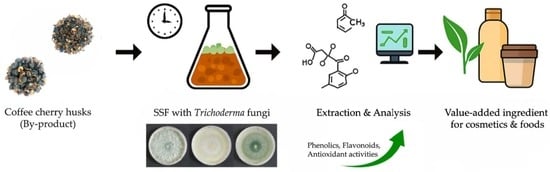
Bio-Enhancement of Phenolic Content and Antioxidant Capacity of Coffee (Coffea arabica L.) Cherry Husks by Solid-State Fermentation with Trichoderma Fungi
Abstract
1. Introduction
2. Materials and Methods
2.1. Microorganisms and Inoculum Preparation
2.2. Plant Material
2.3. Chemicals and Reagents
2.4. Solid-State Fermentation (SSF) of Coffee Cherry Husk
2.5. Preparation of Crude Extracts from Coffee Cherry Husks
2.6. Analyzes
2.6.1. Proximate Analysis
2.6.2. Determination of Extraction Yield
2.6.3. Determination of Total Phenolic Content
2.6.4. Determination of Total Flavonoid Content
2.6.5. Determination of 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Radical Scavenging Activity
2.6.6. Determination of ABTS Radical Scavenging Activity
2.6.7. Determination of Ferric Reducing Antioxidant Power (FRAP)
2.6.8. Identification of Bioactive Compounds by UHPLC-QqQ-MS/MS Analysis
2.7. Cytotoxicity
2.7.1. Cell Line
2.7.2. Cytotoxicity
2.8. Statistical Analysis
3. Results and Discussion
3.1. Growth of Trichoderma Fungi on Coffee Cherry Husk Substrate
3.2. Extraction Yield
3.3. Total Phenolic Content and Total Flavonoid Content
3.4. Antioxidant Activities
3.5. Correlation Analysis
3.6. Identification of Phenolic Compounds and Caffeine
3.7. Cytotoxicity and Cell Viability
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Melo Pereira, G.V.; de Carvalho Neto, D.P.; Magalhães Júnior, A.I.; do Prado, F.G.; Pagnoncelli, M.G.B.; Karp, S.G.; Soccol, C.R. Chemical composition and health properties of coffee and coffee by-products. Adv. Food Nutr. Res. 2020, 91, 65–96. [Google Scholar]
- Murthy, P.S.; Naidu, M.M. Sustainable Management of Coffee Industry By-Products and Value Addition-A Review. Resour. Conserv. Recycl. 2012, 66, 45–58. [Google Scholar] [CrossRef]
- Ramirez-Coronel, M.A.; Marnet, N.; Kolli, V.K.; Roussos, S.; Guyot, S.; Augur, C. Characterization and Estimation of Proanthocyanidins and Other Phenolics in Coffee Pulp (Coffea arabica) by Thiolysis−High-Performance Liquid Chromatography. J. Agric. Food Chem. 2004, 52, 1344–1349. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant Polyphenols as Dietary Antioxidants in Human Health and Disease. Oxidative Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Ali, A.; Bhowmik, S. Bioactive Compounds in Coffee Husk: Extraction, Functional Properties, Applications, and Sustainable Approach in Circular Economy. RSC Sustain. 2025, 3, 4410–4425. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, C.; Zhang, Y.; Zeng, W.; Cesarino, I. Coffee Cell Walls—Composition, Influence on Cup Quality and Opportunities for Coffee Improvements. Food Qual. Saf. 2021, 5, fyab012. [Google Scholar] [CrossRef]
- Rebollo-Hernanz, M.; Aguilera, Y.; Gil-Ramírez, A.; Benitez, V.; Cañas, S.; Braojos, C.; Martin-Cabrejas, M.A. Biorefinery and Stepwise Strategies for Valorizing Coffee By-Products as Bioactive Food Ingredients and Nutraceuticals. Appl. Sci. 2023, 13, 8326. [Google Scholar] [CrossRef]
- Álvarez, A.; Rodríguez, A.; Chaparro, S.; Borrás, L.M.; Rache, L.Y.; Brijaldo, M.H.; Martínez, J.J. Solid-State Fermentation as a Biotechnological Tool to Reduce Antinutrients and Increase Nutritional Content in Legumes and Cereals for Animal Feed. Fermentation 2025, 11, 359. [Google Scholar] [CrossRef]
- Soccol, C.R.; Da Costa, E.S.F.; Letti, L.A.J.; Karp, S.G.; Woiciechowski, A.L.; de Souza Vandenberghe, L.P. Recent Developments and Innovations in Solid State Fermentation. Biotechnol. Res. Innov. 2017, 1, 52–71. [Google Scholar] [CrossRef]
- Janarny, G.; Gunathilake, K.D.P.P. Changes in Rice Bran Bioactives, Their Bioactivity, Bioaccessibility and Bioavailability with Solid-State Fermentation by Rhizopus oryzae. Biocatal. Agric. Biotechnol. 2020, 23, 101510. [Google Scholar] [CrossRef]
- Singh, H.B.; Singh, B.N.; Singh, S.P.; Nautiyal, C.S. Solid-State Cultivation of Trichoderma harzianum NBRI-1055 for Modulating Natural Antioxidants in Soybean Seed Matrix. Bioresour. Technol. 2010, 101, 6444–6645. [Google Scholar] [CrossRef]
- Acosta-Estrada, B.A.; Villela-Castrejón, J.; Perez-Carrillo, E.; Gómez-Sánchez, C.E.; Gutiérrez-Uribe, J.A. Effects of Solid-State Fungi Fermentation on Phenolic Content, Antioxidant Properties and Fiber Composition of Lime Cooked Maize by-Product (Nejayote). J. Cereal Sci. 2019, 90, 102837. [Google Scholar] [CrossRef]
- Cangussu, L.B.; Melo, J.C.; Franca, A.S.; Oliveira, L.S. Chemical Characterization of Coffee Husks, a by-Product of Coffea arabica Production. Foods 2021, 10, 3125. [Google Scholar] [CrossRef]
- Verduzco-Oliva, R.; Gutierrez-Uribe, J.A. Beyond Enzyme Production: Solid State Fermentation (SSF) as an Alternative Approach to Produce Antioxidant Polysaccharides. Sustainability 2020, 12, 495. [Google Scholar] [CrossRef]
- Yao, X.; Guo, H.; Zhang, K.; Zhao, M.; Ruan, J.; Chen, J. Trichoderma and Its Role in Biological Control of Plant Fungal and Nematode Disease. Front. Microbiol. 2023, 14, 1160551. [Google Scholar] [CrossRef] [PubMed]
- Saleh, R.; Kabli, S.; Al-Garni, S.; Al-Ghamdi, M.; Abdel-Aty, A.; Mohamed, S. Solid-state Fermentation by Trichoderma viride for Enhancing Phenolic Content, Antioxidant and Antimicrobial Activities in Ginger. Lett. Appl. Microbiol. 2018, 67, 161–167. [Google Scholar] [CrossRef]
- Serna-Díaz, M.; Mercado-Flores, Y.; Jiménez-González, A.; Anducho-Reyes, M.; Medina-Marín, J.; Tuoh-Mora, J.S.; Téllez-Jurado, A. Use of Barley Straw as a Support for the Production of Conidiospores of Trichoderma harzianum. Biotechnol. Rep. 2020, 26, e00445. [Google Scholar] [CrossRef] [PubMed]
- Kritsadaruangchai, U.; Chaiwut, P.; Chomnunti, P.; Thaochan, N.; Saikeur, A.; Pintathong, P. Effect of Solid State Fermentation with Trichoderma spp. on Phenolic Content and Antioxidant Capacities of Mature Assam Tea Leaves. J. Food Sci. Agric. Technol. 2019, 5, 106–113. [Google Scholar]
- Tangjaidee, P.; Braspaiboon, S.; Singhadechachai, N.; Phongthai, S.; Therdtatha, P.; Rachtanapun, P.; Sommano, S.R.; Seesuriyachan, P. Enhanced Bioactive Coffee Cherry: Infusion of Submerged-Fermented Green Coffee Beans via Vacuum Impregnation. Foods 2025, 14, 1165. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, D.; Zhang, J.; Shen, J.; Cao, J.; Gu, H.; Cui, M.; He, L.; Chen, G.; Liu, S. Improving Soluble Phenolic Profile and Antioxidant Activity of Grape Pomace Seeds through Fungal Solid-State Fermentation. Foods 2024, 13, 1158. [Google Scholar] [CrossRef]
- Yang, F.; Hu, Y.; Wu, M.; Guo, M.; Wang, H. Biologically Active Components and Skincare Benefits of Rice Fermentation Products: A Review. Cosmetics 2025, 12, 29. [Google Scholar] [CrossRef]
- Majchrzak, W.; Motyl, I.; Śmigielski, K. Biological and Cosmetical Importance of Fermented Raw Materials: An Overview. Molecules 2022, 27, 4845. [Google Scholar] [CrossRef]
- Zhao, Y.-S.; Eweys, A.S.; Zhang, J.-Y.; Zhu, Y.; Bai, J.; Darwesh, O.M.; Zhang, H.-B.; Xiao, X. Fermentation Affects the Antioxidant Activity of Plant-Based Food Material through the Release and Production of Bioactive Components. Antioxidants 2021, 10, 2004. [Google Scholar] [CrossRef]
- Yu, H.; Khanum, S.; Ali, M.M.; Rahman, A.; Hayat, Z.; Waqas, M.; Riaz, R.; Sajid, M.; Khan, M.; Zafar, M.S. Research Note: Effects of Fermented Vegetable Extract Supplementation via Drinking Water on Growth Performance, Immune Function, and Antioxidant Status in Broiler Chickens. Poult. Sci. 2025, 104, 105583. [Google Scholar] [CrossRef]
- Leeuwendaal, N.K.; Stanton, C.; O’toole, P.W.; Beresford, T.P. Fermented Foods, Health and the Gut Microbiome. Nutrients 2022, 14, 1527. [Google Scholar] [CrossRef]
- Sarangi, P.K.; Vivekanand, V.; Mohanakrishna, G.; Pattnaik, B.; Muddapur, U.M.; Aminabhavi, T.M. Production of Bioactive Phenolic Compounds from Agricultural By-Products towards Bioeconomic Perspectives. J. Clean. Prod. 2023, 414, 137460. [Google Scholar] [CrossRef]
- Messinese, E.; Pitirollo, O.; Grimaldi, M.; Milanese, D.; Sciancalepore, C.; Cavazza, A. By-Products as Sustainable Source of Bioactive Compounds for Potential Application in the Field of Food and New Materials for Packaging Development. Food Bioprocess Technol. 2024, 17, 606–627. [Google Scholar] [CrossRef]
- Tajik, N.; Tajik, M.; Mack, I.; Enck, P. The Potential Effects of Chlorogenic Acid, the Main Phenolic Components in Coffee, on Health: A Comprehensive Review of the Literature. Eur. J. Nutr. 2017, 56, 2215–2244. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, R.; Oliveira, M.B.P.P.; Alves, R.C. Chlorogenic Acids and Caffeine from Coffee By-Products: A Review on Skincare Applications. Cosmetics 2023, 10, 12. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis of AOAC International, 17th ed.; AOAC International: Gaithersburg, MD, USA, 2000. [Google Scholar]
- Fattahi, S.; Zabihi, E.; Abedian, Z.; Pourbagher, R.; Ardekani, A.M.; Mostafazadeh, A.; Akhavan-Niaki, H. Total Phenolic and Flavonoid Contents of Aqueous Extract of Stinging Nettle and in Vitro Antiproliferative Effect on Hela and BT-474 Cell Lines. Int. J. Mol. Cell. Med. 2014, 3, 102. [Google Scholar] [PubMed]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Byrne, D.H. Comparison of ABTS, DPPH, FRAP, and ORAC Assays for Estimating Antioxidant Activity from Guava Fruit Extracts. J. Food Compos. Anal. 2006, 19, 669–675. [Google Scholar] [CrossRef]
- Solaberrieta, I.; Jiménez, A.; Cacciotti, I.; Garrigós, M.C. Encapsulation of Bioactive Compounds from Aloe Vera Agrowastes in Electrospun Poly (Ethylene Oxide) Nanofibers. Polymers 2020, 12, 1323. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Pramanik, F.; Satari, M.H.; Azhari, A. Cytotoxic Activity of Gambier Leave (Uncaria gambir) Ethyl Acetate Extract on Mouse Embryonic Fibroblast Cell (NIH-3T3) Using MTT Assay. Open Dent. J. 2023, 17, e187421062212300. [Google Scholar] [CrossRef]
- Bulgari, D.; Alias, C.; Peron, G.; Ribaudo, G.; Gianoncelli, A.; Savino, S.; Boureghda, H.; Bouznad, Z.; Monti, E.; Gobbi, E. Solid-State Fermentation of Trichoderma spp.: A New Way to Valorize the Agricultural Digestate and Produce Value-Added Bioproducts. J. Agric. Food Chem. 2023, 71, 3994–4004. [Google Scholar] [CrossRef]
- Vitale, S.; Salzano, F.; Staropoli, A.; Marra, R.; Turrà, D.; Lorito, M.; Vinale, F. Nitrogen Source Orchestrates pH Modulation and Secondary Metabolism in Trichoderma harzianum. Chem. Biol. Technol. Agric. 2025, 12, 19. [Google Scholar] [CrossRef]
- Hoseini, M.; Cocco, S.; Casucci, C.; Cardelli, V.; Corti, G. Coffee By-Products Derived Resources. A Review. Biomass Bioenergy 2021, 148, 106009. [Google Scholar] [CrossRef]
- Harman, G.E.; Howell, C.R.; Viterbo, A.; Chet, I.; Lorito, M. Trichoderma Species—Opportunistic, Avirulent Plant Symbionts. Nat. Rev. Microbiol. 2004, 2, 43–56. [Google Scholar] [CrossRef]
- Belščak-Cvitanović, A.; Komes, D. Extraction and formulation of bioactive compounds. In Handbook of Coffee Processing By-Products: Sustainable Applications, 1st ed.; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 93–140. [Google Scholar]
- García, L.R.P.; Del Bianchi, V.L. The Effect of Fungal Fermentation in Phenolics Content in Robusta Coffee Husk. Semin. Ciências Agrárias 2015, 36, 777–785. [Google Scholar] [CrossRef]
- Meneses, N.G.; Martins, S.; Teixeira, J.A.; Mussatto, S.I. Influence of Extraction Solvents on the Recovery of Antioxidant Phenolic Compounds from Brewer’s Spent Grains. Sep. Purif. Technol. 2013, 108, 152–158. [Google Scholar] [CrossRef]
- Ribeiro, E.F.; Luzia, D.M.M.; Jorge, N. Antioxidant Compounds Extraction from Coffee Husks: The Influence of Solvent Type and Ultrasound Exposure Time. Acta Sci. Technol. 2019, 41, e36451. [Google Scholar] [CrossRef]
- De Silva, M.O.; Honfoga, J.N.B.; de Medeiros, L.L.; Madruga, M.S.; Bezerra, T.K.A. Obtaining Bioactive Compounds from the Coffee Husk (Coffea arabica L.) Using Different Extraction Methods. Molecules 2020, 26, 46. [Google Scholar] [CrossRef]
- Londoño-Hernandez, L.; Ruiz, H.A.; Ramírez, T.C.; Ascacio, J.A.; Rodríguez-Herrera, R.; Aguilar, C.N. Fungal Detoxification of Coffee Pulp by Solid-State Fermentation. Biocatal. Agric. Biotechnol. 2020, 23, 101467. [Google Scholar] [CrossRef]
- Palomino Garcia, L.R.; Biasetto, C.R.; Araujo, A.R.; Bianchi, V.L.D. Enhanced Extraction of Phenolic Compounds from Coffee Industry’s Residues through Solid State Fermentation by Penicillium purpurogenum. Food Sci. Technol. 2015, 35, 704–711. [Google Scholar] [CrossRef]
- Nguyen Thai, H.; Van Camp, J.; Smagghe, G.; Raes, K. Improved Release and Metabolism of Flavonoids by Steered Fermentation Processes: A Review. Int. J. Mol. Sci. 2014, 15, 19369–19388. [Google Scholar] [CrossRef] [PubMed]
- Torino, M.I.; Limón, R.I.; Martínez-Villaluenga, C.; Mäkinen, S.; Pihlanto, A.; Vidal-Valverde, C.; Frias, J. Antioxidant and Antihypertensive Properties of Liquid and Solid State Fermented Lentils. Food Chem. 2013, 136, 1030–1037. [Google Scholar] [CrossRef]
- Torres-Mancera, M.T.; Cordova-López, J.; Rodríguez-Serrano, G.; Roussos, S.; Ramírez-Coronel, M.A.; Favela-Torres, E.; Saucedo-Castañeda, G. Enzymatic Extraction of Hydroxycinnamic Acids from Coffee Pulp. Food Technol. Biotechnol. 2011, 49, 369–373. [Google Scholar]
- Erskine, E.; Ozkan, G.; Lu, B.; Capanoglu, E. Effects of Fermentation Process on the Antioxidant Capacity of Fruit Byproducts. ACS Omega 2023, 8, 4543–4553. [Google Scholar] [CrossRef]
- Schober, P.; Boer, C.; Schwarte, L.A. Correlation Coefficients: Appropriate Use and Interpretation. Anesth. Analg. 2018, 126, 1763–1768. [Google Scholar] [CrossRef]
- Tan, Y.; Wu, H.; Shi, L.; Barrow, C.; Dunshea, F.R.; Suleria, H.A. Impacts of Fermentation on the Phenolic Composition, Antioxidant Potential, and Volatile Compounds Profile of Commercially Roasted Coffee Beans. Fermentation 2023, 9, 918. [Google Scholar] [CrossRef]
- Patial, P.K.; Sharma, A.; Kaur, I.; Cannoo, D.S. Correlation Study among the Extraction Techniques, Phytochemicals, and Antioxidant Activity of Nepeta Spicata Aerial Part. Biocatal. Agric. Biotechnol. 2019, 20, 101275. [Google Scholar] [CrossRef]
- Khasanah, H.; Widianingrum, D.C.; Purnamasari, L.; Wafa, A.; Hwang, S.-G. Evaluation of Coffee Bean Husk Fermented by a Combination of Aspergillus niger, Trichoderma harzianum, and Saccharomyces cerevisiae as Animal Feed. J. Ilmu-Ilmu Peternak. 2022, 32, 416–426. [Google Scholar] [CrossRef]
- Lin, Z.; Wei, J.; Hu, Y.; Pi, D.; Jiang, M.; Lang, T. Caffeine Synthesis and Its Mechanism and Application by Microbial Degradation: A Review. Foods 2023, 12, 2721. [Google Scholar] [CrossRef]
- Mock, M.B.; Summers, R.M. Microbial Metabolism of Caffeine and Potential Applications in Bioremediation. J. Appl. Microbiol. 2024, 135, lxae080. [Google Scholar] [CrossRef]
- Vandeponseele, A.; Draye, M.; Piot, C.; Chatel, G. Study of Influential Parameters of the Caffeine Extraction from Spent Coffee Grounds: From Brewing Coffee Method to the Waste Treatment Conditions. Clean Technol. 2021, 3, 335–350. [Google Scholar] [CrossRef]
- Wu, L. Effect of Chlorogenic Acid on Antioxidant Activity of Flos lonicerae Extracts. J. Zhejiang Univ. Sci. B 2007, 8, 673–679. [Google Scholar] [CrossRef]
- Zhang, Z.; Lv, G.; Pan, H.; Fan, L.; Soccol, C.R.; Pandey, A. Production of Powerful Antioxidant Supplements via Solid-State Fermentation of Wheat (Triticum aestivum L.) by Cordyceps militaris. Food Technol. Biotechnol. 2012, 50, 32–39. [Google Scholar]
- Razak, D.L.A.; Rashid, N.Y.A.; Jamaluddin, A.; Sharifudin, S.A.; Long, K. Enhancement of Phenolic Acid Content and Antioxidant Activity of Rice Bran Fermented with Rhizopus oligosporus and Monascus purpureus. Biocatal. Agric. Biotechnol. 2015, 4, 33–38. [Google Scholar] [CrossRef]
- Buenrostro-Figueroa, J.J.; Velázquez, M.; Flores-Ortega, O.; Ascacio-Valdés, J.A.; Huerta-Ochoa, S.; Aguilar, C.N.; Prado-Barragán, L.A. Solid State Fermentation of Fig (Ficus carica L.) by-Products Using Fungi to Obtain Phenolic Compounds with Antioxidant Activity and Qualitative Evaluation of Phenolics Obtained. Process Biochem. 2017, 62, 16–23. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, Y.; Chen, J.; Tang, H.; Wang, C.; Li, Z.; Xiao, Y. Bioprocessing of Soybeans (Glycine max L.) by Solid-State Fermentation with Eurotium cristatum YL-1 Improves Total Phenolic Content, Isoflavone Aglycones, and Antioxidant Activity. RSC Adv. 2020, 10, 16928–16941. [Google Scholar] [CrossRef]
- Arnous, A.; Meyer, A.S. Grape Skins (Vitis vinifera L.) Catalyze the in Vitro Enzymatic Hydroxylation of p-Coumaric Acid to Caffeic Acid. Biotechnol. Lett. 2009, 31, 1953–1960. [Google Scholar] [CrossRef] [PubMed]
- Ni, H.; Chen, F.; Jiang, Z.D.; Cai, M.Y.; Yang, Y.F.; Xiao, A.F.; Cai, H.N. Biotransformation of tea catechins using Aspergillus niger tannase prepared by solid state fermentation on tea byproduct. LWT–Food Sci. Technol. 2015, 60, 1206–1213. [Google Scholar] [CrossRef]
- Ritthibut, N.; Oh, S.-J.; Lim, S.-T. Enhancement of Bioactivity of Rice Bran by Solid-State Fermentation with Aspergillus Strains. LWT 2021, 135, 110273. [Google Scholar] [CrossRef]
- Haq, H.M.S.U.; Ashfaq, R.; Mehmood, A.; Shahid, W.; Azam, H.G.; Azam, M.; Tasneem, S.; Akram, S.J.; Malik, K.; Riazuddin, S. Priming with Caffeic Acid Enhances the Potential and Survival Ability of Human Adipose-Derived Stem Cells to Counteract Hypoxia. Regen. Ther. 2023, 22, 115–127. [Google Scholar] [CrossRef]
- Kant, V.; Jangir, B.L.; Kumar, V.; Nigam, A.; Sharma, V. Quercetin Accelerated Cutaneous Wound Healing in Rats by Modulation of Different Cytokines and Growth Factors. Growth Factors 2020, 38, 105–119. [Google Scholar] [CrossRef]
- Zhao, B.; Hu, M. Gallic Acid Reduces Cell Viability, Proliferation, Invasion and Angiogenesis in Human Cervical Cancer Cells. Oncol. Lett. 2013, 6, 1749–1755. [Google Scholar] [CrossRef] [PubMed]




| Culture Conditions | Extraction Yield (% w/w) | ||
|---|---|---|---|
| Aqueous Extract | Ethanolic Extract | Acetonic Extract | |
| Unfermented | 32.13 ± 0.49 aA | 23.90 ± 1.31 aB | 13.90 ± 0.17 aC |
| Fermented with CB-Pin-01 | 26.47 ± 1.37 bA | 11.23 ± 1.76 cB | 8.27 ± 0.32 cC |
| Fermented with NTY211 | 20.97 ± 0.35 cA | 9.67 ± 0.25 cB | 8.50 ± 0.61 dC |
| Fermented with PSUT001 | 31.17 ± 0.63 aA | 18.17 ± 0.15 bB | 9.63 ± 0.35 bC |
| TPC | TFC | DPPH | ABTS | |
|---|---|---|---|---|
| TFC | 0.933 ** | |||
| DPPH | 0.966 ** | 0.881 ** | ||
| ABTS | 0.918 ** | 0.868 ** | 0.821 ** | |
| FRAP | 0.979 ** | 0.941 ** | 0.947 ** | 0.905 ** |
| Compounds | Gallic Acid | Protocatechuic Acid | Chlorogenic Acid | Caffeic Acid | p-Coumaric Acid | Quercetin | Caffeine | Total Phenolic Content | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Molecular formula | C7H6O5 | C7H6O4 | C16H18O9 | C9H8O4 | C9H8O3 | C15H10O7 | C8H10N4O2 | |||
| Precursor ion, [M-H]− (m/z) | 169.10 | 153.25 | 352.95 | 178.80 | 162.85 | 300.75 | 195.00 | |||
| Retention time (min) | 2.22 | 3.40 | 5.37 | 6.46 | 9.37 | 12.10 | 4.96 | |||
| Extract | Strain/Isolate | |||||||||
| Concentration (mg/g extract) | Aqueous extract | Unfermented | 0.178 | 0.179 | 11.066 | 0.032 | n.d. | 0.003 | 9.593 | 11.458 |
| CB-Pin-01 | 0.013 | 0.032 | 17.752 | 0.030 | n.d. | 0.005 | 11.215 | 17.832 | ||
| NTY211 | 0.047 | 0.005 | 34.836 | 0.070 | n.d. | 0.003 | 15.815 | 34.961 | ||
| PSUT001 | 0.213 | 0.001 | 21.471 | 0.041 | n.d. | 0.006 | 10.294 | 21.733 | ||
| Ethanolic extract | Unfermented | 0.279 | 0.364 | 26.851 | 0.116 | 0.027 | 0.126 | 15.091 | 27.762 | |
| CB-Pin-01 | 0.044 | 0.080 | 49.547 | 0.117 | 0.003 | 0.213 | 35.887 | 50.005 | ||
| NTY211 | 0.089 | 0.009 | 62.157 | 0.127 | 0.001 | 0.354 | 42.699 | 62.739 | ||
| PSUT001 | 0.398 | 0.399 | 37.826 | 0.117 | 0.011 | 0.183 | 22.970 | 38.935 | ||
| Acetonic extract | Unfermented | 0.636 | 0.789 | 46.968 | 0.141 | 0.041 | 0.321 | 29.737 | 48.896 | |
| CB-Pin-01 | 0.083 | 0.111 | 49.134 | 0.130 | 0.001 | 0.398 | 51.005 | 49.857 | ||
| NTY211 | 0.054 | 0.017 | 60.676 | 0.160 | 0.004 | 0.960 | 74.618 | 61.871 | ||
| PSUT001 | 0.582 | 0.240 | 40.740 | 0.113 | 0.006 | 0.409 | 45.853 | 42.090 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pintathong, P.; Thaochan, N.; Suwannawong, B.; Sangthong, S.; Chaiwut, P. Bio-Enhancement of Phenolic Content and Antioxidant Capacity of Coffee (Coffea arabica L.) Cherry Husks by Solid-State Fermentation with Trichoderma Fungi. Fermentation 2025, 11, 625. https://doi.org/10.3390/fermentation11110625
Pintathong P, Thaochan N, Suwannawong B, Sangthong S, Chaiwut P. Bio-Enhancement of Phenolic Content and Antioxidant Capacity of Coffee (Coffea arabica L.) Cherry Husks by Solid-State Fermentation with Trichoderma Fungi. Fermentation. 2025; 11(11):625. https://doi.org/10.3390/fermentation11110625
Chicago/Turabian StylePintathong, Punyawatt, Narit Thaochan, Benjarat Suwannawong, Sarita Sangthong, and Phanuphong Chaiwut. 2025. "Bio-Enhancement of Phenolic Content and Antioxidant Capacity of Coffee (Coffea arabica L.) Cherry Husks by Solid-State Fermentation with Trichoderma Fungi" Fermentation 11, no. 11: 625. https://doi.org/10.3390/fermentation11110625
APA StylePintathong, P., Thaochan, N., Suwannawong, B., Sangthong, S., & Chaiwut, P. (2025). Bio-Enhancement of Phenolic Content and Antioxidant Capacity of Coffee (Coffea arabica L.) Cherry Husks by Solid-State Fermentation with Trichoderma Fungi. Fermentation, 11(11), 625. https://doi.org/10.3390/fermentation11110625