Abstract
Background: The prognostic trends for adults who undergo fracture surgery for metastatic long bones remain unclear, even as survival among individuals with bone metastasis has improved. Because prognoses after fracture surgery may shift over time, this chronological study aimed to evaluate outcomes in individuals who received surgical treatment for femoral metastasis. Methods: This multicenter retrospective study included 186 adults who underwent fracture surgery for metastatic femoral disease between 2008 and 2023. Individuals were categorized into period 1 (2008–2016) and period 2 (2017–2023). The primary outcome was 1-year overall survival. Prognostic variables included fracture type (impending pathological fracture (IF) or completed pathological fracture (CF)), recovery of ambulation, and receipt of chemotherapy. Results: Individuals in period 2 were older and had a higher rate of solitary bone metastasis; however, period 2 did not demonstrate a higher proportion of IF relative to CF. Multivariate analysis showed that the hazard ratio for period 2 compared with period 1 was 0.57 (95% confidence interval: 0.35 to 0.94, p = 0.02). Conclusions: This chronological assessment demonstrated that the later period was associated with improved 1-year overall survival after fracture surgery for metastatic long bone disease. Because prophylactic fixation for IF is linked to more favorable outcomes, increasing the proportion of IF cases relative to CF should be prioritized.
1. Introduction
The incidence of bone metastasis has increased as individuals with cancer are living longer [1]. Advances in diagnostic and therapeutic strategies have improved the management of bone metastasis. Lung, breast, and prostate cancers are the most common sources of metastatic bone disease, and targeted therapies for these malignancies inhibit proteins, enzymes, or molecules involved in tumor proliferation and spread [2]. Although median survival has improved for individuals with bone metastasis [3,4], nearly half still experience skeletal-related events (SREs), which contribute to morbidity and mortality [5,6]. Because bone metastasis compromises structural integrity, it can lead to pathological fractures, a major SRE [7]. When a lesion is at high risk for a completed pathological fracture (CF), prophylactic stabilization for an impending pathological fracture (IF) is recommended to prevent additional morbidity [7,8,9]. Prophylactic fixation is often less complex and allows broader surgical planning [7], and individuals treated for IF generally recover ambulation better and have lower mortality than those presenting with CF [7,8,10].
Mirels’ score [11], originally described in 1989 and republished in 2010 [11], remains widely used to define IF. It is manually calculated using four variables: lesion site, type, size, and pain. Recent studies still rely on these criteria [7,8,9,12,13,14], and the system remains widely referenced because of its simplicity [15].
Despite improved survival among individuals with bone metastasis [3,4], chronological trends in the characteristics and prognoses of those undergoing fracture surgery for metastatic long bones remain unclear. Diagnostic strategies for identifying IF have remained stable, but prognoses after surgical fixation may have changed. Therefore, this study aimed to evaluate the prognoses of individuals who underwent surgical treatment for femoral metastatic fractures.
2. Materials and Methods
2.1. The Study Design
This multicenter retrospective study reviewed medical records of 216 individuals with malignant tumors who underwent fracture surgery for femoral metastasis between January 2008 and January 2023 (Figure 1). Fracture sites were evaluated preoperatively using magnetic resonance imaging, positron emission tomography–computed tomography, and/or computed tomography (CT) in addition to plain radiography. Fractures were defined using imaging findings and/or pathological assessment, and 186 individuals were included in the final analysis. Exclusion criteria were as follows: no tumor mass at the fracture site on imaging (indicating a non-pathological fracture), revision procedures, surgeries for multiple bone lesions, and a diagnosis of cognitive impairment.
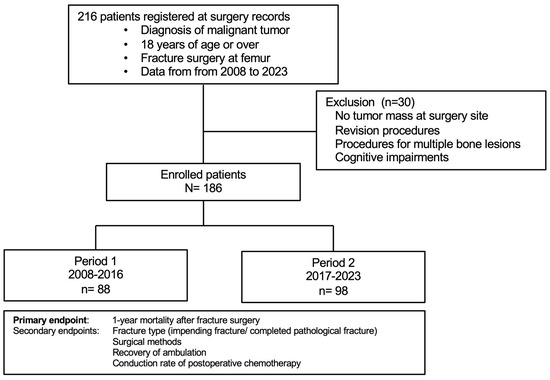
Figure 1.
Study design.
Based on the surgery date, enrolled individuals were grouped into period 1 (2008–2016) and period 2 (2017–2023). The allocation divided the study duration into two equal intervals. The study was approved by the ethics committees of our hospital (022–0036) and all participating centers. Informed consent was obtained via the opt-out method, and the study followed the principles of the Declaration of Helsinki.
2.2. Variables and Outcomes
Medical records of individuals who underwent femoral surgery were reviewed, and demographic and clinicopathologic information was collected as previously described [8] (Table 1). IF was defined as a Mirels’ score of ≥ 8 [11]. The Katagiri–New score includes six prognostic factors: primary tumor type, visceral or cerebral metastasis, abnormal laboratory values, and Eastern Cooperative.

Table 1.
Demographic data.
Oncology Group Performance Status (ECOG PS), previous chemotherapy, and multiple skeletal metastases [16].
The primary outcome of this study was the 1-year mortality rate. As prognostic variables, fracture type (IF/CF), recovery of ambulation, postoperative chemotherapy, and 1-year survival after fracture surgery were evaluated. Recovery of ambulation was defined as the ability to walk with assistive devices within 60 days of postoperative hospitalization [9,10]. Chemotherapy included cytotoxic drugs, immune checkpoint inhibitors, and molecular targeted therapy.
2.3. Statistical Analysis
Categorical variables were evaluated using the chi-squared test for two or more groups, and continuous variables were analyzed using the independent t-test. Survival after surgery was visualized using the Kaplan–Meier method, and differences were assessed with the log-rank test and multivariate proportional hazards. All statistical analyses were performed with JMP Pro version 18 (SAS Institute, Inc., Cary, NC, USA), with the significance level set at 0.05.
3. Results
3.1. Patient Demographics
When clinicopathologic characteristics were compared between the two periods, age was higher in period 2 (61.2 ± 1.5 vs. 66.0 ± 1.4, p = 0.02), and multiple bone metastases were lower (55.7% vs. 31.5%, p = 0.01) (Table 1). Lung, breast, and renal cell carcinoma were the most frequent primary cancers in both groups. Gender distribution (48.9% vs. 43.8%, p = 0.49), body mass index (21.5 ± 0.4 vs. 21.9 ± 0.4, p = 0.48), Katagiri–New score (p = 0.38), primary cancer (p = 0.24), organ metastasis (54.5% vs. 43.9%, p = 0.25), and ECOG PS (p = 0.30) were similar between periods (Table 1).
In period 1, lung metastasis was most frequent at 21.6% (19/88), followed by liver metastasis at 12.5% (11/88). In period 2, lung metastasis was also most frequent at 23.5% (23/98), followed by liver metastasis at 10.2% (10/98).
In period 1, 40.1% (36/88) received conventional chemotherapy, 20.5% (18/88) received hormonal therapy, 6.8% (6/88) received molecular targeted therapy or immune checkpoint inhibitors, and 9.3% (17/88) received bone-modifying agents. In period 2, 40.8% (40/98) received conventional chemotherapy, 13.2% (13/98) received hormonal therapy, 12.2% (12/98) received molecular targeted therapy or immune checkpoint inhibitors, and 19.4% (19/98) received bone-modifying agents.
3.2. Perioperative Information
Period 2 did not show a higher ratio of IF to CF than period 1 (46 to 42 vs. 52 to 46, p = 0.91) (Table 2). The ratio of internal fixation to endoprosthesis was also similar (63 to 25 vs. 72 to 26, p = 0.77). In period 1, 42 individuals had CF, of whom 19.0% (8/42) received endoprosthesis and 81.0% (34/42) received internal fixation. In the same period, 46 individuals had IF, of whom 37.0% (17/46) received endoprosthesis and 63.0% (29/46 received internal fixation. In period 2, 46 individuals had CF, of whom 26.1% (12/46) received endoprosthesis and 73.9% (34/46) received internal fixation. In the same period, 52 individuals had IF, of whom 26.9% (14/52) received endoprosthesis and 73.1% (38/52) received internal fixation.

Table 2.
Perioperative information.
Across both periods, 88 individuals had CF, and 22.7% (20/88) received endoprosthesis, with the remainder treated using internal fixation. A total of 98 individuals had IF, and 31.7% (31/98) received endoprosthesis, with the remainder receiving internal fixation. Thus, surgical strategies did not differ substantially between CF and IF (20 to 68 vs. 31 to 67, p = 0.174).
As postoperative complications in period 1, 9.1% (8/88) had anemia requiring transfusion, 6.8% (6/88) had surgical site infection, 5.7% (5/88) had bronchitis or pneumonia, and 9.1% (8/88) were diagnosed with urinary tract infection. In period 2, 7.1% (7/98) had anemia requiring transfusion, 4.1% (4/98) had surgical site infection, 2.1% (2/98) had bronchitis or pneumonia, and 8.1% (8/98) were diagnosed with urinary tract infection.
Postoperative recovery of ambulation was similar between periods (81.8% vs. 80.6%, p = 0.83), as were rates of home discharge (58.0% vs. 65.3%, p = 0.31). Regarding adjuvant therapy, period 2 did not show a higher rate of postoperative chemotherapy (43.2% vs. 39.8%, p = 0.64) or perioperative radiotherapy to the surgical site (52.2% vs. 38.8%, p = 0.07) (Table 3). Specifically, in period 1, 28 individuals received preoperative radiotherapy and 18 received postoperative radiotherapy. In period 2, 24 received preoperative radiotherapy, and 14 received postoperative radiotherapy. The ratio of preoperative to postoperative radiotherapy did not differ between periods (28 to 18; 24 to 14, p = 0.83).

Table 3.
Cox proportional models for 1-year overall survival in patients after fracture surgery for long bone metastasis.
3.3. Postoperative Prognoses
The rate of loss to follow-up during one year was 5.4% (10/186). The 1-year mortality rate in period 1 was 53.0% (39/83), whereas it was 46.2% (43/93) in period 2 (p = 0.36); the univariate analysis showed a hazard ratio for period 2 compared with period 1 of 0.85 (95% confidence interval: 0.56 to 1.24, p = 0.46).
However, multivariate analysis adjusting for age, sex, body mass index, fracture type, ECOG PS, Katagiri score, and skeletal stabilization showed a hazard ratio for period 2 compared with period 1 of 0.57 (95% confidence interval: 0.35 to 0.94, p = 0.02).
4. Discussion
The chronological analysis revealed that the later period was a favorable prognostic factor for 1-year overall survival after fracture surgery for metastatic long bone. Among prognostic variables, age and the rate of solitary bone metastasis were higher in period 2, whereas the proportion of prophylactic surgery for IF did not differ between the two periods.
These findings suggest a trend toward improved prognoses in individuals undergoing fracture surgery for metastatic long bone, consistent with previous reports indicating that median survival in individuals with bone metastasis has increased over the past decades [2,3]. The primary goal of metastatic femoral surgery is the recovery of ambulation, which is closely related to postoperative rehabilitation. Cancer rehabilitation has been recognized and recommended by organizations such as the American Cancer Society and the American College of Sports Medicine for individuals with cancer [17,18,19]. Beyond surgical stabilization of pathological fractures, chemotherapy plays an essential role in maintaining activities of daily living. Fujii et al. reported that the absence of chemotherapy in individuals with bone metastases was associated with functional decline [20]. Similarly, Kobayashi et al. noted that individuals with bone metastases admitted to rehabilitation wards, where chemotherapy cannot be administered, often experience further reductions in functional capacity [21]. Additionally, Yoshikawa et al. showed that chemotherapy was a significant factor influencing activities of daily living and quality of life, as measured by quantitative indices such as the Barthel Index and the EuroQoL-5 Dimensions [19]. However, evidence regarding rehabilitation specifically for individuals with bone metastases remains limited [22]. Individuals with bone metastases have unique clinical characteristics and varying functional restrictions, requiring thoughtful modification of rehabilitation programs [17,18,19].
The present study did not identify changes in the ratios of IF to CF or in the prognoses of individuals who underwent fracture surgery for femoral metastasis. Although earlier studies reported improved median survival among individuals with bone metastasis over recent decades [3,4], it has remained uncertain whether prognoses have changed specifically in those with pathological fractures. Based on the findings of this study, together with accumulating evidence demonstrating the survival benefits of prophylactic intervention for IF, an increase in the proportion of IF relative to CF may improve prognoses in individuals with pathological fractures [7,8,9].
To increase the proportion of prophylactic interventions, promoting the use of Mirels’ score among non-orthopedic specialists may be beneficial, although substantial inter-rater variability has been reported among medical and radiation oncologists [23]. To reduce such variability and achieve more standardized assessments, developing unbiased automated algorithms that evaluate X-rays of metastatic long bone lesions may be advantageous. Several deep-learning-based models capable of detecting musculoskeletal abnormalities on radiographs have already been described [24,25,26].
CT-based diagnosis of IF may also provide an alternative approach. In clinical practice, individuals with bone metastases often first present to medical or radiation oncologists rather than orthopedic surgeons [27], and these individuals frequently undergo CT examinations. CT-based structural rigidity analysis has shown superior performance to the Mirels’ system [28]. Damron et al. assessed CT-based rigidity analysis for predicting fracture within one year in 125 individuals with femoral metastasis. They compared Mirels’ scores derived from X-ray with CT-based rigidity analysis from bilateral femoral CT scans and found that CT-based rigidity analysis demonstrated higher sensitivity (100% vs. 66.7%), specificity (60.6% vs. 47.9%), positive predictive value (17.6% vs. 9.8%), and negative predictive value (100% vs. 94.4%) compared with the classic Mirels’ threshold for IF (≥9) [28].
Further, the efficacy of longitudinal CT-based rigidity analysis has been demonstrated in preclinical models [29,30]. Bohanske et al. showed that CT-based rigidity analysis could capture real-time, progressive structural changes in rat femurs following intramedullary inoculation of a breast cancer cell line, with or without chemotherapy. In that study, osteolytic progression and fracture risk were quantified, revealing that CT-based rigidity analysis detected temporal changes in bone rigidity and consistently differentiated between chemotherapeutic regimens. The investigators concluded that CT-based rigidity analysis is a promising tool for accurately predicting fractures through objective assessment, compared with conventional X-ray-based methods [30]. Notably, healing status after fracture was also assessed noninvasively using CT-based rigidity analysis integrated with an implantable sensor that monitored healing through implant load. This study demonstrated a significant correlation between sensor data and CT-based rigidity analysis. These systems can objectively evaluate fracture risk and monitor healing progression using longitudinal CT scans, indicating strong potential for clinical application [29,30].
In current clinical practice, the use of cancer boards to coordinate multidisciplinary treatment has become increasingly widespread. Our facilities also hold regular meetings, in which one of the objectives is to maintain and improve the activities of daily living in individuals with bone metastasis. Early diagnosis and intervention before the onset of SREs are essential to ensure continuation of systemic or radiation therapy. However, as the number of individuals with bone metastasis increases, it may become difficult for radiologists to consistently evaluate fracture risk on each follow-up CT scan. In this context, automated risk-evaluation systems using CT-based structural rigidity analysis may assist in identifying candidates for early intervention, potentially improving survival outcomes in individuals with bone metastasis. Given that our teams and facilities have developed or integrated clinically applicable artificial intelligence-based technologies for musculoskeletal disorders [26,31,32], incorporating rigidity analysis into routine practice may be feasible.
As perioperative information, one common reason for perioperative radiotherapy is to reduce pain at rest, whereas surgical intervention primarily aims to reduce pain during motion. In period 2, consultation with the palliative care department may have been more frequent, which could have contributed to the tendency toward decreased perioperative radiotherapy during this period.
The individuals included in this study were those who underwent fracture surgery for metastatic long bone. Individuals who did not undergo surgery for pathological fractures were not evaluated. At our facility, fracture surgery for pathological fractures is indicated when the primary cancer team determines that the individual is expected to live at least three to six months. Survival rates would likely have been lower if nonoperative individuals had been included. Surgery was also canceled when individuals had poor general conditions or other clinical issues, such as serious infectious complications [19]. These factors may have contributed to the lower ratio of CF to IF in the demographic data because bedridden individuals have an increased risk of bacterial infections and physical decline. Acute inactivity during hospitalization poses a significant threat to muscle tissue and functional capacity [33,34], and physical inactivity during hospitalization contributes to decreased ability to perform activities of daily living, particularly in older adults [9].
Median survival duration in individuals with bone metastasis has increased [3,4], attributable to advances in both medical and surgical treatments. Among medical options, bisphosphonates and denosumab are commonly used bone-modifying agents. Bisphosphonates reduce bone resorption by impairing osteoclast function through binding to hydroxyapatite crystals within the bone matrix, thereby reducing SREs and improving quality of life [2]. Denosumab, a monoclonal antibody, inhibits osteoclast-mediated bone resorption by binding to RANKL (Receptor Activator of Nuclear Factor Kappa-B Ligand) and preventing its interaction with osteoclast receptors [2]. Although both agents aim to prevent SREs, Scagliotti et al. performed a subgroup analysis from a randomized phase 3 study evaluating individuals with bone metastasis from non-small-cell or small-cell lung cancer who were randomized 1:1 to receive monthly subcutaneous denosumab 120 mg or intravenous zoledronic acid 4 mg. Denosumab was associated with improved median overall survival compared with zoledronic acid in 811 individuals with any lung cancer (8.9 vs. 7.7 months; hazard ratio 0.80, p = 0.01) and in 702 individuals with non-small-cell lung cancer (9.5 vs. 8.0 months; hazard ratio 0.78, p = 0.01) [35]. Beyond survival benefits in lung cancer, denosumab offers advantages such as subcutaneous administration and a lower risk of renal toxicity, making it suitable for individuals with pre-existing renal impairment [2].
Additionally, the adjuvant use of bone-modifying agents to prevent bone metastasis [35] in individuals with early-stage breast cancer has been discussed in US and European guidelines [36,37]. Randomized controlled trials in postmenopausal individuals with early-stage breast cancer have reported that adjuvant bone-modifying agents improved bone-metastasis-free survival or disease-free survival [38,39,40], although the efficacy of adjuvant BMA therapy in this group remains controversial [38,39,40,41]. The original rationale for adjuvant BMA therapy in postmenopausal individuals was to maintain bone density and prevent osteoporosis [39].
Recent biomolecular research has examined how age-related changes in the host environment influence the metastatic development of secondary lesions [42]. In the lung, one of the most frequent secondary sites, age-induced reprogramming of lung fibroblasts promotes tumor cell activation and proliferation [42,43]. As bone is another common secondary site, osteoclast activity is also influenced by age [44]. Osteolytic metastases, such as those from breast cancer, arise through osteoclast stimulation driven by elevated levels of receptor activator of nuclear factor–κB ligand and inflammatory cytokines [45]. Notably, increased osteoclast activity is also observed in osteoporotic bone, where tissue resorption is accelerated [46,47]. Thus, the pathophysiology of osteoporosis inherently creates a favorable premetastatic niche for tumor cells within bone [48,49]. Supporting this, clinical findings indicate that untreated osteoporosis is associated with accelerated progression of bone metastases when they occur [46]. In addition, bone density itself has emerged as an important risk factor for certain cancers [50]. Therefore, further investigation is needed to clarify the relationship between osteoporosis and the development of bone metastasis.
Bone metastasis commonly arises from lung, breast, and prostate cancer. Targeted therapies suppress specific proteins, enzymes, or signaling pathways involved in cancer cell proliferation and metastatic progression [2], making them more selective than conventional cytotoxic chemotherapy and often associated with fewer systemic adverse effects [51]. Various targeted therapies have demonstrated effectiveness in reducing bone metastases and improving survival across multiple cancer types [2]. In non-small-cell lung cancer, therapies aimed at molecular abnormalities have markedly improved survival. Epidermal Growth Factor Receptor (EGFR) inhibitors, such as erlotinib and gefitinib, block the EGFR pathway, which is essential for tumor cell proliferation [52]. In breast cancer, particularly Human Epidermal Growth Factor Receptor 2 (HER2)-positive disease, therapeutic strategies include monoclonal antibodies that inhibit HER2 signaling (trastuzumab) or disrupt HER2 dimerization (pertuzumab), as well as antibody-drug conjugates that deliver cytotoxic agents directly to HER2-overexpressing cells (ado-trastuzumab emtansine) [2,53]. Cyclin-dependent kinase 4 and 6 (CDK4/6) inhibitors, including palbociclib, ribociclib, and abemaciclib, further suppress tumor proliferation by blocking cell-cycle progression [2]. In prostate cancer, abiraterone inhibits androgen biosynthesis, while enzalutamide blocks androgen receptor signaling, both of which contribute to improved systemic control [2,54]. Other malignancies also benefit from targeted therapies such as tyrosine kinase inhibitors, including sunitinib and pazopanib, which inhibit vascular endothelial growth factor signaling, a pathway essential for tumor angiogenesis and metastatic spread [2,51].
However, despite advances in managing and preventing bone metastasis through newer systemic treatments, significant challenges remain, largely due to the considerable biological heterogeneity of bone metastases and the variable therapeutic responses across tumor types [2]. Additionally, the development of resistance to targeted therapies and immunotherapies within the bone microenvironment represents a major obstacle, often leading to treatment failure and disease progression [2]. Such progression increases the risk of SREs and contributes to a decline in activities of daily living. In these cases, early intervention for IFs is essential, and the occurrence of CFs should be avoided to ensure reliable recovery of ambulation and improve survival outcomes in individuals with pathological fractures.
Certain limitations should be considered when interpreting the present findings. First, this was a retrospective study, introducing the possibility of recall and selection bias. Second, the two cohort periods differed in age distribution. Previous studies have reported that age serves as a prognostic factor in individuals with bone metastasis [21,22]. Although multivariate analysis adjusting for age and other variables (sex, body mass index, fracture type, ECOG PS, Katagiri score, and skeletal stabilization) was conducted, residual confounding may still exist. Third, surgical methods were determined by treating surgeons and, therefore, were not randomized. Lastly, this study did not include individuals with impending or completed pathological fractures who did not undergo surgery. Survival outcomes may have differed if nonoperative individuals had been included. Furthermore, surgery was deferred in individuals with poor general condition or complications such as severe infection, which may have contributed to the lower proportion of CFs relative to IFs, as bedridden individuals have increased susceptibility to infection and functional decline.
5. Conclusions
The chronological analysis demonstrated that the later time period was associated with more favorable 1-year overall survival following fracture surgery for metastatic long bone disease. Although individuals in period 2 were older, the proportion of prophylactic surgery for IF did not differ between the two periods. Increasing the ratio of impending to CFs surgeries remains essential, as earlier intervention has the potential to further improve prognosis in individuals with pathological fractures.
Author Contributions
Conceptualization, H.S. and J.I.; Methodology, H.S.; Validation, M.M.; Formal Analysis, H.S.; Investigation, J.I. and K.I.; Data Curation, T.S. (Tamotsu Soma), D.T., T.S. (Tomohiro Shimizu) and H.H.; Writing—Original Draft Preparation, H.S. and J.I.; Writing—Review and Editing, M.M.; Visualization, H.S.; Supervision, N.I. and A.I.; Project Administration, A.I. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of Hokkaido University Hospital (protocol code 022–0036; approval date: 11 May 2022).
Informed Consent Statement
Informed consent was obtained from all individuals involved in the study via the opt-out method.
Data Availability Statement
Data supporting the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| CF | completed pathological fracture |
| CT | computed tomography |
| ECOG PS | Eastern Cooperative Oncology Group Performance Status |
| IF | impending pathological fracture |
References
- Errani, C. Treatment of Bone Metastasis. Curr. Oncol. 2022, 29, 5195–5197. [Google Scholar] [CrossRef]
- Trovarelli, G.; Rizzo, A.; Zinnarello, F.D.; Cerchiaro, M.; Angelini, A.; Pala, E.; Ruggieri, P. Modern Treatment of Skeletal Metastases: Multidisciplinarity and the Concept of Oligometastasis in the Recent Literature. Curr. Oncol. 2025, 32, 226. [Google Scholar] [CrossRef]
- Rogers, D.L.; Raad, M.; Rivera, J.A.; Wedin, R.; Laitinen, M.; Sørensen, M.S.; Petersen, M.M.; Hilton, T.; Morris, C.D.; Levin, A.S.; et al. Life Expectancy After Treatment of Metastatic Bone Disease: An International Trend Analysis. J. Am. Acad. Orthop. Surg. 2024, 32, e293–e301. [Google Scholar] [CrossRef]
- Matsuda, K.; Shimazu, K.; Shinozaki, H.; Fukuda, K.; Yoshida, T.; Taguchi, D.; Nomura, K.; Shibata, H. Recent Trends in Bone Metastasis Treatments: A Historical Comparison Using the New Katagiri Score System. World J. Clin. Cases 2024, 12, 2499–2505. [Google Scholar] [CrossRef]
- Hong, S.; Youk, T.; Lee, S.J.; Kim, K.M.; Vajdic, C.M. Bone Metastasis and Skeletal-Related Events in Patients with Solid Cancer: A Korean Nationwide Health Insurance Database Study. PLoS ONE 2020, 15, e0234927. [Google Scholar] [CrossRef]
- Leu, J.; Narra, L.R.; Gooley, T.; Cross, N.; Vuong, W.; Khan, H.; Kang, J.; Yang, J.T.; Grassberger, C.; Gillespie, E.F. Evaluating Risk Factors for Skeletal-Related Events Among Bone Metastases from Solid Tumors. Radiother. Oncol. 2025, 211, 111048. [Google Scholar] [CrossRef] [PubMed]
- Groot, O.Q.; Lans, A.; Twining, P.K.; Bongers, M.E.R.; Kapoor, N.D.; Verlaan, J.J.; Newman, E.T.; Raskin, K.A.; Lozano-Calderon, S.A.; Janssen, S.J.; et al. Clinical Outcome Differences in the Treatment of Impending Versus Completed Pathological Long-Bone Fractures. J. Bone Jt. Surg. Am. 2022, 104, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Nishimoto, T.; Shimizu, H.; Matsuoka, M.; Takahashi, D.; Shimizu, T.; Iwasaki, N.; Kuwahara, K.; Soma, T.; Hiraga, H.; Iwata, A. Intervention for Impending Pathological Fractures at Proximal Femur Is Associated with Lower Mortality Rates in Patients with Intermediate-to-High Risk According to the Katagiri-New Score. BMC Musculoskelet. Disord. 2024, 25, 836. [Google Scholar] [CrossRef]
- Ito, K.; Shimizu, H.; Matsuoka, M.; Soma, T.; Takahashi, D.; Shimizu, T.; Nishimoto, T.; Hiraga, H.; Iwasaki, N.; Iwata, A. Recovery of Ambulation in Older Adults Undergoing Fracture Surgery for Femoral Metastasis: A Multicenter Retrospective Cohort Study. Sci. Rep. 2025, 15, 16578. [Google Scholar] [CrossRef]
- Kim, Y.; Krishnan, C.K.; Kim, H.S.; Cho, H.S.; Han, I. Ambulation Recovery After Surgery for Metastases to the Femur. Oncologist 2020, 25, e178–e185. [Google Scholar] [CrossRef] [PubMed]
- Mirels, H. Metastatic Disease in Long Bones: A Proposed Scoring System for Diagnosing Impending Pathologic Fractures. Clin. Orthop. Relat. Res. 1989, 256, 256–264. [Google Scholar] [CrossRef]
- Tat, J.; Bodansky, D.; Sheth, U.; Ung, Y.; Whyne, C.; Nam, D. Predicting Pathological Fractures at Metastatic Humeral Lesions. JBJS Open Access 2023, 8, e23.00012. [Google Scholar] [CrossRef]
- Hoban, K.A.; Downie, S.; Adamson, D.J.A.; MacLean, J.G.; Cool, P.; Jariwala, A.C. Mirels’ Score for Upper Limb Metastatic Lesions: Do We Need a Different Cutoff for Recommending Prophylactic Fixation? JSES Int. 2022, 6, 675–681. [Google Scholar] [CrossRef]
- Hershkovich, O.; Sakhnini, M.; Barkay, G.; Liberman, B.; Friedlander, A.; Lotan, R. Proximal Femoral Multiple Myeloma Pathological Fractures, Impending and Actual Fractures: A Patient Survival Study. BMC Cancer 2024, 24, 917. [Google Scholar] [CrossRef]
- Amendola, R.L.; Miller, M.A.; Kaupp, S.M.; Cleary, R.J.; Damron, T.A.; Mann, K.A. Modification to Mirels Scoring System Location Component Improves Fracture Prediction for Metastatic Disease of the Proximal Femur. BMC Musculoskelet. Disord. 2023, 24, 65. [Google Scholar] [CrossRef] [PubMed]
- Katagiri, H.; Okada, R.; Takagi, T.; Takahashi, M.; Murata, H.; Harada, H.; Nishimura, T.; Asakura, H.; Ogawa, H. New Prognostic Factors and Scoring System for Patients with Skeletal Metastasis. Cancer Med. 2014, 3, 1359–1367. [Google Scholar] [CrossRef]
- Doyle, C.; Kushi, L.H.; Byers, T.; Courneya, K.S.; Demark-Wahnefried, W.; Grant, B.; McTiernan, A.; Rock, C.L.; Thompson, C.; Gansler, T.; et al. Nutrition and Physical Activity During and After Cancer Treatment: An American Cancer Society Guide for Informed Choices. CA Cancer J. Clin. 2006, 56, 323–353. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, K.H.; Courneya, K.S.; Matthews, C.; Demark-Wahnefried, W.; Galvão, D.A.; Pinto, B.M.; Irwin, M.L.; Wolin, K.Y.; Segal, R.J.; Lucia, A.; et al. American College of Sports Medicine Roundtable on Exercise Guidelines for Cancer Survivors. Med. Sci. Sports Exerc. 2010, 42, 1409–1426. [Google Scholar] [CrossRef]
- Yoshikawa, R.; Fujii, Y.; Kashima, R.; Saho, W.; Harada, R.; Makiura, D.; Fujiwara, K.; Inoue, J.; Takeoka, Y.; Sawada, R.; et al. Effectiveness of Rehabilitation Treatment for Bone Metastasis Patients Without Surgical Intervention: A Propensity Score Matching Analysis. J. Bone Oncol. 2025, 53, 100703. [Google Scholar] [CrossRef]
- Fujii, Y.; Yoshikawa, R.; Kashima, R.; Saho, W.; Onishi, H.; Matsumoto, T.; Harada, R.; Takeoka, Y.; Sawada, R.; Fukase, N.; et al. Evaluation of Changes in Activities of Daily Living and Quality of Life in Patients with Bone Metastasis Who Underwent Conservative Therapy Through Bone Metastasis Cancer Boards. Medicina 2024, 60, 906. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Yoshikawa, R.; Harada, R.; Date, A.; Kobayashi, Y.; Kozawa, S.; Sakai, Y. Clinical Outcome of Patients with Bone Metastases in a Convalescent Rehabilitation Ward: A Case Series of Six Patients. Prog. Rehabil. Med. 2022, 7, 20220022. [Google Scholar] [CrossRef] [PubMed]
- Stout, N.L.; Santa Mina, D.; Lyons, K.D.; Robb, K.; Silver, J.K. A Systematic Review of Rehabilitation and Exercise Recommendations in Oncology Guidelines. CA Cancer J. Clin. 2021, 71, 149–175. [Google Scholar] [CrossRef] [PubMed]
- Damron, T.A.; Morgan, H.; Prakash, D.; Grant, W.; Aronowitz, J.; Heiner, J. Critical Evaluation of Mirels’ Rating System for Impending Pathologic Fractures. Clin. Orthop. Relat. Res. 2003, 415, S201–S207. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; He, Y.; Wan, L.; Li, C.; Li, Z.; Li, Z.; Xu, H.; Tu, C. Deep Learning Models in Classifying Primary Bone Tumors and Bone Infections Based on Radiographs. NPJ Precis. Oncol. 2025, 9, 72. [Google Scholar] [CrossRef]
- Beyraghi, S.; Ghorbani, F.; Shabanpour, J.; Lajevardi, M.E.; Nayyeri, V.; Chen, P.Y.; Ramahi, O.M. Microwave Bone Fracture Diagnosis Using Deep Neural Network. Sci. Rep. 2023, 13, 16957. [Google Scholar] [CrossRef]
- Shimizu, H.; Enda, K.; Koyano, H.; Shimizu, T.; Shimodan, S.; Sato, K.; Ogawa, T.; Tanaka, S.; Iwasaki, N.; Takahashi, D. Bimodal Machine Learning Model for Unstable Hips in Infants: Integration of Radiographic Images with Automatically Generated Clinical Measurements. Sci. Rep. 2024, 14, 17826. [Google Scholar] [CrossRef]
- Jawad, M.U.; Scully, S.P. In Brief: Classifications in Brief: Mirels’ Classification: Metastatic Disease in Long Bones and Impending Pathologic Fracture. Clin. Orthop. Relat. Res. 2010, 468, 2825–2827. [Google Scholar] [CrossRef]
- Damron, T.A.; Nazarian, A.; Entezari, V.; Brown, C.; Grant, W.; Calderon, N.; Zurakowski, D.; Terek, R.M.; Anderson, M.E.; Cheng, E.Y.; et al. CT-Based Structural Rigidity Analysis Is More Accurate Than Mirels Scoring for Fracture Prediction in Metastatic Femoral Lesions. Clin. Orthop. Relat. Res. 2016, 474, 643–651. [Google Scholar] [CrossRef]
- Hetreau, C.; Mischler, D.; Schlatter, J.; Valenti, A.; Ernst, M.; Varga, P.; Schwarzenberg, P. Longitudinal CT-Based Finite Element Analyses Provide Objective Fracture Healing Measures in an Ovine Tibia Model. J. Orthop. Res. 2024, 42, 1762–1770. [Google Scholar] [CrossRef]
- Bohanske, M.S.; Momenzadeh, K.; van der Zwaal, P.; Hoogwater, F.J.H.; Cory, E.; Biggane, P.; Snyder, B.D.; Nazarian, A. CT-Based Structural Rigidity Analysis Can Assess Tumor- and Treatment-Induced Changes in Rat Bones with Metastatic Lesions. BMC Cancer 2024, 24, 767. [Google Scholar] [CrossRef]
- Shimizu, H.; Enda, K.; Shimizu, T.; Ishida, Y.; Ishizu, H.; Ise, K.; Tanaka, S.; Iwasaki, N. Machine Learning Algorithms: Prediction and Feature Selection for Clinical Refracture After Surgically Treated Fragility Fracture. J. Clin. Med. 2022, 11, 2021. [Google Scholar] [CrossRef]
- Shimizu, H.; Enda, K.; Koyano, H.; Ogawa, T.; Takahashi, D.; Tanaka, S.; Iwasaki, N.; Shimizu, T. Diagnosis on Ultrasound Images for Developmental Dysplasia of the Hip with a Deep Learning-Based Model Focusing on Signal Heterogeneity in the Bone Region. Diagnostics 2025, 15, 403. [Google Scholar] [CrossRef]
- English, K.L.; Paddon-Jones, D. Protecting Muscle Mass and Function in Older Adults During Bed Rest. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Dall, C.H.; Andersen, H.; Povlsen, T.M.; Henriksen, M. Evaluation of a Technology-Assisted Physical Activity Intervention Among Hospitalised Patients: A Randomised Study. Eur. J. Intern. Med. 2019, 69, 50–56. [Google Scholar] [CrossRef]
- Scagliotti, G.V.; Hirsh, V.; Siena, S.; Henry, D.H.; Woll, P.J.; Manegold, C.; Solal-Celigny, P.; Rodriguez, G.; Krzakowski, M.; Mehta, N.D.; et al. Overall Survival Improvement in Patients with Lung Cancer and Bone Metastases Treated with Denosumab Versus Zoledronic Acid: Subgroup Analysis from a Randomized Phase 3 Study. J. Thorac. Oncol. 2012, 7, 1823–1829. [Google Scholar] [CrossRef] [PubMed]
- Eisen, A.; Somerfield, M.R.; Accordino, M.K.; Blanchette, P.S.; Clemons, M.J.; Dhesy-Thind, S.; Dillmon, M.S.; D’Oronzo, S.; Fletcher, G.G.; Frank, E.S.; et al. Use of Adjuvant Bisphosphonates and Other Bone-Modifying Agents in Breast Cancer: ASCO-OH (CCO) Guideline Update. J. Clin. Oncol. 2022, 40, 787–800. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, F.; Kyriakides, S.; Ohno, S.; Penault-Llorca, F.; Poortmans, P.; Rubio, I.T.; Zackrisson, S.; Senkus, E. Early Breast Cancer: ESMO Clinical Practice Guidelines for Diagnosis, Treatment and Follow-Up. Ann. Oncol. 2019, 30, 1194–1220. [Google Scholar] [CrossRef]
- Gnant, M.; Pfeiler, G.; Steger, G.G.; Egle, D.; Greil, R.; Fitzal, F.; Wette, V.; Balic, M.; Haslbauer, F.; Melbinger-Zeinitzer, E.; et al. Adjuvant Denosumab in Postmenopausal Patients with Hormone Receptor-Positive Breast Cancer (ABCSG-18): Disease-Free Survival Results from a Randomised, Double-Blind, Placebo-Controlled, Phase 3 Trial. Lancet Oncol. 2019, 20, 339–351. [Google Scholar] [CrossRef]
- Gnant, M.; Frantal, S.; Pfeiler, G.; Steger, G.G.; Egle, D.; Greil, R.; Fitzal, F.; Wette, V.; Balic, M.; Haslbauer, F.; et al. Long-Term Outcomes of Adjuvant Denosumab in Breast Cancer. NEJM Evid. 2022, 1, EVIDoa2200162. [Google Scholar] [CrossRef]
- Early Breast Cancer Trialists’ Collaborative Group. Adjuvant Bisphosphonate Treatment in Early Breast Cancer: Meta-Analyses of Individual Patient Data from Randomised Trials. Lancet 2015, 386, 1353–1361. [CrossRef]
- Coleman, R.; Finkelstein, D.M.; Barrios, C.; Martin, M.; Iwata, H.; Hegg, R.; Glaspy, J.; Periañez, A.M.; Tonkin, K.; Deleu, I.; et al. Adjuvant Denosumab in Early Breast Cancer (D-CARE): An International, Multicentre, Randomised, Controlled, Phase 3 Trial. Lancet Oncol. 2020, 21, 60–72. [Google Scholar] [CrossRef]
- Turrell, F.K.; Orha, R.; Guppy, N.J.; Gillespie, A.; Guelbert, M.; Starling, C.; Haider, S.; Isacke, C.M. Age-Associated Microenvironmental Changes Highlight the Role of PDGF-C in ER+ Breast Cancer Metastatic Relapse. Nat. Cancer 2023, 4, 468–484. [Google Scholar] [CrossRef] [PubMed]
- Fane, M.E.; Chhabra, Y.; Alicea, G.M.; Maranto, D.A.; Douglass, S.M.; Webster, M.R.; Rebecca, V.W.; Marino, G.E.; Almeida, F.; Ecker, B.L.; et al. Stromal Changes in the Aged Lung Induce an Emergence from Melanoma Dormancy. Nature 2022, 606, 396–405. [Google Scholar] [CrossRef]
- Møller, A.M.J.; Delaissé, J.M.; Olesen, J.B.; Madsen, J.S.; Canto, L.M.; Bechmann, T.; Rogatto, S.R.; Søe, K. Aging and Menopause Reprogram Osteoclast Precursors for Aggressive Bone Resorption. Bone Res. 2020, 8, 27. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.X.; Bos, P.D.; Massagué, J. Metastasis: From Dissemination to Organ-Specific Colonization. Nat. Rev. Cancer 2009, 9, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.M.; Chen, F.P.; Yang, K.C.; Yuan, S.S. Association of Bone Metastasis with Early-Stage Breast Cancer in Women with and Without Pre-Cancer Osteoporosis According to Osteoporosis Therapy Status. JAMA Netw. Open 2019, 2, e190429. [Google Scholar] [CrossRef]
- Takegahara, N.; Kim, H.; Choi, Y. Unraveling the Intricacies of Osteoclast Differentiation and Maturation: Insight into Novel Therapeutic Strategies for Bone-Destructive Diseases. Exp. Mol. Med. 2024, 56, 264–272. [Google Scholar] [CrossRef]
- Talmadge, J.E.; Fidler, I.J. AACR Centennial Series: The Biology of Cancer Metastasis—Historical Perspective. Cancer Res. 2010, 70, 5649–5669. [Google Scholar] [CrossRef]
- Weilbaecher, K.N.; Guise, T.A.; McCauley, L.K. Cancer to Bone: A Fatal Attraction. Nat. Rev. Cancer 2011, 11, 411–425. [Google Scholar] [CrossRef]
- Dallal, C.M.; Brinton, L.A.; Bauer, D.D.; Buist, D.S.; Cauley, J.A.; Hue, T.F.; Lacroix, A.; Tice, J.A.; Chia, V.M.; Falk, R.; et al. Obesity-Related Hormones and Endometrial Cancer Among Postmenopausal Women: A Nested Case-Control Study Within the B~FIT Cohort. Endocr. Relat. Cancer 2013, 20, 151–160. [Google Scholar] [CrossRef]
- Falvello, V.; Van Poznak, C. Updates in Management of Bone Metastatic Disease in Primary Solid Tumors with Systemic Therapies. Curr. Osteoporos. Rep. 2021, 19, 452–461. [Google Scholar] [CrossRef]
- Mok, T.S.; Wu, Y.L.; Thongprasert, S.; Yang, C.H.; Chu, D.T.; Saijo, N.; Sunpaweravong, P.; Han, B.; Margono, B.; Ichinose, Y.; et al. Gefitinib or Carboplatin-Paclitaxel in Pulmonary Adenocarcinoma. N. Engl. J. Med. 2009, 361, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Miles, D.; Gianni, L.; Krop, I.E.; Welslau, M.; Baselga, J.; Pegram, M.; Oh, D.Y.; Diéras, V.; Guardino, E. Trastuzumab Emtansine for HER2-Positive Advanced Breast Cancer. N. Engl. J. Med. 2012, 367, 1783–1791. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.J.; Heller, G.; Hillman, D.W.; Bobek, O.; Ryan, C.; Antonarakis, E.S.; Bryce, A.H.; Hahn, O.; Beltran, H.; Armstrong, A.J. Randomized Phase III Study of Enzalutamide Compared with Enzalutamide Plus Abiraterone for Metastatic Castration-Resistant Prostate Cancer (Alliance A031201 Trial). J. Clin. Oncol. 2023, 41, 3352–3362. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.