1. Introduction
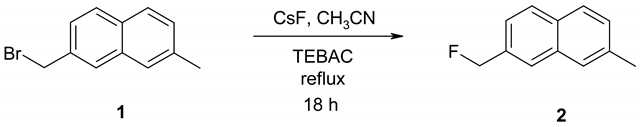
In connection with our investigation of competitive mesolytic cleavages [1] of radical anions we needed 2-fluoromethyl-7-methylnaphthalene, 2, as a gas chromatography standard. Our initial attempt was to prepare 2 via the corresponding known alcohol 2-hydroxymethyl-7-methylnaphthalene [2]. Treatment of the alcohol with the Olah’s reagent (HF/pyridine) [3], according to the procedure of Olah and co-workers for fluorination of alcohols [4] did not result in the desired fluoride. The alternative strategy was to employ halogen exchange using the bromo derivative 1 with a proper source of nucleophilic fluoride. The choice of fluoride source was cesium fluoride, which has appreciable solubility in polar aprotic organic solvents [5], especially when used together with a quaternary ammonium salt [5]. We would like to report a convenient procedure for preparation of 2-fluoromethyl-7-methylnaphthalene.

2. Experimental Section
2.1. General
Melting points were determined using Mel-Temp apparatus and were uncorrected. NMR spectra were recorded on Bruker 400 MHz instrument using deuterated chloroform as solvent and tetramethylsilane as internal standard. The number of hydrogens on each carbon was determined from 13C NMR and 135DEPT spectra. The mass spectra were recorded on Kratos MS-25 RFA double focusing mass spectrometer in chemical ionization (CI) mode. Gas chromatography was performed on a Varian 3700 instrument with packed column. The column was 1/8’’ in diameter and 50 cm in length packed with 5% OV-101 on supelcoport and was purchased from Supelco. The carrier gas was helium (30mL/min flow), the detection was accomplished with flame ionization and monitored with HP- 3390A reporting integrator. Preparative flash chromatography [6] was performed using Merck silica gel 60 (230-400 mesh) and TLC was carried out using Merck pre-coated plates (60 F254, 250 μm).
2.2. 2-Bromomethyl-7-methylnaphthalene (1)
A mixture of 2,7-dimethylnaphthalene [7], (0.981 g, 6.28 mmol), N-bromosuccinimide (1.141 g, 6.42 mmol), benzoyl peroxide (0.038g, 0.16 mmol) in 30 mL of degassed carbon tetrachloride was refluxed under argon for six hours. The mixture was allowed to cool to room temperature then it was cooled on ice and succinimide was removed via filtration. The filtrate was washed with saturated sodium bicarbonate (30 mL), water (2 x 30 mL) and dried over sodium sulfate. The yellow solid that was left behind upon removal of solvent contained 1, unreacted 2,7-dimethylnaphthalene, and 2,7- bis(bromomethyl)naphthalene. The desired product, 2-bromomethyl-7-methylnaphthalene, 1, was isolated by flash chromatography, eluting with 10% methylene chloride in hexanes, as a white solid weighing 0.819 g (55%).
Mp 99-100 °C (lit.[8] 100 °C).
Rf (10% dichloromethane in hexanes) = 0.42.
GC: Rt = 7.93 min (100 °C, 3 min, 8 °C/min to 280 °C).
1H NMR (400 MHz, CDCl3): δ = 7.77 (d, J = 8.4 Hz, 1H), 7.73-7.69 (m, 2H), 7.57 (s, 1H), 7.42 (s, 1H), 7.42 (dd, J = 8.4 Hz, J = 1.7 Hz, 1H), 7.31 (dd, J = 8.4 Hz, J = 1.7 Hz, 1H), 4.65 (s, 2H, Ar-CH2- Br), 2.50 (s, 3 H, Ar-CH3).
13C NMR (100 MHz, CDCl3): δ = 136.6, 135.5, 133.8, 131.7, 129.3, 128.9, 127.9, 127.6, 127.4, 126.3 (Ar), 34.7 (Ar-CH2-Br), 22.1 (Ar-CH3).
2.3. 2-Fluoromethyl-7-methylnaphthalene (2)
A mixture of 2-bromomethyl-7-methylnaphthalene, 1, (0.150 g, 0.605 mmol), dry cesium fluoride (0.396 g, 2.61 mmol), triethylbenzylammonium chloride (TEBAC) (0.003 g, 0.02 mmol) in dry acetonitrile (6 mL) was refluxed under nitrogen for 18 hours. The mixture was cooled to room temperature, filtered and the solid residue was washed with dichloromethane. The crude 2- fluoromethyl-7-methylnaphthalene was dissolved in dichloromethane, 0.3 g of silica gel was added and the solvent was removed. This resulting solid was applied onto an equilibrated silica gel column and eluted with 10% dichloromethane in hexane to yield 0.044 g (42%) of 2 as a white crystalline solid.
Mp 85-87 °C (dec.).
Rf (10 % dichloromethane in hexanes) = 0.34.
GC Rt = 1.46 min (100 °C, 3 min, 8 °C/min to 280 °C).
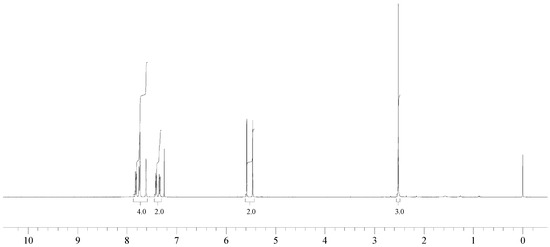
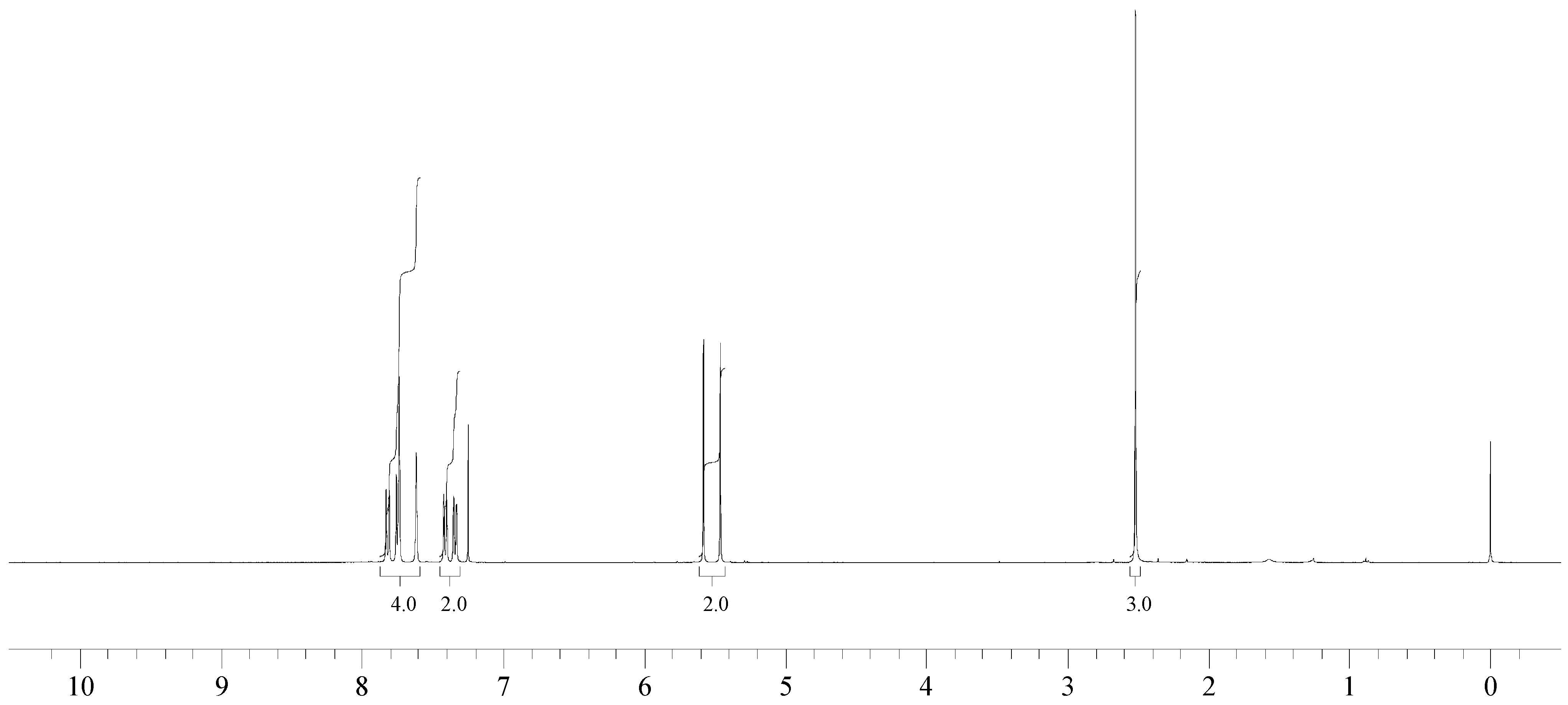
1H NMR (400 MHz, CDCl3): δ = 7.82 (d, 8.4 Hz, 1H), 7.75 (m, 2H), 7.61 (s, 1H), 7.41 (d, 8.4 Hz, 1H), 7.34 (d, 8.4 Hz, 1H), 5.52 (d, 2JH-F = 47.8 Hz, 2H), 2.52 (s, 3H).
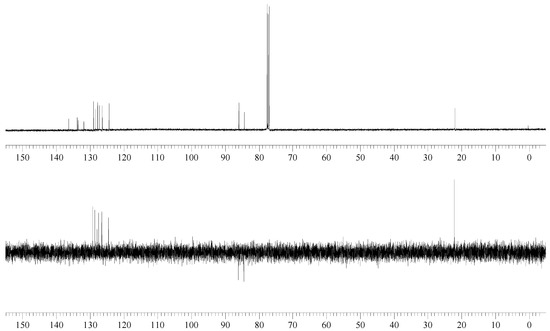
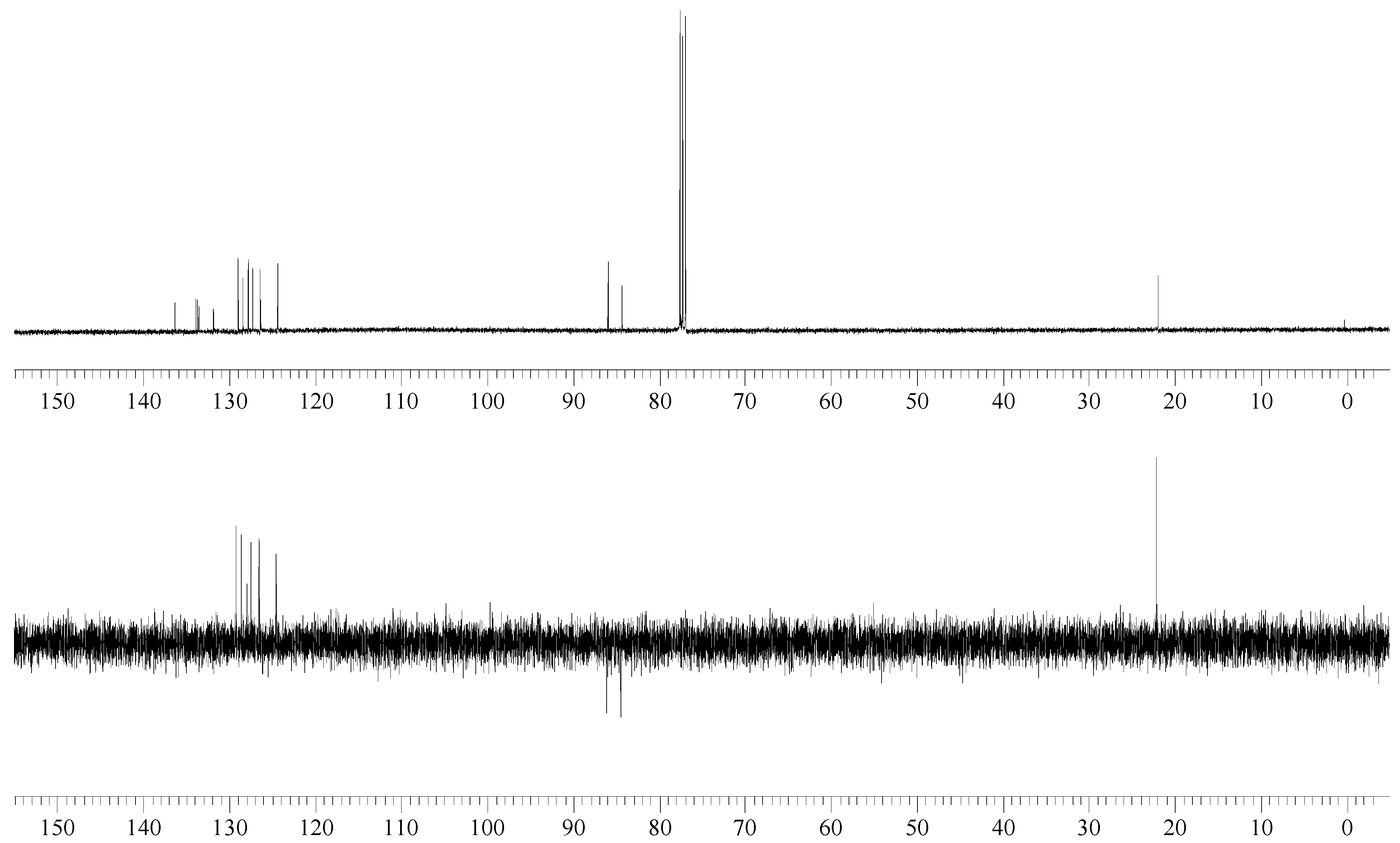
13C NMR (100 MHz): δ = 136.32 (C), 133.83 (d, 2JC-F = 17 Hz, C), 133.60 (C), 131.84 (d, JC-F = 2 Hz, C), 128.99 (d, JC-F = 1 Hz, CH), 128.40 (CH), 127.77 (d, JC-F = 1 Hz, CH), 127.28 (d, JC-F = 1 Hz, CH), 126.36 (d, 3JC-F = 7 Hz, CH), 124.35 (d, JC-F = 5 Hz, CH), (Ar), 85.11 (d, 1JC-F = 165 Hz, Ar-CH2-F), 21.93 (Ar-CH3).
CI-MS (m/z, rel. intensity): 176 (10%), 175 (MH+, 82%), 174 (MH+ - H, 97%), 173 (16%), 159 (16%), 156 (15%), 155, (MH+ - HF, 100%), 141 (11%).
3. Conclusion
The reaction of 2-bromomethyl-7-methylnaphthalene with cesium fluoride in refluxing acetonitrile, in the presence of triethylbenzylammonium chloride gave the desired product 2 as a white crystalline solid. The structure of 2 was unambiguously determined by spectroscopic means (1H NMR, 13C NMR, 135DEPT NMR and MS). The presence of the benzylic fluorine could be seen from the 1H NMR spectrum (doublet centered at 5.52 ppm, arising from two bond hydrogen-fluorine coupling, with coupling constant of 47.8 Hz) and from the 13C NMR spectrum (doublet centered at 85.11 ppm arising from one bond carbon-fluorine coupling with coupling constant of 165 Hz. Both values are in excellent agreement with the literature values for benzylic fluorides [9,10].
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References
- Maslak, P.; Narvaez, J.N. Mesolytic Cleavage of CC Bonds. Com- parison with Homolytic and Heterolytic Processes in the Same Substrate. Angew. Chem. Int. Ed. Engl. 1990, 29, 283–285. [Google Scholar]
- Julia, S.; Julia, M.; Huynh, C. Etude De Cetones Avec Noyau Cyclopropane. 5. Derives Du Methylbenzobicyclo-4.1.0 Heptene. Bull. Soc. Chim. Fr. 1960, 174–178. [Google Scholar]
- Olah, G.A.; Nojima, M.; Kerekes, I. Synthetic Methods and Reactions II1. Hydrofluorination of Alkenes, Cyclopropane and Alkynes with Poly-Hydrogen Fluoride/Pyridine (Trialkylamine) Reagents. Synthesis 1973, 779–780. [Google Scholar] [CrossRef]
- Olah, G.A.; Nojima, M.; Kerekes, I. Synthetic Methods and Reactions IX. Fluorination of Secondary- and Tertiary-Alcohols with Polyhydrogen Fluoride/Pyridine(Trialkylamine) Reagents. Synthesis 1973, 786–787. [Google Scholar] [CrossRef]
- Clark, J.H. Fluoride Ion as a Base in Organic Synthesis. Chem. Rev. 1980, 80, 429–452. [Google Scholar] [CrossRef]
- Still, W.C.; Kahn, M.; Mitra, A. Rapid Chromatographic Technique for Preparative Separations with Moderate Resolution. J. Org. Chem. 1978, 43, 2923–2925. [Google Scholar] [CrossRef]
- Wolinska, J.; Canonne, P.; Leitch, L.C. Synthesis of 2,5,8-Trimethylphenalene. Synthesis 1974, 566–568. [Google Scholar] [CrossRef]
- Buuhoi, N.P.; Lecocq, J. Side-Chain Bromination of Some Alkylnaphthalenes with N-Bromosuccinimide. J. Chem. Soc. 1946, 830–832. [Google Scholar] [CrossRef]
- Silverstein, R.M.; Bassler, G.C.; Morrill, T.C. Spectrometric Identification of Organic Compounds, 5th ed.; Wiley: New York, 1991; p. 281. [Google Scholar]
- Adcock, W.; Abeywickrema, A.N. Conformational Preference of the Fluoromethyl Group in Some Benzyl Fluorides -C-13 NMR-Study. Aust. J. Chem. 1980, 33, 181–187. [Google Scholar] [CrossRef]
© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).