Abstract
Understanding the behavioral strategies that allow freshwater mussels to persist under environmental stress is essential for their conservation, yet burrowing behavior remains poorly quantified. We tested whether valve movement data could be used to detect and characterize burrowing in the endangered Westralunio carteri; a species endemic to a region undergoing severe climatic drying. Mussels from multiple populations were monitored individually under laboratory conditions using Hall effect sensors, and valve movement patterns were analyzed to distinguish between burrowing and non-burrowing behaviors. Burrowing was associated with rapid, high-amplitude valve movements that lengthened as burial progressed, while non-burrowing behaviors showed distinct, slower patterns. These differences indicate that valvometry can reliably identify burrowing behavior, providing a non-invasive method for monitoring mussel activity. This approach has broad applications for ecological research, conservation assessment, and early-warning biomonitoring of imperiled freshwater mussel populations.
1. Introduction
Freshwater mussels (order Unionida) play a critical role in lentic and lotic ecosystems through water filtration, nutrient cycling, and provision of habitat for other organisms [1]. By filtering large volumes of water, they influence suspended particulate concentrations, redistribute nutrients between the water column and benthos, and enhance sediment–water coupling, ultimately supporting diverse aquatic communities [2]. Despite their ecological importance, freshwater mussels are among the most threatened faunal groups globally, with approximately 45% of the ~900 known species listed as near-threatened, threatened, or extinct; a figure that rises to 67% in North America [3]. Population declines have been linked to multiple stressors including habitat alteration, flow regulation, sedimentation, disease, introduction of non-indigenous species, and climate change [3]. Understanding the ecology and behavior of imperiled mussel species is therefore essential for guiding conservation and management efforts [3,4,5].
While population stress is commonly assessed using distribution-based methods, such as presence–absence surveys, density estimates, or mark–recapture studies [6], behavioral and physiological indicators can provide real-time and mechanistic insights into environmental stress responses, such as the closing of a shell when conditions become unfavorable [7]. One such understudied aspect of freshwater mussel ecology is locomotion, particularly the ability of mussels to burrow. Burrowing behavior is a key survival strategy that can mitigate exposure to predators, desiccation, and exposure to environmental stressors, while also contributing functionally to ecosystem processes through bioturbation [8,9,10,11,12].
Despite its importance, the environmental and biological drivers of burrowing behavior in freshwater mussels remain poorly characterized, largely due to the difficulty of observing and quantifying this behavior under natural conditions. Consequently, most existing knowledge derives from controlled laboratory experiments that employ visual observation or time-lapse photography to monitor mussel movement and burial [11,13,14]. These studies have provided valuable insights into how environmental variables such as temperature, flow velocity, and substrate composition influence burial depth, frequency, and timing [9,11]. However, laboratory-based approaches often simplify environmental complexity, limiting their capacity to capture the full range of behavioral variability expressed in situ. Moreover, the episodic nature of observational methods restricts temporal resolution and may miss transient or stimulus-driven behaviors. In this study, we trial a laboratory-based approach designed to be readily replicated in the field, providing a controlled framework to quantify burrowing dynamics while maintaining compatibility with natural environmental conditions. By bridging the gap between highly controlled laboratory studies and potential field applications, this approach improves our ability to understand how mussels dynamically adjust burrowing behavior in response to fluctuating environmental conditions. Recent technological developments, particularly the use of valvometers, devices that non-invasively record valve opening and closing, have opened new avenues for studying unionid behavior and physiology under natural conditions. Valvometry provides continuous, high-resolution records of valve activity, offering insights into feeding rhythms, responses to stressors, and diel patterns of behavior [7,15]. In addition to research applications, valvometry has been implemented in environmental monitoring and biomonitoring programs [16]. For example, in several European countries, species such as Unio tumidus are used as live biosensors in drinking water treatment plants, where abnormal valve closure patterns serve as early-warning indicators of pollution or toxic events [7]. Such facilities in Poland employ mussel-based alert systems that collectively safeguard drinking water supplies for more than ten million people [7]. The ability of these systems to detect rapid behavioral responses highlights their potential utility in ecological and conservation research on freshwater mussels.
The present study focuses on Carter’s Freshwater Mussel (Westralunio carteri; Family Hyriidae), which is endemic to southwestern Australia [17]. Recent integrative taxonomic work has shown that what was previously considered a single, widespread species actually comprises multiple lineages, restricting the true distribution of W. carteri to a narrower set of western coastal drainages and implying that its former range and abundance were overestimated [18]. Contemporary surveys show that many historical populations have contracted, with mussels now occurring at reduced and patchy densities, and several sites experiencing recent local extirpation. Once widespread, these declines, driven by habitat degradation, altered flow regimes, and increasing salinity, and severe regional climatic drying over the past 50 years have led to its Vulnerable conservation status [19,20]. Westralunio carteri is a relatively large and long-lived mussel, with a life span exceeding 40 years, and exhibits burrowing behavior that appears sensitive to hydrological and thermal fluctuations [9,21]. As one of the few native benthic filter feeders in these river systems, it performs important ecosystem functions including biofiltration, nutrient cycling, and providing structural habitat for other benthic organisms. Despite being a key bioindicator species in southwestern Australian freshwater systems, its behavioral ecology remains poorly understood.
The first step in identifying the environmental drivers of burrowing in freshwater mussels is the ability to detect burrowing events reliably. Burrowing in mussels is achieved through rhythmic extension and contraction of the muscular foot, which forces the valves into the substrate [22]. These movements involve characteristic patterns of valve adduction that appear to differ from those associated with normal filtration and feeding activity. Consequently, valvometry may offer a novel approach to detecting and quantifying burrowing behavior.
The aim of this study is to determine the valve movement patterns associated with burrowing in W. carteri and evaluate whether such movements can be distinguished in data collected from telemetered mussel monitoring stations. By establishing a valvometric signature of burrowing, this study seeks to advance the use of behavioral monitoring as a tool for understanding unionid ecology and assessing population responses to environmental change.
2. Materials and Methods
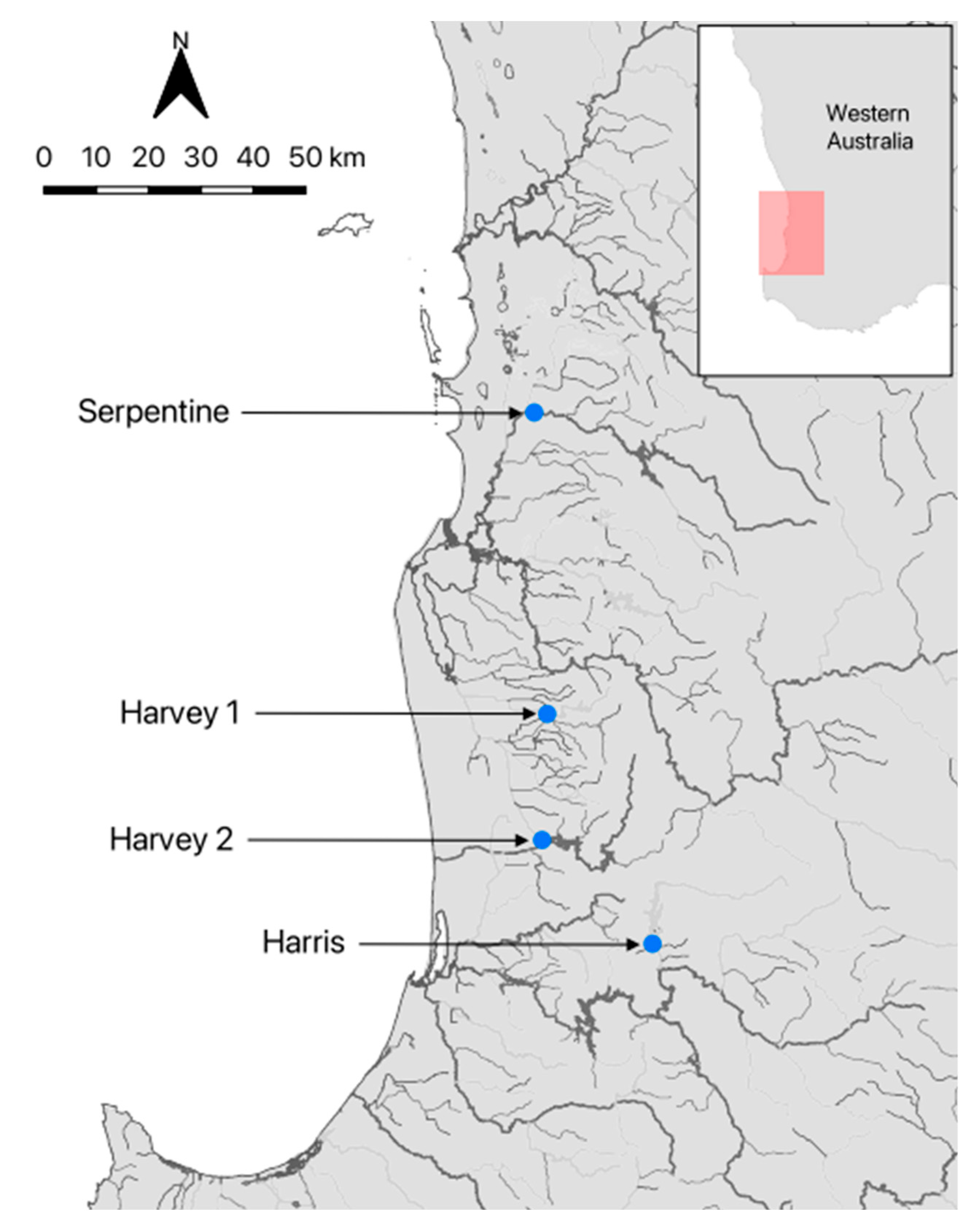
Westralunio carteri (n = 32) of similar shell length (62.5–76.5 mm; Table 1) were collected from the Harris, Serpentine and Harvey rivers in southwestern Australia (Table 1; Figure 1), and transported in insulated coolers to the laboratory. These rivers were chosen as mussels had previously been collected for use in other studies. Individuals collected from the Harris River (under permit TFA 2324-0023) had been in captivity for several months prior. These mussels were originally collected immediately downstream of the Harris River Dam from ~30 cm depth in mixed sand–mud–gravel sediments, and subsequently housed at Murdoch University in three 220 L recirculating aquaria. Each aquarium contained ~5 cm of washed fine sand to promote burrowing, was maintained with deionised water and 30% weekly water changes, and held under a 12 h light cycle (AquaOne LED). Mussels were fed a live culture of Chlorella vulgaris once per week following water changes.

Table 1.
Location and descriptions of sites sampled for Westralunio carteri and shell length range and number of burrowers in each experiment.
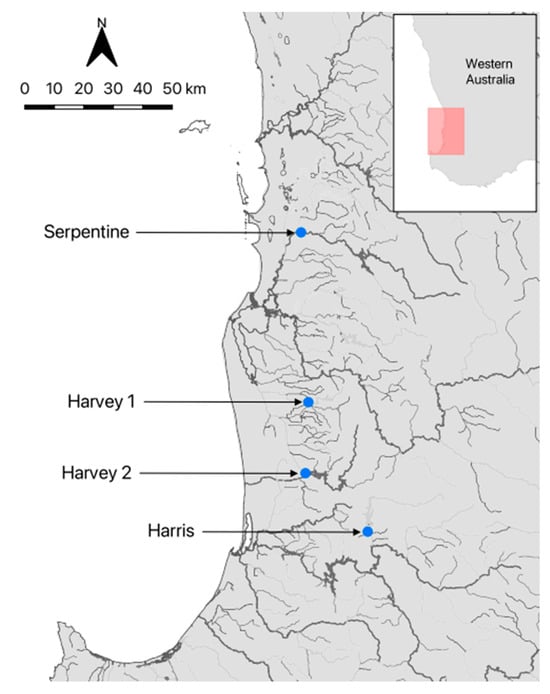
Figure 1.
Map showing the locations of sampling sites in southwestern Australia. Map insert shows location of the sampling area (colored box) in Western Australia.
Mussels collected from the Harvey and Serpentine rivers (under permit TFA 2324-0140) were obtained during a water quality monitoring project that used live-streamed valvometer data (www.spyvalve.com (accessed 9 December 2025)). At the Serpentine River site, sediments consisted of mud, clay, gravel, and organic material, and water temperature at the time of collection was 19.2 °C. At the two Harvey River sites, one consisted of mud, gravel, and organic material (20.2 °C), and the other of medium-grain sand (19.0 °C). For all sites, 45 L of riverine water was collected during sampling, and the same volume was taken from the holding aquaria for the Harris River mussels for use in the burrowing experiments.
The mussels were transported to a temperature-controlled laboratory (20 °C). A Hall effects sensor (encased in silicon) was attached to each mussel via a custom 3D-printed backpack that was glued (Loctite Epoxy Marine) to the posterior region of one valve and a small magnet (5 × 3 mm) glued to the opposing valve so that the sensor and magnet were aligned. During the transportation and attachment of sensors the mussels were exposed to air for ~8 h. Eight mussels with sensors attached were laid out horizontally (to avoid interference between mussels) in a 50 L aquarium held at 20 °C that contained water from the collection site and washed (medium grain) sand ~100 mm deep. Data from the sensors (mV) which vary with magnetic field were read using an Arduino Uno microcontroller board at one second intervals and, along with a timestamp, recorded onto an SD card. Mussel activity was also videoed using a Logitech Full HD Stream Cam attached to a desktop computer and an external hard drive. The first two experiments were run for 17.0 and 16.4 h, and, as it was determined that burial once initiated was typically completed within 5 h, the length of the two subsequent experiments were reduced (i.e., to 11.5 and 7.9 h). We note that as the study was not intended to explore factors driving burrowing behavior, no acclimation period was imposed.
At the end of each experiment the video footage was viewed and used to relate the valvometer data to the activity of each mussel. The time taken to start and complete burrowing was recorded and this was based on each mussel’s extension and contraction of its muscular foot into the sediment.
For each mussel, each data point (recorded at one second intervals) was transformed to between 0 (representing closed valves) and 1 (representing valve open) using the equation: , where is j’th mV reading, and are the minimum and maximum mV value for that mussel.
The data were then interpreted visually on a line graph and statistically analysed in R [23] using wavelet-based analyses (package “WaveletComp” [24]), which employs a Morlet mother wavelet. Wavelet power spectra were computed across the full range of scales supported by the sampling interval using the package’s default scale resolution (dj = 0.1), ensuring adequate frequency detail for behavioral trend detection. As the resultant analyses illustrated an apparent increase in time (delay) between successive digs over the course of burrowing, this was further tested using Pearson’s correlation tests.
3. Results
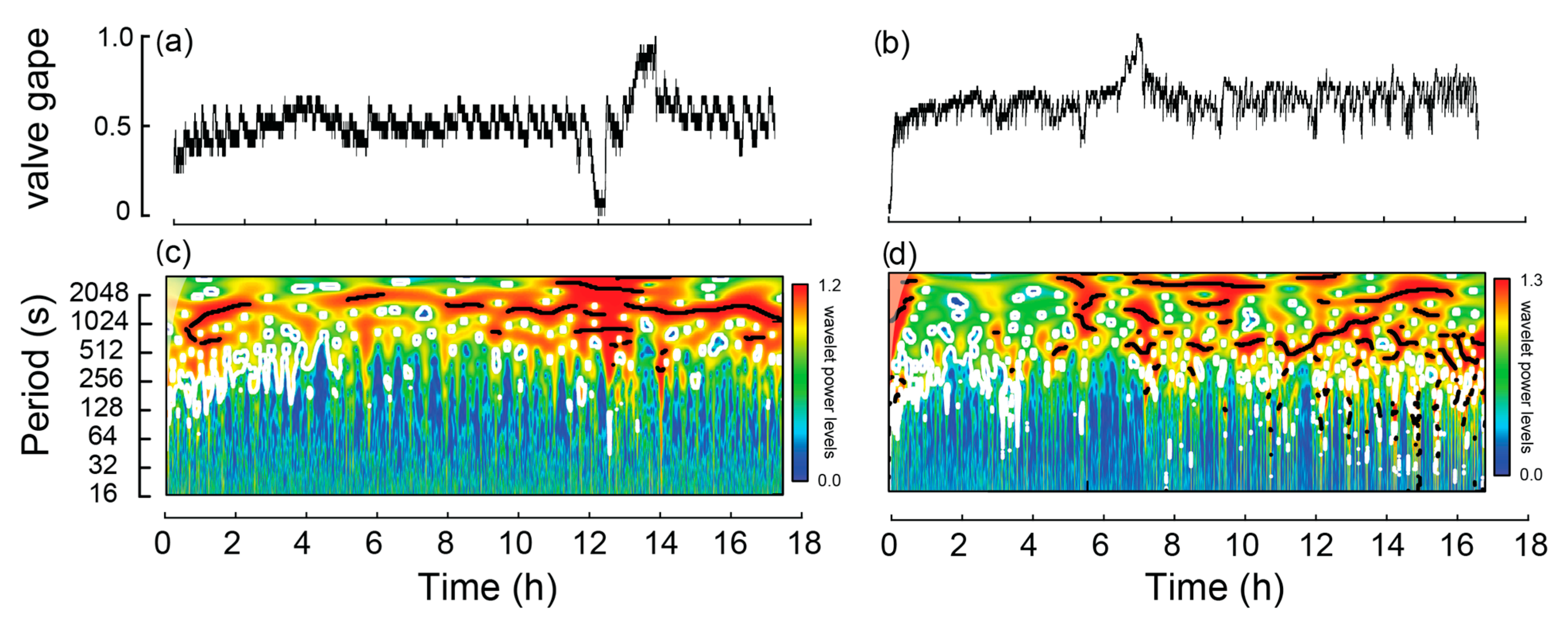
Clear, repeatable movement signatures were detected that distinguished burrowing individuals from those that were non-burrowing. Of the 32 W. carteri over the four experiments, 24 burrowed, one crawled and the remaining seven did not move either vertically or horizontally. Of the non-burrowing mussels, mV alternated by ~10% of the transformed range (0 to 1) at a period of >512 s per cycle, which occurred throughout much of the experiments, as denoted by the significant periods (black lines) in Figure 2c,d.
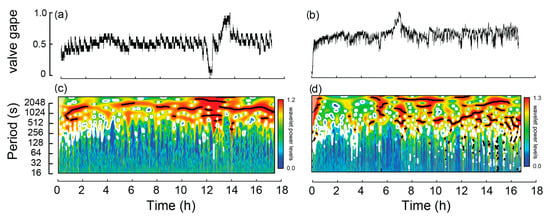
Figure 2.
Example of valve activity from two non-burrowing Westralunio carteri recorded over two ~17 h experiments. A line graph for each mussel is shown in subfigures (a,b), based on changes in mV derived from hall effect sensors (0 represents valves fully closed and 1.0 valves fully open). Corresponding wavelet analyses outputs for each mussel in subfigures (c,d), where color shading indicates wavelet power and black lines denote significant periods.
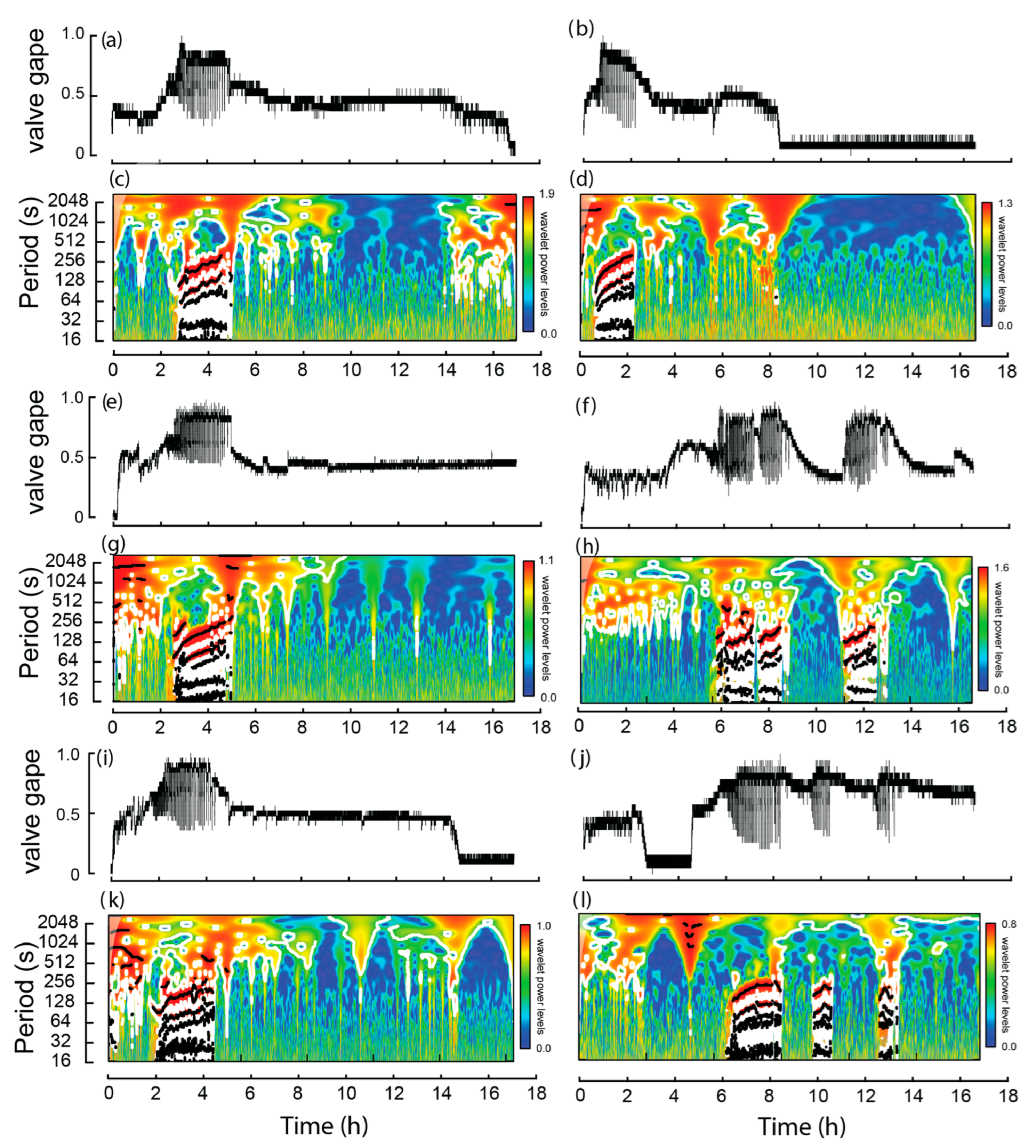
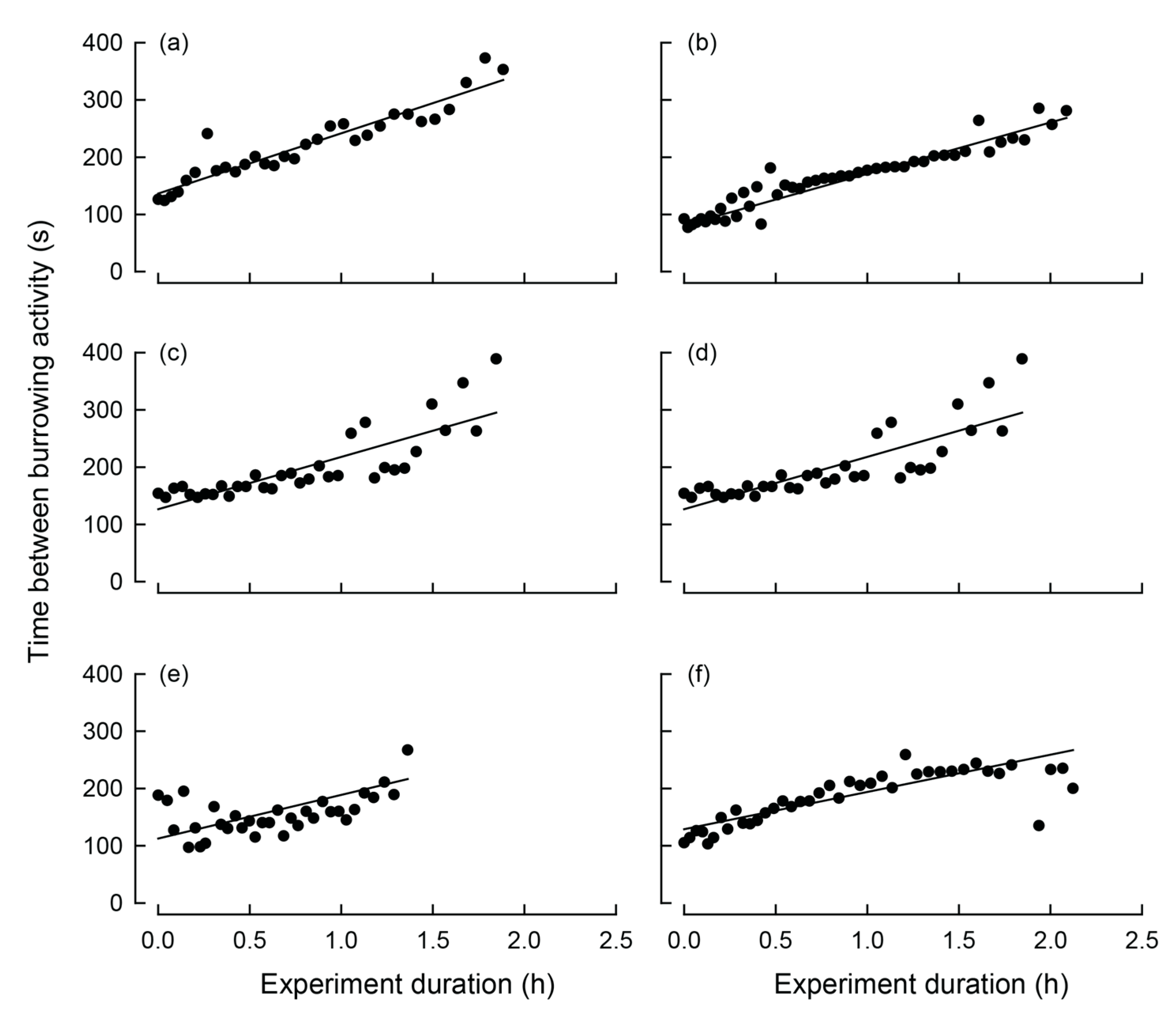
Of the burrowing mussels, burrowing, on average, started 3.5 h (SE = 0.7 h) following the commencement of the experiment and the average burrowing time was 1.8 h (SE = 0.1 h; Figure 3). During extension of the muscular foot into the sediment, i.e., a dig, maximum transformed valve gape values were approximately twice that of the values between the dig (Figure 4). Significant periods of burrowing, representing repeating cycles of valve movement with power exceeding the 95% confidence threshold in the wavelet spectra, were typically <256 s (Figure 3c,d,g,h,k,l). In each case, the time between digs increased significantly over the course of burrowing (Table 2; Figure 4). Thus, based on linear relationships, during the first hour of burrowing, time between digs increased from 124.1 (SE = 10.9) to 216.4 (SE = 20.1) seconds (Table 2, Figure 5).
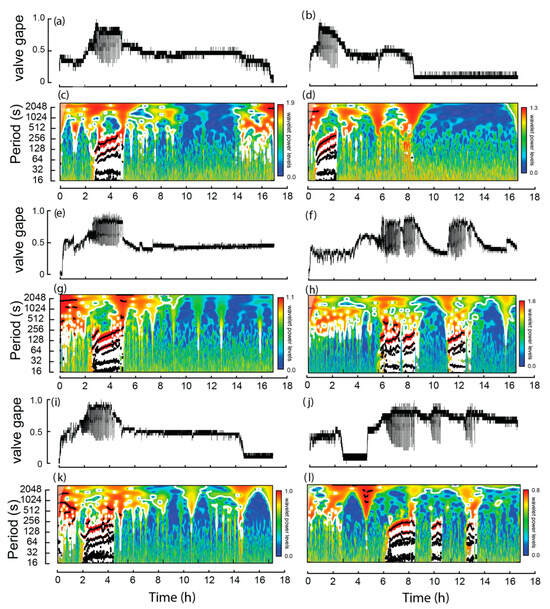
Figure 3.
Valve activity of six burrowing Westralunio carteri recorded over two ~17 h experiments. Line graphs for each mussel shown in subfigures (a,b,e,f,i,j), based on changes in mV derived from hall effect sensors (0 represents valves fully closed and 1.0 valves fully open). Corresponding wavelet analyses outputs for each mussel are shown in subfigures (c,d,g,h,k,l), where color shading indicates wavelet power and black lines denote significant periods.
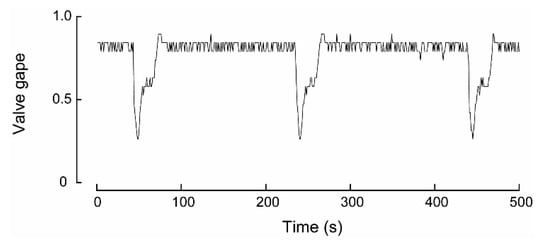
Figure 4.
Valve activity during burrowing activity by Westralunio carteri, based on changes in mV derived from hall effect sensors (0 represents valves fully closed and 1.0 valves fully open).

Table 2.
Slope of the relationship between burrowing activity of Westralunio carteri over time of each regression shown in Figure 5a–f and also for the only individual that crawled (Figure 6d). r is the Pearson correlation coefficient. t-value (test statistic), p-values and df (degrees of freedom) also provided.
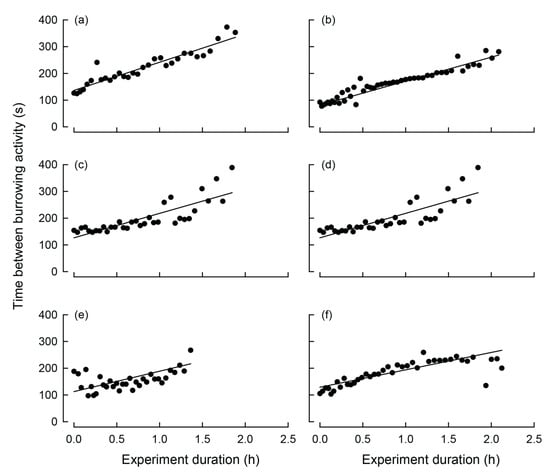
Figure 5.
Relationship between the interval between digging events (seconds) and experimental time (hours) for each of six Westralunio carteri individuals during digging. Points represent the time elapsed between successive digging events as the experiment progressed. Solid lines indicate statistically significant relationships for each mussels (model parameters for each reported in Table 2). Graphs are arranged in the same order as in Figure 3, (a) corresponds to Figure 3a,c; (b) corresponds to Figure 3b,d; (c) corresponds to Figure 3e,g; (d) corresponds to Figure 3f,h; (e) corresponds to Figure 3i,k; (f) corresponds to Figure 3j,l.
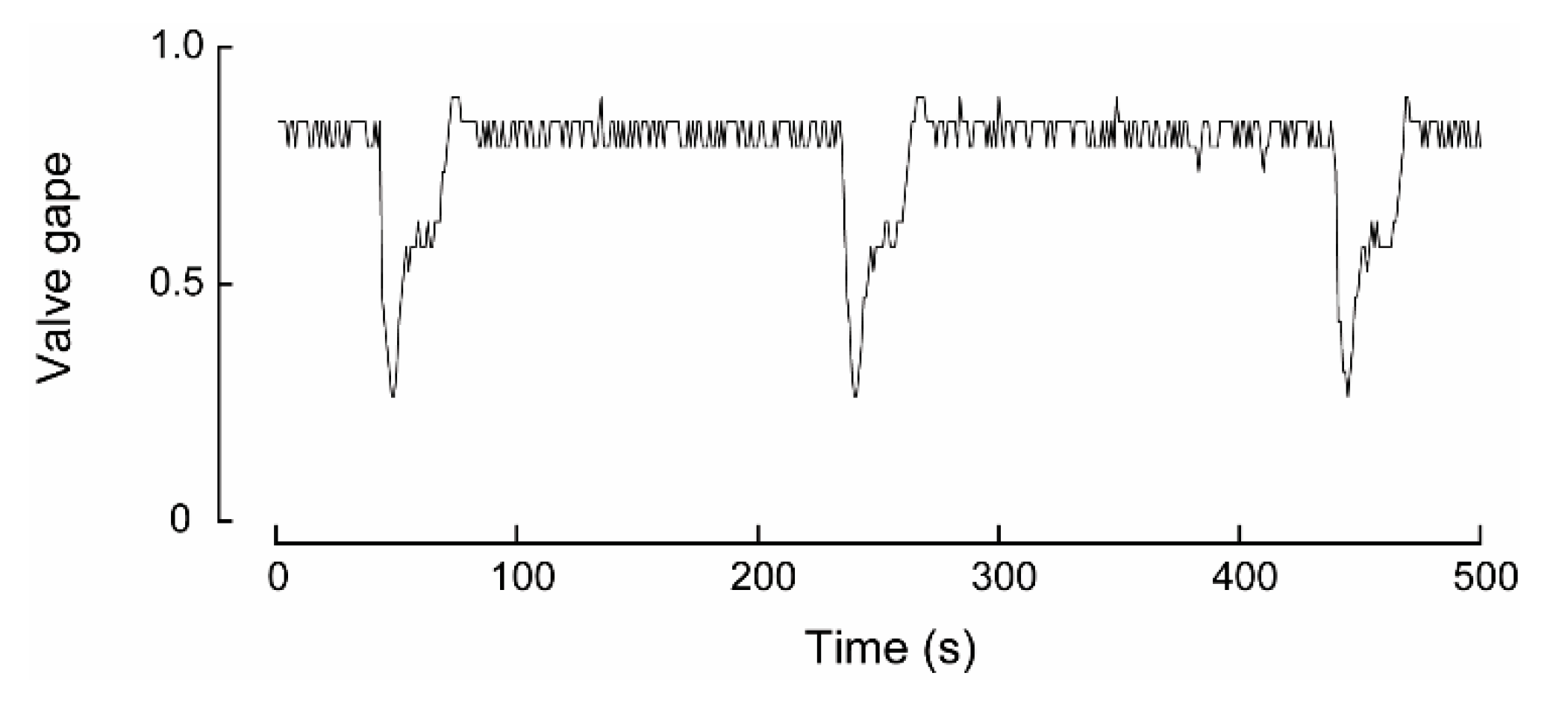
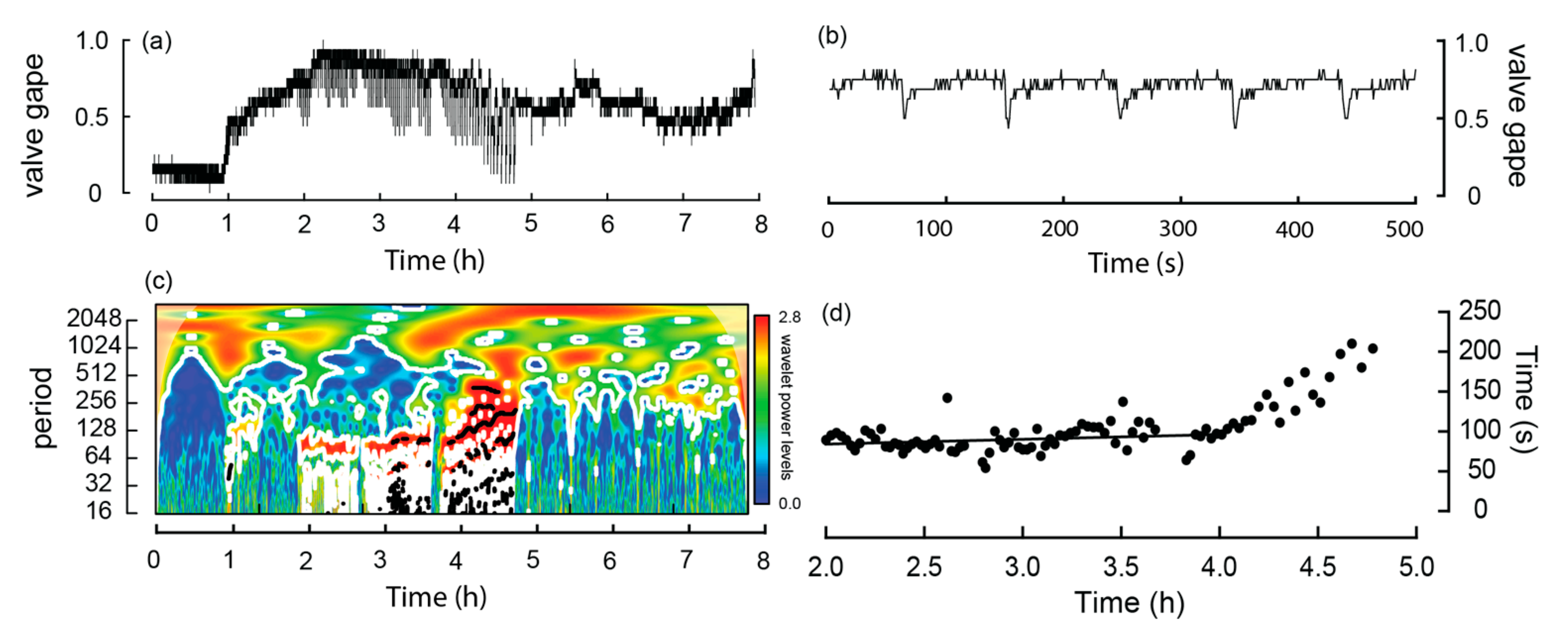
During experiment four, one W. carteri was observed crawling and this behavior differed from burrowing in that each extension and contraction of the muscular foot, which was used to push the mussel across the sediment, occurred over a shorter period. Based on the linear regression, the time taken to complete a ‘step’ increased significantly (p < 0.05) over time from 84.1 to 90.2 s over the first hour (Table 2; Figure 6d). However, during the latter part of the trial the mussel came into contact with other individuals and the side of the aquarium, which noticeably slowed its movement. Because these interactions introduced external constraints on crawl duration, data collected during these periods were excluded from the correlation analysis.
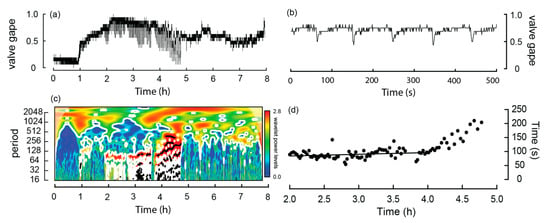
Figure 6.
Valve activity analyses of a Westralunio carteri that crawled during the experiment. (a) line graph of valve gape activity, (b) line graph of valve gape activity during crawling over a 500 s interval, (c) wavelet analyses outputs over the 7.9 h experiment, where color shading indicates wavelet power and black lines denote significant periods, (d) relationship between time (h) and period (s) between steps during the crawl. Dots show time between digs as experiment progressed. Significant linear relationship fitted to the first 2 h of crawl, i.e., from ~2.0 to 4.0 h of the experiment (see also Table 2).
4. Discussion
This study demonstrates that valvometer data can be used to detect and characterize burrowing behavior in Westralunio carteri, providing a methodological foundation for remote monitoring of this ecologically important and endangered freshwater mussel. By combining continuous Hall effect sensor data with video verification, we were able to link distinct patterns in valve movement with behavioral states, particularly burrowing, non-burrowing, and in the case of one individual, crawling. Our results show that burrowing is associated with rapid, high-amplitude valve adductions occurring at relatively short periodicities (<256 s), with a tendency for the time between digging movements to increase as the burrowing sequence progresses. In contrast, non-burrowing mussels exhibited slower, lower-amplitude valve oscillations with much longer periodicities (>512 s). These differences are sufficiently distinct that they could be used as diagnostic indicators in telemetered monitoring applications.
Although burrowing is a key adaptation for mitigating environmental stress, our observations also revealed rare horizontal displacement, with one mussel exhibiting crawling behavior. Such movements may have context-dependent consequences: burrowing near drying banks could prolong survival during drought, whereas crawling into isolated pools may increase exposure to thermal stress or hypoxia, potentially limiting subsequent burrowing capacity [9]. Substrate compaction can also constrain burial, forcing mussels to crawl and risking entrapment in debris [25]. In the present study, mussels were held in uniform, washed sand to provide a consistent and low-compaction substrate that enabled clear detection of burrowing and crawling behaviours. Natural riverine substrates, however, are often more heterogeneous and compacted, and mussels may encounter greater resistance during burial. As such, the relative frequency, speed, and energetic cost of burrowing and crawling observed here may differ under field conditions, particularly in sediments with higher clay content or organic debris loads. The balance between these strategies likely varies among species, as shown by Uniomerus tetralasmus and Lampsilis straminea, which preferentially burrow, versus Pyganodon grandis, which follows receding water levels [8].
Environmental drivers of burrowing remain poorly quantified and are largely inferred from a limited number of observational studies. In W. carteri, larger individuals are more likely to burrow, with frequency declining at greater depths [9]. In other freshwater mussels, such as Unio tumidus, U. pictorum, and Anodonta anatina, current velocity has been suggested as an important influence [11], while photoperiod, temperature, and reproductive state may also play a role. Burrowing has been hypothesized to reduce predation and biofouling by zebra mussels (family Dreissenidae) [10,11], and observational evidence indicates that it can increase with temperature in species, such as Amblema plicata, Leptodea fragilis [10], and Potamilus alatus [26]. However, these patterns are based on a small number of studies, and quantitative, mechanistic understanding of the environmental and biological drivers of burrowing remains lacking.
Our data confirm that valvometers can clearly detect the rapid, sequential adductions associated with burrowing, enabling real-time, non-invasive detection of burrowing. The observed lengthening of inter-dig intervals likely reflects reduced mechanical work as burial depth increases, along with diminished surface cues once partially embedded and possibly the metabolic cost of prolonged digging. While valvometry has often been applied to detect pollutant-induced stress responses [7,27], this study extends its use to natural locomotor behaviors, offering a pathway to distinguish between stress-induced closure and purposeful movement.
Not all individuals burrowed under our controlled conditions, suggesting behavioral variation linked to factors such as captivity duration, site-specific origins, energetic state, body size, or microhabitat preference [13]. Crawling was marked by shorter, more frequent foot extensions than burrowing, suggesting that while it is distinguishable in high-resolution datasets, detection in long-term, lower-frequency records may require supplementary metrics derived from AI-based analyses. Although we cannot directly relate the observed behavioral differences to specific environmental factors at the collection sites, the results highlight the potential for both intrinsic (e.g., energetic state, size) and prior-experience effects (e.g., time in captivity) to influence mussel behavior, emphasizing the need for future studies that systematically investigate these sources of variation.
Behavioral classification from telemetered valvometry has clear applications for conservation and environmental surveillance [28]. In the Spyvalve project (www.spyvalve.com (accessed 9 December 2025)), real-time burrowing detection could serve as an ecological indicator of changes in sediment quality, flow, or predation risk. Increased burrowing might signal thermal or hydrological stress, whereas its absence could indicate unsuitable substrates or impaired locomotor ability. Integrating behavioral detection into existing water-quality alert systems could increase specificity, allowing discrimination between closure caused by contamination and movement linked to habitat adjustment.
5. Conclusions
We provide the first detailed quantification of burrowing in W. carteri using valvometry, identifying clear, repeatable movement signatures that distinguish it from non-burrowing. This capability opens opportunities for automated behavioral monitoring, advancing ecological research, improving population health assessment, and enhancing early-warning systems. Applying this approach in the field could significantly improve understanding of mussel behavioral ecology and support targeted conservation strategies for threatened freshwater mussels.
Author Contributions
Conceptualization, A.C.; methodology, A.C.; formal analysis, A.C.; resources, A.C. and J.D.; data curation, A.C.; writing—original draft preparation, A.C.; all authors assisted in the writing—review and editing; funding acquisition, A.C., J.R.T. and S.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Alcoa Foundation (223429) and the Department of Water and Environmental Regulation (DWER), Western Australia. The authors gratefully acknowledge their financial assistance, which made this study possible.
Data Availability Statement
Data available on request.
Acknowledgments
The authors acknowledge the Noongar People as the Traditional Custodians on the land that the research took place.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Vaughn, C.C. Ecosystem services provided by freshwater mussels. Hydrobiologia 2018, 810, 15–27. [Google Scholar] [CrossRef]
- Howard, J.K.; Cuffey, K.M. The functional role of native freshwater mussels in the fluvial benthic environment. Freshw. Biol. 2006, 51, 460–474. [Google Scholar] [CrossRef]
- Lopes-Lima, M.; Burlakova, L.E.; Karatayev, A.Y.; Mehler, K.; Seddon, M.; Sousa, R. Conservation of freshwater bivalves at the global scale: Diversity, threats and research needs. Hydrobiologia 2018, 810, 1–14. [Google Scholar] [CrossRef]
- Haag, W.R.; Williams, J.D. Biodiversity on the brink: An assessment of conservation strategies for North American freshwater mussels. Hydrobiologia 2014, 735, 45–60. [Google Scholar] [CrossRef]
- Boon, P.J.; Cooksley, S.L.; Geist, J.; Killeen, I.J.; Moorkens, E.A.; Sime, I. Developing a standard approach for monitoring freshwater pearl mussel (Margaritifera margaritifera) populations in European rivers. Aquat. Conserv. Mar. Freshw. Ecosyst. 2019, 29, 1365–1379. [Google Scholar] [CrossRef]
- Sullivan, K.T.; Woolnough, D.A. Water depth, flow variability, and size class influence the movement behavior of freshwater mussels (Unionida) in Great Lakes river drainages. Freshw. Sci. 2021, 40, 328–339. [Google Scholar] [CrossRef]
- Ferreira-Rodríguez, N.; Beggel, S.; Geist, J.P.; Modesto, V.; Österling, M.; Riccardi, N.; Sousa, R.; Urbańska, M. Freshwater mussels as sentinels for safe drinking water supply in Europe. ACS EsT Water 2023, 3, 3730–3735. [Google Scholar] [CrossRef]
- Gough, H.M.; Gascho Landis, A.M.; Stoeckel, J.A. Behaviour and physiology are linked in the responses of freshwater mussels to drought. Freshw. Biol. 2012, 57, 2356–2366. [Google Scholar] [CrossRef]
- Lymbery, A.J.; Ma, L.; Lymbery, S.J.; Klunzinger, M.W.; Beatty, S.J.; Morgan, D.L. Burrowing behavior protects a threatened freshwater mussel in drying rivers. Hydrobiologia 2021, 848, 3141–3152. [Google Scholar] [CrossRef]
- Nichols, S.J.; Wilcox, D.A. Burrowing saves Lake Erie clams. Nature 1997, 389, 921. [Google Scholar] [CrossRef]
- Schwalb, A.N.; Pusch, M.T. Horizontal and vertical movements of unionid mussels in a lowland river. J. N. Am. Benthol. Soc. 2007, 26, 261–272. [Google Scholar] [CrossRef]
- Vaughn, C.C.; Hakenkamp, C.C. The functional role of burrowing bivalves in freshwater ecosystems. Freshw. Biol. 2001, 46, 1431–1446. [Google Scholar] [CrossRef]
- Allen, D.C.; Vaughn, C.C. Burrowing behavior of freshwater mussels in experimentally manipulated communities. J. N. Am. Benthol. Soc. 2009, 28, 93–100. [Google Scholar] [CrossRef]
- Lurman, G.J.; Walter, J.; Hoppeler, H.H. The effect of seasonal temperature variation on behaviour and metabolism in the freshwater mussel (Unio tumidus). J. Therm. Biol. 2014, 43, 13–23. [Google Scholar] [CrossRef]
- Cottingham, A.; Bossie, A.; Valesini, F.; Tweedley, J.R.; Galimany, E. Quantifying the potential water filtration capacity of a constructed shellfish reef in a temperate hypereutrophic estuary. Diversity 2023, 15, 113. [Google Scholar] [CrossRef]
- Kramer, K.J.; Jenner, H.A.; de Zwart, D. The valve movement response of mussels: A tool in biological monitoring. Hydrobiologia 1989, 188, 433–443. [Google Scholar] [CrossRef]
- Klunzinger, M.W.; Beatty, S.J.; Morgan, D.L.; Pinder, A.M.; Lymbery, A.J. Range decline and conservation status of Westralunio carteri Iredale, 1934 (Bivalvia: Hyriidae) from south-western Australia. Aust. J. Zool. 2015, 63, 127–135. [Google Scholar] [CrossRef]
- Klunzinger, M.; Whisson, C.; Zieritz, A.; Benson, J.A.; Stewart, B.A.; Kirkendale, L. Integrated taxonomy reveals new threatened freshwater mussels (Bivalvia: Hyriidae: Westralunio) from southwestern Australia. Sci. Rep. 2022, 12, 20385. [Google Scholar] [CrossRef] [PubMed]
- Klunzinger, M.; Walker, K. Westralunio carteri. In The IUCN Red List of Threatened Species 2014; ICUN: Gland, Switzerland, 2014; p. e.T23073A58526341. [Google Scholar]
- Hope, P. Southern and South-Western Flatlands Cluster Report: Climate Change in Australia Projections for Australia’s NRM Regions; CSIRO: Canberra, Australia, 2015. [Google Scholar]
- Klunzinger, M.W.; Beatty, S.J.; Morgan, D.L.; Lymbery, A.J.; Haag, W.R. Age and growth in the Australian freshwater mussel, Westralunio carteri, with an evaluation of the fluorochrome calcein for validating the assumption of annulus formation. Freshw. Sci. 2014, 33, 1127–1135. [Google Scholar] [CrossRef][Green Version]
- Lewis, J.B.; Riebel, P.N. The effect of substrate on burrowing in freshwater mussels (Unionidae). Can. J. Zool. 1984, 62, 2023–2025. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2025. [Google Scholar]
- Roesch, A.; Schmidbauer, H. WaveletComp: Computational Wavelet Analysis, 2025. Available online: https://CRAN.R-project.org/package=WaveletComp (accessed on 21 August 2025).
- Gagnon, P.M.; Golladay, S.W.; Michener, W.K.; Freeman, M.C. Drought responses of freshwater mussels (Unionidae) in coastal plain tributaries of the Flint River basin, Georgia. J. Freshw. Ecol. 2004, 19, 667–679. [Google Scholar] [CrossRef]
- Block, J.E.; Gerald, G.W.; Levine, T.D. Temperature effects on burrowing behaviors and performance in a freshwater mussel. J. Freshw. Ecol. 2013, 28, 375–384. [Google Scholar] [CrossRef]
- Borcherding, J. Ten years of practical experience with the Dreissena-Monitor, a biological early warning system for continuous water quality monitoring. Hydrobiologia 2006, 556, 417–426. [Google Scholar] [CrossRef]
- Zieritz, A.; Geist, J.; Gum, B. Spatio-temporal distribution patterns of three stream-dwelling freshwater mussel species: Towards a strategy for representative surveys. Hydrobiologia 2014, 735, 123–136. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.