Abstract
The study evaluated the effects of replacing soybean meal (SBM) with marula oilcake (MOC) at equal inclusion (10% fresh weight) levels in whole-crop maize silage treated with or without lactic acid bacteria inoculants on fermentation characteristics, nutritive value, aerobic stability, and in vitro nutrient degradability. Maize was ensiled with SBM or MOC in a non-iso-nitrogenous 2 × 3 factorial design and either inoculated or uninoculated with Lalsil Fresh or Sil-All 4×4 for 90 days. Protein sources differed significantly (p < 0.05). The MOC showed high DM, EE, GE, and ADL, whereas SBM had high CP, ash, and IVOMD. Fibre fractions (aNDF and ADF) were similar (p > 0.05). The SBM control showed significantly high (p < 0.05) LA, NH3-N, CP, IVOMD, propionic acid, and early gas production, indicating efficient fermentation. The SBM + Lalsil maintained low pH, and early OM, CP, and GE degradability. The SBM + Sil-All achieved the highest (p < 0.05) OM, NDF, and ADF degradability and acetic acid production than other treatments. The MOC control showed low (p < 0.05) pH, high fibre and GE, reduced butyric acid, and low 48 h gas production, indicating slower fermentation but improved stability. The MOC + Lalsil had high (p < 0.05) DM, low CO2 and yeasts and moulds, and the highest (p < 0.05) CP degradability, propionic acid, and peak gas production at 12 h. The MOC + Sil-All showed high (p < 0.05) GE and WSC with peak GE degradation at 12 h, but low NDF degradability and reduced gas production. Overall, SBM improved degradability and fermentation efficiency, particularly with Sil-All, whereas MOC enhanced energy density and aerobic stability, with Lalsil optimising protein utilisation. Matching inoculant type to protein source is essential to optimise silage quality and rumen fermentation. Further research should assess different inoculant inclusion rates and include a maize-only control, and evaluate protein source inclusion under iso-nitrogenous conditions to allow more accurate comparisons.
1. Introduction
Forage-based diets underpin ruminant production systems by supplying most nutrients required for animal maintenance, growth, and production. In most ruminant systems, forages contribute between 40 and 90% of total feed intake, highlighting their central role in livestock productivity and sustainability [1,2]. In semi-arid and seasonally variable regions, such as Southern Africa, the preservation of surplus forage during the rainy season is critical to mitigate feed shortages during dry periods and recurrent droughts [3].
Among forage conservation strategies, ensiling has gained prominence over haymaking due to its ability to preserve large quantities of forage rapidly, with reduced reliance on favourable weather conditions and improved palatability resulting from earlier harvest stages [4]. Silage preservation is driven by anaerobic fermentation, during which lactic acid bacteria (LAB) convert water-soluble carbohydrates (WSC) into organic acids, primarily lactic acid, resulting in a rapid decline in pH that stabilises the ensiled biomass [5]. However, the success of this process depends on crop characteristics, microbial dynamics, and management interventions.
Maize (Zea mays L.) is the dominant silage crop in Southern Africa, with South Africa being the principal producer in the region [6]. Its high WSC concentration and favourable fermentability make maize particularly suitable for ensiling and is therefore widely adopted in intensive and semi-intensive livestock systems [7]. Nevertheless, maize silage presents inherent limitations. High lactic acid concentrations and residual sugars, although indicative of efficient primary fermentation, have been strongly associated with poor aerobic stability during feed-out due to rapid proliferation of yeasts and moulds [8]. This challenge is exacerbated under warm climatic conditions, where spoilage microorganisms thrive at temperatures between 20 and 30 °C [9].
In addition to stability concerns, maize silage is intrinsically low in crude protein (CP), typically containing less than 100 g CP/kg dry matter (DM) [10]. This necessitates supplementation with external protein sources to meet ruminant nutritional requirements, particularly in production systems where protein-rich forages are scarce. While soybean meal (SBM) is widely used for this purpose, its increasing cost and reliance on imports have driven interest in alternative plant-based protein supplements. The use of urea has also been explored; however, its narrow safety margin and risk of toxicity limit its practical application.
Microbial inoculants are commonly employed to improve silage fermentation efficiency and aerobic stability. Homofermentative LAB are primarily used to enhance lactic acid production and accelerate pH decline, thereby suppressing undesirable microorganisms [11,12]. In contrast, heterofermentative LAB convert lactic acid to acetic acid and other metabolites that inhibit yeasts and moulds, improving aerobic stability but sometimes at the expense of DM recovery [13]. The relative merits of these inoculants remain debated, particularly when silage composition is altered through nutrient supplementation, as conflicting results have been reported regarding fermentation efficiency, nutrient preservation, and stability.
Agro-industrial by-products with moderate to high CP content have emerged as potential local alternatives to conventional protein supplements. The Sclerocarya birrea spp. (Marula) oilcake (MOC), a by-product of marula oil extraction, contains 340–370 g CP/kg DM and is comparable to other commercial oilseed meals such as canola and sunflower oilcakes [14,15,16]. Despite its nutritional potential, limited information exists on how MOC interacts with maize silage fermentation, particularly when combined with microbial inoculants. The effects of such supplementation on fermentation pathways, aerobic stability, and nutrient digestibility remain poorly understood.
The present study aimed to evaluate the effects of replacing SBM with MOC, with or without microbial inoculation, on the fermentation characteristics, aerobic stability, and in vitro nutrient digestibility of maize silage. By integrating established fermentation theory with emerging alternative protein strategies, this work provides new insight into how silage additives and supplements interact to shape fermentation outcomes.
2. Materials and Methods
2.1. Ethical Approval
All experimental procedures were approved by the Animal Ethics Committee of the Agricultural Research Council–Animal Production (ARC-AP), Irene Campus (APIEC 26/08), and the Tshwane University of Technology Animal Ethics Committee (PO24/04).
2.2. Study Site
Whole-crop maize (Zea mays L.) was planted late October 2024 at Irene Campus, Pretoria, South Africa (longitude 28°12′50″ E, latitude 25°54′0″ S, altitude 1512 m above sea level), and harvested end of February 2025. The maize was harvested at <38% DM using a Feraboli 945 forage harvester (Fondata Nel, Cremona, Italy) adjusted to obtain a 5 mm theoretical chop length. The chopped maize was used to produce maize silage at the ARC–Animal Production, Irene Campus, South Africa.
2.3. Silage Production and Experimental Treatments
Chopped whole-crop maize was divided into two batches (81 kg each). Batch 1 was mixed with 9 kg of SBM, while batch 2 was mixed with 9 kg of marula oilcake (MOC) as a replacement for SBM. Both SBM and MOC were incorporated at an equal inclusion rate without adjustment for CP equivalence. This non-iso-nitrogenous design was intentional, as the study aimed to evaluate the effects of replacing a conventional protein source (SBM; approximately 48% CP) with an alternative agro-industrial by-product (MOC; approximately 34% CP) at equal inclusion levels rather than equivalent nitrogen supply.
From each batch, 30 kg of maize containing SBM was treated by spraying while constantly mixing with either a heterofermentative LAB inoculant, Lalsil Fresh (Lactobacillus buchneri NCIMB 40788; Lallemand Animal Nutrition, Blagnac Cedex, France), or a homofermentative LAB inoculant, Sil-All 4×4® (Lactobacillus plantarum, Pediococcus acidilactici, and Propionibacterium acidipropionici; Danstar Ferment AG, Zug, Switzerland). Inoculants were applied proportionally to forage weight according to manufacturer recommendations. For Lalsil Fresh, 0.15 g inoculant was dissolved in 60 mL of water and treated to 30 kg of fresh whole-crop maize, based on an application rate of 5 g in 2 L water per tonne of forage. For Sil-All 4×4®, 0.06 g inoculant was dissolved in 30 mL of water and applied to 30 kg of fresh whole-crop maize, following the recommended rate of 2 g in 1 L water per tonne of forage.
To standardise moisture addition across the forages, the remaining 30 kg of chopped whole-crop maize received an equivalent volume of distilled water (30 mL per 30 kg fresh forage). Batch 2 (maize containing MOC) was handled similarly to batch 1 with 30 kg sprayed with Lalsil Fresh, another 30 kg with Sil-All 4×4®, and the remaining maize sprayed with distilled water. This experimental setup resulted in 6 treatments, corresponding to 2 protein sources (SBM or MOC) and 3 inoculants (Lalsil Fresh, Sil-All 4×4®, or distilled water as control). Representative samples (800 g) were collected from each treatment prior to ensiling for determination of chemical composition (proximate, fibre fraction as well as WSC, pH, LAB, lactic acid (LA) and yeast and mould (Y & M). Following sampling, each 30 kg of the treated whole-crop maize from both batches were compacted into a total of 18 ensiling jars (1 L). Each treatment was ensiled in triplicate (three jars per treatment), and samples from the jars were used to evaluate fermentation characteristics, nutrient composition, aerobic stability, and in vitro nutrient degradability. In addition to the 18 jars used for fermentation, nutrient and in vitro analyses, intermittent pH measurements were conducted on separate jars on days 3, 7, and 14 of ensiling. For each of the six treatments, three replicate jars were sampled per day, resulting in 54 additional jars (6 treatments × 3 replicates × 3 sampling days), giving a total of 72 jars for the entire experiment. The ensiling jars were considered the experimental units.
The experiment was conducted as a 2 × 3 factorial design in a completely randomised design (CRD) with protein sources (SBM or MOC) and inoculants (Lalsil Fresh, Sil-All® or water as control) as main factors.
The six treatments were prepared as follows:
- Treatment 1: Maize silage supplemented with SBM (positive control).
- Treatment 2: Maize silage supplemented with SBM and inoculated with Lalsil Fresh.
- Treatment 3: Maize silage supplemented with SBM and inoculated with Sil-All 4×4®.
- Treatment 4: Maize silage supplemented with MOC (negative control).
- Treatment 5: Maize silage supplemented with MOC and inoculated with Lalsil Fresh.
- Treatment 6: Maize silage supplemented with MOC and inoculated with Sil-All 4×4®.
Batch 1 was used to prepare treatments 1 to 3, while batch 2 was used to prepare treatments 4 to 6.
2.4. Chemical Composition
2.4.1. Nutrient Composition
Samples of fresh maize collected pre-ensiling, and of silage collected post-ensiling from replicate jars per treatment, were analysed for DM, CP, ether extract (EE), gross energy (GE), organic matter (OM), and fibre fractions [neutral detergent fibre (aNDF), acid detergent fibre (ADF) and acid detergent lignin (ADL)] using AOAC procedures [17,18]. In brief, DM content was determined by oven-drying at 105 °C to constant weight according to [17]. The dried samples were subsequently ground using a Wiley mill (Standard Model 3, Arthur H. Thomas Co., Philadelphia, PA, USA) fitted with a 1 mm screen prior to further analyses.
The CP was determined using the Kjeldahl method following AOAC method 968.06 (1990) [17], while EE was analysed according to [18]. A bomb calorimeter (MC-1000, Energy Instrumentation, Knoppieslaagte, Centurion, South Africa) was used to measure GE. Ash was analysed according to the [17] by heating 2 g of each sample in a muffle furnace at 550 °C overnight to determine OM content in accordance with [17].
Neutral detergent fibre, ADF and ADL contents were analysed following the procedures described by [19]. The aNDF fraction was determined using a heat-stable α-amylase and a Tecator Fibre System (Fibertec™, Tecator, Höganäs, Sweden). Approximately 1 g of each sample was refluxed in 100 mL of neutral detergent solution at 100 °C for 60 min, followed by filtration and rinsing with hot distilled water. Residues were oven-dried at 100 °C overnight, cooled in a desiccator, and weighed.
The ADF fraction was determined using the same procedure as aNDF, except that acid detergent solution was used in place of the neutral detergent solution. The ADL was determined by treating the ADF residue with 72% sulfuric acid for 3 h at room temperature, followed by thorough rinsing with hot distilled water. The remaining residue was then oven-dried at 100 °C for 12 h, cooled, and weighed.
2.4.2. Fermentation Characteristics
For determination of fermentation characteristics, triplicate 40 g samples were collected from both the pre-ensiled maize and from each ensiled treatment. Each sample was homogenized with 360 mL of distilled water in a stomacher bag for 4 min. The pH of each sample was measured using an Accumet pH meter (AB150; Fisher Scientific, Ottawa, ON, Canada) at days 0 (on pre-ensiled fresh maize), 3, 7, 14, 90 and 95 during aerobic exposure. Samples evaluated for pH during day 0 and 90 were then allowed to stand for 24 h at 10 °C before being homogenized again for 4 min. Each sample was filtered through Whatman No. 54 filter paper (G.I.C. Scientific, Midrand, South Africa) to obtain the liquid extract.
The water-soluble carbohydrate (WSC) was determined using the phenol–sulphuric acid method. An aliquot of the filtered extract was mixed with 5% (w/v) phenol solution, followed by rapid addition of concentrated sulphuric acid. The mixture was allowed to react at room temperature for 10 min, after which the absorbance was measured at 490 nm using a double beam UV–VIS Spectrophotometer (Model: LMSP-UV1900; Labman Instruments, Chennai, India). Dextrose standards were used to construct a calibration curve, and WSC concentrations were calculated accordingly.
Ammonia nitrogen (NH3–N) concentration was determined by steam distillation according to [17], based on the volatile nitrogen procedure of [20]. In brief, an aliquot of the silage extract was made alkaline with magnesium oxide (MgO) and steam-distilled using a Büchi 342 distillation unit (Büchi Labortechnik AG, Flawil, Switzerland). The released ammonia was trapped in a boric acid solution containing a mixed indicator and subsequently titrated with standard hydrochloric acid using a Metrohm 655 Dosimat coupled to an E526 titrator (Metrohm AG, Herisau, Switzerland). Ammonia-N concentration was expressed as g NH3-N per kg total nitrogen (TN).
Volatile fatty acid concentrations in maize silage were determined using a Hewlett-Packard 5700A gas–liquid chromatograph (Hewlett-Packard, Palo Alto, CA, USA) equipped with dual-flame ionisation detectors, a Recordall recorder, and a Spectra-Physics Autolab Minigrator integrator, following the procedure of [21]. Separation was achieved using dual glass columns (1.5 m × 2 mm i.d.) packed with 3% PEGPE 3000 on TPA (60–80 mesh), with packing extending to within 10 cm of the column inlet. Triplicate samples (100 g) of oven-dried and ground maize silage from each treatment were mixed with 200 mL of distilled water in 800 mL glass jars, sealed, and stored at 3 °C for 24 h with intermittent shaking to facilitate extraction. Extracts were strained through two layers of cheesecloth and filtered through Whatman No. 42 filter paper. An aliquot (5 µL) of each filtrate was injected directly into the chromatographic column. Columns were conditioned at 190 °C for 24 h prior to analysis. During chromatographic runs, the oven temperature was initially held at 120 °C for 2 min, ramped to 190 °C at 16 °C/min, and maintained at 190 °C for 3 min. Nitrogen was used as the carrier gas at 40 mL/min. Injector and detector temperatures were maintained at 200 °C and 250 °C, respectively. Standard solutions of acetic, propionic, n-butyric, iso-butyric, n-valeric, and iso-valeric acids were prepared from certified reagent-grade chemicals to calibrate the system.
Lactic acid concentration was determined following VFA analysis using the colorimetric method of [22], as modified by [23]. Briefly, 7.9 mL of precipitating reagent was transferred into a centrifuge tube, followed by the addition of 0.10 mL of sample fluid. The mixture was centrifuged at 2000 rpm for 5 min. Thereafter, 1 mL of the supernatant was transferred into a test tube, and 6 mL of concentrated sulphuric acid was rapidly added. The mixture was allowed to stand for 2 min, mixed thoroughly, and cooled under running tap water for 2–3 min to below 20 °C. Subsequently, 0.1 mL of p-hydroxydiphenyl solution was added, mixed, and allowed to react for 10 min. The tube was then placed in a boiling water bath for 90 s, cooled again, and absorbance was measured spectrophotometrically at 564 nm using a V-10 visible spectrophotometer (ONDA, Giorgio Bormac S.r.l., Carpi, Italy) with 1 cm path-length cuvettes. The method is based on the conversion of lactic acid to acetaldehyde upon heating with sulphuric acid, followed by reaction with p-hydroxydiphenyl to form a coloured complex quantified by absorbance. Dissolving p-hydroxydiphenyl in dimethylformamide instead of alkali yields a reagent with indefinite stability [23].
2.4.3. Microbial Analysis
Microbial populations in fresh maize and silage samples were determined according to [24] for the enumeration of lactic acid bacteria (LAB) and fungi. Triplicate samples (10 g) from the pre-ensiled maize and from each ensiled treatment were weighed and homogenised in 90 mL of sterile buffered peptone water containing 0.85% (w/v) sodium chloride to obtain an initial 10−1 dilution. The suspensions were thoroughly mixed using a homogeniser. Serial decimal dilutions (10−1 to 10−9) were then prepared by transferring 1 mL of the homogenate into 9 mL of sterile maximum recovery diluent. From appropriate dilutions, 1 mL aliquots were plated onto selective media for microbial enumeration. Lactic acid bacteria were enumerated on de Man, Rogosa and Sharpe (MRS) agar using the pour-plate technique in accordance with ISO 15214:1998 [24]. Plates were incubated under microaerophilic conditions using CampyGen gas-generating packs (2.5 L) at 37 °C for 72 h. Yeasts and moulds were enumerated on Dichloran Rose-Bengal-Chloramphenicol (DRBC) agar using the pour-plate method, following the ISO method for fungal counts (ISO 21527-2:2008 [25]). Plates were incubated aerobically at 25 °C for 3–5 days. After incubation, colonies were counted on plates containing between 30 and 300 colony-forming units (CFU). Final microbial counts were calculated from the mean values of countable dilution plates. Yeasts and moulds were differentiated based on colony morphology, with yeasts forming smooth unicellular colonies and moulds exhibiting filamentous growth. Microbial populations were expressed as CFU per millilitre (CFU/mL).
2.4.4. Aerobic Stability
Aerobic stability of the silage was evaluated using triplicate 500 g samples from each treatment, which were loosely packed into open plastic jars, with the laboratory temperature maintained at 25 °C. The jars were covered with two layers of cheesecloth to allow air penetration while preventing contamination and were stored at room temperature. Aerobic stability was assessed by exposing the silage samples to air for a period of five days, during which temperature changes, pH variation, and carbon dioxide (CO2) production were monitored. The pH was determined from silage aliquots using an Accumet pH meter. Carbon dioxide production was quantified using a 20% potassium hydroxide (KOH) solution according to the method of [26]. The amount of CO2 absorbed by the KOH solution was determined by titration with 1 N hydrochloric acid. Aerobic stability was defined as the time (h) required for the silage temperature to increase by ≥2 °C above ambient temperature [26].
2.5. In Vitro Nutrient Digestibility and Gas Production
In vitro organic matter digestibility (IVOMD) was determined using the two-stage technique of [27]. Rumen digesta were collected from two rumen-cannulated Mutton Merino ewes (54 ± 2.8 kg body weight) fed a maintenance diet containing 9% crude protein, formulated to meet the nutrient requirements of small ruminants [28]. Animals were individually housed in pens with free access to water, and all procedures were conducted in accordance with the South African National Standard for animal care [29]. Rumen contents were collected 2 h post-feeding, strained through four layers of cheesecloth, pooled on an equal-volume basis, and mixed with CO2-saturated McDougall’s artificial saliva (pH 6.8) at a ratio of 1:4 (v/v). Fresh maize prior to ensiling and maize silage samples from each treatment were dried and ground to pass through a 1 mm screen using a Wiley mill (Standard Model 3, Arthur H. Thomas Co., Philadelphia, PA, USA). Approximately 0.70 g of maize silage from each replicate jar was weighed into 100 mL serum bottles, and 40 mL of buffered rumen fluid was added under continuous CO2 flushing to maintain anaerobic conditions. Bottles were sealed and incubated at 39 °C for 48 h in a shaking water bath (100 strokes/min). Two blank bottles containing inoculum only were included to correct for endogenous residues. Following the initial incubation, 6 mL of 20% hydrochloric acid and 2 mL of 5% pepsin solution were added, and samples were further incubated at 39 °C for 48 h. After digestion, residues were filtered, dried at 105 °C for 12 h, and ashed at 550 °C for 2 h to determine organic matter digestibility. Each treatment was incubated in triplicate serum bottles per run, with each bottle serving as the experimental unit, and the incubation was repeated in three independent runs to ensure reproducibility.
IVOMD was calculated according to [30] as:
where WS is the dry weight of the original maize sample, WR is the dry weight of the residue after incubation, and WB is the dry weight of the blank residue. All samples were analysed in triplicate.
In vitro degradability of nutrient fractions (ash, CP, NDF, ADF, ADL, GE,) was determined using a rumen incubation technique adapted from [27]. Rumen fluid was collected from two rumen-cannulated donor ewes fed a maintenance diet, strained through four layers of cheesecloth under continuous CO2 flushing, and mixed with CO2-saturated McDougall’s artificial saliva (pH 6.8) at a ratio of 1:4 (v/v) to prepare the incubation medium. Ground maize silage samples (0.5 g) from each treatment were placed in triplicate pre-weighed nylon bags (40–50 µm) for each incubation time point and submerged in 100 mL serum bottles containing 40 mL of buffered rumen fluid under anaerobic conditions. Bottles were sealed with rubber stoppers and incubated at 39 °C in a shaking water bath. Each treatment was incubated in independent serum bottles serving as experimental units within each incubation run, and the incubation was repeated across three runs. Incubations were terminated at 3, 6, 12, 24, and 48 h. After incubation, nylon bags were removed, rinsed with distilled water, and dried at 105 °C to constant weight. Residues were analysed for ash, which was used to calculate OM and CP according to [18]. The NDF, ADF, and ADL of residues were analysed using the ANKOM fibre analysis system. Gross energy (GE) of residues was determined using a bomb calorimeter.
Nutrient degradability was calculated as:
where S is the nutrient content in the original sample, R is the nutrient content in the residue after incubation, and B is the blank correction. All treatments were analysed in triplicate.
In vitro gas production was determined using the syringe-based technique described by [31]. Ground maize silage samples from each replicate jar were weighed in triplicate (200 mg) into calibrated 100 mL glass syringes. The fermentation medium was prepared according to [32] and consisted of a buffered mineral solution containing macro- and micro-minerals, resazurin as a redox indicator, and a reducing agent. The medium was continuously flushed with CO2 to maintain anaerobic conditions and warmed to 39 °C prior to inoculation. Rumen fluid was collected from two donor ewes before morning feeding, pooled, and strained through four layers of cheesecloth into a pre-warmed insulated flask. The rumen fluid was mixed with the buffered medium under CO2 stream. A total of 30 mL of the inoculum–buffer mixture was dispensed into each syringe containing substrate. Syringes were sealed and incubated at 39 °C. Gas production was recorded at 3, 6, 12, 24, and 48 h. Blank syringes containing inoculum only were included to correct for endogenous gas production. Each treatment was incubated in three replicate syringes per run, with each syringe serving as the experimental unit. The incubation was repeated in three independent runs. Cumulative gas production was expressed as mL gas per mg DM incubated at each time point.
2.6. Statistical Analysis
Data on silage fermentation indices, nutrient composition, aerobic stability and in vitro nutrient digestibility were analysed using the General Linear Model (GLM) procedure of GenStat as a two-way analysis of variance (ANOVA) in a 2 × 3 factorial arrangement, with protein source (SBM vs. MOC) and inoculant treatment (control, Sil-All, Lalsil) as fixed effects. The model included the main effects of protein source (PS), bacterial inoculant (BI), and their interaction (PS × BI). The model used was:
where Yijk is the observed response, μ is the overall mean, Pi is the effect of protein source, Ij is the effect of inoculant, (PI) ij is the interaction between factors, and εijk is the random residual error.
Yijk = μ + Pi + Ij + (PI) ij + εijk
Variables measured repeatedly over time, including silage pH (0, 3, 7, 14, and 90 days), in vitro gas production (3, 6, 12, 24, and 48 h), and nutrient degradability (0, 3, 6, 12, 24, and 48 h), were analysed using a repeated measures approach within the GenStat mixed model framework, with time included as the within-subject factor.
For all analyses, the experimental unit was defined according to the type of measurement. For silage fermentation, chemical composition, and aerobic stability, each jar served as the experimental unit, with three replicate jars per treatment (n = 3). For in vitro digestibility and nutrient degradability, each bottle was considered the experimental unit, with three replicate bottles per treatment per run (n = 9), and incubations were repeated in three independent runs. For gas production, each syringe served as the experimental unit, with three replicate syringes per treatment per run, and incubations were also repeated in three independent runs (n = 9).
Treatment means were compared using Fisher’s protected least significant difference (LSD) test when significant effects were detected. Statistical significance was declared at p < 0.05. Replication across jars, bottles, or syringes, together with repeated independent runs where applicable, ensured repeatability of the results.
3. Results
3.1. Chemical Composition
The chemical composition of SBM and MOC is presented in Table 1. Compared to SBM, MOC had higher (p < 0.05) DM, EE, GE, and ADL. In contrast, SBM exhibited significantly higher (p < 0.05) CP and ash contents and a higher IVOMD relative to SBM. There was no significant difference (p > 0.05) among the aNDF and ADF contents of SBM and MOC.

Table 1.
Chemical composition of soybean meal (SBM) and marula oilcake (MOC).
The chemical composition of of pre-ensiled freshly chopped whole-crop maize with inclusion of MOC in place of SBM, with or without microbial inoculants, is presented in Table 2. Maize with SBM without bacterial inoculant (treatment 1: positive control) exhibited numerically higher CP, WSC, and LA concentrations than other maize treatments. On the contrary, maize with MOC without bacterial inoculant (treatment 4: negative control) showed higher EE content and LAB population.

Table 2.
Chemical composition of pre-ensiled freshly chopped whole-crop maize with SBM or MOC, with or without bacterial inoculants.
3.2. Silage Fermentation, Nutrient Composition, and Aerobic Stability
Table 3 presents the effects of replacing SBM with MOC, with or without bacterial inoculants, on fermentation characteristics, nutrient composition, and aerobic stability of whole-crop maize silage. There was a significant effect (p < 0.05) of protein source, as well as protein source × inoculant interaction on WSC content. The Y & M counts and NH3-N content were, however, significantly influenced (p < 0.05) by protein source, inoculant and the protein source × inoculant interaction. The WSC content was highest (p < 0.05) in maize with MOC without inoculant (treatment 6) compared to other treatments. While Y & M population were lowest in all SBM-based maize silages (treatments 1, 2 and 3), the population was highest (p < 0.05) in treatment 5. The NH3-N content was significantly (p < 0.05) lower in treatments 4 and 6, but higher (p < 0.05) in treatment 1, which was the same treatment that exhibited the highest (p < 0.05) LAB population and LA concentration compared to other treatments. Although protein source had no significant effect (p > 0.05) on LAB population in the silage, the protein source × inoculant interaction was likewise not significant, despite the inoculant independently reducing (p < 0.05) LAB population. However, protein source × inoculant interaction significantly reduced (p < 0.05) LA concentration in treatments 3, 4 and 5.

Table 3.
Effects of protein source and bacterial inoculants on fermentation characteristics, nutrient composition, and aerobic stability of whole-crop maize silage.
While the effect of protein source was not significant (p > 0.05) on silage DM, inoculant as well as protein source × inoculant interaction reduced DM in treatment 6. Protein source, inoculants, as well as protein source × inoculant interaction significantly affected (p < 0.05) the nutrient composition of the silage. Ash content was significanly reduced (p < 0.05) in all treatments except in treatment 1. While the CP and IVOMD were significantly increased (p < 0.05) in treatment 1, they were significantly reduced (p < 0.05) in treatment 4. The aNDF, ADF, ADL and GE were significantly increased (p < 0.05) in treatmment 4, but significantly reduced (p < 0.05) in treatment 1.
During aerobic stability, pH levels were significantly lower (p < 0.05) in treatments 1 and 4 compared with the other tretaments. The CO2 was significantly reduced (p < 0.05) by the interaction between protein source and inoculant across most treatments, except for treatments 4 and 6, which exhibited significantly higher (p < 0.05) LAB and Y & M populations than the other treatments.
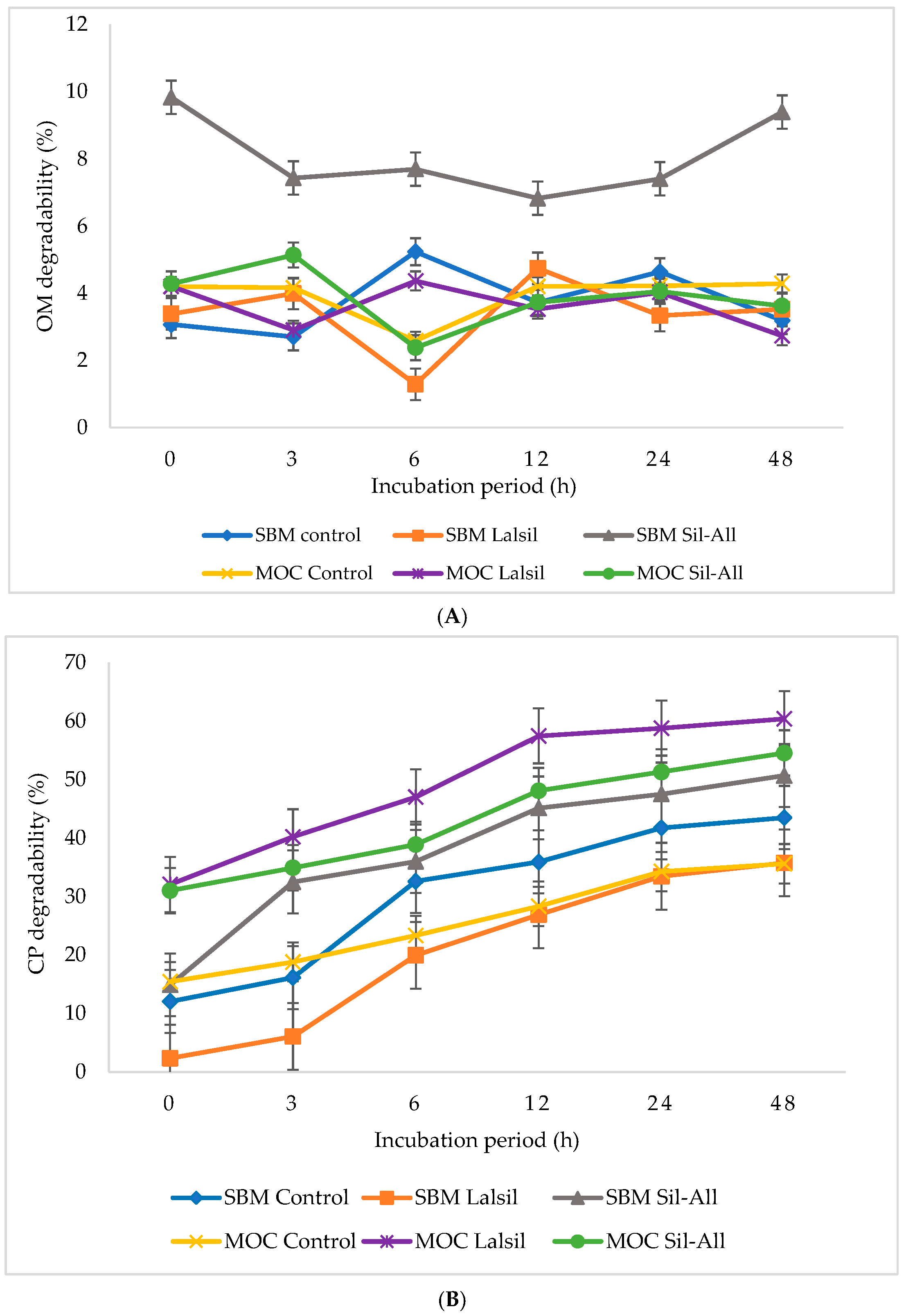
3.3. In Vitro Nutrient Digestibility
Figure 1 (A) OM; (B) CP; (C) aNDF; (D) ADF; (E) ADL; and (F) GE illustrate the effects of protein source and bacterial inoculant on in vitro nutrient degradability of whole-crop maize silage. The interaction between protein source, inoculant, and incubation time was not significant (p > 0.05) for OM degradability. However, treatment 3 consistently showed the highest degradability across incubation times, whereas treatments 2 and 6 recorded lower degradability at 6 h compared with the other treatments.
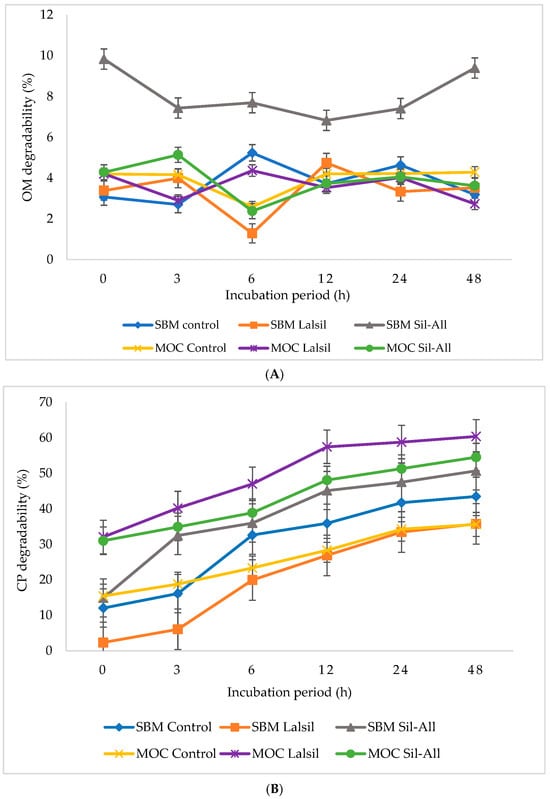
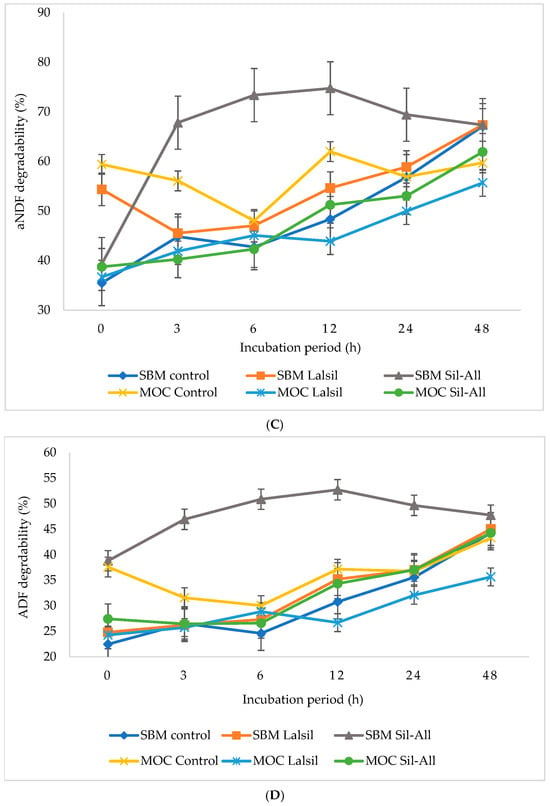
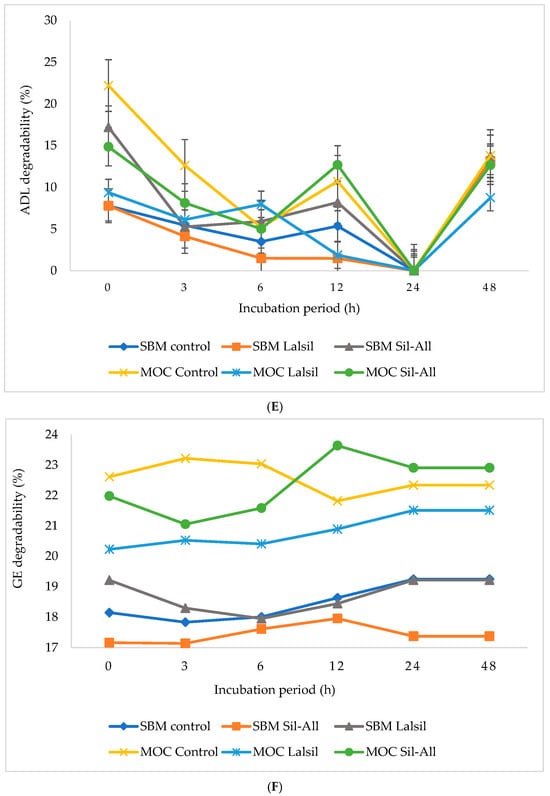
Figure 1.
Effect of protein source inclusion with or without bacterial inoculant on in vitro nurient degradability of chopped whole-crop maize silage. (A) illustrate the effects of protein source and bacterial inoculant on OM degradability of whole-crop maize silage. (B) illustrate the effects of protein source and bacterial inoculant on CP degradability of whole-crop maize silage. (C) show the effects of protein source and bacterial inoculant on aNDF of whole-crop maize silage. (D) demonstrate the effects of protein source and bacterial inoculant on ADF of whole-crop maize silage. (E) demonstrate the effects of protein source and bacterial inoculant on ADL of whole-crop maize silage. (F) illustrate the effects of protein source and bacterial inoculant on GE degradability of whole-crop maize silage.
In contrast, a significant (p < 0.05) three-way interaction between protein source, inoculant, and incubation time was observed for CP degradability. Treatment 2 had the lowest CP degradability, whereas treatment 5 had the highest CP degradability at 24 and 48 h compared with the other treatments. The CP degradability increased with incubation time, reaching its maximum at 48 h.
The interaction between protein source, inoculant, and incubation time was significant (p < 0.05) for aNDF degradability. Treatment 3 showed the highest degradability at 6 and 12 h, with relatively high values persisting at 24 and 48 h. The lowest aNDF degradability occurred in treatment 1 at 0 h.
Similarly, the interaction effect of protein source, inoculant, and incubation time was significant (p < 0.05) for ADF degradability. Treatment 3 exhibited the highest degradability at 12 h, and this higher degradability persisted at 24 and 48 h compared with the other treatments. The lowest ADF degradability was observed in treatment 1 at 6 h.
The ADL degradability was signifiantly (p < 0.05) affected by the three-way interation between protein source, inoculant, and incubation time. Treatment 4 showed the highest degradability at 0 h, followed by treatment 2 at the same incubation time, whereas the lowest ADL degradability across treatments occurred at 24 h.
A significant (p < 0.05) three-way interaction among protein source, inoculant and incubation time was observed for GE degradability. Treatment 6 showed the highest degradability at 12 h, while treatment 4 showed relatively high values at 3 and 6 h. In contrast, treatment 2 consistently showed the lowest GE degradability at 0 and 3 h.
3.4. Volatile Fatty Acids of Whole-Crop Maize Silage
Table 4 shows the effects of replacing SBM with MOC, with or without bacterial inoculant, on VFA profiles of whole-crop maize silage. Acetic acid was affected by a significant protein source × inoculant interaction (p < 0.05), with treatment 3 having the highest concentration compared with other treatments. Propionic acid was significantly influenced (p < 0.05) by protein source inclusion, with a higher concentration in SBM-based silages (treatments 1–3) and treatment 5, while inoculant, and protein source × inoculant interaction had no significant effect (p > 0.05). Butyric and iso-butyric acids were reduced in MOC-based silages (treatments 4–6) and in treatment 3 due to the protein source, inoculant as well as protein source × inoculant interaction effects (p < 0.05). Valeric acid was lower in all MOC-based treatments, with significant main and interaction effects (p < 0.05), than the SBM-based treatments. Iso-valeric acid concentrations were significantly lower (p < 0.05) in treatments 1, 4, and 6 compared with treatments 2, 3 and 5.

Table 4.
Effect of protein source inclusion with or without bacterial inoculant on volatile fatty acid concentrations of whole-crop maize silage.
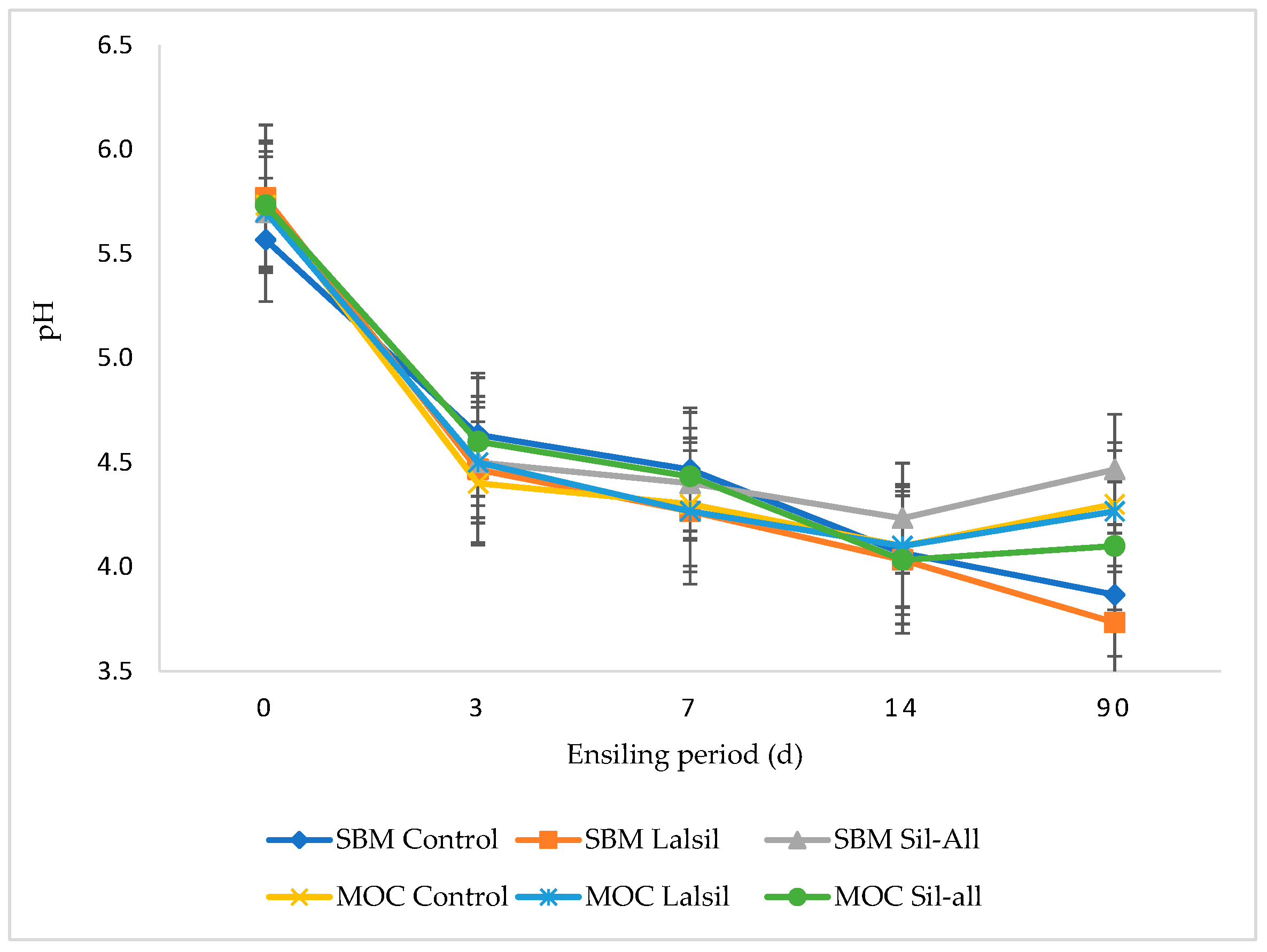
3.5. pH of Whole-Crop Maize Silage
Figure 2 illustrates the effect of replacing SBM with MOC, with or without bacterial inoculant, on the pH of chopped whole-crop maize during 90 days of ensiling. The inclusion of protein source or bacterial inoculant alone did not affect pH across treatments. However, the interaction between protein source, inoculant, and ensiling duration was highly significant (p < 0.05). After 3 days of ensiling, treatment 4 exhibited the lowest pH (4.4), whereas at day 7, treatments 2, 4, and 6 recorded the lowest pH (4.3). From 14 to 90 days, treatment 2 consistently maintained the lowest pH, while treatment 3 showed the highest pH throughout the same period.
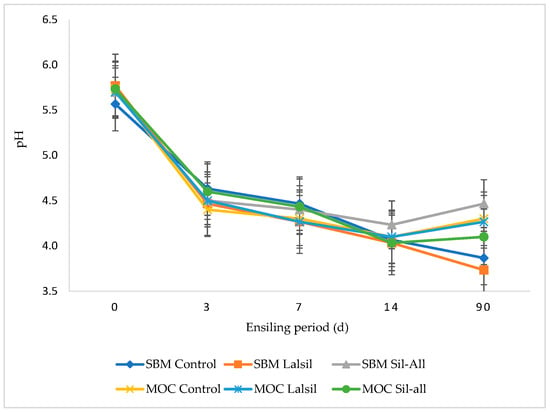
Figure 2.
Effect of protein source inclusion, with or without bacterial inoculant, on the pH of chopped whole-crop maize during 90 days of ensiling.
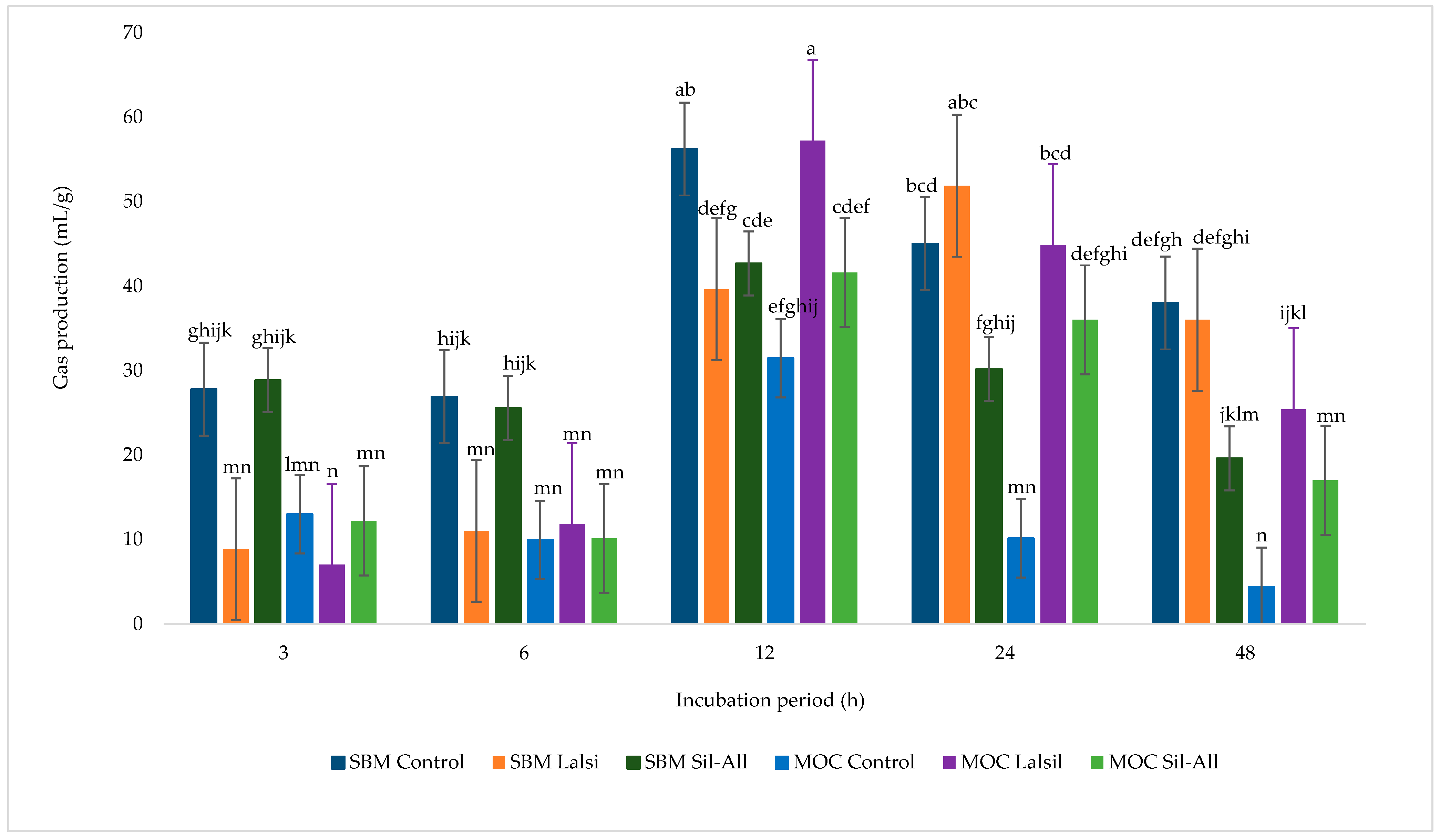
3.6. Whole-Crop Maize Silage Gas Production
Figure 3 illustrates the effects of MOC in place of SBM, with or without bacterial inoculant, on gas production from whole-crop maize silage during 48 h of incubation. Bacterial inoculant alone did not significantly affect gas production across treatments (p > 0.05), whereas incubation time and the interaction between protein source, inoculant, and incubation time were significant (p < 0.05). At 12 h of incubation, the highest gas production was observed in treatment 5, followed by treatment 1. By 24 h, treatments 1, 3, and 5 exhibited the highest gas production values. The lowest gas production occurred at 3 h in treatments 3 and 5, and at 48 h in treatment 4. Overall, gas production increased with incubation time, peaking at 12 h, with treatment 1 producing the most gas and treatment 6 the least.
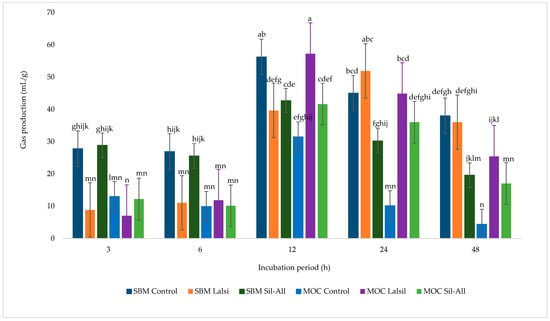
Figure 3.
Effect of protein source inclusion, with or without bacterial inoculant, on gas production from chopped whole-crop maize silage. a–n Means within a row with different superscripts differ significanty (p < 0.05).
4. Discussion
4.1. Chemical Composition
The exploitation of alternative feed resources remains a viable strategy to mitigate feed shortages in South African livestock systems. Chemical composition is a primary determinant of the suitability of novel feedstuffs, as nutrient composition directs dietary inclusion rates and animal performance [33,34]. The results of this study indicate differences in the chemical composition of SBM, a commercial protein source, and MOC, a non-conventional feed resource with potential as an alternative protein source to SBM in maize silage. Compared with SBM, MOC had higher DM, EE, GE, aNDF, ADF, and ADL contents. In contrast, ash, CP, and IVOMD were higher in SBM (Table 1). The higher EE and GE concentrations observed in MOC suggest its potential as an energy-dense feed resource in livestock diets. However, its effectiveness as a supplement in maize silage has not been well established, thereby necessitating the present study.
Although the CP content of MOC (39% DM) was lower than that of SBM (52% DM), the values were approximately similar to commonly used oilseed meals such as canola, cottonseed, and sunflower meals [35,36], supporting its classification as a viable protein supplement. Non-conventional oilseed by-products are increasingly utilised as alternative feed resources or as replacements for conventional oil cakes and meals in animal nutrition. These include, among others, palm kernel meal, macadamia nut cake, copra kernel meal, and rapeseed meal [37]. Slightly higher CP and EE contents for MOC have been reported elsewhere [38,39], suggesting some variability related to processing, growing environment or cultivar differences. The ADL content of MOC (11% DM) was almost similar to the 12% DM reported for macadamia oilcake but lower than that of avocado oilcake meal [40]. Fiber fractions are among the key factors influencing nutrient digestibility [41]. Thus, the relatively lower IVOMD observed in MOC than in SBM may be attributed to its higher ADL content, as lignin is negatively correlated with digestibility [42]. Despite this limitation, the IVOMD contents of both SBM and MOC exceeded 50%, which is considered the minimum threshold for ruminant feeds [43], thereby suggesting their nutritional adequacy. The similarities (p > 0.05) in aNDF and ADF concentrations between SBM and MOC indicate comparable cell wall content. The aNDF and ADF contents of MOC (15 and 7.8% DM, respectively) were almost similar to the 14% DM reported by [44] and the 5.1% DM reported by [45] for pumpkin seed cakes.
4.2. Silage Fermentation, Nutrient Composition and Aerobic Stability
The DM of pre-ensiled maize was between 32 and 38% DM and slightly over the 30 to 35% DM suggested by [46] as adequate for the ensiling process. The higher WSC concentration observed in treatment 1 compared to the other fresh maize treatments prior to ensiling (Table 2) suggests the presence of adequate fermentable substrate for epiphytic LAB to convert it to LA and smaller concentrations of VFA. The availability of WSC, in combination with epiphytic LAB, determines the rate of pH decline during the early stages of ensiling, which is critical to produce stable silage [47]. Indeed, ref. [48] further indicated that an initial WSC content of >6% DM is adequate to produce good-quality grass silage. In the present study, the WSC concentration in treatment 1 was 4.7% DM, whereas that of other maize treatments ranged from 1 to 1.2% DM. The pH values of the pre-ensiled whole maize crop treatments were between 5.0 and 5.8. The LAB microbial count ranged from 1.6 × 106 to 2 × 108 cfu/g, which was above the recommended 1 × 105 to 1 × 106 cfu/g for fresh forage [49]. This suggests that microbial counts of the pre-ensiled maize were sufficient for effective fermentation. The aNDF and ADF contents ranged from 31 to 40% DM and 18 to 22% DM, respectively.
Bacterial inoculants are widely used as silage additives to enhance the ensiling process. Most commercial products contain one or more homofermentative lactic acid bacteria (LAB) species. Lactobacillus plantarum, other Lactobacillus spp., Enterococcus faecium, and various Pediococcus spp. are among the most included microorganisms in silage inoculants. The inclusion of multiple bacterial species in some formulations aims to promote synergistic interactions among strains. For instance, combining Pediococcus and Enterococcus with Lactobacillus plantarum can accelerate the decline in pH and accelerate the transition to conditions that stabilize the forage [50]. Inoculated LAB may also complement the epiphytic LAB naturally present on the forage, thereby facilitating and improving the fermentation process [51,52].
Compared to the WSC content of pre-ensiled fresh maize, that of the ensiled maize was significantly higher, except in treatment 1, which had the highest WSC content prior to ensiling. Although LAB and Y & M populations declined after ensiling, the concentration of LA increased significantly (Table 3), indicating that acid production occurred earlier during active fermentation and remained stable even after microbial populations decreased. A reduction in LAB population during fermentation has been associated with limited tolerance to increasingly acidic conditions [53]. During the ensiling process, even LAB belonging to the Lactobacillus genus may lose viability as fermentation progresses [54]. At 90 days of fermentation, an increase in LA concentration was observed, which likely contributed to the suppression of the Y & M population due to the inhibitory effect of organic acids. Ref. [55] similarly reported that LAB inoculation in maize silage reduced Y & M population.
Although the Y & M population decreased following ensiling in the present study, the population remained above the 1.18 log cfu/g reported by [56] at 90 days of fermentation in silage treated with various inoculants, including Lactobacillus buchneri. Results in this study further show that after 90 days of ensiling, treatment 1 exhibited lower WSC content; however, the LAB population and LA concentration increased. According to [57], LAB population is typically low prior to ensiling but increases during the fermentation process. It is given that as the LAB population increases, so will the LA concentration as an end-product of the anaerobic fermentation to inhibit degradation of WSC by other bacteria [58]. In contrast, Y & M populations declined, whereas NH3-N content increased in treatment 1. This could be an indication of proteolysis due to the high CP content in the silage; however, the NH3-N contents across maize treatments were lower to associate with for proteolysis.
The NH3-N concentrations of treatments in this study were lower than that observed by [59] in maize silage inoculated with homo- and heterofermentative inoculants Lactococcus lactis and Lactobacillus buchneri, respectively. Nonetheless, the concentrations were all below the 10% DM recommended by [60] for a well-preserved silage. Despite a high LAB population in treatment 4, LA concentration did not increase and remained similar to that observed in treatments 3 and 5, treated with a homo- and heterofermentative inoculants, respectively.
Silage pH is a vital indicator to evaluate silage fermentation quality [61], but it is dependent on silage DM, ingredients, and ensiling period. The pH of maize significantly reduced with ensiling from a mean of 5.6 in pre-ensiled fresh maize to 4.1 after 90 days of ensiling. The results in this study show a rapid decline in pH with the lowest pH observed at day 90 of ensiling in treatment 2 (3.7) (Figure 2). The pH values in this study were comparable to those of [59] who reported a mean of 3.6 for maize silage inoculated with homo- and heterofermentative inoculants. This could be attributed to silage DM of >40% in this study. According to [62], pH around five is acceptable when silage is ensiled with high DM content.
4.3. In Vitro Nutrient Degradability
The OM degradability of maize silage after 90 days of ensiling, with MOC or SBM inclusion, and with or without bacterial inoculants, indicated that degradability was highest in treatment 3. Sil-All, a homofermentative multi-species inoculant, is known for enhancing the nutritive value of silage. The response was influenced by protein source inclusion, as OM degradability in treatment 6 was lower compared to its corresponding treatments. Approximately 9.4% of OM was degraded after 48 h of incubation. In contrast, treatment 2 exhibited the lowest degradability, with only 1.3% DM degraded after 6 h of incubation. These results indicate that OM degradability is not solely dependent on inoculation but is strongly mediated by the interaction between inoculant type and substrate composition. Treatments that enhance fermentation efficiency while reducing fiber recalcitrance appear to enhance ruminal OM degradation. A finding in silage research showed that inoculation with L. buchneri and homofermentative bacteria did not significantly affect dry matter (DM) degradability or ruminal degradation values in corn, wheat, or sorghum silages [63]. This suggests that microbial inoculants do not always enhance fiber breakdown or OM degradability under all conditions, possibly due to substrate composition or the limited enzyme repertoire of LAB.
In vitro CP degradability was significantly higher in treatment 5 containing L. buchneri, particularly at 24 and 48 h of incubation (59% and 60%, respectively). Although low NH3-N concentrations indicated limited proteolysis during ensiling, CP degradability remained high, suggesting that preserved protein fractions were still readily accessible to rumen microbes. This contrasts with findings by [59], where reduced proteolysis during fermentation was associated with lower in vitro CP degradability due to increased protein protection. In the present study, however, treatment 5 maintained high degradability despite effective preservation, implying limited lignin–protein complex formation and favorable protein structural characteristics. Comparable trends were observed by [64], in mixed silage, where ruminal degradability characteristics were influenced more by substrate composition and microbial treatment than by fermentation quality alone. Their findings, like the present results, suggest that improved preservation does not necessarily reduce ruminal CP availability, but rather that degradability depends on the interaction between inoculant activity and intrinsic feed protein structure.
In vitro aNDF degradability was highest in treatment 3, with 73% and 75% of aNDF degraded at 6 and 12 h of incubation, respectively. In contrast, treatment 1 showed the lowest early degradability, with only 36% of aNDF degraded at 0 h of incubation. The higher degradability observed in maize silage containing SBM is consistent with reports that homofermentative LAB, such as those in Sil-All, can enhance fiber degradation by promoting favorable fermentation conditions and supporting microbial colonization [65]. However, other studies have shown that LAB inoculation does not always increase NDF degradability, and the response can be influenced by the forage type and substrate characteristics [66]. Moreover, LAB species such as Lactobacillus plantarum have been reported to enhance ruminal fiber fermentation by stimulating cellulolytic microbial activity [67], which may explain the higher degradability observed in Sil-All inoculated treatment and lowest in uninoculated treatment 1.
The degradability of ADF followed a pattern similar to that of OM and aNDF. The highest ADF degradability (53%) at 12 h of incubation was observed in treatment 3. The least degradable silage was treatment 1 (23%) at 0 h of incubation. At 48 h of incubation, degradability was mostly affected, with Sil-All being the dominant inoculant, SBM the primary protein source, and their interaction also showing a significant effect. Contrary to NDF and ADF, in vitro ADL degradability in our study was mainly influenced at 0 and 48 h of incubation, with treatment 4 being the dominant factor affecting lignin degradation. Treatment 4 had the highest initial ADL content (22%), while the lowest degradability (<0%) occurred at 24 h for the same treatment, and treatment 6 degradability increased to 16% at 48 h. This pattern likely reflects the well-established resistance of lignin to microbial fermentation; lignin forms complex, cross-linked aromatic structures within the plant cell wall that physically and chemically inhibit microbial access to cellulose and hemicellulose [68,69], making ADL fractions inherently less degradable than other fiber components such as NDF and ADF in vitro systems. Indeed, lignin content has been shown to correlate negatively with fiber degradability in ruminant feeds [70] and the ruminal microbiota are largely incapable of significant lignin degradation [71]. This is why ADL remains high and resistant in many silages (i.e., low degradability) without specialized fungal or enzyme-producing inoculants. These results are consistent with a study by [64], who reported that mixed silage composition affected rumen degradability of structural components, including ADL, implying that variations in forage composition (e.g., licorice vs. sweet sorghum) and fiber makeup influence how degradability unfolds during incubation. This is consistent with our notion that chemical composition (e.g., lignification) strongly affects in vitro degradability.
In vitro GE degradability was influenced by both protein inclusion and inoculation. At 12 h of incubation, treatment 6 demonstrated the highest GE degradability (24%). The effect of inoculation became more evident at 24 and 48 h, with treatment 4 showing dominant responses at 3 and 6 h of incubation. The lowest GE degradability was observed at 0 and 3 h in treatment 2, where only 17% of GE was degraded. This suggests that, although certain treatments enhanced early energy release, the interaction between inoculant type and substrate composition played a critical role in determining overall energy disappearance during incubation. Treatment 2 also demonstrated low OM and CP degradability, which translated into proportionally low GE degradability. The study by [72] found that, although silage additives improved fermentation quality (e.g., lower pH and reduced DM losses), there were no significant differences in GE values or energy losses among silages regardless of additive treatment. This suggests that GE degradability reflects not only degradation of structural components but also preservation and partitioning of fermentable energy fractions within the silage matrix (e.g., no significant differences (p > 0.05) in GE loss were observed between inoculated and control silages despite improved fermentation parameters).
Overall, nutrient degradability responses were strongly influenced by the interaction between protein source inclusion and inoculation. The OM degradability was highest in treatment 3 (9.3% at 48 h), whereas treatment 2 showed comparatively lower values. In contrast, CP degradability was markedly higher in treatment 5, reaching 59–60% at 24 and 48 h, compared to lower early CP disappearance (6% at 3 h) in treatment 2. Fiber degradability (aNDF and ADF) followed a pattern similar to OM, with treatment 3 generally showing greater structural carbohydrate disappearance, while ADL degradability remained low across treatments and was mainly influenced at 0 and 48 h, reflecting lignin recalcitrance. The GE degradability was highest in treatment 5 at 12 h (24%) but lowest in treatment 2 at 0 and 3 h (17%), indicating that reduced OM and CP degradability corresponded to greater energy disappearance, regardless of supplement. Nutrient breakdown in maize silage depended on both protein source and inoculant. Treatment 3 gave the highest fiber and OM degradability, while treatment 5 provided the best protein breakdown. Lignin was resistant in all treatments, and energy release was associated with the type of inoculant on a substrate.
The detailed superscripts, LSD, SE, and p-Values for protein source inclusion, bacterial inoculant, incubation period, and their interaction are provided in Supplementary Table S1.
4.4. Volatile Fatty Acids
Rumen fibrolytic microorganisms generate VFA during the breakdown of structural carbohydrates, particularly cellulose and hemicellulose [73]. These fermentation end-products consist mainly of short-chain fatty acids (SCFAs), including acetic, propionic, and butyric acids. Such acids are derived directly from the microbial fermentation of plant cell wall components. Short-chain fatty acids have been reported to suppress the growth of Y & M [74], which partly explains why silages treated with homofermentative LAB inoculants may be more susceptible to aerobic deterioration when exposed to air. Once produced in the rumen, VFAs are readily absorbed across the rumen epithelium and serve as a primary energy source for the host animal, thereby contributing substantially to its metabolic functions and overall nutritional status [75]. The VFA concentrations observed in the present study largely did not exceed the typical ranges reported for well-fermented maize silage (acetic acid 1–3% DM; propionic acid < 0.5% DM; butyric acid < 0.1% DM; reported by [76,77]). The higher acetic acid concentration in treatment 3 suggests, in contrast to the homofermentative characteristics, an enhanced heterofermentative activity, which is commonly associated with improved aerobic stability, as also reported by [78]. The relatively low butyric acid concentrations in MOC-based silages indicate limited clostridial activity and desirable fermentation quality, consistent with observations by [79]. Increased propionic acid in SBM-based silages supports the concept that substrate composition influences fermentation end-products, similar to findings by [80]. However, ref. [79] reported reduced acetic acid with certain inoculants, highlighting that additive effects on VFA profiles are substrate dependent.
4.5. Gas Production
In maize silage, in vitro gas production has been used extensively to assess ruminal fermentation and degradability, with studies showing that forage quality and additives significantly influence gas output [29,81,82]. Indeed, gas production patterns in this study closely reflected inoculation rather than degradability responses, which contradicts with the absence of a main inoculant effect. The higher gas output at 12 and 24 h in treatments 5 and 2, respectively, importantly corresponds with the interaction between protein source inclusion, bacterial inoculant, and incubation period indicating enhanced microbial colonization from the heterofermentative inoculant and fermentation of structural carbohydrates. Increased gas production at mid-incubation (12 h) suggests rapid fermentation of soluble and potentially degradable fiber fractions, consistent with the relationship between substrate degradability and gas yield described by [83]. The lower gas production observed at 3 h in treatment 5 and at 48 h in treatment 4 likely reflects limited availability of rapidly fermentable fractions and depletion of fermentable substrate over time.
5. Conclusions
This study evaluated the effects of replacing SBM with MOC and the role of bacterial inoculants on whole-crop maize silage quality and in vitro nutrient degradability. Compared with MOC, SBM contained higher CP, ash, and IVOMD, whereas MOC had higher DM, EE, GE, and lignin content than SBM. Both protein sources produced acceptable silage, with SBM favouring greater nutrient degradability and MOC providing higher energy content and improved aerobic stability. Microbial inoculants modified fermentation and degradability depending on the protein source. The interaction between protein source and inoculant influenced nutrient breakdown and energy availability, indicating that MOC is a viable alternative to SBM in maize silage, with consideration of its effects on fibre utilisation and fermentation dynamics.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/fermentation12030155/s1, Table S1: Superscripts, LSD, SE and p-Values for treatment (supplement), inoculant (additive) and interaction effects for Figure 1A–F.
Author Contributions
M.V., investigation and writing of the original draft; M.M.R., supervision and formal analysis; J.T.T., resources; T.L., data curation; B.D.N., supervision and validation; I.M.M.M., conceptualization, methodology and writing: review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Research Foundation (NRF) of South Africa, grant number TTK190402426559.
Institutional Review Board Statement
The animal study protocol was approved by the Ethics Committees of Agricultural Research Council-Animal Production (APIEC 26/08 and 24 August 2024) and Tshwane University of Technology (PO24/04 and 6 March 2024).
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author due to restricted company policy.
Acknowledgments
The authors gratefully acknowledge Eric Mathebula for his assistance with the statistical analysis of the study data.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ADD | Additive |
| ADF | Acid detergent fiber |
| ADL | Acid detergent lignin |
| aNDF | Neutral detergent fiber (amylase-treated) |
| ARC-AP | Agricultural Research Council–Animal Production |
| BCVFA | Branched-chain volatile fatty acids |
| CFU | Colony-forming units |
| CO2 | Carbon dioxide |
| CP | Crude protein |
| DM | Dry matter |
| EE | Ether extract |
| GE | Gross energy |
| GP | Gas production |
| IVOMD | In vitro organic matter digestibility |
| LAB | Lactic acid bacteria |
| LA | Lactic acid |
| MOC | Marula oilcake |
| NH3-N | Ammonia nitrogen |
| OM | Organic matter |
| SBM | Soybean meal |
| SEM | Standard error of the mean |
| TN | Total nitrogen |
| T1-T6 | Treatment 1 to treatment 6 |
| VFA | Volatile fatty acids |
| WSC | Water-soluble carbohydrates |
| Y & M | Yeast and mould |
References
- Charmley, E. Towards improved silage quality—A review. Can. J. Anim. Sci. 2001, 81, 157–168. [Google Scholar] [CrossRef]
- Isselstein, J.; Kayser, M. Functions of grassland and their potential in delivering ecosystem services. In EGF at 50: The Future of European Grasslands, Proceedings of the 25th General Meeting of the European Grassland Federation, Aberystwyth, Wales, 7–11 September 2014; Hopkins, A., Collins, R.P., Fraser, M.D., King, V.R., Lloyd, D.C., Moorby, J.M., Robson, P.R.H., Eds.; European Grassland Federation: Aberystwyth, Wales, 2014; Volume 19, pp. 199–214. Available online: https://research.aber.ac.uk/en/publications/egf-at-50-the-future-of-european-grasslands-proceedings-of-the-25/ (accessed on 11 March 2026).
- Van De Vyver, W.F.J.; Beukes, J.A.; Meeske, R. Maize silage as a finisher feed for Merino lambs. S. Afr. J. Anim. Sci. 2013, 43, S116–S120. [Google Scholar] [CrossRef]
- Balehegn, M.; Ayantunde, A.; Amole, T.; Njarui, D.; Müller, F.L.; Meeske, R.; Tjelele, T.J.; Malebana, I.M.; Madibela, O.R.; Boitumelo, W.S.; et al. Forage conservation in sub-Saharan Africa: Review of experiences, challenges, and opportunities. Agron. J. 2022, 114, 75–99. [Google Scholar] [CrossRef]
- Muck, R.E. Silage microbiology and its control through additives. Rev. Bras. Zootec. 2010, 39, 183–191. [Google Scholar] [CrossRef]
- Department of Agriculture, Forestry and Fisheries (DAFF). Market Value Chain Profile, 2010–2011; Department of Agriculture, Forestry and Fisheries: Pretoria, South Africa, 2011.
- Nkosi, B.D.; Meeske, R.; Muya, M.C.; Langa, T.; Thomas, R.S.; Malebana, I.M.M.; Motiang, M.D.; Van Niekerk, J.A. Microbial additives affect silage quality and ruminal dry matter degradability of avocado (Persea americana) pulp silage. S. Afr. J. Anim. Sci. 2019, 49, 997–1007. [Google Scholar] [CrossRef]
- Schmidt, R.J.; Kung, L., Jr. The effects of Lactobacillus buchneri with or without a homolactic bacterium on the fermentation and aerobic stability of corn silages made at different locations. J. Dairy Sci. 2010, 93, 1616–1624. [Google Scholar] [CrossRef]
- Ashbell, G.; Weinberg, Z.G.; Hen, Y.; Fily, A. The effects of temperature on the aerobic stability of wheat and corn silages. J. Ind. Microbiol. Biotechnol. 2002, 28, 261–263. [Google Scholar] [CrossRef]
- Contreras-Govea, F.E.; Muck, R.E.; Armstrong, K.L.; Albrecht, K.A. Nutritive value of corn silage in mixture with climbing beans. Anim. Feed Sci. Technol. 2009, 150, 1–8. [Google Scholar] [CrossRef]
- McDonald, P.; Edwards, R.A.; Greenhalgh, J.F.D.; Morgan, C.A.; Sinclair, L.A.; Wilkinson, R.G. Animal Nutrition, 7th ed.; Pearson: Harlow, UK, 2010. [Google Scholar]
- Zahiroddini, H.; Baah, J.; McAllister, T.A. Effects of microbial inoculants on the fermentation, nutrient retention, and aerobic stability of barley silage. Asian-Australas. J. Anim. Sci. 2006, 19, 1429–1436. [Google Scholar] [CrossRef]
- Driehuis, F.; Wilkinson, J.M.; Jiang, Y.; Ogun Ade, I.; Adesogan, A.T. Silage review: Animal and human health risks from silage. J. Dairy Sci. 2018, 101, 4093–4110. [Google Scholar] [CrossRef]
- Malebana, I.M.; Nkosi, B.D.; Erlwanger, K.H.; Chivandi, E. A comparison of the proximate, fiber, mineral content, amino acid and fatty acid profile of marula (Sclerocarya birrea caffra) nut and soybean (Glycine max) meals. J. Sci. Food Agric. 2018, 98, 1381–1387. [Google Scholar] [CrossRef]
- Mthiyane, D.M.N.; Mhlanga, B.S. The nutritive value of marula (Sclerocarya birrea) seed cake for broiler chickens: Nutritional composition, performance, carcass characteristics and oxidative and mycotoxin status. Trop. Anim. Health Prod. 2017, 49, 835–842. [Google Scholar] [CrossRef]
- Singh, R.; Langyan, S.; Sangwan, S.; Rohtagi, B.; Khandelwal, A.; Shivastava, M. Protein for human consumption from oilseed cakes: A review. Front. Sustain. Food Syst. 2022, 6, 856401. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of the Association of Official Analytical Chemists, 15th ed.; The Association of Official Analytical Chemists: Arlington, VA, USA, 1990; Sec. 985.29. [Google Scholar]
- AOAC. Official Methods of Analysis, 18th ed.; Association of Official Analytical Chemists: Gaithersburg, MD, USA, 2006. [Google Scholar]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and non-starch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Pearson, D.; Muslemuddin, M. The accurate determination of volatile nitrogen in meat and fish. J. Assoc. Public Anal. 1968, 6, 117–123. [Google Scholar]
- Suzuki, M.; Lund, C.W. Improved gas-liquid chromatography for simultaneous determination of volatile fatty acids and lactic acids in silage. J. Agric. Food Chem. 1980, 28, 1040–1041. [Google Scholar] [CrossRef]
- Barker, S.B.; Summerson, W.H. The colorimetric determination of lactic acid in biological material. J. Biol. Chem. 1941, 138, 535–554. [Google Scholar] [CrossRef]
- Pryce, J.D. A modification of the Barker-Summerson method for the determination of lactic acid. Analyst 1969, 94, 1151–1152. [Google Scholar] [CrossRef]
- ISO15214; Microbiology of Food and Animal Feeding Stuffs: Horizontal Method for the Enumeration of Mesophilic Lactic Acid Bacteria—Colony-Count Technique at 30 °C. International Organization for Standardization (ISO): Geneva, Switzerland, 1998.
- ISO 21527-2:2008; Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Yeasts and Moulds—Part 2: Colony Count Technique in Products with Water Activity Less than or Equal to 0.95. International Organization for Standardization: Geneva, Switzerland, 1998.
- Ashbell, G.; Weinberg, Z.G.; Azrieli, A.; Hen, Y.; Horev, B. A simple system to study the aerobic deterioration of silages. Can. Agric. Eng. 1991, 33, 391–393. [Google Scholar]
- Tilley, J.M.A.; Terry, R.A. A two-stage technique for the in vitro digestion of forage crops. Grass Forage Sci. 1963, 18, 104–111. [Google Scholar] [CrossRef]
- National Research Council (NRC). Nutrient Requirements of Small Ruminants: Sheep, Goats, Cervids, and New World Camelids; National Academies Press: Washington, DC, USA, 2007. [Google Scholar] [CrossRef]
- Mohr, B.J.; Fakoya, F.A.; Hau, J.; Souilem, O.; Anestidou, L. The governance of animal care and use for scientific purposes in Africa and the Middle East. ILAR J. 2017, 57, 333–346. [Google Scholar] [CrossRef][Green Version]
- Engels, E.A.N.; Van der Merwe, F.J. Application of an in vitro technique to South African forages with special reference to the effect of certain factors on the results. S. Afr. J. Agric. Sci. 1967, 10, 983–995. [Google Scholar]
- Blümmel, M.; Makkar, H.P.S.; Becker, K. In vitro gas production: A technique revisited. J. Anim. Physiol. Anim. Nutr. 1997, 77, 24–34. [Google Scholar] [CrossRef]
- Menke, K.H.; Steingass, H. Estimation of the energetic feed value obtained from chemical analysis and in vitro gas production using rumen fluid. Anim. Res. Dev. 1988, 28, 7–55. [Google Scholar]
- Caisîn, L.; Grosu, N.; Kovalenko, A. The influence of the preparation Primix Bionorm K on the digestibility of the nutrients in the fodders for young pigs. Sci. Pap. Anim. Sci. Biotechnol. 2012, 45, 33. [Google Scholar]
- Khan, A.U.; Khan, M.N.; Ayaz, M.; Khan, M.I.; Khan, F.Z.; Khan, I.U.; Gul, N.; Nawaz, A.; Khan, M.I.; Ullah, R.; et al. Economic comparison of sunflower, canola, and soybean meals in dairy cow diets and their impact on milk yield and composition. J. Anim. Plant Res. 2025, 2, 32–42. [Google Scholar]
- Vasudha, C.; Sarla, L. Nutritional quality analysis of sunflower seed cake (SSC). Pharma Innov. J. 2021, 10, 720–728. [Google Scholar]
- Świątkiewicz, S.; Arczewska-Włosek, A.; Józefiak, D. The use of cottonseed meal as a protein source for poultry: An updated review. World’s Poult. Sci. J. 2016, 72, 473–484. [Google Scholar] [CrossRef]
- Fatufe, A.A.; Akanbi, I.O.; Saba, G.A.; Olowofeso, O.; Tewe, O.O. Growth performance and nutrient digestibility of growing pigs fed a mixture of palm kernel meal and cassava peel meal. Livest. Res. Rural Dev. 2007, 19, 180. [Google Scholar]
- Mlambo, V.; Dlamini, B.J.; Ngwenya, M.D.; Mhazo, N.; Beyene, S.T.; Sikosana, J.L.N. In sacco and in vivo evaluation of marula (Sclerocarya birrea) seed cake as a protein source in commercial cattle fattening diets. Livest. Res. Rural Dev. 2011, 23, 121. [Google Scholar]
- Mdziniso, P.M.; Dlamini, A.M.; Khumalo, G.Z.; Mupangwa, J.F. Nutritional evaluation of marula (Sclerocarya birrea) seed cake as a protein supplement in dairy meal. J. Appl. Life Sci. Int. 2016, 4, 1–11. [Google Scholar] [CrossRef]
- Skenjana, A.; van Ryssen, J.B.J.; van Niekerk, W.A. In vitro digestibility and in situ degradability of avocado meal and macadamia waste products in sheep. S. Afr. J. Anim. Sci. 2006, 36, 445–451. [Google Scholar]
- McDonald, P.; Edwards, R.A.; Greenhalgh, J.F.D. Animal Nutrition, 5th ed.; Longman: London, UK, 1995. [Google Scholar]
- Andualem, D.; Negesse, T.; Tolera, A. Methane concentration, organic matter digestibility, metabolisable energy and short chain fatty acid production of morphological fractions of stinging nettle (Urtica simensis) measured through an in vitro gas test. Glob. Vet. 2016, 16, 276–284. [Google Scholar] [CrossRef]
- Belete, S.; Tolera, A.; Betsha, S.; Dickhöfer, U. Feeding values of indigenous browse species and forage legumes for the feeding of ruminants in Ethiopia: A meta-analysis. Agriculture 2024, 14, 1475. [Google Scholar] [CrossRef]
- Keller, M.; Kreuzer, M.; Reidy, B.; Scheurer, A.; Guggenbühl, B.; Luder, M.; Frank, J.; Giller, K. Effects on performance, carcass and meat quality of replacing maize silage and concentrate by grass silage and corn cob mix in the diet of growing bulls. Meat Sci. 2022, 188, 108795. [Google Scholar] [CrossRef]
- Budzaki, S.; Strelec, I.; Krnic, M.; Alilovic, K.; Tisma, M.; Zelic, B. Proximate analysis of cold press oil cakes after biological treatment with Trametes versicolor and Humicola grisea. Eng. Life Sci. 2018, 18, 924–931. [Google Scholar] [CrossRef]
- Neumann, M. Parâmetros para Análise de Qualidade da Silagem; IEPEC: Maringá, Brazil, 2013. [Google Scholar]
- Yuan, X.J.; Guo, G.; Wen, A.Y.; Desta, S.T.; Wang, J. The effect of different additives on the fermentation quality, in vitro digestibility and aerobic stability of a total mixed ration silage. Anim. Feed Sci. Technol. 2015, 207, 41–50. [Google Scholar] [CrossRef]
- Li, X.; Cheng, Y.; Yang, F.; Hu, J.; Ma, R.; Liu, H.; Shao, T. Improving total mixed ration silage: Effects of lactic acid bacteria inoculants and antimicrobial additives on fermentation quality and aerobic stability. Agronomy 2024, 14, 1602. [Google Scholar] [CrossRef]
- Kung, L.; Shaver, R. Interpretation, and use of silage fermentation analysis reports. Focus Forage 2001, 3, 1–5. [Google Scholar]
- Weinberg, Z.G.; Muck, R.E. New trends and opportunities in the development and use of inoculants for silage. FEMS Microbiol. Rev. 1996, 19, 53–68. [Google Scholar] [CrossRef]
- Yahaya, M.S.; Goto, M.; Yimiti, W.; Smerjai, B.; Kawamoto, Y. Evaluation of fermentation quality of tropical and temperate forage crops ensiled with additives of fermented juice of epiphytic lactic acid bacteria (FJLB). Asian-Australas. J. Anim. Sci. 2004, 17, 942–946. [Google Scholar] [CrossRef]
- Shao, T.; Zhang, Z.X.; Shimojo, M.; Wang, T.; Masuda, Y. Comparison of fermentation characteristics of Italian ryegrass (Lolium multiflorum Lam.) and guineagrass (Panicum maximum Jacq.) during the early stage of ensiling. Asian-Australas. J. Anim. Sci. 2005, 18, 1727–1734. [Google Scholar] [CrossRef]
- Li, Y.; Nishino, N. Bacterial and fungal communities of wilted Italian ryegrass silage inoculated with and without Lactobacillus rhamnosus or Lactobacillus buchneri. Lett. Appl. Microbiol. 2011, 52, 314–321. [Google Scholar] [CrossRef]
- Oude Elferink, S.J.W.H.; Driehuis, F.; Gottschal, J.C.; Spoelstra, S.F. Silage fermentation processes and their manipulation. In FAO Electronic Conference on Tropical Silage; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2000; pp. 1–28. Available online: http://www.fao.org/3/X1736E/x1736e06.htm (accessed on 11 March 2026).
- Ranjit, N.K.; Kung, L., Jr. The effect of Lactobacillus buchneri, Lactobacillus plantarum, or a chemical preservative on the fermentation and aerobic stability of corn silage. J. Dairy Sci. 2000, 83, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Muck, R.E. Effects of corn silage inoculants on aerobic stability. Trans. ASAE 2004, 47, 1011–1016. [Google Scholar] [CrossRef]
- Lin, C.; Bolsen, K.K.; Brent, B.E.; Hart, R.A.; Dickerson, J.T.; Feyerherm, A.M.; Aimutis, W.R. Epiphytic microflora on alfalfa and whole-plant corn. J. Dairy Sci. 1992, 75, 2484–2493. [Google Scholar] [CrossRef] [PubMed]
- McDonald, P.; Edwards, R.A.; Greenhalgh, J.F.D.; Morgan, C.A. Animal Nutrition, 6th ed.; Longman Scientific and Technical, Prentice Hall: Hoboken, NJ, USA, 2002. [Google Scholar]
- Polan, C.E.; Stieve, D.E.; Garrett, J.L. Protein preservation and ruminal degradation of ensiled forage treated with heat, formic acid, ammonia, or microbial inoculant. J. Dairy Sci. 1998, 81, 765–776. [Google Scholar] [CrossRef]
- Liu, C.; Lai, Y.; Lu, X.; Guo, P.; Luo, H. Effect of lactic acid bacteria inoculants on alfalfa (Medicago sativa L.) silage quality: Assessment of degradation (in situ) and gas production (in vitro). J. Integr. Agric. 2016, 15, 2834–2841. [Google Scholar] [CrossRef]
- Muck, R.E.; Weinberg, Z.G.; Contreras Govea, F.E. Silage extracts used to study the mode of action of silage inoculants in ruminants. Agric. Food Sci. 2013, 22, 108–114. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, J.; Shi, S.; Sun, Q. The effects of wilting and storage temperatures on the fermentation quality and aerobic stability of Stylosanthes spp silage. Anim. Sci. J. 2011, 82, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Filya, I. The effect of Lactobacillus buchneri, with or without homofermentative lactic acid bacteria, on the fermentation, aerobic stability and ruminal degradability of wheat, sorghum and maize silages. J. Appl. Microbiol. 2003, 95, 1080–1086. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Wang, J.; Zhang, S.; Chaudhry, A.S.; Khanaki, H. Assessing fermentation quality, aerobic stability, in vitro digestibility, and rumen degradation characteristics of silages mixed with sweet sorghum and aerial parts of licorice. Agriculture 2024, 14, 212. [Google Scholar] [CrossRef]
- Weinberg, Z.G.; Shatz, O.; Chen, Y.; Yosef, E.; Nikbahat, M.; Ben-Ghedalia, D.; Miron, J. Effect of lactic acid bacteria inoculants on in vitro digestibility of wheat and corn silages. J. Dairy Sci. 2007, 90, 4754–4762. [Google Scholar] [CrossRef]
- Yu, M.; Wang, P.; Li, F.; Du, J.; Jin, Y.; Zhao, T.; Yi, Q.; Tang, H.; Yuan, B. Fermentation quality and in vitro digestibility of sweet corn processing by-products silage mixed with millet hull or wheat bran and inoculated with a lactic acid bacteria. Fermentation 2024, 10, 254. [Google Scholar] [CrossRef]
- Chang, L. Effect of Wilting and Lactic acid Bacteria Treatment on Fermentation Pattern of Italian Ryegrass (Lolium multiflorum Lam.) Silage. Ph.D. Thesis, Seoul National University, Seoul, Republic of Korea, 2020. [Google Scholar]
- Inoue, K.; Sewalt, V.J.H.; Ballance, G.M.; Ni, W.T.; Stürzer, C.; Dixon, R.A. Developmental expression and substrate specificities of alfalfa caffeic acid 3-methyltransferase and caffeoyl coenzyme A 3-methyltransferase in relation to lignification. Plant Physiol. 1998, 117, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Casler, M.D.; Vogel, K.P. Accomplishments and impact from breeding for increased forage nutritional value. Crop Sci. 1999, 39, 12–20. [Google Scholar] [CrossRef]
- Van Soest, A.P.; Hermes, G.D.; Berendsen, A.A.; Van De Rest, O.; Zoetendal, E.G.; Fuentes, S.; De Vos, W.M. Associations between pro- and anti-inflammatory gastro-intestinal microbiota, diet, and cognitive functioning in Dutch healthy older adults: The NU-AGE study. Nutrients 2020, 12, 3471. [Google Scholar] [CrossRef]
- Damiran, D.; Biligetu, B.; Lardner, H. Evaluation of rumen degradation kinetics of low-lignin alfalfa ‘Hi-Gest® 360’ in Saskatchewan, Canada. Animals 2023, 13, 1047. [Google Scholar] [CrossRef]
- Sakhawat, I. The effect of silage quality on gross energy losses. In Proceedings of the Agricultural and Food Sciences Conference, Mombasa, Kenya, 7–11 November 2011; Available online: https://api.semanticscholar.org/CorpusID:97981494 (accessed on 11 March 2026).
- Xu, G.; Han, Z.; Wang, S.; Dai, T.; Dong, D.; Zong, C.; Yin, X.; Jia, Y.; Shao, T. Soy sauce residue in total mixed ration silage: Fermentation characteristics, chemical compositions, in vitro digestibility and gas production. J. Anim. Sci. 2022, 21, 1058–1066. [Google Scholar] [CrossRef]
- Moon, N.J. Inhibition of the growth of acid tolerant yeasts by acetate, lactate and propionate and their synergistic mixtures. J. Appl. Microbiol. 1983, 55, 453–460. [Google Scholar] [CrossRef]
- Vasta, V.; Daghio, M.; Cappucci, A.; Buccioni, A.; Serra, A.; Viti, C.; Mele, M. Plant polyphenols and rumen microbiota responsible for fatty acid biohydrogenation, fiber digestion, and methane emission: Experimental evidence and methodological approaches. J. Dairy Sci. 2019, 102, 3781–3804. [Google Scholar] [CrossRef]
- McDonald, P.; Henderson, A.R.; Heron, S.J.E. The Biochemistry of Silage, 2nd ed.; Chalcombe Publications: Marlow, UK, 1991; 340p, Available online: http://books.google.com/books?id=oUcjAQAAMAAJ (accessed on 11 March 2026).
- Kung, L., Jr.; Shaver, R.D.; Grant, R.J.; Schmidt, R.J. Silage review: Interpretation of chemical, microbial, and organoleptic components of silages. J. Dairy Sci. 2018, 101, 4020–4033. [Google Scholar] [CrossRef]
- de Assis, F.G.V.D.; Ávila, C.L.D.S.; Pinto, J.C.; Schwan, R.F. New inoculants on maize silage fermentation. Rev. Bras. Zootec. 2014, 43, 395–403. [Google Scholar] [CrossRef]
- Ma, J.; Lin, L.; Lu, Y.; Weng, B.; Feng, Y.; Du, C.; Gan, S. The influence of silage additives supplementation on chemical composition, aerobic stability, and in vitro digestibility in silage mixed with Pennisetum giganteum and rice straw. Agriculture 2024, 14, 1953. [Google Scholar] [CrossRef]
- Martínez, J.R.P.F.; Huerta, A.G.; Lopez, D.D.J.P.; Cuevas, R.S.; Salem, A.Z.M.; Jimenez, L.E.R.; Ronquillo, M.G. Effect of xylanase, cellulase and natural maguey extract on the chemical composition of corn silage and in vitro rumen gas production. Int. J. Agric. Nat. Resour. 2020, 47, 23–34. [Google Scholar] [CrossRef]
- Hatew, B.; Cone, J.W.; Pellikaan, W.F.; Podesta, S.C.; Bannink, A.; Hendriks, W.H.; Dijkstra, J. Relationship between in vitro and in vivo methane production measured simultaneously with different dietary starch sources and starch levels in dairy cattle. Anim. Feed Sci. Technol. 2015, 202, 20–31. [Google Scholar] [CrossRef]
- Elghandour, M.M.M.Y.; Kholif, A.E.; Marquez-Molina, O.; Vazquez-Armijo, J.F.; Puniya, A.K.; Salem, A.Z.M. Influence of individual or mixed cellulase and xylanase mixture on in vitro rumen gas production kinetics of total mixed rations with different maize silage and concentrate ratios. Turk. J. Vet. Anim. Sci. 2015, 39, 435–442. [Google Scholar] [CrossRef]
- Blümmel, M.; Aiple, K.P.; Steingaß, H.; Becker, K. A note on the stoichiometrical relationship of short chain fatty acid production and gas formation in vitro in feedstuffs of widely differing quality. J. Anim. Physiol. Anim. Nutr. 1999, 81, 157–167. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.