Abstract
Background: Management of penile cancer patients has its grey zones. In particular, no strong evidence or recommendations exist regarding the timing of prophylactic lymphadenectomy. Here, we aim to review the impact that the timing of inguinal and pelvic prophylactic lymph node dissection has on patient outcome. Methods: All relevant databases were searched following the preferred reporting items for systematic reviews and meta-analysis guidelines. A narrative review of indications for lymph node dissection and pathological considerations precede a systematic review of the impact of prophylactic lymph node dissection timing on prognosis. The primary endpoint is disease-free and overall survival in patients undergoing early or late lymph node dissection after penile cancer diagnosis. Results: Four clinical trials, all focusing on the role of inguinal lymph node dissection, are included. Despite the lack of randomized and controlled trials, this review suggests that lymph node dissection should be performed as soon as possible after diagnosis, with 3 months as a realistic cut-off. Conclusions: Survival of penile cancer patients is strictly related to the timing of prophylactic pelvic lymph node dissection. All patients at high risk of nodal metastasis should be offered lymph node dissection within three months of diagnosis, until new predicting tools are validated.
1. Introduction
Penile cancer shows a low incidence in European countries (about 0.5% of all neoplasm in men) with a significant impact on patients’ survival and quality of life [1]. Penile cancer is, usually, more frequent in men aged 50–70 years, even if about 20% of patients are aged 40 years [1,2]. About 95% of all penile neoplasms are squamous cell carcinomas [1,2] and start from the epithelium localized in the inner glans prepuce [1,2]. Penile cancer is frequently associated with human papillomavirus (HPV) infection, even if the association between HPV status and disease-free survival is not totally understood [1,2]. However, several authors highlighted that HPV-positive patients have a better prognosis when compared with HPV-negative ones [3,4]. The association between HPV positivity and lymph node metastasis or survival is still not demonstrated. The natural history of penile cancer is characterized by a starting lesion that commonly originates on the glands and progressively involves the whole glands [3]. If not treated, the lesion could penetrate the Buck’s fascia, the tunica albuginea, and the corpus cavernosum, having the possibility to penetrate the lymphatic vessel [3]. Penile cancer shows an early diffusion through the lymphatic vessels with a high likelihood to penetrate distant organs. On the one hand, the natural history of penile cancer is characterized by aggressive behavior. On the other hand, the most effective treatment is surgical eradication. Chemotherapy and radiation therapy have a limited role in advanced disease [3]. For these reasons, early diagnosis and treatment are crucial for the outcome of penile cancer, in terms of cancer-specific survival and quality of life [1]. The inguinal lymph node status at the moment of penile cancer diagnosis and its early management seem to have a pivotal role in the therapeutic approach to penile cancer patients [2]. Several authors demonstrated that patients’ survival is determined by the inguinal lymph node status at the time of diagnosis [2,3,4]. On the other hand, penile cancer is an oncological disease with a high psychological impact, highly affecting the patients’ general and sexual quality of life. In this sense, it is essential to discover the optimal timing of diagnostic and therapeutic procedures after penile cancer diagnosis in order to improve patients’ disease-specific survival and limit the impact on quality of life. Here, we focused on the role of prophylactic inguinal and/or pelvic lymph node excision. The European Association of Urology (EAU) guidelines on penile cancer recommend a radical inguinal lymph node dissection in all patients with clinically positive lymph nodes (cN1/cN2) [5]. Moreover, the EAU guidelines suggest offering a prophylactic pelvic lymph node dissection to patients with three or more positive inguinal nodes or extra nodal extension of cancer [5]. As regards the timing of the procedure, the EAU guidelines suggest performing surgical inguinal and pelvic nodal extirpation within 3 months of penile cancer diagnosis [5], thereby highlighting the importance of an early lymphadenectomy to improve the patient’s outcome. In a retrospective study, Kroon et al. compared the clinical outcome of early versus delayed lymph node metastases dissection in patients affected by penile carcinoma [4]. By means of multivariate analysis, they demonstrated that early therapeutic dissection of occult inguinal penile cancer metastases was an independent prognostic factor for disease-specific survival [4]. Based on a cohort study of 22 patients, Johnson et al. reported that early therapeutic dissection of lymph nodes was associated with an increased 3- to 5-year survival rate as compared with patients who underwent late dissections [6]. The EAU guidelines highlight that a delay in nodal management of more than 3 to 6 months may reduce disease-free survival [5]. However, the strength of the EAU recommendation for a complete inguinal and pelvic nodal dissection within 3 months of diagnosis is weak, and the level of evidence is 3 [5]. The aim of this article is to review all existing data on the impact of the timing of inguinal and pelvic prophylactic lymph node dissection on the outcomes in patients with penile cancer.
Research Questions
We put forth the following research query: what is the optimal timing of prophylactic inguinal and/or pelvic lymph node dissection after penile cancer diagnosis for the patients’ disease-specific survival rates?
To answer this query, we performed a systematic review of available studies that have been performed to evaluate the impact of prophylactic inguinal and/or pelvic lymph node excision on patients’ survival rates. We have not included in this analysis the role of pre-operative or post-operative radiation therapy.
2. Materials and Methods
Research Strategy
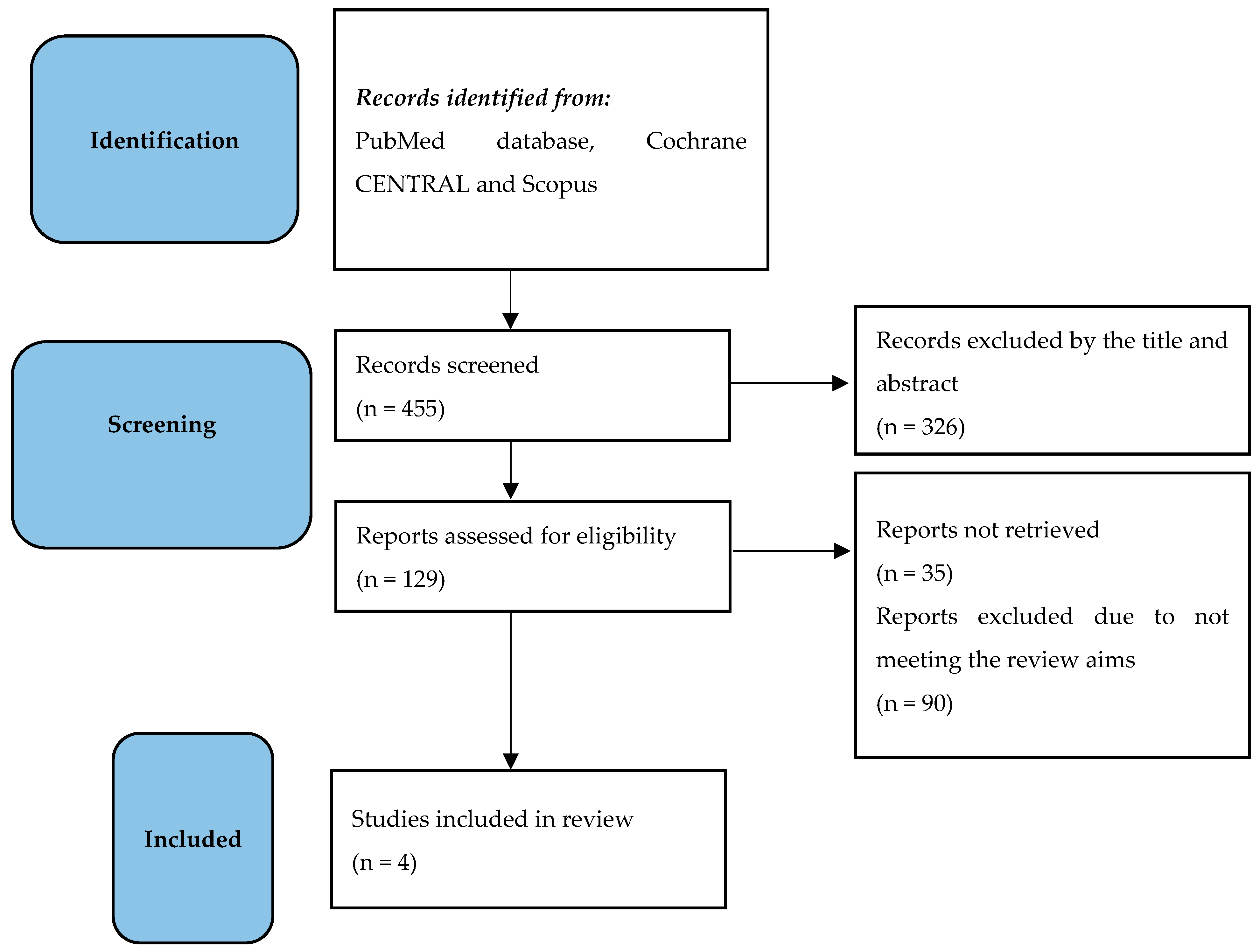
In order to respond to the research questions, from December 2022 to February 2023, two independent urologists (T.C. and M.C.) performed the research in the following databases: PubMed, Cochrane CENTRAL, and Scopus. All data have been collected in a dedicated database and independently analyzed by two urologists (T.C. and M.C.). Any disagreements between the two reviewers were resolved by consulting a supervisor (A.P.). All references cited in relevant articles were also reviewed and analyzed. The search strategy used was (((“Penile Neoplasms” (MeSH)) OR (penile cancer)) OR (penile carcinoma)) AND (lymph node excision (MeSH)) OR (lymph node dissection)) OR (lymphadenectomy)) OR (pelvic lymph node dissection)) OR (pelvic lymphadenectomy)) AND (survival)). As filters, we used clinical trial, humans, English language, and adult. Titles and abstracts were used to screen for the first study inclusion. A full-text review was used where abstracts were insufficient to determine if the study met inclusion or exclusion criteria. Two independent urologists and authors (T.C. and M.C.) performed all data analysis, including evaluation of the study characteristics, risk of bias, and outcome measures, with independent verification performed by the supervisor. The study has been performed in line with the preferred reporting items for systematic reviews and meta-analyses (PRISMA) [7]. All selected trials were used for this systematic review (Figure 1).
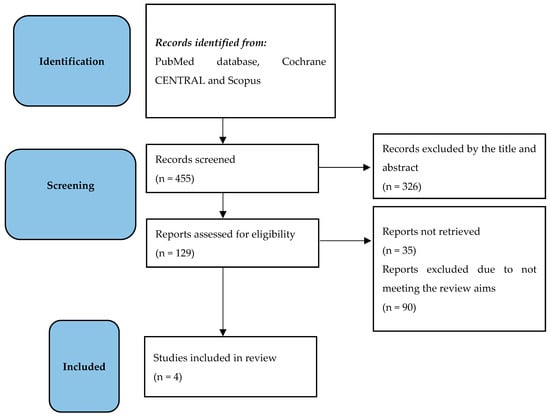
Figure 1.
The flowchart shows the outcome of the literature search, screening, and inclusion in line with the PRISMA statement.
3. Research Evidence
3.1. Indication for Inguinal Lymph Node Dissection
The lymph node metastases from penile carcinoma follows the anatomical drainage way from the primary tumor to inguinal lymph nodes and, then, to the ipsilateral pelvic lymph nodes. Lymphatic spreading to lymph nodes localized at the retroperitoneal level, such as para-aortic and para-caval, is considered a systemic metastatic disease. The overall survival rate is mostly dependent on the extent of the inguinal lymph node disease, with a 5-year survival rate for patients with a single involved inguinal node of about 80%, as compared with 0–12% if deeper pelvic lymph nodes are involved [8]. In this sense, an early lymph node dissection seems to be related to a better outcome. As of today, inguinal lymph node therapeutic excision is recommended for patients with the following characteristics [5,8]:
- -
- Palpable lymph nodes (cN1-cN2);
- -
- pT2 or higher penile cancer stage, even in the case of non-palpable lymph nodes;
- -
- Presence of lymphovascular invasion;
- -
- Poorly differentiated histology.
Imaging is of limited value when palpable lymph nodes are found during clinical examination. Instrumental evaluation with a CT scan or MRI is only indicated to detect distant metastasis. The use of a CT scan or MRI for avoiding unnecessary lymph node excision and reducing the morbidity after surgical staging is not recommended due to the limited accuracy of these instrumentations in lymph node staging. In this sense, CT scan or MRI findings do not alter the patients’ management [8]. Moving to everyday clinical practice, some aspects should be taken into account: first, the role of antimicrobial treatment in the suspicion that enlarged lymph nodes may be due to an infection. The use of antimicrobial agents in this kind of patient is not only supported by international guideline recommendations, but it is dangerous due to the high risk of antibiotic resistance. In select cases, fine needle aspiration cytology is an alternative for clarifying the reason for enlarged lymph nodes, having a sensitivity of 93% and specificity of 91% in predicting metastatic disease [9]. However, the role of fine needle aspiration cytology has been decreasing in recent years due to the risk of overlooking micro-metastasis [8]. Patients with fixed or massive nodal disease diagnosed at the moment of a clinical visit (cN3) gives rise to the suspicion of extensive lymphatic metastatic disease with a poor prognosis. In such cases, a total-body CT scan is required to look for distant metastasis and consider the need for chemotherapy [10]. The role of inguinal lymph nodes dissection is extremely important in the management of penile cancer patients, and the indications for the procedure should be respected and followed duly, as reported by Ren et al. [11].
3.1.1. Timing of Inguinal Lymph Node Dissection
Even if the indications for lymph node therapeutic excision are clear and well described in international guidelines [5,10], there is no consensus on the timing of the dissection. In cN0 patients (non-palpable lymph nodes), an early inguinal lymph node excision is associated with a better survival rate than delayed dissection (within 6 weeks of the first surgical approach vs. surgery following identification of positive nodes during the follow-up visits) [4,5,6]. In the case of pN1-N2 patients, the evidence is not clear. Some authors report a better outcome after early inguinal lymph node dissection (within 3 months of penile cancer diagnosis) [12], while other authors consider a time interval of less than 6 months from primary surgery as critical for the survival rate [13]. Available evidence about the timing of inguinal lymph node dissection does not support a clear recommendation of a specific time interval from diagnosis to lymph node dissection. International guidelines highlight that a delay of lymph node therapeutic dissection of more than 3 to 6 months may affect patients’ survival [4,5,6]. Recently, Ma et al. demonstrated in a retrospective, single-center study that among high-risk patients, 3.5 months is a safe oncological interval before surgical management of inguinal lymph nodes [2]. They also concluded that cN0 penile carcinoma patients at high risk of inguinal lymph node metastasis (≥pT1G3) benefit from simultaneous treatment management of the primary tumor and inguinal lymph nodes or a prophylactic inguinal lymphadenectomy within 3 months of diagnosis [2]. We therefore consider three months as a safe oncological window to ensure an optimal survival rate.
3.1.2. Anatomical Considerations of Inguinal Lymph Node Dissection
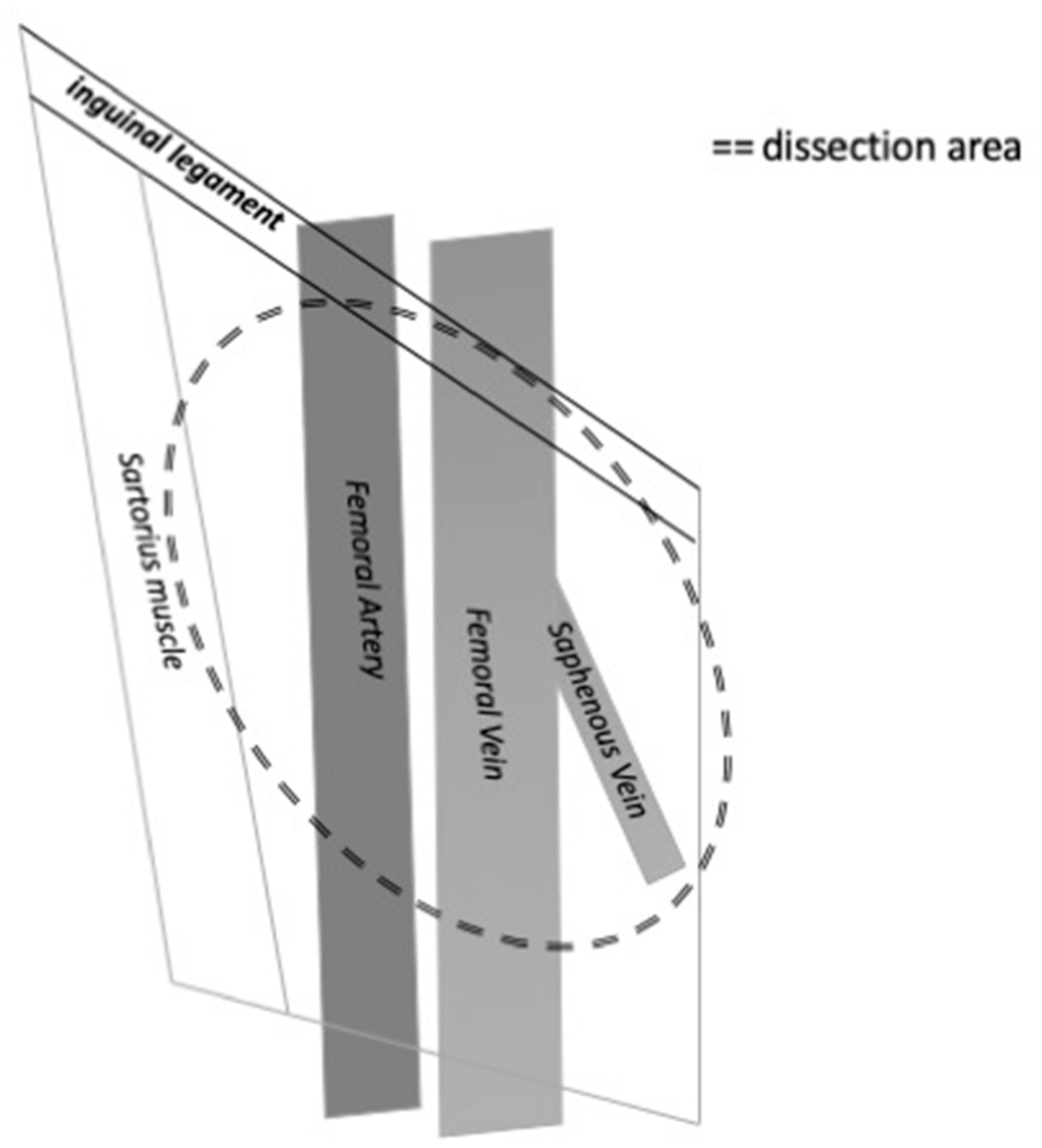
The inguinal lymph nodes are located close to the saphenous vein and femoral vessels within the femoral triangle, demarcated by the inguinal ligament superiorly, the adductor longus muscle medially, and the sartorius muscle laterally [8]. The extension of the lymph node dissection should be balanced with the risk of complications due to the dissection. Several modifications of the dissection technique have been reported to minimize morbidity and complications. It is mandatory to include the central and the superior zone of lymphatic tissue in the dissection. A modified inguinal lymph node dissection includes a smaller skin incision, preservation of the saphenous vein, conservation of the region superior to the fossa ovalis and lateral to the femoral artery, and maintenance of the sartorius muscle in situ [14]. This procedure has been associated with a decreasing rate of major complications [15] while maintaining the rate of diagnostic accuracy and the survival rate. On the other hand, a superficial lymph node dissection has been purposed with interesting results. This procedure is carried out with a 6–8 cm incision parallel and inferior to the inguinal line and includes the excision of flaps deep in the Scarpa’s fascia. The fascia lata is kept intact, while all nodal tissue superficial to it is removed. Compared to the modified inguinal lymph node dissection, the limits of the superficial dissection are extended to the median line and to the lateral margin in order to include the border line of the adductor longus muscle and the lateral boundary of the midpoint of the sartorius muscle. The long saphenous vein is preserved [16] (Figure 2).
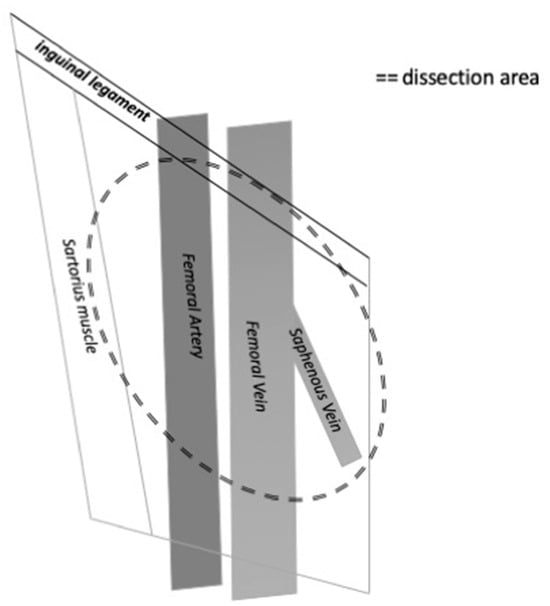
Figure 2.
The figure shows the anatomical limits of inguinal lymph node dissection on the right side.
3.2. Indications for Pelvic Lymph Node Dissection
According to the EAU guidelines, prophylactic pelvic lymph node dissection should be offered to all patients with three or more positive inguinal nodes or with extra nodal extension of cancer in inguinal nodes [5]. The accuracy of a CT scan or PET/CT scan to detect metastases in pelvic lymph nodes is low, just as it is for detecting metastases in inguinal lymph nodes. Sadeghi reported in a systematic review and metanalysis that 18F-FDG PET/CT imaging has relatively low sensitivity for the detection of inguinal lymph node involvement in penile cancer patients and does not recommend this examination for routine use [17]. On the other hand, they suggested performing 18F-FDG PET/CT imaging in patients with palpable inguinal lymph nodes due to its higher sensitivity in this subgroup of patients [17]. However, the presence of palpable inguinal lymph nodes is an indication for inguinal lymph node excision. Recently, a systematic review and metanalysis by Lee et al. showed that 18F-FDG PET/CT had a good diagnostic performance for the detection of metastatic lymph nodes, refreshing the role of this imaging modality in the diagnostic work-up of patients with penile carcinoma [18]. The role of 18F-FDG PET/CT in the diagnostic pathway is justified by enhanced anatomical information as compared to the cross-sectional assessment of CT scans. Lymph nodes of normal size that harbor malignant deposits can be detected by FDG PET/CT due to increased metabolism but not by CT alone [19]. However, in everyday clinical practice, the positive pelvic lymph nodes at PET/CT or CT should not be considered an indication for pelvic lymph node dissection unless there is histopathologically confirmed homolateral inguinal metastasis.
3.2.1. Timing of Pelvic Lymph Node Dissection
International guidelines suggest performing an accurate inguinal and pelvic lymph node excision within 3 months of the initial diagnosis [5]. This recommendation is based on the association between the presence of lymph node metastases and patients’ survival rates. The recommendation is controversial as it is offered even though there is poor evidence that pelvic lymph node dissection improves survival compared to adjuvant radiotherapy or surveillance only. Recently, great attention has been directed toward the percutaneous biopsy of enlarged pelvic lymph nodes. The NCCN Guidelines in Oncology on Penile Cancer suggest performing a percutaneous lymph node biopsy if technically feasible and to decide on pelvic lymph node excision on the basis of biopsy results [20]. However, if pelvic lymph node metastases are suspected through imaging or biopsy, we believe pelvic lymph node dissection should be performed as early as possible in order to avoid the further spread of penile cancer cells.
3.2.2. Anatomical Considerations of Pelvic Lymph Node Dissection
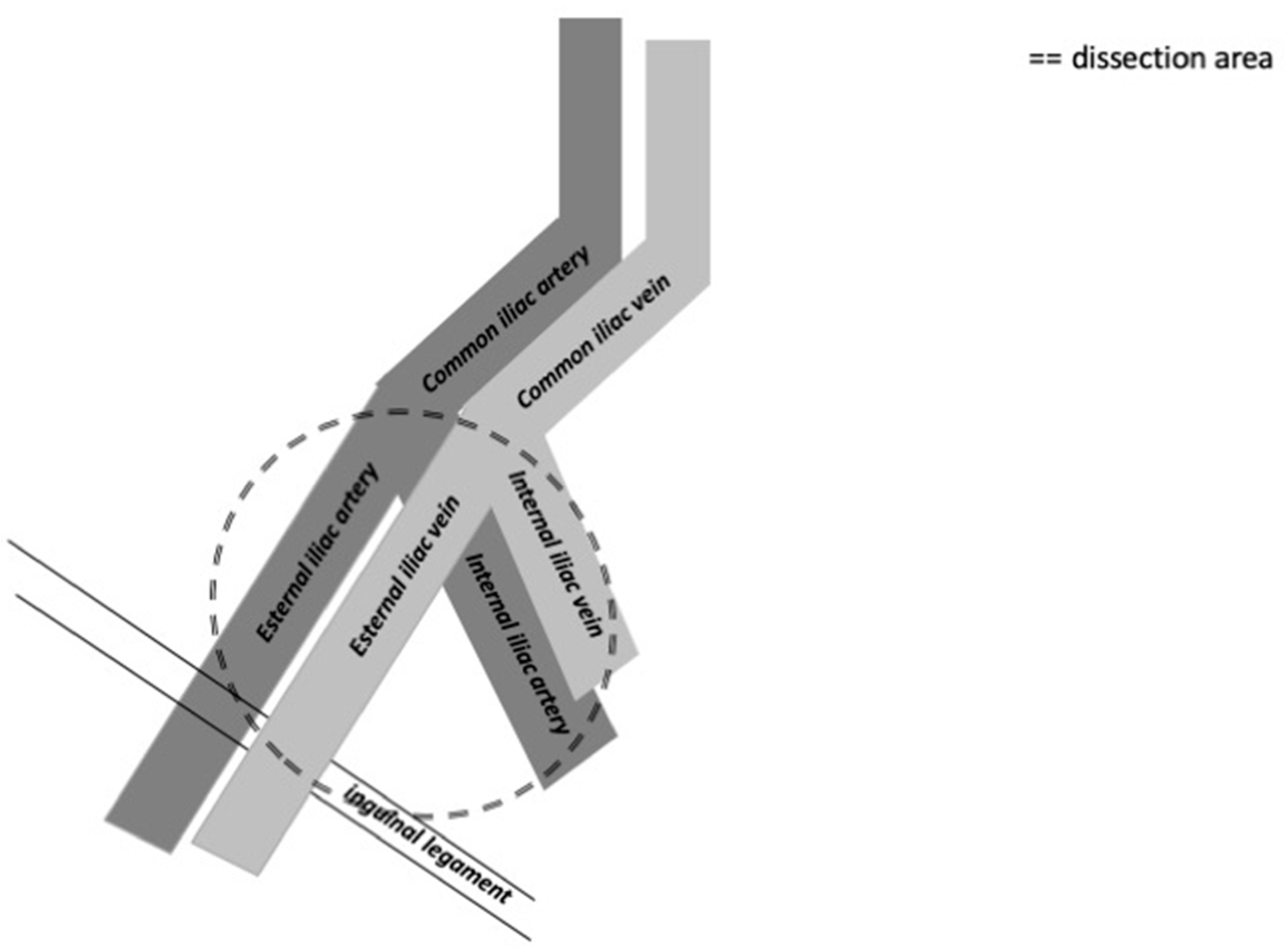
The distribution of the most common positive lymph nodes is in the external iliac, obturator, common iliac, internal iliac, and presacral zones, according to Yao et al. [21]. Crossover metastasis from one inguinal region to the contralateral pelvic region is very rare [21]. The external iliac zone is more often involved than the obturator and common iliac zones [22]. The template used for cystectomy is applicable also for penile cancer and usually includes the distal common iliac, the external iliac, the obturator, and the internal iliac lymph nodes bilaterally [23]. The anatomical limits are the common iliac bifurcation cranially and the inguinal ligament caudally. Laterally, the boundary is the genitofemoral nerve, and medially, it is the bladder wall (Figure 3).
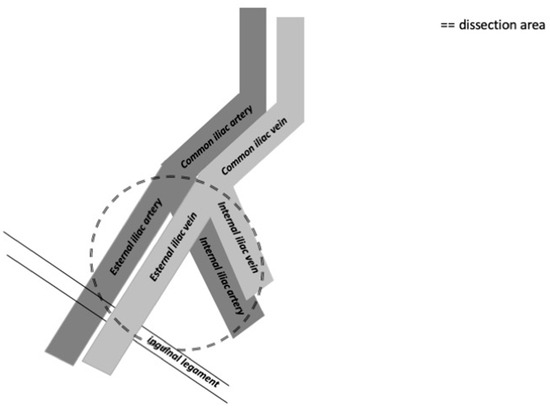
Figure 3.
The figure shows the anatomical limits of pelvic lymph node dissection on the right side.
3.3. Pathological Considerations on Lymph Node Analysis
Due to the high importance of the pathological parameters in decision making for the diagnostic and therapeutic approaches to patients affected by penile cancer, some pathological considerations should be observed in everyday clinical practice. The histologic grade and tumor stage are important prognostic factors in patients affected by penile cancer. Ficarra et al. demonstrated that the histological grade can be considered a predictive prognostic factor in patients with lymph node metastasis, too [24]. In this sense, the correct assessment of histologic grade is essential in the management planning of patients with penile cancer. Moreover, as stated before, the EAU guidelines recommended the performance of a prophylactic pelvic lymph node dissection for all patients with three or more positive inguinal nodes or with an extra nodal extension of cancer in the inguinal nodes [5]. The following parameters of pathological analysis should be considered in the planning of patients’ management:
- -
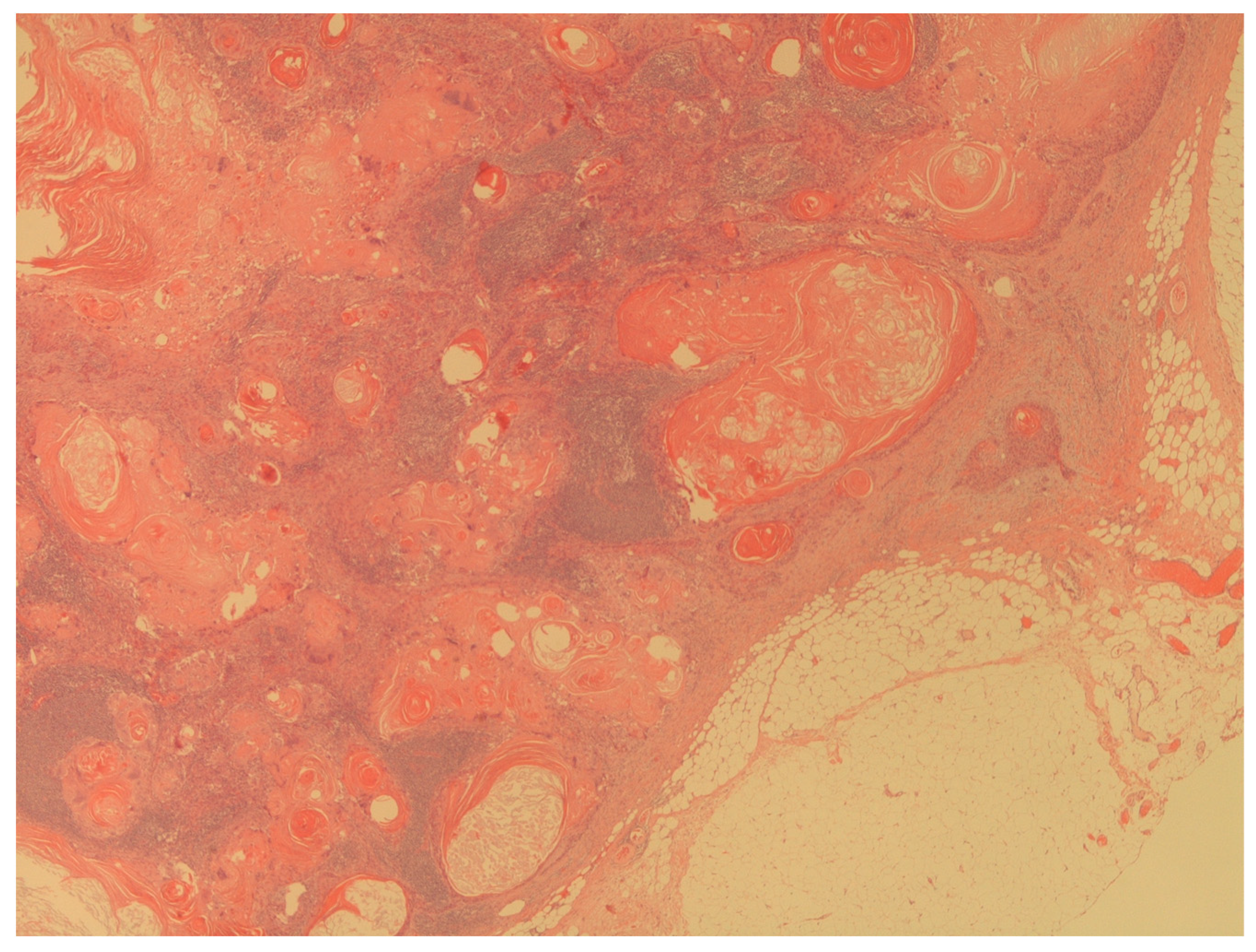
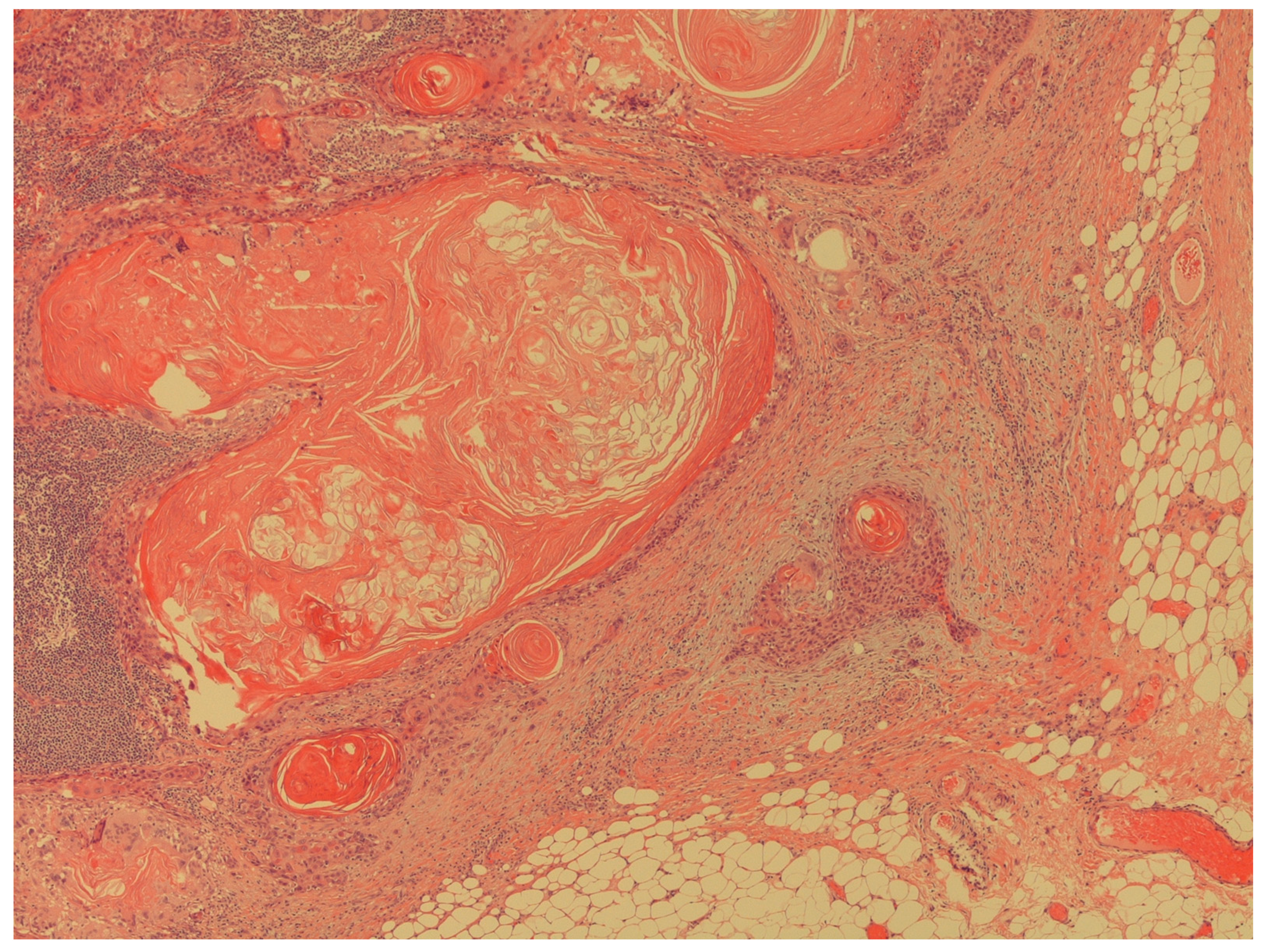
- Presence of penile cancer lymph nodes metastasis (Figure 4);
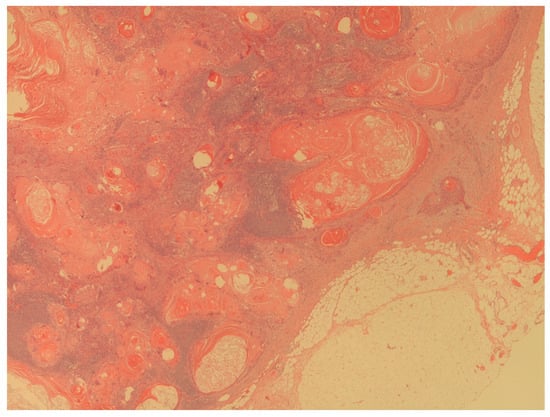 Figure 4. The figure shows a massive lymph node metastasis from squamous cell carcinoma (H&E; 20X).
Figure 4. The figure shows a massive lymph node metastasis from squamous cell carcinoma (H&E; 20X). - -
- Number of lymph nodes involved;
- -
- Presence of extra nodal extension of cancer in lymph nodes (Figure 5).
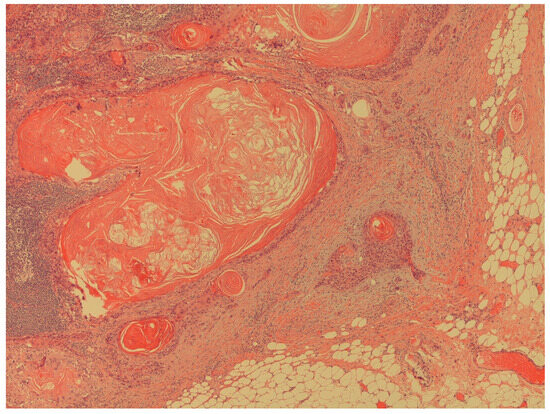 Figure 5. The figure shows an extracapsular neoplastic extension from the foci of squamous cell carcinoma (H&E; 40X).
Figure 5. The figure shows an extracapsular neoplastic extension from the foci of squamous cell carcinoma (H&E; 40X).
The above pathological parameters are essential to guide the diagnostic pathway and to plan the future surgical approach. Moreover, according to the current American Joint Committee on Cancer (AJCC) staging system, any pelvic lymph node involvement or extracapsular extension of all regional lymph nodes is classified as pN3 [25]. The number of nodes that stratify the staging between N1 and N2 are two or more unilateral nodes. The diameter of the largest tumor deposit (not of the lymph node) can influence prognosis in penile cancer and should always be recorded (measured in millimeters). Limited sizes of tumors (up to 0.2 mm) are classified as micrometastases and isolated tumor cells as N0 (i+). The HPV assessment (at least p16 IHC-based) has been required by the international guidelines to be included in the pathology reports of the primary tumor but not in the lymph node evaluation.
3.4. Systematic Review Findings on the Timing of Lymph Node Dissection
In order to demonstrate the impact of the timing of the lymph node dissection on survival rates in patients with penile cancer, we included four papers (Table 1). The lack of randomized clinical trials is the reason for not performing a regular systematic review. All included studies argue for an early lymph node dissection for improving patients’ survival [4,5,6,12,13]. As regards the exact timing of dissection, Kroon et al. identified a cut-off at 6 months as the best interval for the resection of lymph nodes [4]. On the other hand, Chipollini et al. recommended 3 months as the upper time limit for performing inguinal lymph node excision [12], while Gulia et al. suggested performing inguinal lymph node dissection within 2 months of diagnosis [13]. No studies address the best timing for pelvic lymph node dissection. On the basis of these studies, and despite the lack of randomized and controlled trials, we recommend that lymph node dissection should be performed as soon as possible after diagnosis, with 3 months as a realistic upper cut-off time.

Table 1.
The table shows the summary of all included clinical trials in the systematic review.
4. Limitations
The lack of deep knowledge about the role of lymph node dissection in penile cancer is due also to clinical heterogeneity among published clinical studies. Moreover, the time between the glans lesion appearance and the first urological evaluation ranges from a few days to one or two years, as reported by some case reports [26,27]. In these cases, the delayed diagnosis is due to the significant impact of penile cancer on the patient’s self-image and self-esteem. On the other hand, there is no generally accepted indication for lymph node dissection, different lymph node dissection templates are being used, there are a low number of included patients in published series, and the interference of neo/adjuvant treatments varies. No clinically relevant predictive tools are available for assessing the risk of inguinal or pelvic lymph node metastases. The indications for performing lymph node dissection is based on retrospective studies, and recommendations for timing are based on expert opinion, resulting in weak grades of recommendations by international guidelines. No molecular data are available for use in everyday clinical practice to guide the extent of surgery or adjuvant treatment. Genetic analysis of the primary tumor and metastatic cells in lymph nodes might provide us with the molecular parameters to assess the risk of metastatic spread and the survival rate.
5. Future Perspectives
Penile cancer is an aggressive disease that requires early management. The survival rate depends on an early surgical approach in order to prevent the cancer from spreading into regional lymph nodes and to avoid distant metastases [5]. Metastatic involvement of inguinal lymph nodes is the most important prognostic indicator in penile cancer, and understanding the optimal time and extension of prophylactic lymph node dissection is therefore essential. There are also other gray zones related to lymph node therapeutic excision in penile cancer. Recently, Suarez-Ibarrola et al. suggested that despite the rarely found crossover metastasis from inguinal to pelvic lymph nodes, bilateral pelvic lymph node therapeutic excision should be carried out even if the ideal candidate is yet to be determined [28]. Finally, special attention to the impact of lymph node dissection on patients’ quality of life should be considered [29]. The most important outcomes in the management of patients affected by penile cancer are, then, improved survival and quality of life. These two aspects are strictly associated because an early surgical approach to penile cancer is associated with the possibility to perform conservative treatment. Due to the high psychological impact that penile cancer diagnosis has on patients, the adherence to the principles of patient-centered medicine is essential in order to engage the patient and improve the patient–doctor alliance [29]. Appropriate counselling about treatment efficacy and possible complications for urinary and sexual functions has a pivotal role in increasing the patients’ adherence to the treatment [29]. In other words, the preservation of quality of life should be considered an outcome of the treatment, as well as disease-free survival.
6. Conclusions
The survival of patients affected by penile cancer is strictly associated with early diagnosis and surgical treatment. Even though there are no randomized controlled trials, we conclude that in the management of patients affected by penile cancer the following key points should be considered: perform lymph nodes dissection as soon as possible in order to avoid the spread of cancer cells and to improve survival. Moreover, 3 months is regarded as the upper limit for the time interval between diagnosis and lymph node dissection. These recommendations have been provided by international guidelines to improve patients’ survival and preserve quality of life. Surgical treatment should be offered, then, following the recommendations of international guidelines and following patients’ perspectives. A modern approach to genitourinary cancers includes the adherence to the principles of a patient-centered approach and the awareness that the patient’s sexual partner adds an extra dimension to the patients’ QoL concept [29]. According to the present evidence base, lymph node dissection should be offered, the sooner the better, to all patients at high risk of lymph node metastasis.
Author Contributions
Conceptualization, T.C.; methodology, T.C. and M.C.; software, T.C., M.G.Z.; formal analysis, T.C. and M.C.; data curation, T.C., M.C. and M.G.Z.; writing—original draft preparation, T.C. and G.N.; writing—review and editing, A.P. and T.E.B.J.; supervision, M.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Salvioni, R.; Necchi, A.; Piva, L.; Colecchia, M.; Nicolai, N. Penile cancer. Urol. Oncol. 2009, 27, 677–685. [Google Scholar] [CrossRef]
- Ma, S.; Zhao, J.; Liu, Z.; Wu, T.; Wang, S.; Wu, C.; Pan, L.; Jiang, X.; Guan, Z.; Wang, Y.; et al. Prophylactic inguinal lymphadenectomy for high-risk cN0 penile cancer: The optimal surgical timing. Front. Oncol. 2023, 13, 1069284. [Google Scholar] [CrossRef]
- Pow-Sang, M.R.; Ferreira, U.; Pow-Sang, J.M.; Nardi, A.C.; Destefano, V. Epidemiology and natural history of penile cancer. Urology 2010, 76 (Suppl. S1), S2–S6. [Google Scholar] [CrossRef]
- Kroon, B.K.; Horenblas, S.; Lont, A.P.; Tanis, P.J.; Gallee, M.P.; Nieweg, O.E. Patients with penile carcinoma benefit from immediate resection of clinically occult lymph node metastases. J. Urol. 2005, 173, 816–819. [Google Scholar] [CrossRef]
- Available online: https://d56bochluxqnz.cloudfront.net/documents/full-guideline/EAU-ASCO-Guidelines-on-Penile-Cancer-2023_2023-03-08-131333_piyo.pdf (accessed on 3 September 2023).
- Johnson, D.E.; Lo, R.K. Management of regional lymph nodes in penile carcinoma. Five-year results following therapeutic groin dissections. Urology 1984, 24, 308–311. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- The, J.; Duncan, C.; Qu, L.; Guerra, G.; Narasimhan, V.; Pham, T.; Lawrentschuk, N. Inguinal lymph node dissection for penile cancer: A contemporary review. Transl. Androl. Urol. 2020, 9, 3210–3218. [Google Scholar] [CrossRef]
- Saisorn, I.; Lawrentschuk, N.; Leewansangtong, S.; Bolton, D.M. Fine-needle aspiration cytology predicts inguinal lymph node metastasis without antibiotic pretreatment in penile carcinoma. BJU Int. 2006, 97, 1225–1228. [Google Scholar] [CrossRef] [PubMed]
- Clark, P.E.; Spiess, P.E.; Agarwal, N.; Biagioli, M.C.; Eisenberger, M.A.; Greenberg, R.E.; Herr, H.W.; Inman, B.A.; Kuban, D.A.; Kuzel, T.M.; et al. Penile cancer: Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2013, 11, 594–615. [Google Scholar] [CrossRef]
- Ren, S.; Shao, Y.; Bao, Y.; Li, X. Prediction of survival in patients with penile cancer after lymph node dissection. Asian J. Surg. 2022, 45, 1700–1702. [Google Scholar] [CrossRef]
- Chipollini, J.; Tang, D.H.; Gilbert, S.M.; Poch, M.A.; Pow-Sang, J.M.; Sexton, W.J.; Spiess, P.E. Delay to Inguinal Lymph Node Dissection Greater than 3 Months Predicts Poorer Recurrence-Free Survival for Patients with Penile Cancer. J. Urol. 2017, 198, 1346–1352. [Google Scholar] [CrossRef] [PubMed]
- Gulia, A.K.; Mandhani, A.; Muruganandham, K.; Kapoor, R.; Ansari, M.S.; Srivastava, A. Impact of delay in inguinal lymph node dissection in patients with carcinoma of penis. Indian J. Cancer 2009, 46, 214–218. [Google Scholar] [CrossRef]
- Catalona, W.J. Modified inguinal lymphadenectomy for carcinoma of the penis with preservation of saphenous veins: Technique and preliminary results. J. Urol. 1988, 140, 306–310. [Google Scholar] [CrossRef]
- Spiess, P.E.; Hernandez, M.S.; Pettaway, C.A. Contemporary inguinal lymph node dissection: Minimizing complications. World J. Urol. 2009, 27, 205–212. [Google Scholar] [CrossRef]
- Bevan-Thomas, R.; Slaton, J.W.; Pettaway, C.A. Contemporary morbidity from lymphadenectomy for penile squamous cell carcinoma: The M.D. Anderson Cancer Center Experience. J. Urol. 2002, 167, 1638–1642. [Google Scholar] [CrossRef]
- Sadeghi, R.; Gholami, H.; Zakavi, S.R.; Kakhki, V.R.; Horenblas, S. Accuracy of 18F-FDG PET/CT for diagnosing inguinal lymph node involvement in penile squamous cell carcinoma: Systematic review and meta-analysis of the literature. Clin. Nucl. Med. 2012, 37, 436–441. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.W.; Kim, S.J. Diagnostic Performance of 18F-FDG PET/CT for Lymph Node Staging in Penile Cancer. Clin. Nucl. Med. 2022, 47, 402–408. [Google Scholar] [CrossRef]
- Jakobsen, J.K.; Frahm Nielsen, T.; Ipsen, P.; Albrecht-Beste, E.; Cardoso Costa, J.; Alslev, L.; Predbjørn Krarup, K.; Grønkaer Toft, B.; Høyer, S.; Bouchelouche, K.; et al. DaPeCa-7: Comparative assessment of fluorodeoxyglucose positron emission tomography/computed tomography (CT) and conventional diagnostic CT in diagnosis of lymph node metastases, distant metastases and incidental findings in patients with invasive penile cancer. BJU Int. 2021, 127, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www2.tri-kobe.org/nccn/guideline/urological/english/penile.pdf (accessed on 3 September 2023).
- Yao, K.; Chen, Y.; Ye, Y.; Wu, Z.; Chen, D.; Han, H.; Li, Z.; Liu, Z.; Wang, Y.; Qin, Z.; et al. Lymph Node Mapping in Patients with Penile Cancer Undergoing Pelvic Lymph Node Dissection. J. Urol. 2021, 205, 145. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhang, S.L.; Ye, D.W.; Yao, X.D.; Dai, B.; Zhang, H.L.; Shen, Y.J.; Zhu, Y.P.; Shi, G.H.; Ma, C.G. Prospectively packaged ilioinguinal lymphadenectomy for penile cancer: The disseminative pattern of lymph node metastasis. J. Urol. 2009, 181, 2103. [Google Scholar] [CrossRef] [PubMed]
- Perera, M.; McGrath, S.; Sengupta, S.; Crozier, J.; Bolton, D.; Lawrentschuk, N. Pelvic lymph node dissection during radical cystectomy for muscle-invasive bladder cancer. Nat. Rev. Urol. 2018, 15, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Ficarra, V.; Martignoni, G.; Maffei, N.; Cerruto, M.A.; Novara, G.; Cavalleri, S.; Artibani, W. Predictive pathological factors of lymph nodes involvement in the squamous cell carcinoma of the penis. Int. Urol. Nephrol. 2002, 34, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.B.; Edge, S.; Greene, F.L.; Byrd, D.R.; Brookland, R.K.; Washington, M.K.; Gershenwald, J.E.; Compton, C.C.; Hess, K.R.; Sullivan, D.C.; et al. (Eds.) AJCC Cancer Staging Manual, 8th ed.; Springer: New York, NY, USA, 2017. [Google Scholar]
- Kamat, A.M.; Carpenter, S.M.; Czerniak, B.A.; Pisters, L.L. Metastatic penile cancer in a young Caucasian male: Impact of delayed diagnosis. Urol. Oncol. 2005, 23, 130–131. [Google Scholar] [CrossRef] [PubMed]
- Reyes, A.J.; Ramcharan, K.; Goetz, L.; Aboh, S. Penectomy for self-neglected opportunistic carcinoma: A large primary urogenital squamous cell carcinoma as initial manifestation of HIV-AIDS. BMJ Case Rep. 2016, 2016, bcr2016217040. [Google Scholar] [CrossRef] [PubMed]
- Suarez-Ibarrola, R.; Basulto-Martinez, M.; Sigle, A.; Abufaraj, M.; Gratzke, C.; Miernik, A. Is There an Oncological Benefit of Performing Bilateral Pelvic Lymph Node Dissection in Patients with Penile Cancer and Inguinal Lymph Node Metastasis? J. Clin. Med. 2021, 10, 754. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.; Verze, P.; Bjerklund Johansen, T.E. The Quality of Life Definition: Where Are We Going? Uro 2021, 1, 14–22. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).