Homologous Recombination: A GRAS Yeast Genome Editing Tool
Abstract
1. The Yeast Saccharomyces cerevisiae—A Companion through the Ages
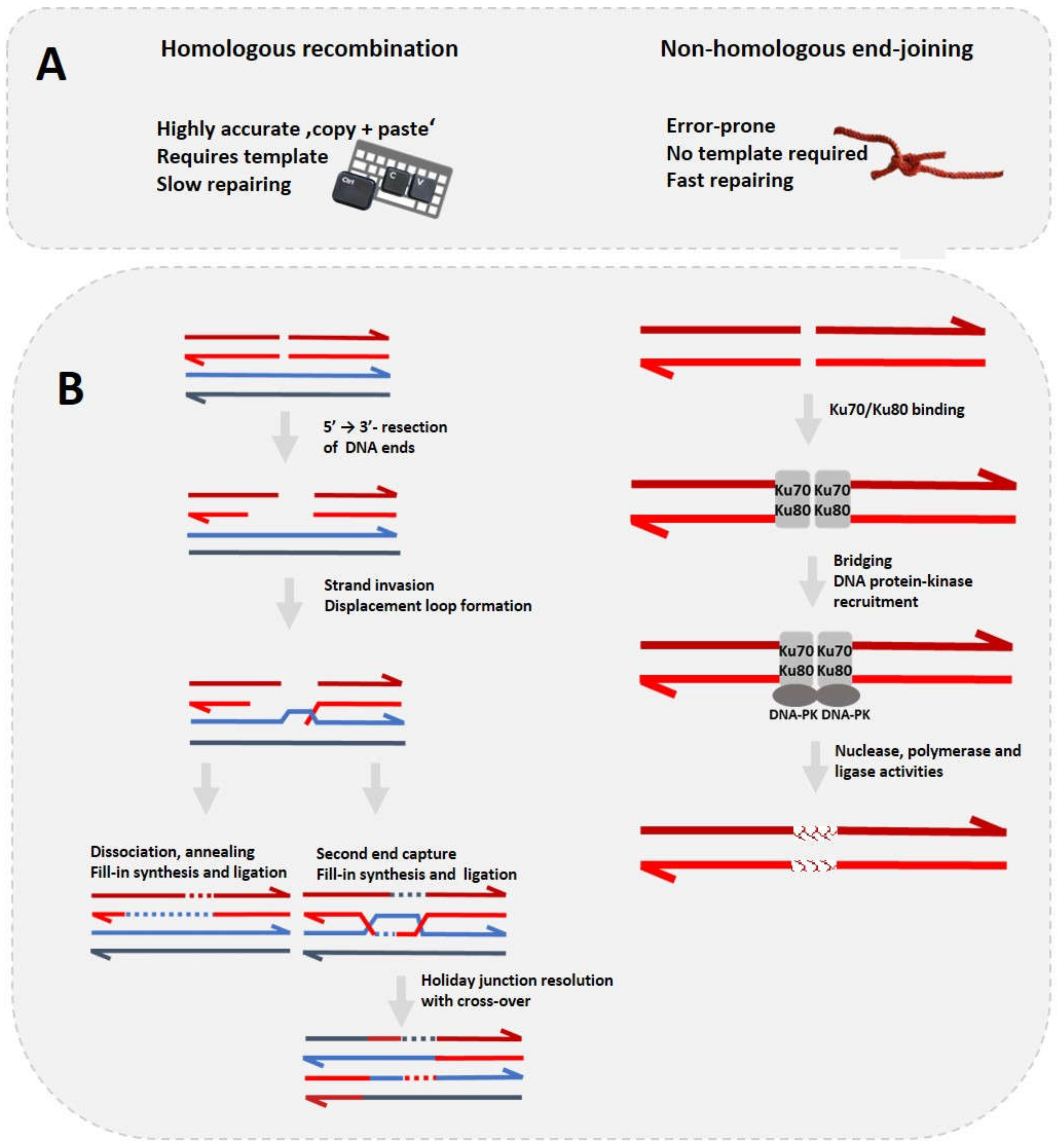
2. Homologous Recombination—A Trusted ‘Copy + Paste’ Genome Editing Tool
3. Progress in Yeast Genome Editing
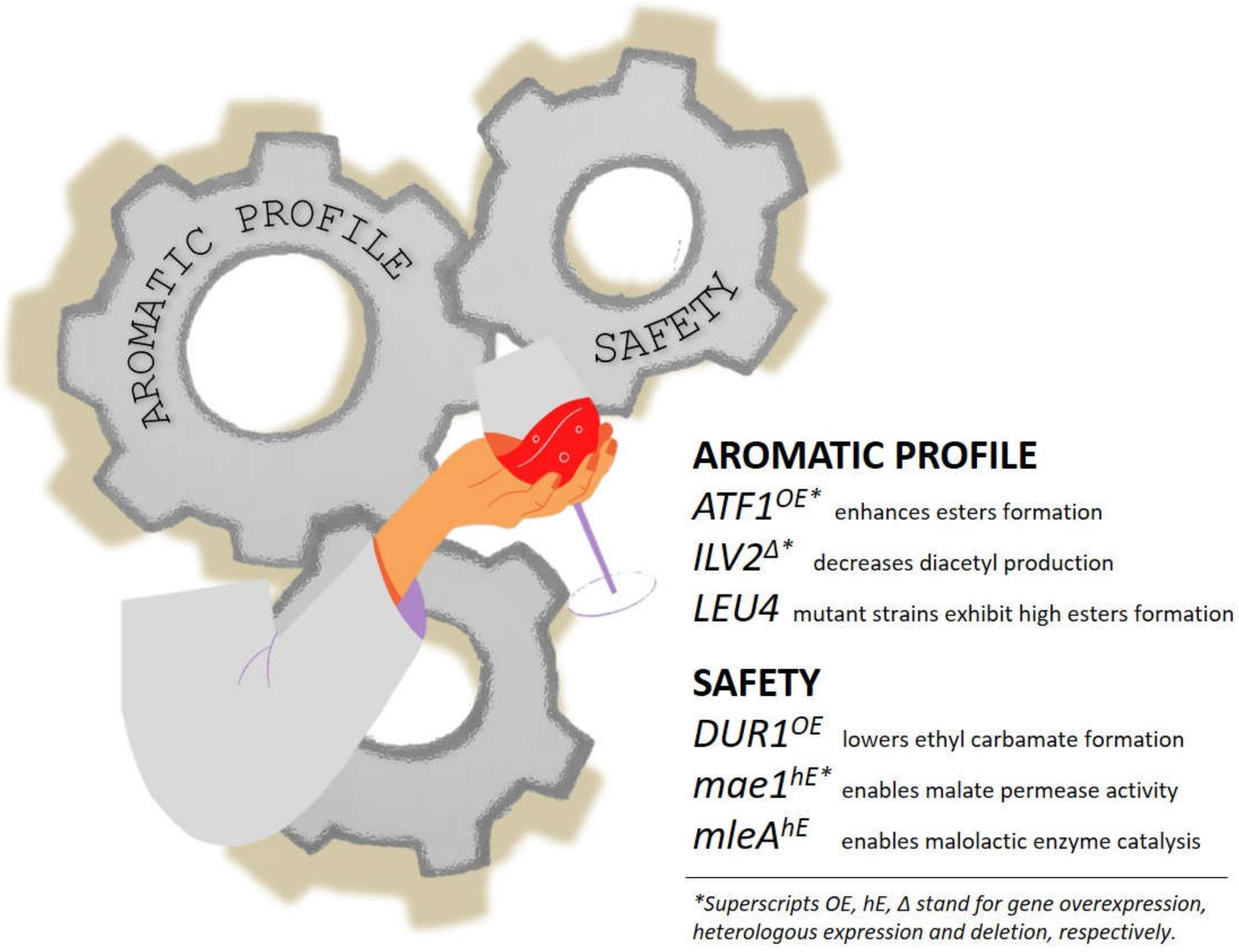
4. Genome Editing in the Beverage Industry
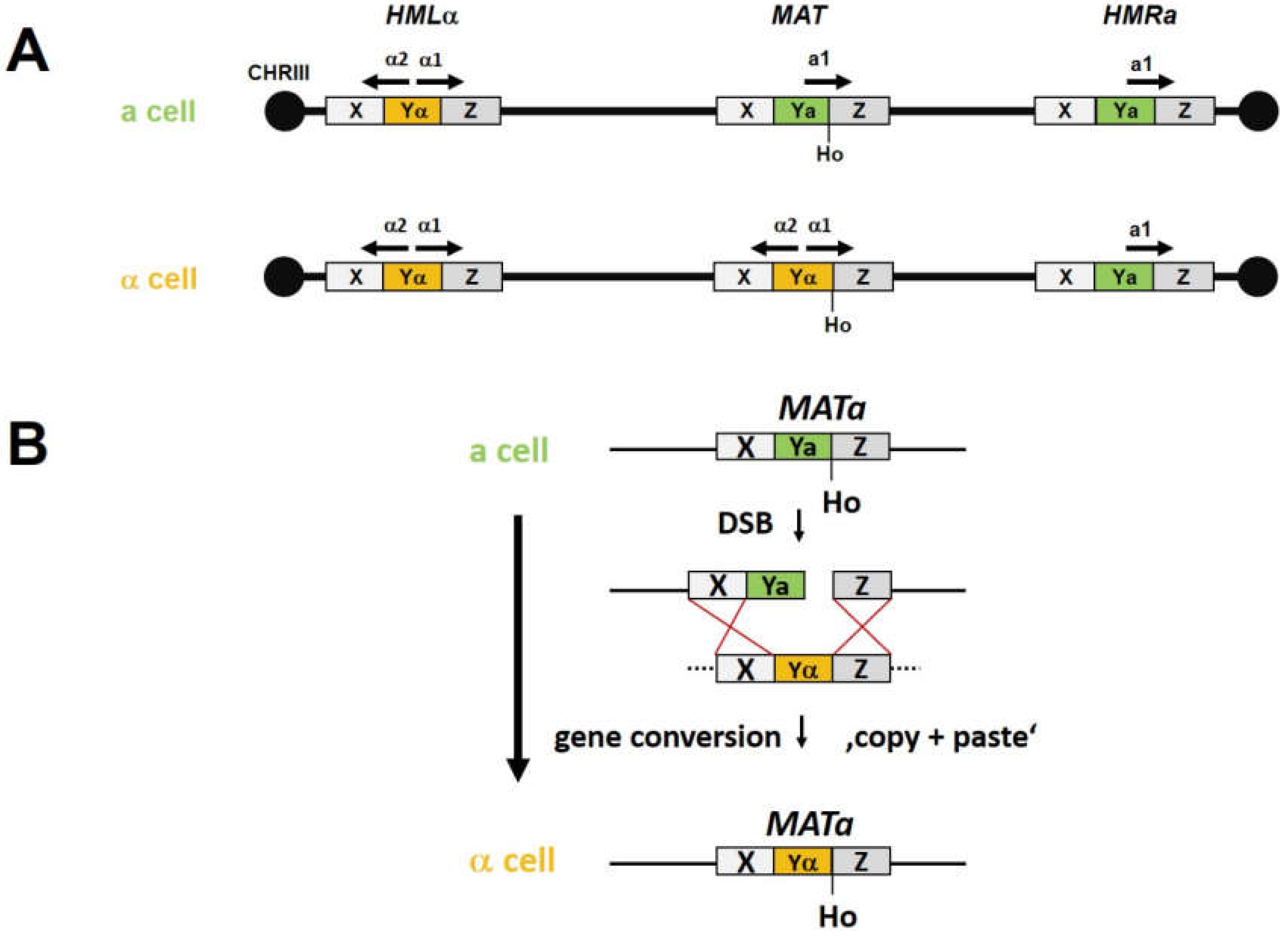
5. Genome Edits in Non-GM Yeasts
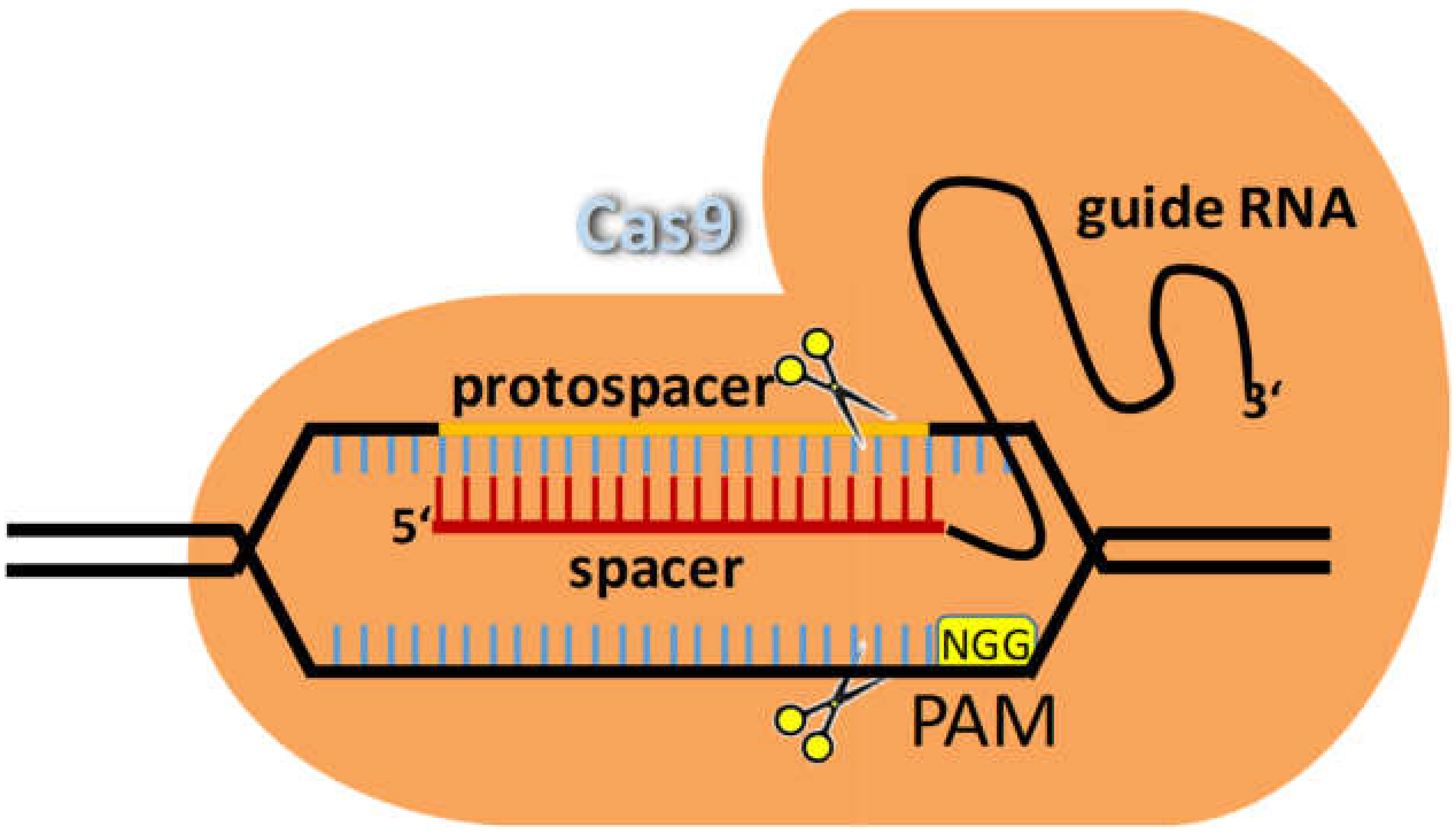
6. Genome Editing in the 21st Century
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Badr, A.; Muller, K.; Schafer-Pregl, R.; El Rabey, H.; Effgen, S.; Ibrahim, H.H.; Pozzi, C.; Rohde, W.; Salamini, F. On the origin and domestication history of barley (Hordeum vulgare). Mol. Biol. Evol. 2000, 17, 499–510. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, O.; Heun, M.; Notroff, J.; Schmidt, K.; Zarnkow, M. The role of cult and feasting in the emergence of Neolithic communities. Antiquity 2012, 86, 674–995. [Google Scholar] [CrossRef]
- Cavalieri, D.; McGovern, P.E.; Hartl, D.L.; Mortimer, R.; Polsinelli, M. Evidence for S. cerevisiae fermentation in ancient wine. J. Mol. Evol. 2003, 57, S226–S232. [Google Scholar] [CrossRef] [PubMed]
- Novo, M.; Bigey, F.; Beyne, E.; Galeote, V.; Gavory, F.; Mallet, S.; Cambon, B.; Legras, J.L.; Wincker, P.; Casaregola, S.; et al. Eukaryote-to-eukaryote gene transfer events revealed by the genome sequence of the wine yeast Saccharomyces cerevisiae EC1118. Proc. Natl. Acad. Sci. USA 2009, 106, 16333–16338. [Google Scholar] [CrossRef]
- Galeote, V.; Novo, M.; Salema-Oom, M.; Brion, C.; Valerio, E.; Goncalves, P.; Dequin, S. FSY1, a horizontally transferred gene in the Saccharomyces cerevisiae EC1118 wine yeast strain, encodes a high-affinity fructose/H+ symporter. Microbiology 2010, 156, 3754–3761. [Google Scholar] [CrossRef]
- Marsit, S.; Mena, A.; Bigey, F.; Sauvage, F.X.; Couloux, A.; Guy, J.; Legras, J.L.; Barrio, E.; Dequin, S.; Galeote, V. Evolutionary advantage conferred by an eukaryote-to-eukaryote gene transfer event in wine yeasts. Mol. Biol. Evol. 2015, 32, 1695–1707. [Google Scholar] [CrossRef]
- Dunn, B.; Richter, C.; Kvitek, D.J.; Pugh, T.; Sherlock, G. Analysis of the Saccharomyces cerevisiae pan-genome reveals a pool of copy number variants distributed in diverse yeast strains from differing industrial environments. Genome Res. 2012, 22, 908–924. [Google Scholar] [CrossRef]
- Will, J.L.; Kim, H.S.; Clarke, J.; Painter, J.C.; Fay, J.C.; Gasch, A.P. Incipient balancing selection through adaptive loss of aquaporins in natural Saccharomyces cerevisiae populations. PLoS Genet. 2010, 6, e1000893. [Google Scholar] [CrossRef]
- Goncalves, M.; Pontes, A.; Almeida, P.; Barbosa, R.; Serra, M.; Libkind, D.; Hutzler, M.; Goncalves, P.; Sampaio, J.P. Distinct domestication trajectories in top-fermenting beer yeasts and wine yeasts. Curr. Biol. 2016, 26, 2750–2761. [Google Scholar] [CrossRef]
- Goto-Yamamoto, N.; Kitano, K.; Shiki, K.; Yoshida, Y.; Suzuki, T.; Iwata, T.; Yamane, Y.; Hara, S. SSU1-R, a sulfite resistance gene of wine yeast, is an allele of SSU1 with a different upstream sequence. J. Ferment. Bioeng. 1998, 86, 427–433. [Google Scholar] [CrossRef]
- Perez-Ortin, J.E.; Querol, A.; Puig, S.; Barrio, E. Molecular characterization of a chromosomal rearrangement involved in the adaptive evolution of yeast strains. Genome Res. 2002, 12, 1533–1539. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, N.; Nakagawa, Y.; Hayakawa, M.; Iimura, Y. Distribution of the sulfite resistance gene ssu1-r and the variation in its promoter region in wine yeasts. J. Biosci. Bioeng. 2004, 98, 394–397. [Google Scholar] [CrossRef]
- Zimmer, A.; Durand, C.; Loira, N.; Durrens, P.; Sherman, D.J.; Marullo, P. Qtl dissection of lag phase in wine fermentation reveals a new translocation responsible for Saccharomyces cerevisiae adaptation to sulfite. PLoS ONE 2014, 9, e86298. [Google Scholar] [CrossRef] [PubMed]
- Adamo, G.M.; Lotti, M.; Tamas, M.J.; Brocca, S. Amplification of the CUP1 gene is associated with evolution of copper tolerance in Saccharomyces cerevisiae. Microbiology 2012, 158, 2325–2335. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Dominska, M.; Petrova, A.; Bagshaw, H.; Kokoska, R.J.; Petes, T.D. Properties of mitotic and meiotic recombination in the tandemly-repeated CUP1 gene cluster in the yeast Saccharomyces cerevisiae. Genetics 2017, 206, 785–800. [Google Scholar] [CrossRef] [PubMed]
- Fay, J.C.; McCullough, H.L.; Sniegowski, P.D.; Eisen, M.B. Population genetic variation in gene expression is associated with phenotypic variation in Saccharomyces cerevisiae. Genome Biol. 2004, 5, R26. [Google Scholar] [CrossRef]
- Borneman, A.R.; Forgan, A.H.; Kolouchova, R.; Fraser, J.A.; Schmidt, S.A. Whole genome comparison reveals high levels of inbreeding and strain redundancy across the spectrum of commercial wine strains of Saccharomyces cerevisiae. G3 2016, 6, 957–971. [Google Scholar] [CrossRef]
- Eldarov, M.A.; Kishkovskaia, S.A.; Tanaschuk, T.N.; Mardanov, A.V. Genomics and biochemistry of Saccharomyces cerevisiae wine yeast strains. Biochemistry 2016, 81, 1650–1668. [Google Scholar] [CrossRef]
- Hansen, E.C. Recherches sur la physiologie et la morphologie des ferments alcooliques V. Methodes pour obtenir des cultures pures de Saccharomyces et de mikroorganismes analogues. C. R. Trav. Lab. Carlsberg 1888, 2, 143–167. [Google Scholar]
- Walther, A.; Hesselbart, A.; Wendland, J. Genome sequence of Saccharomyces carlsbergensis, the world’s first pure culture lager yeast. G3 Genes Genomes Genet. 2014, 4, 783–793. [Google Scholar]
- Wendland, J. Lager yeast comes of age. Eukaryot. Cell 2014, 13, 1256–1265. [Google Scholar] [CrossRef] [PubMed]
- Matti, K.; Bernardi, B.; Brezine, S.; Semmler, H.; von Wallbrunn, C.; Rauhut, D.; Wendland, J. Characterization of old wine yeasts kept for decades under a zero-emission maintenance regime. Fermentation 2020, 6, 9. [Google Scholar] [CrossRef]
- Gonzalez, S.S.; Barrio, E.; Gafner, J.; Querol, A. Natural hybrids from Saccharomyces cerevisiae, Saccharomyces bayanus and Saccharomyces kudriavzevii in wine fermentations. FEMS Yeast Res. 2006, 6, 1221–1234. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, S.S.; Gallo, L.; Climent, M.A.; Barrio, E.; Querol, A. Enological characterization of natural hybrids from Saccharomyces cerevisiae and S. kudriavzevii. Int. J. Food Microbiol. 2007, 116, 11–18. [Google Scholar] [CrossRef]
- Gangl, H.; Batusic, M.; Tscheik, G.; Tiefenbrunner, W.; Hack, C.; Lopandic, K. Exceptional fermentation characteristics of natural hybrids from Saccharomyces cerevisiae and S. kudriavzevii. New Biotechnol. 2009, 25, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Perez-Torrado, R.; Barrio, E.; Querol, A. Alternative yeasts for winemaking: Saccharomyces non-cerevisiae and its hybrids. Crit. Rev. Food Sci. Nutr. 2018, 58, 1780–1790. [Google Scholar] [CrossRef]
- Langdon, Q.K.; Peris, D.; Baker, E.P.; Opulente, D.A.; Nguyen, H.V.; Bond, U.; Goncalves, P.; Sampaio, J.P.; Libkind, D.; Hittinger, C.T. Fermentation innovation through complex hybridization of wild and domesticated yeasts. Nat. Ecol. Evol. 2019, 3, 1576–1586. [Google Scholar] [CrossRef]
- Winge, Ø. On haplophase and diplophase of some Saccharomycetes. C. R. Trav. Lab. Carlsberg 1935, 21, 77–111. [Google Scholar]
- Botstein, D.; Fink, G.R. Yeast: An experimental organism for 21st century biology. Genetics 2011, 189, 695–704. [Google Scholar] [CrossRef]
- Nielsen, J.; Keasling, J.D. Engineering cellular metabolism. Cell 2016, 164, 1185–1197. [Google Scholar] [CrossRef]
- Garcia Sanchez, R.; Solodovnikova, N.; Wendland, J. Breeding of lager yeast with Saccharomyces cerevisiae improves stress resistance and fermentation performance. Yeast 2012, 29, 343–355. [Google Scholar] [CrossRef] [PubMed]
- Bellon, J.R.; Ford, C.M.; Borneman, A.R.; Chambers, P.J. A novel approach to isolating improved industrial interspecific wine yeasts using chromosomal mutations as potential markers for increased fitness. Front. Microbiol. 2018, 9, 1442. [Google Scholar] [CrossRef] [PubMed]
- Bellon, J.R.; Yang, F.; Day, M.P.; Inglis, D.L.; Chambers, P.J. Designing and creating Saccharomyces interspecific hybrids for improved, industry relevant, phenotypes. Appl. Microbiol. Biotechnol. 2015, 99, 8597–8609. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, F.; Krogerus, K.; Vidgren, V.; Sandell, M.; Gibson, B. Improved cider fermentation performance and quality with newly generated Saccharomyces cerevisiae × Saccharomyces eubayanus hybrids. J. Ind. Microbiol. Biotechnol. 2017, 44, 1203–1213. [Google Scholar] [CrossRef] [PubMed]
- Origone, A.C.; Rodriguez, M.E.; Oteiza, J.M.; Querol, A.; Lopes, C.A. Saccharomyces cerevisiae × Saccharomyces uvarum hybrids generated under different conditions share similar winemaking features. Yeast 2018, 35, 157–171. [Google Scholar] [CrossRef]
- Krogerus, K.; Magalhaes, F.; Vidgren, V.; Gibson, B. Novel brewing yeast hybrids: Creation and application. Appl. Microbiol. Biotechnol. 2017, 101, 65–78. [Google Scholar] [CrossRef]
- Chatterjee, N.; Walker, G.C. Mechanisms of DNA damage, repair, and mutagenesis. Env. Mol. Mutagen. 2017, 58, 235–263. [Google Scholar] [CrossRef]
- Zhao, L.; Chang, D.W.; Gong, Y.; Eng, C.; Wu, X. Measurement of DNA damage in peripheral blood by the γ-H2AX assay as predictor of colorectal cancer risk. DNA Repair 2017, 53, 24–30. [Google Scholar] [CrossRef]
- Boiteux, S.; Jinks-Robertson, S. DNA repair mechanisms and the bypass of DNA damage in Saccharomyces cerevisiae. Genetics 2013, 193, 1025–1064. [Google Scholar] [CrossRef]
- Finn, K.; Lowndes, N.F.; Grenon, M. Eukaryotic DNA damage checkpoint activation in response to double-strand breaks. Cell Mol. Life Sci. 2012, 69, 1447–1473. [Google Scholar] [CrossRef]
- Nogueira, A.; Fernandes, M.; Catarino, R.; Medeiros, R. Rad52 functions in homologous recombination and its importance on genomic integrity maintenance and cancer therapy. Cancers 2019, 11, 1622. [Google Scholar] [CrossRef] [PubMed]
- Biehs, R.; Steinlage, M.; Barton, O.; Juhasz, S.; Kunzel, J.; Spies, J.; Shibata, A.; Jeggo, P.A.; Lobrich, M. DNA double-strand break resection occurs during non-homologous end joininging but is distinct from resection during homologous recombination. Mol. Cell 2017, 65, 671–684. [Google Scholar] [CrossRef] [PubMed]
- Karanam, K.; Kafri, R.; Loewer, A.; Lahav, G. Quantitative live cell imaging reveals a gradual shift between DNA repair mechanisms and a maximal use of HR in mid S phase. Mol. Cell 2012, 47, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Saha, J.; Wang, S.Y.; Davis, A.J. Examining DNA double-strand break repair in a cell cycle-dependent manner. Methods Enzym. 2017, 591, 97–118. [Google Scholar]
- Thon, G.; Maki, T.; Haber, J.E.; Iwasaki, H. Mating-type switching by homology-directed recombinational repair: A matter of choice. Curr. Genet. 2019, 65, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Jasin, M.; Rothstein, R. Repair of strand breaks by homologous recombination. Cold Spring Harb. Perspect Biol. 2013, 5, a012740. [Google Scholar] [CrossRef]
- Haber, J.E. A life investigating pathways that repair broken chromosomes. Annu. Rev. Genet. 2016, 50, 1–28. [Google Scholar] [CrossRef]
- Herskowitz, I. Life cycle of the budding yeast Saccharomyces cerevisiae. Microbiol. Rev. 1988, 52, 536–553. [Google Scholar] [CrossRef]
- Sil, A.; Herskowitz, I. Identification of asymmetrically localized determinant, Ash1p, required for lineage-specific transcription of the yeast ho gene. Cell 1996, 84, 711–722. [Google Scholar] [CrossRef]
- Long, R.M.; Singer, R.H.; Meng, X.; Gonzalez, I.; Nasmyth, K.; Jansen, R.P. Mating type switching in yeast controlled by asymmetric localization of ASH1 mRNA. Science 1997, 277, 383–387. [Google Scholar] [CrossRef]
- Lee, C.S.; Haber, J.E. Mating-type gene switching in Saccharomyces cerevisiae. Microbiol. Spectr. 2015, 3, 491–514. [Google Scholar]
- Hinnen, A.; Hicks, J.B.; Fink, G.R. Transformation of yeast. Proc. Natl. Acad. Sci. USA 1978, 75, 1929–1933. [Google Scholar] [CrossRef]
- Goffeau, A.; Barrell, B.G.; Bussey, H.; Davis, R.W.; Dujon, B.; Feldmann, H.; Galibert, F.; Hoheisel, J.D.; Jacq, C.; Johnston, M.; et al. Life with 6000 genes. Science 1996, 274, 547–563. [Google Scholar] [CrossRef]
- Wach, A.; Brachat, A.; Pohlmann, R.; Philippsen, P. New heterologous modules for classical or pcr-based gene disruptions in Saccharomyces cerevisiae. Yeast 1994, 10, 1793–1808. [Google Scholar] [CrossRef] [PubMed]
- Giaever, G.; Chu, A.M.; Ni, L.; Connelly, C.; Riles, L.; Veronneau, S.; Dow, S.; Lucau-Danila, A.; Anderson, K.; Andre, B.; et al. Functional profiling of the Saccharomyces cerevisiae genome. Nature 2002, 418, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Bernardi, B.; Kayacan, Y.; Akan, M.; Wendland, J. Overexpression of RAD51 enables PCR-based gene targeting in lager yeast. Microorganisms 2019, 7, 192. [Google Scholar] [CrossRef]
- Wendland, J. PCR-based methods facilitate targeted gene manipulations and cloning procedures. Curr. Genet. 2003, 44, 115–123. [Google Scholar] [CrossRef]
- Gibson, D.G. Synthesis of DNA fragments in yeast by one-step assembly of overlapping oligonucleotides. Nucleic Acids Res. 2009, 37, 6984–6990. [Google Scholar] [CrossRef]
- Joska, T.M.; Mashruwala, A.; Boyd, J.M.; Belden, W.J. A universal cloning method based on yeast homologous recombination that is simple, efficient, and versatile. J. Microbiol. Methods 2014, 100, 46–51. [Google Scholar] [CrossRef]
- Van Leeuwen, J.; Andrews, B.; Boone, C.; Tan, G. Rapid and efficient plasmid construction by homologous recombination in yeast. Cold Spring Harb. Protoc. 2015, 2015, 085100. [Google Scholar] [CrossRef]
- Gibson, D.G.; Benders, G.A.; Axelrod, K.C.; Zaveri, J.; Algire, M.A.; Moodie, M.; Montague, M.G.; Venter, J.C.; Smith, H.O.; Hutchison, C.A., III. One-step assembly in yeast of 25 overlapping DNA fragments to form a complete synthetic Mycoplasma genitalium genome. Proc. Natl. Acad. Sci. USA 2008, 105, 20404–20409. [Google Scholar] [CrossRef]
- Juhas, M.; Ajioka, J.W. High molecular weight DNA assembly in vivo for synthetic biology applications. Crit. Rev. Biotechnol. 2017, 37, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Pretorius, I.S.; Boeke, J.D. Yeast 2.0—Connecting the dots in the construction of the world’s first functional synthetic eukaryotic genome. FEMS Yeast Res. 2018, 18, foy032. [Google Scholar] [CrossRef] [PubMed]
- Stovicek, V.; Borja, G.M.; Forster, J.; Borodina, I. Easyclone 2.0: Expanded toolkit of integrative vectors for stable gene expression in industrial Saccharomyces cerevisiae strains. J. Ind. Microbiol. Biotechnol. 2015, 42, 1519–1531. [Google Scholar] [CrossRef] [PubMed]
- Boeke, J.D.; Trueheart, J.; Natsoulis, G.; Fink, G.R. 5-fluoroorotic acid as a selective agent in yeast molecular genetics. Methods Enzym. 1987, 154, 164–175. [Google Scholar]
- Haase, M.A.B.; Truong, D.M.; Boeke, J.D. Superloser: A plasmid shuffling vector for saccharomyces cerevisiae with exceedingly low background. G3 Genes Genomes Genet. 2019, 9, 2699–2707. [Google Scholar] [CrossRef]
- Sauer, B. Recycling selectable markers in yeast. Biotechniques 1994, 16, 1086–1088. [Google Scholar]
- Jensen, N.B.; Strucko, T.; Kildegaard, K.R.; David, F.; Maury, J.; Mortensen, U.H.; Forster, J.; Nielsen, J.; Borodina, I. Easyclone: Method for iterative chromosomal integration of multiple genes in Saccharomyces cerevisiae. FEMS Yeast Res. 2014, 14, 238–248. [Google Scholar] [CrossRef]
- Di Carlo, J.E.; Norville, J.E.; Mali, P.; Rios, X.; Aach, J.; Church, G.M. Genome engineering in Saccharomyces cerevisiae using CRISPR-Cas systems. Nucleic Acids Res. 2013, 41, 4336–4343. [Google Scholar] [CrossRef]
- Horvath, P.; Barrangou, R. CRISPR/Cas, the immune system of bacteria and archaea. Science 2010, 327, 167–170. [Google Scholar] [CrossRef]
- Kirchner, M.; Schneider, S. CRISPR-Cas: From the bacterial adaptive immune system to a versatile tool for genome engineering. Angew. Chem. Int. Ed. Engl. 2015, 54, 13508–13514. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Doudna, J.A. CRISPR-Cas9 structures and mechanisms. Annu. Rev. Biophys. 2017, 46, 505–529. [Google Scholar] [CrossRef] [PubMed]
- Knott, G.J.; Doudna, J.A. CRISPR-Cas guides the future of genetic engineering. Science 2018, 361, 866–869. [Google Scholar] [CrossRef]
- Roy, K.R.; Smith, J.D.; Vonesch, S.C.; Lin, G.; Tu, C.S.; Lederer, A.R.; Chu, A.; Suresh, S.; Nguyen, M.; Horecka, J.; et al. Multiplexed precision genome editing with trackable genomic barcodes in yeast. Nat. Biotechnol. 2018, 36, 512–520. [Google Scholar] [CrossRef] [PubMed]
- McCarty, N.S.; Graham, A.E.; Studena, L.; Ledesma-Amaro, R. Multiplexed crispr technologies for gene editing and transcriptional regulation. Nat. Commun. 2020, 11, 1281. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Liang, L.; Choudhury, A.; Garst, A.D.; Eckert, C.A.; Oh, E.J.; Winkler, J.; Gill, R.T. Multiplex navigation of global regulatory networks (minr) in yeast for improved ethanol tolerance and production. Metab. Eng. 2019, 51, 50–58. [Google Scholar] [CrossRef]
- Zetsche, B.; Gootenberg, J.S.; Abudayyeh, O.O.; Slaymaker, I.M.; Makarova, K.S.; Essletzbichler, P.; Volz, S.E.; Joung, J.; van der Oost, J.; Regev, A.; et al. Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-cas system. Cell 2015, 163, 759–771. [Google Scholar] [CrossRef]
- Pretorius, I.S. Tasting the terroir of wine yeast innovation. FEMS Yeast Res. 2020, 20, foz084. [Google Scholar] [CrossRef]
- Kitagaki, H.; Kitamoto, K. Breeding research on sake yeasts in japan: History, recent technological advances, and future perspectives. Annu. Rev. Food Sci. Technol. 2013, 4, 215–235. [Google Scholar] [CrossRef]
- Fischer, S.; Buchner, K.R.; Becker, T. Induced expression of the alcohol acetyltransferase gene ATF1 in industrial yeast Saccharomyces pastorianus tum 34/70. Yeast 2018, 35, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Wang, Z.; Liu, N.; He, X.; Zhang, B. Genetic modification of industrial yeast strains to obtain controllable newflo flocculation property and lower diacetyl production. Biotechnol. Lett. 2008, 30, 2013–2018. [Google Scholar] [CrossRef] [PubMed]
- Kusunoki, K.; Ogata, T. Construction of self-cloning bottom-fermenting yeast with low vicinal diketone production by the homo-integration of ILV5. Yeast 2012, 29, 435–442. [Google Scholar] [CrossRef]
- Coulon, J.; Husnik, J.I.; Inglis, D.L.; van der Merwe, G.K.; Lonvaud, A.; Erasmus, D.J.; van Vuuren, H.J.J. Metabolic engineering of Saccharomyces cerevisiae to minimize the production of ethyl carbamate in wine. Am. J. Enol. Vitic. 2006, 57, 2. [Google Scholar]
- Dahabieh, M.S.; Husnik, J.I.; Van Vuuren, H.J. Functional enhancement of sake yeast strains to minimize the production of ethyl carbamate in sake wine. J. Appl. Microbiol. 2010, 109, 963–973. [Google Scholar] [CrossRef]
- Schuller, D.; Casal, M. The use of genetically modified Saccharomyces cerevisiae strains in the wine industry. Appl. Microbiol. Biotechnol. 2005, 68, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Husnik, J.I.; Volschenk, H.; Bauer, J.; Colavizza, D.; Luo, Z.; van Vuuren, H.J. Metabolic engineering of malolactic wine yeast. Metab. Eng. 2006, 8, 315–323. [Google Scholar] [CrossRef]
- Peris, D.; Perez-Torrado, R.; Hittinger, C.T.; Barrio, E.; Querol, A. On the origins and industrial applications of Saccharomyces cerevisiae × Saccharomyces kudriavzevii hybrids. Yeast 2018, 35, 51–69. [Google Scholar] [CrossRef]
- Steensels, J.; Snoek, T.; Meersman, E.; Picca Nicolino, M.; Voordeckers, K.; Verstrepen, K.J. Improving industrial yeast strains: Exploiting natural and artificial diversity. FEMS Microbiol. Rev. 2014, 38, 947–995. [Google Scholar] [CrossRef]
- Mans, R.; Daran, J.G.; Pronk, J.T. Under pressure: Evolutionary engineering of yeast strains for improved performance in fuels and chemicals production. Curr. Opin. Biotechnol. 2018, 50, 47–56. [Google Scholar] [CrossRef]
- Steensels, J.; Verstrepen, K.J. Taming wild yeast: Potential of conventional and nonconventional yeasts in industrial fermentations. Annu. Rev. Microbiol. 2014, 68, 61–80. [Google Scholar] [CrossRef] [PubMed]
- Varela, C.; Sengler, F.; Solomon, M.; Curtin, C. Volatile flavour profile of reduced alcohol wines fermented with the non-conventional yeast species Metschnikowia pulcherrima and Saccharomyces uvarum. Food Chem. 2016, 209, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Hazelwood, L.A.; Daran, J.M.; van Maris, A.J.; Pronk, J.T.; Dickinson, J.R. The ehrlich pathway for fusel alcohol production: A century of research on Saccharomyces cerevisiae metabolism. Appl. Env. Microbiol. 2008, 74, 2259–2266. [Google Scholar] [CrossRef] [PubMed]
- Cavalieri, D.; Casalone, E.; Bendoni, B.; Fia, G.; Polsinelli, M.; Barberio, C. Trifluoroleucine resistance and regulation of α-isopropyl malate synthase in Saccharomyces cerevisiae. Mol. Gen. Genet. 1999, 261, 152–160. [Google Scholar] [CrossRef]
- Abe, T.; Toyokawa, Y.; Sugimoto, Y.; Azuma, H.; Tsukahara, K.; Nasuno, R.; Watanabe, D.; Tsukahara, M.; Takagi, H. Characterization of a new Saccharomyces cerevisiae isolated from hibiscus flower and its mutant with L-leucine accumulation for awamori brewing. Front. Genet. 2019, 10, 490. [Google Scholar] [CrossRef]
- Cordente, A.G.; Solomon, M.; Schulkin, A.; Leigh Francis, I.; Barker, A.; Borneman, A.R.; Curtin, C.D. Novel wine yeast with ARO4 and TYR1 mutations that overproduce ‘floral’ aroma compounds 2-phenylethanol and 2-phenylethyl acetate. Appl. Microbiol. Biotechnol. 2018, 102, 5977–5988. [Google Scholar] [CrossRef]
- Bloom, B.J.; Nicholson, T.L.; Williams, J.R.; Campbell, S.L.; Bishof, M.; Zhang, X.; Zhang, W.; Bromley, S.L.; Ye, J. An optical lattice clock with accuracy and stability at the 10−18 level. Nature 2014, 506, 71–75. [Google Scholar] [CrossRef]
- Van Wyk, N.; Grossmann, M.; Wendland, J.; von Wallbrunn, C.; Pretorius, I.S. The whiff of wine yeast innovation: Strategies for enhancing aroma production by yeast during wine fermentation. J. Agric. Food Chem. 2019, 67, 13496–13505. [Google Scholar] [CrossRef]
- Valero, E.; Schuller, D.; Cambon, B.; Casal, M.; Dequin, S. Dissemination and survival of commercial wine yeast in the vineyard: A large-scale, three-years study. FEMS Yeast Res. 2005, 5, 959–969. [Google Scholar] [CrossRef]
- Jackson, C.A.; Castro, D.M.; Saldi, G.A.; Bonneau, R.; Gresham, D. Gene regulatory network reconstruction using single-cell RNA sequencing of barcoded genotypes in diverse environments. Elife 2020, 9, e51254. [Google Scholar] [CrossRef]
- Schmidt, S.A.; Kolouchova, R.; Forgan, A.H.; Borneman, A.R. Evaluation of saccharomyces cerevisiae wine yeast competitive fitness in enologically relevant environments by barcode sequencing. G3 Genes Genomes Genet. 2020, 10, 591–603. [Google Scholar] [CrossRef]
- Mitchell, L.A.; Wang, A.; Stracquadanio, G.; Kuang, Z.; Wang, X.; Yang, K.; Richardson, S.; Martin, J.A.; Zhao, Y.; Walker, R.; et al. Synthesis, debugging, and effects of synthetic chromosome consolidation: SynVI and beyond. Science 2017, 355, eaaf4831. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Li, B.Z.; Zhao, M.; Mitchell, L.A.; Xie, Z.X.; Lin, Q.H.; Wang, X.; Xiao, W.H.; Wang, Y.; Zhou, X.; et al. Bug mapping and fitness testing of chemically synthesized chromosome X. Science 2017, 355, eaaf4706. [Google Scholar] [CrossRef] [PubMed]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bernardi, B.; Wendland, J. Homologous Recombination: A GRAS Yeast Genome Editing Tool. Fermentation 2020, 6, 57. https://doi.org/10.3390/fermentation6020057
Bernardi B, Wendland J. Homologous Recombination: A GRAS Yeast Genome Editing Tool. Fermentation. 2020; 6(2):57. https://doi.org/10.3390/fermentation6020057
Chicago/Turabian StyleBernardi, Beatrice, and Jürgen Wendland. 2020. "Homologous Recombination: A GRAS Yeast Genome Editing Tool" Fermentation 6, no. 2: 57. https://doi.org/10.3390/fermentation6020057
APA StyleBernardi, B., & Wendland, J. (2020). Homologous Recombination: A GRAS Yeast Genome Editing Tool. Fermentation, 6(2), 57. https://doi.org/10.3390/fermentation6020057





