Abstract
Forest soil constitutes a critical reservoir within terrestrial carbon pools. Understanding the dynamics of soil organic carbon (SOC) in coniferous forests is crucial for enhancing ecosystem carbon sequestration capacity, yet systematic quantification of SOC characteristics and their driving factors remains limited across critical bioclimatic zones. This study examined SOC features in topsoil and driving factors across eight representative coniferous forest types in Longnan—an ecologically significant transition region of northwestern China. SOC concentrations ranged from 31.76 to 80.86 g·kg−1, where Abies fargesii var. faxoniana exhibited significantly higher concentrations than other conifers. Fungal necromass dominated SOC formation (29%–45% contribution) versus minimal bacterial necromass inputs (3%–5%). Redundancy analysis identified that soil total nitrogen, C/N ratio, and tree evenness showed significant correlations with SOC concentrations and their fractions. Partial least squares path modeling revealed that tree species exerted a direct positive impact on soil total nitrogen, while having an adverse effect on soil pH. Lower soil pH and higher total nitrogen were associated with higher microbial-derived carbon and SOC concentrations. In contrast, plant-derived carbon exerted no direct influence on SOC concentrations, operating exclusively through microbial-derived carbon pathways. These results indicated that coniferous tree species-induced shifts in soil total nitrogen and pH facilitate the accumulation of microbial necromass carbon, rather than plant residues, and thus promote SOC sequestration. A. fargesii var. faxoniana can be regarded as a key strategic tree species for SOC sequestration and sustainable forest management, and its cultivation should be prioritized due to improvements in total nitrogen and microbial-derived carbon.
1. Introduction
Soil organic carbon (SOC), a pivotal index of soil fertility, exerts a crucial function in mediating nutrient supply for plants, promoting soil structure, and mitigating soil and water erosion [1]. Serving as the most substantial pool of organic carbon within terrestrial ecosystems [2,3,4], SOC also performs a vital role in modulating global carbon cycling and climate change [5]. The formation and stability of SOC rely heavily on the transformation, preservation, and relative proportions of carbon fractions originating from plants and microbes [6,7]. Lignin phenols and amino sugars are well-recognized biomarkers corresponding to plant- and microbial-derived carbon, respectively [8,9]. Plant residues containing compounds like lignin phenols are less protected by minerals or aggregates and more accessible to microorganisms, facilitating their synthesis and transformation into microbial necromass carbon (MNC) [10]. MNC is widely recognized as contributing more significantly than plant residues to the long-term sequestration and stabilization of SOC, attributable to its persistent production and comparatively slow decomposition [11]. Globally, MNC contributes an average of 50% to the direct sources of SOC, ranging from 30% to 80% across terrestrial ecosystems [12,13]. This variation arises from differences in precipitation, vegetation type and productivity, and soil microbial community characteristics [14,15]. However, a study across different ecosystems indicates that plant residues contribute more to SOC accumulation than microbial-derived carbon in forests, while the opposite pattern occurs in grasslands [6]. Consequently, there is no consensus on whether plant- or microbial-derived carbon dominates SOC persistence across terrestrial ecosystems [12,14]. Further research is needed to quantify the relative contributions of plant residues and MNC to SOC concentrations, which is vital for comprehending SOC sequestration and responses to global change.
Forests cover roughly 30% of the global terrestrial region and represent a promising climate change mitigation strategy due to their capacity to sequester atmospheric CO2 and store organic carbon in both tree biomass and soils [16,17]. On average, trees and soils hold similar quantities of carbon within forest ecosystems, even more in SOC stocks than those in plant biomass [16,18], and consequently exert a pivotal function in the global carbon cycle [19]. Quantifying the contributions of plant- and microbial-derived carbon to SOC sequestration and identifying their key drivers are essential for assessing forest carbon stocks and dynamics [20]. Extensive research investigate SOC distribution and its controlling factors across terrestrial ecosystems, encompassing grasslands, croplands, wetlands, and forests [21,22]. For example, tree functional traits and forest biomass account for >50% of local SOC variation; climate and soil conditions mediate the impacts of tree functional traits on SOC storage, which are strongest in boreal climates and acidic, nutrient-poor, coarse-textured soils [23]. However, the key drivers of forest SOC sequestration, particularly tree species composition and soil conditions, remain poorly understood. This knowledge gap hinders the implementation of targeted measures to protect and restore forest ecosystems and maintain or enhance their ecological functions, including soil carbon storage.
Longnan lies at the junction of the Qinba Mountains, Qinghai–Tibet Plateau, and Loess Plateau, representing the transition zone between the second and third tiers of China’s topographic ladder. This region is a biodiversity hotspot, with forest coverage averaging >45% and supporting numerous nationally and provincially protected rare plant and animal species [24]. Consequently, Longnan provides an ideal study system for investigating forest SOC sequestration dynamics and identifying key drivers of SOC characteristics. This study quantified the components of plant- and microbial-derived carbon and assessed their relative contributions to SOC sequestration across major coniferous tree species. Among the dominant coniferous species in this region, Abies fargesii var. faxoniana is particularly prominent, typically occurring at higher elevations with cooler and more moist conditions that may favor organic matter accumulation [25,26]. The primary objectives were to: (1) characterize SOC concentrations in coniferous forests, and (2) explore their influencing factors, (3) identify which tree species were superior in SOC sequestration. Based on the existing literature, we tested the following hypotheses: (1) microbial-derived carbon, particularly fungal necromass, contributes more to SOC accumulation than plant-derived carbon in coniferous forest soils [10,12]; (2) tree species influence SOC accumulation indirectly by modulating soil properties (total nitrogen and pH), which in turn affect microbial-derived carbon formation [23]; and (3) Abies fargesii var. faxoniana exhibits the highest SOC sequestration capacity [25,26]. This research establishes baseline data on coniferous forests in Longnan and informs management strategies for forest conservation and restoration. Furthermore, it enhances understanding of the regional forest carbon storage capacity under global climate change.
2. Materials and Methods
2.1. Study Site and Experimental Design
The study area is located in the Longnan area (32°43′–34°10′ N, 104°09′–104°35′ E; elevation 1743–2833 m above sea level). This area has a subtropical monsoon climate, with a mean annual temperature of 2–14°C and mean annual precipitation of 600 mm. The topography is predominantly mountainous, and the soils are classified as Haplic Luvisols, developed from metamorphic rocks [27]. The area harbors numerous species protected at national and provincial levels, including the rare plants Metasequoia glyptostroboides and Taxus wallichiana, as well as animals like Ailuropoda melanoleuca and Rhinopithecus roxellanae.
2.2. Vegetation Surveys and Soil Sample Collection
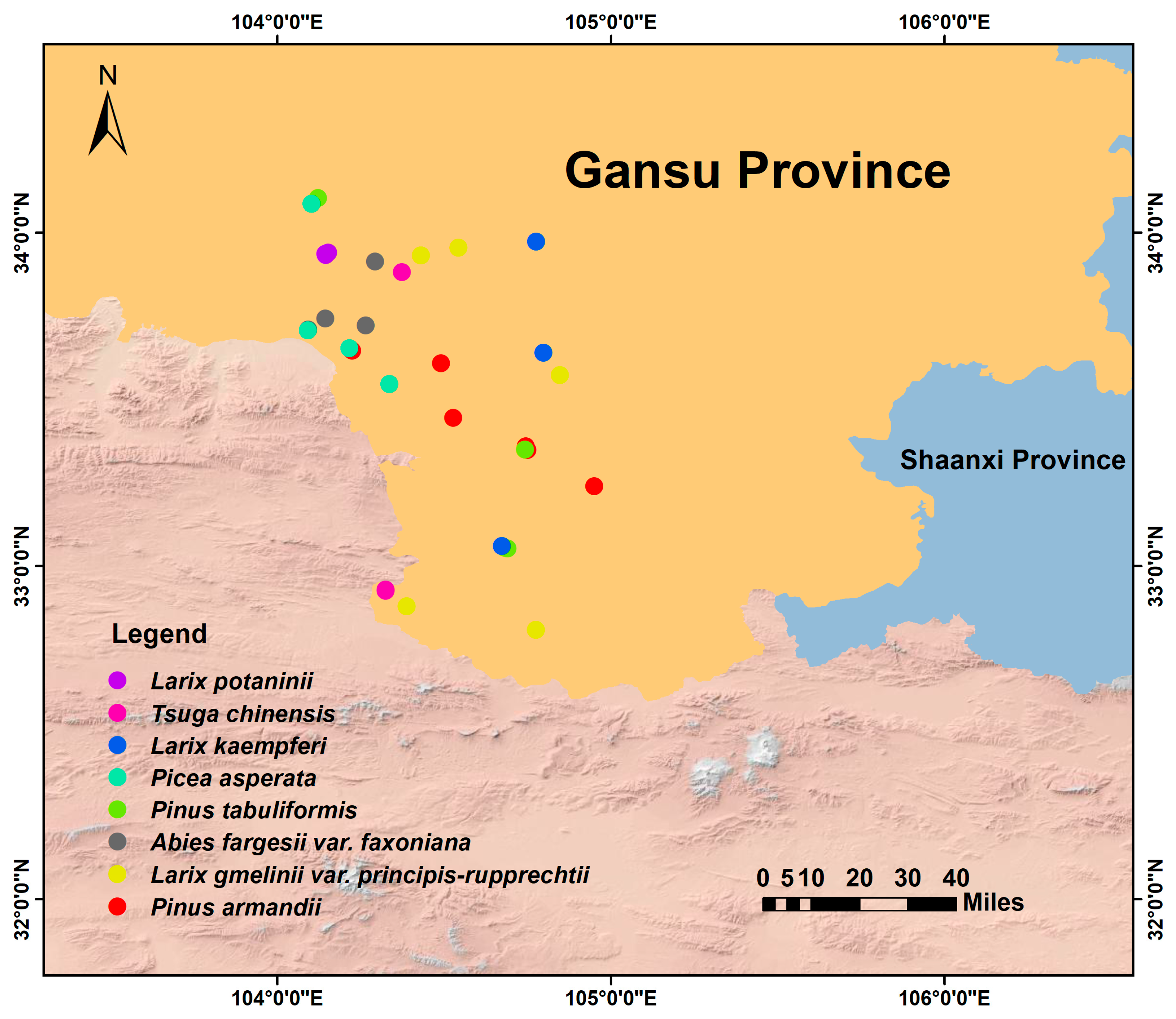
Eight coniferous forest types, each dominated by a single tree species, were selected. The numbers of 20 × 20 m plots established in each forest type (proportional to stand area) were as follows: Pinus armandii (6 plots), Larix gmelinii var. principis-rupprechtii (5 plots), Abies fargesii var. faxoniana (4 plots), P. tabuliformis (4 plots), Picea asperata (4 plots), L. kaempferi (3 plots), Tsuga chinensis (3 plots), and L. potaninii (3 plots). In total, 32 plots were established across the eight forest types. Plots were established following the random systematic sampling method to avoid spatial autocorrelation, with a minimum distance of >100 m between adjacent plots (Figure 1). Each 20 × 20 m plot was divided into four 10 × 10 m quadrats for tree investigation. Within the southwest corner of each quadrat, a 5 × 5 m plot was established for shrub investigation. Similarly, in the southwest corner of each quadrat, a 1 × 1 m plot was used for herb sampling (see Figure S1). For tree species in each plot, all individuals with diameter at breast height (DBH) ≥ 3 cm were measured. Individuals were categorized into two size classes: saplings (3 cm ≤ DBH ≤ 5 cm) and mature trees (DBH > 5 cm). For shrubs, variables of height, basal diameter, clump number, and per cent cover were measured. For herbs, height, clump number, and percent cover were documented.
Figure 1.
Distribution map of forest plots.
Soil sampling was carried out in September 2022. A total of 32 composite soil samples were collected from the eight coniferous forest types. Within each 20 × 20 m plot, five random locations were selected after removing surface litter and crusts. Topsoil samples (0–20 cm depth) were collected from each location using a soil auger and were thoroughly mixed to form one composite sample per plot. The composite samples were placed in sealed plastic bags, transported to the laboratory on ice, and then air-dried at room temperature. The air-dried samples were subsequently sieved through a 2 mm mesh to eliminate roots, stones, and organic debris before analyzing soil physicochemical properties, amino sugar concentrations, and lignin phenols.
Due to the complex mountainous terrain of the study area, the selected plots inherently vary in topographic conditions (e.g., elevation, slope, and aspect). Similarly, soil parent materials and texture may also vary across plots, reflecting the heterogeneous landscape characteristic of the Longnan region. This variation reflects the typical habitat distribution of the different coniferous species in their natural environment.
2.3. Soil Physicochemical Properties Analysis
Soil pH was measured on a pH meter in 1:2.5 (w/v) soil–water suspensions after 30 min shaking [28]. SOC was determined by the external heating potassium dichromate oxidation method [29]. Total nitrogen (TN) and total phosphorus (TP) were determined via the Kjeldahl method and acid-soluble method, respectively [30,31]. Nitrate nitrogen (NO3−-N) and ammonium nitrogen (NH4+-N) were leached from soil using a 2 M potassium chloride solution and subsequently measured via a specific photometric approach with a continuous flow analyzer (SmartChem 160, AMS Alliance, Italy) [32]. Available phosphorus (AP) was leached utilizing HCl-H2SO4 and assessed colorimetrically via the molybdenum blue technique [33].
2.4. Amino Sugar Determination
Soil amino sugars were extracted following the method [34]. Briefly, 0.5 g of air-dried soil was hydrolyzed with 10 mL of 6 M HCl at 105 °C for 6 h. The cooled hydrolysate was filtered, concentrated via rotary evaporation, and a 0.5 mL aliquot was dried under N2 at 45 °C. The residue was reconstituted in 0.5 mL H2O, re-dried under N2, and finally dissolved in 2 mL H2O for analysis. Muramic acid (MurN), galactosamine (GalN), and glucosamine (GluN) were quantified by high-performance liquid chromatography. The concentrations of total amino sugars (TASs), bacterial necromass carbon (BNC), fungal necromass carbon (FNC), and microbial necromass carbon (MNC) were computed following the method of Chen et al. (2020) [35]:
where 31.3 and 10.8 are the bacterial and fungal conversion factors, respectively; 1.16 is the molar ratio of GluN to MurN.
2.5. Lignin Phenol Determination
Lignin phenols were quantified by alkaline cupric oxide oxidation and gas chromatography. Air-dried soil samples, along with CuO, Fe (NH4)2 (SO4)2·6H2O, NaOH, and the internal standard ethylvanillin, were heated in Teflon-lined vessels at 170 °C for 2 h. The resulting solutions were centrifuged, and the supernatant was acidified to pH 1.0 with 6 M HCl, kept in the dark for one hour, and then processed by solid-phase extraction. The final extracts, after drying under N2, were analyzed for lignin phenol concentrations via gas chromatography. Lignin phenol concentrations were regarded as a recalcitrant fraction of plant-originated carbon in SOC. They were computed as the total of vanillyl (V), syringyl (S), and cinnamyl (C) phenols. The S/V and C/V ratios act as indicators of lignin degradation degree and vegetation resources, where lower ratios signify more extensive degradation [36]. The vanillyl and syringyl acid-to-aldehyde ratios, (Ad/Al)v and (Ad/Al)s, are widely used to assess the extent of lignin degradation caused by microbial activity or oxidation; higher ratios indicate more aldehydes being oxidized to acid or more significant lignin degradation by microorganisms [37].
The percentage of plant-derived carbon in SOC concentrations was calculated with the following equation [38]:
where V, S, C represent the carbon concentration of vanillyl, syringyl, and cinnamyl phenols (g kg−1); 33.3% and 90% are release efficiency of vanillyl and syringyl phenols, respectively; 8% is the minimum concentration of lignin in plant residues of most species [39].
2.6. Plant Diversity Assessment
Diversity matrix including species richness (S), Shannon–Wiener diversity index (H), Simpson diversity index (D) and Pielou evenness index (J) for trees, shrubs and herbs, respectively, was calculated according to standard equations:
where Pi2 is the proportion of the i-th species among all species; S is the number of species in the study area.
2.7. Statistical Analysis
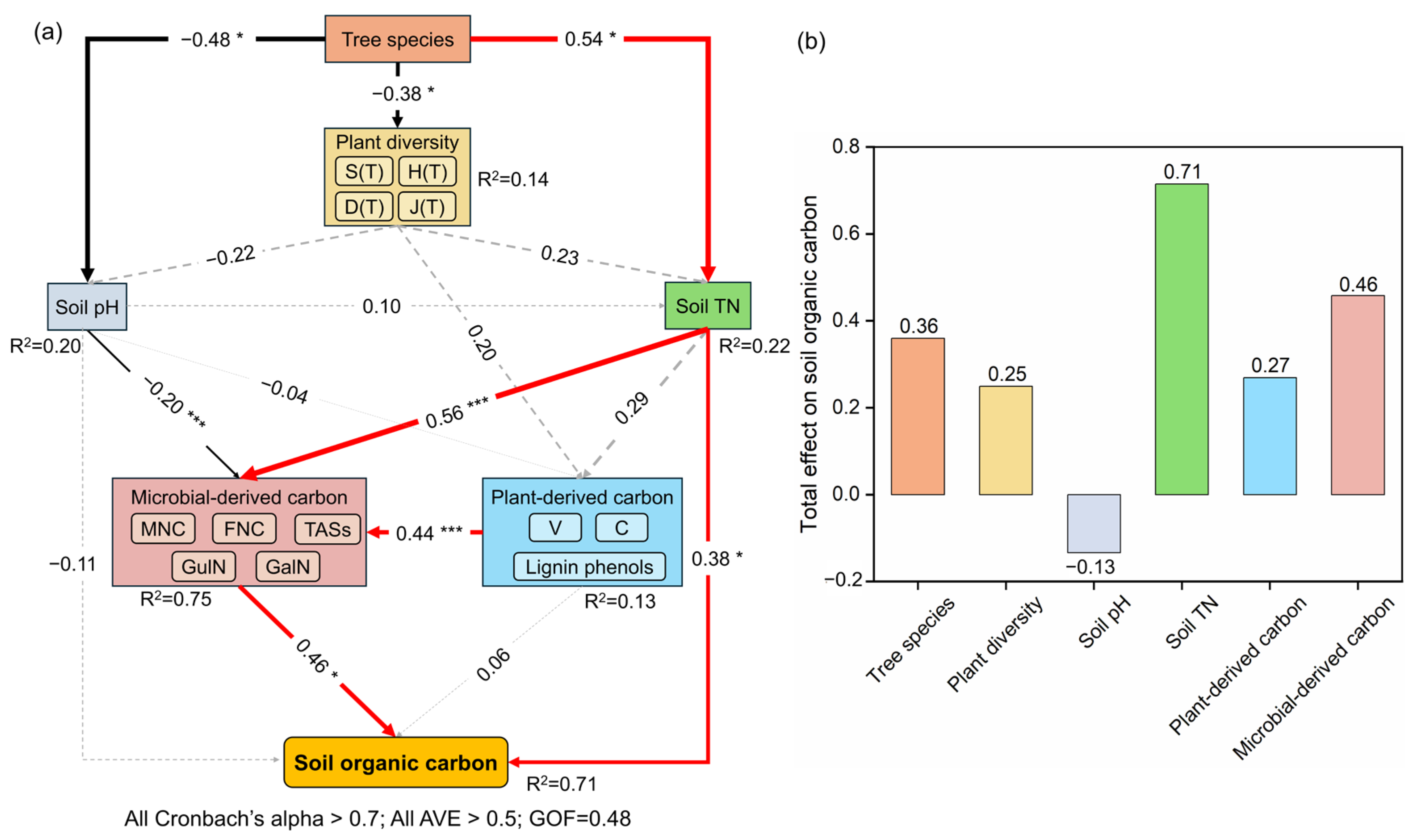
One-way ANOVA was carried out to assess differences in soil properties, amino sugars, lignin phenols, and plant diversity indices across coniferous species using SPSS 27.0 (SPSS Inc., Chicago, IL, USA) software. Duncan’s multiple comparison test was employed to detect significant differences across groups (p < 0.05). Data normality and homogeneity of variances were verified before analysis. Prior to redundancy analysis, multicollinearity diagnostics were performed. The variance inflation factor (VIF) for each predictor was computed, with all values falling within the acceptable threshold of 3 (Table S1) [40], thus validating the assumption of independence among explanatory variables. Redundancy analysis (RDA) with 999 Monte Carlo permutations (Canoco 5.0) was used to quantify the effects of soil properties and plant diversity on SOC concentration and its fractions. The partial least squares path model (PLS-PM) was constructed with the ‘plspm’ package in R 4.5.0 to explore internal links among underlying factors and determine their total effects on SOC sequestration. The model employed non-parametric bootstrapping to assess the statistical significance of the path coefficients, with the following settings: 5000 bootstrap resamples, bias-corrected and accelerated bootstrap confidence intervals at the 95% confidence level, and a two-tailed test with a significance level of α = 0.05. The criteria for determining the statistical significance of a path coefficient were that its 95% bootstrap confidence interval did not include zero, and/or the absolute value of its bootstrap t-statistic exceeded 1.96 (corresponding to p < 0.05). The standard errors and t-values for the path coefficients were derived from the bootstrap distribution. Tree species was specified as a formative construct, as the different coniferous species represent distinct categories that collectively define the species effect. The formative indicators were dummy-coded species variables. Plant diversity, plant-derived carbon, and microbial-derived carbon were specified as reflective constructs, as these measured variables are manifestations of the underlying latent constructs. Soil pH, TN, and SOC concentration was included as a single-item construct. The goodness-of-fit (GOF) index, average variance extracted (AVE), Cronbach’s alpha, and coefficients of determination (R2) of the underlying factors were examined as criteria for assessing model fit [41]. The diversity indices for trees were used as observed variables for plant diversity. V, C, and lignin phenols were used as observed variables for plant-derived carbon. TASs, GluN, GalN, MNC, and FNC were used as observed variables for microbial-derived carbon. Tree species was a categorical variable which was assigned numeric codes according to the order of the target trees. Detailed parameters for error term sources, sample sizes, and degree of freedom calculations are provided in Table 1.

Table 1.
Statistical parameters for error term and degree of freedom calculation.
3. Results
3.1. Soil Physicochemical Properties and Tree Evenness in Coniferous Forests
Basic physicochemical properties of the surface soil, such as pH and TN exhibited notable variations among distinct coniferous forests (p < 0.05) (Table 2). Soil TN concentrations were significantly higher (6.55 g·kg−1) in Abies fargesii var. faxoniana (species 3) than Pinus armandii (species 1) (3.62 g·kg−1), Larix gmelinii var. principis-rupprechtii (species 2) (4.07 g·kg−1), and Larix kaempferi (species 6) (2.30 g·kg−1). However, Abies fargesii var. faxoniana showed the lowest pH value (5.67) among all species (Table 2). For plant diversity, significant differences were observed only in the tree evenness (Pielou index) among different coniferous forests (p < 0.05) (Table S2).

Table 2.
Soil physicochemical properties in coniferous forests.
3.2. Soil Organic Carbon Concentration and Components of Microbial- and Plant-Derived Carbon
3.2.1. Soil Organic Carbon Concentration in Coniferous Forests
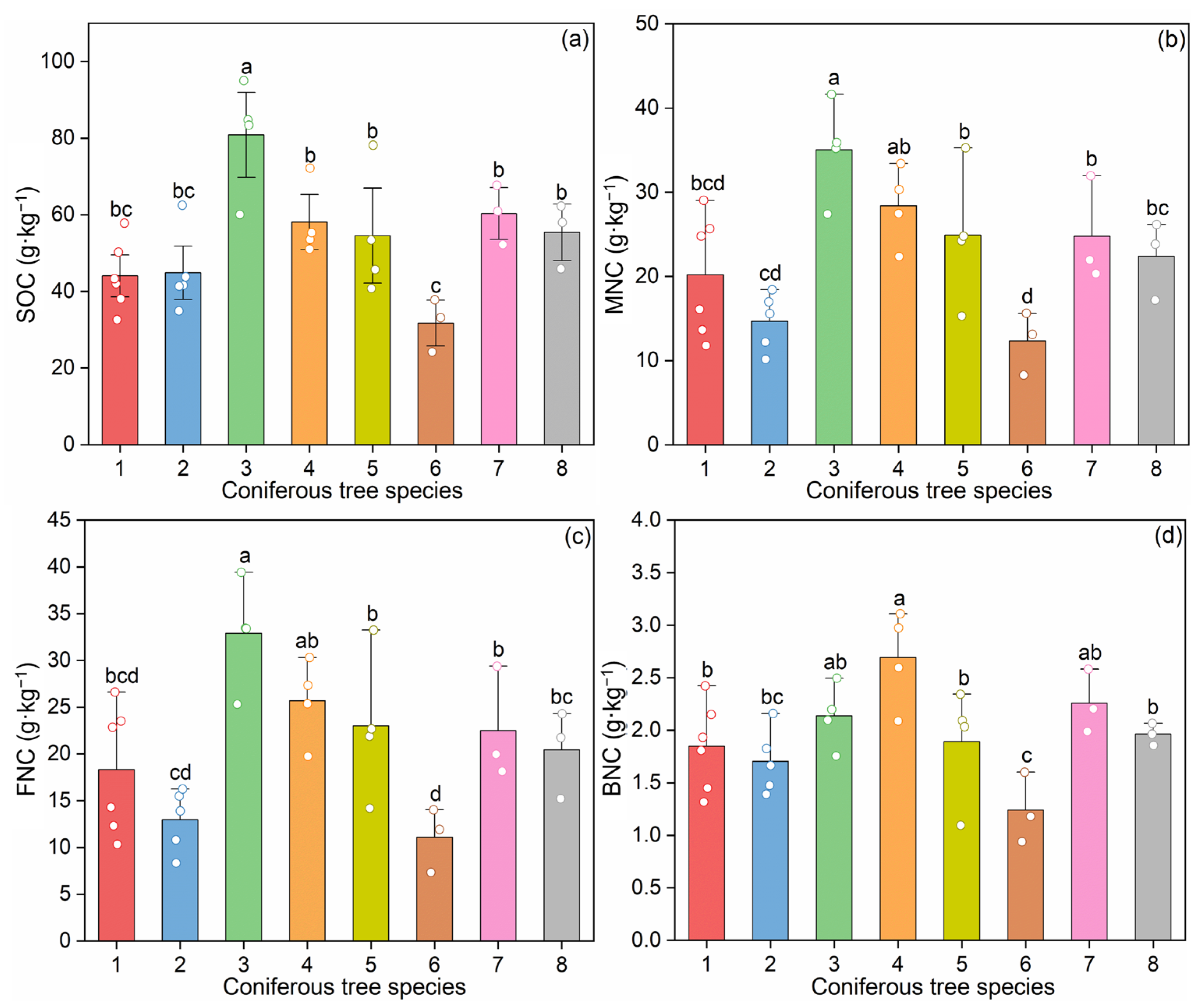
SOC concentrations varied significantly among the eight coniferous forest types, ranging from 31.76 to 80.86 g·kg−1 (Figure 2a). Notably, Abies fargesii var. faxoniana (species 3) exhibited the highest SOC concentration (80.86 g·kg−1), which was significantly higher than all other seven species (p < 0.05) (Figure 2a). In contrast, Larix kaempferi (species 6) showed the lowest SOC concentration (31.76 g·kg−1), significantly lower than species 3, 4 (Pinus tabuliformis), 5 (Picea asperata), 7 (Tsuga chinensis), and 8 (Larix potaninii) (p < 0.05) (Figure 2a). No notable differences in SOC contents were found among the remaining six species (p < 0.05) (Figure 2a).
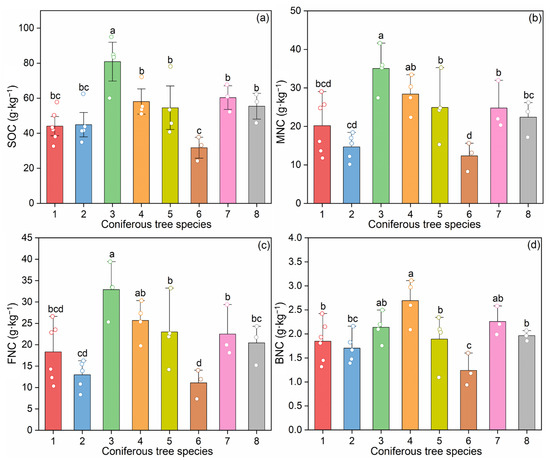
Figure 2.
Soil organic carbon and microbial necromass carbon in the coniferous forest soils: (a) soil organic carbon (SOC); (b) microbial necromass carbon (MNC); (c) fungal necromass carbon (FNC); (d) bacterial necromass carbon (BNC). Data are presented as mean ± standard error (SE). Significant differences among different coniferous tree species are indicated by different letters at the 0.05 level. The circles in different colors represent individual replicate measurements corresponding to each treatment group. Abbreviations: 1: Pinus armandii; 2: Larix gmelinii var. principis-rupprechtii; 3: Abies fargesii var. faxoniana; 4: Pinus tabuliformis; 5: Picea asperata; 6: Larix kaempferi; 7: Tsuga chinensis; 8: Larix potaninii.
3.2.2. Characteristics of Microbial-Derived Carbon in Coniferous Forest Soils
When using amino sugars as biomarkers, significant differences were observed in the contents of total amino sugars and individual amino sugars among the eight coniferous forest types (p < 0.05; Table S3). Total amino sugar concentrations ranged from 1.74 to 5.34 g·kg−1, with Abies fargesii var. faxoniana (species 3) exhibiting the highest concentration, which was significantly higher than all other seven species (p < 0.05; Figure S2). In contrast, Larix kaempferi (species 6) showed the lowest total amino sugar concentration, significantly lower than species 3 and 4 (Pinus tabuliformis) (p < 0.05).
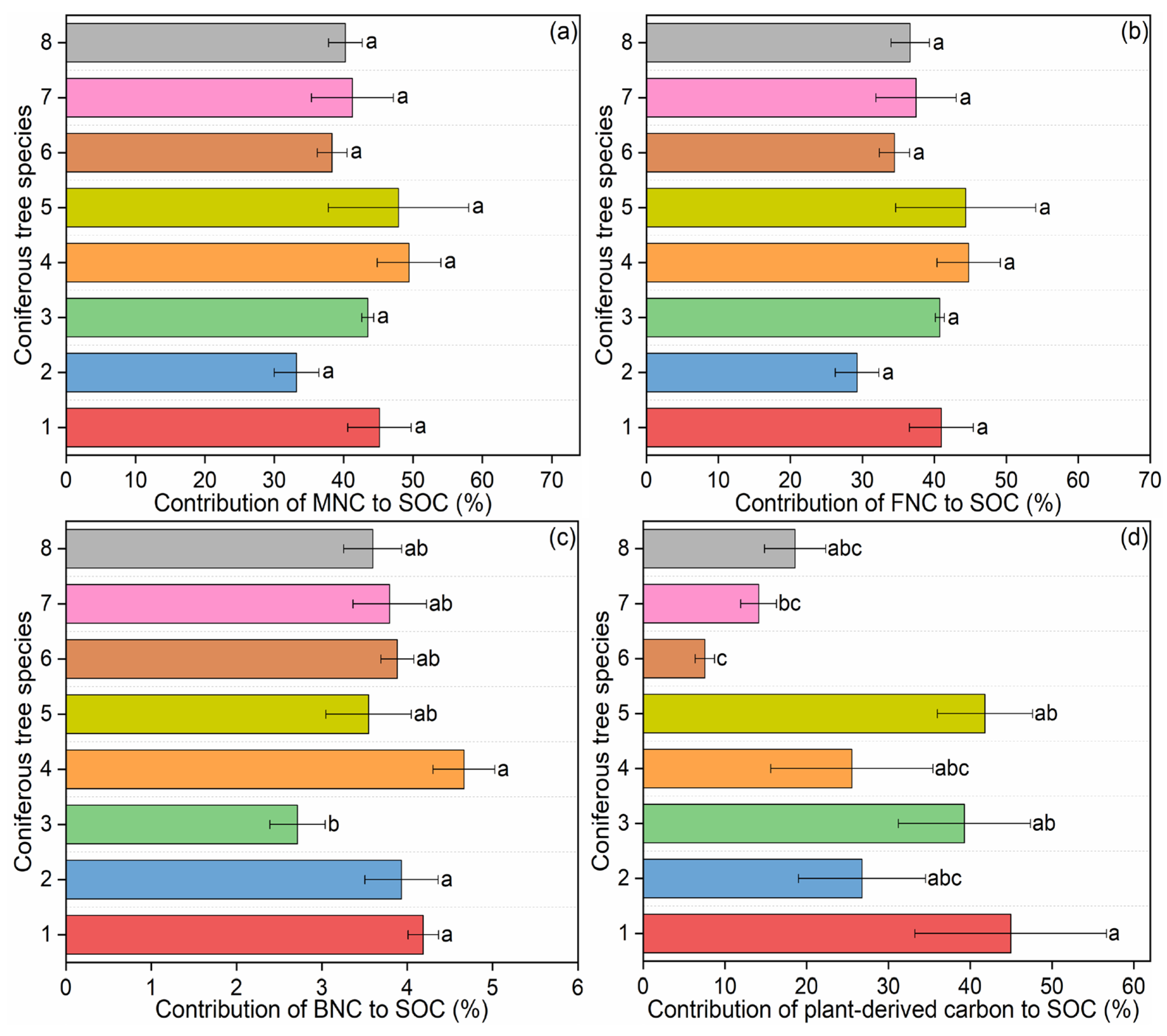
The concentrations of microbial necromass carbon ranged between 12.35 and 35.04 g·kg−1 (Figure 2b–d), which contributed 33.22%–49.45% to SOC contents (Figure 3a), with an average of 9.6 times of those from fungi than from bacteria (Figure 3b,c). When examining the contribution of necromass carbon to SOC (i.e., the percentage of SOC derived from necromass), significant differences among forest types were detected only for bacterial necromass carbon (p < 0.05; Table S3). In contrast, the contributions of total microbial necromass carbon and fungal necromass carbon to SOC showed no significant variation across the eight coniferous species (p > 0.05; Table S3).
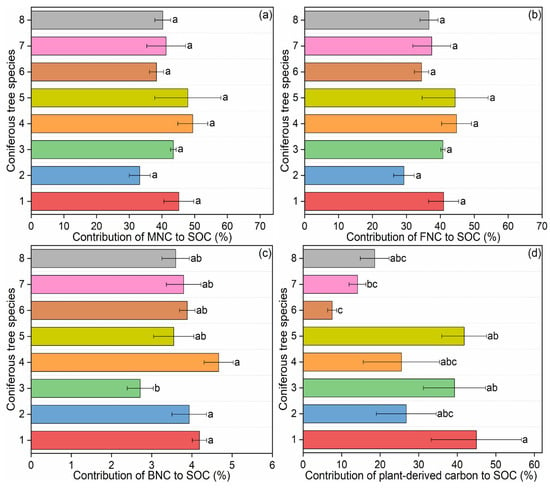
Figure 3.
Contribution of microbial necromass carbon and plant-derived carbon to SOC in coniferous forest soils: (a) contribution of microbial necromass carbon (MNC) to soil organic carbon (SOC); (b) contribution of fungal necromass carbon (FNC) to SOC; (c) contribution of bacterial necromass carbon (BNC) to SOC; (d) contribution of plant-derived carbon to SOC. Data are presented as mean ± standard error (SE). Significant differences among different coniferous tree species are indicated by different letters at the 0.05 level. Abbreviations: 1: Pinus armandii; 2: Larix gmelinii var. principis-rupprechtii; 3: Abies fargesii var. faxoniana; 4: Pinus tabuliformis; 5: Picea asperata; 6: Larix kaempferi; 7: Tsuga chinensis; 8: Larix potaninii.
3.2.3. Characteristics of Plant-Derived Carbon in Coniferous Forest Soils
There are significant differences in lignin phenols among different coniferous tree species in terms of the contents of total lignin phenols and vanillyl, the ratios of cinnamyl/vanillyl and acid/aldehyde in syringyl phenols (p < 0.05) (Table S4; Figure S3). Plant-derived carbon concentrations ranged from 0.2 to 1.2 g·kg−1 (Figure S3i), which accounted for 7.56%–44.96% of SOC concentrations, with the largest and lowest values occurred in P. armandii and L. kaempferi respectively (Figure 3d).
3.3. Factors Affecting Concentrations of Soil Organic Carbon and Its Fractions in Coniferous Forests
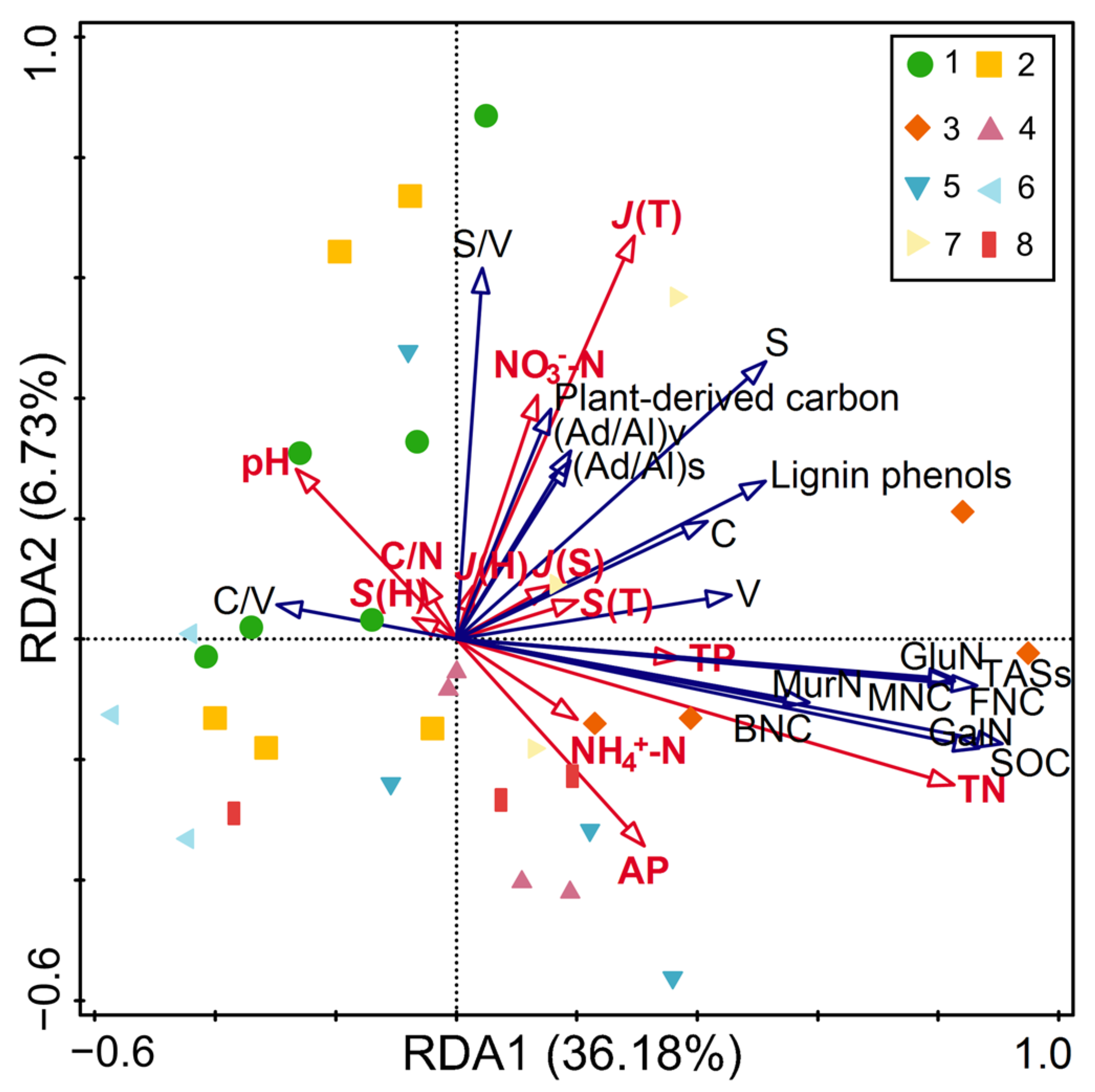
Prior to redundancy analysis, multicollinearity among explanatory variables was examined using VIF. All variables had VIF < 3 (Table S1), indicating no severe multicollinearity issues. Redundancy analysis was then performed to elucidate the controls on SOC concentrations and their fractions by soil properties and plant diversity (Figure 4; Table S5). The model explained 42.91% of total variance, with three key drivers identified: total nitrogen, C/N ratio and tree evenness.
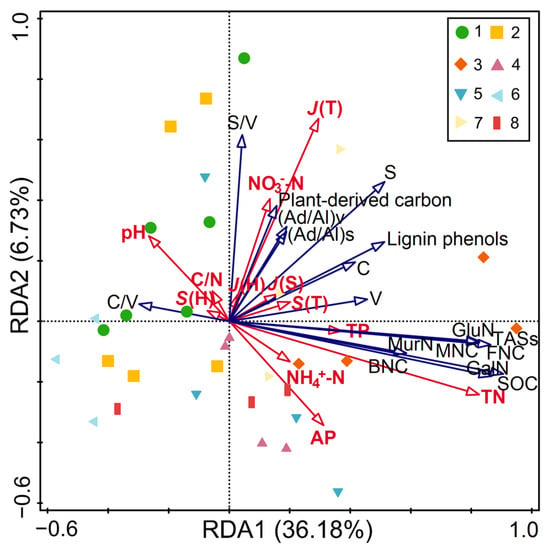
Figure 4.
Redundancy analysis of the effect of soil physicochemical properties and plant diversity on SOC content and components. Abbreviations: SOC: soil organic carbon; MNC: microbial necromass carbon; BNC: bacterial necromass carbon; FNC: fungal necromass carbon; TASs: total amino sugars concentrations; GluN: glucosamine; GalN: galactosamine; MurN: muramic acid; V: vanillyl; S: syringyl; C: cinnamyl; (Ad/Al)v and (Ad/Al)s: acid-aldehyde ratios of vanillyl and syringyl phenols; TN: total nitrogen; C/N: carbon-nitrogen ratio; TP: total phosphorus; NO3−-N: nitrate nitrogen; NH4+-N: ammonium nitrogen; AP: available phosphorus. S(T) and J(T): species richness and Pielou evenness index of the trees; J(S): Pielou evenness index of the shrubs; S(H) and J(H): species richness and Pielou evenness index of the herbs. 1: Pinus armandii; 2: Larix gmelinii var. principis-rupprechtii; 3: Abies fargesii var. faxoniana; 4: Pinus tabuliformis; 5: Picea asperata; 6: Larix kaempferi; 7: Tsuga chinensis; 8: Larix potaninii.
Tree species exerted a direct positive effect on soil total nitrogen standardized path coefficient (β = 0.54, p < 0.05) while negative effects on plant diversity (β = −0.38, p < 0.05) and soil pH (β = −0.48, p < 0.05). In addition, lower pH was indirectly associated with higher microbial-derived carbon (β = −0.20, p < 0.001), which promoted the accumulation of SOC. Meanwhile, elevated total nitrogen levels not only directly increased SOC content (β = 0.38, p < 0.05), but also indirectly promoted SOC sequestration by stimulating microbial-derived carbon production (β = 0.56, p < 0.001). Plant-derived carbon exerted no direct influence on SOC sequestration, operating exclusively through microbial-derived carbon pathways (β = 0.44, p < 0.001) (Figure 5).
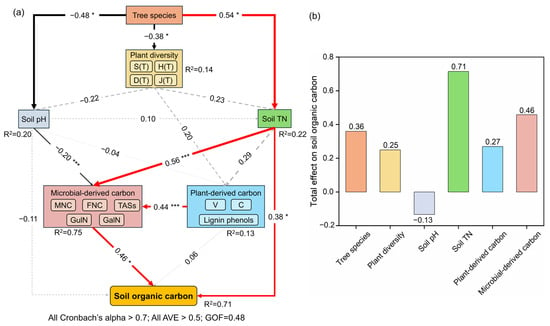
Figure 5.
Results of PLS-PM (a) and total effects on soil organic carbon (b). The red and black lines represent positive and negative effects, respectively, and gray dash line represents insignificant effects. * and *** indicate p < 0.05 and p < 0.001, respectively. Abbreviations: S(T), H(T), D(T), J(T): species richness, Shannon–Wiener diversity index, Simpson diversity index, Pielou evenness index of the trees; TN: total nitrogen; V: vanillyl; C: cinnamyl; MNC: microbial necromass carbon; FNC: fungal necromass carbon; TASs: total amino sugars concentrations; GluN: glucosamine; GalN: galactosamine; SOC: soil organic carbon; AVE: average variance extracted; GOF: goodness-of-fit.
4. Discussion
4.1. Contribution of Microbial-Derived Carbon to Soil Organic Carbon Sequestration
The pattern of individual amino sugars in the topsoil of Longnan coniferous forests (Figure S2) was consistent with the results across diverse coniferous forests [42] but contrary to findings from broadleaf forests [43]. This was ascribed to environmental conditions which favor fungi community but suppress bacterial proliferation and cell wall synthesis: acidic soil (pH 4.5–6.0) [22] and high lignin litter input in coniferous forests [44] and the elevated soil C/N ratios [45]. By contrast, more decomposable litter and near-neutral pH in broadleaf forests enhance bacterial growth and activity and thus muramic acid production [43].
Microbial necromass carbon constituted 33.22%–49.45% of SOC concentrations, with fungal necromass contributing much greater than that from bacteria (Figure 3). This pronounced fungal necromass carbon dominance supported previous observations of fungal-mediated SOC stabilization in coniferous ecosystems [46,47]. Notably, Abies fargesii var. faxoniana exhibited the highest microbial necromass carbon concentration among all species (35.04 g·kg−1), which was 2.8-fold higher than that of Larix kaempferi (12.35 g·kg−1) (Figure 2b). This substantial difference in microbial necromass accumulation underpins the observed variation in total SOC across species [11], highlighting the central role of microbial-derived carbon in driving species-level differences in soil carbon storage. The disparity in contributions of fungal and bacterial necromass to SOC concentrations can be attributed to three factors: (1) recalcitrant fungal cell wall components (e.g., chitin and chitosan) are more decomposition-resistant than bacterial muramic acids [48]; (2) bacterial necromass undergoes preferential reuse due to its lower C/N compared to fungal residues [49]; and (3) fungal hyphae exhibit greater metabolic activity and stronger mineral-association capabilities [50].
Collectively, the characteristics of amino sugars contents and the disproportionate contribution of fungal necromass to SOC pools provide convergent evidence for the central role of fungal necromass carbon in long-term SOC sequestration in coniferous ecosystems. The exceptional performance of A. fargesii var. faxoniana in accumulating microbial necromass suggests that this species creates belowground conditions particularly conducive to microbial processing and stabilization of organic matter. This microbial functional bias offers a mechanistic foundation for SOC persistence and informs strategies for managing forest soil carbon stocks.
4.2. Key Drivers of Soil Organic Carbon Concentrations and Their Fractions in Coniferous Forests
Soil total nitrogen, C/N ratio, and tree evenness were identified as key drivers shaping concentrations of SOC and their fractions in coniferous forests (Figure 4; Table S2), highlighting the complex interplay between soil nutrient status and vegetation structure in regulating SOC dynamics. We acknowledge that total nitrogen and SOC are inherently coupled as major constituents of soil organic matter, and their close association in the ordination partly reflects this fundamental biogeochemical linkage. Nevertheless, total nitrogen (VIF = 1.951, Table S1) provided statistically independent information and served in our analysis as a soil fertility status that sets the background conditions for organic matter dynamics. Within this fertility context, the positive correlations between total nitrogen and microbial necromass carbon, amino sugars, and lignin phenols aligned with previous studies [51]. Consistent with this pattern, A. fargesii var. faxoniana exhibited the highest total nitrogen concentration among all species (Table 1), which likely contributed to its elevated microbial necromass accumulation and ultimately its superior SOC content. In contrast, L. kaempferi showed the lowest total nitrogen levels, corresponding with its minimal microbial necromass and SOC concentrations. This pattern suggests that nitrogen-rich environments support greater microbial community stability and organic matter accumulation, thereby promoting microbial necromass carbon formation and SOC sequestration [52,53,54]. Additionally, nitrogen enrichment has been shown to promote oxidative depolymerization of lignin phenols [55], further linking soil nitrogen status to organic matter composition.
Tree evenness was positively correlated with SOC concentration and most components, which was consistent with prior findings [56,57]. This indicated that forests with greater tree evenness create environments conducive to SOC accumulation and microbial transformation processes. These were likely operated by enhanced tree evenness through three mechanisms: (1) diverse litter chemistry provides broader substrate resources for decomposers [58]; (2) increased root exudate diversity stimulates microbial activity and necromass production [59]; and (3) weakened dominance by single species promotes balanced resource distribution, enhancing SOC sequestration [60].
4.3. Driving Mechanisms of Soil Organic Carbon Sequestration in Coniferous Forests
The positive effects of tree species on soil total nitrogen, while adverse effects on pH (Figure 5), were ascribed to conifer-specific functional traits: (1) the pervasive production of acidic, recalcitrant litter acidifies soils [61]; (2) the slow decomposition rates and efficient nitrogen retention mechanisms under acidic conditions [62]. Such outcomes underscore that the consequences of plant diversity depend fundamentally on trait complementarity within the species pool [63].
This meaningful associations between total nitrogen, microbial-derived carbon, and SOC accumulation observed can be largely attributed to two mechanisms. (1) Microbial cells are primarily composed of nitrogen-rich compounds, including proteins, nucleic acids, fungal chitin, and bacterial peptidoglycan, which together account for more than 60% of the total soil nitrogen pool [47]. (2) Under nitrogen-limited conditions, microbial necromass is more readily decomposed by extracellular enzymes, releasing nitrogen to support the growth of living microbial biomass [49]. The high nitrogen availability in A. fargesii var. faxoniana soils thus creates a positive feedback loop: sufficient nitrogen alleviates microbial nitrogen limitation, reducing the need for necromass recycling and allowing greater necromass accumulation over time. In addition, the microbial-derived carbon increased with the decline in soil pH, which may be due to the fact that lower soil pH can increase binding between protonated carboxylic groups and negatively charged soil particles [64], thereby increasing microbial-derived carbon [52]. The acidic conditions under A. fargesii var. faxoniana (pH 5.67) are particularly favorable for this organo–mineral stabilization mechanism.
Microbial-derived carbon emerged as the non-negotiable engine of SOC persistence (Figure 5) [64], eclipsing direct plant contributions. The insignificance of plant-derived carbon in SOC formation indicated that microbial processing is the indispensable gateway for carbon stabilization [65]. This finding is particularly relevant for understanding species differences: the superior SOC accumulation in A. fargesii var. faxoniana is not driven by greater plant residue inputs per se, but by the species’ ability to foster microbial communities and conditions that efficiently convert plant inputs into stable microbial necromass. Despite significant tree species effects on plant diversity, no detectable influence to soil pH, total nitrogen and plant-derived carbon occurred. This was attributed to two interdependent mechanisms. Firstly, the absence of key functional groups (e.g., nitrogen-fixing plants, deep-rooted hardwoods) limits niche differentiation in resource acquisition [66]. Secondly, conifer-driven soil acidification dissociates plant diversity from soil processes [67]. Consequently, plant diversity functions as an ecological epiphenomenon rather than a functional driver in carbon transformation pathways.
4.4. Contextualizing the Findings and Future Research Directions
The findings of this study should be interpreted within the context of several considerations that also point toward productive directions for future research. As described in Section 2.2, the complex mountainous terrain of the Longnan region means that plots representing different coniferous species inherently vary in topographic conditions (elevation, slope, aspect) and potentially in soil parent materials and texture. This multi-faceted variation faithfully reflects the natural habitat distribution of these species, where different conifers occupy distinct environmental niches. However, this inherent covariation between tree species distribution and abiotic factors means that we cannot entirely rule out potential confounding effects on the observed differences in SOC contents among forest types. Topography influences microclimate, erosion, and hydrology; parent material determines mineral composition and weathering; and soil texture affects water retention, nutrient availability, and physical protection of organic matter—all of which influence SOC dynamics. Thus, the “tree species effects” we report may partially reflect underlying abiotic gradients that covary with species distribution. Future studies could disentangle these intertwined effects through paired sampling along toposequences or within similar parent material units, and through controlled garden experiments using homogenized soils.
Sample size presents another consideration for interpreting our PLS-PM results. With 32 observations and multiple predictors across several constructs, the sample size is modest for path modeling. While PLS-PM is specifically designed for smaller samples and does not require the large sample sizes demanded by covariance-based structural equation modeling, we acknowledge that the current sample size may limit detection of smaller effects and increase the risk of overfitting. The bootstrap procedure with 5000 resamples provides some protection against instability. Nevertheless, larger-scale sampling campaigns with increased plot numbers would enhance statistical power and enable more complex multivariate analyses, including covariance-based structural equation modeling with latent variables.
These considerations notwithstanding, our study provides robust evidence that tree species influence SOC accumulation primarily through microbial pathways mediated by soil nitrogen and pH, with Abies fargesii var. faxoniana demonstrating particularly high carbon sequestration capacity in this mountain region. Future research building on these findings, through experimental manipulations, expanded sampling, and process-based modeling will further clarify the relative contributions of tree species identity versus environmental conditions to SOC dynamics in complex mountain ecosystems like Longnan.
5. Conclusions
SOC concentrations ranged from 31.76 to 80.86 g·kg−1, with A. fargesii var. faxoniana exhibiting the highest values among all species. The relative amino sugar abundance consistently followed glucosamine > galactosamine > muramic acid. Fungal necromass contributed 29%–45% to SOC concentrations—substantially exceeding bacterial necromass inputs (3%–5%). Collectively, these patterns confirm fungal necromass dominance in SOC sequestration. Redundancy analysis identified that soil total nitrogen, C/N ratio, and tree evenness were the main factors affecting SOC concentrations and their fractions. Tree species exerted a direct positive effect on soil total nitrogen and adverse impact on pH. In addition, lower pH was indirectly associated with higher microbial-derived carbon, which promoted the accumulation of SOC. Meanwhile, elevated total nitrogen levels not only directly increased SOC content, but also indirectly promoted SOC sequestration by stimulation of microbial-derived carbon production. Plant-derived carbon functioned exclusively as a precursor to microbial-derived carbon without direct SOC linkage. These findings demonstrate that coniferous tree species promoted SOC sequestration primarily by facilitating the accumulation of microbial necromass carbon, as opposed to plant-derived residues, through modifications in soil total nitrogen and pH. Consequently, promoting A. fargesii var. faxoniana cultivation to improve soil total nitrogen levels and microbial-derived carbon can be an essential approach for optimizing carbon stabilization and achieving sustainable forest management in coniferous forest ecosystems.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/f17030379/s1, Table S1: Multicollinearity diagnostics of explanatory variables; Table S2: Analysis of variance for diversity of trees, shrubs, and herbs in coniferous forests; Table S3: Analysis of variance for microbial necromass carbon and amino sugars in coniferous forests; Table S4: Analysis of variance for plant-derived carbon in coniferous forest soils; Table S5: The goodness-of-fit (R2), adjusted goodness-of-fit (R2), explained fitted variation (contribution), F-value, and p-value of soil physicochemical properties and plant diversity for SOC content and components on Monte Carlo permutation test for RDA (Conditional effect); Figure S1: Schematic diagram of quadrats setup for tree, shrub, and herb; Figure S2: Characteristics of amino sugars in coniferous forest soils; Figure S3: Contents of lignin phenols (plant-derived biomarkers) and plant-derived C in the surface soil of coniferous forests in Longnan region.
Author Contributions
X.W.: Writing—original draft, Visualization, Formal analysis, Data curation. X.Z.: Writing—original draft, Formal analysis. Y.X.: Investigation, Formal analysis. R.F.: Investigation, Formal analysis. J.L.: Writing—review and editing, Conceptualization. C.Z.: Writing—review and editing, Supervision, Validation, Conceptualization. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Second Comprehensive Investigation and Research Topic of the Qinghai–Tibet Plateau (2019QZKK0301).
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Acknowledgments
We extend our gratitude to Mingxin Liu, a Young Researcher from the College of Ecology at Lanzhou University, for his valuable assistance in polishing this manuscript. We also appreciate the editor and the reviewers for their constructive suggestions.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Zhou, S.; Li, P.; Zhang, Y. Factors influencing and changes in the organic carbon pattern on slope surfaces induced by soil erosion. Soil Tillage Res. 2024, 238, 106001. [Google Scholar] [CrossRef]
- Lal, R. Digging deeper: A holistic perspective of factors affecting soil organic carbon sequestration in agroecosystems. Glob. Change Biol. 2018, 24, 3285–3301. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.T.; Yang, R.M.; Zhang, X.; Xu, L.; Zhu, C.M. Understanding drivers of the spatial variability of soil organic carbon in China’s terrestrial ecosystems. Land Degrad. Dev. 2024, 35, 308–320. [Google Scholar] [CrossRef]
- Chen, X.; Luo, G.; Zhu, R.; Zhang, Y.; Huang, Y.; Liao, C. Straw Returning Amount and Duration Influence Paddy Soil Organic Carbon Sequestration by Regulating Plant-and Microbe-Derived Carbon Accumulation. Land Degrad. Dev. 2025, 36, 3729–3741. [Google Scholar] [CrossRef]
- Lal, R.; Monger, C.; Nave, L.; Smith, P. The role of soil in regulation of climate. Philos. Trans. R. Soc. 2021, 376, 20210084. [Google Scholar] [CrossRef]
- Ma, T.; Zhu, S.S.; Wang, Z.H.; Chen, D.M.; Dai, G.H.; Feng, B.W.; Su, X.Y.; Hu, H.F.; Li, K.F.; Han, W.X.; et al. Divergent accumulation of microbial necromass and plant lignin components in grassland soils. Nat. Commun. 2018, 9, 3480. [Google Scholar] [CrossRef]
- Cotrufo, M.F.; Ranalli, M.G.; Haddix, M.L.; Six, J.; Lugato, E. Soil carbon storage informed by particulate and mineral-associated organic matter. Nat. Geosci. 2019, 12, 989–994. [Google Scholar] [CrossRef]
- Cong, M.; Zhang, Z.; Zhao, G.; Dong, X.; Wang, W.; Mu, Z.; Tariq, A.; Graciano, C.; Sardans, J.; Peñuelas, J.; et al. Shrub Afforestation Increases Microbial-Derived Carbon in Arid Regions. Land Degrad. Dev. 2025, 36, 4691–4702. [Google Scholar] [CrossRef]
- Liu, Y.; Halik, Ü.; Teng, Z.; Fu, W.; He, J.; Liu, M.; Wang, X. Stand development promotes the contribution of plant-and microbial-derived carbon to soil organic carbon in Populus euphratica desert forests. Appl. Soil Ecol. 2025, 214, 106350. [Google Scholar] [CrossRef]
- Liang, C.; Zhu, X.F. The soil Microbial Carbon Pump as a new concept for terrestrial carbon sequestration. Sci. China Earth Sci. 2021, 51, 680–695. [Google Scholar] [CrossRef]
- Whalen, E.D.; Grandy, A.S.; Sokol, N.W.; Keiluweit, M.; Ernakovich, J.; Smith, R.G.; Frey, S.D. Clarifying the evidence for microbial- and plant-derived soil organic matter, and the path toward a more quantitative understanding. Glob. Change Biol. 2022, 28, 7167–7185. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Qu, L.; Yang, L.; Liu, D.; Morrissey, E.; Miao, R.; Liu, Z.; Wang, Q.; Fang, Y.; Bai, E. Large-scale importance of microbial carbon use efficiency and necromass to soil organic carbon. Glob. Change Biol. 2021, 27, 2039–2048. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.F.; Ding, J.Z.; Li, J.; Xin, Z.M.; Ren, S.; Wang, T. Necromass-derived soil organic carbon and its drivers at the global scale. Soil Biol. Biochem. 2023, 181, 109025. [Google Scholar] [CrossRef]
- Angst, G.; Mueller, K.E.; Nierop, K.G.J.; Simpson, M.J. Plant- or microbial-derived? A review on the molecular composition of stabilized soil organic matter. Soil Biol. Biochem. 2021, 156, 108189. [Google Scholar] [CrossRef]
- Zhu, E.X.; Cao, Z.J.; Jia, J.; Liu, C.Z.; Zhang, Z.H.; Wang, H.; Dai, G.H.; He, J.S.; Feng, X.J. Inactive and inefficient: Warming and drought effect on microbial carbon processing in alpine grassland at depth. Glob. Change Biol. 2021, 27, 2241–2253. [Google Scholar] [CrossRef]
- Pan, Y.; Birdsey, R.A.; Fang, J.; Houghton, R.; Kauppi, P.E.; Kurz, W.A.; Phillips, O.L.; Shvidenko, A.; Lewis, S.L.; Canadell, J.G.; et al. A large and persistent carbon sink in the world’ s forests. Science 2011, 333, 988–993. [Google Scholar] [CrossRef]
- Jackson, R.B.; Lajtha, K.; Crow, S.E.; Hugelius, G.; Kramer, M.G.; Piñeiro, G. The ecology of soil carbon: Pools, vulnerabilities, and biotic and abiotic controls. Annu. Rev. Ecol. Evol. Syst. 2017, 48, 419–445. [Google Scholar] [CrossRef]
- Mayer, M.; Prescott, C.E.; Abaker, W.E.; Augusto, L.; Cécillon, L.; Ferreira, G.W.; James, J.; Jandl, R.; Katzensteiner, K.; Laclau, J.P.; et al. Tamm Review: Influence of forest management activities on soil organic carbon stocks: A knowledge synthesis. For. Ecol. Manag. 2020, 466, 118127. [Google Scholar] [CrossRef]
- Bradford, M.A.; Wieder, W.R.; Bonan, G.B.; Fierer, N.; Raymond, P.A.; Crowther, T.W. Managing uncertainty in soil carbon feedbacks to climate change. Nat. Clim. Change 2016, 6, 751–758. [Google Scholar] [CrossRef]
- Takele, L.; Yang, S.; Chen, Z.; Yuan, J.; Ding, W. Contribution of microbial necromass to soil organic carbon in profile depths exhibited opposite patterns across ecosystems: A global meta-analysis. Soil Biol. Biochem. 2025, 207, 109842. [Google Scholar] [CrossRef]
- Yang, X.; Song, Z.; Van Zwieten, L.; Guo, L.; Chen, J.; Luo, Z.; Wang, Y.; Luo, Y.; Wang, Z.; Wang, W. Significant accrual of soil organic carbon through long-term rice cultivation in paddy fields in China. Glob. Change Biol. 2024, 30, 17213. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Chen, X.; Zhou, X.; Nie, X.; Liu, G.; Zhuang, G.; Zheng, G.; Fortin, D.; Ma, A. Tibetan Plateau grasslands might increase sequestration of microbial necromass carbon under future warming. Commun. Biol. 2024, 7, 686. [Google Scholar] [CrossRef] [PubMed]
- Augusto, L.; Boca, A. Tree functional traits, forest biomass, and tree species diversity interact with site properties to drive forest soil carbon. Nat. Commun. 2022, 13, 1097. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ding, Y.; Zhang, Y.; Wang, J.; Freedman, Z.B.; Liu, P.; Cong, W.; Wang, J.; Zang, R.; Liu, S. Evenness of soil organic carbon chemical components changes with tree species richness, composition and functional diversity across forests in China. Glob. Change Biol. 2023, 29, 2852–2864. [Google Scholar] [CrossRef]
- Pan, S.A.; Hao, G.; Li, X.; Feng, Q.; Liu, X.; Sun, O.J. Altitudinal variations of hydraulic traits in Faxon fir (Abies fargesii var. faxoniana): Mechanistic controls and environmental adaptability. For. Ecosyst. 2022, 9, 100040. [Google Scholar] [CrossRef]
- Li, F.; Shi, Z.; Liu, S.; Chen, M.; Xu, G.; Chen, J.; Xing, H. Stand structural diversity and edaphic properties regulate aboveground biomass of Abies fargesii var. faxoniana primary forest on the eastern Qinghai-Tibetan Plateau. Eur. J. For. Res. 2024, 143, 1401–1410. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources. International Soil Classification System for Naming Soils and Creating Legends for Soil Maps, 4th ed.; International Union of Soil Sciences (IUSS): Rome, Italy, 2022. [Google Scholar]
- Thomas, G.W. Soil pH and soil acidity. In Methods of Soil Analysis: Part 3 Chemical Methods; Wiley: Hoboken, NJ, USA, 1996; Volume 5, pp. 475–490. [Google Scholar]
- Shi, J.W.; Deng, L.; Gunina, A.; Alharbi, S.; Wang, K.B.; Li, J.W.; Liu, Y.L.; Shangguan, Z.P.; Kuzyakov, Y. Carbon stabilization pathways in soil aggregates during long-term forest succession: Implications from δ13C signatures. Soil Biol. Biochem. 2023, 180, 108988. [Google Scholar] [CrossRef]
- Bremner, J.M.; Mulvaney, C.S. Total nitrogen. In Methods of Soil Analysis, Part 2: Chemical and Microbiological Properties; Page, A.L., Ed.; American Society of Agronomy: Madison, WI, USA, 1982. [Google Scholar]
- Bao, S. Soil and Agricultural Chemistry Analysis; China Agriculture Press: Beijing, China, 2000. (In Chinese) [Google Scholar]
- Xie, P.; Yang, Y.; Su, Q.; He, R.; Liu, W.; Lv, X.; von Gadow, K. Disturbance-Driven Litter-Soil-Microbe Interactions Affect Microbial Carbon Use Efficiency. Eur. J. Soil Sci. 2026, 77, e70291. [Google Scholar] [CrossRef]
- Bray, R.H.; Kurtz, L.T. Determination of total, organic, and available forms of phosphorus in soils. Soil Sci. 1945, 59, 39–46. [Google Scholar] [CrossRef]
- Luo, R.Y.; Kuzyakov, Y.; Chen, J.; Qiang, W.; Zhang, Y.; Pan, X.Y. Phosphorus enrichment mediates the responses of plant lignin and microbial necromass accumulation to nitrogen addition in subalpine forest soil. Geoderma 2025, 458, 117317. [Google Scholar] [CrossRef]
- Chen, G.; Ma, S.; Tian, D.I.; Xiao, W.; Jiang, L.; Xing, A.; Zou, A.; Zhou, L.; Shen, H.; Zheng, C.; et al. Patterns and determinants of soil microbial residues from tropical to boreal forests. Soil Biol. Biochem. 2020, 151, 108059. [Google Scholar] [CrossRef]
- Ma, T.; Yang, Z.; Shi, B.; Gao, W.; Li, Y.; Zhu, J.; He, J.S. Phosphorus supply suppressed microbial necromass but stimulated plant lignin phenols accumulation in soils of alpine grassland on the Tibetan Plateau. Geoderma 2023, 431, 116376. [Google Scholar] [CrossRef]
- Dao, T.T.; Mikutta, R.; Sauheitl, L.; Gentsch, N.; Shibistova, O.; Wild, B.; Schnecker, J.; Bárta, J.; Čapek, P.; Gittel, A.; et al. Lignin preservation and microbial carbohydrate metabolism in permafrost soils. J. Geophys. Res. Biogeosci. 2022, 127, e2020JG006181. [Google Scholar] [CrossRef]
- Chen, L.Y.; Fang, K.; Wei, B.; Qin, S.Q.; Feng, X.H.; Hu, T.Y.; Ji, C.J.; Yang, Y.H. Soil carbon persistence governed by plant input and mineral protection at regional and global scales. Ecol. Lett. 2021, 24, 1018–1028. [Google Scholar] [CrossRef]
- Burgess, M.S.; Mehuys, G.R.; Madramootoo, C.A. Decomposition of grain-corn residues (Zea mays L.): A litterbag study under three tillage systems. Can. J. Soil Sci. 2002, 82, 127–138. [Google Scholar] [CrossRef]
- Bo, Y.; Wang, X.; van Groenigen, K.J.; Linquist, B.A.; Müller, C.; Li, T.; Yang, J.; Jägermeyr, J.; Qin, Y.; Zhou, F. Improved alternate wetting and drying irrigation increases global water productivity. Nat. Food 2024, 5, 1005–1013. [Google Scholar] [CrossRef]
- Xu, H.; He, X.; Chen, J.; Huang, X.; Chen, Y.; Xu, Y.; Xiao, Y.; Liu, T.; He, H.; Wang, Y.; et al. Soil nutrient limitation controls trophic cascade effects of micro-food web-derived ecological functions in degraded agroecosystems. J. Adv. Res. 2025, 77, 43–55. [Google Scholar] [CrossRef]
- Ding, X.; Zhang, B.; Lv, X.; Wang, J.; Horwath, W.R. Parent material and conifer biome influence microbial residue accumulation in forest soils. Soil Biol. Biochem. 2017, 107, 1–9. [Google Scholar] [CrossRef]
- Teng, Q.; Lu, X.; Zhang, Q.; Cai, L.; Sardar, M.F.; Li, Y.; Abbas, T.; Li, Y.; Chang, S.X.; Li, Y. Litterfall quality modulates soil ammonium and nitrate supply through altering microbial function in bamboo encroachment of broadleaf forests. Geoderma 2023, 437, 116592. [Google Scholar] [CrossRef]
- Min, X.; Xiao, L.; Li, Z.; Li, P.; Ma, J.; Wang, B.; Du, D.; Qiu, W. Litter decomposition stage exerted a stronger influence on soil organic carbon fractions than forest litter type. Land Degrad. Dev. 2025, 36, 4643–4657. [Google Scholar] [CrossRef]
- Duan, Y.; Zhang, J.; Petropoulos, E.; Zhao, J.; Jia, R.; Wu, F.; Chen, Y.; Wang, L.; Wang, X.; Li, Y.; et al. Soil acidification destabilizes terrestrial ecosystems via decoupling soil microbiome. Glob. Change Biol. 2025, 31, e70174. [Google Scholar] [CrossRef]
- He, J.; Nie, Y.; Tan, X.; Hu, A.; Li, Z.; Dai, S.; Ye, Q.; Zhang, G.; Shen, W. Latitudinal patterns and drivers of plant lignin and microbial necromass accumulation in forest soils: Disentangling microbial and abiotic controls. Soil Biol. Biochem. 2024, 194, 109438. [Google Scholar] [CrossRef]
- Fu, H.; Chen, H.; Ma, Z.; Liang, G.; Chadwick, D.R.; Jones, D.L.; Wanek, W.; Wu, L.; Ma, Q. Fungal Necromass Carbon Dominates Global Soil Organic Carbon Storage. Glob. Change Biol. 2025, 31, e70413. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wang, X.; Pei, G.; Xia, Z.; Peng, B.; Sun, L.; Wang, J.; Gao, D.; Chen, S.; Liu, D.; et al. Stabilization of microbial residues in soil organic matter after two years of decomposition. Soil Biol. Biochem. 2020, 141, 107687. [Google Scholar] [CrossRef]
- Cui, J.; Zhu, Z.; Xu, X.; Liu, S.; Jones, D.L.; Kuzyakov, Y.; Shibistova, O.; Wu, J.; Ge, T. Carbon and nitrogen recycling from microbial necromass to cope with C: N stoichiometric imbalance by priming. Soil Biol. Biochem. 2020, 142, 107720. [Google Scholar] [CrossRef]
- Fernandez, C.W.; Langley, J.A.; Chapman, S.; McCormack, M.L.; Koide, R.T. The decomposition of ectomycorrhizal fungal necromass. Soil Biol. Biochem. 2016, 93, 38–49. [Google Scholar] [CrossRef]
- Yu, J.; Li, J.; Wu, J.; Shangguan, Z.; Deng, L. Patterns and controlling factors of soil microbial necromass carbon in global ecosystems. J. Environ. Manag. 2025, 385, 125632. [Google Scholar] [CrossRef]
- Chen, Q.; Hu, Y.; Hu, A.; Niu, B.; Yang, X.; Jiao, H.; Ri, X.; Song, L.; Zhang, G. Shifts in the dynamic mechanisms of soil organic matter transformation with nitrogen addition: From a soil carbon/nitrogen-driven mechanism to a microbe-driven mechanism. Soil Biol. Biochem. 2021, 160, 108355. [Google Scholar] [CrossRef]
- Yuan, X.; Qi, Y.; Guo, Y.; Dong, Y.; Peng, Q.; Yan, Z.; Li, Z.; Dong, R.; Zheng, Y. Effect of 9-year water and nitrogen additions on microbial necromass carbon content at different soil depths and its main influencing factors. Sci. Total Environ. 2024, 954, 176825. [Google Scholar] [CrossRef]
- Chen, W.; Yang, Y.; Liang, X.; Chang, S.; Chang, Y.; Miao, N.; Xu, T.; Chen, D.; Wei, Y. Differential contributions of microbial necromass to humic acid during composting of organic wastes. Environ. Res. 2025, 270, 121036. [Google Scholar] [CrossRef]
- Peng, Y.; Li, Y.; Song, S.; Chen, Y.; Chen, G.; Tu, L. Nitrogen addition slows litter decomposition accompanied by accelerated manganese release: A five-year experiment in a subtropical evergreen broadleaf forest. Soil Biol. Biochem. 2022, 165, 108511. [Google Scholar] [CrossRef]
- Chen, X.; Taylor, A.R.; Reich, P.B.; Hisano, M.; Chen, H.Y.; Chang, S.X. Tree diversity increases decadal forest soil carbon and nitrogen accrual. Nature 2023, 618, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Huo, X.; Ren, C.; Wang, D.; Wu, R.; Wang, Y.; Li, Z.; Huang, D.; Qi, H. Microbial community assembly and its influencing factors of secondary forests in Qinling Mountains. Soil Biol. Biochem. 2023, 184, 109075. [Google Scholar] [CrossRef]
- Conti, G.; Díaz, S. Plant functional diversity and carbon storage–an empirical test in semi-arid forest ecosystems. J. Ecol. 2013, 101, 18–28. [Google Scholar] [CrossRef]
- Chen, S.; Wang, W.; Xu, W.; Wang, Y.; Wan, H.; Chen, D.; Tang, Z.; Tang, X.; Zhou, G.; Xie, Z.; et al. Plant diversity enhances productivity and soil carbon storage. Proc. Natl. Acad. Sci. USA 2018, 115, 4027–4032. [Google Scholar] [CrossRef]
- Hillebrand, H.; Bennett, D.M.; Cadotte, M.W. Consequences of dominance: A review of evenness effects on local and regional ecosystem processes. Ecology 2008, 89, 1510–1520. [Google Scholar] [CrossRef]
- De Marco, A.; Berg, B.; Zarrelli, A.; De Santo, A.V. Shifts in soil chemical and microbial properties across forest chronosequence on recent volcanic deposits. Appl. Soil Ecol. 2021, 161, 103880. [Google Scholar] [CrossRef]
- Magdziak, Z.; Gąsecka, M.; Waliszewska, B.; Zborowska, M.; Mocek, A.; Cichy, W.J.; Mazela, B.; Kozubik, T.; Mocek-Płóciniak, A.; Niedzielski, P.; et al. The influence of environmental condition on the creation of organic compounds in Pinus sylvestris L. rhizosphere, roots and needles. Trees 2021, 35, 441–457. [Google Scholar] [CrossRef]
- Yeeles, P.; Lach, L.; Hobbs, R.J.; Didham, R.K. Functional redundancy compensates for decline of dominant ant species. Nat. Ecol. Evol. 2025, 1, 10. [Google Scholar] [CrossRef]
- Zhou, T.; Fan, J.; Zhang, L.; Lv, Q.; Wang, T.; Meng, Y.; Meng, Y.; Hu, H.; Gao, H.; Wang, J.; et al. The accumulation of plant-and microbial-derived carbon and its contribution to soil organic carbon in reclaimed saline-sodic farmland. Appl. Soil Ecol. 2024, 202, 105558. [Google Scholar] [CrossRef]
- Camenzind, T.; Mason-Jones, K.; Mansour, I.; Rillig, M.C.; Lehmann, J. Formation of necromass-derived soil organic carbon determined by microbial death pathways. Nat. Geosci. 2023, 16, 115–122. [Google Scholar] [CrossRef]
- Liu, Z.; Hu, B.; Flemetakis, E.; Franken, P.; Haensch, R.; Rennenberg, H. Interactions between plant-soil feedbacks and climate control root symbioses. Plant Soil 2024, 512, 261–279. [Google Scholar] [CrossRef]
- Tang, T.; Hu, Z.D.; Wang, Y.; Hu, J.; Yuan, D.; Cai, L.; Feng, Q.; Xiao, J. Influence of soil substrate availability and plant species diversity on soil microbial biomass and enzyme activity in a subalpine natural secondary forest. J. Soils Sediments 2025, 25, 1628–1642. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.