Abstract
Background: Staphylococcus aureus is a clinically relevant pathogen with a strong ability to form biofilms on a wide range of surfaces, which markedly reduces the effectiveness of antimicrobial treatments and contributes to therapeutic failure. Although essential oils (EOs) represent effective and economical antimicrobial alternatives, their clinical application is limited by rapid oxidation, volatility, and potential cytotoxicity. In this context, nanoencapsulation emerges as a promising strategy to improve EO stability, control release, and reduce toxicity. In this study, Tetradenia riparia essential oil was encapsulated into poly(lactide) (PLA) nanoparticles (NP) using the nanoprecipitation method. Methods: The physicochemical properties of the nanoparticles were characterized, and their antibacterial, antibiofilm, and cytotoxic activities were evaluated. Antibiofilm and antibacterial effects against S. aureus were assessed by the broth microdilution method, while cytotoxicity was determined using a VERO cell line. Results: The nanoparticles exhibited nanometric size, spherical morphology, and homogeneous structure. Both free EO and EO-loaded nanoparticles demonstrated antibacterial and antibiofilm activity against S. aureus. Importantly, EO-loaded nanoparticles were significantly less cytotoxic than free EO. Nanoencapsulation effectively prevented rapid EO evaporation and degradation, thereby enhancing stability. The nanoparticles exhibited a zeta potential of approximately −23.1 mV, indicating adequate colloidal stability. Differential scanning calorimetry revealed a reduction in melting enthalpy from 429.63 J/g (blank nanoparticles) to 115.83 J/g for EO-loaded nanoparticles, indicating decreased polymer crystallinity and a system favorable for controlled EO release. Conclusions: Overall, these findings demonstrate that nanoencapsulation of T. riparia essential oil into PLA nanoparticles preserves antimicrobial efficacy, reduces cytotoxicity, and improves physicochemical stability, supporting the potential of this nanostructured system as a promising strategy for the treatment of S. aureus biofilm-associated infections.
1. Introduction
Staphylococcus aureus is a Gram-positive, facultative aerobic bacterium and one of the leading causes of skin and soft tissue infections, including dermatitis. Although it asymptomatically colonizes more than 20% of the human population, S. aureus can cause severe infections when the epithelial barrier is compromised [1]. In its planktonic form, this pathogen readily adheres to biotic and abiotic surfaces, proliferates, produces an extracellular polymeric matrix, and develops structured biofilms. Once mature, biofilms can release individual cells or cell clusters, facilitating the dissemination and colonization of new sites. Biofilms are estimated to be involved in approximately 80% microbial infections and exhibit resistance and tolerance to antimicrobial agents that can be up to 1000-fold greater than those observed in planktonic cells [2,3].
Biofilm-associated S. aureus infections remain particularly difficult to eradicate, even when antibiotics are selected based on standard susceptibility essays [4,5]. This limitation arises because routine clinical assays used to determine antibiotic susceptibility are performed on planktonic bacteria and therefore fail to accurately reflect the physiological behavior of biofilm-embedded cells [6]. As a result, therapeutic failure and recurrence are frequently observed, underscoring the urgent need for alternative or complementary antimicrobial strategies capable of targeting biofilm-associated infections.
Conventional antibacterial agents present several limitations, including adverse side effects, poor aqueous solubility, drug–drug interactions, induction of antimicrobial resistance, and reduced long-term efficacy [7]. Consequently, increasing attention has been directed toward natural products, particularly essential oils (EOs) derived from aromatic plants, which have long been used in traditional medicine and exhibit broad-spectrum antimicrobial activity [8]. Essential oils are complex mixtures of volatile secondary metabolites, mainly monoterpenes, sesquiterpenes, and phenylpropanoids, with terpenes largely responsible for their biological effects [9]. Their antimicrobial activity is primarily attributed to their hydrophobic nature, which promotes strong interactions with membrane lipids, leading to disruption of membrane integrity, altered permeability, and ultimately microbial cell death [10].
Poly (lactic acid) (PLA) polymeric nanoparticles have attracted considerable interest in biomedical and microbiological research due to their biocompatibility, biodegradability, and low toxicity, as PLA is metabolized into lactic acid, a naturally occurring compound in the human body [11,12]. Moreover, PLA-based nanocarriers can effectively protect labile bioactive compounds, including essential oils, from volatilization and environmental degradation. These systems also enable controlled and sustained release, improve aqueous dispersibility, and enhance biological efficacy while reducing adverse effects, supporting their application in antimicrobial and antifungal strategies [13].
Nanoparticle-based delivery systems are particularly advantageous for topical applications, as they enhance permeation and penetration of bioactive compounds into deeper skin layers, provide a depot effect with sustained release, and allow for more specific cellular or subcellular targeting. These attributes have positioned nanocarriers as promising tools in dermatological and systemic therapies [14].
Tetradenia riparia (Hochst.) Codd (Lamiaceae) is an herbaceous shrub, 1–3 m in height, widely distributed across Africa and commonly known as false myrrh, lemon verbena, lavandula, misty plume, or incense [15,16]. The species has been traditionally used to treat cough, edema, diarrhea, fever, headache, malaria, and toothache. Its essential oil exhibits diverse biological activities, including larvicidal, insecticidal, antimalarial, antimicrobial, and antinociceptive effects. Chemically, T. riparia essential oil is composed of a complex mixture of monoterpenes, sesquiterpenes, and diterpenes, with compounds such as calyculone, caryophyllene, β,13β-epoxy-7-abietene, fenchone, terpineol, 14-hydroxy-9-caryophyllene, and germacrene-D-4-ol identified as major constituents associated with its biocidal activity [17,18,19].
Despite their strong antimicrobial potential, essential oils are prone to volatilization, oxidative degradation, and loss of activity upon exposure to light, heat, or pressure. Nanoencapsulation of EOs into polymeric nanoparticles represents an effective strategy to preserve their functional properties by enhancing stability, controlling release, reducing cytotoxicity, and improving water solubility [7,20]. Therefore, the aim of this study was to prepare polymeric nanoparticles containing T. riparia essential oil using the nanoprecipitation method, to characterize their physicochemical properties, and to evaluate their cytotoxicity and antibiofilm activity against Staphylococcus aureus.
2. Materials and Methods
2.1. Materials
Anhydrous sodium sulfate (Na2SO4) was used as a drying agent. Poly(L-lactide) (PLA; molecular weight 90,000–120,000) and Pluronic® F68 were purchased from Sigma-Aldrich, St. Louis, MO, USA. Acetone and absolute ethanol (Merck & Co., Rio de Janeiro, RJ, Brazil) were used as organic solvents. Ultrapure water was used throughout all experiments. Hydrogen was employed as the carrier gas for GC–MS analyses. Uranyl acetate (Sigma-Aldrich) (1% w/v) was used as a contrasting agent for transmission electron microscopy, and gold was used for sputter coating prior to scanning electron microscopy analysis. Nitrogen was used as purge gas for differential scanning calorimetry measurements. Phosphate-buffered saline (PBS) (Sigma-Aldrich) was used in release, washing, and biofilm assays. Mueller–Hinton agar (MHA) and Mueller–Hinton broth (MHB) was obtained from Difco and used for antibacterial assays. Tryptic soy broth (TSB) supplemented with glucose was used for biofilm formation. The reagent 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and dimethyl sulfoxide (DMSO) were used in antibiofilm and cytotoxicity assays. Dulbecco’s Modified Eagle Medium (DMEM) and trypsin were used for culturing VERO cells.
2.2. Plant Material, Botanical Identification, and Extraction of Essential Oils
Fresh Tetradenia riparia leaves were collected in western Paraná State, Brazil, in December 2015. The plant was identified by Professor Ezilda Jacomasi, Department of Pharmacy, Paranaense University (UNIPAR), Paraná. A voucher specimen was deposited in the UNIPAR Herbarium (code 2502).
The essential oil (EO) was extracted by hydrodistillation for 3 h using a modified Clevenger-type apparatus, according to the procedure described in [21]. The distilled EO was collected, dried over anhydrous Na2SO4, and stored in amber glass flasks at −4 °C until use, following the method described in [21].
2.3. Chemical Identification of Essential Oils
The chemical composition of T. riparia EO before and after storage was determined by gas chromatography–mass spectrometry (GC–MS). Compounds were identified by comparing their Kovats retention indices with those of known substances and by comparison with mass spectra from reference databases. Relative quantities (%) were calculated directly from GC peak areas.
GC–MS analyses were performed using a Focus GC system (Thermo Electron Corporation, Waltham, MA, USA) equipped with a DBS-MS column (30 m × 0.25 mm × 0.25 µm). The injector and transfer line temperatures were set at 230 °C. The oven temperature program was as follows: initial temperature 60 °C for 1 min, then increased at 3 °C/min to 220 °C and held for 5 min. Hydrogen was used as the carrier gas. The injection volume was 5 µL (split 1:10), and the ionization energy was 70 eV. Mass spectra were obtained at 230 °C using a DSQ II detector (Thermo Scientific, Waltham, MA, USA) in total ion current (TIC) acquisition mode, with a mass range of 50–659 m/z [21].
2.4. Nanoparticle Preparation
Poly(L-lactide) nanoparticles (NPs) were prepared by the nanoprecipitation method using the dropping technique. The organic phase, consisting of 50 mg EO and 50 mg PLA (MW 90,000–120,000; Sigma-Aldrich) in 4 mL acetone, was added dropwise into the aqueous phase (10 mL of 1.0% w/v Pluronic F68; Sigma-Aldrich) under stirring at 1200 rpm for 30 min. Acetone was removed using a rotary evaporator. Blank PLA nanoparticles (without EO) were prepared using the same procedure. The final NP suspension was stored at 4 °C until further analysis [22].
2.5. Nanoparticle Characterization
Particle size and distribution were measured by dynamic light scattering (DLS) via a NanoPlus zeta/nanoparticle analyzer. Nanosuspensions were diluted with ultrapure water for the analysis. The surface morphology of nanoparticles was determined using a Shimadzu SS-550 scanning electron microscope. Nanosuspension samples were prepared by placing one drop on a glass plate and drying under reduced pressure at 25° C [23].
Nanoparticles were coated with gold using a sputtering apparatus before observation under SEM. Transmission electron microscopy (TEM) was performed to observe the nature and size distribution of the NP. Samples were deposited onto a copper grid and then treated with a drop of 1% w/v uranyl acetate. After one minute, excess uranyl acetate was removed, the sample was dried, and its TEM image was obtained [24].
2.6. Thermal Analysis
Thermal properties of blank NP and EO-loaded NP were determined by differential scanning calorimetry (DSC 4000; PerkinElmer, Shelton, CT, USA). Samples (3–5 mg) were placed in aluminum pans and heated from 20 to 200 °C at 20 °C/min under nitrogen flow (50 mL/min) [25].
2.7. Encapsulation Efficiency
The EO content of NP was quantified by UV–visible spectroscopy (Shimadzu UV–VIS, Tokyo, Japan). A standard curve was prepared by serial dilution of EO in absolute ethanol and measuring absorbance at 325 nm [10,14].
For EO quantification in NP, 2 mg of sample was dissolved in 2 mL of absolute ethanol, and absorbance at 325 nm was compared to the standard curve. Encapsulation efficiency was calculated as %EE = (total loaded EO/initial EO) × 100.
2.8. In Vitro Release Profile
Ten milligrams of NP or EO were placed in individual dialysis bags containing 40 mL PBS and incubated on a shaker at 37 °C. A 2 mL aliquot of the release medium was collected at 0, 15, 30, 60, 90, 120, and 360 min, and replaced with fresh PBS. EO release was quantified by UV–visible spectroscopy at 325 nm [26].
2.9. Stability Assays
A stability assay was conducted over 30–45 days at 25 ± 2 °C according to RDC 45 [27]. The antibacterial activity of T. riparia EO and NP was subsequently analyzed using the broth microdilution assay following CLSI guidelines.
2.10. Strain and Growth Conditions
Staphylococcus aureus ATCC 29213 was used as the test strain. The bacterium was maintained on Mueller–Hinton agar (MHA; Difco) at 4 °C and cultured in Mueller–Hinton broth (MHB; Difco) before assays.
2.11. Microdilution Assay
Antibacterial activity was assessed by broth microdilution following CLSI guidelines [27]. Serial twofold dilutions of EO and NP were prepared in 96-well plates containing 100 µL sterile MHB. Bacterial inoculum (105 CFU/mL) was added to each well. Plates were incubated at 37 °C for 24 h.
The minimum inhibitory concentration (MIC) was defined as the lowest concentration preventing visible growth. Minimum bactericidal concentration (MBC) was determined by subculturing 10 µL from wells with no visible growth onto MHA plates, followed by incubation at 37 °C for 24 h.
2.12. Antibiofilm Activity
A 100 µL bacterial suspension (1 × 108 CFU/mL) prepared in tryptic soy broth (TSB) supplemented with 1% glucose was added to wells containing EO or NP dilutions and incubated at 37 °C for 24 h. Wells were washed with PBS.
For the MTT assay, 20 µL MTT solution (2 mg/mL in PBS) was added, and plates were incubated for 2 h at 37 °C. After removing the MTT solution, 100 µL DMSO was added to solubilize formazan crystals. Absorbance was measured at 570 nm [28].
The biofilm inhibitory concentration (BIC50) was defined as the minimum concentration that inhibited ≥50% of biofilm viability relative to untreated controls.
2.13. Cytotoxicity Assay
Cytotoxicity of EO and NP was evaluated using an MTT assay according to Tangarife-Castaño et al. [29]. VERO cells (African green monkey kidney; Cercopithecus aethiops) were cultured in DMEM for 72 h. Cell monolayers were trypsinized, washed, and seeded at 2.5 × 105 cells/well in 96-well plates.
After 24 h, EO and NP (1, 10, 100, and 1000 µg/mL) were added to wells, followed by incubation for 72 h at 37 °C in 5% CO2. Cells were washed with PBS and incubated with 2 mg/mL MTT for 4 h. Formazan crystals were solubilized with 200 µL DMSO, and absorbance was measured at 530 nm using a Biotek PowerWave XS reader.
The IC50 values were obtained by linear regression of dose–response curves generated in R software. Selectivity indices (SIs) were calculated as SI = IC50/MIC.
3. Results and Discussion
3.1. Chemical Composition of Tetradenia riparia Leaf Essential Oil
The antimicrobial activity of essential oils (EOs) is strongly dependent on their chemical composition. The chemical profile of Tetradenia riparia leaf EO was determined by GC–MS analysis, and the extraction yield was 0.33% (w/w) based on fresh plant material. Essential oil yield and composition are known to be influenced by seasonal and environmental factors, including rainfall, temperature, and plant physiological stage [18]. Previous studies have reported higher EO yields from T. riparia leaves collected during winter (0.265%), whereas reduced yields have been observed in spring (0.168%), likely due to increased rainfall during this period [18].
The chemical constituents identified in the free EO prior to nanoencapsulation are listed in Table 1. Oxygenated monoterpenes (28.94%) represented the predominant chemical class, with fenchone (27.19%) as the major compound. Oxygenated sesquiterpenes accounted for 34.30% of the EO composition, mainly α-cadinol (16.12%) and 14-hydroxy-9-epi-caryophyllene (13.09%). Hydrocarbon sesquiterpenes were also present in relevant proportions (11.7%), particularly caryophyllene (8.40%) (Figure S1, Supplementary Data). This chemical profile is consistent with previous reports on T. riparia leaf EO, confirming the reproducibility of the extraction process and the characteristic chemotype of the species [18].

Table 1.
GC-MS analysis of T. riparia leaves essential oil.
3.2. Nanoparticle Characterization
Incorporation of T. riparia EO into poly(L-lactide) (PLA) nanoparticles effectively prevented rapid volatilization and degradation of the EO, thereby enhancing its physicochemical stability. Dynamic light scattering (DLS) analysis revealed nanoparticle sizes ranging from 221.9 to 396.5 nm, with a mean hydrodynamic diameter of 306.7 ± 7.1 nm. Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) confirmed that the nanoparticles were predominantly spherical, exhibited smooth surfaces, and displayed nanometric dimensions (Figure 1).

Figure 1.
(A) Scanning electron microscopy and (B) transmission electron microscopy images of PLA nanoparticles loaded with T. riparia essential oil. The data are representative of one out of three independent experiments.
The polydispersity index (PDI) values reported in this study were obtained directly from DLS measurements using the cumulant analysis method implemented in the instrument software, in accordance with ISO 22412 [49]. Importantly, the PDI was not calculated manually from the mean particle size and standard deviation. While the mean particle size reflects the average hydrodynamic diameter, the PDI describes the width of the size distribution derived from the autocorrelation function. The low PDI values observed indicate a narrow size distribution and high formulation homogeneity, which are critical attributes for reproducible biological performance.
Nanoencapsulation of aromatic bioactive compounds within carriers ranging from 10 to 1000 nm has been shown to significantly enhance biological activity by improving passive cellular uptake, reducing mass transfer resistance, and increasing contact with microbial cells [50]. In addition, TEM images revealed intact nanocapsule surfaces without cracks or pores. Given the high volatility of the major EO constituents, the absence of surface defects suggests effective protection against premature release, oxidative degradation, and environmental interactions. Overall, these findings support nanoencapsulation as a robust strategy to improve EO stability, prolong antimicrobial activity, enhance aqueous dispersibility, and reduce toxicity [10].
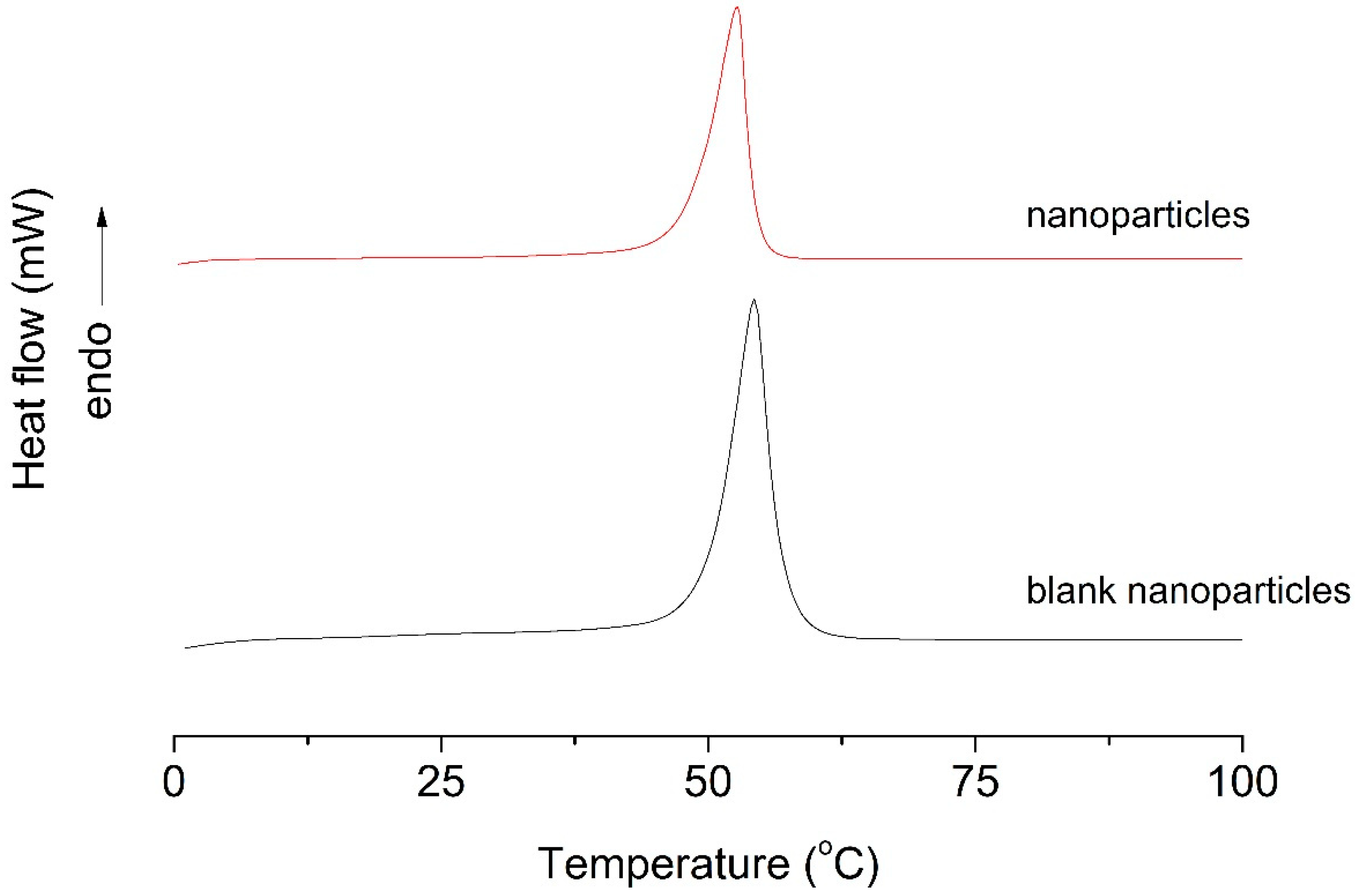
3.3. Differential Scanning Calorimetry (DSC)
Differential scanning calorimetry (DSC) analysis was performed to evaluate the thermal behavior and structural organization of the polymeric nanoparticles. The DSC parameters are summarized in Table 2. Blank PLA nanoparticles exhibited a melting temperature (Tm) of 54.29 °C and a high melting enthalpy (ΔHm) of 429.63 J/g, indicative of a relatively ordered crystalline polymer structure.

Table 2.
Melting temperatures (Tm) and melting enthalpy (ΔHm) of blank NP and NP.
In contrast, EO-loaded nanoparticles showed a slight decrease in melting temperature (52.71 °C) accompanied by a pronounced reduction in melting enthalpy (115.83 J/g). This substantial decrease in ΔHm suggests a marked reduction in polymer crystallinity, indicating that EO incorporation disrupts the crystalline packing of PLA chains. The DSC thermograms (Figure 2) corroborate these observations, as blank nanoparticles displayed a more intense and defined endothermic peak, whereas EO-loaded nanoparticles exhibited a significantly reduced peak area.
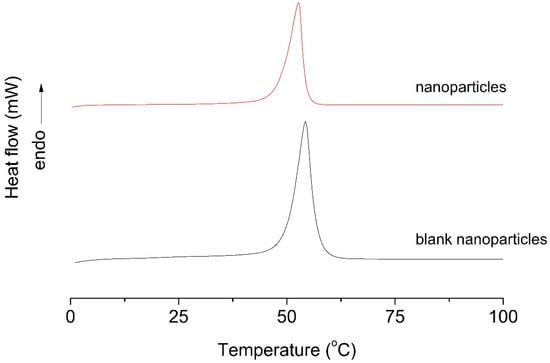
Figure 2.
DSC thermograms of blank nanoparticles and nanoparticles. The data are representative of one out of three independent experiments.
These results indicate strong interactions between the encapsulated EO and the polymer matrix, leading to increased amorphous character and enhanced molecular mobility. Such structural modifications are advantageous for controlled and sustained release of volatile and thermolabile compounds, confirming the successful encapsulation of T. riparia EO and the suitability of PLA nanoparticles as controlled-release delivery systems.
As described in Section 2.8, stability studies conducted for 30–45 days at 25 ± 2 °C in accordance with RDC [45] showed no changes in the antibacterial activity of EO-loaded nanoparticles. Minimum inhibitory concentration (MIC) values remained constant throughout the storage period, demonstrating that nanoencapsulation effectively preserves EO stability and prevents oxidative degradation.
3.4. Zeta Potential and Encapsulation Efficiency
Zeta potential measurements were performed to evaluate the colloidal stability of the nanoparticle suspensions. The EO-loaded nanoparticles exhibited a zeta potential of −23.1 mV, indicating adequate electrostatic stabilization and a reduced tendency toward aggregation. Surface charge is a critical parameter influencing nanoparticle dispersion, flocculation behavior, and interactions with negatively charged bacterial membranes [51,52,53].
Encapsulation efficiency was determined by UV–visible spectrophotometry at 325 nm. The EO-loaded nanoparticles achieved a high encapsulation efficiency of 88.1%, demonstrating the strong affinity between T. riparia EO constituents and the PLA matrix and confirming the effectiveness of the nanoprecipitation method for loading hydrophobic essential oils.
3.5. In Vitro Release Profile
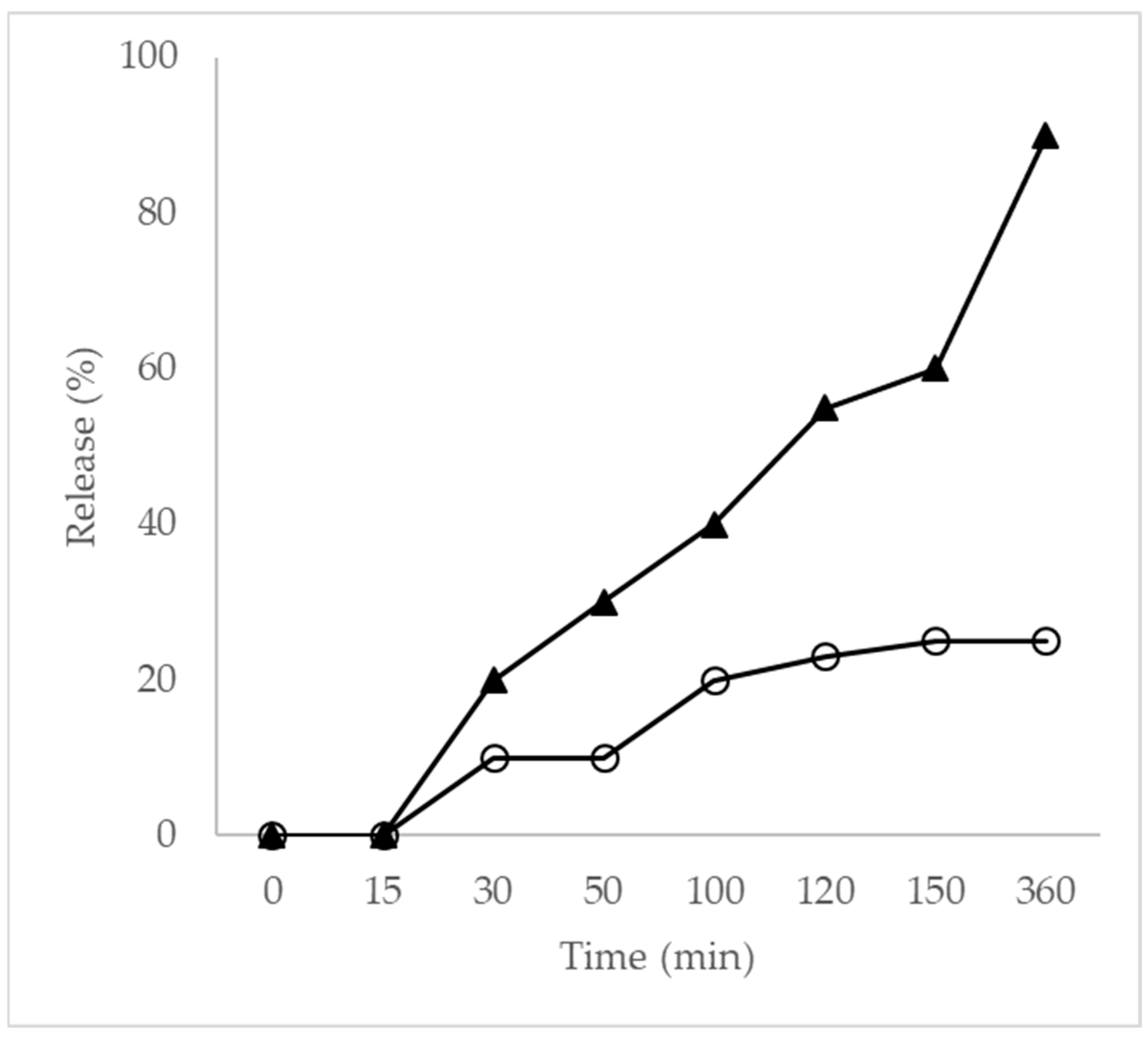
The in vitro release profiles of free EO and EO encapsulated within nanoparticles were evaluated over 360 min (Figure 3). Neither formulation achieved complete release during the assay period. Free EO exhibited rapid release, reaching approximately 90.0% within 360 min, whereas EO-loaded nanoparticles released only 25.5% over the same period, demonstrating a pronounced sustained-release behavior.
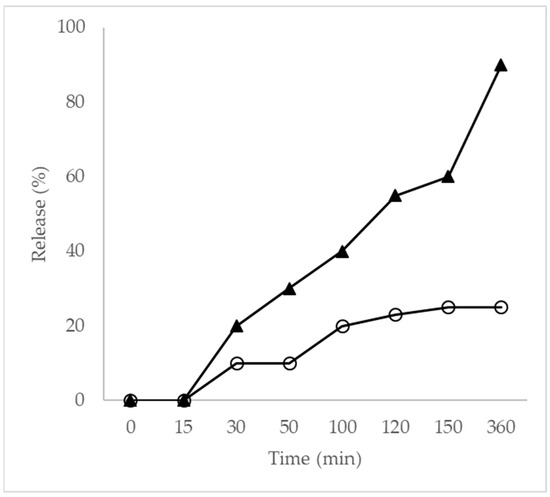
Figure 3.
Release profile curve (▲) EO and (O) NP. The results represent mean values from at least three separate experiments. Standard errors were less than 10% of the means.
Nanoparticles maintained approximately 25.0% release after 100 min, indicating strong retention of EO within the PLA matrix. This controlled-release behavior is attributed to the dense polymeric network formed by poly(L-lactide), which restricts molecular diffusion. In addition, T. riparia EO is rich in sesquiterpenes and diterpenes, compounds with higher molecular weights and lower volatility. Essential oils composed of larger molecules exhibit reduced vapor pressure and stronger interactions with polymeric matrices through hydrogen bonding and van der Waals forces, resulting in slower diffusion and prolonged release [54,55,56]. These findings are consistent with previous reports and highlight the influence of molecular weight and volatility on release kinetics from nanoscale carriers.
3.6. Antibacterial and Antibiofilm Activity
The antibacterial activity of free EO and EO-loaded nanoparticles against Staphylococcus aureus ATCC 29213 was evaluated using broth microdilution assays (Table 3). Based on established criteria, MIC values ≤ 0.5 µg/mL indicate strong antibacterial activity, values between 0.6 and 1.5 µg/mL indicate moderate activity, and values > 1.6 µg/mL indicate inactivity [56].

Table 3.
Minimal inhibitory concentrations (MICs), minimal bactericidal concentrations (MBCs), and 50% biofilm inhibitory concentration (BIC50) in µg/mL of EO, NP, and vancomycin against S. aureus.
Both EO and nanoparticles exhibited comparable antibacterial activity against planktonic and biofilm-associated S. aureus, indicating that nanoencapsulation did not compromise EO antimicrobial efficacy. Although enhanced antibacterial activity after encapsulation would be desirable, maintenance of activity combined with improved stability and reduced toxicity represents a significant pharmacological advantage.
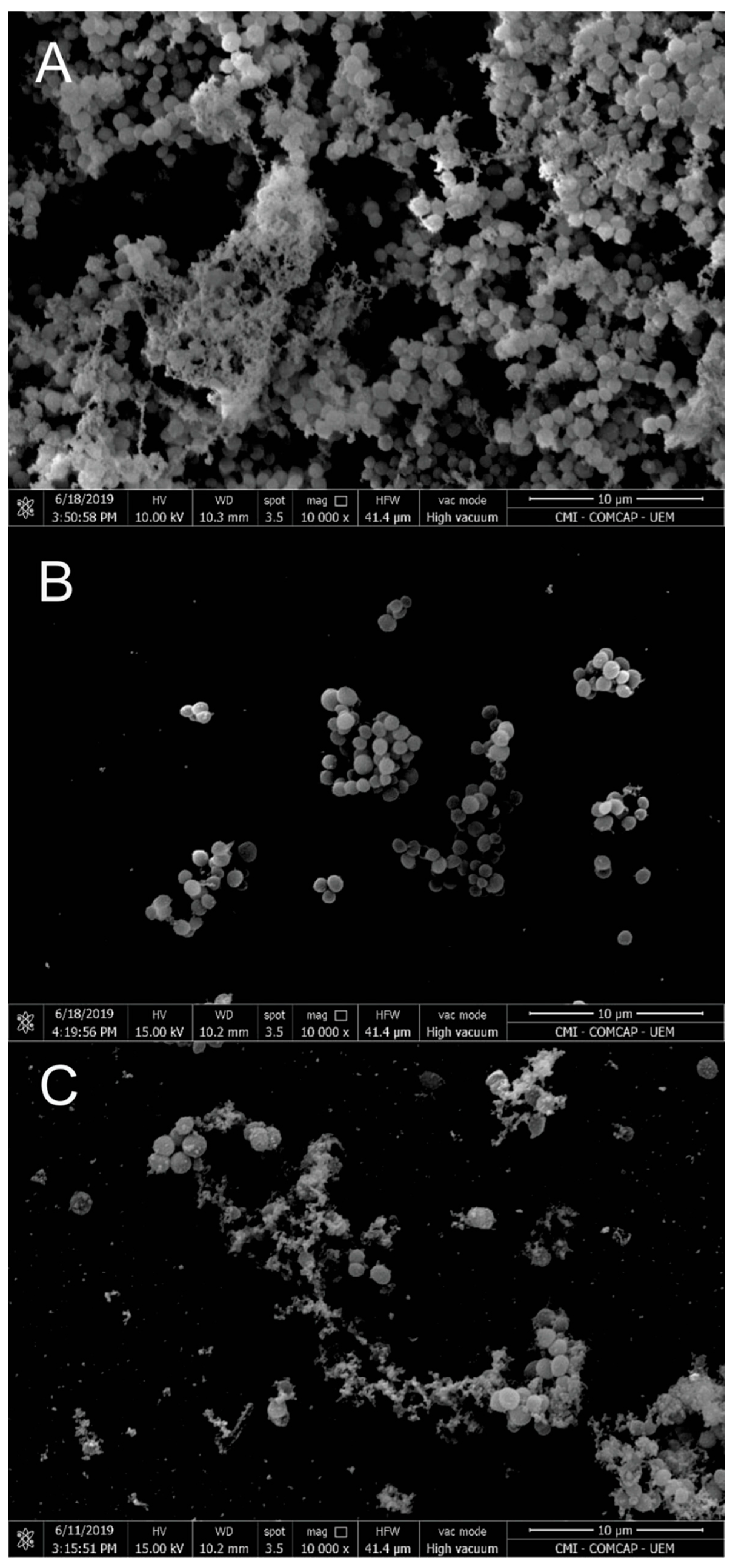
Antibiofilm activity was further evaluated in comparison with vancomycin. The biofilm inhibitory concentration (BIC50) values were 310 µg/mL for free EO and 330 µg/mL for EO-loaded nanoparticles. SEM images of biofilms (Figure 4) revealed that untreated cells exhibited dense biofilm structures with abundant extracellular matrix. In contrast, EO-treated biofilms showed reduced matrix production, altered cell morphology, and decreased cell density. Nanoparticle-treated biofilms also displayed pronounced structural damage, including cell wall and membrane disruption, suggesting irreversible membrane damage, as previously reported for EO-based systems.
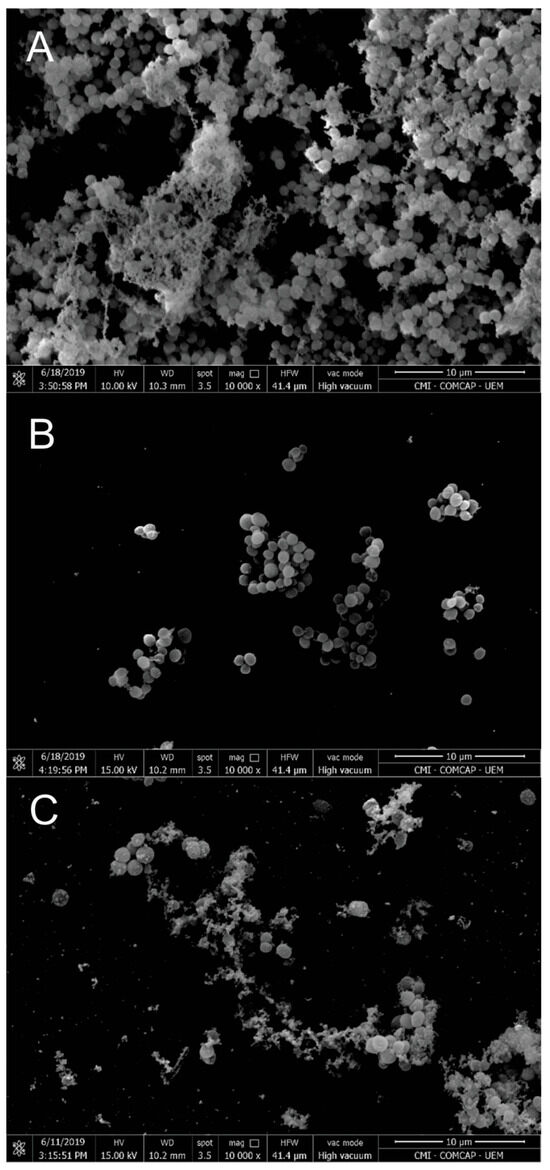
Figure 4.
Scanning electron microscopy: (A) S. aureus biofilm control (untreated); (B) biofilm treated with a subinhibitory concentration of EO; (C) biofilm treated with a subinhibitory concentration of NP. Magnification: 10,000×. Scale bars: 10 µm. The data are representative of one out of three independent experiments.
Similar observations were reported by Kwieciński et al. [57], who demonstrated complete eradication of S. aureus biofilms following treatment with tea tree EO, accompanied by matrix disruption and biofilm detachment. Together, these results support the potential of T. riparia EO and its nanoencapsulated form as antibiofilm agents.
3.7. Cytotoxicity Assessment
Although essential oils are widely investigated as antimicrobial agents, their clinical applicability depends on achieving effective antimicrobial concentrations without inducing host cell toxicity. Cytotoxicity varies according to EO composition, and correlating in vitro toxicity with in vivo safety remains a critical challenge. According to the U.S. National Cancer Institute (NCI), crude extracts with IC50 values ≤ 20 µg/mL are considered cytotoxic [58,59].
As summarized in Table 4, EO-loaded nanoparticles exhibited an IC50 value of 533.96 µg/mL, whereas free EO displayed significantly higher cytotoxicity (IC50 < 125 µg/mL). These results demonstrate that nanoencapsulation substantially reduces EO cytotoxicity toward VERO cells. This reduction is attributed to controlled release and limited direct exposure of cells to high EO concentrations, reinforcing nanoencapsulation as an effective strategy to improve the therapeutic index of EO-based antimicrobial formulations [60].

Table 4.
Cytotoxicity assay of free and nanoencapsulated T. riparia EO on VERO cells.
4. Conclusions
Tetradenia riparia essential oil exhibited a consistent chemical profile rich in oxygenated monoterpenes and sesquiterpenes, supporting its recognized biological potential. Nanoencapsulation into poly(lactide) (PLA) nanoparticles were successful, resulting in high encapsulation efficiency, improved physicochemical stability, and a controlled and sustained release profile. Although nanoencapsulation did not enhance the intrinsic antibacterial activity against Staphylococcus aureus, it effectively preserved the antimicrobial and antibiofilm efficacy of the essential oil while significantly reducing cytotoxicity.
Overall, these findings demonstrate that PLA nanoparticles constitute an efficient and safe delivery system for T. riparia essential oil, optimizing its stability and biocompatibility and reinforcing its potential for future pharmacological and antimicrobial applications, particularly in the context of biofilm-associated infections.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/futurepharmacol6010004/s1, Figure S1: CG-MS chromatogram of T. riparia essential oil. The data are representative of one out of three independent experiments.
Author Contributions
Conceptualization, data curation, formal analysis, investigation, R.Y.M. and E.H.E.; methodology, J.W.M. and C.V.N.; methodology, P.F.R. and F.V.L.; methodology, O.H.G., T.U.-N. and Z.C.G.; conceptualization, funding acquisition, writing—review and editing, E.H.E. and B.P.D.F. All these authors have made substantial contributions to the final manuscript and have approved this submission. All authors are aware of the order of authorship and that no further change in authorship will be performed after submission, except for those previously authorized by the editor-in-chief. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Financiadora de Estudos e Projetos (FINEP), Capacitação e Aperfeiçoamento de Pessoal de Nível Superior (Capes), Fundação Araucária, and Programa de Pós-graduação em Ciências Farmacêuticas da Universidade Estadual de Maringá; Complexo de Centrais de Apoio à Pesquisa (COMCAP/UEM).
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| Eos | Essential oils |
| NPs | Nanoparticles |
| PLA | Poly (L-lactide) |
| DLS | Dynamic light scattering |
| SEM | Scanning electron microscopy |
| TEM | Transmission electron microscopy |
| DSC | Differential scanning calorimetry |
| MHA | Mueller–Hinton agar |
| MHB | Mueller–Hinton broth |
| CFU | Colony-forming unit |
| MIC | Minimum inhibitory concentration |
| MBC | Minimum bactericidal concentration |
| PBS | Phosphate-buffered saline |
| TSB | Tryptic soy broth |
| MTT | Dimethylthiazol-2-yl-2,5-diphenyl tetrazolium bromide |
| DMSO | Dimethyl sulfoxide |
| BIC50 | 50% biofilm inhibitory concentration |
| OD | Optical density |
| DMEM | Dulbecco’s Modified Eagle’s Medium |
| IC50 | 50% inhibitory concentration |
References
- Goldmann, O.; Medina, E. Staphylococcus aureus strategies to evade the host acquired immune response. Int. J. Med. Microbiol. 2018, 308, 625–630. [Google Scholar] [CrossRef]
- Albano, M.; Crulhas, B.P.; Alves, F.C.B.; Pereira, A.F.M.; Andrade, B.F.M.T.; Barbosa, L.N.; Furlanetto, A.; Lyra, L.P.S.; Rall, V.L.M.; Fernandes Júnior, A. Antibacterial and anti-biofilm activities of cinnamaldehyde against Staphylococcus epidermidis. Microb. Pathog. 2019, 126, 231–238. [Google Scholar] [CrossRef]
- Forier, K.; Raemdonck, K.; De Smedt, S.C.; Demeester, J.; Coenye, T.; Braeckmans, K. Lipid and polymer nanoparticles for drug delivery to bacterial biofilms. J. Control. Release 2014, 190, 607–623. [Google Scholar] [CrossRef]
- Kaushik, A.; Kest, H.; Sood, M.; Steussy, B.W.; Thieman, C.; Gupta, S. Biofilm producing methicillin-resistant Staphylococcus aureus infections in humans: Clinical implications and management. Pathogens 2024, 13, 76. [Google Scholar] [CrossRef] [PubMed]
- Brandquist, N.D.; Kielian, T. Immune dysfunction during Staphylococcus aureus biofilm-associated implant infections: Opportunities for novel therapeutic strategies. NPJ Biofilms Microbiomes 2025, 11, 144. [Google Scholar] [CrossRef]
- Sultan, A.R.; Tavakol, M.; Lemmens-den Toom, N.A.; Croughs, P.D.; Verkaik, N.J.; Verbon, A.; van Wamel, W.J.B. Real-time monitoring of Staphylococcus aureus biofilm sensitivity towards antibiotics with isothermal microcalorimetry. PLoS ONE 2022, 17, e0260272. [Google Scholar] [CrossRef]
- Soares, V.B.; Morais, S.M.; Fontenelle, R.O.S.; Queiroz, V.A.; Vila-Nova, N.S.; Pereira, C.M.C.; Brito, E.S.; Neto, M.A.S.; Brito, E.H.S.; Cavalcante, C.S.P.; et al. Antifungal activity, toxicity and chemical composition of the essential oil of Coriandrum sativum L. fruits. Molecules 2012, 17, 8439–8448. [Google Scholar] [CrossRef] [PubMed]
- Raut, J.S.; Karuppayil, S.M. A status review on the medicinal properties of essential oils. Ind. Crops Prod. 2014, 62, 250–264. [Google Scholar] [CrossRef]
- Jamalian, A.; Shams-Ghahfarokhi, M.; Jaimand, K.; Pashootan, N.; Amani, A.; Razzaghi-Abyaneh, M. Chemical composition and antifungal activity of Matricaria recutita flower essential oil. J. Mycol. Med. 2012, 22, 308–315. [Google Scholar] [CrossRef]
- São-Pedro, A.; Espírito-Santo, I.; Silva, V.C.; Albuquerque, E. The use of nanotechnology as an approach for essential oil-based formulations with antimicrobial activity. In Microbial Pathogens and Strategies for Combating Them Science, Technology and Education; Méndez-Vilas, A., Ed.; Formatex Research Center: Badajoz, Spain, 2013. [Google Scholar]
- Makadia, H.K.; Siegel, S.J. Poly (lactic-co-glycolic acid) as biodegradable controlled drug delivery carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Préat, V. PLGA-based nanoparticles: An overview of biomedical applications. J. Control. Release 2012, 161, 505–522. [Google Scholar] [CrossRef] [PubMed]
- Panyam, J.; Labhasetwar, V. Biodegradable nanoparticles for drug and gene delivery to cells and tissues. Adv. Drug Deliv. Rev. 2003, 55, 329–347. [Google Scholar] [CrossRef]
- Raszewska-Famielec, M.; Flieger, J. Nanoparticles for topical application in the treatment of skin dysfunctions. Int. J. Mol. Sci. 2022, 23, 15980. [Google Scholar] [CrossRef] [PubMed]
- Gairola, S.; Naidoo, Y.; Bhatt, A.; Nicholas, A. An investigation of the foliar trichomes of Tetradenia riparia. Flora 2009, 204, 325–330. [Google Scholar] [CrossRef]
- Gazim, Z.C.; Rodrigues, F.; Amorim, A.C.L.; de Rezende, C.M.; Soković, M.; Tešević, V.; Vučković, I.; Krstić, G.; Cortez, L.E.R.; Colauto, N.B.; et al. New natural diterpene-type abietane from Tetradenia riparia essential oil with cytotoxic and antioxidant activities. Molecules 2014, 19, 514–524. [Google Scholar] [CrossRef]
- Gazim, Z.C.; Amorim, A.C.; Hovell, A.M.; Rezende, C.M.; Nascimento, I.A.; Ferreira, G.A.; Cortez, D.A.G. Seasonal variation, chemical composition, analgesic and antimicrobial activities of the essential oil from leaves of Tetradenia riparia. Molecules 2010, 15, 5509–5524. [Google Scholar] [CrossRef]
- Oliveira, P.F.; Alves, J.M.; Damasceno, J.L.; Oliveira, R.A.M.; Dias, H.J.; Crotti, A.E.M.; Tavares, D.C. Cytotoxicity screening of essential oils in cancer cell lines. Rev. Bras. Farmacogn. 2015, 25, 183–188. [Google Scholar] [CrossRef]
- Njau, E.A.; Alcorn, J.; Buza, J.; Chirino-Trejo, M.; Ndakidemi, P. Antimicrobial activity of Tetradenia riparia. Eur. J. Med. Plants 2014, 4, 1462–1472. [Google Scholar] [CrossRef]
- Oliveira, E.F.; Paula, H.C.B.; Paula, R.C.M. Alginate/cashew gum nanoparticles for essential oil encapsulation. Colloids Surf. B Biointerfaces 2014, 113, 146–151. [Google Scholar] [CrossRef]
- Gazim, Z.C.; Demarchi, I.G.; Lonardoni, M.V.C.; Amorim, A.C.L.; Hovell, A.M.C.; Rezende, C.M.; Ferreira, G.A.; de Lima, E.L.; de Cosmo, F.A.; Cortez, D.A.G. Acaricidal activity of the essential oil from Tetradenia riparia. Exp. Parasitol. 2011, 129, 175–178. [Google Scholar] [CrossRef] [PubMed]
- Barwal, I.; Sood, A.; Sharma, M.; Singh, B.; Yadav, S.C. Development of stevioside Pluronic F-68 copolymer-based PLA nanoparticles as an antidiabetic nanomedicine. Colloids Surf. B Biointerfaces 2013, 101, 510–516. [Google Scholar] [CrossRef]
- Granata, G.; Stracquadanio, S.; Leonardi, M.; Napoli, E.; Malandrino, G.; Cafiso, V.; Stefani, S.; Geraci, C. Oregano and thyme essential oils encapsulated in chitosan nanoparticles as antimicrobial agents. Molecules 2021, 26, 4055. [Google Scholar] [CrossRef]
- Koroishi, A.M.; Foss, S.R.; Cortez, D.A.G.; Ueda-Nakamura, T.; Nakamura, C.V.; Dias Filho, B.P. In vitro antifungal activity of extracts and neolignans from Piper regnelli. J. Ethnopharmacol. 2008, 117, 270–277. [Google Scholar] [CrossRef]
- Zhang, S.; Xia, C.; Dong, Y.; Yan, Y.; Li, J.; Shi, S.Q.; Cai, L. Soy protein isolate-based films reinforced by surface-modified cellulose nanocrystal. Ind. Crops Prod. 2016, 80, 207–213. [Google Scholar] [CrossRef]
- Guimarães, G.P. Estabilidade de Medicamentos Sintéticos: Visão Geral da Nova Diretriz da ANVISA. 2020. Available online: https://repositorio.unifesp.br/items/b64d0715-53ed-4643-8273-60f258efec23 (accessed on 3 December 2025).
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 9th ed.; CLSI: Wayne, PA, USA, 2012. [Google Scholar]
- Schillaci, D.; Arizza, V.; Dayton, T.; Camarda, L.; Di Stefano, V. In vitro anti-biofilm activity of Boswellia spp. essential oils. Lett. Appl. Microbiol. 2008, 47, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Tangarife-Castaño, V.; Roa-Linares, V.; Betancur-Galvis, L.A.; Durán-García, D.C.; E-Stashenko, E.; Mesa-Arango, A.C. Antifungal activity of Verbenaceae and Lamiaceae essential oils. Pharmacologyonline 2011, 1, 133–145. [Google Scholar]
- Smadja, J.; Rondeau, P.; Sing, A.S.C. Volatile constituents of five Citrus petitgrain essential oils from Réunion. Flavour Fragr. J. 2005, 20, 399–402. [Google Scholar] [CrossRef]
- Pavlović, M.; Kovačević, N.; Tzakou, O.; Couladis, M. Essential oil composition of Anthemis triumfetti. Flavour Fragr. J. 2006, 21, 297–299. [Google Scholar] [CrossRef]
- Pérez, R.A.; Navarro, T.; de Lorenzo, C. HS-SPME analysis of volatile compounds from spices. Flavour Fragr. J. 2007, 22, 265–273. [Google Scholar] [CrossRef]
- Petrakis, P.V.; Roussis, V.; Papadimitriou, D.; Vagias, C.; Tsitsimpikou, C. Effect of terpenoid extracts from pine species. Behav. Process. 2005, 69, 303–322. [Google Scholar] [CrossRef]
- Zhao, C.X.; Liang, Y.Z.; Fang, H.Z.; Li, X.N. Temperature-programmed retention indices for GC–MS analysis. J. Chromatogr. A 2005, 1096, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Baranauskienė, R.; Venskutonis, P.R.; Viškelis, P.; Dambrauskienė, E. Influence of nitrogen fertilizers on thyme. J. Agric. Food Chem. 2003, 51, 7751–7758. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.X.; Li, X.N.; Liang, Y.Z.; Fang, H.-Z.; Huang, L.-F.; Guo, F.-Q. Comparative analysis of essential oils using chemometrics. Chemom. Intell. Lab. Syst. 2006, 82, 218–228. [Google Scholar] [CrossRef]
- Hazzit, M.; Baaliouamer, A.; Faleiro, M.L.; Miguel, M.G. Composition of Thymus and Origanum essential oils. J. Agric. Food Chem. 2006, 54, 6314–6321. [Google Scholar] [CrossRef]
- Kukić, J.; Petrović, S.; Pavlović, M.; Couladis, M.; Tzakou, O.; Niketić, M. Essential oil of Stachys alpina. Flavour Fragr. J. 2006, 21, 539–542. [Google Scholar] [CrossRef]
- Siani, A.C.; Garrido, I.S.; Monteiro, S.S.; Carvalho, E.S.; Ramos, M.F. Protium icicariba as a source of volatile essences. Biochem. Syst. Ecol. 2004, 32, 477–489. [Google Scholar] [CrossRef]
- Siani, A.C.; Ramos, M.F.S.; Menezes-de-Lima, O.; Ribeiro-Dos-Santos, R.; Fernadez-Ferreira, E.; Soares, R.; Rosas, E.; Susunaga, G.; Guimarães, A.; Zoghbi, M.; et al. Anti-inflammatory activity of Protium essential oils. J. Ethnopharmacol. 1999, 66, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Chorianopoulos, N.; Evergets, E.; Mallouchos, A.; Kalpoutzakis, E.; Nychas, G.-J.; Haroutounian, S.A. Essential oil volatiles of Satureja species. J. Agric. Food Chem. 2006, 54, 3139–3145. [Google Scholar] [CrossRef]
- Weyerstahl, P.; Marschall, H.; Splittgerber, U.; Wolf, D.; Surburg, H. Constituents of Haitian vetiver oil. Flavour Fragr. J. 2000, 15, 395–412. [Google Scholar] [CrossRef]
- Alonzo, G.; Bosco, S.F.D.; Palazzolo, E.; Saiano, F.; Tusa, N. Citrus somatic hybrid leaf essential oil. Flavour Fragr. J. 2000, 15, 258–262. [Google Scholar] [CrossRef]
- Roussis, V.; Tsoukatou, M.; Petrakis, P.V.; Chinou, I.; Skoula, M.; Harborne, J.B. Volatile constituents of Helichrysum species. Biochem. Syst. Ecol. 2000, 28, 163–175. [Google Scholar] [CrossRef]
- Monsef-Esfahani, H.R.; Miri, A.; Amini, M.; Amanzadeh, Y.; Hadjiakhoo, A.; Hajiaghaee, R.; Ajani, Y. Seasonal variations in Teucrium persicum essential oils. Res. J. Biol. Sci. 2010, 5, 492–498. [Google Scholar] [CrossRef]
- Vujišić, L.; Vučković, I.; Tešević, V.; Đoković, D.; Ristić, M.S.; Janaćković, P.; Milosavljević, S. Essential oils of Anthemis species. Flavour Fragr. J. 2006, 21, 458–461. [Google Scholar] [CrossRef]
- Flamini, G.; Tebano, M.; Cioni, P.L.; Bagci, Y.; Dural, H.; Ertugrul, K.; Uysal, T.; Savran, A. Essential oil composition in Centaurea. Plant Syst. Evol. 2006, 261, 217–228. [Google Scholar] [CrossRef]
- Sena, J.D.S.; Rodrigues, S.A.; Sakumoto, K.; Inumaro, R.S.; González-Maldonado, P.; Mendez-Scolari, E.; Piau, R.; Gonçalves, D.D.; Mandim, F.; Vaz, J.; et al. Biological activities of Tetradenia riparia essential oils. Pharmaceuticals 2024, 17, 888. [Google Scholar] [CrossRef]
- ISO 22412:2017; Particle Size Analysis—Dynamic Light Scattering (DLS). International Organization for Standardization (ISO): Geneva, Switzerland, 2017.
- Ali, H.; Al-Khalifa, A.R.; Aouf, A.; Boukhebti, H.; Farouk, A. Effect of nanoencapsulation on Origanum glandulosum essential oil. Sci. Rep. 2020, 10, 2812. [Google Scholar] [CrossRef] [PubMed]
- Feng-Lian, Y.; Zue-Gang, L.; Zhu, F. Structural characterization of nanoparticles loaded with essential garlic oil. J. Agric. Food Chem. 2009, 57, 10156–10162. [Google Scholar] [CrossRef]
- Chen, F.; Shi, Z.; Neoh, K.G.; Kang, E.T. Antioxidant and antibacterial activities of eugenol- and carvacrol-grafted chitosan nanoparticles. Biotechnol. Bioeng. 2009, 104, 30–39. [Google Scholar] [CrossRef]
- Schaffazick, S.R.; Guterres, S.S.; Freitas, L.L.; Pohlmann, A.R. Caracterização e estabilidade físico-química de sistemas nanoparticulados. Quim. Nova 2003, 26, 726–737. [Google Scholar] [CrossRef]
- Anitha, A.; Deepagan, V.G.; Divya Rani, V.V.; Menon, D.; Nair, S.; Jayakumar, R. Curcumin-loaded dextran sulphate–chitosan nanoparticles. Carbohydr. Polym. 2011, 84, 1158–1164. [Google Scholar] [CrossRef]
- Duarte, M.C.T.; Figueira, G.M.; Sartoratto, A.; Rehder, V.L.G.; Delarmelina, C. Anti-Candida activity of Brazilian medicinal plants. J. Ethnopharmacol. 2005, 97, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Jin, W.; Wang, J.; Sun, Y.; Wu, X.; Liu, L. Antibacterial and anti-biofilm activities of peppermint essential oil. LWT–Food Sci. Technol. 2019, 101, 639–645. [Google Scholar] [CrossRef]
- Kwieciński, J.; Eick, S.; Wójcik, K. Effects of tea tree oil on Staphylococcus aureus biofilms. Int. J. Antimicrob. Agents 2009, 33, 343–347. [Google Scholar] [CrossRef]
- Reichling, J.; Schnitzler, P.; Suschke, U.; Saller, R. Essential oils with antibacterial, antifungal and antiviral properties. Forsch. Komplementarmed. 2009, 16, 79–90. [Google Scholar] [CrossRef]
- Sifi, I.; Dzoyem, J.P.; Quinten, M.; Yousfi, M.; McGaw, L.J.; Eloff, J.N. Antimycobacterial and cytotoxic activities of Pistacia atlantica essential oil. Afr. J. Tradit. Complement. Altern. Med. 2015, 12, 150–155. [Google Scholar] [CrossRef]
- Vijayarathna, S.; Sasidharan, S. Cytotoxicity of Elaeis guineensis extracts. Asian Pac. J. Trop. Biomed. 2012, 2, 826–829. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.