Journal Description
Medicines
Medicines
is an international, peer-reviewed, open access journal on drug discovery and clinical application published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within PubMed, PMC, CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 27.7 days after submission; acceptance to publication is undertaken in 7.2 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Medicines is a companion journal of Pharmaceutics.
Latest Articles
The Efficacy of Fixed-Dose Diclofenac and Orphenadrine for Postoperative Pain Management: A Systematic Review
Medicines 2026, 13(2), 17; https://doi.org/10.3390/medicines13020017 - 8 May 2026
Abstract
Background/Objectives: Postoperative pain remains a significant clinical challenge, often requiring multimodal strategies to mitigate opioid-related adverse events. The fixed-dose combination (FDC) of Diclofenac, a non-steroidal anti-inflammatory drug, and Orphenadrine, a muscle relaxant, targets distinct nociceptive pathways to potentially enhance analgesia and reduce opioid
[...] Read more.
Background/Objectives: Postoperative pain remains a significant clinical challenge, often requiring multimodal strategies to mitigate opioid-related adverse events. The fixed-dose combination (FDC) of Diclofenac, a non-steroidal anti-inflammatory drug, and Orphenadrine, a muscle relaxant, targets distinct nociceptive pathways to potentially enhance analgesia and reduce opioid consumption. This systematic review aims to evaluate the analgesic efficacy and safety profile of the fixed-dose combination of Diclofenac and Orphenadrine for postoperative pain management and quantify its opioid-sparing effect compared to standard monotherapies or placebo. Methods: A systematic search of electronic databases (MEDLINE, Scopus) and clinical trial registries (including ClinicalTrials.gov and CTIS) was conducted up to 20 September 2025. Fourteen (14) randomized controlled trials (RCTs) involving 981 adult patients undergoing various surgical procedures were included. Due to high clinical and methodological heterogeneity, a Synthesis Without Meta-analysis (SWiM) approach was utilized. The certainty of evidence was assessed using the GRADE methodology. Results: The synthesis demonstrated that the FDC may improve pain relief (measured by the Visual Analog Scale and Numeric Rating Scale scores) and may reduce opioid consumption compared to active comparators and placebo. The opioid-sparing effect could be correlated with a reduced incidence of dose-dependent adverse events, particularly nausea and vomiting. However, the overall certainty of the evidence was graded as “Very Low” due to the high risk of bias and lack of transparency in the included studies. Conclusions: The FDC of Diclofenac and Orphenadrine is a rational addition to multimodal postoperative analgesic regimens, which may potentially reduce the perioperative opioid burden without compromising pain control. Nevertheless, because almost all included studies suffer from severe methodological flaws, these apparent efficacy findings must be interpreted with caution. Future high-quality, pre-registered, and low-bias randomized controlled trials are required to draw firm clinical conclusions.
Full article
Open AccessReview
Sulforaphane Synergies with Phytochemicals and Pharmaceuticals: Implications for Healthspan
by
Jed W. Fahey and Hua Liu
Medicines 2026, 13(2), 16; https://doi.org/10.3390/medicines13020016 - 6 May 2026
Cited by 1
Abstract
►▼
Show Figures
Sulforaphane, a bioactive isothiocyanate found abundantly in cruciferous vegetables, has attracted significant attention for its chemopreventive and therapeutic potential, particularly in cancer. There is now an abundance of peer-reviewed research documenting true synergies between sulforaphane and (a) cancer treatment drugs, (b) pharmaceuticals in
[...] Read more.
Sulforaphane, a bioactive isothiocyanate found abundantly in cruciferous vegetables, has attracted significant attention for its chemopreventive and therapeutic potential, particularly in cancer. There is now an abundance of peer-reviewed research documenting true synergies between sulforaphane and (a) cancer treatment drugs, (b) pharmaceuticals in development but not yet on the market or in the regulatory pipeline, (c) other phytochemicals, and (d) proprietary mixtures such as leaf extracts and other botanicals, as well as evidence that some cell lines resistant to various cancer drugs become more susceptible when treated with sulforaphane. Most of the published studies demonstrate evidence for synergy in cancer, including cancers of the bladder, blood, brain, breast, colon, esophagus, liver, lung, ovaries, prostate, and skin, where reducing drug dosages could yield substantial patient benefits. Importantly, non-cancer indications have also been reported, such as mitigation of cardiac toxicity, inflammation, obesity, and pain (including antihyperalgesic and antinociceptive effects). Synergistic effects are most often demonstrated in cell line models, with many studies providing robust mechanistic evidence, and some employing the gold-standard Chou–Talalay method for quantifying synergy. Current evidence on the synergistic interactions of sulforaphane with both phytochemicals and pharmaceuticals highlights underlying mechanisms such as modulation of oxidative stress, inflammation, apoptosis, and epigenetic regulation, suggesting significant clinical and therapeutic implications. By providing a comprehensive overview of sulforaphane synergies in both cancer and non-cancer contexts, we aim to inform future research and support the development of integrated therapeutic strategies.
Full article
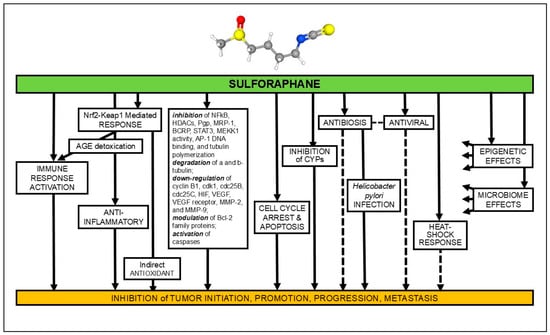
Figure 1
Open AccessReview
Evidence and Tradition in Dialogue: Biological Sex Variability in Phytomedicine Research as a Foundation for Safety, Efficacy, and Robust Evidence Standards
by
Helen Turner, Chad Jansen, Beverly G. Rice, Tiffany Rivera, Julia Howard, Catherine Brockway, Bianca Parisi, Chaker Adra, Andrea Small-Howard and Alexander J. Stokes
Medicines 2026, 13(2), 15; https://doi.org/10.3390/medicines13020015 - 7 Apr 2026
Abstract
►▼
Show Figures
Background: Incorporating sex as a biological variable (SBV) is recognized as essential for improving the reliability, reproducibility, and generalizability of pharmacological research. This principle is codified in international policies and guidelines, yet implementation remains uneven, especially in phytomedicine. Phytomedicines are a major component
[...] Read more.
Background: Incorporating sex as a biological variable (SBV) is recognized as essential for improving the reliability, reproducibility, and generalizability of pharmacological research. This principle is codified in international policies and guidelines, yet implementation remains uneven, especially in phytomedicine. Phytomedicines are a major component of healthcare worldwide, with 65% of the global population relying on them in both regulated and traditional contexts. Globally, phytomedicines are used by males, females, intersex and non-cis gender persons, all of whom may present specific safety and efficacy considerations and warrant full inclusion in pre-clinical to clinical research pipelines. However, in contemporary settings, phytomedicine lags in SBV best practices relative to Western allopathic standards for research design. Methods: We conducted a non-systematic review and in silico data mining to quantify sex/gender representation in recent preclinical and clinical phytomedicine studies, complemented by targeted case studies of sexually dimorphic safety/efficacy. We also summarize the historical role of women and gender-diverse people as users and providers within Traditional and Integrative Medical Systems (TIMSs). Results: Across rodent and human studies, females are under-represented relative to males, and sex is rarely reported for cell lines. Intentional inclusion of intersex and other gender-diverse populations is largely absent. Case studies illustrate plausible sex-associated differences in pharmacokinetics, pharmacodynamics, and adverse event profiles. TIMSs historically address women’s health needs and include substantial participation by female practitioners; however, contemporary SBV practices remain less standardized than in Western allopathic pipelines. Conclusions: SBV integration in phytomedicine is needed to strengthen safety, efficacy, and regulatory-grade evidence. Practical barriers include legacy datasets without sex metadata, limited intersex animal models, and uneven resources across settings. We outline feasible, stepwise practices to improve SBV adoption in a manner compatible with TIMS contexts and recommend expanding current guidelines to better support diverse research environments while maintaining scientific rigor.
Full article

Figure 1
Open AccessArticle
Anticholinergic Burden in Elderly People in Nursing Homes: Cross-Sectional Assessment Using ACB Calculator and CRIDECO Anticholinergic Load Scale
by
Tânia Nascimento, Maria Ana Matos and Ezequiel Pinto
Medicines 2026, 13(2), 14; https://doi.org/10.3390/medicines13020014 - 7 Apr 2026
Abstract
Background/Objectives: Anticholinergic burden is an important risk marker in older adults, associated with cognitive decline, falls, and increased mortality. This study aimed to assess anticholinergic burden in institutionalized elderly individuals using two tools (ACB calculator and CALS—CRIDECO Anticholinergic Load Scale), as well as
[...] Read more.
Background/Objectives: Anticholinergic burden is an important risk marker in older adults, associated with cognitive decline, falls, and increased mortality. This study aimed to assess anticholinergic burden in institutionalized elderly individuals using two tools (ACB calculator and CALS—CRIDECO Anticholinergic Load Scale), as well as to analyze its relationship with pharmacotherapeutic variables like polypharmacy. Methods: A descriptive cross-sectional study was conducted by analyzing the pharmacotherapeutic profiles of institutionalized elderly individuals (≥65 years) utilizing individualized medication preparation services from a community pharmacy in Alentejo (Portugal). Participants agreed to the study and had complete, up-to-date pharmacotherapeutic profiles. Results: The pharmacotherapeutic profiles of 75 institutionalized elderly people were analyzed; the sample comprised mostly women (72%) who had experienced excessive polypharmacy (≥10 medications) (56%) and had an average age of 85.62 ± 7.62 years. It was found that 90.7% (ACB) and 89.3% (CALS—CRIDECO) of the elderly had anticholinergic burden, with mean values of 3.60 ± 2.84 and 3.33 ± 2.51, respectively. Women exhibited higher anticholinergic burden in unadjusted analyses (p < 0.05). The burden correlated moderately with the total number of medications (p < 0.05). Conclusions: The results show high exposure to anticholinergic medications in the institutionalized elderly population, reinforcing the rationale for systematic therapeutic reviews focused on the pharmacological safety of institutionalized older adults in community pharmacies.
Full article
(This article belongs to the Special Issue Drug-Related Problems: Clinical Pharmacy and Safety of Pharmacotherapy)
►▼
Show Figures

Figure 1
Open AccessArticle
The Effect of Oxcarbazepine on the Pharmacokinetics of Single-Dose Aripiprazole in a Rat Model
by
Iulia-Maria Ciocotișan, Dana Maria Muntean and Laurian Vlase
Medicines 2026, 13(2), 13; https://doi.org/10.3390/medicines13020013 - 1 Apr 2026
Abstract
►▼
Show Figures
Background/Objectives: A possible pharmacokinetic interaction between a single dose of aripiprazole and multiple-dose pretreatment with oxcarbazepine was investigated in vivo in Wistar albino rats. Methods: The experiment was conducted on two groups of 12 male rats each. The control group received
[...] Read more.
Background/Objectives: A possible pharmacokinetic interaction between a single dose of aripiprazole and multiple-dose pretreatment with oxcarbazepine was investigated in vivo in Wistar albino rats. Methods: The experiment was conducted on two groups of 12 male rats each. The control group received a single oral dose of aripiprazole (8 mg/kg), while the test group was given oral oxcarbazepine (85 mg/kg/day) for 5 days, followed by a single oral dose of aripiprazole (8 mg/kg). Blood samples were automatically drawn following the administration of aripiprazole to each rat. Noncompartmental analysis was employed to determine the pharmacokinetic parameters of aripiprazole and its active metabolite, dehydroaripiprazole. Results: After the five-day oxcarbazepine pretreatment, aripiprazole’s maximum plasma concentration decreased by 51.37%, and its mean half-life was significantly reduced by 1.51-fold. In contrast, for the metabolite, the mean total area under the concentration–time curve increased by 44.66%, and the mean apparent systemic clearance decreased by 61.84%. Conclusions: Multiple-dose pretreatment with oxcarbazepine resulted in significant changes in the pharmacokinetics of a single oral dose of aripiprazole and its active metabolite in vivo in rats. The clinical implications should be further studied in human subjects, as this interaction may reduce the efficacy of aripiprazole.
Full article
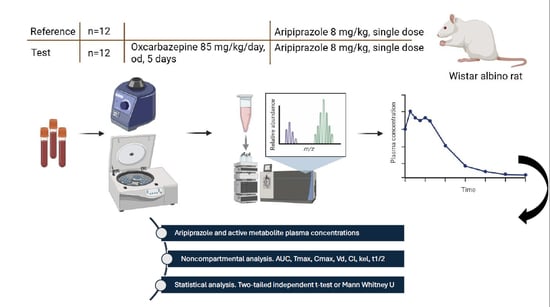
Graphical abstract
Open AccessArticle
Quantification of In Vitro Replicative Lifespan Elongation Activity of Hormones, Antioxidants, Plant Extract and Bacterial Exudate by Updated “Overlay Method”
by
Hiroshi Sakagami, Masayo Abe, Megumi Inomata, Hideki Aoyagi, Takao Tsukahara, Kenjiro Bandow, Shogo Nishino, Hiroshi Kadokura, Yuka Kato and Satoshi Yokose
Medicines 2026, 13(2), 12; https://doi.org/10.3390/medicines13020012 - 30 Mar 2026
Abstract
Background/Objectives: Many products that claim to have anti-aging effects have been reported, but their relative potency is not clear. In this study, the in vitro replicative lifespan extension (RLE) activity of various groups of physiologically active substances was compared by using the
[...] Read more.
Background/Objectives: Many products that claim to have anti-aging effects have been reported, but their relative potency is not clear. In this study, the in vitro replicative lifespan extension (RLE) activity of various groups of physiologically active substances was compared by using the updated “overlay method”. Methods: Human dermal and periodontal ligament fibroblasts (HDFa, HPLF) were inoculated into the inner 60 wells of 96-well microplate, surround by sterile water to prevent the water evaporation. At Day 1 and Day 8, the cells were overlayed with wide ranges of concentrations (0.01–100 µM) of samples without medium change. Viable cell number was measured by the MTT method at Day 15 and then corrected for the variation in cell growth due to the location of inoculated cells. The RLE value was calculated as the maximum cell proliferation rate relative to the control. Results: Cell density of HDFa and HPLFs at subculture decreased with the passage number, and their growth was stopped at 56 or 85 population doubling levels (PDLs), respectively. Hydrocortisone showed the highest RLE values among six hormones, followed by three plant extracts, sodium ascorbate and quercetin. On the other hand, other antioxidants, chlorogenic acid, phenylpropanoids, vanilloids, and bacterial products showed little or no RLE effects. However, for HPLF cells, hydrocortisone did not show RLE effects while oxytocin showed slight stimulation. Conclusions: When differences in proliferation due to cell seeding position were corrected, the biphasic dose response curve of most of the compounds significantly reduced. The present study suggests the significant role of hormones for the regulation of the long-term aging process. To confirm systemic or clinical anti-aging effects, further in vitro and in vivo experiments are needed.
Full article
(This article belongs to the Topic Research in Pharmacological Therapies, 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessReview
Multi-Targeted Mechanisms of Phytochemicals in Mitigating Cadmium-Induced Breast Cancer
by
Fidara F. Fidudusola, Caroline O. Odewumi, Lekan M. Latinwo, Oluwatobi A. Oguntunde, Samia S. Messeha and Karam F. A. Soliman
Medicines 2026, 13(2), 11; https://doi.org/10.3390/medicines13020011 - 24 Mar 2026
Abstract
Cadmium (Cd) is an environmental toxicant originating from both natural processes and human activities. Cd has been strongly associated with multiple diseases, including breast cancer (BC). Background/Objective: Environmental Cd exposure represents a significant contributor to BC onset and progression. Cd-induced breast carcinogenesis
[...] Read more.
Cadmium (Cd) is an environmental toxicant originating from both natural processes and human activities. Cd has been strongly associated with multiple diseases, including breast cancer (BC). Background/Objective: Environmental Cd exposure represents a significant contributor to BC onset and progression. Cd-induced breast carcinogenesis is driven by a constellation of molecular events, including DNA damage, oxidative stress (OS), and the dysregulation of key signaling pathways. These include the ERK/JNK/p38 MAPK cascade, the PI3K/AKT/mTOR axis, NF κB activation, and Wnt signaling, all of which collectively promote tumor initiation, survival, and metastasis. This review underscores the complex interplay between Cd exposure and its effects on cancer-triggering factors. Methods: The complexity of the mechanisms Cd-induced BC, underlying Cd-induced BC makes it challenging to treat, highlighting the need for novel therapeutic strategies that complement or enhance conventional chemotherapy. Therefore, this review was developed by reviewing the literature and presenting the different aspects of the challenge associated with Cd exposure and BC therapy. Results: Phytochemicals, especially phenolics, alkaloids, carotenoids, terpenoids, and related plant-derived compounds, have emerged as promising candidates for mitigating Cd-induced BC. Their antioxidants, anti-estrogenic, and anti-inflammatory properties position them as potential chemopreventive and therapeutic agents capable of counteracting Cd’s molecular toxicity. Conclusions: The review presents current evidence linking Cd exposure to BC development and highlights the protective potential of selected phytochemicals in preventing or attenuating Cd-induced BC. Understanding these interactions reinforces the importance of phytochemical-based interventions as a strategy to reduce Cd-related cancer risk and support breast health.
Full article
(This article belongs to the Topic New Compounds Discovery and Development in Medicine — Advances in Research on Potential Therapeutic Agents and Drug Candidates, 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessReview
Exploring the Pleiotropic Cardioprotective Effects of GLP-1 Receptor Agonists in Preventing Anthracycline-Induced Cardiotoxicity: A Theoretical Proposal for Future Research
by
Matthew L. Repp, Ikeotunye Royal Chinyere, Santiago Teran, Julia Bast and Lavanya Kondapalli
Medicines 2026, 13(1), 10; https://doi.org/10.3390/medicines13010010 - 17 Mar 2026
Abstract
►▼
Show Figures
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) have been shown to reduce morbidity and mortality associated with type II diabetes mellitus, and/or obesity, and/or cardiovascular disease in multiple clinical trials. Their efficacy in reversing cardiovascular disease and mitigating the risk of major adverse cardiac
[...] Read more.
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) have been shown to reduce morbidity and mortality associated with type II diabetes mellitus, and/or obesity, and/or cardiovascular disease in multiple clinical trials. Their efficacy in reversing cardiovascular disease and mitigating the risk of major adverse cardiac and vascular events has been well studied, with outcome trials consistently demonstrating benefits such as reduced systemic inflammation, improved endothelial function, and favorable metabolic effects. These pleiotropic actions have nearly innumerable potential applications, with a progressively growing interest in using GLP-1 RAs to mitigate increased cardiovascular disease risk secondary to other off-target pharmacologic agents. Given these effects, the potential to utilize GLP-1 RAs for prophylactic cardioprotection before, during, and/or after chemotherapy regimens is of great interest. These effects are thought to be mediated in part through anti-inflammatory and antioxidant mechanisms that counter inflammation and reactive oxygen species-driven myocardial injury central to anthracycline-induced cardiotoxicity (AIC). Anthracyclines, a widely used class of chemotherapeutics for various malignancies, are frequently associated with dose-dependent and often irreversible cardiotoxicity, leading to heart failure, reduced quality of life, and adverse long-term outcomes. For the past three decades, dexrazoxane has been the sole Food and Drug Administration-approved agent for cardioprotection in this setting. However, in the current era of novel therapies with multi-system benefits—such as GLP-1 RAs—we propose a theoretical framework exploring their potential role in mitigating AIC and underscore the need for further clinical investigation in this new arena in the field of cardio-oncology.
Full article

Figure 1
Open AccessReview
Advances in Extracellular Matrix Metalloproteinases: Implications for Renal Cell Carcinoma Pathophysiology, Diagnostics, and Therapeutics
by
Evangelia Krikou, Ioanna A. Anastasiou, Panagiotis Sarantis, Hariklia Gakiopoulou, Irini Theochari, Dimitra Grigoriadou, Andreas C. Lazaris and Eleftheria Lakiotaki
Medicines 2026, 13(1), 9; https://doi.org/10.3390/medicines13010009 - 3 Mar 2026
Abstract
Renal cell carcinoma (RCC) is a common tumor that heavily depends on extracellular matrix (ECM) remodeling, an essential process involved not only in normal tissue homeostasis but also in malignant growth. This article reviews the role of matrix metalloproteinases (MMPs, zinc-dependent endopeptidases) in
[...] Read more.
Renal cell carcinoma (RCC) is a common tumor that heavily depends on extracellular matrix (ECM) remodeling, an essential process involved not only in normal tissue homeostasis but also in malignant growth. This article reviews the role of matrix metalloproteinases (MMPs, zinc-dependent endopeptidases) in matrix degradation and ECM reorganization in the setting of RCC. We focus on the specific role of MMP2, MMP7, and MMP9 in clear cell renal cell carcinoma (ccRCC) and major subtypes of RCC. Higher levels of these MMPs are associated with high-grade tumors, increased risk of metastasis, and poorer patient survival rates, indicating that they may have value as prognostic markers. This review also discusses how ECM composition and structure are altered in the tumor microenvironment (TME), thereby preventing cell interactions and promoting cancer growth. Finally, it compiles the existing studies to anticipate a future era in which MMPs could serve as effective prognostic biomarkers and potential treatment targets for RCC, with implications for improving diagnostic and therapeutic interventions targeting ECM remodeling to suppress cancer progression.
Full article
(This article belongs to the Section Cancer Biology and Anticancer Therapeutics)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Sulforaphane Pre-Treatment Improves Alveolar Macrophage Killing After Alcohol-Induced Phagocytic Dysfunction In Vitro and in Galleria mellonella Larvae
by
Caleb Harrop, Nathan Clark, Robert Darby, Dallen James, Scott Quimby, Braydon Black, Vincent Tran, Ethan Ostrom, Tinna Traustadóttir, Fernando P. Monroy and Victor M. Jimenez, Jr.
Medicines 2026, 13(1), 8; https://doi.org/10.3390/medicines13010008 - 19 Feb 2026
Abstract
Background: Alcohol is associated with increased mortality and morbidity globally. Pulmonary infections with opportunistic pathogens can occur in healthy humans; however, binge alcohol intoxication (≥0.08% BAC) is a major risk factor. We have previously shown that a single dose of alcohol comparable to
[...] Read more.
Background: Alcohol is associated with increased mortality and morbidity globally. Pulmonary infections with opportunistic pathogens can occur in healthy humans; however, binge alcohol intoxication (≥0.08% BAC) is a major risk factor. We have previously shown that a single dose of alcohol comparable to binge alcohol intoxication increases infection by reducing alveolar macrophage function in vivo. Sulforaphane (SFN), a phytonutrient, is a potent inducer of antioxidant production through the induction of nuclear factor erythroid 2-related factor 2 (Nrf2) and inhibition of the nuclear factor kappa-light-chain-enhancer (NF-kB) pathway. The aim of this study was to test the therapeutic potential of SFN given as a pretreatment to prevent alcohol-induced phagocytic dysfunction. Methods: Intracellular phagocytic killing was measured via colony-forming units (CFU) and cytokine expression via ELISA. G. mellonella survival was used to determine the therapeutic potential of SFN in vivo. Results: Dose–response curves indicated that SFN concentrations of less than 20 µM were not cytotoxic in either MH-S (murine) or THP-1 (human) cells. Live infection assay results showed that MH-S and THP-1 cells pretreated with SFN (5 µM) and challenged with 0.2% (v/v) alcohol for 3 or 8 h prior to live B. thailandensis or S. epidermidis infection improved intracellular pathogen killing between 12- and 20-fold compared to macrophages treated with alcohol alone. ELISA analysis indicated that SFN significantly reduced levels of Tumor necrosis factor-alpha (TNF-α) expression at 3 and 8 h compared to controls. Additionally, a Galleria mellonella larvae model demonstrated greater survivability in the prophylaxis group compared to larvae exposed to either Gram-positive or Gram-negative pathogens, as well as in groups that received alcohol prior to pathogen inoculation. Conclusions: Taken together, SFN-induced cytoprotection was extended beyond in vitro cell culture to include an in vivo G. mellonella model demonstrating protection against Gram-positive and negative opportunistic pathogens. These data demonstrate that SFN may be an effective pretreatment option to prevent alcohol-mediated innate immune dysfunction and restore macrophage phagocytic killing.
Full article
(This article belongs to the Topic New Compounds Discovery and Development in Medicine — Advances in Research on Potential Therapeutic Agents and Drug Candidates, 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessSystematic Review
Pharmacotherapy for Alcohol Craving Reduction: Efficacy of Short-Term Treatments in Alcohol Use Disorder
by
Matheus Cheibub David Marin, Maria Olivia Pozzolo Pedro, Giuliana Perrotte and João Mauricio Castaldelli-Maia
Medicines 2026, 13(1), 7; https://doi.org/10.3390/medicines13010007 - 14 Feb 2026
Abstract
►▼
Show Figures
Background: Alcohol Use Disorder (AUD) is a major contributor to global morbidity, mortality, and socioeconomic burden. Cravings, defined as intense urges to consume alcohol, play a central role in relapse and are recognized as a diagnostic criterion in DSM-5. Pharmacological strategies targeting cravings
[...] Read more.
Background: Alcohol Use Disorder (AUD) is a major contributor to global morbidity, mortality, and socioeconomic burden. Cravings, defined as intense urges to consume alcohol, play a central role in relapse and are recognized as a diagnostic criterion in DSM-5. Pharmacological strategies targeting cravings may offer immediate or short-term relief, complementing existing long-term approaches. However, evidence on short-term (up to approximately three months) anti-craving interventions remains fragmented. Objective: To systematically review randomized, double-blind, placebo-controlled trials (RCTs) assessing the short-term effects of pharmacological treatments on cue-induced alcohol cravings. Methods: A systematic search was conducted in PubMed and PsycINFO using terms related to alcohol, craving, and randomized controlled designs. Eligibility included clinical trials on alcohol-dependent participants that evaluated craving as an outcome. Exclusion criteria encompassed non-clinical studies, non-pharmacological interventions, animal studies, single-blind trials, and studies with psychiatric comorbidities. Study quality was appraised using Cochrane and Joanna Briggs Institute tools. Results: From 442 studies screened, 26 RCTs fulfilled the inclusion criteria. In total, 1097 participants were enrolled across the trials (range = 16–125 per study; mean = 44), predominantly male outpatients aged 15–65 years. Craving was assessed primarily with the Visual Analog Scale and Alcohol Urge Questionnaire. Intervention duration ranged from 1 to 98 days. Naltrexone consistently reduced cue-induced craving across four trials, with additional benefit observed when combined with ondansetron. Varenicline and acamprosate also demonstrated reductions in craving and drinking. Memantine showed efficacy in craving reduction but was not assessed for abstinence. Topiramate was effective, whereas gabapentin showed limited short-term benefit. Other agents (e.g., citalopram, oxytocin, ondansetron, quetiapine) yielded mixed findings, often limited to single studies. Overall, 58% of trials reported positive anti-craving effects, 23% no difference, and 8% increased craving versus placebo. However, these findings should be interpreted in light of important methodological limitations, including small sample sizes and heterogeneous experimental paradigms. Conclusions: This review suggests that naltrexone and varenicline appear to be the most consistently supported short-term pharmacotherapies for alcohol craving within the available evidence, with promising but less consistent findings for memantine, acamprosate, and topiramate. These results highlight potential candidates for immediate craving management in AUD, while underscoring the need for larger and longer-term trials to confirm their efficacy and safety.
Full article

Figure 1
Open AccessReview
The Role of Metabolites in Acyclovir-Induced Neurotoxicity and Nephrotoxicity
by
Asma Aboelezz and Sherif Hanafy Mahmoud
Medicines 2026, 13(1), 6; https://doi.org/10.3390/medicines13010006 - 2 Feb 2026
Abstract
►▼
Show Figures
Acyclovir is an antiviral drug effective against infections caused by herpes simplex and varicella zoster viruses. It is given intravenously to treat serious infections such as herpes encephalitis. High acyclovir concentrations could cause toxicity, observed mainly as nephrotoxicity and, to a lesser extent,
[...] Read more.
Acyclovir is an antiviral drug effective against infections caused by herpes simplex and varicella zoster viruses. It is given intravenously to treat serious infections such as herpes encephalitis. High acyclovir concentrations could cause toxicity, observed mainly as nephrotoxicity and, to a lesser extent, neurotoxicity. Acyclovir nephrotoxicity is primarily attributed to the crystallization of acyclovir within the renal tubules, although additional mechanisms may also contribute. However, the mechanism of acyclovir-induced neurotoxicity is unknown. Acyclovir is mainly eliminated from the body through renal excretion; however, around 15–20% of acyclovir is metabolized subsequently by alcohol and aldehyde dehydrogenase to the main metabolite 9-carboxymethoxymethylguanine (CMMG), and around 2% is metabolized by aldehyde oxidase to the minor metabolite, 8-hydroxyl acyclovir. It has been suggested that CMMG levels above 10 µmol/mL in the serum and 1 µmol/mL in the cerebrospinal fluid are highly associated with neurotoxicity. Studies have shown that there is a potential contribution of CMMG to acyclovir-induced neurotoxicity and of the acyclovir aldehyde to nephrotoxicity. In this narrative review, we approach the topic of acyclovir metabolites and their association with acyclovir toxicity. Moreover, we identify the research gap of the mechanisms by which these metabolites contribute to toxicity.
Full article

Figure 1
Open AccessCorrection
Correction: Zulfiqar et al. Association of Frailty Status with Risk of Fall among Hospitalized Elderly Patients: A Cross-Sectional Study in an Acute Geriatric Unit. Medicines 2022, 9, 48
by
Abrar-Ahmad Zulfiqar, Perla Habchi, Ibrahima Amadou Dembele and Emmanuel Andres
Medicines 2026, 13(1), 5; https://doi.org/10.3390/medicines13010005 - 2 Feb 2026
Abstract
There was an error in the original publication [...]
Full article
Open AccessArticle
Significant Impact of Previous Major Cardiovascular Events (MACEs) and Viremia on Risk of New MACEs in People Living with HIV on Antiretroviral Therapy
by
Caterina Candela, Alessia Siribelli, Tommaso Clemente, Riccardo Lolatto, Michele Bellomo, Vincenzo Stabile, Hamid Hasson, Vincenzo Spagnuolo, Antonella Castagna, Silvia Nozza and Camilla Muccini
Medicines 2026, 13(1), 4; https://doi.org/10.3390/medicines13010004 - 29 Jan 2026
Abstract
►▼
Show Figures
Background: Major cardiovascular events (MACEs) in people with HIV (PWH) may be partly related to antiretroviral therapy (ART) and persistent inflammation. The aim of the study was to evaluate the association between targeted variables and MACEs. Methods: Retrospective, single-center study conducted on PWH
[...] Read more.
Background: Major cardiovascular events (MACEs) in people with HIV (PWH) may be partly related to antiretroviral therapy (ART) and persistent inflammation. The aim of the study was to evaluate the association between targeted variables and MACEs. Methods: Retrospective, single-center study conducted on PWH receiving ART between January 2010 and April 2024, classified according to HIV-RNA levels: virological suppression (<50 copies/mL), low-level viremia (50–200 or 200–1000 copies/mL), and non-suppression (≥1000 copies/mL). Viremia was considered as a time-dependent variable and by cumulative years in each category. A Cox proportional hazards model for multivariate time-to-event analysis assessed associations between virological status and MACEs. Results: We included 3349 PWH followed for a median time of 14 years (interquartile range, IQR 11.2–14.2). At baseline, 2794 (83.4%) were virologically suppressed, 189 (5.6%) and 90 (2.7%) presented 50–200 and 200–1000 copies/mL, respectively, and 276 (8.2%) were non-suppressed. During the follow-up, virological suppression was documented at least once in 3295 (98.4%), low-level viremia in 1579 (47.1%) with 50–200 copies/mL and 794 (23.7%) with 200–1000 copies/mL, and HIV-RNA > 1000 copies/mL in 844 (25.2%). Overall, 300 MACEs occurred, including 53 (17.7%) repeated events, with total incident rate of 0.00976 events per person-year. The risk of MACEs was significantly associated with previous MACEs (Hazard Ratio, HR 3.385, p-value < 0.001) and viremia > 1000 copies/mL at baseline (HR 2.209, p-value 0.039). Their onset was also significantly associated with greater age at baseline and years on ART, hypertension, diabetes, lower HDL, and higher triglycerides. Conclusions: PWH on ART with HIV-RNA > 1000 copies/mL at baseline and a previous MACE presented higher risk of developing MACEs.
Full article

Figure 1
Open AccessSystematic Review
Anticonvulsant Therapy in Trigeminal Neuralgia: A Class-Oriented Systematic Review
by
Miguel Pinto Moreira, Bruno Daniel Carneiro, Carlos Silva Faria, Daniel Humberto Pozza and Sara Fonseca
Medicines 2026, 13(1), 3; https://doi.org/10.3390/medicines13010003 - 26 Jan 2026
Abstract
Background/Objectives: Trigeminal Neuralgia (TN) is a chronic neuropathic condition characterized by sudden, severe facial pain. Anticonvulsants are the cornerstone of pharmacological management, yet comparative evidence based on pharmacological class remains scarce. This systematic review aimed to evaluate the efficacy and safety of anticonvulsants
[...] Read more.
Background/Objectives: Trigeminal Neuralgia (TN) is a chronic neuropathic condition characterized by sudden, severe facial pain. Anticonvulsants are the cornerstone of pharmacological management, yet comparative evidence based on pharmacological class remains scarce. This systematic review aimed to evaluate the efficacy and safety of anticonvulsants in TN, stratified by their mechanism of action. Methods: A systematic search in PubMed, Scopus and Web of Science was conducted following PRISMA 2020 guidelines. Studies employing a pharmacological approach including human patients with TN, published in English since 2000, were included. Risk of bias was assessed using the Cochrane RoB 2, the ROBINS-I and the ROBINS-E tools, according to the study design. Results: Out of 922 initial records, 12 studies met the eligibility criteria. Sodium channel inhibitors showed high efficacy but frequent adverse effects, particularly hyponatremia and central nervous system symptoms. Calcium channel modulators offered a more favorable safety profile. Combination therapies showed benefits, levetiracetam and topiramate were moderately effective and well tolerated. Although the evidence has limitations, anticonvulsants continue to be the primary treatment for TN. Sodium-channel blockers demonstrate strong efficacy, whereas alternative agents generally provide superior tolerability. Conclusions: These findings support selecting drugs according to their underlying mechanisms of action. Equally important is tailoring therapy to pain phenotype and patient characteristics, balancing mechanism with tolerability and efficacy.
Full article
(This article belongs to the Section Neurology and Neurologic Diseases)
►▼
Show Figures

Figure 1
Open AccessPerspective
Antioxidant Delivery Revisited: The Promise of Nanostructured Lipid Carriers
by
Leif Behar and Holly Siddique
Medicines 2026, 13(1), 2; https://doi.org/10.3390/medicines13010002 - 22 Jan 2026
Abstract
►▼
Show Figures
Natural products have an invaluable therapeutic effect on human health. Natural antioxidants, including beta-carotene, turmeric, and polyphenols, are recognised for their health benefits but face significant barriers related to insufficient solubility, instability, volatility, and diminished bioavailability, which limit their therapeutic efficacy in drug
[...] Read more.
Natural products have an invaluable therapeutic effect on human health. Natural antioxidants, including beta-carotene, turmeric, and polyphenols, are recognised for their health benefits but face significant barriers related to insufficient solubility, instability, volatility, and diminished bioavailability, which limit their therapeutic efficacy in drug delivery systems. Therefore, encapsulation of natural products in a carrier addresses the above concern. Drug delivery systems, such as solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs), are promising carriers for effective release, consisting of solid and liquid lipids, which enhance efficiency, stability, and controlled release, thereby minimising bioavailability limitations. This review consolidates current studies on the formulation methodologies, mechanisms of action, and therapeutic applications of NLCs, emphasizing their use in the treatment of conditions such as cancer, neurological disorders, and cardiovascular diseases. The results demonstrate that NLCs substantially enhance the bioavailability and therapeutic efficacy of antioxidants, thereby improving their targeted administration and clinical effects. Nonetheless, difficulties in clinical translation remain, including drug loading capacity, regulatory authorisation, and the need for pervasive research on cytotoxicity. This article highlights important areas for future inquiry, specifically the optimisation of NLC formulations, the enhancement of targeting accuracy, and the resolution of safety issues to enhance their clinical application.
Full article

Figure 1
Open AccessReview
Treatment of Symptomatic Male Hypogonadism with New Oral Testosterone Therapies: A Comparative Review of Jatenzo, Tlando, and Kyzatrex
by
Samantha H. Rosen and Kian Asanad
Medicines 2026, 13(1), 1; https://doi.org/10.3390/medicines13010001 - 22 Dec 2025
Abstract
Symptomatic male hypogonadism, defined by low serum testosterone with associated clinical symptoms, is increasingly treated with testosterone replacement therapy. Traditional oral formulations were limited by hepatotoxicity and poor bioavailability, leading to reliance on injectable and transdermal routes. Recent advances in oral testosterone undecanoate
[...] Read more.
Symptomatic male hypogonadism, defined by low serum testosterone with associated clinical symptoms, is increasingly treated with testosterone replacement therapy. Traditional oral formulations were limited by hepatotoxicity and poor bioavailability, leading to reliance on injectable and transdermal routes. Recent advances in oral testosterone undecanoate formulations have introduced safer and more effective options. This review compares Jatenzo, Tlando, and Kyzatrex, highlighting their pharmacology, efficacy, safety, and clinical utility. Clinical trial data demonstrate restoration of eugonadal testosterone levels in most patients (80–88%), with shared risks including hypertension, polycythemia, and lipid changes. Differences in dosing regimens, titration requirements, and insurance coverage influence choice of therapy and patient adherence. Kyzatrex offers flexible titration and self-pay access, Tlando provides a fixed-dose regimen, and Jatenzo combines titratability with established clinical data. Collectively, these agents expand the therapeutic landscape of hypogonadism, offering effective, non-invasive alternatives that support individualized treatment strategies.
Full article
(This article belongs to the Topic New Compounds Discovery and Development in Medicine — Advances in Research on Potential Therapeutic Agents and Drug Candidates, 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessArticle
Preliminary Evaluation of an Injectable Therapeutic for Cisplatin Ototoxicity Using Neuronal SH-SY5Y Cells
by
Michelle Hong, Katherine Kedeshian, Larry Hoffman and Ashley Kita
Medicines 2025, 12(4), 30; https://doi.org/10.3390/medicines12040030 - 9 Dec 2025
Abstract
►▼
Show Figures
Background/Objectives: Though ototoxic, cisplatin is a mainstay of chemotherapy for a variety of cancers. One suggested mechanism of cisplatin ototoxicity involves damage to the spiral ganglion afferent neurons in the inner ear. There is a need for a high-throughput model to screen medications
[...] Read more.
Background/Objectives: Though ototoxic, cisplatin is a mainstay of chemotherapy for a variety of cancers. One suggested mechanism of cisplatin ototoxicity involves damage to the spiral ganglion afferent neurons in the inner ear. There is a need for a high-throughput model to screen medications for efficacy against cisplatin and to develop a local therapeutic to mitigate cisplatin’s debilitating side effects. Microparticles encapsulating a therapeutic medication are an injectable and tunable method of sustained drug delivery, and thus a promising treatment. Methods: SH-SY5y human neuroblastoma cells were used as a cell line model for the spiral ganglion neurons. The cells were dosed with cisplatin and four potential therapeutics (melatonin, metformin, cyclosporine, and N-acetylcysteine), with cell viability measured by CCK-8 assay. The most promising therapeutic, N-acetylcysteine (NAC), was then encapsulated into multiple poly(lactic-co-glycolic acid) (PLGA) microparticle subtypes of varied lactide–glycolide (L:G) ratios and NAC amounts. The elution profile of each microparticle subtype was determined over two months. Results: Of the therapeutics screened, only cells dosed with 1 or 10 mM NAC prior to cisplatin injury demonstrated an improvement in cell viability (73.8%, p < 1 × 10−8) when compared to cells dosed with cisplatin alone. The 75:25 L:G microparticles demonstrated an increase in the amount of NAC released compared to the 50:50 L:G microparticles. Conclusions: NAC is a potential therapeutic agent for cisplatin toxicity when tested in a neuronal cell line model. NAC was encapsulated into PLGA microparticles and eluted detectable concentrations of NAC for 6 days, which is a first step towards otoprotection for the weeks long duration of chemotherapy treatment. This work describes a method of screening potential therapeutics and a strategy to develop local drug eluting treatments to protect against cisplatin ototoxicity.
Full article

Graphical abstract
Open AccessArticle
Identification of Active Components in Connarus ruber Extract Exhibiting Anti-Glycation Effects
by
Ryoji Taniguchi, Ryusuke Nakatsuka, Yuka Sasaki, Mariko Takenokuchi, Takashi Maoka, Tomio Iseki, Hirohito Kubo and Tadashige Nozaki
Medicines 2025, 12(4), 29; https://doi.org/10.3390/medicines12040029 - 3 Dec 2025
Abstract
Background: Glycation, a non-enzymatic reaction between sugars and biomolecules, leads to the formation of advanced glycation end-products (AGEs), which are implicated in the progression of chronic diseases. Connarus ruber (Poepp.) Planch (C. ruber), a traditional medicinal plant used for diabetes, has
[...] Read more.
Background: Glycation, a non-enzymatic reaction between sugars and biomolecules, leads to the formation of advanced glycation end-products (AGEs), which are implicated in the progression of chronic diseases. Connarus ruber (Poepp.) Planch (C. ruber), a traditional medicinal plant used for diabetes, has shown anti-glycation activity. This study aimed to identify the active components in C. ruber extract and elucidate their anti-glycation mechanisms. Methods: Using NMR and LC-MS analyses, we identified epicatechin and procyanidin A2 as major polyphenolic constituents. Collagen glycation assays were performed to evaluate the inhibitory effects of these compounds on fructose- and glyceraldehyde (GA)-induced glycation. Additionally, their cytoprotective effects were assessed using GA-induced cytotoxicity assays in dental pulp stem cells (DPSCs). Results: Both epicatechin and procyanidin A2 inhibited fructose- and GA-induced glycation in a dose-dependent manner, showing greater efficacy than aminoguanidine. Furthermore, these compounds significantly alleviated GA-induced cytotoxicity in DPSCs. Conclusions: These findings suggest that epicatechin and procyanidin A2 are candidate contributors to the anti-glycation and cytoprotective effects of C. ruber. The results support the potential of C. ruber extract as a source of therapeutic agents for glycation-related diseases and for enhancing stem cell viability.
Full article
(This article belongs to the Section Oral Medicine and Dentistry)
►▼
Show Figures

Figure 1
Open AccessReview
AΙ-Driven Drug Repurposing: Applications and Challenges
by
Paraskevi Keramida, Nikolaos K. Syrigos, Marousa Kouvela, Garyfallia Poulakou, Andriani Charpidou and Oraianthi Fiste
Medicines 2025, 12(4), 28; https://doi.org/10.3390/medicines12040028 - 13 Nov 2025
Cited by 6
Abstract
►▼
Show Figures
Drug repurposing is the process of discovering new therapeutic indications for already existing drugs. By using already approved molecules with known safety profiles, this approach reduces the time, costs, and failure rates associated with traditional drug development, accelerating the availability of new treatments
[...] Read more.
Drug repurposing is the process of discovering new therapeutic indications for already existing drugs. By using already approved molecules with known safety profiles, this approach reduces the time, costs, and failure rates associated with traditional drug development, accelerating the availability of new treatments to patients. Artificial Intelligence (AI) plays a crucial role in drug repurposing by exploiting various computational techniques to analyze and process big datasets of biological and medical information, predict similarities between biomolecules, and identify disease mechanisms. The purpose of this review is to explore the role of AI tools in drug repurposing and underline their applications across various medical domains, mainly in oncology, neurodegenerative disorders, and rare diseases. However, several challenges remain to be addressed. These include the need for a deeper understanding of molecular mechanisms, ethical concerns, regulatory requirements, and issues related to data quality and interpretability. Overall, AI-driven drug repurposing is an innovative and promising field that can transform medical research and drug development, covering unmet medical needs efficiently and cost-effectively.
Full article

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Healthcare, Hospitals, JCM, Safety, Medicina, Medicines
Drug Use and Patient Safety in Primary and Secondary Care Settings
Topic Editors: Kingston Rajiah, Ashuin Kammar-GarcíaDeadline: 31 October 2026
Topic in
Biomedicines, IJMS, Medicines, Molecules, Pharmaceuticals, Sci. Pharm.
New Compounds Discovery and Development in Medicine — Advances in Research on Potential Therapeutic Agents and Drug Candidates, 2nd Edition
Topic Editors: Monika Wujec, Anna Bogucka-Kocka, Przemysław Kołodziej, Jacek BoguckiDeadline: 31 December 2026
Topic in
Compounds, Medicines, Molecules, Plants, Separations, Applied Biosciences, Life, Pharmaceuticals
Research on Natural Products of Medical Plants
Topic Editors: Cristiane Aguiar Da Costa, Graziele Freitas De BemDeadline: 5 March 2027
Topic in
Biomedicines, IJMS, Medicines, Pharmaceuticals, Sci. Pharm.
Research in Pharmacological Therapies, 2nd Edition
Topic Editors: Juan Gambini, Ángel Luis OrtegaDeadline: 30 March 2027

Conferences
Special Issues
Special Issue in
Medicines
Drug-Related Problems: Clinical Pharmacy and Safety of Pharmacotherapy
Guest Editors: Mariola Drozd, Jakub PawlikowskiDeadline: 30 August 2026
Special Issue in
Medicines
Drug Abuse and Withdrawal Syndromes: From Addiction Pathways to Novel Interventions
Guest Editor: Stephen J. LewisDeadline: 31 December 2026
Conference Reports
Medicines 2017, 4(4), 83; https://doi.org/10.3390/medicines4040083
Medicines 2017, 4(4), 76; https://doi.org/10.3390/medicines4040076






