Abstract
Wood pellets can sustain the growth of Trichoderma spp. in soil; however, little is known about their side effects on the microbiota. The aims of this study were to evaluate the effect of wood pellets on the growth of Trichoderma spp. in bulk soil and on the soil microbial population’s composition and diversity. Trichoderma atroviride SC1 coated wood pellets and non-coated pellets were applied at the level of 10 g∙kg−1 of soil and at the final concentration of 5 × 103 conidia∙g−1 of soil and compared to a conidial suspension applied at the same concentration without the wood carrier. Untreated bulk soil served as a control. The non-coated wood pellets increased the total Trichoderma spp. population throughout the experiment (estimated as colony-forming unit g−1 of soil), while wood pellets coated with T. atroviride SC1 did not. The wood carrier increased the richness, and temporarily decreased the diversity, of the bacterial population, with Massilia being the most abundant bacterial genus, while it decreased both the richness and diversity of the fungal community. Wood pellets selectively increased fungal species having biocontrol potential, such as Mortierella, Cladorrhinum, and Stachybotrys, which confirms the suitability of such carriers of Trichoderma spp. for soil application.
Keywords:
Trichoderma; substrate; soil; metabarcoding; diversity; community composition; wood pellets 1. Introduction
Biological control of soil-borne diseases is a valuable alternative to synthetic chemical fungicides [1], and, within the genus Trichoderma [2], several strains have demonstrated good efficacy against soil-borne pathogens such as Rhizoctonia solani, Fusarium spp., Pythium spp., and nematodes of the genus Meloidogyne [3]. However, the success of treatments with Trichoderma spp. depends highly on the physicochemical and biological traits of the soil, as well as the rhizosphere competence of the strains used. After the soil treatment, the population of Trichoderma spp. normally tends to decrease over time [4]. This problem is usually addressed by applying high quantities of the biocontrol agent and/or by formulating the biocontrol agent (i.e., as a wettable powder, emulsion, pellets, granules, etc.) or adding nutrients to the formulation that can extend its longevity in the soil [5,6,7,8]. Particular formulations of biocontrol agents comprise aids that can preserve them, favor their delivery to targets, and improve their activity [9]. Another limiting factor that prevents the widespread use of Trichoderma spp. in soil treatments is the difficulty in homogenously applying small quantities of conidia in large volumes of soil [10]. Although several authors have addressed the effectiveness of formulations and the addition of nutrients [11,12,13,14,15,16,17,18,19,20], limited information is available on the effect of such components on the soil microbiome [21]. More particularly, the effect of the use of lignocellulosic substrates inoculated with Trichoderma spp. on soil fungal and bacterial populations is unknown.
The use of wood pellets coated with conidia of Trichoderma spp. might represent an interesting approach for soil treatments [10]. For example, Trichoderma atroviride SC1 can easily grow on beech wood pellets and reach high population levels (e.g., 109 cfu∙g−1 of wood pellet), especially if complemented with nitrogen sources, such as soy protein isolates. The advantage of using wood pellets is double: they can be easily spread and incorporated in soil by using standard equipment (e.g., using a fertilizer spreader, followed by harrowing) and support the growth of the fungus, which colonize wood before other microbes and then outcompete them. For example, early or simultaneous inoculation of T. viride or T. harzianum with basidiomycetes that can attack coconut fibers, such as Trametes versicolor and Stereum rugosum, can protect coconuts from white rot decay, mainly by nutrient competition, but also by toxins that can inhibit the growth of the pathogens [22]. On the other hand, carriers can also modify the soil microbial communities’ composition. Since soil microbial communities often act synergistically to control soil-borne pathogens, a change in the soil microbial community structure, and/or a reduction in biodiversity, may affect the occurrence of soil-borne diseases [23].
Many factors can contribute to shifting the microbial populations in the soil, such as the soil type and pH, structure, salinity, and moisture, but most importantly, soil organic matter and plant exudates [24,25]. Generally, adding organic matter to the soil enhances the microbial activity [26,27,28], while the use of mineral fertilizers can reduce fungal diversity [29,30]. The inoculation of Trichoderma spp. without organic matter has a transient effect on the microbial population of the soil [31,32], and combining the application of Trichoderma spp. with organic composts and bio-organic fertilizers has been proposed as an alternative to mineral fertilizers; in fact, adding these species to the substrate can increase soil fertility and microbial biodiversity [33,34].
The aim of this study was to test the effectiveness of a carrier of T. atroviride SC1 made from wood pellets in prolonging the survival of the fungus in the soil and to evaluate its possible impact on the soil microbiota by metabarcoding analysis of the microbial communities. Although this case study is based on the use of a specific carrier (beech wood pellet) and a specific strain (T. atroviride SC1) in a single soil, the protocol could be replicated for other similar combinations, for future comparison.
2. Materials and Methods
2.1. Coating the Wood Pellets with Trichoderma atroviride SC1, Soil Treatments and Experimental Design
Beech wood pellets (Italwood S.r.l., Piovene Rocchette, Italy) were used as carriers to deliver T. atroviride SC1 to the soil. Wood pellets were coated with conidia, according to Chammem et al. [10]. Briefly, beech pellets (100 g) were dried in an oven at 120 °C for 12 h, sprayed with soy protein isolates (30 mg∙mL−1 corresponding to a final rate of 3 mg∙g−1 of pellets) and coated with 0.1 mL of T. atroviride SC1 conidia sterile water suspension (SDW) with a spray bottle, while continuously mixing in a mixer (MUM44R1- BSH Elettrodomestici S.p.A., Milan, Italy) at a speed of 25 rpm. Conidia were prepared according to Longa et al. [35] and adjusted with a hemocytometer at 5 × 106 conidia∙mL−1 to reach a final concentration of 5 × 105 conidia∙g−1 of pellets. The wood pellets were used immediately after coating.
The experiment was carried out under controlled greenhouse conditions, at a temperature of 25 ± 1°C and relative humidity 70 ± 10%, in 2020. The coated wood pellets were applied to a bulk of sandy loam soil (69.7% sand: 26.3% lime: 4% clay, pH 8) collected in San Michele all’Adige, Italy (N 46.182315, E 11.118804), representing a typical agricultural soil of this region (apple orchards). The soil was mixed thoroughly, sieved, and then distributed into 20 plastic pots (Mongardi, Ferriera di Buttigliera Alta, Italy; 2L) at 1 kg of soil∙pot−1. A randomized block design was used, with four treatments and five replicates (pots) each: bulk soil (untreated control; Ctr), soil mixed with non-coated wood pellet (Trt1), soil mixed with a conidial SDW suspension T. atroviride SC1 (Trt2), and soil mixed with of T. atroviride SC1 coated pellets prepared as described above (Trt3). The final estimated concentration of T. atroviride SC1 conidia in Trt2 and Trt3 was 5 × 103 conidia∙g−1 of soil. The wood pellets in Trt1 and Trt3 were applied by laying them on the soil surface (10 g∙pot−1), spraying 50 mL of SDW pot−1, letting them swell and disintegrate (20 min), and gently mixing the broken-down pieces in the soil. The Ctr and Tr2 were sprayed with 50 mL of SDW per pot−1. After calculation of the water holding capacity of the soil using the percolation method, the soil was kept at 60 ± 10% humidity, by weighing the pots every two days and adding the quantity of water that was lost by evaporation. The experiment (E1) was repeated after one week (E2).
To ensure the absence of T. atroviride SC1 in the soil, real-time PCR primers and probes, designed for the detection and quantification of T. atroviride SC1 [36], were used to check the bulk soil before the experiments.
2.2. Soil Sampling
The growth of T. atroviride SC1 was monitored by sampling the soil immediately after completing the treatments (12 h, D0), every 15 days in the first two months (D15, D30, D45, D60), and the final sampling was carried out after ninety days (D90). Samples of soil (5 g) were collected from each pot (replicate) by taking 1 g from the center of the pot and 1 g from each of its four corners. The samples were put in 50 mL sterile Falcon tubes (Merk Life Science S.r.l., Milan, Italy) and thoroughly mixed. Colony forming unit (cfu) counting was performed by suspending 1 g from each falcon tube in 10 mL of SDW, vortexing for 1 min, and plating 100–200 µL from the SDW suspension on a Trichoderma semi-selective medium that contained potato dextrose agar (Oxoid, Basingstoke, UK, 39 g∙L−1), rose bengal (Sigma Aldrich, Anekal Taluk, India, 0.1 g∙L−1), chloramphenicol (Sigma Aldrich, Beijing, China, 0.1 g∙L−1), and streptomycin sulfate (Fluka Biochemika, Milan, Italy, 0.05 g∙L−1). The results are reported as cfu∙g−1 of soil ± the standard deviation.
For metabarcoding analysis, only four replicates from each treatment were considered and were chosen randomly. Samples (1 g) were collected at D0, D15, and D90, lyophilized in a freeze-dryer (HetoLyoLab 3000-Analitica De Mori, Milan, Italy) for 12 h, and stored at −80 °C until use.
2.3. DNA Extraction, Amplification, and Sequencing
Total genomic DNA was extracted from 500 mg of lyophilized soil samples (96 soil samples) using a FastDNA™ Spin kit (MP Biomedicals, Irvine, CA, USA), following the manufacturer’s instructions and was quantified using a NanoDrop™ 8000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA).
The library construction and sequencing were performed on the sequencing platform of the Edmund Mach Foundation. Total genomic DNA was amplified using primers specific to either the bacterial and archaeal 16S rRNA gene or the fungal ITS1 region. The specific bacterial primer set 515F (5′-GTGYCAGCMGCCGCGGTAA-3′) and the 806R (5′-GGACTACNVGGGTWTCTAAT-3′) was used [37], with degenerate bases suggested by Apprill et al. [38] and by Parada et al. [39]. Although no approach based on PCR amplification is free from bias, this primer pair has been shown to guarantee good coverage of known bacterial and archaeal taxa [40]. For the identification of fungi, the internal transcribed spacer 1 (ITS1) was amplified using the primers ITS1F (5′-CTTGGTCATTTAGAGGAAGTAA-3′) [41] and ITS2 (5′-GCTGCGTTCTTCATCGATGC-3′) [42]. All the primers included specific overhang Illumina adapters for the amplicon library construction.
For the 16S V4 region, each sample was amplified by PCR using a 25-μL reaction with one μM of each primer. In more detail, 12.5 μL of 2× KAPA HiFi HotStart ReadyMix and 10 μL forward and reverse primers were used in combination with 2.5 μL of template DNA (5–20 ng∙μL−1). PCR reactions were executed using a GeneAmp PCR System 9700 (Thermo Fisher Scientific) and the following cycling conditions: initial denaturation step at 95 °C for 5 min (1 cycle); 28 cycles at 95 °C for 30 s, 55 °C for 30 s, and 72 °C for 30 s; a final extension step at 72 °C for 5 min (1 cycle).
For the ITS1 region, each sample was amplified by PCR using 25-μL reaction with 10 μM of each primer. In more detail, 22 μL of premix FastStart High Fidelity PCR System (Roche) and 2 μL forward and reverse primers were used in combination with 1 μL of template DNA (5–20 ng∙ul−1). PCR reactions were executed using a GeneAmp PCR System 9700 (Thermo Fisher Scientific) and the following cycling conditions: initial denaturation step at 95 °C for 3 min (1 cycle); 30 cycles at 95 °C for 20 s, 50 °C for 45 s, and 72 °C for 90 s; final extension step at 72 °C for 10 min (1 cycle).
The amplification products were checked on 1.5% agarose gel and purified using a CleanNGS kit (CleanNA, Waddinxveen, The Netherlands), following the manufacturer’s instructions. Afterward, a second PCR was used to apply dual indices and Illumina sequencing adapters Nextera XT Index Primer (Illumina, Berlin, Germany), using seven cycles of PCR (16S Metagenomic Sequencing Library Preparation, Illumina, Berlin, Germany). The amplicon libraries were purified using a CleanNGS kit (CleanNA, Waddinxveen, The Netherlands), and quality control was performed on a Typestation 2200 platform (Agilent Technologies, Santa Clara, CA, USA). Finally, all barcoded libraries were pooled in an equimolar manner and sequenced on an Illumina® MiSeq (PE300) platform (MiSeq Control Software 2.5.0.5 and Real-Time Analysis software 1.18.54.0).
2.4. Bioinformatics and Statistical Analysis
Illumina reads were filtered with Bowtie2 v2.3.4.3 [43] to avoid the presence of Illumina phiX contamination, and quality was preliminarily checked with FastQC v0.11.8 [44]. Primers were stripped using Cutadapt v1.18 [45]. Sequences were quality filtered, trimmed, denoised, and amplicon sequence variants (ASVs) were generated with DADA2 v1.14 [46]. Denoised forward and reverse ASV sequences were merged and chimeras were removed. Filtered ASVs were checked using Metaxa2 v2.2.1 [47] and ITSx v1.1.2 [48] for targeting the presence of the V4 16S rRNA and ITS1 regions, in archaeal and bacterial sequences and fungal sequences, respectively. Taxonomic assignment of 16S rRNA gene ASVs and ITS based ASVs was performed using a RDP classifier, reimplemented in DADA2 against the SILVA v138 database [49] and UNITE 8.2 database [50], respectively. BIOM objects with bacterial and fungal counts, respectively, were built and imported into the R-4.0.3 statistical environment for further analyses [51].
The data of the growth assessment of T. atroviride SC1 cfu counts (Figure 1) were log10 transformed to simplify the data analysis, as is commonly the case for colony counts to avoid data skewness. Bartlett’s test of homogeneity of variances and Shapiro–Wilk’s normality test were used to check the normal distribution of the data. ANOVA and Tukey’s HSD tests were performed on log10 transformed data with a normal distribution (test of the evolution of treatments in time), and the non-parametric Kruskal–Wallis and Dunn post-hoc (Benjamini–Hochberg p-adjustment method α = 0.05) tests were used otherwise (comparing the cfu counts between treatments at each sampling point).
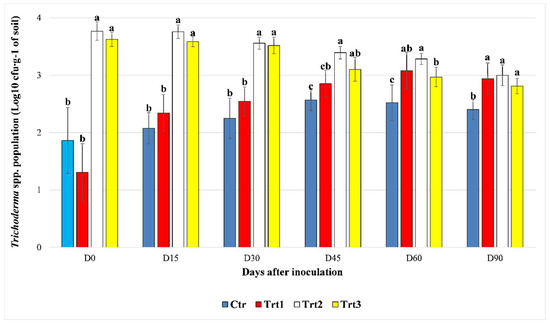
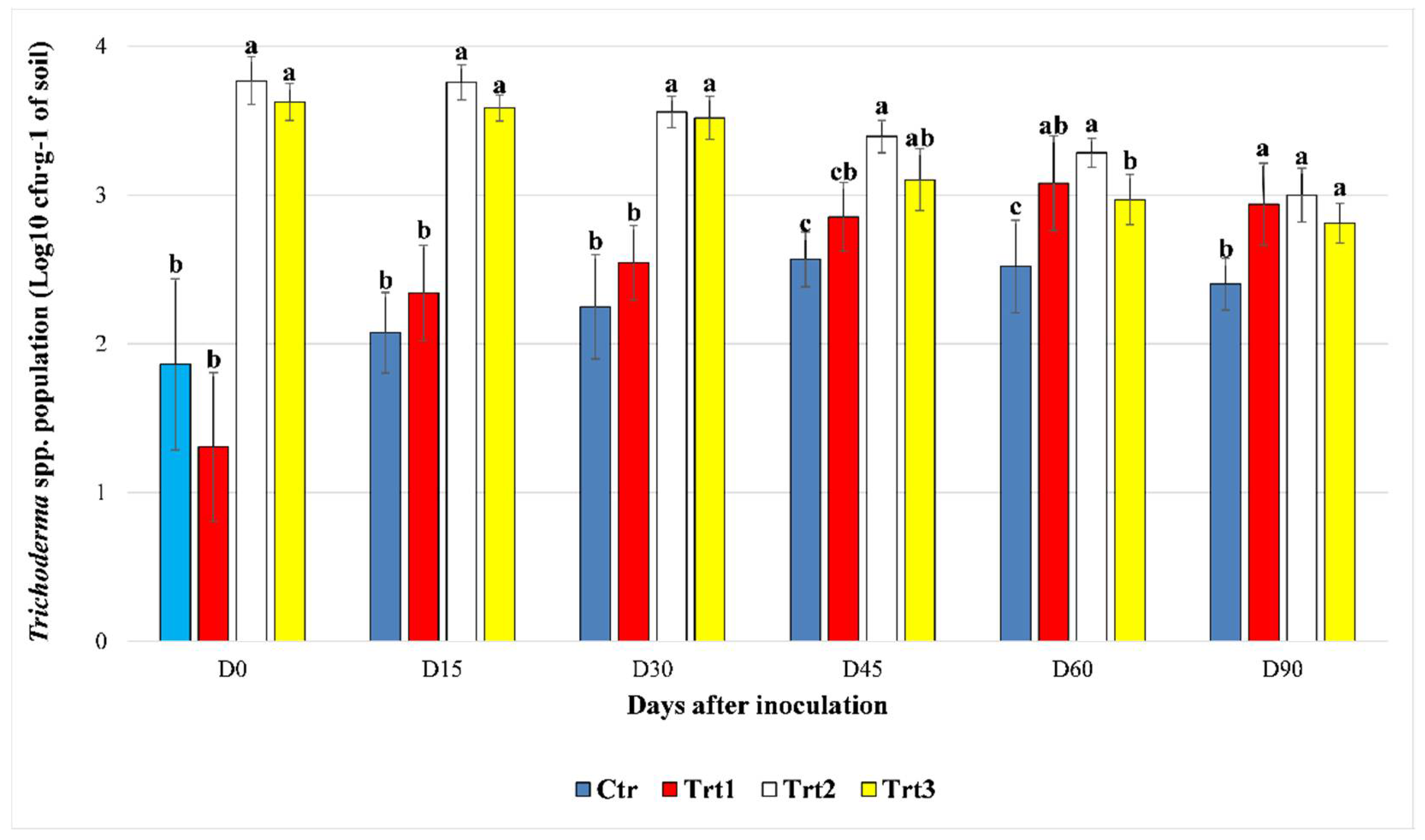
Figure 1.
The effect of beech wood pellets coated with Trichoderma atroviride SC1 and uncoated on the Trichoderma spp. population in the soil at different sampling times post inoculation (D0: after 12h, D15: after 15 days, D30: after 30 days, D45: after 45 days, D60: after 60 days, and D90: after 90 days). Ctr: bulk soil; Trt1: soil with 10 g of beech wood pellets; Trt2: soil with T. atroviride SC1 conidial suspension (5 × 103 conidia∙g−1 of soil); Trt3: soil with 10 g of T. atroviride SC1 coated beech pellets (5 × 105 conidia∙g−1 of beech wood pellets). At each sampling point, different letters indicate significant statistical differences between treatments, according to Dunn’s test (α = 0.05). Colony counts of the two experiment were pooled.
Bacterial and fungal count tables were filtered using the RAM R package, and rare ASVs (relative abundance < 0.1%) were discarded. Relative abundance of taxa at different taxonomic ranks was calculated with the RAM R package [52].
Alpha-diversity values were calculated adopting a multiple rarefaction method, implemented in the rtk R package [53]. In detail, richness (observed ASVs) and diversity values (Simpson’s index) were generated by averaging the results inferred after 999 rarefactions, starting from a minimum number of 38,256 and 13,418 reads, for 16S rRNA gene and ITS data, respectively. A regression analysis based on linear models was carried out on the richness and diversity values, for each dataset, after inspection with the fitdistrplus R package [54]. In more detail, a machine learning approach based on 9999 bootstrap resampling was adopted to evaluate models in which factors (i.e., experiment, time, and treatment) were considered only for their main effects or also with an interaction. The performance of the models was assessed by means of RMSE (root mean squared error) and R-squared, which measure the prediction error and the proportion of variation explained by each model, respectively [55]. An analysis of variance (ANOVA) followed, to evaluate the linear model fit. A post-hoc analysis was carried out with pairwise comparisons, based on the estimated marginal means (EMMs) as implemented in the emmeans R package [56]. Richness and diversity values were graphically represented as boxplots, using the ggplot2 R package (Figure 2 and Figure 3) [57]. A confirmatory analysis based on recursive partitioning [58] was carried out by considering richness and diversity variables together in the same model; with experiment, time, and treatment as factors (Figure S1).
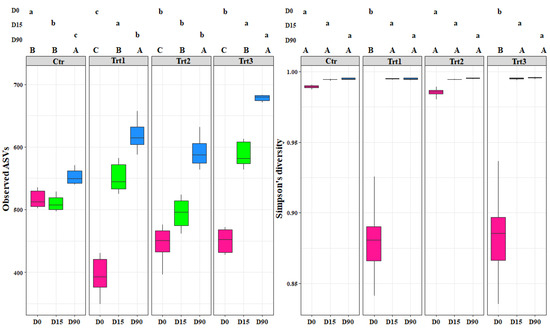
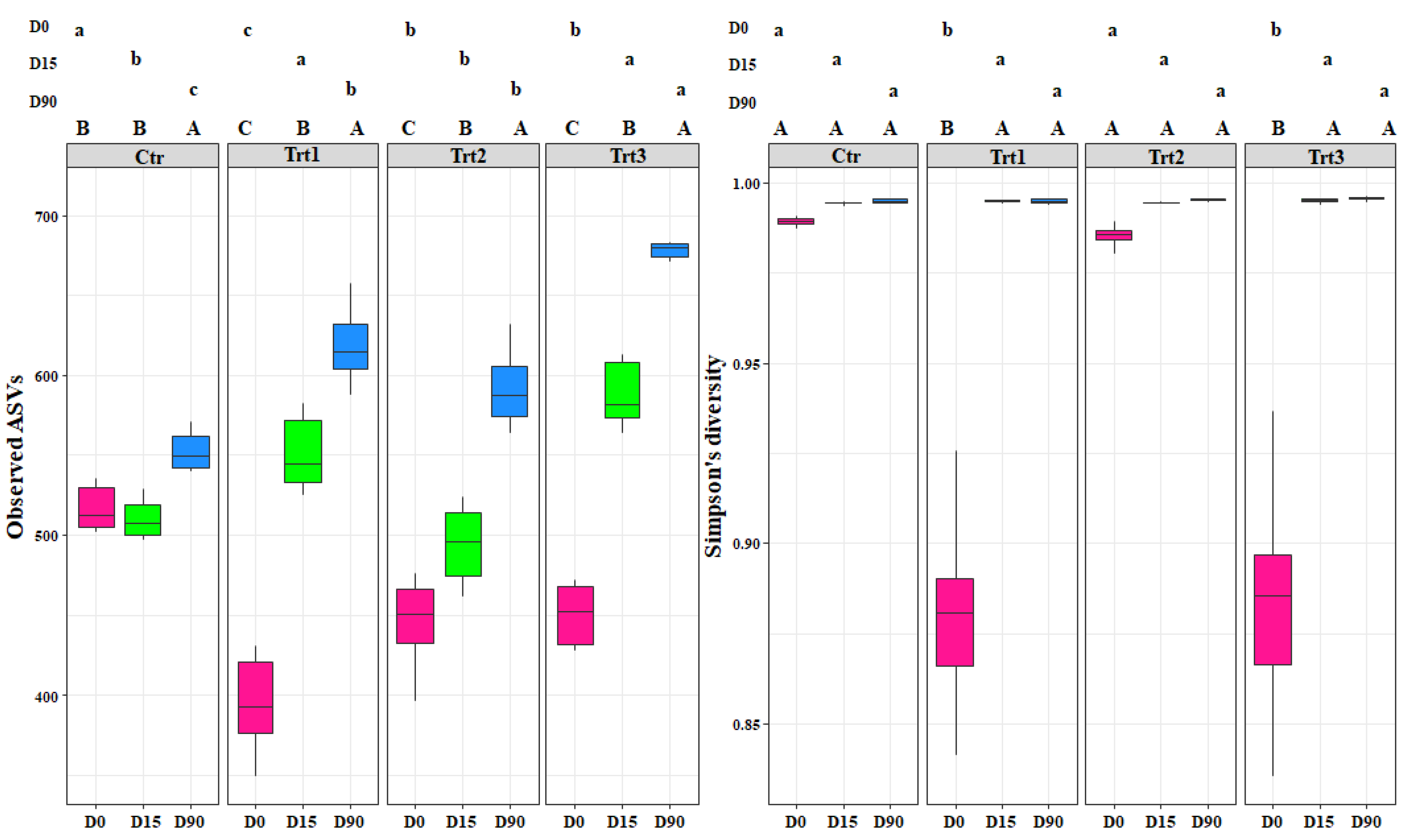
Figure 2.
Boxplots of the bacterial richness (observed ASVs) and bacterial diversity (Simpson’s diversity) at different sampling times (D0: after 12 h; D15: after 15 days; D90: after 90 days). Ctr: bulk soil; Trt1: soil with 10 g of beech wood pellets; Trt2: soil with Trichoderma atroviride SC1 conidial suspension at the rate of 5 × 103 conidia∙g−1 of soil; Trt3: soil with 10 g of T. atroviride SC1 coated beech pellets (5 × 105 conidia∙g−1 of beech wood pellets). Letters indicate significant differences according to the emmeans package (α = 0.05) between treatments at each sampling point (lower case letters) or for the same treatment over time (upper case letters). Data of the two experiments E1 and E2 were pooled.
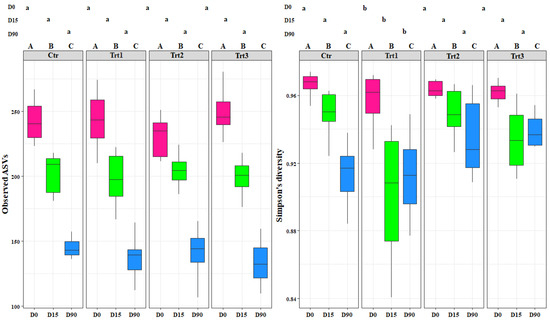
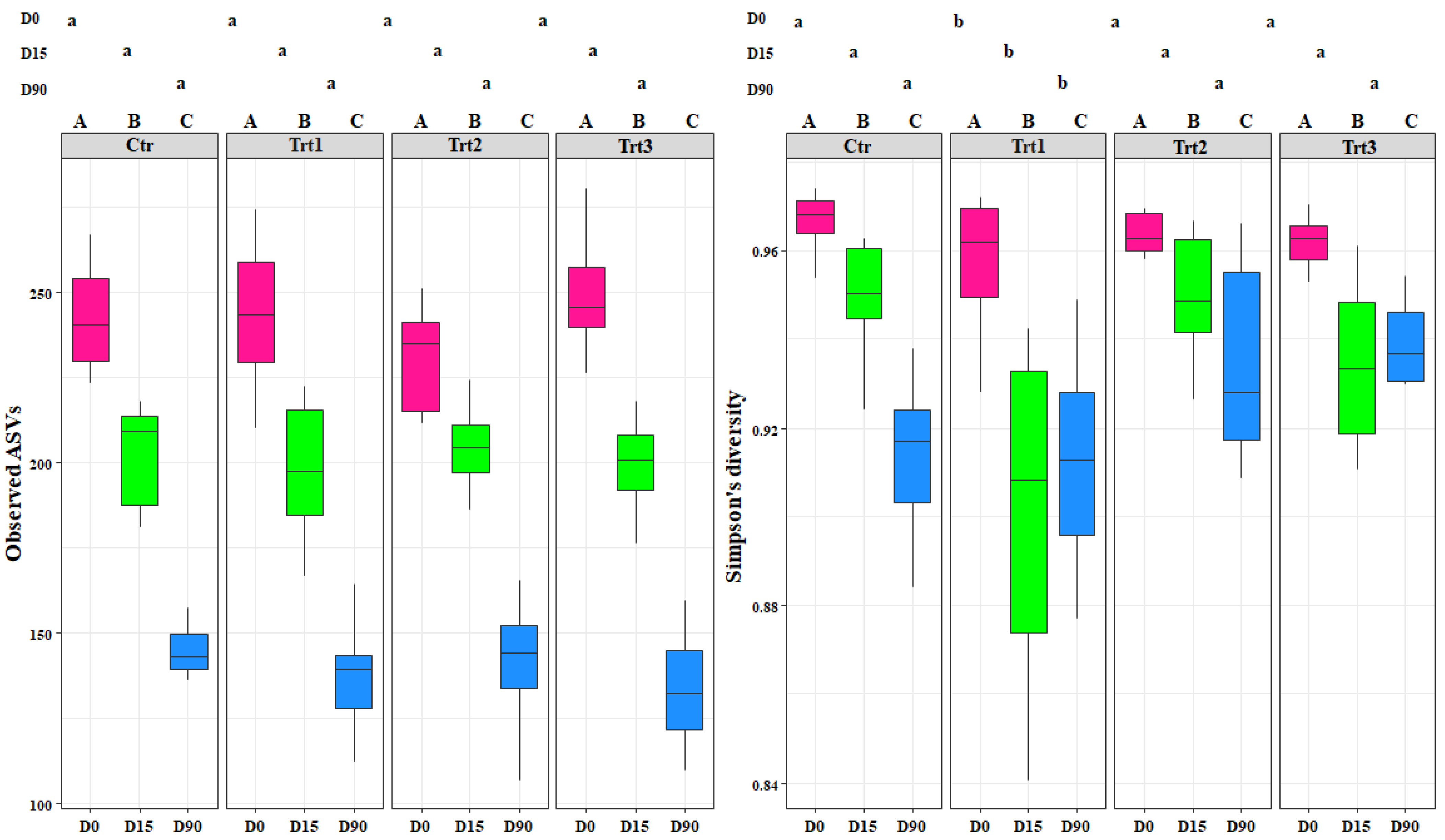
Figure 3.
Boxplots of the fungal richness (observed ASVs) and fungal diversity (Simpson’s diversity) at different sampling times (D0: after 12 h; D15: after 15 days; D90: after 90 days). Ctr: bulk soil; Trt1: soil with 10 g of beech wood pellets; Trt2: soil with Trichoderma atroviride SC1 conidial suspension at the rate of 5 × 103 conidia∙g−1 of soil; Trt3: soil with 10 g of T. atroviride SC1 coated beech pellets (5 × 105 conidia∙g−1 of beech wood pellets). Letters indicate significant differences according to the emmeans package (α = 0.05) between treatments at each sampling point (lower case letters) or for the same treatment over time (upper case letters). Data of the two experiments E1 and E2 were pooled.
Beta diversity calculations were conducted after normalization with the median of ratios method implemented in the DESeq2 R Bioconductor package [59]. Exploratory non-metric multidimensional scaling (NMDS) ordinations were applied to Bray–Curtis dissimilarities. NMDS ordinations were plotted using the ggvegan and ggplot2 R packages (Figure 4) [60]. A multivariate analysis based on PERMANOVA was performed on Bray–Curtis dissimilarities applied to normalized bacterial and fungal count tables, respectively (adonis function, vegan R package) [61]. To confirm the PERMANOVA results, a multivariate generalized model (mGLM) was calculated, including all available factors and based on a negative binomial distribution (confirmed by graphical inspection). The model was assessed by analysis of deviance with a likelihood-ratio-test (manyglm function, mvabund R package) [62]. ASVs that had abundances significantly different (p < 0.05) in at least one factor were extracted from the mGLM results and were used to calculate univariate non-parametric tests for each factor (multtest.gp function, RVAideMemoire R package) [63]. The results of each rank test were corrected by false discovery rate (FDR), and post-hoc pairwise comparisons were performed between the levels in each factor, with a Dunn test followed by Benjamini–Hochberg adjustment (dunntest function, FSA R package) [64]. Bacterial and fungal indicator ASVs, respectively, were collapsed at genus level and relative abundances were plotted with the RAM R package (Figure 5).
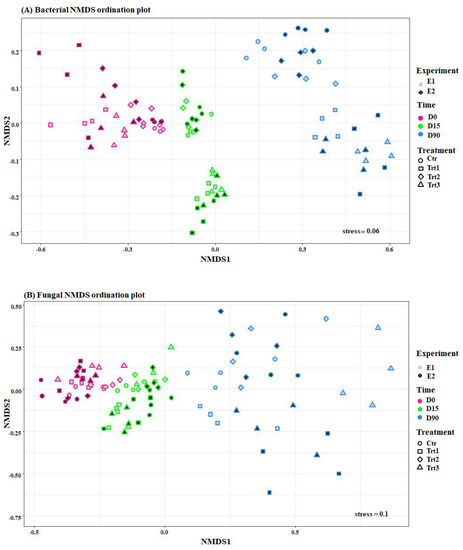
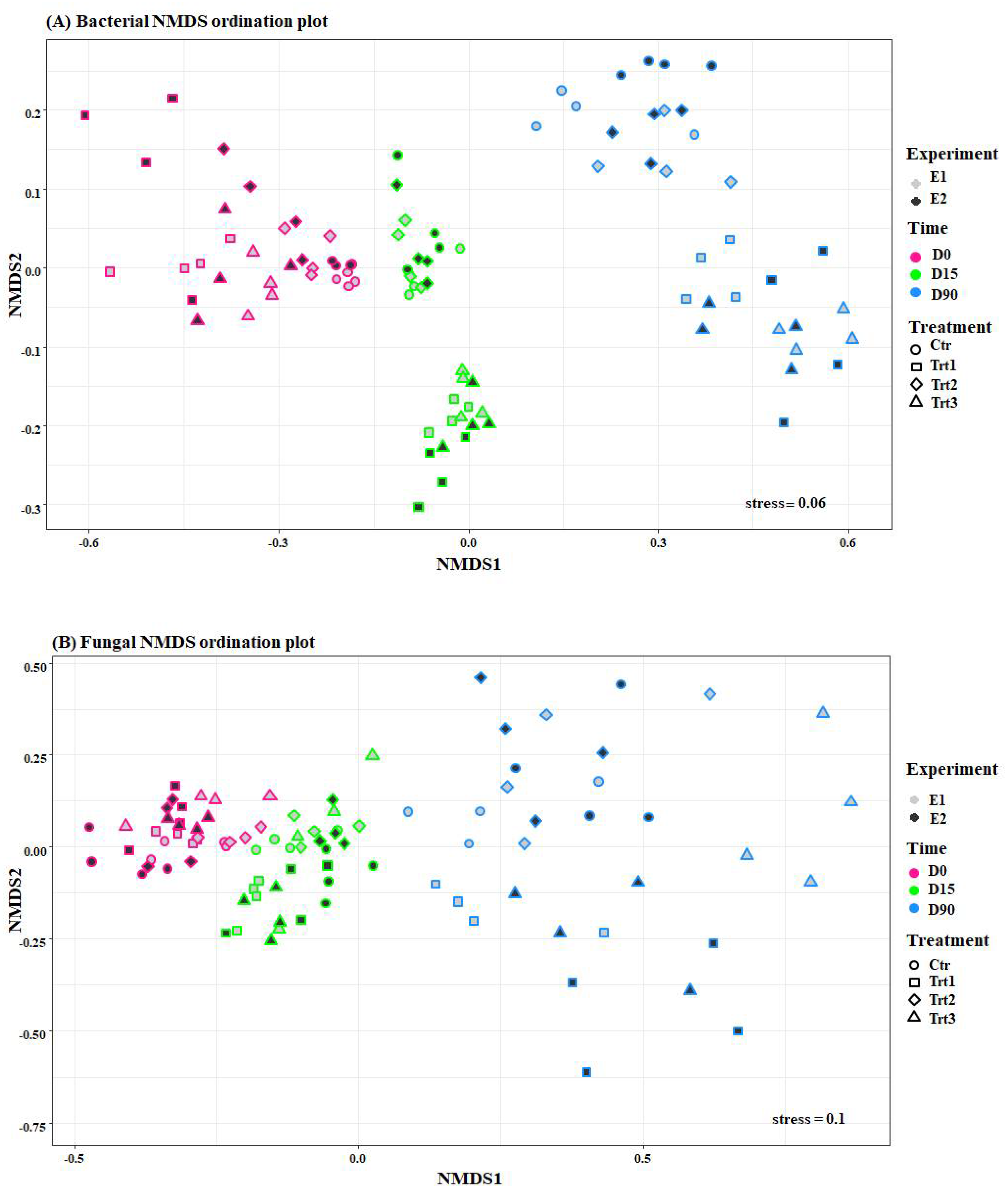
Figure 4.
Ordination plots of non-metric multidimensional scale analysis (NMDS) using the Bray-Curtis dissimilarities of bacterial (A) and fungal (B) communities. Pink, green, and blue colors indicate different sampling times (D0: after 12 h; D15: after 15 days; D90: after 90 days) and show how both fungal and bacterial communities are grouped by time. The colors of the filling, grey and black, represent the two experiments (E1 and E2, respectively), and different shapes represent the treatments that grouped, mainly the bacterial community. Ctr: bulk soil; Trt1: soil with 10 g of beech wood pellets; Trt2: Trichoderma atroviride SC1 applied to the soil as a conidial suspension at the rate of 5 × 103 conidia g−1 of soil; Trt3: soil with 10 g of T. atroviride SC1 coated beech wood pellets at 5 × 105 conidia g−1.
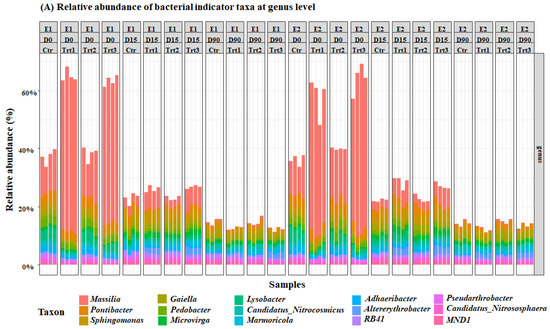
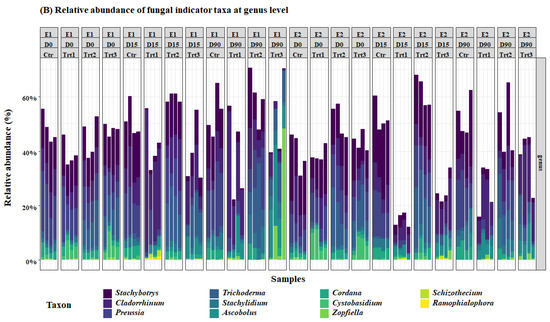
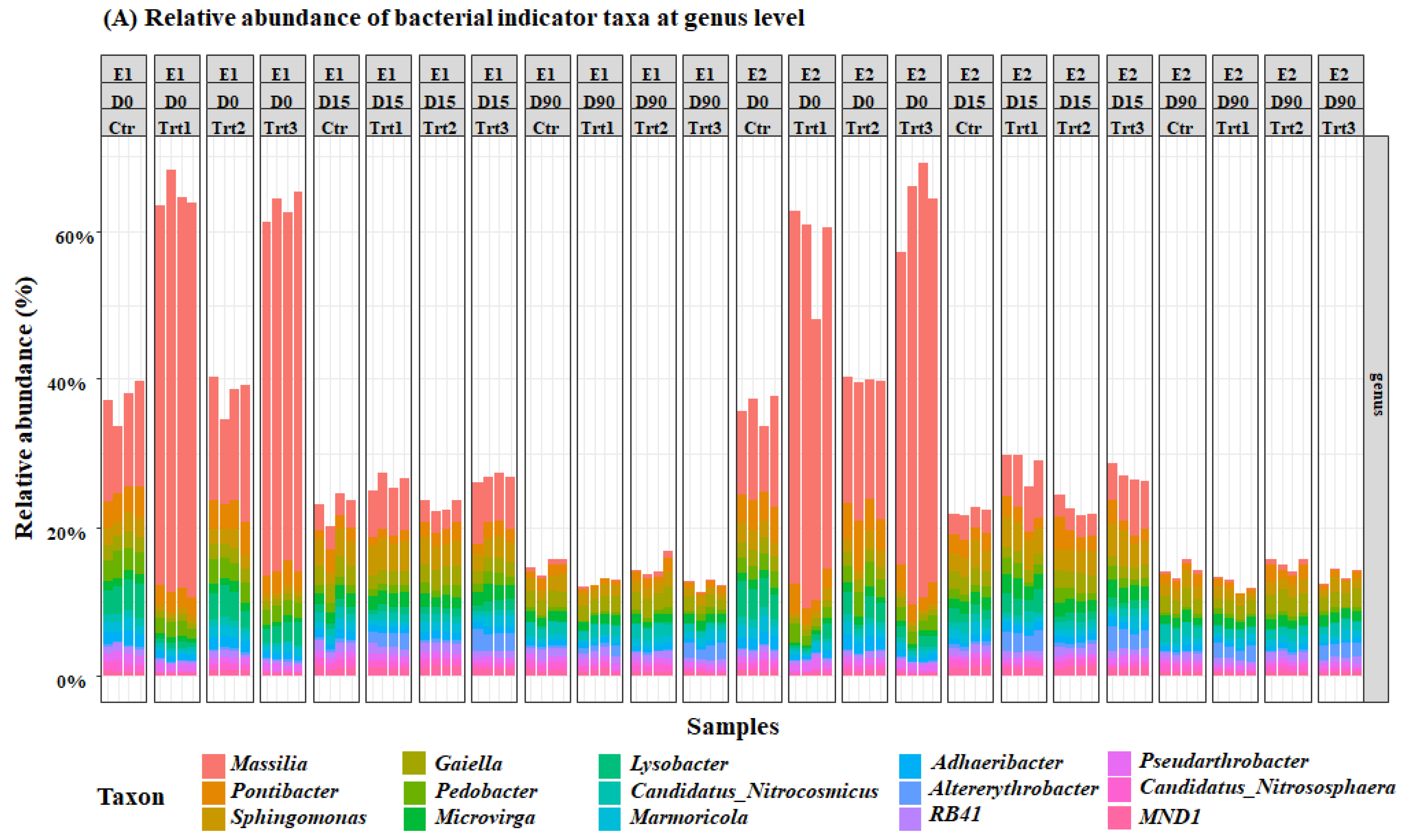
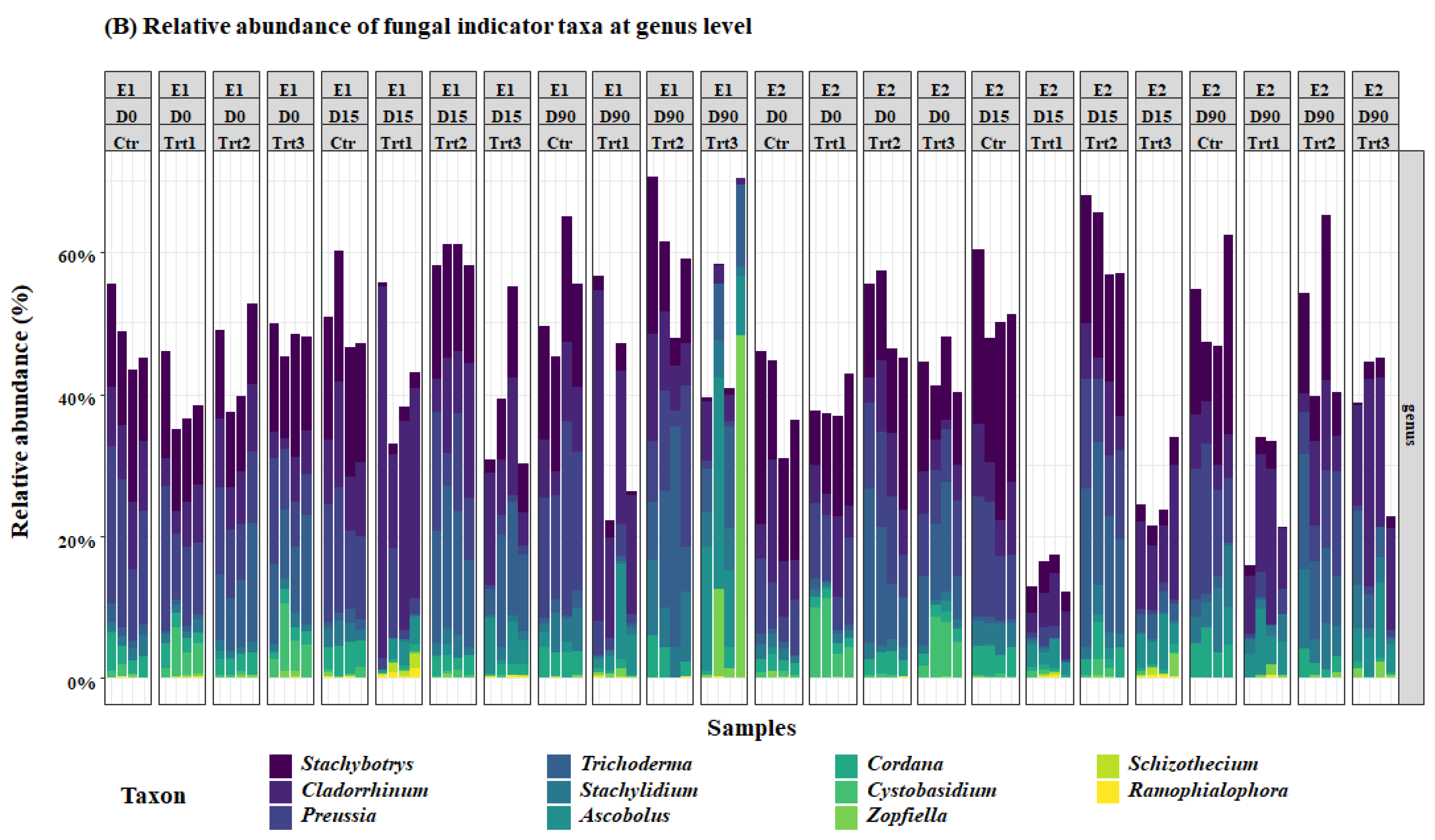
Figure 5.
Relative abundance of the most important bacterial (A) and fungal (B) genera. E1and E2: two experiments with four treatments at different sampling times (D0: after 12 h; D15: after 15 days; D90: after 90 days); Ctr: bulk soil; Trt1: soil with 10 g of beech wood pellets; Trt2: soil with Trichoderma atroviride SC1 conidial suspension at the rate of 5 × 103 conidia∙g−1 of soil; Trt3: soil with 10 g of T. atroviride SC1 coated beech pellets (5 × 105 conidia∙g−1 of beech wood pellets). Data of each replicate are presented in the histograms.
3. Results
3.1. Impact of the Trichoderma atroviride SC1 Coated Beech Wood Carrier on the Growth of Trichoderma spp. in Soil
Trichoderma atroviride SC1 DNA was not detected in the original bulk soil. Since the cfu counting does not allow for distinguishing the species/strains of Trichoderma, T. atroviride SC1 and the indigenous population of Trichoderma, are mentioned as Trichoderma spp. population throughout the paper.
The Trichoderma spp. cfu counts increased rapidly in the first 30 days, until D45 in Ctr and Trt1. For Ctr, the cfu counts were not significantly different at D15 and D30 compared to D0, according to Dunn’s post-hoc test (p = 0.380; p = 0.080). Then at D45, the counts reached levels significantly different from the ones registered at D0 and D15 (p = 0.001; p = 0.002). No significant difference was observed between the cfu counts of the Trichoderma spp. population at D60 compared to D30 (p = 0.074) and D45 (p = 0.370), respectively, while it remained significant for D0 (p = 0.002) and D15 (p = 0.004). At the end of the experiment (D90), a slight decrease in the population was observed (2.69 × 102 ± 102.87 cfu∙g−1 of soil) and a significant difference was recorded only with D0 (p = 0.021). For Trt1, the colonies started to grow faster than Ctr, and a significant difference was detected starting from D30 as compared to D0 (p = 0.010). The population of Trichoderma spp. continued to grow and became significantly different from the levels observed at D30 a month later at D60 (p = 0.009). The levels registered at D90 (1.00 × 103 ± 712.57 cfu∙g−1 of soil) remained significantly different from the cfu counts at D30, but not different from those of D45 (p = 0.275) and D60 (p = 0.338). The treatments Trt2 and Trt3, where T. atroviride SC1 was inoculated at the rate of 5 × 103 conidia∙g−1 of soil, maintained the same level of cfu count in the first 30 days compared to D0, with p = 0.065 and p = 0.206 for Trt2 and Trt3, respectively. Then at D45, the population of Trichoderma spp. started to decline in Trt2 (2.54 × 103 ± 689.00 cfu∙g−1 of soil) and Trt3 (1.40 × 103 ± 681.09 cfu∙g−1 of soil), reaching levels significantly different from the ones registered at D15 (p = 0.003; 0.001), and the same was observed between D60 and D30 (p = 0.012; p = 0.001). The population at D90 was significantly lower than all the other sampling points for Trt2, except for D60 (p = 0.103), while no significant difference was observed between D45, D60, and D90 for Trt3 (p = 0.057; p = 0.218).
Between treatments, at D0, there was a significant difference in the cfu counts between Ctr/Trt1 and Trt2/Trt3 (H = 31.538, df = 3, p < 0.001), which persisted until D45. At D45, no significant difference was observed between the counts of Ctr and Trt1 (p = 0.052), Trt1 and Trt3 (p = 0.054), or Trt2 and Trt3 (p = 0.050). Trt2 and Trt3 were both significantly different from Ctr (H = 29.067, df = 3, p < 0.001). Trt2 registered the highest cfu count among the treatments (2.54 × 103 ± 689.82 cfu∙g−1 of soil).
At D60 the cfu counts of Trt1 continued to rise and became significantly different from the control (p = 0.003). The cfu counts in Trt3 continued to decrease and became significantly different from the cfu counts in Trt2 (p = 0.011).
At D90, the population of Trichoderma spp. showed no statistical difference (p = 0.250; p = 0.140; p = 0.040) between Trt1 (1.03 × 103 ± 712.71 cfu∙g−1 of soil), Trt2 (1.07 × 103 ± 417.05 cfu∙g−1 of soil), and Trt3 (6.76 × 102 ± 232.81 cfu∙g−1 of soil), with cfu counts that were significantly higher than the control (H = 23.766, df = 3, p < 0.001) (Figure 1).
3.2. Impact of the Trichoderma atroviride SC1 Coated Beech Wood Carrier on the Microorganisms of the Soil
A total of 103,970.6 bacterial/archaeal reads and 86,077.48 fungal reads were obtained. The most dominant bacterial phyla, in terms of relative abundance, were Proteobacteria (34%), Crenarchaeota (10%), Actinobacteriota (10%), Bacteroidota (10%), and Acidobacteriota (10%). At genus level, Massilia was the most abundant, with 12%, followed by Pontibacter, Sphingomonas, and Gaiella, with (2%) and finally Microvirga (1%). The fungal taxa were dominated by Ascomycota (82%), then Basidiomycota (6%), and Mortierellomycota (6%), followed by Chytridiomycota (3%) and Aphelidiomycota (3%). Mortierella (6%) was the most dominant fungal genus, followed by Fusarium (4%) and Cladorrhinum (4%), and finally Gibberella (3%), then Stachybotrys (2%).
Bacterial and Fungal Richness and Diversity
The bacterial alpha diversity showed statistical differences in richness (observed ASVs) between the different sampling time points for each treatment (F = 514.48, p < 0.001) and between treatments (F = 45.94, p < 0.001). The increase in the bacterial richness occurred faster for all treatments (15 days after the inoculation) compared to the control, which showed a significant increase after 90 days (Figure 2). This shows that the introduction of the carrier components, the incorporation of T. atroviride SC1 into the soil, and their combined application enhanced the bacterial richness. At D90, the highest effect was observed with Trt3, which presented the highest ASVs (680) among all treatments, while no significant difference was observed between Trt1 and Trt2.
In contrast, the bacterial diversity (Simpson’s index) showed differences between treatments at D0 only (F = 245.07, p < 0.001). The treatments where a carrier was applied with or without T. atroviride SC1 (Trt1 and Trt3) affected the bacterial population community and decreased its diversity, as is shown by the low Simpson’s values (0.88 and 0.87) for Trt1 and Trt3, respectively. The addition of the carrier, which contains carbon and nitrogen sources, clearly favored certain genera that are more adapted to those components and decreased the less competent ones (Figure 2). Generally, the introduction of T. atroviride SC1 did not affect the bacterial population and the most important factor that governed the bacterial dynamics was the addition of the carrier (F = 61.40, p < 0.001).
The fungal alpha diversity showed significant differences in richness between the different sampling time points for each treatment (F = 303.58, p < 0.001), but not between treatments (F = 0.31, p = 0.81). The richness decreased in time for both control and treatments, with no exceptions (Figure 3).
Simpson’s diversity index was significantly different for Trt1 compared to the other treatments, including the control (F = 5.17, p < 0.001) (Figure 3). This shows the effect of combining the addition of wood to the soil with the application of T. atroviride SC1. The fungal diversity in the treatments where T. atroviride SC1 was applied with and without wood pellets were not significantly different from the control (Figure 3). In contrast, the fungal diversity in Trt1, which was supplemented only with wood, was significantly different. This shows that adding T. atroviride SC1 can counterbalance the effects of the wood on the fungal diversity. The addition of wood was the main cause for the differences observed between treatments. The recursive partitioning analysis of the bacterial and fungal richness and diversity confirmed the above-mentioned results (Figure S1).
Unsupervised non-metric multidimensional scaling (NMDS) ordinations applied on Bray–Curtis dissimilarities showed that the dissimilarities observed among the bacterial samples were grouped (Figure 4) according to the factors, time and treatment, while it pooled the fungal communities only according to the factor, time (Figure 4). In fact, the permutational multivariate analyses of variance (PERMANOVA) on Bray–Curtis dissimilarities revealed that time was responsible for the biggest portion of the variation in microbiome beta-diversity. The bacterial community differed very significantly according to the factor time (F = 144.95, R2 = 0.56, p < 0.001) and treatment (F = 20.38, R2 = 0.11, p < 0.001), as well as their interaction (F = 11.37, R2 = 0.13, p < 0.001), and less significantly with the factor experiment (F = 3.47, R2 = 0.006, p = 0.015). The same was observed for the fungal communities, which varied according to time (F = 21.99, R2 = 0.23, p < 0.001), treatment (F = 8.59, R2 = 0.14, p < 0.001), the interaction between the two factors (F = 3.35, R2 = 0.10, p = 0.001), and finally the factor experiment (F = 2.64, R2 = 0.014, p = 0.006). These results indicate a high consistency of the effects of the treatments over time (T. atroviride SC1 coated and uncoated wood pellets and T. atroviride SC1) on soil microbial communities.
Bacterial and fungal indicator ASVs that have significantly different abundances (p < 0.05) in the factor time and treatment were extracted from the mGLM results. The results of the Simpson’s bacterial diversity index correspond with an increase in the population of the genus Massilia, which was significantly different between the two groups Ctr/Trt2 and Trt1/Trt3 (Figure 5). The results also revealed a significant increase in terms of the relative abundances of the genera Pontibacter, Sphingomonas, Gaiella, Pedobacter, and Microvirga (Figure 5).
For the fungal community, at D0, Trichoderma spp. were higher in Trt2 and Trt3 than in Ctr and Trt1, as expected. Contrarily to the cfu counts, however, this did not change until the end of the experiment, according to Dunn’s post-hoc test. The carrier (Trt1/Trt3) selectively and significantly increased the total ASV’s of Cystobasidium, Ascobolus, Stachybotrys, Cladorrhinum, Preussia, and Stachylidium (Figure 5).
4. Discussion
Trichoderma spp. formulations are important, as they can delay the decline of the population of the fungus, protecting conidia from soil fungistasis [65,66], by providing nutrients to selectively stimulate their growth, or by combining both mentioned benefits [67]. In our experiment, we tested the effect of a carrier based on lignocellulosic materials (beech wood pellet supplemented with soy protein isolates) on the growth of T. atroviride SC1 in a sandy loam, and assessed the effect of such a carrier on the microbiota of the soil. Since it was hard to distinguish colonies of our strain from the indigenous population in the soil, we reported the results as the population of the total Trichoderma spp., instead of T. atroviride SC1.
The Trichoderma population showed a steady growth in the Ctr and Trt1 for the first 30 to 45 days, then it stabilized. This growth can be explained by the addition of SDW, which stimulated the indigenous population to germinate and exploit the soil organic matter. This is in concordance with studies that show that water affects the microbial growth in the soil [68,69,70]. Trt2, where T. atroviride SC1 was applied as an SDW conidial suspension, showed the typical population decline that is reported in literature when Trichoderma spp. are applied as conidia [4,35]. The population remained stable for 30 days, then started to decline gradually until D90. This result is in concordance with [31], who reported a decrease in the population of Trichoderma atroviride I-1237 after three weeks in neutral clayey soils and after 13 weeks after its inoculation into an acidic sandy loam. Their research showed that the decline of the population of Trichoderma spp. can be governed by the physicochemical characteristics of a soil. In fact, since the growth of T. atroviride SC1 on wood has been demonstrated by previous studies [10,71], we expected the fungus to grow to higher levels in Trt3 compared to Trt1 and Trt2; however, surprisingly, the total Trichoderma population remained stable for 30 days and then declined to levels similar to the ones registered with Trt1 and Trt2, which suggests a soil fungistatic effect that inhibited the growth of T. atroviride SC1. The growth of T. atroviride SC1 in the soil can be hindered by unfavorable soil conditions (pH 8 in our experiment), as it grows best in acidic conditions [72]. This has also been reported in other studies, which showed that the growth of Trichoderma spp. can be affected by soil texture and pH [73,74,75].
The carrier increased the Trichoderma spp. population in Trt1 steadily in the first 45 days and then it stabilized to levels that were not significantly different to those observed in the treatments where T. atroviride SC1 was added with and without coated beech pellets. Since Trt1 did not contain detectable levels of T. atroviride SC1 (since initially we tested the soil for the presence of this fungal strain using specific primers [36]) the presence of other competitive strains of Trichoderma spp. that are more efficient than our strain in degrading wood is highly plausible. Competition with other Trichoderma spp. could have played an important role in slowing down the growth of T. atroviride SC1 with the carrier in Trt3 as compared to Trt1, where the population of Trichoderma flourished in the first 45 days. Generally, Trichoderma spp. compete with each other in the soil for wood colonization with an effectiveness that depends on the species [76,77]. Several species have been reported in the literature for their high cellulolytic activity, such as T. reesei, T. viride, T. harzianum, T. virens, and T. longibrachiatum [78,79,80]. The difference that was observed between the results of the cfu counts of Trichoderma spp. and the results of the ITS amplicon-based analysis is in concordance with previous studies, which reported that such differences could be due to the fact that a dead propagule that still contains DNA does not develop into a cfu [81].
Since soil experiments can be influenced by a complex of physicochemical factors of a soil, such as temperature, texture, water availability, aeration, and light, as well as other biological factors, consisting mainly of the distribution of microorganisms in the soil and their interactions [69], we repeated the experiment twice. Our results show that the experimental design consistently detected the differences occurring due to time and treatment. The incorporation of T. atroviride SC1 did not affect the bacterial richness and diversity. This is in concordance with other studies that reported a transient effect of Trichoderma spp. on the microbial population [31,36,82,83]. The fungal richness, however, decreased in all treatments, probably due to the growth of fungal genera that are more competitive in growing in conditions either of low organic matter or on woody substrates. Cordier and Alabouvette [31] observed the same effect with the introduction of T. atroviride I-1237 on the fungal community; however, the change only lasted for three months, which was the full length of our experiment. The fungal diversity also decreased in all treatments; however, it decreased the most in Trt1, where pellets were introduced in the absence of T. atroviride SC1. This suggests that the main driver of the change observed between treatments was the introduction of non-coated beech wood pellets and that T. atroviride SC1 contributed in balancing the fungal diversity, probably by increasing the availability of nutrients to other fungi. This is in concordance with Asghar and Kataoka [34], who found that introducing organic amendments into the soil had a negative effect on the diversity of the fungal community. Longa et al. [84] also reported a correlation between the increase in the organic matter and the abundance of microbial functional groups with agricultural importance, such as nitrogen fixing bacteria when soil was supplied with green manure.
In our study, the tested soil was rich in wood-degrading bacteria and fungi in all treatments, as was demonstrated by the analysis of the top ten genera of bacteria and fungi, in terms of relevant abundance. These analyses revealed a dominance of the genus Massila for bacteria in all treatments, including the control. Massilia, which are saprophytic and opportunistic and were significantly more abundant in the presence of the carrier at D0. This can be explained by the early growth of these bacteria on wood pellets. In fact, Oxalobacteraceae in general, and Massilia spp. in particular, are very active in the early stages of bacterial succession in soils [85], and species of this genus can produce cellulases [86,87], which can explain their initial growth on wood and soil amended with fresh plant residues [88,89]. However, this effect is usually transient and only occurs when sufficient carbon and energy sources are present, and before competition with other microorganisms becomes limiting [87]. Overall, the carrier increased the bacterial richness and had a transient effect on its diversity. This is in concordance with other studies that showed that using inorganic nitrogen fertilizers in bulk soils affects their bacterial composition [90]. Illescas et al. [91] found that inorganic fertilizers increased bacterial genera with antagonistic activities, such as Sphingomonas and Pseudomonas, Kaistobacter, and Streptomyces. We observed the same pattern with Sphingomonas and Pseudomonas, but also Lysobacter, which was not reported in their study. Lysobacter spp., named after their lytic effects on other microorganisms and which are often good biocontrol agents, are Gram-negative bacteria that are frequently found in soils. Their increase could be a response to the increase of other microorganisms or to the availability of a nutritional substrate related to the treatments [92].
The carrier also enriched the presence of bacteria involved in the soil nitrogen cycle such as Microvirga and Pedobacter, which confirms the results obtained by Longa et al. [84] when soil was supplemented with green manure. These results suggest a selective effect of a wood-based carrier of T. atroviride SC1. This selective effect was more visible for fungi. In fact, non-coated beech wood pellets decreased both the richness and diversity of the fungal population. This could be the result of promoting fungi that are more adapted to the addition of wood, particularly in bulk soils [90], such as Mortierella, which was the most abundant genus. These fungi are considered good degraders of toxic pollutants, such as pesticides and heavy metals [91,93,94], and they have potential for biocontrol [95,96,97]. They are also important plant growth promoters and can enhance soil conditions under salt stress [98,99], but most importantly, they can degrade cellulose, hemicellulose, and organic matter in periods that range from 30 to 430 days, depending on the substrate and the soil conditions [100,101]. Tamayo-Vélez et al. [100] reported an optimal degradation rate of organic matter by Mortierella 90 days after inoculation. These results can be compared to what we have observed, as 90 days were sufficient to rank Mortierella spp. as the most abundant fungi in the soil. This shows that wood pellets are a suitable substrate for the growth of Mortierella spp. This might have caused the competition that prevented Trichoderma spp. in general, and T. atroviride SC1 in particular, from thriving in the pots supplemented with coated wood pellets.
Another genus that might have played an important role in the competition for wood degradation is Cladorrhinum. Cladorrhinum spp. have been extensively reported in agricultural soils [102,103]. They were found in soil as saprotrophs on dung or plant material [104,105], or in roots as endophytes [106]. It is an ammonia fungus belonging to the early successional phase of fungi involved in the saprotrophic litter decomposition in soil [107]. Moreover, they are more efficient than Trichoderma for the degradation of hardwood [23,108]. This selective abundance of the carrier might be beneficial, as Gasoni and Stegman de Gurfinkel [109] reported a potential antagonistic activity of Cladorrhinum spp. against Rhizoctonia solani. In fact, this is not the only fungus found in our research that can be effective against Rhizoctonia solani. Some species of the genus Stachybotrys, which is also commonly found in soils and on cellulose [110,111], have a strong antagonistic activity against R. solani through mycoparasitism, by the means of chitinases and β-1,3-glucanases [110]. One of the species of this genus, namely, Stachybotrys chartarum was also suspected to play a role in the development of human pulmonary diseases; however, the results are not yet conclusive [112,113]. Generally, drying pellets before coating, as suggested in our protocol, would eliminate any potential development of unwanted molds that could be harmful to animals or humans; however, further research is needed in different soil types and with different wood species. The results of the selective abundance of beneficial microbes is in concordance with Sani et al. [114], Fu et al. [115], and Zhang et al. [99], who reported a decrease in the relative abundance of genera hosting phytopathogens such as Neonectria and Fusarium, improved soil fertility, and an increase in the relative abundance of plant growth-promoting rhizobacteria, respectively, when different Trichoderma isolates where inoculated into the soil. In our research, the results of the two experiments yielded more consistent results with bacteria compared to fungi. This could be explained by the high competition between fungi in colonizing the woody substrate. The decreased prevalence of wood degrading fungi in some samples was replaced by an increase in the relative abundance of the genera Cordana, Cystobasidium, Zopfiella, Schizothecium, and Ramophialophora, which might be due to the heterogeneous distribution of microorganisms in the soil [69].
5. Conclusions
The incorporation of T. atroviride SC1 coated wood pellets in the soil enriched the bacterial community and had a selective effect on the abundance of beneficial fungal species that have biocontrol potential. Although a wider screening of a combination of different wood and soil types is necessary to confirm this effect, these results are promising as they reinforce the suitability of the use of wood pellets as carriers of Trichoderma spp., because, in addition to being good growth substrates for the fungus, they have a mild effect on the microbial community, as they induce temporary side effects on the bacterial community, and favor the growth of fungal species with biocontrol potential. In addition, the carrier did not increase any potential human or plant pathogens. Although wood pellets can be used as carriers of conidia of Trichoderma spp., further studies are needed to assess their efficacy against soil-borne pathogens and to optimize application conditions and dosages in order to reach an efficient biocontrol effect. Possible side-effects on the plant, e.g., phytotoxicity, must also be tested.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/jof7090751/s1, Figure S1: Recursive partitioning analysis of bacterial richness and diversity.
Author Contributions
Conceptualization, H.C., A.N. and I.P.; Data curation, H.C., L.A. and M.P.; Funding acquisition, I.P.; Methodology, H.C., A.N. and I.P.; Project administration, I.P.; Software, L.A.; Supervision, I.P.; Validation, I.P.; Visualization, L.A.; Writing—original draft, H.C.; Writing—review and editing, H.C., L.A., A.N., M.P. and I.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Fondazione Edmund Mach (San Michele all’Adige, Italy). Bi-PA nv (Biological Products for Agriculture, Belgium) funded the Ph.D. grant (197564) of Hamza Chammem at the University of Trento.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
Andrea Nesler is a development engineer at Bi-PA nv. The remaining authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Rahman, A.B.; Syed, S.F.; Singh, E.; Pieterse, C.M.J.; Schenk, P.M. Emerging microbial biocontrol strategies for plant pathogens. Plant Sci. 2018, 267, 102–111. [Google Scholar] [CrossRef] [Green Version]
- Kubicek, C.P.; Mach, R.L.; Peterbauer, C.K.; Lorito, M. Trichoderma: From genes to biocontrol. J. Plant Pathol. 2001, 83, 11–23. Available online: http://www.jstor.org/stable/41998018 (accessed on 26 July 2021).
- Ferreira, F.V.; Musumeci, M.A. Trichoderma as biological control agent: Scope and prospects to improve efficacy. World J. Microbiol. Biotechnol. 2021, 37, 90. [Google Scholar] [CrossRef]
- Papavizas, G.C. Survival of Trichoderma harzianum in soil and in pea and bean rhizosphere. Phytopathology 1982, 72, 121–125. [Google Scholar] [CrossRef]
- Elad, Y.; Chet, I.; Katan, J. Trichoderma harzianum: A Biocontrol Agent Effective against Sclerotium rolfsii and Rhizoctonia solani. Phytopathology 1980, 70, 119–121. [Google Scholar] [CrossRef] [Green Version]
- Lewis, J.A.; Papavizas, G.C. Application of Trichoderma and Gliocladium in alginate pellets for control of Rhizoctonia damping-off. Plant Pathol. 1987, 36, 438–446. [Google Scholar] [CrossRef]
- Papavizas, G.C.; Lewis, J.A. Effect of Gliocladium and Trichoderma on damping-off and blight of snapbean caused by Sclerotium rolfsii in the greenhouse. Plant Pathol. 1989, 38, 277–286. [Google Scholar] [CrossRef]
- Kumar, G.; Maharshi, A.; Patel, J.; Mukherjee, A.; Singh, H.B.; Sarma, B.K. Trichoderma: A potential fungal antagonist to control plant diseases. SATSA Mukhapatra Annu. Tech. 2017, 21, 206–218. [Google Scholar]
- Burges, H.D.; Jones, K.A. Trends in formulation of microorganisms and future research requirements. In Formulation of Microbial Biopesticides: Beneficial Microorganisms, Nematodes and Seed Treatments; Burges, H.D., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1998; pp. 311–332. [Google Scholar]
- Chammem, H.; Nesler, A.; Pertot, I. Wood pellets as carriers for Trichoderma atroviride SC1 for soil application. Fungal Biol. 2021. [Google Scholar] [CrossRef]
- Papavizas, G.C.; Dunn, M.T.; Lewis, J.A.; Beagle-Ristaino, J. Liquid fermentation technology for experimental production of biocontrol fungi. Phytopathology 1984, 74, 1171–1175. [Google Scholar] [CrossRef]
- Jin, X.; Hayes, K.C.; Harman, G.E. Principles in the development of biological control systems employing Trichoderma species against soil-born plant pathogenic fungi. In Frontiers in Industrial Mycology; Lantham, G.F., Ed.; Chapman & Hall Inc.: New York, NY, USA, 1992; pp. 174–195. [Google Scholar]
- Whipps, J.M. Developments in the biological control of soil-borne plant pathogens. Adv. Bot. 1997, 26, 1–134. [Google Scholar]
- Shaban, G.M.; El-Komy, H.M.A. Survival and proliferation of alginate encapsulated Trichoderma spp. in Egyptian soil in comparison with allyl alcohol soil fumigation. Mycopathology 2001, 151, 139–146. [Google Scholar] [CrossRef]
- Thangavelu, R.; Palaniswami, A.; Velazhahan, R. Mass production of Trichoderma harzianum for managing Fusarium wilt of banana. Agric. Ecosyst. Environ. 2004, 103, 259–263. [Google Scholar] [CrossRef]
- Kolombet, L.V.; Zhigletsova, S.K.; Kosareva, N.I.; Bystrova, E.V.; Derbyshev, V.V.; Krasnova, S.P.; Schisler, D. Development of an extended shelf-life, liquid formulation of the biofungicide Trichoderma asperellum. World J. Microbiol. Biotechnol. 2008, 24, 123–131. [Google Scholar] [CrossRef]
- Al-Taweil, H.I.; Osman, M.B.; Abdulhamid, A.; Mohammad, N.; Yussof, W.M.W. Comparison of different delivery system of Trichoderma and Bacillus as biofertilizer. Adv. Environ. Biol. 2010, 4, 31–33. [Google Scholar]
- Sriram, S.; Palanna, K.B.; Ramanujam, B. Effect of chitin on the shelf-life of Trichoderma harzianum in talc formulation. Indian J. Agric. Sci. 2010, 80, 930–932. [Google Scholar]
- Sriram, S.; Roopa, K.P.; Savitha, M.J. Extended shelf-life of liquid fermentation derived talc formulations of Trichoderma harzianum with the addition of glycerol in the production medium. Crop. Prot. 2011, 30, 1334–1339. [Google Scholar] [CrossRef]
- John, R.P.; Tyagi, R.D.; Brar, S.K.; Surampalli, R.Y.; Prévost, D. Bio-encapsulation of microbial cells for targeted agricultural delivery. Crit. Rev. Biotechnol. 2011, 31, 211–226. [Google Scholar] [CrossRef]
- Cumagun, C.J.R. Advances in Formulation of Trichoderma for Biocontrol. In Biotechnology and Biology of Trichoderma; Gupta, V.K., Schmoll, M., Herrera-Estrella, A., Upadhyay, R.S., Druzhinina, I., Tuohy, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; Chapter 39; pp. 527–532. [Google Scholar]
- Antheunisse, J.; Burema, J. Biological and chemical inhibition of lingo-cellulose decay by white rot basidiomycetes. Gen. Appl. Microbiol. 1983, 29, 257–269. [Google Scholar] [CrossRef] [Green Version]
- Mao, T.; Jiang, X. Changes in Microbial Community and Activities in Continuous Pepper Cropping Soil after a Trichoderma Hamatum MHT1134 Application. Research 2021. [Google Scholar] [CrossRef]
- Fontaine, S.; Mariotti, A.; Abbadie, L. The priming effect of organic matter: A question of microbial competition? Soil Biol. Biochem. 2003, 35, 837–843. [Google Scholar] [CrossRef]
- Compant, S.; Samad, A.; Faist, H.; Sessitsch, A. A review on the plant microbiome: Ecology, functions, and emerging trends in microbial application. J. Adv. Res. 2019, 19, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Chirinda, N.; Olesen, J.E.; Porter, J.R. Effects of organic matter input on soil microbial properties and crop yields in conventional and organic cropping systems. In Proceedings of the 16th IFOAM Organic World Congress, Modena, Italy, 16–20 June 2008. [Google Scholar]
- Hou, J.; Li, M.; Mao, X.; Hao, Y.; Ding, J.; Liu, D.; Xi, B.; Liu, H. Response of microbial community of organic-matter-impoverished arable soil to long-term application of soil conditioner derived from dynamic rapid fermentation of food waste. PLoS ONE 2017, 12, e0175715. [Google Scholar] [CrossRef]
- Vermeire, M.L.; Cornélis, J.T.; Van Ranst, E.; Bonneville, S.; Doetterl, S.; Delvaux, B. Soil Microbial Populations Shift as Processes Protecting Organic Matter Change During Podzolization. Front. Environ. Sci. 2018, 6, 70. [Google Scholar] [CrossRef] [Green Version]
- Bärlocher, F.; Boddy, L. Aquatic fungal ecology—How does it differ from terrestrial? Fungal Ecol. 2016, 19, 5–13. [Google Scholar] [CrossRef]
- Cai, Y.; Chen, H.; Yuan, R.; Wang, F.; Chen, Z.; Zhou, B. Toxicity of perfluorinated compounds to soil microbial activity: Effect of carbon chain length, functional group and soil properties. Sci. Total Environ. 2019, 690, 1162–1169. [Google Scholar] [CrossRef] [PubMed]
- Cordier, C.; Alabouvette, C. Effects of the introduction of a biocontrol strain of Trichoderma atroviride on non target soil micro-organisms. Eur. J. Soil Biol. 2009, 45, 267–274. [Google Scholar] [CrossRef]
- Liu, B.; Glenn, D.; Buckley, K. Trichoderma communities in soils from organic, sustainable, and conventional farms, and their relation with Southern blight of tomato. Soil Biol. Biochem. 2008, 40, 1124–1136. [Google Scholar] [CrossRef]
- Ye, L.; Zhao, X.; Bao, E.; Li, J.; Zou, Z.; Cao, K. Bio-organic fertilizer with reduced rates of chemical fertilization improves soil fertility and enhances tomato yield and quality. Sci. Rep. 2020, 10, 1–11. [Google Scholar]
- Asghar, W.; Kataoka, R. Effect of co-application of Trichoderma spp. with organic composts on plant growth enhancement, soil enzymes and fungal community in soil. Arch. Microbiol. 2021, 203, 4281–4291. [Google Scholar] [CrossRef]
- Longa, C.M.O.; Pertot, I. An intact soil-core microcosm method to evaluate the survival and vertical dispersal of Trichoderma atroviride SC1. Lett. Appl. Microbiol. 2009, 49, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Savazzini, F.; Longa, C.M.O.; Pertot, I. Impact of the biocontrol agent Trichoderma atroviride SC1 on soil microbial communities of a vineyard in northern Italy. Soil Biol. Biochem. 2009, 41, 1457–1465. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Lozupone, C.A.; Turnbaugh, P.J.; Fierer, N.; Knight, R. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc. Natl. Acad. Sci. USA 2011, 108, 4516–4522. [Google Scholar] [CrossRef] [Green Version]
- Apprill, A.; McNally, S.; Parsons, R.; Weber, L. Minor revision to V4 region SSU rRNA 806R gene primer greatly increases detection of SAR11 bacterioplankton. Aquat. Microb. Ecol. 2015, 75, 129–137. [Google Scholar] [CrossRef] [Green Version]
- Parada, A.E.; Needham, D.M.; Fuhrman, J.A. Every base matters: Assessing small subunit rRNA primers for marine microbiomes with mock communities, time series and global field samples. Environ. Microbiol. 2016, 18, 1403–1414. [Google Scholar] [CrossRef]
- Walters, W.; Hyde, E.R.; Berg-Lyons, D.; Ackermann, G.; Humphrey, G.; Parada, A. Improved bacterial 16S rRNA gene (V4 and V4–5) and fungal internal transcribed spacer marker gene primers for microbial community surveys. mSystems 2016, 1, e00009-15. [Google Scholar] [CrossRef] [Green Version]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specificity for basidiomycetes—application to the identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal rna genes for phylogenetics. In PCR Protocols; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Elsevier: Amsterdam, The Netherlands; Academic Press: Cambridge, MA, USA, 1990; pp. 315–322. [Google Scholar] [CrossRef]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [Green Version]
- Andrews, S. FASTQC. A Quality Control Tool for High Throughput Sequence Data 2010. Available online: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 9 August 2021).
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bengtsson-Palme, J.; Thorell, K.; Wurzbacher, C.; Sjöling, Å.; Nilsson, R.H. Metaxa2 Diversity Tools: Easing microbial community analysis with Metaxa2. Ecol. Inform. 2016, 33, 45–50. [Google Scholar] [CrossRef]
- Bengtsson-Palme, J.; Ryberg, M.; Hartmann, M.; Branco, S.; Wang, Z.; Godhe, A. Improved software detection and extraction of ITS1 and ITS2 from ribosomal ITS sequences of fungi and other eukaryotes for analysis of environmental sequencing data. Methods Ecol. Evol. 2013, 4, 914–919. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Nilsson, R.H.; Larsson, K.-H.; Taylor, A.F.S.; Bengtsson-Palme, J.; Jeppesen, T.S.; Schigel, D. The UNITE database for molecular identification of fungi: Handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res. 2019, 47, D259–D264. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 10 August 2021).
- Chen, W.; Simpson, J.; Levesque, C.A. RAM: R for Amplicon-Sequencing-Based Microbial-Ecology; Agriculture and Agri-Food Canada: Canada, 2020; Available online: https://cran.r-project.org/package=RAM (accessed on 10 August 2021).
- Saary, P.; Bork, P.; Hildebrand, F. Rtk: Rarefaction Tool Kit. Bioinformatics 2017, 33, 2594–2595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delignette-Muller, M.-L.; Dutang, C.; Pouillot, R.; Denis, J.-B.; Siberchicot, A. Fitdistrplus: Help to Fit of a Parametric Distribution to Non-Censored or Censored Data. 2021. Available online: https://rdrr.io/cran/fitdistrplus/ (accessed on 10 September 2021).
- Kuhn, M. Building Predictive Models in R Using the Caret Package. J. Stat. Softw. 2008, 28, 1–26. [Google Scholar] [CrossRef] [Green Version]
- Lenth, R.; Buerkner, P.; Herve, M.; Love, J.; Riebl, H.; Singmann, H. Emmeans: Estimated Marginal Means, Aka Least-Squares Means. 2020. Available online: https://cran.r-project.org/web/packages/emmeans/index.html (accessed on 10 September 2021).
- Wickham, H.; Chang, W.; Henry, L.; Pedersen, T.L.; Takahashi, K.; Wilke, C.; Woo, K.; Yutani, H.; Dunnington, D. RStudio Ggplot2: Create Elegant Data Visualisations Using the Grammar of Graphics. 2021. Available online: https://cran.r-project.org/web/packages/ggplot2/ggplot2.pdf (accessed on 10 September 2021).
- Hothorn, T.; Seibold, H.; Zeileis, A. Partykit: A Toolkit for Recursive Partytioning. 2020. Available online: https://cran.r-project.org/web/packages/partykit/vignettes/partykit.pdf (accessed on 10 September 2021).
- Love, M.; Ahlmann-Eltze, C.; Forbes, K.; Anders, S.; Huber, W. DESeq2: Differential Gene Expression Analysis Based on the Negative Binomial Distribution; Bioconductor Version: Release (3.12). 2021. Available online: https://bioc.ism.ac.jp/packages/devel/bioc/manuals/DESeq2/man/DESeq2.pdf (accessed on 10 September 2021).
- Simpson, G.L. Ggvegan: Ggplot2-Based Plots for Vegan. Available online: https://gavinsimpson.github.io/ggvegan/ (accessed on 12 March 2021).
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. 2019. Available online: https://cran.r-project.org/web/packages/vegan/index.html (accessed on 10 September 2021).
- Wang, Y.; Naumann, U.; Eddelbuettel, D.; Wilshire, J.; Warton, D.; Byrnes, J.; Silva, R.d.S.; Niku, J.; Renner, I.; Wright, S. Mvabund: Statistical Methods for Analysing Multivariate Abundance Data. 2020. Available online: https://cran.r-project.org/web/packages/mvabund/mvabund.pdf (accessed on 10 September 2021).
- Hervé, M. RVAideMemoire: Testing and Plotting Procedures for Biostatistics. 2020. Available online: https://cran.r-project.org/web/packages/RVAideMemoire/RVAideMemoire.pdf (accessed on 10 September 2021).
- Ogle, D.; Doll, J.; Wheeler, P.; Dinno, A. Provided Base Functionality of FSA: Simple Fisheries Stock Assessment Methods. 2021. Available online: https://cran.r-project.org/web/packages/FSA/FSA.pdf (accessed on 10 September 2021).
- Martínez-Medina, A.; Roldán, A.; Pascual, J.A. Performance of a Trichoderma harzianum bentonite-vermiculite formulation against Fusarium wilt in seedling nursery melon plants. HortScience 2009, 44, 2025–2027. [Google Scholar] [CrossRef] [Green Version]
- Larkin, R.P. Impacts of biocontrol products on Rhizoctonia disease of potato and soil microbial communities, and their persistence in soil. Crop. Prot. 2016, 90, 96–105. [Google Scholar] [CrossRef]
- Prasad, R.D.; Rangeshwaran, R.; Hegde, S.V.; Anuroop, C.P. Effect of soil and seed application of Trichoderma harzianum on pigeonpea wilt caused by Fusarium udum under field conditions. Crop. Prot. 2002, 21, 293–297. [Google Scholar] [CrossRef]
- Griffin, D.M. Soil moisture and the ecology of soil fungi. Biol. Rev. 1963, 38, 141–166. [Google Scholar] [CrossRef] [PubMed]
- Griffin, D.M. Soil water and the ecology of fungi. Annu. Rev. Phytopathol. 1969, 15, 319–329. [Google Scholar] [CrossRef]
- Kaisermann, P.A.; Maron, L.; Beaumelle, J.C. Lata, Fungal communities are more sensitive indicators to non-extreme soil moisture variations than bacterial communities. Appl. Soil Ecol. 2015, 86, 158–164. [Google Scholar] [CrossRef]
- Pellegrini, A.; Prodorutti, D.; Pertot, I. Use of bark mulch pre-inoculated with Trichoderma atroviride to control Armillaria root rot. Crop. Prot. 2014, 64, 104–109. [Google Scholar] [CrossRef]
- Longa, C.M.O.; Pertot, I.; Tosi, S. Ecophysiological requirements and survival of a Trichoderma atroviride isolate with biocontrol potential. J. Basic Microbiol. 2008, 48, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Brockett, B.F.T.; Prescott, C.E.; Grayston, S.J. Soil moisture is the major factor influencing microbial community structure and enzyme activities across seven biogeoclimatic zones in western Canada. Soil Biol. Biochem. 2012, 44, 9–20. [Google Scholar] [CrossRef]
- Van der Bom, F.; Nunes, I.; Raymond, N.S.; Hansen, V.; Bonnichsen, L.; Magid, J.; Nybroe, O.; Jensen, L.S. Long-term fertilisation form, level and duration affect the diversity, structure and functioning of soil microbial communities in the field. Soil Biol. Biochem. 2018, 122, 91–103. [Google Scholar] [CrossRef]
- Mayo-Prieto, S.; Porteous-Álvarez, A.; Mezquita-García, S.; Rodríguez-González, Á.; Carro-Huerga, G.; Del Ser-Herrero, S.; Gutiérrez, S.; Casquero, P. Influence of Physicochemical Characteristics of Bean Crop Soil in Trichoderma spp. Development. Agron. 2021, 11, 274. [Google Scholar] [CrossRef]
- Widden, P.; Hsu, D. Competition between Trichoderma species: Effects of temperature and litter type. Soil Biol. Biochem. 1987, 19, 89–93. [Google Scholar] [CrossRef]
- Klein, D.; Eveleigh, D.E. Trichoderma and Gliocladium; Kubicek, C.P., Harman, G.E., Eds.; Taylor and Francis: London, UK, 1998; Volume 1, pp. 57–69. [Google Scholar]
- Mutschlechner, M.; Illmer, P.; Wagner, A.O. Biological pre-treatment: Enhancing biogas production using the highly cellulolytic fungus Trichoderma viride. Waste Manag. 2015, 43, 98–107. [Google Scholar] [CrossRef]
- Bischof, R.H.; Ramoni, J.; Seiboth, B. Cellulases and beyond: The first 70 years of the enzyme producer Trichoderma reesei. Microb. Cell Factor. 2016, 15, 106. [Google Scholar] [CrossRef] [Green Version]
- Marecik, R.; Błaszczyk, L.; Biegańska-Marecik, R.; Piotrowska-Cyplik, A. Screening and identification of Trichoderma strains isolated from natural habitats with potential to cellulose and xylan degrading enzymes production. Pol. J. Microbiol. 2018, 67, 181–190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Savazzini, F.; Longa, C.M.O.; Pertot, I.; Gessler, C. Real-time PCR for detection and quantification of the biocontrol agent Trichoderma atroviride strain SC1 in soil. J. Microbiol. Methods 2008, 73, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Liu, L.; Wen, T.; Zhang, J.; Shen, Q.; Cai, Z. Reductive soil disinfestations combined or not with Trichoderma for the treatment of a degraded and Rhizoctonia solani infested greenhouse soil. Sci. Hortic. 2016, 206, 51–61. [Google Scholar] [CrossRef]
- Umadevi, P.; Anandaraj, M.; Srivastav, V.; Benjamin, S. Trichoderma harzianum MTCC 5179 impacts the population and functional dynamics of microbial community in the rhizosphere of black pepper (Piper nigrum L.). Braz. J. Microbiol. 2018, 49, 463–470. [Google Scholar] [CrossRef]
- Longa, C.M.O.; Nicola, L.; Antonielli, L.; Mescalchin, E.; Zanzotti, R.; Turco, E.; Pertot, I. Soil microbiota respond to green manure in organic vineyards. J. Appl. Microbiol. 2017, 123, 1547–1560. [Google Scholar] [CrossRef]
- Shrestha, P.M.; Noll, M.; Liesack, W. Phylogenetic identity, growth response time and rRNA operon copy number of soil bacteria indicate different stages of community succession. Environ. Microbiol. 2007, 9, 2464–2474. [Google Scholar] [CrossRef] [PubMed]
- Hrynkiewicz, K.; Baum, C.; Leinweber, P. Density, metabolic activity, and identity of cultivable rhizosphere bacteria on Salix viminalis in disturbed arable and landfill soils. J. Plant Nutr. Soil Sci. 2010, 173, 747–756. [Google Scholar] [CrossRef]
- Ofek, M.; Hadar, Y.; Minz, D. Ecology of Root Colonizing Massilia (Oxalobacteraceae). PLoS ONE 2012, 7, e40117. [Google Scholar] [CrossRef]
- Cheng, W.; Zhang, Q.; Coleman, D.C.; Carroll, R.C.; Hoffman, A. Is available carbon limiting microbial respiration in the rhizosphere? Soil Biol. Biochem. 1996, 28, 1283–1288. [Google Scholar] [CrossRef]
- Bernard, L.; Mougel, C.; Maron, P.-A.; Nowak, V.; Lévêque, J.; Henault, C.; Haichar, F.Z.; Berge, O.; Marol, C.; Balesdent, J.; et al. Dynamics and identification of soil microbial populations actively assimilating carbon from 13C-labelled wheat residue as estimated by DNA- and RNA-SIP techniques. Environ. Microbiol. 2007, 9, 752–764. [Google Scholar] [CrossRef]
- Illescas, M.; Rubio, M.B.; Hernández-Ruiz, V.; Morán-Diez, M.E.; Martínez de Alba, A.E.; Nicolás, C.; Monte, E.; Hermosa, R. Effect of Inorganic N Top Dressing and Trichoderma harzianum Seed-Inoculation on Crop Yield and the Shaping of Root Microbial Communities of Wheat Plants Cultivated Under High Basal N Fertilization. Front. Plant. Sci. 2020, 23, 575861. [Google Scholar] [CrossRef]
- Nakagawa, A.; Osawa, S.; Hirata, T.; Yamagishi, Y.; Hosoda, J.; Horikoshi, T. 2,4-Dichlorophenol Degradation by the Soil Fungus Mortierella sp. Biosci. Biotechnol. Biochem. 2006, 70, 525–527. [Google Scholar] [CrossRef] [Green Version]
- De Bruijn, I.; Cheng, X.; de Jager, V.; Exposito, R.G.; Watrous, J.; Patel, N.; Postma, J.; Dorrestein, P.C.; Kobayashi, D.; Raaijmakers, J.M. Comparative genomics and metabolic profiling of the genus Lysobacter. BMC Genom. 2015, 16, 991. [Google Scholar] [CrossRef]
- Kataoka, R.; Takagi, K.; Sakakibara, F. A new endosulfan-degrading fungus, Mortierella species, isolated from a soil contaminated with organochlorine pesticides, J. Pestic. Sci. 2010, 35, 326–332. [Google Scholar] [CrossRef] [Green Version]
- Cui, Z.; Zhang, X.; Yang, H.; Sun, L. Bioremediation of heavy metal pollution utilizing composite microbial agent of Mucor circinelloides, Actinomucor sp. and Mortierella sp. J. Environ. Chem. Eng. 2017, 5, 3616–3621. [Google Scholar] [CrossRef]
- Lambe, R.C.; Wills, W.H. Fungicides and Mortierella for the protection of azaleas from Phytophthora root rot. Phytopathology 1983, 73, 503. [Google Scholar]
- Tagawa, M.; Tamaki, H.; Manome, A.; Koyama, O.; Kamagata, Y. Isolation and characterization of antagonistic fungi against potato scab pathogens from potato field soils. FEMS Microbiol. Lett. 2010, 305, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Melo, I.S.; Santos, S.N.; Rosa, L.H.; Parma, M.M.; Silva, L.J.; Queiroz, S.C.; Pellizari, V.H. Isolation and biological activities of an endophytic Mortierella alpina strain from the Antarctic moss Schistidium antarctici. Extremophiles 2014, 18, 15–23. [Google Scholar] [CrossRef]
- Zhang, H.; Wu, X.; Li, G.; Qin, P. Interactions between arbuscular mycorrhizal fungi and phosphate-solubilizing fungus (Mortierella sp.) and their effects on Kostelelzkya virginica growth and enzyme activities of rhizosphere and bulk soils at different salinities. Biol. Fertil. Soils 2011, 47, 543. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Tian, C.; Xiao, J.; Wei, L.; Tian, Y.; Liang, Z. Soil inoculation of Trichoderma asperellum M45a regulates rhizosphere microbes and triggers watermelon resistance to Fusarium wilt. AMB Express 2020, 10, 189. [Google Scholar] [CrossRef] [PubMed]
- Tamayo-Vélez, Á.; Correa-Londoño, G.; Osorio, N.W. Inoculation with a soil fungus accelerates decomposition of avocado cv. Hass leaf litter in three plantations in Colombia. AUC Geogr. 2019, 54, 24–36. [Google Scholar] [CrossRef] [Green Version]
- Ozimek, E.; Hanaka, A. Mortierella Species as the Plant Growth-Promoting Fungi Present in the Agricultural Soils. Agriculture 2021, 11, 7. [Google Scholar] [CrossRef]
- Mouchacca, J.; Gams, W. The hyphomycete genus Cladorrhinum and its teleomorph connections. Mycotaxon 1993, 48, 415–440. [Google Scholar]
- Barrera, V.A.; Martin, M.E.; Aulicino, M.; Martinez, S.; Chiessa, G.; Saparrat, M.C.N.; Gasoni, A.L. Carbon-substrate utilization profiles by Cladorrhinum (Ascomycota). Rev. Argent. Microbiol. 2019, 51, 302–306. [Google Scholar] [CrossRef]
- Lewis, J.A.; Larkin, R.P. Formulation of the Biocontrol Fungus Cladorrhinum foecundissimum to Reduce Damping-Off Diseases Caused by Rhizoctonia solani and Pythium ultimum. Biol. Control. 1998, 12, 182–190. [Google Scholar] [CrossRef]
- Madrid, H.; Cano, J.; Gené, J.; Guarro, J. Two new species of Cladorrhinum. Mycologia 2011, 103, 795–805. [Google Scholar] [CrossRef]
- Gasoni, L.; Stegman De Gurfinkel, B. The endophyte Cladorrhinum foecundissimum in cotton roots: Phosphorus uptake and host growth. Mycol. Res. 1997, 101, 867–870. [Google Scholar] [CrossRef]
- Sagara, N. Ammonia fungi—A chemoecological grouping of terrestrial fungi. Contr. Biol. Lab. 1975, 24, 205–276. [Google Scholar]
- Nilsson, T. Studies on wood degradation and cellulolytic activity of micro fungi. Stud. For. Suec. 1973, 104, 1–40. [Google Scholar]
- Gasoni, L.; de Gurfinkel, S. Biocontrol of Rhizoctonia solani by the endophytic fungus Cladorrhinum foecundissimum in cotton plants. Australas. Plant. Pathol. 2009, 38, 389–391. [Google Scholar] [CrossRef]
- Wang, Y.; Hyde, K.D.; McKenzie, E.H.C. Overview of Stachybotrys (Memnoniella) and current species status. Fungal Divers. 2015, 71, 17–83. [Google Scholar] [CrossRef]
- Yasanthika, W.A.E.; Wanasinghe, D.N.; Karunarathna, S.C.; Bhat, D.J.; Samarakoon, S.M.B.C.; Ren, G.C.; Monkai, J.; Mortimer, P.E.; Hyde, K.D. Two new Sordariomycetes records from forest soils in Thailand. Asian J. Mycol. 2020, 3, 456–471. [Google Scholar] [CrossRef]
- Kuhn, D.M.; Ghannoum, M.A. Indoor Mold, Toxigenic Fungi, and Stachybotrys chartarum: Infectious Disease Perspective. Clin. Microbiol. Rev. 2003, 16, 144–172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hossain, M.A.; Ahmed, M.S.; Ghannoum, M.A. Attributes of Stachybotrys charatrum and its association with human disease. J. Allergy Clin. Immunol. 2004, 113, 200–208. [Google Scholar] [CrossRef]
- Sani, M.N.H.; Hasan, M.; Uddain, J.; Subramaniam, S. Impact of application of Trichoderma and biochar on growth, productivity and nutritional quality of tomato under reduced N-P-K fertilization. Ann. Agric. Sci. 2020, 65, 107–115. [Google Scholar] [CrossRef]
- Fu, J.; Xiao, Y.; Wang, Y.-F.; Liu, Z.-H.; Zhang, Y.-F.; Yang, K.-J. Trichoderma asperellum alters fungal community composition in saline-alkaline soil maize rhizospheres. Soil Sci. Soc. Am. J. 2021, 85, 1091–1104. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).