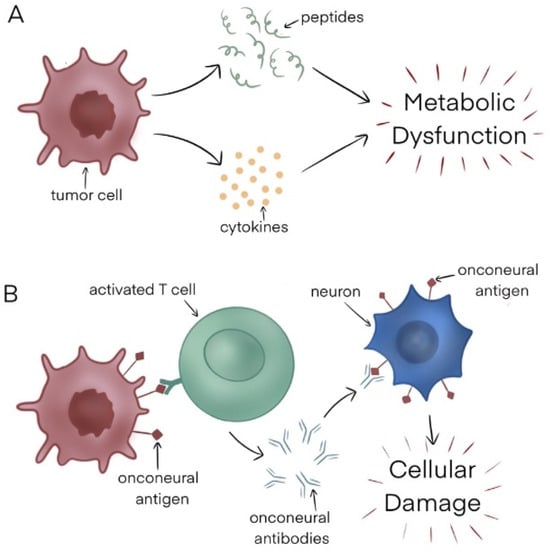
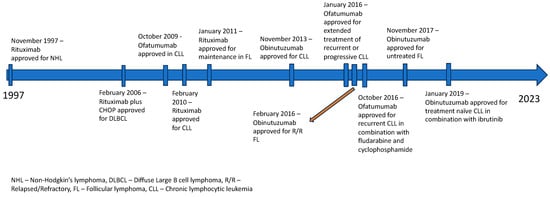
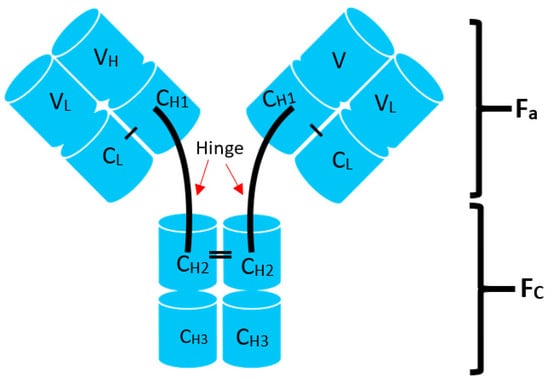
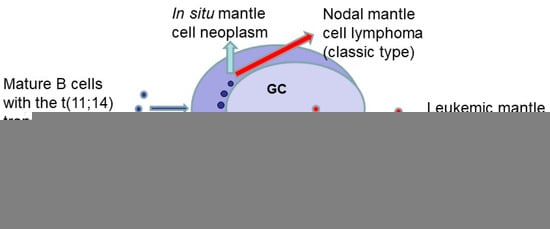
Lymphomas
A topical collection in Lymphatics (ISSN 2813-3307).
Viewed by 65380Editors
Interests: lymphomas
Topical Collection Information
Dear Colleagues,
This Collection of the Lymphatics journal aims to enable the rapid publication of contributions concerning all aspects of lymphoma research, ranging from basic research to clinical applications. Topics include, but are not limited to, the following:
- Improving the assessment of prognosis in lymphomas using clinical and molecular markers;
- Improved understanding of environmental contributors to the development of lymphoma;
- Novel cellular therapies in lymphoma;
- Identification of appropriate patients and cell therapy products;
- Methods of limiting the toxicity of treatment;
- Understanding mechanisms of resistance to current therapies;
- Optimizing post-treatment surveillance strategies in patients with aggressive lymphomas.
Dr. Jonathon B. Cohen
Dr. Rahul Lakhotia
Collection Editors
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Lymphatics is an international peer-reviewed open access quarterly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 1000 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.