- Review
Dermatomyositis with Anti-MDA5 Autoantibodies After SARS-CoV-2 mRNA Vaccination Treated with Tofacitinib: Integrating Literature Evidence and a Novel Observation
- Maurizio Benucci,
- Elisa Cioffi and
- Mariangela Manfredi
- + 7 authors
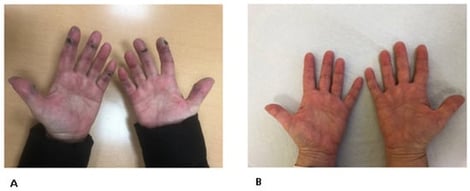
COVID-19 mRNA vaccines activate type I interferon pathways and in genetically or immunologically predisposed individuals may trigger autoimmune responses, including autoantibodies against melanoma differentiation-associated protein 5 (MDA5). Although cases of dermatomyositis (DM), particularly anti-MDA5-positive DM, have been increasingly reported after SARS-CoV-2 vaccination, its clinical spectrum and management remain incompletely defined. We conducted a narrative review of the literature on post-vaccination dermatomyositis, focusing on clinical features, autoantibody profiles, therapeutic approaches, and outcomes. The review was enriched by the inclusion of a new case: a 60-year-old woman who developed anti-MDA5-positive dermatomyositis two weeks after receiving her fourth dose of the BNT162b2 (Pfizer/BioNTech) vaccine. She presented predominantly with cutaneous and articular manifestations in the absence of interstitial lung disease. Treatment with oral prednisone, intravenous alprostadil, and the Janus kinase inhibitor tofacitinib resulted in marked clinical improvement. This case, together with the literature review, illustrates both typical and atypical presentations of vaccine-associated anti-MDA5 DM, highlights diagnostic challenges without lung involvement, and suggests JAK inhibition as a potential therapeutic option, contributing to a more comprehensive understanding of post-vaccination dermatomyositis.
9 March 2026