Magnetic Driven Nanocarriers for pH-Responsive Doxorubicin Release in Cancer Therapy
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterization of the Magnetic Nanocarriers
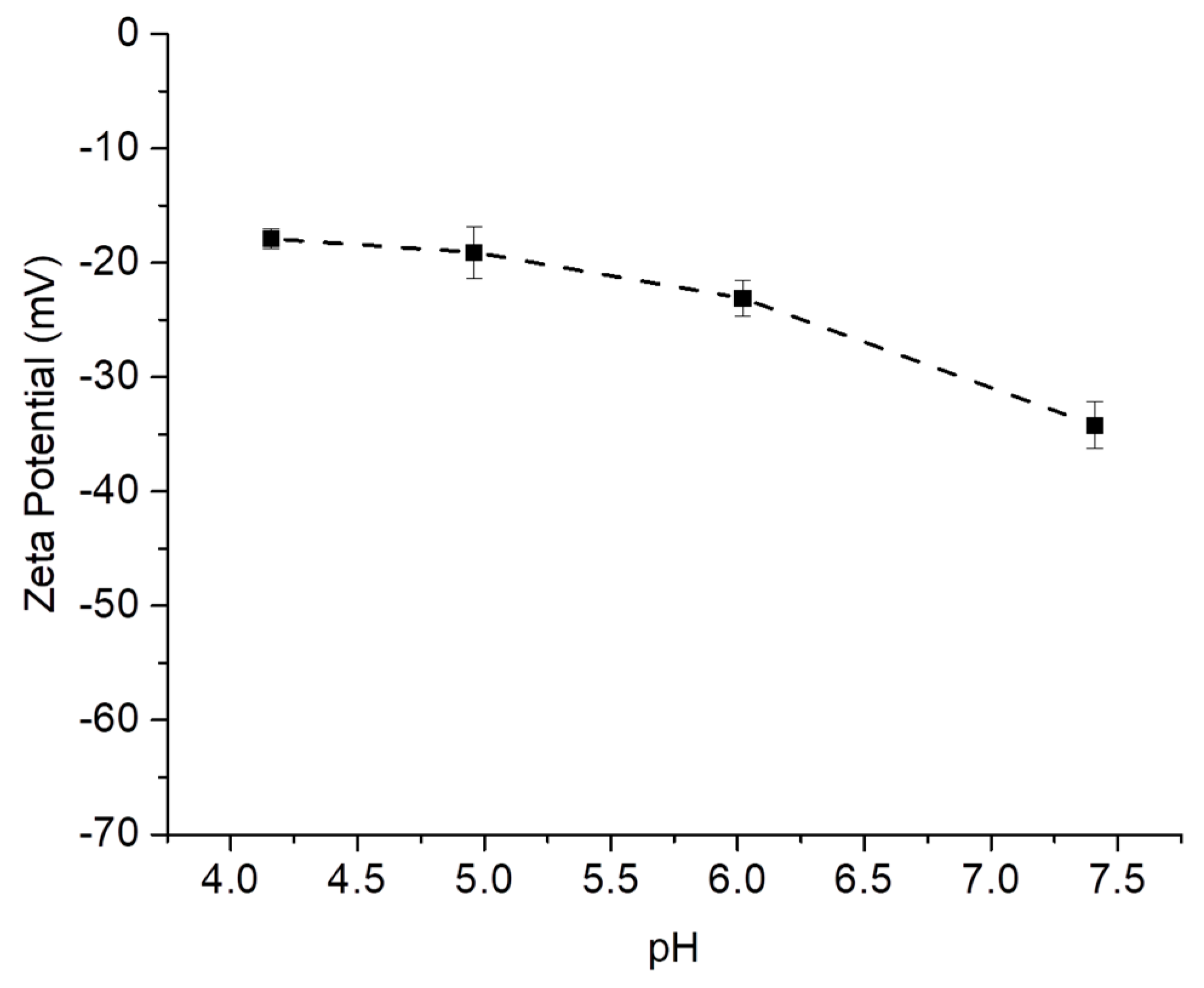
2.2. Doxorubicin Loading
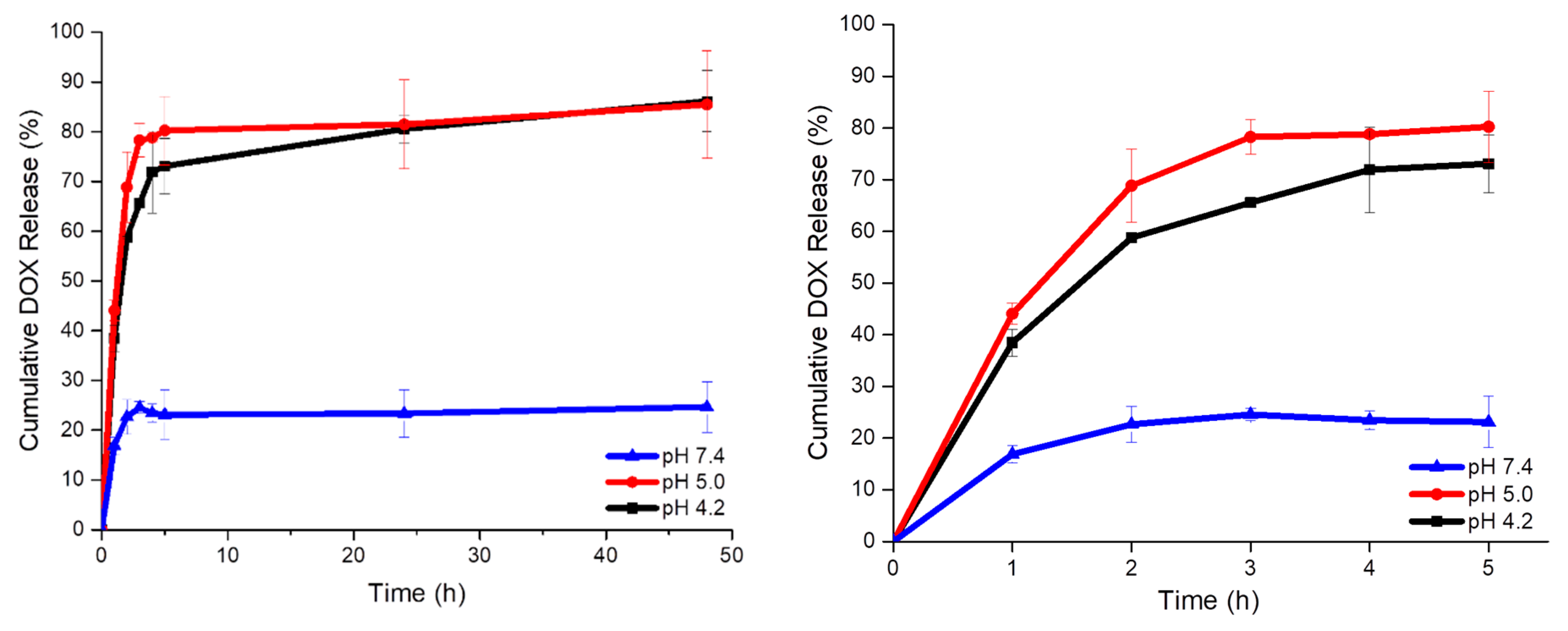
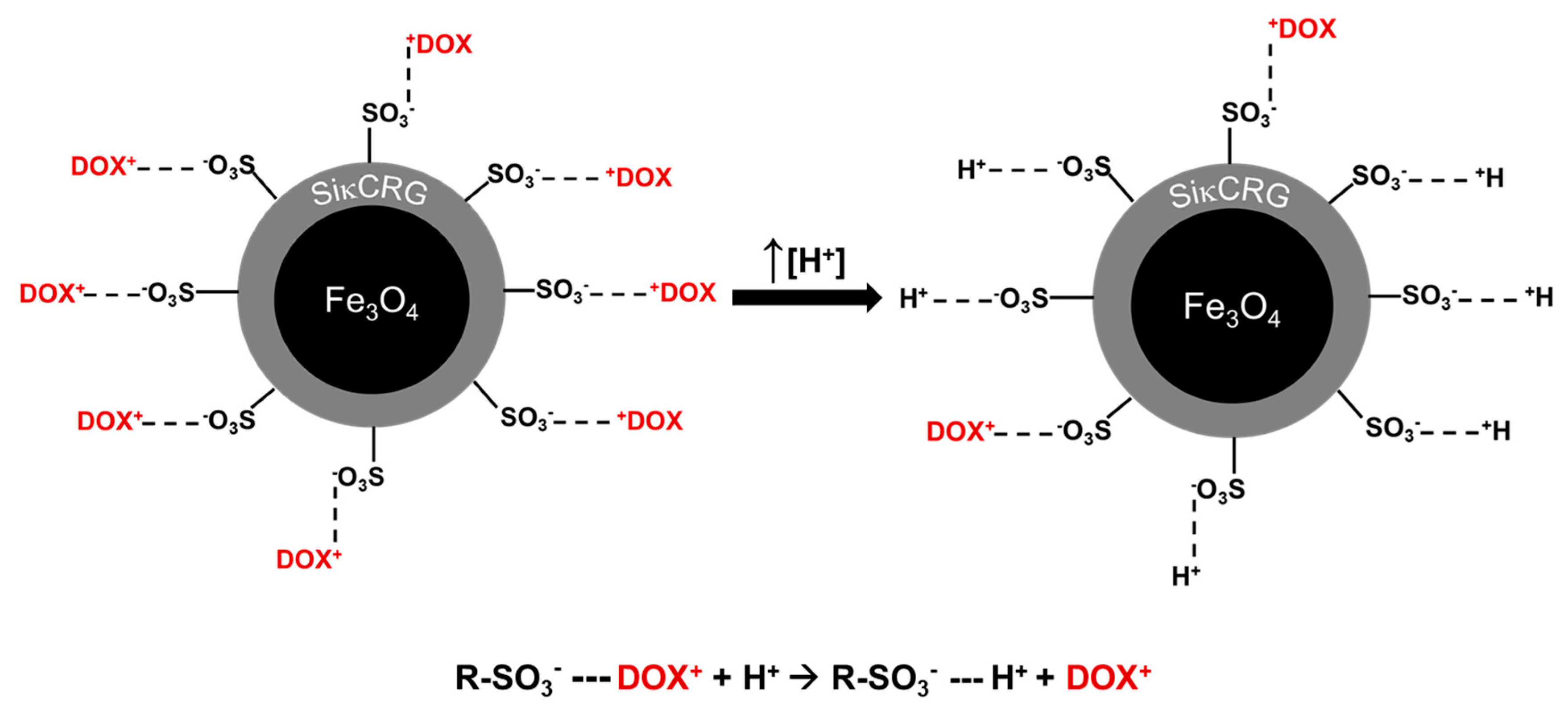
2.3. Doxorubicin pH Mediated Release Studies
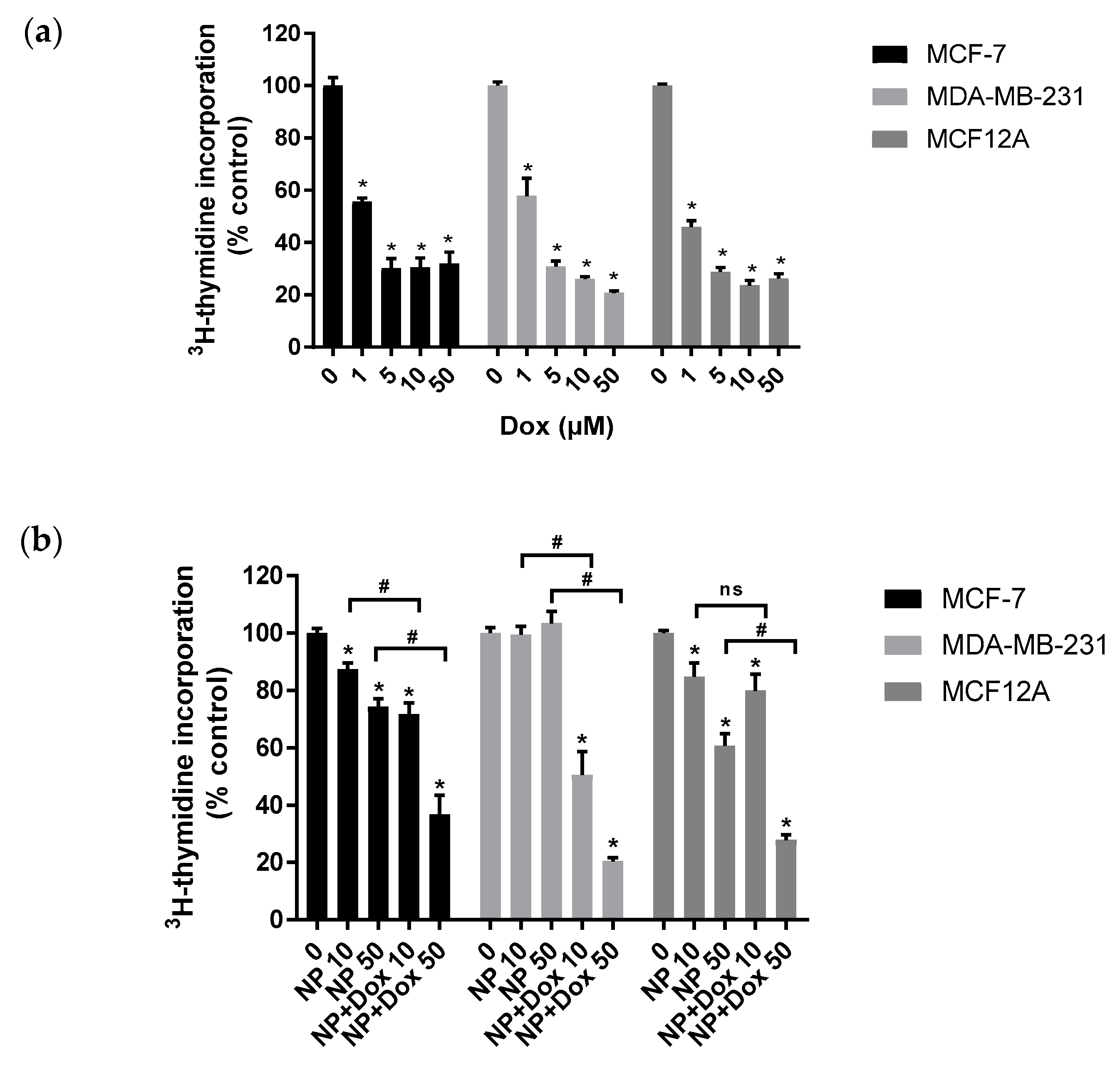
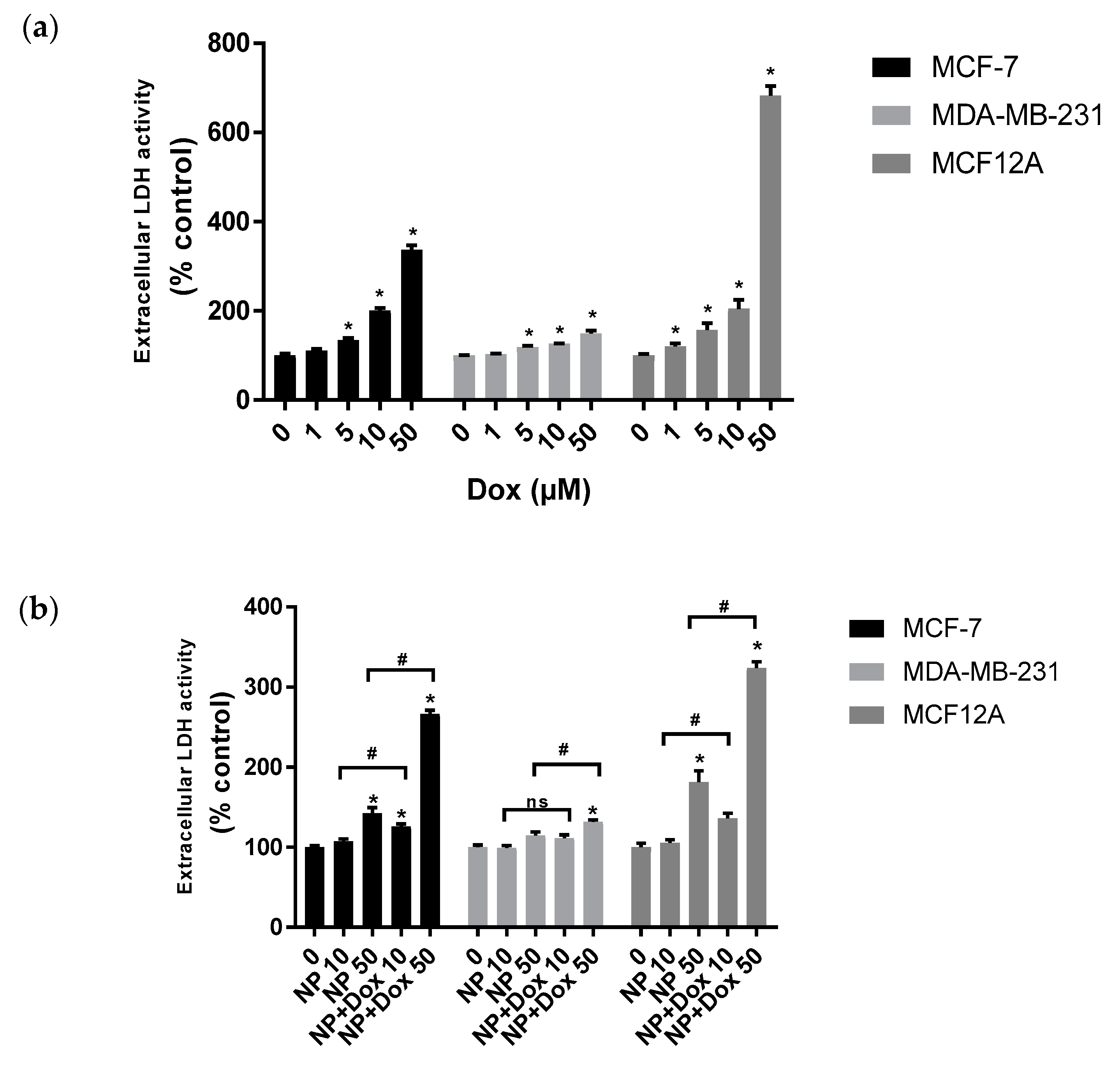
2.4. Antiproliferative and Cytotoxicity In Vitro Evaluation
3. Materials and Methods
3.1. Materials
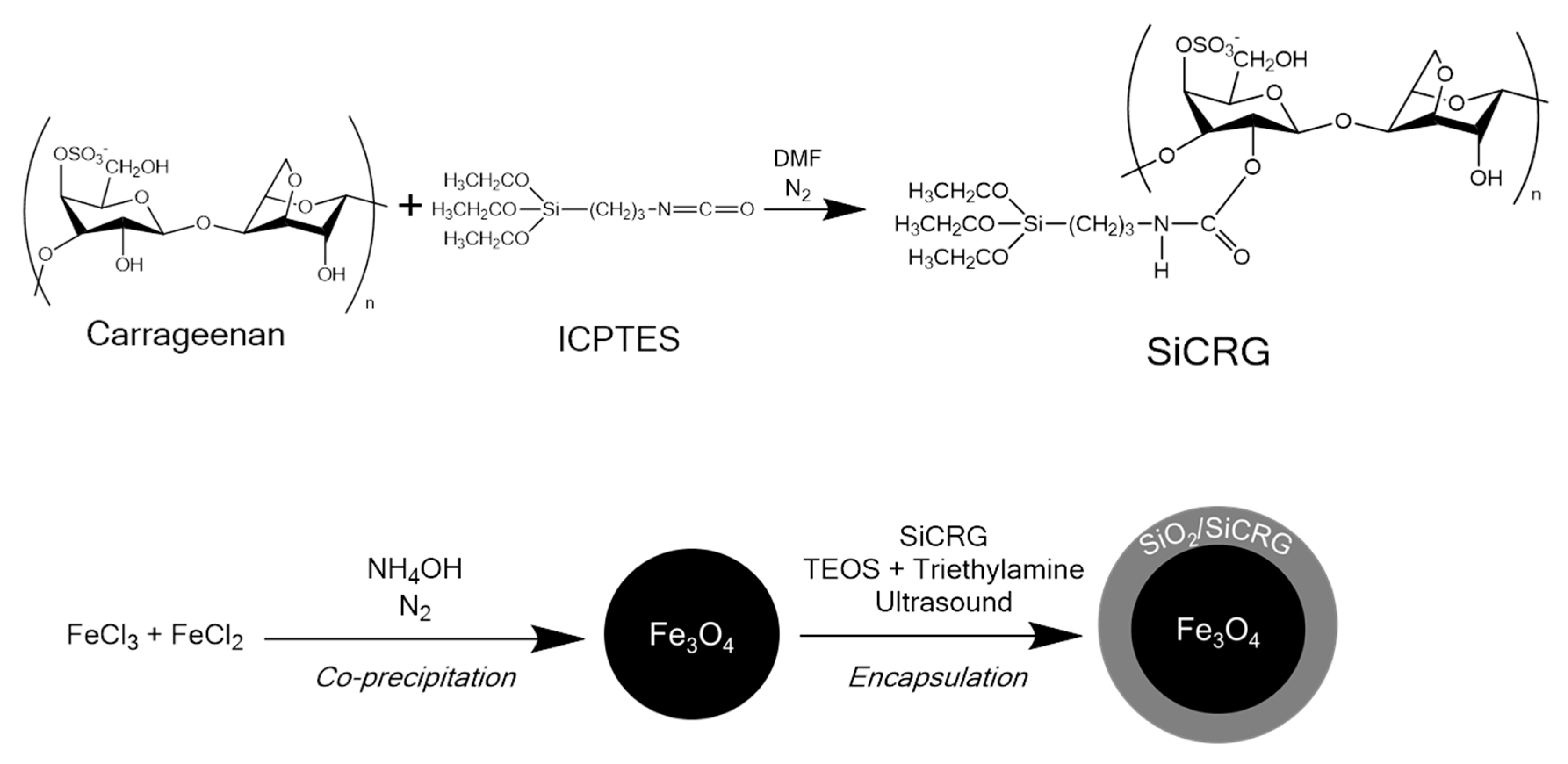
3.2. Synthesis of the Magnetic Nanocarriers
3.3. Nanoparticle Characterization
3.4. Doxorubicin Loading and Release Studies
3.5. Cell Studies
3.5.1. Cell Culture and Treatment
3.5.2. Cytotoxicity and Cell Proliferation Determination
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kostrzewa-Nowak, D.; Paine, M.; Wolf, C.R.; Tarasiuk, J. The role of bioreductive activation of doxorubicin in cytotoxic activity against leukaemia HL60-sensitive cell line and its multidrug-resistant sublines. Br. J. Cancer 2005, 93, 89–97. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shafei, A.; El-bakly, W.; Sobhy, A.; Wagdy, O.; Reda, A.; Aboelenin, O.; Marzouk, A.; El, K.; Mostafa, R.; Ali, M.A.; et al. A review on the efficacy and toxicity of different doxorubicin nanoparticles for targeted therapy in metastatic breast cancer. Biomed. Pharmacother. 2017, 95, 1209–1218. [Google Scholar] [CrossRef] [PubMed]
- Dass, C.R.; Tan, M.L.; Choong, P.F.M. Review: Doxorubicin delivery systems based on chitosan for cancer therapy. J. Pharm. Pharmacol. 2009, 2, 131–142. [Google Scholar] [CrossRef]
- Harake, D.; Franco, V.I.; Henkel, J.M.; Miller, T.L.; Lipshultz, S.E. Cardiotoxicity in Childhood Cancer Survivors Strategies for Prevention and Management. Futur. Cardiol. 2012, 8, 647–670. [Google Scholar] [CrossRef]
- Vandecruys, E.; Mondelaers, V.; de Wolf, D.; Benoit, Y.; Suys, B. Late cardiotoxicity after low dose of anthracycline therapy for acute lymphoblastic leukemia in childhood. J. Cancer Surviv. 2012, 6, 95–101. [Google Scholar] [CrossRef]
- Pein, F.; Sakiroglu, O.; Dahan, M.; Lebidois, J.; Merlet, P.; Shamsaldin, A.; Villain, E.; De Vathaire, F.; Sidi, D.; Hartmann, O. Cardiac abnormalities 15 years and more after adriamycin therapy in 229 childhood survivors of a solid tumour at the Institut Gustave Roussy. Br. J. Cancer 2004, 91, 37–44. [Google Scholar] [CrossRef]
- Luqmani, Y.A. Mechanisms of drug resistance in cancer chemotherapy. Med. Princ. Pract. 2008, 14, 35–48. [Google Scholar] [CrossRef]
- Gottesman, M.M. Mechanisms of drug cancer resistance. Annu. Rev. Med. 2002, 53, 615–627. [Google Scholar] [CrossRef]
- Zeng, X.; Morgenstern, R.; Nyström, A.M. Nanoparticle-directed sub-cellular localization of doxorubicin and the sensitization breast cancer cells by circumventing GST-Mediated drug resistance. Biomaterials 2014, 35, 1227–1239. [Google Scholar] [CrossRef]
- El-Boubbou, K. Magnetic iron oxide nanoparticles as drug carriers: Preparation, conjugation and delivery. Nanomedicine 2018, 13, 929–952. [Google Scholar] [CrossRef]
- Rosenblum, D.; Joshi, N.; Tao, W.; Karp, J.M.; Peer, D. Progress and challenges towards targeted delivery of cancer therapeutics. Nat. Commun. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Jacob, J.; Haponiuk, J.T.; Thomas, S.; Gopi, S. Biopolymer based nanomaterials in drug delivery systems: A review. Mater. Today Chem. 2018, 9, 43–55. [Google Scholar] [CrossRef]
- Gao, Z.; Zhang, L.; Sun, Y. Nanotechnology applied to overcome tumor drug resistance. J. Control. Release 2012, 162, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, Y.; Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent Smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar]
- Maeda, H. The enhanced permeability and retention (EPR) effect in tumor vasculature: The key role of tumor-selective macromolecular drug targeting. Adv. Enzyme Regul. 2001, 41, 189–207. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef]
- Unsoy, G.; Khodadust, R.; Yalcin, S.; Mutlu, P.; Gunduz, U. Synthesis of Doxorubicin loaded magnetic chitosan nanoparticles for pH responsive targeted drug delivery. Eur. J. Pharm. Sci. 2014, 62, 243–250. [Google Scholar] [CrossRef]
- Soares, P.I.P.; Sousa, A.I.; Silva, J.C.; Ferreira, I.M.M.; Novo, C.M.M.; Borges, J.P. Chitosan-based nanoparticles as drug delivery systems for doxorubicin: Optimization and modelling. Carbohydr. Polym. 2016, 147, 304–312. [Google Scholar] [CrossRef]
- Lopes, P.P.; Barroca, N.B.; Daniel-da-silva, A.L.; Ferreira Leite, B. Application of Chitosan Based Materials for Drug Delivery. Front. Biomater. Chitosan Based Mater. Its Appl. 2017, 3, 181–248. [Google Scholar]
- Zheng, F.; Shi, X.W.; Yang, G.F.; Gong, L.L.; Yuan, H.Y.; Cui, Y.J.; Wang, Y.; Du, Y.M.; Li, Y. Chitosan nanoparticle as gene therapy vector via gastrointestinal mucosa administration: Results of an in vitro and in vivo study. Life Sci. 2007, 80, 388–396. [Google Scholar] [CrossRef]
- Tigli Aydin, R.S.; Pulat, M. 5-fluorouracil encapsulated chitosan nanoparticles for pH-stimulated drug delivery: Evaluation of controlled release kinetics. J. Nanomater. 2012, 2012. [Google Scholar] [CrossRef]
- Kievit, F.M.; Wang, F.Y.; Fang, C.; Mok, H.; Wang, K.; Silber, J.R.; Ellenbogen, R.G.; Zhang, M. Doxorubicin loaded iron oxide nanoparticles overcome multidrug resistance in cancer in vitro. J. Control. Release 2011, 152, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Chen, L.; Liu, F.; Qi, X.; Ge, Y.; Shen, S. Remotely controlled drug release based on iron oxide nanoparticles for specific therapy of cancer. Colloids Surf. B Biointerfaces 2017, 152, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Maeng, J.H.; Lee, D.-H.; Jung, K.H.; Bae, Y.-H.; Park, I.-S.; Jeong, S.; Jeon, Y.-S.; Shim, C.-K.; Kim, W.; Kim, J.; et al. Multifunctional doxorubicin loaded superparamagnetic iron oxide nanoparticles for chemotherapy and magnetic resonance imaging in liver cancer. Biomaterials 2010, 31, 4995–5006. [Google Scholar] [CrossRef]
- Fang, C.; Kievit, F.M.; Veiseh, O.; Stephen, Z.R.; Wang, T.; Lee, D.; Ellenbogen, R.G.; Zhang, M. Fabrication of magnetic nanoparticles with controllable drug loading and release through a simple assembly approach. J. Control. Release 2012, 162, 233–241. [Google Scholar] [CrossRef]
- Liang, P.; Chen, Y.-C.; Chiang, C.; Mo, L.-R.; Wei, S.-Y.; Hsieh, W.; Lin, W.-L. Doxorubicin-modified magnetic nanoparticles as a drug delivery system for magnetic resonance chemotherapy. Int. J. Nanomed. 2016, 11, 2021–2037. [Google Scholar]
- Khan, A.K.; Saba, A.U.; Nawazish, S.; Akhtar, F.; Rashid, R.; Mir, S.; Nasir, B.; Iqbal, F.; Afzal, S.; Pervaiz, F.; et al. Carrageenan based bionanocomposites as drug delivery tool with special emphasis on the influence of ferromagnetic nanoparticles. Oxid. Med. Cell. Longev. 2017, 2017. [Google Scholar] [CrossRef]
- Blanco-Andujar, C.; Walter, A.; Cotin, G.; Bordeianu, C.; Mertz, D.; Felder-Flesch, D.; Begin-Colin, S. Design of iron oxide-based nanoparticles for MRI and magnetic hyperthermia. Nanomedicine 2016, 11, 1889–1910. [Google Scholar] [CrossRef]
- Hervault, A.; Dunn, A.E.; Lim, M.; Boyer, C.; Mott, D.; Maenosono, S.; Thanh, N.T.K. Development of a doxorubicin-loaded dual pH- and thermo-responsive magnetic nanocarrier for application in magnetic hyperthermia and drug delivery in cancer therapy. Nanoscale 2016, 8, 12152–12161. [Google Scholar] [CrossRef]
- Cagel, M.; Bernabeu, E.; Moretton, M.A.; Chiappetta, D.A. Doxorubicin: nanotechnological overviews from bench to bedside. Drug Discov. Today 2017, 22, 270–281. [Google Scholar] [CrossRef]
- Solomon, R.; Gabizon, A.A. Clinical pharmacology of liposomal anthracyclines: Focus on pegylated liposomal doxorubicin. Clin. Lymphoma Myeloma 2008, 8, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Szebeni, J.; Muggia, F.; Gabizon, A.; Barenholz, Y. Activation of complement by therapeutic liposomes and other lipid excipient-based therapeutic products: Prediction and prevention. Adv. Drug Deliv. Rev. 2011, 63, 1020–1030. [Google Scholar] [CrossRef]
- Chan, S.; Davidson, N.; Juozaityte, E.; Erdkamp, F.; Pluzanska, A.; Azarnia, N.; Lee, L.W.; Beauduin, M.; Humblet, Y.; Lemmens, J.; et al. Phase III trial of liposomal doxorubicin and cyclophosphamide compared with epirubicin and cyclophosphamide as first-line therapy for metastatic breast cancer. Ann. Oncol. 2004, 15, 1527–1534. [Google Scholar] [CrossRef] [PubMed]
- Chao, T.-C.; Wang, W.-S.; Yen, C.-C.; Chiou, T.-J.; Liu, J.-H.; Chen, P.-M. A dose-escalating pilot study of sterically stabilized, pegylated liposomal doxorubicin (Lipo-Dox®) in patients with metastatic breast cancer. Cancer Investig. 2003, 21, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, X.G.; Li, Y.Y.; Liu, C.S. Self-assembled nanoparticles based on hydrophobically modified chitosan as carriers for doxorubicin. Nanomed. Nanotechnol. Biol. Med. 2007, 3, 258–265. [Google Scholar] [CrossRef]
- Pu, Y.; Chang, S.; Yuan, H.; Wang, G.; He, B.; Gu, Z. The anti-tumor efficiency of poly(l-glutamic acid) dendrimers with polyhedral oligomeric silsesquioxane cores. Biomaterials 2013, 34, 3658–3666. [Google Scholar] [CrossRef] [PubMed]
- Parodi, A.; Quattrocchi, N.; Ven, A.; Chiappini, C.; Evangelopoulous, M.; Martinez, J.; Brown, B.; Khaled, S.; Yazdi, I.; Enzo, M.; et al. Biomimetic functionalization with leukocyte membranes imparts cell like functions to synthetic particles. Nat. Nanotechnol. 2013, 8, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Munnier, E.; Cohen-Jonathan, S.; Linassier, C.; Douziech-Eyrolles, L.; Marchais, H.; Soucé, M.; Hervé, K.; Dubois, P.; Chourpa, I. Novel method of doxorubicin-SPION reversible association for magnetic drug targeting. Int. J. Pharm. 2008, 363, 170–176. [Google Scholar] [CrossRef]
- Wuang, S.C.; Neoh, K.G.; Kang, E.T.; Leckband, D.E.; Pack, D.W. Acid-sensitive magnetic nanoparticles as potential drug depots. AIChE J. 2011, 57, 1638–1645. [Google Scholar] [CrossRef]
- Bakandritsos, A.; Mattheolabakis, G.; Chatzikyriakos, G.; Szabo, T. Doxorubicin nanocarriers based on magnetic colloids with a bio-polyelectrolyte corona and high non-linear optical response: Synthesis, characterization, and properties. Adv. Funct. Mater. 2011, 21, 1465–1475. [Google Scholar] [CrossRef]
- Panlasigui, L.; Baello, O.; Dimatangal, J.; Dumelod, B. Blood cholesterol and lipid-lowering effects of carrageenan on human volunteers. Asia Pac. J. Clin. Nutr. 2003, 12, 209–214. [Google Scholar] [PubMed]
- Necas, J.; Bartosikova, L. Carrageenan: A review. Vet. Med. 2013, 58, 187–205. [Google Scholar] [CrossRef]
- Cunha, L.; Grenha, A. Sulfated seaweed polysaccharides as multifunctional materials in drug delivery applications. Mar. Drugs 2016, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Daniel-da-Silva, A.L.; Ferreira, L.; Gil, A.M.; Trindade, T. Synthesis and swelling behavior of temperature responsive κ-carrageenan nanogels. J. Colloid Interface Sci. 2011, 355, 512–517. [Google Scholar] [CrossRef]
- Li, L.; Ni, R.; Shao, Y.; Mao, S. Carrageenan and its applications in drug delivery. Carbohydr. Polym. 2014, 103, 1–11. [Google Scholar] [CrossRef]
- Kulkarni, R.V.; Boppana, R.; Krishna Mohan, G.; Mutalik, S.; Kalyane, N.V. pH-responsive interpenetrating network hydrogel beads of poly(acrylamide)-g-carrageenan and sodium alginate for intestinal targeted drug delivery: Synthesis, in vitro and in vivo evaluation. J. Colloid Interface Sci. 2012, 367, 509–517. [Google Scholar] [CrossRef]
- Bosio, V.E.; Cacicedo, M.L.; Calvignac, B.; León, I.; Beuvier, T.; Boury, F.; Castro, G.R. Synthesis and characterization of CaCO3-biopolymer hybrid nanoporous microparticles for controlled release of doxorubicin. Colloids Surf. B Biointerfaces 2014, 123, 158–169. [Google Scholar] [CrossRef]
- Cacicedo, M.L.; Cesca, K.; Bosio, V.E.; Porto, L.M.; Castro, G.R. Self-assembly of carrageenin-CaCO3 hybrid microparticles on bacterial cellulose films for doxorubicin sustained delivery. J. Appl. Biomed. 2015, 13, 239–248. [Google Scholar] [CrossRef]
- Raman, M.; Devi, V.; Double, M. Biocompatible ι-carrageenan-γ-maghemite nanocomposite for biomedical applications—Synthesis, characterization and in vitro anticancer efficacy. J. Nanobiotechnol. 2015, 13. [Google Scholar] [CrossRef]
- Oliveira-Silva, R.; Pinto da Costa, J.; Vitorino, R.; Daniel-da-Silva, A.L. Magnetic chelating nanoprobes for enrichment and selective recovery of metalloproteases from human saliva. J. Mater. Chem. B 2015, 3, 238–249. [Google Scholar] [CrossRef]
- Fernandes, T.; Soares, S.; Trindade, T.; Daniel-da-Silva, A. Magnetic hybrid nanosorbents for the uptake of paraquat from water. Nanomaterials 2017, 7, 68. [Google Scholar] [CrossRef] [PubMed]
- Soares, S.F.; Rocha, M.J.; Ferro, M.; Amorim, C.O.; Amaral, J.S.; Trindade, T.; Daniel-da-Silva, A.L. Magnetic nanosorbents with siliceous hybrid shells of alginic acid and carrageenan for removal of ciprofloxacin. Int. J. Biol. Macromol. 2019, 139, 827–841. [Google Scholar] [CrossRef] [PubMed]
- Soares, S.F.; Fernandes, T.; Trindade, T.; Daniel-da-Silva, A.L. Trimethyl chitosan/siloxane-hybrid coated Fe3O4 nanoparticles for the uptake of sulfamethoxazole from water. Molecules 2019, 24, 1958. [Google Scholar] [CrossRef] [PubMed]
- Soares, S.F.; Fernandes, T.; Sacramento, M.; Trindade, T.; Daniel-da-Silva, A.L. Magnetic quaternary chitosan hybrid nanoparticles for the efficient uptake of diclofenac from water. Carbohydr. Polym. 2019, 203, 35–44. [Google Scholar] [CrossRef]
- Soares, S.F.; Simões, T.R.; António, M.; Trindade, T.; Daniel-da-Silva, A.L. Hybrid nanoadsorbents for the magnetically assisted removal of metoprolol from water. Chem. Eng. J. 2016, 302, 560–569. [Google Scholar] [CrossRef]
- PDF 04-009-8435; International Centre for Diffraction Data: Newtown Square, PA, USA, 2011.
- Khalil, M.I. Co-precipitation in aqueous solution synthesis of magnetite nanoparticles using iron(III) salts as precursors. Arab. J. Chem. 2015, 8, 279–284. [Google Scholar] [CrossRef]
- Prado-Fernández, J.; Rodríguez-Vázquez, J.A.; Tojo, E.; Andrade, J.M. Quantitation of κ-, ι- and λ-carrageenans by mid-infrared spectroscopy and PLS regression. Anal. Chim. Acta 2003, 480, 23–37. [Google Scholar] [CrossRef]
- Pereira, L.; Amado, A.M.; Critchley, A.T.; Van de Velde, F.; Ribeiro-Claro, P.J. Identification of selected seaweed polysaccharides (phycocolloids) by vibrational spectroscopy (FTIR-ATR and FT-Raman). Food Hydrocoll. 2009, 23, 1903–1909. [Google Scholar] [CrossRef]
- Soares, S.F.; Trindade, T.; Daniel-da-Silva, A.L. Carrageenan-silica hybrid nanoparticles prepared by a non-emulsion method. Eur. J. Inorg. Chem. 2015, 2015, 4588–4594. [Google Scholar] [CrossRef]
- Silva, S.S.; Ferreira, R.A.S.; Fu, L.; Carlos, D.; Mano, F.; Reis, L. Functional nanostructured chitosan—Siloxane hybrids. J. Mater. Chem. 2005, 15, 3952–3961. [Google Scholar] [CrossRef]
- Daniel-da-silva, A.L.; Salgueiro, A.M.; Trindade, T. Effects of Au nanoparticles on thermoresponsive genipin-crosslinked gelatin hydrogels. Gold Bull. 2013, 46, 25–33. [Google Scholar] [CrossRef]
- Daniel-da-silva, A.L.; Bordado, J.C.M.; Martín-Martínez, J.M. Moisture curing kinetics of isocyanate ended urethane quasi-prepolymers monitored by IR spectroscopy and DSC. J. Appl. Polym. Sci. 2008, 107, 700–709. [Google Scholar] [CrossRef]
- Daniel-da-silva, A.L.; Salgueiro, A.M.; Creaney, B.; Oliveira-Silva, R.; Silva, N.J.O.; Trindade, T. Carrageenan-grafted magnetite nanoparticles as recyclable sorbents for dye removal. J. Nanopart. Res. 2015, 17, 1–15. [Google Scholar] [CrossRef]
- Mascolo, M.C.; Pei, Y.; Ring, T.A. Room temperature co-precipitation synthesis of magnetite nanoparticles in a large pH window with different bases. Materials 2013, 6, 5549–5567. [Google Scholar] [CrossRef]
- Daoush, W.M. Co-precipitation and magnetic properties of magnetite nanoparticles for potential biomedical applications. J. Nanomed. Res. 2017, 5, 3–8. [Google Scholar] [CrossRef]
- Sanson, C.; Diou, O.; Thévenot, J.; Ibarboure, E.; Soum, A.; Brûlet, A.; Miraux, S.; Thiaudière, E.; Tan, S.; Brisson, A.; et al. Doxorubicin loaded magnetic polymersomes: Theranostic nanocarriers for MR imaging and magneto-chemotherapy. ACS Nano 2011, 5, 1122–1140. [Google Scholar] [CrossRef]
- Munnier, E.; Tewes, F.; Cohen-Jonathan, S.; Linassier, C.; Douziech-Eyrolles, L.; Marchais, H.; Soucé, M.; Hervé, K.; Dubois, P.; Chourpa, I. On the interaction of doxorubicin with oleate ions: fluorescence spectroscopy and liquid–liquid extraction study. Chem. Pharm. Bull. 2007, 55, 1006–1010. [Google Scholar] [CrossRef]
- Yousefpour, P.; Atyabi, F.; Farahani, E.; Sakhtianchi, R.; Dinarvand, R. Polyanionic carbohydrate doxorubicin—Dextran nanocomplex as a delivery system for anticancer drugs: In vitro analysis and evaluations. Int. J. Nanomed. 2011, 6, 1487–1496. [Google Scholar]
- Biondi, M.; Fusco, S.; Lewis, A.L.; Netti, P.A. New insights into the mechanisms of the interactions between doxorubicin and the ion-exchange hydrogel DC beadTM for use in Transarterial Chemoembolization (TACE). J. Biomater. Sci. Polym. Ed. 2012, 23, 333–354. [Google Scholar] [CrossRef]
- Bhattacharjee, S. DLS and zeta potential—What they are and what they are not? J. Control. Release 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Bae, Y.; Fukushima, S.; Harada, A.; Kataoka, K. Design of environment-sensitive supramolecular assemblies for intracellular drug delivery: Polymeric micelles that are responsive to intracellular pH change. Angew. Chem. Int. Ed. 2003, 42, 4640–4643. [Google Scholar] [CrossRef] [PubMed]
- Swaine, T.S.; Garcia, P.; Tang, Y.; Lewis, A.L.; Parkes, G.; Waters, L.J. Characterizing drug-polymer bead interactions using isothermal titration calorimetry. J. Pharm. Sci. 2019, 108, 1772–1778. [Google Scholar] [CrossRef] [PubMed]
- Mohamadnia, Z.; Zohuriaan-Mehr, M.J.; Kabiri, K.; Jamshidi, A.; Mobedi, H. pH-sensitive IPN hydrogel beads of carrageenan-alginate for controlled drug delivery. J. Bioact. Compat. Polym. 2007, 22, 342–356. [Google Scholar] [CrossRef]
- Hezaveh, H.; Muhamad, I.I. Controlled drug release via minimization of burst release in pH-response kappa-carrageenan/polyvinyl alcohol hydrogels. Chem. Eng. Res. Des. 2012, 91, 508–519. [Google Scholar] [CrossRef]
- Langenbucher, F. Letters to the Editor: Linearization of dissolution rate curves by the Weibull distribution. J. Pharm. Pharmacol. 1972, 24, 979–981. [Google Scholar] [CrossRef]
- Samaha, D.; Shehayeb, R.; Kyriacos, S. Modeling and comparison of dissolution profiles of diltiazem modified-release formulations. Dissolution Technol. 2009, 16, 41–46. [Google Scholar] [CrossRef]
- Tacar, O.; Sriamornsak, P.; Dass, C.R. Doxorubicin: An update on anticancer molecular action, toxicity and novel drug delivery systems. J. Pharm. Pharmacol. 2013, 65, 157–170. [Google Scholar] [CrossRef]
- Smith, L. The analysis of doxorubicin resistance in human breast cancer cells using antibody microarrays. Mol. Cancer Ther. 2006, 5, 2115–2120. [Google Scholar] [CrossRef]
- Amiri, Z.; Moghadam, M.F.; Sadeghizadeh, M. Anticancer Effects of Doxorubicin-Loaded Micelle on MCF-7 and MDA-MB-231, Breast Cancer Cell Lines. J. Res. Med. Dent. Sci. 2018, 6, 298–304. [Google Scholar] [CrossRef]
- Oncul, S.; Ercan, A. Discrimination of the effects of eoxorubicin on two different breast cancer cell lines on account of multidrug resistance and apoptosis. Indian J. Pharm. Sci. 2017, 79. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). Determination of the Specific Surface Area of Solids by Gas Adsorption—BET Method (ISO 9277); ISO: Geneva, Switzerland, 2010. [Google Scholar]
- Bergmeyer, H.U.; Bernt, E. UV-Assay with Pyruvate and NADH. In Methods of Enzymatic Analysis; Academic Press: New York, NY, USA, 1974; pp. 574–579. [Google Scholar]
Sample Availability: Not available. |
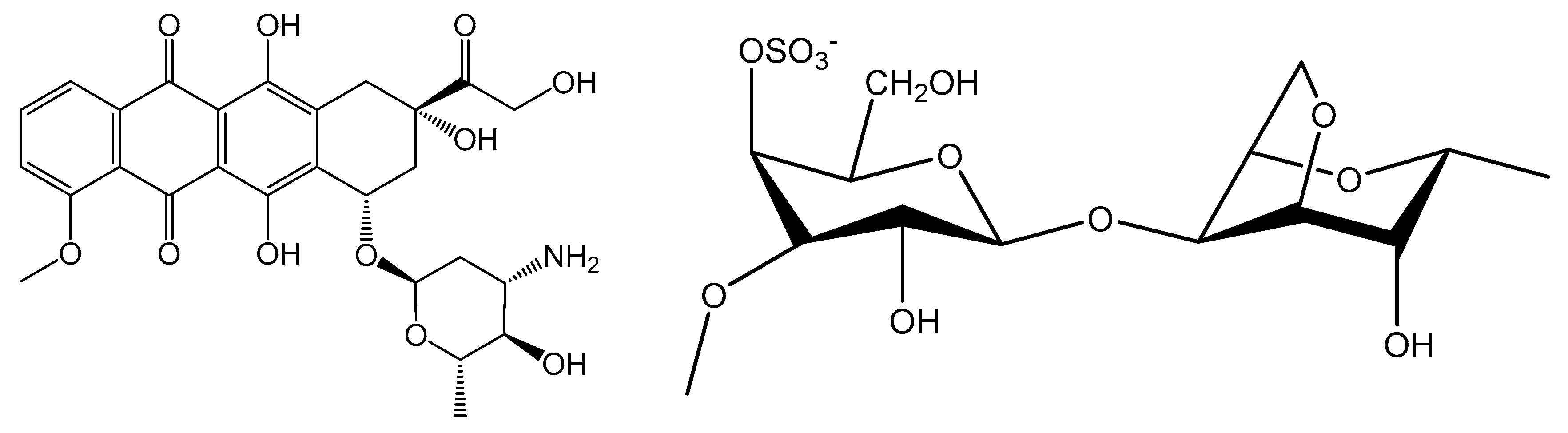
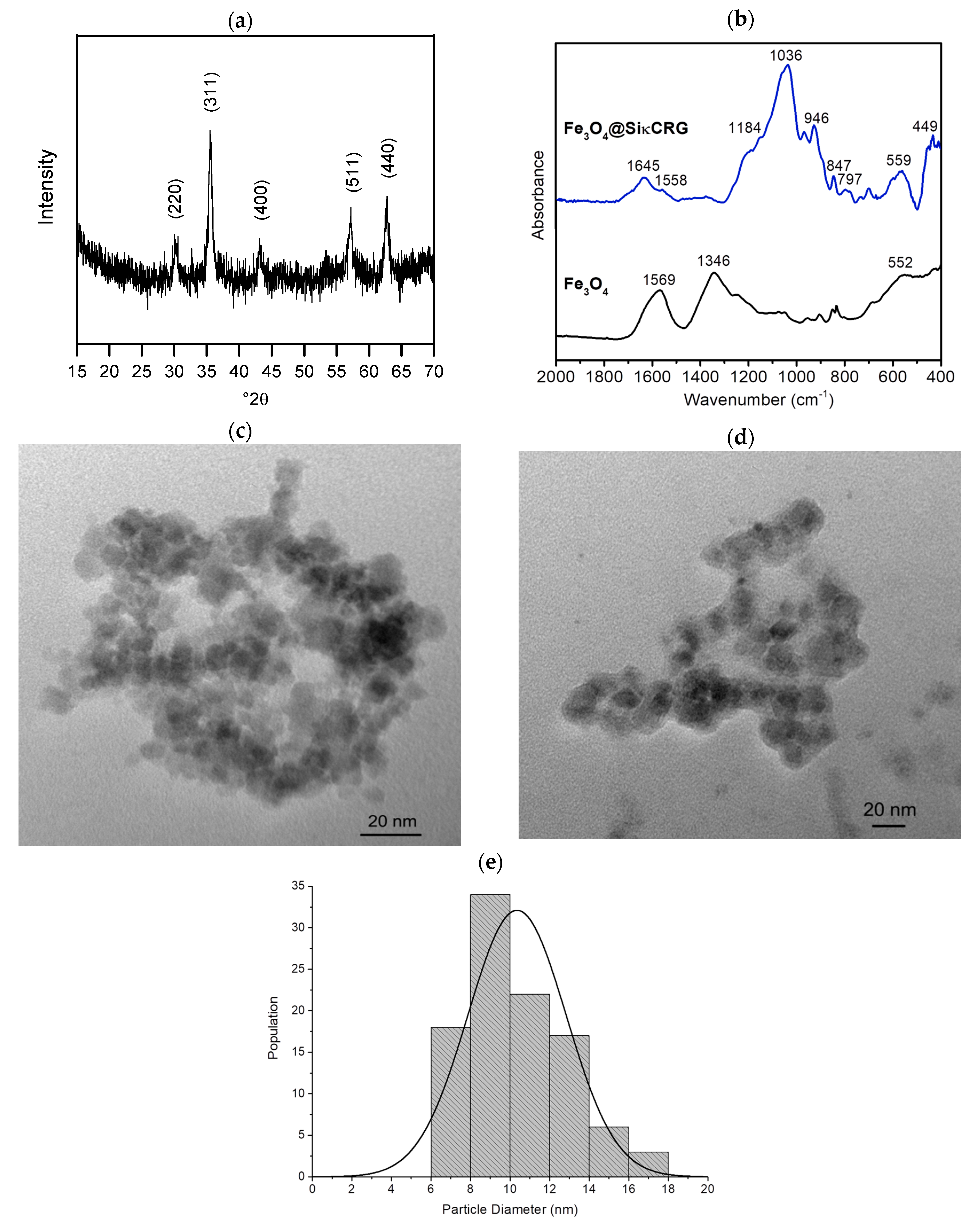
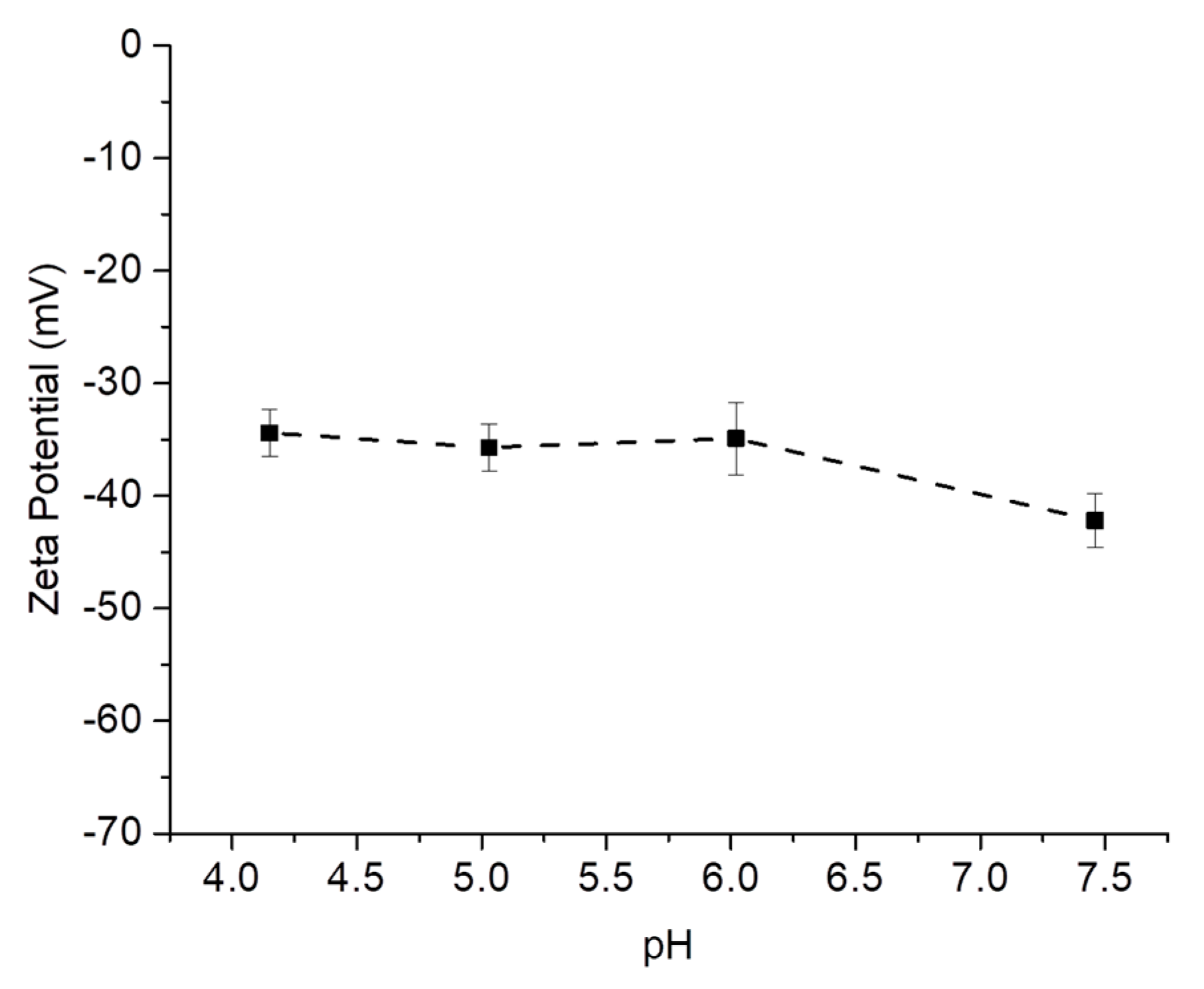
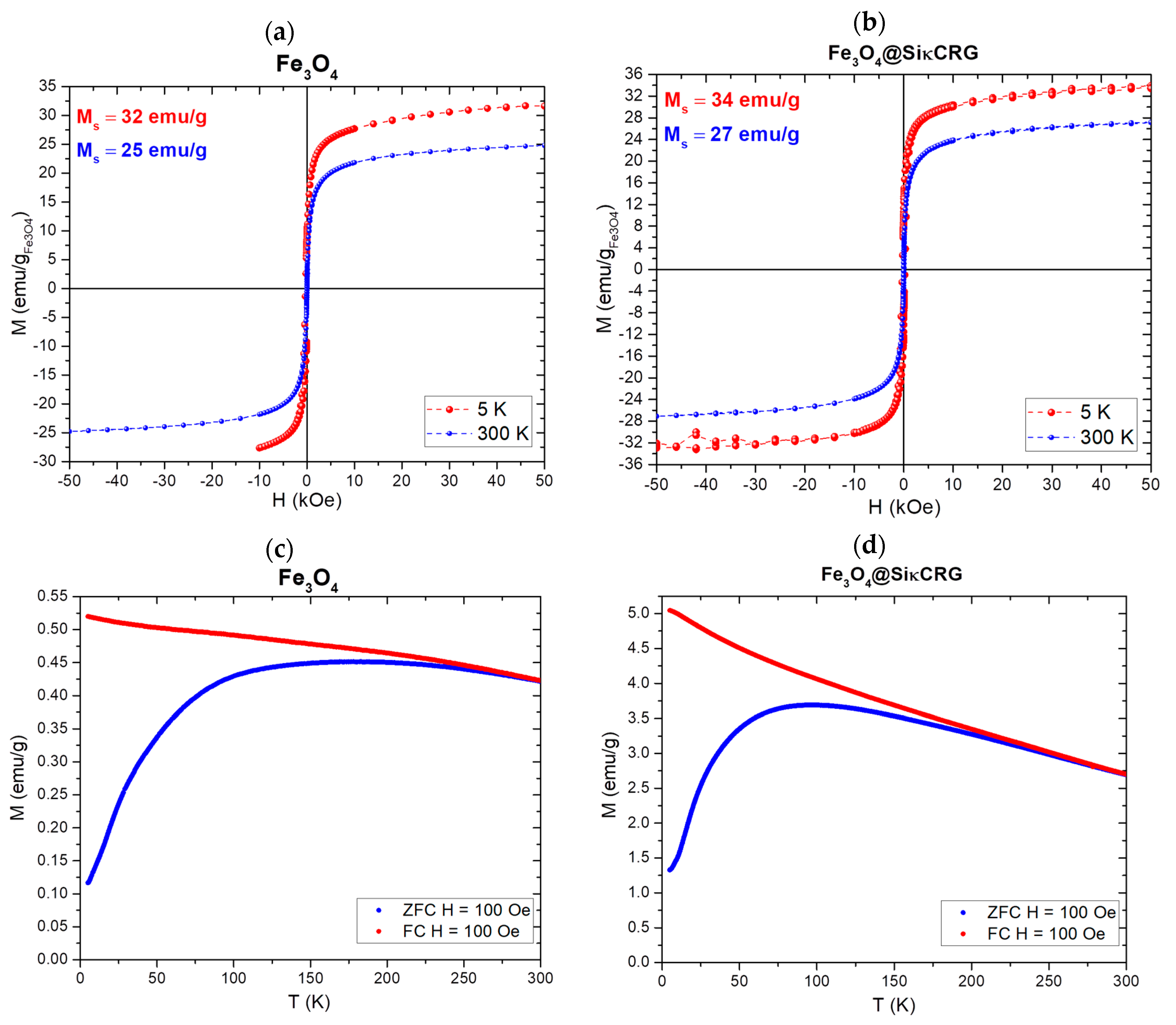

| Sample | D (nm) | T (nm) | C (%) | H (%) | S (%) | SBET (m2·g−1) |
|---|---|---|---|---|---|---|
| Fe3O4 | 10 ± 2.5 | 6.55 | 1.28 | 152.00 | ||
| Fe3O4@SiκCRG | 10 ± 2.5 | 5.2 ± 2.1 | 13.85 | 2.27 | 1.27 | 13.94 |
| Material | Ms (emu/gsample) | Ms (emu/gFe3O4) | Mr (emu/gFe3O4) | Hc (Oe) |
|---|---|---|---|---|
| Fe3O4 | 23 | 25 | 1.2 | 31 |
| Fe3O4@SiĸCRG | 15 | 27 | 1.9 | 32 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nogueira, J.; Soares, S.F.; Amorim, C.O.; Amaral, J.S.; Silva, C.; Martel, F.; Trindade, T.; Daniel-da-Silva, A.L. Magnetic Driven Nanocarriers for pH-Responsive Doxorubicin Release in Cancer Therapy. Molecules 2020, 25, 333. https://doi.org/10.3390/molecules25020333
Nogueira J, Soares SF, Amorim CO, Amaral JS, Silva C, Martel F, Trindade T, Daniel-da-Silva AL. Magnetic Driven Nanocarriers for pH-Responsive Doxorubicin Release in Cancer Therapy. Molecules. 2020; 25(2):333. https://doi.org/10.3390/molecules25020333
Chicago/Turabian StyleNogueira, João, Sofia F. Soares, Carlos O. Amorim, João S. Amaral, Cláudia Silva, Fátima Martel, Tito Trindade, and Ana L. Daniel-da-Silva. 2020. "Magnetic Driven Nanocarriers for pH-Responsive Doxorubicin Release in Cancer Therapy" Molecules 25, no. 2: 333. https://doi.org/10.3390/molecules25020333
APA StyleNogueira, J., Soares, S. F., Amorim, C. O., Amaral, J. S., Silva, C., Martel, F., Trindade, T., & Daniel-da-Silva, A. L. (2020). Magnetic Driven Nanocarriers for pH-Responsive Doxorubicin Release in Cancer Therapy. Molecules, 25(2), 333. https://doi.org/10.3390/molecules25020333











