New Frontiers in Nucleic Acid Chemistry
A topical collection in Molecules (ISSN 1420-3049). This collection belongs to the section "Chemical Biology".
Submission Status: Closed (12 May 2026) | Viewed by 103407Editors
2. Networking Center on Bioengineering, Biomaterials and Nanomedicine (CIBER-BBN), Jordi Girona 18-26, E-08034 Barcelona, Spain
Interests: siRNA; antisense oligonucleotides; G-quadruplex; DNA triplex; i-motif; DNA nanotechnology; gene silencing; aptamers; drug delivery; DNA; RNA; oligonucleotide synthesis
Special Issues, Collections and Topics in MDPI journals
Interests: G quadruplexes; supramolecular chemistry; synthetic organic chemistry of carbohydrates; nucleobases; nucleosides; C-nucleosides; peptide nucleic acids; heterocycles; protecting groups
Special Issues, Collections and Topics in MDPI journals
Interests: oligonucleotides; nucleoside analogues; unusual structures of DNA; G-quadruplex; Ru(III)-complexes; Pt(II)-complexes; metal–DNA interactions; small molecule–DNA interactions
Special Issues, Collections and Topics in MDPI journals
Topical Collection Information
Dear Colleagues,
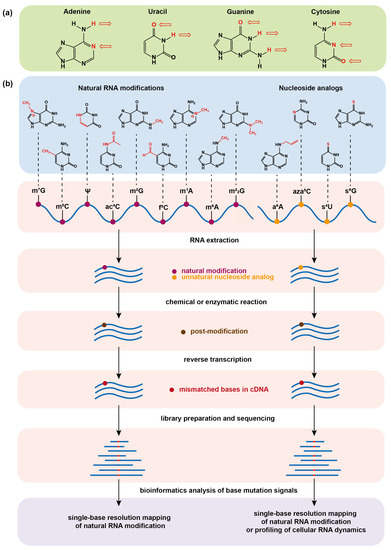
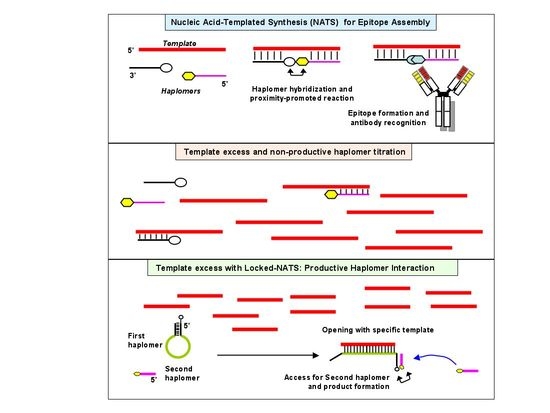
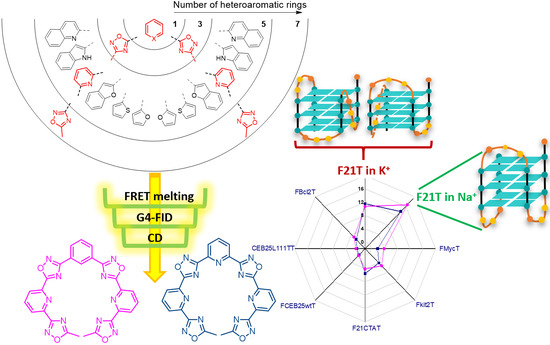
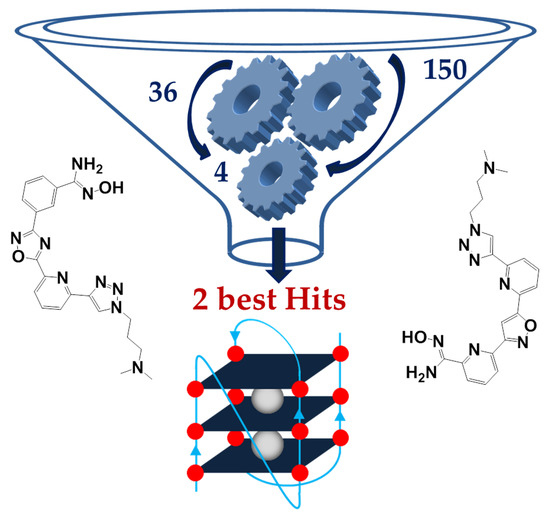
After the success of the Special Issues “Nucleic Acid Analogs”, edited by Molecules in 2012, and “Frontiers in Nucleic Acid Chemistry”, edited in 2015, we are happy to announce the launch of a Topical Collection on these topics. Synthetic oligonucleotides have become essential tools for biological, biomedical, and nanotechnology researches and have also shown promising results in therapeutics and diagnostics. Novel oligonucleotide derivatives with tailored properties are being continuously developed since their specific recognition and unique folding characteristics allow to obtain potential drugs or drug delivery agents, biosensors, diagnostic agents, etc. Along with this development, nucleic acid research is taking new directions as well: synthesis and applications of even more complex molecules, like synthetic RNA analogues (peptide nucleic acids, locked nucleic acids, morpholino oligos, conjugates, etc.), highly informative analytical methods (e.g., those based on mass spectrometry), study of higher-order structures (triple and quadruple helices), investigation of molecular electronic devices based on smart, self-assembling oligonucleotide analogues, to name just a few. The study of the properties of modified nucleic acids brings together many different branches of science, such as molecular biology, chemistry, biochemistry, medicinal chemistry, medicine, materials science, and synthetic biology.
The Topical Collection of Molecules, “New Frontiers in Nucleic Acid Chemistry”, will concentrate on the latest developments in nucleic acids chemistry. We cordially invite all the researchers involved in this exciting field to contribute to the continuing success of the Topical Collection “New Frontiers in Nucleic Acid Chemistry”.
Prof. Dr. Ramon Eritja
Dr. Lajos Kovács
Prof. Dr. Daniela Montesarchio
Collection Editors
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Molecules is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2700 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- modified oligonucleotides
- artificial nucleic acids
- xeno nucleic acids
- synthetic genes
- oligonucleotide conjugates
- nucleoside/nucleotide analogs
- G-quadruplex
- i-motif
- triplex
- antisense
- siRNA
- RNA interference
- antiviral activity
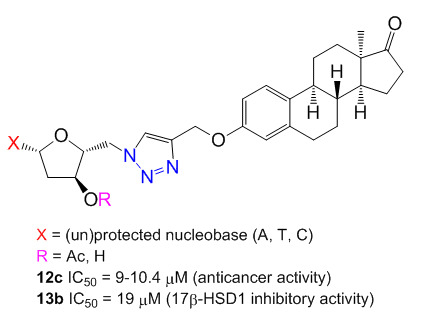
- anticancer activity
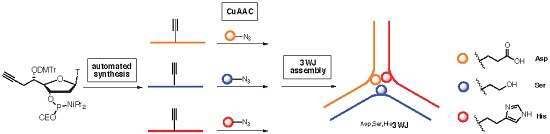
- DNA nanobiotechnology
- nanomaterials
- aptamers
- biosensors
- surface-plasmon resonance
- fluorescent nucleic acids
- locked nucleic acids
- unlocked nucleic acids
- peptide nucleic acids
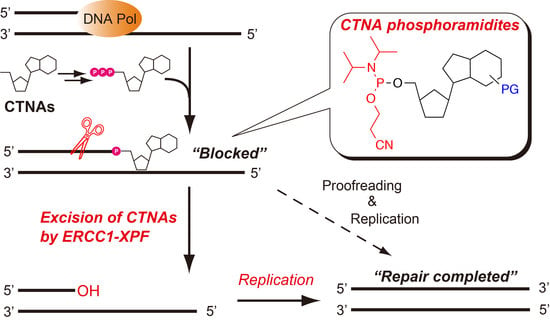
- DNA repair
- base-modified DNA
- DNA polymerase
- circular nucleic acids
- NMR of nucleic acids
- mass spectrometry of nucleic acids
- biophysical studies
Related Special Issues
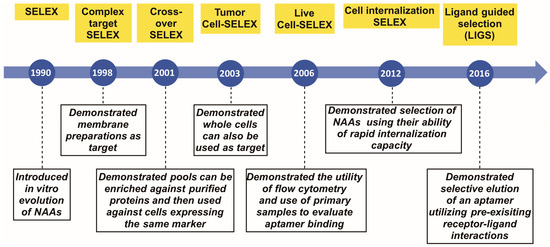
- Aptamers: Past, Present, and Future in Molecules (22 articles - displayed below)
- Frontiers in Nucleic Acid Chemistry in Molecules (24 articles - displayed below)
- Nucleic Acid Analogs in Molecules (30 articles - displayed below)
- Catalytic Nucleic Acids in Molecules (23 articles - displayed below)
- Nucleic Acids in Molecules (26 articles - displayed below)