Abstract
Polyhydroxyalkanoates (PHA) are microbial biopolyesters utilized as “green plastics”. Their production under controlled conditions resorts to bioreactors operated in different modes. Because PHA biosynthesis constitutes a multiphase process, both feeding strategy and bioreactor operation mode need smart adaptation. Traditional PHA production setups based on batch, repeated batch, fed-batch or cyclic fed-batch processes are often limited in productivity, or display insufficient controllability of polyester composition. For highly diluted substrate streams like is the case of (agro) industrial waste streams, fed-batch enhanced by cell recycling has recently been reported as a viable tool to increase volumetric productivity. As an emerging trend, continuous fermentation processes in single-, two- and multi-stage setups are reported, which bring the kinetics of both microbial growth and PHA accumulation into agreement with process engineering and allow tailoring PHA’s molecular structure. Moreover, we currently witness an increasing number of CO2-based PHA production processes using cyanobacteria; these light-driven processes resort to photobioreactors similar to those used for microalgae cultivation and can be operated both discontinuously and continuously. This development is parallel to the emerging use of methane and syngas as abundantly available gaseous substrates, which also calls for bioreactor systems with optimized gas transfer. The review sheds light on the challenges of diverse PHA production processes in different bioreactor types and operational regimes using miscellaneous microbial production strains such as extremophilic Archaea, chemoheterotrophic eubacteria and phototrophic cyanobacteria. Particular emphasis is dedicated to the limitations and promises of different bioreactor–strain combinations and to efforts devoted to upscaling these processes to industrially relevant scales.
1. Introduction
The old definition “La fermentation, c’est la vie sans l’air” (“fermentation is life without air”), as Luis Pasteur once wrote, shows an old ambiguity in the terminus “fermentation” [1]. Originally used sensu stricto exclusively for anaerobic biotechnological processes like manufacturing of ethanol by yeasts, lactic acid by lactic acid bacteria, or acetone, butanol and ethanol (“ABE”) by Clostridia, the terminus “fermentation” is more and more in use also for strictly aerobic cultivations in bioreactors such as for oxygen-demanding production of acetic acid or yeast biomass, or, as the topic of this review, the production of polyhydroxyalkanoate (PHA) biopolyesters [2].
PHA, polyoxoesters of hydroxyalkanoates, are typical prokaryotic reserve materials; they are produced as intracellular products of the secondary metabolism of various Gram-negative or Gram-positive bacteria and several extremophilic Archaea. Here, a currently emerging trend is the isolation of novel halophilic microbial species of marine origin, which often display powerful PHA producers based on the conversion of inexpensive substrates [3,4,5,6]. Stored in the cell’s cytoplasm, spherical in shape and light-refractive, PHA granules consist of an hydrophobic core of coiled PHA chains and water acting as plasticizer; this core is covered by more hydrophilic enzymes and structural proteins, which, as not yet definitively clarified, may form a protein membrane. It is proposed by leading scientists in this field to describe this core–protein system as “carbonosomes”, in order to underline that PHA granules are more than simple polymer inclusions by far, but rather functional organelles-like inclusion bodies with various biological functions [7]. For the producing cells, PHA primarily serve as storage compounds for energy and carbon to better survive periods of starvation. However, other biological roles of PHA were elucidated during the last few years [8]. Besides their role for sporulation in Bacilli, their interaction with the nitrogen fixation metabolism and others, they exert a protective function for cells when exposed to hazardous environmental conditions [9], such as toxic solvents [10], oxidative stress [11,12], heat [12], hyper-salinity [13,14,15], freezing [16], or UV-irradiation [17]. From the application-oriented point of view, PHA attract attention as biodegradable packaging materials, e.g., in the food sector [18,19], for biomedical and pharmaceutical application [20,21,22,23], or, more recently, as materials for slow-carbon release to enhance bioremediation of contaminated environments [24].
Regarding the molecular architecture of PHA, these macromolecules are of helical structure, with the PHA-helix being stabilized by hydrogen bonds between the carbonyl groups of the individual monomers [25]. These monomers are hydroxyalkanoates (or, for some exotic cases, also hydroxyalkenoates) of three to five (“short chain length PHA”, scl-PHA; two to five when considering glycolate as PHA building block sensu stricto) or six and more (“medium chain length PHA”, mcl-PHA) carbon atoms. The best described PHA, namely the homopolyester poly(3-hydroxybutyrate) (PHB), is the most prominent representative of the scl-PHA group, which are materials characterized by rather high crystallinity, thermoplasticity and pronounced brittleness [26]. Crystallinity and brittleness of scl-PHA can be decreased by incorporation of additional monomers into the PHB matrix, e.g., 3-hydroxyvalerate (3HV) or 4-hydroxybutyrate (4HB); the resulting copolymers (recently also referred to as “bipolymers” [25]) and terpolymers display a broader “window of processibility”, which is defined as the difference between melting and decomposition temperature; this makes them more convenient for processing towards marketable products. In most cases, scl-PHA producing microbes convert simple carbon sources like sugars, alcohols or lipids via acetyl-CoA towards 3HB; production of 3HV or 4HB normally requires structurally related precursor substrates such as the 3HV-precursors propionic acid [27], valeric acid [28], levulinic acid [29,30,31], the cocktails of ozonolytically splitted fatty acids [32] or non-oxygenated polyethylene wax [33], or 4HB-precursors like γ–butyrolactone (GBL), 4HB sodium salt, or 1,4-butanediole [34,35]. Among extremophilic Archaea, a number of strains such as Haloferax mediterranei [36] or Halogeometricum borinquense [37] have been identified, which produce copolyesters of 3HB and 3HV (PHBHV) from simple, structurally unrelated substrates such as carbohydrates or glycerol, which in turn saves on precursor costs for copolyester synthesis. Among eubacteria, PHA copolyester production from unrelated substrates is a scarce feature and reported, e.g., for the Gram-positive strain Bacillus licheniformis PL26 [5] and the Gram-negative strain Hydrogenophaga pseudoflava [38]. Mcl-PHA, in most cases, are heteropolyesters of different randomly distributed hydroxyalkanotes with at least six carbon atoms. They are typically synthetized by Gram-negative bacteria from the genus Pseudomonas and, in contrast to scl-PHA, display low crystallinity and low glass transition temperature; macroscopically, they appear as highly elastic “bio-latexes” [39]. The generation of mcl-PHA homopolyesters such as poly(3-hydroxyhexanoate) (PHHx) or poly(3-hydroxyoctanoate (PHO) is mainly described by genetically engineered production strains [40]. Using an engineered Escherichia coli strain, Abe et al. described production of mcl-PHA with side chain length ranging from C3–C9, when starting from 2-alkenoic acids of six to twelve carbon atoms, and studied the impact of side chain length on thermal and crystallization properties of resulting biopolyesters. Melting temperatures (Tm) of mcl-PHA first decreased from 59 °C to 45 °C when the side chain changed from C3 to C4; with side chains up to C9, Tm increased again to 69 °C. This was explained by X-ray diffraction patterns, which indicated different crystallization behavior for mcl-PHA with side chains less than C3 and for those with side chains longer than C4 [41]. A new class of PHA are the so-called “quarterpolymers”, which consist of at least four different types of PHA monomers, among them both chiral and achiral scl-PHA monomers differing in both side chains and backbones and mcl-PHA building blocks. A recently described example is a quarterpolymer produced by Cupriavidus eutrophus strain B10646; this PHA consists of the chiral building blocks 3HB, 3HV (scl-PHA) and 3-hydroxyhexanoate (3HHx) (mcl-PHA) and the achiral scl-PHA building block 4HB [25]. Other researchers referred to such quarterpolymers as “quadripolymers” and designed them by genetic engineering of production strains, as shown for poly(glycolate-co-lactate-co-3HB-co-4HB) production by metabolic engineering of Escherichia coli [42]. In the context of lactic acid as PHA constituent, Taguchi et al. described for the first time the occurrence of this 2-hydroxyalkanoate in microbially produced PHA; in their study, these authors discovered that an engineered Pseudomonas sp. 61–3 PHA synthase accepts lactic acid-CoA as substrate for in vivo polymerization in LA-CoA producing transgenic E. coli cells, which harbor a CoA transferase gene in addition to said engineered PHA synthase [43]. Whether a strain produces scl- or mcl-PHA, respectively, depends on the type of PHA synthase active in the organism. While scl-PHA production is catalyzed by PHA synthases of Class I (prototype organism Cupriavidus necator) and, to a lesser extent, Class III synthases (prototype organism Allochromatium vinosum), mcl-PHA production resorts to Class II synthases (prototype organisms: Pseudomonas sp.). The first organism ever described as a PHA producer, namely Bacillus megaterium, possesses a PHA synthase differing from the others which, therefore, is often referred to as a Class IV synthase. Mixed scl-mcl heteropolymers can be biosynthetized either by strains with specialized Class III synthases, such as Thiocapsia pfeningii or Aeromonas caviae, or by means of genetic engineering, hence, by designing cells harboring both Class I and Class II synthase [44,45]. Figure 1 shows the general chemical structure of PHA and the most important PHA monomers discussed in this review.
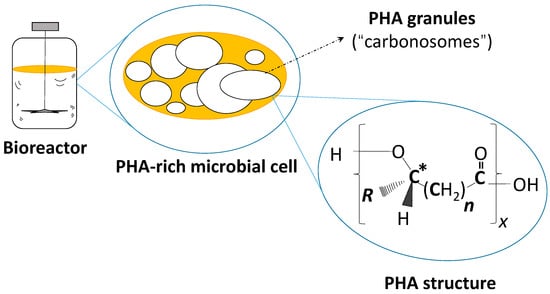
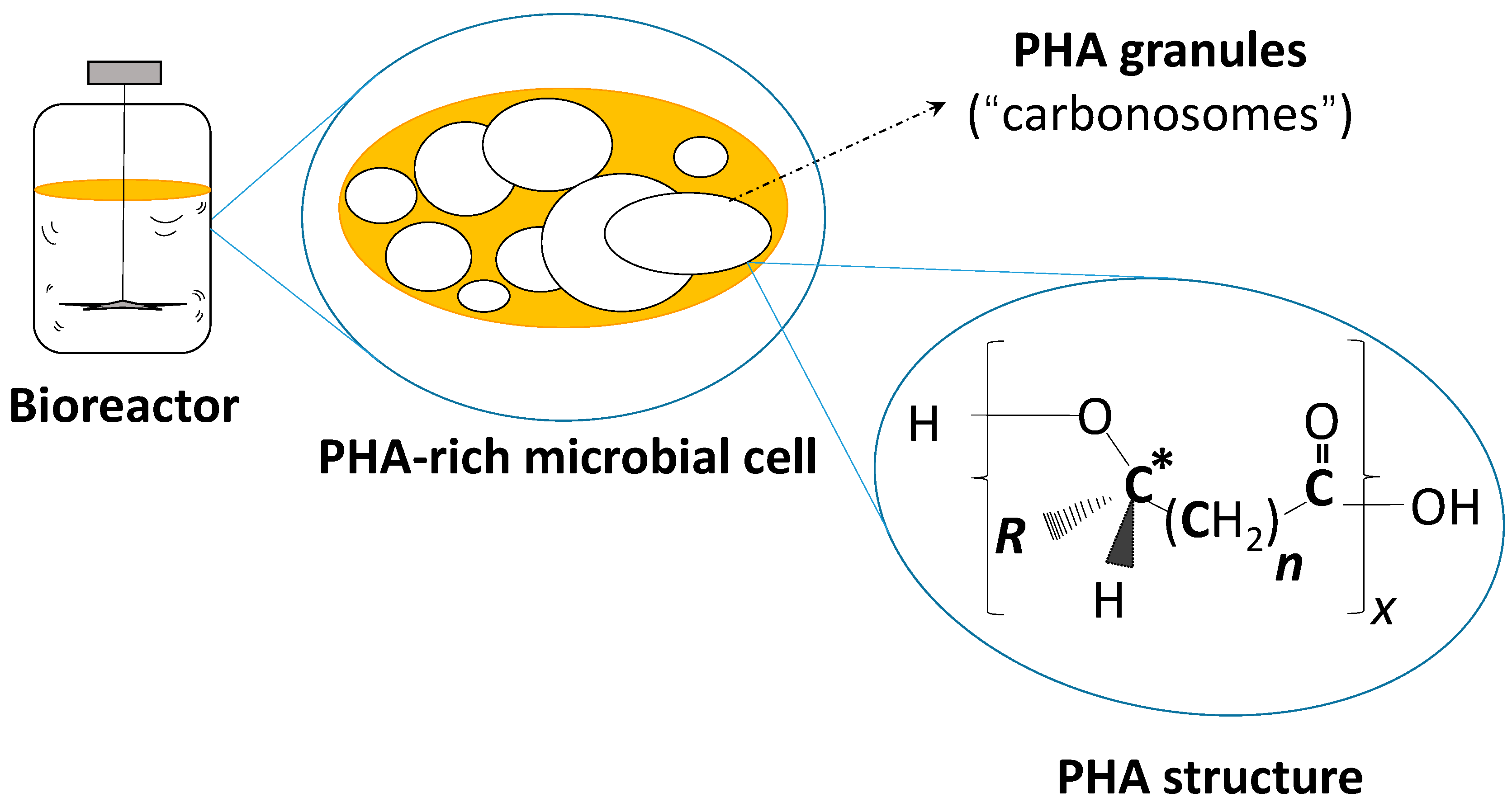
Figure 1.
Schematic of a PHA-rich microbial cell in a schematic bioreactor and the general chemical structure of PHA. X: Degree of polymerization (number of monomers); R: Side chain of monomers; n: Number of methylene groups in monomer´s backbone; *: Asymmetric carbon atom of most monomers (exception: 4HB). 3-hydroxybutyrate (3HB), R = CH3, n = 1, (scl-PHA); 3-hydroxyvalerate (3HV), R = C2H5, n = 1, (scl-PHA); 4-hydroxybutyrate (4HB), R = H, n = 2, (scl-PHA; achiral); 3-hydroxyhexanoate (3HHx), R = C3H7, n = 1, (mcl-PHA); 3-hydroxyoctanoate (3HB), R = C5H11, n = 1, (mcl-PHA).
2. Solid State Fermentation for PHA Production
It has to be stressed that an overwhelming majority of “fermentation” processes dedicated to PHA production is carried out in aqueous phase; solid-state fermentation (SSF) to generate PHA is, although described in the literature [46], still in its infancy as comprehensively summarized by Sindhu et al. [47]. In this context, SSF, which describes the cultivation of microbes on wet solid particles in substrate beds with a low amount of free water present between the solid particles, offers an option to use inexpensive cultivation media, e.g., those based on agro-industrial residues. This strategy provides an intriguing alternative both for waste disposal and for generation of value-added compounds under reduced capex costs if compared to submerse processes [48]. Moreover, for SSF-based products, manufacturing costs can be further reduced when fermented solids can be used directly, hence when downstream processing steps are not needed; in this context, PHA-containing fermented solids could directly be used for manufacturing of easily biodegradable composite materials. Furthermore, SSF provides several additional benefits over submerse fermentation such as higher substrate concentration resulting in smaller reactor volumes, or the easier feasibility of strong ventilation through the spaces between the solid particles [47].
In the case of SSF-based PHA production, Oliveira tested soya cake supplemented with sugarcane molasses for SSF with C. necator. These authors obtained 4.9 g of PHB per kg of solid medium after 60 h of incubation; the PHB content amounted to 0.39 g PHB per g cell dry mass (CDM), which surpassed the results obtained without supplementing with molasses by far [49]. Detailed material investigation of SSF-PHB showed that no major differences occur between PHB produced either via submerged fermentation or via SSF, except the observation of higher molar mass and a lower degree of crystallinity (Xc) for SSF-based biopolyesters [50]. In addition, Rocha et al. supplemented solid biodiesel waste with molasses and achieved better results for SSF-PHB production by C. necator if compared to cultivation on non-supplemented biodiesel waste; these authors report a PHB production of 2.1 g per kg solid substrate [51]. As a Gram-positive production strain, Bacillus sphaericus NII 0838 was tested by Ramadas and colleagues for PHB production on Jack fruit seed hydrolysate; these authors carried out the process using physical inert solid poly(urethane) foam (PUF) supports in order to facilitate the detachment of bacterial cells from the solid substrate after fermentation, which is a general problem of SSF processes. After optimization of the process parameters (inoculum size, nitrogen source concentration and pH-value), a PHB production of 0.17 g per g PUF support was reported under alkaline process conditions (pH-value 9), which was higher than parallel setups using Cassava bagasse or wheat bran as substrates [52].
However, according to Sindhu et al., the biggest obstacle to the market-ready production of PHA via SSF is the difficulty of keeping the process conditions constant, which explains that such processes are still of immature nature and need further development [47]. Table 1 provides an overview of reported SSF processes dedicated to PHA production.

Table 1.
Examples for PHA production based on SSF.
3. Kinetics of PHA Biosynthesis
Talking about secondary metabolites, it is noteworthy that, for most production strains, PHA biosynthesis is boosted under conditions limiting the propagation of active microbial cell mass. Typically, a profile illustrating the time curves for growth and PHA production consists of two easily distinguishable phases; under nutrient-rich conditions, the concentration of microbial biomass increases in accordance with the kinetics of an autocatalytic process until the running out of a growth-essential nutrient component, such as nitrogen source or phosphate. Now, the second cultivation phase is initiated, characterized by an almost constant concentration of catalytically active biomass (“residual biomass” is an often found expression in literature to express the PHA-free part of cells) and a linear increase of intracellular PHA according to 0-order reaction kinetics. In this second phase, PHA concentration increases until the external carbon source is depleted, or by steric factors determined by the cell geometry, which hinder further increase of the intracellular mass fraction of PHA granules [53]. This case is described as the so-called “non-growth associated PHA production”; however, this model does not reflect the total truth; besides some PHA producers that produce high amounts of PHA already under nutritionally balanced conditions (“growth-associated PHA production”), PHA-producers with typically clearly separated phases of biomass and PHA formation can also contain significant amounts of PHA even without nutrient limitation. An extreme situation is described for so-called “PHA hyper-accumulators” such as Pseudomonas 2F, which show extraordinary high PHA accumulation rates after a period of starvation and subsequent refeeding of exogenous carbon sources [54]. This makes it clear that an optimized PHA production process needs to be in accordance with the process kinetics, the feeding regime for the different nutrient components and the process engineering. Independent from the degree of growth association of PHA biosynthesis, the achievement of high cell density (high concentration of active biomass) is indispensable for obtaining a high volumetric PHA productivity as a crucial economic factor; therefore, both the biomass formation and PHA accumulation phase have to be optimized to increase overall productivity [53].
Technologically, PHA are produced under controlled conditions of pH-value, dissolved oxygen tension (pO2) and temperature in bioreactors (“fermenters”) of different sizes and types. The most common type is the cylindrical stirred tank reactor (STR), an apparatus well known from yeast, vinegar, or ethanol production. For PHA production, these STRs can be operated discontinuously (batch, repeated batch, fed-batch, repeated fed-batch) or continuously (CSTR). The entire PHA production process is multifaceted and needs optimization of each single process step, such as strain selection, strain engineering, bioreactor design, process regime, substrate selection, or downstream processing, with the process engineering being among the most decisive factors, especially in large-scale processes [55]. Particularly when inexpensive waste streams are applied as substrates for PHA production, upscaling to industrially relevant scales faces numerous challenges [56]. As a consequence, in order to reduce the number of cultivation experiments during process development and upscaling and to identify metabolic bottlenecks of the bioprocess in development, kinetic analysis and mathematical process modelling gain more and more attention in the PHA-related R&D [57,58]. The subsequent sections discuss in detail the theoretical background of discontinuous and continuous processes, describe the technological challenges in upscaling of different PHA production processes from lab- to large scale and provide successful examples from literature for these approaches. Figure 2 provides simplified schematics of the individual process regimes detailed in the review.
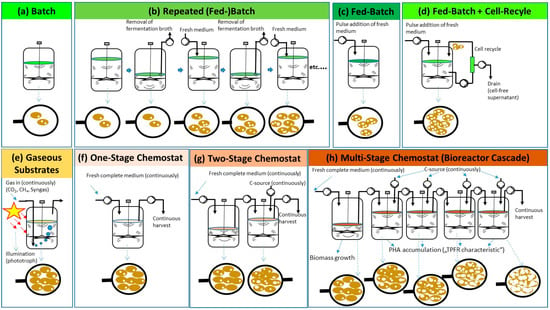
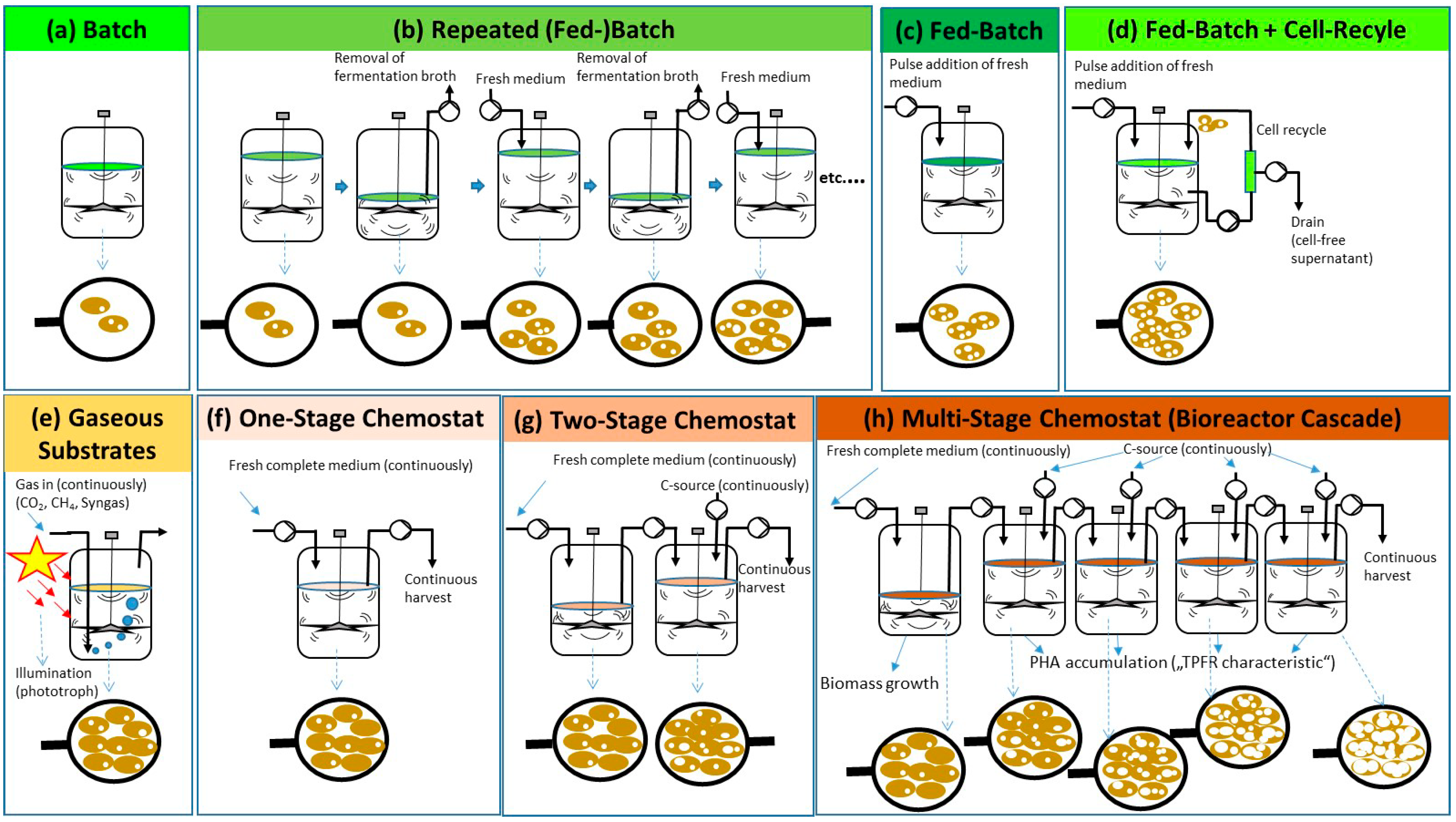
Figure 2.
Schematics of process regimes described for PHA biosynthesis in differently operated bioreactors: (a–d): discontinuous processes; (a) batch; (b) repeated (fed)-batch; (c) fed-batch; (d) fed-batch with cell recycle for biomass retention; (e) PHA production on gaseous substrates (CO2, CH4, syngas); illumination is indicated for photoautotrophic processes on CO2 (cyanobacteria) or syngas (Rhodospirillum rubrum); (f–h): continuous processes; (f) one-stage chemostat process, (g) two-stage chemostat process; (h) multi-stage continuous process in bioreactor cascade. Nota bene: in the illustration below, the bioreactor schemes symbolize microbial cells at the end of the individual processes, observed through symbolically indicated lenses; dependent on the individual process regimes, cells are of different density and of different PHA mass fractions.
4. Discontinuous PHA Production Processes
4.1. Batch Systems
Batch cultivations for PHA production are simple in their operation, but intrinsically of low productivity; the maximum allowed concentration of nitrogen and carbon source at the beginning of the fermentation batch is restricted by physiological preconditions of the production strain; typically, concentration of nitrogen sources at the beginning of PHA production processes amounts to about 2–3 g/L ammonia sulfate for the most important production strains and carbon source concentrations to about 10–30 g/L for simple substrates like glucose, sucrose, or glycerol. In the case of mcl-PHA, for related substrates like fatty acids, inhibiting concentrations often are even below 1 g/L, which drastically conflicts with batch production strategies; the same is valid for 3HV precursor compounds in the case of scl-PHA production [28]. Moreover, the carbon source submitted at the beginning has to be in excess of the available growth-limiting substrate and undergoes, analogous to other PHA-production processes, conversion towards biomass, PHA, CO2 and minor metabolites, thus resulting in a low overall conversion yield of carbon source towards PHA. Considering a theoretical conversion yield below 0.48 g/g towards biomass and PHA, respectively, one can easily imagine that such processes are far away from being economically feasible. As examples reported in literature, high PHA fractions in biomass were obtained in batch processes for cultivation of Chelatococcus daeguensis TAD1 on glycerol (0.81 g/g) [59], Halomonas campisalis on maltose (up to 0.81 g/g) [60], C. necator H16 on jatropha oil (0.9 g/g) [61], or Bacillus firmus NI 0830 on rice straw hydrolysate (0.89 g/g) [62]; however, volumetric productivity of these batch processes was not economically feasible, amounting to only 0.01 (C. daeguensis TAD1), 0.03 (H. campisalis), 0.17 (C. necator), or 0.02 (B. firmus) g/(L h).
Only recently, Gahlawat and colleagues demonstrated a simple “repeated batch” approach to enhance the volumetric productivity of PHB biosynthesis by Azohydromonas australica DSM 1124, a strain formerly known as Alcaligenes latus. This “drain-and-fill” strategy comprised the batching of the culture in a 7 L stirred tank bioreactor until two thirds of the carbon source sucrose and all nitrogen sources (ammonia sulfate) were depleted; at this point, cells were in a stationary growth phase. During this batch phase, which lasted for 36 h, 8.71 g/L CDM and 6.24 g/L PHB were produced, which corresponds to a volumetric productivity of 0.17 g/(L h). Now, 20% of the fermentation broth was removed (“drain”) and replaced by the same volume of fresh cultivation medium, which was composed the same way as the batch medium and contained also nitrogen sources because PHA biosynthesis by this organism occurs in a growth-associated manner. Each time the sucrose concentration dropped below 8 g/L, a replacement of 20% of the fermentation broth was carried out. Whereas the first repeated batch cycle took 21 h and resulted in a final PHB concentration of about 15 g/L, the second and third repeated batch cycle lasted only 12 and 9 h, respectively and generated about 15 g/L and 19 g/L PHB, respectively. After the entire process (69 h), about 27.9 g/L CDM and 20.6 g/L PHB were obtained; volumetric productivity for PHB was highest with 0.31 g/(L h) for the first repeated batch cycle between 36 and 48 h; after this cycle, also the highest intracellular PHB fraction was observed with 0.82 g/g PHB in CDM; the second and third repeated batch cycle displays decreased metabolic activity compared to the first, but still performed superiorly to the batch phase. The entire process achieved a PHB productivity of 0.30 g/(L h). The authors emphasized that not only the increase of volumetric productivity can be considered as a major advantage of this strategy over simple batch processes; moreover, this approach saves non-productive (“dead”) time for revamping of the bioreactor normally needed between individual batches [63].
At the moment, we are witnessing increasing efforts to optimize the application of mixed microbial consortia (MMC) for PHA biosynthesis as an inexpensive alternative to processes based on pure, monoseptic cultures. For such MMC-based processes, selective environmental pressure is exerted to a natural microbial consortium by employing suitable cultivation conditions [64,65,66,67,68,69,70,71]. As PHA are storage polymers naturally accumulated under dynamic environmental conditions, selective pressure to boost PHA biosynthesis can be achieved via so-called “cyclic feast-and-famine feeding regimes”, which consist of a series of alternating availability (feast) and absence of nutrients (famine) [71,72]. In the majority of cases, such systems are also operated in repeated batch cultivation mode [73]. Using MMC, PHA can be produced at high productivity and low costs due to minor sterility demands, reduced requirements for the equipment and control devices, and, most of all, MMC can utilize inexpensive, complex feedstocks such as carbonaceous domestic or industrial effluents [65,72,74,75]. Auspicious yields and specific productivity were described for MMC processes based on either defined substrates like acetate [71], or inexpensive, complex compounds from wastewater [76], olive oil mill effluents [75], sugar cane molasses [77], food waste [78], crude glycerol phase (CGP) [79], or even toxic phenolics [80]. These complex substrates can either be used directly for PHA production, or they have to be fermented in a first, anaerobic step towards volatile fatty acids (VFAs). Furthermore, continuous feeding strategies enable PHA production with the additional effects of a more stable process and altered copolymer composition [65]. However, it should be noted that the MMC process for PHA production have fundamental obstacles because the obtained biopolyesters are either blends of copolymers and/or homopolymers; hence, the polymer microstructure cannot be properly regulated, which hampers the production of PHA with on demand quality by this approach.
4.2. Fed-Batch Systems
4.2.1. General Aspects of Fed-Batch Processes for PHA Production
When running a fed-batch culture, substrate is added via substrate pulses when its concentration drops below a critical value without removing the culture fluid. In the PHA case, both nitrogen and carbon sources can be refed in periodic intervals according to the consumption by the biomass, until a required concentration of active, PHA-poor biomass is reached. For nitrogen feeding during the growth phase, one can resort to coupling the nitrogen feed to the change of the pH-value during the process; this is possible by profiting from the fact that biomass growth is directly proportional to a decline of pH-value. When using a solution of the inorganic nitrogen source ammonia as alkaline pH-regulating agent, both nitrogen level and pH-value are kept constant (“pH-stat”) without the need for excessive analytical control of the nitrogen source level [81,82,83]. To initiate the switch from growth phase to PHA production phase, nitrogen concentration is allowed to drop to zero by exchanging the ammonia solution by a NaOH solution as alkaline pH-corrective agent [83]. Now, carbon sources are fed by adding substrate pulses until the end of the process; the approaching process end may be heralded by a slowing down of the specific PHA production rate. Typical fed-batch PHA production setups resorting to pulses of different carbon sources as a response to substrate depletion are reported for lab-scale bioreactor processes for diverse strain/substrate combinations such as C. necator/soybean oil [83], Burkholderia sacchari/sucrose [34], or Hfx. mediterranei/CGP [84]; all of these processes generated scl-PHA. Similar fed-batch processes for mcl-PHA production were described, e.g., by Muhr and colleagues, who tested different combinations of Pseudomonades (Pseudomonas citronellolis and Pseudomonas chlororaphis) and low-quality biodiesel made of animal waste lipids; due to difficulties in rapid biodiesel determination, substrate feed pulses in these setups were accomplished as a response to increasing signals of the oxygen probe [85,86]. A different fed-batch approach was chosen by Follonier and colleagues, who supplied the carbon source (50/50 mixture of octanoic acid and 10-undecenoic acid) permanently in a linear feeding rate during the PHA accumulation phase of Pseudomonas putida KT2440. Linear substrate feeding matches the 0-order kinetics of PHA accumulation (linear increase of PHA concentration with time) well under conditions of constant concentration of residual biomass. This strategy avoids excessive concentrations of these substrates, which would occur after pulse feeding. Here, it should be noted that substrates like the carboxylic acids used in Follonier’s study on the one hand are needed to produce mcl-PHA of tailored composition, but, on the other hand, are toxic for the production strains already at low concentrations. In addition, these substrates are quite costly; this problem can at least partly be solved by running the biomass growth phase with an alternative, inexpensive substrate. In the case of the study by Follonier et al., this was accomplished by cultivating the bacteria in a first batch phase on the extract of “Gewürztraminer” grape pomace, a waste product of winemaking. Switching from the batch phase on pomace extract to linear feeding of the mcl-PHA precursors was initiated after surmounting defined peak signals for pO2 in fermentation broth and CO2 in the exhaust gas stream. The obtained mcl-PHA consisted of 53 mol-% saturated (deriving from octanoic acid) and 47% unsaturated (from 10-undecenoic acid) monomers, with a mass fraction of PHA in CDM of about 0.41 g/g. From the material point of view, the product was completely amorphous (no melting endotherm observed by thermoanalysis), with an outstandingly low glass transition temperature below −45 °C [87].
In addition, carbon source feeding in fed-batch processes can be automatized by coupling the feed pulse additions to the typical increase of pH-value provoked by carbohydrate depletion; this was demonstrated by Ahn and colleagues, who carried out pH-stat fed-batch cultivation for PHB production using recombinant E. coli as production strain and bovine whey powder solution as carbon source; both nitrogen and carbon feed were automatized as reactions to changing pH-value [81]. Also in the case of MMC applied for PHA production, substrate supply is often done by the above described aerobic dynamic feast/famine feeding strategy in a pulse-wise substrate addition [77].
In the ideal case, a fed-batch process is stopped by the operator at a current carbon concentration of almost zero in order to avoid unnecessary substrate loss, but well before intracellular PHA degradation starts. Knowing the ideal time point to refeed substrates requires a permanent control of the current substrate level. Here, one either resorts to periodic sampling and ex situ substrate analysis, or to modern online sensors enabling in situ substrate control; in the PHA case, such analytical online systems are described for the use of glucose or methanol as substrates. However, more advanced systems resort to the coupling of carbon source feeding to the actual concentration of dissolved oxygen in fermentation broth, or the CO2 level in the gas stream leaving the bioreactor; a lack of carbon source results in increased pO2 levels due to reduced metabolic activity and decreased CO2 levels in exhaust gas due to decreased aerobic respiration [88,89].
Not only for fed-batch processes, but for PHA production in general, tools of mathematical modelling, encompassing low structured (formal kinetic), high structured (metabolic), cybernetic, dynamic, or hybrid-type models, are gaining importance by the scientific community due to their potential to accelerate the upscaling of lab-experiments to pilot- and industrial scale, to clarify preferred metabolic pathways for strain/substrate combinations, to identify metabolic bottlenecks and to reduce the required number of lab-experiments during process development; a comprehensive overview of mathematical modelling approaches in the PHA arena was recently provided by Novak and colleagues [57]. For fed-batch processes, a highly predictive, integrated dynamic model describing PHB production by Azohydromonas lata, accounting for bacterial growth, PHB synthesis and degradation, substrate utilization, cell respiration and molecular mass of PHB was recently developed and validated by Penloglou and associates. Model validation was done by comparison with control experiments and delivered optimization protocols for enhancing productivity and polymer properties. An optimized cultivation regime resulted in a maximum PHB mass fraction in CDM of 0.94 g/g and a volumetric PHB productivity of 4.2 g/(L h). In addition, the model suggested that different PHB qualities with weight average molecular weights (Mw) of more than 1500 kg/mol could be achieved by selecting the optimal operating conditions for the fed-batch process [90].
4.2.2. Fed-Batch Processes with Cell Recycling for Biomass Retention
As the major drawback of fed-batch cultivation systems, the addition of feeding solution increases the fermentation volume, causing a concomitant dilution of the fermentation broth. In order to keep this dilution as low as possible, highly concentrated feed solutions containing several hundred grams of carbon source per liter are used; classical examples are glucose feeding solutions containing 500 g per liter. In the case of (agricultural) waste streams used as carbon sources, the substrate concentration is often considerably lower; e.g., whey from dairy industry, a convenient carbon source for many PHA producers, contains only between 40 and 50 g/L of carbon source (lactose) [36]. Such low substrate contents make a direct use technologically complicated due to the rapid volume increase and the too low cell-densities accessible by the fermentations. A potential way out is to concentrate the substrate by evaporation (e.g., using falling film evaporators like usual in milk industry) or membrane processes, e.g., based on ultrafiltration membrane modules. This is a typical strategy to separate carbohydrate-rich whey permeate from the low concentrated raw material whey. The permeate fraction, a well storable substrate to be used inter alia for PHA production, is about five-fold concentrated if compared to the raw substrate whey and contains more than 200 g/L lactose. Unfortunately, this value is already close to lactose solubility in water; further concentration is only possible after prior hydrolysis of lactose into its monomers glucose and galactose, which requires an additional upstream processing step [36].
In any case, described substrate concentration processes are energy and time demanding. Furthermore, toxic molecules of low molecular mass, e.g., hydroxymethylfurfural, furfural, carboxylic acids, aldehydes, etc., which are formed during thermal processing, can be co-concentrated in parallel to the desired substrate and negatively affect the fermentation performance of the production strain. This effect is especially obvious in the case of lignocellulose hydrolysates; lignocellulosics display the most abundantly organic carbon sources available for biotechnology, with their hydrolyzates typically containing in the range of about 40 g/L convertible carbon sources [91]. However, the removal of toxins generated during hydrolysis requires efficient and inexpensive solutions for a broad implementation of these materials as substrates for PHA production [92]. An alternative strategy is to operate the bioprocess in a cell-recycling mode by coupling a membrane module directly with the bioreactor. In the past, such cell-recycle approaches to enhance PHA production have been described by different authorships. Ahn et al. used highly concentrated whey powder solution for PHA production by the recombinant E. coli strain CGSC 4401; this strain harbored the plasmid pJC4 containing the PHA biosynthesis gene from the well-known PHA producer A. latus (today: A. lata). In the first stage, a classical fed-batch fermentation using a carbon source feed of 210 g/L lactose was carried out in a 6.6-L bioreactor. Here, the nitrogen source feed was coupled to the pH-value increase during the growth phase and, after cell division stops, the lactose feed was performed as a response to the pH-electrode signal (increase of pH-value by depletion of lactose); after reaching a pre-defined volume of fermentation broth, the cell recycle process using a 280 g/L lactose feed was started. Prior to each substrate feed, fermentation broth was pumped by peristaltic pumps through an external crossflow type membrane of poly(sulfone) hollow fibers with a molecular weight cut-off (MWCO) of 500 kDa in order to recycle the bacterial cells and to keep the volume of the culture constant. During cell-recycle operation, the flow rate for cell-recycling was maintained at 350 mL/min, whereas the rate of cell-free permeate removal was increased from 10 mL/min to 50 mL/min. This way, the authors were able to reach a CDM of almost 200 g/L with an intracellular PHB mass fraction of 0.87 g/g and an outstanding volumetric productivity of 4.6 g/(L h), which, according to the available literature, is still the highest value for volumetric PHA productivity [82]. Ienczak et al. reported a repeated fed-batch process with cell-recycling for PHB production by C. necator on a 50/50 mixture glucose/fructose. As a primary aim, high cell density was aspired by these researchers by a repeated batch approach during growth phase with balanced medium (urea as nitrogen source); here, the first batch lasted 22 h, the second 3 h. After the first batch, 15 g/L residual cell mass was reached and 25% of the nutrient-poor fermentation supernatant was withdrawn, cells were recycled and withdrawn supernatant replaced by a fresh concentrated balanced medium. For PHA accumulation, six repeated fed-batch cycles were carried out by replacing nutrient-poor supernatant by nitrogen-free feeding solution containing 90 g/L sugar. In this setup, cell recycling was carried out using two external parallel cross-flow type poly(propylene) membranes, operated at flow rates of 60 L/h. With this repeated fed-batch strategy, the authors reached a CDM of 61.6 g/L with an intracellular PHB mass fraction of 0.69 g/g, which corresponds to a volumetric productivity of 1.0 g/(L h) [93].
A similar approach was presented by Haas and colleagues, who used an external microfiltration membrane unit for cell recycling in C. necator DSM 545 cultivation setups. After a batch phase in a 5 L STR, a synthetic nitrogen-free nutritional medium containing glucose as a sole carbon source at rather low concentration (50 g/L) was fed into the bioreactor at a rate of 720 g/h as a response to the oxygen consumption by the cells; in parallel to re-feed, cell-recycle occurred at the same rate, almost removing the glucose-free supernatant and returning active cells to the bioreactor. By running this approach, a high density of PHB-rich biomass and outstanding volumetric PHB productivity, namely 148 g/L CDM, 0.76 g/g PHB in CDM and 3.1 g/(L h), were achieved. The conversion yield of glucose to PHB amounted to 0.33 g/g, which is in agreement with most reported batch and fed-batch studies using this strain/substrate combination [94].
Lorantfy and colleagues used a similar cell retention system by installing a microfiltration unit for high productive biosynthesis of PHA by Hfx. mediterranei in a corrosion resistant lab-scale bioreactor containing highly saline cultivation medium (156 g/L NaCl); the authors reported a ten-fold increase of volumetric productivity for the cell recycle system if compared to a continuous chemostat culture. Most importantly, this work demonstrates that, at lower cross flow rates, higher extracellular protein concentrations occur due to oxygen limitation, whereas at higher cross flow rates, excessive shear stress caused cell burst, as monitored by detected DNA fragments. Hence, the work can be considered pioneering for optimization of process regimes for extremophilic microbes in terms of robustness and scalability [95].
4.2.3. Repeated Fed-Batch for PHA Production
As another extremophile, the thermophilic PHA producer Chelatococcus sp. MW10 was isolated by screening experiments carried out by Ibrahim and colleagues. In order to obtain high biomass concentrations and high volumetric PHA productivity, the organism was cultured using different advanced cultivation strategies. First, on a 2 L stirred tank bioreactor scale, classical fed-batch fermentation processes were carried out, where glucose as the sole carbon source was added via pulse feeding in order to hold its concentration above 20 g/L. The authors argue that the excess glucose feeding is not only to increase both residual biomass and PHB concentration, but also to avoid intracellular PHA degradation. By this fed-batch strategy, a maximum CDM concentration of 5.2 ± 0.6 g/L was reached after 53 h of cultivation; at that time, 2.9 ± 0.7 g/L PHB were obtained as the maximum product concentration. The authors underlined that, despite the stable substrate availability, the intracellular PHB fraction steadily declined during the cultivation. To handle this deficiency, a repeated batch fermentation (in the article: “cyclic batch fermentation, CBF”) strategy was conceived; this CBF was tested in a 42 L stirred tank bioreactor at an elevated temperature of 50 °C; the glucose concentration amounted to 50 g/L. Based on the optimum results obtained by the fed batch cultivations on a 2 L scale, a cycling time of 50 h was chosen. The starting volume for the CBF was 25 L. In this process, pO2 was kept constant at 20% of air saturation (triggered by adjusting the aeration rate and the stirrer speed). For the first cycle, which lasted 50 h, a high maximum specific growth rate of μmax. = 0.125 1/h and a CDM concentration of up to 12.7 ± 0.9 g/L were obtained; these results were considerably higher than those obtained by the simple fed-batch process. However, the intracellular PHB fraction (55 ± 6 wt.-%) did not outperform the fed-batch cultivation results. After this first cultivation cycle, 23 L of the cultivation broth were removed from the bioreactor and replaced by 23 L of fresh, non-sterilized minimal medium to start this second cycle with a strong inoculum density. A similar CDM concentration like in cycle 1 (about 11 g/L) was obtained in cycle 2; despite the permanent glucose availability, the intracellular PHB fraction dropped to 38 ± 6 wt.-%. A third cycle was carried out analogous to cycle 2; here, the PHB fraction in CDM further dropped to 32 ± 3 wt.-%.
As an alternative strategy, an adjusted semi-continuous cultivation approach, namely a repeated fed-batch (in article: “cyclic fed-batch fermentation, CFBF”), was investigated; a 42 L stirred tank bioreactor was used for this purpose. CFBF is characterized by partially withdrawing the culture broth and subsequently refilling the bioreactor with fresh medium. CFBF prevents an increase in concentrations of toxic by-products and avoids a volume increase typical for fed-batch processes. In the Chelatococcus sp. MW10 cultivation, different volumes (20 to 40%) of a cultivation broth volume were replaced by fresh medium at irregular intervals. The CFBF process was started in batch mode with 30 g/L of glucose. Fresh medium was supplied for the first time after 21 h. After 44 h, when μ was relatively high with 0.070 1/h and no intracellular PHB degradation was noticed yet, 5 L of fermentation broth were replaced by fresh medium. After this first cycling, feeding was performed continuously and subsequent cycles of medium replacement were done as a response to undue volume increase in the vessel and drop of pO2 caused by high biomass concentration. In addition, 10 L of culture volume were removed and replaced by only 5 L of fresh medium in order to prevent extreme dilution of the cultivation broth. The highest intracellular PHB mass fractions (more than 50 wt.-%) were obtained in the second cycle between 82 h and 143 h. Here, a biomass concentration of 43 ± 1 g/L and a PHB fraction in CDM of 17 ± 4 g/L was obtained. After running the third cycle for 14 h, a final addition of 5 L of fresh medium was carried out. Generally, biomass growth significantly increased during the third cycle, in which CDM reached a high value of 115 ± 4 g/L. Despite the rather low intracellular PHB fraction of 12 ± 4 wt.-% at the end of this process, high volumetric PHB productivity for the entire process of 14 ± 5 g/L was reported [96].
4.3. “Continuous Fed-Batch” Systems
4.3.1. Use of Liquid Substrates
A special approach for producing PHA in “continuous fed-batch” was reported by Du and Yu, who coupled PHA production in a 1.6 L airlift-type bubble column bioreactor, as the system operated without mechanical stirring, to an acidogenic, anaerobic bioreactor. In the anaerobic bioreactor, food waste was digested by an MMC, yielding a cocktail of organic acids, predominately acetate, propionate, butyrate and lactate. Technologically, the acidic sludge from the anaerobic bioreactor was recycled by a peristaltic pump through a tubular membrane module; this module was immersed in the fermentation broth of the aerobically operated bubble column airlift reactor and enabled permeation of low molecular mass compounds into the culture broth in the bubble column, but retained biomass. Due to the fact that substrates were supplied permanently in this study as a response to the concentration gradient caused by consumption by the cells, but neither cells nor fermentation broth were removed from the bioreactor, it is suggested to call such systems “continuous fed-batch”. In the case of Du and Yu, two different membrane types were used for molecule diffusion into the bubble column, in which PHA synthesis took place under aerobic conditions by C. necator ATCC 17699. Using a silicon rubber membrane, butyrate and acetate passed through and resulted in accumulation of PHB homopolyester; CDM production amounted to 11.3 g/L, with a PHB mass fraction of about 0.6 g/g. Using a dialysis membrane enhanced the mass transfer of acids and also enabled the passing through of lactate and propionate, which act as precursors not only for 3HB, but also for 3HV biosynthesis. In addition, 22.7 g/L CDM with about 0.73 g PHBHV copolyester containing 0.028 mol/mol 3HV were obtained in this dialysis membrane setup [97].
4.3.2. Use of Gaseous Substrates CH4, CO2 and Syngas
“Continuous fed-batch” processes for PHA biosynthesis are also described in the case of using gaseous carbon sources, namely CH4 (methanotroph production strains) and CO2 (autotrophs like cyanobacteria or “Knallgas-bacteria”). Here, substrate availability is limited by the solubility of CH4 or CO2, respectively, in the aqueous fermentation broth; this solubility of substrates and substrate availability for the cells are strongly influenced by parameters like temperature, pH-value, size of gas bubbles, etc. Similar to the well-known kLa value as quality parameter for oxygen transfer into bioreactor systems, mass transfer of CH4 or CO2 have to be characterized for individual bioreactors and optimized for given strain–bioreactor combinations. Sufficient supply of biomass with gaseous substrates calls for a permanent (continuous) inflow of substrates into the bioreactor. For CH4, solubility in water amounts to about 19 mg/L at room temperature and neutral pH-value; in the case of CO2, this value is reported with 1.7 g/L [98].
CH4 as the major component of natural gas and biogas was suggested by Rostkowski and colleagues as a substrate for PHA biosynthesis due to its potential to decrease PHA production costs [99]. In the meantime, a long-term process for PHA production based on CH4 was described by a Methylocystis-dominated enrichment culture obtained by repeated fed-batch supply with methane and oxygen [100]. Later, the strain Methylocystis sp. WRRC1 was isolated, an organism capable of producing PHB from CH4 alone and PHBHV when co-supplied with CH4 and the 3HV precursors valerate or n-pentanol [101]. Only recenty, Lopéz and colleagues described aerobic microbial growth and PHA biosynthesis on biogas and VFAs using the type II methanotroph Methylocystis hirsuta. The strain was able to thrive on artificial biogas and, under conditions of nitrogen deprivation, accumulated PHA up to 0.45 g per g biomass. When compared to control experiments with pure CH4, the presence of CO2, H2S and VFAs in artificial biogas did not negatively impact growth or PHA biosynthesis. Adding 10% of the carbon source as VFAs resulted in formation of PHBHV copolyesters and even boosted the maximum PHA yield and the intracellular PHA fraction. When using mixtures of biogas and propionic acid, the 3HV fraction in PHA amounted to 0.02 mol/mol, whereas this values increased to 0.25 mol/mol in the case of biogas and valeric acid mixtures [102]. As recently shown, this organism can efficiently be cultivated in gas-recycling bubble column bioreactors [103].
Cyanobacteria are phototrophic bacteria, which, in older literature, are termed as “Cyanophyta”, “blue algae”, or “blue-green algae”; from the microbiological point of view, these old designations should nowadays finally be circumvented in order to underline the prokaryotic nature of these organisms, which are preferably cultivated in light-transparent photobioreactor (PBR) systems. As comprehensively reviewed in the recent years, some cyanobacterial species display considerable potential for PHA production. According to the present state of knowledge, PHA biosynthesis occurs in species belonging to the cyanobacterial orders Chroccocales, Nostocales, Oscillatoriales, Pseudoanabenales and Synechococcales. Here, PHA accumulation can, depending on the strain, occur photoautotrophically (Anabaena cylindrica, Chlorogloea sp., Oscillatoria limosa, Synechocystis sp., Synechococcus sp.), chemoheterotrophically (Spirulina sp., Nostoc muscorum, Aulosira sp., Synechocystis sp.), or mixotrophically (Anabaena cylindrica, Chlorogloea sp., Gloeothece sp., Spirulina sp., Nostoc muscorum, Aulosira sp., Synechocystis sp., Synechococcus sp.). Different bioreactor devices were investigated for cyanobacterial PHA production [104,105,106]. Despite the numerous studies on shaking-flask-scale PHA production by cyanobacteria reported, only very few reports on such processes under controlled conditions in bioreactors are available. One such study investigated PHB biosynthesis by Anabaena solitaria in a flat panel PBR system. Cells in this PBR were airlifted and continuously substrate-supplied by bubbling CO2-enriched compressed air at a rate of 2.5 L/h from the PBR’s bottom, which makes this PBR a “flat plate bubble column” bioreactor. Halogen lamps served for external illumination and thermal energy needed for cyanobacterial growth. A working volume of 9.5 L was used to start the fermentation process. A. solitaria exhibited significant, yet not exceptionally high PHB production potential in this PBR system; PHA concentrations of up to 7 mg/L were reached after twelve days of fermentation at illumination and temperature of 500 µE/(m2 s) and 28 °C; the same output was achieved after three days at the same illumination and 40 °C [107].
Only recently, serious attempts were undertaken to make cyanobacterial PHA production industry-relevant. Researchers around Troschl et al. operated a 200 L pilot plant tubular glass PBR, located in a glass house, for about three years. In this setup, the authors tested robustness and potential of diverse PHA-producing cyanobacteria during long-term fermentations when supplied with CO2 from a local coal power plant; CO2 supply was automated by coupling to the increase of pH-value by substrate depletion. The illumination, supported by gas-discharge lamps, was accomplished by alternating day/night cycles. As a major outcome of their pilot scale experiments, the authors highlighted Synechocystis salina CCALA 192 as the most suitable cyanobacterial production strain because it turned out to be convenient to handle and, under examined cultivation conditions, reached final biomass and fractions of PHB homopolyester in the biomass in the range of 0.9 to 2.1 g/L and 0.05 to 0.09 g/g, respectively [108]. Follow-up studies with this PBR-strain combination revealed the feasibility of using inexpensive stillage digestate supernatant as additional carbon source; this mixotrophic feeding resulted in the production of 1.6 g/L CDM with about 0.055 g/g PHB in CDM [109]. Only recently, the authors presented a holistic study on the downstream processing of S. salina biomass produced in this process; fractionating the biomass into valued pigments, PHA and residual biomass, the latter undergoing application either as fodder or as feedstock for anaerobic biogas production, leads to a sustainable biorefinery concept, which could finally make cyanobacterial PHA production mature for the market [110]. Other current studies describe the recycling of residual cyanobacterial biomass as substrate for subsequent PHA-production processes; this was accomplished by da Silva et al., who cultivated the cyanobacterium Spriulina sp. LEB in a 1.8 L horizontal tubular PBR. After cell harvest and polymer extraction, the remaining liquid phase containing cell debris was fed to follow-up cultures in the same PBR system; cultivations were carried out under artificial illumination (12 kW/m²) and 12 h day/night cycles. As a major result, follow-up cultivations based on Spirulina waste resulted in similar biomass production as observed when using pure autotrophic cultivation medium, whereas PHB content in cells reached its highest value of almost 11% in a minimal medium supplemented with 25% Spirulina waste [111]. Similar recycling experiments were carried out before with the extremophile chemoheterotrophic haloarchaeon Hfx. mediterranei. This organism requires about 200 g/L of NaCl for optimum growth and PHA production [36]; hence, recycling of the highly saline residues accruing when producing PHA by this strain, namely salty spent fermentation broth (supernatant) remaining after cell harvest and salty cell debris as a left-over product of the PHA recovery process, is needed; this reduces the excessive salt lots to be disposed of. As demonstrated by Koller, it is possible to run follow-up cultivations with this organism on spent supernatant and to replace about 30% of expensive complex nitrogen sources like yeast extract by saline cell debris. For the recycling experiments, the PHA fraction in cells amounted to approximately 0.7 g/g, which approaches results obtained by the “original” fermentation using fresh medium on a 300 L pilot scale. Both for the “original” and the “recycling” setup, the produced biopolyesters was identified as PHBHV copolyester with about 10 mol-% 3HV [112].
Photosynthetic purple non-sulphur bacteria are currently also being investigated as PHA producers from gaseous substrates. In particular, Rhodospirillum rubrum attracts attention due to its capacity to produce PHA from syngas, a mixture of CO, CO2, CH4, H2, H2S and N2. Syngas is formed by high-temperature gasification and pyrolysis of organic materials, such as carbon-rich agricultural waste and, after appropriate conditioning, can be used as biotechnological feedstock [113]. R. rubrum possesses the enzymatic machinery enabling the conversion of the toxin CO to less toxic CO2 and bio-hydrogen; here, it should be added that conversion of CO to CO2 occurs via the water–gas shift reaction. CO2, in turn, undergoes anaerobic photosynthetic conversion to biomass and products such as PHA. Revelles and colleagues reported PHB homopolyester production by R. rubrum from syngas [114], whereas, starting from gasification of corn seeds, syngas-based PHA production by R. rubrum delivered PHBHV with 3HV contents of 0.14 mol/mol, however, obtained PHBHV productivity and intracellular fractions only amounted to 0.0002 g/(L h) and 0.09 g/g, respectively [115]. Nevertheless, PHBHV copolyester production from these simple, structurally unrelated C1-compounds, explained by the high intracellular propionyl-CoA pools in R. rubrum, makes this process stimulating. In addition, this strain also utilizes inexpensive heterotrophic substrates for PHA biosynthesis. As an example, Smith et al. cultivated the organism on distiller’s condensed corn soluble (CCS), a by-product of ethanol production; however, these authors reported only inferior PHBHV productivity and modest PHBHV fractions in biomass [116]. Another example for PHA production from waste-originating syngas resorts to household waste collected in the Spanish city Seville. In this process, syngas was generated from waste by microwave-induced pyrolysis in order to save time and energy. This article emphasized that syngas purity does not tremendously impact PHA productivity [117]. Choi and colleagues provided economic calculations, which estimate the production costs per kg of syngas-based PHA with less than $2 (USD) per kg. These calculations are based on a biorefinery concept using switch grass as feedstock for syngas production and generating bio-hydrogen as a marketable by-product of this process; according to these authors, the fermentation process should be carried out in CSTRs [118]. Karmann and colleagues demonstrated a tailored platform for safe production of PHB from syngas using Labfors (Infors, Basel, Switzerland) STRs, which can be operated in fed-batch and chemostat mode; the authors underlined the importance of safety measures to overcome the risks connected to CO as toxic substrate and biohydrogen as a precarious co-product and online analytical tools to make PHA production technologically feasible [119]. More recently, heterologous expression of PHA synthase genes in R. rubrum was studied by Klask and colleagues in order to enhance PHA productivity, and to extend the range of types of PHA accessible by this strain. Different Class I and Class IV PHA synthases were investigated in PHA-negative R. rubrum as the host organism; all of the new strains revealed PHA biosynthesis. As a major outcome, the authors proved that insufficient PHA synthase activity in R. rubrum wild type displays the bottle neck of this process, which can be solved by means of genetic engineering [120]. As a follow up, a genetically engineered R. rubrum strain harboring P. putida KT2440 PHA synthesis genes was successfully used by these authors for mcl-PHA biosynthesis in simple flasks aerated with artificial syngas [121].
Table 2 collects the presented case studies for discontinuous PHA production using different feeding regimes.

Table 2.
Selected case studies for discontinuous PHA production.
5. Continuous PHA Production Processes Operated as Chemostats
5.1. General
Continuous production processes are generally characterized by being operated under so-called “steady state” conditions, where process parameters like substrate and product concentrations, but also factors like pH-value, pO2, working volume, nutrient supply, etc., are kept constant. In accordance with the substrate conversion rate of the active biomass, the substrate is continuously refed and volume increase is compensated by continuous withdrawal of fermentation broth. The ratio between flow rate (volume into and out of the reactor per time) and working volume is known as “dilution rate” (D), which is a decisive parameter for continuous processes; too low D values will lead to insufficient supply of cells with substrates, resulting in low growth rates and productivities, while D exceeding a critical value will lead to “wash out” of the cultivation reactor. By carefully adjusting D, specific growth rate µ and product formation can be fine-tuned; hence, the operator forces the system to have a process-engineering parameter in order to fix a physiological parameter (µ) of biomass. To reach fast cell growth and high productivity, D will be selected slightly below the experimentally determined values for µmax. and qp, respectively. The inverse value of D, the so-called retention time τ, determines the time provided to the cells to convert substrate and accumulate PHA as an intracellular product. Often, “continuous processes” are used in the same meaning as “chemostat” processes; merging these terminations is not correct sensu stricto because “continuous” process regimes also encompass, in addition to chemostats (“chemical environment remaining static”), pH-stat, turbido-stat (turbidity as a parameter for the cell density remaining constant), redox-stat, or volume-stat processes [122,123]. In order to address the current trend in the literature, the expression “continuous PHA-production” in the subsequent paragraphs refers to chemostat studies for PHA production.
In the context of MMC for PHA production, continuous feeding strategies are more and more implemented [68]; such processes allow PHA production with the additional benefit of a more stable process and the possibility to adapt the PHA composition on the monomeric level, as demonstrated by Albuquerque et al., who supplied MMCs both via pulse feeding (fed-batch) and continuously with differently composed mixtures of odd- and even numbered VFAs to study the effect of the feeding regime on PHA productivity, composition of obtained PHBHV copolyesters and their material properties. The authors clearly emphasize the superiority of the continuous regime in terms of flexibility of polyester composition and productivity. In more detail, this process consisted of three stages: a first anaerobic stage was carried out for continuous fermentation of sugar molasses to VFAs in a STR, while a second, aerobic stage in a sequencing batch reactor (SBR) serving for enrichment of PHA-accumulating organisms under dynamic feast-and famine cultivation at a C/N/P ratio in the feed stream amounting to 100/8/1; this second stage was operated for ten months in order to enrich a powerful MMC. The third stage contained enriched sludge from stage 2, which was supplied with micro-filtrated fermented molasses from stage 1, either by pulse feeding or continuously. Polymer composition was triggered by artificially changing the fatty acid profile in the fermented molasses stream [65]. Only recently, Marang et al. studied the impact of continuous substrate feeding on enriching PHA-accumulating microbes in two SBRs. In the first SBR, acetate was supplied continuously, resulting in the enrichment of Zoogloea sp. PHA was accumulated by exposure to excess acetate, but PHA productivity and PHA mass fraction (0.53 g/g) amounted to only about 20% of the values obtained for enrichment cultures in an established, pulse-fed SBR with the PHA producer Plasticicumulans acidivorans as the dominating species. In the second SBR, half of the acetate was supplied at the beginning of the cycle, and the rest was fed continuously. By this true feast phase, the enrichment of P. acidivorans was not inhibited by the continuous supply of acetate; a mass fraction of 0.85 g/g PHA, which was identified as homopolyester PHB, was obtained in CDM. This displays a paradigm shift in running MMC-based PHA production processes under dynamic feast-and-famine feeding by showing that, for enrichment of bacteria with excellent PHA production capacity, a real feast phase by periodic substrate excess is indispensable, but not periodic famine phases [64].
5.2. One-Stage Chemostats
5.2.1. One-Stage Chemostats Based on Pure Cultures
For continuously operated STRs, one-stage (“single-stage”) setups are not very productive for PHA production; as intracellular products of the secondary metabolism, boosted synthesis of PHA requires the limitation of a growth-essential nutrient; hence, it is not possible to continuously supply carbon source plus nitrogen and phosphate source at rates that result in both high biomass formation and expedient PHA accumulation. For high volumetric productivity, both biomass concentration and intracellular PHA mass fraction have to be optimized. The impact of τ on PHA productivity in one-stage continuous processes was, for the first time, demonstrated by Senior and colleagues, who cultivated Azotobacter beijerinkii NCIB 9067 in a continuously operated 2 L STR under restricted oxygen and nitrogen supply. Increasing τ by decreasing D from 0.252 1/h to 0.049 1/h boosted the intracellular PHA fraction in cells from 0.20 to 0.44 g/g. However, these values are still inferior compared to results obtained in batch setups, where the mass fraction of PHA in CDM amounted to 74% [124]. Later, Ramsay and colleagues demonstrated the viability of continuous operation mode to trigger the molar composition of PHA; these authors cultivated C. necator in one-stage continuous mode, supplying glucose and the 3HV precursor propionic acid continuously at a tailored ratio. Using a D of 0.15 1/h, the authors obtained a volumetric PHBHV productivity of 0.3 g/(L h), with 3HV fractions in PHBHV of 0.05 mol/mol and 0.33 g/g PHBHV in CDM. Regarding these productivities and PHA content values, continuous process mode in only one-stage without chronological and/or spatial separation of biomass formation and PHA accumulation was not competitive with fed-batch setups using the same production strain. However, these experiments by Ramsay’s group opened the door for triggering PHA composition by a continuous process regime [125].
5.2.2. Dual Nutrient Limited Chemostat Cultivation to Utilize “Inefficient” Carbon Sources for PHA Biosynthesis
Especially in the case of using toxic substrates like those usually needed for mcl-PHA biosynthesis, dual nutrient (carbon and nitrogen source) limited growth (DNL) in continuous cultures is suggested as a suitable method to produce different types of PHA. The concept of DNL originated from the biological law that growth rate, concentration and composition of biomass are not influenced by one limiting factor alone, but by the concerted action of two or more limiting nutrients (reviewed by [126]). Because toxicity of a substrate depends on its concentration, cell growth is usually not affected when the substrate is immediately metabolized, hence when its concentration does not exceed a certain threshold. This was demonstrated in the case of mcl-PHA production by DNL cultivation of Pseudomonas putida GPo1 on gaseous n-octane [127], Pseudomonas oleovorans (today: P. putida) ATCC 29347 on octanoate [128,129], or, in the case of scl-PHA production, for C. necator DSM 428 on mixtures of butyric acid and valeric acid, which normally display considerable toxicity already at low concentrations. By changing the butyric/valeric acid ratio in the feed stream supplied at D = 0.1 1/h in a one-stage chemostat process, it was possible to produce PHBHV copolyesters of tunable monomeric composition under DNL cultivation, which allowed the reproducible fine-tuning of thermomechanical biopolyesters properties such as melting point (Tm), crystallinity, flexibility, or glass transistion temperature (Tg) [130]. By DNL continuous cultivation of P. putida GPo1 with multiple carbon sources, it was possible to produce mcl-PHA of unprecedented monomeric composition. Mixtures of 5-phenylpentanoate, octanoate and 10-undecenoate were used at D = 0.1 1/h and C/N ratios of 15/1 in the substrate feed stream to produce diverse poly(3-hydroxy-5-phenylvalerate-co-3-hydroxyalkanoate-co-3-hydroxy-ω-alkenoate)s consisting of aromatic, saturated and unsaturated building blocks. The fraction of the aromatic monomer (3-hydroxy-5-phenylvalerate) ranged from 0 to 0.52 mol/mol. It was observed that increasing aromatic fraction results in a linear increase of the polymer’s Tg from −37.6 °C to −6 °C [131]. Later, DNL cultivation in a single-stage chemostat was carried out with mixtures of lactic acid and the aromatic compounds p-methylphenylvaleric acid or phenylvaleric acid [132]. In the case of using p-methylphenylvaleric acid as co-substrate, a poly(3-hydroxy-p-methylphenylvalerate) mcl-PHA homopolyester was obtained, which displayed considerable crystallinity and a sharp Tm at 99 °C, which is in contrast to others described, often completely amorphous mcl-PHA biopolyesters. Poly(3-hydroxy-phenylvalerate), the homopolyester produced by using phenylvaleric acid as co-substrate, displayed material properties typical for mcl-PHA. Again, the authors underlined that chemostat cultivation, especially using DNL regime, is the only possibility to culture PHA producing microbes on such “inefficient” substrates like described aromatic compounds, which exert considerable toxicity to the cells [132].
5.2.3. Non-Sterile Single-Stage Chemostat Processes
Continuous cultivation processes, especially those that run for extended periods, always carry the risk of microbial infection, which endangers the entire cultivation process. As a way out of this dilemma, extremophilic organisms can be used for PHA production under restricted sterility precautions, or even under non-sterile cultivation conditions in open reaction vessels; such processes can also be operated in continuous mode (reviewed by [122,123,133]).
Already in 1990, Lillo and Rodriguez-Valera tested the extremely halophile archaeon Hfx. mediterranei in continuously operated long-term bioreactor experiments on glucose as carbon source and two different phosphate concentration levels at five different D ranging between 0.02 and 0.10 1/h. At D = 0.02 1/h, best results were obtained; here, the intracellular PHA mass fraction amounted to 0.42 g/g, with the PHA concentration and volumetric productivity reaching 1.5 g/L and 0.03 g/(L h), respectively. The high robustness and long-term stability of this organism was revealed by taking only minimal sterility precautions during running the process continuously at D = 0.12 1/h for three months; no microbial contamination was observed during this long period [134]. Years later, this high resistance of Hfx. mediterranei against microbial contamination was confirmed by Hermann-Krauss and colleagues, who cultivated the strain in fed-batch mode on CGP, a waste stream from biodiesel production and 4HB-precursors for about 200 h without sterilization of the bioreactor equipment; only the nutritional medium was heat-sterilized. Also in this process, no microbial contamination occurred [84].
Halomonas campaniensis LS21, an alkalo-halophile Gram-negative bacterium, was cultivated in a seawater-based open, non-sterile and continuous process dedicated to PHA production. This process was based on a substrate cocktail of polysaccharides, lipids and proteins, mimicking nutrient-rich kitchen waste. Both the wild type H. campaniensis LS21 and its recombinant strain equipped with the phbCAB PHB synthesis genes were continuously cultivated for more than two months in artificial seawater containing the mixed substrates and 27 g/L NaCl at a pH-value of 10. At 37 °C, the engineered strain accumulated about 0.7 g/g PHB in biomass until the process end, which is more than double the value of what was reached by the parental strain; both cultures did not reveal any microbial contamination [135].
5.3. Two-Stage Chemostats
5.3.1. Two-Stage Chemostats under Strict Sterility Precautions
Even when using growth-associated PHA production strains like, e.g., A. lata, PHA mass fraction in cells is not exceptionally high when continuously supplying the carbon source and the growth-limiting component like nitrogen source at a balanced ratio; this generates active biomass at relatively high productivity, but with only a modest PHA fraction [125]. Regarding the rates for microbial growth and PHA accumulation for a given strain-substrate combination, it becomes obvious that the autocatalytic growth typically occurs much faster than PHA accumulation; hence, it is impossible to carry out such processes in “one-pot-setups” at sufficient productivity. Therefore, it was demonstrated in the past that two-stage continuous processes are better suitable for this purpose. Here, in a first STR, active biomass is continuously generated by supplying a balanced nutrient medium; this dense cultivation broth is continuously transferred into a second STR, where carbon source is fed continuously, but no more growth limiting substrate is provided [125]. When optimizing such two-stage continuous PHA production processes, it is reasonable to adjust D in the first STR to a value slightly below the maximum specific growth rate µmax. of the strain under given cultivation conditions, in order to select concentrations of both the growth-limiting component and the carbon source slightly above the concentration levels that are converted in the first stage. In the second stage, residues of the growth-limiting component will immediately be consumed by the cells, which are continuously supplied with carbon source in order to provoke enhanced PHA accumulation. In this second stage, it is reasonable to adjust τ and carbon feed in such a way as to give the cells enough time for the biotransformation of carbon source to PHA, which results in higher intracellular PHA fractions and to keep the actual carbon source at almost zero in order to avoid economically disadvantageous substrate losses. Ramsay et al. demonstrated for the first time the superiority of two-stage chemostat processes. Using A. lata as production strain on sucrose and propionic acid in two-stage setups, operated at D = 0.15 1/h in both stages, the authors obtained a mass fraction of PHBHV in CDM of about 0.38 g/g and a 3HV share in PHBHV of 0.15 mol/mol in the first stage; nitrogen source and 3HV precursor were completely utilized by the cells. The second stage, continuously supplied with the fermentation broth from stage 1 containing residual sucrose, but no additional medium, served for ongoing conversion of sucrose to increase PHA fraction in cells; hence, in this stage, cells were allowed to take their time to go on accumulating PHA by completely depleting sucrose. Mass fraction of PHBHV in CDM increased to about 0.55 g/g in this second stage, while the 3HV share in PHBHV decreased to 0.11 mol/mol due to the lacking supply with propionic acid in this stage [121]. In the context of two-stage continuous processes, additional strain/substrate combinations were reported in the past years, resulting in the production of different scl- and mcl-PHA homo- and heteropolyesters (reviewed by [122,123]). As a pioneering example, Du and colleagues were the first who selected drastically higher τ in the second stage than in the first in order to boost intracellular PHA fractions. Using Ralstonia eutropha (today: C. necator) strain WSH3 on glucose and nitrogen deprivation, maximum volumetric productivity for PHB amounted to 1.43 g/(L h) at an overall D = 0.12 1/h; however, under these conditions, PHB mass fractions in biomass did not exceed 0.48 g/g; at an overall D = 0.075 1/h, the mass fraction increased to more than 72%, with only slightly decreasing overall productivity to 1.24 g/(L h) [136]. Attempts to run the first stage at rather high D (0.2 1/h) to approach µmax. and lower D (0.06 1/h) in the second stage to enable high PHA accumulation were carried out by Mothes and Ackermann, who obtained a PHA copolyester consisting of 3HB and 4HB in two-stage continuous processes of Delftia acidovorans on acetic acid and the 4HB precursor GBL. Using the gaseous substrate n-octane, a two-stage continuous process was operated for mcl-PHA production by P. oleovorans (today: P. putida GPo1), a strain featuring strict separation between the phases of microbial growth and PHA biosynthesis during batch cultivation. Also here, D used for the first stage was close to the biological maximum determined by the strain’s µmax. (0.21 1/h), whereas the second stage was operated at a D of 0.16 1/h; 1.06 g/(L h) of poly(3-hydroxyhexanoate-co-3-hydroxyoctanoate) were reported for the volumetric productivity, with cells containing 0.63 g/g of the mcl-PHA [137]. Moreover, P. putida GPo1 was also cultivated by Hartmann and colleagues in a two-stage chemostat system consisting of two chemostats linked in sequence with two different substrates (octanoic acid in the first and 10-undecenoic acid in the second stage). Interestingly, the cells accumulated a blend of two different polymers (PHO and poly(3-hydroxy-10-undecenoate-co-3-hydroxy-8-nonenoate-co-3-hydroxy-6-heptenoate) (PHUE); hardly any blocky structured PHA (b-PHA) were formed [138]. This observation is quite different to previous reports that claimed to have b-PHA produced when switching substrates during the period of PHA accumulation [139].
5.3.2. Non-Sterile Two-Stage Chemostat Cultivation for PHA Production
Similar to the open, single-stage continuous processes by H. campaniensis LS21 described above [140], Halomonas TD01, another Gram-negative bacterial isolate populating Chinese salt lakes, was cultivated by Tan and associates for two weeks in an open, non-sterile and continuous two-stage process. In the first stage, containing a saline glucose and nitrogen medium, CDM reached an average value of 40 g/L; the intracellular PHB fraction in biomass amounted to 0.60 g/g. The fermentation broth was continuously transferred from the first to the second, nitrogen-deficient stage. Although this transfer diluted the biomass concentration, a constant intracellular PHB mass fraction between 0.65 and 0.70 g/g was obtained. The authors explicitly suggested this open, continuous fermentation process as the ultimate step forward to achieve cost-efficient PHA production [140]. This organism was later genetically engineered to make it more stable by inhibiting its DNA restriction/methylation apparatus. Moreover, the stable plasmid pSEVA341 was constructed to induce the expression of multiple pathway genes. This construct was further engineered by deleting genes encoding 2-methylcitrate synthase and three PHA depolymerases within its chromosome. The resulting strain Halomonas TD08 displayed overexpression of enzymes responsible for threonine synthesis, which enabled the strain to accumulate PHBHV copolyesters harboring 4–6 mol.-% 3HV from structurally unrelated carbon sources. Overexpression of the cell division inhibitor MinCD during biomass growth elongated the cells’ shape by about 1.4 times, which resulted in boosting PHA mass fraction in biomass from 0.69 to 0.82 g/g, further enabling a simplified process for cell harvest [141].
5.4. Multi-Stage Chemostats
Described two-stage processes can be expanded to multi-stage processes, hence CSTR-bioreactor cascades. This approach was demonstrated for the first time by Atlić and colleagues, who used a five-stage bioreactor cascade consisting of one CSTR serving for generation of active biomass under permanent feeding of a nutrient-rich medium and four subsequent CSTRs for high productive PHA accumulation under nitrogen-limited conditions [142]. As previously suggested by Braunegg and colleagues, this series of four CSTRs serving the same purpose (PHA accumulation) resembles the process engineering characteristics of a tubular plug flow reactor (TPFR), which, based on theoretical considerations, was suggested almost three decades ago to be highly suitable for generation of intracellular bioproducts of the secondary metabolism; the more vessels the cascade consists of, the more the flow characteristics resemble the plug flow characteristics of a TPFR [53]. Above the potential of a TPFR, the cascade setup offers the additional benefit of adaptability of the process conditions (pH-value, dissolved oxygen concentration, co-substrate supply) in each of the individual cascade stages; this enables the production of PHA biopolyesters of pre-defined composition and molecular mass. When operated with C. necator on glucose as the sole carbon source, the system generated high volumetric and specific PHB productivity of up to 2.31 g/(L h) and 0.105 g/(g h), respectively, with PHB biopolyesters displaying highly uniform molecular masses under steady state conditions. Nitrogen source remaining in the effluent of the first vessel (dedicated to biomass growth) was completely depleted in vessel number two, which was used as a “transient stage” between growth and product formation, whereas vessels 3 to 5 were exclusively dedicated to product formation. Such multi-stage CSTR-bioreactor cascades were suggested by the authors as a precious process-engineering tool for producing tailor-made b-PHA by supplying selected precursor substrates to the individual stages [142].
Based on the obtained experimental data, this multi-stage cascade system was later subjected towards different investigative studies based on diverse mathematical modelling tools [58]. First, a formal kinetic mathematical model was developed by Horvat et al., in order to get deeper insights about how to best operate such systems. Here, the first vessel of the cascade (R1) was considered a nutritionally balanced continuous biomass production system. R2 was modelled as a process governed by two substrates (glucose, nitrogen), while vessels R3–R5 were committed to PHA biosynthesis under continuous glucose fed, but enduring nitrogen limitation. The developed model consisted of 49 parameters and 20 differential equations. Twenty of the parameters (four times five due to five vessels) were accounted for the initial concentrations of PHA, non-PHA biomass, carbon- and nitrogen source, ten parameters correspond to the in- and outflow for the five individual vessels, seven kinetic constants were integrated into the model to address substrate saturation and inhibition and five parameters were accounted to tank volumes R1–R5; moreover, four conversion factors and three inlet stream substrate concentrations were used as additional parameters to complete the model. Three independent cascade cultivations were performed in order to better define these parameters and to validate the predictive power of the model; these three cultivations were carried out at different D in individual reactors and at different feeding regimes (number of stages continuously supplied with glucose). Simulated results predicted well the experimental data for glucose, biomass and PHA if glucose is continuously supplied to all five vessels. The authors provide calculations showing that volumetric productivity in the cascade could be further boosted from experimentally obtained 2.14 g/(L h) to the modeled value of 9.95 g/(L h); this theoretical value is based on suggested optimization of certain experimental conditions, such as the overall D, the substrate inflow rate and substrate concentration in the feed streams to all individual stages. Under such optimized conditions, up to 164 g/L biomass and 123 g/L PHA could be obtained [143].
Insights into the metabolic fluxes of C. necator in such multi-stage cascade PHA production processes were provided by Lopar et al., who developed a highly structured metabolic model of this process. The model covers a metabolic network of 43 mass balance equations for 43 intracellular compounds. The metabolic conditions of bacteria in the individual cascade vessels (R1–R5) were studied via two-dimensional yield space analysis of elementary flux modes for conversion of glucose to biomass and PHA, respectively. Each elementary mode was converted into an overall stoichiometric reaction, which encompasses educts (glucose, ammonia and oxygen) and products (biomass, PHA, and CO2) and the related stoichiometric coefficients for these six compounds. Calculating experimental yields and metabolic fluxes based on these reactions enabled in silico illustration of all possible metabolic conditions of the bacteria in the five individual cascade vessels. The metabolic model revealed excellent agreement with experimental results regarding biomass and PHA yields in all five stages of the cascade. This high-structured model provided additional hints to optimize the operation of the cascade by further adapting the feeding strategy; consistent with the outcomes from the formal kinetic modelling (vide supra), glucose feeding in all five stages turned out to be the most suitable strategy to perform multi-stage continuous PHB synthesis [144].
Only recently, Vadlja and colleagues carried out electron microscopic studies on the statistical distribution of number of PHA granules (“carbonosomes”) per cell, cell size and PHA granule size in all individual cascade stages R1–R5; this opened deep insight into cell morphology and carbonosome formation under balanced (R1), transient (R2) and nutrient-limited (R3–R5) conditions. Electron microscopic pictures of cells from Atlić et al.’s experiments [133] were converted to binary images using the software tool Image J, thus visualizing PHA (white) and non-PHA biomass (black) footprint areas. For each cascade stage, results for μ and qP correlated well with experimentally determined kinetics. Log-normal and gamma distribution best described granule size distribution in the individual cascade stages. In R1, cells containing granules of small size dominated. This situation changed with longer τ; from R2 to R5, the granules gradually increased in sizes, until finally an upper limit has been reached, which appears to have been determined by steric factors, caused by the physiological maximum of the ratio of carbonosomes to cell area. The ratio of carbonosomes to cell area augmented along the system, reaching a final value of 0.64 in R5. In addition, the extent of the rise of the PHA fraction in cells and the increase of the carbonosomes to slow down from R1 to R5; moreover, the number of granules per cell also decreases with increasing τ. These data support the optimization of the cascade system, mainly by evaluating the optimum number of stages [145].
Table 3 collects the presented case studies for PHA production in continuous mode.

Table 3.
Selected case studies for continuous PHA production.
6. Conclusions
The review illustrates the huge variety of different bioreactor systems and diverse cultivation regimes already tested to produce PHA biopolyesters of different molecular architecture by a plethora of different microbes. It becomes clear that different combinations of production strains and substrates require different fermentation equipment, bioreactor facilities and feeding regimes. The cultivation regime and process design have to be adapted to the physiological and kinetic particularities of the biological system in order to optimize PHA productivity and product quality. As a conclusion of the most recent studies, it can be expected that the future trend in fermentative PHA production will combine the operation of continuous chemostat process, the application of robust, extremophilic production strains needed to run these processes with a minimum of energy input, the use of inexpensive carbon-rich feedstocks to safe substrate costs and the large scale implementation of sustainable downstream processing methods for PHA recovery and purification. Only this combination will allow for sustainable and economic production of PHA biopolyesters of on-demand quality as a decisive step forwards into efficient White Biotechnology.
Conflicts of Interest
The author declares no conflict of interest.
References
- Pasteur, L. Mémoire sur la fermentation alcoolique. Ann. Chim. Phys. 1860, 58, 323–426. [Google Scholar]
- Choi, J.; Lee, S.Y. Factors affecting the economics of polyhydroxyalkanoate production by bacterial fermentation. Appl. Microbiol. Biotechnol. 1999, 51, 13–21. [Google Scholar] [CrossRef]
- Kavitha, G.; Rengasamy, R.; Inbakandan, D. Polyhydroxybutyrate production from marine source and its application. Int. J. Biol. Macromol. 2017, 111, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, R.Y.U.; Castilho, N.A.S.; da Silva, M.A.C.; Miotto, M.C.; de Souza Lima, A.O. Prospecting for marine bacteria for polyhydroxyalkanoate production on low-cost substrates. Bioengineering 2017, 4, 60. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Dubey, S.; Singh, P.; Shrivastava, A.; Mishra, S. Biodegradable polymeric substances produced by a marine bacterium from a surplus stream of the biodiesel industry. Bioengineering 2016, 3, 34. [Google Scholar] [CrossRef] [PubMed]
- Kucera, D.; Pernicová, I.; Kovalcik, A.; Koller, M.; Mullerova, L.; Sedlacek, P.; Mravec, F.; Nebesarova, J.; Kalina, M.; Marova, I.; et al. Characterization of the promising poly(3-hydroxybutyrate) producing halophilic bacterium Halomonas halophila. Bioresour. Technol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Jendrossek, D.; Pfeiffer, D. New insights in the formation of polyhydroxyalkanoate granules (carbonosomes) and novel functions of poly(3-hydroxybutyrate). Environ. Microbiol. 2014, 16, 2357–2373. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Sedlacek, P.; Koller, M.; Kucera, D.; Pernicova, I. Involvement of polyhydroxyalkanoates in stress resistance of microbial cells: Biotechnological consequences and applications. Biotechnol. Adv. 2018. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Marova, I.; Svoboda, Z.; Mikulikova, R. Use of controlled exogenous stress for improvement of poly(3-hydroxybutyrate) production in Cupriavidus necator. Folia Microbiol. 2010, 55, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Marova, I.; Snajdar, O.; Mravcova, L.; Svoboda, Z. Production of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by Cupriavidus necator from waste rapeseed oil using propanol as a precursor of 3-hydroxyvalerate. Biotechnol. Lett. 2010, 32, 1925–1932. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Marova, I.; Stankova, M.; Mravcova, L.; Svoboda, Z. Effect of ethanol and hydrogen peroxide on poly (3-hydroxybutyrate) biosynthetic pathway in Cupriavidus necator H16. World J. Microbiol. Biotechnol. 2010, 26, 1261–1267. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Sedlacek, P.; Mravec, F.; Samek, O.; Marova, I. Evaluation of 3-hydroxybutyrate as an enzyme-protective agent against heating and oxidative damage and its potential role in stress response of poly(3-hydroxybutyrate) accumulating cells. Appl. Microbiol. Biotechnol. 2016, 100, 1365–1376. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Sedlacek, P.; Mravec, F.; Krzyzanek, V.; Nebesarova, J.; Samek, O.; Kucera, D.; Benesova, P.; Hrubanova, K.; Milerova, M.; et al. The presence of PHB granules in cytoplasm protects non-halophilic bacterial cells against the harmful impact of hypertonic environments. New Biotechnol. 2017, 39, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Soto, G.; Setten, L.; Lisi, C.; Maurelis, C.; Mozzicafreddo, M.; Cuccioloni, M.; Angeletti, M.; Ayub, N.D. Hydroxybutyrate prevents protein aggregation in the halotolerant bacterium Pseudomonas sp. CT13 under abiotic stress. Extremophiles 2012, 16, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Contreras, A.; Koller, M.; Braunegg, G.; Marqués-Calvo, M.S. Poly[(R)-3-hydroxybutyrate] production under different salinity conditions by a novel Bacillus megaterium strain. New Biotechnol. 2016, 33, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Sedlacek, P.; Krzyzanek, V.; Mravec, F.; Hrubanova, K.; Samek, O.; Kucera, D.; Benesova, P.; Marova, I. Accumulation of poly(3-hydroxybutyrate) helps bacterial cells to survive freezing. PLoS ONE 2016, 11, e0157778. [Google Scholar] [CrossRef] [PubMed]
- Slaninova, E.; Sedlacek, P.; Mravec, F.; Mullerova, L.; Samek, O.; Koller, M.; Hesko, O.; Kucera, D.; Marova, I.; Obruca, S. Light scattering on PHA granules protects bacterial cells against the harmful effects of UV radiation. Appl. Microbiol. Biotechnol. 2018, 102, 1923–1931. [Google Scholar] [CrossRef] [PubMed]
- Koller, M. Poly(hydroxyalkanoates) for food packaging: Application and attempts towards implementation. Appl. Food Biotechnol. 2014, 1, 3–15. [Google Scholar] [CrossRef]
- De Souza, L.; Shivakumar, S. Polyhydroxyalkanoates (PHAs)-biodegradable polymers for ‘green’ food packaging materials. In Recent Advances in Biotechnology; Delhi Printer & Lamination: New Delhi, India, 2017; p. 149. [Google Scholar]
- Koller, M. Biodegradable and biocompatible polyhydroxyalkanoates (PHA): Auspicious microbial macromolecules for pharmaceutical and therapeutic applications. Molecules 2018, 23, 362. [Google Scholar] [CrossRef] [PubMed]
- Peptu, C.; Kowalczuk, M. Biomass-derived polyhydroxyalkanoates: Biomedical applications. In Biomass as Renewable Raw Material to Obtain Bioproducts of High-Tech Value, 1st ed.; Popa, V.I., Volf, I., Eds.; Elsevier, B.V.: Amsterdam, The Netherlands, 2018; pp. 271–313. ISBN 978-0-444-63774-1. [Google Scholar]
- Brigham, C.J.; Sinskey, A.J. Applications of polyhydroxyalkanoates in the medical industry. Int. J. Biotechnol. Well. Ind. 2012, 1, 52–60. [Google Scholar] [CrossRef]
- Junyu, Z.; Shishatskaya, E.I.; Volova, T.G.; da Silva, L.F.; Chen, G.Q. Polyhydroxyalkanoates (PHA) for therapeutic applications. Mater. Sci. Eng. C 2018, 86, 144–150. [Google Scholar] [CrossRef]
- Pierro, L.; Matturro, B.; Rossetti, S.; Sagliaschi, M.; Sucato, S.; Alesi, E.; Bartsch, E.; Arjmand, F.; Papini, M.P. Polyhydroxyalkanoate as a slow-release carbon source for in situ bioremediation of contaminated aquifers: From laboratory investigation to pilot-scale testing in the field. New Biotechnol. 2017, 37, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Zhila, N.; Shishatskaya, E. Properties of PHA bi-, ter-, and quarter-polymers containing 4-hydroxybutyrate monomer units. Int. J. Biol. Macromol. 2018, 111, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Maršálek, L.; Miranda de Sousa Dias, M.; Braunegg, G. Producing microbial polyhydroxyalkanoate (PHA) biopolyesters in a sustainable manner. New Biotechnol. 2017, 37, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, G.; Rocher, M.; Braunegg, G. Effects of low dissolved-oxygen concentrations on poly-(3-hydroxybutyrate-co-3-hydroxyvalerate) production by Alcaligenes eutrophus. Appl. Environ. Microbiol. 1997, 63, 827–833. [Google Scholar] [PubMed]
- Koller, M.; Bona, R.; Chiellini, E.; Fernandes, E.G.; Horvat, P.; Kutschera, C.; Hesse, P.; Braunegg, G. Polyhydroxyalkanoate production from whey by Pseudomonas hydrogenovora. Bioresour. Technol. 2008, 99, 4854–4863. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Hesse, P.; Fasl, H.; Stelzer, F.; Braunegg, G. Study on the effect of levulinic acid on whey-based biosynthesis of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by Hydrogenophaga pseudoflava. Appl. Food Biotechnol. 2017, 4, 65–78. [Google Scholar] [CrossRef]
- Koller, M.; Miranda de Sousa Dias, M.; Rodríguez-Contreras, A.; Kunaver, M.; Žagar, E.; Kržan, A.; Braunegg, G. Liquefied wood as inexpensive precursor-feedstock for bio-mediated incorporation of (R)-3-hydroxyvalerate into polyhydroxyalkanoates. Materials 2015, 8, 6543–6557. [Google Scholar] [CrossRef] [PubMed]
- Ashby, R.D.; Solaiman, D.K.; Nuñez, A.; Strahan, G.D.; Johnston, D.B. Burkholderia sacchari DSM 17165: A source of compositionally-tunable block-copolymeric short-chain poly(hydroxyalkanoates) from xylose and levulinic acid. Bioresour. Technol. 2017, 253, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Salerno, A.; Strohmeier, K.; Schober, S.; Mittelbach, M.; Illieva, V.; Chiellini, E.; Braunegg, E. Novel precursors for production of 3-hydroxyvalerate-containing poly[(R)-hydroxyalkanoate]s. Biocatal. Biotransform. 2014, 32, 161–167. [Google Scholar] [CrossRef]
- Johnston, B.; Jiang, G.; Hill, D.; Adamus, G.; Kwiecień, I.; Zięba, M.; Sikorska, W.; Green, M.; Kowalczuk, M.; Radecka, I. The molecular level characterization of biodegradable polymers originated from polyethylene using non-oxygenated polyethylene wax as a carbon source for polyhydroxyalkanoate production. Bioengineering 2017, 4, 73. [Google Scholar] [CrossRef] [PubMed]
- Miranda de Sousa Dias, M.; Koller, M.; Puppi, D.; Morelli, A.; Chiellini, F.; Braunegg, G. Fed-Batch synthesis of poly(3-hydroxybutyrate) and poly(3-hydroxybutyrate-co-4-hydroxybutyrate) from sucrose and 4-hydroxybutyrate precursors by Burkholderia sacchari strain DSM 17165. Bioengineering 2017, 4, 36. [Google Scholar] [CrossRef] [PubMed]
- Raposo, R.S.; de Almeida, M.C.M.; da Fonseca, M.M.R.; Cesário, M.T. Feeding strategies for tuning poly(3-hydroxybutyrate-co-4-hydroxybutyrate) monomeric composition and productivity using Burkholderia sacchari. Int. J. Biol. Macromol. 2017, 105, 825–833. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Hesse, P.; Bona, R.; Kutschera, C.; Atlić, A.; Braunegg, G. Potential of various archae-and eubacterial strains as industrial polyhydroxyalkanoate producers from whey. Macromol. Biosci. 2007, 7, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Salgaonkar, B.B.; Bragança, J.M. Utilization of sugarcane bagasse by Halogeometricum borinquense strain E3 for biosynthesis of poly(3-hydroxybutyrate-co-3-hydroxyvalerate). Bioengineering 2017, 4, 50. [Google Scholar] [CrossRef] [PubMed]
- Povolo, S.; Romanelli, M.G.; Basaglia, M.; Ilieva, V.I.; Corti, A.; Morelli, A.; Chiellini, E.; Casella, S. Polyhydroxyalkanoate biosynthesis by Hydrogenophaga pseudoflava DSM 1034 from structurally unrelated carbon sources. New Biotechnol. 2013, 30, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Hokamura, A.; Yunoue, Y.; Goto, S.; Matsusaki, H. Biosynthesis of polyhydroxyalkanoate from steamed soybean wastewater by a recombinant strain of Pseudomonas sp. 61–63. Bioengineering 2017, 4, 68. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.Q.; Hajnal, I. The ‘PHAome’. Trends Biotechnol. 2015, 33, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Abe, H.; Ishii, N.; Sato, S.; Tsuge, T. Thermal properties and crystallization behaviors of medium-chain-length poly(3-hydroxyalkanoate)s. Polymer 2012, 53, 3026–3034. [Google Scholar] [CrossRef]
- Li, Z.J.; Qiao, K.; Che, X.M.; Stephanopoulos, G. Metabolic engineering of Escherichia coli for the synthesis of the quadripolymer poly(glycolate-co-lactate-co-3-hydroxybutyrate-co-4-hydroxybutyrate) from glucose. Metab. Eng. 2017, 44, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, S.; Yamada, M.; Matsumoto, K.I.; Tajima, K.; Satoh, Y.; Munekata, M.; Ohno, K.; Kohda, K.; Shimamura, T.; Kambe, H.; et al. A microbial factory for lactate-based polyesters using a lactate-polymerizing enzyme. Proc. Natl. Acad. Sci. USA 2008, 105, 17323–17327. [Google Scholar] [CrossRef] [PubMed]
- Steinbüchel, A.; Lütke-Eversloh, T. Metabolic engineering and pathway construction for biotechnological production of relevant polyhydroxyalkanoates in microorganisms. Biochem. Eng. J. 2003, 16, 81–96. [Google Scholar] [CrossRef]
- Rehm, B.H. Polyester synthases: Natural catalysts for plastics. Biochem. J. 2003, 376, 15–33. [Google Scholar] [CrossRef] [PubMed]
- Castilho, L.R.; Mitchell, D.A.; Freire, D.M. Production of polyhydroxyalkanoates (PHAs) from waste materials and by-products by submerged and solid-state fermentation. Bioresour. Technol. 2009, 100, 5996–6009. [Google Scholar] [CrossRef] [PubMed]
- Sindhu, R.; Pandey, A.; Binod, P. Solid-state fermentation for the production of poly(hydroxyalkanoates). Chem. Biochem. Eng. Q. 2015, 29, 173–181. [Google Scholar] [CrossRef]
- Pandey, A. Solid-state fermentation. Biochem. Eng. J. 2003, 13, 81–84. [Google Scholar] [CrossRef]
- Oliveira, F.C.; Freire, D.M.G.; Castilho, L.R. Production of poly(3-hydroxybutyrate) by solid-state fermentation with Ralstonia eutropha. Biotechnol. Lett. 2004, 26, 1851–1855. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, F.C.; Dias, M.L.; Castilho, L.R.; Freire, D.M. Characterization of poly(3-hydroxybutyrate) produced by Cupriavidus necator in solid-state fermentation. Bioresour. Technol. 2007, 98, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Rocha, P.R.S., Jr. Estudo das Condições de Cultivo para a Produção de PHB por Cupriavidus necator em Fermentação no Estado Sólido. Ph.D. Thesis, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil, 2005. (In Spanish). [Google Scholar]
- Ramadas, N.V.; Singh, S.K.; Soccol, C.R.; Pandey, A. Polyhydroxybutyrate production using agro-industrial residue as substrate by Bacillus sphaericus NCIM 5149. Braz. Arch. Biol. Technol. 2009, 52, 17–23. [Google Scholar] [CrossRef]
- Braunegg, G.; Lefebvre, G.; Renner, G.; Zeiser, A.; Haage, G.; Loidl-Lanthaler, K. Kinetics as a tool for polyhydroxyalkanoate production optimization. Can. J. Microbiol. 1995, 41, 239–248. [Google Scholar] [CrossRef]
- Braunegg, G.; Korneti, L. Pseudomonas 2 F: Kinetics of growth and accumulation of poly-d(-)-3-hydroxybutyric acid (poly-HB). Biotechnol. Lett. 1984, 6, 825–829. [Google Scholar] [CrossRef]
- Kaur, G.; Roy, I. Strategies for large-scale production of polyhydroxyalkanoates. Chem. Biochem. Eng. Q. 2015, 29, 157–172. [Google Scholar] [CrossRef]
- Rodriguez-Perez, S.; Serrano, A.; Pantión, A.A.; Alonso-Fariñas, B. Challenges of scaling-up PHA production from waste streams. A review. J. Environ. Manag. 2018, 205, 215–230. [Google Scholar] [CrossRef] [PubMed]
- Novak, M.; Koller, M.; Braunegg, M.; Horvat, P. Mathematical modelling as a tool for optimized PHA production. Chem. Biochem. Eng. Q. 2015, 29, 183–220. [Google Scholar] [CrossRef]
- Koller, M.; Vadljia, D.; Braunegg, G.; Atlić, A.; Horvat, P. Formal- and high-structured kinetic process modelling and footprint area analysis of binary imaged cells: Tools to understand and optimize multistage-continuous PHA biosynthesis. EuroBiotech J. 2017, 1, 203–211. [Google Scholar] [CrossRef]
- Cui, B.; Huang, S.; Xu, F.; Zhang, R.; Zhang, Y. Improved productivity of poly(3-hydroxybutyrate) (PHB) in thermophilic Chelatococcus daeguensis TAD1 using glycerol as the growth substrate in a fed-batch culture. Appl. Microbiol. Biotechnol. 2015, 99, 6009–6019. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, S.O.; Kanekar, P.P.; Nilegaonkar, S.S.; Sarnaik, S.S.; Jog, J.P. Production and characterization of a biodegradable poly(hydroxybutyrate-co-hydroxyvalerate) (PHB-co-PHV) copolymer by moderately haloalkalitolerant Halomonas campisalis MCM B-1027 isolated from Lonar Lake, India. Bioresour. Technol. 2010, 101, 9765–9771. [Google Scholar] [CrossRef] [PubMed]
- Ng, K.S.; Wong, Y.M.; Tsuge, T.; Sudesh, K. Biosynthesis and characterization of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) and poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) copolymers using jatropha oil as the main carbon source. Proc. Biochem. 2011, 46, 1572–1578. [Google Scholar] [CrossRef]
- Sindhu, R.; Silviya, N.; Binod, P.; Pandey, A. Pentose-rich hydrolysate from acid pretreated rice straw as a carbon source for the production of poly-3-hydroxybutyrate. Biochem. Eng. J. 2013, 78, 67–72. [Google Scholar] [CrossRef]
- Gahlawat, G.; Srivastava, A.K. Enhancing the production of polyhydroxyalkanoate biopolymer by Azohydromonas australica using a simple empty and fill bioreactor cultivation strategy. Chem. Biochem. Eng. Q. 2018, 31, 479–485. [Google Scholar] [CrossRef]
- Marang, L.; van Loosdrecht, M.C.; Kleerebezem, R. Enrichment of PHA-producing bacteria under continuous substrate supply. New Biotechnol. 2018, 41, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, M.G.E.; Martino, V.; Pollet, E.; Avérous, L.; Reis, M.A.M. Mixed culture polyhydroxyalkanoate (PHA) production from volatile fatty acid (VFA)-rich streams: Effect of substrate composition and feeding regime on PHA productivity, composition and properties. J. Biotechnol. 2011, 151, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Kourmentza, C.; Plácido, J.; Venetsaneas, N.; Burniol-Figols, A.; Varrone, C.; Gavala, H.N.; Reis, M.A. M Recent advances and challenges towards sustainable polyhydroxyalkanoate (PHA) production. Bioengineering 2017, 4, 55. [Google Scholar] [CrossRef] [PubMed]
- Raza, Z.A.; Abid, S.; Banat, I.M. Polyhydroxyalkanoates: Characteristics, production, recent developments and applications. Int. Biodeterior. Biodegrad. 2018, 126, 45–56. [Google Scholar] [CrossRef]
- Chen, Z.; Huang, L.; Wen, Q.; Guo, Z. Efficient polyhydroxyalkanoate (PHA) accumulation by a new continuous feeding mode in three-stage mixed microbial culture (MMC) PHA production process. J. Biotechnol. 2015, 209, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Reis, M.A.M.; Serafim, L.S.; Lemos, P.C.; Ramos, A.M.; Aguiar, F.R.; van Loosdrecht, M.C.M. Production of polyhydroxyalkanoates by mixed microbial cultures. Bioproc. Biosyst. Eng. 2003, 25, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Valentino, F.; Karabegovic, L.; Majone, M.; Morgan-Sagastume, F.; Werker, A. Polyhydroxyalkanoate (PHA) storage within a mixed-culture biomass with simultaneous growth as a function of accumulation substrate nitrogen and phosphorus levels. Water Res. 2015, 77, 49–63. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chen, Z.; Wen, Q.; Zhao, L.; Lee, D.J.; Yang, L.; Wang, Y. Insights into Feast-Famine polyhydroxyalkanoate (PHA)-producer selection: Microbial community succession, relationships with system function and underlying driving forces. Water Res. 2018, 131, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.S.; Silva, C.E.; Carvalho, G.; Reis, M.A.M. Strategies for efficiently selecting PHA producing mixed microbial cultures using complex feedstocks: Feast and famine regime and uncoupled carbon and nitrogen availabilities. New Biotechnol. 2017, 37, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Serafim, L.S.; Lemos, P.C.; Oliveira, R.; Reis, M.A.M. Optimization of polyhydroxybutyrate production by mixed cultures submitted to aerobic dynamic feeding conditions. Biotechnol. Bioeng. 2004, 87, 145–160. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, M.G.E.; Eiroa, M.; Torres, C.; Nunes, B.R.; Reis, M.A.M. Strategies for the development of a side stream process for polyhydroxyalkanoate (PHA) production from sugar cane molasses. J. Biotechnol. 2007, 130, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Dionisi, D.; Carucci, G.; Petrangeli Papinia, M.; Riccardi, C.; Majone, M.; Carrasco, F. Olive oil mill effluents as a feedstock for production of biodegradable polymers. Water Res. 2005, 39, 2076–2084. [Google Scholar] [CrossRef] [PubMed]
- Carucci, A.; Dionisi, D.; Majone, M.; Rolle, E.; Smurra, P. Aerobic storage by activated sludge on real wastewater. Water Res. 2001, 35, 3833–3844. [Google Scholar] [CrossRef]
- Carvalho, G.; Pedras, I.; Karst, S.M.; Oliveira, C.S.; Duque, A.F.; Nielsen, P.H.; Reis, M.A.M. Functional redundancy ensures performance robustness in 3-stage PHA-producing mixed cultures under variable feed operation. New Biotechnol. 2018, 40, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Rhu, D.H.; Lee, W.H.; Kim, J.Y.; Choi, E. Polyhydroxyalkanoate (PHA) production from waste. Water Sci. Technol. 2003, 48, 221–228. [Google Scholar] [PubMed]
- Burniol-Figols, A.; Varrone, C.; Le, S.B.; Daugaard, A.E.; Skiadas, I.V.; Gavala, H.N. Combined polyhydroxyalkanoates (PHA) and 1, 3-propanediol production from crude glycerol: Selective conversion of volatile fatty acids into PHA by mixed microbial consortia. Water Res. 2018, 136, 180–191. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wusiman, A.; Liu, X.; Wan, C.; Lee, D.J.; Tay, J. Polyhydroxyalkanoates (PHA) production from phenol in an acclimated consortium: Batch study and impacts of operational conditions. J. Biotechnol. 2018, 267, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Ahn, W.S.; Park, S.J.; Lee, S.Y. Production of poly(3-hydroxybutyrate) by fed-batch culture of recombinant Escherichia coli with a highly concentrated whey solution. Appl. Environ. Microbiol. 2000, 66, 3624–3627. [Google Scholar] [CrossRef] [PubMed]
- Ahn, W.S.; Park, S.J.; Lee, S.Y. Production of poly(3-hydroxybutyrate) from whey by cell recycle fed-batch culture of recombinant Escherichia coli. Biotechnol. Lett. 2001, 23, 235–240. [Google Scholar] [CrossRef]
- Da Cruz Pradella, J.G.; Ienczak, J.L.; Delgado, C.R.; Taciro, M.K. Carbon source pulsed feeding to attain high yield and high productivity in poly(3-hydroxybutyrate) (PHB) production from soybean oil using Cupriavidus necator. Biotechnol. Lett. 2012, 34, 1003–1007. [Google Scholar] [CrossRef] [PubMed]
- Hermann-Krauss, C.; Koller, M.; Muhr, A.; Fasl, H.; Stelzer, F.; Braunegg, G. Archaeal production of polyhydroxyalkanoate (PHA) co-and terpolyesters from biodiesel industry-derived by-products. Archaea 2013, 2013, 129268. [Google Scholar] [CrossRef] [PubMed]
- Muhr, A.; Rechberger, E.M.; Salerno, A.; Reiterer, A.; Schiller, M.; Kwiecień, M.; Adamus, G.; Kowalczuk, M.; Stromeier, K.; Schober, S.; et al. Biodegradable latexes from animal-derived waste: Biosynthesis and characterization of mcl-PHA accumulated by Ps. citronellolis. React. Funct. Polym. 2013, 73, 1391–1398. [Google Scholar] [CrossRef]
- Muhr, A.; Rechberger, E.M.; Salerno, A.; Reiterer, A.; Malli, K.; Strohmeier, K.; Schober, S.; Mittelbach, M.; Koller, M. Novel description of mcl-PHA biosynthesis by Pseudomonas chlororaphis from animal-derived waste. J. Biotechnol. 2013, 165, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Follonier, S.; Riesen, R.; Zinn, M. Pilot-scale production of functionalized mcl-PHA from grape pomace supplemented with fatty acids. Chem. Biochem. Eng. Q. 2015, 29, 113–121. [Google Scholar] [CrossRef]
- Ienczak, J.L.; Quines, L.K.; De Melo, A.A.; Brandellero, M.; Mendes, C.R.; Schmidell, W.; Aragão, G.M.F. High cell density strategy for poly(3-hydroxybutyrate) production by Cupriavidus necator. Braz. J. Chem. Eng. 2011, 28, 585–596. [Google Scholar] [CrossRef]
- Ienczak, J.L.; Schmidell, W.; de Aragão, G.M.F. High-cell-density culture strategies for polyhydroxyalkanoate production: A review. J. Ind. Microbiol. Biotechnol. 2013, 40, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Penloglou, G.; Vasileiadou, A.; Chatzidoukas, C.; Kiparissides, C. Model-based intensification of a fed-batch microbial process for the maximization of polyhydroxybutyrate (PHB) production rate. Bioproc. Biosyst. Eng. 2017, 40, 1247–1260. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Benesova, P.; Marsalek, L.; Marova, I. Use of lignocellulosic materials for PHA production. Chem. Biochem. Eng. Q. 2015, 29, 135–144. [Google Scholar] [CrossRef]
- Kucera, D.; Benesova, P.; Ladicky, P.; Pekar, M.; Sedlacek, P.; Obruca, S. Production of polyhydroxyalkanoates using hydrolyzates of spruce sawdust: Comparison of hydrolyzates detoxification by application of overliming, active carbon, and lignite. Bioengineering 2017, 4, 53. [Google Scholar] [CrossRef] [PubMed]
- Ienczak, J.L.; Schmidt, M.; Quines, L.K.; Zanfonato, K.; da Cruz Pradella, J.G.; Schmidell, W.; de Aragao, G.M.F. Poly(3-hydroxybutyrate) production in repeated fed-batch with cell recycle using a medium with low carbon source concentration. Appl. Biochem. Biotechnol. 2016, 178, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Haas, C.; El-Najjar, T.; Virgolini, N.; Smerilli, M.; Neureiter, M. High cell-density production of poly(3-hydroxybutyrate) in a membrane bioreactor. New Biotechnol. 2017, 37, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Lorantfy, B.; Ruschitzka, P.; Herwig, C. Investigation of physiological limits and conditions for robust bioprocessing of an extreme halophilic archaeon using external cell retention system. Biochem. Eng. J. 2014, 90, 140–148. [Google Scholar] [CrossRef]
- Ibrahim, M.H.; Steinbüchel, A. High-cell-density cyclic fed-batch fermentation of a poly(3-hydroxybutyrate)-accumulating thermophile, Chelatococcus sp. strain MW10. Appl. Environ. Microbiol. 2010, 76, 7890–7895. [Google Scholar] [CrossRef] [PubMed]
- Du, G.; Yu, J. Green technology for conversion of food scraps to biodegradable thermoplastic polyhydroxyalkanoates. Environ. Sci. Technol. 2002, 36, 5511–5516. [Google Scholar] [CrossRef] [PubMed]
- Khosravi-Darani, K.; Mokhtari, Z.B.; Amai, T.; Tanaka, K. Microbial production of poly(hydroxybutyrate) from C1 carbon sources. Appl. Microbiol. Biotechnol. 2013, 97, 1407–1424. [Google Scholar] [CrossRef] [PubMed]
- Rostkowski, K.H.; Criddle, C.S.; Lepech, M.D. Cradle-to-gate life cycle assessment for a cradle-to-cradle cycle: Biogas-to-bioplastic (and back). Environ. Sci. Technol. 2012, 46, 9822–9829. [Google Scholar] [CrossRef] [PubMed]
- Myung, J.; Galega, W.M.; van Nostrand, J.D.; Yuan, T.; Zhou, J.; Criddle, C.S. Long-term cultivation of a stable Methylocystis-dominated methanotrophic enrichment enabling tailored production of poly(3-hydroxybutyrate-co-3-hydroxyvalerate). Bioresour. Technol. 2015, 198, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Cal, A.J.; Sikkema, W.D.; Ponce, M.I.; Franqui-Villanueva, D.; Riiff, T.J.; Orts, W.J.; Pieja, A.J.; Lee, C.C. Methanotrophic production of polyhydroxybutyrate-co-hydroxyvalerate with high hydroxyvalerate content. Int. J. Biol. Macromol. 2016, 87, 302–307. [Google Scholar] [CrossRef] [PubMed]
- López, J.C.; Arnáiz, E.; Merchán, L.; Lebrero, R.; Muñoz, R. Biogas-based polyhydroxyalkanoates production by Methylocystis hirsuta: A step further in anaerobic digestion biorefineries. Chem. Eng. J. 2018, 333, 529–536. [Google Scholar] [CrossRef]
- García-Pérez, T.; López, J.C.; Passos, F.; Lebrero, R.; Revah, S.; Muñoz, R. Simultaneous methane abatement and PHB production by Methylocystis hirsuta in a novel gas-recycling bubble column bioreactor. Chem. Eng. J. 2018, 334, 691–697. [Google Scholar] [CrossRef]
- Drosg, B.; Fritz, I.; Gattermayr, F.; Silvestrini, L. Photo-autotrophic production of poly (hydroxyalkanoates) in cyanobacteria. Chem. Biochem. Eng. Q. 2015, 29, 145–156. [Google Scholar] [CrossRef]
- Koller, M.; Maršalek, L. Cyanobacterial polyhydroxyalkanoate production: Status quo and Quo vadis? Curr. Biotechnol. 2015, 4, 464–480. [Google Scholar] [CrossRef]
- Ghysels, S.; Mozumder, M.S.I.; De Wever, H.; Volcke, E.I.; Garcia-Gonzalez, L. Targeted poly(3-hydroxybutyrate-co-3-hydroxyvalerate) bioplastic production from carbon dioxide. Bioresour. Technol. 2018, 249, 858–868. [Google Scholar] [CrossRef] [PubMed]
- Roberts, M. Screening and Selection of a Cyanobacteria for Production of poly-β-hydroxybutyrate in a Closed Photobioreactor. Ph.D. Thesis, University of Adelaide, Adelaide, Australia, 2009. [Google Scholar]
- Troschl, C.; Meixner, K.; Drosg, B. Cyanobacterial PHA Production—Review of Recent Advances and a Summary of Three Years’ Working Experience Running a Pilot Plant. Bioengineering 2017, 4, 26. [Google Scholar] [CrossRef] [PubMed]
- Kovalcik, A.; Meixner, K.; Mihalic, M.; Zeilinger, W.; Fritz, I.; Fuchs, W.; Kucharczyk, P.; Stelzer, F.; Drosg, B. Characterization of polyhydroxyalkanoates produced by Synechocystis salina from digestate supernatant. Int. J. Biol. Macromol. 2017, 102, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Meixner, K.; Kovalcik, A.; Sykacek, E.; Gruber-Brunhumer, M.; Zeilinger, W.; Markl, K.; Haas, C.; Fritz, I.; Mundigler, N.; Stelzer, F.; et al. Cyanobacteria biorefinery—Production of poly (3-hydroxybutyrate) with Synechocystis salina and utilisation of residual biomass. J. Biotechnol. 2018, 265, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, C.K.; Costa, J.A.V.; de Morais, M.G. Polyhydroxybutyrate (PHB) synthesis by Spirulina sp. LEB 18 using biopolymer extraction waste. Appl. Biochem. Biotechnol. 2018, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Koller, M. Recycling of waste streams of the biotechnological poly(hydroxyalkanoate) production by Haloferax mediterranei on whey. Int. J. Polym. Sci. 2015, 2015, 370164. [Google Scholar] [CrossRef]
- Chaudhari, S.T.; Dalai, A.K.; Bakhshi, N.N. Production of hydrogen and/or syngas (H2 + CO) via steam gasification of biomass-derived chars. Energy Fuel. 2003, 17, 1062–1067. [Google Scholar] [CrossRef]
- Revelles, O.; Calvillo, I.; Prieto, A.; Prieto, M.A. Syngas Fermentation for Polyhydroxyalkanoate Production in Rhodospirillum rubrum. In Hydrocarbon and Lipid Microbiology Protocols, 1st ed.; McGenity, T., Timmis, K., Nogales, B., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 105–119. ISBN 978-3-662-491317. [Google Scholar]
- Do, Y.S.; Smeenk, J.; Broer, K.M.; Kisting, C.J.; Brown, R.; Heindel, T.J.; Bobik, T.A.; DiSpririto, A.A. Growth of Rhodospirillum rubrum on synthesis gas: Conversion of CO to H2 and poly-β-hydroxyalkanoate. Biotechnol. Bioeng. 2007, 97, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.L.; West, T.P.; Gibbons, W.R. Rhodospirillum rubrum: Utilization of condensed corn solubles for poly-(3-hydroxybutyrate-co-3-hydroxyvalerate) production. J. Appl. Microbiol. 2008, 104, 1488–1494. [Google Scholar] [CrossRef] [PubMed]
- Revelles, O.; Beneroso, D.; Menendez, J.A.; Arenillas, A.; García, J.L.; Prieto, M.A. Syngas obtained by microwave pyrolysis of household wastes as feedstock for polyhydroxyalkanoate production in Rhodospirillum rubrum. Microb. Biotechnol. 2017, 10, 1412–1417. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.; Chipman, D.C.; Bents, S.C.; Brown, R.C. A techno-economic analysis of polyhydroxyalkanoate and hydrogen production from syngas fermentation of gasified biomass. Appl. Biochem. Biotechnol. 2014, 160, 1032–1046. [Google Scholar] [CrossRef] [PubMed]
- Karmann, S.; Follonier, S.; Egger, D.; Hebel, D.; Panke, S.; Zinn, M. Tailor-made PAT platform for safe syngas fermentations in batch, fed-batch and chemostat mode with Rhodospirillum rubrum. Microb. Biotechnol. 2017, 10, 1365–1375. [Google Scholar] [CrossRef] [PubMed]
- Klask, C.; Raberg, M.; Heinrich, D.; Steinbüchel, A. Heterologous expression of various PHA synthase genes in Rhodospirillum rubrum. Chem. Biochem. Eng. Q. 2015, 29, 75–85. [Google Scholar] [CrossRef]
- Heinrich, D.; Raberg, M.; Fricke, P.; Kenny, S.T.; Morales-Gamez, L.; Babu, R.P.; O’Connor, K.E.; Steinbüchel, A. Synthesis gas (Syngas)-derived medium-chain-length polyhydroxyalkanoate synthesis in engineered Rhodospirillum rubrum. Appl. Environ. Microbiol. 2016, 82, 6132–6140. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Muhr, A. Continuous production mode as a viable process-engineering tool for efficient poly(hydroxyalkanoate) (PHA) bio-production. Chem. Biochem. Eng. Q. 2014, 28, 65–77. [Google Scholar]
- Koller, M.; Braunegg, G. Potential and prospects of continuous polyhydroxyalkanoate (PHA) production. Bioengineering 2015, 2, 94–121. [Google Scholar] [CrossRef] [PubMed]
- Senior, P.J.; Beech, G.A.; Ritchie, G.A.F.; Dawes, E.A. The role of oxygen limitation in the formation of poly-β-hydroxybutyrate during batch and continuous culture of Azotobacter beijerinckii. Biochem. J. 1972, 128, 1193. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, B.A.; Lomaliza, K.; Chavarie, C.; Dube, B.; Bataille, P.; Ramsay, J.A. Production of poly-(beta-hydroxybutyric-co-beta-hydroxyvaleric) acids. Appl. Environ. Microbiol. 1990, 56, 2093–2098. [Google Scholar] [PubMed]
- Zinn, M.; Witholt, B.; Egli, T. Dual nutrient limited growth: Models, experimental observations, and applications. J. Biotechnol. 2004, 113, 263–279. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.; Hazenberg, W.; Prieto, M.; Witholt, B. Two-stage continuous process development for the production of medium-chain-length poly(3-hydroxyalkanoates). Biotechnol. Bioeng. 2001, 72, 19–24. [Google Scholar] [CrossRef]
- Durner, R.; Witholt, B.; Egli, T. Accumulation of poly[(R)-3-hydroxyalkanoates] in Pseudomonas oleovorans during growth with octanoate in continuous culture at different dilution rates. Appl. Environ. Mmicrobiol. 2000, 66, 3408–3414. [Google Scholar] [CrossRef]
- Durner, R.; Zinn, M.; Witholt, B.; Egli, T. Accumulation of poly[(R)-3-hydroxyalkanoates] in Pseudomonas oleovorans during growth in batch and chemostat culture with different carbon sources. Biotechnol. Bioeng. 2001, 72, 278–288. [Google Scholar] [CrossRef]
- Zinn, M.; Weilenmann, H.U.; Hany, R.; Schmid, M.; Egli, T.H. Tailored synthesis of poly([R]-3-hydroxybutyrate-co-3-hydroxyvalerate) (PHB/HV) in Ralstonia eutropha DSM 428. Eng. Life Sci. 2003, 23, 309–316. [Google Scholar] [CrossRef]
- Hartmann, R.; Hany, R.; Geiger, T.; Egli, T.; Witholt, B.; Zinn, M. Tailored biosynthesis of olefinic medium-chain-length Poly[(R)-3-hydroxyalkanoates] in Pseudomonas putida GPo1 with improved thermal properties. Macromolecules 2004, 37, 6780–6785. [Google Scholar] [CrossRef]
- Hany, R.; Brinkmann, M.; Ferri, D.; Hartmann, R.; Pletscher, E.; Rentsch, D.; Zinn, M. Crystallization of an aromatic biopolyester. Macromolecules 2009, 42, 6322–6326. [Google Scholar] [CrossRef]
- Yin, J.; Chen, J.C.; Wu, Q.; Chen, G.Q. Halophiles, coming stars for industrial biotechnology. Biotechnol. Adv. 2015, 33, 1433–1442. [Google Scholar] [CrossRef] [PubMed]
- Lillo, J.G.; Rodriguez-Valera, F. Effects of culture conditions on poly(β-hydroxybutyric acid) production by Haloferax mediterranei. Appl. Environ. Microb. 1990, 56, 2517–2521. [Google Scholar]
- Yue, H.; Ling, C.; Yang, T.; Chen, X.; Chen, Y.; Deng, H.; Wu, Q.; Chen, J.; Chen, G.Q. A seawater-based open and continuous process for polyhydroxyalkanoates production by recombinant Halomonas campaniensis LS21 grown in mixed substrates. Biotechnol. Biofuels 2014, 7, 108–119. [Google Scholar] [CrossRef]
- Du, G.; Chen, J.; Yu, J.; Lun, S. Continuous production of poly-3-hydroxybutyrate by Ralstonia eutropha in a two-stage culture system. J. Biotechnol. 2001, 88, 59–65. [Google Scholar] [CrossRef]
- Mothes, G.; Ackermann, J.U. Synthesis of poly(3-hydroxybutyrate-co-4-hydrobutyrate) with a target mole fraction of 4-hydroxybutyric acid units by two-stage continuous cultivation of Delftia acidovorans P4a. Eng. Life Sci. 2005, 5, 58–62. [Google Scholar] [CrossRef]
- Hartmann, R.; Hany, R.; Witholt, B.; Zinn, M. Simultaneous biosynthesis of two copolymers in Pseudomonas putida GPo1 using a two-stage continuous culture system. Biomacromolecules 2010, 11, 1488–1493. [Google Scholar] [CrossRef] [PubMed]
- Pederson, E.N.; McChalicher, C.W.; Srienc, F. Bacterial synthesis of PHA block copolymers. Biomacromolecules 2006, 7, 1904–1911. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Xue, Y.S.; Aibaidula, G.; Chen, G.Q. Unsterile and continuous production of polyhydroxybutyrate by Halomonas TD01. Bioresour. Technol. 2011, 102, 8130–8136. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Wu, Q.; Chen, J.C.; Chen, G.Q. Engineering Halomonas TD01 for the low-cost production of polyhydroxyalkanoates. Metab. Eng. 2014, 26, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Atlić, A.; Koller, M.; Scherzer, D.; Kutschera, C.; Grillo-Fernandes, E.; Horvat, P.; Chiellini, E.; Braunegg, G. Continuous production of poly([R]-3-hydroxybutyrate) by Cupriavidus necator in a multistage bioreactor cascade. Appl. Microbiol. Biotechnol. 2011, 91, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Horvat, P.; Vrana Špoljarić, I.; Lopar, M.; Atlić, A.; Koller, M.; Braunegg, G. Mathematical modelling and process optimization of a continuous 5-stage bioreactor cascade for production of poly[-(R)-3-hydroxybutyrate] by Cupriavidus necator. Bioproc. Biosyst. Eng. 2013, 36, 1235–1250. [Google Scholar] [CrossRef] [PubMed]
- Lopar, M.; Vrana Špoljarić, I.; Atlić, A.; Koller, M.; Braunegg, G.; Horvat, P. Five-step continuous production of PHB analyzed by elementary flux, modes, yield space analysis and high structured metabolic model. Biochem. Eng. J. 2013, 79, 57–70. [Google Scholar] [CrossRef]
- Vadlja, D.; Koller, M.; Novak, M.; Braunegg, G.; Horvat, P. Footprint area analysis of binary imaged Cupriavidus necator cells to study PHB production at balanced, transient, and limited growth conditions in a cascade process. Appl. Microbiol. Biotechnol. 2016, 100, 10065–10080. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).