Nutritional Support for Liver Diseases
Abstract
1. Introduction
2. Nutritional Support for Liver Diseases
2.1. Cholangiocarcinoma
2.1.1. Oral Nutritional Supplements (ONS)
2.1.2. Omega-3 Fatty Acids
2.2. Non-Alcoholic Fatty Liver Disease (NAFLD) and Non-Alcoholic Steatohepatitis (NASH)
2.2.1. Overall Recommendation
2.2.2. Mediterranean Diet
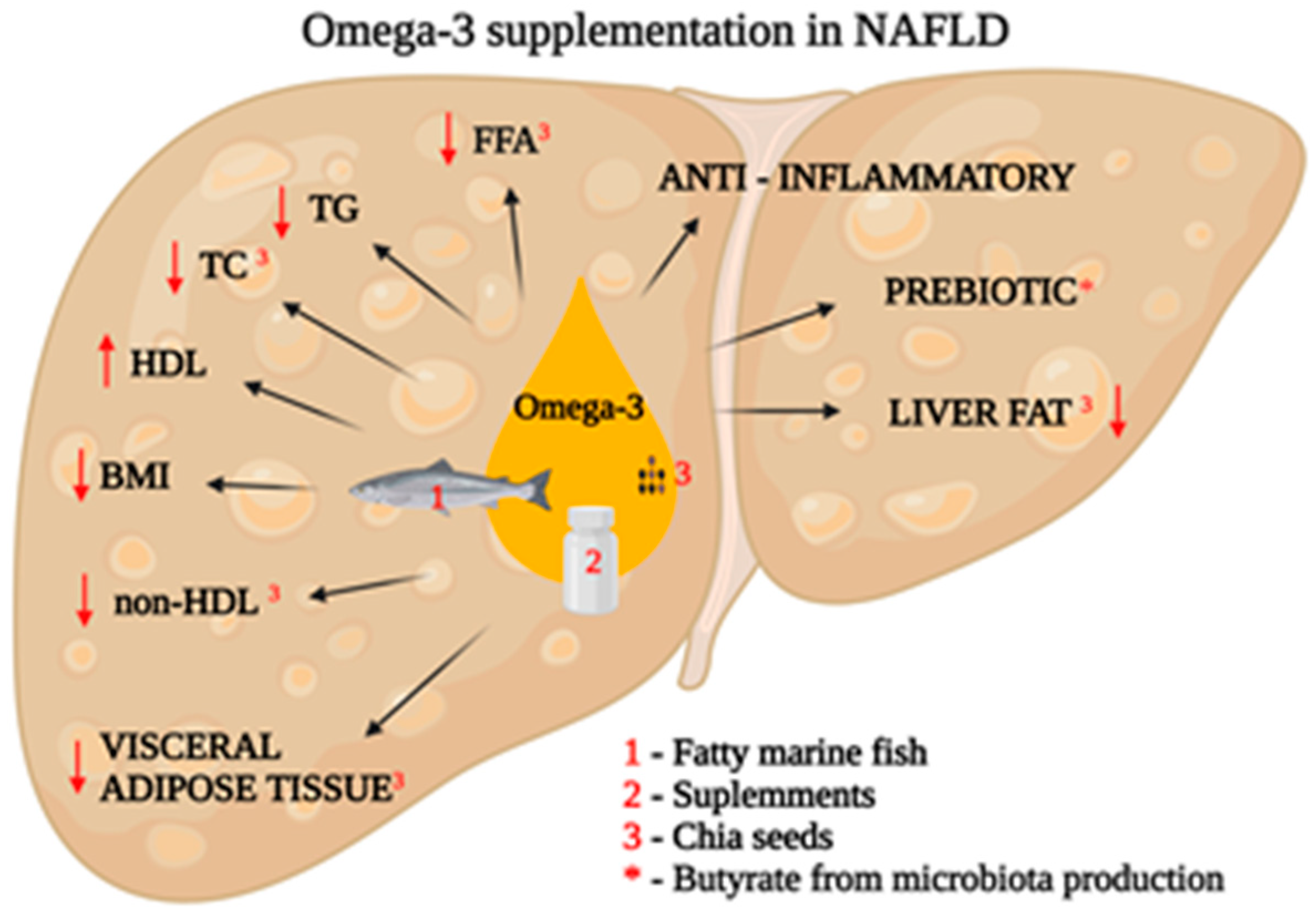
2.2.3. Supplementation
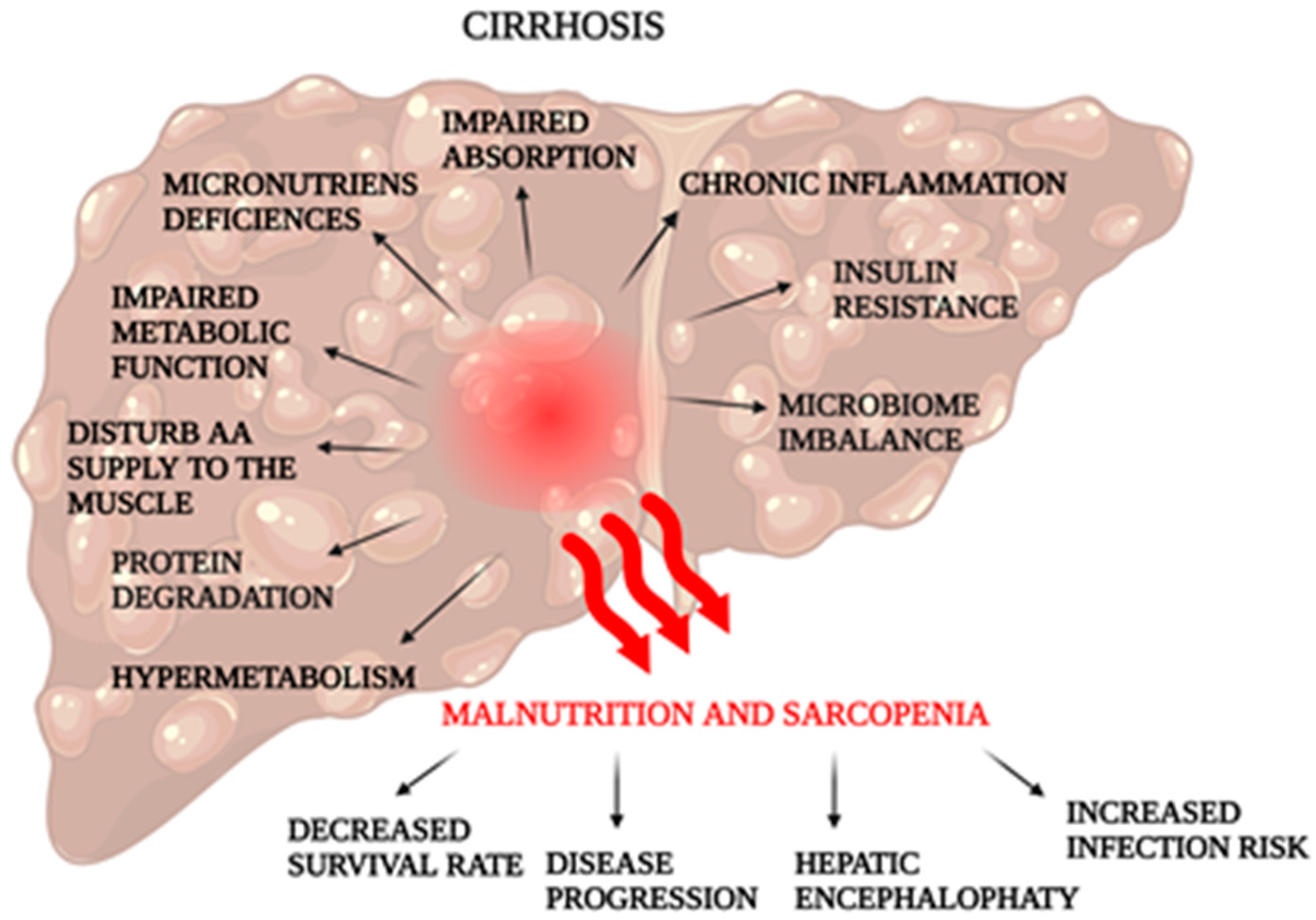
2.3. Cirrhosis
2.3.1. Dietotherapy
2.3.2. Supplementation
2.4. Liver Transplantation
- A reduction in the amount of food consumed by the patient and an inadequate diet (i.a. low calorie diet low in protein (<0.8 g/kg bw) [146];
- metabolic process disorders (patients are characterized by a state of hypermetabolism; there is an acceleration of protein, fat and carbohydrate metabolism);
- the impaired absorption of fats associated with reduced bile secretion. This condition also leads to deficiencies in fat-soluble vitamins;
- the premature feeling of satiety (patients experience a delay in gastric emptying and ascites compressing the gastrointestinal tract) [93].
2.4.1. Nutritional Support before Liver Transplantation
Before the Surgery—ERAS Protocol
Limitations of Implementing the ERAS
2.4.2. Nutritional Support after Liver Transplantation
Early Period (12–24 h)
Later Period
3. Future Perspectives and Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sharma, A.; Nagalli, S. Chronic Liver Disease. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Cheung, K.; Lee, S.S.; Raman, M. Prevalence and Mechanisms of Malnutrition in Patients with Advanced Liver Disease, and Nutrition Management Strategies. Clin. Gastroenterol. Hepatol. 2012, 10, 117–125. [Google Scholar] [CrossRef]
- Younossi, Z.; Tacke, F.; Arrese, M.; Chander Sharma, B.; Mostafa, I.; Bugianesi, E.; Wai-Sun Wong, V.; Yilmaz, Y.; George, J.; Fan, J.; et al. Global Perspectives on Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis. Hepatology 2019, 69, 2672–2682. [Google Scholar] [CrossRef] [PubMed]
- Shergill, R.; Syed, W.; Rizvi, S.A.; Singh, I. Nutritional Support in Chronic Liver Disease and Cirrhotics. World J. Hepatol. 2018, 10, 685–694. [Google Scholar] [CrossRef]
- Bischoff, S.C.; Bernal, W.; Dasarathy, S.; Merli, M.; Plank, L.D.; Schütz, T.; Plauth, M. ESPEN Practical Guideline: Clinical Nutrition in Liver Disease. Clin. Nutr. 2020, 39, 3533–3562. [Google Scholar] [CrossRef]
- Merli, M.; Berzigotti, A.; Zelber-Sagi, S.; Dasarathy, S.; Montagnese, S.; Genton, L.; Plauth, M.; Parés, A. EASL Clinical Practice Guidelines on Nutrition in Chronic Liver Disease. J. Hepatol. 2019, 70, 172–193. [Google Scholar] [CrossRef] [PubMed]
- Anand, A.C. Nutrition and Muscle in Cirrhosis. J. Clin. Exp. Hepatol. 2017, 7, 340–357. [Google Scholar] [CrossRef] [PubMed]
- Banales, J.M.; Marin, J.J.G.; Lamarca, A.; Rodrigues, P.M.; Khan, S.A.; Roberts, L.R.; Cardinale, V.; Carpino, G.; Andersen, J.B.; Braconi, C.; et al. Cholangiocarcinoma 2020: The next Horizon in Mechanisms and Management. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 557–588. [Google Scholar] [CrossRef]
- Zori, A.G.; Yang, D.; Draganov, P.V.; Cabrera, R. Advances in the Management of Cholangiocarcinoma. World J. Hepatol. 2021, 13, 1003–1018. [Google Scholar] [CrossRef]
- Khan, A.S.; Dageforde, L.A. Cholangiocarcinoma. Surg. Clin. N. Am. 2019, 99, 315–335. [Google Scholar] [CrossRef]
- Goyal, L.; Kongpetch, S.; Crolley, V.E.; Bridgewater, J. Targeting FGFR Inhibition in Cholangiocarcinoma. Cancer Treat. Rev. 2021, 95, 102170. [Google Scholar] [CrossRef]
- Noguchi, D.; Kuriyama, N.; Nakagawa, Y.; Maeda, K.; Shinkai, T.; Gyoten, K.; Hayasaki, A.; Fujii, T.; Iizawa, Y.; Tanemura, A.; et al. The Prognostic Impact of Lymphocyte-to-C-Reactive Protein Score in Patients Undergoing Surgical Resection for Intrahepatic Cholangiocarcinoma: A Comparative Study of Major Representative Inflammatory/Immunonutritional Markers. PLoS ONE 2021, 16, e0245946. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.-Q.; Chen, S.-Y.; Jiang, Z.-B.; Wu, B.; He, Y.; Wang, X.-X.; Li, Y.; Gao, P.; Yang, X.-J. Effect of Postoperative Early Enteral Nutrition on Clinical Outcomes and Immune Function of Cholangiocarcinoma Patients with Malignant Obstructive Jaundice. World J. Gastroenterol. 2020, 26, 7405–7415. [Google Scholar] [CrossRef] [PubMed]
- Cereda, E.; Cappello, S.; Colombo, S.; Klersy, C.; Imarisio, I.; Turri, A.; Caraccia, M.; Borioli, V.; Monaco, T.; Benazzo, M.; et al. Nutritional Counseling with or without Systematic Use of Oral Nutritional Supplements in Head and Neck Cancer Patients Undergoing Radiotherapy. Radiother. Oncol. 2018, 126, 81–88. [Google Scholar] [CrossRef]
- August, D.A.; Huhmann, M.B.; American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.) Board of Directors. Clinical Guidelines: Nutrition Support Therapy during Adult Anticancer Treatment and in Hematopoietic Cell Transplantation. JPEN J. Parenter. Enter. Nutr. 2009, 33, 472–500. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, S.M.; Jeung, H.C.; Lee, I.J.; Park, J.S.; Song, M.; Lee, D.K.; Lee, S.-M. The Effect of Nutrition Intervention with Oral Nutritional Supplements on Pancreatic and Bile Duct Cancer Patients Undergoing Chemotherapy. Nutrients 2019, 11, 1145. [Google Scholar] [CrossRef]
- Kaźmierczak-Siedlecka, K.; Daca, A.; Folwarski, M.; Makarewicz, W.; Lebiedzińska, A. Immunonutritional Support as an Important Part of Multidisciplinary Anti-Cancer Therapy. Cent. Eur. J. Immunol. 2020, 45, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, P.; Zhang, Y.; Shou, Q.; Li, H.; Zhu, Y.; He, L.; Chen, J.; Jiao, J. Eicosapentaenoic and Docosahexaenoic Acids Differentially Alter Gut Microbiome and Reverse High-Fat Diet-Induced Insulin Resistance. Mol. Nutr. Food Res. 2020, 64, e1900946. [Google Scholar] [CrossRef]
- Yao, L.; Han, C.; Song, K.; Zhang, J.; Lim, K.; Wu, T. Omega-3 Polyunsaturated Fatty Acids Upregulate 15-PGDH Expression in Cholangiocarcinoma Cells by Inhibiting MiR-26a/b Expression. Cancer Res. 2015, 75, 1388–1398. [Google Scholar] [CrossRef] [PubMed]
- Tai, H.-H.; Chi, X.; Tong, M. Regulation of 15-Hydroxyprostaglandin Dehydrogenase (15-PGDH) by Non-Steroidal Anti-Inflammatory Drugs (NSAIDs). Prostaglandins Other Lipid Mediat. 2011, 96, 37–40. [Google Scholar] [CrossRef]
- Abe, K.; Uwagawa, T.; Haruki, K.; Takano, Y.; Onda, S.; Sakamoto, T.; Gocho, T.; Yanaga, K. Effects of ω-3 Fatty Acid Supplementation in Patients with Bile Duct or Pancreatic Cancer Undergoing Chemotherapy. Anticancer Res. 2018, 38, 2369–2375. [Google Scholar] [CrossRef]
- Pouwels, S.; Sakran, N.; Graham, Y.; Leal, A.; Pintar, T.; Yang, W.; Kassir, R.; Singhal, R.; Mahawar, K.; Ramnarain, D. Non-Alcoholic Fatty Liver Disease (NAFLD): A Review of Pathophysiology, Clinical Management and Effects of Weight Loss. BMC Endocr. Disord. 2022, 22, 63. [Google Scholar] [CrossRef] [PubMed]
- Schattenberg, J.M.; Anstee, Q.M.; Caussy, C.; Bugianesi, E.; Popovic, B. Differences between Current Clinical Guidelines for Screening, Diagnosis and Management of Nonalcoholic Fatty Liver Disease and Real-World Practice: A Targeted Literature Review. Expert Rev. Gastroenterol. Hepatol. 2021, 15, 1253–1266. [Google Scholar] [CrossRef]
- Riazi, K.; Azhari, H.; Charette, J.H.; Underwood, F.E.; King, J.A.; Afshar, E.E.; Swain, M.G.; Congly, S.E.; Kaplan, G.G.; Shaheen, A.-A. The Prevalence and Incidence of NAFLD Worldwide: A Systematic Review and Meta-Analysis. Lancet Gastroenterol. Hepatol. 2022, 7, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Gershuni, V.M.; Yan, S.L.; Medici, V. Nutritional Ketosis for Weight Management and Reversal of Metabolic Syndrome. Curr. Nutr. Rep. 2018, 7, 97–106. [Google Scholar] [CrossRef]
- Sakurai, Y.; Kubota, N.; Yamauchi, T.; Kadowaki, T. Role of Insulin Resistance in MAFLD. Int. J. Mol. Sci. 2021, 22, 4156. [Google Scholar] [CrossRef]
- DiStefano, J.K.; Gerhard, G.S. NAFLD in Normal Weight Individuals. Diabetol. Metab. Syndr. 2022, 14, 45. [Google Scholar] [CrossRef] [PubMed]
- Parra-Vargas, M.; Rodriguez-Echevarria, R.; Jimenez-Chillaron, J.C. Nutritional Approaches for the Management of Nonalcoholic Fatty Liver Disease: An Evidence-Based Review. Nutrients 2020, 12, 3860. [Google Scholar] [CrossRef]
- Bauer, K.C.; Littlejohn, P.T.; Ayala, V.; Creus-Cuadros, A.; Finlay, B.B. Nonalcoholic Fatty Liver Disease and the Gut-Liver Axis: Exploring an Undernutrition Perspective. Gastroenterology 2022, 162, 1858–1875.e2. [Google Scholar] [CrossRef]
- Moore, M.P.; Cunningham, R.P.; Dashek, R.J.; Mucinski, J.M.; Rector, R.S. A Fad Too Far? Dietary Strategies for the Prevention and Treatment of NAFLD. Obesity 2020, 28, 1843–1852. [Google Scholar] [CrossRef]
- Worm, N. Beyond Body Weight-Loss: Dietary Strategies Targeting Intrahepatic Fat in NAFLD. Nutrients 2020, 12, 1316. [Google Scholar] [CrossRef]
- Chen, F.; Esmaili, S.; Rogers, G.B.; Bugianesi, E.; Petta, S.; Marchesini, G.; Bayoumi, A.; Metwally, M.; Azardaryany, M.K.; Coulter, S.; et al. Lean NAFLD: A Distinct Entity Shaped by Differential Metabolic Adaptation. Hepatology 2020, 71, 1213–1227. [Google Scholar] [CrossRef] [PubMed]
- Fitriakusumah, Y.; Lesmana, C.R.A.; Bastian, W.P.; Jasirwan, C.O.M.; Hasan, I.; Simadibrata, M.; Kurniawan, J.; Sulaiman, A.S.; Gani, R.A. The Role of Small Intestinal Bacterial Overgrowth (SIBO) in Non-Alcoholic Fatty Liver Disease (NAFLD) Patients Evaluated Using Controlled Attenuation Parameter (CAP) Transient Elastography (TE): A Tertiary Referral Center Experience. BMC Gastroenterol. 2019, 19, 43. [Google Scholar] [CrossRef] [PubMed]
- Kessoku, T.; Kobayashi, T.; Imajo, K.; Tanaka, K.; Yamamoto, A.; Takahashi, K.; Kasai, Y.; Ozaki, A.; Iwaki, M.; Nogami, A.; et al. Endotoxins and Non-Alcoholic Fatty Liver Disease. Front. Endocrinol. 2021, 12, 770986. [Google Scholar] [CrossRef] [PubMed]
- Safari, Z.; Gérard, P. The Links between the Gut Microbiome and Non-Alcoholic Fatty Liver Disease (NAFLD). Cell. Mol. Life Sci. 2019, 76, 1541–1558. [Google Scholar] [CrossRef]
- Frost, F.; Kacprowski, T.; Rühlemann, M.; Pietzner, M.; Bang, C.; Franke, A.; Nauck, M.; Völker, U.; Völzke, H.; Dörr, M.; et al. Long-Term Instability of the Intestinal Microbiome Is Associated with Metabolic Liver Disease, Low Microbiota Diversity, Diabetes Mellitus and Impaired Exocrine Pancreatic Function. Gut 2021, 70, 522–530. [Google Scholar] [CrossRef] [PubMed]
- An, L.; Wirth, U.; Koch, D.; Schirren, M.; Drefs, M.; Koliogiannis, D.; Nieß, H.; Andrassy, J.; Guba, M.; Bazhin, A.V.; et al. The Role of Gut-Derived Lipopolysaccharides and the Intestinal Barrier in Fatty Liver Diseases. J. Gastrointest. Surg. 2022, 26, 671–683. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.-C.; Yeh, W.-C.; Ohashi, P.S. LPS/TLR4 Signal Transduction Pathway. Cytokine 2008, 42, 145–151. [Google Scholar] [CrossRef]
- Plauth, M.; Bernal, W.; Dasarathy, S.; Merli, M.; Plank, L.D.; Schütz, T.; Bischoff, S.C. ESPEN Guideline on Clinical Nutrition in Liver Disease. Clin. Nutr. 2019, 38, 485–521. [Google Scholar] [CrossRef]
- Abstracts—APASL 2013. Hepatol. Int. 2013, 7 (Suppl. 1), 1–754. [CrossRef]
- AASLD Abstracts. Hepatology 2012, 56, 191A–1144A. [CrossRef]
- Associazione Italiana per lo Studio del Fegato (AISF); Società Italiana di Diabetologia (SID); Società Italiana dell’Obesità (SIO); Members of the Guidelines Panel; Coordinator; AISF Members; SID Members; SIO Members. Metodologists Non-Alcoholic Fatty Liver Disease in Adults 2021: A Clinical Practice Guideline of the Italian Association for the Study of the Liver (AISF), the Italian Society of Diabetology (SID) and the Italian Society of Obesity (SIO). Nutr. Metab. Cardiovasc. Dis. 2022, 32, 1–16. [Google Scholar] [CrossRef] [PubMed]
- da Silva Ferreira, G.; Catanozi, S.; Passarelli, M. Dietary Sodium and Nonalcoholic Fatty Liver Disease: A Systematic Review. Antioxidants 2023, 12, 599. [Google Scholar] [CrossRef] [PubMed]
- Wadhawan, M.; Anand, A.C. Coffee and Liver Disease. J. Clin. Exp. Hepatol. 2016, 6, 40–46. [Google Scholar] [CrossRef]
- Cani, P.D.; Van Hul, M. Mediterranean Diet, Gut Microbiota and Health: When Age and Calories Do Not Add Up! Gut 2020, 69, 1167–1168. [Google Scholar] [CrossRef]
- Abenavoli, L.; Boccuto, L.; Federico, A.; Dallio, M.; Loguercio, C.; Di Renzo, L.; De Lorenzo, A. Diet and Non-Alcoholic Fatty Liver Disease: The Mediterranean Way. Int. J. Environ. Res. Public Health 2019, 16, 3011. [Google Scholar] [CrossRef]
- Cândido, F.G.; Valente, F.X.; Grześkowiak, Ł.M.; Moreira, A.P.B.; Rocha, D.M.U.P.; Alfenas, R.D.C.G. Impact of Dietary Fat on Gut Microbiota and Low-Grade Systemic Inflammation: Mechanisms and Clinical Implications on Obesity. Int. J. Food Sci. Nutr. 2018, 69, 125–143. [Google Scholar] [CrossRef]
- Yaskolka Meir, A.; Rinott, E.; Tsaban, G.; Zelicha, H.; Kaplan, A.; Rosen, P.; Shelef, I.; Youngster, I.; Shalev, A.; Blüher, M.; et al. Effect of Green-Mediterranean Diet on Intrahepatic Fat: The DIRECT PLUS Randomised Controlled Trial. Gut 2021, 70, 2085–2095. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Guisado, J.; Muñoz-Serrano, A. The Effect of the Spanish Ketogenic Mediterranean Diet on Nonalcoholic Fatty Liver Disease: A Pilot Study. J. Med. Food 2011, 14, 677–680. [Google Scholar] [CrossRef]
- Xie, Z.; Sun, Y.; Ye, Y.; Hu, D.; Zhang, H.; He, Z.; Zhao, H.; Yang, H.; Mao, Y. Randomized Controlled Trial for Time-Restricted Eating in Healthy Volunteers without Obesity. Nat. Commun. 2022, 13, 1003. [Google Scholar] [CrossRef]
- Jamshed, H.; Beyl, R.A.; Della Manna, D.L.; Yang, E.S.; Ravussin, E.; Peterson, C.M. Early Time-Restricted Feeding Improves 24-Hour Glucose Levels and Affects Markers of the Circadian Clock, Aging, and Autophagy in Humans. Nutrients 2019, 11, 1234. [Google Scholar] [CrossRef]
- Sutton, E.F.; Beyl, R.; Early, K.S.; Cefalu, W.T.; Ravussin, E.; Peterson, C.M. Early Time-Restricted Feeding Improves Insulin Sensitivity, Blood Pressure, and Oxidative Stress Even without Weight Loss in Men with Prediabetes. Cell Metab. 2018, 27, 1212–1221.e3. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-H.; Fu, Y.; Yang, S.-J.; Chi, C.-C. Effects of Omega-3 Polyunsaturated Fatty Acid Supplementation on Non-Alcoholic Fatty Liver: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 2769. [Google Scholar] [CrossRef]
- Medina-Urrutia, A.; Lopez-Uribe, A.R.; El Hafidi, M.; González-Salazar, M.D.C.; Posadas-Sánchez, R.; Jorge-Galarza, E.; Del Valle-Mondragón, L.; Juárez-Rojas, J.G. Chia (Salvia hispanica)-Supplemented Diet Ameliorates Non-Alcoholic Fatty Liver Disease and Its Metabolic Abnormalities in Humans. Lipids Health Dis. 2020, 19, 96. [Google Scholar] [CrossRef]
- Scorletti, E.; Afolabi, P.R.; Miles, E.A.; Smith, D.E.; Almehmadi, A.; Alshathry, A.; Childs, C.E.; Del Fabbro, S.; Bilson, J.; Moyses, H.E.; et al. Synbiotics Alter Fecal Microbiomes, But Not Liver Fat or Fibrosis, in a Randomized Trial of Patients with Nonalcoholic Fatty Liver Disease. Gastroenterology 2020, 158, 1597–1610.e7. [Google Scholar] [CrossRef]
- Asadi-Samani, M.; Kafash-Farkhad, N.; Azimi, N.; Fasihi, A.; Alinia-Ahandani, E.; Rafieian-Kopaei, M. Medicinal Plants with Hepatoprotective Activity in Iranian Folk Medicine. Asian Pac. J. Trop. Biomed. 2015, 5, 146–157. [Google Scholar] [CrossRef]
- Kaur, A.P.; Bhardwaj, S.; Dhanjal, D.S.; Nepovimova, E.; Cruz-Martins, N.; Kuča, K.; Chopra, C.; Singh, R.; Kumar, H.; Șen, F.; et al. Plant Prebiotics and Their Role in the Amelioration of Diseases. Biomolecules 2021, 11, 440. [Google Scholar] [CrossRef] [PubMed]
- Almatroodi, S.A.; Anwar, S.; Almatroudi, A.; Khan, A.A.; Alrumaihi, F.; Alsahli, M.A.; Rahmani, A.H. Hepatoprotective Effects of Garlic Extract against Carbon Tetrachloride (CCl4)-Induced Liver Injury via Modulation of Antioxidant, Anti-Inflammatory Activities and Hepatocyte Architecture. Appl. Sci. 2020, 10, 6200. [Google Scholar] [CrossRef]
- Dwivedi, S.; Sahrawat, K.; Puppala, N.; Ortiz, R. Plant Prebiotics and Human Health: Biotechnology to Breed Prebiotic-Rich Nutritious Food Crops. Electron. J. Biotechnol. 2014, 17, 238–245. [Google Scholar] [CrossRef]
- Ozougwa, J.C.; Eyo, J.E. Hepatoprotective Effects of Allium Cepa (Onion) Extracts against Paracetamol-Induced Liver Damage in Rats. Afr. J. Biotechnol. 2014, 13, 2679–2688. [Google Scholar] [CrossRef]
- Chakraborty, R.; Vickery, K.; Darido, C.; Ranganathan, S.; Hu, H. Bacterial Antigens Reduced the Inhibition Effect of Capsaicin on Cal 27 Oral Cancer Cell Proliferation. Int. J. Mol. Sci. 2021, 22, 8686. [Google Scholar] [CrossRef]
- Eswar Kumar, K.; Harsha, K.N.; Sudheer, V.; Giri Babu, N. In Vitro Antioxidant Activity and in Vivo Hepatoprotective Activity of Aqueous Extract of Allium Cepa Bulb in Ethanol Induced Liver Damage in Wistar Rats. Food Sci. Hum. Wellness 2013, 2, 132–138. [Google Scholar] [CrossRef]
- Krepkova, L.V.; Babenko, A.N.; Saybel’, O.L.; Lupanova, I.A.; Kuzina, O.S.; Job, K.M.; Sherwin, C.M.; Enioutina, E.Y. Valuable Hepatoprotective Plants—How Can We Optimize Waste Free Uses of Such Highly Versatile Resources? Front. Pharmacol. 2021, 12, 738504. [Google Scholar] [CrossRef] [PubMed]
- Costea, L.; Chițescu, C.L.; Boscencu, R.; Ghica, M.; Lupuliasa, D.; Mihai, D.P.; Deculescu-Ioniță, T.; Duțu, L.E.; Popescu, M.L.; Luță, E.-A.; et al. The Polyphenolic Profile and Antioxidant Activity of Five Vegetal Extracts with Hepatoprotective Potential. Plants 2022, 11, 1680. [Google Scholar] [CrossRef]
- Mahboubi, M.; Mahboubi, M. Hepatoprotection by Dandelion (Taraxacum officinale) and Mechanisms. Asian Pac. J. Trop. Biomed. 2020, 10, 1. [Google Scholar] [CrossRef]
- Kamal, F.Z.; Lefter, R.; Mihai, C.-T.; Farah, H.; Ciobica, A.; Ali, A.; Radu, I.; Mavroudis, I.; Ech-Chahad, A. Chemical Composition, Antioxidant and Antiproliferative Activities of Taraxacum officinale Essential Oil. Molecules 2022, 27, 6477. [Google Scholar] [CrossRef] [PubMed]
- Ghorbel Koubaa, F.; Chaâbane, M.; Choura, B.; Turki, M.; Makni-Ayadi, F.; El Feki, A. Hepatoprotective Effects of Taraxacum officinale Root Extract on Permethrin-Induced Liver Toxicity in Adult Mice. Pharm. Biomed. Res. 2020, 6, 223–236. [Google Scholar] [CrossRef]
- Maletha, D.; Singh, S.P.; Ramanarayanan, S. Hepatoprotective and Nephroprotective Efficacy of Cichorium intybus Following Imidacloprid Induced Subchronic Toxicity in WLH Cockerels. Indian J. Anim. Sci. 2022, 92, 940–945. [Google Scholar] [CrossRef]
- Fatima, N.; Akhtar, T.; Sheikh, N. Prebiotics: A Novel Approach to Treat Hepatocellular Carcinoma. Can. J. Gastroenterol. Hepatol. 2017, 2017, e6238106. [Google Scholar] [CrossRef]
- Yang, L.; Kang, X.; Dong, W.; Wang, L.; Liu, S.; Zhong, X.; Liu, D. Prebiotic Properties of Ganoderma lucidum Polysaccharides with Special Enrichment of Bacteroides ovatus and B. uniformis in Vitro. J. Funct. Foods 2022, 92, 105069. [Google Scholar] [CrossRef]
- Lv, X.-C.; Wu, Q.; Cao, Y.-J.; Lin, Y.-C.; Guo, W.-L.; Rao, P.-F.; Zhang, Y.-Y.; Chen, Y.-T.; Ai, L.-Z.; Ni, L. Ganoderic Acid A from Ganoderma lucidum Protects against Alcoholic Liver Injury through Ameliorating the Lipid Metabolism and Modulating the Intestinal Microbial Composition. Food Funct. 2022, 13, 5820–5837. [Google Scholar] [CrossRef]
- Peng, H.; Zhong, L.; Cheng, L.; Chen, L.; Tong, R.; Shi, J.; Bai, L. Ganoderma lucidum: Current Advancements of Characteristic Components and Experimental Progress in Anti-Liver Fibrosis. Front. Pharmacol. 2023, 13, 1094405. [Google Scholar] [CrossRef]
- Chen, T.Q.; Wu, J.-G.; Kan, Y.-J.; Yang, C.; Wu, Y.-B.; Wu, J.-Z. Antioxidant and Hepatoprotective Activities of Crude Polysaccharide Extracts from Lingzhi or Reishi Medicinal Mushroom, Ganoderma lucidum (Agaricomycetes), by Ultrasonic-Circulating Extraction. Int. J. Med. Mushrooms 2018, 20, 581–593. [Google Scholar] [CrossRef]
- Lu, C.; Lee, B.H.; Ren, Y.; Ji, D.; Rao, S.; Li, H.; Yang, Z. Effects of Exopolysaccharides from Antrodia cinnamomea on Inflammation and Intestinal Microbiota Disturbance Induced by Antibiotics in Mice. Food Biosci. 2022, 50, 102116. [Google Scholar] [CrossRef]
- Tsai, Y.-T.; Ruan, J.-W.; Chang, C.-S.; Ko, M.-L.; Chou, H.-C.; Lin, C.-C.; Lin, C.-M.; Huang, C.-T.; Wei, Y.-S.; Liao, E.-C.; et al. Antrodia cinnamomea Confers Obesity Resistance and Restores Intestinal Barrier Integrity in Leptin-Deficient Obese Mice. Nutrients 2020, 12, 726. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.-Y.; Kuan, C.-M.; Hsu, P.-K. Hepatoprotective Effect of Antrodia cinnamomea Mycelia Extract in Subhealth Japanese Adults: A Randomized, Double-Blind, Placebo-Controlled Clinical Study. J. Diet. Suppl. 2022. [Google Scholar] [CrossRef] [PubMed]
- Chiou, Y.-L.; Chyau, C.-C.; Li, T.-J.; Kuo, C.-F.; Kang, Y.-Y.; Chen, C.-C.; Ko, W.-S. Hepatoprotective Effect of Antrodia cinnamomea Mycelium in Patients with Nonalcoholic Steatohepatitis: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Am. Coll. Nutr. 2021, 40, 349–357. [Google Scholar] [CrossRef]
- Geethangili, M.; Tzeng, Y.-M. Review of Pharmacological Effects of Antrodia Camphorata and Its Bioactive Compounds. Evid. Based Complement. Altern. Med. 2011, 2011, 212641. [Google Scholar] [CrossRef]
- Liu, Y.-W.; Lu, K.-H.; Ho, C.-T.; Sheen, L.-Y. Protective Effects of Antrodia cinnamomea against Liver Injury. J. Tradit. Complement. Med. 2012, 2, 284–294. [Google Scholar] [CrossRef]
- Ye, J.; Zhang, C.; Fan, Q.; Lin, X.; Wang, Y.; Azzam, M.; Alhotan, R.; Alqhtani, A.; Jiang, S. Antrodia cinnamomea Polysaccharide Improves Liver Antioxidant, Anti-Inflammatory Capacity, and Cecal Flora Structure of Slow-Growing Broiler Breeds Challenged with Lipopolysaccharide. Front. Vet. Sci. 2022, 9, 994782. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Nichols, R.G.; Roy, P.; Gui, W.; Smith, P.B.; Zhang, J.; Lin, Y.; Weaver, V.; Cai, J.; Patterson, A.D.; et al. Prebiotic Effects of White Button Mushroom (Agaricus bisporus) Feeding on Succinate and Intestinal Gluconeogenesis in C57BL/6 Mice. J. Funct. Foods 2018, 45, 223–232. [Google Scholar] [CrossRef]
- Hess, J.; Wang, Q.; Gould, T.; Slavin, J. Impact of Agaricus bisporus Mushroom Consumption on Gut Health Markers in Healthy Adults. Nutrients 2018, 10, 1402. [Google Scholar] [CrossRef] [PubMed]
- García-Sanmartín, J.; Bobadilla, M.; Mirpuri, E.; Grifoll, V.; Pérez-Clavijo, M.; Martínez, A. Agaricus Mushroom-Enriched Diets Modulate the Microbiota-Gut-Brain Axis and Reduce Brain Oxidative Stress in Mice. Antioxidants 2022, 11, 695. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zheng, D.; Su, L.; Wang, Q.; Li, Y. Protective Effect of Polysaccharide from Agaricus bisporus in Tibet Area of China against Tetrachloride-Induced Acute Liver Injury in Mice. Int. J. Biol. Macromol. 2018, 118, 1488–1493. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Li, J.; Zhang, J.; Wang, W.; Wang, X.; Jing, H.; Ren, Z.; Gao, Z.; Song, X.; Gong, Z.; et al. The Antioxidative, Antiaging, and Hepatoprotective Effects of Alkali-Extractable Polysaccharides by Agaricus bisporus. Evid. Based Complement. Altern. Med. 2017, 2017, e7298683. [Google Scholar] [CrossRef]
- Huang, Y.; Gao, Y.; Pi, X.; Zhao, S.; Liu, W. In Vitro Hepatoprotective and Human Gut Microbiota Modulation of Polysaccharide-Peptides in Pleurotus citrinopileatus. Front. Cell. Infect. Microbiol. 2022, 12, 892049. [Google Scholar] [CrossRef]
- Sawangwan, T.; Wansanit, W.; Pattani, L.; Noysang, C. Study of Prebiotic Properties from Edible Mushroom Extraction. Agric. Nat. Resour. 2018, 52, 519–524. [Google Scholar] [CrossRef]
- Chi, Q.; Wang, G.; Sheng, Y.; Xu, W.; Shi, P.; Zhao, C.; Huang, K. Ethanolic Extract of the Golden Oyster Mushroom, Pleurotus citrinopileatus (Agaricomycetes), Alleviates Metabolic Syndrome in Diet-Induced Obese Mice. Int. J. Med. Mushrooms 2017, 19, 1001–1008. [Google Scholar] [CrossRef]
- Tena-Garitaonaindia, M.; Ceacero-Heras, D.; Montoro, M.D.M.M.; de Medina, F.S.; Martínez-Augustin, O.; Daddaoua, A. A Standardized Extract of Lentinula Edodes Cultured Mycelium Inhibits Pseudomonas aeruginosa Infectivity Mechanisms. Front. Microbiol. 2022, 13, 814448. [Google Scholar] [CrossRef]
- Song, X.; Cai, W.; Ren, Z.; Jia, L.; Zhang, J. Antioxidant and Hepatoprotective Effects of Acidic-Hydrolysis Residue Polysaccharides from Shiitake Culinary-Medicinal Mushroom Lentinus edodes (Agaricomycetes) in Mice. IJM Int. J. Med. Mushrooms 2021, 23, 85–96. [Google Scholar] [CrossRef]
- Ponnusamy, C.; Uddandrao, V.V.S.; Pudhupalayam, S.P.; Singaravel, S.; Periyasamy, T.; Ponnusamy, P. Lentinula Edodes (Edible Mushroom) as a Nutraceutical: A Review. Biosci. Biotechnol. Res. Asia 2022, 19, 1–11. [Google Scholar] [CrossRef]
- D’Amico, G.; Garcia-Tsao, G.; Pagliaro, L. Natural History and Prognostic Indicators of Survival in Cirrhosis: A Systematic Review of 118 Studies. J. Hepatol. 2006, 44, 217–231. [Google Scholar] [CrossRef] [PubMed]
- Traub, J.; Reiss, L.; Aliwa, B.; Stadlbauer, V. Malnutrition in Patients with Liver Cirrhosis. Nutrients 2021, 13, 540. [Google Scholar] [CrossRef] [PubMed]
- Prieto-Frías, C.; Conchillo, M.; Payeras, M.; Iñarrairaegui, M.; Davola, D.; Frühbeck, G.; Salvador, J.; Rodríguez, M.; Richter, J.Á.; Mugueta, C.; et al. Factors Related to Increased Resting Energy Expenditure in Men with Liver Cirrhosis. Eur. J. Gastroenterol. Hepatol. 2016, 28, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Plank, L.D.; McCall, J.L.; Gillanders, L.K.; McIlroy, K.; Gane, E.J. Body Composition, Muscle Function, and Energy Expenditure in Patients with Liver Cirrhosis: A Comprehensive Study. Am. J. Clin. Nutr. 2007, 85, 1257–1266. [Google Scholar] [CrossRef]
- Guglielmi, F.W.; Panella, C.; Buda, A.; Budillon, G.; Caregaro, L.; Clerici, C.; Conte, D.; Federico, A.; Gasbarrini, G.; Guglielmi, A.; et al. Nutritional State and Energy Balance in Cirrhotic Patients with or without Hypermetabolism. Multicentre Prospective Study by the “Nutritional Problems in Gastroenterology” Section of the Italian Society of Gastroenterology (SIGE). Dig. Liver Dis. 2005, 37, 681–688. [Google Scholar] [CrossRef]
- Bhanji, R.A.; Carey, E.J.; Yang, L.; Watt, K.D. The Long Winding Road to Transplant: How Sarcopenia and Debility Impact Morbidity and Mortality on the Waitlist. Clin. Gastroenterol. Hepatol. 2017, 15, 1492–1497. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European Consensus on Definition and Diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Kim, G.; Kang, S.H.; Kim, M.Y.; Baik, S.K. Prognostic Value of Sarcopenia in Patients with Liver Cirrhosis: A Systematic Review and Meta-Analysis. PLoS ONE 2017, 12, e0186990. [Google Scholar] [CrossRef]
- Janssen, I. Influence of Sarcopenia on the Development of Physical Disability: The Cardiovascular Health Study. J. Am. Geriatr. Soc. 2006, 54, 56–62. [Google Scholar] [CrossRef]
- van Vugt, J.L.A.; Levolger, S.; de Bruin, R.W.F.; van Rosmalen, J.; Metselaar, H.J.; IJzermans, J.N.M. Systematic Review and Meta-Analysis of the Impact of Computed Tomography-Assessed Skeletal Muscle Mass on Outcome in Patients Awaiting or Undergoing Liver Transplantation. Am. J. Transpl. 2016, 16, 2277–2292. [Google Scholar] [CrossRef]
- Chang, K.-V.; Chen, J.-D.; Wu, W.-T.; Huang, K.-C.; Lin, H.-Y.; Han, D.-S. Is Sarcopenia Associated with Hepatic Encephalopathy in Liver Cirrhosis? A Systematic Review and Meta-Analysis. J. Formos. Med. Assoc. 2019, 118, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Lucero, C.; Verna, E.C. The Role of Sarcopenia and Frailty in Hepatic Encephalopathy Management. Clin. Liver Dis. 2015, 19, 507–528. [Google Scholar] [CrossRef] [PubMed]
- Tapper, E.B.; Jiang, Z.G.; Patwardhan, V.R. Refining the Ammonia Hypothesis: A Physiology-Driven Approach to the Treatment of Hepatic Encephalopathy. Mayo Clin. Proc. 2015, 90, 646–658. [Google Scholar] [CrossRef] [PubMed]
- Berzigotti, A.; Garcia-Tsao, G.; Bosch, J.; Grace, N.D.; Burroughs, A.K.; Morillas, R.; Escorsell, A.; Garcia-Pagan, J.C.; Patch, D.; Matloff, D.S.; et al. Obesity Is an Independent Risk Factor for Clinical Decompensation in Patients with Cirrhosis. Hepatology 2011, 54, 555–561. [Google Scholar] [CrossRef]
- Montano-Loza, A.J.; Angulo, P.; Meza-Junco, J.; Prado, C.M.M.; Sawyer, M.B.; Beaumont, C.; Esfandiari, N.; Ma, M.; Baracos, V.E. Sarcopenic Obesity and Myosteatosis Are Associated with Higher Mortality in Patients with Cirrhosis. J. Cachexia Sarcopenia Muscle 2016, 7, 126–135. [Google Scholar] [CrossRef]
- Tantai, X.; Liu, Y.; Yeo, Y.H.; Praktiknjo, M.; Mauro, E.; Hamaguchi, Y.; Engelmann, C.; Zhang, P.; Jeong, J.Y.; van Vugt, J.L.A.; et al. Effect of Sarcopenia on Survival in Patients with Cirrhosis: A Meta-Analysis. J. Hepatol. 2022, 76, 588–599. [Google Scholar] [CrossRef] [PubMed]
- Wijarnpreecha, K.; Panjawatanan, P.; Thongprayoon, C.; Jaruvongvanich, V.; Ungprasert, P. Sarcopenia and Risk of Nonalcoholic Fatty Liver Disease: A Meta-Analysis. Saudi J. Gastroenterol. 2018, 24, 12–17. [Google Scholar] [CrossRef]
- Laube, R.; Wang, H.; Park, L.; Heyman, J.K.; Vidot, H.; Majumdar, A.; Strasser, S.I.; McCaughan, G.W.; Liu, K. Frailty in Advanced Liver Disease. Liver Int. 2018, 38, 2117–2128. [Google Scholar] [CrossRef]
- Periyalwar, P.; Dasarathy, S. Malnutrition in Cirrhosis: Contribution and Consequences of Sarcopenia on Metabolic and Clinical Responses. Clin. Liver Dis. 2012, 16, 95–131. [Google Scholar] [CrossRef]
- Sam, J.; Nguyen, G.C. Protein-Calorie Malnutrition as a Prognostic Indicator of Mortality among Patients Hospitalized with Cirrhosis and Portal Hypertension. Liver Int. 2009, 29, 1396–1402. [Google Scholar] [CrossRef]
- Bunchorntavakul, C.; Supanun, R.; Atsawarungruangkit, A. Nutritional Status and Its Impact on Clinical Outcomes for Patients Admitted to Hospital with Cirrhosis. J. Med. Assoc. Thail. 2016, 99 (Suppl. 2), S47–S55. [Google Scholar]
- Maharshi, S.; Sharma, B.C.; Srivastava, S. Malnutrition in Cirrhosis Increases Morbidity and Mortality. J. Gastroenterol. Hepatol. 2015, 30, 1507–1513. [Google Scholar] [CrossRef]
- Ribeiro, H.S.; Maurício, S.F.; Antônio da Silva, T.; de Vasconcelos Generoso, S.; Lima, A.S.; Toulson Davisson Correia, M.I. Combined Nutritional Assessment Methods to Predict Clinical Outcomes in Patients on the Waiting List for Liver Transplantation. Nutrition 2018, 47, 21–26. [Google Scholar] [CrossRef]
- Huisman, E.J.; Trip, E.J.; Siersema, P.D.; van Hoek, B.; van Erpecum, K.J. Protein Energy Malnutrition Predicts Complications in Liver Cirrhosis. Eur. J. Gastroenterol. Hepatol. 2011, 23, 982–989. [Google Scholar] [CrossRef]
- Ruiz-Margáin, A.; Macías-Rodríguez, R.U.; Ampuero, J.; Cubero, F.J.; Chi-Cervera, L.; Ríos-Torres, S.L.; Duarte-Rojo, A.; Espinosa-Cuevas, Á.; Romero-Gómez, M.; Torre, A. Low Phase Angle Is Associated with the Development of Hepatic Encephalopathy in Patients with Cirrhosis. World J. Gastroenterol. 2016, 22, 10064–10070. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, C.; Majeed, A.; Wahlin, S. Body Composition Assessed by Dual-Energy X-ray Absorptiometry Predicts Early Infectious Complications after Liver Transplantation. J. Hum. Nutr. Diet 2017, 30, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Johnston, H.E.; Takefala, T.G.; Kelly, J.T.; Keating, S.E.; Coombes, J.S.; Macdonald, G.A.; Hickman, I.J.; Mayr, H.L. The Effect of Diet and Exercise Interventions on Body Composition in Liver Cirrhosis: A Systematic Review. Nutrients 2022, 14, 3365. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Wang, J. The Efficacy of Parenteral Nutrition (PN) and Enteral Nutrition (EN) Supports in Cirrhosis: A Systematic Review and Network Meta-Analysis. Medicine 2022, 101, e28618. [Google Scholar] [CrossRef]
- Bowen, T.S.; Schuler, G.; Adams, V. Skeletal Muscle Wasting in Cachexia and Sarcopenia: Molecular Pathophysiology and Impact of Exercise Training. J. Cachexia Sarcopenia Muscle 2015, 6, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Williams, F.R.; Berzigotti, A.; Lord, J.M.; Lai, J.C.; Armstrong, M.J. Review Article: Impact of Exercise on Physical Frailty in Patients with Chronic Liver Disease. Aliment. Pharmacol. Ther. 2019, 50, 988–1000. [Google Scholar] [CrossRef]
- Toshikuni, N.; Arisawa, T.; Tsutsumi, M. Nutrition and Exercise in the Management of Liver Cirrhosis. World J. Gastroenterol. 2014, 20, 7286–7297. [Google Scholar] [CrossRef]
- Plank, L.D.; Gane, E.J.; Peng, S.; Muthu, C.; Mathur, S.; Gillanders, L.; McIlroy, K.; Donaghy, A.J.; McCall, J.L. Nocturnal Nutritional Supplementation Improves Total Body Protein Status of Patients with Liver Cirrhosis: A Randomized 12-Month Trial. Hepatology 2008, 48, 557–566. [Google Scholar] [CrossRef]
- Tsien, C.D.; McCullough, A.J.; Dasarathy, S. Late Evening Snack: Exploiting a Period of Anabolic Opportunity in Cirrhosis. J. Gastroenterol. Hepatol. 2012, 27, 430–441. [Google Scholar] [CrossRef]
- Guo, Y.-J.; Tian, Z.-B.; Jiang, N.; Ding, X.-L.; Mao, T.; Jing, X. Effects of Late Evening Snack on Cirrhotic Patients: A Systematic Review and Meta-Analysis. Gastroenterol. Res. Pract. 2018, 2018, 9189062. [Google Scholar] [CrossRef]
- Verboeket-van de Venne, W.P.; Westerterp, K.R.; van Hoek, B.; Swart, G.R. Energy Expenditure and Substrate Metabolism in Patients with Cirrhosis of the Liver: Effects of the Pattern of Food Intake. Gut 1995, 36, 110–116. [Google Scholar] [CrossRef][Green Version]
- Zenith, L.; Meena, N.; Ramadi, A.; Yavari, M.; Harvey, A.; Carbonneau, M.; Ma, M.; Abraldes, J.G.; Paterson, I.; Haykowsky, M.J.; et al. Eight Weeks of Exercise Training Increases Aerobic Capacity and Muscle Mass and Reduces Fatigue in Patients with Cirrhosis. Clin. Gastroenterol. Hepatol. 2014, 12, 1920–1926.e2. [Google Scholar] [CrossRef]
- Aamann, L.; Dam, G.; Borre, M.; Drljevic-Nielsen, A.; Overgaard, K.; Andersen, H.; Vilstrup, H.; Aagaard, N.K. Resistance Training Increases Muscle Strength and Muscle Size in Patients with Liver Cirrhosis. Clin. Gastroenterol. Hepatol. 2020, 18, 1179–1187.e6. [Google Scholar] [CrossRef]
- Kawaguchi, T.; Izumi, N.; Charlton, M.R.; Sata, M. Branched-Chain Amino Acids as Pharmacological Nutrients in Chronic Liver Disease. Hepatology 2011, 54, 1063–1070. [Google Scholar] [CrossRef]
- Ichikawa, K.; Okabayashi, T.; Maeda, H.; Namikawa, T.; Iiyama, T.; Sugimoto, T.; Kobayashi, M.; Mimura, T.; Hanazaki, K. Oral Supplementation of Branched-Chain Amino Acids Reduces Early Recurrence after Hepatic Resection in Patients with Hepatocellular Carcinoma: A Prospective Study. Surg. Today 2013, 43, 720–726. [Google Scholar] [CrossRef]
- Merli, M.; Iebba, V.; Giusto, M. What Is New about Diet in Hepatic Encephalopathy. Metab. Brain Dis. 2016, 31, 1289–1294. [Google Scholar] [CrossRef]
- Agarwal, A.; Avarebeel, S.; Choudhary, N.S.; Goudar, M.; Tejaswini, C.J. Correlation of Trace Elements in Patients of Chronic Liver Disease with Respect to Child-Turcotte-Pugh Scoring System. J. Clin. Diagn. Res. 2017, 11, OC25–OC28. [Google Scholar] [CrossRef]
- Loguercio, C.; De Girolamo, V.; Federico, A.; Feng, S.L.; Crafa, E.; Cataldi, V.; Gialanella, G.; Moro, R.; Del Vecchio Blanco, C. Relationship of Blood Trace Elements to Liver Damage, Nutritional Status, and Oxidative Stress in Chronic Nonalcoholic Liver Disease. Biol. Trace Elem. Res. 2001, 81, 245–254. [Google Scholar] [CrossRef]
- Yoshida, Y.; Higashi, T.; Nouso, K.; Nakatsukasa, H.; Nakamura, S.I.; Watanabe, A.; Tsuji, T. Effects of Zinc Deficiency/Zinc Supplementation on Ammonia Metabolism in Patients with Decompensated Liver Cirrhosis. Acta Med. Okayama 2001, 55, 349–355. [Google Scholar] [CrossRef]
- Nangliya, V.; Sharma, A.; Yadav, D.; Sunder, S.; Nijhawan, S.; Mishra, S. Study of Trace Elements in Liver Cirrhosis Patients and Their Role in Prognosis of Disease. Biol. Trace Elem. Res. 2015, 165, 35–40. [Google Scholar] [CrossRef]
- Rahelić, D.; Kujundzić, M.; Romić, Z.; Brkić, K.; Petrovecki, M. Serum Concentration of Zinc, Copper, Manganese and Magnesium in Patients with Liver Cirrhosis. Coll. Antropol. 2006, 30, 523–528. [Google Scholar]
- Teriaky, A.; Mosli, M.; Chandok, N.; Al-Judaibi, B.; Marotta, P.; Qumosani, K. Prevalence of Fat-Soluble Vitamin (A, D, and E) and Zinc Deficiency in Patients with Cirrhosis Being Assessed for Liver Transplantation. Acta Gastroenterol. Belg. 2017, 80, 237–241. [Google Scholar]
- Sanders, K.M.; Scott, D.; Ebeling, P.R. Vitamin D Deficiency and Its Role in Muscle-Bone Interactions in the Elderly. Curr. Osteoporos. Rep. 2014, 12, 74–81. [Google Scholar] [CrossRef]
- Nelson, J.E.; Roth, C.L.; Wilson, L.A.; Yates, K.P.; Aouizerat, B.; Morgan-Stevenson, V.; Whalen, E.; Hoofnagle, A.; Mason, M.; Gersuk, V.; et al. Vitamin D Deficiency Is Associated with Increased Risk of Non-Alcoholic Steatohepatitis in Adults with Non-Alcoholic Fatty Liver Disease: Possible Role for MAPK and NF-ΚB? Am. J. Gastroenterol. 2016, 111, 852–863. [Google Scholar] [CrossRef]
- Park, D.; Kwon, H.; Oh, S.W.; Joh, H.K.; Hwang, S.S.; Park, J.H.; Yun, J.M.; Lee, H.; Chung, G.E.; Ze, S.; et al. Is Vitamin D an Independent Risk Factor of Nonalcoholic Fatty Liver Disease? A Cross-Sectional Study of the Healthy Population. J. Korean Med. Sci. 2017, 32, 95–101. [Google Scholar] [CrossRef]
- Betrapally, N.S.; Gillevet, P.M.; Bajaj, J.S. Gut Microbiome and Liver Disease. Transl. Res. 2017, 179, 49–59. [Google Scholar] [CrossRef]
- Les, I.; Doval, E.; García-Martínez, R.; Planas, M.; Cárdenas, G.; Gómez, P.; Flavià, M.; Jacas, C.; Mínguez, B.; Vergara, M.; et al. Effects of Branched-Chain Amino Acids Supplementation in Patients with Cirrhosis and a Previous Episode of Hepatic Encephalopathy: A Randomized Study. Am. J. Gastroenterol. 2011, 106, 1081–1088. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, N. Selection for Liver Transplantation: Indications and Evaluation. Curr. Hepatol. Rep. 2020, 19, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Directive 2010/45/EU of the European Parliament and of the Council of 7 July 2010 on Standards of Quality and Safety of Human Organs Intended for Transplantation; Official Journal of the European Union: Luxembourg, 2010; Volume 207.
- Bakshi, N.; Singh, K. Diet and Nutrition Therapy in Pre-Liver Transplant Patients. Hepatoma Res. 2016, 2, 207–215. [Google Scholar] [CrossRef]
- Ney, M.; Abraldes, J.G.; Ma, M.; Belland, D.; Harvey, A.; Robbins, S.; Den Heyer, V.; Tandon, P. Insufficient Protein Intake Is Associated with Increased Mortality in 630 Patients with Cirrhosis Awaiting Liver Transplantation. Nutr. Clin. Pract. 2015, 30, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Bayramov, N.; Mammadova, S. A Review of the Current ERAS Guidelines for Liver Resection, Liver Transplantation and Pancreatoduodenectomy. Ann. Med. Surg. 2022, 82, 104596. [Google Scholar] [CrossRef]
- Bischoff, S.C.; Barazzoni, R.; Busetto, L.; Campmans-Kuijpers, M.; Cardinale, V.; Chermesh, I.; Eshraghian, A.; Kani, H.T.; Khannoussi, W.; Lacaze, L.; et al. European Guideline on Obesity Care in Patients with Gastrointestinal and Liver Diseases—Joint ESPEN/UEG Guideline. Clin. Nutr. 2022, 41, 2364–2405. [Google Scholar] [CrossRef]
- Ostrowska, J.; Jeznach-Steinhagen, A. Niedożywienie szpitalne. Metody oceny stanu odżywienia. Forum. Med. Rodz. 2017, 11, 54–61. [Google Scholar]
- Hammad, A.; Kaido, T.; Aliyev, V.; Mandato, C.; Uemoto, S. Nutritional Therapy in Liver Transplantation. Nutrients 2017, 9, 1126. [Google Scholar] [CrossRef]
- Brustia, R.; Monsel, A.; Skurzak, S.; Schiffer, E.; Carrier, F.M.; Patrono, D.; Kaba, A.; Detry, O.; Malbouisson, L.; Andraus, W.; et al. Guidelines for Perioperative Care for Liver Transplantation: Enhanced Recovery After Surgery (ERAS) Recommendations. Transplantation 2022, 106, 552–561. [Google Scholar] [CrossRef]
- Melloul, E.; Hübner, M.; Scott, M.; Snowden, C.; Prentis, J.; Dejong, C.H.C.; Garden, O.J.; Farges, O.; Kokudo, N.; Vauthey, J.-N.; et al. Guidelines for Perioperative Care for Liver Surgery: Enhanced Recovery After Surgery (ERAS) Society Recommendations. World J. Surg. 2016, 40, 2425–2440. [Google Scholar] [CrossRef]
- Song, G.-M.; Tian, X.; Zhang, L.; Ou, Y.-X.; Yi, L.-J.; Shuai, T.; Zhou, J.-G.; Zeng, Z.; Yang, H.-L. Immunonutrition Support for Patients Undergoing Surgery for Gastrointestinal Malignancy: Preoperative, Postoperative, or Perioperative? A Bayesian Network Meta-Analysis of Randomized Controlled Trials. Medicine 2015, 94, e1225. [Google Scholar] [CrossRef]
- Joliat, G.-R.; Kobayashi, K.; Hasegawa, K.; Thomson, J.-E.; Padbury, R.; Scott, M.; Brustia, R.; Scatton, O.; Tran Cao, H.S.; Vauthey, J.-N.; et al. Guidelines for Perioperative Care for Liver Surgery: Enhanced Recovery after Surgery (ERAS) Society Recommendations 2022. World J. Surg. 2023, 47, 11–34. [Google Scholar] [CrossRef] [PubMed]
- Weimann, A.; Breitenstein, S.; Breuer, J.P.; Gabor, S.E.; Holland-Cunz, S.; Kemen, M.; Längle, F.; Rayes, N.; Reith, B.; Rittler, P.; et al. Clinical nutrition in surgery: Guidelines of the German Society for Nutritional Medicine. Chirurg 2014, 85, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Fagiuoli, S.; Colli, A.; Bruno, R.; Craxì, A.; Gaeta, G.B.; Grossi, P.; Mondelli, M.U.; Puoti, M.; Sagnelli, E.; Stefani, S.; et al. Management of Infections Pre- and Post-Liver Transplantation: Report of an AISF Consensus Conference. J. Hepatol. 2014, 60, 1075–1089. [Google Scholar] [CrossRef] [PubMed]
- Kaido, T.; Mori, A.; Ogura, Y.; Ogawa, K.; Hata, K.; Yoshizawa, A.; Yagi, S.; Uemoto, S. Pre- and Perioperative Factors Affecting Infection after Living Donor Liver Transplantation. Nutrition 2012, 28, 1104–1108. [Google Scholar] [CrossRef] [PubMed]
- Shirabe, K.; Yoshimatsu, M.; Motomura, T.; Takeishi, K.; Toshima, T.; Muto, J.; Matono, R.; Taketomi, A.; Uchiyama, H.; Maehara, Y. Beneficial Effects of Supplementation with Branched-Chain Amino Acids on Postoperative Bacteremia in Living Donor Liver Transplant Recipients. Liver Transplant. 2011, 17, 1073–1080. [Google Scholar] [CrossRef]
- Grąt, M.; Wronka, K.M.; Lewandowski, Z.; Grąt, K.; Krasnodębski, M.; Stypułkowski, J.; Hołówko, W.; Masior, Ł.; Kosińska, I.; Wasilewicz, M.; et al. Effects of Continuous Use of Probiotics before Liver Transplantation: A Randomized, Double-Blind, Placebo-Controlled Trial. Clin. Nutr. 2017, 36, 1530–1539. [Google Scholar] [CrossRef]
- Plank, L.D.; Mathur, S.; Gane, E.J.; Peng, S.-L.; Gillanders, L.K.; McIlroy, K.; Chavez, C.P.; Calder, P.C.; McCall, J.L. Perioperative Immunonutrition in Patients Undergoing Liver Transplantation: A Randomized Double-Blind Trial. Hepatology 2015, 61, 639–647. [Google Scholar] [CrossRef]
- Sawas, T.; Al Halabi, S.; Hernaez, R.; Carey, W.D.; Cho, W.K. Patients Receiving Prebiotics and Probiotics before Liver Transplantation Develop Fewer Infections than Controls: A Systematic Review and Meta-Analysis. Clin. Gastroenterol. Hepatol. 2015, 13, 1567–1574.e3; quiz e143–e144. [Google Scholar] [CrossRef]
- Germani, G.; Battistella, S.; Ulinici, D.; Zanetto, A.; Shalaby, S.; Pellone, M.; Gambato, M.; Senzolo, M.; Russo, F.P.; Burra, P. Drug Induced Liver Injury: From Pathogenesis to Liver Transplantation. Minerva Gastroenterol. 2021, 67, 50–64. [Google Scholar] [CrossRef]
- Dąbrowska-Bender, M.; Tatara, T. Ocena stanu odżywienia i sposobu odżywiania się pacjentów po przeszczepieniu wątroby. Probl. Hig. Epidemiol. 2011, 92, 247–253. [Google Scholar]
| Common Name (Latin Name) | Family | Used Part | Materials | References |
|---|---|---|---|---|
| Garlic (Allium sativum) | Alliaceae | bulb | Rats | [56,57,58] |
| Onion (Allium cepa) | Alliaceae | bulb | Rats | [57,59,60,61,62] |
| Artichoke (Cynara scolymus) | Apiaceae | leaves, flowers | Rats, clinical trials (100 patients, 17 patients) | [56,57,59,63,64] |
| Dandelion (Taraxacum officinale) | Asteraceae | root, leaves | Mice | [56,57,64,65,66,67] |
| Chicory (Cichorium intybus) | Asteraceae | root, herb | Rats, mice, rabbits, clinical trials (92 patients, 36 patients) | [59,63,64,68] |
| Reishi or Lingzhi (Ganoderma lucidum) | Polyporaceae | fruiting bodies | Mice, rats, | [69,70,71,72,73] |
| Antrodia cinnamomea (syn. A. camphorata) | Fomitopsidaceae | fruiting bodies | Rats, mice, female chicks, human HCC cell lines (i.e., HepG2 and PLC/PRF/5), clinical trials (44 patients, 28 patiensts) | [69,74,75,76,77,78,79,80] |
| Mushroom (Agaricus bisporus) | Agaricaceae | fruiting bodies | Mice | [81,82,83,84,85] |
| Golden oyster mushroom (Pleurotus citrinopileatus) | Pleurotaceae | fruiting bodies | Mice, Human cell line (HepG2 cells) | [86,87,88] |
| Shiitake (Lentinus edodoes) | Marasmiaceae | fruiting bodies | Mice | [87,89,90,91] |
| Symptoms/Patient’s Condition | Recommendation | |
|---|---|---|
| PREOPERATIVE PERIOD | ||
| Current condition of the patient | Diet adapted to the patient’s condition [145]; | |
| optimal supply of energy: 30–35 kcal/kg BW/day [151]; obese patient: EN or PN 25 kcal/kg IBW/day [151] | ||
| optimal supply of protein:1.2–1.5 g/kg) [151]; obese patients: 2.0–2.5 g/kg IBW/day [151]; | ||
| optimal supply of fat-soluble vitamins, B vitamins, zinc, magnesium, antioxidants (selenium, vit E, and vit C) [150]; | ||
| Hepatic encephalopathy | Modified protein intake [145]; | |
| Malnourished patients (BMI < 18.5 kg/m2; weight loss > 10–15% within 6 months; serum albumin < 30 g/L) | Oral nutritional supplements (ONS) for 7 days before surgery [152]; | |
| Severely malnourished patients (more than 10% WL) | Surgery postponed by 2 weeks [152]; | |
| Improving nutritional status [152]; | ||
| Infectious complications risk | Formulas enriched with arginine, omega-3 fatty acids, nucleotides [153]; | |
| Preoperative fasting | Shortening fasting period [151]; | |
| 6 h for solid food and 2 h for fluid before surgery [151]; | ||
| Postoperative insulin resistance | Clear carbohydrate fluid 2 h before surgery [151,154]; | |
| POSTOPERATIVE PERIOD | ||
| Early period 12–24 h | Oral food intake and/or enteral nutrition within 12–24 h after surgery (30–35 kcal/kg/day; protein: 1.2–1.5 g/kg/day) [147,155]; | |
| Malnourished patients OR prolonged postoperative fasting (>5 days) | Additional nutritional support [147,155]; | |
| Oral route impossible | Parenteral nutrition [147]; | |
| Later period | DM risk (immunosuppressive drugs) | Reduce consumption of simple sugars, |
| reduce Tacrolimus dose [150]; | ||
| Increased potassium level (immunosuppressive drugs) | Limit the food products rich in potassium [156]; | |
| Decreased magnesium level | Balanced diet (whole grains, legumes, nuts) [150]; | |
| Excessive body weight (overweight, obesity) | Balanced diet, proper protein intake [150]; | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jamioł-Milc, D.; Gudan, A.; Kaźmierczak-Siedlecka, K.; Hołowko-Ziółek, J.; Maciejewska-Markiewicz, D.; Janda-Milczarek, K.; Stachowska, E. Nutritional Support for Liver Diseases. Nutrients 2023, 15, 3640. https://doi.org/10.3390/nu15163640
Jamioł-Milc D, Gudan A, Kaźmierczak-Siedlecka K, Hołowko-Ziółek J, Maciejewska-Markiewicz D, Janda-Milczarek K, Stachowska E. Nutritional Support for Liver Diseases. Nutrients. 2023; 15(16):3640. https://doi.org/10.3390/nu15163640
Chicago/Turabian StyleJamioł-Milc, Dominika, Anna Gudan, Karolina Kaźmierczak-Siedlecka, Joanna Hołowko-Ziółek, Dominika Maciejewska-Markiewicz, Katarzyna Janda-Milczarek, and Ewa Stachowska. 2023. "Nutritional Support for Liver Diseases" Nutrients 15, no. 16: 3640. https://doi.org/10.3390/nu15163640
APA StyleJamioł-Milc, D., Gudan, A., Kaźmierczak-Siedlecka, K., Hołowko-Ziółek, J., Maciejewska-Markiewicz, D., Janda-Milczarek, K., & Stachowska, E. (2023). Nutritional Support for Liver Diseases. Nutrients, 15(16), 3640. https://doi.org/10.3390/nu15163640







