Abstract
Tick-borne diseases are rising across Europe, yet human-biting tick data from the central Balkans remain limited. We analysed ticks removed from patients seeking care after tick bites at centers in Skopje (North Macedonia) and Novi Sad (Serbia) between January 2022 and December 2024, recording species, developmental stage, month of removal, and anatomical attachment site. A total of 1641 ticks were collected (North Macedonia, n = 157; Serbia, n = 1484) and identified morphologically. Six tick species were detected in North Macedonia and eight in Serbia. Ixodes ricinus was dominant in both countries, comprising ~69% of ticks in North Macedonia and >90% in Serbia, while Hyalomma marginatum was found in North Macedonia. Adult females predominated among ticks in North Macedonia (75.8%), whereas nymphs were most frequent among ticks in Serbia (55.6%); between-center comparisons of developmental stage should be interpreted cautiously given the marked sampling imbalance. In both settings, collections peaked in May–June (~64% of annual ticks), indicating highest bite pressure in late spring/early summer. Attachment sites were most commonly the torso and lower extremities, with significant between-country differences and species-/stage-specific patterns most evident in Serbia. These data refine the epidemiology of human tick exposure in two Balkan countries and support targeted seasonal prevention messaging and strengthened regional surveillance.
1. Introduction
Ticks are blood-feeding ectoparasites that play a critical role as vectors of numerous pathogens affecting humans. They transmit the agents of many diseases, such as Lyme borreliosis (LB), spotted-fever-group (SFG) rickettsiosis, tick-borne encephalitis (TBE), Crimean-Congo hemorrhagic fever (CCHF), and others, impacting public health worldwide [1]. The castor bean tick Ixodes ricinus is the most common human-biting tick species in central and western Europe and serves as the primary vector for LB, TBE, and several other tick-borne pathogens (TBPs) [2]. The incidence and geographic range of tick-borne diseases (TBDs) have increased in many parts of Europe [3] due to climate change, changing land use, and wildlife population shifts on the one side and improved TBP and TBD detection protocols and technological advancement [4,5,6]. This trend has increased the concern about tick bites and associated TBDs, underscoring the need for updated epidemiological data and preventive strategies.
The Balkan Peninsula represents a complex transitional zone between central and southern Europe, characterized by diverse climates, vegetation, and fauna that support a wide range of tick species and TBPs [7]. According to a comprehensive regional review, at least 32 ixodid tick species have been reported across the Western Balkans, including members of the genera Ixodes, Haemaphysalis, Dermacentor, Rhipicephalus, and Hyalomma [8]. These findings position the Balkans as an emerging hotspot for TBDs, where environmental changes, animal movements, and limited surveillance increase the complexity of pathogen transmission.
Within this regional context, Serbia and North Macedonia occupy a particularly important ecological and epidemiological position of the central Balkans, shaped both by their high tick species diversity, where seroprevalence studies revealed the opposing north–south gradients in IgG against Borrelia spp. and Rickettsia spp. among tick-infested and general population. More precisely, it was found that Borrelia exposure toward the south is decreasing, while SFGR exposure in the same direction increases [9]. Data from the region show that ticks removed from humans in Serbia and North Macedonia frequently carry multiple TBPs, including Borrelia afzelii, Anaplasma phagocytophilum and SFGR [7,10]. In addition, it was observed that more than 8% of tick infested individuals develop subclinical bacteremia, confirming a substantial hidden burden of tick-borne infection [7]. However, despite the growing body of serological and molecular evidence [9,10,11], studies specifically focusing on ticks removed from humans, their species composition, life stages, and patterns of body-site attachment, remain limited in both Serbia and North Macedonia [12,13,14].
Tick fauna in North Macedonia comprises about 15 recorded hard-tick species, yet decades without systematic surveys have left major gaps in national biodiversity data, whereas Serbia’s fauna is better characterized, with at least 21 ixodid species reported [8]. Seasonal tick activity in temperate climates peaks in warmer months, and the risk of bites correlates with human outdoor activity patterns. The anatomical location of tick attachment also varies with host age and behavior: adults are most often bitten on the lower extremities, while children are more frequently bitten on the head and neck [12,15]. Whether certain tick species or developmental stages in Balkans show preferences for particular attachment sites remains poorly understood.
This study aimed to provide an updated comparative analysis of ticks removed from humans in Serbia and North Macedonia during the period from 2022 to 2024. The objectives were to determine the diversity and seasonal distribution of human-biting ticks and to assess whether specific tick species or life stages show preferences for particular anatomical attachment sites. The overarching goal was to improve understanding of human–tick interactions in the central Balkans and to contribute to evidence-based public-health measures for tick-bite prevention and surveillance improvement.
2. Materials and Methods
2.1. Ethical Considerations
Ethical approval was obtained from the Ethics Committee of the Medical faculty in Skopje, North Macedonia (Approval No. 03–1835/2), and the Ethics Committee of Medical Faculty Novi Sad (Approval No. 01–39/24/1). All procedures were conducted in accordance with the ethical standards of these institutions and in line with the Declaration of Helsinki. The study also complied with the applicable national legislation on patient rights in both North Macedonia and Serbia.
2.2. Study Design and Period
This cross-sectional study analyzed ticks removed from humans at two centers between January 2022 and December 2024. Tick specimens were extracted from patients seeking consultation after a tick-bite at the Clinic for Infectious Diseases in Skopje (CIDS), North Macedonia and the Pasteur institute in Novi Sad (PINS), Serbia.
Tick removal in healthcare facilities was performed using tweezers, although some patients attempted removal by themselves with tweezers or fingers, and in some cases the tick was collected only after spontaneous detachment. Following removal, patients were instructed to place the tick in a plastic container and deliver it to their respective centers. Each specimen was placed in a sterile plastic tube containing 70% ethanol, labelled with a unique identifier, and accompanied by metadata including the date of removal, patient sex and age, anatomical site of attachment, and possible location of infestation. Samples were kept and transported at room temperature to PINS for further analysis.
Tick attachment sites were categorized into five clinically relevant regions: the head and neck, torso, inguinal-genital area, upper and lower extremities.
If multiple ticks were removed from the same individual, each tick was treated as a separate observation. The month of tick removal was used as a proxy for seasonal activity. The total number of ticks collected each month, categorized by species and developmental stage, were expressed as proportions of the total number of ticks collected annually for each country.
2.3. Morphological Identification
The collected tick specimens were identified taxonomically based on morphological characteristics using standard keys described by Estrada-Peña et al. [16], considering both life stage and sex.
2.4. Statistical Analysis
To address whether nymphs infested the same body areas as adults, attachment-site distributions were compared between nymphs and adults separately for each species within each country using contingency-table methods (chi-squared test when expected counts permitted, otherwise Fisher’s exact test with Monte Carlo simulation). Species–country strata were considered not testable when one developmental stage was absent or when, after excluding empty categories, all observations fell within a single attachment-site category. In these instances, comparisons were restricted to descriptive summaries.
All outputs were generated in R (v4.3.1) using readxl [17], dplyr [18], tidyr [19], stringr [20], purrr [21], ggplot2 [22], ggalluvial [23], and patchwork [24].
Remaining datasets were compiled in Microsoft Excel 2021 and analyzed using GraphPad Prism 9.0.2. Descriptive statistics were expressed as counts and percentages. Associations between categorical variables (country, tick species, developmental stage, attachment site, seasonality and year) were evaluated using the chi-squared (χ2) test or Fisher’s exact test. Statistical significance was set at p < 0.05.
Although patient age was collected as part of the metadata, age data were not uniformly available for all enrolled participants across the study cohort. To avoid potential bias arising from incomplete data coverage, age was not included in the comparative analyses.
3. Results
A total of 1641 ticks were collected during the study period, comprising 157 (157/1641; 9.57%) specimens from North Macedonia and 1484 from Serbia (1484/1641; 90.43%) in the period from 2022 to 2024. All ticks were removed from patients who came for medical evaluation following a tick bite.
3.1. Geographical Distribution of Tick-Bites
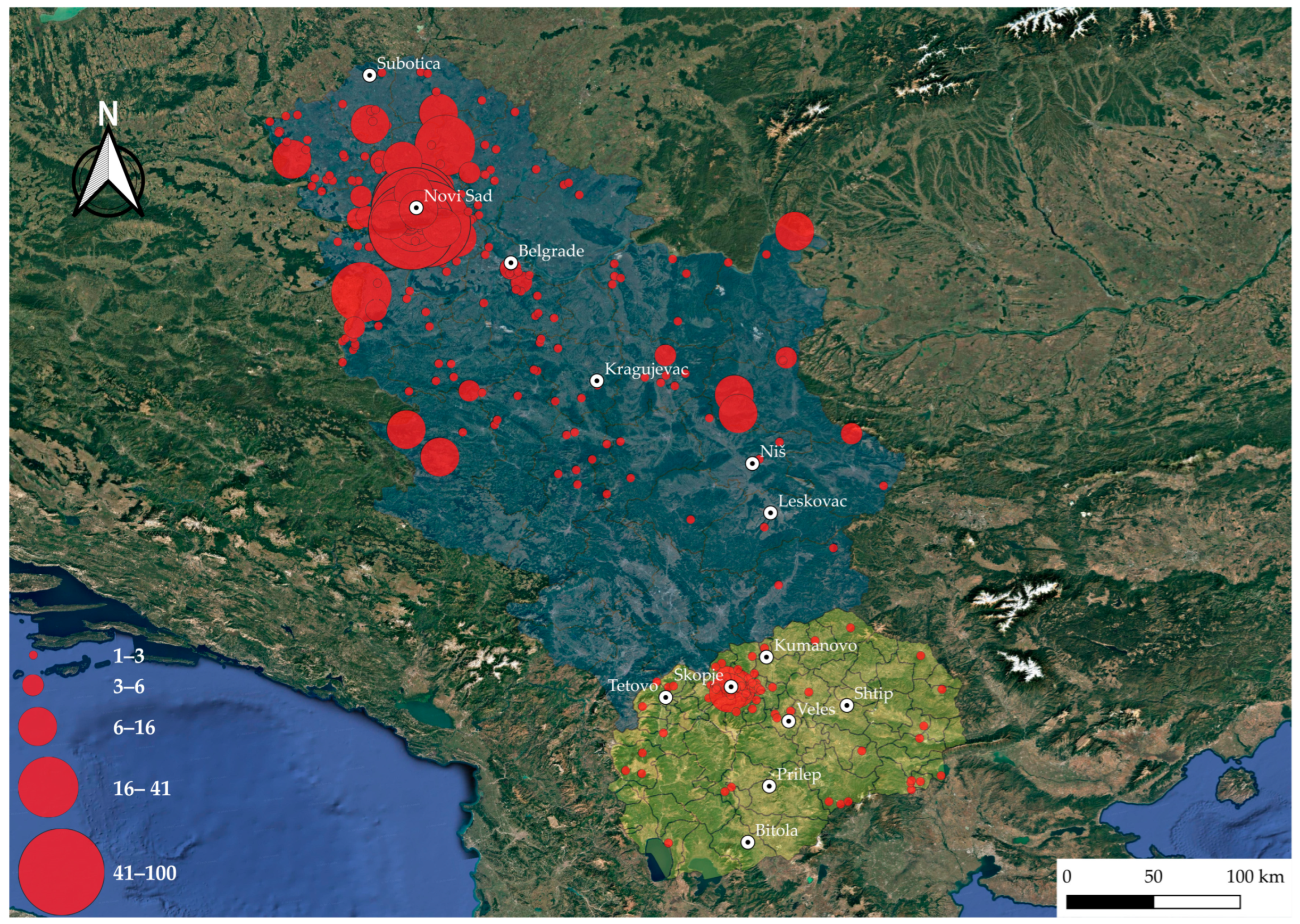
Probable tick-infestation sites reported by patients across North Macedonia are shown in Figure 1. In North Macedonia, the majority of ticks collected came from the northwestern part, particularly the capital, Skopje and nearby municipalities, while only sporadic cases were recorded in the peripheral regions. In contrast, Serbia exhibited a much wider geographic spread of reported tick bites (Figure 1). The highest density was again centered around a major urban area, Novi Sad and its surrounding municipalities. Beyond this hotspot, tick bites were recorded throughout the entire country, including both rural and peri-urban regions. Compared to North Macedonia, Serbia displayed far greater spatial heterogeneity, with tick bites detected across nearly the entire country, reflecting both higher case numbers and a more extensive surveillance coverage.
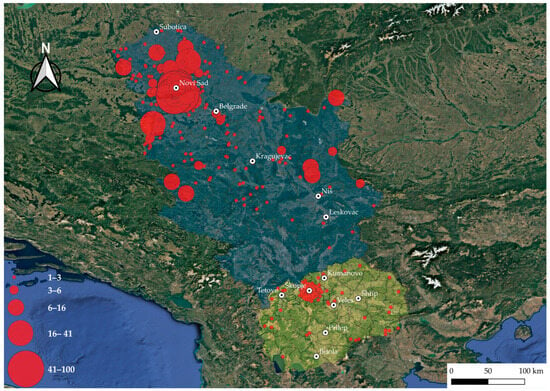
Figure 1.
Geographic distribution of locations where tick infestation occurred in patients from North Macedonia and Serbia (2022–2024). Each point represents a probable tick infestation site reported by patients. Red bubble size indicates the number of ticks collected per site, and color intensity reflects relative abundance. The shape file for mapping at district and municipality levels is available at the GADM database of Global Administrative Areas (v4.1, https://gadm.org/, accessed on 22 July 2022). The map was generated using QGIS v3.12 [25].
3.2. Tick Prevalence
During the study period 2022–2024, 6 tick species were identified from humans in North Macedonia (Table 1). I. ricinus was the dominant species each year, accounting for 68.80% of all ticks collected (108/157), followed by Hyalomma marginatum and Rhipicephalus sanguineus (Table 1). In Serbia, 8 tick species were identified from humans during 2022–2024, with I. ricinus predominating each year and accounting for 92.69% of all ticks collected (1369/1477). Other species were recorded sporadically, including R. sanguineus, Dermacentor reticulatus, Haemaphysalis punctata, Haemaphysalis spp., Dermacentor marginatus, Dermacentor spp. and Ixodes hexagonus (Table 1).

Table 1.
Annual distribution of tick species removed from humans in North Macedonia and Serbia (2022–2024).
3.3. Development Stage
In North Macedonia, most ticks were adults (119/157, 75.80%), followed by nymphs (35/157, 22.29%) and larvae (3/157, 1.91%). I. ricinus was predominantly represented by adult females in Macedonian patients, across all three study years, 83.33% (10 adults of 12 total), 75.41% (46 adults of 61 total), and 91.43% (32 adults of 35 total) in 2022, 2023 and 2024, respectively. In addition, I. ricinus and was significantly more likely to be collected from a Macedonian patient as an adult female in comparison to all other tick species combined (Fisher’s exact test, p = 0.0166). Adult females also comprised 42.9% (13/30) of all H. marginatum and 93.3% (13/14) of R. sanguineus ticks, whereas nymphs accounted for 56.67% and 7.14%, respectively. Other tick species were represented by only a few specimens, and due to their limited number, statistical analysis of developmental stages was not feasible.
In Serbia, nymphs were the most frequently detected developmental stage (821/1477, 55.59%), followed by adults (571/1477, 38.66%) and larvae (85/1477, 5.75%). The distribution of developmental stages differed significantly between years (χ2 = 26.85, df = 4, p < 0.0001). I. ricinus was represented by all developmental stages, with nymphs predominating (Fisher’s exact test, p < 0.0001). Adult females accounted for 44.33% (168/379), 41.39% (195/447), and 61.90% (218/651) in 2022, 2023 and 2024 respectively. On the other hand, nymphs represented 52.96%, 52.66%, and 64.67% respectively.
R. sanguineus showed a significantly different distribution of developmental stages compared with all other tick species (Fisher’s exact test, p < 0.0001), with adults predominating (42/66, 63.64%) over nymphs (24/66, 36.36%) and larvae (0/66; 0%). Other tick species were recorded in very low numbers and were represented almost exclusively by adult female stages, also making statistical comparison of developmental stage distribution across years not feasible.
The distribution of developmental stages differed significantly between the two centers (χ2 = 113.4, df = 2, p < 0.0001), with nymphs significantly more prevalent among ticks from Serbia (p < 0.0001), while adult females significantly predominated in North Macedonia (Fisher’s exact test, p < 0.0001). Adult males of any tick species were not detected during the study period. Frequency of larval ticks did not differ significantly between the two countries (p = 0.0786). I. ricinus showed a significantly different stage distribution between centers (χ2 = 87.61, df = 2, p < 0.0001), with nymphs predominating in Serbia and adults in North Macedonia. In contrast, R. sanguineus showed no significant difference in developmental-stage distribution between the two centers (p = 0.0533).
3.4. Seasonal Dynamics
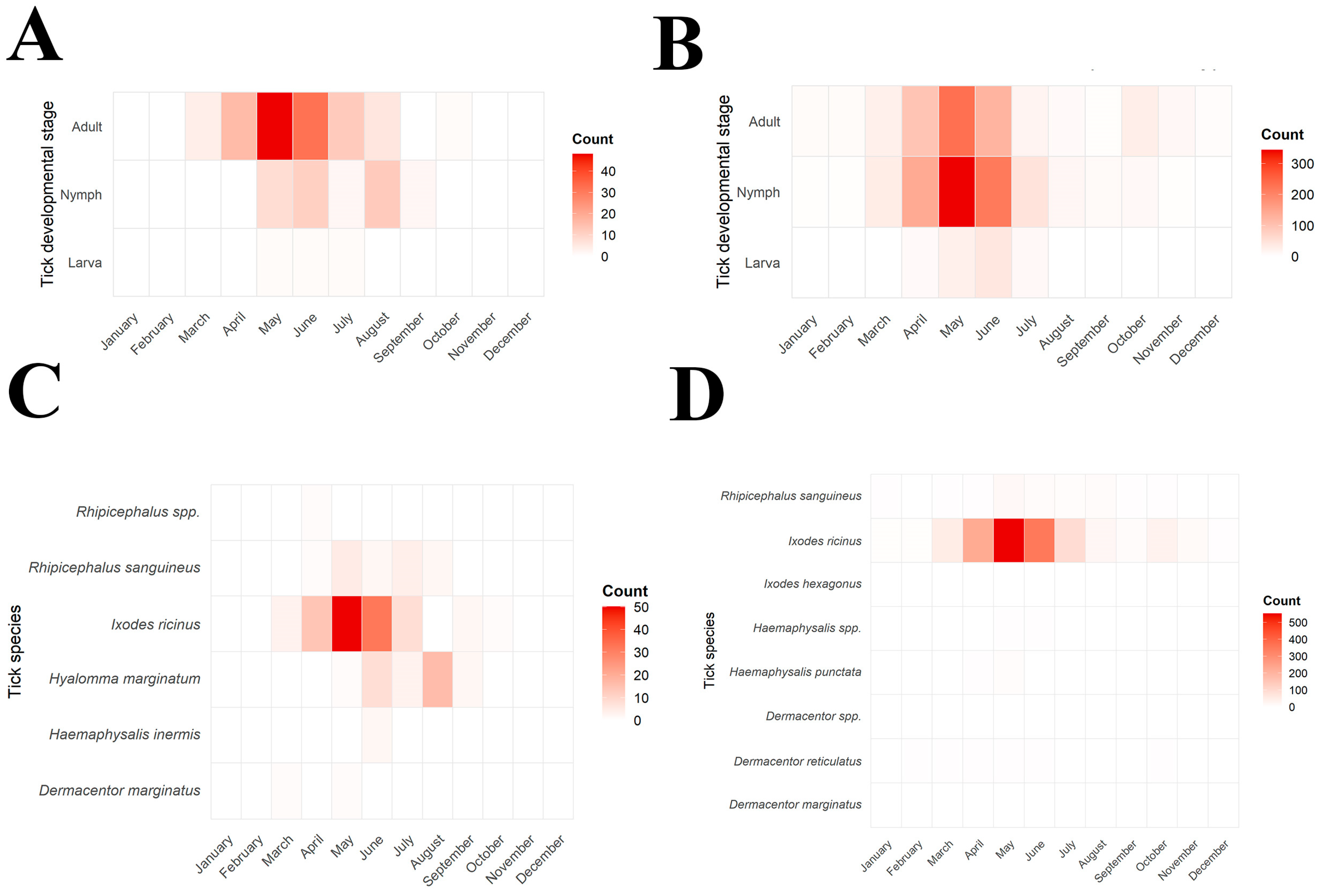
Tick activity exhibited a clear seasonal pattern in both centers during 2022–2024, with consistent trends across years (Figure 2 and Figure 3).
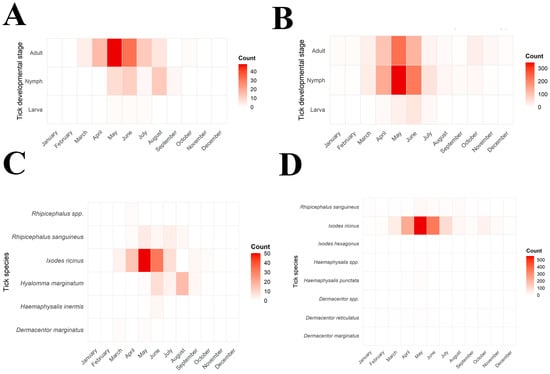
Figure 2.
Monthly distribution of species and development stage of ticks removed from humans in North Macedonia and Serbia (2022–2024). (A): illustration of monthly distribution of human-infesting tick species in North Macedonia; (B): corresponding illustration for Serbia; (C): developmental stage distribution of tick removed from humans in North Macedonia; (D): corresponding illustration for Serbia. Intensive red shades indicate higher numbers of collected specimens.
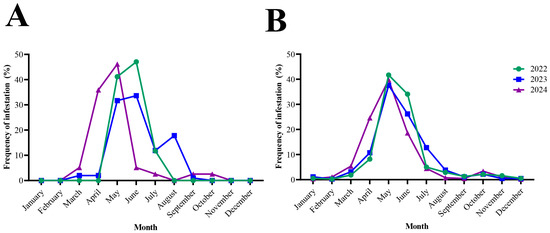
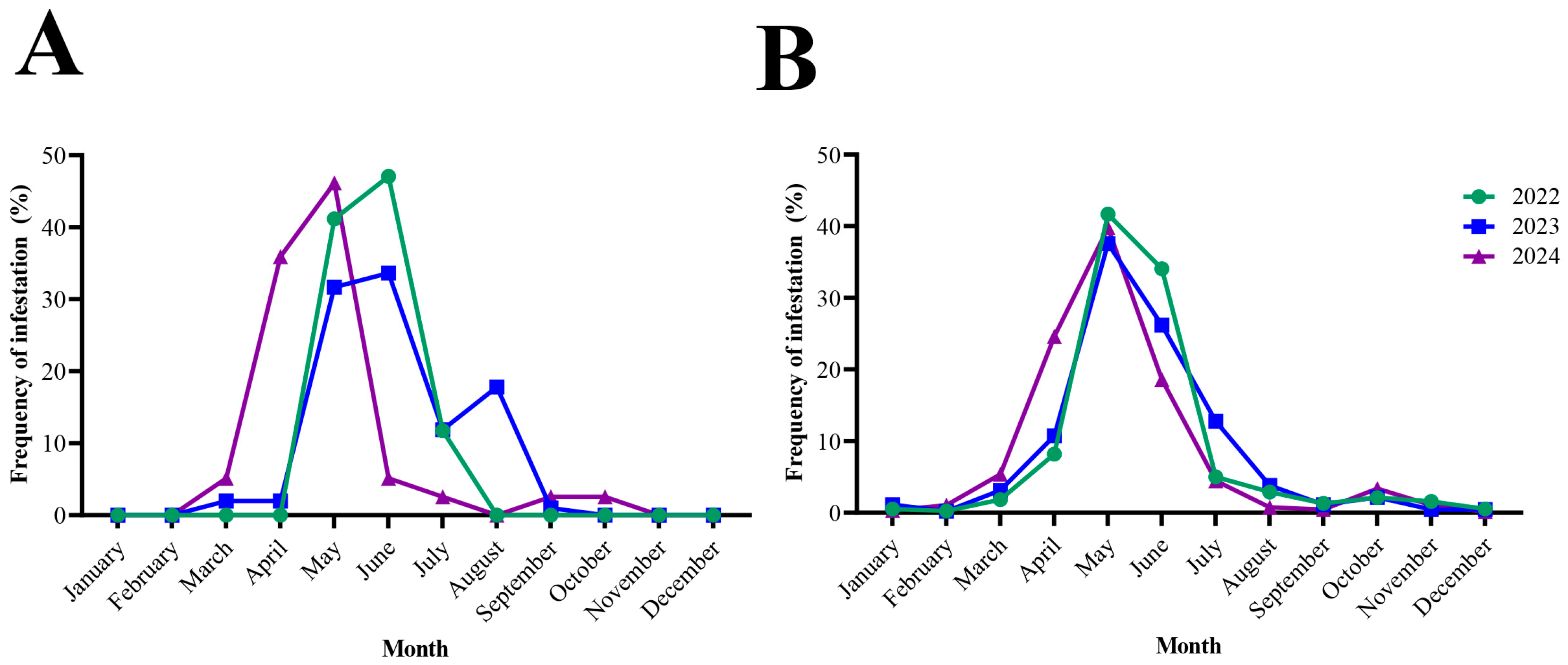
Figure 3.
Monthly distribution of tick infestations in patients from North Macedonia and Serbia (2022–2024). Dataset from every examined year is presented in different color. (A): monthly prevalence of tick-infestation in humans from North Macedonia; (B): corresponding illustration for Serbia.
In North Macedonia, first records of tick activity began in March, increased sharply in April–May, and peaked during May–June (63.5% of all ticks). The monthly distribution of tick species in North Macedonia is presented in Figure 2A,C and Figure 3A. Activity then declined gradually through July and August, with only sporadic findings in September–October. The monthly distribution of I. ricinus differed significantly from that of other tick species (χ2 = 55.34, df = 7, p < 0.0001), peaking in May–June, whereas H. marginatum exhibited a distinct seasonal pattern with predominance later in the season in August (χ2 = 76.00, df = 7, p < 0.0001). The activity of R. sanguineus showed no significant difference from other ticks (χ2 = 8.072, df = 7, p = 0.3263), with activity peaks recorded during the summer months (June–August). Adult ticks were most frequently collected during May and June, while nymphs were mainly active between May and August. Larvae were rarely detected and appeared only sporadically in late spring and early summer. The seasonal distribution of developmental stages in North Macedonia is presented in Figure 2A.
Tick activity in Serbia was recorded throughout the entire year, beginning at low levels in January and February. The monthly distribution of tick species in Serbia is presented in Figure 2B,D. Activity increased sharply in March–April and reached its peak during May–June, when 64.45% of all ticks were collected (Figure 3B). A gradual decline followed through July and August, but tick presence persisted into early autumn, with occasional detections in September–October and isolated specimens still found in November and December. Adults were the predominant developmental stage, peaking in May–June and accounting for most tick activity. Their distribution differed significantly from that of immature stages (χ2 = 67.88, df = 11, p < 0.0001). Nymphs showed a distinct pattern, remaining active later into summer (χ2 = 36.57, df = 11, p < 0.0001), whereas larvae appeared only sporadically during June–August (χ2 = 42.26, df = 11, p = 0.0001). Figure 2B shows the monthly distribution of developmental stages in Serbia.
Seasonal activity differed significantly between North Macedonia and Serbia (χ2 = 153.2, df = 11, p < 0.0001) (Figure 2). When comparing the monthly distribution of all human-biting ticks collected throughout the year (January–December), I. ricinus was the predominant species in both regions, representing 91.2% of all ticks collected in Serbia and 68.8% in North Macedonia. The proportion of I. ricinus among all collected ticks differed significantly between Serbia and North Macedonia (Fisher’s exact test, p < 0.0001). All other tick species showed no statistically significant variation in distribution between Serbia and North Macedonia (p > 0.05).
3.5. Anatomical Distribution of Tick Infestations
In North Macedonia, the lower extremities (53/157; 33.76%) and torso (45/157; 28.66%) predominated, with fewer bites observed on the head and neck (21/157; 13.38%), upper extremities (20/157; 12.74%), and inguinal region (18/157; 11.46%) (Figure 4A). In comparison, in Serbia, tick attachments were most frequent on the torso (468/1477; 31.69%) and lower extremities (413/1477; 27.96%), followed by the head and neck (302/1477; 20.45%), upper extremities (213/1477; 14.42%), and inguinal region (81/1477; 5.48%) (Figure 4B). There is a statistically significant difference in the distribution of tick bites by body region between Serbia and North Macedonia, with p = 0.0063.
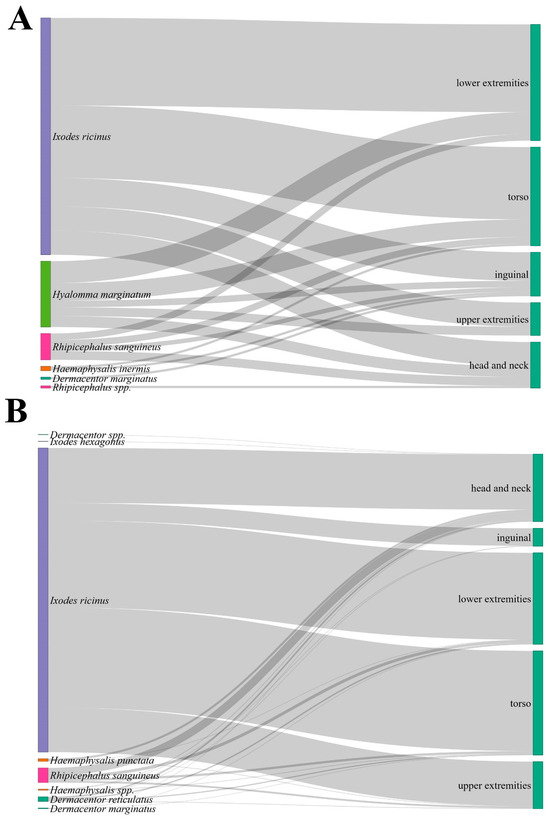
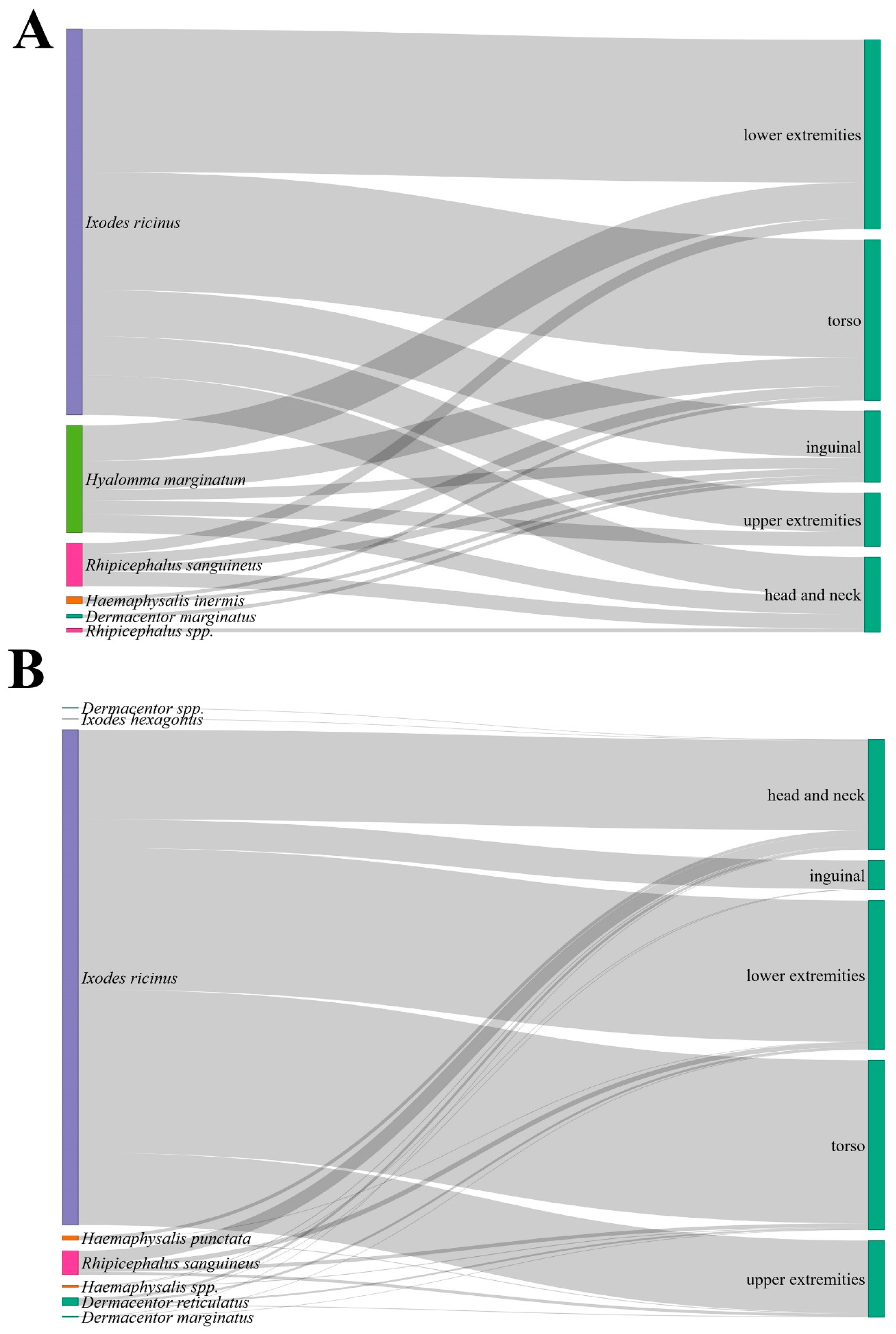
Figure 4.
Sankey diagrams illustrating the distribution of tick species by body region in North Macedonia and Serbia. (A): relative frequency of human tick attachments across body regions for each tick species removed from humans in North Macedonia; (B): corresponding distribution for Serbia. Flow widths represent the proportional contribution of each species to specific body regions.
Statistical comparison of body-site distribution among species in North Macedonia showed no significant association between tick species and attachment site (p > 0.05) (Figure 4). However, there is a statistically significant association between tick species and attachment site in Serbia (χ2 = 93.74, df = 28, p < 0.0001). I. ricinus exhibited a clear preference for the torso (32.94%) and lower extremities (28.56%), whereas other tick species were relatively more often found on the head and neck region (Figure 4). The difference in body-site distribution between I. ricinus and all other species was statistically significant (χ2 = 60.70, df = 4, p < 0.00001). The distribution of I. ricinus attachment sites differed significantly between Serbia and North Macedonia (χ2 = 13.91, df = 4, p = 0.0076), indicating variation in exposure patterns or tick-host interactions between the two settings.
The anatomical distribution of tick attachment sites did not differ significantly between developmental stages in North Macedonia, whereas in Serbia a significant association was observed (p < 0.001). In Serbia, nymphs were predominantly attached to the torso and lower extremities, while adults showed a more even distribution across all body regions. Furthermore, the Macedonian data indicated a similar distribution pattern across stages without clear site preference.
Stage-specific attachment-site frequencies and within-stage percentages in Serbia and North Macedonia are summarized in Supplementary Table S1.
After adjustment for multiple comparisons within each country (Benjamini–Hochberg false discovery rate), a significant difference in attachment-site distribution between nymphs and adults was observed only for I. ricinus in Serbia, indicating stage-dependent attachment-site preferences in this high-frequency species (Supplementary Table S2). In contrast, no species in North Macedonia remained significant after correction, consistent with reduced statistical power and sparse counts across several species-stage combinations (Supplementary Table S2).
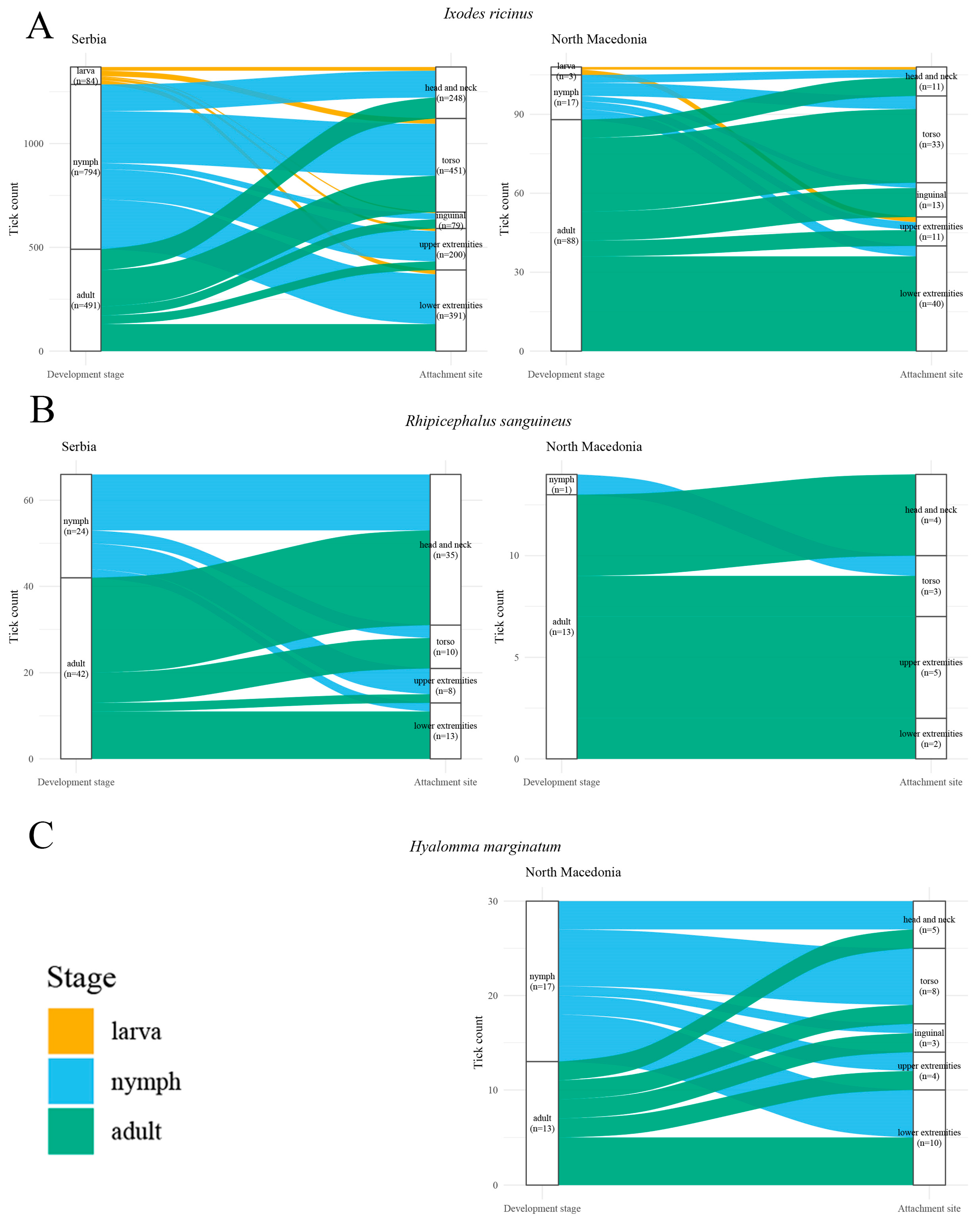
Overall, the combined descriptive and inferential findings suggest that nymph–adult attachment-site distributions are broadly similar for several species-country strata, but that stage-associated differences may occur for specific species in Serbia, most clearly for I. ricinus (Figure 5).
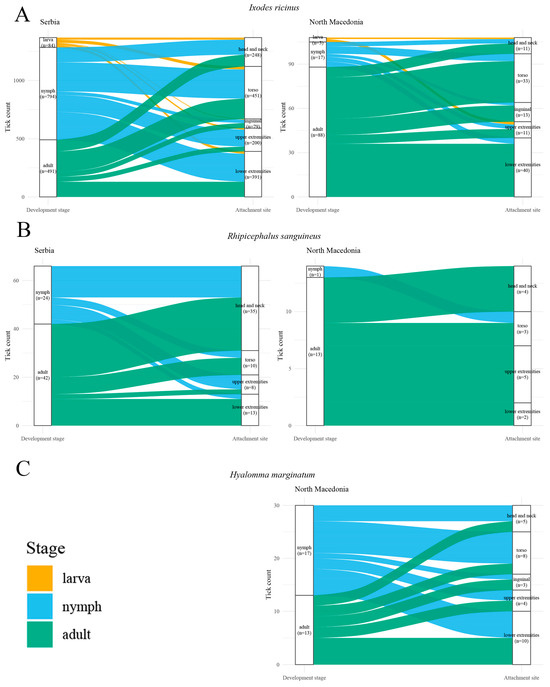
Figure 5.
Sankey diagrams illustrating the distribution of development stages of most common tick species stratified by body region in North Macedonia (panels (A–C)) and Serbia (panels (A,B)). (A): Ixodes ricinus; (B): Rhipicephalus sanguineus; (C): Hyalomma marginatum. Flow widths represent the proportional contribution of each species to specific body regions.
4. Discussion
This study provides one of the first comparative assessments of human tick bites in North Macedonia and Serbia, revealing both shared and divergent patterns in tick species composition. In both countries, I. ricinus was the dominant human-biting species, but its predominance was far greater in Serbia (over 90% of ticks) than in North Macedonia (around 68%). This stable I. ricinus dominance in the northern Serbia site likely reflects the temperate, wooded habitats of the region, which are highly favorable for this species [26]. Notably, H. marginatum, principal vector for CCHFV in the region, was consistently present in North Macedonia (19.1% of ticks overall) but absent in the Serbian sample, although it was previously documented in the Serbian tick fauna [8]. The mid-summer peak of H. marginatum in North Macedonia and the sustained dominance of I. ricinus in Serbia are compatible with climatic and habitat differences between the sampled regions [27,28]. Similar patterns have been observed in temperate regions such as Poland, where I. ricinus can comprise approximately 97% of ticks infesting humans [29]. In contrast, in hotter Mediterranean climates Hyalomma ticks become more prominent. In southwestern Turkey, Hyalomma species accounted for approximately 79% of ticks infesting humans, while Ixodes species represented less than 1% [30]. These findings highlight how climatic and ecological gradients shape tick exposure in the Balkans. Species adapted to more temperate or humid environments (such as I. ricinus) remain abundant in cooler, forested areas year-round, while more thermophilic species like H. marginatum establish in warmer zones [31].
The presence of Hyalomma spp. in North Macedonia is of particularly public health relevance, as H. marginatum is the primary vector of CCHFV in Europe, a pathogen that causes a severe hemorrhagic fever with high fatality rates [32]. While CCHFV historically had only sporadic or absent activity in this region, a fatal locally acquired case occurred in eastern North Macedonia in 2023 after a Hyalomma bite, its first confirmed local transmission in over 50 years, followed by two additional cases in 2024 [33,34]. Given the documented circulation of CCHFV in neighboring Bulgaria and Albania [35,36,37,38] public health authorities in Serbia should remain vigilant and strengthen surveillance in areas where Hyalomma ticks are expected to be present (Serbian territory bordering with Bulgaria, North Macedonia and Albania).
In terms of anatomical attachment sites, the pattern observed in North Macedonia and Serbia aligned with tick exposures reported in Spain, United Kingdom, Northern Europe and Northern Serbia, as ticks tend to attach to lower extremities of the human host [12,15,39,40]. Given that the difference observed in the distribution of bite sites in North Macedonia was not species-related, the lower frequency of ticks on the head and neck may be related to the predominance of adult patients compared to children at this reference center.
In Serbia, I. ricinus showed a tendency to attach to the torso and lower extremities more than other regions, whereas R. sanguineus was relatively more often found on the head/neck, which is consistent with reports describing R. sanguineus frequently attaching to the head region in humans [39]. One-third of the D. reticulatus ticks were found attached to the head and neck, consistent with reports that Dermacentor spp. often bite the scalp or head, frequently producing a necrotic eschar and regional lymphadenopathy [41,42]. Our findings of year-round tick activity, with a pronounced peak in May–June driven by I. ricinus are consistent with studies in Central Europe showing I. ricinus and D. reticulatus activity even during the winter [43].
This study provides a comparative assessment of tick species, developmental stages, seasonal dynamics, and anatomical attachment sites among ticks removed from humans in Serbia and North Macedonia between 2022 and 2024. While I. ricinus predominated in both countries, its relative dominance was substantially higher in Serbia, whereas H. marginatum represented a notable proportion of human-infesting ticks in North Macedonia. Seasonal activity in both regions peaked in late spring and early summer, and species- and stage-specific variation in body-site attachment was observed.
It should be highlighted that our study suffers from several limitations, as tick sampling was clinic-based and restricted to two referral centers, which likely introduced selection and catchment-area biases and limits national representativeness, especially given the marked imbalance in specimen numbers between Serbia and North Macedonia. In particular, the North Macedonia series reflects submissions to a single sentinel center and should not be interpreted as nationally representative; therefore, between-country comparisons of developmental stage should be interpreted cautiously and considered exploratory. The marked imbalance in submitted ticks (North Macedonia: n = 157; Serbia: n = 1484), including I. ricinus (n = 108 and n = 1369, respectively), increases susceptibility to catchment and referral effects, and observed differences in stage composition may reflect differences in healthcare-seeking pathways and tick submission practices between centers.
Additionally, smaller life stages may be differentially detected and recovered (e.g., during self-removal, delayed presentation, or spontaneous detachment), which could bias the apparent proportion of nymphs and larvae in clinic-based collections. Probable infestation sites and some tick removals were self-reported or patient-performed, which may contribute to exposure misclassification and differential recovery of life stages that are smaller in size (i.e., larvae and nymphs).
A key strength of this study is its comparative design, using consistently collected metadata (date of removal, patient age/sex, attachment site, and probable exposure location) and standardized morphological identification to describe species composition, developmental stages, seasonality, and anatomical attachment patterns; the relatively large overall sample (n = 1641) enables robust inference for common taxa, particularly I. ricinus in Serbia, and the visual mapping of categorical relationships (e.g., species-to-body-site distributions) aids interpretation and translation to prevention messaging.
Future research should expand human–tick surveillance across additional clinical and community settings to improve geographic representativeness and reduce catchment-area bias. Molecular confirmation of tick species and comprehensive pathogen screening, including Borrelia, Anaplasma, SFGR, and CCHFV should be incorporated to directly link species and stage-specific patterns with pathogen carriage and human infection risk. Prospective, longitudinal cohort designs with standardized follow-up applied in smaller scale within previous studies [7,10] would strengthen understanding of seasonal bite dynamics, attachment duration, and clinical outcomes, while multivariable or mixed-effects modelling approaches are needed to account for confounding factors such as age, outdoor activity, protection via specialized clothing, and tick exposure via pets/companion animals/farm animals. Integration of ecological and environmental data, such as land use, vegetation indices, wildlife host distributions, and meteorological variables could further elucidate spatial drivers of tick–human contact [44]. Finally, harmonized Pan-Balkan surveillance protocols and public awareness assessments would support regional early-warning systems [45] and more targeted prevention strategies.
Our findings reflect underlying ecological differences across the central Balkans and highlight the need for strengthened, geographically comprehensive surveillance systems. Improved integration of molecular diagnostics, clinical follow-up, and ecological data will be critical for refining public-health interventions and reducing the burden of tick-borne diseases in the region.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/parasitologia6010006/s1. Table S1—Stage-specific attachment-site frequencies and within-stage percentages of ticks removed from humans in Serbia and North Macedonia; Table S2—Attachment-site distribution between larvae, nymphs and adults of ticks removed from humans in Serbia and North Macedonia.
Author Contributions
Conceptualization, D.J. and P.B.; methodology, D.J., A.M.R. and P.B.; resources, S.M., M.N., A.M.R., V.S., I.B., D.M. and S.S.; data curation: D.J., I.B., V.S., A.M.R., S.S., A.S., M.N. and P.B.; writing—original draft preparation: A.M.R., D.J. and P.B.; writing—review and editing, P.B., D.J., V.S., I.B., D.M. and S.S.; supervision, P.B. and D.J.; All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
This study was reviewed and approved by the Ethics Committee of the Faculty of Medicine, Ss. Cyril and Methodius University in Skopje (Approval No. 03–1835/2 from 24 April 2023) and the Ethics Committee of Medical Faculty Novi Sad (Approval No. 01–39/24/1 from 30 June 2021). All procedures were conducted in accordance with the ethical standards of these institutions and in line with the Declaration of Helsinki. The study also complied with the applicable national legislation on patient rights in both North Macedonia and Serbia.
Informed Consent Statement
Patient consent was waived due to the retrospective nature of the study.
Data Availability Statement
All data generated in this manuscript are available in the main text.
Acknowledgments
This research was supported by Pasteur Institute Novi Sad and the COST (European Cooperation in Science and Technology Short Term Scientific Mission [COST Action project nr. CA21170, Prevention, anticipation, and mitigation of tick-borne disease risk applying the DAMA protocol (PRAGMATICK)] and Balkan Association for Vector-Borne Diseases (www.bavbd.org, accessed on 4 December 2025).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- de la Fuente, J.; Estrada-Peña, A.; Rafael, M.; Almazán, C.; Bermúdez, S.; Abdelbaset, A.E.; Kasaija, P.D.; Kabi, F.; Akande, F.A.; Ajagbe, D.O.; et al. Perception of Ticks and Tick-Borne Diseases Worldwide. Pathogens 2023, 12, 1258. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.; Kahl, O.; Zintl, A. Pathogens Transmitted by Ixodes ricinus. Ticks Tick-Borne Dis. 2024, 15, 102402. [Google Scholar] [CrossRef] [PubMed]
- Morea, A.; Schino, V.; Bartolomeo, N.; Ravallese, R.; Sacino, G.; Ravallese, R.; Iatta, R.; Otranto, D.; Lovreglio, P.; Stufano, A. Seroprevalence of Tick-Borne Diseases in Europe in Occupational Settings: A Systematic Review and Metanalysis. Public Health 2025, 248, 105923. [Google Scholar] [CrossRef]
- Dantas-Torres, F. Climate Change, Biodiversity, Ticks and Tick-Borne Diseases: The Butterfly Effect. Int. J. Parasitol. Parasites Wildl. 2015, 4, 452–461. [Google Scholar] [CrossRef]
- Stark, J.H.; Pilz, A.; Jodar, L.; Moïsi, J.C. The Epidemiology of Lyme Borreliosis in Europe: An Updated Review on a Growing Public Health Issue. Vector-Borne Zoonotic Dis. 2023, 23, 139–141. [Google Scholar] [CrossRef]
- Kunze, M.; Banović, P.; Bogovič, P.; Briciu, V.; Čivljak, R.; Dobler, G.; Hristea, A.; Kerlik, J.; Kuivanen, S.; Kynčl, J.; et al. Recommendations to Improve Tick-Borne Encephalitis Surveillance and Vaccine Uptake in Europe. Microorganisms 2022, 10, 1283. [Google Scholar] [CrossRef]
- Banović, P.; Jakimovski, D.; Bogdan, I.; Simin, V.; Mijatović, D.; Bosilkovski, M.; Mateska, S.; Díaz-Sánchez, A.A.; Foucault-Simonin, A.; Zając, Z.; et al. Tick-Borne Diseases at the Crossroads of the Middle East and Central Europe. Infect. Dis. Now 2024, 54, 104959. [Google Scholar] [CrossRef]
- Kapo, N.; Zuber Bogdanović, I.; Gagović, E.; Žekić, M.; Veinović, G.; Sukara, R.; Mihaljica, D.; Adžić, B.; Kadriaj, P.; Cvetkovikj, A.; et al. Ixodid Ticks and Zoonotic Tick-Borne Pathogens of the Western Balkans. Parasit. Vectors 2024, 17, 45. [Google Scholar] [CrossRef]
- Jakimovski, D.; Mateska, S.; Najdovska, M.; Stamenkovska, A.; Pavleva, V.; Bosilkovski, M.; Mijatović, D.; Simin, V.; Bogdan, I.; Grujić, J.; et al. Differential Exposure to Borrelia spp. and Spotted Fever Group Rickettsia in Serbia and North Macedonia: A Comparative Study. Pathogens 2025, 14, 814. [Google Scholar] [CrossRef]
- Banović, P.; Díaz-Sánchez, A.A.; Simin, V.; Foucault-Simonin, A.; Galon, C.; Wu-Chuang, A.; Mijatović, D.; Obregón, D.; Moutailler, S.; Cabezas-Cruz, A. Clinical Aspects and Detection of Emerging Rickettsial Pathogens: A “One Health” Approach Study in Serbia, 2020. Front. Microbiol. 2022, 12, 797399. [Google Scholar] [CrossRef] [PubMed]
- Jakimovski, D.; Mateska, S.; Dimitrova, E.; Bosilkovski, M.; Mijatović, D.; Simin, V.; Bogdan, I.; Grujić, J.; Budakov-Obradović, Z.; Meletis, E.; et al. Tick-Borne Encephalitis Virus and Borrelia burgdorferi Seroprevalence in Balkan Tick-Infested Individuals: A Two-Centre Study. Pathogens 2023, 12, 922. [Google Scholar] [CrossRef]
- Stevanoska, S.; Simin, V.; Bogdan, I.; Gazibara, D.; Mijatović, D.; Stojšić, A.; Banović, P. Prevalencija tvrdih krpelja koji infestiraju ljude u Južnobačkom okrugu: Povezanost starosne dobi i lokalizacije infestacije sa razvojnim stadijumima krpelja. PONS—Med. Časopis 2025, 22, 3–8. [Google Scholar] [CrossRef]
- Banović, P. Faktori Rizika za Infestiranje Krpeljima i Obolevanje od Lajmske Borelioze u Južnobačkom Okrugu i Dijagnostika u Ranoj Fazi Bolesti. Ph.D. Thesis, University in Novi Sad, Novi Sad, Serbia, 2022. [Google Scholar]
- Mladenović, J.L. Epidemiološki Aspekti Lajmske Bolesti na Teritorije Građa Beograda. Ph.D. Thesis, University in Niš, Niš, Serbia, 2014. [Google Scholar]
- Cull, B.; Pietzsch, M.E.; Gillingham, E.L.; McGinley, L.; Medlock, J.M.; Hansford, K.M. Seasonality and Anatomical Location of Human Tick Bites in the United Kingdom. Zoonoses Public Health 2020, 67, 112–121. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Mihalca, A.D.; Petney, T.N. Ticks of Europe and North Africa: A Guide to Species Identification; Illustrations; Springer: Cham, Switzerland, 2017; p. xxi, 404p. [Google Scholar]
- Read Excel Files. Available online: https://readxl.tidyverse.org/ (accessed on 5 January 2026).
- A Grammar of Data Manipulation. Available online: https://dplyr.tidyverse.org/ (accessed on 5 January 2026).
- Tidy Messy Data. Available online: https://tidyr.tidyverse.org/ (accessed on 5 January 2026).
- Simple, Consistent Wrappers for Common String Operations. Available online: https://stringr.tidyverse.org/ (accessed on 5 January 2026).
- Functional Programming Tools. Available online: https://purrr.tidyverse.org/ (accessed on 5 January 2026).
- Create Elegant Data Visualisations Using the Grammar of Graphics. Available online: https://ggplot2.tidyverse.org/ (accessed on 5 January 2026).
- Alluvial Plots in ggplot2. Available online: https://corybrunson.github.io/ggalluvial/ (accessed on 5 January 2026).
- GADM. Database of Global Administrative Areas, version 4.1. 2022. Available online: https://gadm.org (accessed on 5 January 2026).
- The Composer of Plots. Available online: https://patchwork.data-imaginist.com/ (accessed on 5 January 2026).
- Zając, Z.; Kulisz, J.; Bartosik, K.; Woźniak, A.; Dzierżak, M.; Khan, A. Environmental Determinants of the Occurrence and Activity of Ixodes ricinus Ticks and the Prevalence of Tick-Borne Diseases in Eastern Poland. Sci. Rep. 2021, 11, 15472. [Google Scholar] [CrossRef]
- Smailagic, J.; Savovic, A.; Markovic, D.; Nesic, D.; Drakula, B.; Milenkovic, M.; Zdravkovic, S. Climate Characteristics of Serbia; Republic Hydrometeorological Service of Serbia: Belgrade, Serbia, 2013; pp. 1–26. [Google Scholar]
- Petrušev, E.; Stolić, N.; Šajn, R.; Stafilov, T. Geological Characteristics of the Republic of North Macedonia. Geol. Maced. 2021, 35, 49–58. [Google Scholar] [CrossRef]
- Pawełczyk, A.; Bednarska, M.; Hamera, A.; Religa, E.; Poryszewska, M.; Mierzejewska, E.J.; Welc-Falęciak, R. Long-Term Study of Borrelia and Babesia Prevalence and Co-Infection in Ixodes ricinus and Dermacentor recticulatus Ticks Removed from Humans in Poland, 2016–2019. Parasites Vectors 2021, 14, 348. [Google Scholar] [CrossRef]
- Bakırcı, S.; Aysul, N.; Bilgiç, H.B.; Hacılarlıoğlu, S.; Eren, H.; Karagenç, T. Tick Bites on Humans in Southwestern Region of Turkey: Species Diversity. Turk. J. Parasitol. 2019, 43, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Celina, S.S.; Černý, J. Hyalomma marginatum in Europe: The Past, Current Status, and Future Challenges—A Systematic Review. Transbound. Emerg. Dis. 2025, 2025, 7771431. [Google Scholar] [CrossRef]
- Ergönül, Ö. Crimean–Congo haemorrhagic fever. Lancet Infect. Dis. 2006, 6, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Jakimovski, D.; Banović, P.; Spasovska, K.; Rangelov, G.; Cvetanovska, M.; Cana, F.; Simin, V.; Bogdan, I.; Mijatović, D.; Cvetkovikj, A.; et al. One health investigation following a cluster of Crimean-Congo haemorrhagic fever, North Macedonia, July to November 2023. Eurosurveillance 2025, 30, 2400286. [Google Scholar] [CrossRef]
- Jakimovski, D.; Poposki, K.; Dimzova, M.; Cvetanovska, M.; Cana, F.; Bogdan, I.; Cabezas-Cruz, A.; Zana, B.; Lanszki, Z.; Tauber, Z.; et al. Two human infections with diverse Europe-1 Crimean-Congo hemorrhagic fever virus strains, North Macedonia, 2024. Emerg. Infect. Dis. 2025, 31, 313–317. [Google Scholar] [CrossRef]
- Nasirian, H. Ticks infected with Crimean-Congo hemorrhagic fever virus (CCHFV): A decision approach systematic review and meta-analysis regarding their role as vectors. Travel Med. Infect. Dis. 2022, 47, 102309. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Carnell, O.; Wijburg, S.R.; Holding, M.; Sprong, H. Crimean–Congo Haemorrhagic Fever Virus Circulates within Broad Ecological Networks of Ticks and Vertebrates. bioRxiv 2025. [Google Scholar] [CrossRef]
- Sherifi, K.; Rexhepi, A.; Berxholi, K.; Mehmedi, B.; Gecaj, R.M.; Hoxha, Z.; Joachim, A.; Duscher, G.G. Crimean-Congo Hemorrhagic Fever Virus and Borrelia burgdorferi sensu lato in Ticks from Kosovo and Albania. Front. Vet. Sci. 2018, 5, 38. [Google Scholar] [CrossRef] [PubMed]
- Ngoc, K.; Stoikov, I.; Trifonova, I.; Panayotova, E.; Taseva, E.; Trifonova, I.; Christova, I. Molecular and Clinical Characterization of Crimean-Congo Hemorrhagic Fever in Bulgaria, 2015–2024. Pathogens 2025, 14, 785. [Google Scholar] [CrossRef] [PubMed]
- Vieira Lista, M.C.; Belhassen-García, M.; Vicente Santiago, M.B.; Sánchez-Montejo, J.; Pedroza Pérez, C.; Monsalve Arteaga, L.C.; Herrador, Z.; del Álamo-Sanz, R.; Benito, A.; Soto López, J.D.; et al. Identification and Distribution of Human-Biting Ticks in Northwestern Spain. Insects 2022, 13, 469. [Google Scholar] [CrossRef]
- Wilhelmsson, P.; Lindblom, P.; Fryland, L.; Nyman, D.; Jaenson, T.G.; Forsberg, P.; Lindgren, P.-E. Ixodes ricinus Ticks Removed from Humans in Northern Europe: Seasonal Pattern of Infestation, Attachment Sites and Duration of Feeding. Parasites Vectors 2013, 6, 362. [Google Scholar] [CrossRef] [PubMed]
- Parola, P.; Rovery, C.; Rolain, J.M.; Brouqui, P.; Davoust, B.; Raoult, D. Rickettsia slovaca and R. raoultii in Tick-Borne Rickettsioses. Emerg. Infect. Dis. 2009, 15, 1105. [Google Scholar] [CrossRef]
- Oteo, J.A.; Portillo, A. Tick-Borne Rickettsioses in Europe. Ticks Tick-Borne Dis. 2012, 3, 271–278. [Google Scholar] [CrossRef]
- Probst, J.; Springer, A.; Topp, A.-K.; Bröker, M.; Williams, H.; Dautel, H.; Kahl, O.; Strube, C. Winter Activity of Questing Ticks (Ixodes ricinus and Dermacentor reticulatus) in Germany—Evidence from Quasi-Natural Tick Plots, Field Studies and a Tick Submission Study. Ticks Tick-Borne Dis. 2023, 14, 102225. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Cevidanes, A.; Sprong, H.; Millán, J. Pitfalls in Tick and Tick-Borne Pathogens Research, Some Recommendations and a Call for Data Sharing. Pathogens 2021, 10, 712. [Google Scholar] [CrossRef] [PubMed]
- Pateras, K.; Meletis, E.; Denwood, M.; Eusebi, P.; Kostoulas, P. The Convergence Epidemic Volatility Index (cEVI) as an Alternative Early Warning Tool for Identifying Waves in an Epidemic. Infect. Dis. Model. 2023, 8, 484–490. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.