Abstract
(1) Background: Following the discovery of the adipokine/hormone asprosin, a substantial amount of research has provided evidence for its role in the regulation of glucose homeostasis, as well as appetite, and insulin sensitivity. Its levels are dysregulated in certain disease states, including breast cancer. To date, little is known about its role in endometrial cancer (EC). The present study investigated the effects of asprosin on the transcriptome of the Ishikawa and NOU-1 EC cell lines, and assessed the expression of asprosin’s candidate receptors (TLR4, PTPRD, and OR4M1) in health and disease. (2) Methods: tissue culture, RNA extraction, RNA sequencing, reverse transcription-quantitative PCR, gene enrichment and in silico analyses were used for this study. (3) Results: TLR4 and PTPRD were significantly downregulated in EC when compared to healthy controls. TLR4 appeared to have a prognostic role in terms of overall survival (OS) in EC patients (i.e., higher expression, better OS). RNA sequencing revealed that asprosin affected 289 differentially expressed genes (DEGs) in Ishikawa cells and 307 DEGs in NOU-1 cells. Pathway enrichment included apoptosis, glycolysis, hypoxia, and PI3K/AKT/ mTOR/NOTCH signalling for Ishikawa-treated cells. In NOU-1, enriched processes included inflammatory response, epithelial-mesenchymal transition, reactive oxygen species pathways, and interferon gamma responses. Other signalling pathways included mTORC1, DNA repair, and p53, amongst others. (4) Conclusions: These findings underscore the importance of understanding receptor dynamics and signalling pathways in the context of asprosin’s role in EC, and provide evidence for a potential role of TLR4 as a diagnostic biomarker.
1. Introduction
Endometrial cancer (EC) is the most common gynaecological malignancy in developed countries, with its incidence rising significantly over the last two decades. This increase is largely attributed to an ageing population and escalating obesity rates. Systematic reviews and meta-analyses have identified key risk factors for EC development, including elevated body mass index (BMI), diabetes mellitus, polycystic ovary syndrome (PCOS), and nulliparity [1,2,3]. The histopathologic types of ECs according to WHO [4] are endometrioid carcinoma (i.e., adenocarcinoma; adenocarcinoma-variants), mucinous adenocarcinoma, serous adenocarcinoma, clear cell adenocarcinoma, undifferentiated carcinoma, and neuroendocrine tumours. Approximately 70–80% of ECs are of endometrioid subtype, whilst 10% are serous [5].
In terms of its molecular classification, the European Society of Gynaecological Oncology (ESGO), the European Society for Radiotherapy and Oncology (ESTRO) and the European Society of Medical Oncology (ESMO) sought to stratify patients diagnosed with EC by a different prognostic approach [6]. Prognostic features such as depth of myometrial invasion, lymphovascular space invasion (LVSI), and histological subtype have been incorporated into a risk stratification system, which has subsequently been updated in 2020 with the option of adding molecular data to best reflect differences in prognosis, particularly for those with early-stage disease [7].
Recently, we have shown that the components of metabolic syndrome (MetS), both independently and in combination, significantly increase the risk of EC [8]. Asprosin is a newly discovered adipokine that plays a role in regulating glucose metabolism/insulin sensitivity, and appetite, whilst it is dysregulated in cardiometabolic diseases [8]. For example, higher circulating levels of asprosin were measured in females with obesity than in females with normal BMI, while decreased circulating asprosin levels were noted in females 6 months following bariatric surgery [9]. Similarly, Ugur et al. (2022) studied volunteers of all BMI categories from underweight to class 3 obesity and demonstrated elevated circulating asprosin levels amongst patients with obesity in both serum and saliva [10].
Asprosin’s effects are mediated through different receptors. To date, the primary receptors that have been investigated are the Olfactory Receptor Family 4 Subfamily M Member 1 (OR4M1), Toll Like Receptor 4 (TLR4) and Protein Tyrosine Phosphatase Receptor Delta (PTPRD), though limited evidence for a bona fide asprosin receptor exists [11,12,13,14]. Currently, little is known about the effects of asprosin in EC. In this study, we have measured gene and protein expression of asprosin receptors in EC preclinical models and clinical samples, and assessed their prognostic role. We have also investigated the effects of asprosin on the transcriptome of Ishikawa and NOU-1 EC cell lines.
2. Results
2.1. Expression of Asprosin Receptors in Endometrial Cancer (EC) Cell Lines
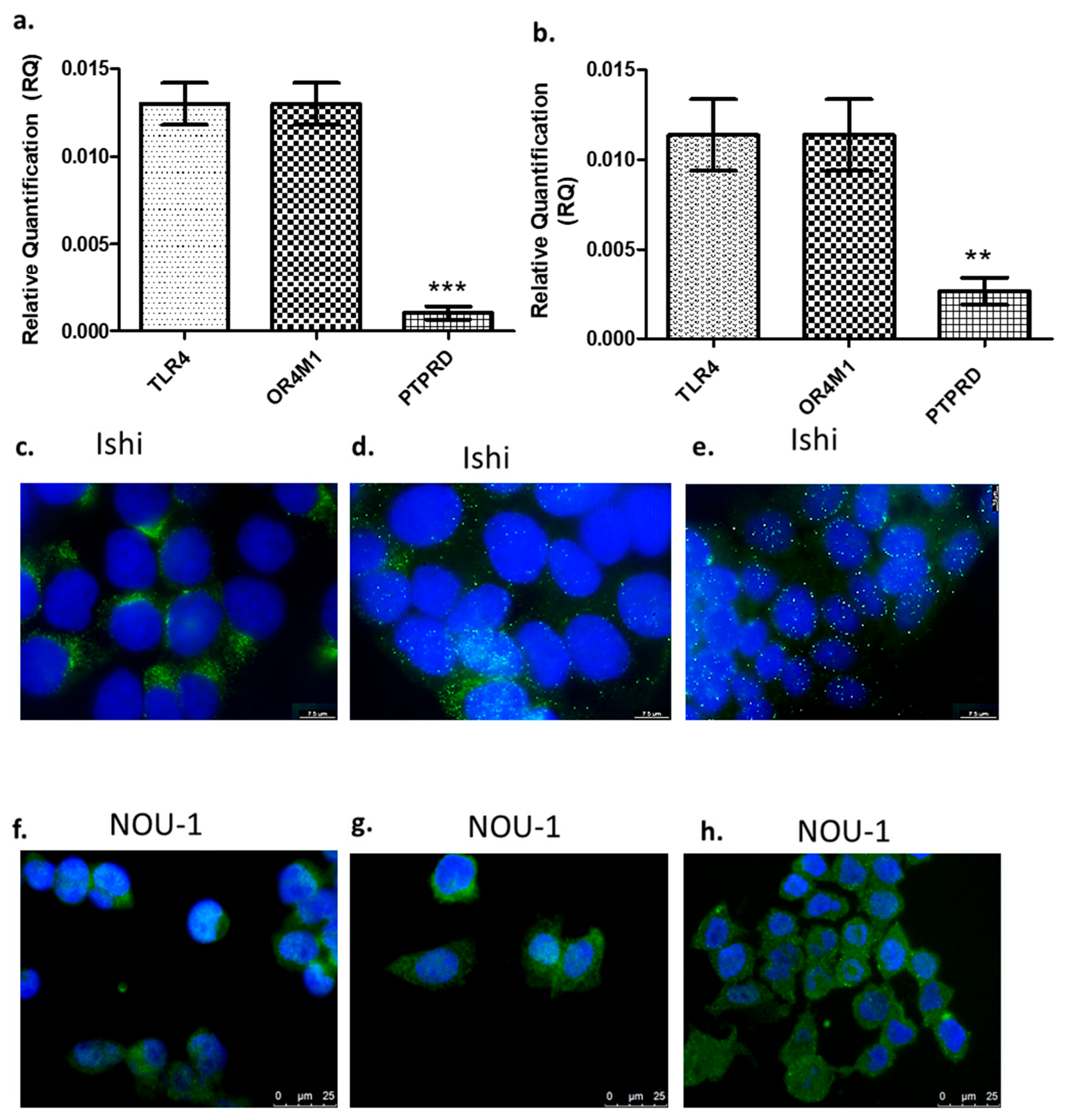
In this study, we have used two different EC cell lines, namely Ishikawa and NOU-1. RT-qPCR confirmed the expression of all asprosin’s putative receptors, i.e., TLR4, PTPRD and OR4M1 (Figure 1a,b). In both cell lines, PTPRD’s expression was significantly lower when compared to TLR4 or OR4M1. Immunofluorescence analysis was also conducted to assess the protein expression of these receptors. A strong cytoplasmic/membrane staining was evident for TLR4 for both cell lines (Figure 1c,f), whereas PTPRD had mainly a scattered cytoplasmic distribution in Ishikawa cells and more membranous in NOU-1 cells (Figure 1d,g). Finally, OR4M1 demonstrated mainly a membrane localisation in Ishikawa cells, in accordance with its role as a G protein-coupled receptor (GPCR; Figure 1e,h).
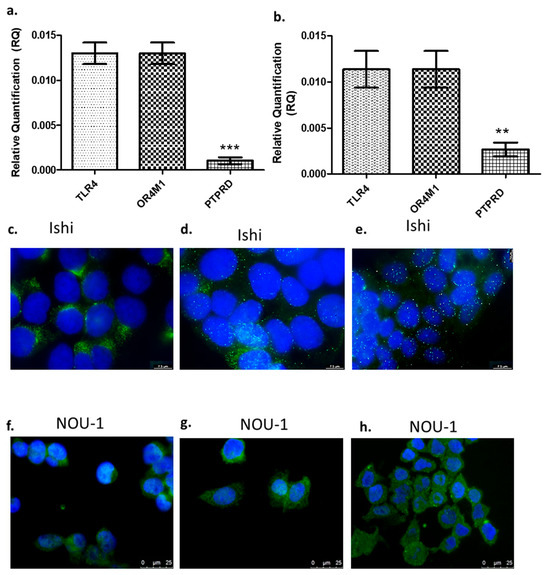
Figure 1.
Gene and protein expression of asprosin receptors. Gene expression of TLR4, PTPRD, and OR4M1 in Ishikawa (Ishi) cells (a) and in NOU-1 cells (b). The relative quantification (RQ) was calculated using the delta Ct (ΔΔCt) method. Protein expression of TLR4, PTPRD and OR4M1 in Ishikawa (Ishi; (c–e)) and NOU-1 cells (f–h). For immunofluorescence, we have used TLR4 (PA5-23124), OR4M1 (NBP2-46853) and PTPRD (NBP2-49153) primary rabbit antibodies. Anti-rabbit Alexa Fluor 488 was used as a secondary antibody (green), and the DAPI nuclear stain (blue) was used to stain the nuclei. Significance was calculated using one-way ANOVA. *** p < 0.0001, ** p < 0.001.
2.2. Differential Expression of TLR4, PTPRD, and OR4M1 in EC: Potential Role of TLR4 as a Biomarker
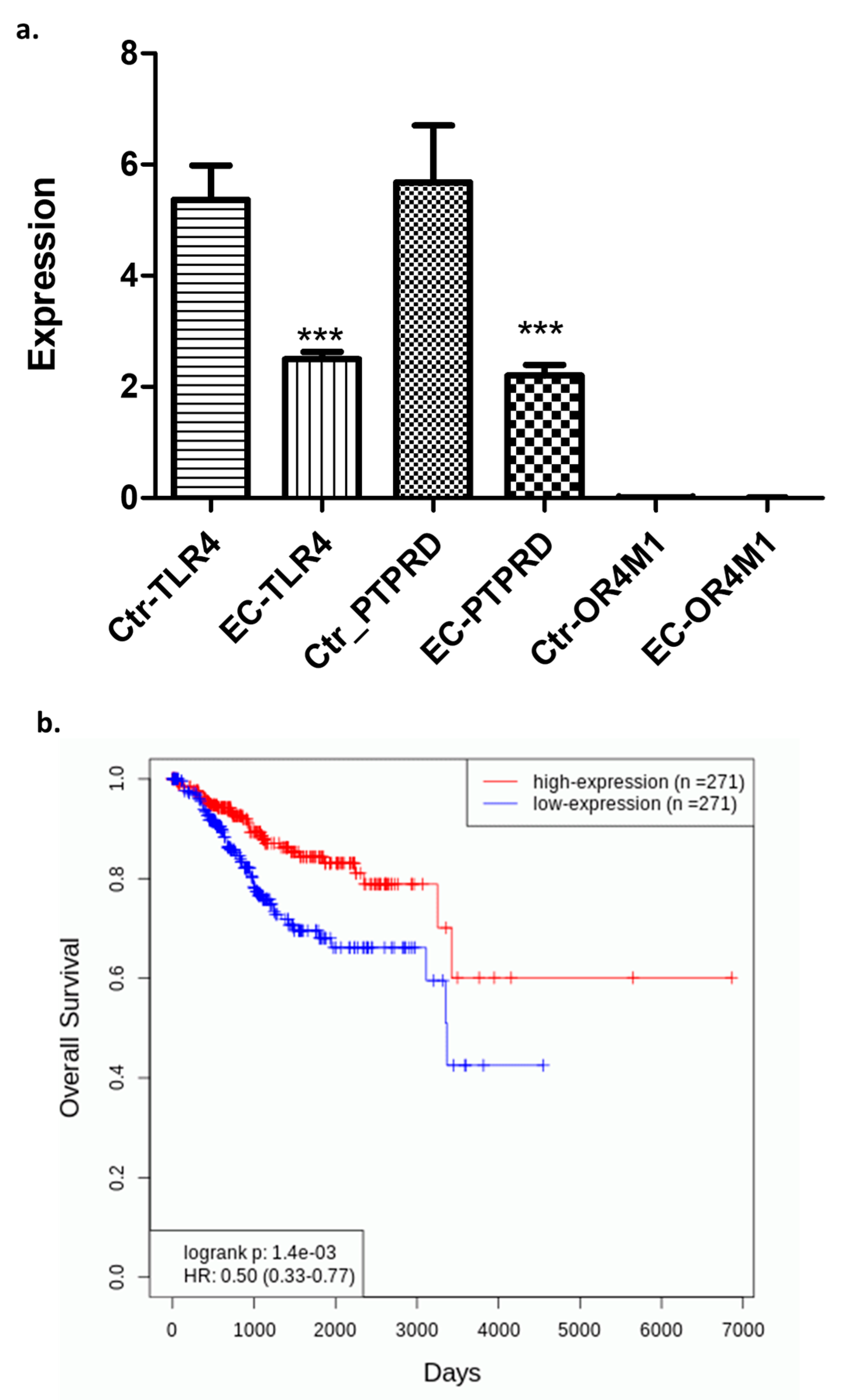
Curating data from the database OncoDb (TCGA and GTEx), we have assessed the expression of the above-mentioned receptors in EC patients (n = 557) and healthy controls (n = 35) (Figure 2a). TLR4 was significantly downregulated in EC patients when compared to controls (p < 0.0001). Similarly, PTPRD was also significantly downregulated in EC patients when compared to controls (p < 0.0001). Very little data were available for OR4M1 to draw any meaningful conclusions, but in general, very low expression was noted in both groups (0.01 to 0.06). Out of all three putative asprosin receptors, only TLR4 appeared to have a prognostic role in terms of overall survival (OS) in EC patients (Figure 2b). Patients with higher expression of TLR4 had higher OS (n = 271), when compared to patients with low TLR4 expression (n = 271; p = 0.0014).
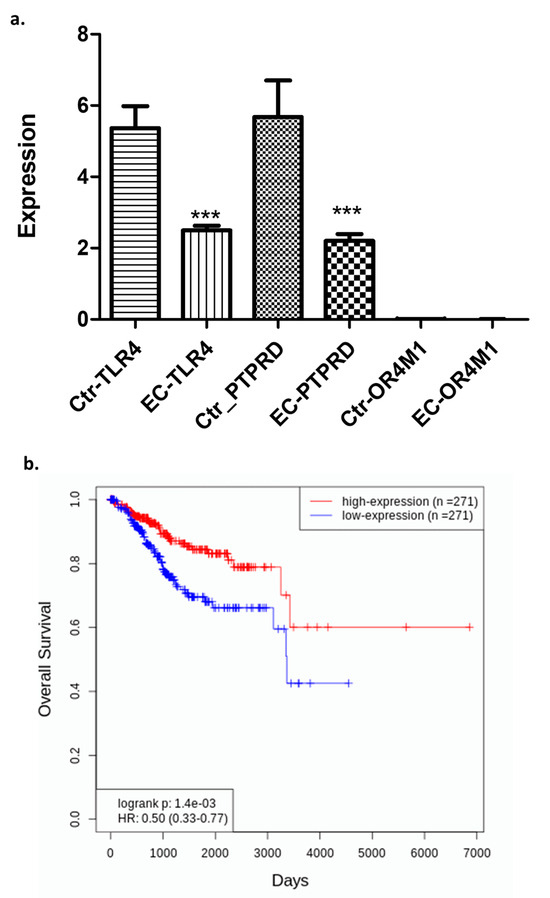
Figure 2.
Expression of the TLR4, PTPRD, and OR4M1 asprosin receptors in EC patients (n = 557) and healthy controls (n = 35). (a) Downregulation of TLR4 and PTPRD, but not OR4M1, in EC patients compared to controls (*** p < 0.0001); (b) EC patients expressing higher levels of TLR4 (n= 271) have better overall survival compared to EC patients with lower TLR4 expression (n = 271) (p = 0.0014).
2.3. Transcriptomic Changes of Ishikawa and NOU-1 Cells Treated Wth Asprosin
Initially, RNA sequencing analysis was performed on asprosin-treated (10 nM for 4 h) Ishikawa cells and compared to non-treated controls. We have chosen this concentration and time-point based on previous studies from our groups demonstrating that asprosin can induce signalling pathways and transcriptomic changes at these points [13,15].
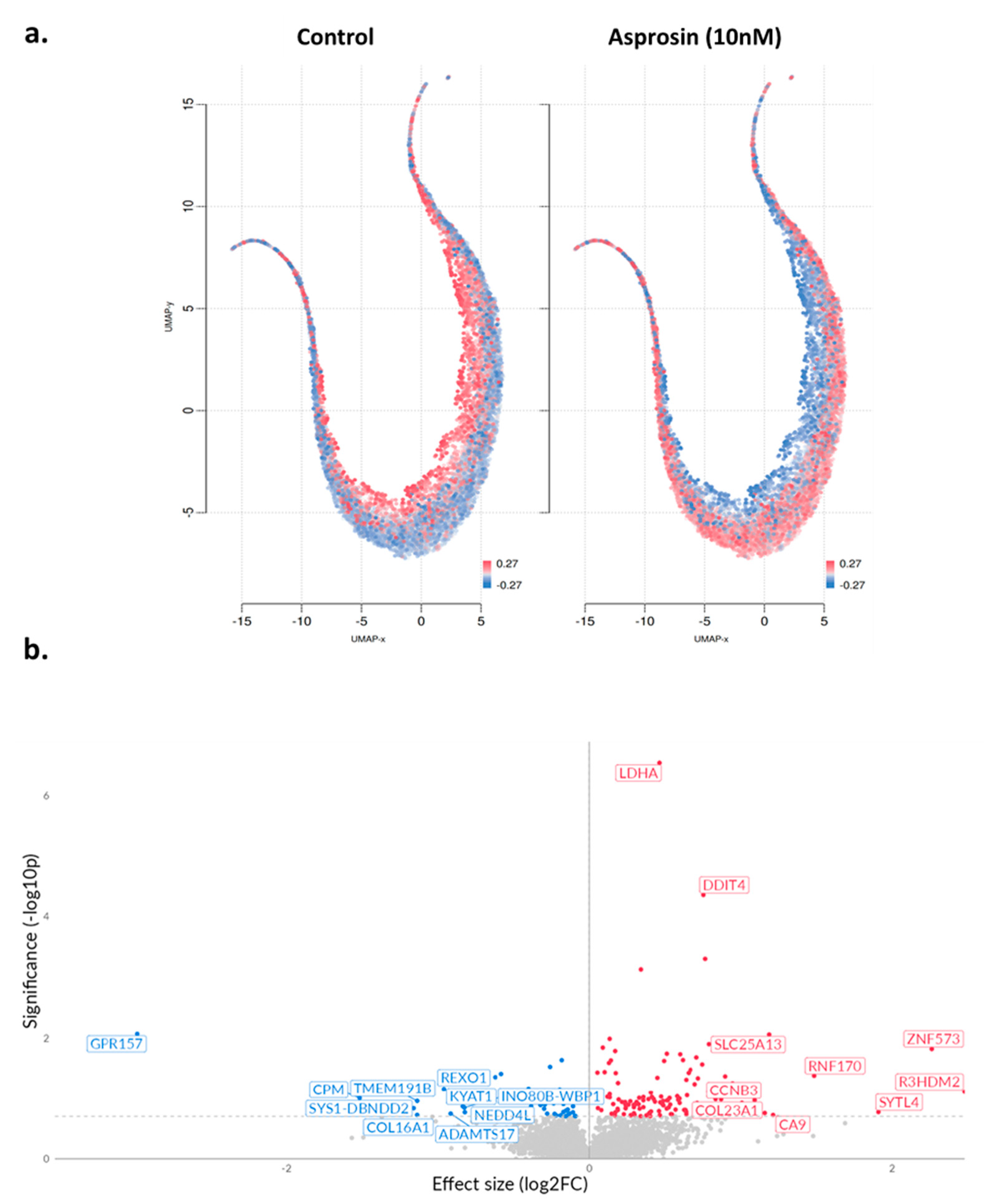
A total of 289 differentially expressed genes (DEGs) were identified, of which 186 were up-regulated and 103 down-regulated (Table A1). Figure 3a provides an overview of UMAP clustering coloured by relative log-expression of genes between the different phenotypic groups (−0.27 to 0.27 Z-score of median transcripts per million; TPM). Subsequently, a volcano plot was generated indicating log fold change (log2FC) between treated and untreated cells (Figure 3b).
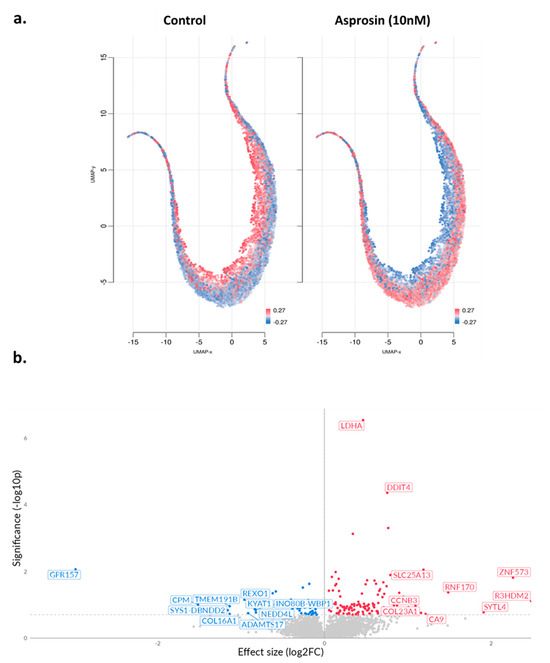
Figure 3.
UMAP projections and volcano plot in asprosin-treated Ishikawa cells. (a) UMAP projections of Ishikawa cells under two conditions: untreated control and asprosin-treated. Each point corresponds to an individual gene, and red colour indicates higher gene expression, whereas blue indicates lower expression. (b) Volcano plot of differentially expressed genes (DEGs) in asprosin-treated Ishikawa cells relative to untreated controls. Upregulated genes with positive log2FC are shown in red, downregulated genes are shown in blue, and non-significant genes are depicted in grey. Selected top hits (e.g., LDHA, DDIT4, GPR157) are labelled. The dashed horizontal line denotes the p-value threshold (p < 0.05).
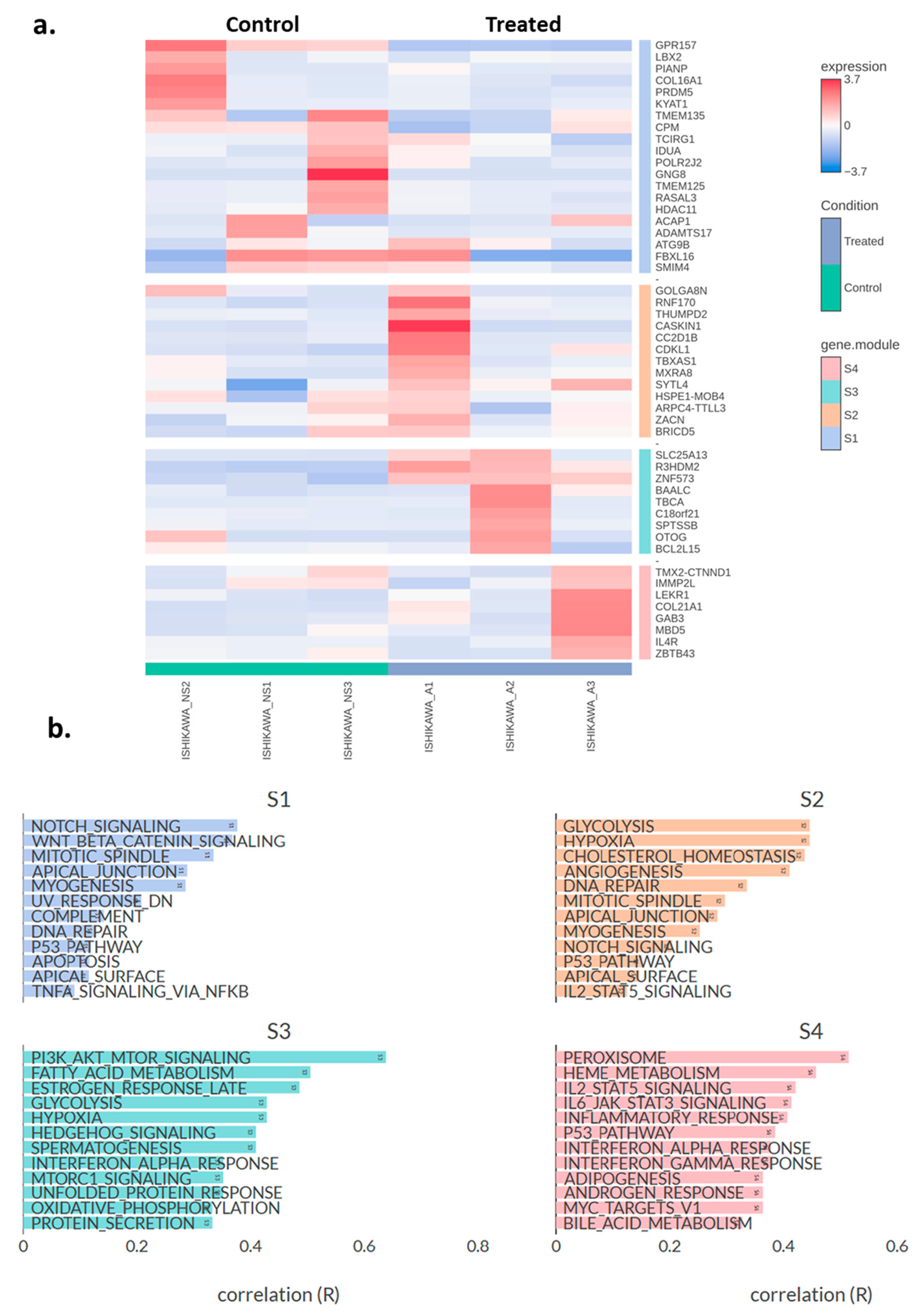
Subsequently, hierarchical clustering was performed on the top 50 DEGs (Ishikawa asprosin-treated vs. control (no supplement; NS)), and the genes were grouped into four distinct clusters S1–S4 (Figure 4a). Under S1 cluster, the most prominent functional annotations related to Notch, and Wnt/Beta Catenin signalling, as well as mitotic spindle, apical junction and myogenesis (Figure 4b). In the S2 cluster, main annotations include glycolysis, hypoxia, cholesterol homeostasis and angiogenesis, whereas in S3, the most enriched annotations include PI3K_AKT_mTOR signalling, fatty acid metabolism, and estrogen response. Finally, the S4 cluster included peroxisome, heme metabolism, IL2_STAT5 and IL6_JAK_STAT3 signalling pathways (Figure 4b).
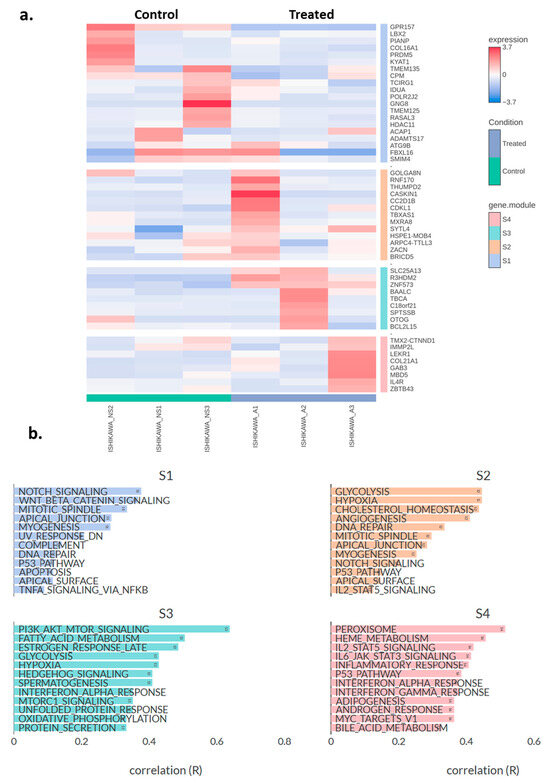
Figure 4.
(a) Heatmap showing the top 50 differentially expressed genes in asprosin-treated Ishikawa cells (blue-labelled samples) compared to untreated controls (green-labelled samples). The colour gradient indicates standardised expression levels, ranging from low (blue, −2.5) to high (red, +3.7). Hierarchical clustering was performed on both genes and samples, with gene clusters (S1–S4) labelled on the right. A: asprosin-treated, NS: no supplement. (b) Pathway correlation analysis for the four gene clusters (S1, S2, S3, and S4). Each bar represents the correlation coefficient (R) between a given hallmark gene set and the module’s gene expression profile. Higher values of R indicate stronger associations between the module and that particular pathway. Ishikawa_A1-A3 and Inshikawa_NS1-3 refer to treated and control samples, respectively (n = 3 per group).
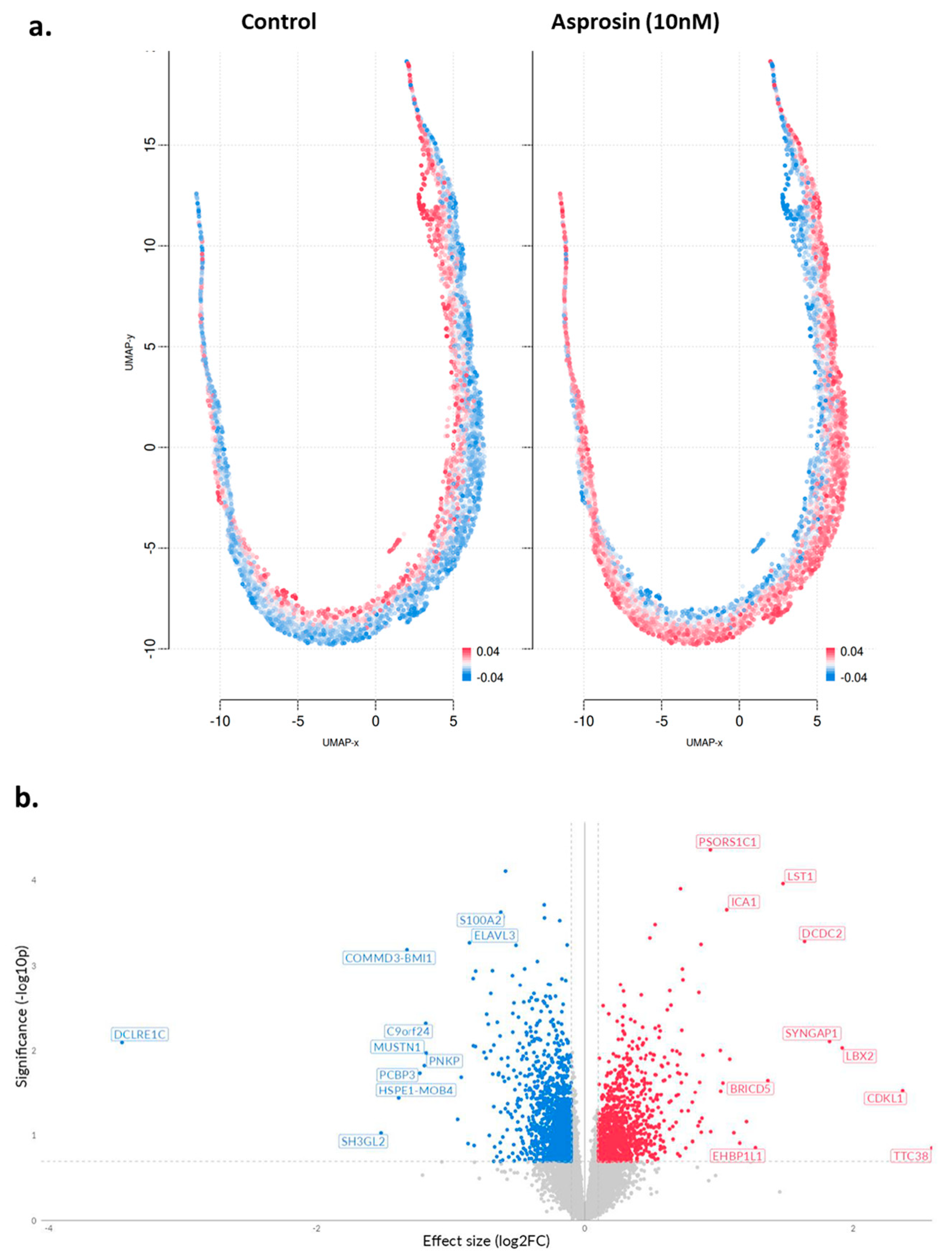
Similar analyses were performed in NOU-1 cells, where 307 DEGs were identified, 202 of which were up-regulated and 105 down-regulated (Table A2). UMAP clustering of genes coloured by relative log-expression of genes between the different groups provided some interesting insights. The treated cells show a distinct shift in expression profiles compared to the control, with clusters of cells exhibiting higher expression (more red regions; Figure 5a). In addition, the volcano plot indicates log2FC changes between asprosin-treated and untreated (control) NOU-1 cells (Figure 5b).
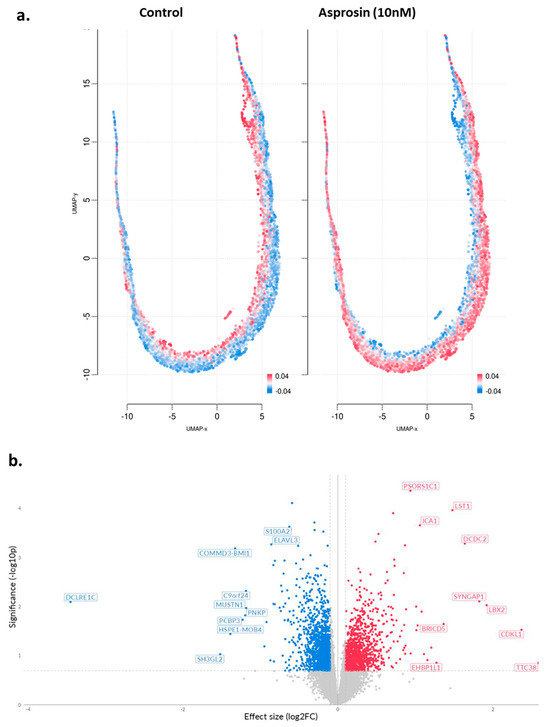
Figure 5.
(a) UMAP projections of asprosin-treated NOU-1 cells; (b) Volcano plot of DEGs in asprosin-treated (10 nM for 4 h) cells when compared to untreated controls. Genes that are significantly upregulated are highlighted in red, whereas significantly downregulated genes are highlighted in blue. Each point represents a single gene; labelled genes (e.g., COMMD3, BMI1, DCDC2, LST1, LBX2) are among the most significantly altered. The dashed vertical lines mark a 0.1 effect size cutoff, while the dashed horizontal line denotes the p-value threshold (p < 0.05).
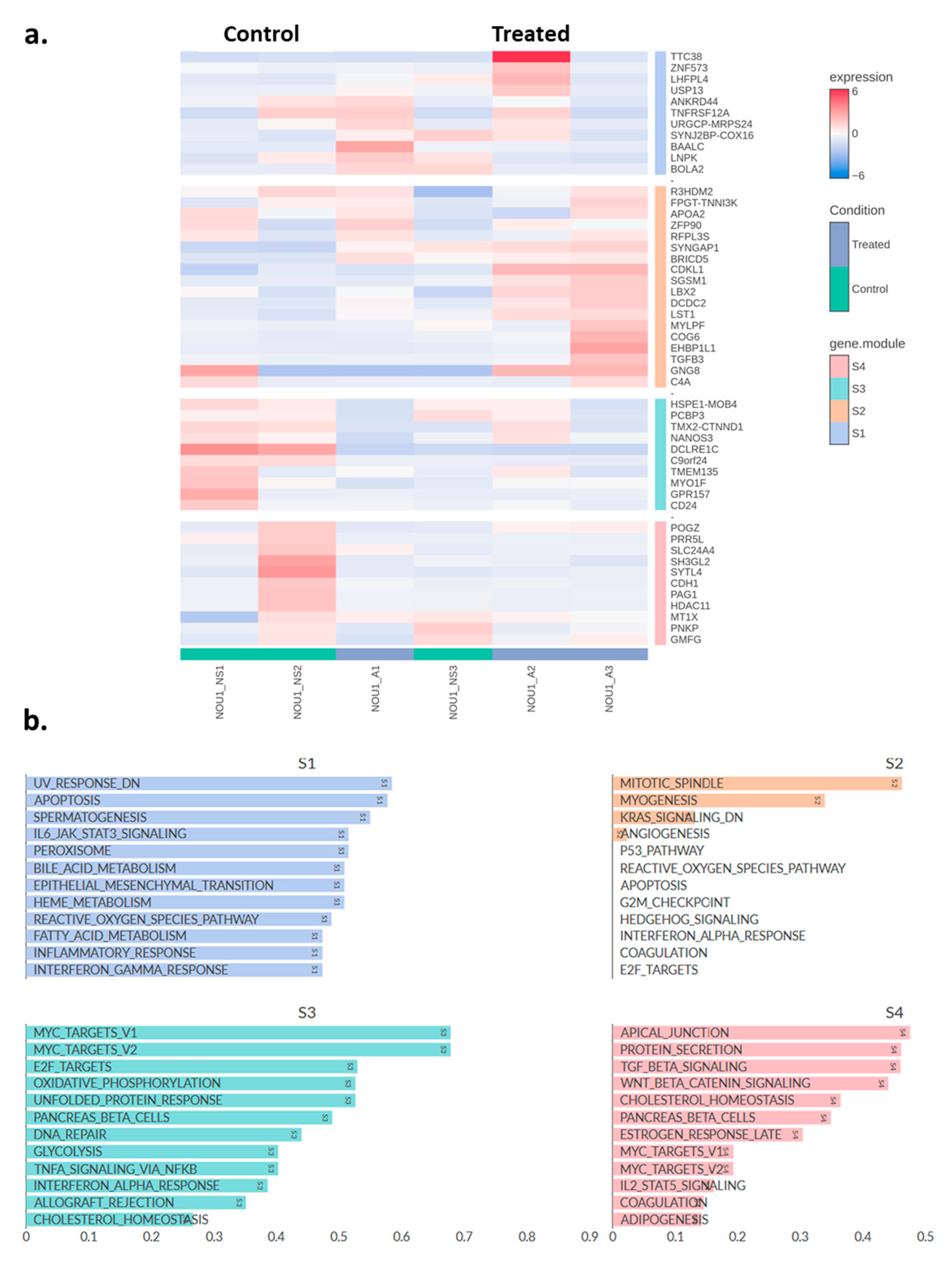
Using a similar methodology as for the Ishikawa cells, a heatmap of the top 50 DEGs in NOU-1 cells was generated, distributing these genes in four different clusters (Figure 6a). Figure 6b depicts pathway enrichment analysis for four gene clusters (S1–S4), highlighting biological processes associated with asprosin treatment. In S1, pathways such as the inflammatory response, epithelial-mesenchymal transition (EMT), reactive oxygen species (ROS) pathways, and interferon gamma response are enriched. These processes suggest asprosin’s involvement in stress responses, immune signalling, and cellular plasticity, potentially influencing the tumour microenvironment. In S2, key pathways include KRAS signalling, myogenesis and mitotic spindle functioning, indicating that asprosin might affect cell cycle regulation, cancer-related signalling, and vascularisation, all of which are critical for tumour growth and progression. S3 reveals enrichment in MYC target pathways, oxidative phosphorylation, DNA repair, and glycolysis. These pathways are fundamental to cancer metabolism, energy production, and genomic stability, suggesting that asprosin may play a role in metabolic reprogramming and enhancing cellular survival mechanisms. Lastly, S4 is enriched in pathways such as TGF-beta signalling, WNT-beta catenin signalling, protein secretion, and cholesterol homeostasis. These pathways implicate asprosin in intercellular communication, lipid metabolism, and processes associated with cancer stemness and metastasis.
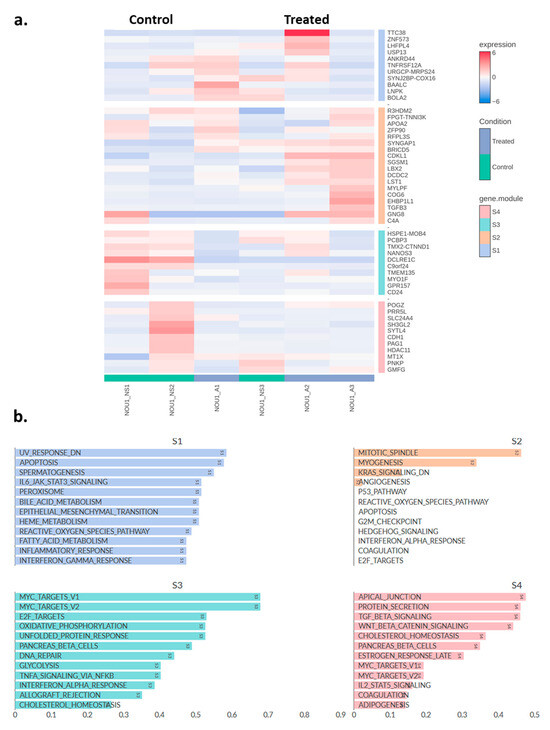
Figure 6.
(a) Heatmap showing the top 50 differentially expressed genes in asprosin-treated NOU-1 cells (blue-labelled samples) compared to untreated controls (green-labelled samples). Hierarchical clustering was performed on both genes (rows) and samples (columns). A: asprosin-treated, NS: no supplement. (b) Pathway correlation analysis for the four gene clusters (S1, S2, S3, and S4) identified in NOU-1 cells treated with asprosin. NOU1_A1-A3 and NOU1_NS1-3 refer to treated and control samples, respectively (n = 3 per group).
3. Discussion
In this study, we provide novel evidence that the human endometrium and two preclinical in vitro models (i.e. Ishikawa and NOU-1 EC cell lines) expresses all three known asprosin receptors (OR4M1, TLR4 and PTPRD), with TLR4 and PTPRD being downregulated in EC. Notably, due to limited data for OR4M1 in EC, it is challenging to draw any conclusions at this stage. We also offer a novel insight into the regulation EC transcriptomes by asprosin in vitro. Although there is not a bona fide asprosin receptor, PTPRD has been identified as a potential orexigenic receptor for asprosin in hypothalamic AgRP neurons, since genetic ablation of PTPRD leads to loss of appetite, resistance to diet-induced obesity, and lack of response to asprosin [11]. Similarly, asprosin appears to impair insulin secretion in response to glucose in human primary islets containing β-cells via a TLR4 pathway [12]. We have also shown that asprosin exerts pro-inflammatory effects in THP-1 macrophages in vitro, via activation of TLR4 [13]. Another candidate receptor for asprosin is the olfactory receptor OR4M1, since it can promote gluconeogenesis and maintains glucose homeostasis via this GPCR [14]. Of note, despite the differences on the gene expression of asprosin receptors in our in vitro models, substantial expression was evident when immunofluorescence was performed. Although the protein expression was not quantitative, these discrepancies can arise from the fact that transcription not always is reflected in translation, given the complexity of the regulation steps that need to be followed, especially in cancer [16].
All these three receptors have been shown to be differentially expressed in certain cancers. We have previously shown that PTPRD is dysregulated in patients with glioblastoma and in EC, whilst its expression is significantly downregulated in patients with obesity [17]. In gastric cancers, loss of PTPRD induced CXCL8 and promoted angiogenic and metastatic events, via STAT3 and ERK signalling pathways [17]. PTPRD was also shown to be involved in colon cancer cell migration via a β-catenin/TCF/CD44 signalling pathway, whereas in lung cancer PTPRD appears to act as a tumour suppressor gene [18,19,20]. Notably, PTPRD is mutated in 11.1% of endometrial samples. In a GWAS meta-analysis, 13 loci were associated with EC and endometriosis, with one particular locus located within the PTPRD gene [21]. Collectively, these data point towards a central role of PTPRD not only as a potential tumour suppressor gene, but also as an orexigenic mediator in EC.
Toll-like receptors (TLRs) are pattern-recognition receptors that detect ligands and initiate downstream signalling involving a number of adaptor molecules (e.g. MyD88) that upon recruitment drives activation of transcription factors (e.g. NF-κB) [22]. For example, TLR4 activation can result in NF-κB translocation to the nucleus, modulating transcription of COX-2, a gene related to inflammation [23]. In the same study it was shown that LPS (a ligand for TLR4) induced a TLR4-dependent stimulation of p38, ERK1/2, and JNK. Of note, inhibition of JNK induced NF-κB activity and expression of COX-2. Recently, using an in vitro EC model, it was demonstrated that heme metabolism reduces phagocytosis by modulating the secretion of TLR4-mediated IFN Iα as well as CD36 expression; contributing to events leading to immune escape in this cancer [24]. The authors have suggested that an imbalanced immune microenvironment promotes EC progression. Previous studies have also implicated TLR4 in EC. For example, when Ishikawa cells were treated with fusaric acid, it led to a decrease in cell proliferation by compromising the expression of TLR4 [25]. Of note, a TLR4 polymorphism (rs4986790), did not appear to be associated with EC risk [26].
Here, we show that TLR4 is downregulated in EC; a finding that corroborates initial studies, where TLR4 was significantly downregulated in endometrial hyperplasia and adenocarcinoma when compared with controls (i.e. postmenopausal women) [27]. Moreover, we have demonstrated that TLR4 appears to have a potential prognostic role, since EC patients with higher expression have better overall survival. Similarly, TLR4 appears to have a prognostic role in ovarian cancer progression [28]. However, it should be noted that in a meta-analysis study, it was suggested that increased TLR4 expression is associated with poor OS in patients with solid tumours [29]. Indeed, the finding that high TLR4 expression correlates with better overall survival is intriguing. In a review article by Lupi et al, the complexity of TLR4 signalling in gynecological cancers was discussed at length [30], where the authors suggested that TLR4 can have a dual role exerting both anti- and pro-tumour responses, depending on the pathway it activates. For example, in most cancers, uncontrolled TLR4-mdiatd signalling tips the tumor microenvironment (TME) towards a proliferative status, and evasion of immune surveillance; involving secretion of proinflammatory cytokines and other molecules. On the other hand, activation of TLR4 can enhance an immune response (e.g. T lymphocytes) that will result to inhibition of cancer cell proliferation. Therefore, more research is needed to provide a better insight into the TLR4 pathway(s) in EC and whether its expression can have any clinical utility as a biomarker. However, there is a higher order of complexity when it comes to the ligand binding and TLR4 signalling. Given the multiple ligands that can bind to and activate TLR4, future studies should also elucidate if activation of TLR4 by asprosin can exert anti-proliferative effects and if it acts in TME-specific manner (i.e. well-differentiated vs. poorly-differentiated EC).
Olfactory receptors can detect a wide range of odorants, as well as numerous endogenous ligands. Over the past years, a growing body of studies has pointed towards involvement of olfactory receptors in a number of diseases, including infectious diseases (e.g. COVID-19), neurological diseases, metabolic diseases and cancer [31,32]. Previous work from our group has also shown that OR4M1 is expressed in the ovaries and is upregulated in early stages of ovarian cancer (I and II) compared to late ones (III and IV) [33]. In the current study, there were limited data available to draw any conclusions regarding the expression or its prognostic value in EC. However, if asprosin influences cAMP signalling through OR4M1, cAMP-Response Element Binding Proteins (CREB) could become activated. CREB binds cAMP-response elements in promoter regions to regulate transcriptional events [34]. A possibility of its low expression, might also have to do with the fact that it belongs to the GPCR family. Indeed, since OR4M1 is a GPCR, it is possible that chronic stimulation by asprosin might lead to desensitization and internalization of this receptor. This reduces the receptor availability on the cell surface and may, in turn, lower mRNA levels which would lead to temporary loss of function.
In the present study, treatment of two in vitro models has provided a novel insight into the regulation of EC by asprosin. The top biomarkers of asprosin-exposure in Ishikawa cells include: GPR157, ZNF573, R3HDM2, TMEM191B, CA9, SLC24A4, CDK11A, CXCR4. Although there is no data on ZNF573 and EC, it appears to have a diagnostic role for ovarian cancer in combination with other biomarkers [35]. Another upregulated gene is CA9, a hypoxia-responsive gene that is upregulated in the hypoxic tumour microenvironment [36]. Overexpression of CA9 correlates with tumour progression, metastasis, and poor prognosis in cancers including renal carcinomas, cervical squamous carcinomas, oesophageal carcinomas, bladder carcinomas and non-small cell lung carcinomas [37]. Asprosin might drive the increase of CA9 expression via induction of pro-inflammatory cytokines (e.g., TNF-α, IL-6), which can upregulate hypoxia-response genes, including CA9, even under normoxic conditions. In contrast, asprosin downregulated GPR157 and SLC24A4. Of note GPR157 has been identified as a potential biomarker for endometrioid endometrial carcinoma [38]. In addition, SLC24A4 was downregulated in colon carcinoma cells; it markedly increased their migration potential [39]. It is possible therefore, that asprosin might influence cell invasion/migration of EC cells via downregulation of such genes. There is some evidence that olfactory receptors can indeed internalize, in similar fashion to other GPCRs, involving GPCR specific kinases (GRKs), and β-arrestin [40]. For example, odorants can induce GRK3 translocation to the cell membrane that can mediate olfactory desensitization [41]. In addition, Lefkowitz’s group has shown that when GRK3 was disrupted, the odorant receptor-mediated desensitization was compromised [42]. Finally, Mashukova et al., argued that prolonged exposure of Hana3A cells to odorants, led to β-arrestin 2 accumulation, within intracellular vesicles [43].
It should be noted that, although DDIT-4 was not featured as one of the top differentiating biomarkers of asprosin treatment it was one of the highest overexpressed DEGs. DDIT4 is rapidly induced by various cellular stresses, including hypoxia, heat shock, or endoplasmic reticulum stress. The primary function of DDIT4 has been linked to its role in suppressing the mechanistic target of rapamycin complex 1 (mTORC1), a crucial regulator of cell growth, tumorigenesis, cell aging, and autophagy [44]. Indeed, in ovarian epithelial cells, increased expression of DDIT4 is associated with a decreased level of pro-apoptotic proteins and an elevation in anti-apoptotic protein levels, particularly when RAS oncogene activity is induced. Collectively, these findings suggest that DDIT4 plays a pivotal role in regulating cell survival and growth, particularly under stress conditions [44]. Of note, high DDIT4 expression correlating with favorable EC prognosis [45]. Contrary, upregulation of DDIT4 can stimulate cell proliferation in gastric epithelial cells [46] and high expression of DDIT4 correlates with more aggressive tumour behaviour and more advanced stages of disease in colorectal cancer patients [44]. Further research is needed to elucidate the relationship between asprosin and DDIT-4 in terms of its involvement in proliferative or apoptotic events in EC. In the present study, the “enriched” genes as biomarkers of asprosin exposure in NOU-1 cells were: DCDC2, BRICD5, PDE11A, PSORS1C1, ACAP1, and PRR11. PRR11 is a gene that plays a significant role in cell cycle progression and is often implicated in cancer progression [47], whilst it is associated with poor prognosis in a number of cancers [48]. For example, when PRR11 was knockdown in ovarian cancer, a decrease in tumour growth was noted [49]. Similarly, suppression of DCDC2 expression led to inhibition of cell proliferation and subsequent metastasis in colon cancer [50]. Table 1 highlights known relevance of top DEGs to EC.

Table 1.
Role of DEGs in EC.
Despite the robustness of the present findings, certain limitations of our study should be acknowledged. We have relied on using in silico data regarding the expression of TLR4, PTPRD and OR4M1 in health and disease (EC). Furthermore, we have assessed asprosin’s effects only in vitro, using two EC cell lines, representing different stages of differentiation. Ishikawa cell line was initially established from an endometrial adenocarcinoma from a 39-year-old female. According to ECACC, these cells induced well-differentiated adenocarcinoma in athymic nude mice. This cell line also expresses steroid receptors (estrogen and progesterone). One the other hand, the NOU-1 cell line is a poorly differentiated lethal human endometrial carcinoma cell line, that lacks both estrogen and progesterone receptors [55]. The properties of these cell lines might account for the differences in the transcriptomic landscape we have observed. To further elucidate the role of asprosin in EC and its broader implications in cancer biology, a number of detailed functional studies could be undertaken, particularly examining asprosin’s effects on cell proliferation, apoptosis and migration at different concentrations and time points. we acknowledge that lack of functional studies is a major limitation for this study. These processes are fundamental to cancer progression and metastasis, making them critical targets for understanding asprosin’s role in cancer pathogenesis.
4. Materials and Methods
4.1. Cell Culture
Endometrial cancer (EC) cells, Ishikawa, and NOU-1 cells were cultured using a complete medium of either Dulbecco’s Modified Eagle’s Medium (DMEM, Gibco, Bleiswijk, The Netherlands), High-Glucose Liquid Medium (Cytiva, Amersham, UK), or RPMI (Cytiva) as per the manufacturers’ instructions and supplemented with 10% Foetal Bovine Serum (FBS, Gibco, Bleiswijk, The Netherlands) and 1% penicillin-streptomycin (Gibco) at 37 °C with 5% CO2.
4.2. Immunofluorescence (IF)
In preparation for immunofluorescence (IF), an 8 mm coverslip was added to each well of a 6-well plate under a laminar flow cabinet. In the same method used for sub-culture, Ishikawa and NOU-1 cells were resuspended in complete media and incubated for 24 h or until they reached a confluence of approximately 80% on the coverslip. The 6-well plate was then removed from cell culture conditions. Media were aspirated, and cells were washed twice with 1 mL of PBS. Fixation of cells was undertaken using 4% paraformaldehyde (PFA) for five minutes. Repeat washings were undertaken by applying the solution away from the coverslip to avoid the detachment of cells. No permeabilisation of the cell was undertaken. Blocking was then undertaken using 100 μL of 1% bovine serum albumin (BSA) diluted in PBS in each well. Parafilm was used to prevent dehydration, and the slides were left for one hour at room temperature. Next, 100 μL of the primary antibody solution (TLR4, PTPRD, OR4M1 antibodies) diluted in 1% BSA in PBS was applied. Following incubation with the primary antibody, 1 mL of PBS was then used to wash the coverslips three times for five minutes each. The secondary antibody, anti-Rabbit Alexa Fluor 488 antibody (Merck Millipore, Watford, UK), was added to each well at a concentration of 1:200 and left for one hour in the dark at room temperature. A final three washes with PBS were then undertaken. Coverslips were removed from the six-well plate and allowed to air-dry. Glass slides were prepared with 5 μL of mounting medium with DAPI nuclear stain (Vectashield), and cover slides were inverted gently onto the mounting media and left for ten minutes to allow the mounting media to dry. Slides were sealed with clear nail varnish and left to air dry before viewing under a LEICA DM4000 Fluorescent Microscope. All IF analyses were carried out using the LAS-X analysis software (version 3.7.0).
4.3. In Silico Tools
The online database OncoDB [56] was accessed to determine expression PTPRD, TLR4 and OR4M1 in UCEC vs. controls, making use of TCGA and GTEx datasets. Enrichment analysis was also performed using Omics playground (v3.44, BigOmics Analytics, Bellinzona, Switzerland) for the function comparison of DEGs in asprosin-treated versus untreated Ishikawa and NOU-1 cells.
4.4. RNA Sequencing
RNA was extracted from three technical replicates from two cell lines, Ishikawa and NOU-1 (3 × no supplement control and 3 × asprosin treated), as previously described [17]. Briefly, indexed libraries were submitted to an Illumina NovaSeq (Novogene, Cambridge, UK). Files were compiled using lllumina package bcl2fastq to convert the base call (BCL) binary results to FASTQ. All samples passed internal quality control before sequencing.
The freely available SSH, PuTTY that connects users to the Linux OS was used in the initial steps of data processing. Three programs, Bowtie, TopHat and Cufflinks [57]. As mentioned, using the BigOmics Analytics platform, CSV files were uploaded to the platform to begin analysis. The clustering module performs a holistic clustering analysis of the samples. The main output of this feature is 2-fold: (i) to generate a heatmap of samples and also to provide a plot of samples obtained by principal components analysis (PCA) or t-distributed stochastic embedding algorithms [58,59,60]. The R program ggplot2 was used to better explore and annotate volcano plots.
4.5. RT-qPCR
The expression of the genes of interest were quantified on the QuantStudio 7 Flex Real-Time PCR Machine (Applied Biosystems™, Loughborough, UK) using SYBR™ Green PCR Master Mix (Applied Biosystems™, Loughborough, UK). Each qPCR reaction was performed in triplicate on a MicroAmp™ Fast Optical 96-Well Reaction Plate with Barcode, 0.1 mL. Primer sequences were acquired from the Harvard Primer Bank and issued by Sigma Aldrich (Merck, Gillingham, UK) [61] (Table 2).

Table 2.
List of primers used.
4.6. Statistical Analysis
Differences identified in RT-qPCR experiments were assessed for statistical significance using one-way ANOVA. All statistical tests were performed using GraphPad Prism® software (GraphPad Software, Inc., San Diego, CA, USA). p-values < 0.05 were considered significant.
5. Conclusions
The present findings underscore the importance of understanding receptor dynamics and signalling pathways in the context of the potential role of asprosin in EC. The outcomes of future functional studies could provide a deeper insight into the biological mechanisms through which asprosin may influence EC, but also identify potential therapeutic targets. For example, if asprosin enhances cell proliferation and migration via TLR4 or PTPRD signalling, inhibiting these receptors could offer a novel strategy for limiting EC progression. Focusing on functional studies (including potential in vivo models) is a critical step in translating such molecular insights on the role of asprosin into actionable therapeutic approaches.
Author Contributions
Conceptualization, J.C. and E.K.; methodology, R.K., C.S. and S.S.; software, R.K., C.S. and E.K.; formal analysis, R.K., S.S., C.S. and E.K.; investigation, R.K., C.S., I.K., H.S.R. and J.C.; resources, I.K., H.S.R. and J.C.; data curation, R.K.; writing—original draft preparation, R.K., C.S. and E.K.; writing—reviewing and editing, R.K., C.S., S.S., I.K., H.S.R., J.C. and E.K.; supervision, J.C. and E.K.; project administration, J.C. and E.K.; funding acquisition, I.K., H.S.R., J.C. and E.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Gynae-oncology Research and Clinical Excellence (GRACE) charity and University Hospital Coventry & Warwickshire (UHCW), NHS Trust, #10418168.
Institutional Review Board Statement
Ethical review and approval were waived for this study due to the use of commercially available cell lines and publicly available data.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets generated and/or analysed during the current study are available upon reasonable request. Researchers interested in accessing the data can contact the corresponding authors. Data on DEGs is provided within Appendix A.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| Endometrial Cancer | EC |
| body mass index | BMI |
| polycystic ovary syndrome | PCOS |
| European Society of Gynaecological Oncology | ESGO |
| European Society for Radiotherapy and Oncology | ESTRO |
| European Society of Medical Oncology | ESMO |
| metabolic syndrome | MetS |
| Olfactory Receptor Family 4 Subfamily M Member 1 | OR4M1 |
| Toll Like Receptor 4 | TLR4 |
| Protein Tyrosine Phosphatase Receptor Delta | PTPRD |
| G protein-coupled receptor | GPCR |
| transcripts per million | TPM |
| differentially expressed genes | DEGs |
| no supplement | NS |
| epithelial-mesenchymal transition | EMT |
| reactive oxygen species | ROS |
| cAMP-Response Element Binding Proteins | CREB |
| mechanistic target of rapamycin complex 1 | mTORC1 |
| paraformaldehyde | PFA |
| bovine serum albumin | BSA |
Appendix A

Table A1.
DEGs in asprosin-treated Ishikawa cells.

Table A2.
DEGs in asprosin-treated NOU-1 cells.
References
- SHutt, D.; Mihaies, E.; Karteris, A.; Michael, A.; Payne, M.; Chatterjee, J. Statistical Meta-Analysis of Risk Factors for Endometrial Cancer and Development of a Risk Prediction Model Using an Artificial Neural Network Algorithm. Cancers 2021, 13, 3689. [Google Scholar] [CrossRef]
- Johnson, J.-E.; Daley, D.; Tarta, C.; Stanciu, P.I. Risk of endometrial cancer in patients with polycystic ovarian syndrome: A meta-analysis. Oncol. Lett. 2023, 25, 168. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, H.; Yang, S.; Zhang, J.; Qian, L.; Chen, X. Overweight, Obesity and Endometrial Cancer Risk: Results from a Systematic Review and Meta-Analysis. Int. J. Biol. Markers 2014, 29, e21–e29. [Google Scholar] [CrossRef]
- Höhn, A.K.; Brambs, C.E.; Hiller, G.G.R.; May, D.; Schmoeckel, E.; Horn, L.C. 2020 WHO Classification of Female Genital Tumors. Geburtshilfe Frauenheilkd. 2021, 81, 1145–1153. [Google Scholar] [CrossRef] [PubMed]
- Murali, R.; Soslow, R.A.; Weigelt, B. Classification of endometrial carcinoma: More than two types. Lancet Oncol. 2014, 15, e268–e278. [Google Scholar] [CrossRef]
- Colombo, N.; Creutzberg, C.; Amant, F.; Bosse, T.; González-Martín, A.; Ledermann, J.; Marth, C.; Nout, R.; Querleu, D.; Mirza, M.R.; et al. ESMO-ESGO-ESTRO Consensus Conference on Endometrial Cancer: Diagnosis, treatment and follow-up. Ann. Oncol. 2016, 27, 16–41. [Google Scholar] [CrossRef]
- Concin, N.; Matias-Guiu, X.; Vergote, I.; Cibula, D.; Mirza, M.R.; Marnitz, S.; Ledermann, J.; Bosse, T.; Chargari, C.; Fagotti, A.; et al. ESGO/ESTRO/ESP guidelines for the management of patients with endometrial carcinoma. Int. J. Gynecol. Cancer 2021, 31, 12–39. [Google Scholar] [CrossRef]
- Karkia, R.; Maccarthy, G.; Payne, A.; Karteris, E.; Pazoki, R.; Chatterjee, J. The Association Between Metabolic Syndrome and the Risk of Endometrial Cancer in Pre- and Post-Menopausal Women: A UK Biobank Study. J. Clin. Med. 2025, 14, 751. [Google Scholar] [CrossRef]
- Wang, C.-Y.; Lin, T.-A.; Liu, K.-H.; Liao, C.-H.; Liu, Y.-Y.; Wu, V.C.-C.; Wen, M.-S.; Yeh, T.-S. Serum asprosin levels and bariatric surgery outcomes in obese adults. Int. J. Obes. 2019, 43, 1019–1025. [Google Scholar] [CrossRef]
- Ugur, K.; Erman, F.; Turkoglu, S.; Aydin, Y.; Aksoy, A.; Lale, A.; Karagöz, Z.K.; Ugur, I.; Akkoc, R.F.; Yalniz, M. Asprosin, visfatin and subfatin as new biomarkers of obesity and metabolic syndrome. Eur. Rev. Med. Pharmacol.Sci. 2022, 26, 2124–2133. [Google Scholar] [CrossRef] [PubMed]
- Mishra, I.; Xie, W.R.; Bournat, J.C.; He, Y.; Wang, C.; Silva, E.S.; Liu, H.; Ku, Z.; Chen, Y.; Erokwu, B.O.; et al. Protein tyrosine phosphatase receptor δ serves as the orexigenic asprosin receptor. Cell Metab. 2022, 34, 549–563.e8. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.; Yun, S.; Jeong, J.H.; Jung, T.W. Asprosin impairs insulin secretion in response to glucose and viability through TLR4/JNK-mediated inflammation. Mol. Cell. Endocrinol. 2019, 486, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Shabir, K.; Gharanei, S.; Orton, S.; Patel, V.; Chauhan, P.; Karteris, E.; Randeva, H.S.; Brown, J.E.; Kyrou, I. Asprosin Exerts Pro-Inflammatory Effects in THP-1 Macrophages Mediated via the Toll-like Receptor 4 (TLR4) Pathway. Int. J. Mol. Sci. 2022, 24, 227. [Google Scholar] [CrossRef] [PubMed]
- Li, E.; Shan, H.; Chen, L.; Long, A.; Zhang, Y.; Liu, Y.; Jia, L.; Wei, F.; Han, J.; Li, T.; et al. OLFR734 Mediates Glucose Metabolism as a Receptor of Asprosin. Cell Metab. 2019, 30, 319–328.e8. [Google Scholar] [CrossRef]
- Kerslake, R.; Sisu, C.; Panfilov, S.; Hall, M.; Khan, N.; Jeyaneethi, J.; Randeva, H.; Kyrou, I.; Karteris, E. Differential Regulation of Genes by the Glucogenic Hormone Asprosin in Ovarian Cancer. J. Clin. Med. 2022, 11, 5942. [Google Scholar] [CrossRef]
- Maggi, L.B.; Weber, J.D. Forget Transcription: Translation Is Where the Action Is. Mol. Cell. Biol. 2013, 33, 1884–1885. [Google Scholar] [CrossRef]
- Orton, S.; Karkia, R.; Mustafov, D.; Gharanei, S.; Braoudaki, M.; Filipe, A.; Panfilov, S.; Saravi, S.; Khan, N.; Kyrou, I.; et al. In Silico and In Vitro Mapping of Receptor-Type Protein Tyrosine Phosphatase Receptor Type D in Health and Disease: Implications for Asprosin Signalling in Endometrial Cancer and Neuroblastoma. Cancers 2024, 16, 582. [Google Scholar] [CrossRef]
- Bae, W.J.; Ahn, J.M.; Byeon, H.E.; Kim, S.; Lee, D. PTPRD-inactivation-induced CXCL8 promotes angiogenesis and metastasis in gastric cancer and is inhibited by metformin. J. Exp. Clin. Cancer Res. 2019, 38, 484. [Google Scholar] [CrossRef]
- Kohno, T.; Otsuka, A.; Girard, L.; Sato, M.; Iwakawa, R.; Ogiwara, H.; Sanchez-Cespedes, M.; Minna, J.D.; Yokota, J. A catalog of genes homozygously deleted in human lung cancer and the candidacy of PTPRD as a tumor suppressor gene. Genes Chromosomes Cancer 2010, 49, 342–352. [Google Scholar] [CrossRef]
- Funato, K.; Yamazumi, Y.; Oda, T.; Akiyama, T. Tyrosine phosphatase PTPRD suppresses colon cancer cell migration in coordination with CD44′. Exp. Ther. Med. 2011, 2, 457–463. [Google Scholar] [CrossRef]
- Painter, J.N.; O’mara, T.A.; Morris, A.P.; Cheng, T.H.; Gorman, M.; Martin, L.; Hodson, S.; Jones, A.; Martin, N.G.; Gordon, S.; et al. Genetic overlap between endometriosis and endometrial cancer: Evidence from cross-disease genetic correlation and GWAS meta-analyses. Cancer Med. 2018, 7, 1978–1987. [Google Scholar] [CrossRef]
- Sameer, A.S.; Nissar, S. Toll-Like Receptors (TLRs): Structure, Functions, Signaling, and Role of Their Polymorphisms in Colorectal Cancer Susceptibility. BioMed Res. Int. 2021, 2021, 1157023. [Google Scholar] [CrossRef]
- CKüper, F.-X.B.; Neuhofer, W. Toll-like receptor 4 activates NF-κB and MAP kinase pathways to regulate expression of proinflammatory COX-2 in renal medullary collecting duct cells. Am. J. Physiol.-Ren. Physiol. 2012, 302, F38–F46. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, Y.X.; Lu, J.J.; Hou, D.Y.; Abudukeyoumu, A.; Zhang, H.W.; Li, M.Q.; Xie, F. Active Heme Metabolism Suppresses Macrophage Phagocytosis via the TLR4/Type I IFN Signaling/CD36 in Uterine Endometrial Cancer. Am. J. Reprod. Immunol. 2024, 92, e13916. [Google Scholar] [CrossRef]
- Gulbay, G.; Secme, M.; Mutlu, D. Fusaric acid inhibits cell proliferation and downregulates expressions of toll-like receptors pathway genes in Ishikawa endometrial cancer cells. Eur. Rev. Med. Pharmacol. Sci. 2023, 27, 7431–7436. [Google Scholar] [CrossRef]
- Ashton, K.A.; Proietto, A.; Otton, G.; Symonds, I.; McEvoy, M.; Attia, J.; Scott, R.J. Toll-Like Receptor (TLR) and Nucleosome-binding Oligomerization Domain (NOD) gene polymorphisms and endometrial cancer risk. BMC Cancer 2010, 10, 382. [Google Scholar] [CrossRef]
- Allhorn, S.; Böing, C.; Koch, A.A.; Kimmig, R.; Gashaw, I. TLR3 and TLR4 expression in healthy and diseased human endometrium. Reprod. Biol. Endocrinol. 2008, 6, 40. [Google Scholar] [CrossRef]
- Hossain, M.A.; Islam, S.M.S.; Quinn, J.M.W.; Huq, F.; Moni, M.A. Machine learning and bioinformatics models to identify gene expression patterns of ovarian cancer associated with disease progression and mortality. J. Biomed. Inform. 2019, 100, 103313. [Google Scholar] [CrossRef] [PubMed]
- Hao, B.; Chen, Z.; Bi, B.; Yu, M.; Yao, S.; Feng, Y.; Yu, Y.; Pan, L.; Di, D.; Luo, G.; et al. Role of TLR4 as a prognostic factor for survival in various cancers: A meta-analysis. Oncotarget 2018, 9, 13088–13099. [Google Scholar] [CrossRef] [PubMed]
- Lupi, L.A.; Cucielo, M.S.; Silveira, H.S.; Gaiotte, L.B.; Cesário, R.C.; Seiva, F.R.F.; de Almeida Chuffa, L.G. The role of Toll-like receptor 4 signaling pathway in ovarian, cervical, and endometrial cancers. Life Sci. 2020, 247, 117435. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.-Q.; Peng, X.-C.; Liu, L.; Yang, F.-Y.; Qian, F. Olfactory receptors and human diseases. Cell Tissue Res. 2025, 401, 1–14. [Google Scholar] [CrossRef]
- Kerslake, R.; Hall, M.; Randeva, H.S.; Spandidos, D.A.; Chatha, K.; Kyrou, I.; Karteris, E. Co-expression of peripheral olfactory receptors with SARS-CoV-2 infection mediators: Potential implications beyond loss of smell as a COVID-19 symptom. Int. J. Mol. Med. 2020, 46, 949–956. [Google Scholar] [CrossRef]
- Kerslake, R.; Hall, M.; Vagnarelli, P.; Jeyaneethi, J.; Randeva, H.S.; Pados, G.; Kyrou, I.; Karteris, E. A pancancer overview of FBN1, asprosin and its cognate receptor OR4M1 with detailed expression profiling in ovarian cancer. Oncol. Lett. 2021, 22, 650. [Google Scholar] [CrossRef] [PubMed]
- Bailey, J.; Tyson-Capper, A.J.; Gilmore, K.; Robson, S.C.; Europe-Finner, G.N. Identification of human myometrial target genes of the cAMP pathway: The role of cAMP-response element binding (CREB) and modulator (CREMα and CREMτ2α) proteins. J. Mol. Endocrinol. 2005, 34, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Tang, Y.; Deng, M.J.; Huang, C.T.; Lan, D.; Nong, W.Z.; Li, L.; Wang, Q. A combined biomarker panel shows improved sensitivity and specificity for detection of ovarian cancer. J. Clin. Lab. Anal. 2022, 36, e24232. [Google Scholar] [CrossRef]
- Pastorekova, S.; Gillies, R.J. The role of carbonic anhydrase IX in cancer development: Links to hypoxia, acidosis, and beyond. Cancer Metastasis Rev. 2019, 38, 65–77. [Google Scholar] [CrossRef]
- Ronca, R.; Supuran, C.T. Carbonic anhydrase IX: An atypical target for innovative therapies in cancer. Biochim. Biophys. Acta (BBA)—Rev. Cancer 2024, 1879, 189120. [Google Scholar] [CrossRef]
- Xie, Q.; Huang, J.; Xie, Y.; Hu, J.; Jin, L. Identification of prognostic biomarkers for endometrioid endometrial carcinoma based on the miRNA and mRNA co-expression network regulated by estradiol. Clinics 2025, 80, 100672. [Google Scholar] [CrossRef]
- Jin, M.; Yin, C.; Yang, J.; Yang, X.; Wang, J.; Zhu, J.; Yuan, J. Identification and validation of calcium extrusion-related genes prognostic signature in colon adenocarcinoma. PeerJ 2024, 12, e17582. [Google Scholar] [CrossRef]
- Sharma, A.; Kumar, R.; Aier, I.; Semwal, R.; Tyagi, P.; Varadwaj, P. Sense of Smell: Structural, Functional, Mechanistic Advancements and Challenges in Human Olfactory Research. Curr. Neuropharmacol. 2019, 17, 891–911. [Google Scholar] [CrossRef] [PubMed]
- Boekhoff, I.; Inglese, J.; Schleicher, S.; Koch, W.J.; Lefkowitz, R.J.; Breer, H. Olfactory desensitization requires membrane targeting of receptor kinase mediated by beta gamma-subunits of heterotrimeric G. proteins. J. Biol. Chem. 1994, 269, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Peppel, K.; Boekhoff, I.; McDonald, P.; Breer, H.; Caron, M.G.; Lefkowitz, R.J. G protein-coupled receptor kinase 3 (GRK3) gene disruption leads to loss of odorant receptor desensitization. J. Biol. Chem. 1997, 272, 25425–25428. [Google Scholar] [CrossRef]
- Mashukova, A.; Spehr, M.; Hatt, H.; Neuhaus, E.M. β-Arrestin2-Mediated Internalization of Mammalian Odorant Receptors. J. Neurosci. 2006, 26, 9902–9912. [Google Scholar] [CrossRef]
- Fattahi, F.; Saeednejad Zanjani, L.; Habibi Shams, Z.; Kiani, J.; Mehrazma, M.; Najafi, M.; Madjd, Z. High expression of DNA damage-inducible transcript 4 (DDIT4) is associated with advanced pathological features in the patients with colorectal cancer. Sci. Rep. 2021, 11, 13626. [Google Scholar] [CrossRef]
- Yoshikawa, N.; Yoshida, K.; Liu, W.; Matsukawa, T.; Hattori, S.; Yoshihara, M.; Tamauchi, S.; Ikeda, Y.; Yokoi, A.; Shimizu, Y.; et al. The prognostic significance of DDIT4 in endometrial cancer. Cancer Biomark. 2023, 37, 217–225. [Google Scholar] [CrossRef]
- Chang, B.; Liu, G.; Yang, G.; Mercado-Uribe, I.; Huang, M.; Liu, J. REDD1 is required for RAS-mediated transformation of human ovarian epithelial cells. Cell Cycle 2009, 8, 780–786. [Google Scholar] [CrossRef]
- Ji, Y.; Xie, M.; Lan, H.; Zhang, Y.; Long, Y.; Weng, H.; Li, D.; Cai, W.; Zhu, H.; Niu, Y.; et al. PRR11 is a novel gene implicated in cell cycle progression and lung cancer. Int. J. Biochem. Cell Biol. 2013, 45, 645–656. [Google Scholar] [CrossRef]
- Ni, W.; Yi, L.; Dong, X.; Cao, M.; Zheng, J.; Wei, Q.; Yuan, C. PRR11 is a prognostic biomarker and correlates with immune infiltrates in bladder urothelial carcinoma. Sci. Rep. 2023, 13, 2051. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Wu, X.; Zheng, G.; Jin, J.; Li, C.; Yu, G.; Li, W. Proline-rich protein 11 overexpression is associated with a more aggressive phenotype and poor overall survival in ovarian cancer patients. World J. Surg. Oncol. 2020, 18, 318. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.; Liu, Y.; Zhang, T.; Huang, Z.; Xu, X.; Zhao, Z.; Liu, J.; Zhai, E.; Cai, S.; Chen, J. Spindle function and Wnt pathway inhibition by PBX1 to suppress tumor progression via downregulating DCDC2 in colorectal cancer. Oncogenesis 2023, 12, 3. [Google Scholar] [CrossRef]
- Maclean, A.; Adishesh, M.; Button, L.; Richards, L.; Alnafakh, R.; Newton, E.; Drury, J.; Hapangama, D.K. The effect of pre-analytical variables on downstream application and data analysis of human endometrial biopsies. Hum. Reprod. Open 2022, 2022, hoac026. [Google Scholar] [CrossRef]
- Medina-Gutierrez, E.; Céspedes, M.V.; Gallardo, A.; Rioja-Blanco, E.; Pavon, M.A.; Asensio-Puig, L.; Farre, L.; Alba-Castellon, L.; Unzueta, U.; Villaverde, A.; et al. Novel Endometrial Cancer Models Using Sensitive Metastasis Tracing for CXCR4-Targeted Therapy in Advanced Disease. Biomedicines 2022, 10, 1680. [Google Scholar] [CrossRef]
- Ueda, H.; Ishiguro, T.; Mori, Y.; Yamawaki, K.; Okamoto, K.; Enomoto, T.; Yoshihara, K. Glycolysis-mTORC1 crosstalk drives proliferation of patient-derived endometrial cancer spheroid cells with ALDH activity. Cell Death Discov. 2024, 10, 435. [Google Scholar] [CrossRef]
- Kim, M.; Lee, S.; Park, W.H.; Suh, D.H.; Kim, K.; Kim, Y.B.; No, J.H. Silencing Bmi1 expression suppresses cancer stemness and enhances chemosensitivity in endometrial cancer cells. Biomed. Pharmacother. 2018, 108, 584–589. [Google Scholar] [CrossRef]
- Faruqi, S.A.; Satyaswaroop, P.G.; LiVolsi, V.A.; Deger, R.B.; Noumoff, J.S. Establishment and characterization of a poorly differentiated lethal human endometrial carcinoma cell line (NOU-1) with karyotype 46,XX. Cancer Genet. Cytogenet. 2002, 138, 44–49. [Google Scholar] [CrossRef]
- Tang, G.; Cho, M.; Wang, X. OncoDB: An interactive online database for analysis of gene expression and viral infection in cancer. Nucleic Acids Res. 2022, 50, D1334–D1339. [Google Scholar] [CrossRef] [PubMed]
- Trapnell, C.; Roberts, A.; Goff, L.; Pertea, G.; Kim, D.; Kelley, D.R.; Pimentel, H.; Salzberg, S.L.; Rinn, J.L.; Pachter, L. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 2012, 7, 562–578. [Google Scholar] [CrossRef]
- Akhmedov, M.; Martinelli, A.; Geiger, R.; Kwee, I. Omics Playground: A comprehensive self-service platform for visualization, analytics and exploration of Big Omics Data. NAR Genom. Bioinform. 2020, 2, lqz019. [Google Scholar] [CrossRef]
- Van Der Maaten, L.; Courville, A.; Fergus, R.; Manning, C. Accelerating t-SNE Using Tree-Based Algorithms. 2014. Available online: https://jmlr.org/papers/v15/vandermaaten14a.html (accessed on 15 July 2025).
- Witten, D.M.; Tibshirani, R.; Hastie, T. A penalized matrix decomposition, with applications to sparse principal components and canonical correlation analysis. Biostatistics 2009, 10, 515–534. [Google Scholar] [CrossRef]
- Wang, X.; Spandidos, A.; Wang, H.; Seed, B. PrimerBank: A PCR primer database for quantitative gene expression analysis, 2012 update. Nucleic Acids Res. 2012, 40, D1144–D1149. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).