Exposure to Different Amounts of Dietary Gluten in Patients with Non-Celiac Gluten Sensitivity (NCGS): An Exploratory Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Intervention
2.2. Diets
2.3. Gastrointestinal Symptoms and Quality of Life
2.4. Statistical Analysis
3. Results
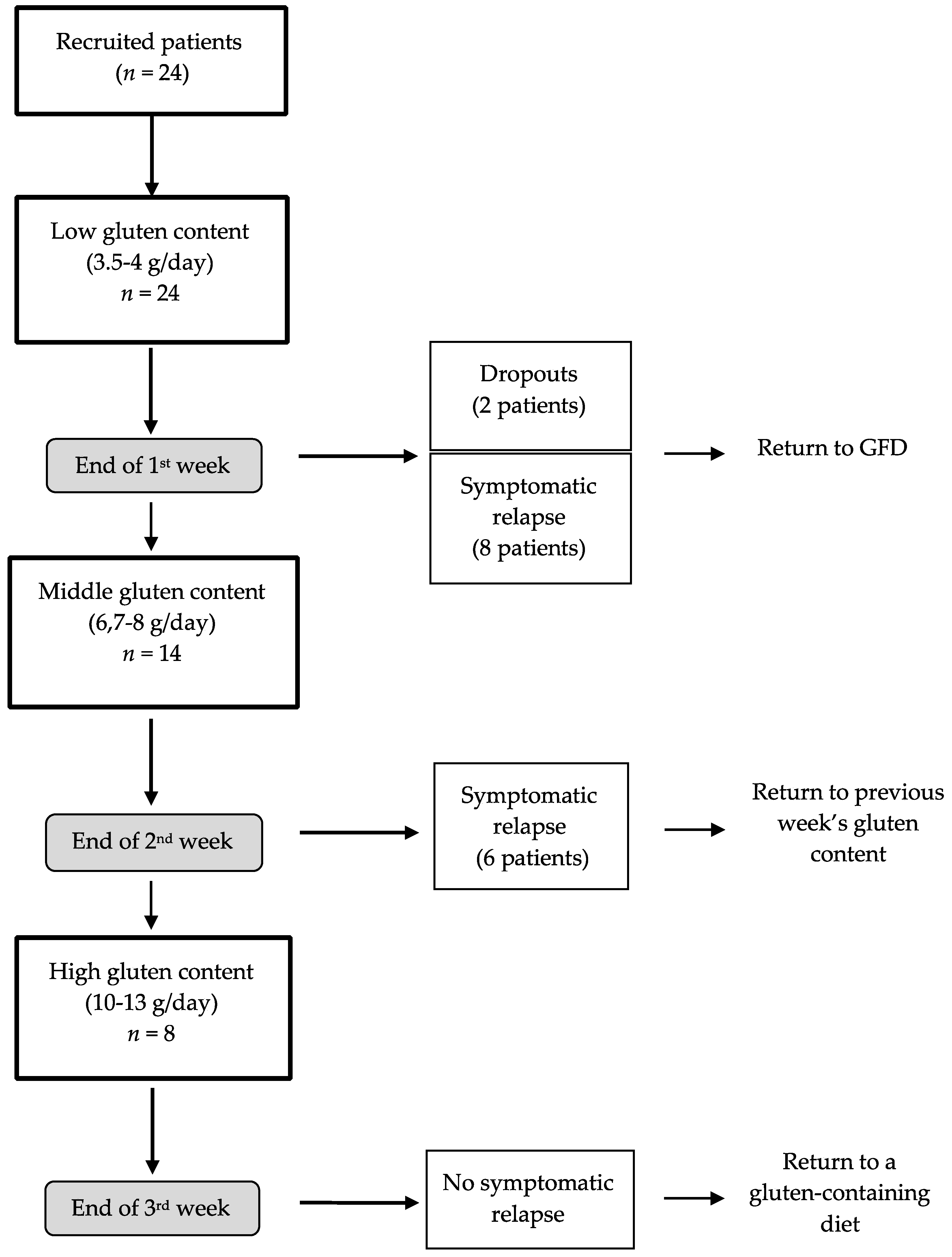
3.1. Patients
3.2. Quality of Life
3.3. Gastrointestinal Symptoms
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Sapone, A.; Bai, J.C.; Ciacci, C.; Dolinsek, J.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Rostami, K.; Sanders, D.S.; Schumann, M.; et al. Spectrum of gluten-related disorders: Consensus on new nomenclature and classification. BMC Med. 2012, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Bascuñán, K.A.; Roncoroni, L.; Branchi, F.; Doneda, L.; Scricciolo, A.; Ferretti, F.; Aray, M.; Elli, L. The 5 Ws of a gluten challenge for gluten-related disorders. Nutr. Rev. 2018, 76, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Bai, J.C.; Bonaz, B.; Bouma, G.; Calabrò, A.; Carroccio, A.; Castillejo, G.; Ciacci, C.; Cristofori, F.; Dolinsek, J.; et al. Non-celiac gluten sensitivity: The new frontier of gluten related disorders. Nutrients 2013, 5, 3839–3853. [Google Scholar] [CrossRef]
- Catassi, C.; Elli, L.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; Cellier, C.; Cristofori, F.; de Magistris, L.; Dolinsek, J.; et al. Diagnosis of non-celiac gluten sensitivity (NCGS): The salerno experts’ criteria. Nutrients 2015, 7, 4966–4977. [Google Scholar] [CrossRef]
- Biesiekierski, J.R.; Newnham, E.D.; Irving, P.M.; Barrett, J.S.; Haines, M.; Doecke, J.D.; Shepherd, S.J.; Muir, J.G.; Gibson, P.R. Gluten Causes Gastrointestinal Symptoms in Subjects Without Celiac Disease: A Double-Blind Randomized Placebo-Controlled Trial. Am. J. Gastroenterol. 2011, 106, 508. [Google Scholar] [CrossRef] [PubMed]
- Volta, U.; Bardella, M.T.; Calabrò, A.; Troncone, R.; Corazza, G.R.; Bagnato, C.; Belcari, C.; Bellantoni, A.; Caio, G.; Calella, F.; et al. An Italian prospective multicenter survey on patients suspected of having non-celiac gluten sensitivity. BMC Med. 2014, 12, 85. [Google Scholar] [CrossRef] [PubMed]
- Parrish, C.R. Non-Celiac Gluten Sensitivity; Where are we now in 2015? Pract. Gastroenterol. 2015, 142, 40–48. [Google Scholar]
- Fasano, A.; Sapone, A.; Zevallos, V.; Schuppan, D. Nonceliac gluten sensitivity. Gastroenterology 2015, 148, 1195–1204. [Google Scholar] [CrossRef]
- Lionetti, E.; Pulvirenti, A.; Vallorani, M.; Catassi, G.; Verma, A.K.; Gatti, S.; Catassi, C. Re-challenge Studies in Non-celiac Gluten Sensitivity: A Systematic Review and Meta-Analysis. Front. Physiol. 2017, 8, 621. [Google Scholar] [CrossRef]
- Molina-Infante, J.; Carroccio, A. Suspected Nonceliac Gluten Sensitivity Confirmed in Few Patients After Gluten Challenge in Double-Blind, Placebo-Controlled Trials. Clin. Gastroenterol. Hepatol. 2017, 15, 339–348. [Google Scholar] [CrossRef]
- Rubio-Tapia, A.; Hill, I.D.; Kelly, C.P.; Calderwood, A.H.; Murray, J.A. ACG clinical guidelines: Diagnosis and management of celiac disease. Am. J. Gastroenterol. 2013, 108, 656–676, quiz 677. [Google Scholar] [CrossRef] [PubMed]
- Sverker, A.; Hensing, G.; Hallert, C. “Controlled by food”—Lived experiences of coeliac disease. J. Hum. Nutr. Diet. 2005, 18, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Bascuñán, K.A.; Vespa, M.C.; Araya, M. Celiac disease: Understanding the gluten-free diet. Eur. J. Nutr. 2017, 56, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Hill, I.D.; Fasano, A.; Guandalini, S.; Hoffenberg, E.; Levy, J.; Reilly, N.; Verma, R. NASPGHAN clinical report on the diagnosis and treatment of gluten-related disorders. J. Pediatr. Gastroenterol. Nutr. 2016, 63, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Alaedini, A.; Bojarski, C.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; De Magistris, L.; Dieterich, W.; Di Liberto, D.; et al. The overlapping area of non-celiac gluten sensitivity (NCGS) and wheat-sensitive irritable bowel syndrome (IBS): An update. Nutrients 2017, 9, 1268. [Google Scholar] [CrossRef] [PubMed]
- Uhde, M.; Ajamian, M.; Caio, G.; De Giorgio, R.; Indart, A.; Green, P.H.; Verna, E.C.; Volta, U.; Alaedini, A. Intestinal cell damage and systemic immune activation in individuals reporting sensitivity to wheat in the absence of coeliac disease. Gut 2016, 65, 1930–1937. [Google Scholar] [CrossRef] [PubMed]
- Pinto-Sánchez, M.I.; Verdú, E.F. Non-coeliac gluten sensitivity: Are we closer to separating the wheat from the chaff? Gut 2016. [Google Scholar] [CrossRef]
- Junker, Y.; Zeissig, S.; Kim, S.-J.; Barisani, D.; Wieser, H.; Leffler, D.A.; Zevallos, V.; Libermann, T.A.; Dillon, S.; Freitag, T.L.; et al. Wheat amylase trypsin inhibitors drive intestinal inflammation via activation of toll-like receptor 4. J. Exp. Med. 2012, 209, 2395–2408. [Google Scholar] [CrossRef]
- de Punder, K.; Pruimboom, L. The dietary intake of wheat and other cereal grains and their role in inflammation. Nutrients 2013, 5, 771–787. [Google Scholar] [CrossRef]
- Tatham, A.S.; Shewry, P.R. Allergens to wheat and related cereals. Clin. Exp. Allergy 2008, 38, 1712–1726. [Google Scholar]
- Pellegrina, C.D.; Perbellini, O.; Scupoli, M.T.; Tomelleri, C.; Zanetti, C.; Zoccatelli, G.; Fusi, M.; Peruffo, A.; Rizzi, C.; Chignola, R. Effects of wheat germ agglutinin on human gastrointestinal epithelium: Insights from an experimental model of immune/epithelial cell interaction. Toxicol. Appl. Pharmacol. 2009, 237, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Manning, L.P.; Biesiekierski, J.R. Use of dietary interventions for functional gastrointestinal disorders. Curr. Opin. Pharmacol. 2018, 43, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Roncoroni, L.; Bascuñán, K.A.; Doneda, L.; Scricciolo, A.; Lombardo, V.; Branchi, F.; Ferretti, F.; Dell’osso, B.; Montanari, V.; Bardella, M.T.; et al. A low FODMAP gluten-free diet improves functional gastrointestinal disorders and overall mental health of celiac disease patients: A randomized controlled trial. Nutrients 2018, 10, 8. [Google Scholar] [CrossRef]
- Carroccio, A.; Mansueto, P.; Iacono, G.; Soresi, M.; D’Alcamo, A.; Cavataio, F.; Brusca, I.; Florena, A.M.; Ambrosiano, G.; Seidita, A.; et al. Non-celiac wheat sensitivity diagnosed by double-blind placebo-controlled challenge: Exploring a new clinical entity. Am. J. Gastroenterol. 2012, 107, 1898–1906, quiz 1907. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.L.; Biesiekierski, J.R.; Yelland, G.W.; Muir, J.G.; Gibson, P.R. Randomised clinical trial: Gluten may cause depression in subjects with non-coeliac gluten sensitivity—An exploratory clinical study. Aliment. Pharmacol. Ther. 2014, 39, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Zanini, B.; Baschè, R.; Ferraresi, A.; Ricci, C.; Lanzarotto, F.; Marullo, M.; Villanacci, V.; Hidalgo, A.; Lanzini, A. Randomised clinical study: Gluten challenge induces symptom recurrence in only a minority of patients who meet clinical criteria for non-coeliac gluten sensitivity. Aliment. Pharmacol. Ther. 2015, 42, 968–976. [Google Scholar] [CrossRef]
- Capannolo, A.; Viscido, A.; Barkad, M.A.; Valerii, G.; Ciccone, F.; Melideo, D.; Frieri, G.; Latella, G. Non-Celiac Gluten Sensitivity among Patients Perceiving Gluten-Related Symptoms. Digestion 2015, 92, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Rosinach, M.; Fernández-Bañares, F.; Carrasco, A.; Ibarra, M.; Temiño, R.; Salas, A.; Esteve, M. Double-Blind Randomized Clinical Trial: Gluten versus Placebo Rechallenge in Patients with Lymphocytic Enteritis and Suspected Celiac Disease. PLoS ONE 2016, 11, e0157879. [Google Scholar] [CrossRef] [PubMed]
- Picarelli, A.; Borghini, R.; Di Tola, M.; Marino, M.; Urciuoli, C.; Isonne, C.; Puzzono, M.; Porowska, B.; Rumi, G.; Lonardi, S.; et al. Intestinal, systemic, and oral gluten-related alterations in patients with nonceliac gluten sensitivity. J. Clin. Gastroenterol. 2016, 50, 849–858. [Google Scholar] [CrossRef]
- Elli, L.; Tomba, C.; Branchi, F.; Roncoroni, L.; Lombardo, V.; Bardella, M.T.; Ferretti, F.; Conte, D.; Valiante, F.; Fini, L.; et al. Evidence for the presence of non-celiac gluten sensitivity in patients with functional gastrointestinal symptoms: Results from a multicenter randomized double-blind placebo-controlled gluten challenge. Nutrients 2016, 8, 84. [Google Scholar] [CrossRef] [PubMed]
- Skodje, G.I.; Sarna, V.K.; Minelle, I.H.; Rolfsen, K.L.; Muir, J.G.; Gibson, P.R.; Veierød, M.B.; Henriksen, C.; Lundin, K.E.A. Fructan, Rather Than Gluten, Induces Symptoms in Patients With Self-Reported Non-Celiac Gluten Sensitivity. Gastroenterology 2018, 154, 529–539.e2. [Google Scholar] [CrossRef] [PubMed]
- Dale, H.F.; Hatlebakk, J.G.; Hovdenak, N.; Ystad, S.O.; Lied, G.A. The effect of a controlled gluten challenge in a group of patients with suspected non-coeliac gluten sensitivity: A randomized, double-blind placebo-controlled challenge. Neurogastroenterol. Motil. 2018, e13332. [Google Scholar] [CrossRef] [PubMed]
- Volta, U.; Caio, G.; Tovoli, F.; De Giorgio, R. Non-celiac gluten sensitivity: Questions still to be answered despite increasing awareness. Cell. Mol. Immunol. 2013, 10, 383. [Google Scholar] [CrossRef] [PubMed]
- Drossman, D.A.; Dumitrascu, D.L. Rome III: New standard for functional gastrointestinal disorders. J. Gastrointest. Liver Dis. 2006, 15, 237–241. [Google Scholar]
- Leffler, D.A.; Dennis, M.; Edwards George, J.B.; Jamma, S.; Magge, S.; Cook, E.F.; Schuppan, D.; Kelly, C.P. A Simple Validated Gluten-Free Diet Adherence Survey for Adults With Celiac Disease. Clin. Gastroenterol. Hepatol. 2009, 7, 530–536.e2. [Google Scholar] [CrossRef]
- Schalk, K.; Lexhaller, B.; Koehler, P.; Scherf, K.A. Isolation and characterization of gluten protein types from wheat, rye, barley and oats for use as reference materials. PLoS ONE 2017, 12, e0172819. [Google Scholar] [CrossRef]
- Ware, J.E.; Sherbourne, C.D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 1992, 473–483. [Google Scholar] [CrossRef]
- Di Sabatino, A.; Volta, U.; Salvatore, C.; Biancheri, P.; Caio, G.; De Giorgio, R.; Di Stefano, M.; Corazza, G.R. Small Amounts of Gluten in Subjects With Suspected Nonceliac Gluten Sensitivity: A Randomized, Double-Blind, Placebo-Controlled, Cross-Over Trial. Clin. Gastroenterol. Hepatol. 2015, 13, 1604–1612.e3. [Google Scholar] [CrossRef]
- Shahbazkhani, B.; Sadeghi, A.; Malekzadeh, R.; Khatavi, F.; Etemadi, M.; Kalantri, E.; Rostami-Nejad, M.; Rostami, K. Non-Celiac Gluten Sensitivity Has Narrowed the Spectrum of Irritable Bowel Syndrome: A Double-Blind Randomized Placebo-Controlled Trial. Nutrients 2015, 7, 4542–4554. [Google Scholar] [CrossRef]
- Sanders, D.S.; Aziz, I. Editorial: Non-celiac wheat sensitivity: Separating the wheat from the chat! Am. J. Gastroenterol. 2012, 107, 1908–1912. [Google Scholar] [CrossRef]
- Carroccio, A.; Rini, G.; Mansueto, P. Non-celiac wheat sensitivity is a more appropriate label than non-celiac gluten sensitivity. Gastroenterology 2014, 146, 320–321. [Google Scholar] [CrossRef] [PubMed]
| Study (Reference) | Patients | Study Design | No. of Patients | Challenge Duration | Methods | Main Findings |
|---|---|---|---|---|---|---|
| Biesiekierski et al. [5] | NCGS w/IBS | DBRPCT | 34 patients | up to 6 weeks | Gluten or placebo (two bread slices plus one muffin per day) | 68% (13) of patients in the gluten group reported inadequate symptoms control compared with 40% (6) under placebo. |
| Carroccio et al. [24] | NCGS | DBRPCT | 2 weeks | Capsules with wheat (20 g) vs. placebo | 30% (276) of patients diagnosed as non-celiac wheat sensitivity. Wheat challenge induced >30% increase in symptoms. | |
| Peters et al. [25] | NCGS w/IBS symptoms | DBRPCT | 22 patients | 3 days | Dietary challenge: gluten-free food supplemented with gluten (16 g/day), whey (16 g/day) or not supplemented (placebo) | Higher overall Spielberger State Trait Personality Inventory state depression scores compared to placebo but not whey, after gluten. Similar gastrointestinal symptoms induced after all treatments |
| Zanini et al. [26] | NCGS | DBRPCT | 35 non-CD patients | 10 days | Gluten-containing flour or gluten-free flour for 10 days, followed by a 2-week washout period | 34% of patients diagnosed as having NCGS |
| Capannolo et al. [27] | NCGS | DBRPCT | 364 patients (27 with NCGS and 337 with no specific diagnosis) | 1 month | Challenge with dietary gluten vs. a GFD. | 85.96% did not experience a change in symptomatology after a GFD. Low value of a gluten-containing diet for an increase in symptoms. |
| Rosinach et al. [28] | NCGS | DBRPCT | 18 non-CD patients | 6 months | 11 gluten (20 g/day) and 7 placeboes | 91% of patients with clinical relapse during gluten challenge (vs. 28.5% after placebo) |
| Picarelli et al. [29] | NCGS | DBRPCT | 26 patients | 1 day | Food challenge (oral provocation test) with a gluten-containing croissant (10 g of gluten per croissant) to 13 patients and a gluten-free croissant to the other 13 patients. | No difference was found between the severity of symptoms with gluten-containing croissants compared to a group of patients with gluten-free croissants. |
| Elli et al. [30] | NCGS w/functional gastrointestinal symptoms | DBRPCT | 98 patients | 7 days | Gluten intake (5.6 g/day) or placebo | 28 patients reported symptomatic relapse with deteriorated quality of life |
| Skodje et al. [31] | Subjects with self-reported NCGS | DBRPCT | 59 subjects | 7 days | Diets containing gluten (5.7 g), fructans (2.1 g), or placebo | Overall Gastrointestinal Symptom Rating Scale for Irritable Bowel Syndrome scores increased after fructans rather than gluten. |
| Dale et al. [32] | Patients w/suspected NCGS | DBRPCT | 20 patients | 4 days | Two muffins a day (11/0 g gluten). (4 periods w/double-blinded provocation, 2 w/gluten, 2 w/placebo) | Most severe symptoms found after placebo. Only 4/20 patients correctly identified periods w/gluten. |
| Meal | Low-Gluten (3.5–4 g Gluten) | Mid-Gluten (6.7–8 g Gluten) | High-Gluten (10–13 g Gluten) |
|---|---|---|---|
| Breakfast | 1 small cup of coffee, 300 mL partly skimmed milk, 1 gluten-free croissant | 1 small cup of coffee, 300 mL partly skimmed milk, 4 gluten-free biscuits | 1 small cup of coffee, 300 mL partly skimmed milk, 4 gluten-free rusks |
| Morning snack | 1 kiwifruit | 1 apple | 1 banana |
| Lunch | 100 g gluten-free pasta, 90 g mixed vegetables, 1 portion of chard, 4 mandarins | 100 g gluten-free pasta, 40 g cow ricotta cheese, 100 g potatoes, 2 bananas | 60 g wheat pasta with broccoli, 2 teaspoonfuls grated Parmesan cheese, 1 portion of mixed salad, 2.5 glasses of fruit salad |
| Afternoon snack | 1 bowl of strawberries | 1 bowl of strawberries | 1 pear |
| Dinner | 50 g wheat pasta with zucchini, 100 g turkey thigh 200 g potatoes, 5 slices of pineapple | 50 g wheat pasta with tomato sauce, 1 spoon of fresh peas 120 g pork, 1 portion of chard 5 mandarins | 60 g wheat pasta with tomato sauce, 100 g halibut, 200 g potatoes, 2 glasses of fruit salad |
| During the day | 40 g gluten-free bread, 6.5 teaspoonfuls virgin olive oil | 50 g of white wheat bread, 3.5 teaspoonfuls virgin olive oil | 30 g white wheat bread, 8 teaspoonfuls virgin olive oil |
| Low-Gluten (3.5–4 g/day, n = 8) | Mid-Gluten (6.7–8 g/day, n = 6) | High-Gluten (10–13 g/day, n = 8) | p-Value † | |
|---|---|---|---|---|
| Age, years | 44.6 ± 4.5 | 45.5 ± 3.1 | 44.6 ± 5.2 | 0.98 |
| Gender, F/M | 7/1 | 6/0 | 7/1 | 0.99 |
| BMI, kg/m2 | 23.0 ± 1.7 | 23.8 ± 1.8 | 21.8 ± 0.7 | 0.65 |
| Diarrhea, n (%) | 1 (12.5) | 0 (0) | 1 (12.5) | 0.99 |
| Bloating, n (%) | 0 (0) | 2 (33.3) | 1 (12.5) | 0.24 |
| Constipation, n (%) | 3 (37.5) | 5 (83.3) | 3 (37.5) | 0.21 |
| Abdominal pain, n (%) | 5 (62.5) | 4 (66.6) | 3 (37.5) | 0.64 |
| Dyspepsia, n (%) | 6 (75) | 1 (16.6) | 2 (25) | 0.08 |
| Low-Gluten (3.5–4 g/day, n = 8) | Mid-Gluten (6.7–8 g/day, n = 6) | High-Gluten (10–13 g/day, n = 8) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | 7-Day | p-Value † | Baseline | 14-Day | p-Value † | Baseline | 21-Day | p-Value † | p-Value ‡ | |
| Physical functioning | 100.0 (5.0) | 95.0 (10.0) | 0.10 | 100.0 (20.0) | 82.5 (45.0) | 0.17 | 100.0 (2.5) | 100.0 (2.5) | 0.94 | 0.13 |
| Role physical | 100.0 (25.0) | 50.0 (37.5) | 0.03 | 87.5 (25.0) | 100.0 (50.0) | 0.72 | 87.5 (37.5) | 87.5 (62.5) | 0.65 | 0.19 |
| Role emotional | 79.0 (26.0) | 41.0 (31.0) | 0.004 | 79.0 (26.0) | 62.5 (32.0) | 0.92 | 84.0 (27.5) | 67.5 (40.5) | 0.83 | 0.01 |
| Bodily pain | 76.0 (22.5) | 53.5 (47.5) | 0.008 | 76.0 (22.5) | 64.0 (11.0) | 0.87 | 66.5 (50.0) | 69.5 (44.0) | 0.30 | 0.15 |
| Mental health | 65.0 (20) | 47.5 (17.5) | 0.12 | 65.0 (20.0) | 55.0 (20.0) | 0.74 | 72.5 (15.0) | 57.5 (37.5) | 0.79 | 0.48 |
| Vitality | 87.0 (12.5) | 56.0 (18.5) | 0.02 | 87.0 (12.5) | 62.5 (38.0) | 0.50 | 100.0 (31.5) | 68.5 (44.0) | 0.34 | 0.67 |
| Social interaction | 100.0 (17.0) | 33.0 (33.0) | 0.007 | 100.0 (67.0) | 100.0 (34.0) | 0.10 | 100.0 (33.5) | 100.0 (17.0) | 0.39 | 0.30 |
| General health | 82.0 (18.0) | 62.0 (34.0) | 0.15 | 76.0 (20.0) | 72.0 (16.0) | 0.46 | 78.0 (10.0) | 72.0 (24.0) | 0.99 | 0.49 |
| Low-Gluten (3.5–4 g/day, n = 8) | Mid-Gluten (6.7–8 g/day, n = 6) | High-Gluten (10–13 g/day, n = 8) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | 7-Day | p-Value † | Baseline | 14-Day | p-Value † | Baseline | 21-Day | p-Value † | p-Value ‡ | |
| Abdominal pain | 0 (1.3) | 2.6 (6.1) | 0.16 | 5.3 (4.5) | 4.9 (3.6) | 0.87 | 0 (0.4) | 0.5 (7.8) | 0.22 | 0.66 |
| Stool satisfaction | 3.8 (9.1) | 0 (3.4) | 0.16 | 7.0 (9.8) | 3.2 (5.4) | 0.25 | 7.6 (8.8) | 4.1 (8.8) | 0.99 | 0.21 |
| Abdominal bloating | 0 (3.9) | 3.0 (8.4) | 0.19 | 5.4 (5.5) | 5.2 (6.0) | 0.87 | 0 (2.2) | 3.5 (8.8) | 0.19 | 0.68 |
| Postprandial fullness | 0 (4.4) | 4.3 (7.4) | 0.12 | 0 (6.7) | 0 (2.9) | 0.84 | 0 (0.0) | 2.5 (6.6) | 0.29 | 0.37 |
| Early satiety feeling | 0.2 (1.8) | 3.5 (5.2) | 0.13 | 0 (1.0) | 0 (1.4) | 0.84 | 0 (0.0) | 1.3 (6.4) | 0.09 | 0.36 |
| Epigastric burn | 0 (0.9) | 2.0 (8.2) | 0.07 | 0 (2.0) | 0 (4.4) | 0.85 | 0 (1.9) | 0.7 (5.8) | 0.41 | 0.42 |
| General well-being | 9.1 (2.9) | 2.1 (2.9) | 0.01 | 7.9 (3.1) | 5.9 (4.5) | 0.14 | 8.5 (1.9) | 5.1 (4.3) | 0.21 | 0.05 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roncoroni, L.; Bascuñán, K.A.; Vecchi, M.; Doneda, L.; Bardella, M.T.; Lombardo, V.; Scricciolo, A.; Branchi, F.; Elli, L. Exposure to Different Amounts of Dietary Gluten in Patients with Non-Celiac Gluten Sensitivity (NCGS): An Exploratory Study. Nutrients 2019, 11, 136. https://doi.org/10.3390/nu11010136
Roncoroni L, Bascuñán KA, Vecchi M, Doneda L, Bardella MT, Lombardo V, Scricciolo A, Branchi F, Elli L. Exposure to Different Amounts of Dietary Gluten in Patients with Non-Celiac Gluten Sensitivity (NCGS): An Exploratory Study. Nutrients. 2019; 11(1):136. https://doi.org/10.3390/nu11010136
Chicago/Turabian StyleRoncoroni, Leda, Karla A. Bascuñán, Maurizio Vecchi, Luisa Doneda, Maria T. Bardella, Vincenza Lombardo, Alice Scricciolo, Federica Branchi, and Luca Elli. 2019. "Exposure to Different Amounts of Dietary Gluten in Patients with Non-Celiac Gluten Sensitivity (NCGS): An Exploratory Study" Nutrients 11, no. 1: 136. https://doi.org/10.3390/nu11010136
APA StyleRoncoroni, L., Bascuñán, K. A., Vecchi, M., Doneda, L., Bardella, M. T., Lombardo, V., Scricciolo, A., Branchi, F., & Elli, L. (2019). Exposure to Different Amounts of Dietary Gluten in Patients with Non-Celiac Gluten Sensitivity (NCGS): An Exploratory Study. Nutrients, 11(1), 136. https://doi.org/10.3390/nu11010136





