Abstract
To better understand recent adolescent (10–19 years) HIV trends in Central America, we analyzed routine data from countries supported by the United States President’s Emergency Plan for AIDS Relief (PEPFAR): Guatemala, El Salvador, Honduras, Panama, and Nicaragua, over the period from October 2020 to September 2024. Key PEPFAR indicators included HIV testing, HIV positivity rates, new treatment initiations, advanced HIV disease (AHD) at diagnosis, viral load coverage (VLC), viral load suppression (VLS), and multi-month dispensing (MMD) uptake for children and adolescents living with HIV (CALHIV) from 10–19 years of age. Since October 2020, the number of HIV tests conducted among adolescents has increased; however, the positivity rate has remained stable at approximately 2%. The number of adolescents initiating treatment increased by 21%. At the same time, VLS has shown steady regional improvement (from 73% to 90%), though VLC is a persistent challenge (80%). Treatment interruption rates have been relatively stable, fluctuating between 2% and 3%. Advanced HIV is high in adolescents new to treatment (34%), especially among females (40%), though cluster of differentiation 4 (CD4) testing at diagnosis has only been collected recently and coverage is not complete. The high prevalence of AHD among adolescents underscores the need to reinforce earlier and more targeted interventions for adolescents, especially in countries with greater HIV prevalence such as Panama and Guatemala.
1. Introduction
Despite progress in reducing the global burden of HIV, adolescents living with HIV (ALHIV) remain an overlooked population, with persistent gaps across the HIV care continuum. They experience higher rates of delayed diagnosisand poorer outcomes in retention and viral suppression compared to adults [1].
International guidelines emphasize the need for adolescent-responsive services to address transitions into adulthood, HIV status disclosure, and differentiated services appropriate for adolescents [1,2] Additionally, many programs do not consider challenges beyond HIV, such as the prevention of pregnancies, the prevention of sexually transmitted infections, substance abuse, violence, and the need to establish life goals [3,4,5].
According to 2024 global data from the Joint United Nations Programme on HIV/AIDS (UNAIDS), the HIV epidemic in Central America is heterogeneous. Guatemala has the largest estimated number of people living with HIV (PLHIV), with 33,000 individuals and a prevalence of 0.2%. Panama, while having a slightly smaller number of PLHIV (31,000), demonstrates the highest prevalence at 1.1%. El Salvador follows with 21,000 PLHIV and a prevalence of 0.4%, followed by Honduras with 19,000 PLHIV and a prevalence of 0.2%. These patterns suggest a concentrated epidemic in specific networks and geographies [6,7,8,9,10] During this same period, adolescents (10–19 years) accounted for 4% of PLHIV globally (1.6 million), of which about 4000 were in Central America. Recent reports from UNAIDS and HIV researchers have described progress with regards to the HIV response in this region; however, gaps remain in testing and treatment coverage [11,12,13,14].
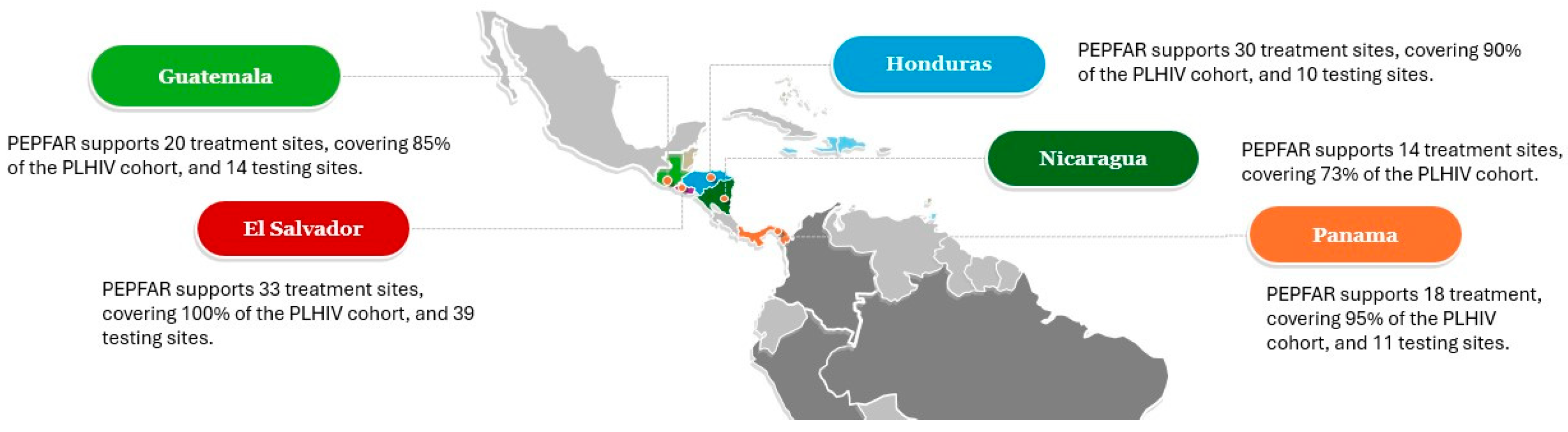
The U.S. President’s Emergency Plan for AIDS Relief (PEPFAR) supports five countries in Central America: Guatemala, El Salvador, Honduras, Panama, and Nicaragua, reaching 70,983 people living with HIV in 2024 (89% coverage of all PLHIV in this region), of whom approximately 2% were adolescents aged 10–19 years (Figure 1). Most care and treatment sites in Guatemala, El Salvador, and Panama receive some support through PEPFAR programming, whereas in Honduras and Nicaragua, most of these sites are supported by the Ministries of Health. In contrast, PEPFAR does not offer support to the majority of national HIV testing sites across the five countries. PEPFAR plays a crucial role in addressing the HIV/AIDS epidemic, collaborating with Ministries of Health and implementing a comprehensive approach through partners. This approach includes HIV prevention, testing, treatment, the capacity building of HIV services, and the monitoring of key indicators.
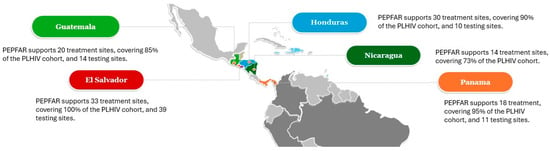
Figure 1.
Number of PEPFAR-supported sites across Central American in 2024. Created by the authors using data from DATIM (Data for Accountability, Transparency, and Impact).
Our analysis evaluates recent trends in HIV testing, treatment initiation, and viral load suppression among adolescents aged 10–19 years in Central America from 2020 to 2024.
2. Materials and Methods
We conducted a retrospective, descriptive analysis of secondary data using routinely collected PEPFAR program data [15] from Guatemala, El Salvador, Honduras, Nicaragua, and Panama, covering the period from October 2020 to September 2024. This analysis included adolescents aged 10–19 years and used PEPFAR fiscal years, which run from October 1 to September 30 of the following year. Accordingly, the fiscal years were categorized as follows: October 2020–September 2021 (Year 1), October 2021–September 2022 (Year 2), October 2022–September 2023 (Year 3), and October 2023–September 2024 (Year 4).
PEPFAR program data were extracted from the DATIM (Data for Accountability, Transparency, and Impact) database [16], the official PEPFAR reporting system for the MER (Monitoring, Evaluation, and Reporting) indicators. DATIM contains de-identified, aggregate MER data reported by implementing partners from the sites supported, in whole or in part, by PEPFAR.
We conducted a descriptive year-on-year analysis of aggregate indicators and did not perform individual-level analyses, as DATIM does not include patient-level identifiers or longitudinal follow-up time. To compare trends across the years, between genders, across age groups, advanced stage diagnosis, and countries, χ2 and two-sample proportion tests were employed, with a cutoff of α = 0.05 for significance. Missing data was omitted from the analyses; given the aggregate nature of the data source, imputation was not possible. PEPFAR supports data collection, cleaning, and validation across supported countries; aggregate results are reported quarterly or semi-annually, according to PEPFAR MER guidelines.
Indicators Used for Analysis
Key PEPFAR MER indicators included HIV testing (HTS_TST), HIV positive tests (HTS_TST_POS), new treatment initiations (TX_NEW), viral load coverage (VLC), viral load suppression (VLS), and multi-month dispensing (MMD) uptake [17] The definitions and additional indicators may be seen in Table 1. Indicators were selected in alignment with the conceptual framework of the HIV care cascade [18] and the UNAIDS 95-95-95 targets [19].

Table 1.
Definitions of Core PEPFAR Monitoring, Evaluation, and Reporting (MER) indicators for HIV for the reporting periods.
These indicators were reported for the entire cohort supported by PEPFAR funding in each country. For the purpose of this analysis, we limited results to indicators for which age disaggregation for ages 10–19 years was available. This analysis was reviewed by the U.S. Centers for Disease Control and Prevention and determined not to constitute human subject research.
To describe treatment continuity, we used interruption in treatment and MMD (months of ART dispensed) as programmatic proxies, recognizing that these do not constitute formal cohort retention estimates.
For this analysis we derived Advanced HIV Disease (AHD) among new ART initiators using the cluster of differentiation 4 (CD4) results reported in DATIM. Adolescents were classified as having AHD if their most recent CD4 result at or within three months of ART initiation was <200 cells/mm3 or if they were recorded as WHO stage 3 or 4 at initiation, consistent with WHO/UNAIDS definitions.
3. Results
3.1. HIV Testing
Since Year 1, the total number of HIV tests supported among adolescents increased across most of Central America, from 11,415 to 17,237 (Table 2). Regionally, 90% of adolescent HIV tests were performed in adolescents aged 15–19. El Salvador and Guatemala have consistently increased the number of tests conducted in adolescents from 2020 to 2024, whereas Honduras, Nicaragua, and Panama showed fluctuating patterns in testing volumes. An increase and subsequent decrease in testing were reported in Nicaragua each year of the analysis among those aged 15 to 19 years.

Table 2.
HIV testing outcomes in Central America from October 2020 to September 2024.
During Year 4, adolescents represented approximately 3.1% of all tested individuals in the region. El Salvador recorded the highest number of adolescent tests, totaling 5251, with all new adolescent diagnoses (32) in El Salvador occurring among those aged 15–19. Additional testing data can be found in Supplemental Table S1.
3.1.1. Testing Modalities
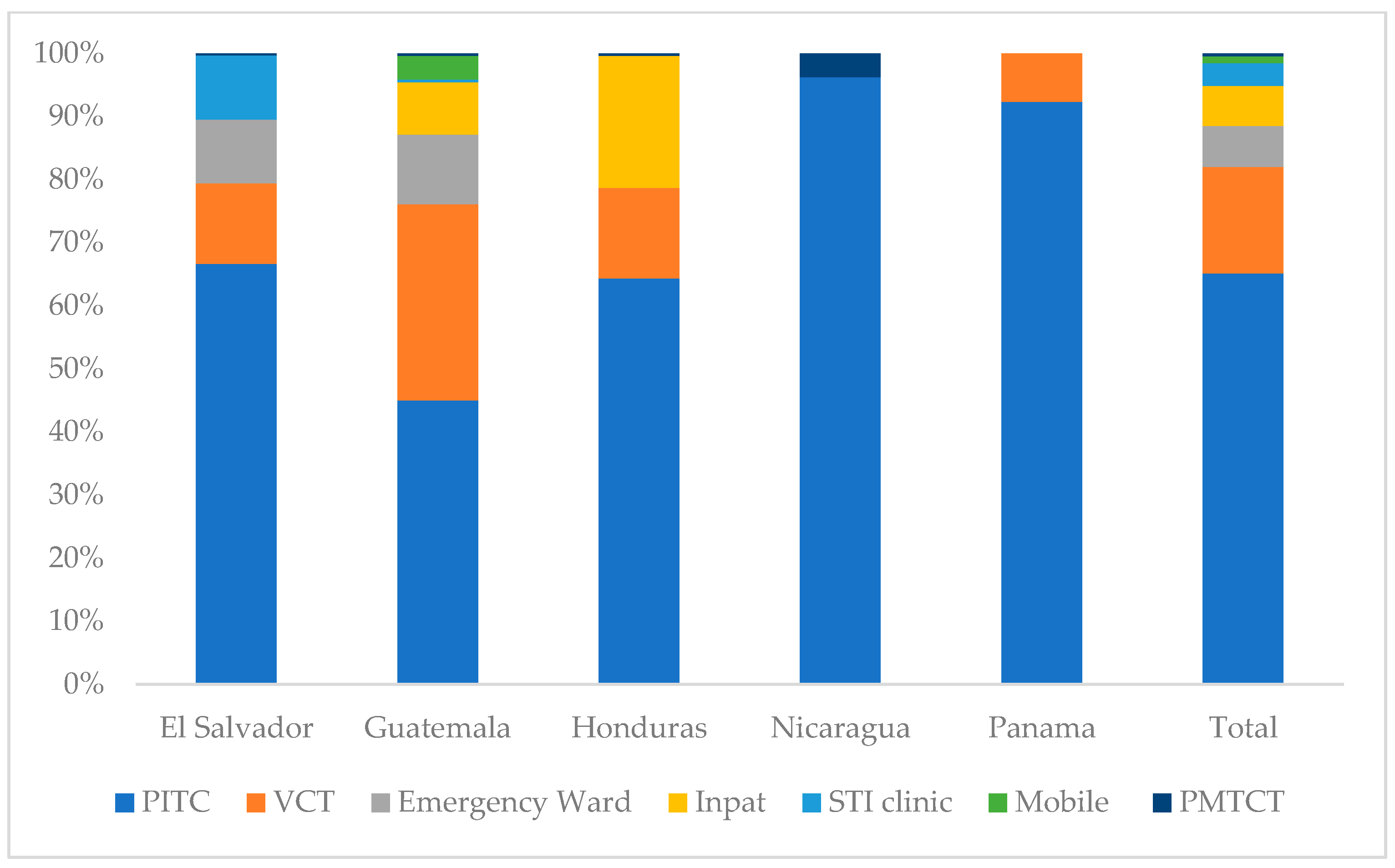
Across all countries, most HIV testing was conducted through facility-based, provider-initiated approaches, with varying contributions from voluntary and community-based modalities (Figure 2).
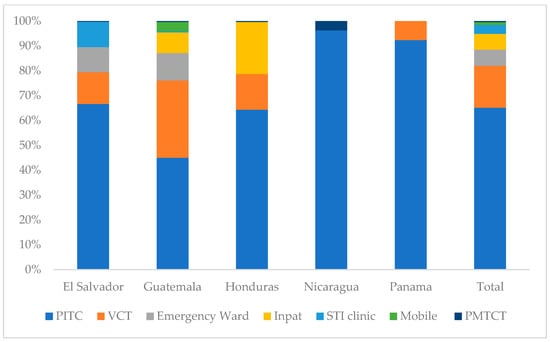
Figure 2.
Test modality distribution by country, October 2023 to September 2024. Testing modalities: Emergency Ward = HIV testing in hospital emergency services; Inpat = HIV testing in inpatient hospital wards; PITC = provider-initiated testing and counseling during routine clinical visits; Mobile = community/mobile outreach testing services; PMTCT = testing in prevention of mother-to-child transmission services; STI clinic = testing in sexually transmitted infection clinics; VCT = voluntary counseling and testing services.
- In El Salvador 45% of tests were conducted through routine provider-initiated testing in health facilities (Other PITC), 22% through voluntary counseling and testing (VCT), and 11% in sexually transmitted infection (STI) clinics.
- In Guatemala, around 34% of HIV tests were conducted through routine health services in clinics and hospitals, 31% through voluntary testing services, and 11% through mobile outreach services.
- In Honduras, 54% of all HIV tests were conducted through routine health services, 24% through voluntary testing services, and 13% among inpatient clients.
- In Nicaragua 49% of all HIV tests were conducted in routine health services, 29% through voluntary testing services, and 10% through community-based outreach.
- Panama’s large majority of HIV tests were conducted through routine health services (71%), whereas only 11% were provided through voluntary testing services.
3.1.2. HIV Positive Testing
Between Year 1 and Year 4, a total of 56,662 adolescents were tested: 1243 tested positive, yielding a regional positivity of 2.0%. Ninety-six percent of diagnoses occurred among adolescents between 15 and 19 years of age. Guatemala (3.1%) and Panama (4%) were the only countries reporting positivity rates above the regional average and required the fewest tests to identify one positive case, indicating higher yields compared with neighboring countries. By sex, adolescent males had a positivity proportion twice that of females (Table 2). When age was also considered, positivity was higher in both male age groups, 10–14 and 15–19 (1.6% and 2.9%, respectively), compared with females in same age groups (0.2% and 1.0%, respectively); however, these differences were not statistically significant (Supplemental Table S2). The highest positivity among males aged 15–19 was observed in Panama (4.9%).
3.1.3. Country-Level Trends in Year 4
Country-level analysis revealed distinct patterns.
- El Salvador consistently reported positivity below 1% throughout the period (Table 2) despite having the highest number of tests performed (5251), yielding 32 new diagnoses (0.6% positivity), all among those aged 15–19 year. Within the country, the highest percentages of positivity were, in descending order, Chalatenango, La Union, and La Paz (3.3%, 2.7%, and 1.6%, respectively).
- Guatemala had the second largest testing volume (4699), with its highest testing activity in the Guatemala department, whereas positivity was highest in Retalhuleu, Sacatepéquez, and Alta Verapaz (7.4%, 7.0%, and 5.6%, respectively). Chiquimula, which borders with Retalhuleu, had only 15 tests performed and 0% positivity, as did San Marcos.
- Honduras reported its highest proportion of tests conducted in Francisco Morazan (28%) and in Cortes (24%), with positivity being 1.3% and 1.7%, respectively; the highest positivity in Honduras was found in Colon (9.0%).
- Nicaragua observed the greatest variability over time, with positivity fluctuating from 7.0% in Year 1 to 1.4% in Year 4. The largest number of tests were performed in the capital, Managua, but positivity was only 1.7%. Atlantico Norte, with only 144 tests performed, had a positivity of 7.6%.
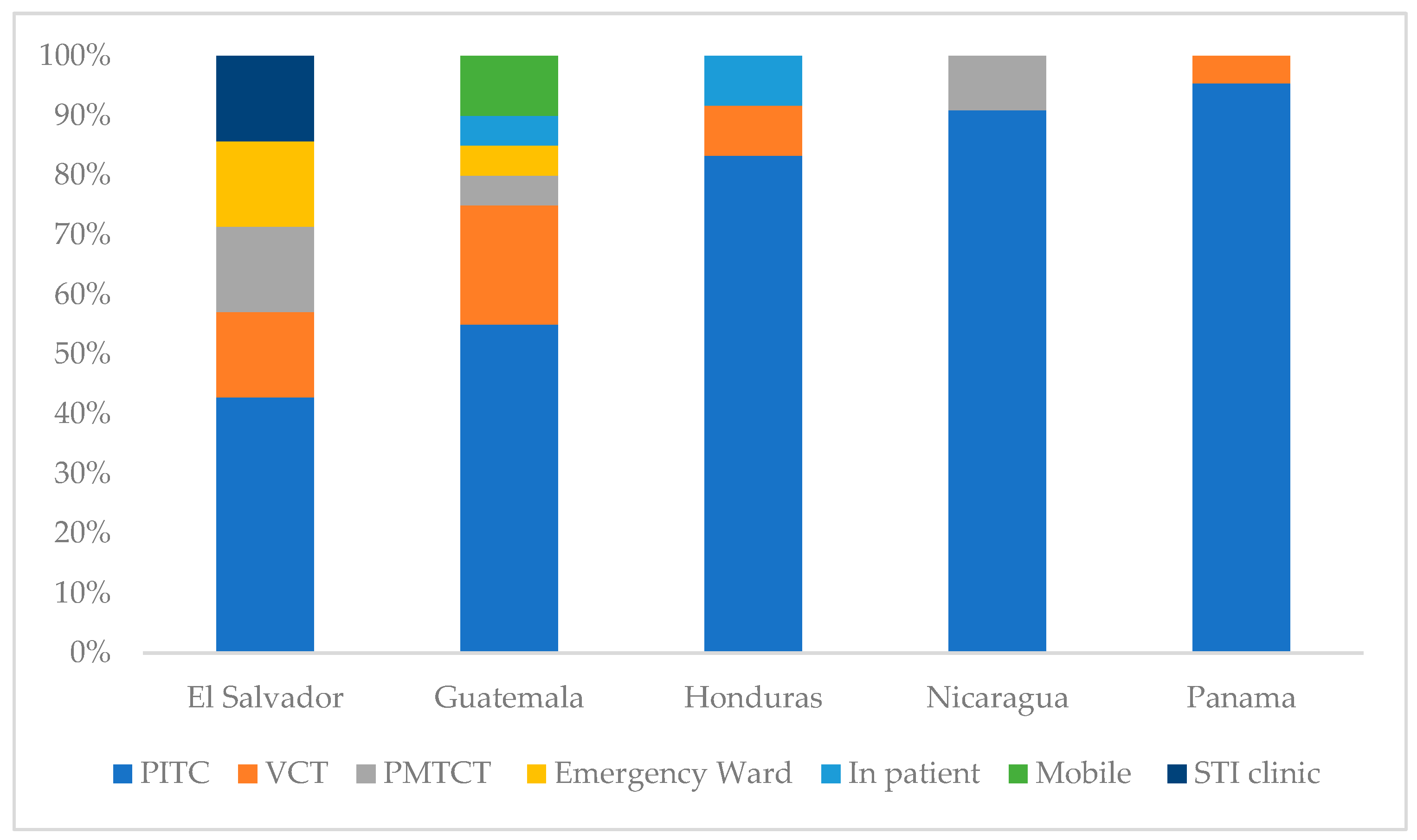
- Across all five countries, provider-initiated testing and counseling (PITC) accounted for the largest proportion of HIV-positive diagnoses, representing the predominant testing modality among adolescents with newly identified HIV infection (Figure 3).
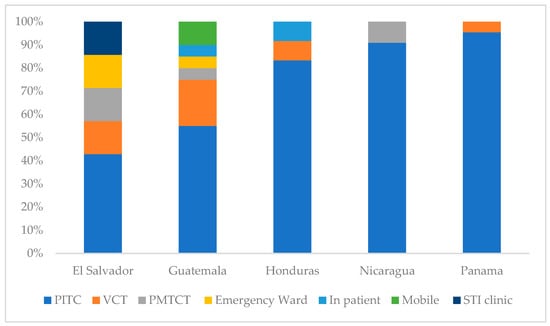 Figure 3. HIV positive test modality distribution by country from October 2023 to September 2024. Testing modalities: Emergency Ward = HIV testing in hospital emergency services; Inpat = HIV testing in inpatient hospital wards; PITC = provider-initiated testing and counseling during routine clinical visits; Mobile = community/mobile outreach testing services; PMTCT = prevention of mother-to-child transmission services; STI clinic = sexually transmitted infection; VCT = voluntary counseling and testing services.
Figure 3. HIV positive test modality distribution by country from October 2023 to September 2024. Testing modalities: Emergency Ward = HIV testing in hospital emergency services; Inpat = HIV testing in inpatient hospital wards; PITC = provider-initiated testing and counseling during routine clinical visits; Mobile = community/mobile outreach testing services; PMTCT = prevention of mother-to-child transmission services; STI clinic = sexually transmitted infection; VCT = voluntary counseling and testing services. - Panama conducted 2707 tests, with the highest number in its capital (1464 tests, 1.5% positivity). The highest positivity was recorded in Chiriqui (14.8%), followed by Bocas del Toro (9.7%) and Veraguas (5.3%); the Comarca Embera had no positive results (from 11 tests).
3.2. Treatment
3.2.1. Adolescent Representation Within HIV Treatment Cohorts
Throughout the analysis period, an average of 1370 adolescents (10–19) were reported at year-end as being in PEPFAR-supported active HIV treatment cohorts in El Salvador, Guatemala, Honduras, Nicaragua, and Panama. This number reflects the average annual cohort size and does not represent unique individuals across the years. Regionally, these adolescents represent approximately 2.0% of the cohort in supported sites (Table 3, Supplemental Table S2).

Table 3.
Distribution of adolescents on treatment in comparison with the general treatment cohort in PEPFAR supported sites from October 2023 to September 2024.
Males represented 58% of all adolescents in the regional cohort. The highest proportions of male adolescents were observed in Guatemala (61%) and Panama (70%), whereas Honduras was the only country where females represented the majority (59%) of adolescents on treatment. The proportion of adolescents in the Honduras treatment cohorts have declined from 4.3% to 2.3% (1.9% decrease, p < 0.001).
In Year 4 (Table 3), adolescents represented 1.9% of the cohort in the Central American region, ranging from 1.2% in El Salvador to 2.7% in Nicaragua. Guatemala had the highest number of adolescents in FY24, representing 32% of all adolescents in the region.
3.2.2. New to Treatment
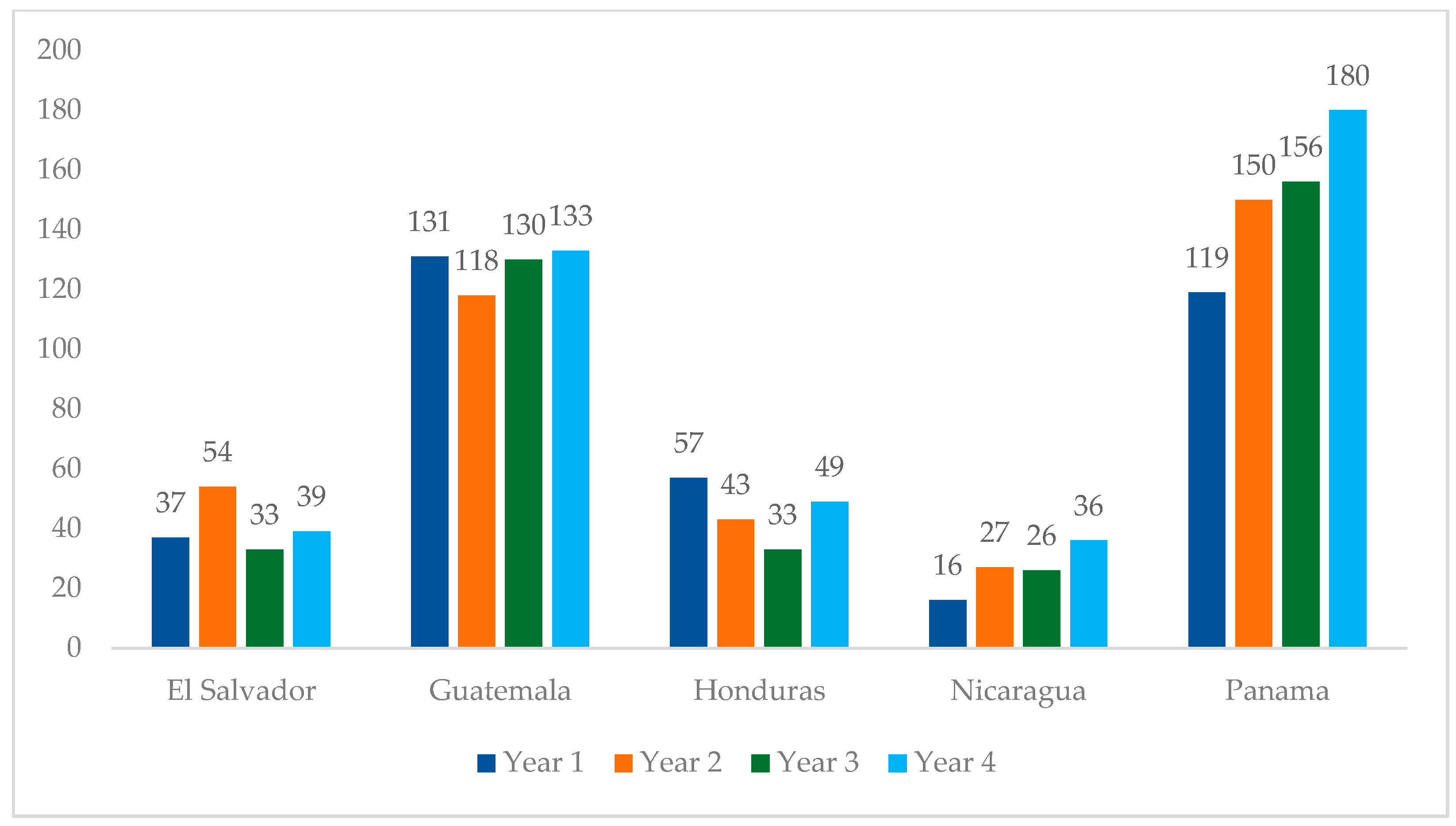
From Year 1 to Year 4 of this analysis, the number of adolescents initiating treatment (see Table 1 for indicator definitions) increased from 360 to 437, representing a 21% increase (Figure 4). This increase was mainly driven by Panama, which observed a 51% increase in treatment initiation during this period. Honduras was the only country in the region that reported fewer treatment initiations in Year 4 compared to Year 1, with a 14% decrease in treatment initiations. Nicaragua reported a 125% increase in this period. El Salvador and Guatemala observed marginal changes in treatment initiation from Year 1 to 4.
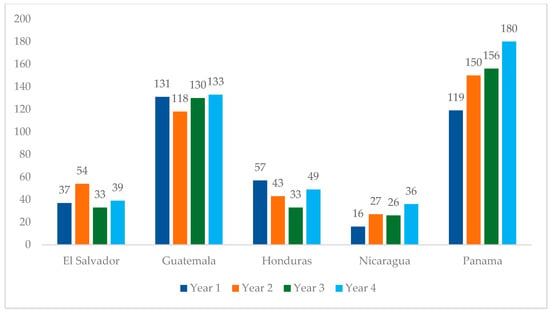
Figure 4.
Regional overview of adolescent treatment initiations at PEPFAR-supported sites in Central America, October 2020–September 2024.
3.2.3. Continuity of Treatment
Treatment interruption rates have remained relatively stable, fluctuating between 2% and 3% on average, and are generally lower and more stable than those observed in adults. Adult interruption peaked at 5.5% in Year 2, then gradually declined to 2.6% by the final quarter of Year 4. Nicaragua and Panama reported the highest average treatment interruptions, at 7.3% and 4.8%, respectively. The multi-month dispensing (MMD) of antiretroviral therapy (ART) increased, especially for 3–5-month refills, whereas 6-month dispensing remained low. Guatemala and Panama led in 3–5-month MMD periods, with each reporting over 50% MMD for adolescents in Year 4.
3.3. Viral Load Coverage and Suppression
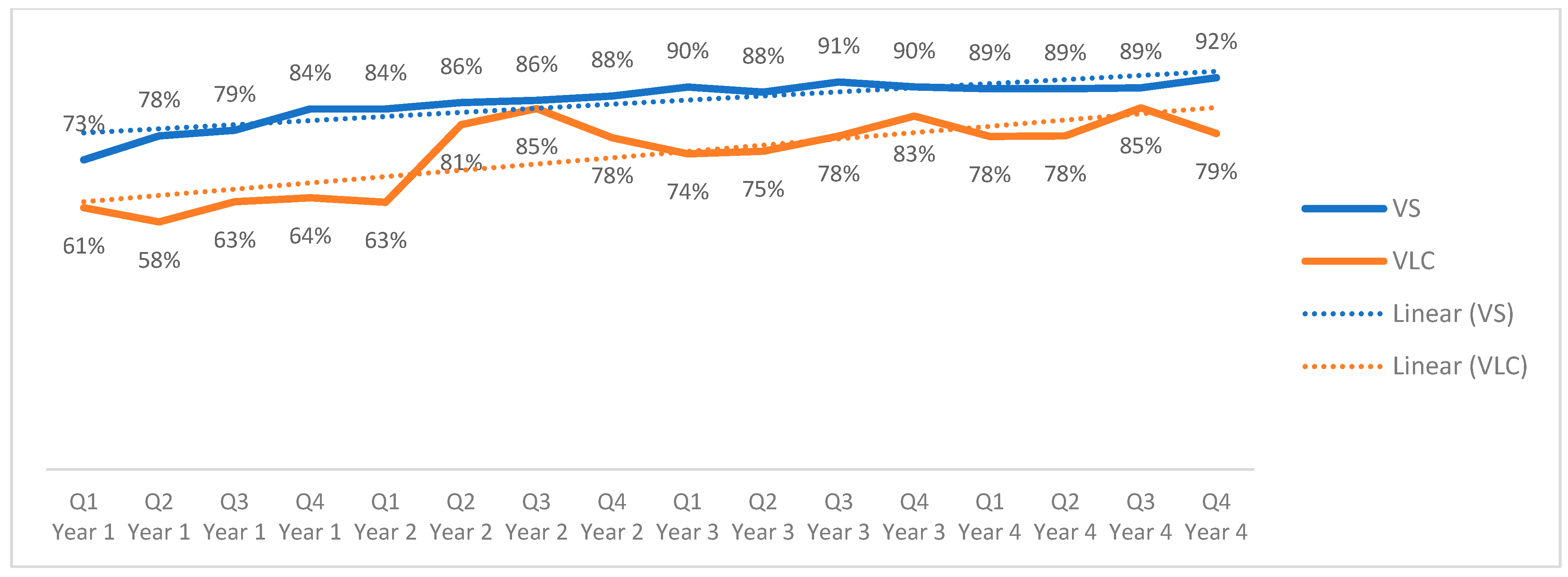
Regional viral load coverage (Figure 5) showed a variable but overall upward trend among adolescents, increasing from 61% to 79% over the four-year period (an 18-percentage point increase, p > 0.10). At the regional level, viral load suppression among adolescents with a documented viral load result increased steadily from 73% in the first trimester of Year 1 to 92% in the last trimester of Year 4 (19-percentage point increase, p > 0.10). Because viral load suppression is calculated only among adolescents with a documented viral load result, these percentages reflect suppression among those with viral load testing and not among all adolescents on ART.
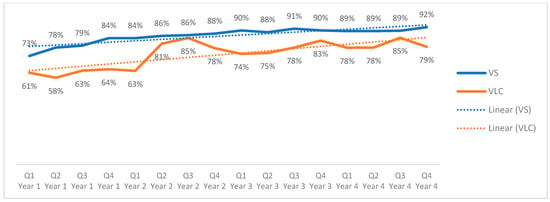
Figure 5.
Regional viral load coverage and suppression in adolescents across quarters, October 2020–to September 2024.
Viral load coverage and suppression demonstrated similar trends among adolescents aged 10–14 and 15–19 years. Viral load suppression among tested adolescents started below 90% in El Salvador, Guatemala, and Honduras in Year 1, but each reached 90% or above by the end of Year 4. The cohorts from Nicaragua reported a VLS of 86% in Year 1, which decreased to 81% by the end of the analyzed period. Panama reported the greatest improvement in VLS among those tested, with 60% virologic suppression reported in Year 1 and 86% reported in Year 4. Panama, although reporting an average of 56% coverage during this period, has seen significant improvements from 30% to 69% coverage. Although viral load coverage and suppression reflect changes across the analysis period, these results are not considered statistically significant with the available PEPFAR data (see Supplemental Table S3).
On average, viral load coverage among adolescents has varied across the region, with 74% reported regionally over the four-year period, indicating that roughly one quarter of adolescents on ART did not have a recent VL result recorded.
3.4. Advanced HIV
Data for advanced HIV disease (AHD) was only available for the final year of the analysis period given that this indicator was not included in previous PEPFAR guidance. Of adolescents new to treatment, 30% were categorized as having AHD. A difference between sexes (40% for women and 28% for men) was noted in the proportion of late-stage diagnoses; however, these differences were not statistically significant (see Supplemental Table S2). Panama and Guatemala have the highest percentages of AHD for both sexes (44% and 33%, respectively) and the highest percentage for women (44% and 53%, respectively). Honduras reported lower AHD among 15–19 years olds (10% in females; 15% in males). However, these values are likely underestimated, as the availability of CD4 results for adolescents in the region ranged from 8% missing in El Salvador to 56% missing in Panama, making age-specific comparisons difficult.
Overall, adolescent treatment outcomes showed modest improvements in initiation and continuity, whereas retention and viral load monitoring remain critical areas for strengthening across all programs.
4. Discussion
This study provides a multi-country descriptive analysis of adolescent HIV cascade outcomes across PEPFAR-supported programs in Central America, highlighting both progress and persistent gaps in testing, treatment, and viral suppression.
Although global estimates show that the burden of adolescent HIV is largely concentrated in sub-Saharan Africa (SSA) [14], the patterns observed in Central America remain relevant for understanding how small but concentrated epidemics respond to prevention and treatment intervention and how global averages can overshadow regional disparities [13,14,21].
4.1. Testing Patterns
Central America has maintained testing with a stable 2% positivity, unlike a previous analysis that documented declines in adolescent HIV testing attributed to service disruptions during COVID and shifts in PEPFAR guidance over similar time periods [22]. This could partly reflect how the involvement of regions with different epidemiological and programmatic contexts [5,23] can obscure trends in regions with smaller cohorts [13]. Despite the near parity in testing between males and females, the proportion of positives results was consistently higher in males. This pattern is consistent with the concentrated epidemic of the region [24,25] and differs from sub-Saharan Africa, where positivity is higher in females [14,26,27].
As we examined individual country patterns, both Guatemala and Panama maintained high HIV testing and positivity rates throughout the analyzed years, whereas Honduras also reports elevated positivity in specific populations. Beyond the context of a concentrated epidemic and the known vulnerability of adolescents to HIV [14,27], existing literature from these countries underscores the importance of considering indigenous populations such as the Ngäbe-Buglé, Garífuna, and Maya Q’eqchi’, who may also face vulnerabilities due to structural barriers, cultural and linguistic differences, and limited health services [13,28,29,30,31,32,33,34,35,36]. Although findings are sometimes inconsistent, these examples highlight both the importance of and the gaps in knowledge needed to tailor the HIV response to the realities of indigenous populations. Evidence from qualitative research suggests that age and ethnicity can act synergistically to deepen vulnerabilities to HIV. El Salvador had a 3% decrease in the incidence of HIV from 2000 to 2019 [37], which may reflect the cumulative impact of sustained national efforts to strengthen multiple prevention strategies [38], testing, and treatment within a primary health care framework, similar to decreasing trends reported in SSA [22]. Another indication of El Salvador’s epidemic control is the elimination of mother-to-child transmission of HIV, which may serve as a proxy indicator for the functionality of the country’s HIV program [39].
National HIV prevalence rates reported by UNAIDS in the analyzed countries range from 0.2 to 1.1 [40]. Positivity proportions among the adolescents included in this analysis ranged from 0.4% to 7.0%, which suggests some degree of targeted testing toward higher-risk adolescents. Although there is a predominance in provider-initiated and facility-based testing, such models may not fully align with adolescents’ preferences and needs [40,41,42]. In Sub-Saharan Africa, a systematic review of HIV uptake and yield among children and adolescents found that, although facility-based testing models reached higher-risk adolescents (late in disease progression) and therefore yielded more positive results, community- and family-based testing models facilitated earlier diagnosis [43]. Notably, index testing was not reported across the programs analyzed. This absence may reflect programmatic and ethical challenges in implementing contact testing among adolescents, similar to findings from a recent Ugandan study, which highlighted that less than one-third (28.7%) of adolescents living with HIV utilized provider-assisted partner notification, primarily due to potential violence and confidentiality concerns [44]. However, evidence from other high-burden settings demonstrates that index testing can achieve substantially higher yields than other testing modalities [45]. Peer- and network-based approaches, including social network strategy, leverage referrals within existing youth networks and have been associated with increased testing and case identification among young and high-risk populations [46] Because age shapes both risk and availability, strategies should differentiate between younger adolescents (10–14 years), more often reached though family/index testing and pediatric services, and older adolescents (15–19 years), who are more likely to seek services themselves and engage via peer/network approaches [47]. These findings underscore the need to expand adolescent-friendly outreach through strategies such as home visits, peer networks, family-based testing, self-testing, and potential adaptations of age-appropriate approaches to close remaining gaps in HIV detection among undiagnosed adolescents [42,48,49]. Additionally, restrictive adolescent consent laws in the region continue to limit the availability of HIV testing and prevention services, leaving healthcare providers uncertain about whether to prioritize the protection of minors or the promotion of self-care [50,51,52].
4.2. Treatment Pattern
Despite a 21% increase in new treatment initiation over the analyzed years, the overall proportion of adolescents within the HIV treatment cohorts remained stable. This suggest that although linkage to treatment has improved in some countries such as Panama and Nicaragua, many new initiations may have been balanced out by the transition to adult services. The consistently small cohorts in El Salvador and Panama could also reflect challenges in early diagnosis and long-term retention.
This pattern of increased ART initiation without a proportional increase in the cohort is not unique to Central America. In Lima, Peru, adolescents and young adults experienced low retention despite initiating ART, a trend largely attributed to structural barriers such as clinic hours, mental health morbidity, and the ineffective transition to adult care. Many adolescents’ initiating treatment between the ages of 15 and 19 were enrolled directly into adult services [53]. The age of initiation and lack of transitioning services in this age group should be considered in Central America.
Treatment continuity trends revealed decreasing interruption rates and an expansion of multi-month dispensing of ART. By year 4, interruption rates among adolescents and adults converged, suggesting some narrowing of the gap in program continuity. However, these improvements may still be fragile, and we were not able to calculate individual-level retention with aggregate MER data. The higher proportion of males on treatment (58%) may also highlight the cumulative effect of testing patterns, where adolescent males, although less likely to test, were more likely to test positive and thus appear in treatment cohorts. This pattern emphasizes the need for differentiated care models that address the needs of both sexes. Sustained retention among adolescents requires multiple layered approaches, including community and virtual clinical services, psychosocial support, peer support, and trained providers to address age-specific needs [48]. Although retention issues in our region remain modest, a combination of interventions could stabilize and progressively strengthen retention over time, particularly among male adolescents.
4.3. Viral Load Coverage and Suppression Patterns
Adolescents persistently lag behind adults in virologic suppression worldwide because of their unique barriers in adhering to treatment: developing executive function, lack of systemic support via school-based programs, and psychosocial challenges that require adolescent-specific treatment strategies and specialized support approaches beyond those traditionally offered for adults [54,55]. In our analysis, virologic suppression among those with a viral load result improved over time; however, viral load coverage remained incomplete, meaning that a substantial proportion of adolescents on ART did not receive a documented viral load test. Because suppression is typically calculated only among those tested, viral load results may overestimate true population-level suppression when coverage is suboptimal. Therefore, interpreting adolescent viral load outcomes requires considering both coverage and suppression together, rather than either indicator alone. Continued efforts to scale up viral-load testing and coverage remain essential to achieve more complete monitoring of adolescent treatment outcomes and to support the accurate assessment of program progress [56].
In Panama, where adolescents exhibited high positivity but smaller treatment cohorts, drug resistance may complicate viral suppression outcomes. A recent study by Ventosa-Cubillo et al. (2023) found that more than half of children and adolescents living with HIV carried major drug resistance mutations, primarily to NRTIs and NNRTIs [57] Some ART-naïve adolescents were already infected with resistant strains, indicating the ongoing transmission of resistant viruses. These findings underscore the need for routine surveillance and robust adherence support, highlighting that improvements in viral load suppression will not only depend on programmatic adherence but also on the availability of effective regimens [58].
Beyond pharmacologic monitoring, stronger adolescent support is still essential. Regional efforts to increase peer support programs and leverage adolescent-friendly mobile health technologies are needed to improve adolescent virologic suppression. Similarly, differentiated service delivery models shown to support adolescent adherence and retention should be implemented throughout the region, including MMD for adolescents and adolescent friendly clinics [1]. Family dynamics, school schedules, transportation limitations, and developmental needs should all be factored into “adolescent-friendly” services [59,60].
4.4. Advanced HIV Disease at Diagnosis
The high prevalence of AHD and the fact that PITC is the highest-yield testing modality among adolescents new to treatment (34%) highlight missed opportunities for early detection. Late presentation and advanced HIV disease has already been documented in Latin America [61]. The sex difference (40% females and 28% males) suggest that women may be facing additional barriers, contrary to a study that found that sex was a significant predictor of advanced HIV disease (AHD) among young people in Sierra Leone, with females demonstrating significantly lower odds of having AHD compared to males [62]. As noted above, most testing takes place at health facilities, whereas current research suggests that adolescents prefer more community-based or home-based testing [1]. For epidemics such as Central America’s, which are concentrated primarily among high-risk populations, integrating testing and prevention into community spaces may be a future focus to better address issues related to privacy and confidentiality concerns. Integral to testing programs in community are focused resources to link adolescents who test positive for HIV to care. Without this crucial step, AHD rates will remain elevated. Higher rates of AHD among women may reveal gaps in community-based testing for young women or structural barriers to prevention, testing, and linkage to care among adolescent young women. Efforts to screen for and address violence may be a crucial component of HIV testing outreach for adolescent women [62,63].
It is important to highlight that the interpretation of advanced HIV disease among adolescents requires caution because data were only available for the final year. Similar to VL documentation, data completeness varies substantially across countries, which could lead to the underestimation or overestimation of values.
4.5. Limitations and Strengths
This analysis is subject to several limitations:
- It relies on routine programmatic data from DATIM, which are de-identified, aggregated, and not patient-level, limiting the ability to explore individual outcomes across the HIV cascade or evaluate person-level retention or adherence. This introduces the possibility that aggregated numbers may not represent individual data.
- Given the aggregate nature of these data, the timing of infection remains unclear in these adolescents. If these are missed perinatal transmissions, regulations should be focused on improved testing for infants and toddlers with HIV exposure. If these are primarily new infections from sexual activity, guidelines should focus on HIV prevention among adolescents. Further, patient-level data is needed to help guide these initiatives.
- Viral load suppression is reported only among those with a documented viral load result, whereas viral load coverage was incomplete, varying across countries and years. This could lead to an overestimation of true population-level suppression.
- Data completeness varies across countries, particularly for CD4 testing, which is essential to determine AHD status. CD4 results were available only for the final year and missing data limits our ability to assess AHD reliably or make comparisons across sexes or age groups.
- Key disaggregations by high-risk populations, migration status, and ethnicity were not available, restricting further analysis of vulnerable populations. Other indicators associated with the adolescent HIV cascade, such as behavioral, socioeconomic, or structural barriers, are not included in routine MER reporting, which may limit interpretation.
- Finally, because the analysis includes only PEPFAR-supported sites, findings may not represent all adolescents receiving HIV services nationally.
Despite these limitations, the study’s strengths include its multi-country scope, standardized MER definitions, and the use of core cascade indicators to provide a regional perspective for adolescents in Central America, which is rarely available in our region.
5. Conclusions
Across Central America, adolescent HIV outcomes showed areas of progress alongside persistent programmatic gaps. Testing volume was maintained over the four-year period, yet case detection remained concentrated among older adolescents and males, and a substantial proportion of new diagnoses presented with advanced HIV disease. Although viral load suppression among those tested improved, incomplete viral load coverage may indicate that many adolescents on ART are not yet fully captured in monitoring systems. These patterns underscore that further gains in adolescent outcomes will depend on continued attention to earlier diagnosis, consistent viral load monitoring, and age-responsive service delivery. The analysis also highlights the diversity of testing modalities and treatment cohorts across countries, shaped by differences in service availability, reporting practices, and epidemic profiles. Approaches such as confidential, age-appropriate outreach, social and peer-network strategies, re-evaluation of the age of testing consent laws, and adaptations of partner-based testing—already used in other settings—illustrate potential avenues through which programs in the region can expand reach and support adolescent engagement.
Although this study was not designed to evaluate specific interventions, the findings point to opportunities for countries to draw on existing national platforms and experience to strengthen adolescent-focused components of their HIV programs as they pursue progress toward the 95-95-95 goals.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/tropicalmed11010005/s1, Supplemental Table S1: HIV testing and treatment outcomes in Central America, October 2020 to September 2024; Supplemental Table S2: Comparison of HIV positivity yield, advanced HIV disease results, and Honduras adolescent and adult cohorts, by sex, Supplemental Table S3: Viral load coverage and suppression proportions by country, Year 1 vs. Year 4, Supplemental Table S4: Adolescent and total cohort size by country, October 2020 to September 2024, Supplemental Figure S1: HIV testing among adolescents in Central American countries supported by PEPFAR, October 2020 to September 2024, Supplemental Figure S2: Positive HIV tests among adolescents in Central American countries supported by PEPFAR, October 2020 to September 2024, Supplemental Figure S3: Adolescent users new to treatment in Central American countries supported by PEPFAR, October 2020 to September 2024, Supplemental Figure S4: Total adolescents on treatment in Central American countries supported by PEPFAR, October 2020 to September 2024, Supplemental Figure S5: Treatment interruptions among adolescents in Central American countries supported by PEPFAR, October 2020 to September 2024.
Author Contributions
L.R.C. conceived the study, conducted the formal analysis and visualization, and drafted the original manuscript. J.R. contributed to the study methodology and conducted the formal data analysis. C.G. and N.A. assisted in data analysis, preparation, and writing of the original draft. A.A.R., N.F., C.C., and B.R.P. provided validation and critical review of the analysis. C.C., N.F., A.A.R., and B.R.P. contributed to the review and editing of the final manuscript. B.R.P. supervised the project. All authors have read and agreed to the published version of the manuscript.
Funding
This manuscript was supported by the President’s Emergency Plan for AIDS Relief (PEPFAR) through the Centers for Disease Control and Prevention (CDC) under Cooperative Agreement NU2GGH002261 and NU2GGH002262.
Institutional Review Board Statement
Ethical approval for the study was obtained from Centers for Disease Control and Prevention (CDC) Division of Global HIV & TB on 30 October 2025 (Approval No. 0900f3eb825f5abb).
Informed Consent Statement
We have no need to obtain informed consent because the routine data used in this study were collected in aggregate form, ensuring that individual-level data cannot be identified.
Data Availability Statement
The data analyzed in this study were obtained from the U.S. President’s Emergency Plan for AIDS Relief (PEPFAR) Data for Accountability, Transparency, and Impact Monitoring (DATIM) system. Access to DATIM data is restricted to authorized users and implementing partners. Therefore, the data is not publicly available due to programmatic confidentiality agreements. Aggregated data supporting the findings of this study may be available from the corresponding author upon reasonable request and with permission from PEPFAR.
Acknowledgments
During the preparation of this manuscript, the authors used ChatGPT (GPT-5, OpenAI, 2025) to assist with English language editing, summarizing scientific literature, and improving clarity and structure. The authors reviewed, verified, and edited all AI-generated content and take full responsibility for the accuracy and integrity of the final text.
Conflicts of Interest
The authors declare no conflicts of interest. CDC disclaimer: The findings and conclusions in this manuscript are those of the authors and do not necessarily represent the official position of the funding agencies.
Abbreviations
The following abbreviations are used in this manuscript:
| AHD | Advanced HIV Disease |
| ART | Antiretroviral Therapy |
| CDC | U.S. Centers for Disease Control and Prevention |
| CD4 | Cluster of Differentiation 4 (T lymphocyte count) |
| ELS | El Salvador |
| FY | Fiscal Year |
| GUA | Guatemala |
| HIV | Human Immunodeficiency Virus |
| HON | Honduras |
| HTS | HIV Testing Services |
| MMD | Multi-Month Dispensing (of ART) |
| MER | Monitoring, Evaluation, and Reporting (PEPFAR reporting framework) |
| NIC | Nicaragua |
| PAN | Panama |
| PEPFAR | U.S. President’s Emergency Plan for AIDS Relief |
| PITC | Provider-Initiated Testing and Counselling |
| PMTCT | Prevention of Mother-to-Child Transmission |
| SSA | Sub-Saharan Africa |
| STI | Sexually Transmitted Infection |
| UNAIDS | Joint United Nations Programme on HIV/AIDS |
| VCT | Voluntary Counselling and Testing |
| VLC | Viral Load Coverage |
| VLS | Viral Load Suppression |
| WHO | World Health Organization |
References
- WHO. Implementing WHO Evidence-Based Interventions for Adolescents and Young Adults Living with and Affected by HIV; World Health Organization: Geneva, Switzerland, 2024; Available online: https://iris.who.int/bitstream/handle/10665/379634/9789240100411-eng.pdf?sequence=1 (accessed on 21 September 2025).
- WHO. Adolescent-Friendly Health Services for Adolescents Living with HIV: From Theory to Practice; World Health Organization: Geneva, Switzerland, 2019; Available online: https://iris.who.int/bitstream/handle/10665/329993/WHO-CDS-HIV-19.39-eng.pdf?sequence=1 (accessed on 23 July 2025).
- Machado, D.M.; Duda, S.N.; Celia de Menezes Succi, R.; Kim, A.; Ranadive, P.; Rouzier, V.; Crabtree-Ramirez, B.; Luque, M.T.; Mejia, F.; Rodriguez, F.; et al. HIV Care Continuum Outcomes Among Adolescents and Young Adults Living With HIV in Latin America and the Caribbean: Association With Depression and Substance Use. Open Forum Infect. Dis. 2025, 12, ofaf353. [Google Scholar] [CrossRef]
- Embleton, L.; Sudjaritruk, T.; Machado, D.M.; Chihota, B.; Musabyimana, F.; Jesson, J.; Apondi, E.; Puthanakit, T.; Luque, M.T.; van Dongen, N.E.; et al. Characterizing adolescent and youth-friendly HIV services: A cross-sectional assessment across 16 global sites. J. Int. AIDS Soc. 2025, 28, e26437. [Google Scholar] [CrossRef]
- Bailey, H.; Cruz, M.L.S.; Songtaweesin, W.N.; Puthanakit, T. Adolescents with HIV and transition to adult care in the Caribbean, Central America and South America, Eastern Europe and Asia and Pacific regions. J. Int. AIDS Soc. 2017, 20, 21475. [Google Scholar] [CrossRef]
- UNAIDS. Country Factsheets: Guatemala; UNAIDS: Geneva, Switzerland, 2024; Available online: https://www.unaids.org/en/regionscountries/countries/guatemala (accessed on 23 September 2025).
- UNAIDS. Country Factsheets: Honduras; UNAIDS: Geneva, Switzerland, 2024; Available online: https://www.unaids.org/en/regionscountries/countries/honduras (accessed on 23 September 2025).
- UNAIDS. Country Factsheets: El Salvador; UNAIDS: Geneva, Switzerland, 2024; Available online: https://www.unaids.org/en/regionscountries/countries/elsalvador (accessed on 23 September 2025).
- UNAIDS. Country Factsheets: Nicaragua; UNAIDS: Geneva, Switzerland, 2024; Available online: https://www.unaids.org/en/regionscountries/countries/nicaragua (accessed on 23 September 2025).
- UNAIDS. Country Factsheets: Panama; UNAIDS: Geneva, Switzerland, 2024; Available online: https://www.unaids.org/en/regionscountries/countries/panama (accessed on 23 September 2025).
- UNAIDS. Global Data on HIV Epidemiology and Response. Available online: https://aidsinfo.unaids.org/ (accessed on 21 September 2025).
- UNAIDS. Latin America 2024 Global AIDS Update; The Joint United Nations Programme on HIV/AIDS: Geneva, Switzerland, 2024; Available online: https://www.unaids.org/fr/node/58697?utm_source=chatgpt.com (accessed on 22 September 2025).
- Chen, S. The statistical illusion of a global HIV endgame: Disparities in regional progress toward 95-95-95 targets. Front. Public Health 2025, 13, 1602711. [Google Scholar] [CrossRef] [PubMed]
- Slogrove, A.L.; Sohn, A.H. The global epidemiology of adolescents living with HIV: Time for more granular data to improve adolescent health outcomes. Curr. Opin. HIV AIDS 2018, 13, 170–178. [Google Scholar] [CrossRef] [PubMed]
- PEPFAR. Panorama Platform. U.S. Department of State. Available online: https://data.pepfar.gov (accessed on 23 September 2025).
- PEPFAR. Data for Accountability, Transparency and Impact Monitoring (DATIM). Available online: https://www.datim.org (accessed on 2 September 2025).
- Monitoring, Evaluation, and Reporting Indicator Reference Guide. 2025. Available online: https://help.datim.org/hc/en-us/articles/360000084446-MER-Indicator-Reference-Guides (accessed on 2 September 2025).
- Gardner, E.M.; Young, B. The HIV care cascade through time. Lancet Infect. Dis. 2014, 14, 5–6. [Google Scholar] [CrossRef]
- UNAIDS. Draft Recommendations—Global Task Team for Setting 2030 HIV Targets. 2025. Available online: https://www.unaids.org/sites/default/files/2025-05/20250328_recommended_2030_HIV_targets_livedocument_en_13_May_2025.pdf (accessed on 3 September 2025).
- Waldrop, G.; Doherty, M.; Vitoria, M.; Ford, N. Stable patients and patients with advanced disease: Consensus definitions to support sustained scale up of antiretroviral therapy. Trop. Med. Int. Health 2016, 21, 1124–1130. [Google Scholar] [CrossRef] [PubMed]
- UNAIDS. What the Data Tell Us: Projection for the HIV Epidemic in Latin America in 2030; Joint United Nations Programme on HIV/AIDS: Geneva, Switzerland, 2024; Available online: https://www.unaids.org/sites/default/files/2025-03/la_2030-projections-hiv-epidemic_en.pdf (accessed on 3 September 2025).
- Hrapcak, S.; Hast, M.; Okegbe, T.; Gross, J.; Williams, J.; Patel, M.; Wolf, H.; Siberry, G.; Lee, L.; Wiersma, S.; et al. The Status of Adolescent Testing and Treatment in PEPFAR-Supported Programs, October 2017 to September 2020. J. Acquir. Immune Defic. Syndr. 2023, 93, 15–24. [Google Scholar] [CrossRef]
- Benyumiza, D.; Amongin, J.F.; Ochaba, I.; Adupa, M.; Abuch, N.; Banula, C.B.; Udho, S. Factors Associated with Utilization of HIV Testing Services among Adolescents Aged 10-19 Years in Lira District, Northern Uganda: A Cross-Sectional Study. Biomed. Res. Int. 2021, 2021, 9568148. [Google Scholar] [CrossRef]
- Boyce, S.C.; Morales-Miranda, S.; Ritter, J.; Triplett, D.P.; Loya-Montiel, I.; Silverman, J.G. HIV Infection and Risk Heightened Among Female Sex Workers Who Entered the Sex Trade as Adolescents in Guatemala. AIDS Behav. 2020, 24, 2906–2917. [Google Scholar] [CrossRef]
- Coelho, L.E.; Torres, T.S.; Veloso, V.G.; Grinsztejn, B.; Jalil, E.M.; Wilson, E.C.; McFarland, W. The Prevalence of HIV Among Men Who Have Sex With Men (MSM) and Young MSM in Latin America and the Caribbean: A Systematic Review. AIDS Behav. 2021, 25, 3223–3237. [Google Scholar] [CrossRef]
- Mhungu, A.; Sixsmith, J.; Burnett, E. Adolescent Girls and Young Women’s Experiences of Living with HIV in the Context of Patriarchal Culture in Sub-Saharan Africa: A Scoping Review. AIDS Behav. 2023, 27, 1365–1379. [Google Scholar] [CrossRef]
- Murewanhema, G.; Musuka, G.; Moyo, P.; Moyo, E.; Dzinamarira, T. HIV and adolescent girls and young women in sub-Saharan Africa: A call for expedited action to reduce new infections. IJID Reg. 2022, 5, 30–32. [Google Scholar] [CrossRef]
- Gabster, A.; Mayaud, P.; Pascale, J.M.; Cislaghi, B. Gender norms and sexual behaviours among Indigenous youth of the Comarca Ngabe-Bugle, Panama. Cult. Health Sex. 2020, 22, 1032–1046. [Google Scholar] [CrossRef] [PubMed]
- Gabster, A.; Mayaud, P.; Jhangimal, M.; Pascale, J.M.; Francis, S.C.; Cislaghi, B. Social norms that sustain transactional sex and associations with sexual health outcomes: A mixed-methods study in the Comarca Ngabe-Bugle, a rural-Indigenous region of Panama. PLoS ONE 2024, 19, e0304805. [Google Scholar] [CrossRef]
- Dunleavy, V.O.; Phillips, J.R.; Chudnovskaya, E.V. A Community-Based Approach to HIV Prevention: Engaging Mayan Young Adults in Rural Guatemala. J. Health Care Poor Underserved 2019, 30, 1001–1023. [Google Scholar] [CrossRef]
- United Nations Human Rights Council. Addendum to the Report of the Working Group on the Universal Periodic Review: Honduras (A/HRC/33/42/Add.2); United Nations: New York, NY, USA, 2016; Available online: https://docs.un.org/en/A%2FHRC%2F33%2F42%2FAdd.2 (accessed on 25 August 2025).
- SESAL. Informe Estadistico de la Epidemia de VIH en Honduras; Secretaria de Salud: Tegucigalpa, Honduras, 2023; Available online: https://salud.gob.hn/sshome/index.php/component/jdownloads/?task=download.send&id=1371 (accessed on 26 August 2025).
- SESAL. Informe Estadistico de la Epidemia de VIH en Honduras Periodo 1985-Marzo 2024; Secretaria de Salud: Tegucigalpa, Honduras, 2024; Available online: https://salud.gob.hn/sshome/index.php/component/jdownloads/?task=download.send&id=1522 (accessed on 26 August 2025).
- MINSA. Boletín Informativo 2023; Ministry of Health: Panama City, Panama, 2023. Available online: https://www.minsa.gob.pa/sites/default/files/programas/boletin_noviembre_2023_1.pdf (accessed on 8 September 2025).
- MINSA. Informe del Monitoreo Nacional de Campo de los Indicadores Reportados por las CTARV y Diferentes Programas del MINSA, CSS, CLAMS, Regiones de Salud, ONG y Otros; Ministry of Health: Panama City, Panama, 2024; Available online: https://www.minsa.gob.pa/sites/default/files/programas/informe_nacional_de_mye_2024-2025.pdf (accessed on 8 September 2025).
- Taylor, T.M.; Hembling, J.; Bertrand, J.T. Ethnicity and HIV risk behaviour, testing and knowledge in Guatemala. Ethn. Health 2015, 20, 163–177. [Google Scholar] [CrossRef]
- PAHO. Health in the Americas 2023: El Salvador Country Profile; Pan American Health Organization: Washington, DC, USA, 2023; Available online: https://hia.paho.org/pt/node/216?utm_source=chatgpt.com#situation (accessed on 26 August 2025).
- Dickson-Gomez, J.; Tarima, S.; Glasman, L.; Cuellar, W.; de Mendoza, L.R.; Bodnar, G. Cumulative Effects of Adding a Small Group Intervention to Social Network Testing on HIV Testing Rates Among Crack Users in San Salvador, El Salvador. AIDS Behav. 2021, 25, 2316–2323. [Google Scholar] [CrossRef]
- WHO. Global Guidance on Criteria and Processes for Validation: Elimination of Mother-to-Child Transmission of HIV and Syphilis; World Health Organization: Geneva, Switzerland, 2021; Available online: https://www.who.int/publications/i/item/9789240039360 (accessed on 23 July 2025).
- Edwards, P.V.; Roberts, S.T.; Chelwa, N.; Phiri, L.; Nyblade, L.; Mulenga, D.; Brander, C.; Musheke, M.; Mbizvo, M.; Subramanian, S. Perspectives of Adolescent Girls and Young Women on Optimizing Youth-Friendly HIV and Sexual and Reproductive Health Care in Zambia. Front. Glob. Womens Health 2021, 2, 723620. [Google Scholar] [CrossRef] [PubMed]
- Mwaba, K.; Mannell, J.; Burgess, R.; Sherr, L. Uptake of HIV testing among 15-19-year-old adolescents in Zambia. AIDS Care 2020, 32, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Inwani, I.; Chhun, N.; Agot, K.; Cleland, C.M.; Rao, S.O.; Nduati, R.; Kinuthia, J.; Kurth, A.E. Preferred HIV Testing Modalities Among Adolescent Girls and Young Women in Kenya. J. Adolesc. Health 2021, 68, 497–507. [Google Scholar] [CrossRef]
- Govindasamy, D.; Ferrand, R.A.; Wilmore, S.M.; Ford, N.; Ahmed, S.; Afnan-Holmes, H.; Kranzer, K. Uptake and yield of HIV testing and counselling among children and adolescents in sub-Saharan Africa: A systematic review. J. Int. AIDS Soc. 2015, 18, 20182. [Google Scholar] [CrossRef]
- Kinera-Kagimu, I.; Katuramu, R.; Nangendo, J.; Semitala, F.C.; Nabukeera-Barungi, N.; Dhabangi, A. Provider assisted partner notification utilization and associated factors among adolescents living with HIV in Uganda, a cross-sectional study. BMC Pediatr. 2024, 24, 785. [Google Scholar] [CrossRef]
- Shamu, S.; Farirai, T.; Kuwanda, L.; Slabbert, J.; Guloba, G.; Khupakonke, S.; Johnson, S.; Masihleho, N.; Kamera, J.; Nkhwashu, N. Comparison of community-based HIV counselling and testing (CBCT) through index client tracing and other modalities: Outcomes in 13 South African high HIV prevalence districts by gender and age. PLoS ONE 2019, 14, e0221215. [Google Scholar] [CrossRef] [PubMed]
- Paiboon, N.; Songtaweesin, W.N.; Wongharn, P.; Moonwong, J.; Khamthi, S.; Premgamone, A.; Theerawit, T.; Saisaengjan, C.; Kawichai, S.; Anugulruengkitt, S.; et al. Social Network Strategy to Promote HIV Testing and Linkage to HIV Services among Young men who Have sex with men and Transgender Women in Thailand. J. Int. Assoc. Provid. AIDS Care 2022, 21, 23259582221143672. [Google Scholar] [CrossRef]
- Aheron, S.; Paredes-Vincent, A.; Patton, M.E.; Gross, J.; Medley, A.; Mona, G.; Mtimkulu, N.; Nkuna, K.; Grund, J.M. Improving HIV Case Finding Through Index Testing: Findings from Health Facilities in 12 Districts of South Africa, October 2019–September 2021. AIDS Behav. 2024, 28, 1708–1718. [Google Scholar] [CrossRef]
- Zanoni, B.C.; Elliott, R.J.; Neilan, A.M.; Haberer, J.E. Screening for HIV and linkage to care in adolescents: Insights from a systematic review of recent interventions in high- versus low- and middle-income settings. Adolesc. Health Med. Ther. 2018, 9, 211–235. [Google Scholar] [CrossRef] [PubMed]
- Zanoni, B.; Archary, M.; Sibaya, T.; Ramos, T.; Donenberg, G.; Shahmanesh, M.; Celum, C.; Pettifor, A.; Bekker, L.G.; Haberer, J. Interventions addressing the adolescent HIV continuum of care in South Africa: A systematic review and modified Delphi analysis. BMJ Open 2022, 12, e057797. [Google Scholar] [CrossRef]
- McKinnon, B.; Vandermorris, A. National age-of-consent laws and adolescent HIV testing in sub-Saharan Africa: A propensity-score matched study. Bull. World Health Organ. 2019, 97, 42–50. [Google Scholar] [CrossRef]
- Legislativa, A. Ley de prevención y control de la infección provocada por el virus de inmunodeficiencia humana; Republica de Panama Asamblea Legistlativa: Panama City, Panama, 2001. [Google Scholar]
- Legislativa, A. Que Regula los Derechos y Obligaciones de los Pacientes, en Materia de Informacion y de Decision Libre Informada; Gaceta Oficial: Panama City, Panama, 2003; Volume 68. [Google Scholar]
- Niwa, M.; Matos, E.; Rodriguez, C.; Wong, M.; Errea, R.A.; Ramos, A.; Senador, L.; Contreras, C.; Galea, J.T.; Lindeborg, A.; et al. Retention and Viral Suppression Among Adolescents Newly Initiating Antiretroviral Therapy in Adult HIV Care in Lima, Peru: A Retrospective Cohort Study. J. Adolesc. Health 2024, 74, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Wong, V.J.; Murray, K.R.; Phelps, B.R.; Vermund, S.H.; McCarraher, D.R. Adolescents, young people, and the 90-90-90 goals: A call to improve HIV testing and linkage to treatment. AIDS 2017, 31, S191–S194. [Google Scholar] [CrossRef]
- Mulawa, M.I.; Knippler, E.T.; Al-Mujtaba, M.; Wilkinson, T.H.; Ravi, V.K.; Ledbetter, L.S. Interventions to Improve Adolescent HIV Care Outcomes. Curr. HIV/AIDS Rep. 2023, 20, 218–230. [Google Scholar] [CrossRef]
- Lecher, S.L.; Fonjungo, P.; Ellenberger, D.; Toure, C.A.; Alemnji, G.; Bowen, N.; Basiye, F.; Beukes, A.; Carmona, S.; de Klerk, M.; et al. HIV Viral Load Monitoring Among Patients Receiving Antiretroviral Therapy—Eight Sub-Saharan Africa Countries, 2013–2018. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 775–778. [Google Scholar] [CrossRef] [PubMed]
- Ventosa-Cubillo, J.; Pinzon, R.; Gonzalez-Alba, J.M.; Estripeaut, D.; Navarro, M.L.; Holguin, A. Drug resistance in children and adolescents with HIV in Panama. J. Antimicrob. Chemother. 2023, 78, 423–435. [Google Scholar] [CrossRef]
- Alonso, S.; Salazar, E.; Galvez, N.; Navas, J.; Quintana, C.; Orozco, N.; Prieto, L.; Navarro, M.L.; Guillen, S.; Gomez-Alba, J.M.; et al. Prevalence of acquired resistance to antiretrovirals in children and adolescents living with HIV under clinical follow-up at the Roosevelt Hospital in Guatemala. J. Infect. Public. Health 2025, 18, 102827. [Google Scholar] [CrossRef]
- Zanoni, B.C.; Sibaya, T.; Cairns, C.; Lammert, S.; Haberer, J.E. Higher retention and viral suppression with adolescent-focused HIV clinic in South Africa. PLoS ONE 2017, 12, e0190260. [Google Scholar] [CrossRef]
- Wilson, K.; Onyango, A.; Mugo, C.; Guthrie, B.; Slyker, J.; Richardson, B.; John-Stewart, G.; Inwani, I.; Bukusi, D.; Wamalwa, D.; et al. Kenyan HIV Clinics With Youth-Friendly Services and Trained Providers Have a Higher Prevalence of Viral Suppression Among Adolescents and Young Adults: Results From an Observational Study. J. Assoc. Nurses AIDS Care 2022, 33, 45–53. [Google Scholar] [CrossRef]
- Belaunzaran-Zamudio, P.F.; Caro-Vega, Y.N.; Shepherd, B.E.; Rebeiro, P.F.; Crabtree-Ramirez, B.E.; Cortes, C.P.; Grinsztejn, B.; Gotuzzo, E.; Mejia, F.; Padgett, D.; et al. The Population Impact of Late Presentation With Advanced HIV Disease and Delayed Antiretroviral Therapy in Adults Receiving HIV Care in Latin America. Am. J. Epidemiol. 2020, 189, 564–572. [Google Scholar] [CrossRef]
- Baldeh, M.; Kizito, S.; Lakoh, S.; Sesay, D.; Williams, S.A.; Barrie, U.; Dennis, F.; Robinson, D.R.; Lamontagne, F.; Amahowe, F.; et al. Advanced HIV disease and associated factors among young people aged 15–24 years at a tertiary hospital in Sierra Leone: A cross-sectional study. BMC Infect. Dis. 2024, 24, 611. [Google Scholar] [CrossRef] [PubMed]
- Chabikuli, O.N.; Ditekemena, J.D.; Sigwadhi, L.N.; Mulenga, A.; Mboyo, A.; Bidashimwa, D.; Nachega, J.B. Advanced HIV Disease at Antiretroviral Therapy Initiation and Treatment Outcomes Among Children and Adolescents Compared to Adults Living With HIV in Kinshasa, Democratic Republic of the Congo. J. Int. Assoc. Provid. AIDS Care 2023, 22, 23259582231221955. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.