Diurnal Variation in Biomarkers of Exposure to Endocrine-Disrupting Chemicals and Their Association with Oxidative Damage in Norwegian Adults: The EuroMix Study
Abstract
1. Introduction
2. Materials and Methods
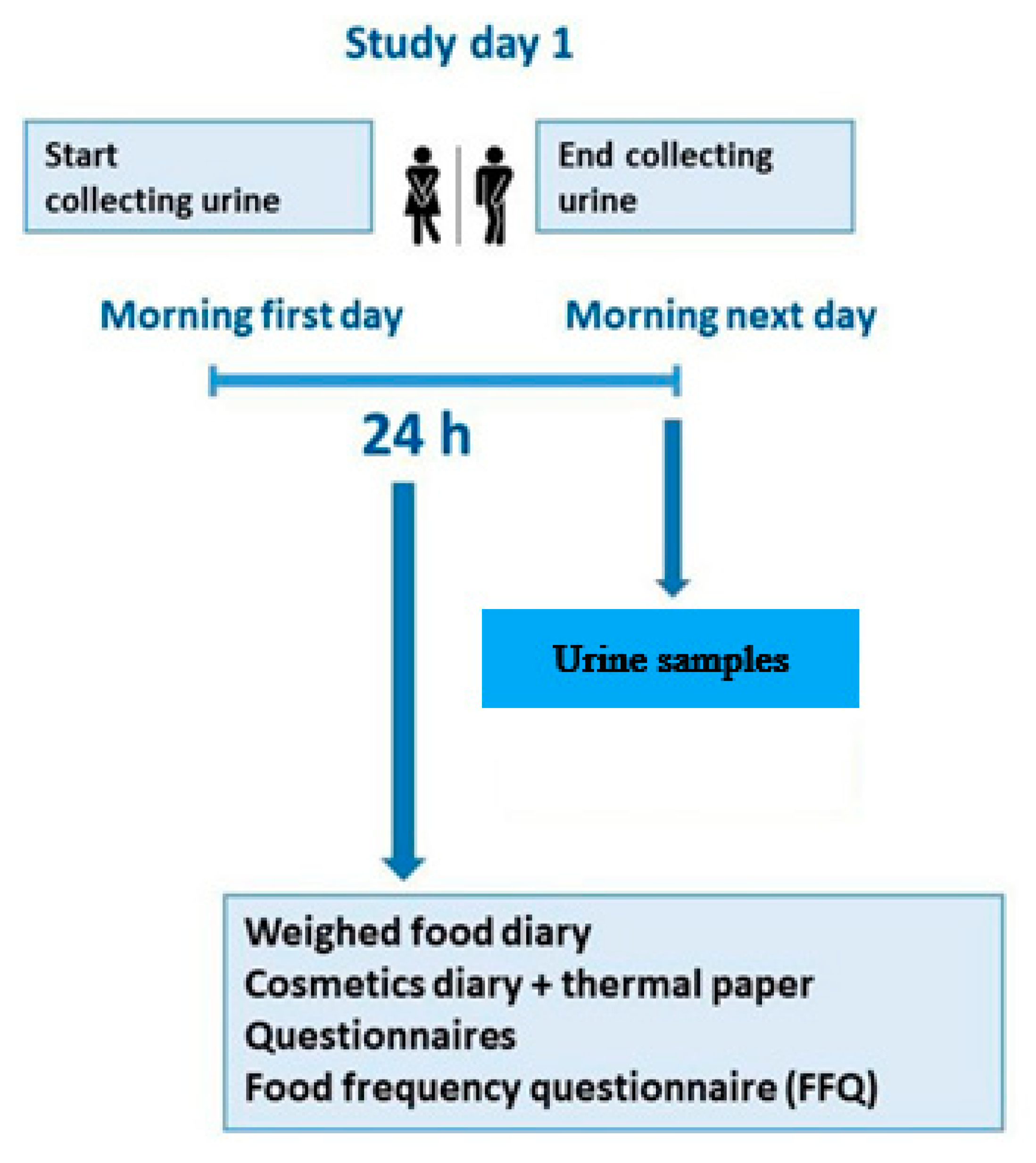
2.1. Data Collection
2.2. Biomarkers
2.3. Data Handling
2.3.1. Data Cleaning
2.3.2. Dealing with Missing Values
2.3.3. Data below Limit of Detection
2.3.4. Data Adjustment
2.4. Statistical Analysis
3. Results
3.1. Population Characteristics
3.2. Biomarkers of Exposure/Effect
3.2.1. Urinary Biomarkers of Exposure to Phenols, Phthalates, and Parabens
3.2.2. Association between Time of the Day and the Biomarkers of EDC Exposure and Effect
3.2.3. Association between Biomarkers of Exposure to EDCs and the Biomarker of Oxidative Damage
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Gore, A.C.; Chappell, V.A.; Fenton, S.E.; Flaws, J.A.; Nadal, A.; Prins, G.S.; Toppari, J.; Zoeller, R.T. EDC-2: The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr. Rev. 2015, 36, E1–E150. [Google Scholar] [CrossRef] [PubMed]
- OECD. Revised Guidance Document 150 on Standardised Test Guidelines for Evaluating Chemicals for Endocrine Disruption; OECD Series on Testing and Assessment, No. 150; OECD Publishing: Paris, France, 2018. [Google Scholar] [CrossRef]
- Trasande, L.; Zoeller, R.T.; Hass, U.; Kortenkamp, A.; Grandjean, P.; Myers, J.P.; DiGangi, J.; Bellanger, M.; Hauser, R.; Legler, J.; et al. Estimating Burden and Disease Costs of Exposure to Endocrine-Disrupting Chemicals in the European Union. J. Clin. Endocrinol. Metab. 2015, 100, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Bourguignon, J.-P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-Disrupting Chemicals: An Endocrine Society Scientific Statement. Endocr. Rev. 2009, 30, 293–342. [Google Scholar] [CrossRef] [PubMed]
- LaKind, J.; Idri, F.; Naiman, D.Q.; Verner, M.-A. Biomonitoring and Nonpersistent Chemicals—Understanding and Addressing Variability and Exposure Misclassification. Curr. Environ. Health Rep. 2019, 6, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Frederiksen, H.; Søgaard Nielsen, J.K.; Aarøe Mørck, T.; Hansen, P.W.; Jensen, J.F.; Nielsen, O.; Andersson, A.-M.; Knudsen, L.E. Urinary excretion of phthalate metabolites, phenols and parabens in rural and urban Danish mother–child pairs. Int. J. Hyg. Environ. Health 2013, 216, 772–783. [Google Scholar] [CrossRef] [PubMed]
- Larsson, K.; Björklund, K.L.; Palm, B.; Wennberg, M.; Kaj, L.; Lindh, C.H.; Jönsson, B.A.; Berglund, M. Exposure determinants of phthalates, parabens, bisphenol A and triclosan in Swedish mothers and their children. Environ. Int. 2014, 73, 323–333. [Google Scholar] [CrossRef]
- Koch, H.M.; Bolt, H.M.; Preuss, R.; Angerer, J. New metabolites of di(2-ethylhexyl)phthalate (DEHP) in human urine and serum after single oral doses of deuterium-labelled DEHP. Arch. Toxicol. 2005, 79, 367–376. [Google Scholar] [CrossRef]
- Morgan, M.K.; Nash, M.; Barr, D.B.; Starr, J.M.; Clifton, M.S.; Sobus, J. Distribution, variability, and predictors of urinary bisphenol A levels in 50 North Carolina adults over a six-week monitoring period. Environ. Int. 2017, 112, 85–99. [Google Scholar] [CrossRef]
- Völkel, W.; Colnot, T.; Csanády, G.A.; Filser, J.G.; Dekant, W. Metabolism and Kinetics of Bisphenol A in Humans at Low Doses Following Oral Administration. Chem. Res. Toxicol. 2002, 15, 1281–1287. [Google Scholar] [CrossRef]
- Ye, X.; Wong, L.-Y.; Bishop, A.M.; Calafat, A.M. Variability of Urinary Concentrations of Bisphenol A in Spot Samples, First Morning Voids, and 24-Hour Collections. Environ. Health Perspect. 2011, 119, 983–988. [Google Scholar] [CrossRef]
- Kwak, E.S.; Just, A.; Whyatt, R.; Miller, R.L. Phthalates, pesticides, and bisphenol-A exposure and the development of nonoccupational asthma and allergies: How valid are the links? Open Allergy J. 2009, 2, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Crinnion, W.J. The CDC fourth national report on human exposure to environmental chemicals: What it tells us about our toxic burden and how it assists environmental medicine physicians. Altern. Med. Rev. 2010, 15, 101–109. [Google Scholar] [PubMed]
- Benjamin, S.; Masai, E.; Kamimura, N.; Takahashi, K.; Anderson, R.C.; Faisal, P.A. Phthalates impact human health: Epidemiological evidences and plausible mechanism of action. J. Hazard. Mater. 2017, 340, 360–383. [Google Scholar] [CrossRef] [PubMed]
- Lottrup, G.; Andersson, A.-M.; Leffers, H.; Mortensen, G.K.; Toppari, J.; Skakkebaek, N.E.; Main, K.M. Possible impact of phthalates on infant reproductive health. Int. J. Androl. 2006, 29, 172–180. [Google Scholar] [CrossRef]
- Ramadan, M.; Cooper, B.; Posnack, N.G. Bisphenols and phthalates: Plastic chemical exposures can contribute to adverse cardiovascular health outcomes. Birth Defects Res. 2020, 112, 1362–1385. [Google Scholar] [CrossRef]
- Leppert, B.; Strunz, S.; Seiwert, B.; Schlittenbauer, L.; Schlichting, R.; Pfeiffer, C.; Röder, S.; Bauer, M.; Borte, M.; Stangl, G.I.; et al. Maternal paraben exposure triggers childhood overweight development. Nat. Commun. 2020, 11, 561. [Google Scholar] [CrossRef]
- Ben-Jonathan, N.; Hugo, E.R. Bisphenols Come in Different Flavors: Is “S” Better Than “A”? Endocrinology 2016, 157, 1321–1323. [Google Scholar] [CrossRef][Green Version]
- Anderson, G.; Beischlag, T.V.; Vinciguerra, M.; Mazzoccoli, G. The circadian clock circuitry and the AHR signaling pathway in physiology and pathology. Biochem. Pharmacol. 2013, 85, 1405–1416. [Google Scholar] [CrossRef]
- Vernet, C.; Pin, I.; Giorgis-Allemand, L.; Philippat, C.; Benmerad, M.; Quentin, J.; Calafat, A.M.; Ye, X.; Annesi-Maesano, I.; Siroux, V.; et al. In Utero Exposure to Select Phenols and Phthalates and Respiratory Health in Five-Year-Old Boys: A Prospective Study. Environ. Health Perspect. 2017, 125, 097006. [Google Scholar] [CrossRef]
- Hwang, M.; Choi, K.; Park, C. Urinary levels of phthalate, bisphenol, and paraben and allergic outcomes in children: Korean National Environmental Health Survey 2015–2017. Sci. Total Environ. 2021, 818, 151703. [Google Scholar] [CrossRef]
- Pugazhendhi, D.; Pope, G.S.; Darbre, P.D. Oestrogenic activity of p-hydroxybenzoic acid (common metabolite of paraben esters) and methylparaben in human breast cancer cell lines. J. Appl. Toxicol. 2005, 25, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.; E Arbuckle, T.; Mallick, R.; Leblanc, A.; Hauser, R.; Feeley, M.; Koniecki, D.; Ramsay, T.; Provencher, G.; Bérubé, R.; et al. Bisphenol A and phthalate metabolite urinary concentrations: Daily and across pregnancy variability. J. Expo. Sci. Environ. Epidemiol. 2014, 25, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Gängler, S.; Charisiadis, P.; Seth, R.; Chatterjee, S.; Makris, K.C. Time of the day dictates the variability of biomarkers of exposure to disinfection byproducts. Environ. Int. 2018, 112, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kang, D.R.; Kwak, J.M.; Lee, J.K. Concentration and Variability of Urinary Phthalate Metabolites, Bisphenol A, Triclosan, and Parabens in Korean Mother–Infant Pairs. Sustainability 2020, 12, 8516. [Google Scholar] [CrossRef]
- Vernet, C.; Philippat, C.; Calafat, A.M.; Ye, X.; Lyon-Caen, S.; Siroux, V.; Schisterman, E.; Slama, R. Within-Day, Between-Day, and Between-Week Variability of Urinary Concentrations of Phenol Biomarkers in Pregnant Women. Environ. Health Perspect. 2018, 126, 037005. [Google Scholar] [CrossRef] [PubMed]
- Makris, K.C. Desynchronized circadian clock and exposures to xenobiotics are associated with differentiated disease phenotypes. BioEssays 2021, 43, 2100159. [Google Scholar] [CrossRef]
- Kang, S.; Kim, S.; Park, J.; Kim, H.-J.; Lee, J.; Choi, G.; Choi, S.; Kim, S.; Kim, S.Y.; Moon, H.-B.; et al. Urinary paraben concentrations among pregnant women and their matching newborn infants of Korea, and the association with oxidative stress biomarkers. Sci. Total Environ. 2013, 461–462, 214–221. [Google Scholar] [CrossRef]
- Zheng, R.; Dragomir, A.-C.; Mishin, V.; Richardson, J.R.; Heck, D.E.; Laskin, D.L.; Laskin, J.D. Differential metabolism of 4-hydroxynonenal in liver, lung and brain of mice and rats. Toxicol. Appl. Pharmacol. 2014, 279, 43–52. [Google Scholar] [CrossRef]
- Zarkovic, K. 4-Hydroxynonenal and neurodegenerative diseases. Mol. Asp. Med. 2003, 24, 293–303. [Google Scholar] [CrossRef]
- Dalleau, S.; Baradat, M.; Guéraud, F.; Huc, L. Cell death and diseases related to oxidative stress: 4-hydroxynonenal (HNE) in the balance. Cell Death Differ. 2013, 20, 1615–1630. [Google Scholar] [CrossRef]
- Paradis, V.; Kollinger, M.; Fabre, M.; Holstege, A.; Poynard, T.; Bedossa, P. In situ detection of lipid peroxidation by-products in chronic liver diseases. Hepatology 1997, 26, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Rahman, I.; Van Schadewijk, A.A.M.; Crowther, A.J.L.; Hiemstra, P.S.; Stolk, J.; MacNee, W.; de Boer, W. 4-Hydroxy-2-Nonenal, a Specific Lipid Peroxidation Product, Is Elevated in Lungs of Patients with Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2002, 166, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Husøy, T.; Andreassen, M.; Hjertholm, H.; Carlsen, M.; Norberg, N.; Sprong, C.; Papadopoulou, E.; Sakhi, A.; Sabaredzovic, A.; Dirven, H. The Norwegian biomonitoring study from the EU project EuroMix: Levels of phenols and phthalates in 24-hour urine samples and exposure sources from food and personal care products. Environ. Int. 2019, 132, 105103. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, E.; Sabaredzovic, A.; Namork, E.; Nygaard, U.C.; Granum, B.; Haug, L.S. Exposure of Norwegian toddlers to perfluoroalkyl substances (PFAS): The association with breastfeeding and maternal PFAS concentrations. Environ. Int. 2016, 94, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Sakhi, A.K.; Sabaredzovic, A.; Cequier, E.; Thomsen, C. Phthalate metabolites in Norwegian mothers and children: Levels, diurnal variation and use of personal care products. Sci. Total Environ. 2017, 599, 1984–1992. [Google Scholar] [CrossRef]
- Sabaredzovic, A.; Sakhi, A.K.; Brantsæter, A.L.; Thomsen, C. Determination of 12 urinary phthalate metabolites in Norwegian pregnant women by core-shell high performance liquid chromatography with on-line solid-phase extraction, column switching and tandem mass spectrometry. J. Chromatogr. B 2015, 1002, 343–352. [Google Scholar] [CrossRef]
- Sakhi, A.K.; Sabaredzovic, A.; Papadopoulou, E.; Cequier, E.; Thomsen, C. Levels, variability and determinants of environmental phenols in pairs of Norwegian mothers and children. Environ. Int. 2018, 114, 242–251. [Google Scholar] [CrossRef]
- Karangwa, I. Using the Markov Chain Monte Carlo Method to Make Inferences on Items of Data Contaminated by Missing Values. Am. J. Theor. Appl. Stat. 2013, 2, 48–53. [Google Scholar] [CrossRef]
- Moon, J.-Y.; Jung, H.-J.; Moon, M.H.; Chung, B.C.; Choi, M.H. Heat-map visualization of gas chromatography-mass spectrometry based quantitative signatures on steroid metabolism. J. Am. Soc. Mass Spectrom. 2009, 20, 1626–1637. [Google Scholar] [CrossRef]
- Sifakis, S.; Androutsopoulos, V.P.; Tsatsakis, A.M.; Spandidos, D.A. Human exposure to endocrine disrupting chemicals: Effects on the male and female reproductive systems. Environ. Toxicol. Pharmacol. 2017, 51, 56–70. [Google Scholar] [CrossRef]
- Bray, M.S.; Shaw, C.A.; Moore, M.W.S.; Garcia, R.A.P.; Zanquetta, M.M.; Durgan, D.J.; Jeong, W.J.; Tsai, J.-Y.; Bugger, H.; Zhang, D.; et al. Disruption of the circadian clock within the cardiomyocyte influences myocardial contractile function, metabolism, and gene expression. Am. J. Physiol. Circ. Physiol. 2008, 294, H1036–H1047. [Google Scholar] [CrossRef] [PubMed]
- Schindler, B.K.; Esteban, M.; Koch, H.M.; Castano, A.; Koslitz, S.; Cañas, A.; Casteleyn, L.; Kolossa-Gehring, M.; Schwedler, G.; Schoeters, G.; et al. The European COPHES/DEMOCOPHES project: Towards transnational comparability and reliability of human biomonitoring results. Int. J. Hyg. Environ. Health 2014, 217, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.C.Y.; Pasikanti, K.K.; Nicholson, J. Global urinary metabolic profiling procedures using gas chromatography–mass spectrometry. Nat. Protoc. 2011, 6, 1483–1499. [Google Scholar] [CrossRef] [PubMed]
- Curtin, P.; Austin, C.; Curtin, A.; Gennings, C.; Arora, M.; Tammimies, K.; Willfors, C.; Berggren, S.; Siper, P.; Rai, D.; et al. Dynamical features in fetal and postnatal zinc-copper metabolic cycles predict the emergence of autism spectrum disorder. Sci. Adv. 2018, 4, eaat1293. [Google Scholar] [CrossRef]
- Carroll, R.; Metcalfe, C.; Gunnell, D.; Mohamed, F.; Eddleston, M. Diurnal variation in probability of death following self-poisoning in Sri Lanka--evidence for chronotoxicity in humans. Int. J. Epidemiology 2012, 41, 1821–1828. [Google Scholar] [CrossRef]
- Imai, T. Human Carboxylesterase Isozymes: Catalytic Properties and Rational Drug Design. Drug Metab. Pharmacokinet. 2006, 21, 173–185. [Google Scholar] [CrossRef]
- Jewell, C.; Prusakiewicz, J.J.; Ackermann, C.; Payne, N.A.; Fate, G.; Voorman, R.; Williams, F.M. Hydrolysis of a series of parabens by skin microsomes and cytosol from human and minipigs and in whole skin in short-term culture. Toxicol. Appl. Pharmacol. 2007, 225, 221–228. [Google Scholar] [CrossRef]
- Obringer, C.; Wu, S.; Troutman, J.; Karb, M.; Lester, C. Effect of chain length and branching on the in vitro metabolism of a series of parabens in human liver S9, human skin S9, and human plasma. Regul. Toxicol. Pharmacol. 2021, 122, 104918. [Google Scholar] [CrossRef]
- Aubert, N.; Ameller, T.; Legrand, J.-J. Systemic exposure to parabens: Pharmacokinetics, tissue distribution, excretion balance and plasma metabolites of [14C]-methyl-, propyl- and butylparaben in rats after oral, topical or subcutaneous administration. Food Chem. Toxicol. 2012, 50, 445–454. [Google Scholar] [CrossRef]
- Zhang, Y.-K.J.; Yeager, R.L.; Klaassen, C.D. Circadian Expression Profiles of Drug-Processing Genes and Transcription Factors in Mouse Liver. Drug Metab. Dispos. 2008, 37, 106–115. [Google Scholar] [CrossRef]
- Zhao, M.; Zhang, T.; Yu, F.; Guo, L.; Wu, B. E4bp4 regulates carboxylesterase 2 enzymes through repression of the nuclear receptor Rev-erbα in mice. Biochem. Pharmacol. 2018, 152, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, H.; Sugihara, K.; Watanabe, Y.; Ohta, S.; Kitamura, S. Cytochrome P450-inhibitory activity of parabens and phthalates used in consumer products. J. Toxicol. Sci. 2016, 41, 551–560. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pollock, T.; Weaver, R.E.; Ghasemi, R.; Decatanzaro, D. Butyl paraben and propyl paraben modulate bisphenol A and estradiol concentrations in female and male mice. Toxicol. Appl. Pharmacol. 2017, 325, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Cederbaum, A.I. CYP2E1 and oxidative liver injury by alcohol. Free Radic. Biol. Med. 2008, 44, 723–738. [Google Scholar] [CrossRef] [PubMed]
- Seth, R.K.; Kumar, A.; Das, S.; Kadiiska, M.B.; Michelotti, G.; Diehl, A.M.; Chatterjee, S. Environmental Toxin–Linked Nonalcoholic Steatohepatitis and Hepatic Metabolic Reprogramming in Obese Mice. Toxicol. Sci. 2013, 134, 291–303. [Google Scholar] [CrossRef]
- Weber, D.; Milkovic, L.; Bennett, S.J.; Griffiths, H.R.; Zarkovic, N.; Grune, T. Measurement of HNE-protein adducts in human plasma and serum by ELISA—Comparison of two primary antibodies. Redox Biol. 2013, 1, 226–233. [Google Scholar] [CrossRef]
- Zhong, H.; Yin, H. Role of lipid peroxidation derived 4-hydroxynonenal (4-HNE) in cancer: Focusing on mitochondria. Redox Biol. 2014, 4, 193–199. [Google Scholar] [CrossRef]
- Łuczaj, W.; Gindzienska-Sieskiewicz, E.; Jarocka-Karpowicz, I.; Andrisic, L.; Sierakowski, S.; Zarkovic, N.; Waeg, G.; Skrzydlewska, E. The onset of lipid peroxidation in rheumatoid arthritis: Consequences and monitoring. Free Radic. Res. 2016, 50, 304–313. [Google Scholar] [CrossRef]
- Castro, J.P.; Jung, T.; Grune, T.; Siems, W. 4-Hydroxynonenal (HNE) modified proteins in metabolic diseases. Free Radic. Biol. Med. 2017, 111, 309–315. [Google Scholar] [CrossRef]
- Steffensen, I.-L.; Dirven, H.; Couderq, S.; David, A.; D’Cruz, S.C.; Fernández, M.F.; Mustieles, V.; Rodríguez-Carrillo, A.; Hofer, T. Bisphenols and Oxidative Stress Biomarkers—Associations Found in Human Studies, Evaluation of Methods Used, and Strengths and Weaknesses of the Biomarkers. Int. J. Environ. Res. Public Health 2020, 17, 3609. [Google Scholar] [CrossRef]
- Wang, Y.-X.; Liu, C.; Shen, Y.; Wang, Q.; Pan, A.; Yang, P.; Chen, Y.-J.; Deng, Y.-L.; Lu, Q.; Cheng, L.-M.; et al. Urinary levels of bisphenol A, F and S and markers of oxidative stress among healthy adult men: Variability and association analysis. Environ. Int. 2018, 123, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Asimakopoulos, A.G.; Xue, J.; De Carvalho, B.P.; Iyer, A.; Abualnaja, K.O.; Yaghmoor, S.S.; Kumosani, T.A.; Kannan, K. Urinary biomarkers of exposure to 57 xenobiotics and its association with oxidative stress in a population in Jeddah, Saudi Arabia. Environ. Res. 2016, 150, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, K.; Cantonwine, D.E.; McElrath, T.F.; Mukherjee, B.; Meeker, J.D. Repeated measures analysis of associations between urinary bisphenol-A concentrations and biomarkers of inflammation and oxidative stress in pregnancy. Reprod. Toxicol. 2016, 66, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Watkins, D.J.; Ferguson, K.; Del Toro, L.V.A.; Alshawabkeh, A.N.; Cordero, J.F.; Meeker, J.D. Associations between urinary phenol and paraben concentrations and markers of oxidative stress and inflammation among pregnant women in Puerto Rico. Int. J. Hyg. Environ. Health 2014, 218, 212–219. [Google Scholar] [CrossRef]
- Rocha, B.A.; Asimakopoulos, A.G.; Honda, M.; da Costa, N.L.; Barbosa, R.M.; Barbosa, F.; Kannan, K. Advanced data mining approaches in the assessment of urinary concentrations of bisphenols, chlorophenols, parabens and benzophenones in Brazilian children and their association to DNA damage. Environ. Int. 2018, 116, 269–277. [Google Scholar] [CrossRef]
- Lv, Y.; Rui, C.; Dai, Y.; Pang, Q.; Li, Y.; Fan, R.; Lu, S. Exposure of children to BPA through dust and the association of urinary BPA and triclosan with oxidative stress in Guangzhou, China. Environ. Sci. Process. Impacts 2016, 18, 1492–1499. [Google Scholar] [CrossRef]
- Ding, S.; Qi, W.; Xu, Q.; Zhao, T.; Li, X.; Yin, J.; Zhang, R.; Huo, C.; Zhou, L.; Ye, L. Relationships between di-(2-ethylhexyl) phthalate exposure and lipid metabolism in adolescents: Human data and experimental rat model analyses. Environ. Pollut. 2021, 286, 117570. [Google Scholar] [CrossRef]
- Aseervatham, G.S.B.; Sivasudha, T.; Jeyadevi, R.; Ananth, D.A. Environmental factors and unhealthy lifestyle influence oxidative stress in humans—An overview. Environ. Sci. Pollut. Res. 2013, 20, 4356–4369. [Google Scholar] [CrossRef]
- Rytilä, P.; Rehn, T.; Ilumets, H.; Rouhos, A.; Sovijärvi, A.; Myllärniemi, M.; Kinnula, V.L. Increased oxidative stress in asymptomatic current chronic smokers and GOLD stage 0 COPD. Respir. Res. 2006, 7, 69. [Google Scholar] [CrossRef][Green Version]
- Kode, A.; Yang, S.-R.; Rahman, I. Differential effects of cigarette smoke on oxidative stress and proinflammatory cytokine release in primary human airway epithelial cells and in a variety of transformed alveolar epithelial cells. Respir. Res. 2006, 7, 132. [Google Scholar] [CrossRef]
- Sbrana, E.; Suter, M.A.; Abramovici, A.R.; Hawkins, H.K.; Moss, J.E.; Patterson, L.; Shope, C.; Aagaard-Tillery, K. Maternal tobacco use is associated with increased markers of oxidative stress in the placenta. Am. J. Obstet. Gynecol. 2011, 205, 246.e1–246.e7. [Google Scholar] [CrossRef] [PubMed]
- Minato, T.; Tsutsumi, M.; Tsuchishima, M.; Hayashi, N.; Saito, T.; Matsue, Y.; Toshikuni, N.; Arisawa, T.; George, J. Binge Alcohol Consumption Aggravates Oxidative Stress and Promotes Pathogenesis of NASH from Obesity-Induced Simple Steatosis. Mol. Med. 2014, 20, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Watt, J.; Schuon, J.; Davis, J.; Ferguson, T.F.; Welsh, D.A.; Molina, P.E.; Ronis, M.J.J. Reduced Serum Osteocalcin in High-Risk Alcohol Using People Living With HIV Does Not Correlate With Systemic Oxidative Stress or Inflammation: Data From the New Orleans Alcohol Use in HIV Study. Alcohol. Clin. Exp. Res. 2019, 43, 2374–2383. [Google Scholar] [CrossRef] [PubMed]
- Wegner, S.A.; Pollard, K.A.; Kharazia, V.; Darevsky, D.; Perez, L.; Roychowdhury, S.; Xu, A.; Ron, R.; Nagy, L.E.; Hopf, F.W. Limited Excessive Voluntary Alcohol Drinking Leads to Liver Dysfunction in Mice. Alcohol. Clin. Exp. Res. 2017, 41, 345–358. [Google Scholar] [CrossRef]
- Raza, H.; John, A. 4-Hydroxynonenal induces mitochondrial oxidative stress, apoptosis and expression of glutathione S-transferase A4-4 and cytochrome P450 2E1 in PC12 cells. Toxicol. Appl. Pharmacol. 2006, 216, 309–318. [Google Scholar] [CrossRef]
- Das, S.; Kumar, A.; Seth, R.; Tokar, E.J.; Kadiiska, M.B.; Waalkes, M.P.; Mason, R.P.; Chatterjee, S. Proinflammatory adipokine leptin mediates disinfection byproduct bromodichloromethane-induced early steatohepatitic injury in obesity. Toxicol. Appl. Pharmacol. 2013, 269, 297–306. [Google Scholar] [CrossRef]
- Tomasi, A.; Albano, E.; Biasi, F.; Slater, T.F.; Vannini, V.; Dianzani, M.U. Activation of chloroform and related trihalomethanes to free radical intermediates in isolated hepatocytes and in the rat in vivo as detected by the ESR-spin trapping technique. Chem. Interact. 1985, 55, 303–316. [Google Scholar] [CrossRef]
- Seth, R.K.; Das, S.; Dattaroy, D.; Chandrashekaran, V.; Alhasson, F.; Michelotti, G.; Nagarkatti, M.; Nagarkatti, P.; Diehl, A.M.; Bell, P.D.; et al. TRPV4 activation of endothelial nitric oxide synthase resists nonalcoholic fatty liver disease by blocking CYP2E1-mediated redox toxicity. Free Radic. Biol. Med. 2016, 102, 260–273. [Google Scholar] [CrossRef]
- Gkrillas, A.; Dirven, H.; Papadopoulou, E.; Andreassen, M.; Hjertholm, H.; Husøy, T. Exposure estimates of phthalates and DINCH from foods and personal care products in comparison with biomonitoring data in 24-hour urine from the Norwegian EuroMix biomonitoring study. Environ. Int. 2021, 155, 106598. [Google Scholar] [CrossRef]
- Haddad, N.; Andrianou, X.D.; Makris, K.C. A Scoping Review on the Characteristics of Human Exposome Studies. Curr. Pollut. Rep. 2019, 5, 378–393. [Google Scholar] [CrossRef]
- Vermeulen, R.; Schymanski, E.L.; Barabási, A.-L.; Miller, G.W. The exposome and health: Where chemistry meets biology. Science 2020, 367, 392–396. [Google Scholar] [CrossRef] [PubMed]


| Basic Characteristics | Overall (n = 144) | Males (n = 44) | Females (n = 100) |
|---|---|---|---|
| Age (years, mean ± SD) | 42.56± 12.05 | 43.36 ± 11.65 | 42.20 ± 12.26 |
| Weight (kg, mean ± SD) | 70.45 ± 11.71 | 81.98 ± 8.54 | 65.22 ± 8.88 |
| Height (m, mean ± SD) | 1.72 ± 0.08 | 1.81 ± 0.06 | 1.68 ± 0.06 |
| BMI (kg/m2, mean ± SD) | 23.47 ± 3.52 | 24.94 ± 2.33 | 22.82 ± 3.77 |
| Smoking status (n, %) | |||
| Non-smokers | 90 (62.5) | 26 (59.1) | 64 (64) |
| Ex-smokers | 35 (24.3) | 11 (25.0) | 24 (24) |
| Occasional smokers | 19 (13.2) | 7 (15.9) | 12 (12) |
| Education (n, %) | |||
| University/college | |||
| up to 4 years | 30 (20.8) | 8 (18.2) | 22 (22) |
| >4 years | 114 (79.2) | 36 (81.8) | 78 (78) |
| Biomarkers | Time Group | Min | 5th | 25th | 50th | 75th | 90th | 95th | Max |
|---|---|---|---|---|---|---|---|---|---|
| MeP (ug) | 6:00–12:00 | 0.427 | 1.075 | 3.013 | 5.937 | 12.582 | 23.521 | 44.215 | 418 |
| 12:00–18:00 | 0.406 | 0.815 | 1.751 | 3.872 | 8.524 | 18.277 | 34.046 | 1990 | |
| 18:00–6:00 | 0.607 | 1.923 | 4.099 | 8.219 | 18.517 | 34.438 | 60.88 | 364 | |
| Overall | 0.406 | 2.801 | 5.955 | 12.715 | 1990 | ||||
| MiBP (ug) | 6:00–12:00 | 0.504 | 1.084 | 2.394 | 4.341 | 6.969 | 11.789 | 17.681 | 106 |
| 12:00–18:00 | 0.11 | 0.822 | 1.54 | 2.569 | 5.005 | 7.906 | 10.947 | 23.5 | |
| 18:00–6:00 | 0.917 | 2.214 | 3.907 | 5.738 | 9.371 | 17.15 | 24.399 | 78.4 | |
| Overall | 0.110 | 2.289 | 4.370 | 7.340 | 106.4 | ||||
| MnBP (ug) | 6:00–12:00 | 0.809 | 1.528 | 3.119 | 5.174 | 9.147 | 17.033 | 21.591 | 42.1 |
| 12:00–18:00 | 0.479 | 0.979 | 2.129 | 3.431 | 5.378 | 9.061 | 11.97 | 20.6 | |
| 18:00–6:00 | 1.476 | 2.537 | 5.036 | 8.036 | 14.621 | 21.362 | 24.603 | 30.5 | |
| Overall | 0.480 | 2.995 | 5.192 | 9.531 | 42.1 | ||||
| MBzP (ug) | 6:00–12:00 | 0.064 | 0.221 | 0.418 | 0.686 | 1.167 | 2.017 | 3.247 | 40.9 |
| 12:00–18:00 | 0.043 | 0.133 | 0.308 | 0.478 | 0.818 | 1.505 | 2.174 | 8.7 | |
| 18:00–6:00 | 0.166 | 0.363 | 0.710 | 1.068 | 1.911 | 2.545 | 3.532 | 40.6 | |
| Overall | 0.043 | 0.425 | 0.733 | 1.248 | 40.9 | ||||
| SumDEHP (ug) | 6:00–12:00 | 2.379 | 3.52 | 8.463 | 12.526 | 22.372 | 33.672 | 42.43 | 64.7 |
| 12:00–18:00 | 1.455 | 3.079 | 6.041 | 9.537 | 14.741 | 26.007 | 35.354 | 176.8 | |
| 18:00–6:00 | 2.665 | 6.971 | 12.979 | 19.201 | 30.559 | 44.156 | 58.501 | 127.7 | |
| Overall | 1.455 | 8.467 | 13.295 | 23.315 | 176.8 | ||||
| SumDiNP (ug) | 6:00–12:00 | 0.568 | 1.368 | 2.916 | 5.448 | 10.466 | 20.743 | 38.159 | 526 |
| 12:00–18:00 | 0.661 | 1.187 | 2.435 | 4.805 | 7.998 | 14.915 | 29.127 | 156 | |
| 18:00–6:00 | 0.644 | 2.645 | 5.196 | 8.271 | 15.653 | 37.221 | 69.239 | 1276 | |
| Overall | 0.568 | 3.185 | 6.219 | 11.136 | 1276 | ||||
| SumDINCH (ug) | 6:00–12:00 | 0.066 | 0.158 | 0.378 | 0.691 | 1.239 | 2.704 | 5.526 | 12.5 |
| 12:00–18:00 | 0.025 | 0.12 | 0.295 | 0.483 | 0.957 | 1.637 | 2.345 | 7.8 | |
| 18:00–6:00 | 0.124 | 0.222 | 0.555 | 0.941 | 1.654 | 2.762 | 4.389 | 93.3 | |
| Overall | 0.025 | 0.399 | 0.710 | 1.216 | 93.3 | ||||
| oh-MPHP (ug) | 6:00–12:00 | 0.005 | 0.02 | 0.078 | 0.196 | 0.381 | 0.582 | 0.771 | 2.7 |
| 12:00–18:00 | 0.007 | 0.017 | 0.07 | 0.16 | 0.299 | 0.507 | 0.684 | 1.18 | |
| 18:00–6:00 | 0.018 | 0.038 | 0.135 | 0.309 | 0.538 | 0.851 | 1.315 | 5.95 | |
| Overall | 0.005 | 0.084 | 0.208 | 0.410 | 5.95 | ||||
| MEPA (ug/L) | 6:00–12:00 | 0.816 | 1.512 | 3.394 | 6.305 | 37.192 | 99.038 | 158.275 | 18519 |
| 12:00–18:00 | 0.336 | 1.11 | 2.954 | 5.85 | 24.273 | 81.163 | 134.853 | 3960 | |
| 18:00–6:00 | 0.756 | 1.456 | 3.277 | 7.320 | 27.653 | 87.744 | 123.885 | 6728 | |
| Overall | 0.336 | 3.246 | 6.467 | 25.362 | 18519 | ||||
| ETPA (ug/L) | 6:00–12:00 | 0.024 | 0.164 | 0.520 | 1.235 | 3.432 | 14.118 | 26.195 | 761.7 |
| 12:00–18:00 | 0.026 | 0.144 | 0.390 | 1.083 | 3.194 | 12.177 | 36.465 | 271.1 | |
| 18:00–6:00 | 0.029 | 0.145 | 0.348 | 0.841 | 3.269 | 19.471 | 40.127 | 2038 | |
| Overall | 0.024 | 0.422 | 1.083 | 3.264 | 2038 | ||||
| PRPA (ug/L) | 6:00–12:00 | 0.008 | 0.012 | 0.024 | 0.251 | 2.827 | 20.076 | 46.206 | 384.9 |
| 12:00–18:00 | 0.008 | 0.012 | 0.024 | 0.095 | 1.159 | 10.660 | 19.738 | 225.7 | |
| 18:00–6:00 | 0.008 | 0.012 | 0.027 | 0.169 | 3.404 | 16.416 | 32.363 | 175.7 | |
| Overall | 0.008 | 0.024 | 0.173 | 2.366 | 384.9 | ||||
| BUPA (ug/L) | 6:00–12:00 | 0.014 | 0.018 | 0.032 | 0.065 | 0.179 | 0.528 | 1.083 | 9.82 |
| 12:00–18:00 | 0.016 | 0.019 | 0.041 | 0.074 | 0.177 | 0.347 | 0.585 | 5.45 | |
| 18:00–6:00 | 0.014 | 0.019 | 0.032 | 0.051 | 0.162 | 0.364 | 0.681 | 47.1 | |
| Overall | 0.014 | 0.035 | 0.065 | 0.176 | 47.1 | ||||
| BPA (ug/L) | 6:00–12:00 | 0.014 | 0.219 | 0.621 | 1.393 | 2.439 | 3.965 | 5.646 | 21.1 |
| 12:00–18:00 | 0.011 | 0.060 | 0.602 | 1.059 | 2.175 | 4.176 | 4.983 | 24.6 | |
| 18:00–6:00 | 0.014 | 0.184 | 0.678 | 1.170 | 1.965 | 3.774 | 5.319 | 19.6 | |
| Overall | 0.011 | 0.621 | 1.163 | 2.194 | 24.6 | ||||
| BPS (ug/L) | 6:00–12:00 | <LOD | LOD | <LOD | <LOD | <LOD | 0.501 | 1.083 | 2.8 |
| 12:00–18:00 | <LOD | <LOD | <LOD | <LOD | 0.182 | 0.455 | 0.693 | 7.04 | |
| 18:00–6:00 | <LOD | <LOD | <LOD | <LOD | 0.165 | 0.691 | 1.082 | 55.7 | |
| Overall | <LOD | <LOD | <LOD | <LOD | 0.138 | 55.7 | |||
| BPF (ug/L) | 6:00–12:00 | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | 6.0 |
| 12:00–18:00 | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | 7.7 | |
| 18:00–6:00 | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD | 0.580 | 12.8 | |
| Overall | <LOD | <LOD | <LOD | <LOD | 12.8 | ||||
| 4HNE (ug/L) | 6:00–12:00 | 3.028 | 3.262 | 3.763 | 4.042 | 4.36 | 4.583 | 4.784 | 5.6 |
| 12:00–18:00 | 2.766 | 3.289 | 3.813 | 4.083 | 4.458 | 4.771 | 5.026 | 5.8 | |
| 18:00–6:00 | 2.905 | 3.336 | 3.762 | 4.044 | 4.294 | 4.618 | 4.948 | 6.5 | |
| Overall | 2.766 | 3.775 | 4.048 | 4.375 | 6.5 |
| Log(4HNE) | ||||
|---|---|---|---|---|
| Parent EDCs | Biomarker | Estimate | 95% CI | q Value |
| Phthalates | MeP | 0.09 | 0.05–0.13 | <0.001 |
| MiBP | 0.14 | 0.09–0.20 | <0.001 | |
| MnBP | 0.143 | 0.088–0.199 | <0.001 | |
| MBzP | 0.111 | 0.059–0.163 | <0.001 | |
| SumDEHP | 0.183 | 0.125–0.240 | <0.001 | |
| SumDiNP | 0.103 | 0.061–0.146 | <0.001 | |
| SumDINCH | 0.113 | 0.065–0.161 | <0.001 | |
| oh-MPHP | 0.125 | 0.083–0.167 | <0.001 | |
| Parabens | MEPA | 0.13 | 0.10–0.17 | <0.001 |
| ETPA | 0.096 | 0.064–0.128 | <0.001 | |
| PRPA | 0.039 | 0.017–0.061 | <0.001 | |
| BUPA | 0.123 | 0.064–0.181 | <0.001 | |
| Bisphenols | BPA | 0.128 | 0.091–0.165 | <0.001 |
| BPS | −0.015 | −0.114–0.084 | 0.767 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abimbola, S.O.; Xeni, C.; Sakhi, A.K.; Heibati, B.; Husøy, T.; Dirven, H.; Makris, K.C. Diurnal Variation in Biomarkers of Exposure to Endocrine-Disrupting Chemicals and Their Association with Oxidative Damage in Norwegian Adults: The EuroMix Study. Toxics 2022, 10, 181. https://doi.org/10.3390/toxics10040181
Abimbola SO, Xeni C, Sakhi AK, Heibati B, Husøy T, Dirven H, Makris KC. Diurnal Variation in Biomarkers of Exposure to Endocrine-Disrupting Chemicals and Their Association with Oxidative Damage in Norwegian Adults: The EuroMix Study. Toxics. 2022; 10(4):181. https://doi.org/10.3390/toxics10040181
Chicago/Turabian StyleAbimbola, Samuel Olushola, Christina Xeni, Amrit Kaur Sakhi, Behzad Heibati, Trine Husøy, Hubert Dirven, and Konstantinos C. Makris. 2022. "Diurnal Variation in Biomarkers of Exposure to Endocrine-Disrupting Chemicals and Their Association with Oxidative Damage in Norwegian Adults: The EuroMix Study" Toxics 10, no. 4: 181. https://doi.org/10.3390/toxics10040181
APA StyleAbimbola, S. O., Xeni, C., Sakhi, A. K., Heibati, B., Husøy, T., Dirven, H., & Makris, K. C. (2022). Diurnal Variation in Biomarkers of Exposure to Endocrine-Disrupting Chemicals and Their Association with Oxidative Damage in Norwegian Adults: The EuroMix Study. Toxics, 10(4), 181. https://doi.org/10.3390/toxics10040181







