Abstract
Tourette syndrome (TS), or Tourette’s, is a tic disorder (TD) belonging to a group of neuropsychiatric conditions marked by recurrent motor movements or vocalizations known as tics. TD, including TS, typically begins in childhood between 4 and 18 years of age and affects approximately 3% of children and adolescents. The etiology and pathogenesis of TD are multifactorial, involving genetic, immunologic, psychological, and environmental factors. Evidence suggests that neurotransmitter dysregulation, particularly within the cortical dopaminergic networks of the basal ganglia and limbic system, which support motor control and cognition, may be involved in the development of TD. Nutritional factors may modulate TD through various mechanisms, including effects on neurotransmitter synthesis and metabolism, neurodevelopment, neural architecture, and neuroimmune activity. This review integrates current evidence on the roles of vitamins D, B6, and A, as well as iron, magnesium, zinc, and copper, in TD. For each micronutrient, its physiological and neurobiological functions are discussed, along with possible mechanistic links to TD pathophysiology. Additionally, we summarize the impact of nutrient deficiencies and assess available evidence regarding their potential therapeutic potential role in TD management. Overall, this synthesis highlights how nutritional status may influence TD onset and symptom severity, suggesting that nutrient-based interventions could potentially serve as valuable adjunctive strategies in treatment.
Keywords:
Tourette’s syndrome; tic disorder; micronutrients; vitamin D; vitamin B6; vitamin A; iron; magnesium; zinc; copper 1. Introduction
Tic disorders (TDs) are common neurodevelopmental conditions characterized by the occurrence of tics, which are brief, repetitive motor movements or vocalizations often preceded by a premonitory urge [1,2,3,4,5,6,7]. Tics show considerable variability both between individuals and within the same individual over time, including differences in intensity, complexity, location, and frequency [4]. TDs can be further classified based on clinical course and symptom presentation. The main subtypes include transient tic disorder (TTD), chronic motor or vocal tic disorder (CTD), and Tourette syndrome (TS) [8,9]. The prevalence of TD varies, with TS being less common than other persistent tic disorders. A CDC report estimated that approximately 0.3% of children aged 3 to 17 years in the United States, about 174,000 between 2016 and 2019, were diagnosed with TS [10,11]. TS is also frequently associated with comorbid neuropsychiatric conditions. Over two-thirds of affected individuals experience co-occurring disorders such as obsessive–compulsive disorder (OCD), autism spectrum disorder (ASD), attention-deficit/hyperactivity disorder (ADHD), sleep disturbances, depression, and anxiety [12,13,14,15].
The etiology and pathogenesis of TD remain incompletely understood, but multiple interacting factors are contributing, including genetic, microbiological, immunologic, psychological, and environmental components [16,17,18]. A widely supported hypothesis proposes disinhibition within the cortico-striato-thalamo-cortical (CSTC) circuit, a neural loop central to motor control and cognitive regulation [19]. Such disinhibition may be partly caused by excessive dopaminergic activity in the striatum, arising from increased dopamine (DA) release or enhanced postsynaptic DA receptor sensitivity [19,20]. This concept aligns with extensive evidence linking dopaminergic imbalance in cortico-basal ganglia pathways to the pathophysiology of CTD [21,22]. Neuroimaging studies have shown increased DA transporter binding in individuals with TD, while immunologic investigations have detected anti-dopamine D2 receptor autoantibodies correlated with symptom severity [23]. Some researchers further suggest that spontaneous fluctuations in tic manifestation may reflect transient alterations in dopaminergic signaling [22]. In addition to DA, other neurotransmitter systems have been implicated in TD pathology, including glutamate, γ-aminobutyric acid (GABA), serotonin, norepinephrine, acetylcholine, and histamine [24,25,26,27].
Growing evidence and anecdotal reports suggest that environmental influences, particularly diet and nutrition, may contribute to tic symptomatology [28]. Nutritional factors can influence the dopaminergic system by altering neurotransmitter synthesis and metabolism, modulating gut microbiota composition, and driving neuroinflammatory processes, highlighting a complex interplay relevant to TD [29]. Although the importance of nutrition has been established in various neurological and psychiatric conditions, its specific relationship with TS and other TD remains underexplored. Evidence shows that adjuvant treatment including micronutrients may have therapeutic benefits against neurological disorders [30]. This review summarizes current evidence on the potential involvement of key nutrients in TD, including vitamin D, vitamin B6, vitamin A, iron, magnesium, zinc, and copper. By consolidating findings, it aims to advance understanding of TD pathophysiology and explore the potential of targeted nutritional strategies in its management and prevention.
2. Vitamin D (Vit D)
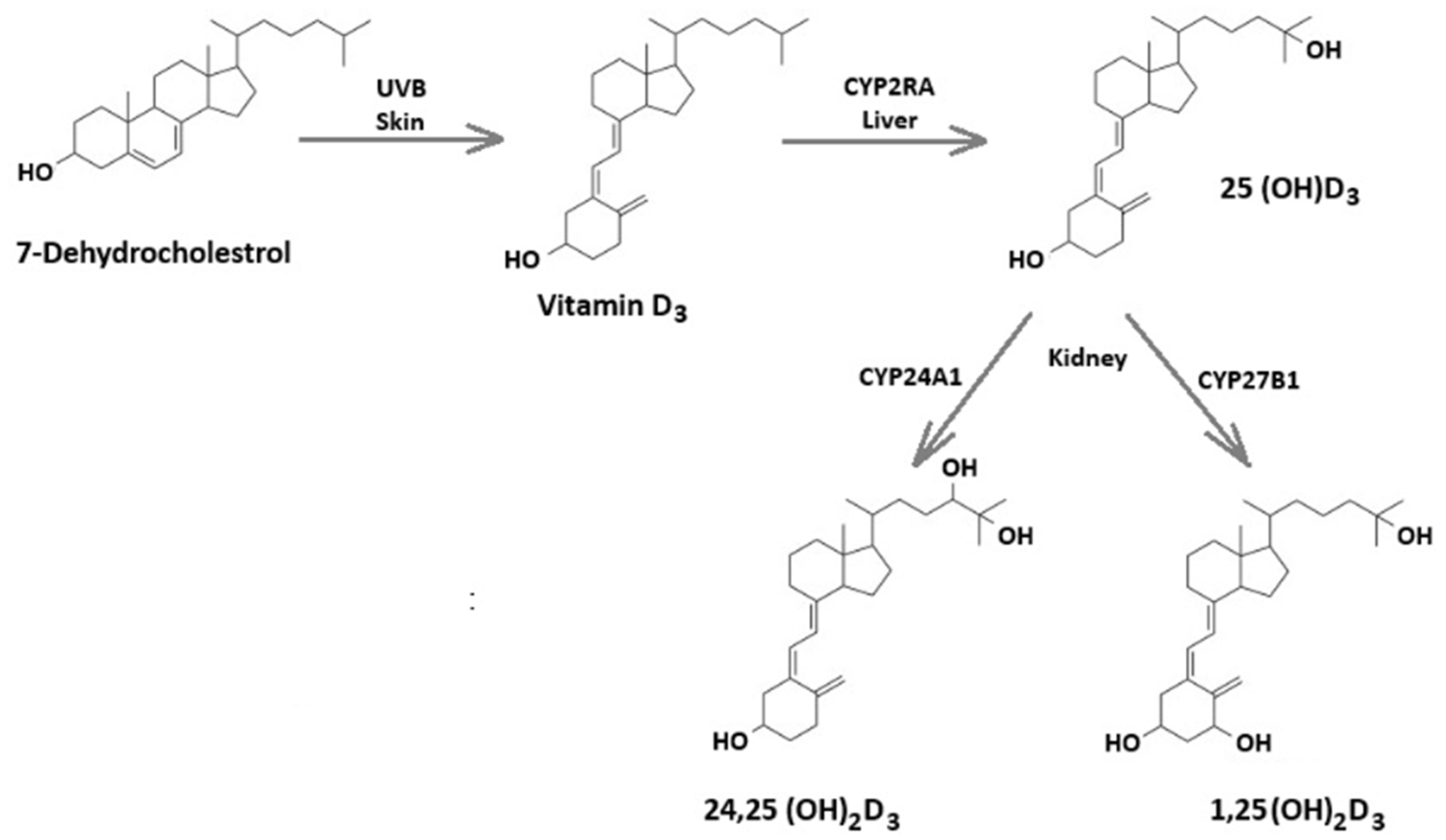
Vitamin D is a neuroactive steroid hormone essential for skeletal health, immune regulation, and neural function [31,32]. Its biologically active precursor, vitamin D3 (cholecalciferol), is synthesized in the skin from 7-dehydrocholesterol upon exposure to ultraviolet B (UVB) radiation from sunlight. It can also be obtained through diet or supplementation [33,34]. Once produced or ingested, vitamin D3 undergoes hydroxylation in the liver, mainly by the cytochrome P450 enzyme CYP2R1, which forms the circulating metabolite 25-hydroxyvitamin D3 (25(OH)D3) [34]. In the kidneys, a second hydroxylation catalyzed by CYP27BI generates 1,25(OH)2D3, the most physiologically active form of vitamin D3 (Figure 1) [34]. Vit D receptors (VDR) and the activating enzyme 1α-hydroxylase are widely distributed throughout the brain, suggesting roles for Vit D in multiple neurological functions [35,36]. High VDR expression in the hippocampus has prompted research into its potential role in cognitive processes, supported by associations between low Vit D levels and cognitive decline [37]. In developing brains, Vit D regulates neuronal proliferation, differentiation, and maturation [38,39,40,41]. More broadly, Vit D exerts neuroprotective effects by modulating neurotransmission, calcium signaling, and synaptic plasticity, as well as antioxidant responses and neuroimmune regulation [37].
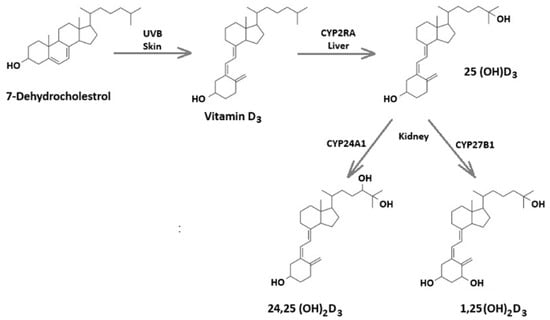
Figure 1.
Overview of vitamin D metabolic enzymes and sites of metabolism.
Dysfunction in cortico-basal ganglia dopaminergic pathways is implicated TD pathophysiology, and evidence suggests that Vit D may help maintain dopaminergic system integrity [4,22]. In experimental models of dopaminergic toxicity, the active form of vitamin D3 (1,25(OH)2D3) enhances dopaminergic neuron survival [31]. Vit D may exert neuroprotective effects in developing dopaminergic neurons [42,43] by modulating the RET receptor. RET is a key component of the glial cell line-derived neurotrophic factor (GDNF) signaling pathway, which governs neuronal differentiation and cell survival [44,45,46,47]. Beyond its influence on DA, Vit D may also interact with other neurotransmitter systems relevant to TD. For example, one proposed mechanism involves modulation of the GABAergic system. In a study by Lerner et al., impaired GABA-A receptor binding was observed across several brain regions in individuals with TS, with particularly marked abnormalities in the insula and cerebellum, regions implicated in tic generation [48]. Vit D also regulates neuroinflammatory processes, exerting anti-inflammatory effects through modulation of both cellular and humoral immune responses [49,50]. Low Vit D levels may therefore promote neuroinflammation, a mechanism potentially contributing to TD neuropathology [51]. This relationship is supported by evidence linking low serum Vit D to elevated inflammatory markers in individuals with TD [52].
A growing body of research suggests that serum Vit D levels are lower in patients with TD when compared with healthy controls (Table 1) and that Vit D deficiency may represent a risk factor for neurological disorders, particularly those associated with dopaminergic dysregulation [31,39,53]. A study of children with CTD (n = 176; median age of 9 years) and healthy children (n = 154) measured 25-hydroxyvitamin D (25[OH]D) levels using high-performance liquid chromatography (HPLC) and tandem mass spectrometry (MS/MS) [52]. Tic status was also measured using the Yale Global Tic Severity Scale (YGTSS). Children with CTD showed significantly lower average 25(OH)D levels (21.7 ng/mL) than healthy children (24.1 ng/mL; p = 0.01). These levels were inversely related to motor tic stores on the YGTSS [52]. Vitamin D deficiency was also found to be significantly more common in CTD children (75 ng/mL) than in healthy controls (44 ng/mL) [52]. This finding was replicated by another study that measured serum 25(OH)D levels in children with TD (n = 132) and healthy peers (n = 144) using HLPC-MS/MS [54]. The researchers then used 25(OH)D levels to categorize each participant’s vitamin D status, which they defined as normal (>30 ng/mL), insufficient (10–30 ng/mL), or deficient (<10 ng/mL) [54]. They found that mean serum 25(OH)D levels were significantly lower (p < 0.01) and that vitamin D deficiency or insufficiency rates were significantly higher (p < 0.01) in the TD group than in the control group [54]. Another study included children with TD (n = 179, 31 females, 148 males, mean age at diagnosis: 8.0 ± 2.7 years old) and without TD (n = 189, 35 females, 154 males, mean age at diagnosis: 8.1 ± 2.6 years old). Those with TD had lower mean serum 25(OH)D levels (33 nmol/L) than their healthy peers (84 nmol/L; p < 0.001) [55]. The authors also found that 25(OH)D status was significantly associated with the presence of TD, and that among children with TD, 25(OH)D levels were negatively correlated with tic severity scores on the YGTSS [55]. Finally, the rate of 25(OH)D deficiency was significantly higher in the TD group than in the healthy control group [55]. Similar associations have been documented in several independent pediatric cohorts [19,56,57,58,59]. In a larger study, children aged 5–14 years with TD (n = 2960, 2355 boys, 560 girls) were compared with age-matched healthy controls (n = 2665, 1912 boys, 753 girls), and 25(OH)D levels were measured in both groups [19]. Average levels of 25(OH)D were lower in the TD group (38.47 nmol/L) than in the control group (50.05 nmol/L; p < 0.001) [19]. Additionally, 25(OH)D deficiency was significantly more common in those with TD than in those without TD (p < 0.001) [19].

Table 1.
Comparison of serum 25-hydroxyvitamin D levels between TD group and healthy controls.
Vit D supplementation is increasingly being investigated as a potential adjunctive therapy for TD [60,61]. For example, a rat study examined effects of vitamin D supplementation in animals (n = 10) with TS-like symptoms induced by 3,3-Iminodipropionitrile (IDPN) [60]. Administering vitamin D continuously by gavage for 8 weeks at 10 μg/kg/day led to a significant reduction in the tic phenotype over the course of treatment (p < 0.001) [60]. Another randomized controlled trial involving 83 children with TDs aged 4–15 years evaluated the comparative efficacy of high-dose (5000 IU/day) and low-dose (1000 IU/day) Vit D supplementation over 3 months [61]. At both dosages, participants showed increased 25(OH)D concentration (p < 0.05) and significantly reduced tic severity scores on the YGTSS (p < 0.05) [61]. These effects were particularly pronounced in the high-dose group, whose YGTSS scores decreased from 28.85 ± 7.19 at baseline to 18.13 ± 4.30 following treatment (p < 0.05). Symptom improvements were accompanied by an increase in 25(OH)D levels from 20.25 ± 7.02 ng/mL to 48.65 ± 10.07 ng/mL (p < 0.05) [61]. Moreover, multivariate linear regression analysis revealed a significant negative association between 25(OH) levels and tic severity scores (p < 0.05), supporting a dose-dependent relationship between vitamin D status and symptom improvement [61]. In another study comparing 140 controls aged 5 to 14 years with 120 children of the same age with CTD, 36 of whom received Vit D treatment for 3 months, baseline serum 25(OH)D concentrations were significantly lower in CTD participants and negatively correlated with symptom severity [62]. Children with CTD who received vitamin D3 supplementation (300 IU/kg/day; maximum 5000 IU/day) showed symptom improvement as their serum 25(OH)D levels approached the target range of 55–70 ng/mL [62]. Following supplementation, children with CTD exhibited normalized serum 25(OH)D levels, which increased from 21 mg/mL at baseline, consistent with vitamin D insufficiency, to 51 ng/mL, falling within the optimal range of 30–90 ng/mL. These changes were accompanied by marked improvements in motor and phonic tics and overall YGTSS scores, without any reported adverse effects [62]. Overall, these findings suggest that Vit D supplementation may constitute a safe and effective adjunctive strategy for managing TD symptoms, particularly in pediatric populations (Table 2).

Table 2.
Summary of studies that support the role of micronutrients in tic alleviation.
Caution is warranted when considering Vit D supplementation, as excessive intake may result in toxicity. Because Vit D enhances calcium absorption in the gastrointestinal tract, toxicity is typically characterized by pronounced hypercalcemia, defined as total serum calcium concentrations exceeding 11.1 mg/dL (normal range: 8.4–10.3 mg/dL) [72,73]. Vit D toxicity can also lead to hypercalciuria and markedly elevated serum 25(OH)D concentrations, often exceeding 375 nmol/L (150 ng/mL) [72,73]. Symptoms of hypercalcemia may present as gastrointestinal complaints such as nausea and vomiting. More systemic effects can include dehydration, loss of appetite, increased thirst and urination, kidney stones, neuropsychiatric disturbances, and musculoskeletal pain and weakness [74]. Current guidelines indicate that the tolerable upper intake level for Vit D varies by age, ranging from 25–100 mcg (1000–4000 IU/day) (Table 3) [74].

Table 3.
Tolerable upper intake levels (ULs) for vitamins D, A, B6, magnesium, iron, zinc, and copper.
3. Vitamin B6 (Vit B6)
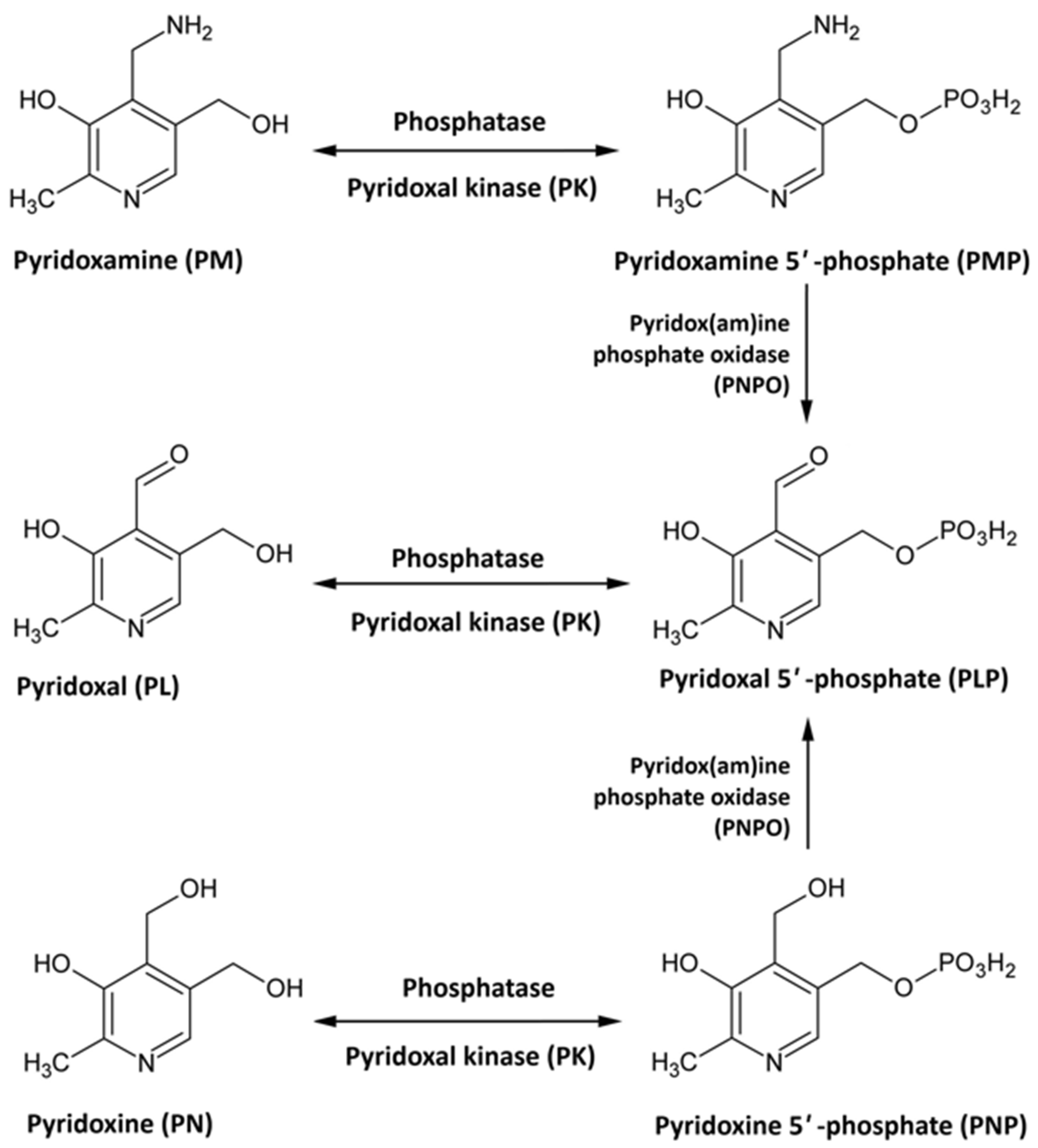
Vit B6 is a water-soluble vitamin that functions as a coenzyme in more than 150 enzymatic reactions, primarily involved in the metabolism of amino acids, carbohydrates, and lipids [76,77,78,79]. Beyond its metabolic roles, Vit B6 supports the synthesis of neurotransmitters, heme, and several bioactive metabolites [76,77,78,79]. It also contributes to DNA and RNA synthesis and modulates gene expression [80]. Additionally, Vit B6 maintains chromosomal stability and regulates inflammation, oxidative stress, angiogenesis, and cell cycle progression [81,82,83]. Vit B6 exists in several interconvertible forms, including pyridoxine (PN), pyridoxamine (PM), and pyridoxal (PL) [84,85]. Each vitamer can be phosphorylated by pyridoxal kinase to produce its respective 5′-phosphate derivative. Pyridoxine 5′-phosphate (PNP) and pyridoxamine 5′-phosphate (PMP) are then oxidized by pyridoxine 5′-phosphate oxidase to generate pyridoxal-5′-phosphate (PLP), the biologically active coenzyme form of Vit B6 (Figure 2) [84,85]. Owing to its broad biological functions, particularly in neurotransmitter synthesis, Vit B6 is essential for normal brain function [86,87,88]. PLP acts as a cofactor for glutamate decarboxylase, the enzyme responsible for converting glutamate, the principal excitatory neurotransmitter, into GABA, the main inhibitory neurotransmitter [84,85]. Other neurotransmitter systems also depend on Vit B6, as it is required for the synthesis of serotonin, dopamine, noradrenaline, histamine, glycine, and D-serine [63,79,89,90,91,92,93].
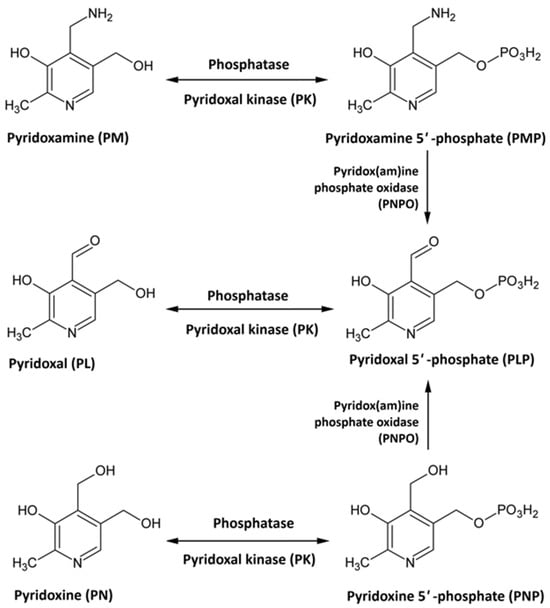
Figure 2.
Vitamin B6 metabolism.
Consequently, Vit B6 deficiency can reduce neurotransmitter levels, potentially contributing to neurological and psychiatric disorders [94]. Multiple lines of evidence link diminished Vit B6 activity to biological abnormalities, including increased sympathetic activation, heightened glucocorticoid sensitivity, and elevated kynurenine levels [63]. Each of these changes can induce neurotoxicity and immune dysregulation [63]. Regarding immune function, Vit B6 deficiency impairs cell-mediated immunity and decreases antibody activity [94]. Deficits in Vit B6 have also been associated with behavioral and motor abnormalities such as hyperirritability, abnormal head movements, and spasmodic motor activity [63]. With respect to its effect on neurotransmission, increasing Vit B6 levels may enhance DA synthesis and restore dopaminergic balance [79,95].
Accordingly, several studies suggest that Vit B6 supplementation may support the treatment of neurological disorders including TS and other TD. A double-blind randomized controlled trial reported that daily Vit B6 at 2 mg/kg reduced TS symptoms in children aged 7–14 years, reflected in lower YGTSS scores and marked reductions in both motor and phonic tics [63]. Another open-label randomized trial of children aged 4–17 years with TS or CTD (n = 34, 30 boys and 4 girls) found decreased tic and anxiety symptoms following Vit B6-based nutritional supplementation [64]. One group of participants (n = 17) was randomly assigned to receive Vit B6 at 2.8 mg/day for two months, while the other group (n = 17) did not receive supplementation. In the Vit B6 group, mean YGTSS scores decreased from 20.35 (±5.8) to 11.5 (±6.1) after treatment, with an average score reduction of 8.85 (43.5%) [64]. This represented a significantly greater improvement in tic severity over the course of the study compared to the control group [64]. Similarly, in a study by Garcia-López et al., supplementation of Vit B6 along with Mg correlated with lower YGTSS scores during symptomatic exacerbations in children aged 7 to 14 years over 90 days of treatment [65]. The total effect on the YGTSS was a decrease from 58.1 at baseline to 12.9 post-treatment, and total tic scores lowered from 26.7 to 12.9, both changes being statistically significant [65]. Collectively, these findings indicate that Vit B6 supplementation may represent a safe, viable adjunctive approach for managing TD symptoms (Table 2).
Safety is an important consideration for Vit B6 supplementation. Overconsumption of Vit B6 can cause neuropathy, which may lead to impaired motor control [96,97]. To minimize the risk of toxicity, the tolerable upper intake level of Vit B6 for adults is set to 100 mg per day (Table 3) [97].
4. Vitamin A (Vit A)
Vit A is a vital micronutrient required for the normal functioning of several body systems, including the central nervous system [60,95]. Its active metabolite, retinoic acid (RA), is essential for neuronal developmental processes such as differentiation and maturation [52,60,98]. RA also exhibits antioxidant properties and regulates inflammatory signaling [99,100,101]. In addition, Vit A influences gene expression through nuclear retinoic acid receptor (RAR) complexes [102,103]. In the brain, Vit A supports neuroplasticity, with animal studies showing that Vit A deficiency impairs hippocampal plasticity through mechanisms involving RA nuclear receptor-α (RARα) [104,105,106]. RARs are abundantly expressed in striatal neurons, where RA signaling promotes neurogenesis and striatal differentiation [107,108]. By modulating gene transcription, RA can affect DA pathways, suggesting a potential role for Vit A in TD pathogenesis, which is consistently linked to dopaminergic dysfunction [105,109,110].
Several clinical studies corroborate this association. In one study comparing children with CTD (n = 176, median age of 9 years) and healthy controls (n = 154, median age of 9 years), Vit A deficiency and combined micronutrient insufficiency were significantly more prevalent in the CTD group [52]. Vit A status was determined by circulating retinol levels, which were measured by HPLC and MS/MS [52]. The CTD group, whose tic status was confirmed with the YGTSS, had a mean serum retinol level of 1.09 μmol/L, while the control group had a significantly higher mean of 1.23 μmol/L (p = 0.001) [52]. Another study involving 198 children with TD and 50 controls found markedly lower Vit A levels and higher deficiency rates among affected participants, with the lowest concentrations observed in those with TS compared to CTD or TTD (p < 0.05) [56]. A larger study of 245 children with TD and 63 controls further revealed an inverse correlation between serum Vit A concentrations and tic severity (p < 0.01) [66]. Collectively, these findings suggest that insufficient Vit A status may contribute to TD pathogenesis and suggest possible benefits from Vit A supplementation. In one intervention study, 50 children with TD and 50 neurotypical peers received Vit A drops combined with five-dimensional lysine granules, which resulted in short-term tic reduction [111]. This outcome highlights the possibility of a potential therapeutic role for Vit A supplementation in managing TD symptoms (Table 2).
Excessive Vit A intake, referred to as hypervitaminosis A, can result in significant hepatotoxicity [112]. Toxicity may occur acutely, following a very high single dose, or chronically, arising from prolonged overconsumption. Clinical manifestations include liver damage, as well as nausea and bone pain [113]. Mechanistically, Vit A toxicity contributes to oxidative stress, inflammation, and progressive liver damage which may advance from steatosis to fibrosis and ultimately cirrhosis [113,114]. The tolerable upper intake level for adults has been established as 3000 retinol activity equivalents (RAE) per day, corresponding to 3000 mcg or 9000 IU (Table 3) [115].
5. Iron
Iron is a trace element that serves as a cofactor for numerous enzymes involved in hydroxylation, oxidation, and peroxidation reactions [70,116]. Consequently, it is indispensable for several biological processes, including oxygen transport and mitochondrial energy production [117,118]. Iron also plays a central role in neurotransmitter systems, particularly for monoamines such as dopamine, norepinephrine, epinephrine, and serotonin. These systems regulate emotion, attention, reward processing, and motor control [119]. Iron is essential for neurotransmitter synthesis, transport, and metabolism [120], particularly within dopaminergic circuits, where it facilitates DA synthesis, receptor development, and transporter function [121,122]. Additionally, iron helps maintain glutamate and γ-aminobutyric acid (GABA) equilibrium, thereby sustaining the excitatory–inhibitory balance critical for normal neural communication [123,124,125,126]. Iron deficiency has been associated with alterations in GABA metabolism, including elevated GABA concentrations in the hippocampus, striatum, and globus pallidus, as well as changes in GABA receptor binding at synaptic membranes [127,128,129,130]. In addition to neurotransmitter metabolism, iron supports neuronal development by promoting dendritic and synaptic maturation, as well as white matter myelination [131,132,133].
Low iron levels have been associated with central nervous system dysfunction across several neuropsychiatric disorders, including TS [134,135,136,137]. A neuroimaging study reported that, relative to matched controls (n = 40), individuals with TS (n = 25) exhibited significantly reduced subcortical magnetic susceptibility, indicative of lower iron content, in regions such as the caudate, pallidum, subthalamic nucleus, thalamus, red nucleus, and substantia nigra [116]. These alterations were accompanied by dopaminergic dysregulation, including decreased availability of dopamine D1 receptors in the dorsal striatum, which positively correlated with tic severity [116]. Together, these findings suggest that disturbances in multiple dopaminergic components, particularly including synaptic dopamine release and D1 receptor binding, may contribute to TS neuropathology [116]. The co-occurrence of these abnormalities with disrupted iron homeostasis further implicates iron dysregulation as one factor in TS pathogenesis.
Other studies indicate a possible association between reduced iron levels and structural brain alterations that may contribute to TD development [28,67,68]. Evidence suggests that inadequate iron availability could lead to hypoplasia, or incomplete maturation, of the caudate putamen, potentially increasing susceptibility to tic formation [28]. Lower iron concentrations may also be linked to decreased cortical volumes, resulting in diminished inhibitory control of motor output [67]. One study measured serum iron in 41 children and adults with TS and 32 controls. Ferritin, a key iron-storage protein, was measured in 63 individuals with TS and 44 controls [67]. Consistent with prior research, the investigators observed lower serum iron and ferritin levels in participants with TS, though values remained within physiological norms and were not significantly associated with symptom severity [67]. Mean serum ferritin in the TS group was 55.0 ng/mL (SD = 54.3), significantly lower than 72.2 ng/mL (SD = 72.8) in the control group (t = 2.28, df = 106, p = 0.03) [67]. As with ferritin, serum iron levels were within the normal range in both groups (60–180 μg/dL in males or 50–170 μg/dL in females). Still, mean serum iron was lower in the TS group at 81.6 μg/dL (SD = 24.8) than in the control group at 90.6 μg/dL (SD = 24.8), and this difference was statistically significant (t = 2.38, df = 72, p = 0.02) [67]. Neuroimaging analyses further explored mechanisms linking iron availability to TS pathology. Magnetic resonance imaging (MRI) revealed contrasting correlations between ferritin levels and brain morphology in the TS and control groups: in the TS group, ferritin levels positively correlated with caudate volume, whereas in the controls, the relationship was inverse [67]. Across both cohorts, ferritin levels positively correlated with cortical volumes in the sensorimotor, midtemporal, and subgenual cortices [67], further supporting the idea that iron availability can influence brain structure. A large retrospective study comparing medical records from children with TD (n = 1204) and controls (n = 1220) found that those with TD had lower whole blood iron levels (p < 0.01) [68]. The average Fe level in the TD group was 8.47 ± 0.86 mmol/L, compared to 8.80 ± 0.94 mmol/L in the control group [68]. Another study conducted in a pediatric neurology clinic assessed iron store balance by measuring serum ferritin in drug-naïve children receiving a first-time diagnosis of a TD (n = 47, 32 boys and 15 girls, aged 3.17–8.66 years), compared to age- and sex- matched children seen in the same clinic for headaches (n = 100, 62 boys and 38 girls, aged 3.17–8.66 years) [69]. Mean serum ferritin levels were 32% lower in children with TD than in their peers with headaches (p = 0.01) [69].
Building on evidence linking iron balance to TD, multiple studies have demonstrated significant correlations between reduced iron availability and increased tic severity. A case–control study of 54 children with TD and 54 matched healthy controls reported significantly lower serum iron and ferritin levels in the TD group, alongside strong negative correlations between these measures and YGTSS scores [59]. This trend extended to clinical subtypes of CTD and TS, where lower iron and ferritin concentrations corresponded with more severe tic manifestations [59]. In one study, children with TD (n = 57) who exhibited serum ferritin levels below 50 ng/mL (n = 37) displayed higher tic severity and greater life impact scores than those maintaining normal ferritin levels (n = 20) [70]. Within 12 months of the initial measurement, 26 patients were seen for follow-up, including 12 who were iron-deficient and 14 who were iron-sufficient at baseline [70]. Iron-deficient children who received iron supplementation (n = 5) showed an improvement in tic severity scores from 2.70 to 1.90 after 12 months, whereas those who did not receive supplementation (n = 7) saw an increase from 2.36 to 2.70 [70]. Similar effects were seen in the iron-sufficient group, though less pronounced. Iron-sufficient children supplemented with iron (n = 10) showed a reduction in tic severity scores from 2.40 to 1.95, while children who were not supplemented (n = 4) showed no change, remaining at an average score of 2.88 upon follow-up [70]. This emerging evidence base suggests that iron supplementation may be a promising adjunctive therapeutic approach for TD management. Although further longitudinal trials are warranted to confirm efficacy and safety, emerging evidence provides encouraging support for the role of iron supplementation as an adjunctive intervention in TD treatment (Table 2).
Toxic amounts of iron consumption can lead to various gastrointestinal issues, including nausea, vomiting, diarrhea, constipation, and gastric upset [138]. At very high doses, more severe effects may be seen, including corrosive necrosis of the intestine or multisystem organ failure, which can be fatal [139]. Excess iron accumulates in the liver, heart, and endocrine glands, where it promotes the production of reactive oxygen species, leading to oxidative stress, cell damage, and organ dysfunction [140]. Toxicity can occur due to high individual doses or from gradual toxic accumulation. The latter is particularly of concern in patients who receive chronic transfusions for sickle cell disease, thalassemia, or hematologic malignancies [141]. To minimize the risk of toxicity, the tolerable upper intake level for iron is set at 45 mg per day in adults. For infants, children, and adolescents, the upper intake level varies by age, ranging from 40–45 mg per day (Table 3) [139].
6. Magnesium (Mg)
Mg is an essential mineral that acts as a cofactor in more than 600 biochemical reactions, making it indispensable for maintaining physiological stability and overall health [142,143,144,145,146,147]. One of Mg’s primary functions is regulating intracellular calcium and potassium, two electrolytes critical for neural excitability and synaptic signaling [148]. Within the nervous system, Mg modulates the body’s stress response, partly by influencing the hypothalamic–pituitary–adrenal (HPA) axis [149]. It decreases adrenocorticotropic hormone (ACTH) secretion from the pituitary and cortisol release from the adrenal glands, thereby mitigating anxiety [150,151]. Beyond its endocrine effects, Mg attenuates presynaptic glutamate release and glutamatergic hyperactivity, processes that are implicated in fear and panic responses [149,152,153,154,155].
Given its extensive neuro-modulatory functions, low Mg levels can contribute to psychiatric manifestations such as anxiety and depression, as well as neurological manifestations including headaches, seizures, muscle spasms, and tics [156,157,158]. Studies indicate that Mg deficiency induces neuromuscular hyperexcitability, often accompanied by convulsions and involuntary movements such as chorea and athetosis [159]. Mg insufficiency has also been linked to increased DA release, altered serotonin receptor modulation, and heightened defensive reactivity [28]. In the context of TD, Mg deficiency appears more prevalent among children with TS than among healthy peers [159]. However, a large retrospective analysis of medical records from 1204 children with TD and 1220 controls found no significant differences in whole-blood Mg concentrations between groups [68]. Children with TD had a mean Mg level of 1.58 ± 0.19 mmol/L, whereas controls had a mean Mg level of 1.57 ± 0.17 mmol/L (p = 0.318) [68].
Despite these inconsistencies, several studies suggest that Mg supplementation may alleviate tic severity. A randomized, double-blind controlled trial in children aged 7–14 years experiencing TS exacerbations found that daily Mg administration (0.5 mEq/kg) significantly reduced motor and phonic tics and improved overall impairment on the YGTSS [63]. In this study, the Mg group showed a 50% improvement from baseline, with a mean total tic count of 12.9 (SD = 11.20), compared with 26.7 (SD = 7.38) in the control group [63]. The authors presented a power calculation indicating that mean final total tic scores of 13 (SD = 10) for the treatment group and 23 (SD = 10) for the control group yielded a statistical power of 0.8 and a significance level of 0.05 [63]. Another study involving children aged 7–14 with TS reported progressive tic reduction over 90 days following oral administration of Mg and Vit B6, with symptom relief observed during clinical exacerbations [65]. Total tic scores lowered from 26.7 at baseline to 12.9 after 90 days of treatment, and the total effect on the YGTSS was a decrease from 58.1 to 18.8 [65]. Collectively, these findings highlight the therapeutic potential of Mg supplementation as an adjunctive intervention for TS and other TD, although further trials are warranted to establish efficacy and optimal dosing parameters (Table 2).
Excessive intake of magnesium can result in hypermagnesemia, which may manifest as gastrointestinal symptoms including diarrhea, nausea, and abdominal cramping [160]. At extremely high intakes, magnesium toxicity can cause serious cardiovascular effects, including arrhythmias and, in severe cases, cardiac arrest [161]. Individuals with impaired renal function or kidney failure are particularly at risk of toxicity, as their ability to remove excess magnesium is reduced or absent [161]. The tolerable upper intake level of magnesium for adults is set at 350 mg per day (Table 3) [162].
7. Zinc (Zn)
Zn is an essential trace element that acts as a cofactor for more than 300 enzymes involved in carbohydrates, protein, fatty acid, and nucleic acid metabolism [163,164,165,166]. These processes are vital for central nervous system development and maintenance [167,168]. In addition to neurogenesis, Zn exerts protective effects throughout the body by supporting immune function and antioxidant defenses [169,170]. At the molecular level, Zn acts as a cofactor for zinc-finger proteins, which contribute to neuronal metabolism by regulating gene expression and enzymatic activation [169]. Proper Zn homeostasis is essential for brain health, as both deficiency and excess can induce neural dysfunction [171]. Zn deficiency has been linked to oxidative stress and cognitive impairment, whereas accumulation beyond physiological levels can trigger neurotoxicity and neuronal apoptosis [169,171]. Within the brain, Zn contributes to melatonin synthesis in the pineal gland, thereby modulating DA activity [172]. This is noteworthy since dopaminergic dysregulation remains a defining feature of TD pathophysiology, and downregulating DA signaling is considered a central therapeutic strategy. Zn may also influence TD symptoms by modulating GABA neurotransmission, which exerts inhibitory effects on tic generation. Accordingly, Zn deficiency, which has been associated with impaired GABA synthesis in rat studies, may exacerbate tics [173]. Several studies have investigated Zn status in TD populations [68,174]. A large retrospective review of medical records from 1204 children with TD and 1220 healthy controls revealed significantly lower whole-blood Zn concentrations in the TD group (78.90 ± 11.50 μmol/L) compared to controls (83.90 ± 12.10 μmol/L; p < 0.01) [68]. Similarly, an outpatient study involving 161 children with TD and 178 healthy peers reported lower serum Zn levels among those with TD [71]. Collectively, these studies support an association between Zn deficiency and TD. However, evidence for the therapeutic efficacy of Zn supplementation remains inconclusive, underscoring the need for controlled interventional research to clarify its role in symptom modulation (Table 2).
While Zn is essential in trace amounts, excessive intake can result in toxicity, affecting the nervous, gastrointestinal, and respiratory systems [175]. Overconsumption of Zn may also induce relative copper deficiency, which can cause neurological symptoms such as numbness and weakness in the limbs [176]. To reduce the risk of toxicity, the European Food Safety Authority (ESFA) recommends a tolerable upper intake level of 25 mg per day for adults, whereas the U.S. Food and Drug Administration (FDA) sets the upper intake level at 40 mg per day (Table 3) [177].
8. Copper (Cu)
Cu is an essential trace element that plays a pivotal role in brain function. It supports enzymatic processes involved in DA metabolism, norepinephrine synthesis, catecholamine degradation, and cellular antioxidant defense [178,179,180]. Cu acts as a cofactor for multiple cuproenzymes, including cytochrome c oxidase, dopamine β-hydroxylase, amine oxidase, tyrosinase, and Cu-dependent superoxide dismutase [181,182]. These enzymes facilitate oxidation, hydroxylation, and disproportionation reactions vital to cellular energy metabolism [181,182]. Through these enzymatic functions, Cu contributes to mitochondrial respiration, oxidative stress regulation, and the biosynthesis of pigments, connective tissue, and neurotransmitters [183]. Dysregulation of Cu homeostasis has been implicated in several neurological disorders, particularly those affecting motor control. Elevated Cu concentrations may promote neurotoxicity through the formation of Cu–dopamine complexes, which oxidize DA and generate reactive oxygen species, ultimately causing dopaminergic neuronal injury [184,185]. This mechanism provides a plausible link between excess Cu and disorders characterized by dopaminergic dysregulation, including TD [184,185]. Although evidence on Cu involvement in TD remains limited, some studies suggest a potential association. In the same large retrospective study of 1204 children with TD and 1220 controls that identified lower Zn levels in the TD group, children with TD also exhibited significantly reduced whole blood Cu concentrations compared with healthy peers [68]. Cu levels were 17.80 ± 3.28 μmol/L in the TD group and 18.50 ± 3.54 μmol/L in the control group (p < 0.01) [68]. At present, there is insufficient evidence to support Cu supplementation as a therapeutic intervention for TD, highlighting the need for further mechanistic and interventional research (Table 2).
Cu toxicity resulting from excessive consumption may cause liver damage and gastrointestinal symptoms, including abdominal pain, nausea, vomiting, and diarrhea [186,187]. Maintaining an appropriate balance between Cu and Zn is important for overall health [188]. Normal serum levels are 10–25 µmol/L for copper and 12–15 µmol/L for zinc [189,190]. Disturbances in the copper-to-zinc ratio have been associated with serious diseases and health problems, highlighting the need for careful consideration when supplementing with either trace element [190]. The tolerable upper intake level for Cu in adults is set at 10,000 mcg per day (Table 3) [190].
9. Non-Nutritional Strategies
The management of tic disorders is complex and can generally be divided into three main approaches: (1) pharmacological therapy, (2) behavioral therapy, and (3) neurostimulation [191,192].
Pharmacologic treatment options include alpha-2-agonists, anticonvulsants, dopamine receptor antagonists, dopamine depletors, and muscle relaxants [192,193]. Other options include cannabis and traditional Chinese herbal therapies [192,193]. When first-line options are insufficient, second-line strategies often involve the use of antipsychotics such as fluphenazine, aripiprazole, risperidone, and ziprasidone [191]. While generally effective, these medications carry the risk of inducing side effects such as metabolic syndrome and tardive dyskinesia [191]. Only three antipsychotic agents (haloperidol, pimozide, and aripiprazole) have been approved by the FDA for managing TD symptoms [194]. Of these, aripiprazole belongs to the class of atypical antipsychotics, which are more selective dopamine D2 receptor blockers, although they may also influence serotonin signaling [195]. Other atypical antipsychotics include risperidone, clozapine, olanzapine, and quetiapine [195].
Non-pharmacologic strategies provide alternatives for patients who do not respond to drug therapy or who wish to avoid systemic side effects [196]. The American Academy of Neurology (AAN) recognizes behavioral therapy and deep brain stimulation (DBS) as the two primary non-pharmacologic interventions [194]. Behavioral therapy is widely considered the most effective psychotherapeutic approach for tic disorders [197,198]. This is often delivered as Comprehensive Behavioral Intervention for Tics (CBIT), which integrates three previously separate interventions: habit reversal training, relaxation therapy, and awareness training [191]. CBIT has received a “high confidence” recommendation in the AAN’s clinical practice guidelines and is regarded as a non-invasive first-line treatment for TS [196]. DBS is a relatively new intervention in which electrodes are surgically implanted into targeted brain regions, enabling modulation of neural activity through electrical stimulation [194,197]. In tic disorders, this targeted modulation may help normalize dysfunctional neural circuits involved in generating tics.
10. Conclusions
TDs are neurodevelopmental conditions characterized by persistent motor and/or vocal tics, with diagnostic categories including TS, CTD, and TTD. This review examined the association between TD, particularly TS and CTD, and serum concentrations of several key micronutrients (Vit D, Vit B6, Vit A, iron, Mg, Zn, and Cu). These micronutrients play critical neurobiological roles, and their insufficiencies may contribute to TD pathophysiology by disrupting neurotransmitter regulation, neurodevelopmental processes, and cellular redox homeostasis. Accumulating evidence suggests association of deficiencies in these micronutrients with both the occurrence and severity of TDs. Preliminary clinical studies indicate that supplementation with specific nutrients including Vit D, Vit B6, Vit A, iron, Mg, Zn, and Cu may alleviate tic symptoms, although some findings are variable across studies. Further neurochemical, genetic, and longitudinal research is warranted to clarify the mechanisms underlying these associations and to evaluate the efficacy of nutrient-based interventions as adjunctive strategies for TD management and prevention.
Author Contributions
Conceptualization, S.M. (Samskruthi Madireddy) and S.M. (Sahithi Madireddy); methodology, S.M. (Samskruthi Madireddy) and S.M. (Sahithi Madireddy); validation, S.M. (Samskruthi Madireddy) and S.M. (Sahithi Madireddy); investigation, S.M. (Samskruthi Madireddy) and S.M. (Sahithi Madireddy); resources, S.M. (Samskruthi Madireddy) and S.M. (Sahithi Madireddy); data curation, S.M. (Samskruthi Madireddy) and S.M. (Sahithi Madireddy); writing—original draft preparation, S.M. (Samskruthi Madireddy) and S.M. (Sahithi Madireddy); writing—review and editing, S.M. (Samskruthi Madireddy) and S.M. (Sahithi Madireddy); project administration, S.M. (Samskruthi Madireddy) and S.M. (Sahithi Madireddy). All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| TS | Tourette syndrome |
| TD | Tic disorder |
| TTD | Transient tic disorder |
| CTD | Chronic motor or vocal tic disorder |
| OCD | Obsessive–compulsive disorder |
| ASD | Autism spectrum disorder |
| ADHD | Attention-deficit/hyperactivity disorder |
| CSTC | Cortico-striato-thalamo-cortical |
| DA | Dopamine |
| GABA | γ-aminobutyric acid |
| Vit D | Vitamin D |
| VDR | Vitamin D receptor |
| UVB | Ultraviolet B |
| GDNF | Glial cell line-derived neurotrophic factor |
| YGTSS | Yale Global Tic Severity Scale |
| HPLC | High-performance Liquid Chromatography |
| MS/MS | Tandem Mass Spectrometry |
| HC | Healthy Controls |
| IU | International Units |
| Vit B6 | Vitamin B6 |
| PLP | Pyridoxal-5′-phosphate |
| PN | Pyridoxine |
| PM | Pyridoxamine |
| PL | Pyridoxal |
| PNP | Pyridoxine 5′-phosphate |
| PMP | Pyridoxamine 5′-phosphate |
| RA | Retinoic acid |
| RAR | Retinoic acid receptor |
| UL | Upper intake level |
| HPA | Hypothalamic–pituitary–adrenal |
| ACTH | Adrenocorticotropic hormone |
| Mg | Magnesium |
| Zn | Zinc |
| Cu | Copper |
| MRI | Magnetic resonance imaging |
| FDA | Food and Drug Administration |
| AAN | American Academy of Neurology |
| DBS | deep brain stimulation |
| CBIT | Comprehensive Behavioral Intervention |
References
- Widomska, J.; DeWitte, W.; Buitelaar, J.K.; Glennon, J.C.; Poelmans, G. Molecular Landscape of Tourette’s Disorder. Int. J. Mol. Sci. 2023, 24, 1428. [Google Scholar] [CrossRef] [PubMed]
- Serajee, F.J.; Mahbubul Huq, A.H.M. Advances in Tourette syndrome: Diagnoses and treatment. Pediatr. Clin. N. Am. 2015, 62, 687–701. [Google Scholar] [CrossRef]
- Du, J.C.; Chiu, T.F.; Lee, K.M.; Wu, H.L.; Yang, Y.C.; Hsu, S.Y.; Sun, C.S.; Hwang, B.; Leckman, J.F. Tourette syndrome in children: An updated review. Pediatr. Neonatol. 2010, 51, 255–264. [Google Scholar] [CrossRef]
- Buse, J.; Schoenefeld, K.; Münchau, A.; Roessner, V. Neuromodulation in tourette syndrome: Dopamine and beyond. Neurosci. Biobehav. Rev. 2013, 37, 1069–1084. [Google Scholar] [CrossRef]
- Hoekstra, P.J.; Kallenberg, C.G.M.; Korf, J.; Minderaa, R.B. Is Tourette’s syndrome an autoimmune disease? Mol. Psychiatry 2002, 7, 437–445. [Google Scholar] [CrossRef]
- Häge, A.; Krämer, R.; Dunlap, M.; Mechler, K.; Müller-Vahl, K.R.; Nagy, P. Emerging therapeutic approaches for Tourette syndrome and other tic disorders—A systematic review of current clinical trials. Eur. Child Adolesc. Psychiatry 2025, 34, 1719–1729. [Google Scholar] [CrossRef] [PubMed]
- Mittal, S.O. Tics and Tourette’s syndrome. Drugs Context 2020, 9, 2019–2020. [Google Scholar] [CrossRef]
- Yang, C.; Zhang, L.; Zhu, P.; Zhu, C.; Guo, Q. The prevalence of tic disorders for children in China: A systematic review and meta-analysis. Medicine 2016, 95, e4354. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.-S.; Cui, Y.-H.; Sun, D.; Lu, Q.; Jiang, Y.-W.; Jiang, L.; Wang, J.-Q.; Luo, R.; Fang, F.; Zhou, S.-Z.; et al. Current status, diagnosis, and treatment recommendation for tic disorders in China. Front. Psychiatry 2020, 11, 774. [Google Scholar] [CrossRef]
- Bitsko, R.H.; Claussen, A.H.; Lichstein, J.; Black, L.I.; Jones, S.E.; Danielson, M.L.; Hoenig, J.M.; Davis Jack, S.P.; Brody, D.J.; Gyawali, S.-G.; et al. Mental Health Surveillance Among Children—United States, 2013–2019. MMWR Suppl. 2022, 71, 1–42. [Google Scholar] [CrossRef]
- CDC’s Data and Statistics on Tourette Syndrome. Available online: https://www.cdc.gov/tourette-syndrome/data/index.html (accessed on 27 September 2025).
- Eapen, V.; Robertson, M.M. Are there distinct subtypes in Tourette syndrome? Pure-Tourette syndrome versus Tourette syndrome-plus, and simple versus complex tics. Neuropsychiatr. Dis. Treat. 2015, 11, 1431–1436. [Google Scholar] [CrossRef]
- Set, K.K.; Warner, J.N. Tourette syndrome in children: An update. Curr. Probl. Pediatr. Adolesc. Health Care 2021, 51, 101032. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Trescher, W.; Byler, D. Tourette Syndrome and Comorbid Neuropsychiatric conditions. Curr. Dev. Disord. Rep. 2016, 3, 217–221. [Google Scholar] [CrossRef]
- Robertson, M.M.; Eapen, V.; Singer, H.S.; Martino, D.; Scharf, J.M.; Paschou, P.; Roessner, V.; Woods, D.W.; Hariz, M.; Mathews, C.A.; et al. Gilles de la Tourette syndrome. Nat. Rev. Dis. Prim. 2017, 3, 16097. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.J.; Wong, L.C.; Lee, W.T. Immunological dysfunction in Tourette syndrome and related disorders. Int. J. Mol. Sci. 2021, 22, 853. [Google Scholar] [CrossRef] [PubMed]
- Gloor, F.T.; Walitza, S. Tic disorders and Tourette syndrome: Current concepts of etiology and treatment in children and adolescents. Neuropediatrics 2016, 47, 84–96. [Google Scholar] [CrossRef]
- Qi, Y.; Zheng, Y.; Li, Z.; Liu, Z.; Xiong, L. Genetic studies of tic disorders and tourette syndrome. Methods. Mol. Biol. 2019, 2011, 547–571. [Google Scholar] [CrossRef]
- Wang, S.; Xu, Q.; Wang, A.; Yuan, F.; Luo, X.; Wang, Y.; Guo, M.; Zhang, Y.; Zhang, W.; Ji, X.; et al. Correlation Between Tic Disorders and Serum 25-Hydroxyvitamin D Levels in Chinese Children. Front. Pediatr. 2022, 10, 833371. [Google Scholar] [CrossRef]
- Qing, L.; Dan, S.; Zhisheng, L. Interpretation of expert consensus for diagnosis and treatment of tic disorders in China. Chin. J. Appl. Clin. Pediatr. 2021, 36, 647–652. [Google Scholar] [CrossRef]
- Yael, D.; Vinner, E.; Bar-Gad, I. Pathophysiology of tic disorders. Mov. Disord. 2015, 30, 1171–1178. [Google Scholar] [CrossRef]
- Müller-Vahl, K.R.; Loeber, G.; Kotsiari, A.; Müller-Engling, L.; Frieling, H. Gilles de la tourette syndrome is associated with hypermethylation of the dopamine D2 receptor gene. J. Psychiatr. Res. 2016, 86, 1–8. [Google Scholar] [CrossRef]
- Bai, L.; Jin, M.; Zhang, Q.; Sun, S. Progress in research on nutrition, neuroinflammation and dopaminergic alterations in Tic disorders. Front. Pediatr. 2025, 13, 1526117. [Google Scholar] [CrossRef] [PubMed]
- Deeb, W.; Malaty, I.A.; Mathews, C.A. Tourette disorder and other tic disorders. Handb. Clin. Neurol. 2019, 165, 123–153. [Google Scholar] [CrossRef] [PubMed]
- Kanaan, A.S.; Gerasch, S.; García-García, I.; Lampe, L.; Pampel, A.; Anwander, A.; Near, J.; Möller, H.E.; Müller-Vahl, K. Pathological glutamatergic neurotrans- mission in Gilles de la Tourette syndrome. Brain 2017, 140, 218–234. [Google Scholar] [CrossRef]
- Mogwitz, S.; Buse, J.; Ehrlich, S.; Roessner, V. Clinical pharmacology of dopamine-modulating agents in Tourette’s syndrome. Int. Rev. Neurobiol. 2013, 112, 281–349. [Google Scholar] [CrossRef]
- Draper, A.; Stephenson, M.C.; Jackson, G.M.; Pepes, S.; Morgan, P.S.; Morris, P.G. Increased GABA contributes to enhanced control over motor excitability in Tourette syndrome. Curr. Biol. 2014, 24, 2343–2347. [Google Scholar] [CrossRef]
- Ludlow, A.K.; Rogers, S. Understanding the impact of diet and nutrition on symptoms of Tourette syndrome: A scoping review. J. Child Health Care 2018, 22, 68–83. [Google Scholar] [CrossRef] [PubMed]
- van Galen, K.A.; Schrantee, A.; Ter Horst, K.W.; la Fleur, S.E.; Booij, J.; Constable, R.T.; Schwartz, G.J.; DiLeone, R.J.; Serlie, M.J. Brain responses to nutrients are severely impaired and not reversed by weight loss in humans with obesity: A randomized crossover study. Nat. Metab. 2023, 5, 1059–1072. [Google Scholar] [CrossRef]
- Madireddy, S.; Madireddy, S. Regulation of Reactive Oxygen Species-Mediated Damage in the Pathogenesis of Schizophrenia. Brain Sci. 2020, 10, 742. [Google Scholar] [CrossRef]
- Cui, X.; Pertile, R.; Liu, P.; Eyles, D.W. Vitamin D regulates tyrosine hydroxylase expression: N-cadherin a possible mediator. Neurosci. 2015, 304, 90–100. [Google Scholar] [CrossRef]
- di Michele, F.; Talamo, A.; Niolu, C.; Siracusano, A. Vitamin D and N-Acetyl Cysteine Supplementation in Treatment-Resistant Depressive Disorder Patients: A General Review. Curr. Pharm. Des. 2020, 26, 2442–2459. [Google Scholar] [CrossRef]
- Mora, J.R.; Iwata, M.; von Andrian, U.H. Vitamin effects on the immune system: Vitamins A and D take centre stage. Nat. Rev. Immunol. 2008, 8, 685–698. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Skv, M.; Abraham, S.M.; Eshwari, O.; Golla, K.; Jhelum, P.; Maity, S.; Komal, P. Tremendous Fidelity of Vitamin D3 in Age-related Neurological Disorders. Mol. Neurobiol. 2024, 61, 7211–7238. [Google Scholar] [CrossRef]
- Athanassiou, L.; Mavragani, C.P.; Koutsilieris, M. The immunomodulatory properties of vitamin D. Mediterr. J. Rheumatol. 2022, 33, 7–13. [Google Scholar] [CrossRef]
- Orme, R.P.; Middleditch, C.; Waite, L.; Fricker, R.A. The Role of Vitamin D3 in the Development and Neuroprotection of Midbrain Dopamine Neurons. Vitam. Horm. 2016, 100, 273–297. [Google Scholar] [CrossRef] [PubMed]
- Eyles, D.W.; Burne, T.H.J.; McGrath, J.J. Vitamin D, effects on brain development, adult brain function and the links between low levels of vitamin D and neuropsychiatric disease. Front. Neuroendocrinol. 2013, 34, 47–64. [Google Scholar] [CrossRef] [PubMed]
- Groves, N.J.; McGrath, J.J.; Burne, T.H. Vitamin D as a neurosteroid affecting the developing and adult brain. Annu. Rev. Nutr. 2014, 34, 117–141. [Google Scholar] [CrossRef]
- Muscogiuri, G.; Altieri, B.; Annweiler, C.; Balercia, G.; Pal, H.B.; Boucher, B.J.; Cannell, J.J.; Foresta, C.; Grübler, M.R.; Kotsa, K.; et al. Vitamin D and chronic diseases: The current state of the art. Arch. Toxicol. 2017, 91, 97–107. [Google Scholar] [CrossRef]
- Lin, X.; Jiang, J.; Chen, X.; Chen, Y. Vitamin D status and tic disorder: A systematic review and meta-analysis of observational studies. Front. Pediatr. 2023, 11, 1173741. [Google Scholar] [CrossRef]
- Cui, X.; Pelekanos, M.; Burne, T.H.J.; McGrath, J.J.; Eyles, D.M. Maternal vitamin D deficiency alters the expression of genes involved in dopamine specification in the developing rat mesencephalon. Neurosci. Lett. 2010, 486, 220–223. [Google Scholar] [CrossRef]
- Eyles, D.W.; Liu, P.Y.; Josh, P.; Cui, X. Intracellular distribution of the vitamin D receptor in the brain: Comparison with classic target tissues and redistribution with development. Neuroscience 2014, 268, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pertile, R.A.N.; Cui, X.; Hammond, L.; Eyles, D.W. Vitamin D regulation of GDNF/Ret signaling in dopaminergic neurons. FASEB J. 2018, 32, 819–828. [Google Scholar] [CrossRef]
- Brown, J.; Bianco, J.I.; Mcgrath, J.J.; Eyles, D.W. 1,25-dihydroxyvitamin D3 induces nerve growth factor, promotes neurite outgrowth and inhibits mitosis in embryonic rat hippocampal neurons. Neurosci. Lett. 2003, 343, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Kalueff, A.V.; Eremin, K.O.; Tuohimaa, P. Mechanisms of neuroprotective action of vitamin D3. Biochemistry 2004, 69, 738–741. [Google Scholar] [CrossRef]
- Kočovská, E.; Fernell, E.; Billstedt, E.; Minnis, H.; Gillberg, C. Vitamin D and autism: Clinical review. Res. Dev. Disabil. 2012, 33, 1541–1550. [Google Scholar] [CrossRef] [PubMed]
- Lerner, A.; Bagic, A.; Simmons, J.M.; Mari, Z.; Bonne, O.; Xu, B.; Kazuba, D.; Herscovitch, P.; Carson, R.E.; Murphy, D.L.; et al. Widespread abnormality of the γ-aminobutyric acid-ergic system in Tourette syndrome. Brain 2012, 135, 1926–1936. [Google Scholar] [CrossRef]
- Madireddy, S.; Madireddy, S. Therapeutic Interventions to Mitigate Mitochondrial Dysfunction and Oxidative Stress-Induced Damage in Patients with Bipolar Disorder. Int. J. Mol. Sci. 2022, 23, 1844. [Google Scholar] [CrossRef]
- Menéndez, S.G.; Manucha, W. Vitamin D as a modulator of neuroinflammation: Implications for brain health. Curr. Pharm. Des. 2024, 30, 323–332. [Google Scholar] [CrossRef]
- Kočovská, E.; Gaughran, F.; Krivoy, A.; Meier, U.C. Vitamin-D deficiency as a potential environmental risk factor in multiple sclerosis, schizophrenia, and autism. Front. Psych. 2017, 8, 47. [Google Scholar] [CrossRef]
- Wang, C.-X.; Wang, B.; Sun, J.-J.; Xiao, C.-Y.; Ma, H.; Jia, F.-Y.; Li, H.-H. Circulating retinol and 25(OH)D contents and their association with symptoms in children with chronic tic disorders. Eur. Child Adolesc. Psychiatry 2024, 33, 1017–1028. [Google Scholar] [CrossRef]
- Pertile, R.A.N.; Brigden, R.; Raman, V.; Cui, X.; Du, Z.; Eyles, D. Vitamin D: A potent regulator of dopaminergic neuron differentiation and function. J. Neurochem. 2023, 166, 779–789. [Google Scholar] [CrossRef]
- Li, H.-H.; Wang, B.; Shan, L.; Wang, C.-X.; Jia, F.-Y. Serum levels of 25-hydroxyvitamin D in children with tic disorders. Chin. J. Contemp. Pediatr. 2017, 19, 1165–1168. [Google Scholar]
- Li, H.-H.; Shan, L.; Wang, B.; Du, L.; Xu, Z.-D.; Jia, F.-Y. Serum 25-hyroxyvitamin d levels and tic severity in Chinese children with tic disorders. Psychiatry Res. 2018, 267, 80–84. [Google Scholar] [CrossRef]
- Wang, A.; Zhang, Z.; Li, Y.; Tian, Y. Correlation between Vitamin A, D and E and Severity of Tic Disorder. J. Pediatr. Pharm. 2022, 28, 31–35. [Google Scholar] [CrossRef]
- Ge, L.; Jin, X.; Zhu, L. Expression and correlation of 25 hydroxyvitamin D and nerve growth factor in serum of children with tourette syndrome. Labeled Immunoass. Clin. Med. 2020, 27, 1943–1946. [Google Scholar]
- You, H.; Zhou, Y.; Xie, J.; Jin, Z.; Wang, G.; Sun, K. Correlation between serum vitamin D and tic disorder. Chin. J. Child Health Care 2022, 30, 904–907. [Google Scholar] [CrossRef]
- Wang, Y.M.; Jia, R.M.; Song, H.F. Relationship between tic disorder in children and serum iron, ferritin and 25-hydroxyvitamin D. Med. Innov. China 2022, 19, 45–50. [Google Scholar] [CrossRef]
- Li, L.-N.; Xu, Y.; He, X.-J.; Zhang, W.-X.; Zhang, H.-F.; Song, L.; Shi, H.-S.; Tian, X.-Y.; Yan, X. Effects and mechanisms of vitamins A and D on behavior associated with Tourette syndrome in rats. Front. Nutr. 2025, 12, 1561693. [Google Scholar] [CrossRef]
- Mohamed, Z.A.; Bai, M.; Dong, H.; Xue, Y.; Jia, F.; Feng, J. Efficacy of high-dose vs. low-dose vitamin D3 supplementation in children with chronic tic disorders: A randomized controlled trial. Nutr. J. 2025, 24, 44. [Google Scholar] [CrossRef]
- Li, H.-H.; Xu, Z.-D.; Wang, B.; Feng, J.-Y.; Dong, H.-Y.; Jia, F.-Y. Clinical improvement following vitamin D3 supplementation in children with chronic tic disorders. Neuropsychiatr. Dis. Treat. 2019, 15, 2443–2450. [Google Scholar] [CrossRef]
- Garcia-Lopez, R.; Perea-Milla, E.; Garcia, C.R.; Rivas-Ruiz, F.; Romero-Gonzalez, J.; Moreno, J.L.; Faus, V.; Aguas Gdel, C.; Diaz, J.C. New therapeutic approach to Tourette Syndrome in children based on a randomized placebo-controlled double-blind phase IV study of the effectiveness and safety of magnesium and vitamin B6. Trials 2009, 10, 16. [Google Scholar] [CrossRef]
- Rizzo, R.; Prato, A.; Scerbo, M.; Saia, F.; Barone, R.; Curatolo, P. Use of Nutritional Supplements Based on L-Theanine and Vitamin B6 in Children with Tourette Syndrome, with Anxiety Disorders: A Pilot Study. Nutrients 2022, 14, 852. [Google Scholar] [CrossRef]
- García-López, R.; Romero-González, J.; Perea-Milla, E.; Ruiz-García, C.; Rivas-Ruiz, F.; de Las Mulas Béjar, M. An open study evaluating the efficacy and security of magnesium and vitamin B6 as a treatment of Tourette syndrome in children. Med. Clin. 2008, 131, 689–691. [Google Scholar] [CrossRef]
- Hou, C.; Yang, Z.C.; Liu, X.M.; Sun, M.X.; Yi, M.J. Correlation between serum vitamin A/D and tic disorder symptom severity and clinical type. Chin. J. Behav. Med. Brain Sci. 2020, 29, 308–314. [Google Scholar] [CrossRef]
- Gorman, D.A.; Zhu, H.; Anderson, G.M.; Davies, M.; Peterson, B.S. Ferritin Levels and Their Association with Regional Brain Volumes in Tourette’s Syndrome. Am. J. Psychiatry 2006, 163, 1264–1272. [Google Scholar] [CrossRef] [PubMed]
- Qian, R.; Ma, Y.; You, L.; Zhao, Y.; Li, S.; Shen, J.; Jiang, L.; Yang, C.; Jiang, P.; Yuan, Z.; et al. The Blood Levels of Trace Elements Are Lower in Children with Tic Disorder: Results from a Retrospective Study. Front. Neurol. 2019, 10, 1324. [Google Scholar] [CrossRef]
- Avrahami, M.; Barzilay, R.; HarGil, M.; Weizman, A.; Watemberg, N. Serum ferritin levels are lower in children with tic disorders compared with children without tics: A cross-sectional study. J. Child Adolesc. Psychopharmacol. 2017, 27, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, D.; Burkman, E. Relationship of serum ferritin level and tic severity in children with Tourette syndrome. Child. Nerv. Syst. 2017, 33, 1373–1378. [Google Scholar] [CrossRef]
- Luo, M.; Xiao, H.; Rao, M.; Li, J.; Zhao, Y. Levels and significance of serum ferritin, zinc and vitamin D in children with tic disorders. Lab. Med. Clin. 2023, 20, 3611–3614. [Google Scholar] [CrossRef]
- Galior, K.; Grebe, S.; Singh, R. Development of vitamin D toxicity from overcorrection of vitamin D deficiency: A review of case reports. Nutrients 2018, 10, 953. [Google Scholar] [CrossRef]
- Rizzoli, R. Vitamin D supplementation: Upper limit for safety revisited? Aging Clin. Exp. Res. 2021, 33, 19–24. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health (NIH), Office of Dietary Supplements (ODS). Vitamin D. Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/vitamind-HealthProfessional/ (accessed on 9 December 2025).
- National Institutes of Health (NIH), Office of Dietary Supplements (ODS). Dietary Supplement Fact Sheets. Available online: https://ods.od.nih.gov/factsheets/ (accessed on 9 December 2025).
- Zhao, Y.; Zhao, T.; Liu, X.; Li, R.; Xiao, H. Vitamin B6 Nutrition, Metabolism, and the Relationship of Diseases: Current Concepts and Future Research. J. Future Foods 2025, 6, 765–779. [Google Scholar] [CrossRef]
- Bird, R.P. Chapter Four—The Emerging Role of Vitamin B6 in Inflammation and Carcinogenesis. Adv. Food. Nutr. Res. 2018, 83, 151–194. [Google Scholar] [CrossRef]
- Mocellin, S.; Briarava, M.; Pilati, P. Vitamin B6 and Cancer Risk: A Field Synopsis and Meta-Analysis. J. Natl. Cancer Inst. 2017, 109, djw230. [Google Scholar] [CrossRef] [PubMed]
- Sato, K. Why is vitamin B6 effective in alleviating the symptoms of autism? Med. Hypotheses 2018, 115, 103–106. [Google Scholar] [CrossRef]
- Spinneker, A.; Sola, R.; Lemmen, V.; Castillo, M.J.; Pietrzik, K.; González-Gross, M. Vitamin B6 status, deficiency and its consequences—An overview. Nutr. Hosp. 2007, 22, 7–24. [Google Scholar]
- Komatsu, S.; Yanaka, N.; Matsubara, K.; Kato, N. Antitumor effect of vitamin B6 and its mechanisms. Biochim. Biophys. Acta 2003, 1647, 127–130. [Google Scholar] [CrossRef]
- Wondrak, G.T.; Jacobson, E.L. Vitamin B6: Beyond coenzyme functions. Subcell. Biochem. 2012, 56, 291–300. [Google Scholar] [CrossRef]
- Marzio, A.; Merigliano, C.; Gatti, M.; Verni, F. Sugar and chromosome stability: Clastogenic effects of sugars in vitamin B6-deficient cells. PLoS Genet. 2014, 10, e1004199. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.S.; Roh, S.Y.; Jeong, E.H.; Kim, B.S.; Sunwoo, M.K. Ginkgotoxin Induced Seizure Caused by Vitamin B6 Deficiency. J. Epilepsy Res. 2015, 5, 104–106. [Google Scholar] [CrossRef]
- Rivero, M.; Novo, N.; Medina, M. Pyridoxal 5′-Phosphate Biosynthesis by Pyridox-(am)-ine 5′-Phosphate Oxidase: Species-Specific Features. Int. J. Mol. Sci. 2024, 25, 3174. [Google Scholar] [CrossRef]
- Salam, R.A.; Zuberi, N.F.; Bhutta, Z.A. Pyridoxine (vitamin B6) supplementation during pregnancy or labour for maternal and neonatal outcomes. Cochrane Database Syst. Rev. 2015, 2015, CD000179. [Google Scholar] [CrossRef]
- Schellack, N.; Yotsombut, K.; Sabet, A.; Nafach, J.; Hiew, F.L.; Kulkantrakorn, K. Expert Consensus on Vitamin B6 Therapeutic Use for Patients: Guidance on Safe Dosage, Duration and Clinical Management. Drug Healthc. Patient Saf. 2025, 17, 97–108. [Google Scholar] [CrossRef]
- Miodownik, C.; Lerner, V.; Vishne, T.; Sela, B.-A.; Levine, J. High-dose Vitamin B6 Decreases Homocysteine Serum Levels in Patients with Schizophrenia and Schizoaffective Disorders: A Preliminary Study. Clin. Neuropharmacol. 2007, 30, 13–17. [Google Scholar] [CrossRef]
- Lerner, V.; Miodownik, C.; Kaptsan, A.; Cohen, H.; Loewenthal, U.; Kotler, M. Vitamin B6 as add-on treatment in chronic schizophrenic and schizoaffective patients: A double-blind, placebo-controlled study. J. Clin. Psychiatry 2002, 63, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Han, A.; Almeida, L.; Anand, N.; Salloum, I.M.; Kanaan, S.; Gadad, B.S.; Daher, J.P.L. Exploring neuropsychiatric manifestations of vitamin B complex deficiencies. Front. Psychiatry 2025, 16, 1569826. [Google Scholar] [CrossRef] [PubMed]
- Orywal, K.; Socha, K.; Iwaniuk, P.; Kaczyński, P.; Farhan, J.A.; Zoń, W.; Łozowicka, B.; Perkowski, M.; Mroczko, B. Vitamins in the Prevention and Support Therapy of Neurodegenerative Diseases. Int. J. Mol. Sci. 2025, 26, 1333. [Google Scholar] [CrossRef] [PubMed]
- Przybelski, A.G.; Bendlin, B.B.; Jones, J.E.; Vogt, N.M.; Przybelski, R.J. Vitamin B6 and vitamin D deficiency co-occurrence in geriatric memory patients. Alzheimer’s Dement. Diagn. Assess. Dis. Monit. 2024, 16, e12525. [Google Scholar] [CrossRef]
- Indika, N.; Frye, R.E.; Rossignol, D.A.; Owens, S.C.; Senarathne, U.D.; Grabrucker, A.M.; Perera, R.; Engelen, M.P.; Deutz, N.E. The Rationale for Vitamin, Mineral, and Cofactor Treatment in the Precision Medical Care of Autism Spectrum Disorder. J. Pers. Med. 2023, 13, 252. [Google Scholar] [CrossRef]
- Malouf, R.; Grimley, E.J. Vitamin B6 for cognition. Cochrane Database Syst. Rev. 2003, 4, CD004393. [Google Scholar] [CrossRef]
- Parra, M.; Stahl, S.; Hellmann, H. Vitamin B6 and Its Role in Cell Metabolism and Physiology. Cells 2018, 7, 84. [Google Scholar] [CrossRef]
- Hadtstein, F.; Vrolijk, M. Vitamin B-6-Induced Neuropathy: Exploring the Mechanisms of Pyridoxine Toxicity. Adv. Nutr. 2021, 12, 1911–1929. [Google Scholar] [CrossRef]
- National Institutes of Health (NIH), Office of Dietary Supplements (ODS). Vitamin B6. Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/vitaminb6-HealthProfessional/ (accessed on 9 December 2025).
- Olson, C.R.; Mello, C.V. Significance of vitamin A to brain function, behavior and learning. Mol. Nutr. Food. Res. 2010, 54, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Marie, A.; Darricau, M.; Touyarot, K.; Parr-Brownlie, L.C.; Bosch-Bouju, C. Role and Mechanism of Vitamin A Metabolism in the Pathophysiology of Parkinson’s Disease. J. Park. Dis. 2021, 11, 949–970. [Google Scholar] [CrossRef]
- Maden, M. Retinoic acid in the development, regeneration and maintenance of the nervous system. Nat. Rev. Neurosci. 2007, 8, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Das, B.C.; Dasgupta, S.; Ray, S.K. Potential therapeutic roles of retinoids for prevention of neuroinflammation and neurodegeneration in Alzheimer’s disease. Neural Regen. Res. 2019, 14, 1880–1892. [Google Scholar] [CrossRef]
- Parastouei, K.; Mirshafiey, A.; Eshraghian, M.R.; Shiri-Shahsavar, M.R.; Solaymani-Mohammadi, F.; Chahardoli, R.; Alvandi, E.; Saboor-Yaraghi, A.A. The effect of 1, 25(OH)2 D3 (calcitriol) alone and in combination with all-trans retinoic acid on ROR-γt, IL-17, TGF-β, and FOXP3 gene expression in experimental autoimmune encephalomyelitis. Nutr. Neurosci. 2018, 21, 210–218. [Google Scholar] [CrossRef]
- Chesney, R.W.; Han, X. Differential regulation of TauT by calcitriol and retinoic acid via VDR/RXR in LLC-PK1 and MCF-7 cells. Adv. Exp. Med. Biol. 2013, 776, 291–305. [Google Scholar] [CrossRef] [PubMed]
- Hou, N.; Ren, L.; Gong, M.; Bi, Y.; Gu, Y.; Dong, Z.; Liu, Y.; Chen, J.; Li, T. Vitamin A deficiency impairs spatial learning and memory: The mechanism of abnormal CBP-dependent histone acetylation regulated by retinoic acid receptor alpha. Mol. Neurobiol. 2015, 51, 633–647. [Google Scholar] [CrossRef]
- Chen, B.-W.; Zhang, K.-W.; Zhang, K.-W.; Chen, S.-J.; Chun, Y.; Li, P.-G. Vitamin A Deficiency Exacerbates Gut Microbiota Dysbiosis and Cognitive Deficits in Amyloid Precursor Protein/Presenilin 1 Transgenic Mice. Front. Aging Neurosci. 2021, 13, 753351. [Google Scholar] [CrossRef]
- Pallet, V.; Touyarot, K. Vitamin A and cognitive processes. Nutr. Aging 2015, 3, 21–31. [Google Scholar] [CrossRef]
- Rataj-Baniowska, M.; Niewiadomska-Cimicka, A.; Paschaki, M.; Szyszka-Niagolov, M.; Carramolino, L.; Torres, M.; Dollé, P.; Krężel, W. Retinoic acid receptor β controls development of striatonigral projection neurons through FGF-dependent and Meis1-dependent mechanisms. J. Neurosci. 2015, 35, 14467–14475. [Google Scholar] [CrossRef]
- Pan, J.; Yu, J.; Sun, L.; Xie, C.; Chang, L.; Wu, J.; Hawes, S.; Saez-Atienzar, S.; Zheng, W.; Kung, J.; et al. ALDH1A1 regulates postsynaptic μ-opioid receptor expression in dorsal striatal projection neurons and mitigates dyskinesia through transsynaptic retinoic acid signaling. Sci. Rep. 2019, 9, 3602. [Google Scholar] [CrossRef] [PubMed]
- Niewiadomska-Cimicka, A.; Krzyżosiak, A.; Ye, T.; Podleśny-Drabiniok, A.; Dembélé, D.; Dollé, P.; Krężel, W. Genome-wide analysis of RARβ transcriptional targets in mouse striatum links retinoic acid signaling with Huntington’s disease and other neurodegenerative disorders. Mol. Neurobiol. 2017, 54, 3859–3878. [Google Scholar] [CrossRef]
- Lane, M.A.; Bailey, S.J. Role of retinoid signalling in the adult brain. Prog. Neurobiol. 2005, 75, 275–293. [Google Scholar] [CrossRef] [PubMed]
- Bian, Q.X.; Jiang, B.B.; Tian, W.T.; Chen, D.Y. The application value of vitamin A, D and five-dimension lysine granules in children with tic disorders. Hebei Med. J. 2020, 42, 3601–3604. [Google Scholar] [CrossRef]
- Olson, J.M.; Ameer, M.A.; Goyal, A. Vitamin A Toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Pestalardo, M.L.; Bevilacqua, C.S.; Amante, M.F. Vitamin A toxicity and hepatic pathology: A comprehensive review. World J. Hepatol. 2025, 17, 107738. [Google Scholar] [CrossRef]
- Penniston, K.L.; Tanumihardjo, S.A. The acute and chronic toxic effects of vitamin A. Am. J. Clin. Nutr. 2006, 83, 191–201. [Google Scholar] [CrossRef]
- National Institutes of Health (NIH), Office of Dietary Supplements (ODS). Vitamin A and Carotenoids. Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/vitamina-HealthProfessional/ (accessed on 9 December 2025).
- Gkotsoulias, D.G.; Rullmann, M.; Schmitt, S.; Bujanow, A.; Zientek, F.; Messerschmidt, K.; Pampel, A.; Büttner, A.P.; Schildan, A.; Sabri, O.; et al. Abnormalities of iron homeostasis and the dopaminergic system in Tourette syndrome revealed by 7T MRI and PET. Brain Commun. 2025, 7, fcaf104. [Google Scholar] [CrossRef]
- Möller, H.E.; Bossoni, L.; Connor, J.R.; Crichton, R.R.; Does, M.D.; Ward, R.J.; Zecca, L.; Zucca, F.A.; Ronen, I. Iron, myelin, and the brain: Neuroimaging meets neurobiology. Trends Neurosci. 2019, 42, 384–401. [Google Scholar] [CrossRef]
- Obeagu, E.I. Iron homeostasis and health: Understanding its role beyond blood health—A narrative review. Ann. Med. Surg. 2025, 87, 3362–3371. [Google Scholar] [CrossRef]
- Hare, D.; Ayton, S.; Bush, A.; Lei, P. A delicate balance: Iron metabolism and diseases of the brain. Front. Aging Neurosci. 2013, 5, 34. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, K.; Jiao, F.; Xu, C. Serum iron status and risk of Tourette’s syndrome: A Mendelian randomization study. Eur. J. Pediatr. 2025, 184, 454. [Google Scholar] [CrossRef] [PubMed]
- Bianco, L.; Unger, E.; Beard, J. Iron deficiency and neuropharmacology. In Iron Deficiency and Overload; Yehuda, S., Mostofsky, D., Eds.; Humana Press: Totowa, NJ, USA, 2010; pp. 141–158. [Google Scholar] [CrossRef]
- Klein, M.O.; Battagello, D.S.; Cardoso, A.R.; Hauser, D.N.; Bittencourt, J.C.; Correa, R.G. Dopamine: Functions, signaling, and association with neurological diseases. Cell Mol. Neurobiol. 2019, 39, 31–59. [Google Scholar] [CrossRef]
- Felt, B.T.; Beard, J.L.; Schallert, T.; Shao, J.; Aldridge, J.W.; Connor, J.R.; Georgieff, M.K.; Lozoff, B. Persistent neurochemical and behavioral abnormalities in adulthood despite early iron supplementation for perinatal iron deficiency anemia in rats. Behav. Brain Res. 2006, 171, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Burhans, M.S.; Dailey, C.; Beard, Z.; Wiesinger, J.; Murray-Kolb, L.; Jones, B.C.; Beard, J.L. Iron deficiency: Differential effects on monoamine transporters. Nutr. Neurosci. 2005, 8, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Kim, J.; Buckett, P.D.; Bohlke, M.; Maher, T.J.; Wessling-Resnick, M. Severe postnatal iron deficiency alters emotional behavior and dopamine levels in the prefrontal cortex of young male rats. J. Nutr. 2011, 141, 2133–2138. [Google Scholar] [CrossRef]
- Ward, K.L.; Tkac, I.; Jing, Y.; Felt, B.; Beard, J.; Connor, J.; Schallert, T.; Georgieff, M.K.; Rao, R. Gestational and lactational iron deficiency alters the developing striatal metabolome and associated behaviors in young rats. J. Nutr. 2007, 137, 1043–1049. [Google Scholar] [CrossRef]
- Batra, J.; Seth, P.K. Effect of iron deficiency on developing rat brain. Indian J. Clin. Biochem. 2002, 17, 108–114. [Google Scholar] [CrossRef]
- Rao, R.; Tkac, I.; Townsend, E.L.; Gruetter, R.; Georgieff, M.K. Perinatal iron deficiency alters the neurochemical profile of the developing rat hippocampus. J. Nutr. 2003, 133, 3215–3221. [Google Scholar] [CrossRef]
- Agarwal, K.N. Iron and the brain: Neurotransmitter receptors and magnetic resonance spectroscopy. Br. J. Nutr. 2001, 85, S147–S150. [Google Scholar] [CrossRef]
- Erikson, K.M.; Shihabi, Z.K.; Aschner, J.L.; Aschner, M. Manganese accumulates in iron-deficient rat brain regions in a heterogeneous fashion and is associated with neurochemical alterations. Biol. Trace Elem. Res. 2002, 87, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Ruan, Z.; Li, Y.; Qiu, H.; Deng, C.; Qian, L.; Cui, W.; Tang, W.; Yang, Z.; Cheng, Y.; et al. Brain Iron Alteration in Pediatric Tourette Syndrome: A Quantitative Susceptibility Mapping Study. Eur. J. Neurol. 2025, 32, e70054. [Google Scholar] [CrossRef]
- Cusick, S.E.; Georgieff, M.K. The Role of Nutrition in Brain Development: The Golden Opportunity of the ‘First 1000 Days’. J. Pediatr. 2016, 175, 16–21. [Google Scholar] [CrossRef]
- Levi, S.; Ripamonti, M.; Moro, A.S.; Cozzi, A. Iron imbalance in neurodegeneration. Mol. Psychiatry 2024, 29, 1139–1152. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.Y.; Wen, F. Serum ferritin levels in children with attention deficit hyperactivity disorder and tic disorder. World J. Clin. Cases 2022, 10, 7749–7759. [Google Scholar] [CrossRef] [PubMed]
- Berglund, S.; Domellöf, M. Meeting iron needs for infants and children. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 267–272. [Google Scholar] [CrossRef]
- Beard, J. Iron Deficiency Alters Brain Development and Functioning. J. Nutr. 2003, 133, 1468S–1472S. [Google Scholar] [CrossRef]
- Kim, J.; Wessling-Resnick, M. Iron and mechanisms of emotional behavior. J. Nutr. Biochem. 2014, 25, 1101–1107. [Google Scholar] [CrossRef]
- Aggett, P.J. Iron. In Present Knowledge in Nutrition, 11th ed.; Marriott, B.P., Birt, D.F., Stallings, V.A., Yates, A.A., Eds.; Elsevier: Cambridge, MA, USA, 2020; Volume 1, pp. 375–392. [Google Scholar]
- National Institutes of Health (NIH) Office of Dietary Supplements (ODS). Iron. Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/iron-HealthProfessional/ (accessed on 9 December 2025).
- Baddam, S.; Chen, R.J. Iron Overload and Toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Rasel, M.; Hashmi, M.F.; Mahboobi, S.K. Transfusion Iron Overload. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Reddy, S.T.; Soman, S.S.; Yee, J. Magnesium Balance and Measurement. Adv. Chronic Kidney Dis. 2018, 25, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Cepeda, V.; Ródenas-Munar, M.; García, S.; Bouzas, C.; Tur, J.A. Unlocking the Power of Magnesium: A Systematic Review and Meta-Analysis Regarding Its Role in Oxidative Stress and Inflammation. Antioxidants 2025, 14, 740. [Google Scholar] [CrossRef]
- Fiorentini, D.; Cappadone, C.; Farruggia, G.; Prata, C. Magnesium: Biochemistry, Nutrition, Detection, and Social Impact of Diseases Linked to Its Deficiency. Nutrients 2021, 13, 1136. [Google Scholar] [CrossRef]
- Konrad, M.; Schlingmann, K.P.; Gudermann, T. Insights into the molecular nature of magnesium homeostasis. Am. J. Physiol. Renal. Physiol. 2004, 286, F599–F605. [Google Scholar] [CrossRef]
- Pouteau, E.; Kabir-Ahmadi, M.; Noah, L.; Mazur, A.; Dye, L.; Hellhammer, J.; Pickering, G.; Dubray, C. Superiority of magnesium and vitamin B6 over magnesium alone on severe stress in healthy adults with low magnesemia: A randomized, single-blind clinical trial. PLoS ONE 2018, 13, e0208454. [Google Scholar] [CrossRef]
- de Baaij, J.H.; Hoenderop, J.G.; Bindels, R.J. Magnesium in man: Implications for health and disease. Physiol. Rev. 2015, 95, 1–46. [Google Scholar] [CrossRef]
- Laires, M.J.; Monteiro, C.P.; Bicho, M. Role of cellular magnesium in health and human disease. Front. Biosci. 2004, 9, 76. [Google Scholar] [CrossRef]
- Murck, H. Magnesium and affective disorders. Nutr. Neurosci. 2002, 5, 375–389. [Google Scholar] [CrossRef] [PubMed]
- Held, K.; Antonijevic, I.A.; Künzel, H.; Uhr, M.; Wetter, T.C.; Golly, I.C.; Steiger, A.; Murck, H. Oral Mg2+ supplementation reverses age-related neuroendocrine and sleep EEG changes in humans. Pharmacopsychiatry 2002, 35, 135–143. [Google Scholar] [CrossRef]
- Dmitrašinović, G.; Pešić, V.; Stanić, D.; Plećaš-Solarović, B.; Dajak, M.; Ignjatović, S. ACTH, Cortisol and IL-6 Levels in Athletes following Magnesium Supplementation. J. Med. Biochem. 2016, 35, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Papadopol, V.; Nechifor, M. Magnesium in neuroses and neuroticism. In Magnesium in the Central Nervous System; Vink, R., Nechifor, M., Eds.; University of Adelaide Press: Adelaide, Australia, 2011. [Google Scholar]
- Boyle, N.B.; Lawton, C.; Dye, L. The Effects of Magnesium Supplementation on Subjective Anxiety and Stress—A Systematic Review. Nutrients 2017, 9, 429. [Google Scholar] [CrossRef] [PubMed]
- Clerc, P.; Young, C.A.; Bordt, E.A.; Grigore, A.M.; Fiskum, G.; Polster, B.M. Magnesium sulfate protects against the bioenergetic consequences of chronic glutamate receptor stimulation. PLoS ONE 2013, 8, e79982. [Google Scholar] [CrossRef]
- Smith, B.L.; Ludlow, A.K. Patterns of Nutritional Supplement Use in Children with Tourette Syndrome. J. Diet. Suppl. 2021, 20, 28–43. [Google Scholar] [CrossRef]
- Kirkland, A.E.; Sarlo, G.L.; Holton, K.F. The Role of Magnesium in Neurological Disorders. Nutrients 2018, 10, 730. [Google Scholar] [CrossRef] [PubMed]
- Fatima, G.; Dzupina, A.; Alhmadi, H.B.; Magomedova, A.; Siddiqui, Z.; Mehdi, A.; Hadi, N. Magnesium Matters: A Comprehensive Review of Its Vital Role in Health and Diseases. Cureus 2024, 16, e71392. [Google Scholar] [CrossRef]
- Pickering, G.; Mazur, A.; Trousselard, M.; Bienkowski, P.; Yaltsewa, N.; Amessou, M.; Noah, L.; Pouteau, E. Magnesium Status and Stress: The Vicious Circle Concept Revisited. Nutrients 2020, 12, 3672. [Google Scholar] [CrossRef]
- Grimaldi, B.L. The central role of magnesium deficiency in Tourette’s syndrome: Causal relationships between magnesium deficiency, altered biochemical pathways and symptoms relating to Tourette’s syndrome and several reported comorbid conditions. Med. Hypotheses 2002, 58, 47–60. [Google Scholar] [CrossRef]
- Azmeen, A.; Condit, D.; Rosenberg, E.; Mcpeck, S.; Deengar, A.; Sathyan, S.; Grover, P.; Nadler, E. A Case of Catastrophic Magnesium Overdose. CHEST 2021, 160, A954–A955. [Google Scholar] [CrossRef]
- Musso, C.G. Magnesium metabolism in health and disease. Int. Urol. Nephrol. 2009, 41, 357–362. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health (NIH), Office of Dietary Supplements (ODS). Magnesium. Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/Magnesium-HealthProfessional/ (accessed on 9 December 2025).
- McCall, K.A.; Huang, C.; Fierke, C.A. Function and mechanism of zinc metalloenzymes. J. Nutr. 2000, 130, 1437S–1446S. [Google Scholar] [CrossRef]
- Reed, S.; Qin, X.; Ran-Ressler, R.; Brenna, J.T.; Glahn, R.P.; Tako, E. Dietary zinc deficiency affects blood linoleic acid: Dihomo-γ-linolenic acid (LA:DGLA) ratio; a sensitive physiological marker of zinc status in vivo (Gallus gallus). Nutrients 2014, 6, 1164–1180. [Google Scholar] [CrossRef]
- Zastrow, M.L.; Pecoraro, V.L. Designing hydrolytic zinc metalloenzymes. Biochemistry 2014, 53, 957–978. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Chen, H. Aberrance of Zinc Metalloenzymes-Induced Human Diseases and Its Potential Mechanisms. Nutrients 2021, 13, 4456. [Google Scholar] [CrossRef]
- Floriańczyk, B. Role of Zinc in nervous system cells. J. Pre-Clin. Clin. Res. 2011, 5, 12–15. [Google Scholar]
- Choi, S.; Hong, D.K.; Choi, B.Y.; Suh, S.W. Zinc in the Brain: Friend or Foe? Int. J. Mol. Sci. 2020, 21, 8941. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, Y.; Wei, R.; Yong, V.W.; Xue, M. The Important Role of Zinc in Neurological Diseases. Biomolecules 2022, 13, 28. [Google Scholar] [CrossRef]
- Madireddy, S.; Madireddy, S. Therapeutic Strategies to Ameliorate Neuronal Damage in Epilepsy by Regulating Oxidative Stress, Mitochondrial Dysfunction, and Neuroinflammation. Brain Sci. 2023, 13, 784. [Google Scholar] [CrossRef] [PubMed]
- Wessells, K.R.; Brown, K.H. Estimating the global prevalence of zinc deficiency: Results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 2012, 7, e50568. [Google Scholar] [CrossRef]
- Hettiarachchi, P.; Shigemoto, A.; Hickey, E.E.; Burdette, S.C.; Johnson, M.A. Zinc-Dependent Modulation of Dopamine Release and Uptake Is Altered in Parkinson’s Disease Model Zebrafish. ACS Chem. Neurosci. 2025, 16, 1872–1882. [Google Scholar] [CrossRef]
- Takeda, A.; Hirate, M.; Oku, N. Release of glutamate and GABA in the hippocampus under zinc deficiency. J. Neurosci. Res. 2003, 72, 537–542. [Google Scholar] [CrossRef]
- Landau, Y.; Steinberg, T.; Richmand, B.; Leckman, J.F.; Apter, A. Involvement of immunologic and biochemical mechanisms in the pathogenesis of Tourette’s syndrome. J. Neural. Transm. 2012, 119, 621–626. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schoofs, H.; Schmit, J.; Rink, L. Zinc Toxicity: Understanding the Limits. Molecules 2024, 29, 3130. [Google Scholar] [CrossRef]
- Duncan, A.; Yacoubian, C.; Watson, N.; Morrison, I. The risk of copper deficiency in patients prescribed zinc supplements. J. Clin. Pathol. 2015, 68, 723–725. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health (NIH), Office of Dietary Supplements. Zinc, Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/zinc-HealthProfessional/ (accessed on 9 December 2025).
- Yu, W.R.; Jiang, H.; Wang, J.; Xie, J.X. Copper (Cu2+) induces degeneration of dopaminergic neurons in the nigrostriatal system of rats. Neurosci. Bull. 2008, 24, 73–78. [Google Scholar] [CrossRef]
- Kondo, M.; Hara, H.; Kamijo, F.; Kamiya, T.; Adachi, T. 6-Hydroxydopamine disrupts cellular copper homeostasis in human neuroblastoma SH-SY5Y cells. Metallomics 2021, 13, mfab041. [Google Scholar] [CrossRef]
- García, C.R.; Angelé-Martínez, C.; Wilkes, J.A.; Wang, H.C.; Battin, E.E.; Brumaghim, J.L. Prevention of iron- and copper-mediated DNA damage by catecholamine and amino acid neurotransmitters, L-DOPA, and curcumin: Metal binding as a general antioxidant mechanism. Dalton Trans. 2012, 41, 6458–6467. [Google Scholar] [CrossRef]
- Kaler, S.; Holmes, C. Catecholamine metabolites affected by the copper-dependent enzyme dopamine-beta-hydroxylase provide sensitive biomarkers for early diagnosis of Menkes Disease and Viral-mediated ATP7A gene therapy. Adv. Pharmacol. 2013, 68, 223–233. [Google Scholar] [CrossRef]
- Goez, H.R.; Jacob, F.D.; Yager, J.Y. Lingual Dyskinesia and Tics: A Novel Presentation of Copper-Metabolism Disorder. Pediatrics 2011, 127, e505–e508. [Google Scholar] [CrossRef]
- Kodama, H.; Fujisawa, C.; Bhadhprasit, W. Inherited copper transport disorders: Biochemical mechanisms, diagnosis and treatment. Curr. Drug Metab. 2012, 13, 237–250. [Google Scholar] [CrossRef] [PubMed]
- Llanos, R.M.; Mercer, J.F. The molecular basis of copper homeostasis copper-related disorders. DNA Cell Biol. 2002, 21, 259–270. [Google Scholar] [CrossRef]
- Viktorinova, A.; Ursinyova, M.; Trebaticka, J.; Uhnakova, I.; Durackova, Z.; Masanova, V. Changed plasma levels of zinc and copper to zinc ratio and their possible associations with parent- and teacher-rated symptoms in children with attention-deficit hyperactivity disorder. Biol. Trace. Elem. Res. 2016, 169, 1–7. [Google Scholar] [CrossRef]
- Royer, A.; Sharman, T. Copper Toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Chen, H.; Li, D.; Zhang, H.; Zhang, M.; Lin, Y.; He, H.; Liu, A.; Shen, S.; Wang, Y.; Han, Z. Mechanisms of copper metabolism and cuproptosis: Implications for liver diseases. Front. Immunol. 2025, 16, 1633711. [Google Scholar] [CrossRef] [PubMed]
- Eom, S.Y.; Yim, D.H.; Huang, M.; Park, C.H.; Kim, G.B.; Yu, S.D.; Choi, B.S.; Park, J.D.; Kim, Y.D.; Kim, H. Copper-zinc imbalance induces kidney tubule damage and oxidative stress in a population exposed to chronic environmental cadmium. Int. Arch. Occup. Environ. Health 2020, 93, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.S.; Hess, S.Y.; Hotz, C.; Brown, K.H. Indicators of zinc status at the population level: A review of the evidence. Br. J. Nutr. 2008, 99, S14–S23. [Google Scholar] [CrossRef]
- National Institutes of Health (NIH), Office of Dietary Supplements (ODS). Copper. Available online: https://ods.od.nih.gov/factsheets/Copper-HealthProfessional/ (accessed on 9 December 2025).
- Billnitzer, A.; Jankovic, J. Current Management of Tics and Tourette Syndrome: Behavioral, Pharmacologic, and Surgical Treatments. Neurotherapeutics 2020, 17, 1681–1693. [Google Scholar] [CrossRef] [PubMed]
- Baizabal-Carvallo, J.F.; Joseph Jankovic, J. Chapter Six—The pharmacological treatment of Tourette syndrome. Int. Rev. Mov. Disord. 2022, 4, 163–201. [Google Scholar] [CrossRef]
- Deekae, A. Pharmacological management of Tourette syndrome. Pharm. J. 2025, 315, 7999. [Google Scholar] [CrossRef]
- Seideman, M.F.; Seideman, T.A. A Review of the Current Treatment of Tourette Syndrome. J. Pediatr. Pharmacol. Ther. 2020, 25, 401–412. [Google Scholar] [CrossRef]
- Eddy, C.M.; Rickards, H.E.; Cavanna, A.E. Treatment strategies for tics in Tourette syndrome. Ther. Adv. Neurol. Disord. 2011, 4, 25–45. [Google Scholar] [CrossRef]
- Pringsheim, T.; Okun, M.S.; Müller-Vahl, K.; Martino, D.; Jankovic, J.; Cavanna, A.E.; Woods, D.W.; Robinson, M.; Jarvie, E.; Roessner, V.; et al. Practice guideline recommendations summary: Treatment of tics in people with Tourette syndrome and chronic tic disorders. Neurology 2019, 92, 896–906. [Google Scholar] [CrossRef]
- Barzyk, K.; Buczek, W.; Kurek, M.; Chrościńska-Krawczyk, M. Different treatment methods for tics and Tourette Syndrome. J. Pre-Clin. Clin. Res. 2025, 19, 29–35. [Google Scholar] [CrossRef]
- Berzosa-Gonzalez, I.; Martinez-Horta, S.; Pérez-Pérez, J.; Kulisevsky, J.; Pagonabarraga, J. Therapeutic Approach to Primary Tic Disorders and Associated Psychiatric Comorbidities. Brain Sci. 2024, 14, 1231. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.