Abstract
Background: Hyponatremia (serum sodium levels below 135 mEq/L) is the most prevalent electrolyte imbalance, with the syndrome of inappropriate antidiuresis (SIAD) being the most common cause among inpatients. Fluid restriction is the primary treatment for SIAD, yet its efficacy is inconsistent. A novel therapeutic approach involves the use of oral vaptans, such as tolvaptan (TLV), which are non-peptide antagonists of arginine vasopressin receptors. The recommended daily dose of TLV is 15 mg; however, the risk of overcorrection and osmotic demyelination syndrome must be considered. Methods: Consequently, a more cautious approach involving a 7.5 mg dose of TLV was studied in SIAD patients to determine its safety and efficacy compared with a 15 mg dose. Results: The findings of our investigation show that the results obtained from the two doses are highly similar. However, it is important to note that the risk of overcorrection was lower in the 7.5 mg TLV group than in the 15 mg group. Furthermore, a more gradual increase in serum Na was observed in the 7.5 mg group than in the 15 mg group after the most critical first 24 h. Conclusions: TLV therapy can be initiated with a 7.5 mg dose, with serum sodium levels monitored at 12 and 24 h to confirm or adjust the TLV dose as required.
1. Introduction
Hyponatremia is defined as a serum sodium (Na) concentration below 135 mEq/L. It is the most common electrolyte imbalance, affecting up to 25–30% of hospitalized patients, and is most frequently caused by the syndrome of inappropriate antidiuresis (SIAD), accounting for almost 35% of cases [1,2]. Classical Schwartz and Barter’s criteria are used for diagnosis [3], and international guidelines propose several algorithms to support the differential diagnosis of hypotonic hyponatremia [4,5]. Furthermore, new markers such as fractional uric acid and potassium excretions are considered useful in challenging differential diagnoses of SIAD in patients treated with diuretics [6]. Although a low serum sodium level is recognized as a risk factor for increased mortality and length of hospital stay, hyponatremia is often underdiagnosed and undertreated. Greenberg et al. found that 17% of hyponatremic inpatients were not treated appropriately and that 78% of patients were still hyponatremic at discharge [7]. Tolvaptan (TLV), a vasopressin V2 receptor antagonist, has been approved in Italy since 2010 for the treatment of hyponatremia due to SIAD. The recommended initial TLV dose is 15 mg, which can be gradually increased up to a maximum of 60 mg per day. TLV therapy has been shown to be as effective as hypertonic saline administration and fluid restriction in increasing serum sodium levels. Real-world data confirmed the efficacy of TLV, reporting a high risk of overly rapid sodium increase, particularly in cases of severe hyponatremia. However, there is currently disagreement between the European Clinical Practice Guidelines and the United States Expert Panel recommendations regarding the treatment of SIAD. This is because European experts advise against the use of TLV for SIAD, highlighting the potential for overcorrection [4,5]. Overcorrection of serum sodium by tolvaptan therapy can induce osmotic demyelination syndrome (ODS), although only one case of ODS has been reported thus far [8]. Thus, previous studies have investigated the efficacy and safety of 7.5 mg of TLV, with controversial results [9]. Lewellyn’s meta-analysis suggests the efficacy and safety of low-dose TLV [10]. Initiating therapy with low-dose TLV (3.75 mg or 7.5 mg) effectively corrects serum sodium levels, reducing the risk of overcorrection and ODS. Our study confirms this evidence in a real-world Italian cohort [10,11,12], while other studies, such as that by Chatzimavridou et al. (39%), have found an increased rate of overcorrection [13].
Many guidelines recommend an optimal increase in serum Na of less than 12 mEq/L within 24 h and less than 18 mEq/L within 48 h in SIAD patients [4,5]. Thus, our study aimed to further evaluate the differences in the efficacy and safety of the recommended 15 mg TLV dose versus (vs.) the 7.5 mg dose once daily in the first 48 h of treatment.
2. Materials and Methods
A retrospective consecutive analysis of case series was performed. Fifty-six patients met the criteria for moderate or severe hyponatremia by SIAD (serum Na < 130 mEq/L, serum osmolality < 270 mOsm/kg, urine osmolality > 100 mOsm/kg, urine sodium > 30 mmol/L, FEUA > 12% [14]. Moderate hyponatremia was characterized by a serum Na value between 129 mEq/L and 125 mEq/L, and severe hyponatremia below 125 mEq/L. The etiologies of SIAD are shown in Table 1.

Table 1.
Likely causes of SIAD in the investigated patients.
Non-eligible diseases and conditions included (1) hypovolemic hyponatremia, psychogenic polydipsia, head trauma, postoperative conditions, uncontrolled hypothyroidism, or adrenal insufficiency; (2) myocardial infarction, sustained ventricular tachycardia or fibrillation, severe angina, cerebrovascular accident, or stroke during the previous three months; (3) systolic blood pressure < 90 mmHg; (4) fasting blood glucose greater than 200 mg/dL; (5) severe liver disease; (6) pregnant or breastfeeding women; (7) use of hypertonic saline; and (8) use of thiazides.
Laboratory parameters were investigated with automated clinical analysis, using ion-selective electrodes (indirect measurement, COBAS 6000 Analyzer Series, Roche, Basel, Switzerland). A blood sample was taken in the morning, contemporary to a urine spot sample, to test Na, uric acid, and creatinine (Cr) levels and then calculate the fractional uric acid excretion (FEUA). Urine and serum osmolality (mOsm/kg) were evaluated with a freezing point depression osmometer (Gonotec, Osmomat 3000 Basic, ELItech Group SPA, Torino, Italy). Patients were treated orally with a starting dose of 7.5 mg (half-dose) or 15 mg (full-dose) of TLV, without fluid restriction. Eventual medications, such as diuretics (usually furosemide), were withdrawn.
On the first day of TLV therapy, serum Na levels were tested at 0, 6, 12, and 24 h.
At 24 h, a serum Na level > 12 mEq/L indicated overcorrection, usually treated with dextrose 5% administration (20 mL/kg) and dose reduction or discontinuation for 24 h. The maximum TLV dose used in our study was 15 mg once daily. After 48 h of TLV therapy, a serum Na < 130 mEq/L indicated the need for continuing therapy. No cases of ODS were observed.
At 24 h, we identified four different categories of correction of serum Na levels, such as:
(A) low degree of correction: <4 mEq/L/24 h; (B), optimal: 4–8 mEq/L/24 h; (C), low risk of overcorrection: 9–12 mEq/L/24 h; (D), severe: >12 mEq/L/24 h [5].
We also analyzed hospital length of stay and the need to continue TLV therapy after discharge.
Statistical Analysis
Continuous data were analyzed using the Mann-Whitney U test or an unpaired 2-tailed t-test, while categorical data were analyzed using the two-tailed Fisher’s exact test. Statistical significance was considered at p < 0.05.
3. Results
Table 2 shows the baseline characteristics of the studied groups and the time-dependent variations of serum sodium levels after TLV administration.

Table 2.
Baseline characteristics of the patients studied and time-dependent serum Na response to TLV therapy (7.5 mg vs. 15 mg).
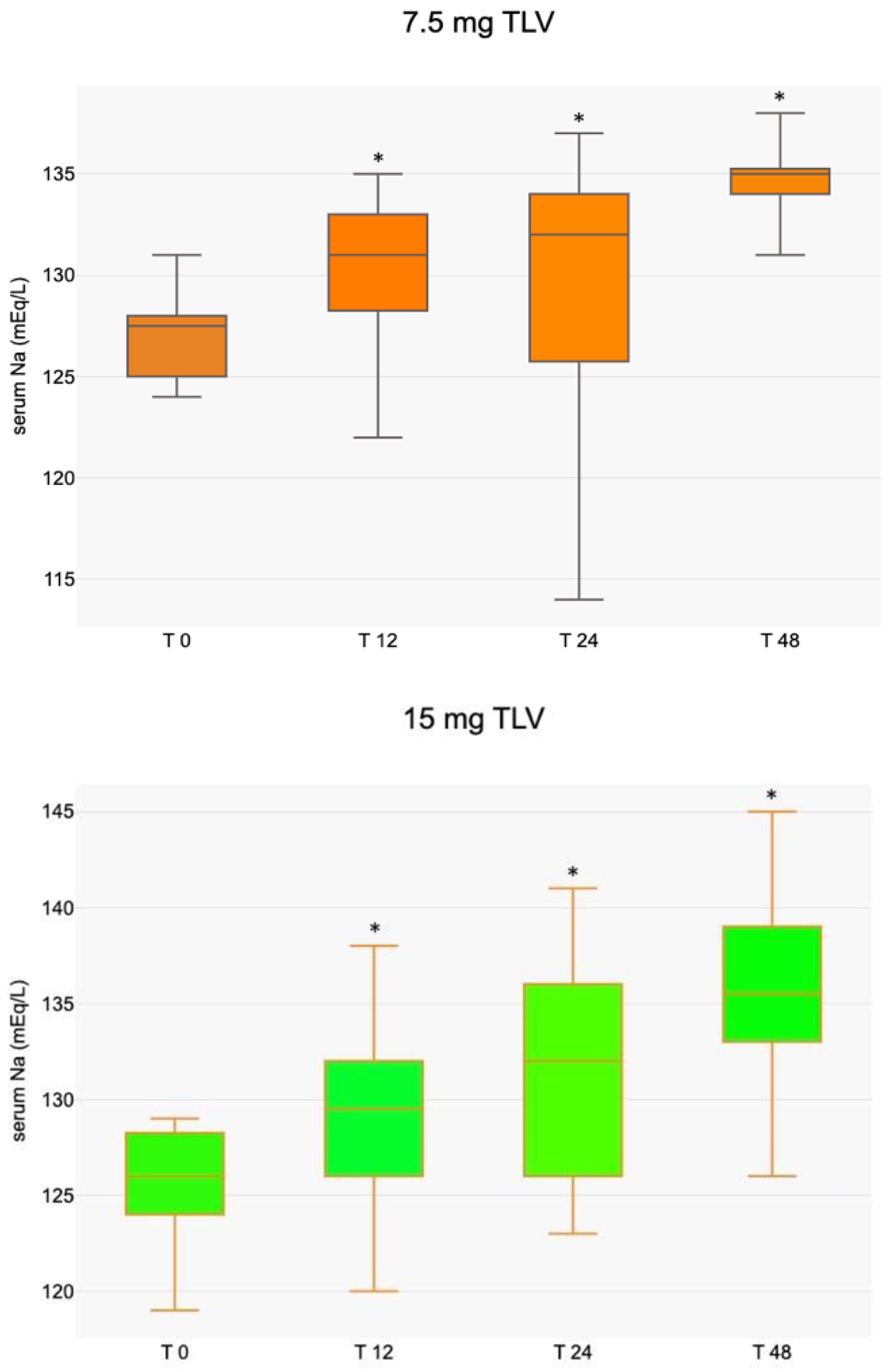
The only significant differences between the groups were in urine osmolality and Na levels. One possible cause is the increased use of furosemide in the 15 mg TLV group (28 mg/d vs. 20 mg/d and 5 patients vs. 4 patients in the 15 mg and 7.5 mg TLV groups, respectively). The time-dependent increase in serum Na induced by TLV was similar between the 7.5 mg and 15 mg treatments, and the maximum effect of TLV was recorded within 12/24 h of the first TLV dose (Figure 1).
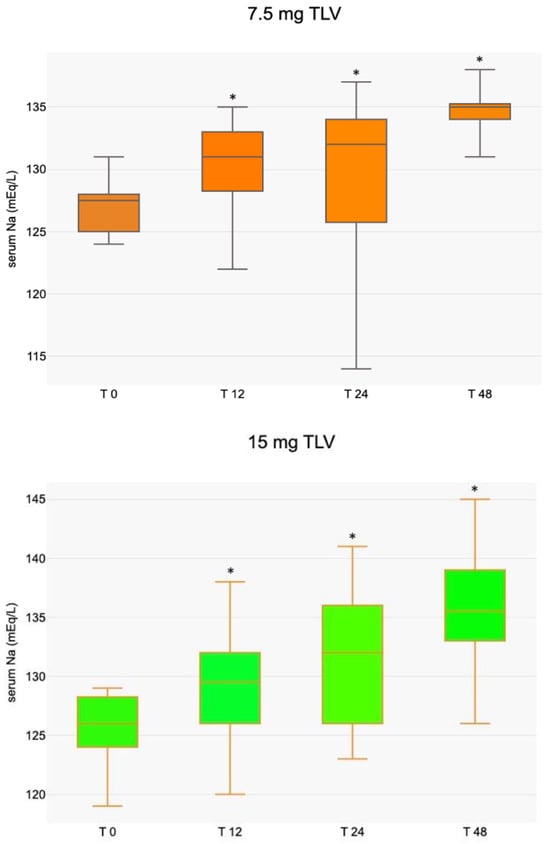
Figure 1.
Median (with interquartile range) serum Na increase following TLV dose at different times (* p < 0.05 compared to T 0).
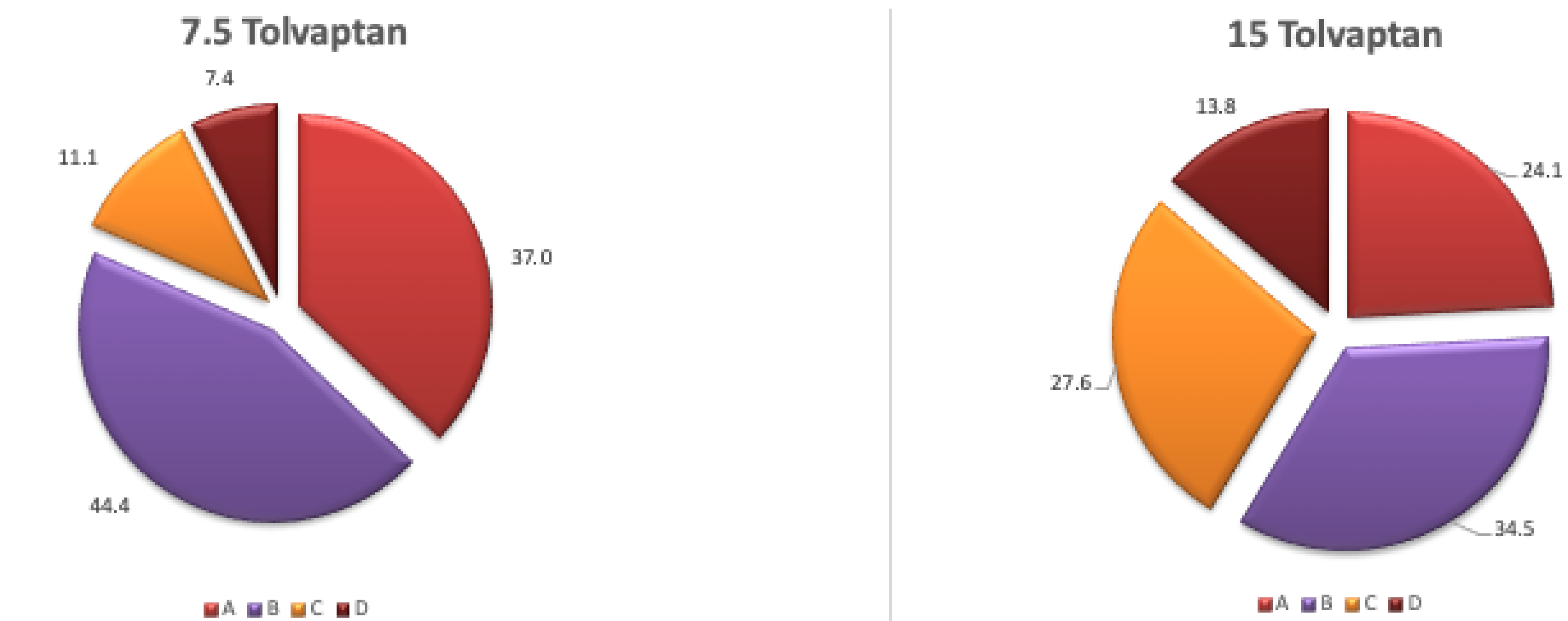
After 24 h, a more gradual increase in serum sodium (Na) is observed in patients treated with a 7.5 mg dose of TLV, with a lower rate of increase compared to those treated with a 15 mg dose (categories A + B%, 81.4% vs. 58.6%, respectively). Conversely, the higher risk categories resulted in a decrease in the 7.5 mg group (C + D%, 18.5% vs. 41.4%). Severe overcorrection (>12 mEq/L) occurred in 7.4% and 13.8% of patients treated with 7.5 mg and 15 mg of TLV, respectively (see Figure 2).
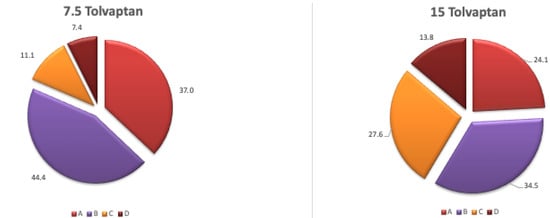
Figure 2.
Increase in serum sodium (%) at 24 h with different doses of TLV: A: <4 mEq/L, B: 4–8 mEq/L, C: 9–12 mEq/L, D: >12 mEq/L.
No cases of overcorrection were observed at 48 h at both TLV doses. The need to increase the dose to 15 mg was observed in only one patient who was initially treated with 7.5 mg of TLV. Patients treated with 7.5 mg experienced a slightly longer hospital stay than those treated with 15 mg (8.2 vs. 6.3 days). Following discharge, additional TLV treatment was required for persistent hyponatremia in 21 patients treated with 7.5 mg versus 18 patients treated with 15 mg.
4. Discussion
Several studies have demonstrated the efficacy of TLV in correcting hyponatremia. However, as with all treatment options for SIAD, its clinical efficacy is questionable. The EVEREST (Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study With TLV) study found that TLV was effective in increasing serum sodium, reducing body weight, and improving dyspnea in hospitalized heart failure patients. However, no effect was demonstrated on long-term mortality, cardiovascular morbidity, or rehospitalization rates [15,16].
In Italy, TLV is used exclusively to treat SIAD. The recommended starting dose is 15 mg once daily, leading to a significant correction in the patient’s serum sodium concentration [4]. However, the high therapeutic potency of TLV is associated with an increased likelihood of ODS due to an inappropriate increase in serum sodium. Thus, the risk of overcorrection induced by the full dose has led to suggestions to test a reduced dose of TLV. Studies investigating the efficacy and safety of a 7.5 mg dose of TLV have yielded conflicting results regarding the risk of sodium overcorrection, ranging from 0% to 30.9% [9,10,11,12,13]. Our study confirms the efficacy of both TLV therapy doses, with similar increases in serum sodium at 24 and 48 h in patients treated with 7.5 mg and 15 mg of TLV (5 vs. 7 mEq/L at 24 h and 8 vs. 10 mEq/L at 48 h, respectively).
In terms of safety, previous studies have shown overcorrection occurring at 24 h in patients treated with 15 mg of TLV, with values ranging from 5.9% to 25% [17,18]. Moreover, the magnitude of TLV-induced Na increase is higher the lower the basal serum level. Our data showed an overcorrection comparison (13.8%) with 15 mg of TLV close to the previous reported values, while in our investigation, the 7.5 mg group, also including several cases of severe hyponatremia vs. the 15 mg group (59.3% vs. 35%, respectively), demonstrated a reduced risk (7.4%; see Figure 1).
The most consistent increase in serum Na was observed within the first 12/24 h, in line with other studies [19,20]. Furthermore, a second dose of TLV (whether 7.5 mg or 15 mg) was much less likely to lead to overcorrection than the initial dose. Thus, we recommend strict control of serum Na levels during this period, whereas afterwards, control at 24 and 48 h appears to be satisfactory.
Moreover, the difference in length of hospital stay (8.6 vs. 6.3 days) could be misleading, as it could suggest that the full dose is too strong, inducing a higher incidence of overcorrection and increasing the risk of ODS in patients. Interestingly, no cases of ODS were recorded in our patients with serum Na overcorrection, in line with the findings of several studies [9]. The increased use of furosemide in the 15 mg TLV group may be the cause of the significant differences in urine osmolality and Na levels between the groups.
However, the necessity of dividing 15 mg tablets to administer a 7.5 mg dose may result in limitations, such as an imprecise dose. The future availability of pre-constituted 7.5 mg doses in Italy provides a valuable opportunity to validate the results obtained in this study. A lower starting dose of 3.75 mg has been anecdotally used with good efficacy results, but the available data from only 10 patients are insufficient to support its therapeutic use [21].
In conclusion, our investigation shows that the half-dose of 7.5 mg is as effective as the full dose of 15 mg of TLV in treating hyponatremia dependent on SIAD. Then, given the heterogeneity of patients presenting with severe hyponatremia and the associated increased risk of ODS, we recommend using the half-dose of TLV as it is safer than the full dose.
Furthermore, the present study grouped all SIAD patients together in a single cohort. However, it is not possible to exclude the possibility that different causes of SIAD may affect the response to TLV. In consideration of the relatively small number of patients enrolled, the retrospective nature of the study, and the lack of randomization, further investigations are needed to confirm these results.
Author Contributions
Conceptualization, data collection, writing—original draft preparation, V.B.; Drafting, bibliography, and statistical analysis; V.A., C.S. and O.F. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Ethical review and approval were waived for this study due to the retrospective, observational nature of the study.
Informed Consent Statement
Written informed consent was obtained from all the patients before participation.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Upadhyay, A.; Jaber, B.L.; Madias, N.E. Incidence and prevalence of hyponatremia. Am. J. Med. 2006, 119, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Verbalis, J.G. The syndrome of inappropriate antidiuretic hormone secretion and other hyposmolar disorders. In Diseases of the Kidney and Urinary Tract, 8th ed.; Schrier, R.W., Ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006; pp. 2511–2548. [Google Scholar]
- Schwartz, W.B.; Bennett, W.; Curelop, S.; Bartter, F.C. A syndrome of renal sodium loss and hyponatremia probably resulting from inappropriate secretion of antidiuretic hormone. Am. J. Med. 1957, 23, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Spasovski, G.; Vanholder, R.; Allolio, B.; Annane, D.; Ball, S.; Bichet, D.; Decaux, G.; Fenske, W.; Hoorn, E.J.; Ichai, C.; et al. Clinical practice guideline on diagnosis and treatment of hyponatraemia. Nephrol. Dial. Transplant. 2014, 29, 1–39. [Google Scholar] [CrossRef] [PubMed]
- Verbalis, J.G.; Goldsmith, S.R.; Greenberg, G.; Korzelius, C.; Schrier, R.W.; Sterns, R.H.; Thompson, C.J. Diagnosis, Evaluation, and Treatment of Hyponatremia: Expert Panel Recommendations. Am. J. Med. 2013, 126, S1–S42. [Google Scholar] [CrossRef] [PubMed]
- Bassi, V.; Fattoruso, O. The Combined Use of Fractional Urate and Potassium Excretion in the Diagnosis of Diuretic-Induced Hyponatremia. Cureus 2021, 13, e15308. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, A.; Verbalis, J.G.; Amin, A.N.; Burst, V.R.; Chiodo, J.A., 3rd; Chiong, J.R.; Dasta, J.F.; Friend, K.E.; Hauptman, P.J.; Peri, A.; et al. Current treatment practice and outcomes. Report of the hyponatremia registry. Kidney Int. 2015, 88, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.H.; Hsu, Y.J.; Chiu, J.S.; Chu, S.J.; Davids, M.R.; Halperin, M.L. Osmotic demyelination syndrome: A potentially avoidable disaster. QJM 2003, 96, 935–947. [Google Scholar] [CrossRef] [PubMed]
- Tzoulis, P.; Kaltsas, G.; Baldeweg, S.E.; Bouloux, P.-M.; Grossma, A.B. Tolvaptan for the treatment of the syndrome of inappropriate antidiuresis (SIAD). Ther. Adv. Endocrinol. Metab. 2023, 14, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Lewellyn, D.; Nuamek, T.; Ostarijas, E.; Logan Ellis, H.; Drakou, E.E.; Aylwin, S.J.B.; Dimitriadis, G.K. Low-Dose Tolvaptan for the Treatment of Syndrome of Inappropriate Antidiuretic Hormone-Associated Hyponatremia: A Systematic Review, Meta-Analysis, and Meta-Regression Analysis of Clinical Effectiveness and Safety. Endocr. Pract. 2025, 31, 956–964. [Google Scholar] [CrossRef] [PubMed]
- Castello, L.; Baldrighi, M.; Panizza, A.; Avanzi, G. Efficacy and safety of two different tolvaptan doses in the treatment of hyponatremia in the Emergency Department. Intern. Emerg. Med. 2017, 12, 993–1001. [Google Scholar] [PubMed]
- Hanna, R.M.; Velez, J.C.; Rastogi, A.; Nguyen, M.K.; Kamgar, M.K.; Moe, K.; Arman, F.; Hasnain, H.; Nobakht, N.; Selamet, U.; et al. Equivalent Efficacy and Decreased Rate of Overcorrection in Patients With Syndrome of Inappropriate Secretion of Antidiuretic Hormone Given Very Low-Dose Tolvaptan. Kidney Med. 2019, 2, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Chatzimavridou-Grigoriadou, V.; Al-Othman, S.; Brabant, G.; Kyriacou, A.; King, J.; Blackhall, F.; Trainer, P.J.; Higham, C.E. Clinical experience of the efficacy and safety of low-dose tolvaptan therapy in a UK tertiary oncology setting. J. Clin. Endocrinol. Metab. 2021, 106, 4766–4775. [Google Scholar] [CrossRef] [PubMed]
- Bassi, V.; Fattoruso, O. The role of fractional excretion of uric acid in the differential diagnosis of hypotonic hyponatraemia in patients with diuretic therapy. Cureus 2020, 12, e7762. [Google Scholar] [CrossRef] [PubMed]
- Konstam, M.A.; Gheorghiade, M.; Burnett, J.C., Jr.; Grinfeld, L.; Maggioni, A.P.; Swedberg, K.; Udelson, J.E.; Zannad, F.; Cook, T.; Ouyang, J.; et al. Effects of oral tolvaptan in patients hospitalized for worsening heart failure: The EVEREST Outcome Trial. JAMA 2007, 297, 1319–1331. [Google Scholar] [CrossRef] [PubMed]
- Gheorghiade, M.; Konstam, M.A.; Burnett, J.C., Jr.; Grinfeld, L.; Maggioni, A.P.; Swedberg, K.; Udelson, J.E.; Zannad, F.; Cook, T.; Ouyang, J.; et al. Short-term clinical effects of tolvaptan, an oral vasopressin antagonist, in patients hospitalized for heart failure: The EVEREST Clinical Status Trials. JAMA 2007, 297, 1332–1343. [Google Scholar] [CrossRef] [PubMed]
- Verbalis, J.G.; Ellison, H.; Hobart, M.; Krasa, H.; Ouyang, J.; Czerwiec, F.S.; Investigation of the Neurocognitive Impact of Sodium Improvement in Geriatric Hyponatremia: Efficacy and Safety of Tolvaptan (INSIGHT) Investigators. Tolvaptan and neurocognitive function in mild to moderate chronic hyponatremia: A randomized trial (INSIGHT). Am. J. Kidney Dis. 2016, 67, 893–901. [Google Scholar] [PubMed]
- Morris, J.H.; Bohm, N.M.; Nemecek, B.D.; Crawford, R.; Kelley, D.; Bhasin, B.; Nietert, P.J.; Velez, J.C.Q. Rapidity of correction of hyponatremia due to syndrome of inappropriate secretion of antidiuretic hormone following tolvaptan. Am. J. Kidney Dis. 2018, 71, 772–782. [Google Scholar] [CrossRef] [PubMed]
- Tzoulis, P.; Waung, J.A.; Bagkeris, E.; Carr, H.; Khoo, B.; Cohen, M.; Bouloux, P.M. Real-life experience of tolvaptan use in the treatment of severe hyponatraemia due to syndrome of inappropriate antidiuretic hormone secretion. Clin. Endocrinol. 2016, 84, 620–626. [Google Scholar] [CrossRef] [PubMed]
- Han, S.W.; Yi, J.H.; Kang, K.P.; Kim, H.Y.; Kim, S.W.; Choi, H.Y.; Ha, S.K.; Kim, G.H.; Kim, Y.W.; Jeong, K.H.; et al. Safety and Efficacy of Tolvaptan in Korean Patients with Hyponatremia Caused by the Syndrome of Inappropriate Antidiuretic Hormone. J. Korean Med. Sci. 2018, 33, e112. [Google Scholar] [CrossRef] [PubMed]
- Shoaf, S.E.; Bricmont, P.; Dandurand, A. Low dose tolvaptan PK/PD: Comparison of patients with hyponatremia due to syndrome of inappropriate antidiuretic hormone secretion to healthy adults. Eur. J. Clin. Pharmacol. 2017, 73, 1399–1408. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.