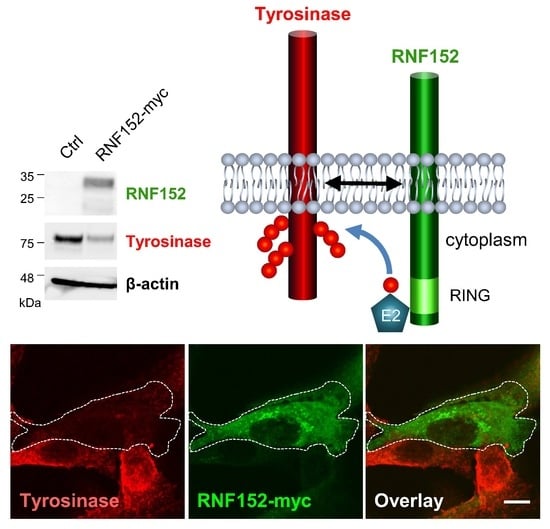
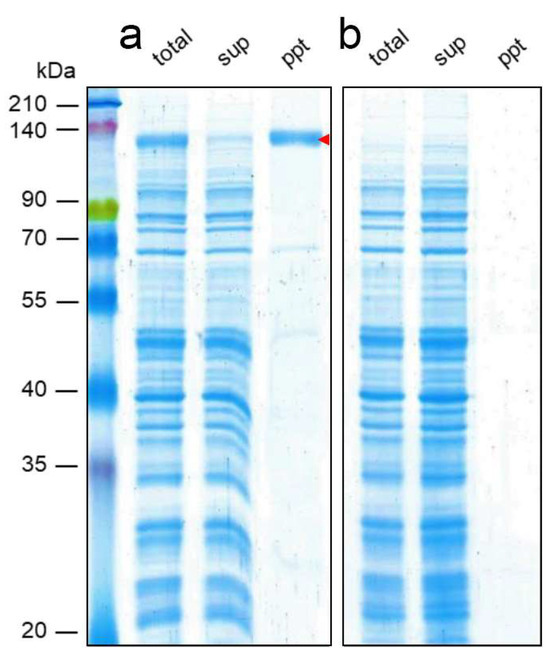
Feature Papers in Biological Membrane Composition and Structures
A topical collection in Membranes (ISSN 2077-0375). This collection belongs to the section "Biological Membranes".
Viewed by 10889Editor
2. Data Science Center, Nara Institute of Science and Technology, Nara 630-0192, Japan
3. Center for Digital Green-Innovation, Nara Institute of Science and Technology, Nara 630-0192, Japan
Interests: mechanisms of cell shaping and cell fate determination; cell biology
Special Issues, Collections and Topics in MDPI journals
Topical Collection Information
Dear Colleagues,
Biological membranes, which include cellular membranes and membrane particles derived from cells, are an essential component of life. Biological membranes consist of various lipids and embedded or associated proteins with or without modifications by sugar and different other materials. The structure of the biological membrane is the result of the assembly of such a variety of materials and exhibits the structure for their functions. This feature paper collection will include but are not limited to:
- The reconstitution studies of the biological membrane components,
- The subcellular organelle studies,
- The extracellular vesicles studies,
- The membrane-embedded/associated protein studies
- Membrane shaping of cells
- Membrane shaping of subcellular organelles
- Membrane shaping of artificial liposomes
- Transport across membranes
- Cell function related to membrane morphogenesis
- Transport by membrane vesicles
- Membrane interaction with proteins, sugars, and other biological materials
- Biological membrane synthesis
- Application of biological membranes and mimicry in industry and research
- Visualization of membranes.
Prof. Dr. Shiro Suetsugu
Collection Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Membranes is an international peer-reviewed open access monthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2200 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.