Abstract
Background: During the COVID-19 pandemic, Thailand prioritized hemodialysis patients for vaccination. Due to limited supply, heterologous regimens were used. This study evaluates the mortality rate and risk factors in hemodialysis patients who received heterologous versus homologous vaccine regimens. Methods: We retrospectively reviewed data of hemodialysis patients in Thailand from January 2021 to December 2022, using data from the Department of Medical Sciences, Ministry of Public Health, and Thailand Renal Replacement Therapy Registry. Mortality was defined as death within 30 days of a positive RT-PCR or rapid antigen test for SARS-CoV-2. Multivariate logistic regression was used to identify mortality risk factors. Results: The associated risks of mortality in hemodialysis patients with COVID-19 were female sex, age ≥ 50 years, diabetes, and BMI ≥ 25.0 kg/m2. Regarding vaccination regimens, the inactivated–Viral vector–mRNA regimen was associated with lower mortality compared with the mRNA–mRNA regimen (OR 0.29, 95% CI 0.08–0.99). In contrast, no vaccination (OR 16.95, 95% CI 7.86–36.54) and single-dose vaccination with inactivated vaccine (OR 17.54, 95% CI 7.01–43.88) or Viral vector vaccine (OR 20.74, 95% CI 9.38–45.86) were associated with markedly higher mortality risk. Conclusion: The inactivated–Viral vector–mRNA vaccine regimen was associated with a decreased mortality risk among this population.
1. Introduction
Hemodialysis (HD) patients are at a significantly elevated risk of Coronavirus disease 2019 (COVID-19) infection. These individuals typically travel to dialysis centers two to three times per week, often relying on public transportation. Furthermore, the dialysis procedure itself often occurs in crowded settings. This combination of factors exposes them to crowded environments, thereby increasing their risk of contracting the virus [1,2]. During the pandemic, efforts to reduce crowding led some dialysis centers to decrease the number of dialysis sessions, resulting in delays in or suboptimal treatment for certain patients [3,4,5].
In addition to the higher risk of infection, HD patients who are infected with COVID-19 frequently require hospitalization due to severe symptoms. Hemodialysis is an established risk factor for severe COVID-19 symptoms, and it is associated with high mortality [6]. Mortality among HD patients with COVID-19 has been reported to be four times higher than in non-dialysis patients [4]. A meta-analysis revealed a mortality rate of 24% in this population, with the male gender, chronic obstructive pulmonary disease (COPD), and coronary artery disease (CAD) identified as significant risk factors for non-survival [7]. Proposed mechanisms for poor outcomes include accelerated immunosenescence and inflammaging, which contribute to immune dysregulation and may exacerbate COVID-19 in this population [4].
In response to the pandemic, numerous vaccines have been developed to combat COVID-19. A systematic review revealed that only 41% of dialysis patients developed antibodies after the first dose of a COVID-19 vaccine, and 89% developed antibodies after the second dose, resulting in an overall immunogenicity rate of 86% [8]. This response is lower than that observed in individuals without kidney disease, raising concerns about adequate protection against COVID-19 infection. Moreover, some studies report that a third dose may be necessary to prevent mortality [9]. In Thailand, various COVID-19 vaccination regimens have been implemented, potentially leading to different outcomes among the HD patient population.
Currently, the large data on HD patients with COVID-19 infection remains limited and often lacks detailed information on vaccine regimens. Consequently, it is challenging to provide definitive data on the importance of vaccination and the optimal number of vaccine doses for this vulnerable population.
This study aims to report on the mortality rates and risk factors associated with COVID-19 infection among a large cohort of HD patients in Thailand, with a specific focus on the details of the vaccination regimens administered. By offering a comprehensive analysis of the impact of vaccination on this vulnerable population, this study seeks to assist healthcare providers in developing more targeted strategies to mitigate the risk and severity of COVID-19 infections among HD patients, ultimately striving to enhance their overall outcomes and quality of life.
2. Material and Methods
2.1. Study Design and Participants
This retrospective study encompassed all HD patients aged 18 years and older between 1 January 2021 and 31 December 2022. Data were sourced from the Ministry of Public Health of Thailand and the Thailand Renal Replacement Therapy Registry. Patient data were anonymized prior to sharing. COVID-19 diagnoses were confirmed using PCR tests of nasopharyngeal or oropharyngeal swabs.
We collected data on patient demographics and clinical characteristics, including age, gender, comorbidities, body mass index (BMI), hemoglobin (Hb) levels, serum albumin, HD adequacy, admission ward, and COVID-19 vaccination status. The primary objective was to identify the risk factors for mortality, which was defined as mortality within 30 days after the first positive PCR test, in HD patients with COVID-19. Secondary objectives included evaluating the benefits of different vaccination doses and regimens on all-cause mortality.
During the study period, COVID-19 vaccination in Thailand followed national policy from the Ministry of Public Health and the Department of Disease Control. Individuals at high risk of severe COVID-19 outcomes, including adults aged ≥ 60 years and those with major chronic diseases such as chronic kidney disease stage 5 or receiving maintenance dialysis, were prioritized for vaccination. Because vaccine supply varied during the national rollout, different vaccine platforms were administered according to availability, and heterologous vaccination regimens were implemented as part of the national vaccination strategy. The available vaccines in Thailand included inactivated vaccine [CoronaVac (Sinovac® (Sinovac Biotech Ltd., Beijing, China) and Sinopharm® (Beijing Institute of Biological Products, Beijing, China)], Viral vector vaccine [ChAdOx1 (AstraZeneca plc, Cambridge, UK)], mRNA vaccine [BNT162b2 (Comirnaty® (Pfizer Inc., New York, NY, USA and BioNTech Manufacturing GmbH, Mainz, Germany), mRNA-1273 (Moderna, Moderna Inc., Cambridge, MA, USA)], protein-based vaccine [NVX-CoV2373 (Covovax®, Serum Institute of India Pvt. Ltd., Pune, India)], and Ad26.COV2.S (AdVac® (Johnson & Johnson, Leiden, The Netherland).
2.2. Statistical Analysis
Numerical data were reported as means with standard deviations (SD) or medians with interquartile ranges (IQR), as appropriate. Categorical variables were presented as frequencies and percentages. To evaluate differences between the two groups, Fisher’s exact test was used for categorical data, and Wilcoxon’s rank-sum test was applied for continuous data.
Logistic regression analyses, both univariate and multivariate, were conducted to determine the risk factors for mortality. Variables suspected of being associated with mortality were included in the multivariate logistic regression analysis. In addition, multivariable Cox proportional hazards regression analysis was performed to evaluate the association between vaccination regimens and 30-day mortality, adjusting for the same covariates included in the multivariate logistic regression model. Hazard ratios (HRs) with 95% confidence intervals (CIs) were reported. Statistical significance was defined as a p-value less than 0.05. All statistical analyses were performed using Stata version 17.0/BE (StataCorp, College Station, TX, USA).
3. Results
3.1. Baseline Characteristics
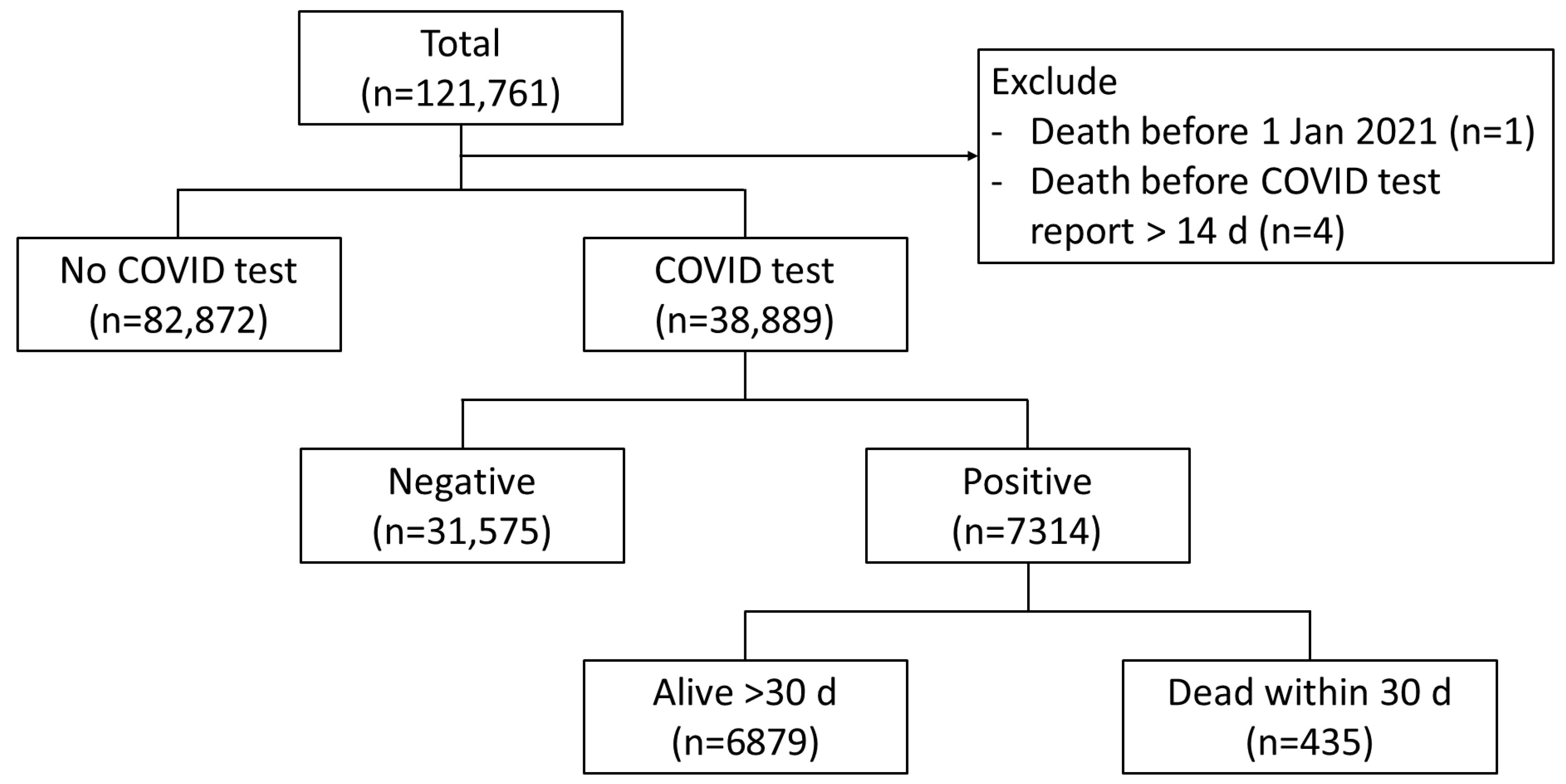
In a cohort of 121,761 HD patients, 7314 individuals (6.0%) were infected with COVID-19. The mean age of the infected patients was 58.1 ± 14.5 years. The 30-day mortality rate among HD patients with COVID-19 was observed to be 6% (Figure 1). Notably, the highest prevalence of COVID-19 infection was recorded in the central region, where non-surviving COVID-19 HD patients were predominantly located.
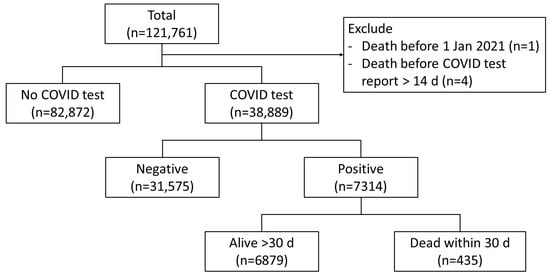
Figure 1.
Study flow.
The non-survival group exhibited several distinguishing characteristics: they were generally older and had a higher prevalence of diabetes and diabetic nephropathy (DN). A significant proportion of these patients were admitted to cohort wards. Additionally, this group demonstrated lower levels of serum albumin, normalized protein catabolic rate (nPCR), and spKt/V, alongside a higher BMI (Table 1). Additional data categorized by pandemic waves are presented in Supplementary Table S1.

Table 1.
Baseline characteristics.
The majority of deaths among infected patients occurred prior to the administration of the first dose of the COVID-19 vaccine. Following the commencement of vaccination, a decline in mortality rates was observed, corresponding with the increase in the number of vaccine doses administered (Table 1).
3.2. Risk-Associated Mortality Among HD Patients with COVID-19 Infection
In a univariate analysis of HD patients with COVID-19, factors associated with increased mortality risk included female gender, advanced age, diabetes, BMI ≥ 25 kg/m2, and Hb > 11.5 g/dL. In multivariate analysis, the factors significantly associated with increased mortality were the female gender, advanced age, diabetes, and BMI ≥ 25 kg/m2 (Table 2).

Table 2.
Mortality risk in HD patients with COVID-19.
The protective factor identified in univariate and multivariate analyses was the number of vaccinations received prior to COVID-19 infection. Notably, an increasing number of vaccine doses was associated with a decreased mortality rate following COVID-19 infection (Table 2).
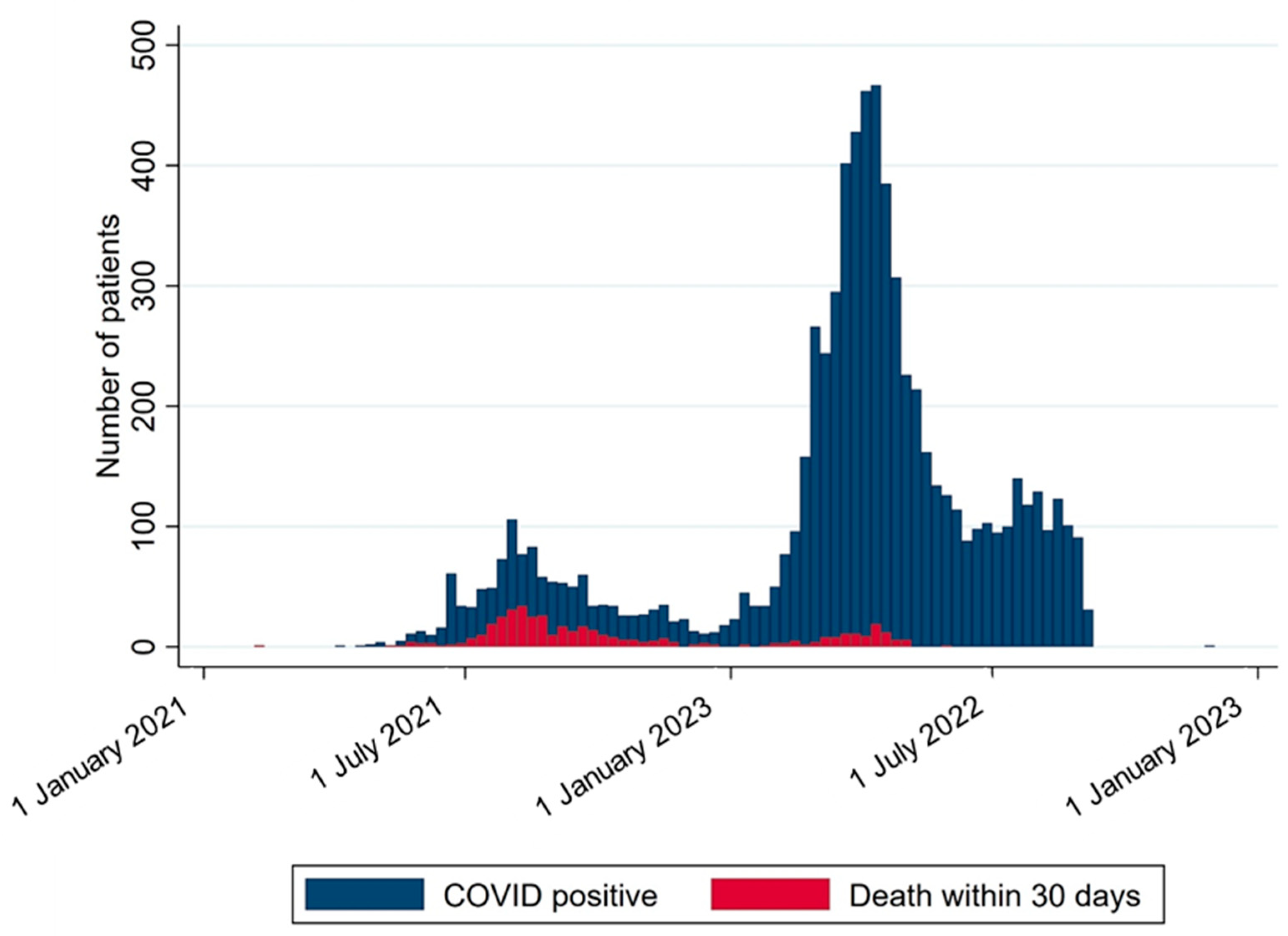
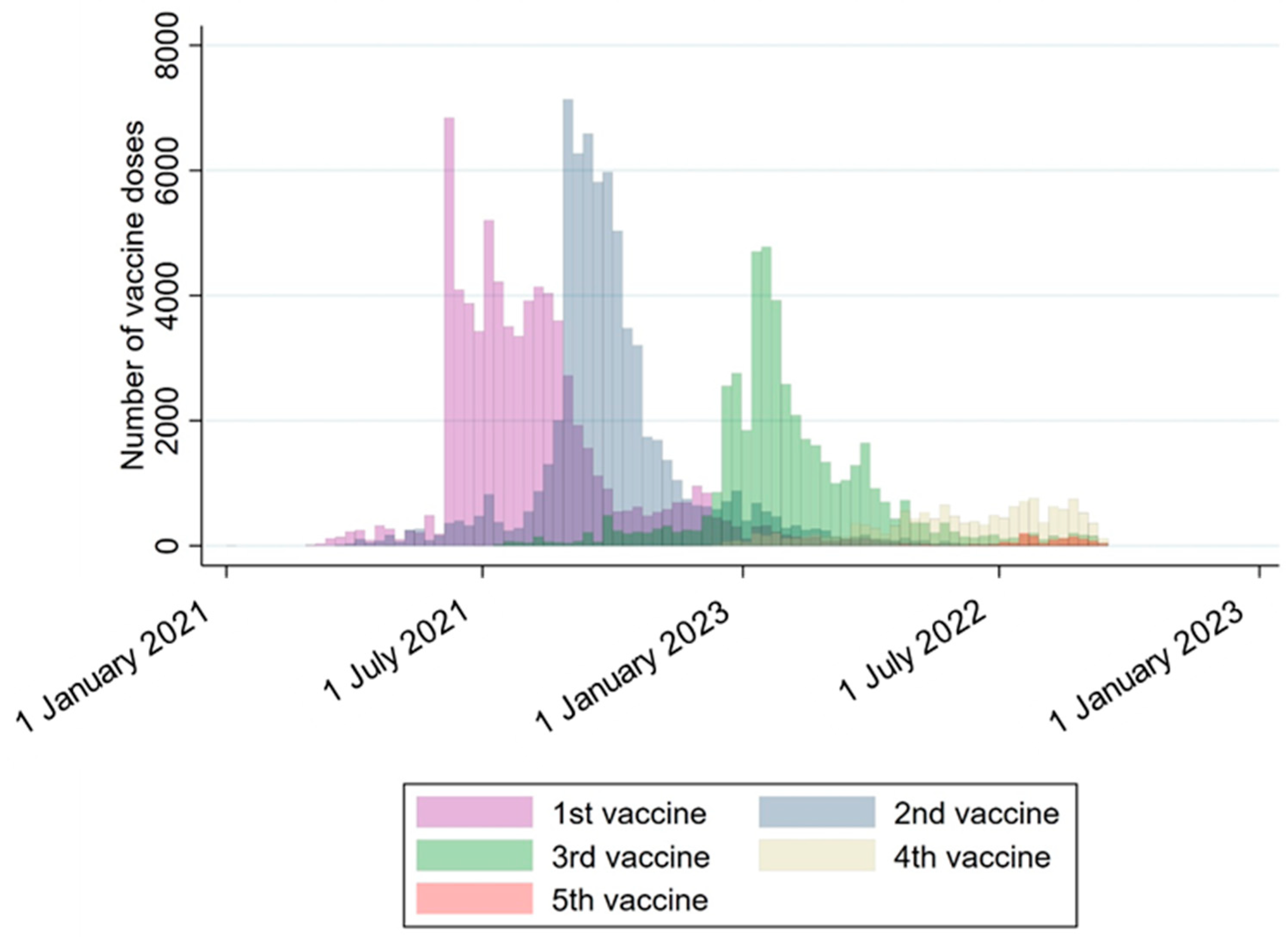
Between 1 January 2021 and 31 December 2022, there were two waves of COVID-19 infection among HD patients. However, the proportion of deaths was lower in the second wave (Figure 2). During the second wave, there was a large number of patients who received the third dose of the COVID-19 vaccine (Figure 3).
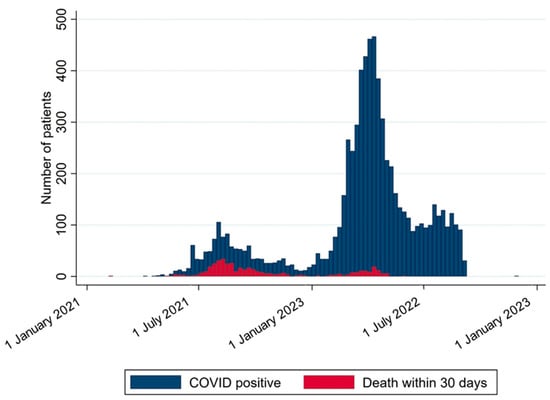
Figure 2.
The number of infections and non-surviving patients at each time point. This figure illustrates the number of hemodialysis patients who were infected with COVID-19 and the number of those who died within 30 days after the infection during the follow-up period from 1 January 2021 to 31 December 2022. The blue bars represent the total number of infected patients, while the red bars indicate the number of deaths. Each bar corresponds to a one-week interval.
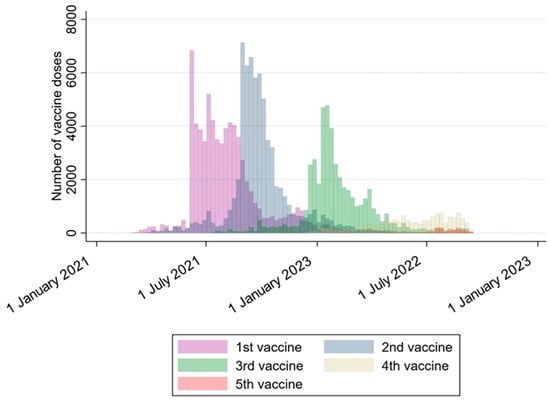
Figure 3.
The number of vaccine doses at each time point. This figure illustrates the total number of vaccine doses administered between 1 January 2021 and 31 December 2022. Different colors are used to distinguish the sequence of doses, including the 1st, 2nd, 3rd, 4th, and 5th doses. Each bar represents a one-week interval. The majority of vaccines were distributed as the 1st, 2nd, and 3rd doses.
3.3. The Efficacy of Each Vaccination Regimen
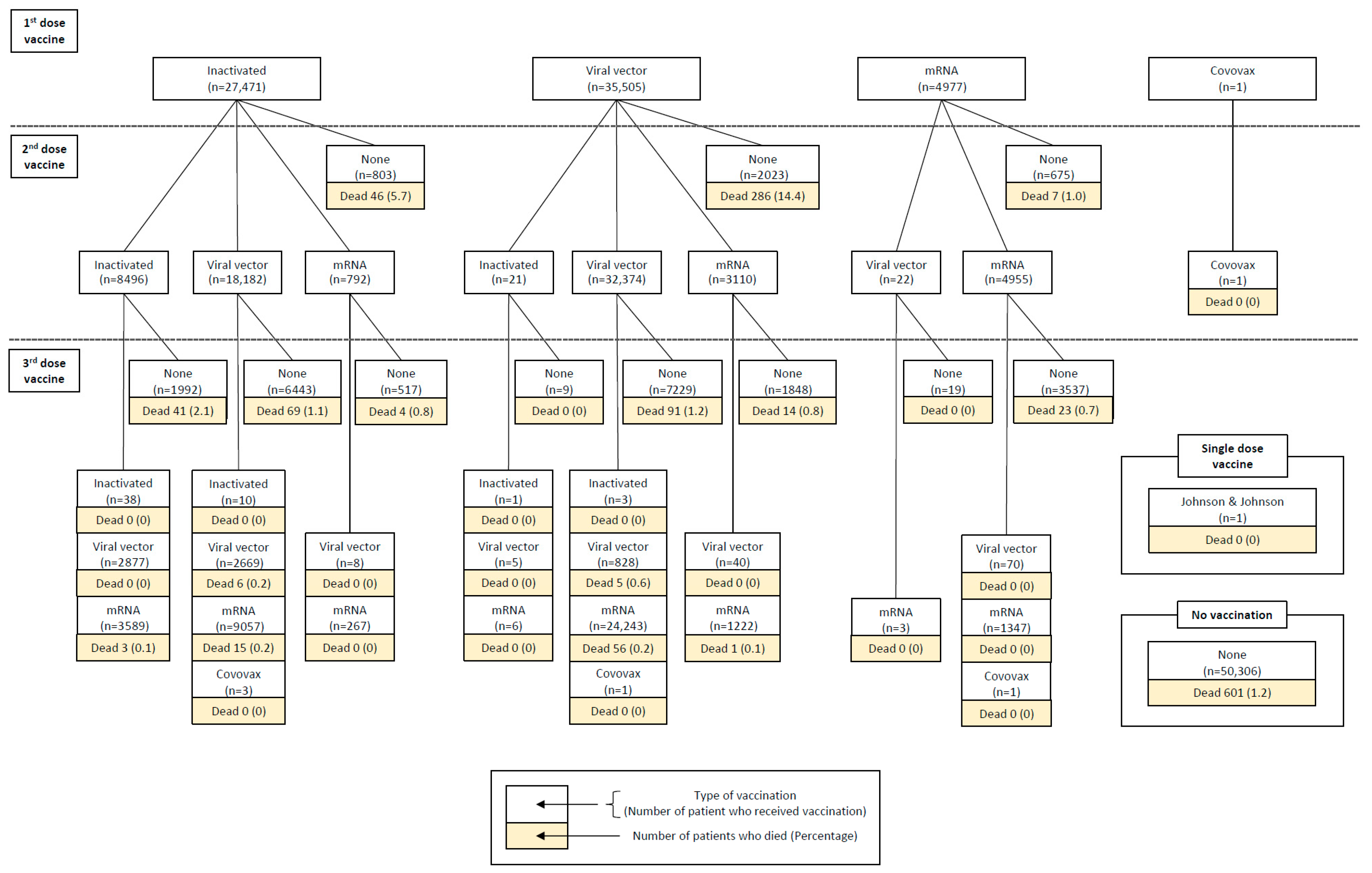
Among 121,761 HD patients, 67,954 individuals (55.8%) received at least one dose of the vaccine. All vaccination regimens demonstrated a subsequent decrease in mortality with each additional dose, approaching near-zero mortality after the third dose with any regimen. Notably, after the first dose, an mRNA vaccine was associated with the lowest mortality rate from the outset. The mortality rate appeared to be comparable across all regimens but remained highest in the inactivated–inactivated regimen (Figure 4).
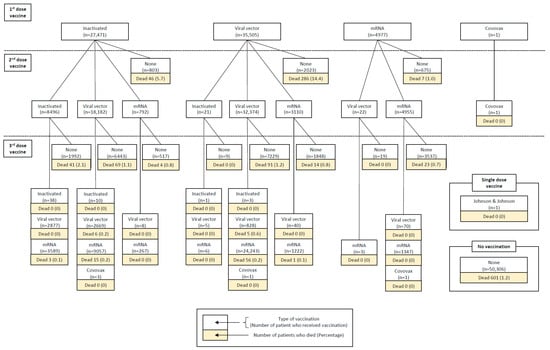
Figure 4.
Vaccine regimens and mortality. This figure represents the number of vaccinated hemodialysis patients between 1 January 2021 and 31 December 2022. A total of 67,954 hemodialysis patients received vaccination. In each block, the upper section represents the total number of vaccinated patients, while the lower section indicates the number of patients who died from any cause. Each step in the figure illustrates subsequent vaccine doses, categorized by vaccine type. The maximum number of doses displayed is three, as the proportion of patients receiving a 4th or 5th dose was too small to be shown. The distribution of deaths by specific vaccine regimens during each epidemic wave is presented in Supplementary Tables S2 and S3.
However, after adjusting for other factors, including gender, age, comorbidities, BMI, Hb, serum albumin, and Kt/V, the inactivated–Viral vector–mRNA regimen significantly reduced mortality compared to the mRNA–mRNA regimen (OR 0.29, 95% CI 0.08–0.99, p-value 0.049). In contrast, the patients who received no vaccination or a single dose of an inactivated or Viral vector vaccine had an increased risk of mortality (Table 3).

Table 3.
Vaccine regimens and mortality.
Multivariable Cox regression analysis, adjusting for the same covariates as the multivariable logistic regression model, demonstrated that patients who received no vaccination, a single dose of an inactivated vaccine, or a Viral vector vaccine had a significantly increased risk of 30-day mortality. In contrast, the inactivated–Viral vector–mRNA regimen was not associated with a statistically significant reduction in mortality compared with the mRNA–mRNA regimen (HR 0.40, 95% CI 0.11–1.49, p = 0.17) (Table 4).

Table 4.
Multivariable Cox proportional hazards analysis for 30-day mortality among hemodialysis patients with COVID-19.
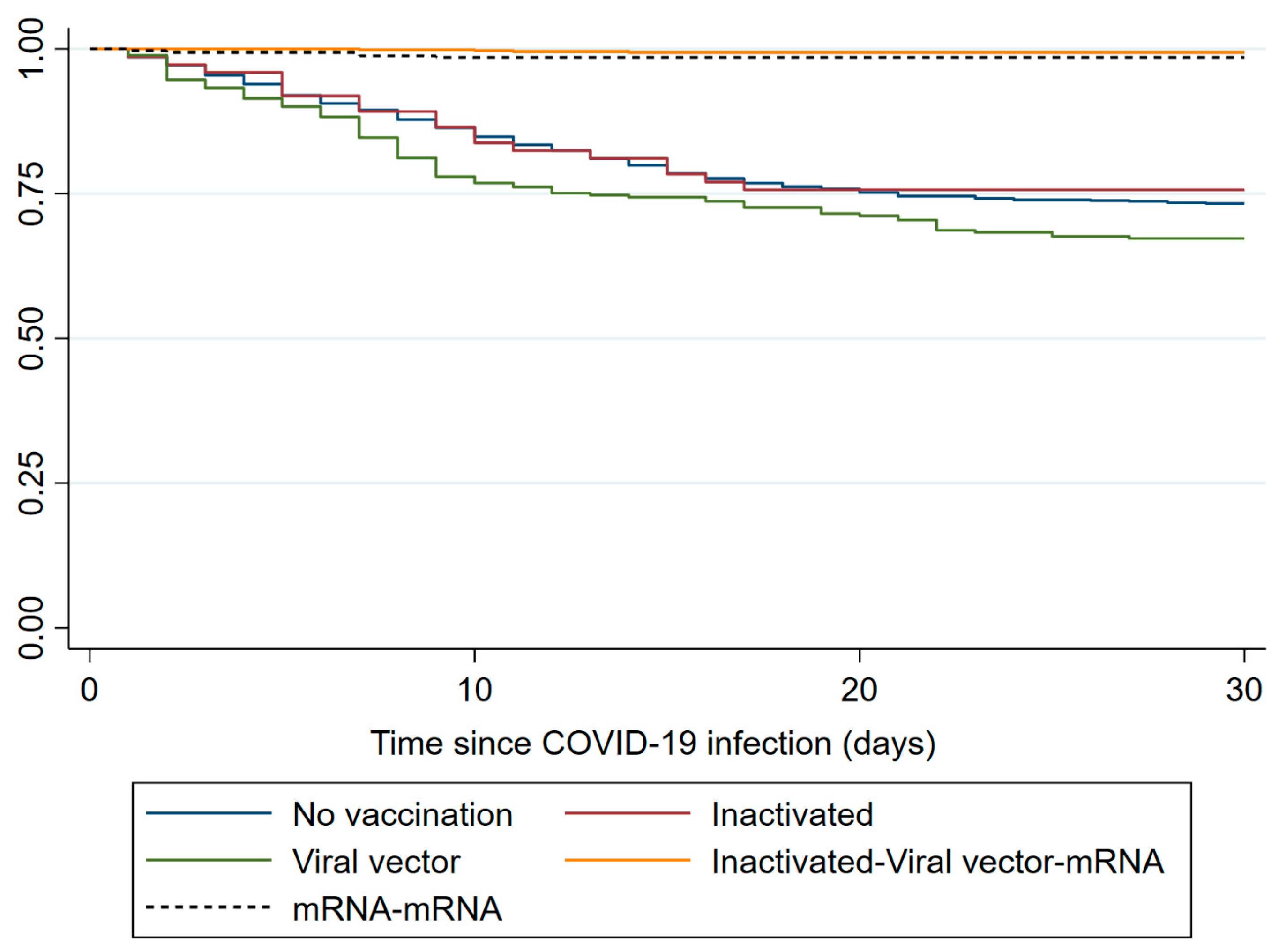
The Kaplan–Meier survival curves (Figure 5) illustrate differences in 30-day survival across vaccination regimens. Patients who received no vaccination or only Viral vector vaccination showed lower survival probabilities over time. The inactivated–Viral vector–mRNA regimen demonstrated a trend toward improved survival compared with the mRNA–mRNA regimen; however, this difference did not reach statistical significance in the adjusted Cox regression model.
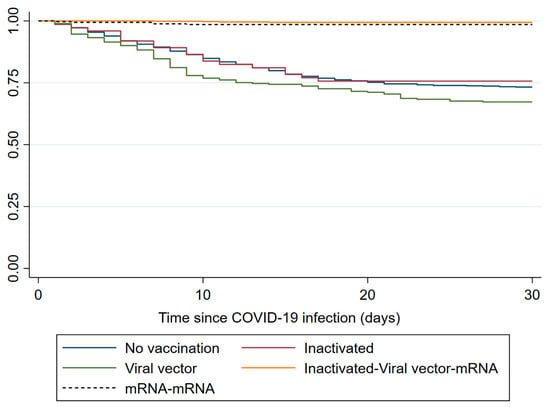
Figure 5.
Kaplan–Meier survival curves for 30-day mortality according to COVID-19 vaccination regimen in hemodialysis patients.
4. Discussion
Our study demonstrated that the COVID-19 infection rate among HD patients was 6%, and the mortality rate was also 6%. The risk factors for mortality among HD patients with COVID-19 included female gender, advanced age, diabetes, and a BMI ≥ 25 kg/m2. Most non-survivors were infected before receiving the first dose of the vaccine. Our data showed that the inactivated–Viral vector–mRNA regimen provided the most protective benefit compared to the two-dose mRNA regimen.
Based on the baseline characteristics, our study found that most non-surviving patients were initially admitted to cohort wards rather than the intensive care unit (ICU), where more severe cases are typically managed. This pattern suggests that patients who deteriorated significantly may have been transferred to the ICU after initial admission. Additionally, during the pandemic, ICU capacity, which requires negative-pressure rooms, was limited in our country, leading to a preference for admitting patients to cohort wards initially. Furthermore, a notable proportion of non-surviving patients were admitted to home isolation, possibly due to delays in recognizing the severity of their condition or the need for palliative care. These factors may explain why many non-surviving patients were not admitted to the ICU. However, we did not have data on patient transfers or informed consent for palliative care.
There is considerable variability in mortality rates reported across studies. For instance, Can et al. [10] reported an infection rate of 23% among 151 HD patients, while Min et al. [11] found an infection rate of 11.8% among 627 HD patients. Similarly, Turgutalp et al. [12] reported a 16.3% infection rate among 567 HD patients. All these studies were conducted in 2020. However, subsequent studies reported a decrease in mortality rates. Tylicki et al. [13] observed a reduction to 6.7% during the fourth wave (September 2021–January 2022), and Shuller et al. [14] reported a further decline to 1.2% in the fifth wave (January 2022–August 2022). The variation in infection rates across studies may be attributed to differences in prevention strategies, including vaccine implementation and COVID-19 variants.
Our study, conducted between 2021 and 2022, benefited from established prevention measures such as social distancing, reorganizing HD schedules to reduce crowding, and, in some instances, decreasing the number of HD sessions to avoid overcrowding. Furthermore, our cohort comprised all HD patients nationwide, allowing for the generalization of protective strategies and potentially reflecting the overall effectiveness of these prevention measures.
During the study period, Thailand implemented a risk-based COVID-19 vaccination strategy that prioritized individuals at high risk of severe disease and mortality, including older adults and patients with major chronic diseases such as chronic kidney disease stage 5 and those receiving maintenance dialysis. Consequently, HD patients were eligible for vaccination throughout the rollout period and were prioritized regardless of age. Because vaccine availability varied over time during the early phases of the national vaccination program, the type of vaccine administered was often determined by supply rather than strict allocation by age group. This policy also contributed to the widespread use of heterologous vaccination regimens in Thailand. Therefore, the distribution of vaccine platforms observed in our cohort likely reflects vaccine availability during different phases of the pandemic rather than systematic age-based vaccine selection. Nevertheless, temporal changes in vaccine availability and policy could still introduce residual confounding.
During the COVID-19 pandemic in Thailand, the dominant SARS-CoV-2 variants changed over time across successive epidemic waves. The first wave in March 2020 was primarily associated with early SARS-CoV-2 lineages, including A and B.1. The second wave, beginning in January 2021, was mainly driven by lineage B.1.36.16. This was followed by the third wave in April 2021, during which the B.1.1.7 (Alpha) variant became predominant. Subsequently, the fourth wave in July 2021 was dominated by the B.1.617.2 (Delta) variant. In early 2022, the Omicron variant emerged and drove the fifth wave, with the B.1.1.529 Omicron BA.1 sublineage predominating in January 2022 and later being replaced by the BA.2 sublineage in March 2022 [15]. These patterns are consistent with publicly available genomic surveillance data from the CoVariants platform [16]. However, because variant sequencing was not available for individual patients in our cohort, this data may not fully represent the specific variants infecting patients undergoing hemodialysis.
The risk factors for mortality in this study may differ from those identified in other studies. Zeng et al. [6] reported that the male gender, advanced age, and chronic diseases were significant risk factors for mortality. The ERA-EDTA registry [17] also highlighted the male gender and older age as major risk factors. Additionally, Li et al. [7] conducted a meta-analysis showing that the male gender, CAD, and COPD were associated with higher mortality rates. In contrast, our study found that the female gender was associated with a higher risk of death, which differs from the aforementioned studies. However, our findings regarding advanced age as a risk factor are consistent with other studies. We also found that high BMI and diabetes were significant risk factors.
Age, BMI, and diabetes remain significant risk factors for mortality even with vaccine implementation. Jahn et al. [18]. reported a reduced immune response after two doses of an mRNA vaccine in patients over 60 years of age. Similarly, diabetes and high BMI have been associated with delayed innate and adaptive immune responses [19,20]. These factors may contribute to the continued elevated risk despite vaccination.
The number of vaccine doses is a crucial factor for HD patients. As is widely recognized, many HD patients have compromised immune responses, including deficiencies in both innate and adaptive immunity, which impair their ability to mount a robust vaccine response. Guipil et al. [21] found that HD patients with a history of COVID-19 infection exhibited reduced immunity following the first dose of an mRNA vaccine. They reported that naïve HD patients required a second vaccine dose to achieve a significant increase in cellular immune responses, as measured by IL-2 and IFN-γ secretion, with a similar need for a second dose to enhance the humoral response. In Taiwan, Chen et al. [22] demonstrated that a third vaccine dose resulted in the highest antibody levels and lowest mortality rates compared to individuals who did not complete all three doses, aligning with Brkovic et al. [9]’s recommendation that at least three doses of vaccination are necessary to effectively reduce mortality. Our findings support this observation that while the first vaccine dose may lower mortality, it is not the most effective. Subsequent doses appear to be more effective, likely due to the initial low immune response elicited by the first dose and the enhanced immune response generated by subsequent doses, which can significantly reduce mortality.
Moreover, our results demonstrated that, despite vaccination, there remained a high rate of COVID-19 infection among patients. However, the proportion of non-surviving patients decreased with each subsequent dose of the vaccine. This suggests that, although patients continued to be at risk of infection post-vaccination, the severity of the disease declined, correlating with the observed reduction in mortality, aligning with Korst’s report [23].
In Thailand, various vaccine regimens have been utilized. However, the majority of HD patients received three doses of the vaccine, with the predominant regimens being Viral vector–Viral vector–mRNA. Our findings indicate that the inactivated–Viral vector–mRNA regimen was the most effective in reducing mortality, with the mRNA–mRNA regimen used as a reference reflecting the global standard. Due to its limited application within our population, the mRNA–mRNA regimen was not directly compared in our study.
It is important to acknowledge that our results may be subject to bias, as the distribution of patients across different vaccine regimens was not evenly balanced. This imbalance could impact the observed outcomes and should be considered when interpreting the findings. Nonetheless, our study confirmed that both no vaccination and a single vaccine dose were associated with higher mortality.
The strengths of our study include the large cohort encompassing all HD patients in Thailand, which allows for generalization to our specific ethnicity. Additionally, we provided detailed information on vaccine regimens, which is particularly limited in HD patients. This highlights the importance of vaccination in this specific group. Lastly, our study demonstrated the benefits of various vaccine regimens, showing that any regimen decreases mortality with subsequent doses. However, our study has some limitations. We were unable to categorize the severity of surviving patients’ conditions based on their admission site, as the admission location does not accurately reflect severity. There was also an imbalance in the distribution of vaccine regimens, which prevents us from definitively identifying the best regimen. Moreover, we did not adjust for time since vaccination, which limits the interpretation of the onset and durability of vaccine-induced protection. Potential confounding by calendar period, infection wave, and baseline disease severity may also have influenced the observed associations. Moreover, changes in dominant SARS-CoV-2 variants during the pandemic in Thailand may have influenced mortality outcomes. Because data regarding specific viral variants were not available in our dataset, the observed differences in mortality between vaccine regimens may partly reflect the impact of circulating variants during different periods of the study. Furthermore, changes in quarantine policies and healthcare system organization during the pandemic may have influenced mortality outcomes. Modifications in dialysis service delivery, patient management protocols, and access to care over time could have affected clinical outcomes and should be considered when interpreting the results. Therefore, although our findings suggest potential differences in mortality among vaccine regimens, these results should be interpreted with caution, and further studies are needed to confirm the superiority of any specific vaccination strategy.
5. Conclusions
COVID-19 infection in HD patients who are female, elderly, have diabetes, or have a high BMI is associated with a high risk of mortality. However, each additional vaccine dose helps reduce mortality. The inactivated–Viral vector–mRNA vaccine regimen was associated with a decreased mortality risk among this population.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/covid6030049/s1. Table S1: Baseline characteristics of hemodialysis patients across COVID-19 epidemic waves; Table S2.: Distribution of deaths by three-dose COVID-19 vaccine regimen across epidemic waves among hemodialysis patients. Data are presented as number of deaths and percentage within each vaccine regimen; Table S3: Distribution of deaths by five-dose COVID-19 vaccine regimen across epidemic waves among hemodialysis patients. Data are presented as number of deaths and percentage within each vaccine regimen.
Author Contributions
P.P., S.A., M.R., S.R., S.S. and T.S. contributed to conceptualization. P.P. performed data curation. P.P. and S.A. carried out formal analysis. T.S. was responsible for funding acquisition. S.S., T.S. and the Nephrology Society of Thailand conducted the investigation. P.P., S.A., M.R., S.R., S.S. and T.S. developed the methodology. S.S. and T.S. managed project administration and provided resources. S.S. and T.S. supervised the project. P.P., S.A., S.S. and T.S. performed validation. P.P. and S.A. prepared the visualization. P.P. and S.A. wrote the original draft. P.P., S.A., M.R., S.R., S.S. and T.S. reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Foundation of Thai Gerontology Research and Development Institute (TGRI).
Institutional Review Board Statement
This study was conducted in accordance with the Declaration of Helsinki and approved by Siriraj Institutional Review Board, Mahidol University, under SIRB Protocol number MU-MOU 383/2565(IRB1) (13 May 2024). Informed consent was waived due to the retrospective cohort study design.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
We extend our gratitude to the Thailand Renal Registry Therapy Registry and the Department of Medical Sciences, Ministry of Public Health, Thailand, for providing the data essential for use in this research. We used ChatGPT-4o solely for grammar-checking purposes and did not utilize it for analysis or creation of any other part of the research.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Ikizler, T.A. COVID-19 and Dialysis Units: What Do We Know Now and What Should We Do? Am. J. Kidney Dis. 2020, 76, 1–3. [Google Scholar] [CrossRef]
- Basile, C.; Combe, C.; Pizzarelli, F.; Covic, A.; Davenport, A.; Kanbay, M.; Kirmizis, D.; Schneditz, D.; van der Sande, F.; Mitra, S. Recommendations for the prevention, mitigation and containment of the emerging SARS-CoV-2 (COVID-19) pandemic in haemodialysis centres. Nephrol. Dial. Transplant. 2020, 35, 737–741. [Google Scholar] [CrossRef]
- Weiner, D.E.; Watnick, S.G. Hemodialysis and COVID-19: An Achilles’ Heel in the Pandemic Health Care Response in the United States. Kidney Med. 2020, 2, 227–230. [Google Scholar] [CrossRef]
- El Karoui, K.; De Vriese, A.S. COVID-19 in dialysis: Clinical impact, immune response, prevention, and treatment. Kidney Int. 2022, 101, 883–894. [Google Scholar] [CrossRef] [PubMed]
- Anumas, S.; Kunawathanakul, S.; Tantiyavarong, P.; Krisanapan, P.; Pattharanitima, P. Predictors for Unsuccessful Reductions in Hemodialysis Frequency during the Pandemic. J. Clin. Med. 2023, 12, 2550. [Google Scholar] [CrossRef]
- Zeng, B.; Zhou, J.; Peng, D.; Dong, C.; Qin, Q. The prevention and treatment of COVID-19 in patients treated with hemodialysis. Eur. J. Med. Res. 2023, 28, 410. [Google Scholar] [CrossRef]
- Li, P.; Guan, Y.; Zhou, S.; Wang, E.; Sun, P.; Fei, G.; Zeng, D.; Wang, R. Mortality and risk factors for COVID-19 in hemodialysis patients: A systematic review and meta-analysis. Sci. Prog. 2022, 105, 368504221110858. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Liu, C.F.; Shen, Y.T.; Kuo, Y.-T.; Ko, C.-C.; Chen, T.-Y.; Wu, T.-C.; Shih, Y.-J. Development of real-time individualized risk prediction models for contrast associated acute kidney injury and 30-day dialysis after contrast enhanced computed tomography. Eur. J. Radiol. 2023, 167, 111034. [Google Scholar] [CrossRef]
- Brkovic, V.; Nikolic, G.; Baralic, M.; Kravljaca, M.; Milinkovic, M.; Pavlovic, J.; Lausevic, M.; Radovic, M. A Study on Mortality Predictors in Hemodialysis Patients Infected with COVID-19: Impact of Vaccination Status. Vaccines 2024, 12, 2. [Google Scholar] [CrossRef] [PubMed]
- Can, Ö.; Bilek, G.; Sahan, S. Risk factors for infection and mortality among hemodialysis patients during COVID-19 pandemic. Int. Urol. Nephrol. 2022, 54, 661–669. [Google Scholar] [CrossRef]
- Min, Y.; Cheng, L.; Tu, C.; Li, H.; He, D.; Huang, D.; Chen, D.; Huang, X.; Chen, F.; Xiong, F. Clinical characteristics of deceased hemodialysis patients affected by COVID-19. Int. Urol. Nephrol. 2021, 53, 797–802. [Google Scholar] [CrossRef]
- Turgutalp, K.; Ozturk, S.; Arici, M.; Eren, N.; Gorgulu, N.; Islam, M.; Uzun, S.; Sakaci, T.; Aydin, Z.; Sengul, E.; et al. Determinants of mortality in a large group of hemodialysis patients hospitalized for COVID-19. BMC Nephrol. 2021, 22, 29. [Google Scholar] [CrossRef] [PubMed]
- Tylicki, L.; Biedunkiewicz, B.; Puchalska-Reglińska, E.; Gellert, R.; Burnier, M.; Wolf, J.; Dȩbska-Ślizień, A. COVID-19 vaccination reduces mortality in patients on maintenance hemodialysis. Front. Med. 2022, 9, 937167. [Google Scholar] [CrossRef]
- Schuller, M.; Ginthör, N.E.; Paller, A.; Waller, M.; Köstenbauer, M.; Schreiber, N.G.O.; Schabhüttl, C.; Mischinger, K.; Hafner-Giessauf, H.; Rosenkranz, A.R.; et al. Reduced COVID-19 morbidity and mortality in hemodialysis patients across the various Omicron sublineages-A retrospective analysis. Front. Public Health 2023, 11, 1218188. [Google Scholar] [CrossRef]
- Puenpa, J.; Rattanakomol, P.; Saengdao, N.; Chansaenroj, J.; Yorsaeng, R.; Suwannakarn, K.; Thanasitthichai, S.; Vongpunsawad, S.; Poovorawan, Y. Molecular characterisation and tracking of severe acute respiratory syndrome coronavirus 2 in Thailand, 2020–2022. Arch. Virol. 2023, 168, 26. [Google Scholar] [CrossRef] [PubMed]
- CoVariants: Per Country. Available online: https://covariants.org/per-country (accessed on 10 March 2026).
- Jager, K.J.; Kramer, A.; Chesnaye, N.C.; Couchoud, C.; Sánchez-Álvarez, J.E.; Garneata, L.; Collart, F.; Hemmelder, M.H.; Ambühl, P.; Kerschbaum, J.; et al. Results from the ERA-EDTA Registry indicate a high mortality due to COVID-19 in dialysis patients and kidney transplant recipients across Europe. Kidney Int. 2020, 98, 1540–1548. [Google Scholar] [CrossRef]
- Jahn, M.; Korth, J.; Dorsch, O.; Anastasiou, O.E.; Sorge-Hädicke, B.; Tyczynski, B.; Gäckler, A.; Witzke, O.; Dittmer, U.; Dolff, S.; et al. Humoral Response to SARS-CoV-2-Vaccination with BNT162b2 (Pfizer-BioNTech) in Patients on Hemodialysis. Vaccines 2021, 9, 360. [Google Scholar] [CrossRef]
- Wilder-Smith, A.; Frahsa, A. Impact of BMI on COVID-19 vaccine effectiveness. Lancet Diabetes Endocrinol. 2022, 10, 551–552. [Google Scholar] [CrossRef] [PubMed]
- He, Y.F.; Ouyang, J.; Hu, X.D.; Wu, N.; Jiang, Z.-G.; Bian, N.; Wang, J. Correlation between COVID-19 vaccination and diabetes mellitus: A systematic review. World J. Diabetes 2023, 14, 892–918. [Google Scholar] [CrossRef]
- Gonzalez-Perez, M.; Montes-Casado, M.; Conde, P.; Cervera, I.; Baranda, J.; Berges-Buxeda, M.J.; Perez-Olmeda, M.; Sanchez-Tarjuelo, R.; Utrero-Rico, A.; Lozano-Ojalvo, D.; et al. Development of Potent Cellular and Humoral Immune Responses in Long-Term Hemodialysis Patients After 1273-mRNA SARS-CoV-2 Vaccination. Front. Immunol. 2022, 13, 845882. [Google Scholar] [CrossRef]
- Chen, T.H.; Ho, Y.; Tai, H.L.; Chu, Y.-C.; Lin, Y.-P.; Yang, C.-Y.; Tseng, W.-C.; Ou, S.-M.; Tsai, M.-T.; Chen, J.-Y.; et al. Survival benefit of a third dose of the COVID-19 vaccine among hemodialysis patients: A prospective cohort study. J. Microbiol. Immunol. Infect. 2023, 56, 1198–1206. [Google Scholar] [CrossRef] [PubMed]
- Korst, U.K.H. COVID-19 Vaccination for Patients Undergoing Long-Term Hemodialysis. Clin. J. Am. Soc. Nephrol. 2022, 17, 767–768. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.