Abstract
Rechargeable lithium-, sodium-, and potassium-ion batteries are utilized as essential energy storage devices for portable electronics, electric vehicles, and large-scale energy storage systems. In these systems, anode materials play a vital role in determining energy density, cycling stability, and safety of various batteries. However, the complex electrochemical reactions and dynamic changes that occur in anode materials during charge–discharge cycles generate major challenges for performance optimization and understanding failure mechanisms. In situ characterization techniques, capable of real-time tracking of microstructures, composition, and interface dynamics under operating conditions, provide critical insights that bridge macroscopic performance and microscopic mechanisms of anodes. This review systematically summarizes the applications of such techniques in studying anodes for lithium-, sodium-, and potassium-ion batteries, with a focus on their contributions across different anode types. It also indicates current challenges and future directions of these techniques, aiming to offer valuable references for relevant applications and the design of high-performance anodes.
1. Introduction
The past three decades have witnessed rapid advances in electrochemical energy storage. While lithium-ion batteries have achieved remarkable commercial and academic success [1,2,3], this has spurred interest in alternative technologies. Sodium-ion batteries have re-emerged as a cost-effective solution with mitigated supply chain risks [4,5,6,7], and potassium-ion batteries have gained significant interest owing to their high natural abundance, competitive energy density, fast kinetics, and low cost [8,9]. A key tool driving the understanding of all three systems is in situ characterization, which is widely used to probe anode materials and monitor real-time battery behavior during operation [10].
In situ characterization techniques are critically important in the studies of anode materials. They overcome the limitations of traditional ex situ methods by dynamically and directly establishing the “structure–composition–performance” relationship of anode materials under real operating conditions. These techniques provide direct evidence for understanding intrinsic mechanisms behind core issues such as capacity fading and poor cycling stability [11]. Furthermore, they guide the design optimization and process improvement of anode materials toward higher capacity, longer lifespan, and enhanced safety, thereby accelerating the development and industrialization of novel anodes for energy storage applications [12,13,14].
In recent years, in situ characterization techniques have been increasingly employed in the study of various anode materials (Figure 1). While several reviews have summarized the application of individual in situ techniques across different anodes, they often lack comprehensive coverage of multiple common methodologies [15]. Others have surveyed multiple characterization methods but focused only on a specific type or category of anode materials [16,17,18,19,20]. Although a limited number of reviews have discussed the use of multiple in situ techniques in anode studies, they are confined to a single battery type [21]. Therefore, a comprehensive review summarizing the applications of various in situ characterization techniques across different battery anode materials is urgently needed. Such work would help consolidate recent research findings and provide valuable guidance for future studies.
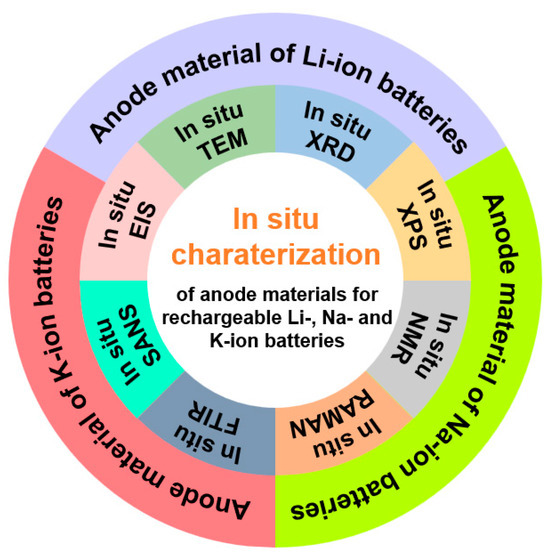
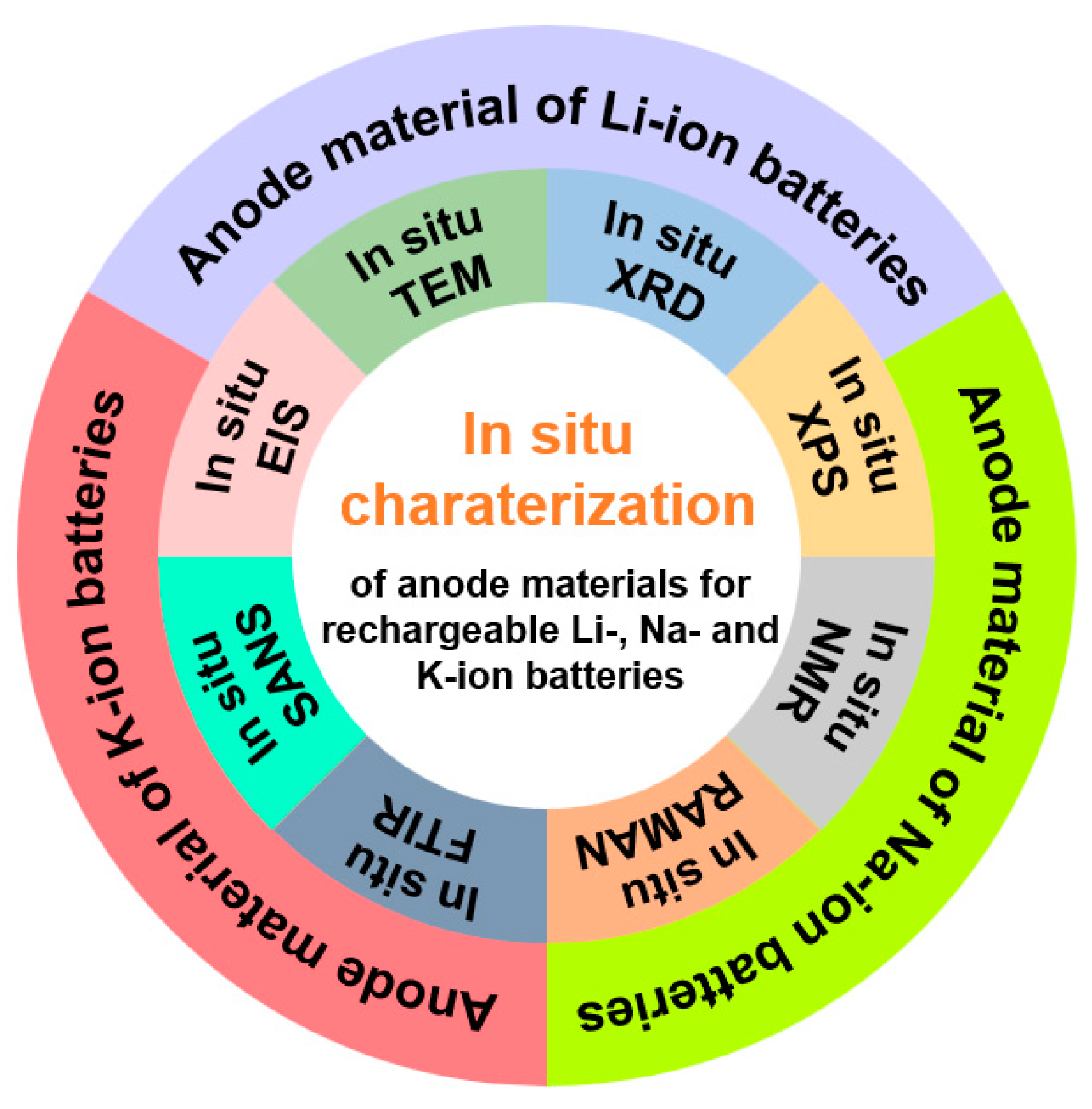
Figure 1.
Scheme of in situ techniques for the studies of anode materials in Li-, Na- and K-ion batteries.
The current review systematically summarizes the applications of diverse in situ characterization techniques in the research of anode materials for lithium-ion, sodium-ion, and potassium-ion batteries, addressing the gap of insufficient comprehensive coverage in existing reviews. It first outlines the core advantages, spatial–temporal resolution, applicable configurations, contributions and limitations of eight mainstream in situ techniques. Subsequently, via specific research cases, it elaborates on how each technique reveals the electrochemical reaction mechanisms, dynamic structural evolution, interfacial behavior, and ion transport kinetics of different anode materials during charge–discharge cycles, and discusses the complementary application of combined techniques to overcome single-method limitations. Finally, the review summarizes current progress, identifies key challenges, and proposes forward-looking perspectives on future research directions. By integrating novel insights for anode performance optimization and structural design, this work aims to provide a practical reference for alkali metal ion battery researchers, accelerating the development of high-performance anode materials and related technologies.
2. In Situ Characterization Technique
In situ characterization techniques offer significant advantages in anode materials studies. Their core strength lies in the ability to track real-time changes in morphology, chemical composition, and interfacial states of anode materials under dynamic operating conditions that simulate or are found in real battery charge–discharge processes [22,23]. This capability overcomes the limitations of traditional ex situ methods, which often miss transient reaction stages and are susceptible to artifacts introduced during sample transfer. Table 1 systematically sorts out the characterization contributions, restrictions and applicable battery configurations of eight mainstream in situ techniques, clarifying their basic application characteristics [24]. It is worth noting that different characterization techniques have significant differences in terms of spatial and temporal resolution, the information obtained, and the physical parameters. Researchers need to select appropriate in situ characterization methods based on different detection purposes.

Table 1.
Resolution, characterization information, and physical parameters of in situ techniques.
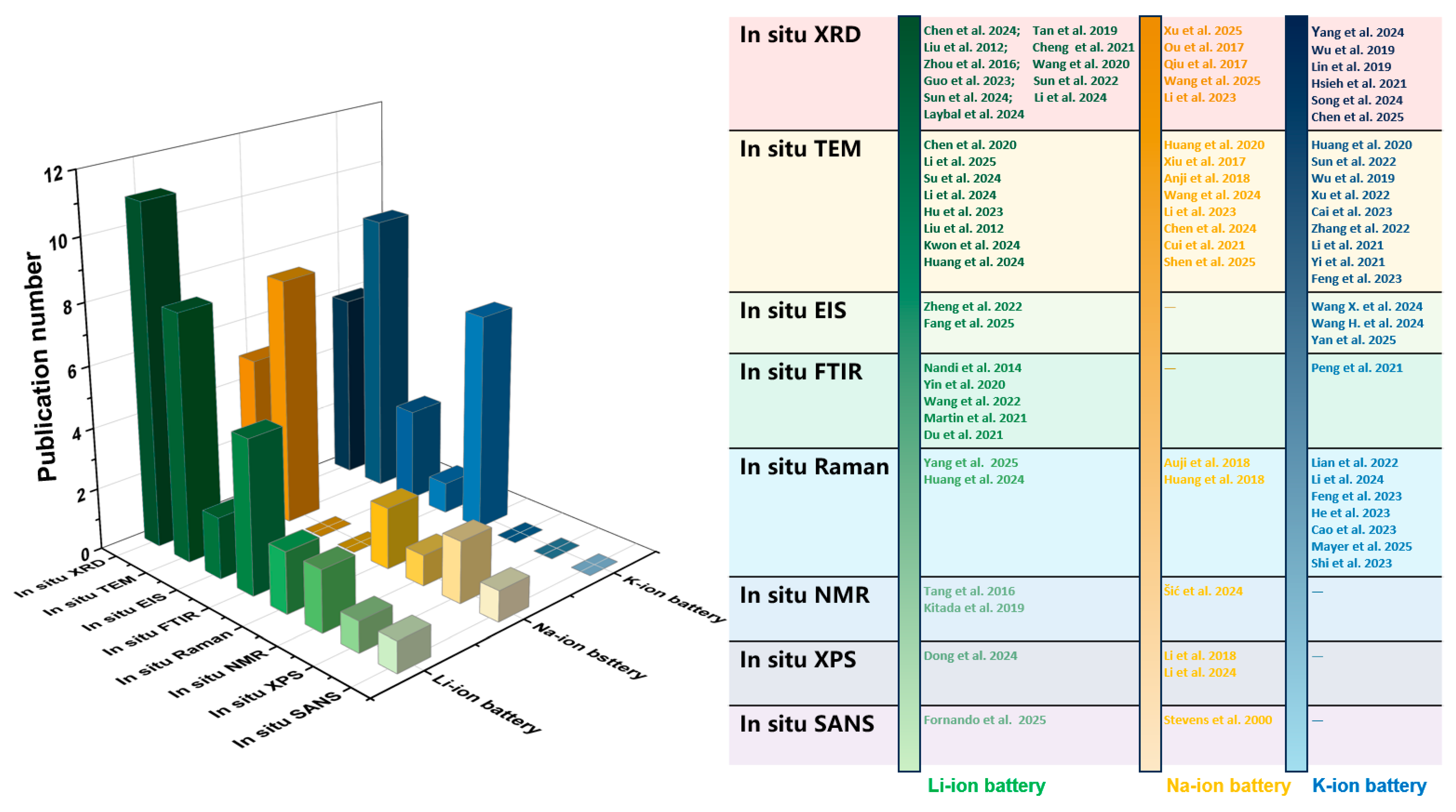
To facilitate researchers’ intuitive understanding of different characterization techniques, in addition to Table 1 which summarizes the fundamental information of each technique as presented above, the paper further compiles Table 2 to illustrate the various roles of these characterization techniques in studying different anode materials. The usage of these in situ characterization techniques in the development of anode materials for the three battery systems is summarized in Figure 2. At present, all eight types of in situ characterization techniques have been applied in the research of lithium-ion batteries. However, when studying sodium-ion batteries and potassium-ion batteries, some of these in situ characterization techniques are rarely involved, which demonstrates the mature application of in situ characterization technology in lithium-ion battery studies. Furthermore, the in situ XRD and in situ TEM techniques are applied more frequently in the three batteries compared to the other six in situ characterization methods, suggesting that in future studies, there is a significant potential to utilize different types of in situ characterization to further advance the study of different types of batteries.

Table 2.
The specific roles of several in situ characterization techniques in the development of anode materials for Li-, Na-, and K-ion batteries.
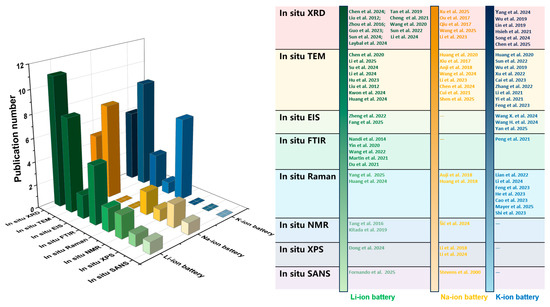
Figure 2.
Statistics regarding application status of various in situ characterization techniques in anode material studies for Li-, Na-, and K-ion batteries in recent years [49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95].
Each in situ technique will be elaborated in following sections, focusing on their core advantages and illustrating their application in battery mechanism research and performance optimization with relevant research examples so as to provide support for the efficient application of in situ characterization techniques in the battery field.
2.1. In Situ XRD
In situ XRD enables real-time monitoring of lattice parameter and phase composition changes in the electrode or at the electrode–electrolyte interface during battery operation. Its critical applications in anode research include the following: (1) For intercalation-type anodes, it helps identify intermediate phase formation, clarify reaction pathways, and verify intercalation mechanisms and structural stability [85]. (2) For alloy-type anodes, it tracks alloying reactions and monitors volume expansion and structural degradation [86,96]. (3) For oxide-type anodes, it elucidates redox reaction mechanisms and assesses structural reversibility during cycling [87,97,98,99]. These insights provide essential theoretical support for structural optimization and performance enhancement of battery materials.
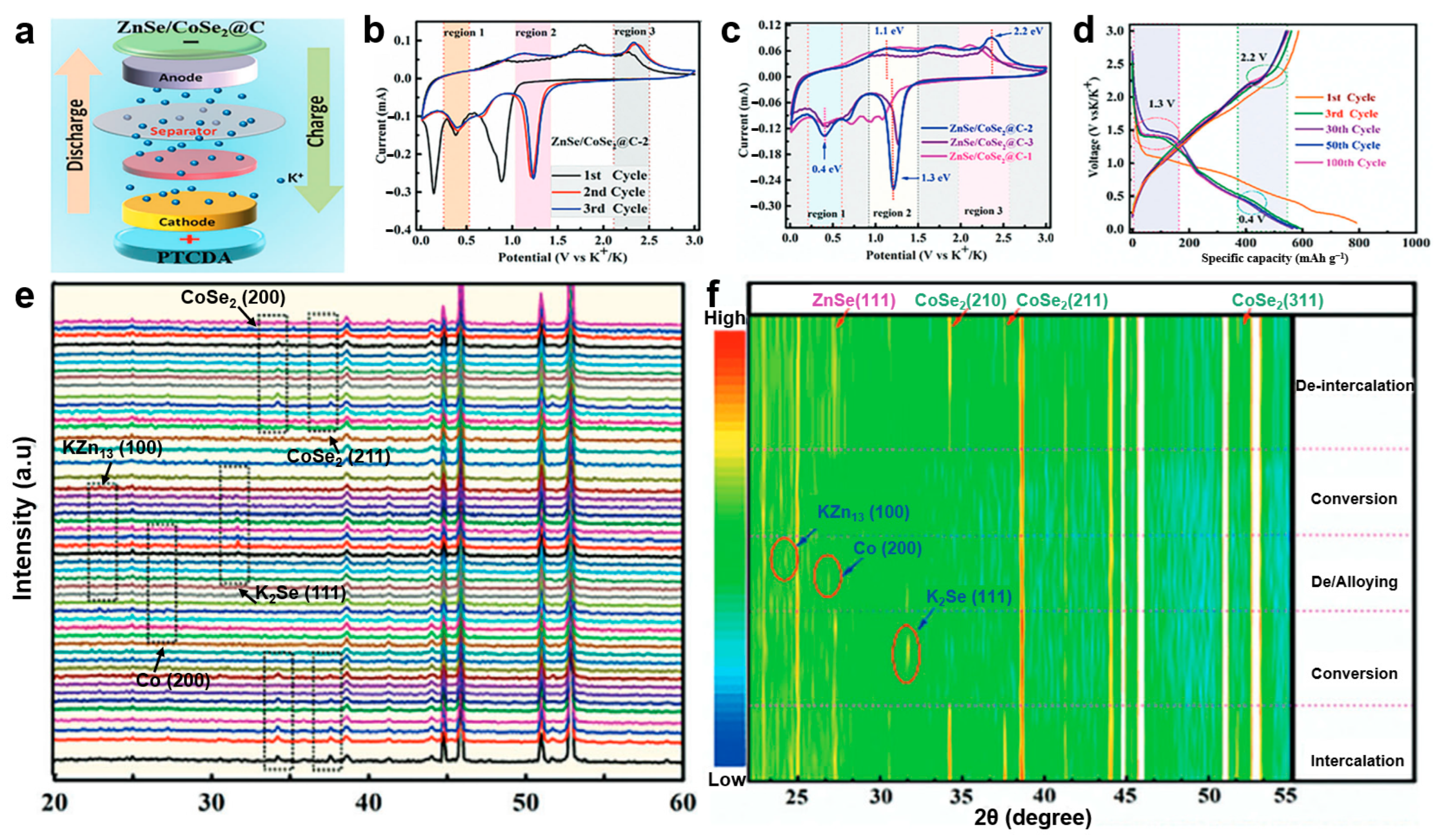
Iqbal et al. [88] synthesized a two-dimensional selenium-rich ZnSe/CoSe2@C heterostructure via hydrothermal and selenization methods as a high-performance anode for potassium-ion batteries (Figure 3a). Based on the CV and charge/discharge curve analysis (Figure 3b–d), the highly reversible potassium-ion insertion storage process in the heterostructure has been confirmed. However, to clarify the specific potassium storage mechanism, in-depth investigation using further characterization techniques such as in situ XRD is necessary. In situ XRD analysis (Figure 3e,f) provides further insight into the potassium storage mechanism. During discharge, the characteristic peaks of ZnSe and CoSe2 remain stable until 1.3 V, then gradually weaken and disappear, indicating the formation of KxZnSe/CoSe2. A strong K2Se peak emerges at 0.8 V, reflecting the high crystallinity enabled by the heterointerface. At 0.01 V, new peaks corresponding to KZn13 and metallic Co confirm the alloying and reduction processes observed in CV. Upon charging, these intermediate phases disappear at 2.2 V, and the original ZnSe and CoSe2 peaks reappear and intensify at 3.0 V, demonstrating the excellent structural reversibility of the heterostructure anode.
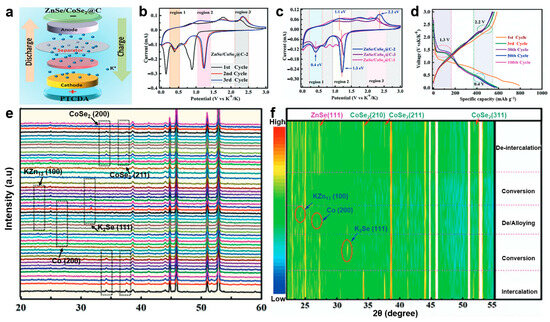
Figure 3.
(a) Schematic model of the ZnSe/CoSe2@C-2||PTCDA full cell; (b) CV at different scan rates; (c) CV curves at a scan rate of 0.1 mV s−1; (d) charge/discharge curves for different cycles; (e,f) in situ XRD patterns of ZnSe/CoSe2@C-2. (Reproduced from [88] with permission from Wiley, 2024).
In the aforementioned study, in situ XRD primarily served to monitor phase transitions in the electrode material during battery operation in real-time. However, its utility extends beyond alloy-type anodes and includes other anode categories, with capabilities surpassing mere phase tracking. A summary of these multifunctional applications is presented in Table 2, suggesting that in situ XRD characterization will play an important role in the study of charging/discharging processes of various batteries.
2.2. In Situ TEM
In situ TEM enables real-time observation of microstructural and interfacial evolution in electrode materials during electrochemical cycling, providing critical insights for designing high-performance anodes. A key challenge lies in accurately replicating the electrode’s operational state within the high-vacuum TEM environment [100]. Two primary approaches exist for environmental control. One is environmental TEM, which employs a differential pumping system to maintain a localized gas environment (Figure 4a). This configuration minimizes gas interference for high-resolution imaging yet is incompatible with liquid electrolytes. Alternatively, specialized in situ sample holders create a sealed electrochemical cell within the holder itself. This widely adopted method offers a low-cost use, compatibility with standard TEM instruments, and flexibility in experimental design.
For battery studies, two kinds of cell designs are commonly used: open-cell and closed-cell configurations. Open-cell setups provide higher spatial resolution and simpler fabrication but poorly simulate realistic battery conditions. In contrast, closed-cell systems enable compatibility with various electrolytes and facilitate nanomaterial observation in liquid environments, though they present greater technical challenges in implementation [89,101].
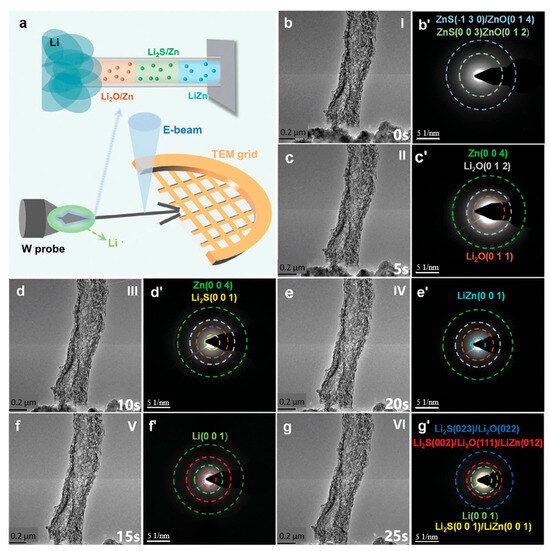
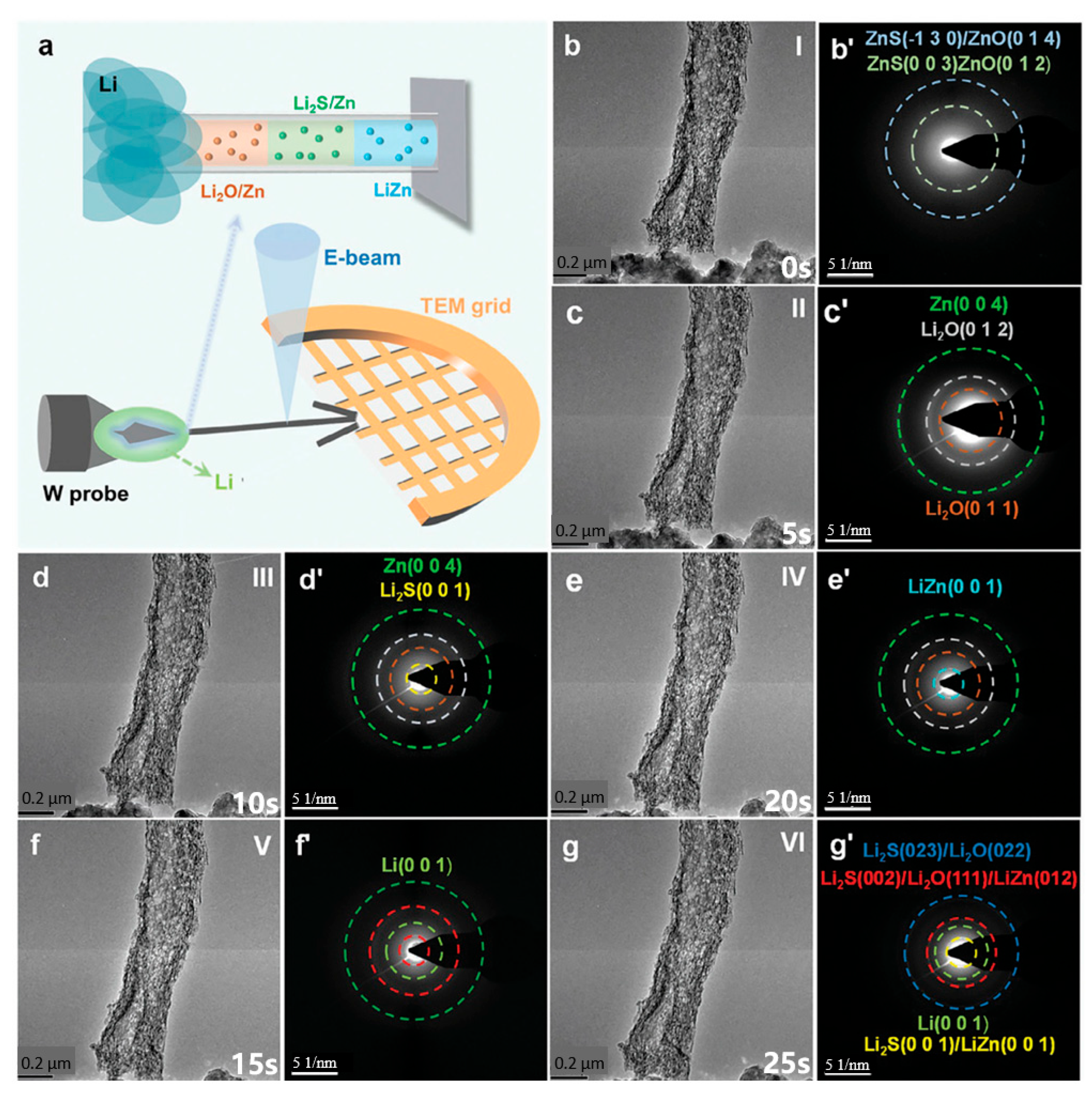

Figure 4.
(a) Schematic illustration of the in situ TEM setup employed for the lithiation of ZOS-CF; (b–g) in situ TEM snapshots and (b’–g’) their corresponding SAED patterns of ZOS-CF captured at different times during the lithiation process. (Reproduced from [102] with permission from Wiley).
Figure 4.
(a) Schematic illustration of the in situ TEM setup employed for the lithiation of ZOS-CF; (b–g) in situ TEM snapshots and (b’–g’) their corresponding SAED patterns of ZOS-CF captured at different times during the lithiation process. (Reproduced from [102] with permission from Wiley).

To gain deeper insights into the spontaneous reaction between ZnO/ZnS nanoparticles and lithium metal, Liu et al. [102] employed in situ TEM to characterize their lithiation process (Figure 4a). Throughout the entire lithiation process (Figure 4a–g), the carbon fibers exhibit no significant dimensional changes, indicating that the three-dimensional carbon-based scaffold can effectively mitigate volume expansion. Figure 4b,b’ presents the original TEM and the selected area electron diffraction (SAED) pattern of ZOS-CF prior to lithiation, respectively (Stage I), where two distinct diffraction rings are attributed to ZnO and ZnS. At the early lithiation stage (Stage II), new diffraction rings corresponding to the Zn phase and Li2O phase emerged in the SAED pattern (Figure 4c’), confirming the spontaneous reaction between lithium and zinc. With progressive lithiation (Stages III→V), the formation of Li2S and LiZn alloy evidences the conversion reaction and alloying reaction of ZnS (Figure 4d’,e’). After full lithiation (Stage VI), the diffraction rings in the SAED pattern (Figure 4g’) verify that ZOS-CF can spontaneously react with lithium metal to generate in situ-formed Li2O/Li2S phases and a LiZn alloy.
In situ TEM has proven effective in monitoring the microstructural evolution of anode materials in real-time, as demonstrated in elucidating the lithiation mechanism of molybdenum disulfide [103]. Moreover, this technique offers broader capabilities in studying other material systems, with specific applications summarized in Table 2.
2.3. In Situ EIS
In situ EIS serves as a powerful tool in anode material studies by monitoring dynamic interfacial changes during cycling. It captures critical parameters such as SEI film impedance and charge transfer resistance evolution. Through analyzing impedance variations at different cycling stages, this technique helps identify the primary causes of electrochemical performance degradation—whether originating from interfacial issues or deteriorated ion diffusion within the bulk material. Furthermore, in situ EIS enables quantitative comparison of interface impedance and charge transfer kinetics before and after material modification, providing direct evaluation of modification effectiveness and component synergy [90]. The acquired insights into SEI formation dynamics also guide the optimization of electrolyte composition, cycling protocols, and electrode fabrication processes, ultimately contributing to enhanced battery performance.
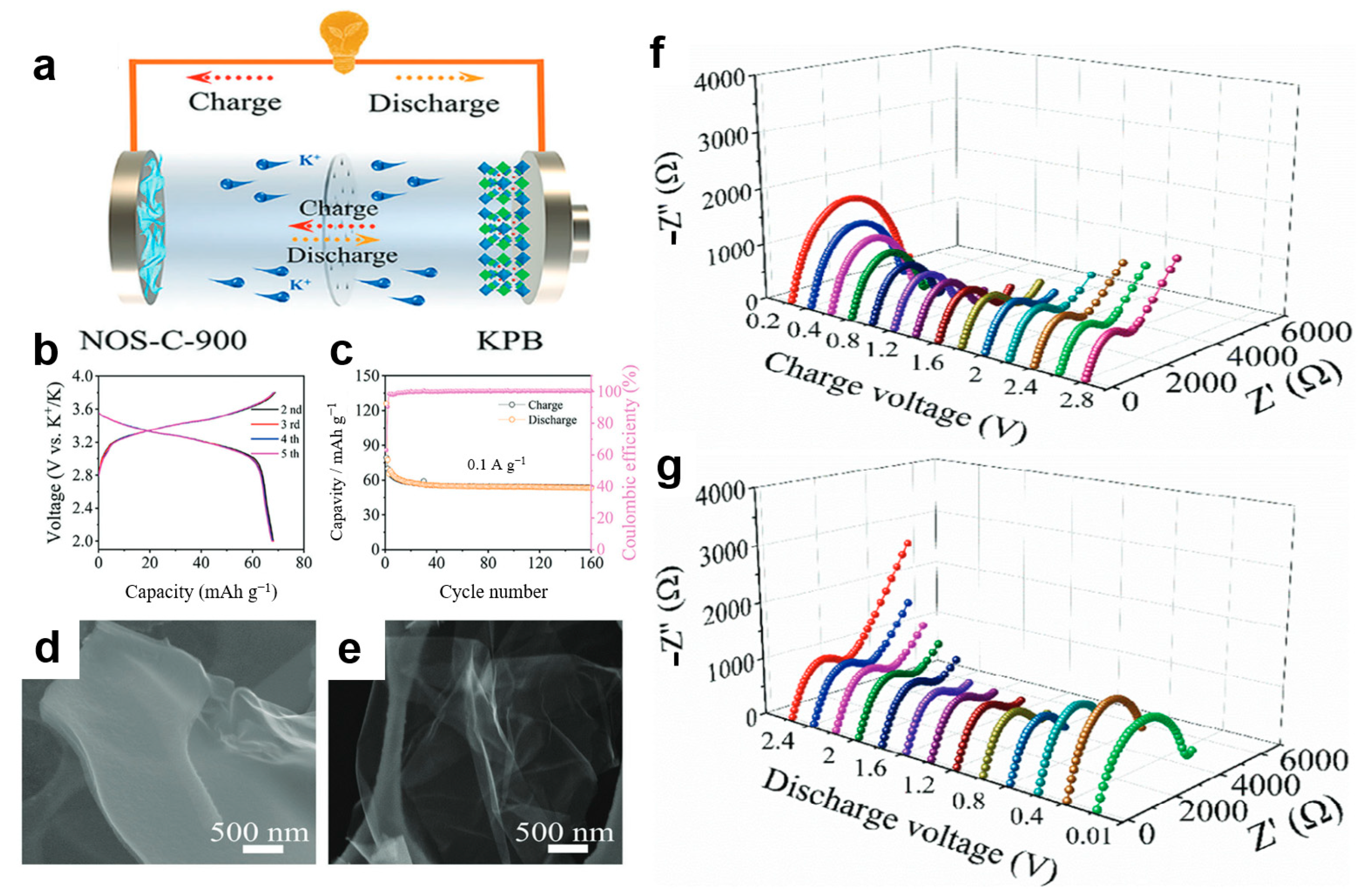
Xu et al. [104] employed in situ EIS to gain deeper insights into the potassium storage mechanism and electrochemical kinetics of the NOS-C-900 anode. The study begins by illustrating the battery configuration schematic (Figure 5a) and the electrode’s microstructure (Figure 5d,e). Figure 5f,g presents the in situ EIS response of the electrode during charge and discharge under a specified current density. The results indicate that as the discharge voltage drops into a lower range, the charge transfer resistance increases significantly, corresponding to the intercalation of a large number of potassium ions into the electrode. Conversely, during the charging process, this resistance shows a clear decreasing trend, indicating effective structural recovery of the electrode after depotassiation and confirming its highly reversible charge–discharge behavior. Further electrochemical performance (Figure 5b,c) further reveals the high reversibility and stability of the anode, providing circumstantial evidence for its excellent electrochemical kinetic performance and supporting the proposed surface–interlayer synergistic potassium storage mechanism.
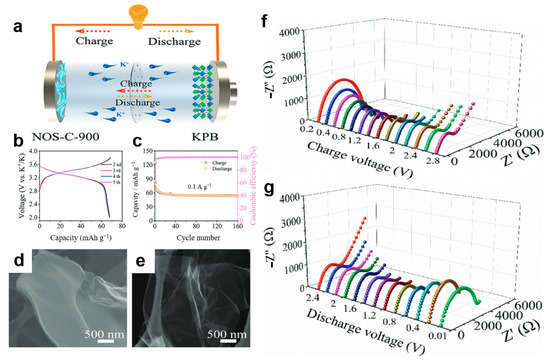
Figure 5.
(a) Cell configuration to coin cell; (b,c) GCD curves and cycle performance of KPB cathodes at 0.1 A g−1; (d,e) SEM images of NOS-C-900 and NOS-C-1000; (f,g) in situ EIS plots of discharge and charge processes of the NOS-C-900 anode at 0.1 A g−1. (Reproduced from [104] with permission from Elsevier).
In situ EIS fulfills distinct functions across different anode categories. For intercalation-type anodes, it primarily monitors SEI film stability and interfacial charge transfer resistance, thereby assisting in optimization of electrode fabrication processes [105]. For alloy-type anodes, it tracks impedance fluctuations induced by volume expansion and ion diffusion barriers, enabling verification of modification strategies [106]. For oxide-type anodes, it focuses on charge transfer resistance evolution during redox reactions and the impedance impact of newly formed interfaces, helping to differentiate the origins of performance degradation [107]. A summary of these multifunctional applications is presented in Table 2.
2.4. In Situ FTIR
In situ FTIR plays a vital role in anode material studies by enabling real-time tracking of functional group evolution during electrochemical processes. This capability provides critical insights into reaction mechanisms, including pathway identification and intermediate detection [108]. The technique effectively monitors characteristic functional group changes during solid electrolyte interphase (SEI) formation, determining its chemical composition and structural evolution across cycling stages to guide SEI optimization [109]. Furthermore, in situ FTIR assesses material stability through characteristic peak variations, detects structural degradation, and identifies side-reaction byproducts along with their triggering conditions, thereby contributing to enhanced battery performance and safety [110].
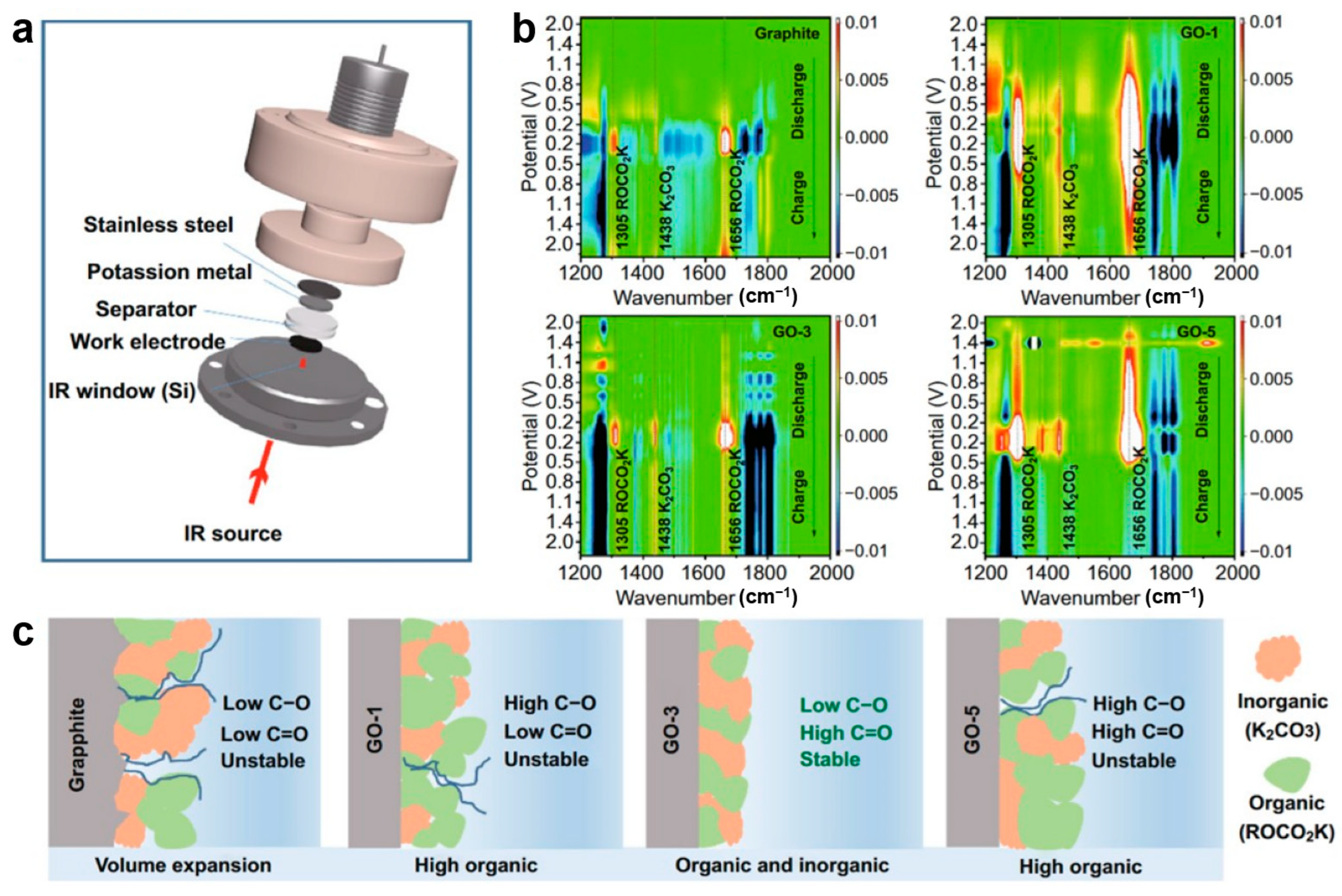
Peng et al. [91] synthesized oxygen-doped graphite anodes for potassium-ion batteries and employed in situ FTIR to investigate how oxygen-containing functional groups enhance potassium storage. Using a specially designed in situ cell with a silicon crystal infrared window and MCT detector (Figure 6a), they characterize the electrode/electrolyte interface evolution of GO and graphite electrodes. The results reveal that oxygen functional groups catalyze SEI formation. On GO electrodes, SEI generation commences at ~2.0 V, evidenced by the decomposition of EC/DEC solvents appearing as new peaks at 1305, 1438, and 1661 cm−1 (Figure 6c), whereas graphite requires much lower potential (~0.8 V, Figure 6b). SEI thickness follows the order GO-1 > GO-5 > GO-3 > graphite, correlating with expansion rates at the 10th charge (12.0%, 6.7%, 5.5% vs. graphite). This trend aligns with improved initial coulombic efficiency. Mechanistically, C–O groups promote ROCO2K formation (most prevalent in GO-1), while C=O and COOH facilitate the generation of K2CO3. The latter contributes to a more stable and ion-conductive SEI. Combined with its relatively thin SEI, GO-3 demonstrates optimal rate capability and cycling stability. Notably, SEI component peaks attenuate but persist after charging, confirming dynamic stability in maintaining electrode integrity.
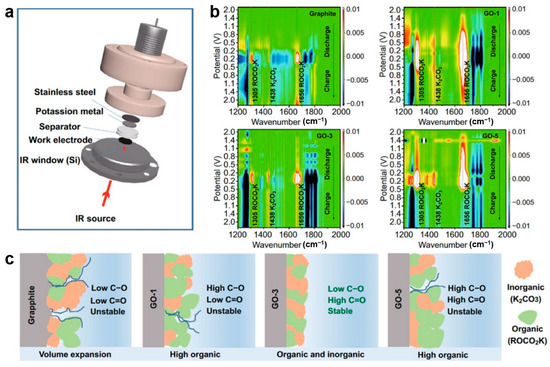
Figure 6.
(a) Schematic representative of the in situ FTIR spectra electrochemistry cell; (b) FTIR spectra of graphite, GO-1, GO-3, GO-5; (c) schematic diagram of the SEI components (reproduced from [91] with permission from Springer).
In situ FTIR provides distinct analytical capabilities for different anode material categories. For intercalation-type materials, it reveals ion intercalation mechanisms through functional group vibration shifts while simultaneously tracking the formation and dynamic evolution of the solid electrolyte interphase (SEI) [111]. For alloy-type materials, it monitors characteristic peak variations during alloying reactions to elucidate reaction pathways while capturing SEI damage and self-repair processes under volume fluctuation [36]. For conversion-type materials, it tracks oxygen-containing functional group transformations during redox processes to analyze reaction mechanisms while characterizing SEI composition and stability to support performance optimization [38]. A summary of these multifunctional applications is presented in Table 2.
2.5. In Situ Raman
In situ Raman spectroscopy plays a crucial role in anode material studies by enabling real-time monitoring of phase transitions, chemical bond evolution, and interfacial reactions under operational conditions [112]. This capability provides direct insight into lithium/sodium storage mechanisms, structural stability during cycling, and failure origins, thereby offering essential experimental evidence for optimizing the composition, structure, and performance of anode materials.
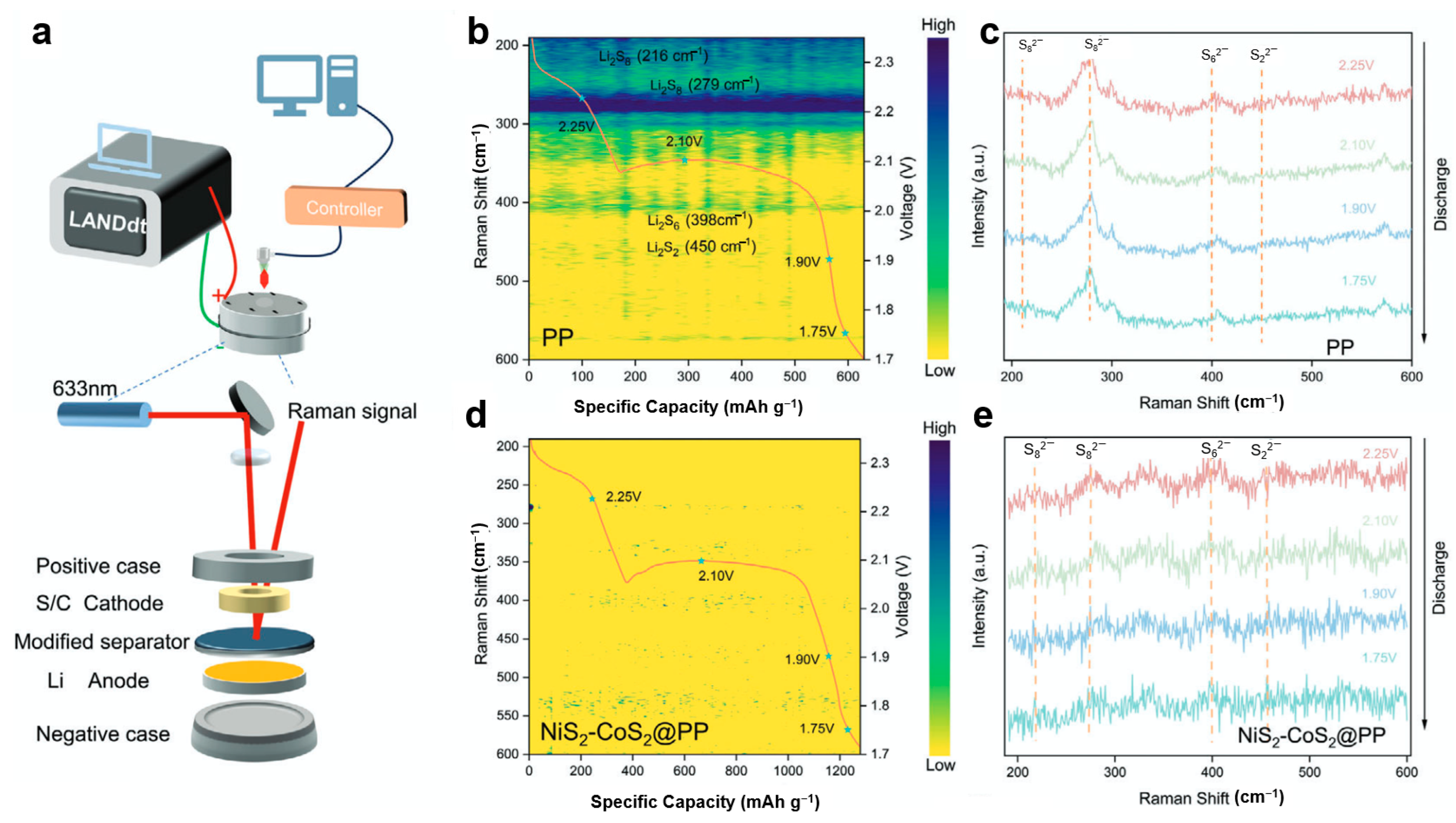
To address the sluggish reaction kinetics in lithium–sulfur batteries, Ye et al. [113] developed a NiS2-CoS2 heterojunction material and employed in situ Raman spectroscopy to investigate its mechanism in suppressing polysulfide shuttling. Using a customized in situ platform integrating a Li-S cell with 633 nm laser Raman spectroscopy and an electrochemical workstation (Figure 7a), the researchers monitored polysulfide evolution under operational conditions. For cells with conventional PP separators (Figure 7b,c), discharge profiles and the corresponding Raman spectra reveal distinct signals at 216 and 279 cm−1 (long-chain Li2S8) during the first plateau. As the discharge progresses to the second plateau (~2.1 V), new peaks emerge at 398 and 450 cm−1 (Li2S6 and Li2S2), indicating significant polysulfide accumulation and shuttle effect. In contrast, cells with NiS2-CoS2@PP separators (Figure 7d,e) show barely detectable polysulfide signals throughout discharge, demonstrating the heterojunction’s effectiveness in capturing soluble polysulfides, accelerating redox kinetics, and preventing their migration across the separator.
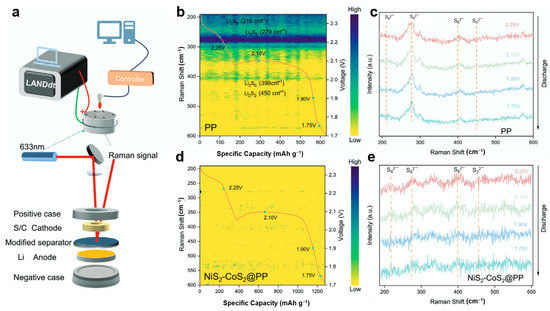
Figure 7.
(a) Schematic diagram of the in situ Raman spectroscopy platform; (b,c) capacity-resolved Raman spectra and discharge profiles of the Li-S battery with PP separator; (d,e) capacity-resolved Raman spectra and discharge profiles of the Li-S battery with NiS2-CoS2@PP separator (reproduced from [113] with permission from Wiley).
In the development of intercalation-type, conversion-type, and alloy-type anode materials, in situ Raman spectroscopy serves the fundamental function of tracking microstructural evolution and chemical reactions in real-time during electrochemical cycling, thereby providing critical insights for performance optimization and failure analysis. Specifically, for intercalation-type anodes, it detects characteristic peak shifts during ion intercalation/deintercalation to assess reaction homogeneity and identify SEI components [114]. For conversion-type anodes, it monitors bond vibration changes to track metal ion redox states and structural phase transitions [115]. For alloy-type anodes, it follows characteristic peak evolution during alloying reactions to investigate structural degradation from volume expansion and cycle performance decay [116]. A summary of these multifunctional applications is shown in Table 2.
2.6. In Situ NMR
In situ NMR provides crucial insights into anode material behavior by directly tracking nuclear spin signal variations in guest ions under operating conditions. This technique enables real-time observation of ion diffusion pathways, site occupancy, and dynamic migration behavior. Simultaneously, it identifies material phase transformations, monitors electrode–electrolyte interfacial evolution, and quantifies ion populations in different states [46]. These capabilities deliver molecular-level evidence for elucidating storage mechanisms, correlating structural degradation with performance decay, and guiding rational material design.
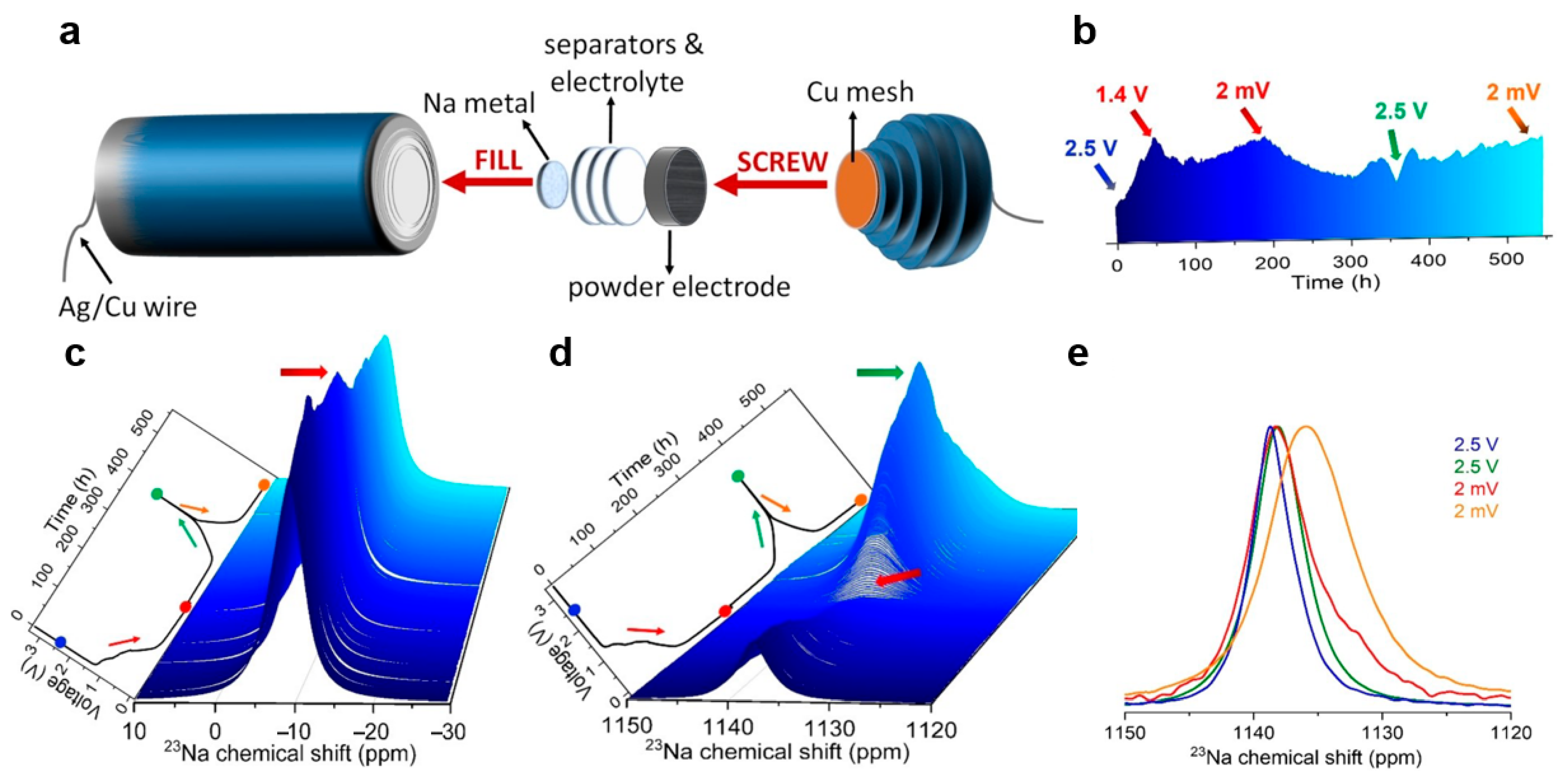
Šić et al. [92] developed an optimized cylindrical cell design for in situ 23Na solid-state NMR studies, particularly suitable for powder electrode materials (Figure 8a). Using hard carbon (HC) anodes and sodium metal reference electrodes, they conducted real-time NMR measurements during electrochemical cycling. Figure 8b,c illustrates the dynamic evolution of sodium ion behavior during cycling. The intensity of the electrolyte sodium ion signal varies progressively with the voltage, reflecting the ions’ reversible migration and indicating continuous interfacial reactions and SEI formation processes. Simultaneously, the corresponding signal changes in the hard carbon region further confirm the reversibility of the sodium storage mechanism within the electrode (Figure 8d,e). In this process, in situ NMR acts like a “real-time microscope,” allowing us to directly observe the dynamic migration of sodium ions during charge and discharge, as well as the ongoing chemical reactions at the electrode interface, thereby confirming the reversibility of the sodium storage mechanism within the battery.
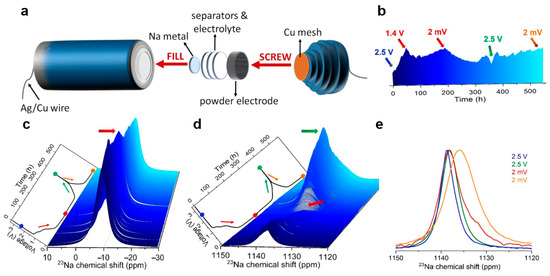
Figure 8.
(a) Schematic representative of Na|NaPF6|HC cell for 23Na in situ NMR studies. (b) Projection of the enlarged 3D view of the 23Na in situ NMR spectra. (c) Enlarged 3D view of the 23Na in situ NMR spectra in the range of 10 to −30 ppm for the Na|NaPF6|HC cell. (d) Enlarged 3D view of the 23Na in situ NMR spectra in the range of 1150 to 1120 ppm. (e) Extracted and normalized 23Na in situ NMR spectra in the range of 1150 to 1120 ppm at the initial charge state of approximately 2.5 V, as well as at the highest voltage of 2.5 V and the lowest voltage of 2 mV (reproduced from [92] with permission from Wiley).
For intercalation-type anodes, it tracks real-time ion (de)intercalation processes, site occupancy, and structural evolution, thereby revealing storage mechanisms and dynamic behavior [117]. For conversion-type anodes, it identifies reaction intermediates and products, clarifying redox pathways and interface transformation [118]. For alloy-type anodes, it monitors phase transitions, compositional changes, and local atomic environments during alloying reactions, helping us to understand reaction mechanisms and address challenges such as volume expansion. Collectively, these capabilities provide fundamental evidence for optimizing performance and elucidating reaction mechanisms across anode material systems. These multifunctional applications are summarized in Table 2.
2.7. In Situ XPS
In situ XPS provides critical insights into anode material behavior by enabling the real-time monitoring of surface and near-surface chemical states during electrochemical cycling. This technique tracks dynamic changes in elemental composition, chemical environment, and valence states, facilitating the investigation of ion intercalation mechanisms, solid electrolyte interphase (SEI) formation and evolution, and interfacial reaction dynamics. These capabilities help establish fundamental correlations between electrochemical performance and surface chemistry, thereby offering essential experimental support for optimizing anode material design and enhancing battery performance [44].
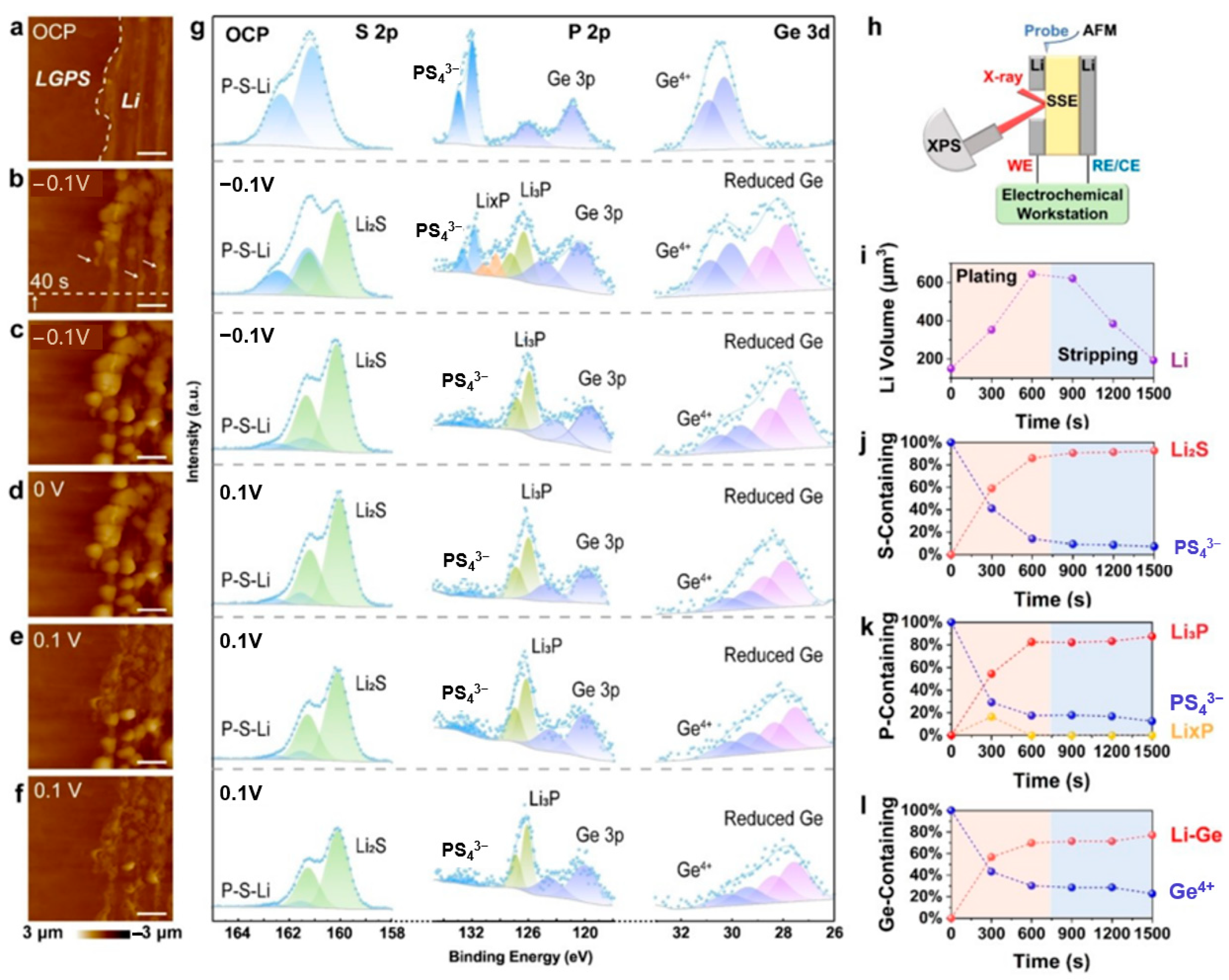
Zhang et al. [119] employed coupled in situ AFM and XPS to investigate the morphological and chemical evolution at the Li-LGPS interface under low bias voltages. Figure 8 illustrates the key observations. At open circuit potential (OCP, Figure 9a), AFM identifies the Li-LGPS boundary while XPS (Figure 9g) primarily detects LGPS components. Upon applying a −0.1 V cathodic overpotential, particle nucleation occurs within 40 s (Figure 9b), evolving into dense Li deposition after 600 s (Figure 9c). Concurrent XPS analysis reveals the formation of a ternary-component SEI during deposition, as indicated by the increasing signals of Li2S and reduced Ge species, alongside the transient appearance of LixP followed by sustained Li3P growth. During subsequent anodic polarization (+0.1 V), Li particles progressively dissolve (Figure 9d,e), leaving residual wrinkles after 600 s (Figure 9f). XPS spectra correlated with these morphological changes confirm the persistence of SEI components (Figure 9i–l). The experimental configuration (Figure 9h) featured a symmetric cell with precisely polished interfaces for simultaneous AFM morphology tracking and XPS chemical analysis. XPS quantification provides critical insights into the dynamic SEI composition. Throughout the deposition and stripping processes, the evolution of components such as Li2S, Li3P, and the Li-Ge alloy is clearly tracked, revealing the interface’s chemical transformation. In particular, XPS analysis demonstrates that the SEI maintains remarkable compositional stability during Li stripping despite the volume decrease, highlighting its interfacial robustness. This multimodal approach, as showcased in Figure 8, successfully correlates deposition kinetics with chemical evolution at the buried solid-state interface.
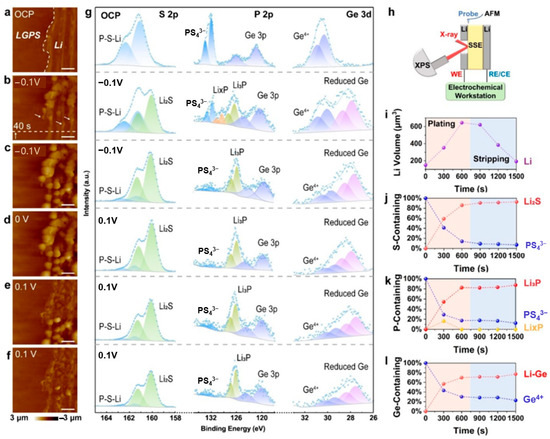
Figure 9.
(a–f) AFM images: (a) Open Circuit Potential (OCP), (b,c) −0.1 V, (d) 0 V, (e,f) 0.1 V, with all scale bars being 4 μm and the scanning direction from bottom to top. (g) In situ XPS spectra of the Li-LGPS interface at different potentials, where the capture time of each AFM image and the collection time of each XPS spectrum are both 300 s. (h) Schematic diagram of the combination of in situ AFM and in situ XPS. (i–l) Changes in Li volume, S-containing species, P-containing species, and Ge-containing species during Li plating and stripping, respectively (reproduced from [119] with permission from Wiley).
For intercalation-type anodes, it dynamically tracks binding energy shifts in host atoms and guest ions during deintercalation/intercalation, identifying insertion sites, elucidating chemical state evolution, and verifying reaction reversibility [120]. For conversion-type anodes, it enables real-time monitoring of transition metal redox behavior and lithium-containing product formation, helping validate conversion mechanisms and analyze irreversibility from incomplete reduction or side reactions [121]. For alloy-type anodes, it captures metal atom binding energy changes during alloying, detects irreversible byproducts, and reveals capacity fading mechanisms associated with irreversible phase transformations, thereby providing direct experimental evidence for optimizing material structure and performance [122]. A summary of these multifunctional applications is displayed in Table 2.
2.8. In Situ SANS
In situ SANS provides unique capabilities for investigating anode materials by exploiting differences in neutron scattering properties between electrolyte and electrode components. This technique quantifies ion concentrations within electrode pores through scattered neutron intensity measurements under varying potentials, enabling detailed analysis of ion adsorption/desorption processes [123]. Furthermore, it monitors ion behavior across different pore sizes, with enhanced sensitivity in smaller pores due to more pronounced scattering variations, offering valuable insights into pore-size-dependent storage mechanisms. However, since scattering signals depend on combined contributions from cations, anions, and solvent molecules, SANS faces inherent challenges in achieving fully quantitative speciation of pore ion populations [124].
Bridges et al. [125] employed in situ SANS to investigate the stabilization mechanism of high-capacity hard carbon during initial cycling in ionic liquid electrolytes (Figure 10a). A comparative analysis of hard carbon electrodes in EMIM·TFSI and MPPY·TFSI electrolytes, via SANS measurements, reveals distinct differences in their interfacial evolution. As shown in Figure 10d–f, the MPPY·TFSI system exhibits a marked increase in peak area during initial discharge, corresponding to electric double layer formation and Li+ desolvation/intercalation processes within specific voltage windows. In contrast, the EMIM·TFSI system shows a similar but weaker response (Figure 10g–i). This divergence is attributed to cation-specific behavior: both EMIM+ and MPPY+ participate in co-intercalation or reduction reactions. Obviously, the MPPY·TFSI system demonstrates more pronounced SEI formation in the first cycle, with its coulombic efficiency rapidly stabilizing at a high level by the fifth cycle (Figure 10b,c). These SANS observations align with previously reported solvated Li+ adsorption behavior in carbonate electrolytes, confirming that variations in scattering length density directly reflect changes in Li+ concentration within the carbon pores. Thus, this study provides nanoscale structural evidence for understanding the interfacial chemical evolution of hard carbon across different ionic liquid environments.
Figure 10.
(a) Schematic view of the operando SANS cell (reproduced from [126] with permission from ACS Publications, 2019). (b,c) Charge−discharge profiles for ex situ half-cells under a current density of 25 μA cm−2. (d–g) In situ SANS data for cycling of half-cells containing ordered mesoporous hard carbon cathodes, 7Li anodes, and 0.5 M LiTFSI/MPPY·TFSI (d–f) or LiTFSI/EMIM·TFSI (g–i) electrolyte. Darker background for discharge regions, lighter background for charged regions; (reproduced from [125] with permission from ACS Publications).
In situ SANS has exceptional sensitivity to light elements and the unique capability of being able to probe buried interfaces, making it particularly valuable for elucidating complex electrochemical mechanisms. By correlating structural parameters with electrochemical performance, in situ SANS enables the fundamental understanding of degradation pathways and facilitates the rational design of advanced electrode architectures with enhanced stability and kinetics. The technique’s non-destructive nature and statistical sampling capabilities further distinguish it from other characterization methods in battery research. A summary of these multifunctional applications is presented in Table 2.
2.9. Application Limitations of In Situ Characterization Techniques
Although individual in situ characterization techniques have played important roles in various battery studies, their application still has some limitations. Table 3 summarizes the current contributions and limitations of various in situ characterization methods. More practical issues still need to be taken into consideration when selecting and employing different in situ characterization techniques for specific applications.

Table 3.
Summary of contributions and limitations of in situ characterization techniques.
In situ characterization techniques exhibit distinct trade-offs in sample preparation complexity, cost, and applicability for alkali metal ion battery anode studies. In situ XRD demands precise control of electrode thickness and low-absorption window materials with moderate preparation costs and low testing expenses, making it ideal for batch dynamic monitoring of crystalline phase transitions. In situ TEM requires advanced micro–nano cell packaging to reconcile vacuum conditions with liquid electrolytes, leading to high preparation and testing costs with relatively low sample success rates and expensive equipment maintenance. In situ EIS features low preparation complexity and cost, as it only requires stable electrode–electrolyte interfaces and leak-free cell packaging, enabling long-term impedance tracking at minimal expense. In situ Raman spectroscopy relies on flat electrode surfaces and light-transmitting windows, with moderate preparation costs and low testing fees, suitable for surface component dynamics. In situ FTIR faces challenges from infrared light penetration limits and brittle window materials, resulting in slightly higher preparation costs than Raman and moderate testing costs, with strict humidity control required during analysis. In situ NMR necessitates non-magnetic cell packaging and high-purity electrolytes to avoid signal distortion under strong magnetic fields, leading to high preparation costs and extremely expensive testing due to the large-scale nature of NMR equipment. In situ XPS requires ultra-high vacuum-compatible cells and an inert atmosphere transfer to prevent electrode oxidation, with high preparation and testing costs stemming from vacuum system maintenance and long pumping times. In situ SANS depends on optimized scattering contrast between electrodes and electrolytes, as well as low-background packaging, with moderate preparation costs but prohibitive testing expenses due to limited access to synchrotron radiation or neutron sources.
To address the current issues, a significant amount of research and technological breakthroughs are still required, such as reducing the manufacturing cost of the testing devices, innovating and developing new materials for key devices, designing and developing in situ testing systems based on new working principles, etc. It is believed that through continuous efforts, costs and technical limitations in the application of in situ characterization technology will be continuously overcome. In the future, in situ characterization techniques will play a more widespread role in the laboratory research and industrial application of Li-, Na-, and K-ion batteries.
2.10. Combined Use of In Situ Characterization Techniques
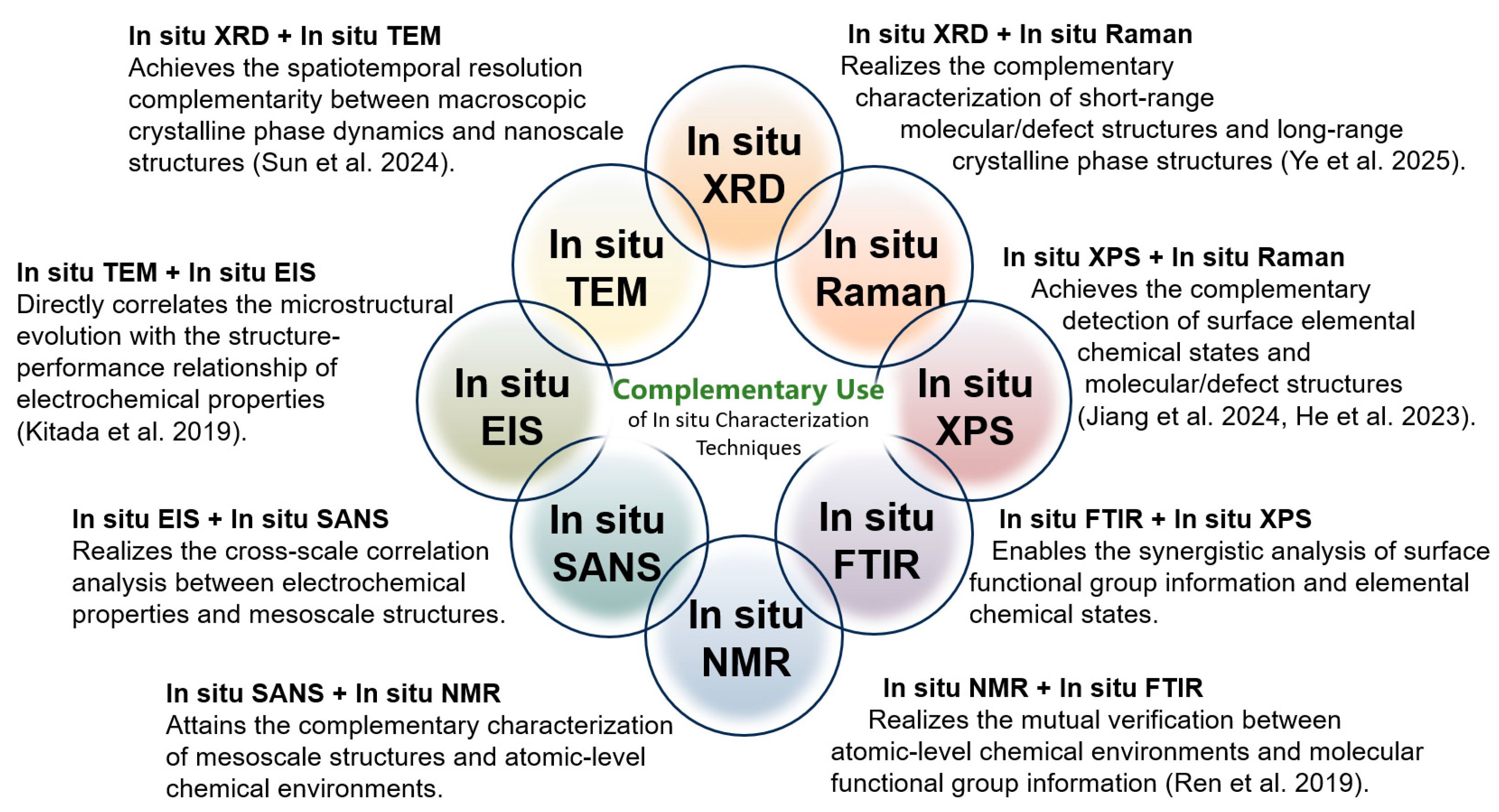
A single in situ characterization technique obtains limited detection information during its use. If we want to fully and comprehensively reveal the various details changes during the battery charging/discharging process from multiple perspectives, the combined use of two in situ characterization techniques is a very effective strategy. Several complementary application schemes for in situ characterization techniques are proposed in Figure 11, some of which have been validated by successful cases in relevant studies, suggesting their important role in revealing the new principles and phenomena of battery reactions.
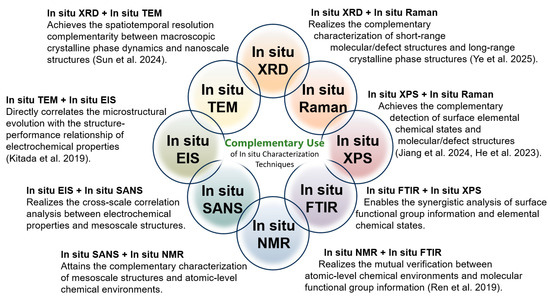
Figure 11.
Combination utilization of different in situ characterization techniques [46,86,113,127,128,129].
In situ XRD + in situ TEM: The core advantage of the combination of in situ XRD and in situ TEM is that it achieves the complementarity of spatiotemporal resolution between macroscopic crystalline phase dynamics and nanoscale structures. With this combined approach, it is possible to simultaneously reflect the dynamic change information of macroscopic crystalline phases and the structural characteristics at the nanoscale, enabling researchers to observe the dynamic processes of material structures across different scales. It is reported that when Cu2S@NSC is used as an anode for sodium-ion batteries, in situ XRD tracks the evolution of the crystal structure during cycling, confirming the reversible conversion reaction between Cu2S, Cu, and Na2S. While in situ TEM directly presents the morphological changes in nanosheets, the sodium insertion process, and the volume expansion buffering effect of the carbon layer, verifying that structural integrity is maintained. This compensates for the lack of macro phase laws or the micro intuitiveness in single characterization, providing comprehensive and rigorous experimental support for explaining the mechanism of the material’s excellent sodium storage performance [110].
In situ XRD + in situ Raman: The advantage of the combination of in situ XRD and in situ Raman lies in its complementary characterization of short-range molecular/defect structures and long-range crystalline phase structures, thus completing the correlation analysis of material structures at different scales. In the study of potassium storage in graphite foam, in situ XRD tracks the long-range crystal structure evolution of K-GICs, confirming reversible stage transitions and identifying the intermediate phase KC16. In situ Raman characterizes short-range charge transfer and local structural changes, revealing the transformation in the middle stage. This process makes up for the limitations of single characterization and offers rigorous support for explaining the potassium storage mechanism [87].
In situ TEM + in situ EIS: When Fe3N@NCNT is used as an anode for Li-ion batteries, in situ TEM directly observes that Fe3N nanoparticles are effectively encapsulated by NCNTs, with the composite structure remaining stable after long-term cycling, while reference samples show particle detachment or pulverization. At the same time, in situ EIS quantifies its electron/ion transport efficiency and stability, confirming excellent electrochemical performance [46]. Their core complementarity lies in the following aspects: TEM directly presents the microscopic phenomenon of “structural stability” and EIS quantifies the electrochemical result of “superior performance”, directly correlating “NCNT encapsulation induced structural stability” with “low impedance and high diffusion efficiency induced excellent cycling/rate performance”. This compensates for the lack of intuitiveness or quantifiability in single characterization, making the demonstration of the material’s advantages more rigorous.
Recently, the combined application of two or more in situ characterization techniques has been increasingly adopted in various battery studies. With the complementary use of various in situ characterization techniques, more and more new discoveries will be revealed during the electrochemical charging and discharging process of batteries. With the continuous technological updates and the reduction in manufacturing costs, this combined in situ characterization strategy will be adopted more widely in the future.
3. Summary and Perspectives
In situ characterization techniques are indispensable for understanding the complex electrochemical processes within rechargeable batteries, providing real-time insights into structural and chemical evolution during operation [130]. This review systematically examines their application in anode materials for a range of alkali metal ion batteries (AMIBs), including Li-, Na-, and K-ion battery systems [131,132,133]. These in situ characterization techniques can facilitate the elucidation of the related electrochemical mechanisms. Individual in situ techniques play distinct roles. In situ XRD tracks crystalline phase transitions, TEM visualizes nanoscale structural evolution, EIS quantifies interfacial impedance, and Raman/FTIR probes chemical bond changes, while NMR, XPS, and SANS complement these methods with insights into ion transport, surface chemistry, and nanoscale morphology. However, each method has limitations. Synergistic use of techniques overcomes single-method drawbacks, enabling comprehensive understanding of anode behavior. These advances facilitate anode optimization, yet future efforts should address extreme condition adaptability and intelligent data analysis to further advance battery technology. For future development, the following perspectives are proposed.
- Advancing characterization capabilities for complex operational scenarios remains a critical challenge. Under extreme battery conditions—such as high voltage (>4.5 V) and wide temperature ranges (−40 °C to 80 °C)—current in situ setups often suffer from limited stability and signal interference. Future development should focus on designing electrochemical cells resistant to high-voltage corrosion, detection systems with minimized thermal drift, and enhanced data correction algorithms. These improvements will mitigate environmental interference and expand the applicability of in situ techniques under realistic operating conditions.
- Advancing intelligent characterization represents a crucial direction for evolution. Current in situ methodologies predominantly rely on manual data processing, leading to inefficiencies and potential oversight of critical information. Future development should integrate artificial intelligence to establish comprehensive data systems capable of real-time characterization, automated data acquisition, intelligent analysis, and mechanistic prediction.
- Developing more robust and versatile in situ cells that are compatible with diverse characterization techniques while accurately mimicking the internal environment of practical batteries is a key direction for obtaining scientifically relevant and industrially applicable insights [96,134].
- There is a critical need to bridge the gap between model materials and practical cells by applying these advanced in situ diagnostics directly to commercially relevant electrodes under realistic cycling conditions, including high mass loading, limited electrolyte, and wide voltage windows.
- Besides its application in the field of anode materials, in situ characterization technology can also play a significant role in other research areas of batteries. The application of in situ characterization techniques in artificial and biomimetic protective coatings will provide real-time insights into the dynamic evolution of coating microstructures, interfacial interactions, and degradation processes under service conditions. This will deepen the understanding of structure–function relationships and guide the rational design of high-performance, long-life protective coatings [135,136].
Author Contributions
Conceptualization, Z.W. and S.M.; formal analysis, J.G. and X.L.; investigation, J.G.; writing—original draft preparation, J.G.; writing—review and editing, Z.W. and S.M.; visualization, J.G. and X.L.; supervision, S.M., X.L. and Z.W.; funding acquisition, Z.W. All authors have read and agreed to the published version of the manuscript.
Funding
The authors would like to acknowledge the financial support from the Tianjin Municipal Science and Technology Program, China (24YDTPJC00140) and the Natural Science Foundation of Hebei Province, China (E2023202253).
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Acknowledgments
During the preparation of this manuscript, the authors used DeepSeek (version V3.2) for English polishing only. The authors declare that AI tools were not used for any content generation. The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Li, L.; Li, T.; Sha, Y.; Ren, B.; Zhang, L.; Zhang, S. A web-like three-dimensional binder for silicon anode in lithium-ion batteries. Energy Environ. Mater. 2024, 7, e12482. [Google Scholar] [CrossRef]
- Zhou, Y.; Pan, W.; Ma, L.; Chen, T.; Sun, Y. Application and challenges of coating technology in cathode materials of lithium ion batteries. China Powder Sci. Technol. 2025, 31, 74–86. [Google Scholar]
- Li, X.; Wang, J.; Han, C.; Zeng, K.; Wu, Z.; Wang, D. Surface engineering of nickel-rich single-crystal layered oxide cathode enables high-capacity and long cycle-life sulfide all-solid-state batteries. Adv. Powder Mater. 2024, 3, 100228. [Google Scholar] [CrossRef]
- Liu, C.; Jia, S.; Yang, T.; Liu, J.; Zhou, X.; Wang, Z.; Dong, H.; Shi, Z.; Zhang, Y.; Chen, Z. Scalable and ultrathin dual entangled network polymer electrolytes for safe solid-state sodium batteries. Angew. Chem. Int. Ed. 2025, 64, e202505938. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wu, Y. Fundamental issues and optimization strategies of solid-state electrolyte-powder electrode interface. China Powder Sci. Technol. 2025, 31, 22–38. [Google Scholar]
- Wang, S.; Li, J.; Xu, L.; Sun, M.; Huang, W.; Liu, Q.; Ren, F.; Sun, Y.; Duan, L.; Ma, H.; et al. Manipulation of Na3V2(PO4)2F3 via aluminum doping to alter local electron states toward an advanced cathode for sodium-ion batteries. Rare Met. 2024, 43, 4253–4262. [Google Scholar] [CrossRef]
- Sun, Z.; Wang, Y.; Yang, T.; Wang, Z.G.; Wang, Z.F.; Shi, Z.; Zhang, Y.; Chen, Z. Constructing low-strain cation storage via high-entropy doping to stabilize layered oxide cathodes for advanced sodium-ion batteries. Small 2025, 21, 2502861. [Google Scholar] [CrossRef]
- Liu, Z.; Feng, H.; Wang, Y.; Wang, F.; Liu, Y.; Yang, J.; Gu, Y.; Tan, J.; Ye, C.; Zhang, C. Nanopore design of sulfur doped hollow carbon nanospheres for superior potassium-ion battery anodes. Rare Met. 2024, 43, 2103–2114. [Google Scholar] [CrossRef]
- Pasanaje, A.; Singh, N. Evolutionary prediction of novel biphenylene networks as an anode material for lithium and potassium-ion batteries. Nano Mater. Sci. 2025, 7, 83–89. [Google Scholar] [CrossRef]
- Vanpeene, V.; King, A.; Maire, E.; Roué, L. In situ characterization of Si-based anodes by coupling synchrotron X-ray tomography and diffraction. Nano Energy 2019, 56, 799–812. [Google Scholar] [CrossRef]
- Kumar, A.; Gowthami, C.; Vijay, R.; Rao, T.N.; Anandan, S. Mini-review on advanced characterization techniques for insights into the sodium storage mechanism of hard carbon anodes: Recent advances and future perspectives. Energy Fuels 2024, 38, 18153–18168. [Google Scholar] [CrossRef]
- Li, S.; Wang, K.; Zhang, G.; Li, S.; Xu, Y.; Zhang, X.; Zhang, X.; Zheng, S.; Sun, X.; Ma, Y. Fast charging anode materials for lithium-ion batteries: Current status and perspectives. Adv. Funct. Mater. 2022, 32, 2200796. [Google Scholar] [CrossRef]
- Bai, Z.; Yao, Q.; Wang, M.; Meng, W.; Dou, S.; Liu, H.K.; Wang, N. Low-temperature sodium-ion batteries: Challenges and progress. Adv. Energy Mater. 2024, 14, 2303788. [Google Scholar] [CrossRef]
- Santos, É.A.; Amaral, M.M.; Damasceno, B.S.; Da Silva, L.M.; Zanin, H.G.; Weker, J.N.; Rodella, C.B. Advanced in situ/operando characterizations of lithium-sulfur batteries: A sine qua non. Nano Energy 2024, 130, 110098. [Google Scholar] [CrossRef]
- Cheng, X.; Li, H.; Zhao, Z.; Wang, Y.; Wang, X. The use of in-situ Raman spectroscopy in investigating carbon materials as anodes of alkali metal-ion batteries. New Carbon Mater. 2021, 36, 93–105. [Google Scholar] [CrossRef]
- Cui, J.; Zhang, J.; Jing, J.; Wang, Y.; Du, G.; Yang, Y.; Yan, L.; Su, Q. Advances in in situ TEM for dynamic studies of carbon-based anodes in alkali metal-ion batteries. Adv. Funct. Mater. 2025, 35, 2418059. [Google Scholar] [CrossRef]
- Cheng, D.; Hong, J.; Lee, D.; Lee, S.; Zheng, H. In situ TEM characterization of battery materials. Chem. Rev. 2025, 125, 1840–1896. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Ke, C.; Gong, Z.; Li, D.; Ci, L.; Zhang, L.; Zhang, Q. Application of in-situ characterization techniques in all-solid-state lithium batteries. Acta Phys. Sin. 2021, 70, 198102. [Google Scholar] [CrossRef]
- Lei, H.; Li, J.; Zhang, X.; Ma, L.; Ji, Z.; Wang, Z.; Pan, L.; Tan, S.; Mai, W. A review of hard carbon anode: Rational design and advanced characterization in potassium ion batteries. InfoMat 2022, 4, e12272. [Google Scholar] [CrossRef]
- Ding, R.; Huang, Y.; Li, G.; Liao, Q.; Wei, T.; Liu, Y.; Huang, Y.; He, H. Carbon anode materials for rechargeable alkali metal ion batteries and in-situ characterization techniques. Front. Chem. 2020, 8, 607504. [Google Scholar] [CrossRef]
- Liu, D.; Shadike, Z.; Lin, R.; Qian, K.; Li, H.; Li, K.; Wang, S.; Yu, Q.; Liu, M.; Ganapathy, S. Review of recent development of in situ/operando characterization techniques for lithium battery research. Adv. Mater. 2019, 31, 1806620. [Google Scholar] [CrossRef]
- Xiang, S.; Zhu, L.; Fu, L.; Wang, M.; Zhang, X.; Tang, Y.; Sun, D.; Wang, H. Cryogenic and in situ characterization techniques for electrode interphase analysis. eScience 2025, 5, 100291. [Google Scholar] [CrossRef]
- Mi, C.; Wang, Z.; Yang, S.; Liu, X.; Wang, Y.; Wang, Z. Porous Al11Ce3 intermetallics as effective sulphur host networks for stable lithium–sulphur batteries. J. Mater. Chem. C 2025, 13, 9014–9026. [Google Scholar] [CrossRef]
- Wang, Z.F.; Wang, H.Y.; Liu, X.L.; Chen, Y.X.; Zhao, Y.; Zhang, Y.G.; Han, Q.Q.; Qin, C.L.; Bakenov, Z.; Wang, Y.C. Single Zn atoms anchored on hollow carbon nanofiber network for dendrite-free lithium metal anode of flexible Li–S full cell. Rare Met. 2023, 42, 3705–3717. [Google Scholar] [CrossRef]
- Yang, W.; Huang, J.; Zheng, Q.; Chen, L.; Orita, A.; Saito, N.; Zhang, Z.; Zhang, Y.; Yang, L. A bandgap-tuned tetragonal perovskite as zero-strain anode for potassium-ion batteries. Angew. Chem. Int. Ed. 2024, 63, e202412706. [Google Scholar] [CrossRef]
- Hsieh, Y.-Y.; Chen, K.-T.; Tuan, H.-Y. A Synergetic SnSb-amorphous carbon composites prepared from polyesterification process as an ultrastable potassium-ion battery anode. Chem. Eng. J. 2021, 420, 130451. [Google Scholar] [CrossRef]
- Chen, M.; Liu, F.-M.; Zhao, M.-Y.; Qian, X.; Yuan, Z.-Y.; Wan, R.; Li, C.-S.; Zhang, X.; Wang, S. Controllable synthesis and growth mechanism of cracked cowpea-like NCNT encapsulated with Fe3N nanoparticles for high-performance anode material in lithium-ion batteries. Carbon 2024, 230, 119579. [Google Scholar] [CrossRef]
- Xu, H.; Li, H.; Gao, M.; Yi, W.; Zhao, Z.; Wang, X. Dual-confinement and oxygen vacancy strategies for ultra-stable sodium storage and mechanisms in high-entropy spinel oxides. J. Energy Storage 2025, 113, 115641. [Google Scholar] [CrossRef]
- Huang, C.; Guo, B.; Wang, X.; Cao, Q.; Zhang, D.; Huang, J.; Jiang, J. Alkali-ion batteries by carbon encapsulation of liquid metal anode. Adv. Mater. 2024, 36, 2309732. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Cheng, Y.; Liu, H.; Zhang, Q.; Petrova, V.; Chen, H.; Liu, P.; Peng, D.-L.; Liu, M.; Wang, M.-S. Hierarchical design of Mn2P nanoparticles embedded in N,P-codoped porous carbon nanosheets enables highly durable lithium storage. ACS Appl. Mater. Int. 2020, 12, 36247–36258. [Google Scholar] [CrossRef]
- Xia, X.; Wang, Q.; Zhu, Q.; Xie, J.; Wang, J.; Zhuang, D.; Zhang, S.; Cao, G.; Zhao, X. Improved Na-storage cycling of amorphous-carbon-sheathed Ni3S2 arrays and investigation by in situ TEM characterization. Mater. Today Energy 2017, 5, 99–106. [Google Scholar] [CrossRef]
- Huang, H.; Wang, J.; Yang, X.; Hu, R.; Liu, J.; Zhang, L.; Zhu, M. Unveiling the advances of nanostructure design for alloy-type potassium-ion battery anodes via in situ TEM. Angew. Chem. Int. Ed. 2020, 59, 14504–14510. [Google Scholar] [CrossRef]
- Zheng, Y.; Xu, Y.; Guo, J.; Li, J.; Shen, J.; Guo, Y.; Bao, X.; Huang, Y.; Zhang, Q.; Xu, J. Cobalt sulfide nanoparticles restricted in 3D hollow cobalt tungstate nitrogen-doped carbon frameworks incubating stable interfaces for Li-ion storage. Electrochim. Acta 2022, 431, 141134. [Google Scholar] [CrossRef]
- Wang, X.; Wang, J.; Wang, Y.; Chen, R.; Qin, J.; Ouyang, T.; Wei, H.; Jing, H.; Yuan, G.; Wang, B. Mechanism influence on the coexistence of two anions on redox kinetics and volume effect of CoPSe as anode material for potassium-ion batteries and potassium-ion hybrid capacitors. Chem. Eng. J. 2024, 499, 156606. [Google Scholar] [CrossRef]
- Nandi, D.K.; Sen, U.K.; Choudhury, D.; Mitra, S.; Sarkar, S.K. Atomic layer deposited molybdenum nitride thin film: A promising anode material for Li ion batteries. ACS Appl. Mater. Int. 2014, 6, 6606–6615. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Lv, L.-P.; Tang, X.; Chen, X.; Sun, W.; Wang, Y. Designing cobalt-based coordination polymers for high-performance sodium and lithium storage: From controllable synthesis to mechanism detection. Mater. Today Energy 2020, 17, 100478. [Google Scholar] [CrossRef]
- Wang, G.; Wang, H.; Ma, G.; Du, X.; Du, L.; Jing, P.; Wang, Y.; Wu, K.; Wu, H.; Wang, Q. Investigation on process mechanism of a novel energy-saving synthesis for high performance Li4Ti5O12 Anode Material. J. Energy Chem. 2022, 70, 266–275. [Google Scholar] [CrossRef]
- Martin, T.R.; Pekarek, R.T.; Coyle, J.E.; Schulze, M.C.; Neale, N.R. Understanding why poly (acrylic acid) works: Decarbonylation and cross-linking provide an ionically conductive passivation layer in silicon anodes. J. Mater. Chem. A 2021, 9, 21929–21938. [Google Scholar] [CrossRef]
- Yang, J.; Dai, Q.; Hou, S.; Han, C.; Zhao, L. Anti-self-discharge capability of Zn-halogen batteries through an entrapment-adsorption-catalysis strategy built upon separator. Adv. Mater. 2025, 37, 2418258. [Google Scholar] [CrossRef]
- Lian, X.; Sun, Z.; Mei, Q.; Yi, Y.; Zhou, J.; Rümmeli, M.H.; Sun, J. Biomass template derived boron/oxygen Co-doped carbon particles as advanced anodes for potassium-ion batteries. Energy Environ. Mater. 2022, 5, 344–352. [Google Scholar] [CrossRef]
- Anji Reddy, M.; Helen, M.; Groß, A.; Fichtner, M.; Euchner, H. Insight into sodium insertion and the storage mechanism in hard carbon. ACS Energy Lett. 2018, 3, 2851–2857. [Google Scholar] [CrossRef]
- Huang, S.; Li, Z.; Wang, B.; Zhang, J.; Peng, Z.; Qi, R.; Wang, J.; Zhao, Y. N-doping and defective nanographitic domain coupled hard carbon nanoshells for high performance lithium/sodium storage. Adv. Funct. Mater. 2018, 28, 1706294. [Google Scholar] [CrossRef]
- Li, H.; Qian, K.; Qin, X.; Liu, D.; Shi, R.; Ran, A.; Han, C.; He, Y.-B.; Kang, F.; Li, B. The different Li/Na ion storage mechanisms of nano Sb2O3 anchored on graphene. J. Power Sources 2018, 385, 114–121. [Google Scholar] [CrossRef]
- Dong, L.; Wang, Z.; Mi, C.; Zhao, W.; Qin, C.; Luo, C.; Wang, Z. Defect-rich hierarchical porous spinel MFe2O4 (M = Ni, Co, Fe, Mn) as high-performance anode for lithium ion batteries. Mater. Today Chem. 2024, 35, 101853. [Google Scholar] [CrossRef]
- Tang, W.; Goh, B.-M.; Hu, M.Y.; Wan, C.; Tian, B.; Deng, X.; Peng, C.; Lin, M.; Hu, J.Z.; Loh, K.P. In situ Raman and nuclear magnetic resonance study of trapped lithium in the solid electrolyte interface of reduced graphene oxide. J. Phys. Chem. C 2016, 120, 2600–2608. [Google Scholar] [CrossRef]
- Kitada, K.; Pecher, O.; Magusin, P.C.M.M.; Groh, M.F.; Weatherup, R.S.; Grey, C.P. Unraveling the reaction mechanisms of SiO anodes for Li-ion batteries by combining in situ7 Li and ex situ7 Li/29 Si solid-state NMR spectroscopy. J. Am. Chem. Soc. 2019, 141, 7014–7027. [Google Scholar] [CrossRef]
- Reynolds, E.M.; Fitzpatrick, J.; Jones, M.O.; Tapia-Ruiz, N.; Playford, H.Y.; Hull, S.; McClelland, I.; Baker, P.J.; Cussen, S.A.; Pérez, G.E. Investigation of sodium insertion in hard carbon with operando small angle neutron scattering. J. Mater. Chem. A 2024, 12, 18469–18475. [Google Scholar] [CrossRef]
- Didier, C.; Gilbert, E.P.; Mata, J.; Peterson, V.K. Direct In situ determination of the surface area and structure of deposited metallic lithium within lithium metal batteries using ultra small and small angle neutron scattering. Adv. Energy Mater. 2023, 13, 2301266. [Google Scholar] [CrossRef]
- Ou, X.; Liang, X.; Zheng, F.; Wu, P.; Pan, Q.; Xiong, X.; Yang, C.; Liu, M. In situ X-ray diffraction investigation of CoSe2 anode for Na-ion storage: Effect of cut-off voltage on cycling stability. Electrochim. Acta 2017, 258, 1387–1396. [Google Scholar] [CrossRef]
- Tan, X.F.; McDonald, S.D.; Gu, Q.; Hu, Y.; Wang, L.; Matsumura, S.; Nishimura, T.; Nogita, K. Characterisation of lithium-ion battery anodes fabricated via in-situ Cu6Sn5 growth on a copper current collector. J. Power Sources 2019, 415, 50–61. [Google Scholar] [CrossRef]
- Cheng, Y.; Xie, H.; Zhou, L.; Shi, B.; Guo, L.; Huang, J. In-Situ liquid-phase transformation of SnS2/CNTs composite from SnO2/CNTs for high performance lithium-ion battery anode. Appl. Surf. Sci. 2021, 566, 150645. [Google Scholar] [CrossRef]
- Liu, N.; Wu, H.; McDowell, M.T.; Yao, Y.; Wang, C.; Cui, Y. A yolk-shell design for stabilized and scalable Li-ion battery alloy anodes. Nano Lett. 2012, 12, 3315–3321. [Google Scholar] [CrossRef]
- Kwon, O.; Kim, T.Y.; Kim, T.; Kang, J.; Jang, S.; Eom, H.; Choi, S.; Shin, J.; Park, J.; Seol, M.L. Intelligent stress-adaptive binder enabled by shear-thickening property for silicon electrodes of lithium-ion batteries. Adv. Energy Mater. 2024, 14, 2304085. [Google Scholar] [CrossRef]
- Zhou, L.; Shen, S.Y.; Peng, X.X.; Wu, L.; Wang, Q.; Shen, C.H.; Tu, T.T.; Huang, L.; Li, J.T.; Sun, S.G. New insights into the structure changes and interface properties of Li3VO4 anode for lithium-ion batteries during the initial cycle by in-situ techniques. ACS Appl. Mater. Interfaces 2016, 8, 23739–23745. [Google Scholar] [CrossRef]
- Sun, Z.; Zhao, Y.; Sun, C.; Ni, Q.; Wang, C.; Jin, H. High entropy spinel-structure oxide for electrochemical application. Chem. Eng. J. 2022, 431, 133448. [Google Scholar] [CrossRef]
- Wang, D.; Jiang, S.; Duan, C.; Mao, J.; Dong, Y.; Dong, K.; Wang, Z.; Luo, S.; Liu, Y.; Qi, X. Spinel-structured high entropy oxide (FeCoNiCrMn)3O4 as anode towards superior lithium storage performance. J. Alloys Compd. 2020, 844, 156158. [Google Scholar] [CrossRef]
- Guo, X.; Xu, H.; Li, W.; Liu, Y.; Shi, Y.; Li, Q.; Pang, H. Embedding atomically dispersed iron sites in nitrogen-doped carbon frameworks-wrapped silicon suboxide for superior lithium storage. Adv. Sci. 2023, 10, 2206084. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.F.; Sun, G.H.; Li, Y.; Cheng, J.Y.; Chen, J.P.; Song, G.; Kong, Q.Q.; Xie, L.J.; Chen, C.M. Pre-oxidation of lignin precursors for hard carbon anode with boosted lithium-ion storage capacity. Carbon 2021, 178, 243–255. [Google Scholar] [CrossRef]
- Qiu, S.; Xiao, L.; Sushko, M.L.; Han, K.S.; Shao, Y.; Yan, M.; Liang, X.; Mai, L.; Feng, J.; Cao, Y. Manipulating adsorption–insertion mechanisms in nanostructured carbon materials for high-efficiency sodium ion storage. Adv. Energy Mater. 2017, 7, 1700403. [Google Scholar] [CrossRef]
- Wu, Y.; Hu, S.; Xu, R.; Wang, J.; Peng, Z.; Zhang, Q.; Yu, Y. Boosting potassium-ion battery performance by encapsulating red phosphorus in free-standing nitrogen-doped porous hollow carbon nanofibers. Nano Lett. 2019, 19, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, M.; Zhu, Z.; Ai, W.; Wu, H.; Wang, B.; He, P.; Xie, D.; Wu, J.; Huang, W. Hard carbon with embedded graphitic nanofibers for fast-charge sodium-ion batteries. Nano Energy 2024, 124, 109459. [Google Scholar] [CrossRef]
- Tout, W.; Mateos, M.; Zhang, J.; Oubla, M.; Emery, N.; Leroy, E.; Dubot, P.; El Moursli, F.C.; Edfouf, Z.; Cuevas, F. Unraveling the energy storage mechanism in nanostructured SnHPO3 anode through advanced operando and ex-situ characterizations. J. Energy Storage 2025, 112, 115502. [Google Scholar] [CrossRef]
- Huang, N.; Zhang, H.; Zhu, G.; Tang, C.; Cheng, Z.; Du, A.; Wang, M.S.; Zhang, H. Yolk-shell structured FeS0.5Se0.5@N-Doped mesoporous carbon composite as high-performance lithium-ion battery anodes. Chem. Eng. J. 2024, 502, 157959. [Google Scholar] [CrossRef]
- Li, Q.; Wang, T.; Guo, B.; Qiao, X.; Meng, X.; Jin, D.; Qiu, H. Design and synthesis of FeS2/graphite sandwich structure with enhanced lithium-storage performance for lithium-ion and solid-state lithium batteries. J. Colloid Interface Sci. 2025, 680, 890–900. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Lei, X.; Chen, W.; Su, Y.; Liu, H.; Ren, S.; Tong, R.; Lin, Y.; Jiang, W.; Liu, X. Si-based all-lithium-reactive high-entropy alloy for thin-film lithium-ion battery anode. Chem. Eng. J. 2024, 500, 157197. [Google Scholar] [CrossRef]
- Shen, H.; Shi, Y.; Bian, W.; Wu, H.; Chen, Y.; Zhao, P.; Kotobuki, M.; Zhang, W.; Chen, X.; Pan, Z. Revisiting the failure mechanism of layered germanium phosphide anode for lithium/sodium-ion batteries: Decisive role of mechanical robustness. J. Power Sources 2025, 630, 236171. [Google Scholar] [CrossRef]
- Wang, C.; Yan, J.; Li, T.; Tang, Y.; Zheng, Q.; Li, X. Single-atom generation inducing electrochemical transformation during cycling in transition metal sulfides for Na-ion batteries. Chem. Eng. J. 2025, 507, 160355. [Google Scholar] [CrossRef]
- Li, Y.; He, K.; Ren, Q.; Zeng, X.; Li, G.; Zhao, S.; Song, W.; He, X.; Wang, S.; Yuan, Y. Mechanically differentiated lithium versus sodium ion storage in an alloying-type bismuth anode. Adv. Funct. Mater. 2024, 34, 2410840. [Google Scholar] [CrossRef]
- Li, Y.; Guo, Y.; You, K.; Guo, C.; Li, C.; Zeng, X.; Wang, W.; Tang, Q.; Yuan, Y. Porous bismuth nanocrystals with advanced sodium ion storage property. Prog. Nat. Sci. Mater. Int. 2023, 33, 92–99. [Google Scholar] [CrossRef]
- Chen, D.; Ye, Z.; Jia, P.; Zhao, Z.; Lin, J.; Wang, X.; Ye, Z.; Li, T.; Zhang, L.; Lu, J. Design of ion channel confined binary metal Cu-Fe selenides for all-climate, high-capacity sodium ion batteries. Small Methods 2024, 8, 2301423. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, H.; Ding, T.; Tian, R.; Sun, D.; Wang, M.-S.; Zhou, X. Synthesis of Yolk-shell Bi2O3@TiO2 submicrospheres with enhanced potassium storage. Sci. China Chem. 2022, 65, 1807–1816. [Google Scholar] [CrossRef]
- Cai, R.; Bao, L.; Zhang, W.; Xia, W.; Sun, C.; Dong, W.; Chang, X.; Hua, Z.; Shao, R.; Fukuda, T. In situ atomic-scale observation of size-dependent (de)potassiation and reversible phase transformation in tetragonal FeSe anodes. InfoMat 2023, 5, e12364. [Google Scholar] [CrossRef]
- Zhang, H.; Li, W.; Pan, J.; Sun, Z.; Xiao, B.; Ye, W.; Ke, C.; Gao, H.; Cheng, Y.; Zhang, Q. Synergistic coupling of amorphous carbon and graphitic domains toward high-rate and long-life K+ storage. J. Energy Chem. 2022, 73, 533–541. [Google Scholar] [CrossRef]
- Li, H.; Ma, Q.; Yuan, Y.; Wang, R.; Wang, Z.; Zhang, Q.; Zhang, L.; Zhu, J.; Zhang, S.; Mao, J. Mesoporous N,S-rich carbon hollow nanospheres controllably prepared from poly (2-aminothiazole) with ultrafast and highly durable potassium storage. Adv. Funct. Mater. 2024, 34, 2301987. [Google Scholar] [CrossRef]
- Yi, Z.; Jiang, S.; Du, Y.; Ma, L.; Qian, Y.; Tian, J.; Jia, C.; Chen, S.; Lin, N.; Qian, Y. Coordinatively and spatially coconfining high-loading atomic Sb in sulfur-rich 2D carbon matrix for fast K+ diffusion and storage. ACS Mater. Lett. 2021, 3, 790–798. [Google Scholar] [CrossRef]
- Cui, J.; Zheng, H.; Zhang, Z.; Hwang, S.; Yang, X.-Q.; He, K. Origin of anomalous high-rate Na-ion electrochemistry in layered bismuth telluride anodes. Matter 2021, 4, 1335–1351. [Google Scholar] [CrossRef]
- Yan, B.; Yu, Y.; Sun, H.; Liu, X.; Li, Y.; Zhang, L.; Yang, X.; Zhong, S.; Wang, R. Flexible potassium-ion batteries enabled by encapsulating hollow NiSe/SnSe nanocubes within freestanding N-Doped carbon nanofibers. Energy Storage Mater. 2025, 74, 103908. [Google Scholar] [CrossRef]
- Li, L.; Huang, A.; Jiang, H.; Li, Y.; Pan, X.; Chen, T.; Chen, H.; Peng, S. Encapsulation of Sn sub-nanoclusters in multichannel carbon matrix for high-performance potassium-ion batteries. Angew. Chem. Int. Ed. 2024, 63, e202412077. [Google Scholar] [CrossRef]
- Song, H.; Xu, C.; Li, R.; Gao, M.; Liu, P.; Wang, F.; Wang, Z.; Deng, J. In Situ catalytic formation of graphite-like carbon with superior potassium storage performance. J. Energy Storage 2024, 101, 113812. [Google Scholar] [CrossRef]
- Chen, Z.; Cheng, L.; Lin, H.; Li, H. Self-hybridization structures of BiOBr0.5Cl0.5: An ultra-high capacity anode material for half/full potassium-ion batteries. J. Colloid Interface Sci. 2025, 677, 769–779. [Google Scholar] [CrossRef] [PubMed]
- Feng, W.; Pan, C.; Wang, H.; Zhang, B.; Luo, W.; Shen, C.; Wang, J.; Cheng, C.; Xv, X.; Yu, R. Molecular carbon skeleton with self−regulating ion−transport channels for long−life potassium ion batteries. Energy Storage Mater. 2023, 63, 102975. [Google Scholar] [CrossRef]
- He, X.; Zhong, L.; Qiu, X.; Wen, F.; Sun, S.; Zu, X.; Zhang, W. Sustainable polyvinyl chloride-derived soft carbon anodes for potassium-ion storage: Electrochemical behaviors and mechanism. ChemSusChem 2023, 16, e202300646. [Google Scholar] [CrossRef]
- Shi, M.; Wang, Y.; He, J.; Zhao, L.; Dai, K.; Yan, C. Multiple redox-active cyano-substituted organic compound integrated with Mxene for high-performance flexible aqueous K-ion battery. Chem. Eng. J. 2022, 450, 138238. [Google Scholar] [CrossRef]
- Cao, M.; Jin, X.; Zhao, J.; Wang, X. Triggering hollow carbon nanotubes via dual doping for fast pseudocapacitive potassium-ion storage. Appl. Mater. Today 2023, 30, 101694. [Google Scholar] [CrossRef]
- Fernando, W.T.R.S.; Amaraweera, T.H.N.G.; Jayathilaka, K.M.D.C.; Kumara, L.S.R.; Seo, O.; Osaka, K.; Sakata, O.; Wijesundera, R.P.; Wijayasinghe, H.W.M.A.C. Engineering electron density and structural properties of CuO anode material via calcination and in situ synchrotron XRD for enhanced lithium-ion battery performance. J. Mater. Sci. 2025, 60, 25386–25408. [Google Scholar] [CrossRef]
- Sun, X.; Qin, C.; Zhao, B.; Jia, S.; Wang, Z.; Yang, T.; Liu, X.; Pan, L.; Zheng, L.; Luo, D. A cation and anion dual-doping strategy in novel Li-Rich Mn-Based cathode materials for high-performance Li metal batteries. Energy Storage Mater. 2024, 70, 103559. [Google Scholar] [CrossRef]
- Liu, J.; Yin, T.; Tian, B.; Zhang, B.; Qian, C.; Wang, Z.; Zhang, L.; Liang, P.; Chen, Z.; Yan, J. Unraveling the potassium storage mechanism in graphite foam. Adv. Energy Mater. 2019, 9, 1900579. [Google Scholar] [CrossRef]
- Iqbal, S.; Wang, L.; Kong, Z.; Zhai, Y.; Wang, F.; Jing, Z.; Sun, X.; Wang, B.; He, X.; Dou, J. 2D Se-rich ZnSe/CoSe2@C heterostructured composite as ultrastable anodes for alkaline-ion batteries. Small 2024, 20, 2404193. [Google Scholar] [CrossRef]
- Hu, Y. α-LiFe5O8: A promising iron-based anode material for lithium-ion batteries. Mater. Sci. Eng. B 2023, 297, 116792. [Google Scholar] [CrossRef]
- Wang, H.; Ding, H.; Wang, Z.; Zhu, Y.; Chen, Z.; Song, B. Heteroatoms-doped mesoporous carbon nanosheets with dual diffusion pathways for highly efficient potassium ion storage. Small 2024, 20, 2310908. [Google Scholar] [CrossRef]
- Peng, Y.; Chen, Z.; Zhang, R.; Zhou, W.; Gao, P.; Wu, J.; Liu, H.; Liu, J.; Hu, A.; Chen, X. Oxygen-containing functional groups regulating the carbon/electrolyte interfacial properties toward enhanced K+ storage. Nano-Micro Lett. 2021, 13, 192. [Google Scholar] [CrossRef]
- Šić, E.; Schutjajew, K.; Haagen, U.; Breitzke, H.; Oschatz, M.; Buntkowsky, G.; Gutmann, T. Electrochemical sodium storage in hard carbon powder electrodes implemented in an improved cell assembly: Insights from in-situ and ex-situ solid-state NMR. ChemSusChem 2024, 17, e202301300. [Google Scholar] [CrossRef]
- Fang, X.; Hu, F.; Lu, J.; Han, X.; Pu, J.; Li, Y.; Yue, C.; Yang, Y. Laser-induced graphene as an effective supporting structure for high performance Ge anode applied in Li-ion batteries. J. Colloid Int. Sci. 2025, 688, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Stevens, D.A.; Dahn, J.R. An in situ small-angle X-ray scattering study of sodium insertion into a nanoporous carbon anode material within an operating electrochemical cell. J. Electrochem. Soc. 2000, 147, 4428. [Google Scholar] [CrossRef]
- Meyer, L.C.; Thiagarajan, A.K.; Koposov, A.; Balducci, A. Unravelling the mechanism of potassium-ion storage into graphite through electrolyte engineering. Energy Storage Mater. 2025, 75, 104021. [Google Scholar] [CrossRef]
- Llewellyn, A.V.; Matruglio, A.; Brett, D.J.L.; Jervis, R.; Shearing, P.R. Using in-situ laboratory and synchrotron-based X-Ray diffraction for lithium-ion batteries characterization: A review on recent developments. Condens. Matter 2020, 5, 75. [Google Scholar] [CrossRef]
- Chang, C.B.; Chen, K.T.; Tuan, H.Y. Large-scale synthesis of few-layered copper antimony sulfide nanosheets as electrode materials for high-rate potassium-ion storage. J. Colloid Interface Sci. 2022, 608, 984–994. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Yu, B.; Ma, W.; Fei, X.; Cheng, G.; Gao, H.; Zhang, Z. Facile preparation of MnO/Mn3O4 anode from commercial manganese(ii) oxalate dihydrate and its advanced lithium storage performance. Electrochim. Acta 2024, 504, 144947. [Google Scholar] [CrossRef]
- Liu, K.; Guo, D.; Zhao, D.; Zhao, P.; Ma, R.; Geng, F.; Zhang, Q.; Zhang, S.; Song, Y.; Sun, J. A simple strategy for the controllable synthesis of Tin-based anode materials and their lithium storage performances. J. Energy Storage 2024, 100, 113600. [Google Scholar] [CrossRef]
- He, Y.; Wang, C.; Lin, R.; Hu, E.; Trask, S.E.; Li, J.; Xin, H.L. A self-healing, flowable, yet solid electrolyte suppresses Li-metal morphological instabilities. Adv. Mater. 2024, 36, 2406315. [Google Scholar] [CrossRef]
- Liu, X.; Wang, Y.; Yang, Y.; Lv, W.; Lian, G.; Golberg, D.; Wang, X.; Zhao, X.; Ding, Y. A MoS2/Carbon hybrid anode for high-performance Li-Ion batteries at low temperature. Nano Energy 2020, 70, 104550. [Google Scholar] [CrossRef]
- Liu, X.; Tan, H.; Li, Y.; Wu, H.; Wu, S.; Yang, L.; Liang, Y.; Xu, G.; Huang, J.; Wang, G. Constructing fast ion/electron conducting pathway within 3D stable scaffold for dendrite-free lithium metal anode. Adv. Funct. Mater. 2025, 35, 2420382. [Google Scholar] [CrossRef]
- Liu, X.H.; Huang, J.Y. In situ TEM electrochemistry of anode materials in lithium ion batteries. Energy Environ. Sci. 2011, 4, 3844. [Google Scholar] [CrossRef]
- Xu, C.; Yang, J.; Chen, K.; Ma, G.; Wang, Y.; Li, Z.; Zhou, Z.; Wu, Z.; Che, S.; Ding, C. CoSe2-modified multidimensional porous carbon frameworks as high-performance anode for fast-charging sodium-ion batteries. Chem. Eng. J. 2024, 497, 154875. [Google Scholar] [CrossRef]
- Chen, L.; Ma, J.; Wang, M.; Dai, W.; Zhou, Y.; Li, G.; Li, Q.; Feng, Y.; Li, J.; Zou, B. Electrochemical dynamic regulation of in-situ copper ion doped CoV2S4 nanohollow cubic boxes for fast electrochemical activation and high efficient sodium ion storage. J. Alloys Compd. 2025, 1018, 179087. [Google Scholar] [CrossRef]
- Ding, X.; Yuan, W.; Fan, Y.; Chen, Y.; Yang, Y.; Liu, L.; Han, L. Tremendously excess Na-Storage capacity of Sb@C anode towards high-performance sodium-ion batteries. Chem. Eng. J. 2025, 505, 159402. [Google Scholar] [CrossRef]
- Xu, K.; Li, Y.H.; Wang, X.; Cao, Y.P.; Wang, S.T.; Cao, L.; Zhang, Q.T.; Wang, Z.F.; Yang, J. Unlocking the structure and anion synergistic modulation of MoSe2 anode for ultra-stable and high-rate sodium-ion storage. Rare Met. 2025, 44, 1661–1673. [Google Scholar] [CrossRef]
- Wang, R.; Wu, Y.; Niu, Y.; Yang, Q.; Li, H.; Chen, T.; Song, Y.; Zhong, B.; Wu, Z. Favored amorphous LixSi process with restrained volume change enabling long cycling quasi-solid-state SiOx anode. ChemSusChem 2025, 18, e202400168. [Google Scholar] [CrossRef]
- Queiroz, B.D.; Vital, P.L.S.; Budke, K.O.; Rey-Raap, N.; Arenillas, A.; Barra, G.M.O.; Ferreira, D.S.; Camara, G.A.; Wender, H.; Martins, C.A. A comprehensive investigation of methanol electrooxidation on copper anodes: Spectroelectrochemical insights and energy conversion in microfluidic fuel cells. ACS Appl. Mater. Interface 2024, 16, 35255–35267. [Google Scholar] [CrossRef] [PubMed]
- Amaral, M.M.; Real, C.G.; Yukuhiro, V.Y.; Doubek, G.; Fernandez, P.S.; Singh, G.; Zanin, H. In situ and operando infrared spectroscopy of battery systems: Progress and opportunities. J. Energy Chem. 2023, 81, 472–491. [Google Scholar] [CrossRef]
- Yang, J.; Solomatin, N.; Kraytsberg, A.; Ein-Eli, Y. In-situ spectro–electrochemical insight revealing distinctive silicon anode solid electrolyte interphase formation in a lithium–ion battery. ChemistrySelect 2016, 1, 572–576. [Google Scholar] [CrossRef]
- Sun, Y.; Liang, S.; Li, Y.; Miao, J.; Liu, S.; Chen, D.; Shao, W.; He, J.; Liu, P. Fiber-based anode for lithium metal battery: Ion deposition behavior, interface stabilization mechanisms, and advanced characterization. J. Alloys Compd. 2025, 1010, 178118. [Google Scholar] [CrossRef]
- Ye, X.; Wu, F.; Xue, Z.; Yuan, H.; Mei, S.; Wang, J.; Yang, R.; Wu, X.; Zhao, X.; Pan, H. Accelerated polysulfide conversion by rationally designed NiS2-CoS2 heterostructure in lithium–sulfur batteries. Adv. Funct. Mater. 2025, 35, 2417776. [Google Scholar] [CrossRef]
- Hu, M.; Key, J.; Zhu, M.; He, H.; Wang, N.; Zhu, Y.; Shen, P.K.; Zhu, J. Two-dimensional nanosheet arrays with chemically bonded Cu2S@NSC heterostructure for efficient sodium-ion storage. J. Energy Storage 2024, 85, 111140. [Google Scholar] [CrossRef]
- Matsuda, Y.; Kuwata, N.; Okawa, T.; Dorai, A.; Kamishima, O.; Kawamura, J. In situ Raman spectroscopy of LiCoO2 cathode in Li/Li3PO4/LiCoO2 all-solid-state thin-film lithium battery. Solid State Ion. 2019, 335, 7–14. [Google Scholar] [CrossRef]
- Hardwick, L.J.; Ruch, P.W.; Hahn, M.; Scheifele, W.; Kötz, R.; Novák, P. In situ Raman spectroscopy of insertion electrodes for lithium-ion batteries and supercapacitors: First cycle effects. J. Phys. Chem. Solids 2008, 69, 1232–1237. [Google Scholar] [CrossRef]
- Yao, X.; Zhao, Z.; Hou, G. Development of in situ MAS NMR and its applications in material synthesis and heterogeneous catalysis. Chin. J. Struct. Chem. 2022, 41, 2210045–2210055. [Google Scholar]
- Gomes, B.F.; Lobo, C.M.S.; Colnago, L.A. Monitoring electrochemical reactions in situ with low field NMR: A mini-review. Appl. Sci. 2019, 9, 498. [Google Scholar] [CrossRef]
- Zhang, X.; Wan, J.; Shen, Z.; Lang, S.; Xin, S.; Wen, R.; Guo, Y.; Wan, L. In Situ analysis of interfacial morphological and chemical evolution in all-solid-state lithium-metal batteries. Angew. Chem. 2024, 136, e202409435. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Y.; Li, Y.; Yang, Y.; Ning, M.; Wu, J.; Zhu, K.; Liu, W. A 2-volt aqueous battery enabled by synergistic effect of Cu2+/Cu+ redox and intercalation/deintercalation in copper hexacyanoferrate cathodes. Energy Storage Mater. 2025, 75, 104088. [Google Scholar] [CrossRef]
- Cui, Y.; Liao, Y.; Sun, Y.; Wang, W.; Wu, J.; Dai, W.; Huang, T. Advanced XPS-based techniques in the characterization of catalytic materials: A mini-review. Catalysts 2024, 14, 595. [Google Scholar] [CrossRef]
- Huo, H.; Jiang, M.; Mogwitz, B.; Sann, J.; Yusim, Y.; Zuo, T.; Moryson, Y.; Minnmann, P.; Richter, F.H.; Veer Singh, C. Interface design enabling stable polymer/thiophosphate electrolyte separators for dendrite-free lithium metal batteries. Angew. Chem. Int. Ed. 2023, 62, e202218044. [Google Scholar] [CrossRef]
- Prehal, C.; Von Mentlen, J.M.; Drvarič Talian, S.; Vizintin, A.; Dominko, R.; Amenitsch, H.; Porcar, L.; Freunberger, S.A.; Wood, V. On the nanoscale structural evolution of solid discharge products in lithium-sulfur batteries using operando scattering. Nat. Commun. 2022, 13, 6326. [Google Scholar] [CrossRef]
- Urbain, F.; Smirnov, V.; Becker, J.P.; Lambertz, A.; Yang, F.; Ziegler, J.; Kaiser, B.; Jaegermann, W.; Rau, U.; Finger, F. Multijunction Si photocathodes with tunable photovoltages from 2.0 V to 2.8 V for light induced water splitting. Energy Environ. Sci. 2016, 9, 145–154. [Google Scholar] [CrossRef]
- Bridges, C.A.; Sun, X.G.; Guo, B.; Heller, W.T.; He, L.; Paranthaman, M.P.; Dai, S. Observing framework expansion of ordered mesoporous hard carbon anodes with ionic liquid electrolytes via in situ small-angle neutron scattering. ACS Energy Lett. 2017, 2, 1698–1704. [Google Scholar] [CrossRef]
- Risse, S.; Härk, E.; Kent, B.; Ballauff, M. Operando analysis of a lithium/sulfur battery by small-angle neutron scattering. ACS Nano 2019, 13, 10233–10241. [Google Scholar] [CrossRef]
- Jiang, J.; Li, M.; Liu, X.; Yi, J.; Jiang, Y.; Wu, C.; Liu, H.; Zhao, B.; Li, W.; Sun, X. Multifunctional additives to realize dendrite-free lithium deposition in carbonate electrolytes toward low-temperature Li metal batteries. Adv. Energy Mater. 2024, 14, 2400365. [Google Scholar] [CrossRef]
- He, X.; Lai, W.; Liang, Y.; Zhao, J.; Yang, Z.; Peng, J.; Liu, X.; Wang, Y.; Qiao, Y.; Li, L. Achieving all-plateau and high-capacity sodium insertion in topological graphitized carbon. Adv. Mater. 2023, 35, 2302613. [Google Scholar] [CrossRef]
- Ren, W.; Wang, Y.; Tan, Q.; Yu, J.; Etim, U.J.; Zhong, Z.; Su, F. Nanosized Si particles with rich surface organic functional groups as high-performance Li-battery anodes. Electrochim. Acta 2019, 320, 134625. [Google Scholar] [CrossRef]
- Bridges, C.A.; Sun, X.G.; Zhao, J.; Paranthaman, M.P.; Dai, S. In situ observation of solid electrolyte interphase formation in ordered mesoporous hard carbon by small-angle neutron scattering. J. Phys. Chem. C 2012, 116, 7701–7711. [Google Scholar] [CrossRef]
- Yang, Q.; Liu, X.; Zhu, J.; Wen, Y.; Zhang, Y.; Chen, G.; Li, K.; Li, Y.; Zhao, Y. Research progress of metal selenides as anode materials for potassium ion batteries: Materials classification, theoretical calculation, and advanced characterization. Chem. Eng. J. 2025, 522, 167105. [Google Scholar] [CrossRef]
- Klvač, O.; Trochta, D.; Novák, L.; Priecel, P.; Bornhöfft, M.; Kazda, T.; Liu, Z. Tracking structural evolution in lithium-ion batteries via operando scanning electron microscopy. Energy Storage Mater. 2025, 81, 104516. [Google Scholar] [CrossRef]
- Li, J.; He, Y.; Zhang, Y.; He, Y.; Dai, Y.; Gao, T.; Zhou, G. Recent advances of cobalt-based bimetallic selenides anodes for high-performance rechargeable alkali metal–ion batteries. Chem. Eng. J. 2025, 505, 159623. [Google Scholar] [CrossRef]
- Peng, H.; Miao, W.; Cui, S.; Liu, Z.; Wang, X.; Tao, B.; Hou, W.; Zhang, Z.; Ma, G. In situ confined growth of Cu2-xSe nanoparticles in highly defective nitrogen-doped carbon for high-rate sodium-ion battery anodes. Chem. Eng. J. 2024, 487, 150701. [Google Scholar] [CrossRef]
- Belmesov, A.A.; Glukhov, A.A.; Kayumov, R.R.; Podlesniy, D.N.; Latkovskaya, E.M.; Repina, M.A.; Ivanov, N.P.; Tsvetkov, M.V.; Shichalin, O.O. Using aquatic plant-derived biochars as carbon materials for the negative electrodes of Li-ion batteries. Coatings 2023, 13, 2075. [Google Scholar] [CrossRef]
- Shichalin, O.O.; Priimak, Z.E.; Seroshtan, A.; Marmaza, P.A.; Ivanov, N.P.; Shurygin, A.V.; Tsygankov, D.K.; Korneikov, R.I.; Efremov, V.V.; Ognev, A.V. Sol–Gel synthesis of carbon-containing Na3V2(PO4)3: Influence of the NASICON Crystal structure on cathode material properties. J. Compos. Sci. 2025, 9, 543. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.