Abstract
Epstein–Barr virus (EBV) is a key oncogenic pathogen implicated in the development of lymphomas, particularly among HIV-positive and immunocompromised individuals. While the association between EBV and lymphoma is well established, the mechanisms underlying progression from infection to malignancy—especially in the head and neck region—remain incompletely understood. This review offers a comprehensive analysis of the pathophysiological pathways by which EBV and HIV contribute to lymphomagenesis, with an emphasis on latency patterns, immune evasion, and epigenetic “hit and run” oncogenesis. Notably, it integrates novel findings on the diagnostic implications of EBV latency proteins, explores HIV-mediated B-cell dysregulation, and evaluates the emerging landscape of targeted therapies, including monoclonal antibodies and lytic cycle inducers. By focusing specifically on head and neck lymphomas, this review underscores a clinically underrepresented domain and offers insights that may guide future diagnostics, surveillance, and treatment strategies in vulnerable patient populations. This review also highlights the pressing need for improved animal models and continued research into EBV-specific therapeutic targets.
1. Introduction
Human oncogenic viruses have been recognized for more than a century as contributors to carcinogenesis. Several of these viruses, including Epstein–Barr virus (EBV) and human immunodeficiency virus (HIV), promote malignancy either through direct oncogenic mechanisms or by inducing states of immunosuppression that increase vulnerability to additional tumor-associated infections [1,2,3]. Immunocompromised individuals, particularly those living with HIV, are therefore disproportionately affected by virus-driven lymphoid neoplasms arising in various anatomical sites, including the head and neck region.
EBV is a ubiquitous gammaherpesvirus infecting more than 90% of adults worldwide [4]. Although primary infection is often asymptomatic in early childhood, infection during adolescence or young adulthood commonly causes infectious mononucleosis, characterized by fever, sore throat, fatigue, cervical lymphadenopathy, and splenomegaly. Beyond causing acute illness, EBV plays a well-established role in the development of several lymphoid malignancies, including Burkitt lymphoma, diffuse large B-cell lymphoma (DLBCL), extranodal NK/T-cell lymphoma (ENKTL), and Hodgkin lymphoma [5,6,7,8]. EBV was first identified in association with Burkitt lymphoma in 1964 by Epstein and Barr [5], providing the foundation for decades of research into its oncogenic potential. Given the predominance of EBV-related malignancies in the head and neck region, early recognition of disease manifestations is of particular importance for otorhinolaryngologists.
Clinicians in otorhinolaryngology frequently encounter symptoms that may reflect the initial presentation of lymphoid malignancies, including recurrent epistaxis, persistent nasal obstruction, cervical lymphadenopathy, or, in severe cases, acute airway compromise [6]. Although EBV- and HIV-associated lymphomas can arise in numerous extranodal sites, this review focuses specifically on presentations within the head and neck—the anatomical region most relevant to ENT practice. Understanding these patterns is essential for timely diagnosis and multidisciplinary management.
EBV-associated lymphomas encompass a heterogeneous group of diseases, including T-cell and B-cell non-Hodgkin lymphomas and the various subtypes of classical Hodgkin lymphoma [7,8]. Epidemiologic patterns vary across populations, with higher EBV-positive case proportions reported in low- and middle-income countries, and survival outcomes influenced by disease subtype, treatment access, and geographic region [8,9,10].
HIV infection significantly increases the risk of EBV-mediated lymphomagenesis through mechanisms that include chronic immune activation, disruption of germinal center dynamics, and impaired cytotoxic T-cell surveillance [11,12,13]. Although antiretroviral therapy (ART) has reduced the incidence of some AIDS-defining malignancies, HIV-positive individuals continue to experience elevated rates of aggressive lymphomas. Globally, approximately 37.9 million people are living with HIV, emphasizing the public health relevance of understanding these interactions [11,12].
Recent years have seen notable advances in characterizing the molecular pathways involved in EBV- and HIV-associated lymphomas. These include identification of novel inhibitory immune receptors (CD47, LAG-3, TIM-3, VISTA, DDR1) involved in immune escape, as well as the emerging use of immune checkpoint inhibitors such as nivolumab and pembrolizumab, particularly in relapsed or refractory disease [14,15]. These advancements underscore the need for updated clinical frameworks that integrate evolving molecular knowledge with practical ENT-focused considerations for diagnosis and management.
This review, therefore, synthesizes current evidence on the roles of EBV and HIV in lymphomagenesis within the head and neck region, incorporating virologic, immunologic, diagnostic, and therapeutic perspectives. By emphasizing aspects most pertinent to otorhinolaryngologists, we aim to strengthen early clinical suspicion, support timely evaluation, and promote interdisciplinary care in this vulnerable patient population.
2. Epstein–Barr
Epstein–Barr virus (EBV), formally known as human herpesvirus 4, infects more than 90% of adults worldwide [16]. It is typically transmitted through saliva but can also spread via semen during sexual contact, organ transplantation, and blood transfusion [17,18]. When infection occurs early in life, it may remain asymptomatic; however, primary infection later in life can present symptoms of infectious mononucleosis [17].
Infectious mononucleosis (IM) warrants consideration for surveillance due to its increased potential for carcinogenesis. A retrospective study assessed the risk of developing neoplasms in 25,582 patients with a history of mononucleosis infection, compared with a control group of the same size. During the follow-up period, newly diagnosed cases of lymphoma were significantly higher in the IM group than in the control group (p < 0.05), along with other types of cancers. Patients with IM exhibited a fivefold higher risk of developing lymphoma compared to healthy controls [19].
Hjalgrim et al. reported that lymphoma often develops within an average of four years after the onset of IM [20]. This timeframe can create the impression of rapid malignancy progression, influenced by several factors. The overlapping symptoms of infectious mononucleosis (IM) and lymphoma—such as fever, cervical lymphadenopathy, and tonsillar enlargement—can result in diagnostic confusion or short-term misdiagnoses. Persistent or worsening symptoms in IM patients often lead to further evaluation, including lymph node biopsies, which may uncover lymphoma [19].
These observations highlight the importance of closely monitoring IM-infected patients, particularly those with unresolved or progressing symptoms. Early detection of potential malignancy is essential for timely diagnosis and treatment, ultimately improving patient outcomes.
2.1. Pathophysiology
EBV exhibits a strong tropism for oropharyngeal epithelial cells and B lymphocytes [17]. A remarkable feature of EBV is its ability to immortalize normal resting B lymphocytes in vitro, showcasing its potent transforming potential [21,22]. EBV exists in two types: Type 1 and Type 2. Type 1 is more prevalent worldwide and has a greater capacity for malignant transformation [23]. Entry into human B lymphocytes—the primary reservoir for EBV—is facilitated by the viral glycoprotein gp350/220 binding to the complement receptor 2 (CR2/CD21) and the interaction of the glycoprotein complex gH/gL/gp42 with HLA class II molecules [17,24,25].
After primary infection, EBV adopts one of two infection patterns: latent or lytic. The lytic infection pattern propagates the virus and promotes its spread [26,27], while latent infection allows EBV to persist in the host with minimal impact, effectively evading the immune system. During latency, EBV resides in the circulating B-cell pool of peripheral blood, minimizing viral production. The primary human host cells for EBV include lymphocytes, T lymphocytes, NK cells, epithelial cells, and myocytes [17].
Genetic predispositions, such as variations in specific HLA alleles, may significantly affect the likelihood of developing EBV-associated Hodgkin’s lymphoma. Certain HLA class I alleles are linked to either an elevated or reduced risk of this condition, highlighting the pivotal role of the host immune response in its development. One study identified an increased risk associated with the HLA-A01 allele (odds ratio [OR] per allele: 2.15; 95% CI: 1.60–2.88), while the HLA-A02 allele was linked to a reduced risk (OR per allele: 0.70; 95% CI: 0.51–0.97). Notably, the observed connection between IM and Hodgkin’s lymphoma risk was present in individuals carrying the HLA-A01 allele but not in those with the HLA-A*02 allele [28].
It is not yet clear how IM progresses to lymphoma, but one hypothesis suggests that alterations in immune function following IM weaken immune surveillance against malignant cells. This immune tolerance may diminish the body’s anti-tumor response [29]. Nevertheless, research has shown that even patients with normal or reduced immunity may still progress to lymphoma [30,31].
EBV antigens play crucial roles in promoting cellular growth and contributing to various mechanisms that favor malignancy. These include resistance to apoptosis, reduced responsiveness to differentiation signals, and enhanced cellular invasion [32,33]. The virus encodes six nuclear antigens—EBNA-1, EBNA-2, EBNA-3A, EBNA-3B, EBNA-3C, and EBNA-LP—as well as three latent membrane proteins: LMP-1, LMP-2A, and LMP-2B [26,30]. Each of these proteins performs distinct functions that are critical to EBV’s ability to drive lymphoma pathogenesis (Table 1).

Table 1.
Function of each EBV antigen.
EBV DNA levels in whole blood are considered a superior prognostic and monitoring factor in DLBCL compared to EBV-encoded small RNA (EBER). The presence of EBV DNA is associated with poorer overall survival, a trend also observed in NK/T-cell lymphoma, follicular lymphoma, and Hodgkin lymphoma [36,37]. However, combining both markers (EBV DNA and EBER) provides a more accurate assessment of prognosis than using either marker alone [36,38].
In Hodgkin lymphoma, plasma EBV DNA has been shown to predict treatment outcomes [39]. The expression of cMYC and BCL2, particularly when both are overexpressed (double expression), correlates with worse overall survival (OS) in patients with DLBCL. The study by Zeng et al. demonstrated that a cMYC expression cutoff of 47.5% effectively predicts high-grade DLBCL [40].
The period following infection, but preceding cell division, is referred to as the pre-latent phase [41] (Figure 1 and Table 2). During this phase, as lytic mRNA levels decrease, EBNA2 and EBNA-LP accumulate, directly activating the major upstream EBV latency C promoter (Cp) between 48 and 72 h post-infection [42]. Approximately 84 h post-infection, non-coding RNAs and mRNAs associated with latency IIb are expressed. The initial four cell divisions after infection occur rapidly, with each cycle lasting 8–12 h [43]. Cells that subsequently reduce their proliferation rate become immortalized [41].
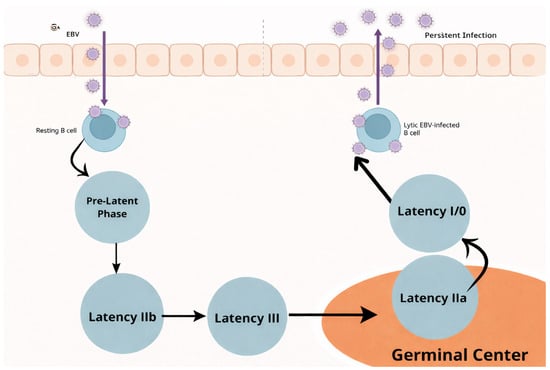
Figure 1.
Diagram illustrating the progression of EBV latency states in B cells. Created with Canva.

Table 2.
Antigens expressed in each latency pattern.
Full latency III is marked by the expression of all EBNAs and LMPs and is typically observed in immunosuppressed conditions. Survival during latency III relies heavily on elevated NFκB activity, predominantly mediated by LMP1 [44,45]. However, during the initial two weeks following infection, LMP1-induced NFκB activity is low, raising the unresolved question of how these infected cells persist during this phase [41].
Latency IIa is observed in germinal center cells of healthy individuals and in EBV-positive Hodgkin lymphoma [46]. The mechanisms driving the transition to this state remain unclear, though cytokines such as IL-21 are believed to downregulate Cp and EBNA expression [47]. Cells expressing Latency IIa are also considered precursors of EBV-associated Hodgkin lymphomas [41].
The primary reservoir of EBV-infected B cells consists of resting memory cells [48]. In Latency 0, no viral proteins are expressed except during cell division (Latency I). This limited expression allows these cells to evade detection by T cells [41].
Molecular markers also play a crucial role in prognosis. For instance, p53 overexpression, a high Ki67 index, advanced disease stage, T-cell phenotype, and the presence of the del-LMP1 mutation are associated with poor outcomes [29]. These findings highlight the importance of integrating molecular and clinical markers into prognostic models to refine risk stratification and guide treatment decisions.
2.2. Hit and Run Theory
Epigenetic mechanisms, such as DNA methylation, histone modification, and chromatin conformation alterations, play a significant role in tumor development. Altered DNA methylation states are frequently observed in malignancies linked to EBV. Notably, tumor progression in these cases can occur even in the absence of the virus, indicating that EBV’s influence on tumorigenesis does not rely on its continuous presence. This phenomenon is described as the “hit and run” mechanism [49,50].
The “hit and run” process begins when EBV infects a B lymphocyte, delivering the necessary instructions for the cell’s growth and survival, effectively making the cell oncogenic. This initial stage is termed the “hit.” Under normal circumstances, the immune system, when functioning effectively, identifies these infected B lymphocytes through the viral antigens displayed on their surfaces and eliminates them [50].
However, EBV has evolved strategies to evade immune detection by inducing epigenetic changes in the host lymphocytes. These changes silence the expression of viral antigens on the cell surface, marking the “run” phase of the mechanism. By doing so, EBV prevents immune recognition and eradication of the infected cells. These epigenetic modifications become heritable, enabling the oncogenic characteristics of the cells to persist even after the virus is no longer present in the host [50].
This “hit and run” mechanism highlights the intricate role of epigenetics in EBV-associated malignancies and provides a framework for understanding how viral infections can have lasting oncogenic effects.
3. HIV
HIV-infected individuals are at significantly higher risk of developing malignancies compared to the general population, with lymphoma being the second most frequent HIV-associated neoplasm [48]. This increased risk underscores the profound impact of HIV on immune function and its interaction with oncogenic viruses such as EBV [48,51]. Notably, plasmablastic lymphoma, a rare and aggressive subtype, nearly always occurs in HIV-positive patients [48]
Most lymphomas in HIV-infected patients are B-cell-derived, with immunoblastic large cell lymphomas and diffuse large B-cell lymphomas (DLBCLs) being the most common non-Hodgkin lymphomas (NHLs). These lymphomas typically arise in the setting of moderate to severe immunodeficiency, particularly when CD4+ T-cell counts fall below 100 cells/mm3, and they are often associated with EBV [48,51]. The risk of developing NHL in HIV-infected individuals is starkly elevated, with a 1000-fold increase for Burkitt lymphoma [52,53] and a 400-fold increase for other aggressive lymphoma [54] compared to the general population [52,53].
Persistent immune activation and inflammation play a crucial role in the development of HIV-associated lymphomas. HIV infection induces chronic immune activation, partly driven by microbial translocation. This persistent activation leads to sustained B-cell stimulation, which promotes DNA alterations such as oncogene mutations and chromosomal translocations [55]. Chronic stimulation also disrupts normal B-cell receptor (BCR) signaling, resulting in uncontrolled B-cell proliferation and facilitating lymphomagenesis [56].
The underlying mechanisms involve HIV-induced alterations in BCR gene structure and rearrangement through the increased activity of activation-induced cytidine deaminase (AID) and recombination-activating genes (RAGs). This process upregulates the BCR signaling pathway, enhancing PI3K/AKT and RAS/MAPK signaling. The consequent extensive proliferation and accumulation of mutations render B cells more susceptible to malignant transformation [56].
Phosphatase and tensin homolog (PTEN) acts as a negative regulator of the PI3K pathway. PI3K signaling supports the survival of mature B cells even in the absence of BCR expression. The HIV-1 matrix protein p17 has been implicated in this pathway, contributing to lymphomagenesis by maintaining PTEN in its inactive form [57]. Notably, certain p17 variants, such as S75X from the Ugandan HIV-1 strain, can directly activate the PI3K/AKT signaling pathway by binding to p17 receptors on B cells [58,59].
RAG expression is thought to be influenced by the levels of the HIV trans-activator of transcription (Tat) protein—a key regulator of HIV infection and viral reactivation. Beyond its transcriptional role, Tat can also induce DNA damage, further contributing to genomic instability [60].
AID is essential for immunoglobulin class-switch recombination. Nevertheless, its overexpression can result in abnormal somatic mutations that ultimately lead to leukemogenesis and lymphomagenesis. AID introduces mutations within the variable (V) gene segments of immunoglobulin heavy chain genes, thereby altering the specificity and affinity of the BCR. AID activity predominantly occurs in the germinal center, and most HIV-associated lymphomas are believed to originate from this region [56,61].
The upregulation of AID in HIV-infected individuals is driven by multiple mechanisms. CD40 ligand (CD40L) enhances AID expression, and HIV infection induces CD40 activation [56]. Additionally, B-cell activating factor (BAFF) upregulates AID expression, and elevated BAFF levels persist even in patients receiving effective antiretroviral therapy (ART) [62]. Direct interactions between viral proteins and B-cell membranes can also stimulate AID overexpression [56]. Furthermore, a decrease in the expression of CD300—an immunoregulatory receptor—has been observed in HIV infection and appears irreversible despite ART. Reduced CD300 expression may promote BCR hyperactivation and proliferation, thereby amplifying downstream signaling pathways associated with AID overexpression and B-cell dysregulation [63].
The tumor microenvironment in HIV-associated lymphomas, marked by cytokine imbalances and the presence of reactive inflammatory cells, significantly supports the growth and progression of these malignancies [64].
In the head and neck region, sinonasal lymphoid neoplasms represent less than 1% of all cancers [65,66], but extranodal lymphomas in general account for 25% of lymphomas in this area. Infectious agents, including EBV, HIV-1, and other pathogens such as Helicobacter pylori and Human T-cell lymphotropic virus-1 (HTLV-1), are implicated in these malignancies [54]. A retrospective study examining 243 cases of head and neck DLBCL revealed that Waldeyer’s ring was the most common site of origin, accounting for 63.7% of cases. The tonsils were most frequently involved (28.8%), followed by the oral cavity (16.04%), salivary glands (8.64%), and the nasal cavity (3.7%). Less frequently affected sites included the larynx/hypopharynx (2.05%) and the mandible (1.23%) [67].
HIV positivity significantly affected survival and increased the risk of death in patients with head and neck lymphomas [66], though no direct correlation was observed between HIV status and the anatomical distribution of tumors. Among these cases, nasopharyngeal and gingival lymphomas had the highest proportion of HIV-positive patients, with 20.8% and 40% positivity rates, respectively [67].
Antiretroviral therapy (ART), in addition to restoring immune function, which is crucial for preventing oncogenesis, has shown efficacy in reducing EBV viral loads. This provides the additional benefit of lowering the risk of EBV oral transmission [35,68]. However, despite ART, HIV-infected patients remain more susceptible to EBV-associated malignancies than the general population [69].
Patients with HIV-associated Hodgkin lymphoma (HL) typically present with more adverse prognostic indicators and risk factors compared to their immunocompetent counterparts [69,70]. Nevertheless, the introduction of combination antiretroviral therapy (cART) alongside standard chemotherapy regimens such as adriamycin, bleomycin, vinblastine, and dacarbazine (ABVD) has resulted in comparable outcomes between HIV-positive and HIV-negative patients. In one study, the 2-year progression-free survival (PFS) was 86% in HIV-negative patients and 89% in HIV-positive patients. Similarly, another study reported 5-year overall survival rates of 83% and 85%, respectively, and an identical 5-year event-free survival rate of 64% [70].
Survival outcomes for other HIV-associated lymphomas have also improved with the advent of highly active antiretroviral therapy (HAART). Several studies have demonstrated 2-year survival rates exceeding 60% for HIV-associated Burkitt lymphoma (BL) and diffuse large B-cell lymphoma (DLBCL) [71,72]. However, treatment-related toxicity remains an important consideration.
Integrase inhibitors share structural and functional similarities with RAG1, potentially downregulating RAG expression and thereby reducing lymphoma risk [73].
A longitudinal study found that HIV-infected patients with a low CD4/CD8 ratio had the highest risk of non-Hodgkin lymphoma (hazard ratio 1.53), with the association detectable up to 24 months before diagnosis [74]. Similarly, a CD4/CD8 ratio below 0.5 was linked to infection-related malignancies (aHR 2.03; 95% CI, 1.24–3.33), while CD4 counts below 350 cells/μL correlated with an increased risk of all studied cancers. Regular monitoring of CD4 counts and CD4/CD8 ratios may thus aid early cancer detection, particularly in patients initiating ART late or failing to achieve immune reconstitution above 350 cells/μL [75].
HIV-associated lymphomas are a prominent and aggressive complication of HIV infection, and they are often linked to EBV co-infection. The interaction between these pathogens amplifies the oncogenic potential, underscoring the critical need for effective HIV management and lymphoma surveillance in this vulnerable population.
4. Prevention
Given the significant role of EBV in the pathogenesis of various neoplasms, the concept of developing a vaccine against EBV has gained traction. One promising target for such a vaccine is the glycoprotein gp350/220. This protein is abundantly expressed on the plasma membranes of lytically infected cells and constitutes the predominant protein on the virus’s outer coat. It plays a critical role in EBV’s infection mechanism by binding to the CD21 receptor on B cells, initiating the infection process. Furthermore, the majority of human EBV-neutralizing antibodies are directed against gp350/220, making it an optimal candidate for a subunit vaccine. Research efforts are currently focused on this glycoprotein as the basis for potential immunization strategies [76,77].
Additionally, implementing the EBV vaccine in immunocompromised individuals faces several challenges. One of the key issues is the selection of appropriate antigens and vaccine platforms that can stimulate both humoral and cellular immunity effectively in immunosuppressed populations. The use of virus-like particles (VLPs) has been explored as they can mimic the structure of the virus without containing its genome, potentially offering a safer option for these patients [78]. However, the ability of VLPs to induce a robust immune response in immunocompromised individuals remains to be fully validated. Furthermore, the safety profile of any EBV vaccine must be carefully evaluated in immunocompromised individuals to avoid potential adverse effects, such as exacerbation of their condition or induction of autoimmunity. This necessitates rigorous clinical trials specifically designed for this high-risk group, which can be logistically and ethically complex.
In parallel, the principles of active surveillance programs employed for other oncogenic viruses, such as those targeting oncoviruses, could be extended to EBV-associated conditions. Such measures might include routine HIV screening, as well as ensuring efficient and widespread access to antiretroviral therapies. These efforts not only reduce HIV-associated immunosuppression but may also decrease EBV loads and mitigate its oncogenic potential.
5. Diagnostic Approach
Lymphoma presents with a variety of clinical manifestations that can differ based on the subtype and stage of the disease. For Hodgkin lymphoma, the most common presentation is painless lymphadenopathy, often in the cervical region. Systemic symptoms, known as B-symptoms, such as fever, night sweats, and unexplained weight loss, are also common and tend to occur in more advanced stages [79]. The Lugano classification system is used for staging, which incorporates these symptoms and imaging findings to guide treatment decisions [80].
Non-Hodgkin lymphoma (NHL), which encompasses a variety of subtypes, can manifest in different ways. Typical presentations include enlargement of peripheral lymph nodes and involvement of extranodal sites, such as the gastrointestinal tract or skin. B-symptoms are also seen in NHL, especially in more aggressive forms [79]. In pediatric and adolescent cases, B-cell lymphomas frequently present with swollen lymph nodes in the neck or periphery, abdominal masses, and systemic symptoms like fever and weight loss [81].
Certain subtypes of NHL can exhibit distinct clinical presentations. For example, lymphomas associated with AIDS often involve systemic effects and may include primary central nervous system lymphoma or primary effusion lymphoma [82]. Follicular lymphoma, a frequently encountered indolent subtype of NHL, usually presents with enlarged lymph nodes and may be associated with constitutional symptoms or blood cell deficiencies [83]. Oral lymphomas, though rarer, may present as painful, ulcerative lesions that are frequently linked to bone destruction, especially in high-grade subtypes [84].
The main task of otorhinolaryngologists is diagnosis and follow-up in order to determine recurrences. Neck nodes require thorough examination as they can serve as the initial indication of cancer in the head and neck region. In HIV-positive individuals, the neck is the most common site for lymphoma development, accounting for 50% of cases. These lymphomas often develop in younger patients and may be the first sign that prompts suspicion of HIV infection [85]. Therefore, the diagnostic workup for head and neck lymphomas should routinely include HIV testing. On average, lymphoma develops approximately 1.5 years after an HIV diagnosis [86].
Enlarged neck nodes also warrant endoscopic examination to rule out cavum lymphoma, particularly in HIV-infected patients. Cavum lymphomas often present initially as isolated neck node enlargement [87] and include subtypes such as Burkitt lymphoma, immunoblastic lymphoma, and DLBCL [48].
Pathology results are critical, and an excisional lymph node biopsy is the preferred method for obtaining a definitive diagnosis of lymphoma. This approach provides adequate tissue sampling for histopathological and immunophenotypic analyses. When an excisional biopsy is not feasible, a core-needle biopsy may be considered [79,80], although it may not provide as comprehensive a sample. Ultrasound-guided core needle biopsy (US-CNB) is a minimally invasive alternative to surgical excision biopsy, offering high diagnostic accuracy, with sensitivity and specificity often exceeding 90% for both Hodgkin and non-Hodgkin lymphomas [88,89]. However, US-CNB may have limitations in certain scenarios, such as the subclassification of low-grade lymphomas and Hodgkin lymphoma, where histological architecture is crucial for an accurate diagnosis [90]. In such cases, if the US-CNB results are inconclusive or if there is strong clinical suspicion of malignancy despite a benign biopsy result, a follow-up excisional biopsy may be necessary to confirm the diagnosis [89].
Extranodal lesions require deep biopsies close to healthy tissue to avoid necrotic tissue. Open biopsy of neck masses, paranasal sinus lesions, and excision of affected glands or tonsils may also be used, depending on the tumor’s location. Immunohistochemical staining is necessary for accurate diagnosis [48].
Half of extranodal lymphomas in the head and neck occur in the Waldeyer ring. Common symptoms include dysphagia and sore throat [48,91]. The decision to excise asymmetrical tonsils should be guided by clinical context and risk factors. Rapid unilateral tonsil enlargement, rather than asymmetry alone, is significantly associated with malignancy [92].
Oral lymphomas may present as swelling, pain, or ulcers in the gingiva, palate, or tongue [48,93]. In HIV-positive patients, plasmablastic lymphoma is the most common subtype in this location [94,95], followed by B-cell lymphomas with high proportions of plasmablastic cells [48]. Parotid gland lymphomas present as unilateral enlargement with or without facial paresis, with subtypes including marginal zone B-cell lymphoma of the mucosa-associated lymphoid tissue (MALT), follicular lymphoma, and DLBCL [48]. Laryngeal lymphoma, typically located in the supraglottic region, often manifests as dysphagia, hoarseness, and dyspnea [48].
The presence of elevated EBV antibody titers often precedes the onset of tumors such as Burkitt and Hodgkin’s lymphoma by several years [96]. IgG antibodies to EBNA begin to appear 6 to 12 weeks after the onset of symptoms and persist throughout life. In situ hybridization for EBV-encoded small RNA (EBER) is considered the most sensitive method for detecting EBV-infected cells [26,97], with EBV-specific antibodies being the diagnostic gold standard for infectious mononucleosis [98]. However, some EBER-negative samples may still contain EBV, resulting in a sensitivity of approximately 67.8% for this marker [49,99,100]. Novel molecular biology techniques, such as microRNA-based markers, are under investigation but require further validation.
HL tends to arise in the jugular chain, whereas NHL is more commonly found in retropharyngeal, parotid, occipital, or submandibular nodes. Extranodal lymphomas in the Waldeyer ring often involve gastrointestinal sites, making barium studies essential for staging [101].
Key prognostic factors include neutropenia, lymphopenia, anemia, elevated serum alkaline phosphatase, and calcium levels, which may indicate bone infiltration. Bone marrow involvement is more frequent in immunodeficiency and advanced stages, warranting biopsies in patients with B symptoms, cytopenias, or bulky disease. PET imaging is the gold standard for staging aggressive lymphomas and HL [48]. Additionally, CD4 count, particularly in HIV patients or specific lymphoma subtypes, provides important prognostic information.
The Lugano classification system is widely used to stage both Hodgkin and non-Hodgkin lymphomas. It combines clinical assessments, imaging results, and the presence of B-symptoms to establish the disease stage, which is essential for determining the appropriate treatment. The modified Ann Arbor staging system is also utilized, especially for Hodgkin lymphoma, where the suffixes A and B indicate the absence or presence of B-symptoms, respectively [79,80].
6. General Treatment Approaches for Lymphomas
Treatment for lymphomas broadly depends on the type of lymphoma, its stage, and the patient’s overall health [48,102]. Below is an overview of treatments across various lymphoma subtypes.
For diffuse large B-cell lymphoma (DLBCL), combination chemotherapy with CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) is the standard approach, with the addition of rituximab for CD20-positive cases and in whom the CD4 cell count is >50 cells/μL, has been suggested [103,104]. Dose-adjusted EPOCH (etoposide, vincristine, doxorubicin, prednisone, and cyclophosphamide) plus rituximab is often preferred for cases with high proliferation rates (Ki 67 > 80%) or plasmablastic histology [105,106], considering prophylactic for opportunistic infections and in patients with affectation of paranasal sinuses, epidural space, or bone marrow are involved, or if disease involvement includes more than two extranodal sites lumbar puncture should be done as part of workup [48].
In Burkitt lymphoma, highly aggressive regimens like CODOX-M/IVAC (cyclophosphamide, vincristine, doxorubicin, methotrexate alternating with ifosfamide, etoposide, and cytarabine) [107,108,109], Hyper CVAD (cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with high-dose methotrexate and cytarabine) [87,110,111], or CDE (cyclophosphamide, doxorubicin, etoposide) plus rituximab [112,113,114] for most patients with AID related BL are used. Intrathecal chemotherapy is crucial due to the high risk of CNS involvement [48]. Plasmablastic lymphoma is treated similarly to Burkitt lymphoma [48].
Hodgkin lymphoma is typically managed with ABVD (adriamycin, bleomycin, vinblastine, dacarbazine) as the standard chemotherapy regimen, or BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone) followed by field radiation therapy [115].
For NK-/T-cell lymphomas, combined chemotherapy and radiotherapy are preferred for localized disease due to the high systemic relapse rate. Regimens like SMILE (dexamethasone, methotrexate, ifosfamide, L-asparaginase, etoposide) [116,117,118,119], GELOX (gemcitabine, L-asparaginase, oxaliplatin), or VIPD (etoposide, ifosfamide, cisplatin, dexamethasone) are commonly employed. Radiotherapy doses of at least 50 Gy are necessary to minimize in-field failure rates [120].
EBV-driven lymphomas often exhibit increased drug resistance, which can worsen prognosis and complicate management, particularly in immunocompromised hosts such as those with HIV infection. For example, extranodal NK/T-cell lymphoma (ENKTCL) is resistant to anthracycline-based regimens due to multidrug resistance phenotypes, necessitating the use of asparaginase-based protocols and radiotherapy for localized disease, and more intensive regimens for advanced stages [121].
Relapsed lymphoma is associated with a poor prognosis, with a median survival of less than one year in most cases. For patients with relapsed non-Hodgkin lymphoma (NHL) of B-cell origin, a second-line treatment option is the ESHAP regimen, which includes etoposide, methylprednisolone, cytarabine, and cisplatin. In certain cases, especially with advancements in HIV control and immune reconstitution, autologous stem cell transplantation may be considered a viable therapeutic option. This approach offers a chance for extended remission or potential cure in select patients, depending on their overall health, response to salvage therapy, and lymphoma characteristics [48].
Rituximab has improved the prognosis of lymphoma; however, its application in HIV patients is limited due to immunosuppression, drug interactions, and co-infections [122].
The main limitations in treating lymphomas of the ear, nose, head, and neck in patients with EBV and HIV co-infection are increased toxicity, drug resistance, and the need for individualized supportive care. While standard regimens are generally used, careful consideration of immunosuppression, potential drug interactions, and the unique biology of EBV-driven lymphomas is essential [122,123,124].
Targeted Therapies for EBV-Associated Lymphomas
The association of EBV with lymphomas has spurred research into therapies that target EBV-specific mechanisms to improve outcomes for affected patients. Despite progress, challenges such as the lack of suitable animal models limit the development of effective treatments, as EBV exclusively infects humans [125]. Nonetheless, promising strategies are emerging, focusing on pathways and molecules crucial to EBV-driven oncogenesis.
One venue of targeted therapy involves exploiting the interplay between EBV and apoptosis-related proteins. EBNA3C, an EBV nuclear antigen, disrupts Bcl-6 activity, leading to the derepression of Bcl-2 expression, a key event in lymphomagenesis [126]. While ABT-199, a Bcl-2-selective inhibitor, has shown efficacy in some tumors [127], it has proven ineffective in EBV-immortalized cells [128], and its efficacy in early EBV infection must be confirmed.
Conversely, the EBV-encoded Bcl-2 homolog, BHRF1, blocks apoptosis by binding the Bcl-2-related protein Bim [129]. An inhibitor specifically designed to target BHRF1 has demonstrated significant efficacy, suppressing tumor growth and prolonging survival in a mouse model of EBV-positive B-cell lymphoma [130].
Zidovudine (AZT), a nucleoside analog, has been investigated for its potential to trigger EBV lytic gene expression, resulting in the apoptosis of EBV-infected cells. Regimens that include AZT, often combined with other agents like hydroxyurea that induce lytic activity, have demonstrated effectiveness in treating EBV-positive lymphomas, such as primary central nervous system lymphoma (PCNSL) in patients with AIDS [131,132,133]. However, due to its unfavorable toxicity profile, zidovudine is no longer recommended as a first-line therapy and is currently reserved as a second-line agent [134].
Proteasome inhibition has also emerged as a therapeutic strategy. The FDA-approved proteasome inhibitor ixazomib has shown promise in EBV-positive lymphoma cells by inducing apoptosis and cell cycle arrest [135]. Similarly, targeting spleen tyrosine kinase (SYK), a critical component of the B-cell receptor (BCR) signaling pathway, has proven effective. TAK-659, a novel SYK inhibitor, induces cell death and inhibits tumor development in EBV-associated lymphomas in vivo [136,137], showcasing the potential of signaling pathway inhibitors in treating these malignancies.
Various stimuli, including histone deacetylase (HDAC) inhibitors [138], 12-O-tetradecanoylphorbol-13-acetate (TPA) [139], sodium butyrate [140,141], and anti-immunoglobulin [142,143], can switch EBV-infected cells from latent to lytic infection. This approach allows for the selective targeting of infected cells. A clinical trial demonstrated the efficacy of combining arginine butyrate with ganciclovir [144]. Arginine butyrate induced the lytic cycle, activating the EBV thymidine kinase (EBV-TK), which rendered infected cells susceptible to ganciclovir, a nucleoside antiviral agent that inhibits viral replication [144,145]. However, another study revealed that the EBV protein kinase BGLF4, rather than EBV-TK, mediated the ganciclovir response [146].
Monoclonal antibodies and targeted agents directed against key molecular pathways in Epstein–Barr virus (EBV)-associated lymphomas are under active investigation. Among the most promising strategies are histone deacetylase inhibitors (HDACis), which induce EBV lytic reactivation and sensitize tumor cells to antiviral therapy while exerting direct cytotoxic effects [147,148].
Selective class I HDACis—such as chidamide, romidepsin, vorinostat, and depsipeptide—have shown preclinical efficacy. Chidamide, a selective class I HDAC inhibitor, was shown to induce EBV lytic gene expression in EBV-positive Burkitt lymphoma (BL) cell lines (Raji, Namalwa) while suppressing latent transcripts. This reactivation sensitized tumor cells to tenofovir, a nucleotide analog that inhibits EBV lytic DNA replication. The combination of chidamide and tenofovir produced synergistic antitumor activity both in vitro and in vivo (Raji-GFP-Luc xenograft model in NSG mice), promoting apoptosis and modulating MAPK signaling pathways [149]. Similarly, romidepsin induces EBV lytic reactivation across diverse models, including lymphoblastoid and EBV-positive BL cell lines, through histone acetylation at the BZLF1 promoter in a reactive oxygen species-dependent manner [150].
Together, these findings support a dual-target approach combining HDAC inhibitors with antiviral agents to eradicate EBV-infected tumor cells by inducing viral reactivation while blocking replication.
TCR-like monoclonal antibodies (mAbs) mimic T-cell receptors by recognizing EBV peptides presented on HLA molecules, thereby selectively targeting EBV-transformed B lymphoblastoid cells. Antibodies such as E1, L1, and L2 bind to epitopes derived from EBNA1, LMP1, and LMP2A presented by HLA-A*0201, inducing apoptosis and reducing tumor burden in preclinical models [151,152].
Lai et al. evaluated the E1 antibody, which targets an HLA-A*0201-restricted EBNA1 epitope, in models of EBV-associated lymphoproliferative disease (LPD). In severe combined immunodeficiency (SCID) mice engrafted with EBV-transformed lymphoblastoid cell lines, E1 treatment significantly prolonged survival and reduced tumor burden compared to controls. E1 showed high epitope specificity, binding exclusively to EBNA1 peptide–HLA complexes on EBV-positive cells, while sparing EBV-negative, HLA-matched cells. Mechanistically, its antitumor effects were mediated through both antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC) [151].
Monoclonal antibodies targeting the EBV glycoprotein 42 (gp42), such as mAbs A10 and 4C12, have been developed to inhibit the interaction between gp42 and HLA class II molecules, thereby preventing viral fusion with B cells. Among these, mAb A10 has demonstrated strong neutralizing activity, effectively protecting against EBV infection and lymphoma development in humanized mouse models [153].
In a related study, antibodies 1A7 and 6G7 were isolated against the C-type lectin domain of gp42. Mice treated with 6G7 exhibited markedly lower viral DNA levels, maintained body weight, and were protected from EBV infection and lymphoma formation compared to controls. Two distinct gp42 epitopes targeted by 1A7 and 6G7 were identified. Interestingly, despite their affinity for gp42, these antibodies did not interfere with gp42–gH/gL interactions essential for viral entry, but significantly reduced gp42 binding to HLA class II molecules, thereby impairing B-cell fusion and infection [154].
Monoclonal antibodies targeting the gH/gL glycoprotein complex of EBV have been identified as effective in neutralizing the virus by blocking the fusion of the viral membrane with host cells. Research has revealed several antigenic sites on the gH/gL complex that are susceptible to neutralization, paving the way for the development of therapeutic antibodies to combat EBV-related diseases [155,156].
A bispecific antibody targeting the viral envelope protein gp350 and CD89 on immune cells has been developed to promote phagocytosis and clearance of EBV-infected cells. Preclinical studies have shown its effectiveness in lowering viral load and preventing tumor development in EBV-related hematological disorders [157].
The ongoing research into targeted therapies offers hope for more effective and tailored treatments for EBV-associated lymphomas. Continued efforts to refine these approaches and address existing gaps will be pivotal in advancing patient care.
The fundamental problem in developing any therapeutic option against EBV is the lack of well-defined animal models for studies, as EBV infects only humans.
Current research using different animal models is being developed. Rabbit models have been employed to study the early stages of primary EBV infection and the corresponding immune response. They have also been used to evaluate the efficacy of virus-like particle (VLP)-based vaccines against EBV [158,159,160].
Tree shrews, which share a close phylogenetic relationship with primates, represent another valuable model. Their complement receptor 2 (CR2) exhibits high sequence similarity to the human counterpart, facilitating EBV entry. EBV infection in tree shrews closely mimics the clinical and immunological features of human infection [161,162].
In addition, murine models utilizing murid gammaherpesvirus 68 (MHV-68), an EBV-related virus, have been developed to investigate virus–host interactions, particularly within germinal center B cells [163,164,165]. These models provide important insights into viral latency, reactivation, and lymphomagenesis.
Anti-PD-1 antibodies, nivolumab and pembrolizumab, have also recently been approved by the FDA for treating relapsed or refractory HLs due to promising results across multiple clinical trials demonstrating an overall response rate of more than 65% [166,167,168].
7. Prognosis
The prognosis of lymphomas, particularly those associated with Epstein–Barr virus (EBV), depends on various factors, including the type of lymphoma, patient characteristics, and EBV’s influence on the disease’s biology. The International Prognostic Index (IPI) remains a cornerstone for assessing prognosis in aggressive lymphomas. It categorizes patients into low-, intermediate-, and high-risk groups based on baseline characteristics such as age, Eastern Cooperative Oncology Group (ECOG) performance status, lactate dehydrogenase (LDH) levels, Ann Arbor stage, and extranodal involvement [53]. In HIV patients, who frequently experience extranodal involvement, the prognosis is often poor [169].
Research suggests that localized malignant lymphomas in the head and neck region, particularly diffuse large B-cell lymphoma (DLBCL), have demonstrated a better prognosis with contemporary treatment strategies. One study reported a 5-year overall survival rate of 83% for patients with localized lymphomas in this area, reflecting significant progress compared to historical data [170].
For head and neck DLBCL, treatment with R-CHOP chemotherapy (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) followed by radiotherapy has shown high rates of local control and survival. This approach has been associated with a 5-year overall survival rate of 61% and a relapse-free survival rate of 72% [171]. Radiotherapy after chemotherapy has further improved local control, achieving a final local control rate of 94% [171].
Extranodal non-Hodgkin lymphoma of the head and neck has a 5-year overall survival rate of 63.2%, with factors such as ECOG performance status, Ann Arbor stage, and IPI risk stratification being significant predictors of survival [172]. Additionally, ill-defined tumor margins on imaging have been associated with poorer clinical outcomes [173].
HIV-positive individuals face a 20- to 60-fold increased risk of developing lymphoma, with B-cell lymphomas being predominant [173,174]. In these cases, EBV is integrated into the neoplastic cells in 40% to 60% of systemic lymphomas and nearly all primary central nervous system lymphomas [175,176].
In ENKTL, a review of 153 cases revealed no association between EBV status and prognosis, although EBV-positive patients exhibited a higher propensity for metastasis [177]. Conversely, a systematic review and meta-analysis highlighted that EBV positivity correlates with poorer overall survival (OS) and disease-specific survival (DSS) in Hodgkin lymphoma (HL). Specifically, EBV-positive HL patients had a hazard ratio (HR) of 1.443 for OS and 2.312 for DSS compared to EBV-negative patients. However, age plays a significant role. Among children and adolescents, EBV positivity was linked to favorable OS outcomes, though this trend reversed in older adults, where EBV positivity was associated with significantly poorer OS (HR = 1.905) and DSS [178].
The Chelsea and Westminster cohort study of 111 patients treated for AIDS-related NHL identified critical prognostic factors, including age, tumor stage, LDH levels, ECOG status, and the number of extranodal sites. Additionally, a CD4 count below 100 × 106 cells/L was a strong predictor of mortality. While survival has improved in the combined antiretroviral therapy (cART) era, prognostic scores in the highest quartile showed a sevenfold increased risk of death compared to the lowest quartile. Nonetheless, the study’s limited sample size and variability in treatment regimens warrant further investigation [179].
Patients with higher CD4 counts tend to have better survival rates, as effective HIV management with cART can mitigate some of the adverse effects of immunosuppression on lymphoma prognosis. Therefore, maintaining a higher CD4 count through effective antiretroviral therapy is crucial for improving the prognosis of lymphoma in HIV-infected patients [180,181].
8. Conclusions
The roles of EBV and HIV in the development of various lymphomas are crucial and underscore the complexity of their pathogenesis. EBV’s ability to manipulate cellular pathways emphasizes the importance of targeted therapies that address both viral and host factors driving lymphoma formation. Advances in diagnostic methods, including molecular and immunohistochemical tools, have enhanced the detection of EBV-associated lymphomas. The progression from EBV infection to lymphoma remains poorly understood, particularly in immunosuppressed populations and in older patients, where the risk is elevated. Prognosis varies based on clinical, biological, and molecular markers and age. While significant therapeutic progress has been achieved, especially with the introduction of ART, challenges persist in managing relapsed and refractory lymphomas. Continued research into novel therapies, such as inhibitors targeting specific EBV-related pathways and personalized treatment approaches, is vital. Bridging the gaps in understanding EBV’s role in lymphoma development and addressing the absence of effective animal models are essential steps toward improving the management and outcomes of EBV-associated lymphomas.
Author Contributions
Conceptualization, S.R.-G. and J.d.J.L.-T.; writing—original draft preparation, S.R.-G.; writing—review and editing, S.R.-G., J.d.J.L.-T. and C.B.E.-A.; visualization, S.R.-G., J.d.J.L.-T. and C.B.E.-A.; supervision, S.R.-G., J.d.J.L.-T. and C.B.E.-A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Acknowledgments
The authors acknowledge Lynna Marie Kiere, a native English speaker and with expertise in biological sciences, for proofreading and English revision of the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| EBV | Epstein–Barr Virus |
| HIV | Human Immunodeficiency Virus |
| ART | Antiretroviral Treatment |
References
- Woolhouse, M.; Scott, F.; Hudson, Z.; Howey, R.; Chase-Topping, M. Human Viruses: Discovery and Emergence. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 2864–2871. [Google Scholar] [CrossRef] [PubMed]
- Mesri, E.A.; Feitelson, M.A.; Munger, K. Human Viral Oncogenesis: A Cancer Hallmarks Analysis. Cell Host Microbe 2014, 15, 266–282. [Google Scholar] [CrossRef]
- Engels, E.A. Epidemiologic Perspectives on Immunosuppressed Populations and the Immunosurveillance and Immunocontainment of Cancer. Am. J. Transplant. 2019, 19, 3223–3232. [Google Scholar] [CrossRef]
- Bakkalci, D.; Jia, Y.; Winter, J.R.; Lewis, J.E.; Taylor, G.S.; Stagg, H.R. Risk Factors for Epstein Barr Virus-Associated Cancers: A Systematic Review, Critical Appraisal, and Mapping of the Epidemiological Evidence. J. Glob. Health 2020, 10, 010405. [Google Scholar] [CrossRef]
- Epstein, M.A.; Achong, B.G.; Barr, Y.M. VIRUS PARTICLES IN CULTURED LYMPHOBLASTS FROM BURKITT’S LYMPHOMA. Lancet Lond. Engl. 1964, 1, 702–703. [Google Scholar] [CrossRef]
- Ley-Tomas, J.d.J.; Perez-Delgadillo, G.M.; Espinosa-Arce, C.; Ramirez-Gil, L.S. Laryngotracheal Stenosis Secondary to Mantle Cell Lymphoma. Cureus 2024, 16, e61900. [Google Scholar] [CrossRef]
- Wong, Y.; Meehan, M.T.; Burrows, S.R.; Doolan, D.L.; Miles, J.J. Estimating the Global Burden of Epstein–Barr Virus-Related Cancers. J. Cancer Res. Clin. Oncol. 2022, 148, 31–46. [Google Scholar] [CrossRef]
- Chantada, G.L.; Felice, M.S.; Zubizarreta, P.A.; Diaz, L.; Gallo, G.; Sackmann-Muriel, F. Results of a BFM-Based Protocol for the Treatment of Childhood B-Non-Hodgkin’s Lymphoma and B-Acute Lymphoblastic Leukemia in Argentina. Med. Pediatr. Oncol. 1997, 28, 333–341. [Google Scholar] [CrossRef]
- Martelli, M.; Ferreri, A.J.M.; Agostinelli, C.; Di Rocco, A.; Pfreundschuh, M.; Pileri, S.A. Diffuse Large B-Cell Lymphoma. Crit. Rev. Oncol. Hematol. 2013, 87, 146–171. [Google Scholar] [CrossRef]
- Friedberg, J.W.; Fisher, R.I. Diffuse Large B-Cell Lymphoma. Hematol. Oncol. Clin. N. Am. 2008, 22, 941–952. [Google Scholar] [CrossRef] [PubMed]
- Mahy, M.; Marsh, K.; Sabin, K.; Wanyeki, I.; Daher, J.; Ghys, P.D. HIV Estimates through 2018: Data for Decision-Making. AIDS 2019, 33, S203. [Google Scholar] [CrossRef] [PubMed]
- Fettig, J.; Swaminathan, M.; Murrill, C.S.; Kaplan, J.E. Global Epidemiology of HIV. Infect. Dis. Clin. N. Am. 2014, 28, 323–337. [Google Scholar] [CrossRef]
- Carbone, A.; Cesarman, E.; Spina, M.; Gloghini, A.; Schulz, T.F. HIV-Associated Lymphomas and Gamma-Herpesviruses. Blood 2009, 113, 1213–1224. [Google Scholar] [CrossRef]
- Zhang, F.; Li, W.; Zheng, X.; Ren, Y.; Li, L.; Yin, H. The Novel Immune Landscape of Immune-Checkpoint Blockade in EBV-associated Malignancies. FASEB J. 2024, 38, e70139. [Google Scholar] [CrossRef]
- Ribatti, D.; Cazzato, G.; Tamma, R.; Annese, T.; Ingravallo, G.; Specchia, G. Immune Checkpoint Inhibitors Targeting PD-1/PD-L1 in the Treatment of Human Lymphomas. Front. Oncol. 2024, 14, 1420920. [Google Scholar] [CrossRef]
- Dowd, J.B.; Palermo, T.; Brite, J.; McDade, T.W.; Aiello, A. Seroprevalence of Epstein-Barr Virus Infection in U.S. Children Ages 6–19, 2003–2010. PLoS ONE 2013, 8, e64921. [Google Scholar] [CrossRef]
- Yu, H.; Robertson, E.S. Epstein-Barr Virus History and Pathogenesis. Viruses 2023, 15, 714. [Google Scholar] [CrossRef] [PubMed]
- Tonoyan, L.; Vincent-Bugnas, S.; Olivieri, C.-V.; Doglio, A. New Viral Facets in Oral Diseases: The EBV Paradox. Int. J. Mol. Sci. 2019, 20, 5861. [Google Scholar] [CrossRef]
- Kang, S.H.; Lee, Y.-H.; Myong, J.-P.; Kwon, M. The Impact of Infectious Mononucleosis History on the Risk of Developing Lymphoma and Nasopharyngeal Carcinoma: A Retrospective Large-Scale Cohort Study Using National Health Insurance Data in South Korea. Cancer Res. Treat. 2024, 56, 1077–1083. [Google Scholar] [CrossRef] [PubMed]
- Hjalgrim, H.; Askling, J.; Rostgaard, K.; Hamilton-Dutoit, S.; Frisch, M.; Zhang, J.-S.; Madsen, M.; Rosdahl, N.; Konradsen, H.B.; Storm, H.H.; et al. Characteristics of Hodgkin’s Lymphoma after Infectious Mononucleosis. N. Engl. J. Med. 2003, 349, 1324–1332. [Google Scholar] [CrossRef]
- Henle, W.; Diehl, V.; Kohn, G.; Zur Hausen, H.; Henle, G. Herpes-Type Virus and Chromosome Marker in Normal Leukocytes after Growth with Irradiated Burkitt Cells. Science 1967, 157, 1064–1065. [Google Scholar] [CrossRef] [PubMed]
- Diehl, V.; Henle, G.; Henle, W.; Kohn, G. Demonstration of a Herpes Group Virus in Cultures of Peripheral Leukocytes from Patients with Infectious Mononucleosis. J. Virol. 1968, 2, 663–669. [Google Scholar] [CrossRef]
- Zimber, U.; Adldinger, H.K.; Lenoir, G.M.; Vuillaume, M.; Knebel-Doeberitz, M.V.; Laux, G.; Desgranges, C.; Wittmann, P.; Freese, U.K.; Schneider, U. Geographical Prevalence of Two Types of Epstein-Barr Virus. Virology 1986, 154, 56–66. [Google Scholar] [CrossRef]
- Fingeroth, J.D.; Weis, J.J.; Tedder, T.F.; Strominger, J.L.; Biro, P.A.; Fearon, D.T. Epstein-Barr Virus Receptor of Human B Lymphocytes Is the C3d Receptor CR2. Proc. Natl. Acad. Sci. USA 1984, 81, 4510–4514. [Google Scholar] [CrossRef]
- Tanner, J.; Weis, J.; Fearon, D.; Whang, Y.; Kieff, E. Epstein-Barr Virus Gp350/220 Binding to the B Lymphocyte C3d Receptor Mediates Adsorption, Capping, and Endocytosis. Cell 1987, 50, 203–213. [Google Scholar] [CrossRef]
- Hatano, Y.; Sato, K.; Imai, S.; Teshima, T.; Inoue, D.; Shiozaki, A.; Tanaka, H.; Suzuki, M. Virus-driven carcinogenesis. Cancers 2021, 13, 2625. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Watanabe, T.; Suzuki, C.; Abe, Y.; Masud, H.M.A.A.; Inagaki, T.; Yoshida, M.; Suzuki, T.; Goshima, F.; Adachi, J.; et al. S-like-Phase Cyclin-Dependent Kinases Stabilize the Epstein-Barr Virus BDLF4 Protein To Temporally Control Late Gene Transcription. J. Virol. 2019, 93, e01707–e01718. [Google Scholar] [CrossRef]
- Hjalgrim, H.; Rostgaard, K.; Johnson, P.C.D.; Lake, A.; Shield, L.; Little, A.-M.; Ekstrom-Smedby, K.; Adami, H.-O.; Glimelius, B.; Hamilton-Dutoit, S.; et al. HLA-A alleles and infectious mononucleosis suggest a critical role for cytotoxic T-cell response in EBV-related Hodgkin lymphoma. Proc. Natl. Acad. Sci. USA 2010, 107, 6400–6405. [Google Scholar] [CrossRef]
- Bahnassy, A.A.; Zekri, A.-R.N.; Asaad, N.; El-Houssini, S.; Khalid, H.M.; Sedky, L.M.; Mokhtar, N.M. Epstein-Barr Viral Infection in Extranodal Lymphoma of the Head and Neck: Correlation with Prognosis and Response to Treatment. Histopathology 2006, 48, 516–528. [Google Scholar] [CrossRef]
- Münz, C. Latency and Lytic Replication in Epstein-Barr Virus-Associated Oncogenesis. Nat. Rev. Microbiol. 2019, 17, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Kerr, J.R. Epstein-Barr Virus (EBV) Reactivation and Therapeutic Inhibitors. J. Clin. Pathol. 2019, 72, 651–658. [Google Scholar] [CrossRef]
- Ward, B.J.H.; Schaal, D.L.; Nkadi, E.H.; Scott, R.S. EBV Association with Lymphomas and Carcinomas in the Oral Compartment. Viruses 2022, 14, 2700. [Google Scholar] [CrossRef]
- Yee, J.; White, R.E.; Anderton, E.; Allday, M.J. Latent Epstein-Barr Virus Can Inhibit Apoptosis in B Cells by Blocking the Induction of NOXA Expression. PLoS ONE 2011, 6, e28506. [Google Scholar] [CrossRef]
- Li, J.S.Z.; Abbasi, A.; Kim, D.H.; Lippman, S.M.; Alexandrov, L.B.; Cleveland, D.W. Chromosomal Fragile Site Breakage by EBV-Encoded EBNA1 at Clustered Repeats. Nature 2023, 616, 504–509. [Google Scholar] [CrossRef]
- Labrecque, L.G.; Xue, S.A.; Kazembe, P.; Phillips, J.; Lampert, I.; Wedderburn, N.; Griffin, B.E. Expression of Epstein-Barr Virus Lytically Related Genes in African Burkitt’s Lymphoma: Correlation with Patient Response to Therapy. Int. J. Cancer 1999, 81, 6–11. [Google Scholar] [CrossRef]
- Shen, Z.; Hu, L.; Yao, M.; He, C.; Liu, Q.; Wang, F.; Gu, W.; Wang, Y.; Dong, M.; Zhu, T.; et al. Disparity Analysis and Prognostic Value of Pretreatment Whole Blood Epstein-Barr Virus DNA Load and Epstein-Barr Encoding Region Status in Lymphomas: A Retrospective Multicenter Study in Huaihai Lymphoma Working Group. Int. J. Cancer 2022, 150, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.-H.; Lu, T.-X.; Tian, T.; Wang, L.; Fan, L.; Xu, J.; Zhang, R.; Gong, Q.-X.; Zhang, Z.-H.; Li, J.-Y.; et al. Epstein-Barr Virus (EBV) DNA in Whole Blood as a Superior Prognostic and Monitoring Factor than EBV-Encoded Small RNA in Situ Hybridization in Diffuse Large B-Cell Lymphoma. Clin. Microbiol. Infect. 2015, 21, 596–602. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Kim, J.Y.; Kim, S.; Park, Y. Utility of Epstein-Barr Viral Load in Blood for Diagnosing and Predicting Prognosis of Lymphoma: A Comparison with Epstein-Barr Virus-Encoded RNA in Situ Hybridization. J. Mol. Diagn. 2022, 24, 977–991. [Google Scholar] [CrossRef]
- Kanakry, J.A.; Li, H.; Gellert, L.L.; Lemas, M.V.; Hsieh, W.; Hong, F.; Tan, K.L.; Gascoyne, R.D.; Gordon, L.I.; Fisher, R.I.; et al. Plasma Epstein-Barr Virus DNA Predicts Outcome in Advanced Hodgkin Lymphoma: Correlative Analysis from a Large North American Cooperative Group Trial. Blood 2013, 121, 3547–3553. [Google Scholar] [CrossRef]
- Zeng, M.; Jia, Q.; Chen, J. Enhanced Prognostic Evaluation of Diffuse Large B-Cell Lymphoma: A Comprehensive Surveillance Study Incorporating Epstein–Barr Virus Infection Status and Immunohistochemical Markers. J. Med. Virol. 2024, 96, e29834. [Google Scholar] [CrossRef] [PubMed]
- Price, A.M.; Luftig, M.A. To Be or Not IIb: A Multi-Step Process for Epstein-Barr Virus Latency Establishment and Consequences for B Cell Tumorigenesis. PLoS Pathog. 2015, 11, e1004656. [Google Scholar] [CrossRef]
- Alfieri, C.; Birkenbach, M.; Kieff, E. Early Events in Epstein-Barr Virus Infection of Human B Lymphocytes. Virology 1991, 181, 595–608. [Google Scholar] [CrossRef]
- Nikitin, P.A.; Yan, C.M.; Forte, E.; Bocedi, A.; Tourigny, J.P.; White, R.E.; Allday, M.J.; Patel, A.; Dave, S.S.; Kim, W.; et al. An ATM/Chk2-Mediated DNA Damage-Responsive Signaling Pathway Suppresses Epstein-Barr Virus Transformation of Primary Human B Cells. Cell Host Microbe 2010, 8, 510–522. [Google Scholar] [CrossRef] [PubMed]
- Cahir-McFarland, E.D.; Davidson, D.M.; Schauer, S.L.; Duong, J.; Kieff, E. NF-Kappa B Inhibition Causes Spontaneous Apoptosis in Epstein-Barr Virus-Transformed Lymphoblastoid Cells. Proc. Natl. Acad. Sci. USA 2000, 97, 6055–6060. [Google Scholar] [CrossRef]
- Cesarman, E. Gammaherpesviruses and Lymphoproliferative Disorders. Annu. Rev. Pathol. 2014, 9, 349–372. [Google Scholar] [CrossRef] [PubMed]
- Thorley-Lawson, D.A.; Gross, A. Persistence of the Epstein-Barr Virus and the Origins of Associated Lymphomas. N. Engl. J. Med. 2004, 350, 1328–1337. [Google Scholar] [CrossRef] [PubMed]
- Kis, L.L.; Salamon, D.; Persson, E.K.; Nagy, N.; Scheeren, F.A.; Spits, H.; Klein, G.; Klein, E. IL-21 Imposes a Type II EBV Gene Expression on Type III and Type I B Cells by the Repression of C- and Activation of LMP-1-Promoter. Proc. Natl. Acad. Sci. USA 2010, 107, 872–877. [Google Scholar] [CrossRef]
- Oishi, N.; Bagán, J.V.; Javier, K.; Zapater, E. Head and Neck Lymphomas in HIV Patients: A Clinical Perspective. Int. Arch. Otorhinolaryngol. 2017, 21, 399–407. [Google Scholar] [CrossRef]
- Guidry, J.; Birdwell, C.; Scott, R. Epstein–Barr Virus in the Pathogenesis of Oral Cancers. Oral Dis. 2018, 24, 497–508. [Google Scholar] [CrossRef]
- Migliaro, M.; Massuh, D.; Infante, M.F.; Brahm, A.M.; San Martín, M.T.; Ortuño, D. Role of Epstein-Barr Virus and Human Papilloma Virus in the Development of Oropharyngeal Cancer: A Literature Review. Int. J. Dent. 2022, 2022, 3191569. [Google Scholar] [CrossRef]
- Carbone, A. AIDS-Related Non-Hodgkin’s Lymphomas: From Pathology and Molecular Pathogenesis to Treatment. Hum. Pathol. 2002, 33, 392–404. [Google Scholar] [CrossRef]
- Re, A.; Cattaneo, C.; Rossi, G. Hiv and Lymphoma: From Epidemiology to Clinical Management. Mediterr. J. Hematol. Infect. Dis. 2019, 11, e2019004. [Google Scholar] [CrossRef]
- Tirelli, U.; Spina, M.; Gaidano, G.; Vaccher, E.; Franceschi, S.; Carbone, A. Epidemiological, Biological and Clinical Features of HIV-Related Lymphomas in the Era of Highly Active Antiretroviral Therapy. AIDS Lond. Engl. 2000, 14, 1675–1688. [Google Scholar] [CrossRef]
- Lin, F.Y.; Patel, Z.M. (Eds.) ENT Board Prep: High Yield Review for the Otolaryngology in-Service and Board Exams; Springer International Publishing: Cham, Switzerland, 2023; ISBN 9783031260476. [Google Scholar]
- Epeldegui, M.; Hussain, S.K. The Role of Microbial Translocation and Immune Activation in AIDS-Associated Non-Hodgkin Lymphoma Pathogenesis: What Have We Learned? Crit. Rev. Immunol. 2020, 40, 41–51. [Google Scholar] [CrossRef]
- Liang, Y.; Chen, X.; Zhang, X.; Guo, C.; Zhang, Y. Virus-Driven Dysregulation of the BCR Pathway: A Potential Mechanism for the High Prevalence of HIV Related B-Cell Lymphoma. Ann. Hematol. 2024, 103, 4839–4849. [Google Scholar] [CrossRef]
- Giagulli, C.; D’Ursi, P.; He, W.; Zorzan, S.; Caccuri, F.; Varney, K.; Orro, A.; Marsico, S.; Otjacques, B.; Laudanna, C.; et al. A Single Amino Acid Substitution Confers B-Cell Clonogenic Activity to the HIV-1 Matrix Protein P17. Sci. Rep. 2017, 7, 6555. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Zhou, F.; Qin, D.; Zeng, Y.; Lv, Z.; Yao, S.; Lu, C. Human Immunodeficiency Virus Type 1 Induces Lytic Cycle Replication of Kaposi’s-Sarcoma-Associated Herpesvirus: Role of Ras/c-Raf/MEK1/2, PI3K/AKT, and NF-κB Signaling Pathways. J. Mol. Biol. 2011, 410, 1035–1051. [Google Scholar] [CrossRef]
- Dolcetti, R.; Giagulli, C.; He, W.; Selleri, M.; Caccuri, F.; Eyzaguirre, L.M.; Mazzuca, P.; Corbellini, S.; Campilongo, F.; Marsico, S.; et al. Role of HIV-1 Matrix Protein P17 Variants in Lymphoma Pathogenesis. Proc. Natl. Acad. Sci. USA 2015, 112, 14331–14336, Erratum in Proc. Natl. Acad. Sci. USA 2015, 112, E7033. [Google Scholar] [CrossRef]
- Germini, D.; Tsfasman, T.; Klibi, M.; El-Amine, R.; Pichugin, A.; Iarovaia, O.V.; Bilhou-Nabera, C.; Subra, F.; Bou Saada, Y.; Sukhanova, A.; et al. HIV Tat Induces a Prolonged MYC Relocalization next to IGH in Circulating B-Cells. Leukemia 2017, 31, 2515–2522. [Google Scholar] [CrossRef] [PubMed]
- Oppezzo, P.; Navarrete, M.; Chiorazzi, N. AID in Chronic Lymphocytic Leukemia: Induction and Action During Disease Progression. Front. Oncol. 2021, 11, 634383. [Google Scholar] [CrossRef] [PubMed]
- Doyon-Laliberté, K.; Aranguren, M.; Poudrier, J.; Roger, M. Marginal Zone B-Cell Populations and Their Regulatory Potential in the Context of HIV and Other Chronic Inflammatory Conditions. Int. J. Mol. Sci. 2022, 23, 3372. [Google Scholar] [CrossRef]
- Silva, R.; Moir, S.; Kardava, L.; Debell, K.; Simhadri, V.R.; Ferrando-Martínez, S.; Leal, M.; Peña, J.; Coligan, J.E.; Borrego, F. CD300a Is Expressed on Human B Cells, Modulates BCR-Mediated Signaling, and Its Expression Is down-Regulated in HIV Infection. Blood 2011, 117, 5870–5880. [Google Scholar] [CrossRef]
- Doshi, D.V.; Tripathi, U.; Dave, R.I.; Pandya, S.J.; Shukla, H.K.; Parikh, B.C. Rare Tumors of Sinonasal Track. Indian J. Otolaryngol. Head Neck Surg. 2010, 62, 111–117. [Google Scholar] [CrossRef][Green Version]
- Das, S.; Kirsch, C.F.E. Imaging of Lumps and Bumps in the Nose: A Review of Sinonasal Tumours. Cancer Imaging 2005, 5, 167–177. [Google Scholar] [CrossRef]
- Bernardo, P.S.; Hancio, T.; Vasconcelos, F.d.C.; Nestal de Moraes, G.; de Sá Bigni, R.; Wernersbach Pinto, L.; Thuler, L.C.S.; Maia, R.C. Primary Diffuse Large B-Cell Lymphoma of the Head and Neck in a Brazilian Single-Center Study. Oral Dis. 2023, 29, 968–977. [Google Scholar] [CrossRef]
- Miller, C.S.; Berger, J.R.; Mootoor, Y.; Avdiushko, S.A.; Zhu, H.; Kryscio, R.J. High Prevalence of Multiple Human Herpesviruses in Saliva from Human Immunodeficiency Virus-Infected Persons in the Era of Highly Active Antiretroviral Therapy. J. Clin. Microbiol. 2006, 44, 2409–2415. [Google Scholar] [CrossRef] [PubMed]
- Shmakova, A.; Germini, D.; Vassetzky, Y. HIV-1, HAART and Cancer: A Complex Relationship. Int. J. Cancer 2020, 146, 2666–2679. [Google Scholar] [CrossRef]
- Besson, C.; Lancar, R.; Prevot, S.; Brice, P.; Meyohas, M.-C.; Marchou, B.; Gabarre, J.; Bonnet, F.; Goujard, C.; Lambotte, O.; et al. High Risk Features Contrast with Favorable Outcomes in HIV-Associated Hodgkin Lymphoma in the Modern cART Era, ANRS CO16 LYMPHOVIR Cohort. Clin. Infect. Dis. 2015, 61, 1469–1475. [Google Scholar] [CrossRef] [PubMed]
- Montoto, S.; Shaw, K.; Okosun, J.; Gandhi, S.; Fields, P.; Wilson, A.; Shanyinde, M.; Cwynarski, K.; Marcus, R.; de Vos, J.; et al. HIV Status Does Not Influence Outcome in Patients with Classical Hodgkin Lymphoma Treated with Chemotherapy Using Doxorubicin, Bleomycin, Vinblastine, and Dacarbazine in the Highly Active Antiretroviral Therapy Era. J. Clin. Oncol. 2012, 30, 4111–4116. [Google Scholar] [CrossRef] [PubMed]
- Meister, A.; Hentrich, M.; Wyen, C.; Hübel, K. Malignant Lymphoma in the HIV-Positive Patient. Eur. J. Haematol. 2018, 101, 119–126. [Google Scholar] [CrossRef]
- Schommers, P.; Hentrich, M.; Hoffmann, C.; Gillor, D.; Zoufaly, A.; Jensen, B.; Bogner, J.R.; Thoden, J.; Wasmuth, J.-C.; Wolf, T.; et al. Survival of AIDS-Related Diffuse Large B-Cell Lymphoma, Burkitt Lymphoma, and Plasmablastic Lymphoma in the German HIV Lymphoma Cohort. Br. J. Haematol. 2015, 168, 806–810. [Google Scholar] [CrossRef]
- Nilavar, N.M.; Raghavan, S.C. HIV Integrase Inhibitors That Inhibit Strand Transfer Interact with RAG1 and Hamper Its Activities. Int. Immunopharmacol. 2021, 95, 107515. [Google Scholar] [CrossRef] [PubMed]
- Castilho, J.L.; Bian, A.; Jenkins, C.A.; Shepherd, B.E.; Sigel, K.; Gill, M.J.; Kitahata, M.M.; Silverberg, M.J.; Mayor, A.M.; Coburn, S.B.; et al. CD4/CD8 Ratio and Cancer Risk Among Adults With HIV. J. Natl. Cancer Inst. 2022, 114, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Chammartin, F.; Mocroft, A.; Egle, A.; Zangerle, R.; Smith, C.; Mussini, C.; Wit, F.; Vehreschild, J.J.; d’Arminio Monforte, A.; Castagna, A.; et al. Measures of Longitudinal Immune Dysfunction and Risk of AIDS and Non-AIDS Defining Malignancies in Antiretroviral-Treated People With Human Immunodeficiency Virus. Clin. Infect. Dis. 2024, 78, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Thorley-Lawson, D.A.; Poodry, C.A. Identification and Isolation of the Main Component (Gp350-Gp220) of Epstein-Barr Virus Responsible for Generating Neutralizing Antibodies In Vivo. J. Virol. 1982, 43, 730–736. [Google Scholar] [CrossRef]
- North, J.R.; Morgan, A.J.; Thompson, J.L.; Epstein, M.A. Purified Epstein-Barr Virus Mr 340,000 Glycoprotein Induces Potent Virus-Neutralizing Antibodies When Incorporated in Liposomes. Proc. Natl. Acad. Sci. USA 1982, 79, 7504–7508. [Google Scholar] [CrossRef]
- Ruiss, R.; Jochum, S.; Wanner, G.; Reisbach, G.; Hammerschmidt, W.; Zeidler, R. A Virus-Like Particle-Based Epstein-Barr Virus Vaccine. J. Virol. 2011, 85, 13105–13113. [Google Scholar] [CrossRef]
- Lewis, W.D.; Lilly, S.; Jones, K.L. Lymphoma: Diagnosis and Treatment. Am. Fam. Physician 2020, 101, 34–41. [Google Scholar] [CrossRef]
- Cheson, B.D.; Fisher, R.I.; Barrington, S.F.; Cavalli, F.; Schwartz, L.H.; Zucca, E.; Lister, T.A.; Alliance, Australasian Leukaemia and Lymphoma Group; Eastern Cooperative Oncology Group; European Mantle Cell Lymphoma Consortium; et al. Recommendations for Initial Evaluation, Staging, and Response Assessment of Hodgkin and Non-Hodgkin Lymphoma: The Lugano Classification. J. Clin. Oncol. 2014, 32, 3059–3068. [Google Scholar] [CrossRef]
- Saatci, D.; Zhu, C.; Harnden, A.; Hippisley-Cox, J. Presentation of B-Cell Lymphoma in Childhood and Adolescence: A Systematic Review and Meta-Analysis. BMC Cancer 2024, 24, 718. [Google Scholar] [CrossRef]
- Sparano, J.A. Clinical Aspects and Management of AIDS-Related Lymphoma. Eur. J. Cancer 2001, 37, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Mozas, P.; Sorigué, M.; López-Guillermo, A. Follicular Lymphoma: An Update on Diagnosis, Prognosis, and Management. Med. Clin. 2021, 157, 440–448. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, E.M.; de Cáceres, C.V.B.L.; Santos-Silva, A.R.; Vargas, P.A.; Lopes, M.A.; Pontes, H.A.R.; Pontes, F.S.C.; Mesquita, R.A.; de Sousa, S.F.; Abreu, L.G.; et al. Clinical Diagnostic Approach for Oral Lymphomas: A Multi-Institutional, Observational Study Based on 107 Cases. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2023, 136, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Zapater, E.; Bagan, J.V.; Campos, A.; Armengot, M.; Abril, V.; Basterra, J. Non-Hodgkin’s lymphoma of the head and neck in association with HIV infection. Ann. Otolaryngol. Chir. Cervicofac. 1996, 113, 69–72. [Google Scholar]
- Finn, D.G. Lymphoma of the Head and Neck and Acquired Immunodeficiency Syndrome: Clinical Investigation and Immunohistological Study. Laryngoscope 1995, 105, 1–18. [Google Scholar] [CrossRef]
- Samra, B.; Khoury, J.D.; Morita, K.; Ravandi, F.; Richard-Carpentier, G.; Short, N.J.; El Hussein, S.; Thompson, P.; Jain, N.; Kantarjian, H.; et al. Long-Term Outcome of Hyper-CVAD-R for Burkitt Leukemia/Lymphoma and High-Grade B-Cell Lymphoma: Focus on CNS Relapse. Blood Adv. 2021, 5, 3913–3918, Erratum in Blood Adv. 2022, 6, 2452. [Google Scholar] [CrossRef]
- Wilczynski, A.; Görg, C.; Timmesfeld, N.; Ramaswamy, A.; Neubauer, A.; Burchert, A.; Trenker, C. Value and Diagnostic Accuracy of Ultrasound-Guided Full Core Needle Biopsy in the Diagnosis of Lymphadenopathy: A Retrospective Evaluation of 793 Cases. J. Ultrasound Med. 2020, 39, 559–567. [Google Scholar] [CrossRef]
- Groneck, L.; Quaas, A.; Hallek, M.; Zander, T.; Weihrauch, M.R. Ultrasound-Guided Core Needle Biopsies for Workup of Lymphadenopathy and Lymphoma. Eur. J. Haematol. 2016, 97, 379–386. [Google Scholar] [CrossRef]
- Ribeiro, A.; Pereira, D.; Escalón, M.P.; Goodman, M.; Byrne, G.E. EUS-Guided Biopsy for the Diagnosis and Classification of Lymphoma. Gastrointest. Endosc. 2010, 71, 851–855. [Google Scholar] [CrossRef]
- Oluwasanmi, A.F.; Wood, S.J.; Baldwin, D.L.; Sipaul, F. Malignancy in Asymmetrical but Otherwise Normal Palatine Tonsils. Ear. Nose. Throat J. 2006, 85, 661–663. [Google Scholar] [CrossRef]
- Edwards, D.; Sheehan, S.; Ingrams, D. Unilateral Tonsil Enlargement in Children and Adults: Is Routine Histology Tonsillectomy Warranted? A Multi-Centre Series of 323 Patients. J. Laryngol. Otol. 2023, 137, 1022–1026. [Google Scholar] [CrossRef]
- Kemp, S.; Gallagher, G.; Kabani, S.; Noonan, V.; O’Hara, C. Oral Non-Hodgkin’s Lymphoma: Review of the Literature and World Health Organization Classification with Reference to 40 Cases. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2008, 105, 194–201. [Google Scholar] [CrossRef]
- Levine, A.M. Acquired Immunodeficiency Syndrome-Related Lymphoma. Blood 1992, 80, 8–20. [Google Scholar] [CrossRef]
- Corti, M.; Villafañe, M.; Bistmans, A.; Narbaitz, M.; Gilardi, L. Primary Extranodal Non-Hodgkin Lymphoma of the Head and Neck in Patients with Acquired Immunodeficiency Syndrome: A Clinicopathologic Study of 24 Patients in a Single Hospital of Infectious Diseases in Argentina. Int. Arch. Otorhinolaryngol. 2014, 18, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Geser, A.; de Thé, G.; Lenoir, G.; Day, N.E.; Williams, E.H. Final Case Reporting from the Ugandan Prospective Study of the Relationship between EBV and Burkitt’s Lymphoma. Int. J. Cancer 1982, 29, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Randhawa, P.S.; Jaffe, R.; Demetris, A.J.; Nalesnik, M.; Starzl, T.E.; Chen, Y.Y.; Weiss, L.M. Expression of Epstein-Barr Virus-Encoded Small RNA (by the EBER-1 Gene) in Liver Specimens from Transplant Recipients with Post-Transplantation Lymphoproliferative Disease. N. Engl. J. Med. 1992, 327, 1710–1714. [Google Scholar] [CrossRef]
- Bruu, A.L.; Hjetland, R.; Holter, E.; Mortensen, L.; Natås, O.; Petterson, W.; Skar, A.G.; Skarpaas, T.; Tjade, T.; Asjø, B. Evaluation of 12 Commercial Tests for Detection of Epstein-Barr Virus-Specific and Heterophile Antibodies. Clin. Diagn. Lab. Immunol. 2000, 7, 451–456. [Google Scholar] [CrossRef]
- Mundo, L.; Ambrosio, M.R.; Picciolini, M.; Lo Bello, G.; Gazaneo, S.; Del Porro, L.; Lazzi, S.; Navari, M.; Onyango, N.; Granai, M.; et al. Unveiling Another Missing Piece in EBV-Driven Lymphomagenesis: EBV-Encoded MicroRNAs Expression in EBER-Negative Burkitt Lymphoma Cases. Front. Microbiol. 2017, 8, 229. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.-L.; Han, X.-Q.; Hu, J.; Wang, G.-H.; Gao, J.-W.; Wang, X.; Liang, D.-Y. Comparison of Three Methods for the Detection of Epstein-Barr Virus in Hodgkin’s Lymphoma in Paraffin-Embedded Tissues. Mol. Med. Rep. 2013, 7, 89–92. [Google Scholar] [CrossRef]
- Zapater, E.; Bagán, J.V.; Carbonell, F.; Basterra, J. Malignant Lymphoma of the Head and Neck. Oral Dis. 2010, 16, 119–128. [Google Scholar] [CrossRef]
- Swerdlow, S.H.; Campo, E.; Harris, N.L.; Jaffe, E.S.; Pileri, S.A.; Stein, H.; Thiele, J. (Eds.) WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues; IARC Press: Lyon, France, 2017; ISBN 978-92-832-4494-3. [Google Scholar]
- Boué, F.; Gabarre, J.; Gisselbrecht, C.; Reynes, J.; Cheret, A.; Bonnet, F.; Billaud, E.; Raphael, M.; Lancar, R.; Costagliola, D. Phase II Trial of CHOP plus Rituximab in Patients with HIV-Associated Non-Hodgkin’s Lymphoma. J. Clin. Oncol. 2006, 24, 4123–4128. [Google Scholar] [CrossRef]
- Ribera, J.-M.; Oriol, A.; Morgades, M.; González-Barca, E.; Miralles, P.; López-Guillermo, A.; Gardella, S.; López, A.; Abella, E.; García, M.; et al. Safety and Efficacy of Cyclophosphamide, Adriamycin, Vincristine, Prednisone and Rituximab in Patients with Human Immunodeficiency Virus-Associated Diffuse Large B-Cell Lymphoma: Results of a Phase II Trial. Br. J. Haematol. 2008, 140, 411–419. [Google Scholar] [CrossRef]
- Little, R.F.; Pittaluga, S.; Grant, N.; Steinberg, S.M.; Kavlick, M.F.; Mitsuya, H.; Franchini, G.; Gutierrez, M.; Raffeld, M.; Jaffe, E.S.; et al. Highly Effective Treatment of Acquired Immunodeficiency Syndrome-Related Lymphoma with Dose-Adjusted EPOCH: Impact of Antiretroviral Therapy Suspension and Tumor Biology. Blood 2003, 101, 4653–4659. [Google Scholar] [CrossRef]
- Barta, S.K.; Lee, J.Y.; Kaplan, L.D.; Noy, A.; Sparano, J.A. Pooled Analysis of AIDS Malignancy Consortium Trials Evaluating Rituximab plus CHOP or Infusional EPOCH Chemotherapy in HIV-Associated Non-Hodgkin Lymphoma. Cancer 2012, 118, 3977–3983. [Google Scholar] [CrossRef]
- Wang, E.S.; Straus, D.J.; Teruya-Feldstein, J.; Qin, J.; Portlock, C.; Moskowitz, C.; Goy, A.; Hedrick, E.; Zelenetz, A.D.; Noy, A. Intensive Chemotherapy with Cyclophosphamide, Doxorubicin, High-Dose Methotrexate/Ifosfamide, Etoposide, and High-Dose Cytarabine (CODOX-M/IVAC) for Human Immunodeficiency Virus-Associated Burkitt Lymphoma. Cancer 2003, 98, 1196–1205. [Google Scholar] [CrossRef]
- Barnes, J.A.; Lacasce, A.S.; Feng, Y.; Toomey, C.E.; Neuberg, D.; Michaelson, J.S.; Hochberg, E.P.; Abramson, J.S. Evaluation of the Addition of Rituximab to CODOX-M/IVAC for Burkitt’s Lymphoma: A Retrospective Analysis. Ann. Oncol. 2011, 22, 1859–1864. [Google Scholar] [CrossRef] [PubMed]
- Noy, A.; Kaplan, L.; Lee, J.; Cesarman, E.; Tam, W. Modified Dose Intensive R-CODOX-M/IVAC for HIV-Associated Burkitt (BL) (AMC 048) Shows Efficacy and Tolerability, and Predictive Potential of IRF4/MUM1 Expression. Infect. Agent. Cancer 2012, 7, O14. [Google Scholar] [CrossRef]
- Cortes, J.; Thomas, D.; Rios, A.; Koller, C.; O’Brien, S.; Jeha, S.; Faderl, S.; Kantarjian, H. Hyperfractionated Cyclophosphamide, Vincristine, Doxorubicin, and Dexamethasone and Highly Active Antiretroviral Therapy for Patients with Acquired Immunodeficiency Syndrome-Related Burkitt Lymphoma/Leukemia. Cancer 2002, 94, 1492–1499. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.A.; Faderl, S.; O’Brien, S.; Bueso-Ramos, C.; Cortes, J.; Garcia-Manero, G.; Giles, F.J.; Verstovsek, S.; Wierda, W.G.; Pierce, S.A.; et al. Chemoimmunotherapy with Hyper-CVAD plus Rituximab for the Treatment of Adult Burkitt and Burkitt-Type Lymphoma or Acute Lymphoblastic Leukemia. Cancer 2006, 106, 1569–1580. [Google Scholar] [CrossRef]
- Sparano, J.A.; Lee, S.; Chen, M.G.; Nazeer, T.; Einzig, A.; Ambinder, R.F.; Henry, D.H.; Manalo, J.; Li, T.; Von Roenn, J.H. Phase II Trial of Infusional Cyclophosphamide, Doxorubicin, and Etoposide in Patients with HIV-Associated Non-Hodgkin’s Lymphoma: An Eastern Cooperative Oncology Group Trial (E1494). J. Clin. Oncol. 2004, 22, 1491–1500. [Google Scholar] [CrossRef]
- Spina, M.; Jaeger, U.; Sparano, J.A.; Talamini, R.; Simonelli, C.; Michieli, M.; Rossi, G.; Nigra, E.; Berretta, M.; Cattaneo, C.; et al. Rituximab plus Infusional Cyclophosphamide, Doxorubicin, and Etoposide in HIV-Associated Non-Hodgkin Lymphoma: Pooled Results from 3 Phase 2 Trials. Blood 2005, 105, 1891–1897. [Google Scholar] [CrossRef]
- Hentrich, M.; Berger, M.; Wyen, C.; Siehl, J.; Rockstroh, J.K.; Müller, M.; Fätkenheuer, G.; Seidel, E.; Nickelsen, M.; Wolf, T.; et al. Stage-Adapted Treatment of HIV-Associated Hodgkin Lymphoma: Results of a Prospective Multicenter Study. J. Clin. Oncol. 2012, 30, 4117–4123. [Google Scholar] [CrossRef]
- Kim, W.S.; Song, S.Y.; Ahn, Y.C.; Ko, Y.H.; Baek, C.H.; Kim, D.Y.; Yoon, S.S.; Lee, H.G.; Kang, W.K.; Lee, H.J.; et al. CHOP Followed by Involved Field Radiation: Is It Optimal for Localized Nasal Natural Killer/T-Cell Lymphoma? Ann. Oncol. 2001, 12, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Tobinai, K.; Oguchi, M.; Ishizuka, N.; Kobayashi, Y.; Isobe, Y.; Ishizawa, K.; Maseki, N.; Itoh, K.; Usui, N.; et al. Concurrent Chemoradiotherapy for Localized Nasal Natural Killer/T-Cell Lymphoma: An Updated Analysis of the Japan Clinical Oncology Group Study JCOG0211. J. Clin. Oncol. 2012, 30, 4044–4046. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Zhang, H.; Jiang, Y.; Yang, Q.; Xie, L.; Liu, W.; Zhang, W.; Ji, X.; Li, P.; Chen, N.; et al. Phase 2 Trial of “Sandwich” L-Asparaginase, Vincristine, and Prednisone Chemotherapy with Radiotherapy in Newly Diagnosed, Stage IE to IIE, Nasal Type, Extranodal Natural Killer/T-Cell Lymphoma. Cancer 2012, 118, 3294–3301. [Google Scholar] [CrossRef]
- Kim, S.J.; Kim, K.; Kim, B.S.; Kim, C.Y.; Suh, C.; Huh, J.; Lee, S.-W.; Kim, J.S.; Cho, J.; Lee, G.-W.; et al. Phase II Trial of Concurrent Radiation and Weekly Cisplatin Followed by VIPD Chemotherapy in Newly Diagnosed, Stage IE to IIE, Nasal, Extranodal NK/T-Cell Lymphoma: Consortium for Improving Survival of Lymphoma Study. J. Clin. Oncol. 2009, 27, 6027–6032. [Google Scholar] [CrossRef]
- Kim, S.J.; Yoon, S.E.; Kim, W.S. Treatment of Localized Extranodal NK/T Cell Lymphoma, Nasal Type: A Systematic Review. J. Hematol. Oncol. 2018, 11, 140. [Google Scholar] [CrossRef]
- Tse, E.; Zhao, W.-L.; Xiong, J.; Kwong, Y.-L. How We Treat NK/T-Cell Lymphomas. J. Hematol. Oncol. 2022, 15, 74. [Google Scholar] [CrossRef] [PubMed]
- Kusano, Y.; Yokoyama, M.; Terui, Y.; Nishimura, N.; Mishima, Y.; Ueda, K.; Tsuyama, N.; Hirofumi, Y.; Takahashi, A.; Inoue, N.; et al. Low Absolute Peripheral Blood CD4+ T-Cell Count Predicts Poor Prognosis in R-CHOP-Treated Patients with Diffuse Large B-Cell Lymphoma. Blood Cancer J. 2017, 7, e558, Erratum in Blood Cancer J. 2017, 7, e561. [Google Scholar] [CrossRef]
- Roschewski, M.; Staudt, L.M.; Wilson, W.H. Burkitt’s Lymphoma. N. Engl. J. Med. 2022, 387, 1111–1122. [Google Scholar] [CrossRef]
- Witte, H.M.; Merz, H.; Biersack, H.; Bernard, V.; Riecke, A.; Gebauer, J.; Lehnert, H.; von Bubnoff, N.; Feller, A.C.; Gebauer, N. Impact of Treatment Variability and Clinicopathological Characteristics on Survival in Patients with Epstein-Barr-Virus Positive Diffuse Large B Cell Lymphoma. Br. J. Haematol. 2020, 189, 257–268. [Google Scholar] [CrossRef]
- Torne, A.S.; Robertson, E.S. Epigenetic Mechanisms in Latent Epstein-Barr Virus Infection and Associated Cancers. Cancers 2024, 16, 991. [Google Scholar] [CrossRef]
- Pei, Y.; Banerjee, S.; Jha, H.C.; Sun, Z.; Robertson, E.S. An Essential EBV Latent Antigen 3C Binds Bcl6 for Targeted Degradation and Cell Proliferation. PLoS Pathog. 2017, 13, e1006500. [Google Scholar] [CrossRef] [PubMed]
- Souers, A.J.; Leverson, J.D.; Boghaert, E.R.; Ackler, S.L.; Catron, N.D.; Chen, J.; Dayton, B.D.; Ding, H.; Enschede, S.H.; Fairbrother, W.J.; et al. ABT-199, a Potent and Selective BCL-2 Inhibitor, Achieves Antitumor Activity While Sparing Platelets. Nat. Med. 2013, 19, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Price, A.M.; Dai, J.; Bazot, Q.; Patel, L.; Nikitin, P.A.; Djavadian, R.; Winter, P.S.; Salinas, C.A.; Barry, A.P.; Wood, K.C.; et al. Epstein-Barr Virus Ensures B Cell Survival by Uniquely Modulating Apoptosis at Early and Late Times After Infection. eLife 2017, 6, e22509. [Google Scholar] [CrossRef]
- Desbien, A.L.; Kappler, J.W.; Marrack, P. The Epstein-Barr Virus Bcl-2 Homolog, BHRF1, Blocks Apoptosis by Binding to a Limited Amount of Bim. Proc. Natl. Acad. Sci. USA 2009, 106, 5663–5668. [Google Scholar] [CrossRef]
- Procko, E.; Berguig, G.Y.; Shen, B.W.; Song, Y.; Frayo, S.; Convertine, A.J.; Margineantu, D.; Booth, G.; Correia, B.E.; Cheng, Y.; et al. A Computationally Designed Inhibitor of an Epstein-Barr Viral Bcl-2 Protein Induces Apoptosis in Infected Cells. Cell 2014, 157, 1644–1656. [Google Scholar] [CrossRef]
- Bayraktar, U.D.; Diaz, L.A.; Ashlock, B.; Toomey, N.; Cabral, L.; Bayraktar, S.; Pereira, D.; Dittmer, D.P.; Ramos, J.C. Zidovudine-based lytic-inducing chemotherapy for Epstein–Barr virus-related lymphomas. Leuk. Lymphoma 2014, 55, 786–794. [Google Scholar] [CrossRef]
- Roychowdhury, S.; Peng, R.; Baiocchi, R.A.; Bhatt, D.; Vourganti, S.; Grecula, J.; Gupta, N.; Eisenbeis, C.F.; Nuovo, G.J.; Yang, W.; et al. Experimental Treatment of Epstein-Barr Virus-Associated Primary Central Nervous System Lymphoma. Cancer Res. 2003, 63, 965–971. [Google Scholar] [PubMed]
- Slobod, K.S.; Taylor, G.H.; Sandlund, J.T.; Furth, P.; Helton, K.J.; Sixbey, J.W. Epstein-Barr Virus-Targeted Therapy for AIDS-Related Primary Lymphoma of the Central Nervous System. Lancet Lond. Engl. 2000, 356, 1493–1494. [Google Scholar] [CrossRef]
- Paton, N.I.; Musaazi, J.; Kityo, C.; Walimbwa, S.; Hoppe, A.; Balyegisawa, A.; Kaimal, A.; Mirembe, G.; Tukamushabe, P.; Ategeka, G.; et al. Dolutegravir or Darunavir in Combination with Zidovudine or Tenofovir to Treat HIV. N. Engl. J. Med. 2021, 385, 330–341. [Google Scholar] [CrossRef]
- Ganguly, S.; Kuravi, S.; Alleboina, S.; Mudduluru, G.; Jensen, R.A.; McGuirk, J.P.; Balusu, R. Targeted Therapy for EBV-Associated B-Cell Neoplasms. Mol. Cancer Res. MCR 2019, 17, 839–844. [Google Scholar] [CrossRef]
- Cen, O.; Kannan, K.; Huck Sappal, J.; Yu, J.; Zhang, M.; Arikan, M.; Ucur, A.; Ustek, D.; Cen, Y.; Gordon, L.; et al. Spleen Tyrosine Kinase Inhibitor TAK-659 Prevents Splenomegaly and Tumor Development in a Murine Model of Epstein-Barr Virus-Associated Lymphoma. mSphere 2018, 3, e00378-18. [Google Scholar] [CrossRef]
- Lu, J.; Lin, W.-H.; Chen, S.-Y.; Longnecker, R.; Tsai, S.-C.; Chen, C.-L.; Tsai, C.-H. Syk Tyrosine Kinase Mediates Epstein-Barr Virus Latent Membrane Protein 2A-Induced Cell Migration in Epithelial Cells. J. Biol. Chem. 2006, 281, 8806–8814. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.-K.; Liu, S.-T. Activation of the BRLF1 Promoter and Lytic Cycle of Epstein–Barr Virus by Histone Acetylation. Nucleic Acids Res. 2000, 28, 3918–3925. [Google Scholar] [CrossRef] [PubMed]
- zur Hausen, H.; O’Neill, F.J.; Freese, U.K.; Hecker, E. Persisting Oncogenic Herpesvirus Induced by the Tumour Promotor TPA. Nature 1978, 272, 373–375. [Google Scholar] [CrossRef]
- Westphal, E.M.; Blackstock, W.; Feng, W.; Israel, B.; Kenney, S.C. Activation of Lytic Epstein-Barr Virus (EBV) Infection by Radiation and Sodium Butyrate in Vitro and in Vivo: A Potential Method for Treating EBV-Positive Malignancies. Cancer Res. 2000, 60, 5781–5788. [Google Scholar] [PubMed]
- Luka, J.; Kallin, B.; Klein, G. Induction of the Epstein-Barr Virus (EBV) Cycle in Latently Infected Cells by n-Butyrate. Virology 1979, 94, 228–231. [Google Scholar] [CrossRef]
- Tovey, M.G.; Lenoir, G.; Begon-Lours, J. Activation of Latent Epstein-Barr Virus by Antibody to Human IgM. Nature 1978, 276, 270–272. [Google Scholar] [CrossRef]
- Shimizu, N.; Takada, K. Analysis of the BZLF1 Promoter of Epstein-Barr Virus: Identification of an Anti-Immunoglobulin Response Sequence. J. Virol. 1993, 67, 3240–3245. [Google Scholar] [CrossRef]
- Perrine, S.P.; Hermine, O.; Small, T.; Suarez, F.; O’Reilly, R.; Boulad, F.; Fingeroth, J.; Askin, M.; Levy, A.; Mentzer, S.J.; et al. A Phase 1/2 Trial of Arginine Butyrate and Ganciclovir in Patients with Epstein-Barr Virus-Associated Lymphoid Malignancies. Blood 2007, 109, 2571–2578. [Google Scholar] [CrossRef]
- Faller, D.V.; Mentzer, S.J.; Perrine, S.P. Induction of the Epstein-Barr Virus Thymidine Kinase Gene with Concomitant Nucleoside Antivirals as a Therapeutic Strategy for Epstein-Barr Virus-Associated Malignancies. Curr. Opin. Oncol. 2001, 13, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Hagemeier, S.R.; Fingeroth, J.D.; Gershburg, E.; Pagano, J.S.; Kenney, S.C. The Epstein-Barr Virus (EBV)-Encoded Protein Kinase, EBV-PK, but Not the Thymidine Kinase (EBV-TK), Is Required for Ganciclovir and Acyclovir Inhibition of Lytic Viral Production. J. Virol. 2010, 84, 4534–4542. [Google Scholar] [CrossRef]
- Jiang, Z.; Li, W.; Hu, X.; Zhang, Q.; Sun, T.; Cui, S.; Wang, S.; Ouyang, Q.; Yin, Y.; Geng, C.; et al. Tucidinostat plus Exemestane for Postmenopausal Patients with Advanced, Hormone Receptor-Positive Breast Cancer (ACE): A Randomised, Double-Blind, Placebo-Controlled, Phase 3 Trial. Lancet Oncol. 2019, 20, 806–815. [Google Scholar] [CrossRef]
- Eckschlager, T.; Plch, J.; Stiborova, M.; Hrabeta, J. Histone Deacetylase Inhibitors as Anticancer Drugs. Int. J. Mol. Sci. 2017, 18, 1414. [Google Scholar] [CrossRef]
- Xu, L.; Zhang, M.; Tu, D.; Lu, Z.; Lu, T.; Ma, D.; Zhou, Y.; Zhang, S.; Ma, Y.; Yan, D.; et al. Chidamide Induces Epstein-Barr Virus (EBV) Lytic Infection and Acts Synergistically with Tenofovir to Eliminate EBV-Positive Burkitt Lymphoma. J. Pharmacol. Exp. Ther. 2023, 387, 288–298. [Google Scholar] [CrossRef]
- Hui, K.F.; Cheung, A.K.L.; Choi, C.K.; Yeung, P.L.; Middeldorp, J.M.; Lung, M.L.; Tsao, S.W.; Chiang, A.K.S. Inhibition of Class I Histone Deacetylases by Romidepsin Potently Induces Epstein-Barr Virus Lytic Cycle and Mediates Enhanced Cell Death with Ganciclovir. Int. J. Cancer 2016, 138, 125–136. [Google Scholar] [CrossRef]
- Lai, J.; Tan, W.J.; Too, C.T.; Choo, J.A.L.; Wong, L.H.; Mustafa, F.B.; Srinivasan, N.; Lim, A.P.C.; Zhong, Y.; Gascoigne, N.R.J.; et al. Targeting Epstein-Barr Virus-Transformed B Lymphoblastoid Cells Using Antibodies with T-Cell Receptor-like Specificities. Blood 2016, 128, 1396–1407. [Google Scholar] [CrossRef][Green Version]
- Lai, J.; Choo, J.A.L.; Tan, W.J.; Too, C.T.; Oo, M.Z.; Suter, M.A.; Mustafa, F.B.; Srinivasan, N.; Chan, C.E.Z.; Lim, A.G.X.; et al. TCR-like Antibodies Mediate Complement and Antibody-Dependent Cellular Cytotoxicity against Epstein-Barr Virus-Transformed B Lymphoblastoid Cells Expressing Different HLA-A*02 Microvariants. Sci. Rep. 2017, 7, 9923. [Google Scholar] [CrossRef] [PubMed]
- Bu, W.; Kumar, A.; Board, N.L.; Kim, J.; Dowdell, K.; Zhang, S.; Lei, Y.; Hostal, A.; Krogmann, T.; Wang, Y.; et al. Epstein-Barr Virus Gp42 Antibodies Reveal Sites of Vulnerability for Receptor Binding and Fusion to B Cells. Immunity 2024, 57, 559–573.e6. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Zhong, L.; Wei, D.; Zhang, W.; Hong, J.; Kang, Y.; Chen, K.; Huang, Y.; Zheng, Q.; Xu, M.; et al. Neutralizing Antibodies against EBV Gp42 Show Potent in Vivo Protection and Define Novel Epitopes. Emerg. Microbes Infect. 2023, 12, 2245920. [Google Scholar] [CrossRef]
- Chen, W.-H.; Kim, J.; Bu, W.; Board, N.L.; Tsybovsky, Y.; Wang, Y.; Hostal, A.; Andrews, S.F.; Gillespie, R.A.; Choe, M.; et al. Epstein-Barr Virus gH/gL Has Multiple Sites of Vulnerability for Virus Neutralization and Fusion Inhibition. Immunity 2022, 55, 2135–2148.e6. [Google Scholar] [CrossRef]
- Snijder, J.; Ortego, M.S.; Weidle, C.; Stuart, A.B.; Gray, M.D.; McElrath, M.J.; Pancera, M.; Veesler, D.; McGuire, A.T. An Antibody Targeting the Fusion Machinery Neutralizes Dual-Tropic Infection and Defines a Site of Vulnerability on Epstein-Barr Virus. Immunity 2018, 48, 799–811.e9. [Google Scholar] [CrossRef]
- He, H.; Lei, F.; Huang, L.; Wang, K.; Yang, Y.; Chen, L.; Peng, Y.; Liang, Y.; Tan, H.; Wu, X.; et al. Immunotherapy of Epstein-Barr Virus (EBV) Infection and EBV-Associated Hematological Diseases with Gp350/CD89-Targeted Bispecific Antibody. Biomed. Pharmacother. 2023, 163, 114797. [Google Scholar] [CrossRef]
- Osborne, A.J.; Atkins, H.M.; Balogh, K.K.; Brendle, S.A.; Shearer, D.A.; Hu, J.; Sample, C.E.; Christensen, N.D. Antibody-Mediated Immune Subset Depletion Modulates the Immune Response in a Rabbit (Oryctolagus cuniculus) Model of Epstein-Barr Virus Infection. Comp. Med. 2020, 70, 312–322. [Google Scholar] [CrossRef]
- Reguraman, N.; Hassani, A.; Philip, P.S.; Pich, D.; Hammerschmidt, W.; Khan, G. Assessing the Efficacy of VLP-Based Vaccine against Epstein-Barr Virus Using a Rabbit Model. Vaccines 2023, 11, 540. [Google Scholar] [CrossRef]
- Reguraman, N.; Hassani, A.; Philip, P.; Khan, G. Uncovering Early Events in Primary Epstein-Barr Virus Infection Using a Rabbit Model. Sci. Rep. 2021, 11, 21220. [Google Scholar] [CrossRef] [PubMed]
- Shi, N.; Chen, H.; Lai, Y.; Luo, Z.; Huang, Z.; He, G.; Yi, X.; Xia, W.; Tang, A. Cyclosporine A Induces Epstein-Barr Virus Reactivation in Tree Shrew (Tupaia belangeri chinensis) Model. Microbes Infect. 2023, 25, 105212. [Google Scholar] [CrossRef]
- Xia, W.; Chen, H.; Feng, Y.; Shi, N.; Huang, Z.; Feng, Q.; Jiang, X.; He, G.; Xie, M.; Lai, Y.; et al. Tree Shrew Is a Suitable Animal Model for the Study of Epstein Barr Virus. Front. Immunol. 2021, 12, 789604. [Google Scholar] [CrossRef] [PubMed]
- Wirtz, T.; Weber, T.; Kracker, S.; Sommermann, T.; Rajewsky, K.; Yasuda, T. Mouse Model for Acute Epstein–Barr Virus Infection. Proc. Natl. Acad. Sci. USA 2016, 113, 13821–13826. [Google Scholar] [CrossRef] [PubMed]
- Rambold, U.; Sperling, S.; Chew, Z.; Wang, Y.; Steer, B.; Zeller, K.; Strobl, L.J.; Zimber-Strobl, U.; Adler, H. A Mouse Model to Study the Pathogenesis of γ-Herpesviral Infections in Germinal Center B Cells. Cells 2023, 12, 2780. [Google Scholar] [CrossRef]
- Huang, S.; Yasuda, T. Pathologically Relevant Mouse Models for Epstein–Barr Virus–Associated B Cell Lymphoma. Front. Immunol. 2021, 12, 639844. [Google Scholar] [CrossRef]
- Bryan, L.J.; Casulo, C.; Allen, P.B.; Smith, S.E.; Savas, H.; Dillehay, G.L.; Karmali, R.; Pro, B.; Kane, K.L.; Bazzi, L.A.; et al. Pembrolizumab Added to Ifosfamide, Carboplatin, and Etoposide Chemotherapy for Relapsed or Refractory Classic Hodgkin Lymphoma: A Multi-Institutional Phase 2 Investigator-Initiated Nonrandomized Clinical Trial. JAMA Oncol. 2023, 9, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Armand, P.; Engert, A.; Younes, A.; Fanale, M.; Santoro, A.; Zinzani, P.L.; Timmerman, J.M.; Collins, G.P.; Ramchandren, R.; Cohen, J.B.; et al. Nivolumab for Relapsed/Refractory Classic Hodgkin Lymphoma After Failure of Autologous Hematopoietic Cell Transplantation: Extended Follow-Up of the Multicohort Single-Arm Phase II CheckMate 205 Trial. J. Clin. Oncol. 2018, 36, 1428–1439, Erratum in J. Clin. Oncol. 2018, 36, 2748. [Google Scholar] [CrossRef]
- Chen, R.; Zinzani, P.L.; Lee, H.J.; Armand, P.; Johnson, N.A.; Brice, P.; Radford, J.; Ribrag, V.; Molin, D.; Vassilakopoulos, T.P.; et al. Pembrolizumab in Relapsed or Refractory Hodgkin Lymphoma: 2-Year Follow-Up of KEYNOTE-087. Blood 2019, 134, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Hermans, J.; Krol, A.D.; van Groningen, K.; Kluin, P.M.; Kluin-Nelemans, J.C.; Kramer, M.H.; Noordijk, E.M.; Ong, F.; Wijermans, P.W. International Prognostic Index for Aggressive Non-Hodgkin’s Lymphoma Is Valid for All Malignancy Grades. Blood 1995, 86, 1460–1463. [Google Scholar] [CrossRef] [PubMed]
- Aanaes, K.; Kristensen, E.; Ralfkiaer, E.M.; von Buchwald, C.; Specht, L. Improved Prognosis for Localized Malignant Lymphomas of the Head and Neck. Acta Otolaryngol. 2010, 130, 626–631. [Google Scholar] [CrossRef]
- Kwak, Y.-K.; Choi, B.-O.; Kim, S.H.; Lee, J.H.; Kang, D.G.; Lee, J.H. Treatment Outcome of Diffuse Large B-Cell Lymphoma Involving the Head and Neck: Two-Institutional Study for the Significance of Radiotherapy after R-CHOP Chemotherapy. Medicine 2017, 96, e7268. [Google Scholar] [CrossRef]
- Lv, J.; Jiang, Y.; Yu, T.; Gao, S.; Yin, W. Clinical Characteristics and Prognostic Analysis of Primary Extranodal Non-Hodgkin Lymphoma of the Head and Neck. Aging 2024, 16, 6796–6808. [Google Scholar] [CrossRef]
- Zhou, C.; Duan, X.; Lan, B.; Liao, J.; Shen, J. Prognostic CT and MR Imaging Features in Patients with Untreated Extranodal Non-Hodgkin Lymphoma of the Head and Neck Region. Eur. Radiol. 2015, 25, 3035–3042. [Google Scholar] [CrossRef]
- Scully, C. Oral Cancer: New Insights into Pathogenesis. Dent. Update 1993, 20, 95–100. [Google Scholar]
- Scully, C. Oncogenes, Tumor Suppressors and Viruses in Oral Squamous Carcinoma. J. Oral Pathol. Med. 1993, 22, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Feigal, E.G. AIDS-Associated Malignancies: Research Perspectives. Biochim. Biophys. Acta (BBA)-Rev. Cancer 1999, 1423, C1–C9. [Google Scholar] [CrossRef]
- Goldenberg, D.; Golz, A.; Netzer, A.; Rosenblatt, E.; Rachmiel, A.; Goldenberg, R.F.; Joachims, H.Z. Epstein-Barr virus and cancers of the head and neck. Am. J. Otolaryngol. 2001, 22, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Tvedten, E.; Richardson, J.; Motaparthi, K. What Effect Does Epstein-Barr Virus Have on Extranodal Natural Killer/T-Cell Lymphoma Prognosis? A Review of 153 Reported Cases. Cureus 2021, 13, e17987. [Google Scholar] [CrossRef]
- Hu, J.; Zhang, X.; Tao, H.; Jia, Y. The prognostic value of Epstein-Barr virus infection in Hodgkin lymphoma: A systematic review and meta-analysis. Front Oncol. 2022, 12, 1034398. [Google Scholar] [CrossRef]
- Bower, M.; Gazzard, B.; Mandalia, S.; Newsom-Davis, T.; Thirlwell, C.; Dhillon, T.; Young, A.M.; Powles, T.; Gaya, A.; Nelson, M.; et al. A prognostic index for systemic AIDS-related non-Hodgkin lymphoma treated in the era of highly active antiretroviral therapy. Ann. Intern. Med. 2005, 143, 265–273, Erratum in Ann. Intern. Med. 2006, 144, 620. [Google Scholar] [CrossRef] [PubMed]
- Barta, S.K.; Samuel, M.S.; Xue, X.; Wang, D.; Lee, J.Y.; Mounier, N.; Ribera, J.-M.; Spina, M.; Tirelli, U.; Weiss, R.; et al. Changes in the Influence of Lymphoma- and HIV-Specific Factors on Outcomes in AIDS-Related Non-Hodgkin Lymphoma. Ann. Oncol. 2015, 26, 958–966. [Google Scholar] [CrossRef]
- Castillo, J.J.; Bower, M.; Brühlmann, J.; Novak, U.; Furrer, H.; Tanaka, P.Y.; Besson, C.; Montoto, S.; Cwynarski, K.; Abramson, J.S.; et al. Prognostic Factors for Advanced-Stage Human Immunodeficiency Virus-Associated Classical Hodgkin Lymphoma Treated with Doxorubicin, Bleomycin, Vinblastine, and Dacarbazine plus Combined Antiretroviral Therapy: A Multi-Institutional Retrospective Study. Cancer 2015, 121, 423–431. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.