Abstract
This review article summarizes the recent progress in dental applications of natural products. Catechin gel showed selective antimicrobial activity, whereas the alkaline extract of various plant species rich in lignin carbohydrate complex (LCC) showed much higher antiviral activity than lower molecular weight polyphenols. Mouthwash with the alkaline extract of a plant classified as OTC effectively reduced halitosis. Unexpectedly, many polyphenolic compounds purified from the natural kingdom showed much lower tumor-specificity against human oral squamous cell lines as compared with antitumor agents, although they showed apoptosis-inducing activity. The alkaline extract of bamboo leaf, which exerted various common biological activities with LCC, showed osteogenic activity by stimulating differentiation toward osteoblasts while inhibiting differentiation toward osteoclasts. LCC enhanced the dectin-2 mRNA expression in macrophages, whereas glucan showed anti-osteoblastic action via dectin-1. These data suggest that natural products exert their biological activity by interacting with these molecules.
1. Introduction
It is generally accepted that many natural products effectively reduce oxidative stress and show chemoprevention activity in cell cultures and preclinical animal models [1,2,3]. However, due to inefficient systematic delivery and bioavailability, their favorable in vitro and in vivo effects are not reproducible clinically [4]. On the other hand, orally administered products directly contact the oral tissues or cells, and exert their effects without the loss of activity. There is accumulating evidence that the improvement of oral functions by periodontal treatment or the insertion of dentures and implants elevates general health and quality of life [5,6]. This article reviews the recent progress in the study of dental applications of natural products, focusing particularly on polyphenols.
2. Classification of Natural Polyphenols
Polyphenols present in the natural kingdom are roughly classified into the following three groups: tannins, flavonoids, and lignin-carbohydrate complexes (LCC) [7]. Tannins are classified into two large groups: hydrolysable tannins (in which a polyalcohol is esterified with a polyphenolic carboxylic acid such as a galloyl, hexahydroxydiphenoyl, valoneoyl, or dehydrohexahydroxydiphenoyl group) and condensed tannins (composed of flavan units, mostly catechin, epicatechin, or their analogs, condensed with each other via carbon–carbon bonds).
Flavonoids are secondary metabolites synthesized from chalcones and categorized into flavonols, flavones, flavanones, isoflavones, pterocarpan, and coumestan. Resveratrol is classified as a stilbenoid.
Lignins are formed through phenolic oxidative coupling processes. Lignin macromolecules are formed by the dehydrogenative polymerization of three monolignols: p-coumaryl, p-conifery, and sinapyl alchohols. Some polysaccharides in the cell walls of lignified plants are linked to lignin, and recovered as LCC after extraction with an alkaline solution.
3. Antibacterial Activity
The oral cavity contains nearly half of the commensal bacterial population of the human body. An increase in the number of these microorganisms may produce systemic diseases such as infective endocarditis and aspiration pneumonia as well as oral infections. In order to suppress the onset of diseases, it is important to control the total numbers of these microorganisms. Green tea catechin showed a bactericidal effect against Gram-negative anaerobic rods, and the slow-release, local delivery of catechin combined with mechanical treatment improved periodontal status [8].
In order to maintain the moistness in the oral cavity of elderly patients who require nursing care, gel-entrapped catechin (GEC) was manufactured by mixing catechins (epigallocatechin, epigallocatechin gallate, epicatechin, epicatechin gallate, gallocatechin, catechin, and gallocatechin gallate) with polysaccharide, dextrin, citric acid, potassium chloride, and stevia [9]. GEC inhibited the growth of the Actinomyces, periodontopathic bacteria, and certain tested Candida strains, possibly due to the produced hydrogen peroxide, while it did not inhibit the growth of the oral streptococci that are important in the normal oral flora [9].
5. Anticancer Activity against Oral Squamous Cell Carcinoma
5.1. Polyphenols Show Very Low Tumor-Specificity as Compared with Anticancer Drugs
Most of previous studies of polyphenols have focused on their ability to induce apoptosis in cancer cells, rather than their tumor-specificity [16,17,18]. There is a belief among researchers that apoptosis-inducing activity is a certificate of an anticancer drug. Since there is a morphological similarity between the apoptosis induced by anticancer drugs and that observed during = developmental stages (which eliminates unnecessary or harmful tissues or cells), many people have been engaged in the study of apoptosis. However, we should keep in mind that normal cells are also subjected to apoptosis by anticancer drugs.
In order to quantify the antitumor potency of polyphenols, we established an in vitro assay system for the quantification of tumor-specificity. We used four epithelial human oral squamous cell carcinoma (OSCC) lines (Ca9-22, HSC-2, HSC-3, HSC-4), three mesenchymal human normal oral cells (gingival fibroblast (HGF), periodontal ligament fibroblast (HPLF), pulp cell (HPC)), and two epithelial human normal oral cells (buccal mucosal keratinocytes (HOKs) and primary gingival epithelial cell (HGEP)) [7]. The following two different sets of cells were used: OSCC vs. normal mesenchymal normal oral cells (System 1); OSCC vs. epithelial normal oral cells (System 2). The tumor-selectivity index (TS) was determined by dividing the mean of CC50 (concentration that reduced the viable cell number by 50%) against normal cells by the mean CC50 against tumor cells. First, we calculated the TS values of anticancer drugs (positive control) using System 1. As expected, many anticancer drugs (docetaxel, 5-fluorouracil, methotrexate, mitomycin C, etoposide, daunorubicin, doxorubicin, SN-38, camptothecin, and gefitinib) showed excellent tumor-specificity (TS = 10–1000), validating this in vitro assay system. On the other hand, polyphenols (LCC, flavonoids, tannins, terpenoids, and their glycosides) (TS = 1~4.8) and antioxidants (sodium ascorbate, gallic acid, catechin, epigallocatechin gallate, chlorogenic acid, daidzein, genistein, quercetin, isoliquiritigenin, kaempferol, resveratrol, and curcumin) (TS = 1.0~4.1) showed disappointingly lower tumor-selectivity, although most of them induced apoptosis in cancer cells [10].
5.2. Induction of Keratinocyte Toxicity by Anticancer Drugs
Administration of anticancer agents has been reported to induce skin toxicity [19,20,21,22,23,24,25]. This prompted us to re-evaluate the cytotoxicity and tumor-specificity of anticancer drugs, using System 2. We demonstrated for the first time that anticancer agents such as doxorubicin, daunorubicin, etoposide, mitomycin C, 5-fluorouracil, melphalan, and gefitinib showed comparable cytotoxicity to both epithelial cancer and normal cells, producing very low TS values (TS = 0.1~1.5) [26]. We found that doxorubicin induced apoptosis (loss of cell surface microvilli, chromatin condensation, nuclear fragmentation, and caspase-3 activation) in HOKs [11]. It is therefore urgent to explore new anticancer drugs with less keratinocyte toxicity [26]
5.3. Search for New Type Antitumor Agents that Have Higher Tumor-Specificity but Lower Kereatinocyte Toxicity
Sugita’s group synthesized nine groups of compounds to search for new types of anticancer drugs that shower much lower keratinocyte toxicity [26]. Among a total of 133 compounds, (E)-3-[2-(4-hydroxyphenyl)ethenyl]-6-methoxy-4H-1-benzopyran-4-one (Compound 1; classified as 3-styrylchromones), (E)-3-[2-(4-chlorophenyl)ethenyl]-7-methoxy-2H-1-benzopyran (Compound 2; classified as 3-styryl-2H-chromenes) showed the highest tumor-specificity with the least keratinocyte toxicity (TS = 69.0 and 59.9, respectively, in System 1; TS = 204.5 and >85.1, respectively, in System 2) [27]. Compound 1 induced apoptotic cell death in a human OSCC cell line, possibly by downregulating the glycerophospholipid pathway [28]. Quantitative structure−activity relationship (QSAR) analysis demonstrated that the tumor-specificities of Compounds 1 and 2 were well correlated with chemical descriptors related to their molecular size and lipophilicity [27]. Chemical modification of these lead compounds by the introduction of appropriate functional groups is a crucial step towards manufacturing new types of anticancer drugs with reduced keratinocyte toxicity.
6. Antiosteoporotic Activity
Bone homeostasis is maintained by the balance between bone formation by osteoblasts and bone resorption by osteoclasts. When osteoclast differentiation and activation are enhanced, bone structure impairment and bone fracture occur, which are common characteristics of patients with osteoporosis, rheumatoid arthritis, and bone metastatic disease. Osteoporosis-related bone mass reduction accelerates the alveolar bone resorption caused by periodontitis [29]. We found that rhinacanthin C, a naphthoquinone ester isolated from the root and aerial part of Rhinacanthus nasutus, potently inhibited the receptor activator of nuclear factor-κB ligand (RANKL)-stimulated osteoclast formation in mouse bone marrow macrophage cultures [30] and in mouse calvarial bone in vivo [31]. Rhinacanthin C inhibited the RANKL-stimulated nuclear factor of activated T cells cytoplasmic 1 (NFATc1) expression, the phosphorylation of ERK, JNK, and NF-κB, and the formation of TRAF6-TAK1 complex [31]. These results suggest that rhinacanthin C inhibits osteoclastogenesis via suppressing RANKL-induced TRAF6-TAK1 association followed by its downstream signaling of the MAPKs/NF-κB/NFATc1 pathway (Figure 1). Rhinacanthin C also suppressed LPS-stimulated osteoclastogenesis and bone resorption [31].

Figure 1.
Action point of rhinacanthin C.
Upon RANKL treatment, mouse-macrophage-like RAW264.7 cells can be differentiated towards TRAP-positive multinuclear osteoclasts [32]. SE completely inhibited the RANKL-induced formation of TRAP-positive osteoclasts and multinuclear cells. The inhibition of mononuclear osteoclast formation was detected at 1% of SE, and complete inhibition of osteoclastogenesis was observed at 2.5% [14].
On the other hand, SE dose-dependently enhanced the alkaline phosphatase (ALP) activity, an early differentiation biomarker of osteogenesis, without significant affecting cell proliferation and cytotoxicity [33]. SE treatment of mouse osteoblastic cell line MC3T3-E1 also stimulated the expression of osteogenic-specific Runt-related transcription factor 2 (Runx2), and other osteogenic biomarkers bone sialoprotein 2 (BSP2) and collagen type 1 protein. Alizarin red and von Kossa staining demonstrated that the terminal step of osteoblast differentiation (calcification) was augmented by SE treatment. This indicated that SE promoted osteoblast differentiation and mineralization.
Taken together, these data suggest that SE reciprocally regulates the cell differentiation of bone-resorbing osteoclasts and bone-forming osteoblasts in vitro. SE may have therapeutic potential for the treatment of bone diseases such as osteoporosis.
7. Target Molecules
We previously reported that one of the seven LCC fractions isolated from LEM (Fr4) enhanced the expression of dectin-2 (4.2-fold) and toll-like receptor (TLR)-2 (2.5-fold) prominently, but only slightly modified the expression of dectin-1 (0.8-fold), complement receptor 3 (0.9-fold), TLR1, TLR3, TLR4, TLR9, and TRK13 (0.8~1.7-fold), spleen tyrosine kinase (Syk)b, zeta-chain (TCR)-associated Zap70, Jak2 (1.0~1.2-fold), NFкB1, NFкB2, RELA, RELB (1.0~1.6-fold), NFкB1A, NFкB1B, NFкB1E, NFкBIl2, and NFкBIZ (0.8~2.3-fold) [34]. Fr4 contains lignin precursors such as vanillic acid (25.9 μg/g), syringic acid (25.7 μg/g), p-coumaric acid (157.7 μg/g), and ferulic acid (13.4 μg/g) as well as a negligible amount (0.0395 μg/g) of LPS, possibly produced during extraction with NaOH [35]. However, LPS did not affect the expression of dectin-2 or TLR-2. Dectin-2 is a specific receptor for α-mannan, and plays a significant role in the regulation of the protection of the body from Candida infection via induction of the differentiation of IL-17-producing T-cells [36]. This suggests that the activation of the dectin-2 signaling pathway may play a significant role in the action of LCC on macrophages [34].
Dectin-1 was identified as a receptor for β-glucan [37]. The dectin-1 agonist curdlan inhibited osteoclastogenesis via the inhibition of NFATc1 through Syk kinase [38,39]. These data suggest that natural products exert their biological activity by interacting with these molecules.
8. Future Direction
This review demonstrated that most flavonoids have very low tumor-specificity, although they can induce apoptosis in cancer cells. Even normal keratinocytes are subjected to apoptosis upon doxorubicin treatment, suggesting that apoptosis-inducing activity itself does not guarantee antitumor potential. It is essential to determine the compounds that are highly tumor-specific, but have low keratinocyte toxicity. 4H-1-benzopyran-4-ones (chromones) are an important class of oxygenated heterocyclic compounds since the chromone core structure is found in flavones, isoflavones, and 2-styrylchromones. We found that Compound 1 (classified as 3-styrylchromones) and Compound 2 (classified as 3-styryl-2H-chromenes) fit into this category. QSAR analysis can be applied to estimate the most potent chemical structures. Synthesis of the estimated structure, reconfirmation of its activity, and repeated cycling of this process will surely yield more active compounds (Figure 2).
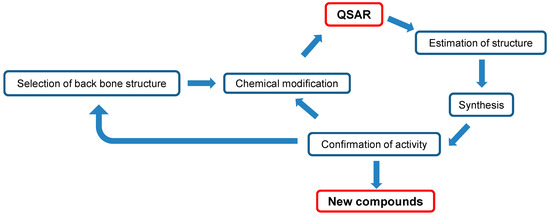
Figure 2.
Diagram of manufacturing new antitumor agents.
We recently found that SE protected amyloid β-peptide-injured neuronal cells [40] and doxorubicin-injured human keratinocytes [26], possibly by its growth-stimulating activity. Hormesis refers to the adaptive responses of biological systems to moderate environmental or self-imposed challenges through which the system improves its functionality and/or tolerance to more severe challenges [41]. Many drugs and radiations produce the stimulatory (i.e., low dose) and inhibitory (i.e., high dose) components of the hormetic dose response [42,43]. It remains to be investigated whether dectin-2 is involved in not only the growth-stimulating action of SE, but also the expression of biological actions of various groups of polyphenols.
We found that the alkaline extraction of green tea leaf, black tea leaf [44], and licorice root [45] produced higher yields of anti-HIV substances, the potency of which was comparable with that of LCC. Thus, alkaline extraction is useful for the efficient utilization of plant resources.
However, there are several problems or limitations concerning the dental applications of natural products. One is the poor absorption of higher molecular weight substances such as LCC and glucans through the stomach and intestine. Actually, we previously reported that only 6% of orally administered [125I] LCC isolated from pine cone extract appeared in the blood of mice after 3 h, and thereafter was excreted into the urine and feces [46]. Since LCC and SE (which have LCC as a major constituent) show excellent antiviral and anti-inflammatory activities, they are recommended to be directly applied to mucous membranes and periodontal tissues for longer periods of time. LCC-vitamin C tablets may be efficacious in improving the condition of HSV-1 patients [47]. On the other hand, cytotoxic lower molecular weight substances can be applicable as a mouthwash or acute treatment. Since many plant materials may be contaminated with LPS from soil bacteria (usually ranging from 10.4 to 18.8 ng/g, sometimes >200 ng/g) [48], extensive washing before extraction and purification is needed.
Acknowledgments
This work was partially supported by KAKENHI from the Japan Society for the Promotion of Science (JSPS): Sakagami H, 16K11519; Tomomura M, 25462898.
Author Contributions
H.S. and M.T. designed and performed the experiments, analyzed the data, and wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Mao, X.; Gu, C.; Chen, D.; Yu, B.; He, J. Oxidative stress-induced diseases and tea polyphenols. Oncotarget 2017, 8, 81649–81661. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Suh, J.; Surh, Y.J.; Na, H.K. Regulation of the tumor suppressor PTEN by natural anticancer compounds. Ann. N. Y. Acad. Sci. 2017, 1401, 136–149. [Google Scholar] [CrossRef] [PubMed]
- Castaneda, O.A.; Lee, S.C.; Ho, C.T.; Huang, T.C. Macrophages in oxidative stress and models to evaluate the antioxidant function of dietary natural compounds. J. Food Drug Anal. 2017, 25, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, I.A.; Sanna, V. Impact of nanotechnology on the delivery of natural products for cancer prevention and therapy. Mol. Nutr. Food Res. 2016, 60, 1330–1341. [Google Scholar] [CrossRef] [PubMed]
- Olsen, I.; Singhrao, S.K. Can oral infection be a risk factor for Alzheimer’s disease? J. Oral Microbiol. 2015, 7, 29143. [Google Scholar] [CrossRef] [PubMed]
- Furuta, M.; Yamashita, Y. Oral Health and Swallowing Problems. Curr. Phys. Med. Rehabil. Rep. 2013, 1, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Sakagami, H. Biological activities and possible dental application of three major groups of polyphenols. J. Pharmacol. Sci. 2014, 126, 92–106. [Google Scholar] [CrossRef] [PubMed]
- Hirasawa, M.; Takada, K.; Makimura, M.; Otake, S. Improvement of periodontal status by green tea catechin using a local delivery system: A clinical pilot study. J. Periodontal Res. 2002, 37, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Tamura, M.; Saito, H.; Kikuchi, K.; Ishigami, T.; Toyama, Y.; Takami, M.; Ochiai, K. Antimicrobial activity of Gel-entrapped catechins toward oral microorganisms. Biol. Pharm l. Bull. 2011, 34, 638–643, ISSN: 1347-5215. [Google Scholar] [CrossRef]
- Sakagami, H.; Sheng, H.; Yasui, T.; Fukuchi, K.; Oizumi, T.; Ohno, H.; Yamamoto, M.; Fukuda, T.; Kotohda, K.; Yoshida, H.; et al. Therapeutic potential of solubilized nanolignin against oral diseases. In Nanostructures for Oral Medicicne; Elsevier: Amsterdam, The Netherlands, 2017; pp. 545–576, Chapter 18; ISBN 978-0-323-47720-8. [Google Scholar]
- Sakagami, H.; Kushida, T.; Oizumi, T.; Nakashima, H.; Makino, T. Distribution of lignin-carbohydrate complex in plant kingdom and its functionality as alternative medicine. Pharmacol. Ther. 2010, 128, 91–105. [Google Scholar] [CrossRef] [PubMed]
- Sakagami, H.; Amano, S.; Yasui, T.; Satoh, K.; Shioda, S.; Kanamoto, T.; Terakubo, S.; Nakashima, H.; Watanabe, K.; Sugiura, T.; et al. Biological interaction between Sasa senanensis Rehder leaf extract and toothpaste ingredients. In Vivo 2013, 27, 275–284. [Google Scholar] [PubMed]
- Sakagami, H.; Fukuchi, K.; Kanamoto, T.; Terakubo, S.; Nakashima, H.; Natori, T.; Suguro-Kitajima, M.; Oizumi, H.; Yasui, T.; Oizumi, T. Synergism of alkaline extract of the leaves of Sasa senanensis Rehder and antiviral agents. In Vivo 2016, 30, 421–426. [Google Scholar] [PubMed]
- Matsuta, T.; Sakagami, H.; Tanaka, S.; Machino, M.; Tomomura, M.; Tomomura, A.; Yasui, T.; Itoh, K.; Sugiura, T.; Kitajima, M.; et al. Pilot clinical study of Sasa senanensis Rehder leaf extract treatment on lichenoid dysplasia. In Vivo 2012, 26, 957–962. [Google Scholar] [PubMed]
- Sakagami, H.; Sheng, H.; Ono, K.; Komine, Y.; Miyadai, T.; Terada, Y.; Nakada, D.; Tanaka, S.; Matsumoto, M.; Yasui, T.; et al. Anti-halitosis effect of toothpaste supplemented with alkaline extract of the leaves of Sasa senanensis Rehder. In Vivo 2016, 30, 107–111. [Google Scholar] [PubMed]
- Losada-Echeberría, M.; Herranz-López, M.; Micol, V.; Barrajón-Catalán, E. Polyphenols as Promising Drugs against Main Breast Cancer Signatures. Antioxidants (Basel) 2017, 6, 88. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kaur, M.; Katnoria, J.K.; Nagpal, A.K. Polyphenols in Food: Cancer Prevention and Apoptosis Induction. Curr. Med. Chem. 2017. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.H.; Sethi, G.; Um, J.Y.; Shanmugam, M.K.; Arfuso, F.; Kumar, A.P.; Bishayee, A.; Ahn, K.S. The Role of Resveratrol in Cancer Therapy. Int. J. Mol. Sci. 2017, 18, 2589. [Google Scholar] [CrossRef] [PubMed]
- Lulli, D.; Carbone, M.L.; Pastore, S. Epidermal growth factor receptor inhibitors trigger a type I interferon response in human skin. Oncotarget 2016, 7, 47777–47793. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, D.; Codecà, C.; Bocci, B.; Crepaldi, F.; Violati, M.; Viale, G.; Careri, C.; Caldiera, S.; Bordin, V.; Luciani, A.; et al. Anti-epidermal growth factor receptor skin toxicity: A matter of topical hydration. Anticancer Drugs 2016, 27, 144–146. [Google Scholar] [CrossRef] [PubMed]
- Benjakul, R.; Kongkaneramit, L.; Sarisuta, N.; Moongkarndi, P.; Müller-Goymann, C.C. Cytotoxic effect and mechanism inducing cell death of α-mangostin liposomes in various human carcinoma and normal cells. Anticancer Drugs 2015, 26, 824–834. [Google Scholar] [CrossRef] [PubMed]
- Do, N.; Weindl, G.; Grohmann, L.; Salwiczek, M.; Koksch, B.; Korting, H.C.; Schäfer-Korting, M. Cationic membrane-active peptides–anticancer and antifungal activity as well as penetration into human skin. Exp. Dermatol. 2014, 23, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Moreno Garcia, V.; Thavasu, P.; Blanco Codesido, M.; Molife, L.R.; Vitfell Pedersen, J.; Puglisi, M.; Basu, B.; Shah, K.; Iqbal, J.; de Bono, J.S.; et al. Association of creatine kinase and skin toxicity in phase I trials of anticancer agents. Br. J. Cancer 2012, 107, 1797–1800. [Google Scholar] [CrossRef] [PubMed]
- Benedict, A.L.; Knatko, E.V.; Dinkova-Kostova, A.T. The indirect antioxidant sulforaphane protects against thiopurine-mediated photo-oxidative stress. Carcinogenesis 2012, 33, 2457–2466. [Google Scholar] [CrossRef] [PubMed]
- Fischel, J.L.; Formento, P.; Ciccolini, J.; Etienne-Grimaldi, M.C.; Milano, G. Lack of contribution of dihydrofluorouracil and alpha-fluoro-beta-alanine to the cytotoxicity of 5′-deoxy-5-fluorouridine on human keratinocytes. Anticancer Drugs 2004, 15, 969–974. [Google Scholar] [CrossRef] [PubMed]
- Sakagami, H.; Okudaira, N.; Masuda, Y.; Amano, O.; Yokose, S.; Kanda, Y.; Suguro, M.; Natori, T.; Oizumi, H.; Oizumi, T. Induction of apoptosis in human oral keratinocyte by doxorubicin. Anticancer Res. 2017, 37, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Sugita, Y.; Takao, K.; Uesawa, Y.; Sakagami, H. Search for new type of anticancer drugs with high tumor-specificity and less keratinocyte toxicity (Review). Anticancer Res. 2017, 37, 5919–5924. [Google Scholar] [CrossRef] [PubMed]
- Sakagami, H.; Shimada, C.; Kanda, Y.; Amano, O.; Sugimoto, M.; Ota, S.; Soga, T.; Tomita, M.; Sato, A.; Tanuma, S.; et al. Effects of 3-styrylchromones on metabolic profiles and cell death in oral squamous cell carcinoma cells. Toxocol. Rep. 2015, 2, 1281–1290. [Google Scholar] [CrossRef] [PubMed]
- Von Wowern, N.; Klausen, B.; Kollerup, G. Osteoporosis: A risk factor in periodontal disease. J. Periodontol. 1994, 65, 1134–1138. [Google Scholar] [CrossRef] [PubMed]
- Horii, H.; Suzuki, R.; Sakagami, H.; Tomomura, M.; Tomomura, A.; Shirataki, Y. New biological activities of Rhinacanthins from the root of Rhinacanthus nasutus. Anticancer Res. 2013, 33, 453–459. [Google Scholar] [PubMed]
- Tomomura, M.; Suzuki, R.; Shirataki, Y.; Sakagami, H.; Tomomura, A. Rhinacanthin C inhibits RANK ligand-induced osteoclast differentiation by preventing TRAF6-TAK1 formation and MAPK/NF-kappaB/NFATc1 pathways. PLoS ONE 2015, 10, e0130174. [Google Scholar] [CrossRef]
- Ishida, N.; Hayashi, K.; Hoshijima, M.; Ogawa, T.; Koga, S.; Miyatake, Y.; Kumegawa, M.; Kimura, T.; Takeya, T. Large-scale gene expression analysis of osteoclastogenesis in vitro and elucidation of NFAT2 as a key regulator. J. Biol. Chem. 2002, 277, 41147–41156. [Google Scholar] [CrossRef] [PubMed]
- Tomomura, M.; Tomomura, A.; Oizumi, T.; Yasui, T.; Sakagami, H. Extract of Sasa senanensis Rehder Leaf Promotes Osteoblast Differentiation in MC3T3-E1 cells. J. Meikai Dent. Med. 2017, 46, 111–116. [Google Scholar]
- Kushida, T.; Makino, T.; Tomomura, M.; Tomomura, A.; Sakagami, H. Enhancement of dectin-2 gene expression by lignin-carbohydrate complex from Lendinus edodes extract (LEM) in mouse macrophage-like cell line. Anticancer Res. 2011, 31, 1241–1248. [Google Scholar] [PubMed]
- Kawano, M.; Thet, M.M.; Makino, T.; Kushida, T.; Sakagami, H. DNA microarray analysis of signaling pathway in macrophages stimulated by lignin-carbohydrate complex from lentinus edodes mycelia (LEM) extract. Anticancer Res. 2010, 30, 2567–2576. [Google Scholar] [PubMed]
- Saijo, S.; Ikeda, S.; Yamabe, K.; Kakuta, S.; Ishigame, H.; Akitsu, A.; Fujikado, N.; Kusaka, T.; Kubo, S.; Chung, S.H.; et al. Dectin-2 recognition of alpha-mannans and induction of Th17 cell differentiation is essential for host defense against Candida albicans. Immunity 2010, 32, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.R.; Tsoni, S.V.; Willment, J.A.; Dennehy, K.M.; Rosas, M.; Findon, H.; Haynes, K.; Steele, C.; Botto, M.; Gordon, S.; et al. Dectin-1 is required for β-glucan recognition and control of fungal infection. Nat. Immunol. 2007, 8, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, T.; Ariyoshi, W.; Okinaga, T.; Adachi, Y.; Hosokawa, R.; Mochizuki, S.; Sakurai, K.; Nishihara, T. The dectin 1 agonist curdlan regulates osteoclastogenesis by inhibiting nuclear factor of activated T cells cytoplasmic 1 (NFATc1) through Syk kinase. J. Biol. Chem. 2014, 289, 9191–9203. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Zhao, Y.; Jiang, Y.; Qin, T.; Chen, J.; Chu, X.; Yi, Q.; Gao, S.; Wang, S. Dectin-1 signaling inhibits osteoclastogenesis via IL-33-induced inhibition of NFATc1. Oncotarget 2017, 8, 53366–53374. [Google Scholar] [CrossRef] [PubMed]
- Sakagami, H.; Tsuji, M.; Tomomura, M.; Masuda, Y.; Iwama, S.; Nakagawa, M.; Suzuki, H.; Tanaka, K.; Abe, T.; Tamura, N.; et al. Protection of differentiating neuronal cells from amyloid β-peptide-induced injury by alkaline extract of leaves of Sasa senanensis Rehder. In Vivo 2018, 32. in press. [Google Scholar]
- Calabrese, E.J.; Mattson, M.P. How does hormesis impact biology, toxicology, and medicine? NPJ Aging Mech. Dis. 2017, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Calabrese, E.J.; Lian, B.; Lin, Z.; Calabrese, V. Hormesis as a mechanistic approach to understanding herbal treatments in traditional Chinese medicine. Pharmacol. Ther. 2017. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Dhawan, G.; Kapoor, R.; Iavicoli, I.; Calabrese, V. What is hormesis and its relevance to healthy aging and longevity? Biogerontology 2015, 16, 693–707. [Google Scholar] [CrossRef] [PubMed]
- Sakagami, H.; Ohkoshi, E.; Amano, S.; Satoh, K.; Kanamoto, T.; Terakubo, S.; Nakashima, H.; Sunaga, K.; Otsuki, T.; Ikeda, H.; et al. Efficient utilization of plant resources by alkaline extraction. Altern. Integr. Med. 2013, 2. [Google Scholar] [CrossRef]
- Ohno, H.; Miyoshi, S.; Araho, D.; Kanamoato, T.; Terakubo, S.; Nakashima, H.; Tsuda, T.; Sunaga, K.; Amano, S.; Ohkoshi, E.; et al. Efficient utilization of licorice root by alkaline extraction. In Vivo 2014, 28, 785–794. [Google Scholar] [PubMed]
- Sakagami, H.; Asano, K.; Yoshida, T.; Kawazoe, Y. Organ distribution and toxicity of lignin. In Vivo 1999, 13, 41–44. [Google Scholar] [PubMed]
- Lopez, B.S.; Yamamoto, M.; Utsumi, K.; Aratsu, C.; Sakagami, H. A clinical pilot study of lignin–ascorbic acid combination treatment of herpes simplex virus. In Vivo 2009, 23, 1011–1016. [Google Scholar] [PubMed]
- Kato, T.; Segami, N.; Sakagami, H. Anti-inflammatory activity of Hangeshashinto in IL-1β-stimulated gingival and periodontal ligament fibroblasts. In Vivo 2016, 30, 257–263. [Google Scholar] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).