3-([4-(Acetylamino)phenyl]methoxy-1-carbonyl)-7-oxabicyclo[2.2.1]heptane-2-carboxylic Acid
Abstract
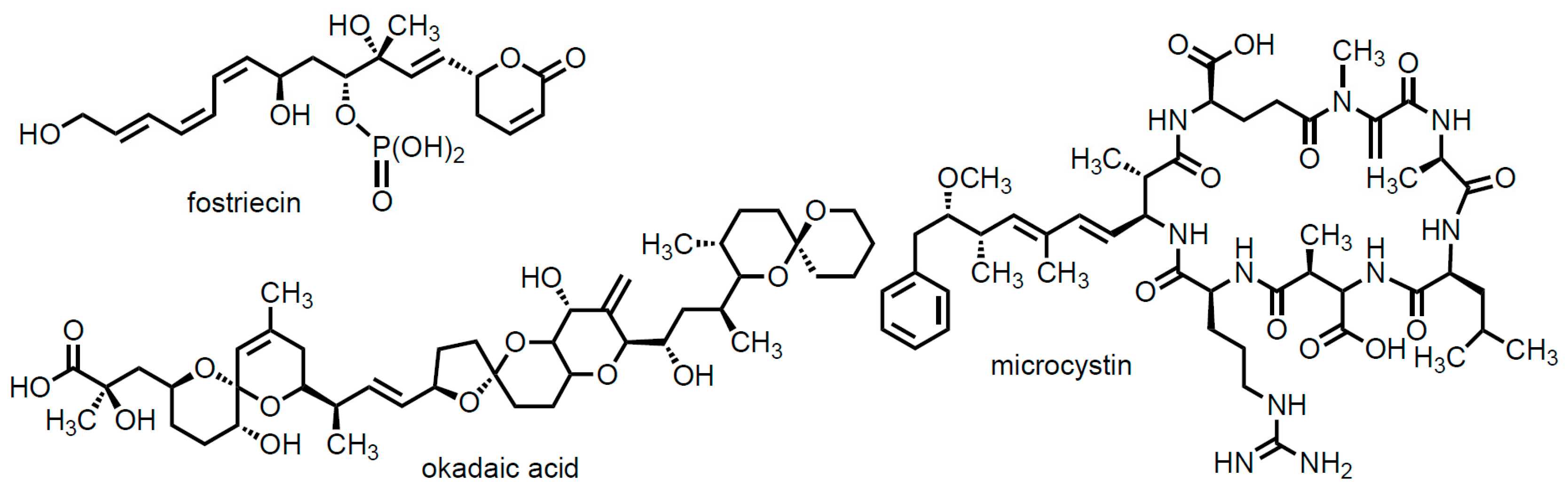
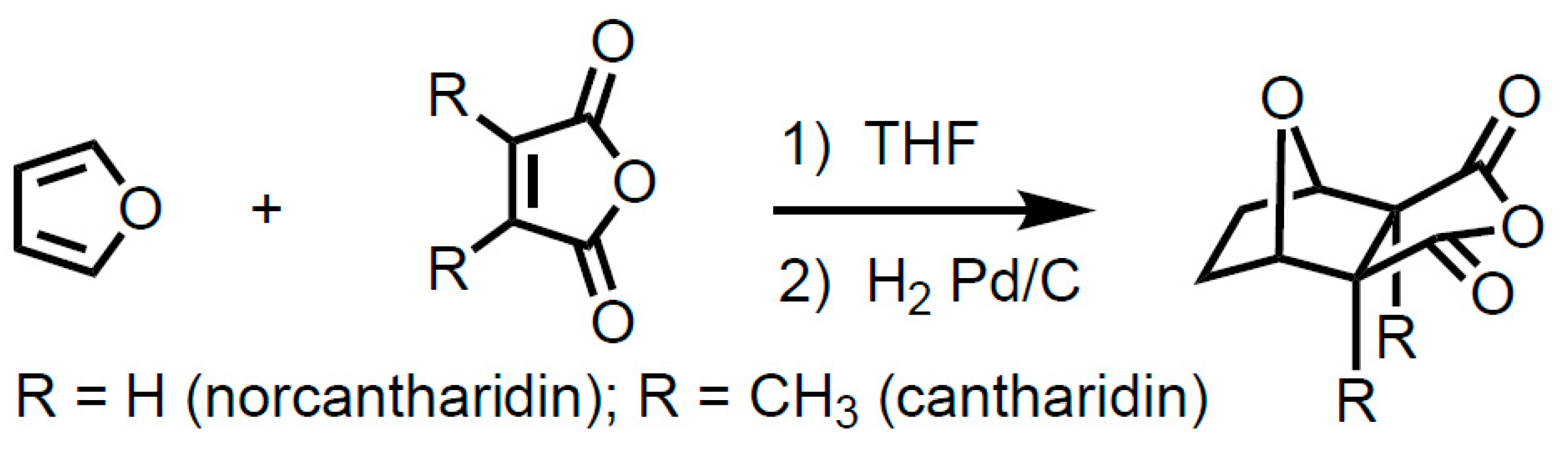
1. Introduction
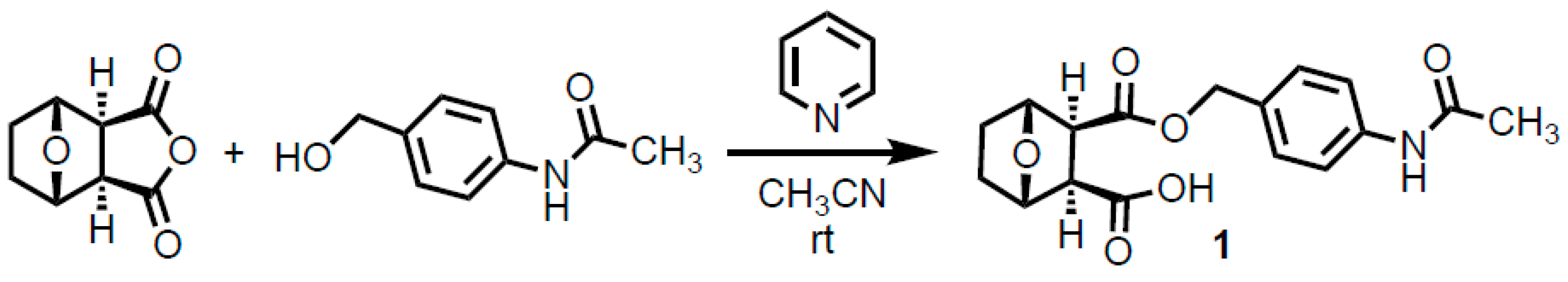
2. Results and Discussion
3. Materials and Methods
4. Experimental
3-([4-(Acetylamino)phenyl]methoxy-1-carbonyl)-7-oxabicyclo[2.2.1]heptane-2-carboxylic Acid (1)
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Golden, T.; Swingle, M.R.; Honkanen, R.E. The role of serine/threonine protein phosphatase type 5 (PP5) in the regulation of stress-induced signaling networks and cancer. Cancer Metastasis Rev. 2008, 27, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-S.; Silverstein, A.M.; Pratt, W.B.; Chinkers, M. The tetratricopeptide repeat domain of protein phosphatase 5 mediates binding to glucocorticoid receptor heterocomplexes and acts as a dominant negative mutant. J. Biol. Chem. 1996, 271, 32315–32320. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Z.; Urban, G.; Scammell, J.G.; Dean, N.M.; McLean, T.K.; Aragon, I.; Honkanen, R.E. Ser/Thr protein phosphatase type 5 (PP5) is a negative regulator of glucocorticoid receptor-mediated growth arrest. Biochemistry 1999, 38, 8849–8857. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Golden, T.; Aragon, I.V.; Honkanen, R.E. Ser/Thr protein phosphatase 5 inactivates hypoxia-induced activation of an apoptosis signal-regulating kinase 1/MKK-4/JNK signaling cascade. J. Biol. Chem. 2004, 279, 46595–46605. [Google Scholar] [CrossRef] [PubMed]
- Swingle, M.R.; Honkanen, R.E. Small Molecule Inhibitors of Ser/Thr Protein Phosphatases: Specificity, Use and Common Forms of Abuse; Methods in Molecular Biology; Springer: Totowa, NJ, USA, 2007; Volume 365, pp. 23–38. [Google Scholar]
- Swingle, M.R.; Honkanen, R.E. Inhibitors of serine/threonine protein phosphatases: Biochemical and structural studies provide insight for further development. Curr. Med. Chem. 2019, 26, 2634–2660. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, D.; Swingle, M.R.; Salter, E.A.; Wood, E.; D’Arcy, B.; Zivanov, C.; Abney, K.; Musiyenko, A.; Rusin, S.F.; Kettenbach, A.; et al. Crystal Structures and Mutagenesis of PPP-Family Ser/Thr Protein Phosphatases Elucidate the Selectivity of Cantharidin and Novel Norcantharidin-Based Inhibitors of PP5C. Biochem. Pharmacol. 2016, 109, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.C.; Mock, A.L.; Nguyen, I.B.; Patel, S.D.; Forbes, D.C. 2-Furanylmethyl N-(2-Propenyl)carbamate. Molbank 2022, 2022, M1510. [Google Scholar] [CrossRef]
- Hill, H.P.; Baker, N.C.; Nguyen, I.B.; Forbes, D.C. Structural Elucidation of Next Generation Inhibitors of Protein Phosphatase 5 Using NMR Spectroscopy. J. Undergrad. Chem. Res. 2022, 21, 19. [Google Scholar]
- Smith, A.; Jones, D. Chemical Stability and Metabolic Resistance of Aromatic Compounds. Bioorg. Med. Chem. 2018, 26, 617–624. [Google Scholar]
- Lee, H.J.; Kim, J.; Park, S. Role of Aromatic Interactions in Drug-Target Binding. Chem. Rev. 2020, 120, 6678–6716. [Google Scholar]
- Brown, L.; Green, R. Functionalization Strategies in Aromatic Scaffolds for Drug Discovery. J. Med. Chem. 2019, 62, 6739–6752. [Google Scholar]
- Taylor, P.; Wilson, K.; Davies, T. Synthetic Methods for Benzene Derivatives in Medicinal Chemistry. Org. Lett. 2017, 19, 2050–2053. [Google Scholar]
- Garcia, M.; Patel, S. Aromatic Rings in Approved Drugs: Trends and Implications. Drug Dev. Res. 2021, 82, 239–248. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mayeaux, K.N.; Baxter, B.N.; Lawley, H.K.; Lopansri, C.N.; Marmande, M.H.; Orr, L.A.; Forbes, D.C. 3-([4-(Acetylamino)phenyl]methoxy-1-carbonyl)-7-oxabicyclo[2.2.1]heptane-2-carboxylic Acid. Molbank 2025, 2025, M2056. https://doi.org/10.3390/M2056
Mayeaux KN, Baxter BN, Lawley HK, Lopansri CN, Marmande MH, Orr LA, Forbes DC. 3-([4-(Acetylamino)phenyl]methoxy-1-carbonyl)-7-oxabicyclo[2.2.1]heptane-2-carboxylic Acid. Molbank. 2025; 2025(3):M2056. https://doi.org/10.3390/M2056
Chicago/Turabian StyleMayeaux, Kathryn N., Bailey N. Baxter, Hannah K. Lawley, Caleb N. Lopansri, Mary Helene Marmande, Lucy A. Orr, and David C. Forbes. 2025. "3-([4-(Acetylamino)phenyl]methoxy-1-carbonyl)-7-oxabicyclo[2.2.1]heptane-2-carboxylic Acid" Molbank 2025, no. 3: M2056. https://doi.org/10.3390/M2056
APA StyleMayeaux, K. N., Baxter, B. N., Lawley, H. K., Lopansri, C. N., Marmande, M. H., Orr, L. A., & Forbes, D. C. (2025). 3-([4-(Acetylamino)phenyl]methoxy-1-carbonyl)-7-oxabicyclo[2.2.1]heptane-2-carboxylic Acid. Molbank, 2025(3), M2056. https://doi.org/10.3390/M2056





