Abstract
Chronic subdural hematoma (CSDH) in frail older adults is increasingly recognized as a sentinel event, with mortality often driven by medical complications rather than neurosurgical factors. We report a failure-to-rescue case in which rapid postoperative deterioration occurred after burr-hole drainage for bilateral CSDH in a frail older adult with diabetes. A clinical picture consistent with sepsis was suspected, and a gastrointestinal source was considered, but the infectious focus could not be confirmed due to limited diagnostic work-up. On admission, chest-computed tomography showed mild right lower-lobe pneumonia, and incidental transverse colonic dilatation was also visible. Burr-hole drainage was uneventful and oxygenation rapidly normalized on room air. On postoperative day (POD) 3, the patient developed a high fever (39 °C), rising C-reactive protein (CRP; 14 mg/dL), abrupt leukopenia (15,300 → 3300/µL), and, several hours later, profuse watery diarrhea. At that time, an evaluation for an infectious source and escalation of therapy (e.g., blood cultures, serum lactate, and abdominal imaging) were not performed. In the early hours of POD 4, he suffered sudden desaturation, shock, and cardiac arrest, and died despite resuscitation. A portable radiograph after intubation showed no new diffuse pulmonary infiltrates but marked colonic gas distension. This case highlights the need to reassess diagnostic framing when discordant postoperative “red flags” emerge and proposes practical triggers for early sepsis evaluation and escalation—prioritizing early recognition and timely rescue rather than a definitive determination of the cause of death—in high-risk CSDH patients.
1. Introduction
Chronic subdural hematoma (CSDH) is a common neurosurgical condition in older adults, and its incidence increases with age and is expected to rise further in super-aged societies [1,2]. Although traditionally regarded as a “benign” entity with good prognosis, recent epidemiological studies have reported 12-month mortality rates in the range of 15–20%, and even over 30% in some cohorts, positioning CSDH as a sentinel event in frail older adults, comparable to hip fracture [1]. Burr-hole drainage remains the standard treatment and is considered a relatively low-risk procedure with perioperative mortality rates of approximately 3–5%; however, medical complications occur in about 20% of patients, and in-hospital deaths are more often attributable to systemic complications than to neurosurgical factors per se [2,3].
Among these systemic complications, sepsis and septic shock are particularly lethal. Postoperative sepsis is associated not only with increased in-hospital mortality, but also with a sustained excess risk of death for more than one year after discharge compared with non-septic surgical patients [4]. Older adults are especially vulnerable to sepsis due to immunosenescence, multimorbidity, and polypharmacy [5], and they frequently present with atypical features, lacking classic signs such as high fever, tachycardia, or marked leukocytosis, which contributes to diagnostic delay [5]. Intra-abdominal sepsis in the elderly is often characterized by minimal abdominal pain and absent peritoneal signs despite advanced disease and is associated with worse outcomes than in younger patients [6].
Diabetes mellitus is a common comorbidity in patients with CSDH and is closely linked to gastrointestinal (GI) dysmotility via autonomic neuropathy. Diabetic enteropathy can manifest as delayed gastric emptying, chronic constipation alternating with sudden watery diarrhea, small and large bowel dilatation, and even chronic intestinal pseudo-obstruction [7]. In frail older adults with such background GI dysfunction, sepsis originating from subileus, infectious enteritis, or other abdominal pathology may initially be masked by respiratory or neurological symptoms, leading clinicians to focus on the lung or brain while an abdominal assessment is deferred.
The concept of failure-to-rescue (FTR)—death after a treatable postoperative complication—has been proposed as a key quality indicator that better explains differences in surgical mortality between hospitals than complication rates alone [8]. Contributing factors include both system issues (escalation pathways, staffing, and monitoring) and cognitive factors such as anchoring bias, where clinicians remain fixated on an initial diagnosis and fail to broaden the differential when new, discordant data emerge [8,9]. In the context of CSDH surgery, however, there are few detailed case reports that explicitly link the delayed recognition of a possible abdominal source of infection, cognitive bias, and FTR in frail, multimorbid patients.
Here, we report a frail 87-year-old man with diabetes who underwent bilateral burr-hole drainage for CSDH and subsequently died after rapidly progressive shock with a clinical picture compatible with sepsis. In this case, pre-existing transverse colonic dilatation on the admission computed tomography (CT) and later developments of watery diarrhea, leukopenia, and colonic gas distension were potential warning signs of a possible abdominal source; however, abdominal assessment and sepsis work-up were limited while clinical attention remained focused on pneumonia and the expected neurosurgical postoperative course. Through this case, we discuss diagnostic and system factors that may delay the recognition of sepsis in postoperative CSDH patients, the role of anchoring bias in the diagnostic process, and practical implications for preventing failure-to-rescue in frail older adult neurosurgical patients.
2. Case Presentation
An 87-year-old man with known type 2 diabetes mellitus (diet-controlled, no medications) and a baseline of independent ambulation with a walker was brought to our hospital by ambulance because of progressive gait disturbance and poor oral intake. Two months earlier, he had sustained a fall and traumatic subarachnoid hemorrhage, which had been treated conservatively at another hospital. Three days before admission, his gait had worsened, and for two days he had been unable to take sufficient fluids. On the day of admission, he was found unable to get out of bed, and emergency medical services were activated.
On first contact, the emergency medical team documented a Japan Coma Scale (JCS) score of 3, respiratory rate of 18/min, heart rate of 56/min, and peripheral oxygen saturation (SpO2) of 96% on room air. Head CT at the referring hospital showed bilateral chronic subdural hematomas (CSDHs), and he was transferred to our neurosurgical center.
On arrival at our emergency department, his level of consciousness was JCS I-3. Vital signs were blood pressure level of 117/77 mmHg, heart rate of 120/min, respiratory rate of 18/min, body temperature in the 36 °C range, and SpO2 of 80% on room air. Oxygen supplied via a face mask at 6 L/min raised SpO2 to only 85%; therefore, the flow was increased to 8 L/min and then switched to a reservoir mask at 10 L/min. Arterial blood gas analysis on 6 L/min oxygen revealed a pH of 7.427, PaO2 of 51.7 mmHg, PaCO2 of 29.6 mmHg, HCO3− of 19 mmol/L, SaO2 of 88%, and base excess of −4, indicating moderate-to-severe hypoxemia with respiratory alkalosis. Laboratory tests showed plasma glucose at 352 mg/dL, glycated hemoglobin A1c (HbA1c) at 7.9%, white blood cell (WBC) count at 13,700/µL, C-reactive protein (CRP) at 1.2 mg/dL, blood urea nitrogen (BUN) at 22.7 mg/dL, creatinine at 0.68 mg/dL, sodium at 140 mEq/L, potassium at 3.5 mEq/L, and D-dimer at 5.7 µg/mL, and N-terminal pro-B-type natriuretic peptide (NT-proBNP) at 92 pg/mL (Table 1). Twelve-lead electrocardiography (ECG) demonstrated sinus tachycardia with PR shortening, complete right bundle branch block, left axis deviation, ST-T changes in V4, and QT prolongation.

Table 1.
Summary of vital signs and laboratory data from admission to just before cardiopulmonary arrest.
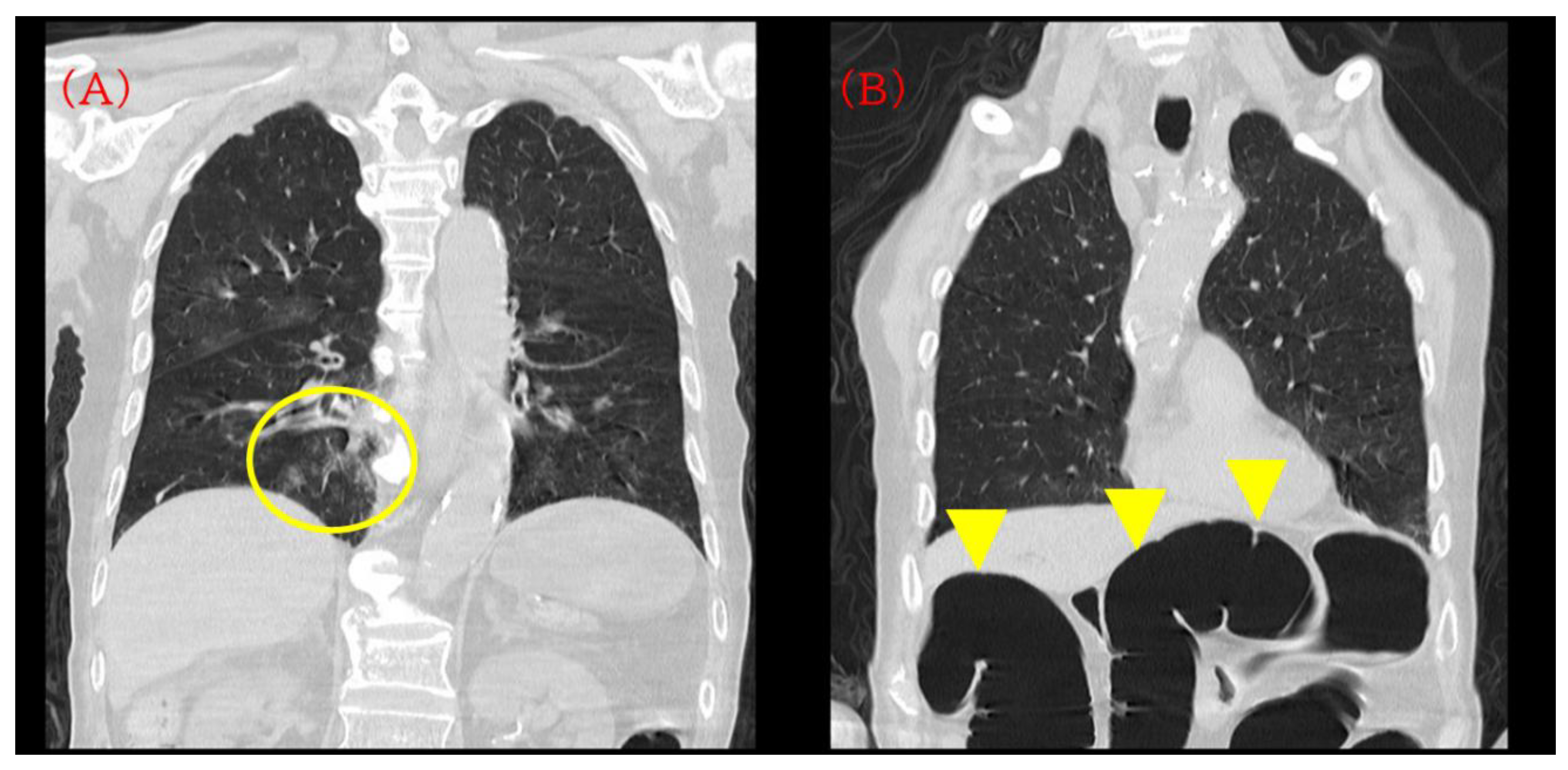
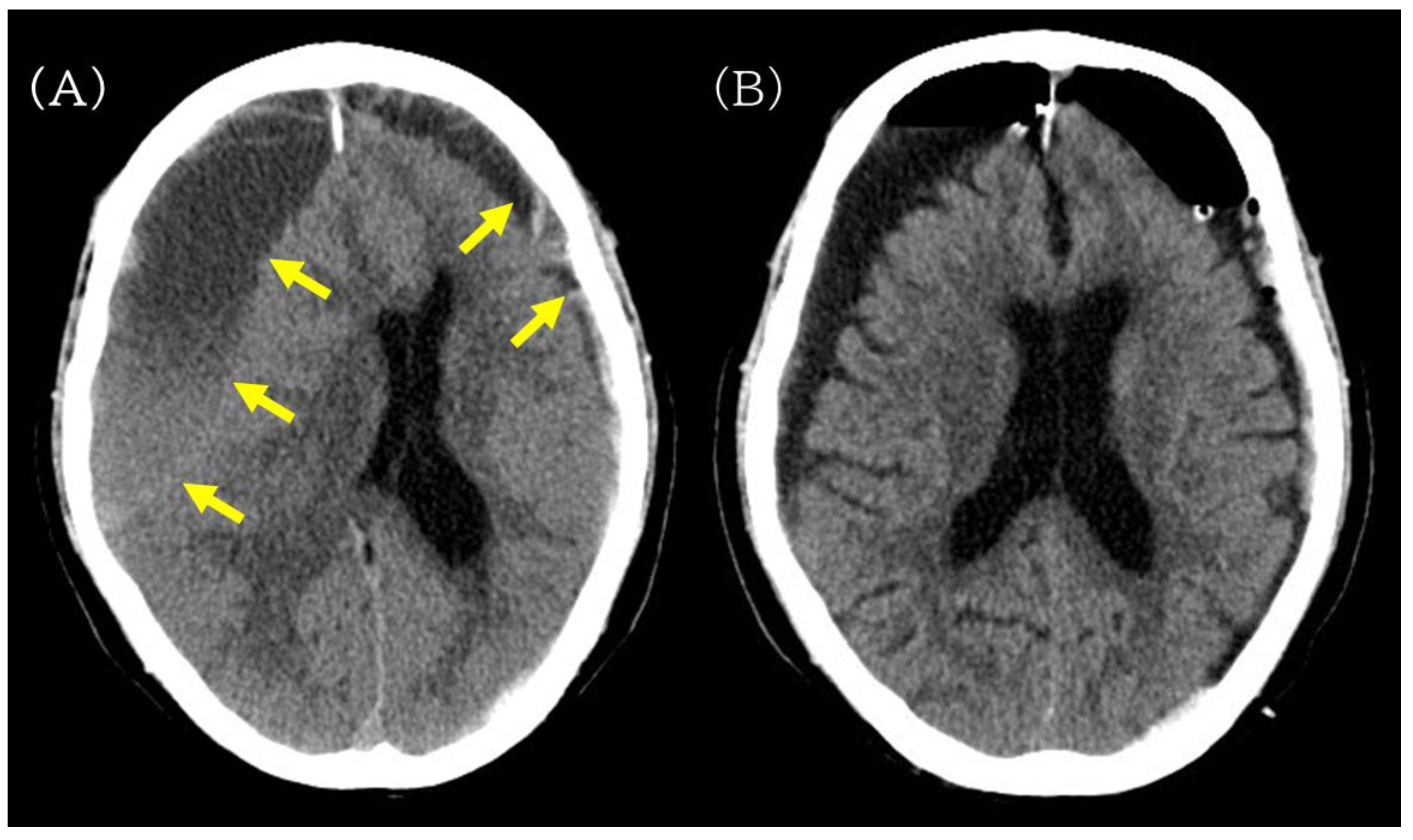
Chest CT showed mild bronchopneumonia predominantly in the right lower lobe without extensive consolidation or large pleural effusions (Figure 1A). On the same CT series, a moderately distended transverse colon with intraluminal gas was incidentally visualized within the scan range, suggesting pre-existing colonic dilatation at admission (Figure 1B). Non-contrast head CT confirmed bilateral CSDHs, more prominent on the right, compressing both cerebral hemispheres (Figure 2A).
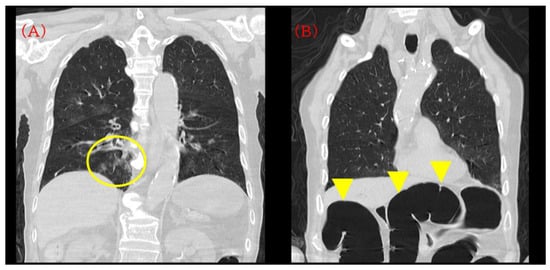
Figure 1.
Initial computed tomography (CT) on admission. (A) Chest CT demonstrating mild bronchopneumonia predominantly in the right lower lobe without extensive consolidation or pleural effusion (yellow circle). (B) The same CT series incidentally showing a moderately distended transverse colon with intraluminal gas (yellow arrowhead). This finding was visible on the admission CT but was not pursued with dedicated abdominal evaluation at that time and was later revisited during case review.
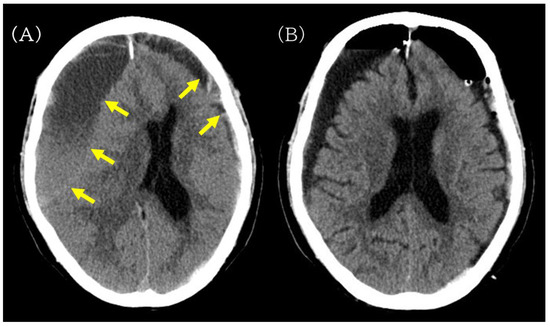
Figure 2.
Preoperative and postoperative non-contrast head CT images illustrating hematoma evacuation after burr-hole drainage. (A) Preoperative non-contrast head CT showing bilateral chronic subdural hematomas (yellow arrow), more prominent on the right, compressing both cerebral hemispheres. (B) Postoperative head CT obtained after burr-hole drainage showing reduced hematoma volume without new intracranial hemorrhage.
Based on these findings, we diagnosed impaired consciousness due to bilateral CSDH with concomitant right lower-lobe pneumonia and admitted the patient to the neurosurgical ward. To manage intracranial pressure and prevent symptomatic seizures, intravenous glycerol (200 mL) and fosphenytoin (750 mg) were administered. A bisoprolol transdermal patch (4 mg) was applied for sinus tachycardia. With oxygen and fluid resuscitation, his SpO2 improved to 96% on 3 L/min oxygen before surgery. Therapeutic antibiotic treatment for pneumonia was intravenous cefazolin (2 g/day on POD 0 and 4 g/day on POD 1–3, then discontinued).
On the day of admission (postoperative day [POD] 0), right-sided burr-hole drainage of the CSDH was performed. The procedure was uneventful, and postoperative head CT confirmed adequate hematoma evacuation without new hemorrhage or acute brain swelling (Figure 2B). Cefazolin was continued perioperatively (2 g/day on POD 0 and 4 g/day on POD 1–3).
On POD 1, his consciousness level remained JCS I-3. Blood pressure was in the range of 130–140/55–78 mmHg, heart rate 95–110/min, and respiratory rate 15–20/min. With 0.5 L/min of supplemental oxygen via nasal cannula, SpO2 was in the range of 97–98%; body temperature ranged from 36.4 to 37.2 °C. On the same day, left-sided burr-hole drainage was additionally performed without intraoperative complications. Postoperative laboratory tests showed BUN at 18.5 mg/dL, creatinine at 0.67 mg/dL, sodium at 144 mEq/L, potassium at 2.9 mEq/L, CRP at 10.0 mg/dL, and WBC at 15,300/µL, consistent with postoperative inflammatory response and pre-existing pneumonia, as well as mild hypokalemia (Table 1). Potassium replacement was not administered on POD 1, and potassium was not remeasured after subsequent supplementation before collapse.
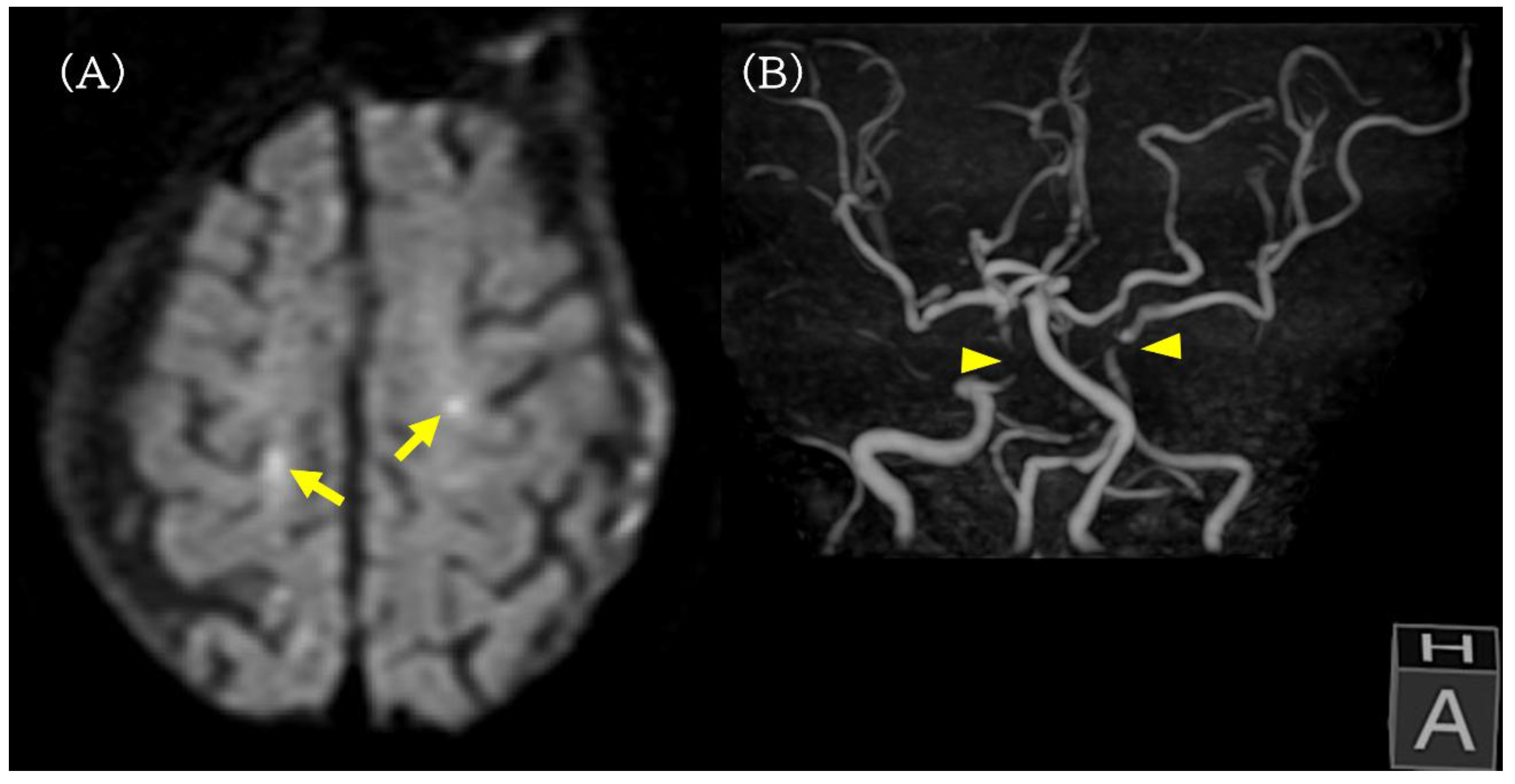
On POD 2, oxygen was discontinued and his SpO2 remained in the range of 95–98% on room air. He was able to converse and eat during the day. During sleep at night, transient desaturation into the 80% range was observed, and low-flow oxygen at 0.5 L/min was restarted, but other vital signs remained stable with systolic blood pressure in the range of 120–130 mmHg and a heart rate in the range of 80–100/min. Brain magnetic resonance imaging (MRI) performed on POD 2 revealed multiple small acute cortical infarcts in both cerebral hemispheres, and magnetic resonance angiography (MRA) demonstrated poor visualization of the bilateral internal carotid artery (ICA) siphon segments and a severe stenosis of the left ICA, indicating advanced carotid atherosclerotic disease with reduced cerebrovascular reserve (Figure 3).
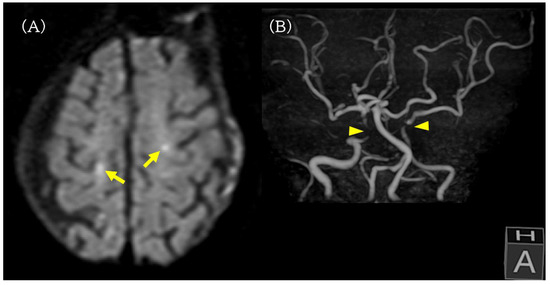
Figure 3.
Brain MRI and MRA on postoperative day 2. (A) Diffusion-weighted MRI revealing multiple small acute cortical infarcts in both cerebral hemispheres (yellow arrow). (B) MR angiography showing poor visualization of the bilateral internal carotid artery siphon segments (yellow arrowhead) and a severe stenosis of the left internal carotid artery, indicating advanced carotid atherosclerotic disease and reduced cerebrovascular reserve.
On POD 3, the patient ate a paste-like diet at lunch without aspiration, and SpO2 remained 99% on room air, allowing discontinuation of oxygen. Blood pressure stayed at around 120–130 mmHg systolic, heart rate at 80–100/min, respiratory rate at approximately 15–20/min, and temperature remained in the 36 °C range. Nursing documentation did not describe any obvious change in urine output or abdominal distension during the day.
At 22:00 on POD 3, his body temperature abruptly rose to 39.0 °C and heart rate increased to the 120/min range, prompting an evaluation by the on-call physician. His consciousness remained JCS I-3, blood pressure showed no documented hypotension, and SpO2 on the bedside monitor was 98%. Intravenous acetaminophen (1000 mg) was administered. By 22:30, his temperature had decreased to 37.0 °C, but tachypnea was noted, and ST depression in V3 was seen on the monitor tracing. A 12-lead ECG confirmed ST depression in V3 without new ST-segment elevation.
Laboratory tests obtained at that time showed BUN at 16.2 mg/dL, creatinine at 1.05 mg/dL (mild renal dysfunction compared with baseline), sodium at 144 mEq/L, potassium at 2.6 mEq/L (further hypokalemia), CRP at 14.0 mg/dL (increasing from POD 1), and a marked drop in WBC to 3300/µL from 15,300/µL two days earlier (Table 1). No blood cultures, chest or abdominal CT, or abdominal ultrasound were performed at this point. Intravenous potassium supplementation (10 mEq) was administered, but potassium was not remeasured before collapse. The fever was initially attributed to a noninfectious postoperative inflammatory response, and management consisted of increased intravenous fluids and antipyretics, without antibiotic escalation or an abdominal work-up.
At 03:11 on POD 4, the nursing record documented SpO2 at 99%, heart rate at 100/min, respiratory rate in the range of 20–30/min, and a large volume of watery diarrhea. Abdominal findings at that time were not examined and not documented in the nursing record. At 04:03, his SpO2 suddenly dropped to 60%; systolic blood pressure was initially recorded at 150 mmHg. A reservoir mask at 10 L/min was applied, but SpO2 did not improve; therefore, the on-call physician was urgently summoned. Soon thereafter, systolic blood pressure rapidly fell to 60 mmHg. Norepinephrine infusion via a peripheral venous line was attempted, but line occlusion and extravasation prevented effective administration. At the time of deterioration, serum lactate, procalcitonin, arterial blood gas analysis, cardiac biomarkers (e.g., troponin, BNP), and bedside echocardiography were not obtained.
Around 05:00, his blood pressure was 60/40 mmHg and SpO2 remained at 80%. The cardiac monitor showed severe bradycardia progressing to pulseless electrical activity/asystole, and cardiopulmonary resuscitation (CPR) was initiated. After bag-mask ventilation, endotracheal intubation was performed at 05:17. Despite ongoing chest compressions, the rhythm remained asystole; transient rises in blood pressure were observed, but sustained return of spontaneous circulation was not achieved.
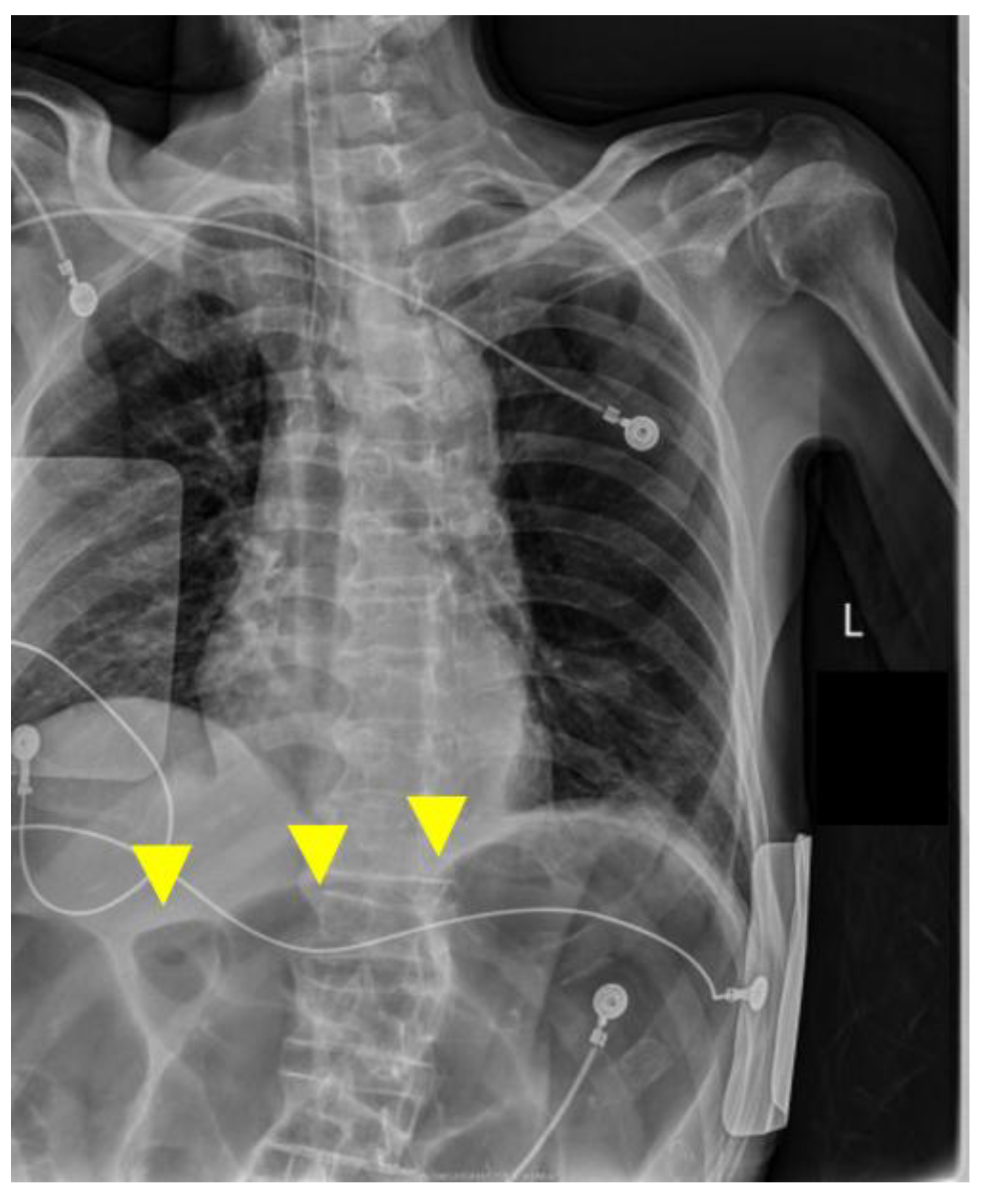
A portable anteroposterior chest radiograph obtained immediately after intubation confirmed appropriate endotracheal tube position and showed no new diffuse bilateral infiltrates suggestive of acute respiratory distress syndrome (ARDS) or marked progression of pneumonia. In contrast, the upper abdomen demonstrated markedly dilated gas-filled bowel loops consistent with colonic distension, likely involving the transverse colon (Figure 4). Following a discussion with the family, a do-not-resuscitate order was confirmed, CPR was discontinued at 05:44, and death was pronounced at 06:15. The death certificate listed acute heart failure as the immediate cause of death.
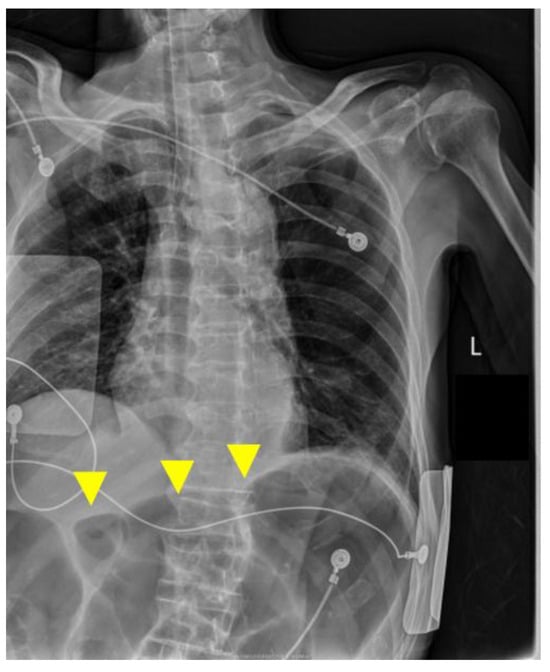
Figure 4.
Portable chest radiograph at the time of intubation on postoperative day 4. Yellow arrows indicate markedly dilated gas-filled bowel loops in the upper abdomen, consistent with colonic gas distension.
The anteroposterior bedside chest X-ray confirmed the appropriate position of the endotracheal tube and showed no new diffuse bilateral infiltrates suggestive of acute respiratory distress syndrome (ARDS) or marked progression of pneumonia. The upper abdomen shows markedly dilated gas-filled bowel loops, most prominent in the transverse colon region; although interpretation is limited on a portable chest radiograph, this finding is considered supportive of possible abdominal pathology in a clinical picture compatible with sepsis.
No blood cultures, stool cultures, or C. difficile toxin assays were obtained during the febrile episode or at the time of deterioration, and no autopsy was performed because the family declined. Written informed consent for publication of this case was obtained from the patient’s family.
3. Discussion
This case illustrates a fatal failure-to-rescue event in a frail older adult with CSDH, in whom rapid postoperative deterioration occurred after an apparently uneventful burr-hole drainage procedure. A clinical picture consistent with sepsis was suspected, and a gastrointestinal source was considered, but it could not be confirmed because key diagnostic tests (e.g., cultures, serum lactate, and abdominal imaging) were not obtained. Clinical attention remained focused on pre-existing pneumonia and the expected postoperative neurosurgical course. Although CSDH has long been regarded as a “benign” condition, recent data show that its 12-month mortality can reach a range of 15–20%, and even higher in some cohorts, with a substantial proportion of deaths attributable to medical, rather than neurosurgical, causes [1,2,3,10].
Our patient fits this pattern: burr-hole drainage was technically uneventful, but a rapidly evolving postoperative clinical picture compatible with sepsis may have contributed substantially to the fatal outcome.
3.1. Hypothetical Interpretation of Admission Hypoxemia and Pre-Existing Colonic Dilatation
On admission, chest CT demonstrated only mild right lower-lobe bronchopneumonia, yet the patient exhibited moderate-to-severe hypoxemia (PaO2 51.7 mmHg while receiving oxygen at 6 L/min). Because FiO2 was not precisely measured, PaO2/FiO2 was not calculated. Within a few days, his oxygenation normalized on room air and remained stable until POD 3. This combination of “disproportionately severe” initial hypoxemia and “rapid recovery” is difficult to explain by mild pneumonia alone.
Importantly, the admission CT also showed a moderately distended transverse colon. In long-standing diabetes, autonomic neuropathy and myopathic changes can cause GI dysmotility, manifesting as delayed gastric emptying, chronic constipation alternating with sudden watery diarrhea, small and large bowel dilatation, and even chronic intestinal pseudo-obstruction [7,11]. Acute and chronic intestinal pseudo-obstruction are well-recognized in older, postoperative, or critically ill patients and are associated with abdominal distension, colonic dilatation, and metabolic abnormalities such as hypokalemia [12].
Abdominal distension and elevated intra-abdominal pressure can mechanically impair respiratory function by elevating the diaphragm, reducing functional residual capacity (FRC), decreasing lung and chest wall compliance, and promoting dependent atelectasis. Experimental work in animal models has shown that abdominal distension can reduce FRC and total lung capacity by approximately 40% and markedly decrease chest wall compliance [13], and perioperative reviews list “intra-abdominal hypertension (ileus, ascites, abdominal compartment syndrome)” as risk factors for postoperative atelectasis and hypoxemia [14].
Taken together, it is plausible that this patient’s admission hypoxemia was multifactorial. In addition to mild pneumonia and reduced physiologic reserve, baseline colonic distension (possibly related to diabetic enteropathy or pseudo-obstruction) may have contributed mechanically by increasing intra-abdominal pressure and limiting diaphragmatic excursion; however, this mechanism remains speculative and cannot be confirmed in the present case. The relative contribution of these factors may have changed over time as the patient’s overall condition, positioning, and pulmonary findings evolved. This case therefore underscores the importance of considering non-pulmonary contributors, including abdominal pathology, when the severity of hypoxemia appears disproportionate to the radiographic extent of pneumonia.
3.2. Red Flags Suggestive of Sepsis and Possible Abdominal Pathology on POD 3
Up to POD 2, the postoperative course appeared stable: the patient was eating, conversing, and maintaining adequate oxygenation on room air. On the night of POD 3, however, he developed an abrupt 39 °C fever, a rise in CRP to 14 mg/dL, and a striking fall in WBC count from 15,300 to 3300/µL, followed by profuse watery diarrhea and radiographic worsening of colonic gas distension.
In Sepsis-3, sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection, and organ dysfunction can be operationalized as an increase in the Sequential Organ Failure Assessment (SOFA) score of 2 points or more [15]. Although SOFA scoring and serum lactate were not available or not obtained in this case, this POD 3 cluster represents discordant findings that should prompt active evaluation for sepsis and possible abdominal emergencies in older adults [16]. In older adults, severe infections may present without leukocytosis; leukopenia or a fall from a previously elevated WBC count can be a marker of advanced sepsis and bone marrow exhaustion [5,17]. Moreover, intra-abdominal sepsis in older adults is often characterized by minimal abdominal pain and few peritoneal signs despite advanced disease, and is associated with high mortality [6,18].
Spangler et al. emphasize that in geriatric abdominal emergencies, relying on “classic” signs such as severe pain, guarding, or marked leukocytosis is dangerous, because many older patients with life-threatening abdominal pathology appear deceptively mild at presentation [18]. In our case, the constellation of high fever, CRP re-elevation, abrupt leukopenia, pre-existing colonic dilatation, new-onset watery diarrhea, and radiographic colonic distension (Figure 4) should be viewed as a cluster of red flags suggestive of sepsis with a possible abdominal source. Differential considerations include infectious enteritis/colitis (including C. difficile), bowel ischemia, and acute colonic pseudo-obstruction with secondary mucosal injury and bacterial translocation, but the etiology could not be confirmed in the absence of cultures, serum lactate, dedicated abdominal imaging, and autopsy [12].
At this juncture, the combination of high fever, rising CRP, abrupt leukopenia, and new gastrointestinal symptoms warranted a focused evaluation for sepsis and potential abdominal emergencies in an older adult, consistent with established sepsis definitions and early management principles (e.g., prompt clinical assessment, blood cultures prior to antibiotics when feasible, serum lactate measurement, and source evaluation including abdominal examination and imaging when indicated) [15,16]. Although early postoperative fever within the first 48 h is often noninfectious, fever occurring after this period—especially high-grade fever accompanied by inflammatory marker rise or organ dysfunction—should prompt active exclusion of infection [19,20]. In this case, the episode occurred overnight and the patient had appeared relatively stable earlier that day; the fever was interpreted as potentially noninfectious postoperative inflammation, and dedicated abdominal evaluation or escalation was not performed at that time.
3.3. Anchoring Bias and Failure to Rescue
A central feature of this case is the role of cognitive bias—specifically anchoring—in the delayed recognition of a possible infectious source. From admission onward, the clinical frame was “CSDH with mild pneumonia,” and after surgery, “expected postoperative course.” Once this mental model was established, subsequent discordant data—new leukopenia, diarrhea, and worsening colonic distension—were interpreted within that frame rather than prompting timely reframing and escalation.
Iwai et al. reported a pediatric case in which an initial diagnosis of “viral infection” anchored the diagnostic process, leading to a delayed recognition of severe polymicrobial sepsis [9]. Etchells and others have highlighted that anchoring and related cognitive biases contribute to a large proportion of diagnostic errors, particularly when new, discordant findings are prematurely forced into an existing diagnosis rather than triggering reconsideration [21]. In our patient, the presence of known pneumonia and the common teaching that CSDH can be accompanied by a benign postoperative inflammatory response likely lowered the perceived probability of a new abdominal source of infection, delaying appropriate investigation.
The outcome can also be viewed through the lens of failure to rescue (FTR). FTR—death after potentially treatable complications—has emerged as a key quality indicator in surgery, explaining inter-hospital differences in mortality better than complication rates alone [8,22]. High-performing hospitals tend not necessarily to have fewer complications, but to detect and manage them more effectively once they occur [22]. In this case, the patient developed acute clinical deterioration compatible with sepsis and shock; however, timely recognition, diagnostic evaluation, and escalation were limited. System factors such as out-of-hours staffing, challenges in securing reliable intravenous access for vasopressors, and limited abdominal assessment may have contributed to reduced rescue opportunities.
We acknowledge the risk of hindsight bias when retrospectively discussing investigations that were not obtained. At the time of initial deterioration, the clinical changes occurred during an out-of-hours period, and the patient had appeared relatively stable earlier, with non-specific findings that can be challenging to interpret in frail older adults. These limitations reflect the common realities of clinical practice and underscore the value of robust escalation pathways and team-based situational awareness rather than attributing outcomes to an individual clinician alone.
From a failure-to-rescue perspective, concrete system-level interventions that may mitigate similar events include the routine use of early warning scores (EWSs) to trigger escalation, structured nighttime escalation protocols (e.g., rapid response activation), early interdisciplinary consultation when new gastrointestinal signs emerge in postoperative patients, and timely access to diagnostic imaging when clinical trajectories change. Such team-based approaches can support earlier recognition and coordinated action in high-risk postoperative patients.
3.4. Risk Profile of Elderly CSDH Patients with Diabetes and Cerebrovascular Disease
The FINISH trial post hoc analysis showed that mortality after CSDH surgery is strongly influenced by age and comorbidities, including cardiovascular disease, dementia, cerebrovascular disease, and hypertension [10]. Our patient also had advanced carotid atherosclerotic disease with bilateral ICA siphon involvement and multiple cortical infarcts on MRI, indicating severely reduced cerebrovascular reserve. In such patients, even modest hypotension or hypoxemia can precipitate rapid neurological and systemic deterioration.
Diabetic enteropathy may further predispose to colonic dysmotility, acute colonic pseudo-obstruction, and bacterial overgrowth [11,12]. In the presence of additional insults—surgery, immobility, and electrolyte disturbances such as hypokalemia—these “vulnerable” bowels are at risk of progressing from subclinical dilatation to clinically significant ileus or colitis and, ultimately, sepsis. Our case highlights how the convergence of advanced age, diabetes, cerebrovascular disease, and CSDH creates a narrow margin of safety: seemingly “minor” postoperative complications (e.g., enteritis, pseudo-obstruction) can quickly become lethal if not recognized and treated early.
3.5. Limitations
This report has several limitations. First, the causative pathogen and definitive source of infection could not be identified. No blood or stool cultures, C. difficile toxin assays, serum lactate, procalcitonin, or abdominal CT/ultrasound were obtained during the febrile episode or at the time of deterioration, and no autopsy was performed. Accordingly, sepsis (and any specific source such as a gastrointestinal focus) remains a clinical suspicion rather than a microbiologically confirmed diagnosis. Second, key data at the time of collapse were limited: arterial blood gas analysis, detailed oxygenation parameters (FiO2), and echocardiography/cardiac biomarkers were not available or not obtained at the time of deterioration (serum lactate, repeat arterial blood gas analysis, troponin, repeat NT-proBNP/BNP, and echocardiography were not obtained), and therefore alternative mechanisms such as pulmonary embolism, acute coronary syndrome, or acute heart failure cannot be excluded. We have revised the manuscript to emphasize diagnostic uncertainty and to focus on the practical recognition of postoperative “red flags” and escalation triggers, rather than definitive attribution of the cause of death.
3.6. Implications for Practice
In summary, this case demonstrates how, in a frail elderly CSDH patient with diabetes and cerebrovascular disease, postoperative critical illness consistent with sepsis can be difficult to recognize when clinicians remain anchored on pre-existing pneumonia and expected neurosurgical trajectories. Disproportionate hypoxemia relative to mild pneumonia, incidentally noted colonic dilatation on admission imaging, and the POD 3 cluster of high fever, rising CRP, abrupt leukopenia, watery diarrhea, and worsening colonic gas represent discordant “red flags” that warrant reframing of the working diagnosis and prompt evaluation.
For CSDH patients with similar risk profiles, perioperative teams should: (1) interpret “mild” pneumonia cautiously when hypoxemia is severe or rapidly changing, (2) treat early CT evidence of bowel dilatation as a potential warning sign rather than a benign incidental finding, (3) regard “high fever + CRP rise/re-elevation + abrupt leukopenia + new-onset diarrhea” as red flags that warrant the evaluation for postoperative sepsis and abdominal emergencies, and (4) consciously re-examine initial diagnostic frames when new, discordant data emerge. Strengthening these cognitive and system safeguards may help prevent future failure-to-rescue events in frail neurosurgical patients.
4. Conclusions
This case describes a frail older adult with diabetes and CSDH who underwent apparently uncomplicated bilateral burr-hole drainage but died after sudden postoperative deterioration. A clinical picture compatible with septic shock was suspected based on high fever, rising CRP, abrupt leukopenia, watery diarrhea, and progressive colonic gas distension; however, the infectious focus and exact mechanism of death could not be confirmed because key diagnostic tests (e.g., cultures, lactate, abdominal imaging, and autopsy) were not obtained. The primary aim of this case report is to highlight early recognition of warning signs and practical escalation triggers to reduce the risk of failure-to-rescue in frail postoperative neurosurgical patients, rather than to establish a definitive cause of death.
In older CSDH patients with diabetes and cerebrovascular disease, even common infections such as pneumonia or enteritis can rapidly progress to shock with a clinical picture consistent with sepsis because cerebral and systemic reserve are markedly reduced. This report highlights three practical implications for perioperative care in such patients: (1) when hypoxemia appears disproportionate to mild radiographic pneumonia, clinicians should reassess for non-pulmonary contributors, including abdominal pathology; (2) postoperative “high fever + CRP re-elevation + leukopenia + new-onset diarrhea” should be treated as a red-flag pattern for sepsis rather than a benign postoperative inflammatory response; and (3) when new, discordant findings emerge, the initial diagnostic frame should be consciously set aside and the differential diagnosis rebuilt from first principles, explicitly considering abdominal sources of infection. Strengthening both cognitive vigilance and system-level escalation pathways may help prevent similar failure-to-rescue events in frail neurosurgical patients.
Author Contributions
Conceptualization, N.K. and Y.A.; Investigation, Y.A., T.N. and K.M.; Resources, K.G.; Writing—original draft preparation, Y.A. and K.M.; Writing—review and editing, N.K. and K.G.; Supervision, N.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Ethical review and approval were waived for this case report in accordance with the institutional policy of Fukujukai Adachi Tobu Hospital regarding the ethics review process for case reports. Under this policy, review is waived when a report describes a single patient’s clinical course retrospectively, involves no research-driven intervention or additional procedures beyond standard care, and is prepared using de-identified clinical information; publication is permitted only after removal of potentially identifying details and confirmation of consent for publication.
Informed Consent Statement
Written informed consent for publication of this case report and the accompanying images was obtained from the patient’s family.
Data Availability Statement
Data sharing is not applicable to this article due to patient privacy and ethical restrictions. All relevant clinical information is included in the manuscript. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| ARDS | acute respiratory distress syndrome |
| BUN | blood urea nitrogen |
| CPR | cardiopulmonary resuscitation |
| CRP | C-reactive protein |
| CSDH | chronic subdural hematoma |
| CT | computed tomography |
| ECG | electrocardiogram/electrocardiography |
| FRC | functional residual capacity |
| FTR | failure to rescue |
| GI | Gastrointestinal |
| HbA1c | glycated hemoglobin A1c |
| ICA | internal carotid artery |
| ICU | intensive care unit |
| JCS | Japan Coma Scale |
| MRA | magnetic resonance angiography |
| MRI | magnetic resonance imaging |
| NT-proBNP | N-terminal pro-B-type natriuretic peptide |
| POD | postoperative day |
| RA | room air |
| SpO2 | peripheral oxygen saturation |
| WBC | white blood cell |
| SOFA | Sequential Organ Failure Assessment |
References
- Blaauw, J.; Jacobs, B.; den Hertog, H.M.; van der Gaag, N.A.; Jellema, K.; Dammers, R.; Kho, K.H.; Groen, R.J.M.; van der Naalt, J.; Lingsma, H.F. Mortality after chronic subdural hematoma is associated with frailty. Acta Neurochir. 2022, 164, 3133–3141. [Google Scholar] [CrossRef] [PubMed]
- Tabuchi, S.; Kadowaki, M. Chronic subdural hematoma in patients over 90 years old in a super-aged society. J. Clin. Med. Res. 2014, 6, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Hendrix, P.; Goren, O.; Dalal, S.; Kanmounye, U.S.; Weiner, G.M.; Schirmer, C.M.; Melamed, I. In-hospital mortality rates, complication rates, length of stay, and total costs of >14,000 chronic subdural hematomas treated in the U.S. between 2016 and 2020: Query of the premier health-care database. Surg. Neurol. Int. 2022, 13, 364. [Google Scholar] [CrossRef] [PubMed]
- Ou, L.; Chen, J.; Hillman, K.; Flabouris, A.; Parr, M.J.; Creighton, R.J.K. The impact of post-operative sepsis on mortality after hospital discharge among elective surgical patients: A population-based cohort study. Crit. Care 2017, 21, 34. [Google Scholar] [CrossRef] [PubMed]
- Rowe, T.A.; McKoy, J.M. Sepsis in older adults. Infect. Dis. Clin. N. Am. 2017, 31, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Podnos, Y.D.; Jimenez, J.C.; Wilson, S.E. Intra-abdominal sepsis in elderly persons. Clin. Infect. Dis. 2002, 35, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Shakil, A.; Church, R.J.; Rao, S.S.C. Gastrointestinal complications of diabetes. Am. Fam. Physician 2008, 77, 1697–1702. [Google Scholar] [PubMed]
- Hatchimonji, J.S.; Kaufman, E.J.; Sharoky, C.E.; Ma, L.; Garcia Whitlock, A.E.; Holena, D.N. Failure to rescue in surgical patients: A review for acute care surgeons. J. Trauma. Acute Care Surg. 2019, 87, 699–706. [Google Scholar] [CrossRef] [PubMed]
- Iwai, K.; Tetsuhara, K.; Ogawa, E.; Kubota, M. Hidden diagnosis behind viral infection: The danger of anchoring bias. BMJ Case Rep. 2018, 2018, bcr-2018-226613. [Google Scholar] [CrossRef] [PubMed]
- Tommiska, P.; Knuutinen, O.; Lönnrot, K.; Luoto, T.; Leinonen, V.; Koivisto, T.; Tetri, S.; Posti, J.P.; Raj, R. Mortality and causes of death after surgery for chronic subdural hematoma: A post hoc study of the FINISH randomized trial. Acta Neurochir. 2025, 167, 310. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, B.; Babu, S.; Walker, J.; Walker, A.B.; Pappachan, J.M. Gastrointestinal complications of diabetes mellitus. World J. Diabetes 2013, 4, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Sharman, T. Intestinal Pseudo-Obstruction. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 3 July 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK560669/ (accessed on 16 December 2025).
- Mutoh, T.; Lamm, W.J.E.; Embree, L.J.; Hildebrandt, J.; Albert, R.K. Abdominal distension alters regional pleural pressures and chest wall mechanics in pigs in vivo. J. Appl. Physiol. 1991, 70, 2611–2618. [Google Scholar] [CrossRef] [PubMed]
- Lagier, D.; Zeng, C.; Fernandez-Bustamante, A.; Melo, M.F.V. Perioperative pulmonary atelectasis—Part II: Clinical implications. J. Cardiothorac. Vasc. Anesth. 2022, 36, 784–800. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Evans, L.; Rhodes, A.; Alhazzani, W.; Antonelli, M.; Coopersmith, C.M.; French, C.; Machado, F.R.; Mcintyre, L.; Ostermann, M.; Prescott, H.C.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Intensive Care Med. 2021, 47, 1181–1247. [Google Scholar] [CrossRef] [PubMed]
- Malone, M.L.; Perry, A.; Weeks, R. Emergency Department Care of an Older Patient with Sepsis—A Brief Topic Review. J. Geriatr. Emerg. Med. 2020, 1, 1–6. [Google Scholar]
- Spangler, R.; Pham, T.V.; Khoujah, D.; Martinez, J.P. Abdominal emergencies in the geriatric patient. Int. J. Emerg. Med. 2014, 7, 43. [Google Scholar] [CrossRef] [PubMed]
- Pile, J.C. Evaluating postoperative fever: A focused approach. Clevel. Clin. J. Med. 2006, 73, S62–S66. [Google Scholar] [CrossRef] [PubMed]
- Garibaldi, R.A.; Brodine, S.; Matsumiya, S.; Coleman, M. Evidence for the non-infectious etiology of early postoperative fever. Infect. Control 1985, 6, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Etchells, E. Anchoring Bias with Critical Implications. AHRQ PSNet (WebM&M). 2015. Available online: https://psnet.ahrq.gov/web-mm/anchoring-bias-critical-implications (accessed on 16 December 2025).
- Ghaferi, A.A.; Birkmeyer, J.D.; Dimick, J.B. Variation in hospital mortality associated with inpatient surgery. N. Engl. J. Med. 2009, 361, 1368–1375. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.