scholar] [CrossRef]
- Otuki, M.F.; Bernardi, C.A.; Prudente, A.S.; Laskoski, K.; Gomig, F.; Horinouchi, C.D.; Cabrini, D.A. Garcinia gardneriana (Planchon & Triana) Zappi. (Clusiaceae) as a Topical Anti-inflammatory Alternative for Cutaneous Inflammation. BCPT 2011, 109, 56–62. [Google Scholar] [CrossRef]
- Demenciano, S.D.C.; Silva, M.C.B.L.E.; Alexandrino, C.A.F.; Kato Junior, W.H.; Figueiredo, P.D.O.; Garcez, W.S.; Campos, R.P.; Guimarães, R.C.A.; Sarmento, U.C.; Bogo, D. Antiproliferative activity and antioxidant potential of extracts of Garcinia gardneriana. Molecules 2020, 25, 3201. [Google Scholar] [CrossRef] [PubMed]
- Verdi, L.G.; Pizzolatti, M.G.; Montanher, A.B.P.; Brighente, I.M.C.; Smânia, J.A.; Smânia, E.F.A.; Simionatto, E.L.; Monache, F.D. Antibacterial and brine shrimp lethality tests of biflavonoids and derivatives of Rheedia gardneriana. Fitoterapia 2004, 75, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Castardo, J.A.; Prudente, A.S.; Ferreira, J.; Guimarães, C.L.; Delle Monache, F.; Cechinel Filho, V.; Otuki, M.F.; Cabrini, D.A. Anti-inflammatory effects of hydroalcoholic extract and two biflavonoids from Garcinia gardneriana leaves in mouse paw oedema. J. Ethnopharmacol. 2008, 118, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Melo, M.S.; Quintans, J.S.; Araújo, A.A.; Duarte, M.C.; Bonjardim, L.R.; Nogueira, P.C.; Moraes, V.R.; Araújo-Júnior, J.X.; Ribeiro, E.A.; Quintans-Júnior, L.J. A systematic review for anti-inflammatory property of Clusiaceae family: A preclinical approach. J. Evid. Based Complement. Altern. Med. 2014, 2014, 960258. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Obesity and Overweight. Fact Sheet. Updated October. 2017. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 9 October 2022).
- Zobel, E.H.; Hansen, T.W.; Rossing, P.; Von Scholten, B.J. Global changes in food supply and the obesity epidemic. Curr. Obes. Rep. 2016, 5, 449–455. [Google Scholar] [CrossRef]
- BRASIL. Ministério da Saúde. Vigitel Brasil 2018: Vigilância de Fatores de Risco e Proteção Para Doenças Crônicas Por Inquérito Telefônico. Brasília: Ministério da Saúde. 2019. Available online: http://portalarquivos2.saude.gov.br/images/pdf/2019/julho/25/vigitel-brasil-2018.pdf (accessed on 9 October 2022).
- Sarapio, E.; De Souza, S.K.; Model, J.F.; Trapp, M.; Da Silva, R.S. Stanniocalcin-1 and-2 effects on glucose and lipid metabolism in white adipose tissue from fed and fasted rats. Can. J. Physiol. Pharmacol. 2019, 97, 916–923. [Google Scholar] [CrossRef]
- Rubio-Ruiz, M.E.; Hafidi, M.E.; Pérez-Torres, I.; Banos, G.; Guarner, V. Medicinal agents and metabolic syndrome. Curr. Med. Chem. 2013, 20, 2626–2640. [Google Scholar] [CrossRef]
- Tan, J.Q.; Zhang, L.; Xu, H.X. Garcinia oligantha: A comprehensive overview of ethnomedicine, phytochemistry and pharmacology. J. Ethnopharmacol. 2023, 5, 116130. [Google Scholar] [CrossRef]
- Rodrigues, D.A.; de Sousa, B.L.; da Silva, J.G.; Pereira, G.A.; Bousada, G.M.; da Silva, A.A.; Demuner, A.J.; Costa, E.D.M.; Pilau, E.J.; Silva, E.; et al. Phytotoxic property of metabolites isolated from Garcinia gardneriana. Comput. Biol. Chem. 2021, 92, 107460. [Google Scholar] [CrossRef]
- dos Santos, A.G.; Sivieri, K.; da Mata, B.P.M. Bacupari Fruit (Garcinia brasiliensis Mart). Handb. Phytonutrients Indig. Fruits Veg. 2022, 1, 298. [Google Scholar] [CrossRef]
- John, O.D.; Brown, L.; Panchal, S.K. Garcinia fruits: Their potential to combat metabolic syndrome. In Nutraceuticals and Natural Product Derivatives: Disease Prevention & Drug Discovery; Ullah, M.F., Ahmad, A., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2019; Volume 1, pp. 39–80. ISBN 9781119436690. [Google Scholar]
- Kalita, A.; Das, M.; Das, B.; Baro, M.R. Molecular docking prediction and in vitro studies elucidate anti-inflammatory effect of Garcinia extract against inducible nitric oxide synthase and cyclooxygenase-2 targets. Beni Suef Univ. J. Basic Appl. Sci. 2022, 11, 1–17. [Google Scholar] [CrossRef]
- Prado, J.; Rostagno, M. Natural Product Extraction: Principles and Applications; Royal Society of Chemistry: London, UK, 2022; Volume 71, pp. 10–95. ISBN 1757-7047. [Google Scholar]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C.J.R. AIN-93 purified diets for laboratory rodents: Final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [CrossRef]
- Od-Ek, P.; Deenin, W.; Malakul, W.; Phoungpetchara, I.; Tusophon, S. Anti-obesity effect of Carica papaya in high-fat diet fed rats. Biomed. Rep. 2020, 13, 30. [Google Scholar] [CrossRef]
- Lee, S.; Keirsey, K.I.; Kirkland, R.; Grunewald, Z.I.; Fischer, J.G.; De La Serre, C.B. Blueberry supplementation influences the gut microbiota, inflammation, and insulin resistance in high-fat-diet–fed rats. J. Nutr. 2018, 148, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Liu, Q.; Zhang, Q.; Tuo, X.; Fan, D.; Deng, J.; Yang, H. Kiwifruit seed oil prevents obesity by regulating inflammation, thermogenesis, and gut microbiota in high-fat diet-induced obese C57BL/6 mice. Food Chem. Toxicol. 2019, 125, 85–94. [Google Scholar] [CrossRef]
- Sripradha, R.; Magadi, S.G. Efficacy of garcinia cambogia on body weight, inflammation and glucose tolerance in high fat fed male wistar rats. J. Clin. Diagn. Res. 2015, 9, 1–12. [Google Scholar] [CrossRef]
- Sripradha, R.; Sridhar, M.G.; Maithilikarpagaselvi, N. Antihyperlipidemic and antioxidant activities of the ethanolic extract of Garcinia cambogia on high fat diet-fed rats. J. Complement. Integr. Med. 2016, 13, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Lenquiste, S.A.; da Silva Marineli, R.; Moraes, É.A.; Dionísio, A.P.; de Brito, E.S.; Junior, M.R.M. Jaboticaba peel and jaboticaba peel aqueous extract shows in vitro and in vivo antioxidant properties in obesity model. Food Res. Int. 2015, 77, 162–170. [Google Scholar] [CrossRef]
- Santana, L.F.; do Espírito Santo, B.L.S.; Tatara, M.B.; Negrão, F.J.; Croda, J.; Alves, F.M.; de Oliveira Filiú, W.F.; Cavalheiro, L.F.; Nazário, C.E.D.; Asato, M.A.; et al. Effects of Carica papaya Linn seed oil on food intake, adiposity, metabolic and inflammatory profile of mice using a high-fat diet. Molecules 2022, 27, 6705. [Google Scholar] [CrossRef]
- Santana, L.F.; Santo, B.L.S.E.; Sasso, S.; Aquino, D.F.S.; Cardoso, C.A.L.; Pott, A.; Soares, F.L.P.; Freitas, K.C. Efeitos do extrato etanólico das folhas de cagaiteira (Eugenia dysenterica DC.) em camundongos diabéticos induzidos por estreptozotocina. Int. J. Dev. Res. 2018, 8, 1355–1362. [Google Scholar]
- Taylor, B.A.; Phillips, S.J. Detection of obesity QTLs on mouse chromosomes 1 and 7 by selective DNA pooling. Genomics 1996, 34, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Agada, R.; Usman, W.A.; Shehu, S.; Thagariki, D.; Agada, R. In vitro and in vivo inhibitory effects of Carica papaya seed on α-amylase and α-glucosidase enzymes. Heliyon 2020, 6, e03618. [Google Scholar] [CrossRef] [PubMed]
- Tai, M.M. A mathematical model for the determination of total area under glucose tolerance and other metabolic curves. Diabetes Care 1994, 17, 152–154. [Google Scholar] [CrossRef] [PubMed]
- Kunle, O.F.; Egharevba, H.O.; Ahmadu, P.O. Standardization of herbal medicines—A review. Int. J. Biodivers. Conserv. 2012, 4, 101–112. [Google Scholar] [CrossRef]
- Shameer, P.S.; Rameshkumar, K.B.; Mohanan, N. Diversity of garcinia in the Western Ghats. Phytochemical perspective. India: Jawaharlal Nehru Trop. Bot. Gard. Res. Inst. 2016, 6, 1–18. [Google Scholar]
- Koudoufio, M.; Desjardins, Y.; Feldman, F.; Spahis, S.; Delvin, E.; Levy, E. Insight into Polyphenol and Gut Microbiota Crosstalk: Are Their Metabolites the Key to Understand Protective Effects against Metabolic Disorders? Antioxidants 2020, 9, 982. [Google Scholar] [CrossRef]
- Rains, T.M.; Agarwal, S.; Maki, K.C. Antiobesity effects of green tea catechins: A mechanistic review. J. Nutr. Biochem. 2011, 22, 1–7. [Google Scholar] [CrossRef]
- Espirito Santo, B.L.S.D.; Santana, L.F.; Kato Junior, W.H.; de Araújo, F.O.; Bogo, D.; Freitas, K.C.; Guimarães, R.C.A.; Hiane, P.A.; Pott, A.; Filiú, W.F.O.; et al. Medicinal Potential of Garcinia Species and Their Compounds. Molecules 2020, 25, 4513. [Google Scholar] [CrossRef]
- Lim, W.F.; Nasir, S.M.; Teh, L.K.; James, R.J.; Izhar, M.H.M.; Salleh, M.Z. The methanolic extract of Garcinia atroviridis (MeGa) reduces body weight and food intake, and improves lipid profiles by altering the lipid metabolism: A rat model. Turk. J. Biol. 2020, 44, 437–448. [Google Scholar] [CrossRef]
- Muhamad Adyab, N.S.; Rahmat, A.; Abdul Kadir, N.A.A.; Jaafar, H.; Shukri, R.; Ramli, N.S. Mangosteen (Garcinia mangostana) flesh supplementation attenuates biochemical and morphological changes in the liver and kidney of high fat diet-induced obese rats. BMC Complement. Altern. Med. 2019, 19, 344. [Google Scholar] [CrossRef]
- Chae, H.S.; Kim, E.Y.; Han, L.; Kim, N.R.; Lam, B.; Paik, J.H. Xanthones with pancreatic lipase inhibitory activity from the pericarps of Garcinia mangostana L. (Guttiferae). Eur. J. Lipid. Sci. Technol. 2016, 118, 1416–1421. [Google Scholar] [CrossRef]
- Chuah, L.O.; Ho, W.Y.; Beh, B.K.; Yeap, S.K. Updates on antiobesity effect of Garcinia origin (-)-HCA. Evid. Based. Complement. Altern. Med. 2013, 2013, 751658. [Google Scholar] [CrossRef]
- Carling, D. The AMP-activated protein kinase cascade—A unifying system for energy control. Trends Biochem. Sci. 2004, 29, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Bijland, S.; Mancini, S.J.; Salt, I.P. Role of AMP-activated protein kinase in adipose tissue metabolism and inflammation. Clin. Sci. 2013, 124, 491–507. [Google Scholar] [CrossRef] [PubMed]
- Patil, M.M.; Appaiah, K.A. Garcinia: Bioactive compounds and health benefits. Introd. Funct. Food Sci. 2015, 1, 110–125. [Google Scholar]
- Guerra, J.F.C.; Abreu, I.C.M.E.; Pereira, R.R.; Silva, M.; Cardoso, L.M.; Lima, W.G. Dietary açai attenuates hepatic steatosis via adiponectin-mediated effects on lipid metabolism in high-fat diet mice. J. Funct. Foods 2015, 14, 192–202. [Google Scholar] [CrossRef]
- Rothblat, G.H.; Phillips, M.C. High-density lipoprotein heterogeneity and function in reverse cholesterol transport. Curr. Opin. Lipidol. 2011, 21, 229–238. [Google Scholar] [CrossRef]
- Klop, B.; Elte, J.W.F.; Cabezas, M.C. Dyslipidemia in obesity: Mechanisms and potential targets. Nutrients 2013, 5, 1218–1240. [Google Scholar] [CrossRef]
- Daoud, E.; Scheede-Bergdahl, C.; Bergdahl, A. Effects of dietary micronutrients on plasma lipid levels and the consequence for cardiovascular disease. J. Cardiovasc. Dev. Dis. 2014, 1, 201–213. [Google Scholar] [CrossRef]
- Arora, R.; Vig, A.P.; Arora, S. Lipid peroxidation: A possible marker of diabetes. J Diabetes Metab. 2013, S11, 1–6. [Google Scholar] [CrossRef]
- Chung, K.W.; Ha, S.; Kim, S.M.; Kim, D.H.; An, H.J.; Lee, E.K.; Chung, H.Y. PPARα/β Activation Alleviates Age-Associated Renal Fibrosis in Sprague Dawley Rats. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in inflammation and metabolic disease. Nat. Rev. Immunol. 2011, 11, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Cabral-Santos, C.; de Lima Junior, E.A.; Fernandes, I.M.D.C.; Pinto, R.Z.; Rosa-Neto, J.C.; Bishop, N.C.; Lira, F.S. Interleukin-10 responses from acute exercise in healthy subjects: A systematic review. J. Cell Physiol. 2019, 234, 9956–9965. [Google Scholar] [CrossRef] [PubMed]
- Azman, K.F.; Amom, Z.; Azlan, A.; Esa, N.M.; Ali, R.M.; Shah, Z.M.; Kadir, K.K.A. Antiobesity effect of Tamarindus indica L. pulp aqueous extract in high-fat diet-induced obese rats. J. Nat. Med. 2012, 66, 333–342. [Google Scholar] [CrossRef]
- Chawla, A.; Chawla, R.; Jaggi, S. Microvascular and macrovascular complications in diabetes mellitus: Distinct or continuum? Indian J. Endocrinol. Metab. 2016, 20, 546–551. [Google Scholar] [CrossRef]
- Su, L.; Mittal, R.; Ramgobin, D.; Jain, R.; Jain, R. Current management guidelines on hyperlipidemia: The silent killer. J. Lipids 2021, 31, 9883352. [Google Scholar] [CrossRef]
- Chhabria, S.; Mathur, S.; Vadakan, S.; Sahoo, D.K.; Mishra, P.; Paital, B. A review on phytochemical and pharmacological facets of tropical ethnomedicinal plants as reformed DPP-IV inhibitors to regulate incretin activity. Front. Endocrinol. (Lausanne) 2022, 11, 1027237. [Google Scholar] [CrossRef]
- Ilango, S.; Sahoo, D.K.; Paital, B.; Kathirvel, K.; Gabriel, J.I.; Subramaniam, K.; Jayachandran, P.; Dash, R.K.; Hati, A.K.; Behera, T.R.; et al. A Review on Annona muricata and Its Anticancer Activity. Cancers (Basel) 2022, 19, 4539. [Google Scholar] [CrossRef]
- Calixto, J.B. Twenty-five years of research on medicinal plants in Latin America: A personal view. J. Ethnopharmacol. 2005, 100, 131–134. [Google Scholar] [CrossRef]
- Sánchez, A.; Rojas, P.; Basfi-Fer, K.; Carrasco, F.; Inostroza, J.; Codoceo, J.; Valencia, A.; Papapietro, K.; Csendes, A.; Ruz, M. Micronutrient deficiencies in morbidly obese women prior to bariatric surgery. Obes. Surg. 2016, 26, 361–368. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santo, B.L.S.d.E.; Santana, L.F.; Kato Junior, W.H.; Araújo, F.d.O.d.; Tatara, M.B.; Croda, J.; Bogo, D.; Freitas, K.d.C.; Guimarães, R.d.C.A.; Hiane, P.A.; et al. Effects of Ethanolic and Aqueous Extracts of Garcinia gardneriana Leaves in an In Vivo Experimental Model Induced by a Hyperlipidic Diet. Nutrients 2023, 15, 1308. https://doi.org/10.3390/nu15061308
Santo BLSdE, Santana LF, Kato Junior WH, Araújo FdOd, Tatara MB, Croda J, Bogo D, Freitas KdC, Guimarães RdCA, Hiane PA, et al. Effects of Ethanolic and Aqueous Extracts of Garcinia gardneriana Leaves in an In Vivo Experimental Model Induced by a Hyperlipidic Diet. Nutrients. 2023; 15(6):1308. https://doi.org/10.3390/nu15061308
Chicago/Turabian StyleSanto, Bruna Larissa Spontoni do Espirito, Lidiani Figueiredo Santana, Wilson Hino Kato Junior, Felipe de Oliveira de Araújo, Mariana Bento Tatara, Júlio Croda, Danielle Bogo, Karine de Cássia Freitas, Rita de Cássia Avellaneda Guimarães, Priscila Aiko Hiane, and et al. 2023. "Effects of Ethanolic and Aqueous Extracts of Garcinia gardneriana Leaves in an In Vivo Experimental Model Induced by a Hyperlipidic Diet" Nutrients 15, no. 6: 1308. https://doi.org/10.3390/nu15061308
APA StyleSanto, B. L. S. d. E., Santana, L. F., Kato Junior, W. H., Araújo, F. d. O. d., Tatara, M. B., Croda, J., Bogo, D., Freitas, K. d. C., Guimarães, R. d. C. A., Hiane, P. A., Pott, A., Filiú, W. F. d. O., Faria, B. B. d., Figueiredo, P. d. O., do Nascimento, V. A., Ayres, F. L., & Bastos, P. R. H. d. O. (2023). Effects of Ethanolic and Aqueous Extracts of Garcinia gardneriana Leaves in an In Vivo Experimental Model Induced by a Hyperlipidic Diet. Nutrients, 15(6), 1308. https://doi.org/10.3390/nu15061308





