Lactic Acid Fermentation as a Valorising Agent for Brewer’s Spent Yeast—Improving the Sensory Quality and Nutritional Potential
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material and Bacterial Strain
2.2. BSY Fermentation
2.3. Characterisation of the Fermentation Profiles
The pH, TTA, Alcohol and Microbial Growth Determination
2.4. Dried Ingredient Characterisation
2.4.1. Compositional Analysis
2.4.2. Sensory Attributes
2.4.3. Aroma Compounds
2.4.4. Protein Profile
2.4.5. Metabolomics
2.4.6. International Bitterness Units
2.4.7. Colour
2.4.8. Ultrastructure
2.5. Statistical Analysis
3. Results
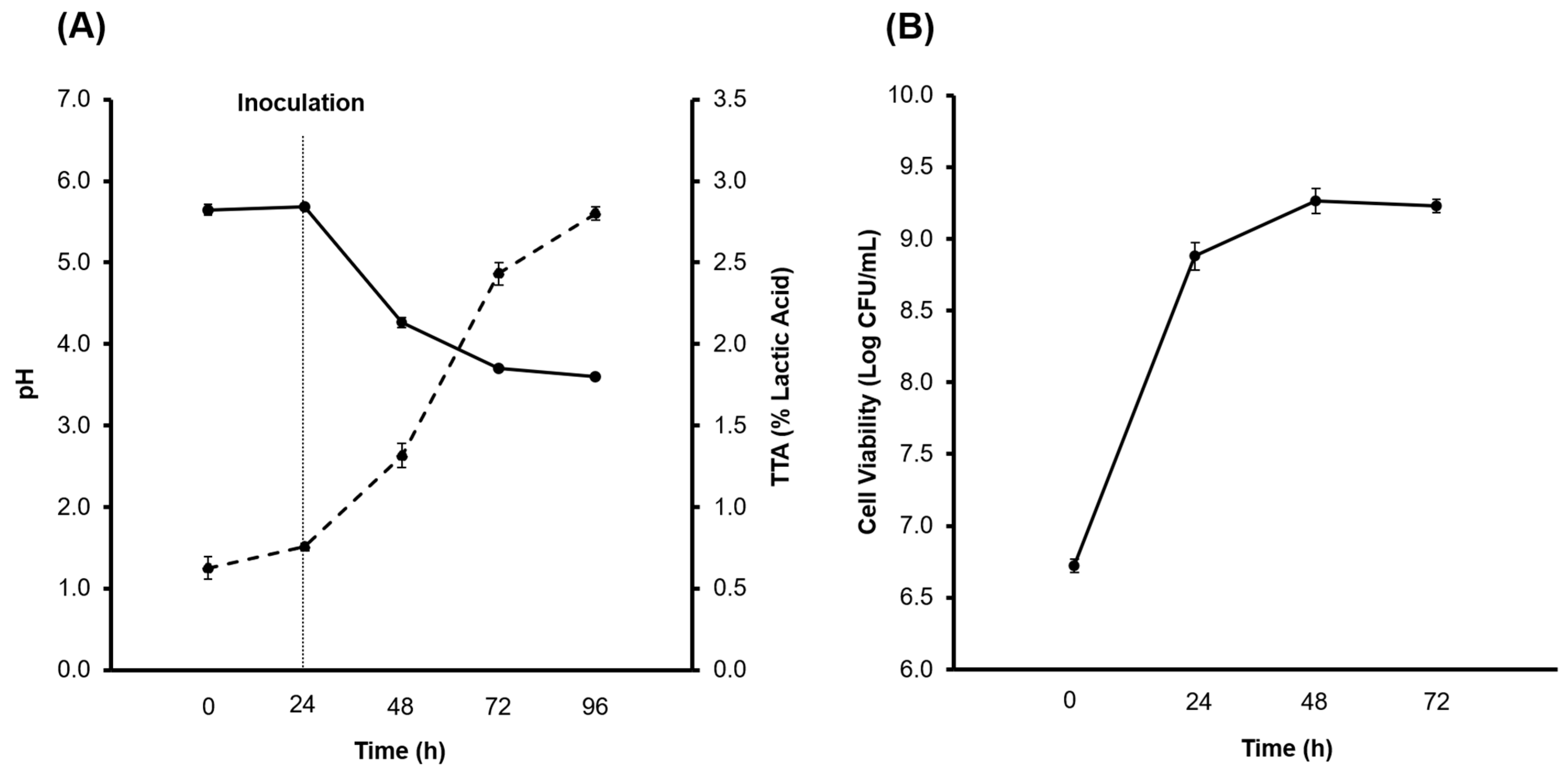
3.1. Production of the CBSY and PBSY Ingredients
3.2. Compositional Analysis
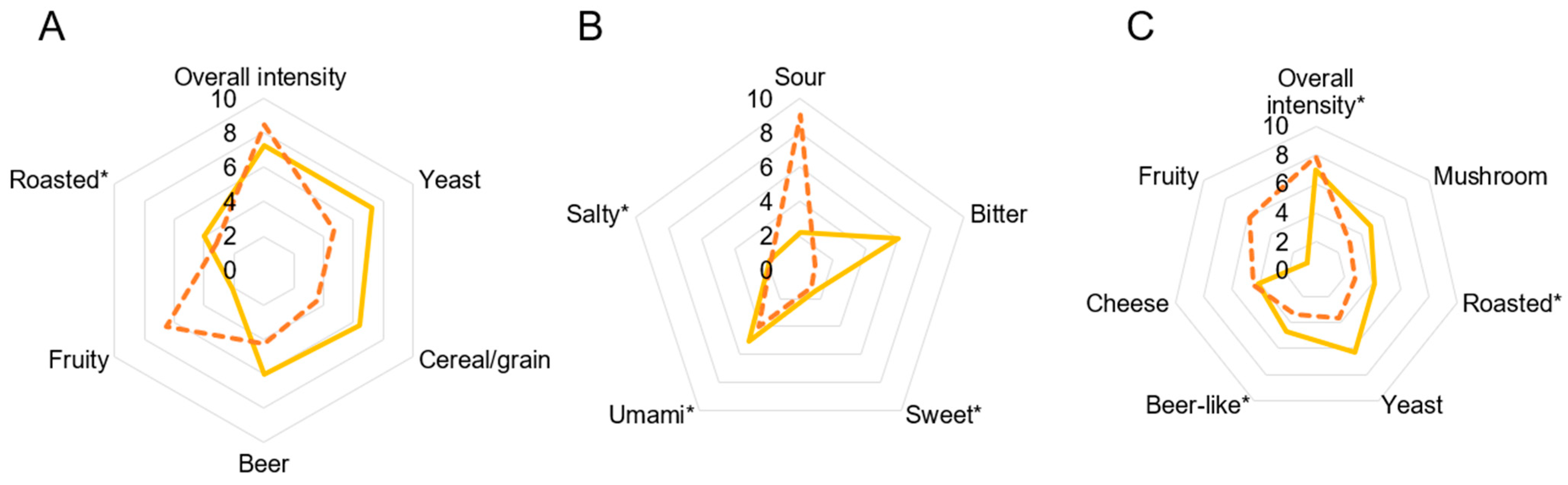
3.3. Sensory Analysis
3.4. Aroma Compounds
3.5. Free and Total Amino Acid Composition
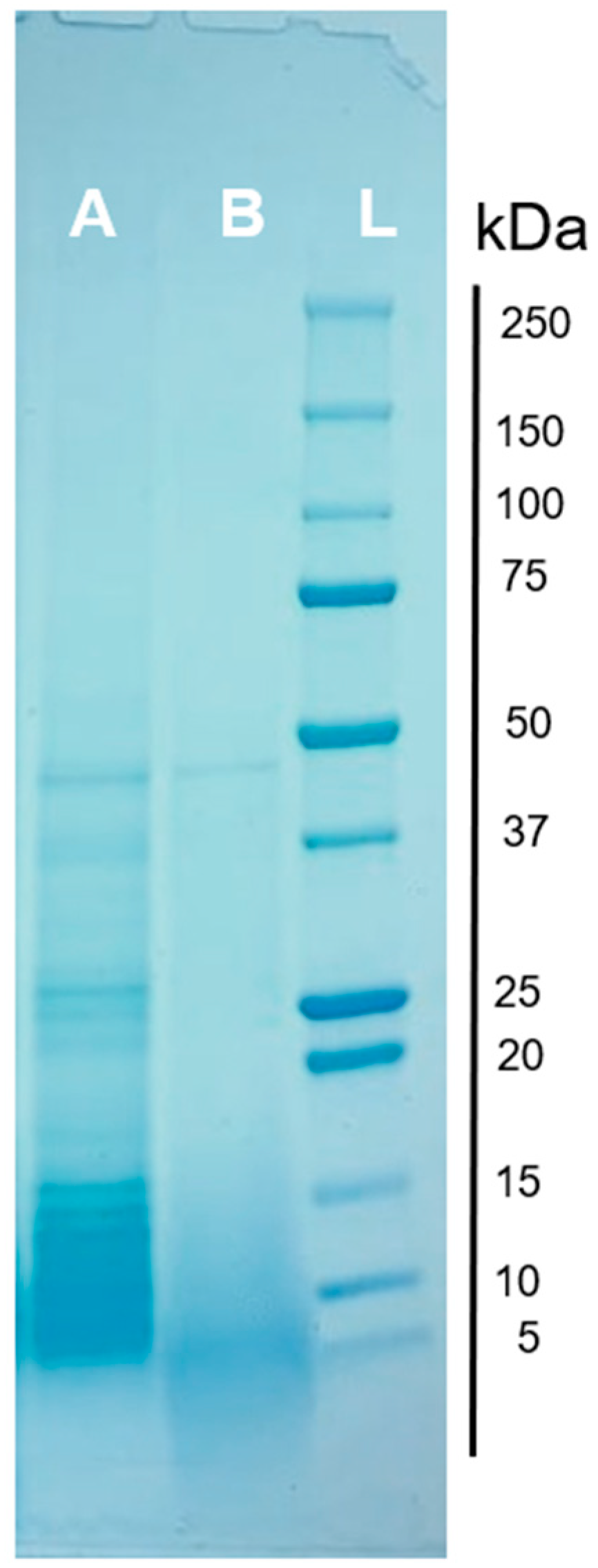
3.6. Protein Profile
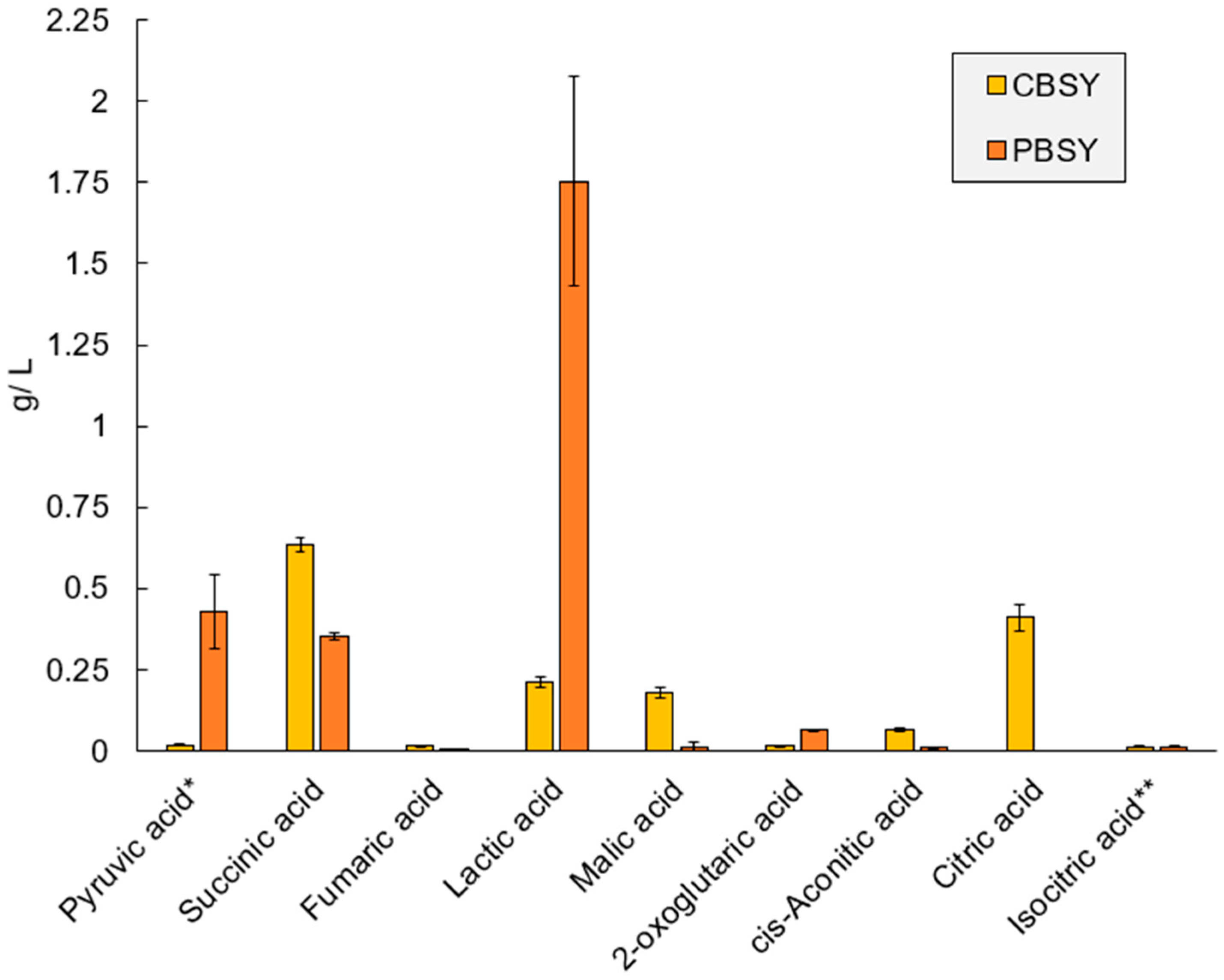
3.7. Metabolomic Analysis
3.8. Appearance and Ultrastructure
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kordialik-Bogacka, E.; Diowksz, A. Physiological State of Reused Brewing Yeast. Czech J. Food Sci. 2013, 31, 264–269. [Google Scholar] [CrossRef]
- Ferreira, I.M.P.L.V.O.; Pinho, O.; Vieira, E.; Tavarela, J.G. Brewer ’s Saccharomyces Yeast Biomass: Characteristics and Potential Applications. Trends Food Sci. Technol. 2010, 21, 77–84. [Google Scholar] [CrossRef]
- Kunze, W. Technology Brewing and Malting, 2nd ed.; VLB Berlin: Berlin, Germany, 1999. [Google Scholar]
- Jaeger, A.; Arendt, E.K.; Zannini, E.; Sahin, A.W. Brewer’s Spent Yeast (BSY), an Underutilized Brewing By-Product. Fermentation 2020, 6, 123. [Google Scholar] [CrossRef]
- Harlow, B.E.; Bryant, R.W.; Cohen, S.D.; O’Connell, S.P.; Flythe, M.D. Degradation of Spent Craft Brewer’s Yeast by Caprine Rumen Hyper Ammonia-Producing Bacteria. Lett. Appl. Microbiol. 2016, 63, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.L.; Oliveira, R.J.F.; Bezerra, L.R.; Nascimento, T.V.C.; de Pellegrini, C.B.; de Freitas Neto, M.D.; do Nascimento, N.G.; de Souza, W.F. Substitution of Corn Meal with Dry Brewer’s Yeast in the Diet of Sheep. Rev. Colomb. Ciencias Pecu. 2016, 29, 99–107. [Google Scholar] [CrossRef]
- Estévez, A.; Padrell, L.; Iñarra, B.; Orive, M.; Martin, D.S. Brewery By-Products (Yeast and Spent Grain) as Protein Sources in Gilthead Seabream (Sparus Aurata) Feeds. Aquaculture 2021, 543, 736921. [Google Scholar] [CrossRef]
- San Martin, D.; Orive, M.; Iñarra, B.; Castelo, J.; Estévez, A.; Nazzaro, J.; Iloro, I.; Elortza, F.; Zufía, J. Brewers’ Spent Yeast and Grain Protein Hydrolysates as Second-Generation Feedstuff for Aquaculture Feed. Waste Biomass Valorization 2020, 11, 5307–5320. [Google Scholar] [CrossRef]
- Nand, K. Debittering of Spent Brewer’s Yeast for Food Purposes. Food/Nahrung 1987, 31, 127–131. [Google Scholar] [CrossRef]
- Shotipruk, A.; Kittianong, P.; Suphantharika, M.; Muangnapoh, C. Application of Rotary Microfiltration in Debittering Process of Spent Brewer’s Yeast. Bioresour. Technol. 2005, 96, 1851–1859. [Google Scholar] [CrossRef]
- Dwivedi, B.K.; Gibson, D.I. Processing of Spent Brewers ’ Yeast for Food Use. Can. Inst. Food Technol. J. 1970, 3, 110–112. [Google Scholar] [CrossRef]
- Ganzle, M.G. Lactic Metabolism Revisited: Metabolism of Lactic Acid Bacteria in Food Fermentations and Food Spoilage. Curr. Opin. Food Sci. 2015, 2, 106–117. [Google Scholar] [CrossRef]
- Montel, M.C.; Masson, F.; Talon, R. Bacterial Role in Flavour Development. Meat Sci. 1998, 49, S111–S123. [Google Scholar] [CrossRef]
- Caplice, E.; Fitzgerald, G.F. Food Fermentations: Role of Microorganisms in Food Production and Preservation. Int. J. Food Microbiol. 1999, 50, 131–149. [Google Scholar] [CrossRef] [PubMed]
- Ryan, L.A.M.; Zannini, E.; Dal, F.; Pawlowska, A.; Koehler, P.; Arendt, E.K. Lactobacillus Amylovorus DSM 19280 as a Novel Food-Grade Antifungal Agent for Bakery Products. Int. J. Food Microbiol. 2011, 146, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Peyer, L.C.; Zarnkow, M.; Jacob, F.; De Schutter, D.P.; Arendt, E.K. Sour Brewing: Impact of Lactobacillus Amylovorus FST2.11 on Technological and Quality Attributes of Acid Beers. J. Am. Soc. Brew. Chem. 2017, 75, 207–216. [Google Scholar] [CrossRef]
- Lowe, D.P.; Ulmer, H.M.; Barta, R.C.; Goode, D.L.; Arendt, E.K. Biological Acidification of a Mash Containing 20% Barley Using Lactobacillus Amylovorus FST 1.1: Its Effects on Wort and Beer Quality. J. Am. Soc. Brew. Chem. 2005, 63, 96–106. [Google Scholar] [CrossRef]
- Lynch, K.M.; Pawlowska, A.M.; Brosnan, B.; Coffey, A.; Zannini, E.; Furey, A.; Mcsweeney, P.L.H.; Waters, D.M.; Arendt, E.K. Application of Lactobacillus Amylovorus as an Antifungal Adjunct to Extend the Shelf-Life of Cheddar Cheese. Int. Dairy J. 2014, 34, 167–173. [Google Scholar] [CrossRef]
- Gold, R.S.; Meagher, M.M.; Hutkins, R.; Conway, T. Ethanol Tolerance and Carbohydrate Metabolism in Lactobacilli. J. Ind. Microbiol. 1992, 10, 45–54. [Google Scholar] [CrossRef]
- Neylon, E.; Nyhan, L.; Zannini, E.; Monin, T.; Münch, S.; Sahin, A.W.; Arendt, E.K. Food Ingredients for the Future: In-Depth Analysis of the Effects of Lactic Acid Bacteria Fermentation on Spent Barley Rootlets. Fermentation 2023, 9, 78. [Google Scholar] [CrossRef]
- Nyhan, L.; Sahin, A.W.; Arendt, E.K. Co-Fermentation of Non-Saccharomyces Yeasts with Lactiplantibacillus Plantarum FST 1.7 for the Production of Non-Alcoholic Beer. Eur. Food Res. Technol. 2023, 249, 167–181. [Google Scholar] [CrossRef]
- AACC International Approved Methods of Analysis, 11th Edn. Method 02-31.01-Titratable Acidity 2011. Available online: https://www.cerealsgrains.org/resources/Methods/Pages/02Acidity.aspx (accessed on 13 December 2023).
- Bellut, K.; Michel, M.; Hutzler, M.; Zarnkow, M.; Jacob, F.; De Schutter, D.P.; Daenen, L.; Lynch, K.M.; Zannini, E.; Arendt, E.K. Investigation into the Potential of Lachancea Fermentati Strain KBI 12.1 for Low Alcohol Beer Brewing. J. Am. Soc. Brew. Chem. 2019, 77, 157–169. [Google Scholar] [CrossRef]
- Baldini, M.; Fabietti, F.; Giammarioli, S.; Onori, R.; Orefice, L.; Stacchini, A. Metodi Di Analisi Utilizzati per Il Controllo Chimico Degli Alimenti; Istituto Superiore Di Sanita: Rome, Italy, 1996. [Google Scholar]
- AOAC International AOAC Official Method 2015.06-2015 Minerals and Trace Elements in Infant Formula 2015. Available online: http://www.aoacofficialmethod.org/index.php?main_page=product_info&products_id=2937 (accessed on 13 December 2023).
- UNE EN 15763:2010; Determination of Trace Elements—Determination of Arsenic, Cadmium, Mercury and Lead in Foodstuffs by Inductively Coupled Plasma Mass Spectrometry (ICP-MS) after Pressure Digestion. iTeh, Inc.: Newark, DE, USA, 2010.
- Environmental Protection Agency Analytical Method EPA Method 6020B (SW-846) Inductively Coupled Plasma—Mass Spectrometry 2014. Available online: https://www.epa.gov/esam/epa-method-6020b-sw-846-inductively-coupled-plasma-mass-spectrometry (accessed on 13 December 2023).
- AACC International AACC Approved Methods of Analysis, 11th Edition 32-05.01 Total Dietary Fiber 2011. Available online: https://www.cerealsgrains.org/resources/Methods/Pages/32Fiber.aspx (accessed on 13 December 2023).
- Smart, K.F.; Aggio, R.B.M.; Van Houtte, J.R.; Villas-Bôas, S.G. Analytical Platform for Metabolome Analysis of Microbial Cells Using Methyl Chloroformate Derivatization Followed by Gas Chromatography–Mass Spectrometry. Nat. Protoc. 2010, 5, 1709–1729. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, L.G.; Skou, P.B.; Khakimov, B.; Bro, R. Gas Chromatography Mass Spectrometry Data Processing Made Easy. J. Chromatogr. A 2017, 1503, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Jacob, F. Wort, Beer, Beer-Based Beverages: Collection of Brewing Analysis Methods of the Mitteleuropäische Brautechnische Analysenkommission; MEBAK: Freising-Weienstephan, Germany, 2013; ISBN 9783980581479/3980581470. [Google Scholar]
- Atzler, J.J.; Sahin, A.W.; Gallagher, E.; Zannini, E.; Arendt, E.K. Investigation of Different Dietary-Fibre-Ingredients for the Design of a Fibre Enriched Bread Formulation Low in FODMAPs Based on Wheat Starch and Vital Gluten. Eur. Food Res. Technol. 2021, 247, 1939–1957. [Google Scholar] [CrossRef]
- The Good Scents Company. Available online: https://www.thegoodscentscompany.com/index.html (accessed on 8 December 2021).
- FAO. Dietary Protein Quality Evaluation in Human Nutrition; FAO: Rome, Italy, 2013; ISBN 9789251074176. [Google Scholar]
- Neylon, E.; Arendt, E.K.; Zannini, E.; Sahin, A.W. Fermentation as a Tool to Revitalise Brewers’ Spent Grain and Elevate Techno-Functional Properties and Nutritional Value in High Fibre Bread. Foods 2021, 10, 1639. [Google Scholar] [CrossRef]
- dos Santos Mathias, T.R.; Alexandre, V.M.F.; Cammarota, M.C.; de Mello, P.P.M.; Sérvulo, E.F.C. Characterization and Determination of Brewer’s Solid Wastes Composition. J. Inst. Brew. 2015, 121, 400–404. [Google Scholar] [CrossRef]
- Vieira, E.F.; Carvalho, J.; Pinto, E.; Cunha, S.; Almeida, A.A.; Ferreira, I.M.P.L.V.O. Nutritive Value, Antioxidant Activity and Phenolic Compounds Profile of Brewer’s Spent Yeast Extract. J. Food Compos. Anal. 2016, 52, 44–51. [Google Scholar] [CrossRef]
- Pinto, M.; Coelho, E.; Nunes, A.; Brandão, T.; Coimbra, M.A. Valuation of Brewers Spent Yeast Polysaccharides: A Structural Characterization Approach. Carbohydr. Polym. 2015, 116, 215–222. [Google Scholar] [CrossRef]
- Van Steenwijk, H.P.; Bast, A.; de Boer, A. Immunomodulating Effects of Fungal Beta-Glucans: From Traditional Use to Medicine. Nutrients 2021, 13, 1333. [Google Scholar] [CrossRef]
- Du, B.; Lin, C.; Bian, Z.; Xu, B. An Insight into Anti-Inflammatory Effects of Fungal Beta-Glucans. Trends Food Sci. Technol. 2014, 41, 49–59. [Google Scholar] [CrossRef]
- Jawhara, S.; Habib, K.; Pignede, G.; Vandekerckove, P.; Maes, E.; Dubuquoy, L.; Fontaine, T.; Guerardel, Y.; Poulain, D. Modulation of Intestinal Inflammation by Yeasts and Cell Wall Extracts: Strain Dependence and Unexpected Anti-Inflammatory Role of Glucan Fractions. PLoS ONE 2012, 7, e40648. [Google Scholar] [CrossRef] [PubMed]
- Sempio, R.; Sahin, A.W.; Walter, J.; Arendt, E.K.; Zannini, E. Impact of Isolated and Chemically Modified Dietary Fiber on Bakery Products: Current Knowledge and Future Directions. Cereal Chem. 2023. [Google Scholar] [CrossRef]
- O’Keefe, S.J. The Association between Dietary Fibre Deficiency and High-Income Lifestyle-Associated Diseases: Burkitt’s Hypothesis Revisited. Lancet Gastroenterol. Hepatol. 2019, 4, 984–996. [Google Scholar] [CrossRef] [PubMed]
- Barber, T.M.; Kabisch, S.; Pfei, A.F.H.; Weickert, M.O. The Health Benefits of Dietary Fibre. Nutrients 2020, 12, 3209. [Google Scholar] [CrossRef]
- MacLeod, R.A.; Snell, E.E. Some Mineral Requirements of the Lactic Acid Bacteria. Inorg. Ions Bact. Growth 1947, 170, 351–365. [Google Scholar] [CrossRef]
- Kulshrestha, S.; Tyagi, P.; Sindhi, V.; Sharma, K. Invertase and Its Applications e A Brief Review. JOPR J. Pharm. Res. 2013, 7, 792–797. [Google Scholar] [CrossRef]
- Kappler, S.; Krahl, M.; Geissinger, C.; Becker, T.; Krottenthaler, M. Degradation of Iso-α-Acids During Wort Boiling. J. Inst. Brew. 2010, 116, 332–338. [Google Scholar] [CrossRef]
- Li, H.; Liu, F. Changes in Organic Acids during Beer Fermentation. J. Am. Soc. Brew. Chem. 2015, 73, 275–279. [Google Scholar] [CrossRef]
- Shibamoto, T. Diacetyl: Occurrence, Analysis, and Toxicity. J. Agric. Food Chem. 2014, 62, 4048–4053. [Google Scholar] [CrossRef]
- Laëtitia, G.; Pascal, D.; Yann, D. The Citrate Metabolism in Homo- and Heterofermentative LAB: A Selective Means of Becoming Dominant over Other Microorganisms in Complex Ecosystems *. Food Nutr. Sci. 2014, 5, 953–969. [Google Scholar] [CrossRef]
- Czerny, M.; Christlbauer, M.; Christlbauer, M.; Fischer, A.; Granvogl, M.; Hammer, M.; Hartl, C.; Hernandez, N.M.; Schieberle, P. Re-Investigation on Odour Thresholds of Key Food Aroma Compounds and Development of an Aroma Language Based on Odour Qualities of Defined Aqueous Odorant Solutions. Eur. Food Res. Technol. 2008, 228, 265–273. [Google Scholar] [CrossRef]
- Rhlid, R.B.; Fleury, Y.; Blank, I.; Fay, L.B.; Welti, D.H.; Vera, F.A.; Juillerat, M.A. Generation of Roasted Notes Based on 2-Acetyl-2-Thiazoline and Its Precursor, 2-(1-Hydroxyethyl)-4, 5-Dihydrothiazole, by Combined Bio and Thermal Approaches. J. Agric. Food Chem. 2002, 50, 2350–2355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, L.; Yu, X.; Xu, Y. Chemistry and Biology of Aroma and Taste The Biosynthesis Mechanism Involving 2,3-Pentanedione and Aminoacetone Describes the Production of 2-Ethyl-3,5-Dimethylpyrazine and 2-Ethyl-3,6-Dimethylpyrazine by Bacillus Subtilis. J. Agric. Food Chem. 2020, 68, 3558–3567. [Google Scholar] [CrossRef] [PubMed]
- Alim, A.; Song, H.; Liu, Y.; Zou, T.; Zhang, Y.; Zhang, S. Flavour-Active Compounds in Thermally Treated Yeast Extracts. J. Sci. Food Agric. 2018, 98, 3774–3783. [Google Scholar] [CrossRef]
- Hill, A.E.; Stewart, G.G. Free Amino Nitrogen in Brewing. Fermentation 2019, 5, 22. [Google Scholar] [CrossRef]
- Schieberle, P. Original Paper The Role of Free Amino Acids Present in Yeast as Precursors of the Odorants 2-Acetyl-l-Pyrroline and 2-Acetyltetrahydropyridine in Wheat Bread Crust. Zeitschrift Lebensmittel-Untersuchung Forschung 1990, 191, 206–209. [Google Scholar] [CrossRef]
- Jacob, F.F.; Striegel, L.; Rychlik, M.; Hutzler, M.; Methner, F.J. Spent Yeast from Brewing Processes: A Biodiverse Starting Material for Yeast Extract Production. Fermentation 2019, 5, 51. [Google Scholar] [CrossRef]
- Hernawan, T.; Fleet, G. Chemical and Cytological Changes during the Autolysis of Yeasts. J. Ind. Microbiol. 1995, 14, 440–450. [Google Scholar] [CrossRef]
- Agboola, J.O.; Lapeña, D.; Øverland, M.; Arntzen, M.Ø.; Mydland, L.T.; Hansen, J.Ø. Yeast as a Novel Protein Source - Effect of Species and Autolysis on Protein and Amino Acid Digestibility in Atlantic Salmon (Salmo Salar). Aquaculture 2022, 546, 737312. [Google Scholar] [CrossRef]
- Lamoolphak, W.; Goto, M.; Sasaki, M.; Suphantharika, M.; Muangnapoh, C.; Prommuag, C.; Shotipruk, A. Hydrothermal Decomposition of Yeast Cells for Production of Proteins and Amino Acids. J. Hazard. Mater. 2006, 137, 1643–1648. [Google Scholar] [CrossRef]
- Petrova, P. Lactic Acid Fermentation of Cereals and Pseudocereals: Ancient Nutritional Biotechnologies. Nutrients 2020, 12, 1118. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.J.; Schieber, A.; Gänzle, M.G. Formation of Taste-Active Amino Acids, Amino Acid Derivatives and Peptides in Food Fermentations—A Review. Food Res. Int. 2016, 89, 39–47. [Google Scholar] [CrossRef]
- Liu, M.; Bayjanov, J.R.; Renckens, B.; Nauta, A.; Siezen, R.J. The Proteolytic System of Lactic Acid Bacteria Revisited: A Genomic Comparison. BMC Genom. 2010, 11, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Savijoki, M.K.; Ingmer, H.; Varmanen, P. Proteolytic Systems of Lactic Acid Bacteria. Appl. Microbiol. Biotechnol. 2006, 71, 394–406. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Wang, J.; Yang, S.; Feng, J.; Jia, F.; Zhang, C. Protein Degradation in Wheat Sourdough Fermentation with Lactobacillus Plantarum M616. Interdiscip. Sci. Comput. Life Sci. 2015, 7, 205–210. [Google Scholar] [CrossRef]
- Kato, H.; Rhue, M.R.; Nishimura, T. Role of Free Amino Acids and Peptides in Food Taste; American Chemical Society: Washington, DC, USA, 1989. [Google Scholar]
- Kohl, S.; Behrens, M.; Dunkel, A.; Hofmann, T.; Meyerhof, W. Amino Acids and Peptides Activate at Least Five Members of the Human Bitter Taste Receptor Family. J. Agric. Food Chem. 2013, 61, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Temussi, P.A. The Good Taste of Peptides. J. Pept. Sci. 2012, 18, 73–82. [Google Scholar] [CrossRef]
- Konings, W.N. The Cell Membrane and the Struggle for Life of Lactic Acid Bacteria. Antonie van Leeuwenhoek Int. J. Gen. Mol. Microbiol. 2002, 82, 3–27. [Google Scholar] [CrossRef]
- Teixeira, J.S.; Seeras, A.; Sanchez-maldonado, A.F.; Zhang, C.; Su, M.S.; Gänzle, M.G. Glutamine, Glutamate, and Arginine-Based Acid Resistance in Lactobacillus Reuteri. Food Microbiol. 2014, 42, 172–180. [Google Scholar] [CrossRef]
- Dhakal, R.; Bajpai, V.K.; Baek, K.H. Production of GABA (γ-Aminobutyric Acid) by Microorganisms: A Review. Braz. J. Microbiol. 2012, 43, 1230–1241. [Google Scholar] [CrossRef]
- Hepsomali, P.; Groeger, J.A.; Nishihira, J.; Scholey, A. Effects of Oral Gamma-Aminobutyric Acid (GABA) Administration on Stress and Sleep in Humans: A Systematic Review. Front. Neurosci. 2020, 14, 923. [Google Scholar] [CrossRef]
- Siragusa, S.; De Angelis, M.; Di Cagno, R.; Rizzello, C.G.; Coda, R.; Gobbetti, M. Synthesis of γ-Aminobutyric Acid by Lactic Acid Bacteria Isolated from a Variety of Italian Cheeses. Appl. Environ. Microbiol. 2007, 73, 7283–7290. [Google Scholar] [CrossRef]
- Li, S.; Zhang, Y.; Yin, P.; Zhang, K.; Liu, Y.; Gao, Y.; Li, Y.; Wang, T.; Lu, S.; Li, B. Probiotic Potential of γ-Aminobutyric Acid (GABA)–Producing Yeast and Its Influence on the Quality of Cheese. J. Dairy Sci. 2021, 104, 6559–6576. [Google Scholar] [CrossRef]
- Weijzen, M.E.G.; van Gassel, R.J.J.; Kouw, I.W.K.; Trommelen, J.; Gorissen, S.H.M.; van Kranenburg, J.; Goessens, J.P.B.; van de Poll, M.C.G.; Verdijk, L.B.; van Loon, L.J.C. Ingestion of Free Amino Acids Compared with an Equivalent Amount of Intact Protein Results in More Rapid Amino Acid Absorption and Greater Postprandial Plasma Amino Acid Availability without Affecting Muscle Protein Synthesis Rates in Young Adults in a Double-Blind Randomized Trial. J. Nutr. 2021, 152, 59–67. [Google Scholar]
- Rønnestad, I.; Conceicao, L.E.C.; Aragao, C.; Dinis, M.T. Nutrient Metabolism—Research Communication Free Amino Acids Are Absorbed Faster and Assimilated More Efficiently than Protein in Postlarval Senegal Sole (Solea Senegalensis) 1. J. Nutr. 2000, 130, 2809–2812. [Google Scholar] [CrossRef]
- Whitley, K.; Marshall, V.M. Heterofermentative Metabolism of Glucose and Ribose and Utilisation of Citrate by the Smooth Biotype of Lactobacillus Amylovorus NCFB 2745. Antonie van Leeuwenhoek Int. J. Gen. Mol. Microbiol. 1999, 75, 217–223. [Google Scholar] [CrossRef]
- Raab, A.M.; Gebhardt, G.; Bolotina, N.; Weuster-botz, D.; Lang, C. Metabolic Engineering of Saccharomyces Cerevisiae for the Biotechnological Production of Succinic Acid. Metab. Eng. 2010, 12, 518–525. [Google Scholar] [CrossRef]
- Morishita, T.; Yajima, M. Incomplete Operation of Biosynthetic and Bioenergetic Functions of the Citric Acid Cycle in Multiple Auxotrophic Lactobacilli. Biosci. Biotechnol. Biochem. 1995, 59, 251–255. [Google Scholar] [CrossRef][Green Version]
- Tsuji, A.; Okada, S.; Hols, P.; Satoh, E. Enzyme and Microbial Technology Metabolic Engineering of Lactobacillus Plantarum for Succinic Acid Production through Activation of the Reductive Branch of the Tricarboxylic Acid Cycle. Enzyme Microb. Technol. 2013, 53, 97–103. [Google Scholar] [CrossRef]
- Bartowsky, E.J.; Costello, P.J.; Chambers, P.J. Emerging Trends in the Application of Malolactic Fermentation. Aust. J. Grape Wine Res. 2015, 21, 663–669. [Google Scholar] [CrossRef]
- Zakhartsev, M.; Reuss, M. Cell Size and Morphological Properties of Yeast Saccharomyces Cerevisiae in Relation to Growth Temperature. FEMS Yeast Res. 2018, 18, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Rodríguez, A.J.; Polo, M.C.; Carrascosa, A.V. Structural and Ultrastructural Changes in Yeast Cells during Autolysis in a Model Wine System and in Sparkling Wines. Int. J. Food Microbiol. 2001, 71, 45–51. [Google Scholar] [CrossRef] [PubMed]


| g/100 g DM | ||
|---|---|---|
| CBSY | PBSY | |
| Proteins | 35.588 ± 1.501 | 38.736 ± 1.638 |
| Total Fats | 1.361 ± 0.089 | 1.393 ± 0.001 |
| Carbohydrates | ||
| Insoluble Dietary Fibre | 44.367 ± 1.972 | 36.723 ± 1.767 |
| High MW Soluble | 3.130 ± 0.482 | 2.095 ± 0.386 |
| Low MW Soluble | 2.412 ± 0.064 | 0.925 ± 0.140 |
| Total Dietary Fibre | 49.909 ± 2.122 | 39.731 ± 1.603 |
| Total Sugars | 0.301 ± 0.019 | 10.048 ± 0.644 |
| Ash | 5.628 ± 0.322 | 3.429 ± 0.211 |
| Moisture | 6.710 ± 0.380 | 14.530 ± 0.380 |
| Energy value (kcal) | 314 ± 10 | 294 ± 10 |
| Energy value (kJ) | 1322 ± 45 | 1237 ± 47 |
| mg/kg | ||
| Calcium | 2320 ± 190 | 2700 ± 220 |
| Iron | 65 ± 13 | 68 ± 14 |
| Phosphorus | 9860 ± 790 | 6600 ± 530 |
| Magnesium | 2060 ± 230 | 1310 ± 140 |
| Manganese | 5.7 ± 1.3 | 5.5 ± 1.2 |
| Potassium | 14,900 ± 1500 | 4950 ± 480 |
| Copper | 3.16 ± 0.67 | 3.36 ± 0.71 |
| Sodium | 271 ± 26 | 166 ± 21 |
| Zinc | 72 ± 14 | 78 ± 15 |
| Total | 29,556 | 15,880 |
| g/100 g DM | ||
| Glucose | 0.166 ± 0.014 | n.d. |
| Fructose | 0.090 ± 0.009 | 9.924 ± 0.644 |
| Lactose | n.d. | n.d. |
| Sucrose | n.d. | n.d. |
| Maltose | 0.045 ± 0.010 | 0.124 ± 0.020 |
| Total Sugars | 0.301 ± 0.019 | 10.048 ± 0.644 |
| IBU (mg/L) | ||
| Iso-α-acids (IBU) | 129 | 100 |
| Compound | Aroma | Flavour | Intensity | |
|---|---|---|---|---|
| CBSY | PBSY | |||
| Ethyl-3-methyl butanoate | fruity, sweet, apple | sweet, fruity, spicy | 1.75 | 1.5 |
| 2,3,-butanedione (diacetyl) | buttery, creamy, sweet | buttery, creamy, sweet | 0.5 | 1.75 |
| Acetic acid | sour, acidic | sour, acidic | 1.5 | 3 |
| 2-ethyl-3,5/6-dimethyl pyrazine | earthy/burnt | earthy/burnt | 2 | n.d. |
| Methional | boiled potato, vegetable | boiled potato, vegetable | 2.75 | 2.25 |
| Butanoic acid (butyric acid) | cheesy, sharp, acetic | sour, acidic, cheesy | 2 | 1.5 |
| Phenylacetaldehyde | green, sweet, floral | honey, floral, sweet, cocoa | 1 | 2 |
| 2-/3-methyl butanoic acid | cheesy, sour, tropical | cheesy, fruity, fermented | 3 | 2.5 |
| 2-acetyl-2-thiazoline | corn, potato, toasted | corn, popcorn, roasted, grain | 2 | n.d. |
| Hexanoic acid | fatty, sour, cheesy | cheesy, fruity, phenolic, goat | 2 | 1.25 |
| Furaneol | caramel, sweet, strawberry | caramel, sweet, burnt sugar, maple | 3 | 3 |
| Octanoic Acid | fatty, waxy, cheesy, | soapy, rancid, cheesy, fatty | 2 | 2 |
| 4-methylphenol | phenolic, narcissus, animal, mimosa | phenolic | 1.5 | 1.5 |
| Lactic acid | lightly acidic | sour, acidic | n.d. | 2 |
| Sotolon | caramel-like, sweet, maple | caramel-like, sweet, maple | 2 | 2.5 |
| Decanoic acid | fatty, rancid, sour, citrus | soapy, waxy, fruity | 2.5 | 2.25 |
| Phenylacetic acid | honey, sweet, floral | honey, sweet, floral | 2.5 | 2.25 |
| Vanillin | vanilla, sweet, creamy | vanilla, sweet, creamy | 3 | 2.25 |
| Phenylpropionic acid | floral, sweet, fatty | sweet, balsamic | 2.25 | 2.5 |
| Essential Amino Acids | CBSY | % of Requirement mg/g Protein | PBSY | % of Requirement mg/g Protein |
|---|---|---|---|---|
| g/100 g | g/100 g | |||
| Isoleucine | 1.17 ± 0.19 | 117 | 1.49 ± 0.23 | 150 |
| Leucine | 2.02 ± 0.27 | 100 | 2.15 ± 0.27 | 106 |
| Valine | 1.38 ± 0.22 | 104 | 1.87 ± 0.25 | 141 |
| Lysine | 2.51 ± 0.30 | 158 | 2.67 ± 0.32 | 168 |
| Tryptophan | 0.32 ± 0.04 | 146 | 0.27 ± 0.03 | 123 |
| Threonine | 1.47 ± 0.23 | 177 | 1.70 ± 0.24 | 205 |
| Histidine | 0.67 ± 0.11 | 126 | 0.65 ± 0.11 | 123 |
| Sulphur AA | 105 | 98 | ||
| Cysteine and Cystine | 0.37 ± 0.06 | 0.25 ± 0.04 | ||
| Methionine | 0.43 ± 0.07 | 0.43 ± 0.08 | ||
| Aromatic AA | 165 | 177 | ||
| Tyrosine | 0.98 ± 0.16 | 1.07 ± 0.17 | ||
| Phenylalanine | 1.26 ± 0.21 | 1.33 ± 0.22 | ||
| Nonessential Amino Acids | CBSY | PBSY | ||
| g/100 g | g/100 g | |||
| Aspartic acid | 3.06 ± 0.35 | 3.02 ± 0.35 | ||
| Glutamic acid | 4.89 ± 0.52 | 3.66 ± 0.40 | ||
| Alanine | 2.08 ± 0.27 | 2.03 ± 0.27 | ||
| Arginine | 2.01 ± 0.26 | 1.67 ± 0.24 | ||
| Glycine | 1.40 ± 0.22 | 1.33 ± 0.22 | ||
| Proline | 1.71 ± 0.24 | 1.59 ± 0.23 | ||
| Serine | 1.85 ± 0.25 | 1.82 ± 0.25 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaeger, A.; Nyhan, L.; Sahin, A.W.; Zannini, E.; Arendt, E.K. Lactic Acid Fermentation as a Valorising Agent for Brewer’s Spent Yeast—Improving the Sensory Quality and Nutritional Potential. Fermentation 2024, 10, 54. https://doi.org/10.3390/fermentation10010054
Jaeger A, Nyhan L, Sahin AW, Zannini E, Arendt EK. Lactic Acid Fermentation as a Valorising Agent for Brewer’s Spent Yeast—Improving the Sensory Quality and Nutritional Potential. Fermentation. 2024; 10(1):54. https://doi.org/10.3390/fermentation10010054
Chicago/Turabian StyleJaeger, Alice, Laura Nyhan, Aylin W. Sahin, Emanuele Zannini, and Elke K. Arendt. 2024. "Lactic Acid Fermentation as a Valorising Agent for Brewer’s Spent Yeast—Improving the Sensory Quality and Nutritional Potential" Fermentation 10, no. 1: 54. https://doi.org/10.3390/fermentation10010054
APA StyleJaeger, A., Nyhan, L., Sahin, A. W., Zannini, E., & Arendt, E. K. (2024). Lactic Acid Fermentation as a Valorising Agent for Brewer’s Spent Yeast—Improving the Sensory Quality and Nutritional Potential. Fermentation, 10(1), 54. https://doi.org/10.3390/fermentation10010054







