Abstract
Body conformation traits are linked to the health, longevity, reproductivity, and production performance of cattle. These traits are also crucial for herd selection and developing new breeds. This study utilized pedigree information and phenotypic (1185 records) and genomic (The resequencing of 496 Xinjiang Brown cattle generated approximately 74.9 billion reads.) data of Xinjiang Brown cattle to estimate the genetic parameters, perform factor analysis, and conduct a genome-wide association study (GWAS) for these traits. Our results indicated that most traits exhibit moderate to high heritability. The principal factors, which explained 59.12% of the total variance, effectively represented body frame, muscularity, rump, feet and legs, and mammary system traits. Their heritability estimates range from 0.17 to 0.73, with genetic correlations ranging from −0.53 to 0.33. The GWAS identified 102 significant SNPs associated with 12 body conformation traits. A few of the SNPs were located near previously reported genes and quantitative trait loci (QTLs), while others were novel. The key candidate genes such as LCORL, NCAPG, and FAM184B were annotated within 500 Kb upstream and downstream of the significant SNPs. Therefore, factor analysis can be used to simplify multidimensional conformation traits into new variables, thus reducing the computational burden. The identified candidate genes from GWAS can be incorporated into the genomic selection of Xinjiang Brown cattle, enhancing the reliability of breeding programs.
1. Introduction
The breeding industry of Xinjiang Brown cattle accounts for a large proportion of the local economic development as well as farmers’ and herders’ income. In 2023, the population of Xinjiang Brown cattle reached 2 million. The development of Xinjiang Brown cattle, a dual-purpose breed, is challenged by the limited scale of linear type traits evaluation, incomplete genetic evaluation systems, and underutilization of genomic breeding technologies. The Xinjiang conformation evaluation method integrates linear scoring from dairy breeds and specific conformation traits from beef breeds. Most countries employ a 9-point scoring system for conformation traits, with the German Brown cattle system encompassing 25 traits across five categories: body frame, muscularity, rump, feet and legs, and mammary system. The latest Chinese dual-purpose cattle total performance index allocates 10% to conformation traits, highlighting their importance in breeding. Conformation traits, while non-productive, are correlated with economically significant traits, such as milk yield [1], reproductive performance [2], health [3], economic efficiency [4], and longevity [5]. Therefore, selecting for body size traits can accelerate genetic progress in other economic traits. Body size appearance identification is also indispensable for selection and breeding [6], where assessors analyze identification data to determine herd defects and formulate selection and mating plans [7,8].
Genetic parameters of body size traits are important for formulating and implementing breeding plans. There are phenotypic and genetic correlations among body conformation traits [9]. For example, Mazza et al. found strong correlations between rear udder height and width [10], while Wang Dan et al. detected a significant correlation between the body structure traits of Xinjiang Brown cattle [11]. The correlations between body size traits indicate redundant information among traits, and reducing the analysis of highly correlated traits is a concern for breeders because, in breeding selection, the highly correlated traits can be selected indirectly from the remaining highly correlated traits by selecting one of them. Principal component analysis (PCA) and factor analysis can explore relationships among traits by reducing data dimensions with minimal information loss [12]. These methods can transform the linear combination of original variables into composite variables using variable weights obtained from the correlation matrix of the original data [13].
Genetic analysis of complex traits, such as body conformation in cattle, is a challenging problem in animal genetic breeding. Due to its recent advances and affordability, resequencing technology has become essential for identifying the loci associated with complex traits in cattle. A genome-wide association study (GWAS), based on linkage disequilibrium, identifies molecular markers associated with phenotypic variations in complex traits by screening thousands of high-density molecular markers within a population. GWAS has been successfully applied in animal breeding [14,15,16,17,18]. Zhou Jinghang et al. reported 12 SNPs associated with milk production and reproductive performance in Xinjiang Brown cattle in 2019 [19]. Globally, the selection indices in countries such as the USA, Canada, Australia, and Europe include body conformation traits as breeding targets [20]. The genetic evaluation models can be continuously optimized by adding SNPs associated with breeding target traits identified through GWAS. Currently, genomic genetic evaluation for dual-purpose cattle in China is still underdeveloped, and molecular markers related to body size traits need to be investigated.
The evaluation method of body conformation traits of Xinjiang Brown Cattle (dual-purpose breed) is formed by absorbing the linear scoring method of Chinese Holstein cattle (dairy breed), referring to the “code of practice of type classification in Chinese Holstein”, and introducing the special body size evaluation method of meat breeds. However, these details have been scarcely researched. Given the need to explain different biological meanings and avoid redundant information among body conformation traits, this study aims to estimate and compare the genetic parameters of Xinjiang Brown cattle using factor analysis. A GWAS was also conducted on body conformation traits based on whole-genome resequencing data. This research provides a scientific basis for the selection and breeding of Xinjiang Brown cattle.
2. Results
2.1. Descriptive Statistics, Variance Components, and Heritability Estimates of Body Size Traits
Table 1 lists the descriptive statistics, variance components, and heritability estimates of body conformation traits in Xinjiang Brown cattle. The top three body conformation traits with the highest coefficients of variation were rump angle, udder depth, and central ligament, indicating large individual differences in their phenotypic values. The heritability estimates of the body conformation traits ranged from 0.01 to 0.59. Front teat diameter and udder balance showed low heritability, while other body conformation traits exhibited medium to high heritability.

Table 1.
Descriptive statistics, results of variance components and heritability of body conformation traits in Xinjiang Brown cattle.
2.2. Factor Analysis Result
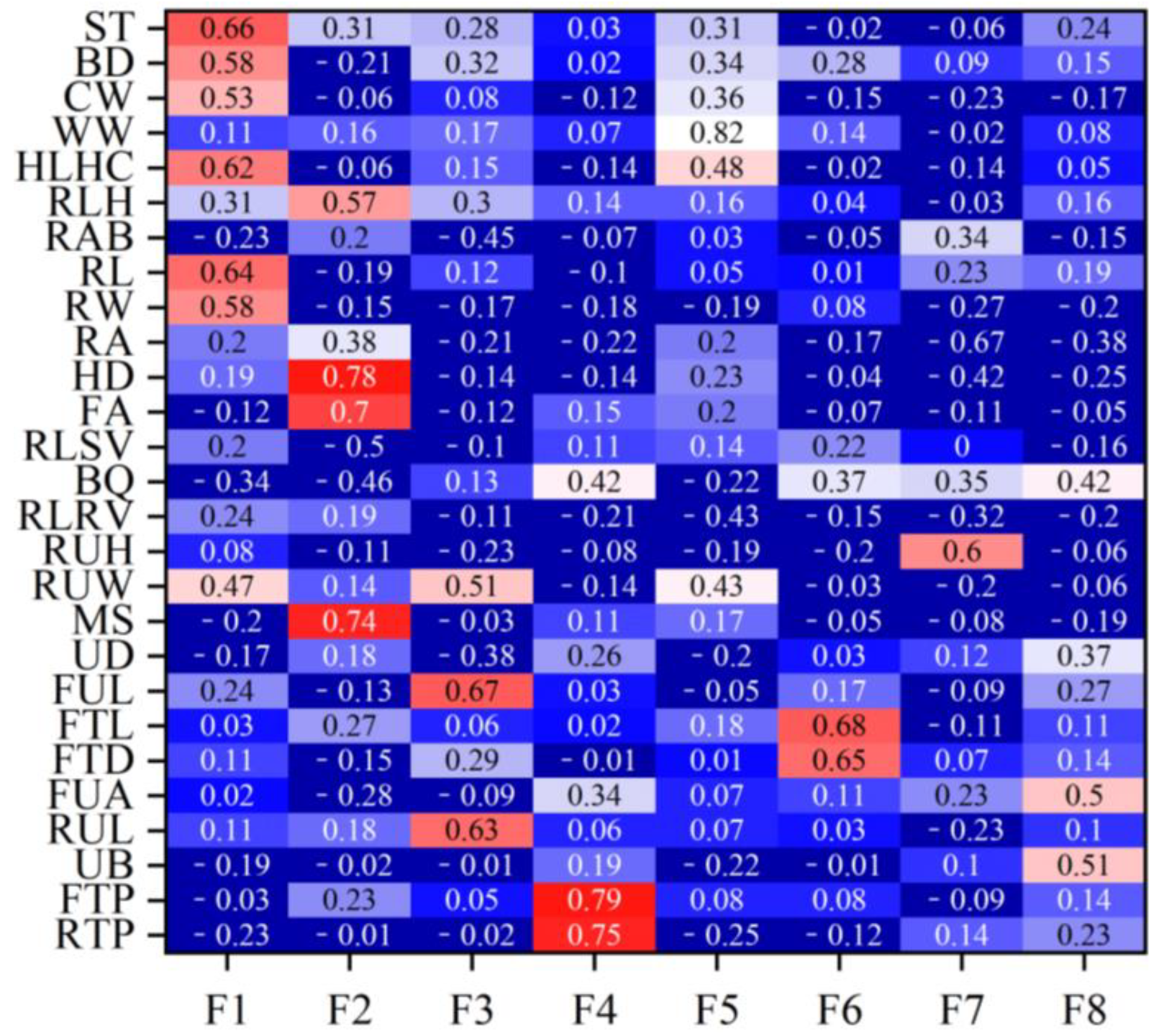
Table 2 lists the eigenvalues and proportions of phenotypic variance explained by each factor in the Xinjiang Brown cattle. Eight latent factors with eigenvalues > 1 explained 59.12% of the information, with the first factor (F1) accounting for the largest proportion of total variance at 16.14%. Figure 1 shows the factor loadings obtained using the varimax rotation method. In F1, rump height (0.69), body depth (0.67), and rump width (0.67) had high loadings, reflecting the body frame and rump information. In F2, foot angle (0.64) and heel depth (0.63) had high loadings, reflecting the limb and hoof information. F3, indicating udder length information, had high loadings for the lengths of the front (0.71) and rear (0.76) udders. F4, reflecting teat position information, had high loadings for the front (0.82) and rear (0.81) teat positions. F5 mainly reflected muscle development information, with loadings of 0.62 and 0.85 for rear leg girth and withers width, respectively. F6, mainly reflecting teat size information, had high loadings for front teat diameter (0.76) and front teat length (0.81). F7 and F8 mainly reflected udder traits.

Table 2.
Eigenvalues, proportional variance, and cumulative variance explained by factor analysis of the phenotypic values of body conformation traits in Xinjiang Brown cattle.
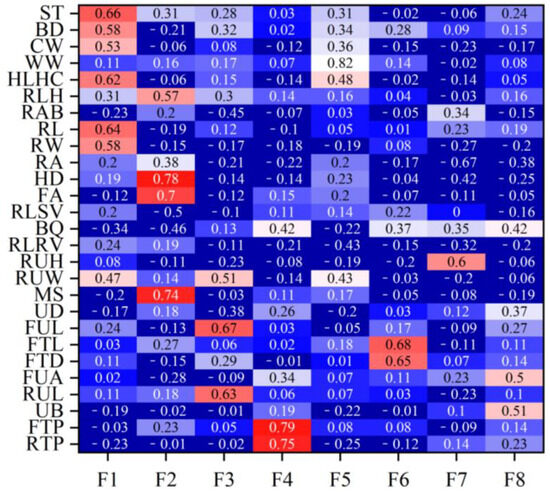
Figure 1.
Rank correlation coefficients between the estimated breeding values for each body conformation trait and the estimated breeding values for factor scores for Xinjiang Brown cattle.Note: ST is stature; BD is body depth; CW is chest width; WW is wither width; HLHC is half of leg circumstance; RLH is rear leg height; RAB is rib and bone; RL is rump length; RA is rump angle; HD is heel depth; FA is feet angle; RLSV is rear leg side view; RUH is rear udder height; RUW is rear udder width; MS is medium; UD is udder depth; FUL is fore udder length; FTL is fore teat length; FTD is fore teat diameter; FUA is fore udder attachment; RUL is rear udder length; UB is udder balance; FTP is fore teat placement; RTP is rear teat placement; DC is dairy character; and LS is loin strength. Red means that the correlation coefficient is greater than 0.3, and the darker the color, the greater the correlation coefficient; blue means that the correlation coefficient is less than 0.3, and the darker the color, the smaller the correlation coefficient.
2.3. Heritability and Genetic Correlation of Factor Scores
Table 3 shows the variance component and heritability estimate for all eight factors in Xinjiang Brown cattle, with the latter ranging from 0.17 (F6) to 0.73 (F2), all being medium to high heritability traits ( > 0.1).

Table 3.
Results of variance components and heritability estimates for each factor in Xinjiang Brown cattle.
2.4. Rank Correlation between Estimated Breeding Values of Body Conformation Traits and Factor Scores
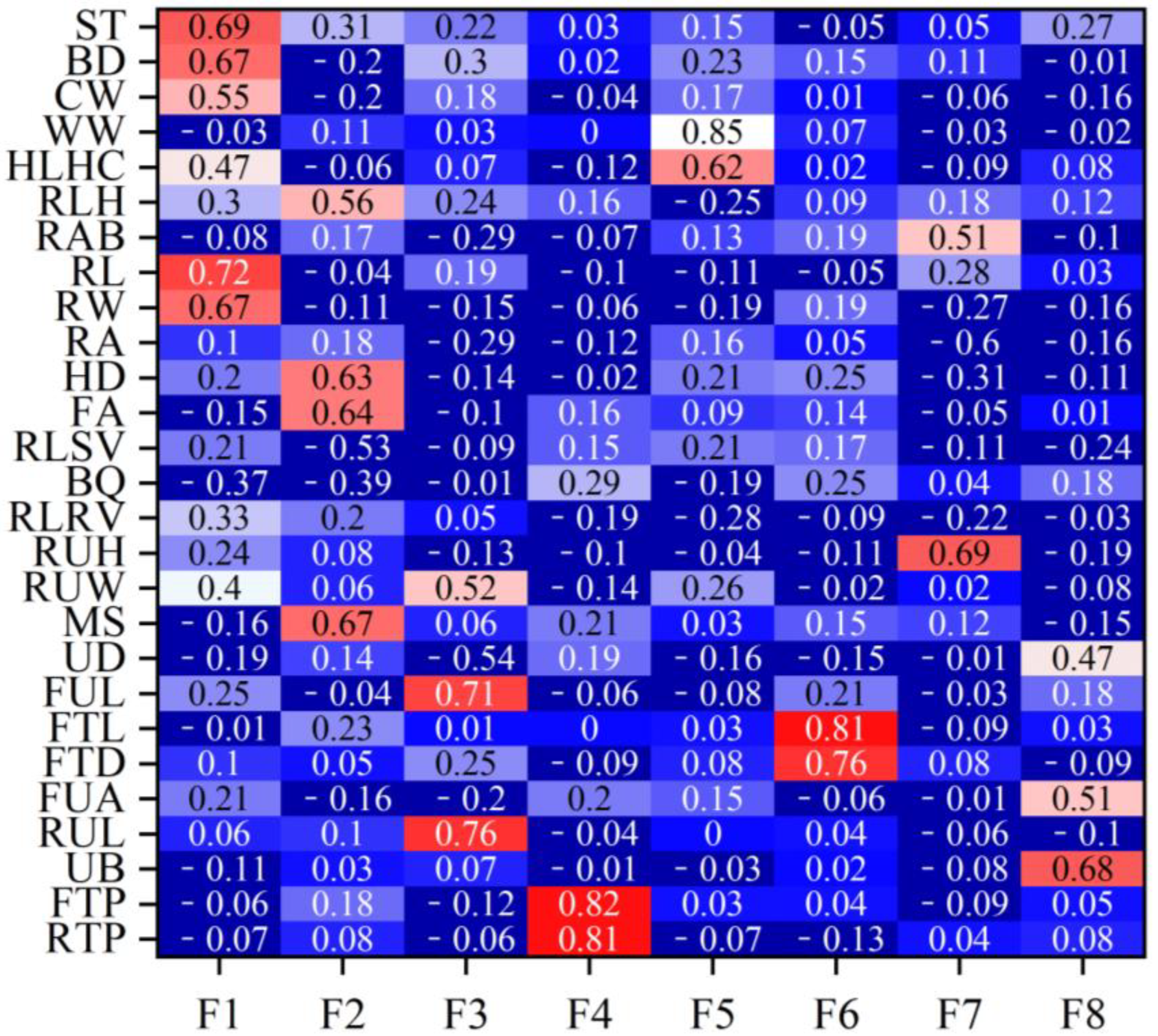
Figure 1 shows the rank correlation coefficients between the estimated breeding values (EBVs) of body conformation traits and factor scores in Xinjiang Brown cattle. Notably, the rank correlation coefficients have similar patterns to the factor loadings of body conformation traits in Figure 2. The results indicate that the EBVs of F1 had high positive correlations with the EBVs of two body frame traits and two rump traits. The EBVs of F2 in Xinjiang Brown cattle had high correlations with those of two feet and legs traits, showing similar research results to Figure 2. Other factors in this study showed similar results; for example, the correlations for the EBVs of F3 were similar to the loading coefficients of body conformation traits in Figure 2. Similar results were observed in F3, F4, F5, F6, F7, and F8.
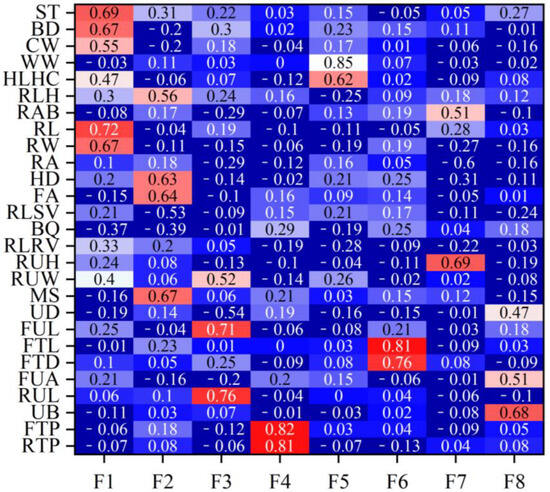
Figure 2.
Factor loading coefficients of body conformation traits for Xinjiang Brown cattle. Note: ST is stature; BD is body depth; CW is chest width; WW is wither width; HLHC is half of leg circumstance; RLH is rear leg height; RAB is rib and bone; RL is rump length; RA is rump angle; HD is heel depth; FA is feet angle; RLSV is rear leg side view; RUH is rear udder height; RUW is rear udder width; MS is medium; UD is udder depth; FUL is fore udder length; FTL is fore teat length; FTD is fore teat diameter; FUA is fore udder attachment; RUL is rear udder length; UB is udder balance; FTP is fore teat placement; RTP is rear teat placement; DC is dairy character; and LS is loin strength. Red means that the correlation coefficient is greater than 0.3, and the darker the color, the greater the correlation coefficient; blue means that the correlation coefficient is less than 0.3, and the darker the color, the smaller the correlation coefficient.
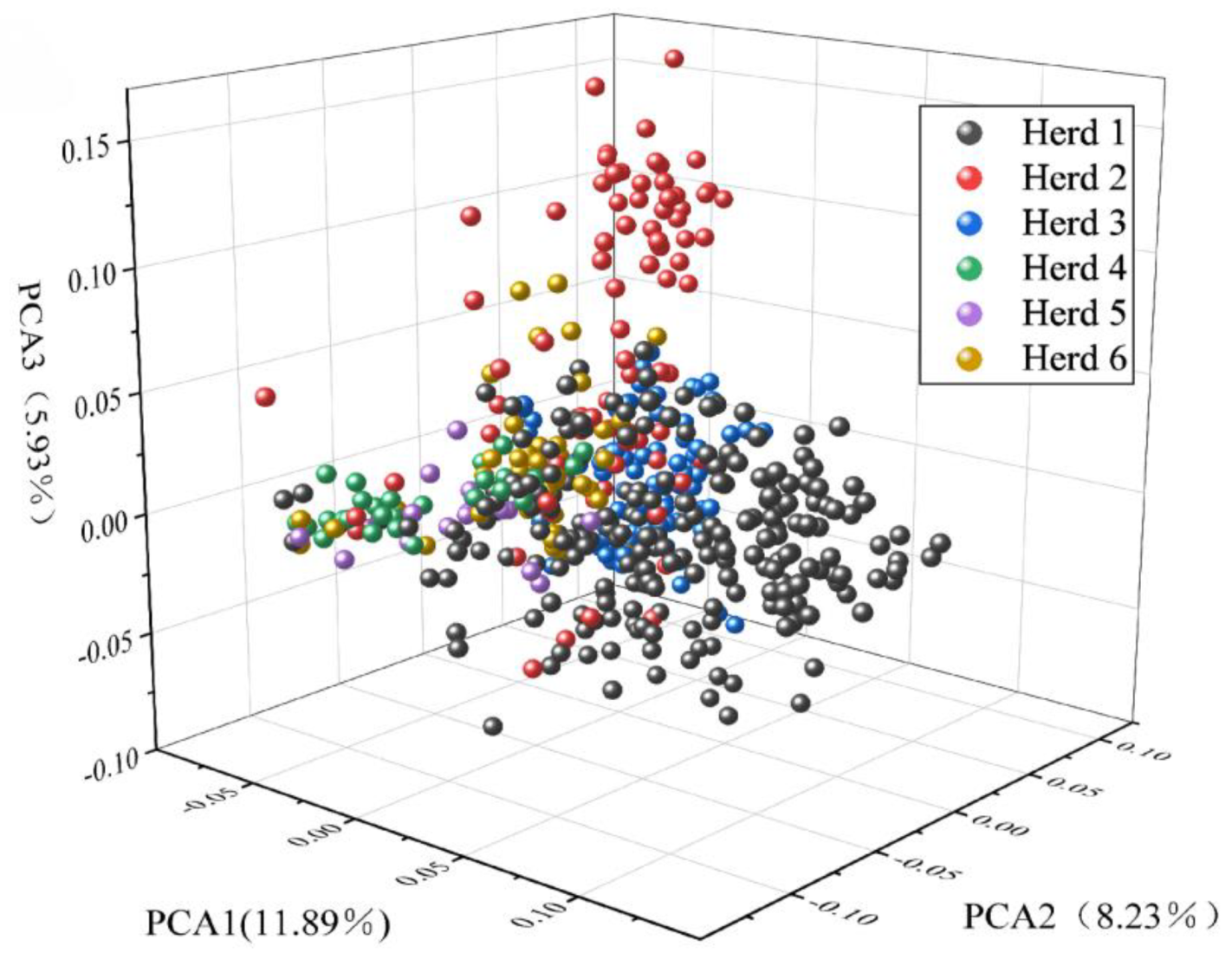
Figure 3 shows the first three principal components, accounting for 11.89%, 8.23%, and 5.93% of the total variance, respectively. By comparing the first three principal components (PCs), individuals from farm 1 (black) and farm 2 (red) could be distinguished. Although the 496 individuals came from different farms, some overlaps existed among farms, such as between farm 3 (blue) and farms 1 (black) and 2 (red), and between farm 3 (green) and farms 5 (purple) and 6 (yellow). The PCA results of 496 lactating Xinjiang Brown cattle showed some genetic connections and distances among the farms.
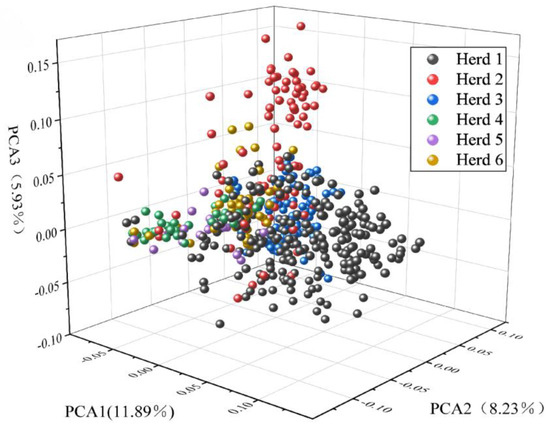
Figure 3.
Population structure map showing the first three principal components of Xinjiang Brown cattle.
2.5. Population Analysis of Body Conformation Traits
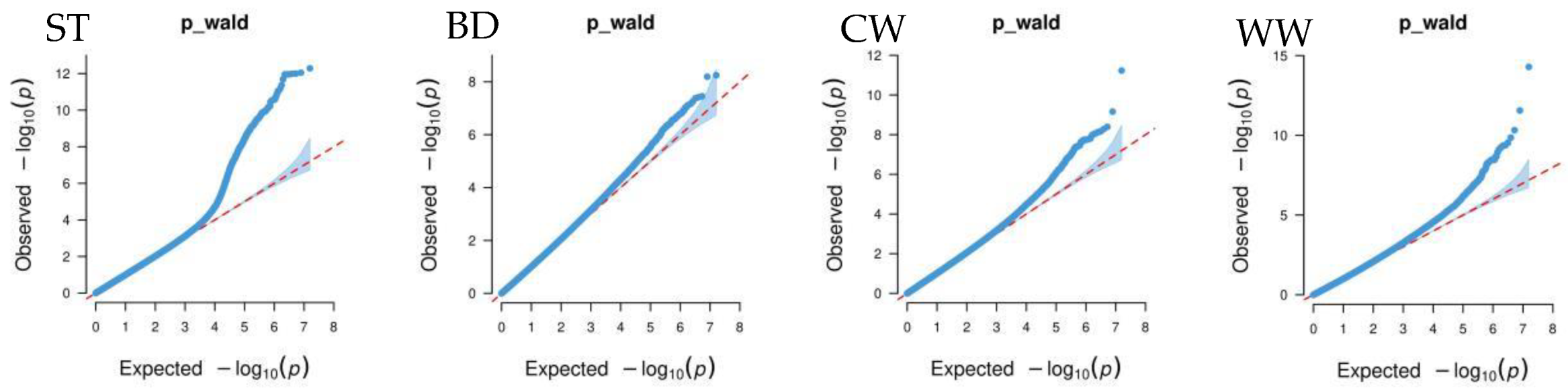
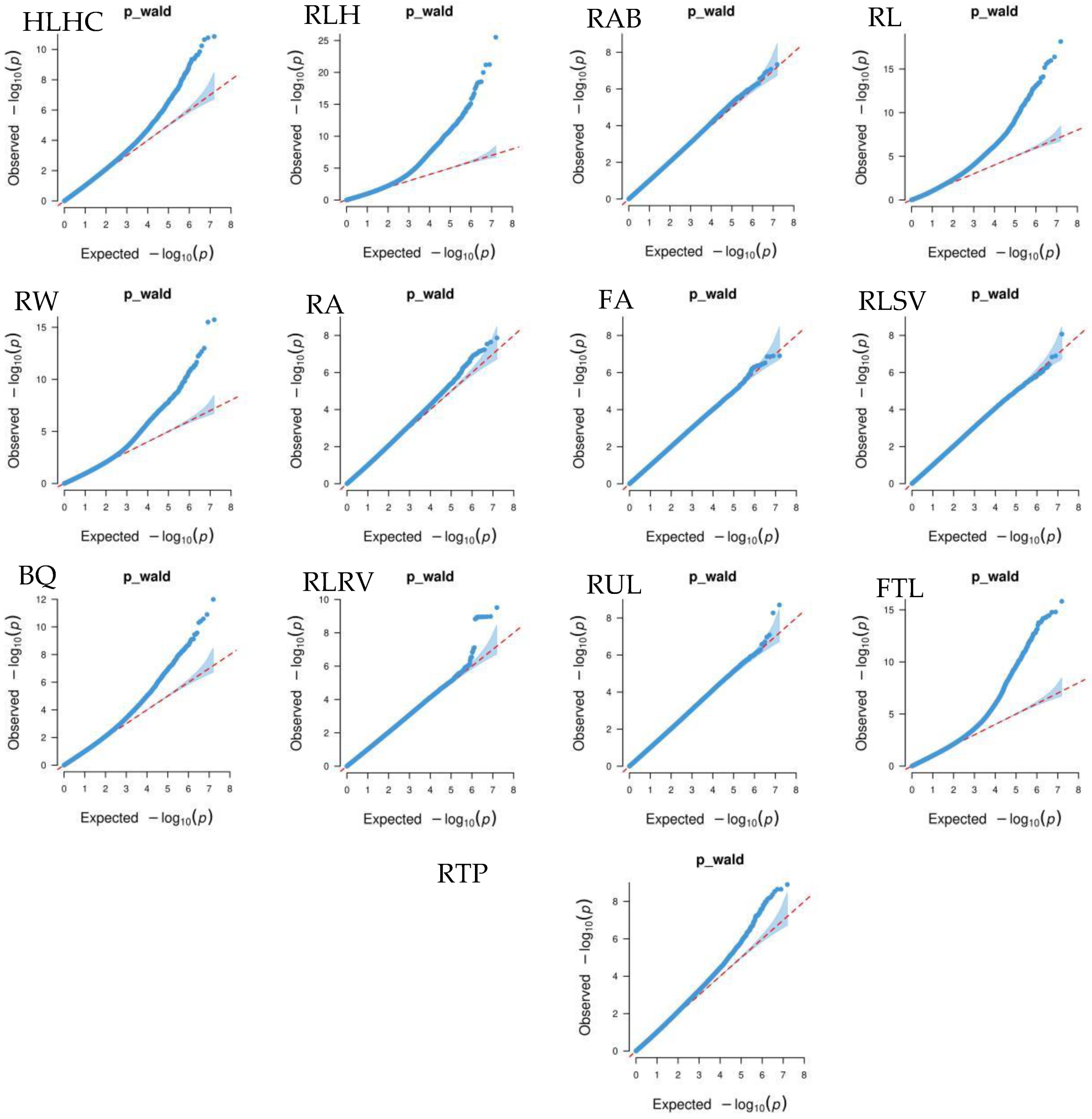
Figure 4 shows that the GWAS of body conformation traits based on the single-trait mixed linear model in GEMMA v0.98.5 software had no systemic bias. The model considered population effects and kinship, so the PCs were not included in the model. Additionally, the genomic inflation factors of body conformation traits in Xinjiang Brown cattle ranged from 0.859 to 1.022, indicating no significant genomic inflation.
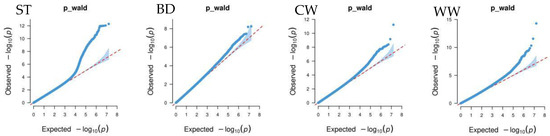
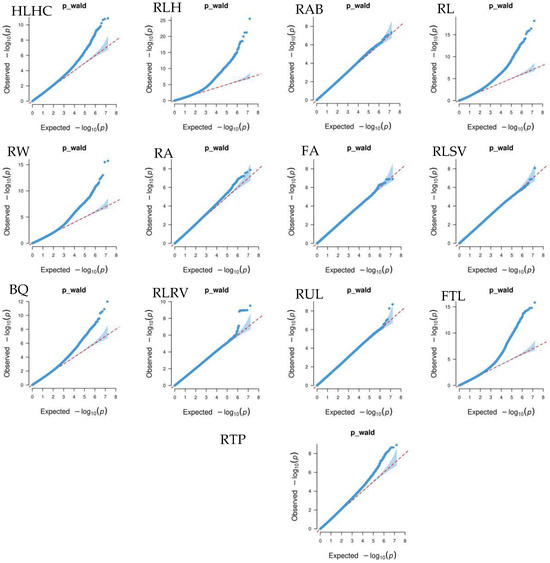
Figure 4.
Quantile–quantile (QQ) plot of GWAS analysis for body conformation traits of Xinjiang Brown cattle. Note: ST is stature; CW is chest width; BD is body depth; WW is wither width; HLHC is half of leg circumstance; RLH is rear leg height; RAB is rib and bone; RL is rump length; RW is rump width; RA is rump angle; FA is rump angle; RLSV is rear leg side view; BQ is bone quality; and RLRV is rear leg rear view.
2.6. QTL Mapping of Body Conformation Traits in Xinjiang Brown Cattle
2.6.1. QTL Mapping of Body Conformation Traits
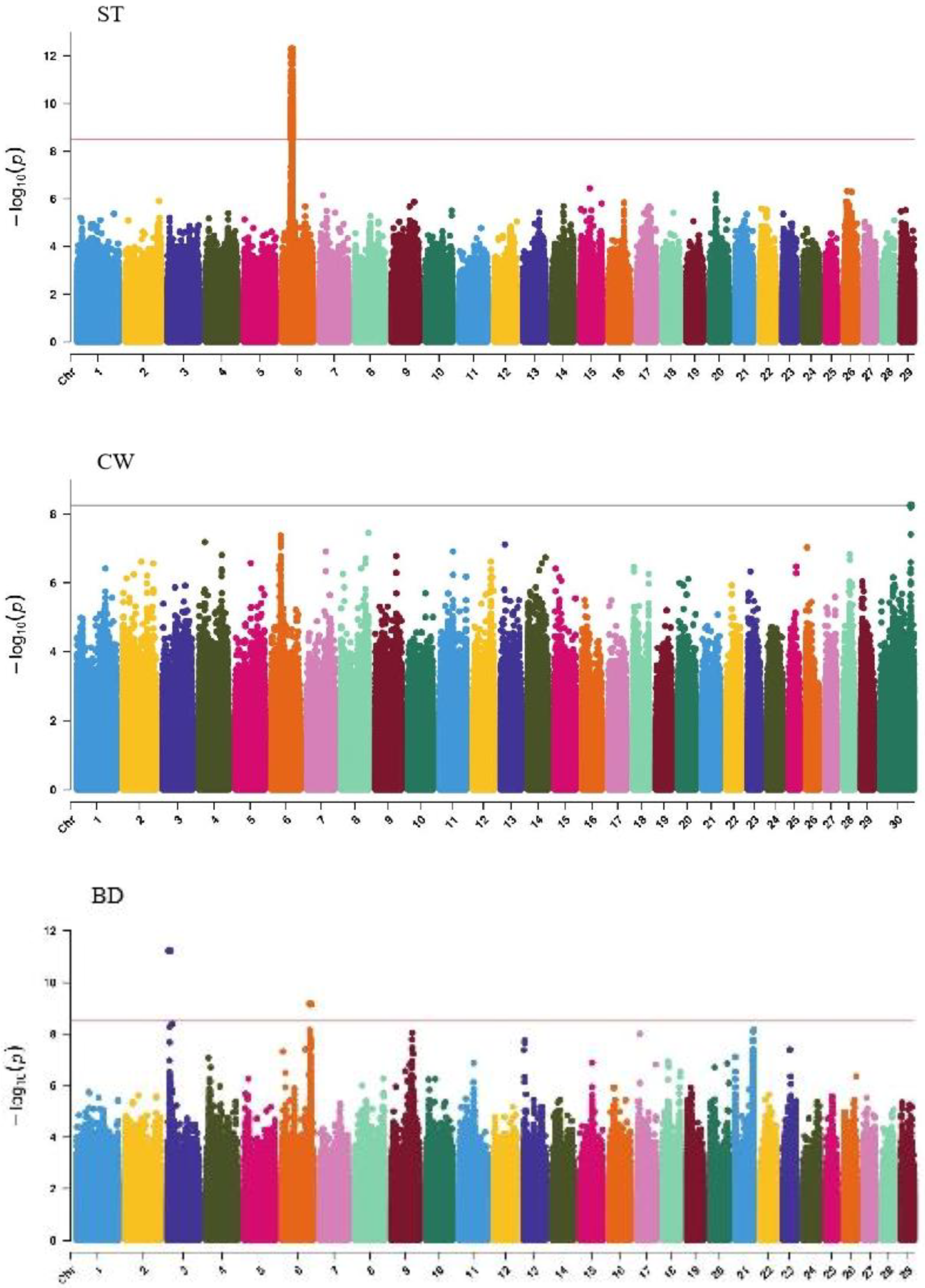
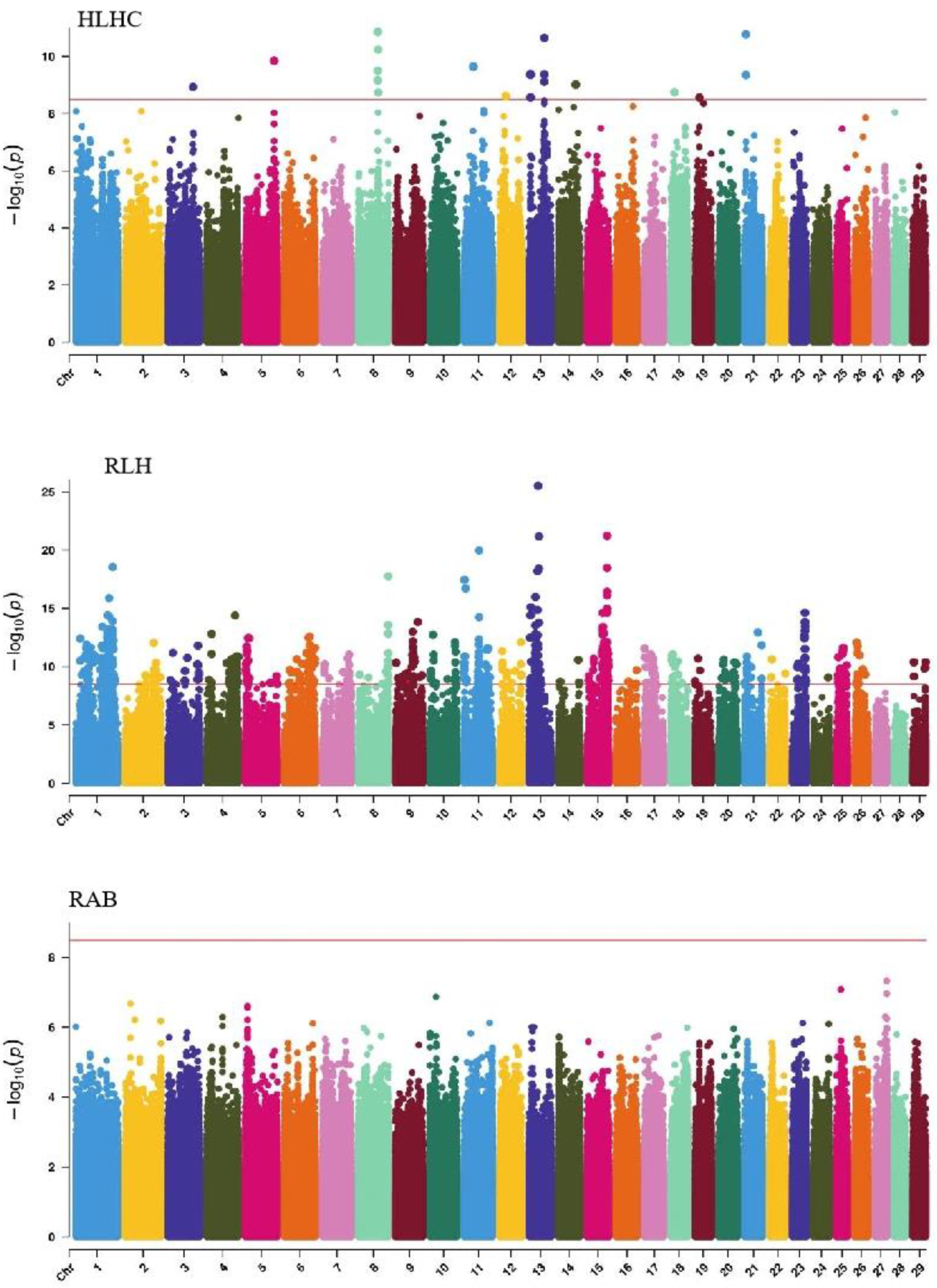
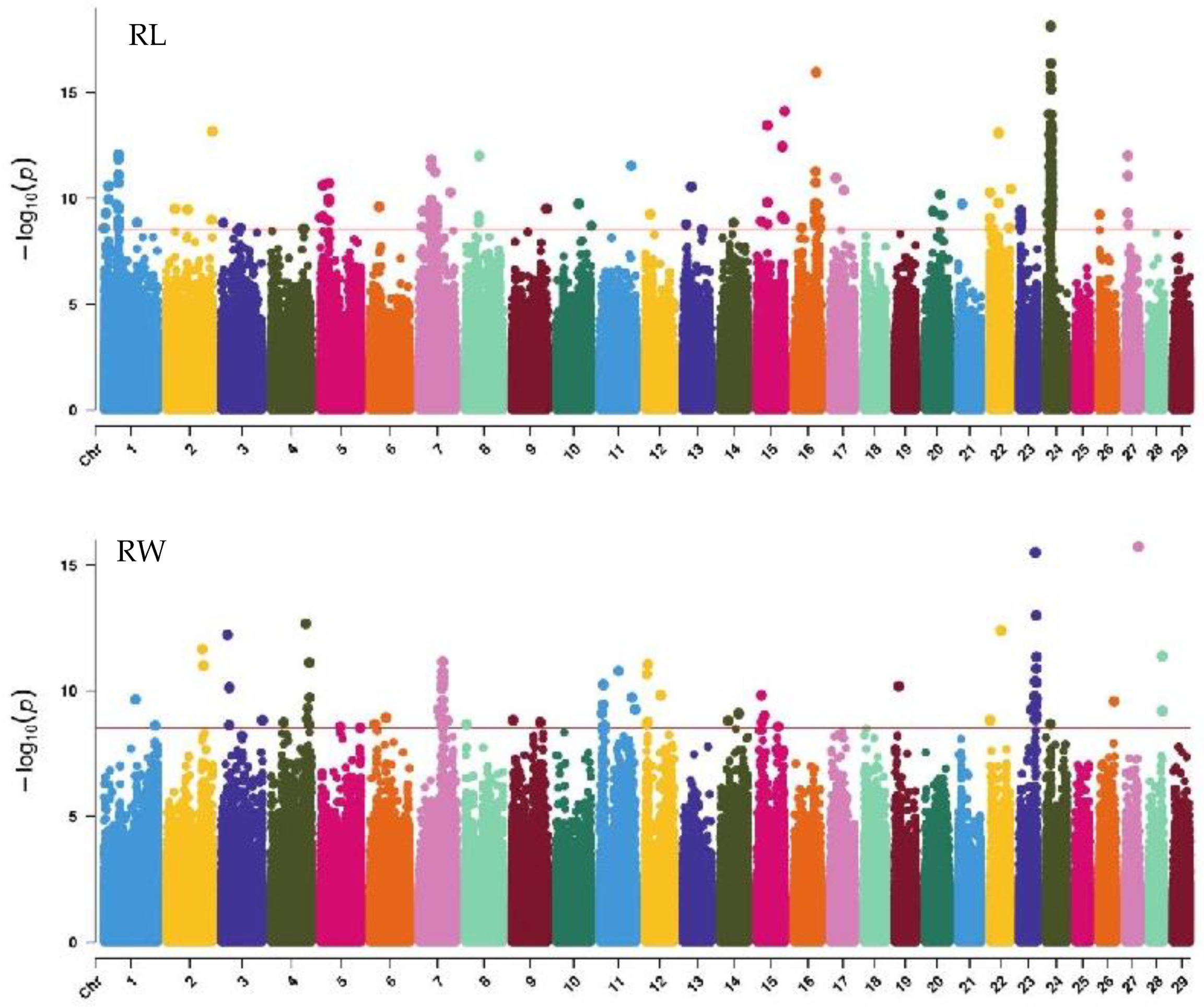
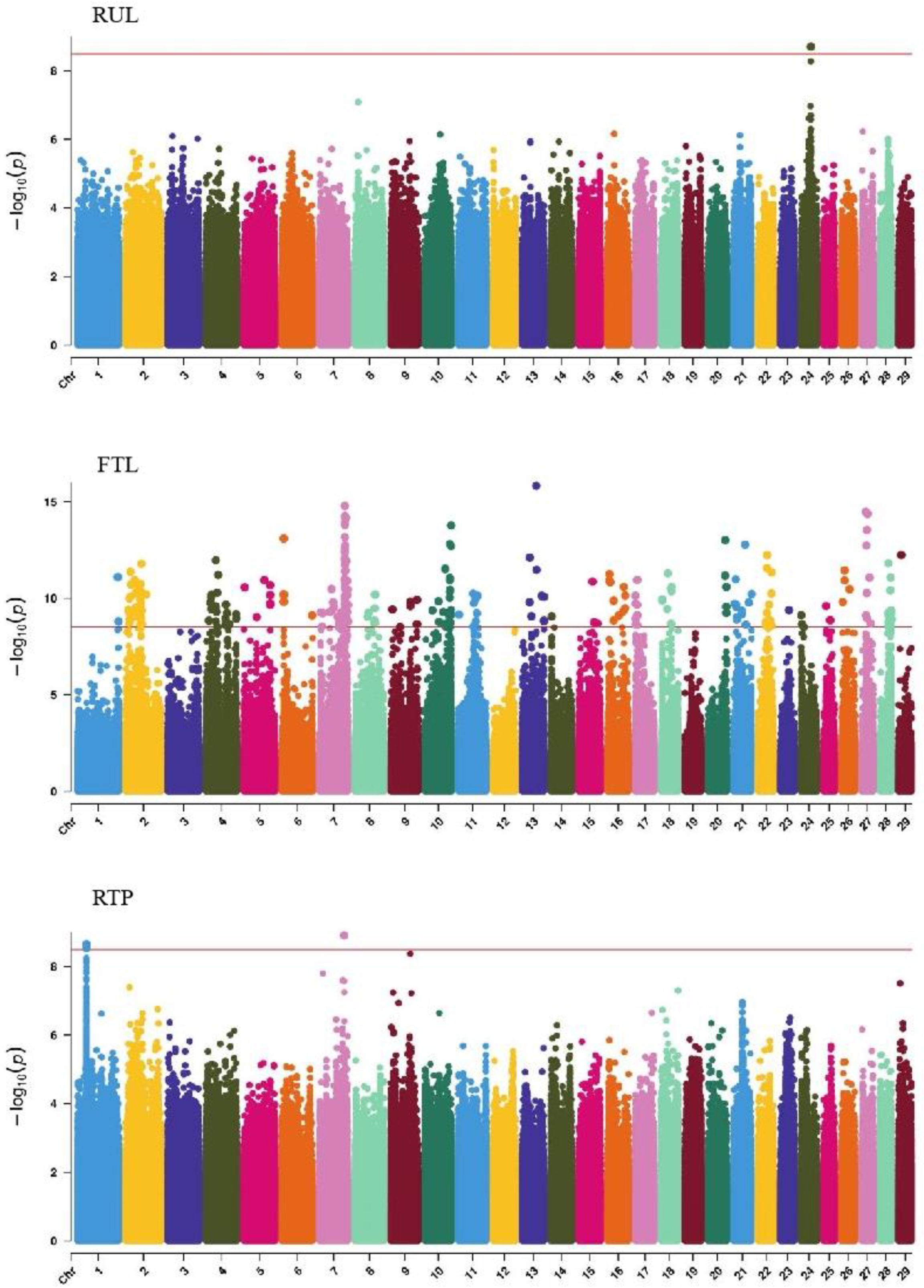
GWAS analysis of body conformation traits (stature, body depth, chest width) in Xinjiang Brown cattle identified 142, 0, and 2 significant SNPs. SNPs significantly associated with rump height and chest width were located on chromosomes 3 and 6 (Figure 5) and chromosome 6 (Figure 6), respectively.
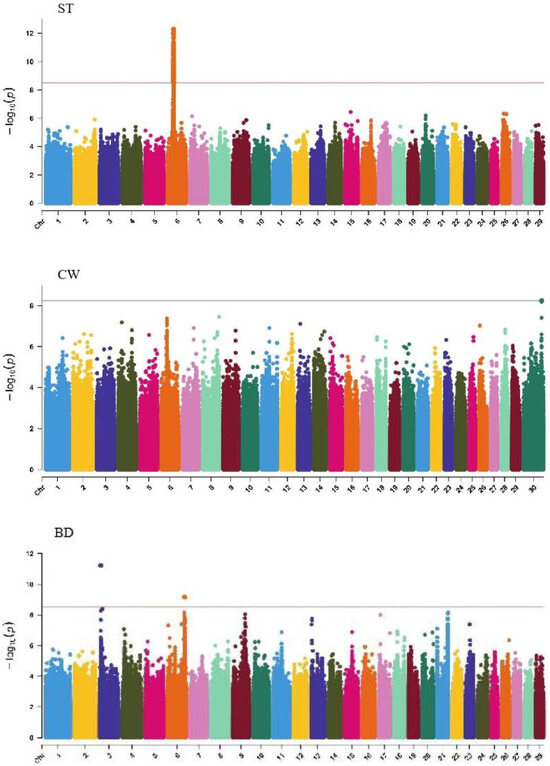
Figure 5.
Manhattan plots of body frame traits (stature, body depth, and chest width) in Xinjiang Brown cattle. Note: ST is stature of body frame traits, BD is body depth of body frame traits and CW is chest width body frame traits. Different colors represent different chromosomes, and the number on the horizontal coordinate is the chromosome number.
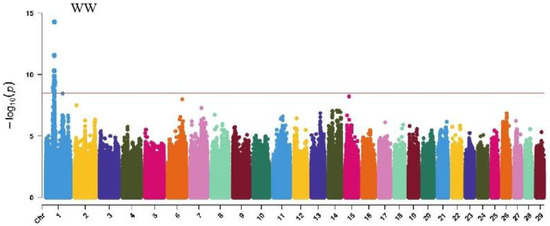
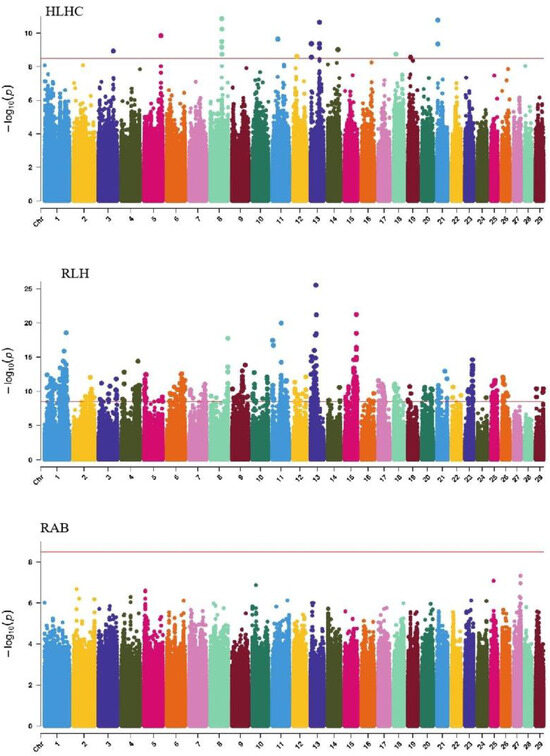
Figure 6.
Manhattan plots of muscularity traits (wither width, half of leg circumstance, rear leg height, and rib and bone) in Xinjiang Brown cattle. Note: WW is wither width; HLHC is half of leg circumstance; RLH is rear leg height; and RAB is rib and bone. Different colors represent different chromosomes, and the number on the horizontal coordinate is the chromosome number.
The SNP with the lowest p-value within each locus was defined as the lead (Table 4). According to the annotation by ANNOVAR software, the most significant locus for rump height was located on chromosome 6, with six leading SNPs in the intronic regions of the LAP3, MED28, LCORL, and NCAPG genes and one leading SNP upstream and downstream of IBSP and TRNAA-CGC, explaining 9%–11.3% of the phenotypic variance. The most significant locus for chest width was on chromosome 3, with the leading SNP in the intronic region of NOS1AP, explaining 9.1% of the phenotypic variance.

Table 4.
Descriptive summary of GWAS results for body frame traits in Xinjiang Brown cattle.
2.6.2. QTL Mapping of Muscle Development Traits
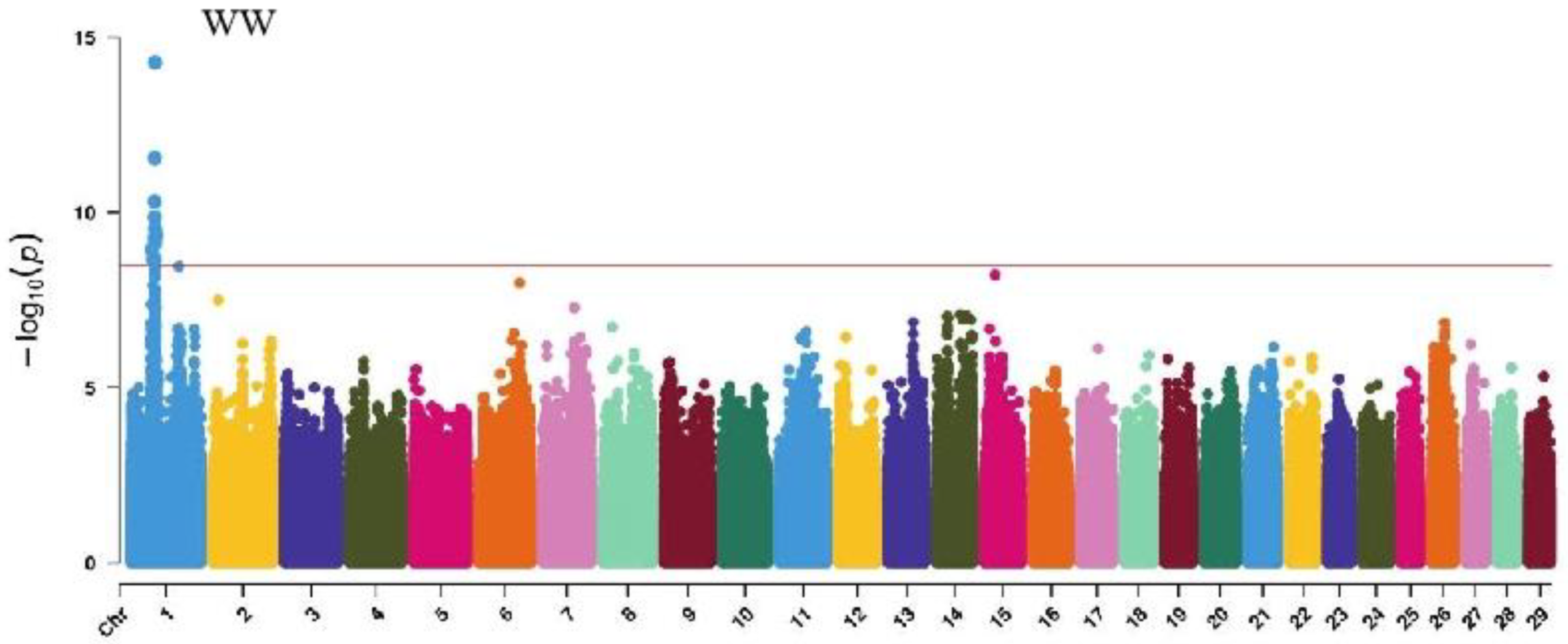
GWAS analysis of muscle development traits in Xinjiang Brown cattle identified 13, 21, 707, and 0 significant SNPs. The significantly associated SNPs were located on chromosome 1 (Figure 6) for withers width and chromosomes 3, 5, 8, 11, 12, 13, 14, 18, 19, and 21 (Figure 6) for rear leg girth. The SNPs for rear leg height were on all chromosomes except 27 and 28, with the most significant SNPs (118) on chromosome 1 and strong signals observed on chromosome 13 (Figure 6).
The SNP with the lowest p-value within each locus was defined as the leading SNP (Table 5). According to the annotation by ANNOVAR software, the most significant locus for withers width was located on chromosome 1, with leading SNPs upstream and downstream of DPPA4 and TRNAE-UUC, explaining 11.6% of the phenotypic variance. The most significant locus for rear leg girth was located on chromosome 13, with two leading SNPs upstream and downstream of CDH4, CDH26, LOC112449365, and BTBD3, explaining 8.6% and 7.6% of the phenotypic variance, respectively. The 51 leading SNPs significantly associated with rear leg height explained 6.9% to 17.1% of the phenotypic variance, with the most significant SNPs upstream and downstream of SYT13 and LOC112441655.

Table 5.
Descriptive summary of GWAS results for muscularity traits in Xinjiang Brown cattle.
2.6.3. QTL Mapping of Rump Traits
GWAS analysis of rump traits in Xinjiang Brown cattle identified 216, 81, and 0 significant SNPs. The SNPs significantly associated with rump length were distributed on all chromosomes except 18, 19, 25, 28, and 29, with the most significant SNPs on chromosome 24 (98) and strong signals on chromosome 24 (Figure 7). SNPs significantly associated with rump width were distributed on all chromosomes except 10, 13, 16, 17, 18, 20, 21, 25, and 29, with the most significant SNPs on chromosome 7 (17) (Figure 7).
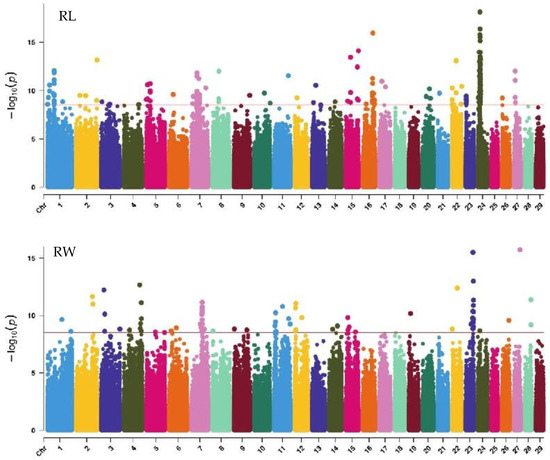
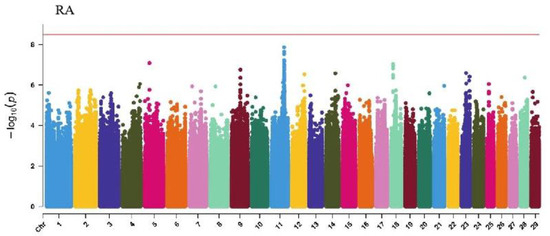
Figure 7.
Manhattan plots of rump traits (rump length, rump width, and rump angle) in Xinjiang Brown cattle. Note: RL is rump length; RW is rump width; and RA is rump angle. Different colors represent different chromosomes, and the number on the horizontal coordinate is the chromosome number.
The SNP with the lowest p-value within each locus was considered the lead (Table 6). According to the annotation by ANNOVAR software, the most significant locus for rump length was located on chromosome 24, with three leading SNPs upstream and downstream of LOC112444152, TRNAC-GCA, TRNAK-UUU, and CDH7, explaining 10.6% to 14.7% of the phenotypic variance. The most significant locus for rump width was located on chromosome 23, with the leading SNP in the intronic region of OFCC1, explaining 12.6% of the phenotypic variance.

Table 6.
Descriptive summary of GWAS results for rump traits in Xinjiang Brown cattle.
2.6.4. QTL Mapping of Limb Traits
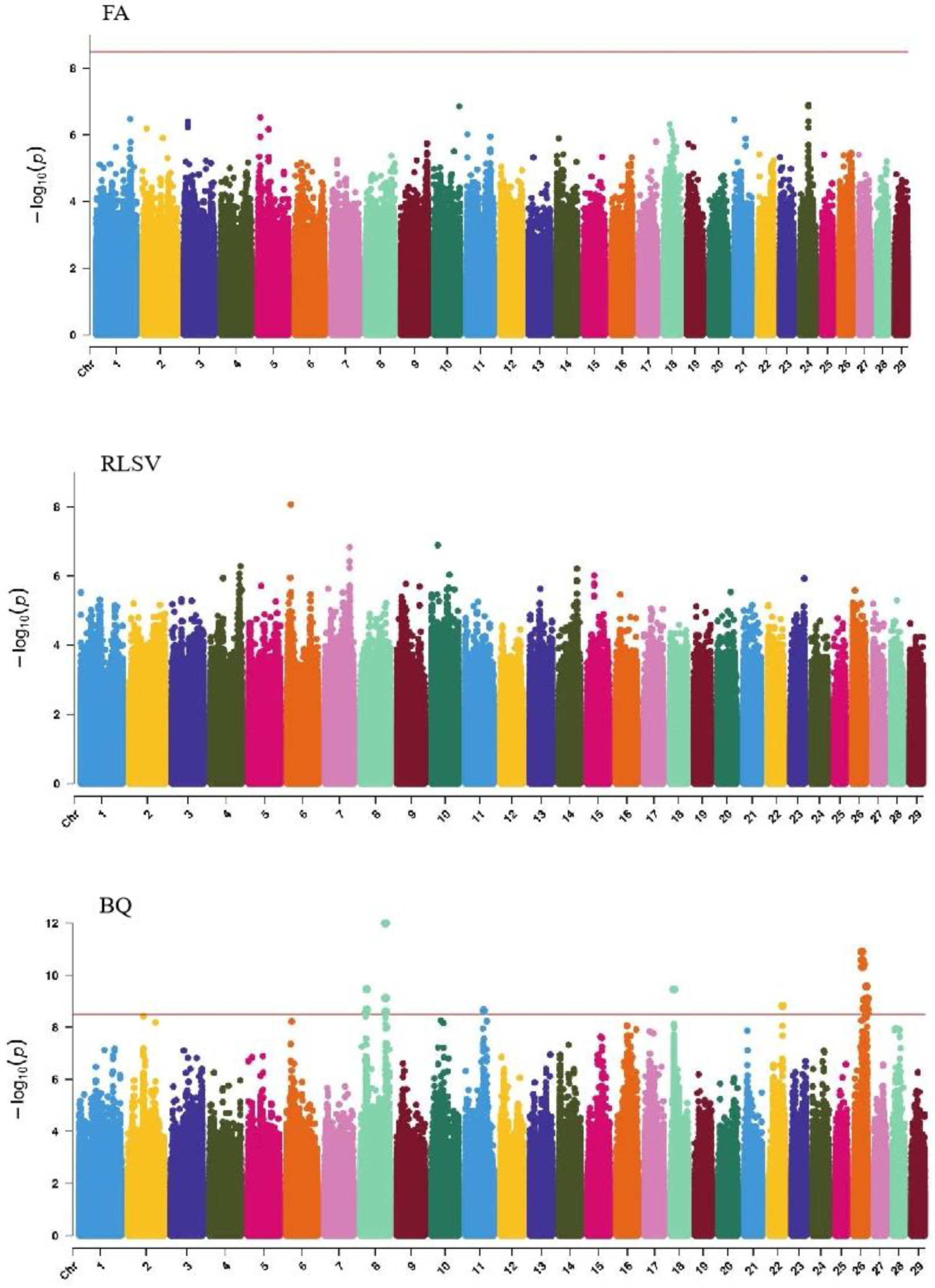
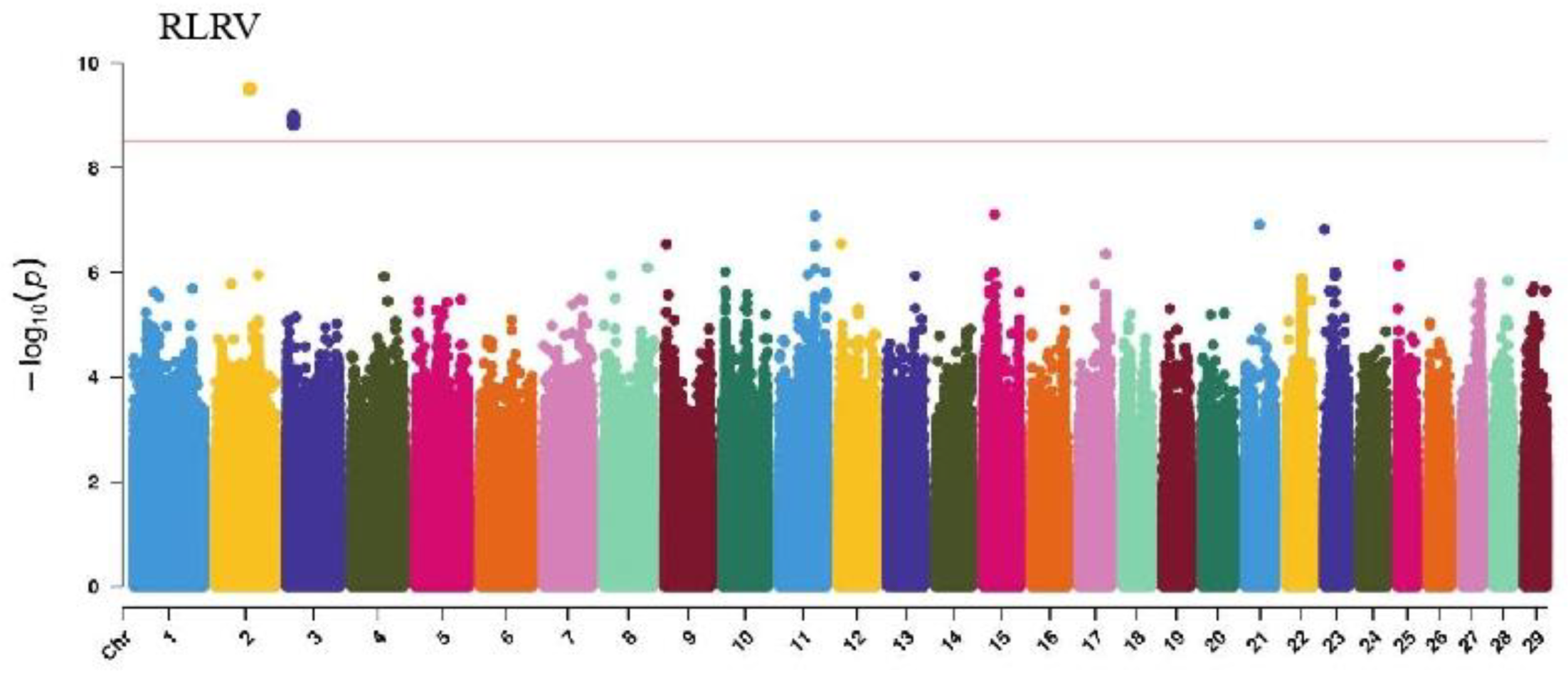
GWAS analysis of limb traits in Xinjiang Brown cattle identified 0, 0, 21, and 11 significant SNPs (p < 3.18 × 10−9). SNPs significantly associated with hock texture were distributed on chromosomes 8, 11, 18, 22, and 26, with the most significant SNPs on chromosome 26 (11) (Figure 8). SNPs significantly associated with the rear view of the rear legs were distributed on chromosomes 2 and 3, with the most significant SNPs on chromosome 3 (10) (Figure 9).
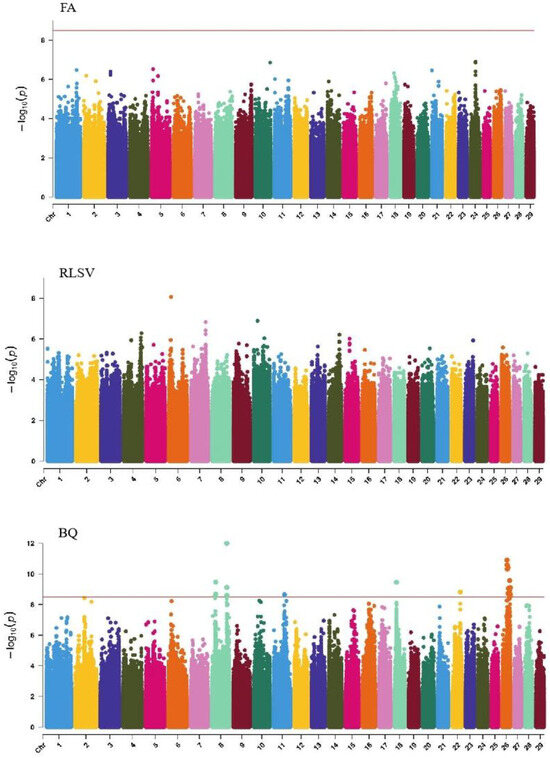
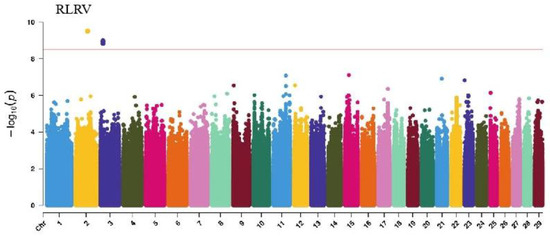
Figure 8.
Manhattan plots of rump traits (feet angle, rear leg side view, bone quality, and rear leg rear view) in Xinjiang Brown cattle. Note: FA is feet angle; RLSV is rear leg side view; BQ is bone quality; and RLRV is rear leg rear view. Different colors represent different chromosomes, and the number on the horizontal coordinate is the chromosome number.
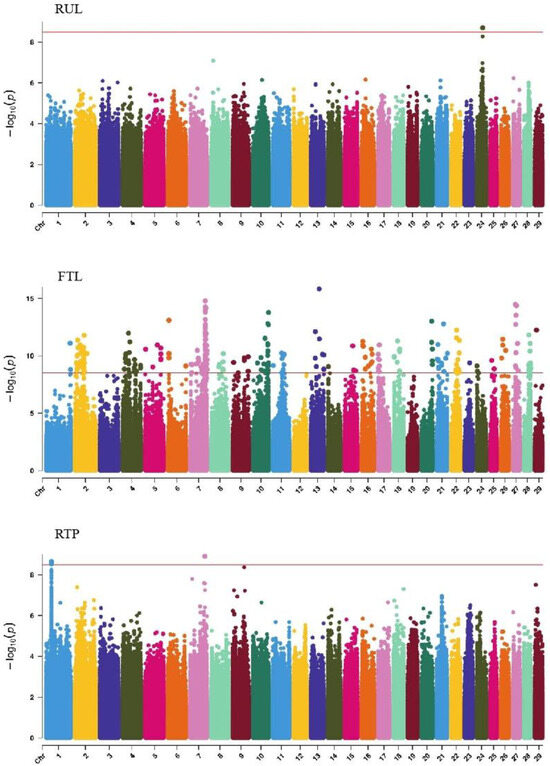
Figure 9.
Manhattan plots of udder traits (rear udder length, fore teat length, and rear teat placement) in Xinjiang Brown cattle. Note: RUL is rear udder length; FTL is fore teat length; and RTP is rear teat placement. Different colors represent different chromosomes, and the number on the horizontal coordinate is the chromosome number.
The SNP with the lowest p-value within each locus was considered the leading SNP (Table 7). According to the annotation by ANNOVAR software, the most significant locus for hock texture was located on chromosome 8, with the leading SNP in the intronic region of ABCA1, explaining 9.7% of the phenotypic variance. Two leading SNPs on chromosome 26 were upstream and downstream of ADRA2A, GPAM, MKI67, and MGMT, explaining 8.8% and 7.7% of the phenotypic variance, respectively. Another leading SNP on chromosome 26 was in the intronic region of KNDC1, explaining 7% of the phenotypic variance. For the rear view of the rear legs, the leading SNP on chromosome 3 was upstream and downstream of KCNN3 and ADAR.

Table 7.
Descriptive summary of GWAS results for feet and leg traits in Xinjiang Brown cattle.
2.6.5. QTL Mapping of Udder Traits
GWAS analysis of udder traits in Xinjiang Brown cattle identified 1, 325, and 4 significant SNPs for rear udder length, front teat length, and rear teat position, respectively. The significantly associated SNPs were located on chromosome 24 for rear udder length and on all chromosomes except 3, 12, and 19, with the most significant SNPs on chromosome 7 for front teat length (Figure 9). SNPs significantly associated with rear teat position were distributed on chromosomes 1 and 7, with three significant SNPs on chromosome 1 and one on chromosome 7.
The SNP with the lowest p-value within each locus was defined as the lead (Table 8). According to the annotation by ANNOVAR software, the most significant locus for rear udder length was located on chromosome 24, with the leading SNP upstream of DLGAP1, explaining 7.1% of the phenotypic variance. For front teat length, two leading SNPs with strong signals on chromosome 7 were upstream and downstream of RGMB and CHD1, explaining 12.0% and 11.4% of the phenotypic variance, respectively. Another leading SNP was in the intronic region of FBXL17. For the rear teat position trait, two leading SNPs on chromosomes 7 and 1 were upstream and downstream of ELL2, PCSK1, and CADM2.

Table 8.
Descriptive summary of GWAS results for udder traits in Xinjiang Brown cattle.
3. Materials and Methods
3.1. Data Source and Processing
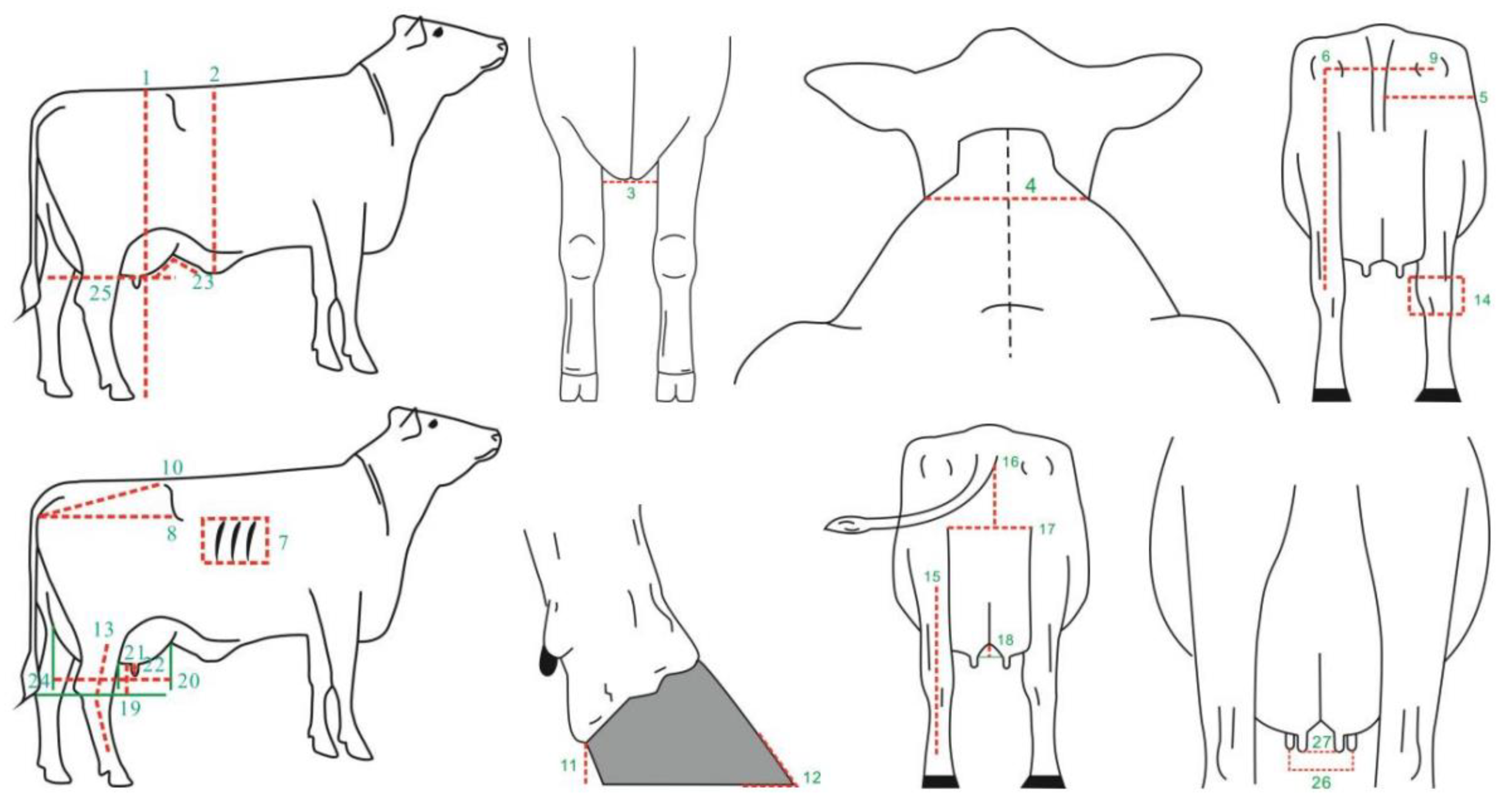
The body conformation data for Xinjiang Brown cattle were collected from nine core breeding farms in Xinjiang from July 2018 to July 2022. A total of 27 body size traits were measured, including 17 measurable traits and 10 scored traits (1–9 points). The specific assessment parts are shown in Figure 10. Scored traits were assessed by two certified Chinese dairy cattle body size assessors (certificate numbers: DAC-TY-083, DAC-TY-019). A total of 1816 lactating Xinjiang Brown cows were assessed, with abnormal records excluded based on the principle of “mean ± 3 standard deviations” and cows without pedigree records removed. Finally, the data from 1185 lactating Xinjiang Brown cows were further analyzed. The pedigree records of Xinjiang Brown cattle consisted of 186 bulls and 2067 cows, checked and sorted using the chkPed() function of ASReml v4.2 software (VSNC, Asian-Pacific region, UK).
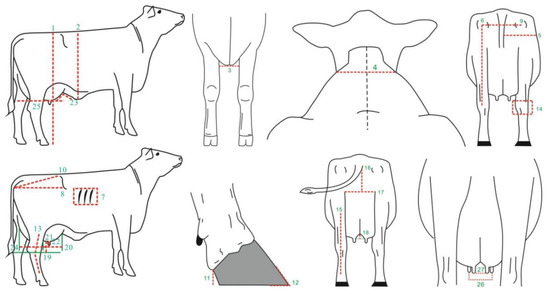
Figure 10.
Description and measurement sites of 27 body conformation traits. Note: 1. stature (cm); 2. body depth (cm); 3. chest width (cm); 4. withers width (cm); 5. hind leg half circumference (cm); 6. rear leg height (cm); 7. rib and bone (points); 8. rump length (cm); 9. rump width (cm); 10. rump angle (cm); 11. heel depth (cm); 12. foot angle (points); 13. rear legs side view (points); 14. bone quality (points); 15. rear legs rear view (points); 16. rear udder height (cm); 17. rear udder width (cm); 18. median suspensory (cm); 19. udder depth (cm); 20. fore udder length (cm); 21. front teat length (cm); 22. front teat diameter (cm); 23. fore udder attachment (points); 24. rear udder length (points); 25. udder balance (points); 26. fore teat placement (points); and 27. rear teat placement (points).
3.2. Blood Sample Collection and Whole-Genome Resequencing
Blood samples (10 mL) were collected from the tail vein of the test animals and the anticoagulated whole blood was stored at −20 °C for genomic testing. Genomic DNA was extracted from 496 Xinjiang Brown cattle and sequenced using the MGISEQ-2000 platform by BGI (MGI Tech, Shenzhen, China). The resequencing of 496 Xinjiang Brown cattle generated approximately 74.9 billion reads, with an average sequencing depth of 8.04× and an average alignment rate of 99.87%.
3.3. Read Alignment and SNP Detection
The raw reads from the resequencing data of 496 Xinjiang Brown cattle were first quality-controlled using Trimmomatic v0.40 software (http://USADELLAB.org) [21]. Clean reads were aligned to the ARS-UCD1.2 reference genome using the MEME module of BWA v0.7.17 software [22] with default parameters. Duplicate reads were removed using the Sortsam and MarkDuplicates modules of Picard v2.25.5 software to reduce PCR amplification bias. Base quality score recalibration was performed using the BaseRecalibrator and PrintReads modules of GATK. The SNPs were detected using the HaplotypeCaller, CombineGVCFs, GenotypeGVCFs, and SelectVariants modules of GATK v3.8 [23]. Low-quality SNPs were filtered using the VariantFiltration module with the following parameters: QD < 2.0, FS > 60.0, MQ < 40.0, MQRankSum < −12.5, ReadPosRankSum < −8.0, and SOR > 3.0. SNPs were aligned to the ARS-UCD1.2 reference genome, and functional annotation of filtered SNPs was performed using ANNOVAR software [24]. SNPs were excluded based on the following criteria: (1) individual genotype call rate < 90%, (2) single SNP genotype call rate < 90%, (3) significant deviation from Hardy–Weinberg equilibrium (p < 10−6), (4) minor allele frequency (MAF) < 0.05, and (5) unknown chromosome or genome information. A total of 15,700,670 SNPs were retained in Xinjiang Brown cattle.
3.4. Statistical Analysis
3.4.1. Estimation of Genetic Parameters
Using ASReml v4.2 software, the variance components of 27 body conformation traits, the PCs, and factors were estimated using the average information restricted maximum likelihood method in an animal model. The model used was:
where represents the observed vector of 27 body conformation traits, β is the fixed effects vector including farm effect (9 levels), days in milk effect (9 levels: 10–40 days, 41–80 days, 81–120 days, 121–160 days, 161–200 days, 201–240 days, 241–280 days, 281–320 days, >320 days), and parity effect (4 levels: 1st parity, 2nd parity, 3rd parity, ≥4th parity), is the additive genetic effect vector, is the random residual, and and are the incidence matrices for fixed and random effects, respectively.
A two-trait animal model was constructed using PCs and factor scores as cap Y to estimate their genetic correlations. The two-trait animal model matrix form is:
where y1 and y2 are the observed values of each factor and principal component, b1 and b2 are the fixed effects vectors, with fixed effects consistent, g1 and g2 are the additive genetic effects vectors, e1 and e2 are the random residuals, and X1, X2, Z1, and Z2 are the corresponding incidence matrices.
The estimated variance components were used to calculate the genetic parameters [25]:
where is heritability, is additive genetic variance, is residual variance, and is the additive genetic covariance between two traits.
3.4.2. Factor Analysis
Factor analysis was conducted using the FACTOR procedure in SAS v9.1 software, which synthesizes the information contained in a set of n observed variables by seeking a set of new variables, known as common latent factors. The varimax rotation method was used to maintain the orthogonality of extracted factors, retaining only factors with eigenvalues ≥1 [26]. Factor loadings of each body conformation trait were observed to interpret analysis results biologically. Standardized factor scores were calculated for each cow to facilitate the analysis of common factors as analysis variables instead of the original variables before dimension reduction.
As described previously [27], the factor analysis model is expressed as:
where yn is the nth original variable, bnp is the loading of each variable n on each factor, xp is the pth common factor of the nth variable, and en reflects the specific factor of the nth variable.
3.4.3. Genome-Wide Association Study
Haplotype inference and missing allele imputation for GWAS analysis were performed using the method described in BEAGLE v4.1 software [28], which included a total of 15,700,670 SNPs for Xinjiang Brown cattle.
Here, GWAS analysis for 27 body conformation traits of Xinjiang Brown cattle was conducted using the single-trait mixed linear model in GEMMA v0.98.5 software [29]. The GWAS model is:
where represents the observed vector of 27 body conformation traits, is the covariant matrix, including farm effect (9 levels), days in milk effect (9 levels: 10–40 days, 41–80 days, 81–120 days, 121–160 days, 161–200 days, 201–240 days, 241–280 days, 281–320 days, >320 days), and parity effect (4 levels: 1st parity, 2nd parity, 3rd parity, ≥4th parity), is the covariant vector, is the marker genotype, is the marker genotype effect; is the genetic relationship coefficient matrix, is the genetic relationship coefficient effect, and is the random residual vector.
Bonferroni correction was applied to multiple hypothesis testing, setting the significance threshold p-value at 3.18 × 10−9. GWAS visualization was performed using the CMplot package in R v4.4.1 software.
To further determine the genetic effects of significant SNPs, the proportion of phenotypic variance explained by significant SNPs was calculated using the formula reported by Shim et al. [30]:
where is the effect size of the SNP marker, MAF is the minor allele frequency, is the residual variance, and N is the sample size for GWAS analysis.
Genomic inflation factor λ was calculated using R v4.4.1 software, converting association test statistics p-values to , and dividing the median statistic by the expected median of the distribution (0.4549).
After completing the GWAS analysis, linkage disequilibrium among significant SNPs was calculated, and the critical value for associated loci was defined. The LD is denoted by R2 (). Loci with fewer than three significant SNPs were excluded from further analysis to reduce false-positive signals. The SNP with the smallest p-value within each locus was defined as the leading SNP. Functional annotation of SNPs related to body size traits was performed using ANNOVAR software [24] based on the ARS-UCD1.2 reference genome. Candidate genes within a 500 Kb range upstream and downstream of significant SNPs were considered for further analysis [30].
4. Discussion
4.1. Estimation of Genetic Parameters for Body Size Traits and Factor Scores
The rump height of lactating Xinjiang Brown cattle was similar to that of Swiss Brown cattle (139 cm) [31], whereas the body depth was similar to that of Slovenian Brown cattle (76.20 cm) [32]. The rump length and width were higher than those of Slovenian Brown cattle (48.40 cm and 17.59 cm, respectively) [32]. These two body conformation traits had medium to high heritability, but chest width had low heritability. Several studies have reported lower heritability for chest width compared with rump height and body depth [33]. Rump traits showed higher heritability, consistent with studies on Italian Brown cattle [34], while most feet and legs traits have been reported to have low to medium heritability [33,35]. This study analyzed more body conformation traits than previous studies in different cattle breeds [36,37], somewhat increasing the data dimension. Generally, cows with deep and tall bodies and wide and large udders are better milk producers [31,38]. Body conformation traits with high loadings in F1 are usually associated with milk production performance, consistent with the findings of Kern et al. [38]. Thus, F1 can be included in selection indices to improve milk production performance.
In this study, the rank correlation results between the EBVs of body conformation traits and the factor scores exhibited patterns similar to those of the factor loadings of body conformation traits. Mazza S et al. reported similar results in two dual-purpose cattle breeds [39]. This further indicates the consistency of using factor score phenotypes for genetic evaluation of body conformation parts. Although factor analysis can eliminate redundant information in correlated variables, it is ambiguous in explaining single traits. When conducting genetic selection for multiple traits in a population, the factor score can effectively eliminate redundancy among traits of interest under genetic selection.
4.2. QTL for Body Frame Traits
Body frame traits include stature, body depth, and chest width. In this study, LCORL and NCAPG were significantly associated with stature in Xinjiang Brown cattle. An Bingsing et al. reported a significant association between LAP3, which regulates hormone secretion levels and protein maturation [40], and stature in Wagyu cattle [41]. Additionally, LAP3 and FAN184B have been reported to be significantly associated with bone weight in beef cattle [42,43]. Shen Jiafei et al. found that IBSP is significantly associated with the ear margin area in Brahman and Yunling cattle [44]. LCORL encodes a transcription factor that may function in spermatogenesis in the testes [45], while NCAPG encodes a non-histone chromatin-associated protein G in mammals, which is part of the condensin I complex involved in chromatin compression and regulation, especially during mitosis [46]. LCORL and NCAPG are key genes affecting height in many species, such as horses [18], cattle [47], and dogs [48]. LCORL and NCAPG have been identified as height-related loci in European, Japanese, and African populations [49,50,51,52]. However, due to the proximity of LCORL and NCAPG and high linkage disequilibrium among SNPs in these regions, it is still uncertain which gene has a greater impact on height, requiring further verification.
4.3. QTL for Muscle Development Traits
Candidate genes related to muscle development traits include CPNE4, RUS1, DNAJC1TBX18, REG3G, AHCYL2, FSTL5, NPFFR2, and FAT3, which have been reported to be associated with growth, muscle development, body size, and meat performance. CPNE4 is a gene related to glycogen content, regulating muscle glycogen content by affecting glucose metabolism and being associated with growth performance, body size, muscle, and skeletal development in cattle [53,54,55]. RUS1 was shown to influence weaning weight in sheep [56]. DNAJC1 was reported to have pleiotropy in weaning and yearling weight in Angus beef cattle [57]. However, TBX18 has not been previously found to be related to body size traits. However, a recent study showed an association between TBX18 and growth performance in Simmental dual-purpose cattle [58]. Additionally, TBX18 is related to obesity in humans and mice [59,60]. Lee et al. reported that TBX18 regulates processes related to skeletal muscle metabolism, affecting body size in animals [61]. Studies on Nellore cattle showed that REG3G is associated with growth performance [62], while AHCYL2 is related to carcass backfat thickness [63]. In a study on Limousin and Jersey cattle crossbreeds, Novianti et al. reported that FSTL5 on bovine chromosome 17 is a key gene affecting muscle development [64]. While NPFFR2 was shown to be significantly associated with the udder morphology traits of German Simmental cattle [65], it was significantly associated with withers width in this study. Regarding meat performance, Riley et al. found that the FAT3 gene is related to beef palatability [66].
4.4. QTL for Rump Traits
EBF1, NSMCE2, TASOR, NEDD4, and PGF, associated with rump traits in Xinjiang Brown cattle, are related to cattle reproduction and rump traits. Cole et al. reported that EBF1 affects rump width and calving ease in Holstein cattle [67]. Rump width has been used as an indirect selection trait for calving ease. The bovine QTL database search results also showed that EBF1 is associated with rump width and calving ease. In this study, this gene was associated with rump traits in Xinjiang Brown cattle, making it a candidate gene for rump trait selection. Moreover, in addition to affecting rump width in dairy cattle [67], NEDD4 is also related to the eye muscle area in beef cattle. Cao et al. demonstrated that NEDD4 deficiency reduces IGF-1 and insulin signaling in knockout mice [68]. PGF, a member of the vascular endothelial growth factor family, is related to early embryo development in yaks [69]. In a study on German Holstein cattle, PGF was shown to be associated with calving ease and stillbirth [70].
4.5. QTL for Feet and Legs Traits
In the GWAS study of feet and legs traits in Xinjiang Brown cattle, six candidate genes were annotated, with MGMT being noteworthy for its association with body size, reproduction, and longevity traits, including overall body score, rear legs, rear view, limb score, and hoof angle. The loci associated with MGMT showed the most significant effect on limb traits [67]. Recent studies have reported that GATB is related to white-line disease in dairy cattle [71]. The white line is a soft keratinized area at the junction of the hoof sole and wall. Pérez-Cabal et al. indicated that foot angle significantly influences the incidence of white-line disease [72], with higher linear scores and larger angles being associated with a lower disease incidence. Other candidate genes identified in this study have not been reported in limb-related traits but are considered important candidate genes in cattle behavior and milk production traits. Michenet et al. [73] found that ADRA2 is associated with maternal protective behavior in cows and encodes an adrenergic receptor related to various behavioral traits in humans, mice, and rats [74]. Hence, this might be a candidate gene affecting cattle behavior.
4.6. QTL for Udder Traits
Various GWAS reports on udder traits in different cattle populations have identified several candidate loci and genes [65,75,76,77]. However, studies identifying genomic regions related to udder traits are inconsistent, including this one. These differences in identified genomic regions are possibly influenced by statistical methods and population differences. Although candidate genes annotated to leading SNPs associated with udder traits in this study have been rarely reported, significant SNPs discovered were adjacent to previously reported SNP loci. For example, SNP rs441400818 on BTA7 and rs380411477 on BTA17 were close to the SNPs rs29023522 and rs42313276 associated with teat length reported by Cole et al., with annotated genes including RGMB, CHD1, CPE, and MSMO1. The SNP rs109736435 on BTA7 was close to the SNP rs110574421 associated with teat position, annotated to ELL2 and PCSK1 [67]. An Animal QTLdb database search indicated that CHD1, CPE, and ELL2 are related to a productive life, overall body score, and somatic cell score, directly associated with teat length and position [78,79].
5. Conclusions
Most body conformation traits and factor scores in Xinjiang Brown cattle were medium to highly heritable. The rank correlation results between the estimated breeding values of body conformation traits and the factor scores exhibited patterns similar to those of the factor loadings of body conformation traits, suggesting that the factor scores should be included in selection indices. Using latent factors in body conformation trait evaluation can simplify multidimensional body conformation traits into new variables, reducing the computational burden of analyzing large datasets. In Xinjiang Brown cattle, 102 leading SNPs were significantly associated with 12 body conformation traits. Some SNPs were located within or near previously reported genes and QTL regions, whereas others were newly discovered. These significant SNPs identified through GWAS can be included in future genomic genetic evaluation models to continuously optimize these models and provide a scientific basis for the selection and breeding of Xinjiang Brown cattle.
Author Contributions
Conceptualization, X.H. and L.X.; methodology, Q.C.; formal analysis, M.Z.; investigation, L.X, D.W., X.Z. and M.Z.; resources, X.H.; data curation, L.X. and Q.C.; writing—original draft preparation, M.Z.; writing—review and editing, M.Z, L.X., Y.W. and X.H.; supervision, X.H. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by The Xinjiang Uygur Autonomous Region Science and Technology Aid Xinjiang Project (Grant No. 2018E02052), the Xinjiang Brown Cattle Joint Breeding Population Improvement and Promotion Action Plan (Grant No. 2023XJHN-16), and the National Modern Agriculture Industrial System Project (Grant No. CARS-36).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The study did not report any data.
Acknowledgments
We greatly thank the MDPI, and we greatly thank the Xinjiang Uygur Autonomous Region local state-owned Urumqi cattle farm, Yili Xinjiang Brown cattle farm, and Tacheng Agriculture and Animal Husbandry Technology Co. Ltd., China for providing us with working conditions.
Conflicts of Interest
We certify that there are no conflicts of interest with any financial organizations regarding the material discussed in the manuscript.
References
- Stefani, G.U.; El Faro, L.; Santana Júnior, M.L.; Tonhati, H.U. Association of longevity with type traits, milk yield and udder health in Holstein cows. Livest. Sci. 2018, 218, 1–7. [Google Scholar] [CrossRef]
- Zink, V.; Štípková, M.; Lassen, J. Genetic parameters for female fertility, locomotion, body condition score, and linear type traits in Czech Holstein cattle. J. Dairy Sci. 2011, 94, 5176–5182. [Google Scholar] [CrossRef] [PubMed]
- Gibson, K.D.; Dechow, C.D. Genetic parameters for yield, fitness, and type traits in US Brown Swiss dairy cattle. J. Dairy Sci. 2018, 101, 1251–1257. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Cabal, M.A.; Alenda, R. Genetic Relationships between Lifetime Profit and Type Traits in Spanish Holstein Cows. J. Dairy Sci. 2002, 85, 3480–3491. [Google Scholar] [CrossRef]
- Török, E.; Komlósi, I.; Szőnyi, V.; Béri, B.; Mészáros, G.; Posta, J. Combinations of Linear Type Traits Affecting the Longevity in Hungarian Holstein-Friesian Cows. Animals 2021, 11, 3065. [Google Scholar] [CrossRef]
- Liang, D.; Tang, S.; Gao, W.; Yang, Z.; Ma, H.; Liu, L.; Xiao, W.; Zhang, Y. Current Status of Mating Program in Dairy Farms: A Questionnaire Survey in Beijing Area. China Dairy Cattle 2016, 4, 17–22. [Google Scholar]
- Hu, Y. The Formulation and Application of Seed Selection and Matching Plan of Simmental Cattle in Core Breeding Farms in Xinjiang; Xinjiang Agricultural University: Urumqi, China, 2022. [Google Scholar]
- Wang, C. Formulation and Application of Seed Selection and Mating Program for Dairy Line of Xinjiang Brown Cattle; Xinjiang Agricultural University: Urumqi, China, 2022. [Google Scholar]
- Simčič, M.; Luštrek, B.; Štepec, M.; Logar, B.; Potočnik, K. Estimation of Genetic Parameters of Type Traits in First Parity Cows of the Autochthonous Cika Cattle in Slovenia. Front. Genet. 2021, 12, 724058. [Google Scholar] [CrossRef]
- Mazza, S.; Guzzo, N.; Sartori, C.; Berry, D.P.; Mantovani, R. Genetic parameters for linear type traits in the Rendena dual-purpose breed. J. Anim. Breed. Genet. 2014, 131, 27–35. [Google Scholar] [CrossRef]
- Wang, D.; Fan, S.; Geng, J.; Yang, G.; Dong, M.; Xu, L.; Yan, M.; Li, Y.; Huang, X.; Wang, Y. Body Size Canonical Correlation Analysis and Body Weight Estimation Formula Formulation of Xinjiang Brown Cattle. Chin. J. Anim. Sci. 2021, 57, 82–86. [Google Scholar]
- Schneider, P.; Fikse, W.F. Short Communication: Exploring Patterns of Variation in Conformation Data by Means of Principal Components Analysis. J. Dairy Sci. 2008, 91, 4440–4443. [Google Scholar]
- Macciotta, N.P.P.; Vicario, D.; Cappio-Borlino, A. Use of multivariate analysis to extract latent variables related to level of production and lactation persistency in dairy cattle. J. Dairy Sci. 2006, 89, 3188–3194. [Google Scholar] [CrossRef] [PubMed]
- Cesar, A.S.; Regitano, L.C.; Mourão, G.B.; Tullio, R.R.; Lanna, D.P.; Nassu, R.T.; Mudado, M.A.; Oliveira, P.S.; Nascimento, M.L.D.; Chaves, A.S. Genome-wide association study for intramuscular fat deposition and composition in Nellore cattle. BMC Genet. 2014, 15, 39. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Zhang, L.; Li, W.; Liu, M. BIEC2-808543 SNP in the LCORL Gene is Associated with Body Conformation in the Yili Horse. Anim. Biotechnol. 2015, 26, 289–291. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Uemoto, Y.; Kikuchi, T.; Egawa, S.; Kohira, K.; Saito, T.; Sakuma, H.; Miyashita, S.; Arata, S.; Kojima, T. SNP- and haplotype-based genome-wide association studies for growth, carcass, and meat quality traits in a Duroc multigenerational population. BMC Genet. 2016, 17, 60. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, R.; Zhao, G.; Zheng, M.; Sun, Y.; Yu, X.; Li, P.; Wen, J. Genome-Wide Linkage Analysis Identifies Loci for Physical Appearance Traits in Chickens. G3 Genes Genomes Genet. 2015, 5, 2037–2041. [Google Scholar] [CrossRef]
- Tetens, J.; Widmann, P.; Kühn, C.; Thaller, G. A genome-wide association study indicates LCORL/NCAPG as a candidate locus for withers height in German Warmblood horses. Anim. Genet. 2013, 44, 467–471. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, L.; Chen, C.J.; Zhang, M.; Lu, X.; Zhang, Z.; Huang, X.; Shi, Y. Genome-wide association study of milk and reproductive traits in dual-purpose Xinjiang Brown cattle. BMC Genom. 2019, 20, 827. [Google Scholar] [CrossRef]
- Zheng, W.; Li, H.; Su, D.; Yan, Q.; Chen, S.; Zhang, S.; Sun, D. General Status of International Genetic Evaluation System of Dairy Cattle. Chin. J. Anim. Sci. 2020, 56, 161–168. [Google Scholar]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina Sequence Data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2010, 25, 1754–1760. [Google Scholar] [CrossRef]
- McKenna, A.; Hanna, M.; Banks, E. The Genome Analysis Toolkit: A MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2010, 20, 1297–1303. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, M.; Hakonarson, H. ANNOVAR: Functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010, 38, e164. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. Animal Breeding; China Agriculture Press: Beijing, China, 2018. [Google Scholar]
- Kaiser, H.F. The Application of Electronic Computers to Factor Analysis. Educ. Psychol. Meas. 1960, 20, 141–151. [Google Scholar] [CrossRef]
- Morrison, D.F. Multivariate Statistical Methods; McGraw-Hill: New York, NY, USA, 1976. [Google Scholar]
- Browning, S.R.; Browning, B.L. Rapid and accurate haplotype phasing and missing-data inference for whole-genome association studies by use of localized haplotype clustering. Am. J. Hum. Genet. 2007, 81, 1084–1097. [Google Scholar] [CrossRef]
- Zhou, X.; Stephens, M. Genome-wide efficient mixed-model analysis for association studies. Nat. Genet. 2012, 7, 821–824. [Google Scholar] [CrossRef]
- Purfield, D.C.; Bradley, D.G.; Kearney, J.F.; Berry, D.P. Genome-wide association study for calving traits in Holstein–Friesian dairy cattle. Animal 2014, 8, 224–235. [Google Scholar] [CrossRef]
- De Haas, Y.; Janss, L.L.G.; Kadarmideen, H.N. Genetic and phenotypic parameters for conformation and yield traits in three Swiss dairy cattle breeds. J. Anim. Breed. Genet. 2007, 124, 12–19. [Google Scholar] [CrossRef]
- Špehar, M.; Štepec, M.; Potočnik, K. Variance components estimation for type traits in Slovenian brown Swiss cattle. Acta Agric. Slov. 2012, 100, 107–115. [Google Scholar] [CrossRef]
- Roveglia, C.; Niero, G.; Bobbo, T.; Penasa, M.; Finocchiaro, R.; Visentin, G.; Lopez-Villalobos, N.; Cassandro, M. Genetic parameters for linear type traits including locomotion in Italian Jersey cattle breed. Livest. Sci. 2019, 229, 131–136. [Google Scholar] [CrossRef]
- Dal Zotto, R.; De Marchi, M.; Dalvit, C.; Cassandro, M.; Gallo, L.; Carnier, P.; Bittante, G. Heritabilities and genetic correlations of body condition score and calving interval with yield, somatic cell score, and linear type traits in Brown Swiss cattle. J. Dairy Sci. 2007, 90, 5737–5743. [Google Scholar] [CrossRef]
- Tarres, J.; Liu, Z.; Ducrocq, V.; Reinhardt, F.; Reents, R. Data transformation for rank reduction in multi-trait MACE model for international bull comparison. Genet. Sel. Evol. 2008, 40, 295–308. [Google Scholar] [PubMed]
- Liu, J.; Yang, G.; Li, J.; Nai, B.; Huang, X.; Wang, Y. Study on Body Conformation Linear Assessment of Xinjiang Brown Cattlee. Chin. J. Anim. Sci. 2020, 56, 64–69. [Google Scholar]
- Gutiérrez, J.P.; Goyache, F. Estimation of genetic parameters of type traits in Asturiana de los Valles beef cattle breed. J. Anim. Breed. Genet. 2002, 119, 93–100. [Google Scholar] [CrossRef]
- Kern, E.L.; Uacute, J.A.; Cobuci, J.; Aacute, C.; Costa, U.N.; Pimentel, C.M.M. Factor analysis of linear type traits and their relation with longevity in brazilian holstein cattle. Asian Austral. J. Anim. 2014, 27, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Mazza, S.; Guzzo, N.; Sartori, C.; Mantovani, R. Factor analysis for genetic evaluation of linear type traits in dual-purpose autochthonous breeds. Animal 2016, 10, 372–380. [Google Scholar] [CrossRef][Green Version]
- Yamahara, N.; Nomura, S.; Suzuki, T.; Itakura, A.; Ito, M.; Okamoto, T.; Tsujimoto, M.; Nakazato, H.; Mizutani, S. Placental leucine aminopeptidase/oxytocinase in maternal serum and placenta during normal pregnancy. Life Sci. 2000, 66, 1401–1410. [Google Scholar] [CrossRef]
- An, B.; Xia, J.; Chang, T.; Wang, X.; Xu, L.; Zhang, L.; Gao, X.; Chen, Y.; Li, J.; Gao, H. Genome-wide association study reveals candidate genes associated with body measurement traits in Chinese Wagyu beef cattle. Anim. Genet. 2019, 50, 386–390. [Google Scholar] [CrossRef]
- Niu, Q.; Zhang, T.; Xu, L.; Wang, T.; Wang, Z.; Zhu, B.; Gao, X.; Chen, Y.; Zhang, L.; Gao, H.; et al. Identification of Candidate Variants Associated with Bone Weight Using Whole Genome Sequence in Beef Cattle. Front. Genet. 2021, 12, 750746. [Google Scholar] [CrossRef]
- Xia, J.; Fan, H.; Chang, T.; Xu, L.; Zhang, W.; Song, Y.; Zhu, B.; Zhang, L.; Gao, X.; Chen, Y.; et al. Searching for new loci and candidate genes for economically important traits through gene-based association analysis of Simmental cattle. Sci. Rep.-UK 2017, 7, 42048. [Google Scholar] [CrossRef]
- Shen, J.; Xia, X.; Sun, L.; Ma, X.; Huang, B.; Hanif, Q.; Chen, N.; Qu, K.; Zhang, J.; Chen, H.; et al. Genome-wide association study reveals that the IBSP locus affects ear size in cattle. Heredity 2023, 130, 394–401. [Google Scholar] [CrossRef]
- Kunieda, T.; Park, J.M.; Takeuchi, H.; Kubo, T. Identification and characterization of Mlr1,2: Two mouse homologues of Mblk-1, a transcription factor from the honeybee brain. FEBS Lett. 2003, 535, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Dej, K.J.; Ahn, C.; Orr-Weaver, T.L. Mutations in the Drosophila condensin subunit dCAP-G: Defining the role of condensin for chromosome condensation in mitosis and gene expression in interphase. Genetics 2004, 168, 895–906. [Google Scholar] [CrossRef] [PubMed]
- Setoguchi, K.; Watanabe, T.; Weikard, R.; Albrecht, E.; Kühn, C.; Kinoshita, A.; Sugimoto, Y.; Takasuga, A. The SNP c.1326T>G in the non-SMC condensin I complex, subunit G (NCAPG) gene encoding a p.Ile442Met variant is associated with an increase in body frame size at puberty in cattle. Anim. Genet. 2011, 42, 650–655. [Google Scholar] [CrossRef] [PubMed]
- Hayward, J.J.; Castelhano, M.G.; Oliveira, K.C.; Corey, E.; Balkman, C.; Baxter, T.L.; Casal, M.L.; Center, S.A.; Fang, M.; Garrison, S.J.; et al. Complex disease and phenotype mapping in the domestic dog. Nat. Commun. 2016, 7, 10460. [Google Scholar] [CrossRef]
- Allen, H.L.; Estrada, K.; Lettre, G.; Berndt, S.I.; Weedon, M.N.; Rivadeneira, F.; Willer, C.J.; Jackson, A.U.; Vedantam, S.; Raychaudhuri, S.; et al. Hundreds of variants clustered in genomic loci and biological pathways affect human height. Nature 2010, 467, 832–838. [Google Scholar] [CrossRef]
- Gudbjartsson, D.F.; Walters, G.B.; Thorleifsson, G.; Stefansson, H.; Halldorsson, B.V.; Zusmanovich, P.; Sulem, P.; Thorlacius, S.; Gylfason, A.; Steinberg, S.; et al. Many sequence variants affecting diversity of adult human height. Nat. Genet. 2008, 40, 609–615. [Google Scholar] [CrossRef]
- Weedon, M.N.; Lango, H.; Lindgren, C.M.; Wallace, C.; Evans, D.M.; Mangino, M.; Freathy, R.M.; Perry, J.R.; Stevens, S.; Hall, A.S.; et al. Genome-wide association analysis identifies 20 loci that influence adult height. Nat. Genet. 2008, 40, 575–583. [Google Scholar] [CrossRef]
- Wood, A.R.; Esko, T.; Yang, J.; Vedantam, S.; Pers, T.H.; Gustafsson, S.; Chu, A.Y.; Estrada, K.; Luan, J.A.; Kutalik, Z.; et al. Defining the role of common variation in the genomic and biological architecture of adult human height. Nat. Genet. 2014, 46, 1173–1186. [Google Scholar] [CrossRef]
- Barbato, M.; Hailer, F.; Upadhyay, M.; Del Corvo, M.; Colli, L.; Negrini, R.; Kim, E.S.; Crooijmans, R.P.; Sonstegard, T.; Ajmone-Marsan, P. Adaptive introgression from indicine cattle into white cattle breeds from Central Italy. Sci. Rep. 2020, 10, 1279. [Google Scholar] [CrossRef]
- de Simoni Gouveia, J.J.; Gualberto Barbosa Da Silva, M.V.; Paiva, S.R. Identification of selection signatures in livestock species. Genet. Mol. Biol. 2014, 37, 330–342. [Google Scholar] [CrossRef]
- Jahuey-Martínez, F.J.; Parra-Bracamonte, G.M.; Sifuentes-Rincón, A.M.; Martínez-González, J.C.; Gondro, C.; García-Pérez, C.A.; López-Bustamante, L.A. Genomewide association analysis of growth traits in Charolais beef cattle. J. Anim. Sci. 2016, 94, 4570–4582. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili-Fard, S.M.; Gholizadeh, M.; Hafezian, S.H.; Abdollahi-Arpanahi, R. Genes and Pathways Affecting Sheep Productivity Traits: Genetic Parameters, Genome-Wide Association Mapping, and Pathway Enrichment Analysis. Front. Genet. 2021, 12, 710613. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.L.; Wilson, M.L.; Nilson, S.M.; Rowan, T.N.; Oldeschulte, D.L.; Schnabel, R.D.; Decker, J.E.; Seabury, C.M. Genome-wide association and genotype by environment interactions for growth traits in U.S. Red Angus cattle. BMC Genom. 2022, 20, 517. [Google Scholar] [CrossRef] [PubMed]
- Doyle, J.L.; Berry, D.P.; Veerkamp, R.F.; Carthy, T.R.; Walsh, S.W.; Evans, R.D.; Purfield, D.C. Genomic Regions Associated with Skeletal Type Traits in Beef and Dairy Cattle Are Common to Regions Associated with Carcass Traits, Feed Intake and Calving Difficulty. Front. Genet. 2020, 11, 20. [Google Scholar] [CrossRef] [PubMed]
- Ejarque, M.; Ceperuelo-Mallafré, V.; Serena, C.; Maymo-Masip, E.; Duran, X.; Díaz-Ramos, A.; Millan-Scheiding, M.; Núñez-Álvarez, Y.; Núñez-Roa, C.; Gama, P.; et al. Adipose tissue mitochondrial dysfunction in human obesity is linked to a specific DNA methylation signature in adipose-derived stem cells. Int. J. Obesity 2018, 43, 1256–1268. [Google Scholar] [CrossRef]
- Sun, W.; Zhao, X.; Wang, Z.; Chu, Y.; Mao, L.; Lin, S.; Gao, X.; Song, Y.; Hui, X.; Jia, S.; et al. Tbx15 is required for adipocyte browning induced by adrenergic signaling pathway. Mol. Metab. 2019, 28, 48–57. [Google Scholar] [CrossRef]
- Lee, K.Y.; Singh, M.K.; Ussar, S.; Wetzel, P.; Hirshman, M.F.; Goodyear, L.J.; Kispert, A.; Kahn, C.R. Tbx15 controls skeletal muscle fibre-type determination and muscle metabolism. Nat. Commun. 2015, 6, 8054. [Google Scholar] [CrossRef]
- Terakado, A.P.N.; Costa, R.B.; De Camargo, G.M.F.; Irano, N.; Bresolin, T.; Takada, L.; Carvalho, C.V.D.; Oliveira, H.N.; Carvalheiro, R.; Baldi, F.; et al. Genome-wide association study for growth traits in Nelore cattle. Animal 2018, 12, 1358–1362. [Google Scholar] [CrossRef]
- Martins, R.; Machado, P.C.; Pinto, L.F.B.; Silva, M.R.; Schenkel, F.S.; Brito, L.F.; Pedrosa, V.B. Genome-wide association study and pathway analysis for fat deposition traits in nellore cattle raised in pasture-based systems. J. Anim. Breed. Genet. 2021, 138, 360–378. [Google Scholar] [CrossRef]
- Novianti, I. Molecular Genetics of Cattle Muscularity; The University of Adelaide: Adelaide, Australia, 2011. [Google Scholar]
- Pausch, H.; Emmerling, R.; Schwarzenbacher, H.; Fries, R. A multi-trait meta-analysis with imputed sequence variants reveals twelve QTL for mammary gland morphology in Fleckvieh cattle. Genet. Sel. Evol. GSE 2016, 48, 14. [Google Scholar] [CrossRef]
- Riley, D.G.; Mantilla-Rojas, C.; Miller, R.K.; Nicholson, K.L.; Gill, C.A.; Herring, A.D.; Riggs, P.K.; Sawyer, J.E.; Savell, J.W.; Sanders, J.O. Genome association of carcass and palatability traits from Bos indicus-Bos taurus crossbred steers within electrical stimulation status and correspondence with steer temperament 3. Aroma and flavor attributes of cooked steaks. Livest. Sci. 2020, 233, 103943. [Google Scholar] [CrossRef]
- Cole, J.B.; Wiggans, G.R.; Ma, L. Genome-wide association analysis of thirty one production, health, reproduction and body conformation traits in contemporary U.S. Holstein cows. BMC Genom. 2011, 12, 408. [Google Scholar] [CrossRef]
- Cao, X.R.; Lill, N.L.; Boase, N.; Shi, P.P.; Croucher, D.R.; Shan, H.; Qu, J.; Sweezer, E.M.; Place, T.; Kirby, P.A.; et al. Nedd4 controls animal growth by regulating IGF-1 signaling. Sci. Signal. 2008, 1, ra5. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Pan, Y.; He, H.; Zheng, H.; Chen, P.; Li, G.; Xu, G.; Yu, S. Analysis of characteristics of yak PGF gene and detection of expression of the gene in different blastocysts. Chin. Vet. Sci. 2016, 46, 1195–1200. [Google Scholar]
- Seidenspinner, T.; Tetens, J.; Habier, D.; Bennewitz, J.; Thaller, G. The placental growth factor ( PGF)—A positional and functional candidate gene influencing calving ease and stillbirth in German dairy cattle. Anim. Genet. 2011, 42, 22–27. [Google Scholar] [CrossRef]
- Li, B.; Barden, M.; Kapsona, V.; Sánchez-Molano, E.; Anagnostopoulos, A.; Griffiths, B.E.; Bedford, C.; Dai, X.; Coffey, M.; Psifidi, A.; et al. Single-step genome-wide association analyses of claw horn lesions in Holstein cattle using linear and threshold models. Genet. Sel. Evol. GSE 2023, 55, 16. [Google Scholar] [CrossRef]
- Pérez-Cabal, M.A.; Charfeddine, N. Short communication: Association of foot and leg conformation and body weight with claw disorders in Spanish Holstein cows. J. Dairy Sci. 2016, 99, 9104–9108. [Google Scholar] [CrossRef]
- Michenet, A.; Saintilan, R.; Venot, E.; Phocas, F. Insights into the genetic variation of maternal behavior and suckling performance of continental beef cows. Genet. Sel. Evol. GSE 2016, 48, 45. [Google Scholar] [CrossRef]
- Schmidt, K.T.; Weinshenker, D. Adrenaline rush: The role of adrenergic receptors in stimulant-induced behaviors. Mol. Pharmacol. 2014, 85, 640–650. [Google Scholar] [CrossRef]
- Nazar, M.; Abdalla, I.M.; Chen, Z.; Ullah, N.; Liang, Y.; Chu, S.; Xu, T.; Mao, Y.; Yang, Z.; Lu, X. Genome-Wide Association Study for Udder Conformation Traits in Chinese Holstein Cattle. Animals 2022, 12, 2542. [Google Scholar] [CrossRef]
- Sinha, R.; Sinha, B.; Kumari, R.; MR, V.; Sharma, N.; Verma, A.; Gupta, I.D. Association of udder type traits with single nucleotide polymorphisms in Sahiwal (Bos indicus) and Karan Fries (Bos taurus × Bos indicus) cattle. Anim. Biotechnol. 2023, 34, 2745–2756. [Google Scholar] [CrossRef] [PubMed]
- Tribout, T.; Croiseau, P.; Lefebvre, R.; Barbat, A.; Boussaha, M.; Fritz, S.; Boichard, D.; Hoze, C.; Sanchez, M.P. Confirmed effects of candidate variants for milk production, udder health, and udder morphology in dairy cattle. Genet. Sel. Evol. GSE 2020, 52, 55. [Google Scholar] [CrossRef] [PubMed]
- Nemcova, E.; Stipkova, M.; Zavadilová, L.; Bouska, J.; Vacek, M. The relationship between somatic cell count, milk production and six linearly scored type traits in Holstein cows. Czech J. Anim. Sci. 2007, 52, 437–446. [Google Scholar] [CrossRef]
- Varona, L.; Moreno, C.; Altarriba, J. Genetic correlation of longevity with growth, post-mortem, docility and some morphological traits in the Pirenaica beef cattle breed. Animal 2012, 6, 873–879. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).