Abstract
Bone marrow involvement in B-cell non-Hodgkin lymphoma is an adverse prognostic factor; therefore, its detection is necessary at the time of diagnosis and during follow-up. This study evaluates the concordance between flow cytometry (FC) and bone marrow biopsy (BMB) in detecting bone marrow involvement in B-cell non-Hodgkin lymphomas as well as their prognostic relevance in a Chilean cohort. A total of 202 samples from 172 patients with diffuse large B-cell (DLBCL), follicular (FL), marginal-zone (MZL), and mantle cell (MCL) lymphoma were retrospectively analyzed; all patients underwent simultaneous BMB and FC. Bone marrow involvement was identified in 29% of samples via BMB and in 40% via FC, with an overall concordance of 89% (kappa: 0.75), which was lower in mantle cell lymphoma. Eleven percent of cases showed BMB-FC+ discordance, generally associated with low tumor burden. In survival analyses, the BMB+/FC+ group exhibited shorter overall and progression-free survival, and concordant involvement was associated with a higher risk of mortality and progression, particularly among patients with an intermediate or high IPI. Involvement detected exclusively by FC did not have a significant prognostic impact. These findings support the role of FC as a complementary or alternative diagnostic tool in settings with limited resources, improving sensitivity for detecting bone marrow involvement without compromising clinical relevance.
1. Introduction
Non-Hodgkin lymphomas (NHLs) comprise a broad and heterogeneous group of malignant neoplasms derived from mature lymphoid cells, 85–90% of which originate from B cells [1,2]. Their diagnosis relies on the integration of clinical, morphological, immunophenotypic, genetic, and molecular information [3]. In Chile, the most frequent B-cell lymphoma subtypes include diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), marginal-zone lymphoma (MZL), and mantle cell lymphoma (MCL) [4].
Bone marrow (BM) involvement in B-cell NHL has been associated with advanced disease stages and observed in approximately 20–40% of cases in Latin American studies [5,6], with considerable variability across subtypes (e.g., around 10–30% for DLBCL, and the rate for indolent lymphomas such as FL ≈ 40–70%). Its detection may influence therapeutic strategies in certain cases [7,8,9]. Traditionally, the diagnosis is based on a bone marrow biopsy (BMB), an invasive procedure associated with patient discomfort and potential complications. Therefore, imaging techniques such as positron emission tomography/computed tomography (PET-CT) scans are increasingly used in clinical practice [10]; however, their use is limited in the public healthcare setting. In this context, bone marrow aspiration with flow cytometry (FC) represents a less invasive and complementary method for detecting bone marrow infiltration that can be performed on aspirated bone marrow obtained at the same time as a BMB, providing faster results at a potentially lower cost [11]. Although the concordance between both techniques is high (60–80%) [12,13], the broader implementation of FC for this purpose in routine clinical practice remains limited.
This report describes the experience of a Chilean public center regarding the concordance between FC and BMB in the diagnosis of BM involvement in B-cell NHL as well as the prognostic significance of BM infiltration across the main B-cell non-Hodgkin lymphoma subtypes.
2. Results
2.1. Patients, Demographics, and Clinical Characteristics
A total of 202 samples from 172 patients were included, 84 (49%) of whom were male, and the mean age of the overall population was 61 years (range: 18–91). Of the total number of samples, 133 were collected at diagnosis, and 69 were collected during follow-up. At diagnosis, 58% of patients presented B symptoms, and 67% showed extranodal involvement, most commonly with one or two sites affected. Seventy-five percent of patients were in advanced stages (Ann Arbor III–IV) and had an intermediate–high or high International Prognostic Index (IPI). The characteristics of the cohort are summarized in Table 1.

Table 1.
Baseline demographic and clinical characteristics of the study cohort.
Of all the samples, 90 (45%) corresponded to DLBCL, 48 (24%) corresponded to FL, 32 (16%) corresponded to MCL, and 30 (15%) corresponded to MZL.
2.2. Concordance Between Flow Cytometry and Bone Marrow Biopsy
Overall, bone marrow infiltration was detected in 58 of 202 samples (29%) via BMB and in 81 of 202 samples (40%) via FCM (p = 0.016). A total of 23 of 202 samples (11%) showed discordant results, all of them BMB-FCM+, with a mean infiltration of 0.44% (range: 0.003–3%), which is significantly lower than the 22.33% (range: 0.01–90%) observed in BMB+/FCM+ cases (p < 0.0001). Most discordant cases were detected at the time of diagnosis (16/23) (p = 0.54).
The overall concordance between both techniques for detecting bone marrow involvement was 89% (179/202), with a Kappa index of 0.75 (substantial). According to histological subtype, substantial concordance was observed for MZL, 90% (κ = 0.80); DLBCL, at 92% (κ = 0.73); and FL, at 88% (κ = 0.73). In the case of MCL, concordance was moderate, at 78% (κ = 0.47). A summary of the concordance between flow cytometry and bone marrow biopsy is presented in Table 2.

Table 2.
Concordance between bone marrow biopsy (BMB) and flow cytometry (FC) according to histological subtype.
2.3. Clinical Outcomes in the Overall Cohort
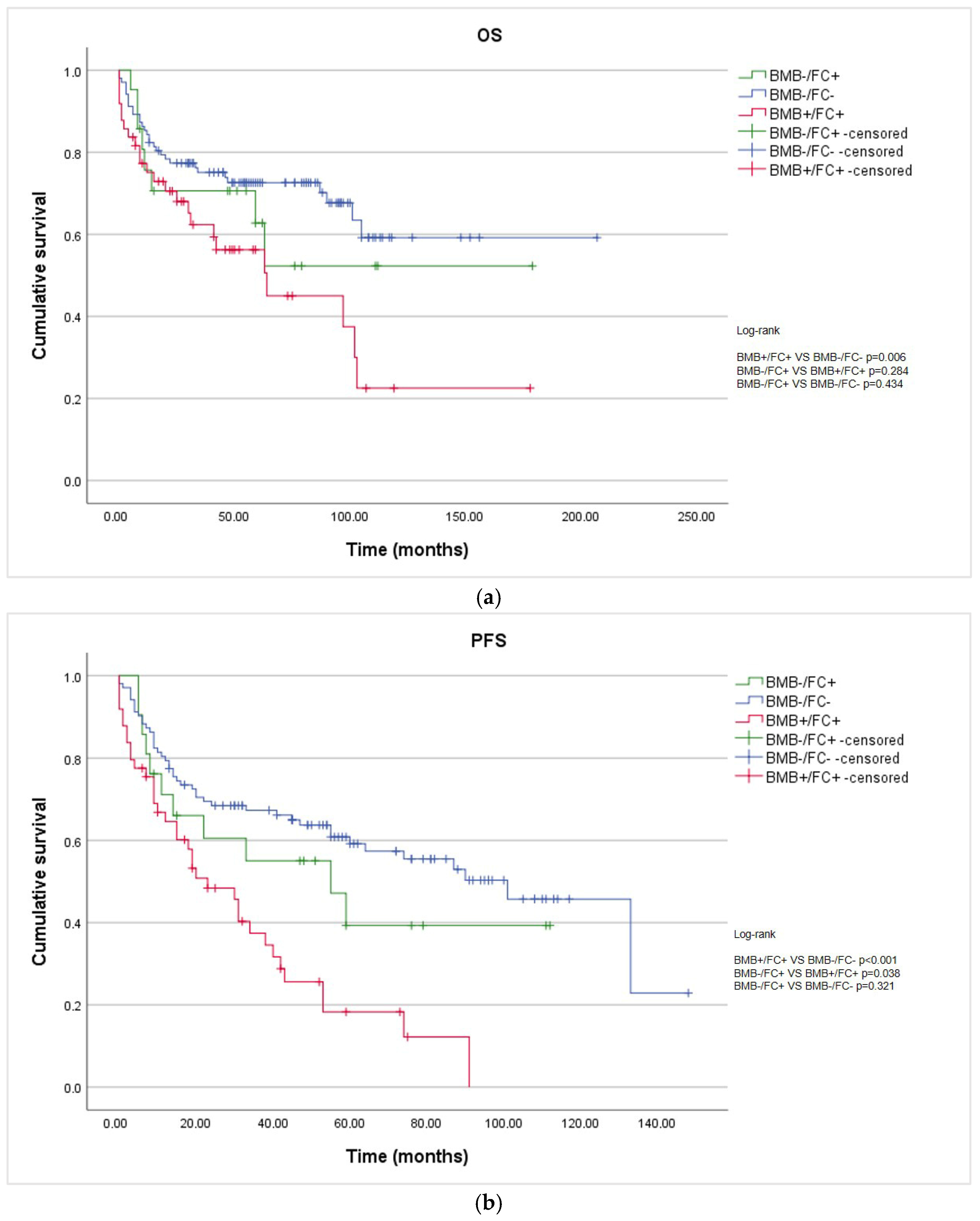
In the overall cohort, the median overall survival (OS) was 105 months, while the median progression-free survival (PFS) was 55 months (95% CI: 34.6–75.4). The median OS for the BMB+/FC+ group was 64 months (95% CI: 27.7–100.3), whereas the median OS for the BMB-/FC+ and BMB-/FC- groups was not reached. The median PFS for the BMB+/FC+, BMB-/FC+, and BMB-/FC- groups was 23 months (95% CI: 12.2–33.8), 55 months (95% CI: 15.8–94.2), and 101 months (95% CI: 76.1–125.9), respectively. The BMB+/FC+ group showed significantly shorter OS and PFS compared with the BMB-/FC- group (log-rank p = 0.006 and p < 0.001, respectively) (Figure 1a,b). Bone marrow infiltration detected exclusively via FC (BMB-/FC+) did not show prognostic significance for OS compared with the other groups (p = 0.43 and p = 0.28, respectively). However, for PFS, this subgroup showed a statistically significant difference when compared with the infiltrated group (BMB+/FC+) p = 0.038.
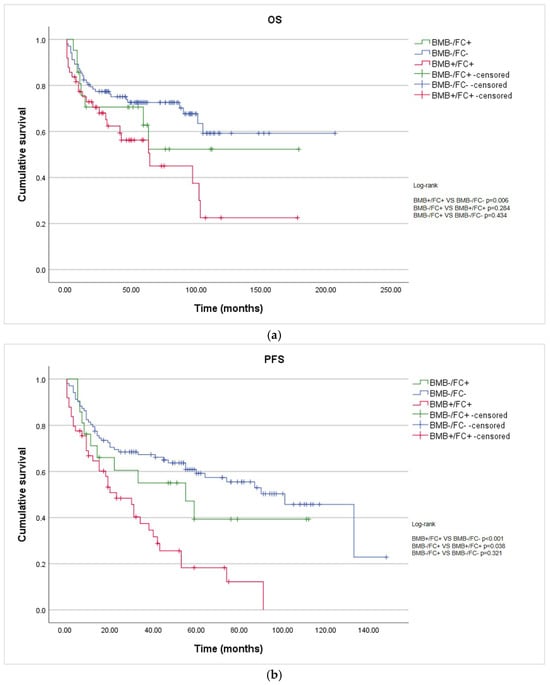
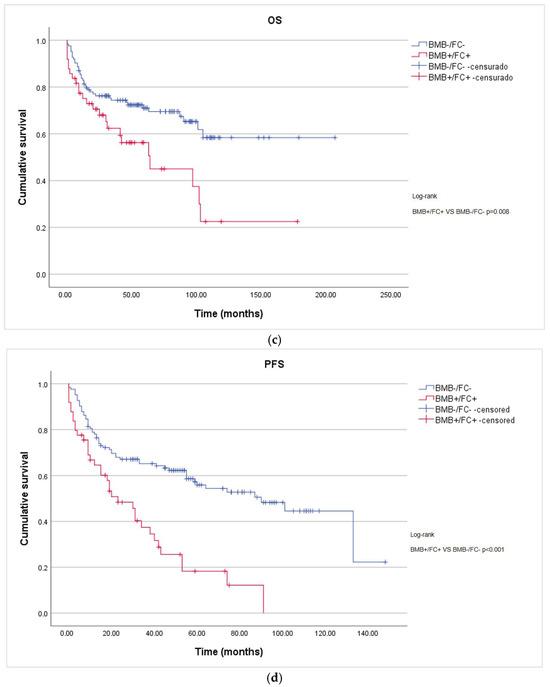
Figure 1.
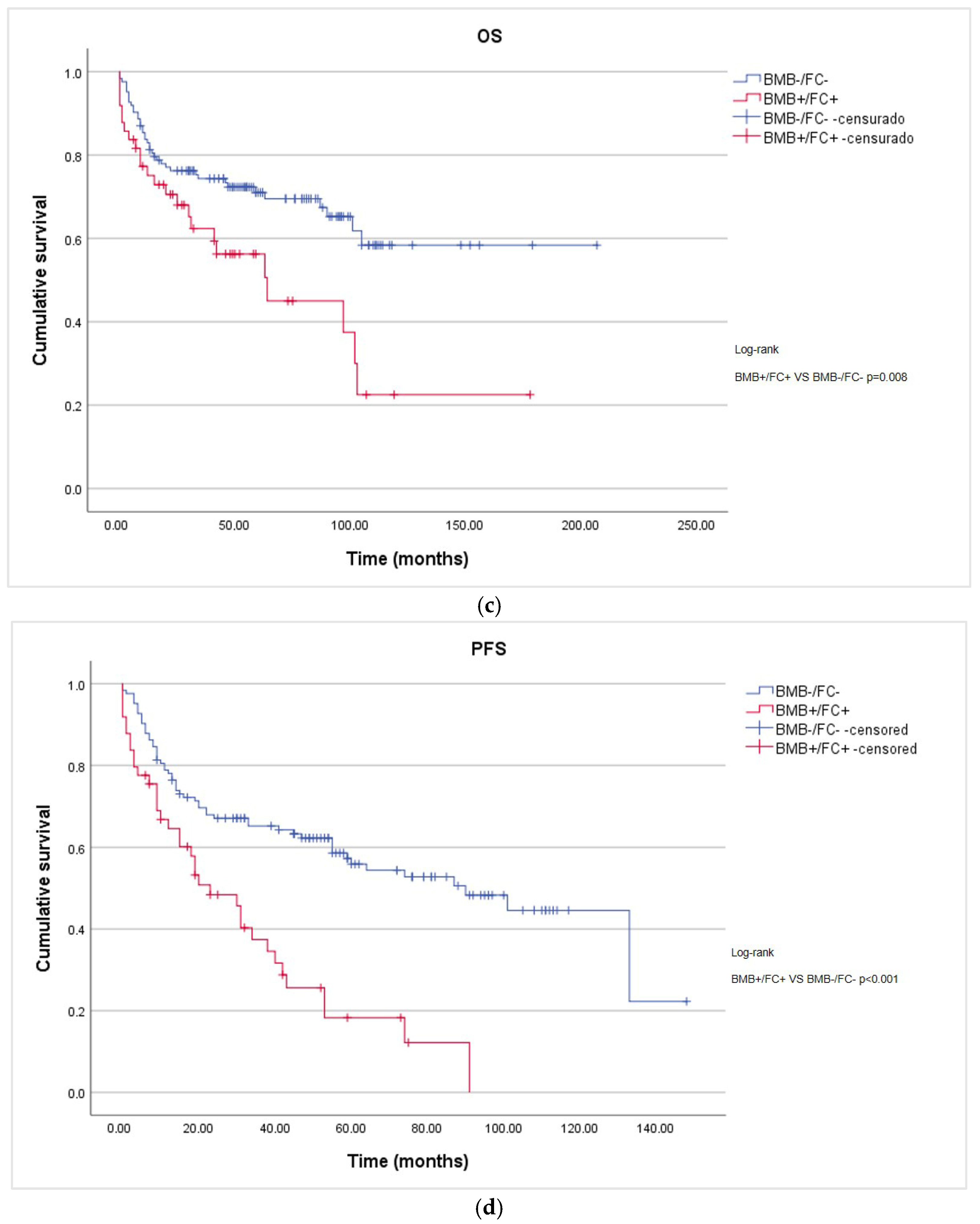
Kaplan–Meier survival curves according to bone marrow involvement assessed via BMB and FC: (a) OS of non-infiltrated (BMB-FC-), discordant (BMB-/FC+), and infiltrated (BMB+/FC+) patients; (b) PFS of non-infiltrated (BMB-/FC-), discordant (BMB-/FC+), and infiltrated (BMB+/FC+) patients; (c) OS considering discordant cases among the non-infiltrated patients; and (d) PFS considering discordant cases among the non-infiltrated patients.
Reclassifying BMB-/FC+ cases as BMB-/FC- resulted in better separation of the survival curves than when they were classified as positive (OS p = 0.008 and PFS p < 0.001). (Figure 1c,d)
2.4. Clinical Outcomes According to Histological Subtype
In regard to FL, patients in the BMB+/FC+ group showed significantly shorter OS (p = 0.039) and PFS (p < 0.001) compared with the BMB-/FC- group (Supplementary Material Figure S1). In the remaining histological subtypes, bone marrow involvement did not demonstrate a statistically significant prognostic impact (Supplementary Material Figures S2–S4).
2.5. Univariate and Multivariate Analysis
In the univariate analysis, bone marrow infiltration (BMB+/FC+) was associated with a significantly increased risk of both mortality and disease progression compared with the BMB-/FC- group (OS: HR = 1.96; 95% CI 1.18–3.27; p = 0.01; PFS: HR = 2.60; 95% CI 1.69–4.00; p < 0.0001).
In the multivariate analysis, which included diagnostic concordance and IPI values, patients without bone marrow infiltration (BMB-/FC-) showed a progressive increase in the risk of both OS and PFS as the IPI category increased. For OS, the intermediate-risk group (including both low and high categories) had an HR of 2.45 (95% CI 1.08–5.55; p = 0.032), and the high-risk group had an HR of 3.71 (95% CI 1.47–9.36; p = 0.005). For PFS, the HRs were 2.10 (95% CI 1.10–3.99; p = 0.024) and 3.59 (95% CI 1.70–7.58; p = 0.001), respectively. In the BMB+/FC+ group, an additional increase in risk was observed. The remaining variables included in the model did not reach statistical significance (p > 0.05). A summary of the multivariate analysis is presented in Table 3.

Table 3.
Multivariate regression model of bone marrow involvement and IPI scores for OS and PFS.
3. Discussion
The results of this study highlight the high diagnostic concordance between FC and BMB (89%) for the detection of bone marrow involvement in B-cell lymphomas at diagnosis or during follow-up, findings consistent with previously published rates (69–92%) [12,13,14,15,16]. Sorigué et al. conducted a systematic review assessing agreement using Cohen’s kappa in B-NHL, stratified by histological subtype, reporting overall concordance within the substantial range (0.65–0.73). We found slightly higher values, with a kappa of 0.75. When analyzing by histology, we again observed concordance levels above those previously reported, particularly in regard to DLBCL, for which we obtained a kappa of 0.73 (substantial), higher than the 0.38–0.59 range described by Sorigué et al. In contrast, MCL exhibited the lowest concordance (kappa: 0.47), whereas the literature reports values ranging from 0.56 to 0.73 [17]. The scarcity of studies evaluating concordance between FC and BMB by histological subtype, especially in Latin America, underscores the relevance of our series. Taken together, these results support the clinical use of FC as a complementary method with respect to BMB.
Discordance between FC and BMB reached 11%, corresponding exclusively to cases determined to be positive via FC and negative via BMB. This proportion is comparable to that reported by Kim B. et al. (8.5%), a difference that may be partly explained by the higher technical resolution of the eight-color FC used in our study compared with the six-color assay used in theirs [16]. Ramanlingam et al. reported a discordance of 12% using a 10-color FC and also found no statistically significant difference between the tumor burdens of BMB+/FC+ and BMB_/FC+ cases, suggesting that negative BMB results are not solely attributable to tumor load. They may also reflect focal or patchy infiltration patterns where involvement is not uniformly distributed and may be missed at the biopsy site. In addition, the cited authors noted that detection of a low-level tumor burden requires >100,000 acquired events [15], a threshold far exceeded in our study (range, 165,568–5,000,000; median, 1,000,000), reinforcing the validity of results determined to be positive via FC in discordant cases.
Regarding clinical outcomes, in our cohort, we observed significantly worse OS and PFS in the BMB+/FC+ group compared with the BMB_/FC- group, indicating that simultaneous detection of marrow involvement via both techniques identifies a subgroup with poorer prognosis, a finding also reported by Okamoto et al. [18]. In our multivariate analysis, this increased risk of mortality and progression doubled or tripled among patients with higher IPI scores. Although the BMB-/FC+ group showed a trend toward shorter OS and PFS, statistical significance was not reached, likely due to the limited number of cases and the low tumor burden detected. The mean infiltration among discordant cases was 0.44% (range 0.003–3%), which provoked us to classify these cases within the BMB-/FC- group. Cheson et al. similarly reported that infiltration <2% detected exclusively by FC may be considered clinically insignificant, with no impact on patient outcomes [10,19].
Prognostic information regarding bone marrow involvement detected via FC and/or BMB in individual histological subgroups remains controversial. In our series, only for FL was a statistically significant decrease in OS and PFS observed in the BMB+/FC+ group, a finding not previously reported. In regard to DLBCL, Arima et al. noted that minimal marrow involvement detected by FC or PCR in cases determined to be negative via BMB did not significantly differ from BMB-/FC- patients prior to treatment; however, it predicted severe hematologic toxicity and negatively influenced PFS and OS following R-CHOP therapy [20]. Similar findings were reported by Martín-Moro et al. and Greenbaum et al., where minimal involvement detected only by FC was associated with poorer OS and PFS and represented an independent prognostic factor [21,22]. These associations were not observed in our cohort (p = 0.4 and p = 0.06, respectively).
The main limitations of our report include its single-center design, the lack of uniformity of FC panels over time, and the limited number of patients in each histological subgroup, restricting the conclusions regarding prognostic impact. Future multicenter studies with larger sample sizes are needed to confirm our findings.
4. Materials and Methods
4.1. Study Design and Patients
This was a retrospective, single-center study. Medical records of patients aged ≥18 years with a diagnosis of DLBCL, FL, MZL, or MCL confirmed by tissue biopsy and treated at Hospital Guillermo Grant Benavente (HGGB) between 1 January 2014 and 31 December 2024 were reviewed. Patients were included if they had undergone simultaneous BMB and bone marrow aspirate for immunophenotyping by FC, either as part of the initial staging work-up or during follow-up. Demographic and clinical data, including age, sex, Ann Arbor stage, IPI, and laboratory findings, were collected. The study was approved by the Ethics Committee of HGGB and Universidad de Concepción.
The primary endpoint was the concordance between FC and hematopathology in detecting bone marrow involvement in lymphoma. The secondary endpoints included the prognostic significance of marrow involvement with respect to OS and PFS.
4.2. Flow Cytometry Immunophenotyping
FC analysis was performed using an 8-color panel consisting of CD20-V450, CD45-V500, Lambda-FITC, Kappa-PE, CD5-PerCP-Cy5.5, CD19-PE-Cy7, CD10-APC, and CD38-APC-H7 in 168 of 202 cases (83.2%). In 8 cases (4%), the EuroFlow Lymphoid Screening Tube (LST) panel was used [23]. In 26 cases (12.8%), a modified panel for MCL was applied, including CD20-V450, CD45-V500, Lambda-FITC, Kappa-PE, CD5-PerCP-Cy5.5, CD19-PE-Cy7, CD200-APC, and CD38 or CD43-APC-H7. The selection of monoclonal antibodies was based on those suggested by the EuroFlow consortium [23]. Samples were acquired on a FACSCanto II flow cytometer (BD Biosciences, Sa Jose, CA, USA), with a median of 1,000,000 acquired events per sample (range: 165,568–5,000,000). Data analysis was performed using Infinicyt® software, version 2.0 (Cytognos SL, Salamanca, Spain). A sample was considered positive for lymphoma infiltration if the presence of clonal mature B cells with light chain restriction was demonstrated. A value of at least 20 events was considered positive for detection, and a value of 50 events was considered positive for quantification. Accordingly, the limit of detection was 0.01% and the limit of quantification was 0.03% for samples with the lowest number of acquired events, whereas the limit of detection was 0.0004% and the limit of quantification was 0.001% for samples with event numbers at the upper limit of the acquisition range.
4.3. Bone Marrow Biopsy
BMB samples were fixed in formalin, decalcified in EDTA, processed, sectioned, and stained with routine hematoxylin and eosin in the Pathology Department. Immunohistochemistry was performed in all cases. A sample was considered positive if cells with a morphology and immunophenotype, as assessed by immunohistochemistry, compatible with lymphoma were identified.
4.4. Statistical Analysis
Statistical analyses were performed using SPSS software version 26.0. A p-value < 0.05 (95% CI) was considered statistically significant. Categorical variables were compared using the chi-square test. Concordance between techniques was assessed using Cohen’s kappa coefficient, interpreted according to standard qualitative categories: no agreement (<0), slight (0.01–0.20), fair (0.21–0.40), moderate (0.41–0.60), substantial (0.61–0.80), and almost perfect (0.81–1.00) [24].
Survival analyses were estimated using the Kaplan–Meier method, with comparisons between curves performed using the log-rank test. To identify independent predictors of survival, a Cox proportional hazards model was applied. Initially, univariate analyses were conducted for each variable of interest, followed by analysis using a multivariate model that included variables with statistical significance or clinical relevance. Results are reported as HRs with 95% CIs.
5. Conclusions
Traditionally, BMB has been considered the gold standard for assessing lymphoid infiltration in bone marrow. However, more sensitive techniques such as flow cytometry (FC) have demonstrated the ability to detect minimal residual disease or subclinical involvement that may not be evident through conventional histology. Unlike BMB, FC provides diagnostic information in a shorter time. In addition, FC allows rapid and quantitative evaluation of millions of cells, offering detailed immunophenotypic characterization, which makes it a highly valuable diagnostic tool. Collectively, the lower invasiveness of the bone marrow aspirate, faster turnaround time, and analytical capacity position FC as a complementary or alternative approach to traditional histopathological assessment.
Considering the Chilean and Latin American context, where the availability of diagnostic resources such as PET-CT remains highly variable, this study provides evidence supporting the utility of integrating FC as a complementary or alternative tool with respect to BMB for evaluating bone marrow involvement in B-cell non-Hodgkin lymphomas.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/lymphatics4010014/s1. Figure S1: Kaplan–Meier survival curves according to bone marrow involvement assessed by BMB and FC in FL. Figure S2: Kaplan–Meier survival curves according to bone marrow involvement assessed by BMB and FC in DLBCL. Figure S3: Kaplan–Meier survival curves according to bone marrow involvement assessed by BMB and FC in MCL. Figure S4: Kaplan–Meier survival curves according to bone marrow involvement assessed by BMB and FC in MZL.
Author Contributions
Conceptualization, M.C.; methodology, M.C., C.O., L.V. and P.H.; investigation, L.V. and C.O.; validation, S.V. and C.C.; formal analysis, P.H.; resources, M.C. and C.C.; data curation, L.V.; writing—original draft preparation, L.V. and M.C.; writing—review and editing, L.V. and M.C.; visualization, S.V. and P.H.; supervision, M.C.; project administration, L.V.; funding acquisition, L.V. and M.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Facultad deE Medicina, Universidad de Concepción Fondos de Apoyo a la Investigación (FAI 2025), under grant number FMED.INV.2025.029.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by Comité Ético Científico del Servicio de Salud Concepción (protocol code 24.12.87 on 28 January 2025) and Comité Ético Científico Facultad de Medicina Universidad de Concepción (protocol code CEC 14/2025 on 6 March 2025).
Informed Consent Statement
The collection of informed consent in this study was considered unnecessary by the institutional ethics committee, as the clinical information was derived from an anonymized database that was provided to the study authors.
Data Availability Statement
The generated data are not publicly available due to ethics committee restrictions and current legal regulations.
Acknowledgments
We would like to express our sincere gratitude to flow cytometry laboratory staff for their valuable support in sample processing and cytometry analysis. We also extend our appreciation to the professional team of the Hematology Department at Guillermo Grant Benavente Hospital, whose dedication and collaboration were essential for the development of this work.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
| FC | Flow cytometry |
| BMB | Bone marrow biopsy |
| DLBCL | Diffuse large B-cell lymphoma |
| FL | Follicular lymphoma |
| MZL | Marginal-zone lymphoma |
| MCL | Mantle cell lymphoma |
| IPI | International prognostic index |
| NHL | Non-Hodgkin lymphoma |
References
- Martin-Moro, F.; Lopez-Jimenez, J.; Garcia-Marco, J.A.; Garcia-Vela, J.A. Comparative Review of the Current and Future Strategies to Evaluate Bone Marrow Infiltration at Diffuse Large B-Cell Lymphoma Diagnosis. Diagnostics 2024, 14, 658. [Google Scholar] [CrossRef]
- Guerra-Soto Ade, J.; Rebolloso-Zúñiga, E.; González-Sánchez, A.G.; Rubio-Jurado, B.; Nava, A. Linfoma no Hodgkin. Conceptos Generales. El Resid. 2013, 8, 23–34. [Google Scholar]
- Alaggio, R.; Amador, C.; Anagnostopoulos, I.; Attygalle, A.D.; de Oliveira Araujo, I.B.; Berti, E.; Bhagat, G.; Borges, A.M.; Boyer, D.; Calaminici, M.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia 2022, 36, 1720–1748, Correction in Leukemia 2022, 36, 1720–1748. https://doi.org/10.1038/s41375-023-01962-5. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, M.E.; Martinez, V.; Nathwani, B.N.; Muller-Hermelink, H.K.; Diebold, J.; MacLennan, K.A.; Armitage, J.; Weisenburger, D.D. Non-Hodgkin lymphoma in Chile: A review of 207 consecutive adult cases by a panel of five expert hematopathologists. Leuk. Lymphoma 2012, 53, 1311–1317. [Google Scholar] [CrossRef]
- Pavlovsky, M.; Cubero, D.; Agreda-Vásquez, G.P.; Enrico, A.; Mela-Osorio, M.J.; Sebastián, J.A.S.; Fogliatto, L.; Ovilla, R.; Avendano, O.; Machnicki, G.; et al. Clinical Outcomes of Patients with B-Cell Non-Hodgkin Lymphoma in Real-World Settings: Findings from the Hemato-Oncology Latin America Observational Registry Study. JCO Glob. Oncol. 2022, 8, e2100265. [Google Scholar] [CrossRef] [PubMed]
- Benavente, R.; Peña, C.; Soto, A.; Valladares, X.; Puga, B.; Cabrera, M.E. Overall survival in Chilean patients with lymphoma and human immunodeficiency virus: A retrospective cohort study. Hematol. Transfus. Cell Ther. 2022, 44, 497–503. [Google Scholar] [CrossRef]
- Cabrera, M.E.; Peña, C.; Vega, V.; Rojas, H.; Pizarro, A.; Rojas, C.; Calderon, S.; Oliva, J.; Hales, C.; Rojas, B.; et al. Follicular Lymphoma in Chile in the Adult Public Cancer Program: The Impact of Chemoimmunotherapy. Cancer Rep. 2024, 7, e2126. [Google Scholar] [CrossRef] [PubMed]
- Pavlovsky, M.A.; Fogliatto, L.; Agreda Vásquez, G.P.; Enrico, A.; Osorio, M.J.M.; San-Sebastian, J.A.A.; Ovilla, R.; Avendaño, O.; López, L.; Santos, T.; et al. Results from a Hemato-Oncology Latin America Observational Registry (HOLA) Providing Real World Outcomes for the Treatment of Patients with NHL. Blood 2016, 128, 5327. [Google Scholar] [CrossRef]
- Alyamany, R.; El Fakih, R.; Alnughmush, A.; Albabtain, A.; Kharfan-Dabaja, M.A.; Aljurf, M. A comprehensive review of the role of bone marrow biopsy and PET-CT in the evaluation of bone marrow involvement in adults newly diagnosed with DLBCL. Front. Oncol. 2024, 14, 1301979. [Google Scholar] [CrossRef]
- Cheson, B.D.; Fisher, R.I.; Barrington, S.F.; Cavalli, F.; Schwartz, L.H.; Zucca, E.; Lister, T.A. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: The Lugano classification. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2014, 32, 3059–3068. [Google Scholar] [CrossRef]
- Carulli, G.; Marini, A. Diagnosis and classification of B-cell non-Hodgkin lymphomas. The role of multiparameter flow cytometry. Clin. Ter. 2012, 163, 47–57. [Google Scholar]
- Wolach, O.; Fraser, A.; Luchiansky, M.; Shapiro, C.; Radnay, J.; Shpilberg, O.; Lishner, M.; Lahav, M. Can flow cytometry of bone marrow aspirate predict outcome of patients with diffuse large B cell lymphoma? A retrospective single centre study. Hematol. Oncol. 2015, 33, 42–47. [Google Scholar] [CrossRef]
- Statuto, T.; Valvano, L.; Calice, G.; Villani, O.; Pietrantuono, G.; Mansueto, G.; D’Arena, G.; Vita, G.; Lalinga, V.; Possidente, L.; et al. Cytofluorimetric and immunohistochemical comparison for detecting bone marrow infiltration in non-Hodgkin lymphomas: A study of 354 patients. Leuk. Res. 2020, 88, 106267. [Google Scholar] [CrossRef]
- Sah, S.P.; Matutes, E.; Wotherspoon, A.C.; Morilla, R.; Catovsky, D. A comparison of flow cytometry, bone marrow biopsy, and bone marrow aspirates in the detection of lymphoid infiltration in B cell disorders. J. Clin. Pathol. 2003, 56, 129–132. [Google Scholar] [CrossRef]
- Ramalingam, T.R.; Muthu, A.; Lakshmanan, A.; Narla, S.; Subramanyan, A.; Simon, S.; Govindaraj, J.; Vaidhyanathan, L.; Easow, J.; Raja, T. Role of high acquisition flow cytometry in the detection of marrow involvement in patients with extramedullary B cell non-Hodgkins lymphoma: A comparison with marrow aspirate cytology, trephine biopsy, and PET. Leuk. Lymphoma 2022, 63, 2589–2596. [Google Scholar] [CrossRef]
- Kim, B.; Lee, S.T.; Kim, H.J.; Kim, S.H. Bone Marrow Flow Cytometry in Staging of Patients With B-cell Non-Hodgkin Lymphoma. Ann. Lab. Med. 2015, 35, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Sorigue, M.; Cañamero, E.; Miljkovic, M.D. Systematic review of staging bone marrow involvement in B cell lymphoma by flow cytometry. Blood Rev. 2021, 47, 100778. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, H.; Uoshima, N.; Muramatsu, A.; Isa, R.; Fujino, T.; Matsumura-Kimoto, Y.; Tsukamoto, T.; Mizutani, S.; Shimura, Y.; Kobayashi, T.; et al. Combination of Bone Marrow Biopsy and Flow Cytometric Analysis: The Prognostically Relevant Central Approach for Detecting Bone Marrow Invasion in Diffuse Large B-Cell Lymphoma. Diagnostics 2021, 11, 1724. [Google Scholar] [CrossRef] [PubMed]
- Cheson, B.D.; Pfistner, B.; Juweid, M.E.; Gascoyne, R.D.; Specht, L.; Horning, S.J.; Coiffier, B.; Fisher, R.I.; Hagenbeek, A.; Zucca, E.; et al. Revised response criteria for malignant lymphoma. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2007, 25, 579–586. [Google Scholar] [CrossRef]
- Arima, H.; Maruoka, H.; Nasu, K.; Tabata, S.; Kurata, M.; Matsushita, A.; Imai, Y.; Takahashi, T.; Ishikawa, T. Impact of occult bone marrow involvement on the outcome of rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisone therapy for diffuse large B-cell lymphoma. Leuk. Lymphoma 2013, 54, 2645–2653. [Google Scholar] [CrossRef]
- Martín-Moro, F.; Piris-Villaespesa, M.; Marquet-Palomanes, J.; García-Cosío, M.; Villarrubia, J.; Lario, A.; García, I.; Michael, B.; Roldán, E.; García-Vela, J.A.; et al. Bone marrow infiltration by flow cytometry at diffuse large B-cell lymphoma NOS diagnosis implies worse prognosis without considering bone marrow histology. Cytom. B Clin. Cytom. 2020, 98, 525–528. [Google Scholar] [CrossRef] [PubMed]
- Greenbaum, U.; Levi, I.; Madmoni, O.; Lior, Y.; Al-Athamen, K.; Perry, Z.H.; Hatzkelzon, L.; Shubinsky, G. The prognostic significance of bone marrow involvement in diffuse large B cell lymphoma according to the flow cytometry. Leuk. Lymphoma 2019, 60, 2477–2482. [Google Scholar] [CrossRef] [PubMed]
- Kalina, T.; Flores-Montero, J.; van der Velden, V.H.J.; Martin-Ayuso, M.; Böttcher, S.; Ritgen, M.; Almeida, J.; Lhermitte, L.; Asnafi, V.; Mendonça, A.; et al. EuroFlow standardization of flow cytometer instrument settings and immunophenotyping protocols. Leukemia 2012, 26, 1986–2010. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. A Coefficient of Agreement for Nominal Scales. Educ. Psychol. Meas. 1960, 20, 37–46. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.