Abstract
Pediococcus acidilactici has gained research and commercial interest due to its outstanding probiotic properties, yet its survival during storage and consumption requires improvement. This study aims to enhance P. acidilactici survival using spray drying encapsulation. Different inlet air temperatures (120 °C, 150 °C, and 170 °C) and whey protein isolate (WPI):gum arabic (GA) ratios (1:1, 3:1, 1:3) were tested. Cell viability was significantly (p < 0.05) affected by the inlet temperature but not the WPI:GA ratio. Increasing the inlet temperature to 170 °C significantly decreased P. acidilactici viability by 1.36 log cycles, from 8.61 log CFU/g to 7.25 log CFU/g. The inlet temperature of 150 °C resulted in a powder yield (63.12%) higher than at 120 °C (58.97%), as well as significantly (p < 0.05) lower moisture content (5.71%) and water activity (aw 0.21). Viable cell counts in all encapsulated P. acidilactici were maintained at 5.24–6.75 log CFU/g after gastrointestinal tract (GIT) simulation, with WPI:GA of 3:1 and inlet temperature 150 °C having the smallest log reduction (0.3 log cycles). All samples containing different WPI:GA ratios maintained sufficient viability (>7 log CFU/g) during the first three weeks of storage at 25 °C. These results could provide insights for further developing P. acidilactici as commercial probiotic products.
1. Introduction
Probiotics are living microorganisms that provide health benefits to their host (or consumer) by maintaining the gut microbiome balance. Their benefits may include intestinal health improvement, immune response enhancement, and better serum cholesterol control [1]. Nowadays, probiotics are commercially available as dietary supplements (e.g., probiotic capsules and tablets [2]) and functional foods (e.g., yogurt [3], cheese [4], cereal [5], chocolate [6], and fruit juice [7]). However, to deliver their health benefits, a sufficient cell concentration must be administered in the food products. In general, it is recommended to incorporate 6–7 log probiotic cells per mL or gram product [1,8,9,10], and a total of 9–10 log probiotic cells should be consumed daily [1,11].
Pediococcus acidilactici are homofermentative gram-positive lactic acid bacteria known to have a great capacity to survive the harsh environment in the animal and human digestive systems (e.g., acidic pH, pepsin, and bile salt), enabling gut colonization [12,13]. Over the past decades, it has considerably gained research interest due to its potent probiotic characteristics, such as good antimicrobial activity [14,15], good adherence to intestinal cells [14,15], and the capability to produce useful metabolites such as bacteriocin and gamma-aminobutyric acid [16,17]. In addition, it has wide applications in the food industry, ranging from fish feed supplementation [18,19], starter culture for traditional sausage production [20], orange juice supplementation [21,22], and bio-preservative agents in food products [23]. However, during processing, storage, and consumption in the gastrointestinal tract (GIT), the probiotics are exposed to several environmental stresses, including heat, desiccation, and low pH, which reduce their viability [21,24,25,26,27]. The probiotics’ survival against such environmental stress could be improved by encapsulation [24,25].
Spray drying is a commonly used encapsulation technique [28,29,30], as it is simple, fast, cost-effective, and scalable compared to other drying encapsulation methods [31,32]. In spray drying encapsulation, probiotics are added to the encapsulation material solution, known as feed solution, then atomized into small particles, exposed to hot air (150 °C to 250 °C), and transformed into powders [33,34]. The resulting powder will contain probiotics encapsulated within the coating matrix that protects the cells from upcoming environmental stress [35]. However, spray drying exposes the probiotics to heat and desiccation stress, which could reduce the viable cell number [31,36,37]. Hence, selecting appropriate parameters used in spray drying is important to maintain a high encapsulation efficiency [38,39,40].
One of the critical parameters in spray drying is inlet air temperature, which refers to the pre-heated drying air entering the drying chamber [41]. Choosing a suitable inlet air temperature is critical in obtaining good quality powder, such as low moisture content and water activity, which is required to prevent microbial growth or contamination and maintain product stability during storage [38,40]. Studies by Flores et al. [39] and Ortega and Vandeker [40] have shown that high inlet air temperature is favourable as it results in low moisture content and water activity. However, such high temperature also induces viable cell loss as it gives relatively high stress that could damage the cell wall, DNA, and RNA and disrupt their metabolic activity [8,40,42].
Besides the inlet temperature, selecting an appropriate encapsulation material is also crucial to obtain a powder with high viable cell concentration. No single biopolymer can provide all the ideal criteria for encapsulation materials (e.g., edible, low-cost, idle in nature, and good physicochemical properties) [43]. Hence, two or three materials are often used to obtain synergic properties in maintaining a high viable probiotic cell count [44]. Whey protein isolate (WPI) and gum arabic (GA) have been shown to have good properties as spray drying encapsulation materials, as they are able to construct physically strong and stable matrices [45]. Furthermore, their interactions demonstrate excellent interfacial activity and emulsifying properties that can protect the probiotic cells during spray drying [46]. Another study demonstrates that WPI, in combination with GA, exhibited the highest probiotic survival during GIT simulation compared to WPI combined with other materials, such as locust bean gum and maltodextrin [47]. Moreover, using response surface methodology, the use of WPI and GA was predicted to encapsulate Lactobacillus acidophilus with a high encapsulation efficiency of 93.95% [44].
As of now, the effect of inlet air temperature and the use of WPI and GA as spray drying encapsulation material for P. acidilactici has never been studied. Hence, this study investigated the effect of varying inlet temperature (120 °C, 150 °C and 170 °C) and WPI:GA ratio (1:1, 3:1, and 1:3) on P. acidilactici survivability during spray drying, storage, and GIT simulation. Additionally, the production yield and physicochemical properties of all samples were analysed, including moisture content, water activity, Fourier-transform infrared spectroscopy (FT-IR), and scanning electron microscopy (SEM).
2. Materials and Methods
2.1. Materials and Culture
Encapsulation materials were food grade, and the other materials for survival and physicochemical analysis were analytical grades. Whey protein isolate 90 (WPI) (JFO store, Jakarta, Indonesia) and gum arabic (GA) (ALMA Chemical, Demak, Indonesia) were purchased using local e-commerce in Indonesia. Microbiological growth media used were de Man-Rogosa-Sharpe (MRS) broth (MERCK, Darmstadt, Germany) and MRS agar (MERCK, Darmstadt, Germany). For GIT simulation, the MRS broth was supplemented with glucose (MERCK, Darmstadt, Germany), KH2PO4 (MERCK, Darmstadt, Germany), CaCl2 (MERCK, Darmstadt, Germany), and KCl (MERCK, Darmstadt, Germany). The rehydration media for spray-dried probiotic and an additional supplement to gastrointestinal simulation media was NaCl (Himedia, Mumbai, India). The pH of gastrointestinal simulation media was maintained by adding HCl (MERCK, Darmstadt, Germany) and NaOH (ROFA, Bandung, Indonesia). P. acidilactici culture was obtained from Universitas Gadjah Mada (UGM), Food and Nutrition Culture Collection. The bacterial identity was confirmed through gram-staining and 16S rRNA sequencing.
2.2. Culture Preparation for Spray Drying
P. acidilactici from MRS agar was subcultured twice in fresh 50 mL MRS broth, followed by incubation at 30 °C for 18 h until it entered the late log phase. Then, 50 mL culture (OD600 0.7 equal to 7–8 log CFU/mL) was harvested and washed twice with 25 mL 0.9% NaCl solution. Cell harvest and wash were done by centrifugation at 2438× g, 25 °C, for 15 min. Lastly, the culture was concentrated by removing the supernatant, and the pellet was resuspended in 10 mL 0.9% NaCl solution to get a higher cell concentration (9–10 log CFU/mL).
2.3. Viable Cell Counting
The viable cell counting was performed using the Miles and Misra method with adjustments [48]. First, 100 µL culture or 1 g of spray-dried sample was transferred to 900 µL or 9 mL 0.9% NaCl solution, respectively, followed by serial dilution. Then three drops of 10 µL culture from each dilution were dropped onto the MRS agar and allowed to set. The agar plates were incubated at 30 °C for 36–48 h, and the number of colonies was calculated.
2.4. Spray Drying
Feed solution containing probiotic culture (1% v/v) and 20% w/v WPI-GA was prepared to be subjected to spray drying. The effect of inlet temperature was investigated by keeping the WPI-GA ratio fixed at 1:1, while varying the inlet temperature at 120 °C, 150 °C and 170 °C. To evaluate the effect of WPI:GA ratio, the inlet temperature was fixed at 150 °C, while WPI:GA ratio was varied to 1:3, 1:1, and 3:1.
After the culture was prepared (described in Section 2.2), feed solutions containing encapsulation material were prepared. A total of 200 g WPI and GA were dissolved in 1 L mineral water and the solution was homogenized using a hand blender (Bamix Deluxe hand blender, Mettlen, Switzerland) for 5 min at maximum speed. Once homogenized, the 10 mL culture prepared in Section 2.2 was added and the feed solution was homogenized for another 2 min. The spray drying was carried out using a pilot-scale spray dryer (LPG 5, Changzhou Huaihai Drying Equipment Co., Ltd., Changzhou, China). The solutions were fed into the chamber through a peristaltic pump at a constant flow rate of 15 rpm/min. Other parameters were fan speed at 45 Hz, atomisation at 250 Hz, and air hammer within 1 s every 20 s. Spray-dried samples were cooled down and further stored in a Ziplock plastic bag. The Ziplock bag was kept inside an aluminum bag, added with silica gel, and sealed with a heat sealer until further analysis.
2.5. Viability Loss
The viability loss was defined as a log reduction of viable cell concentration as described in Equation (1) [37,49,50]. N0 is the viable cell concentration (log CFU/g) before spray drying and Nt is the viable cell concentration (log CFU/g) after spray drying. For gastrointestinal simulation, N0 is the initial viable cell concentration (log CFU/g) before the simulation, and Nt is the viable concentration (log CFU/g) after the simulation.
Log reduction = log N0 − log Nt
2.6. Gastrointestinal Simulation
The survival during GIT simulation was studied to monitor viability loss during consumption. The method was adapted from a previous study with modifications [44]. In general, spray-dried and free cells of P. acidilactici (control) were sequentially exposed to simulated gastric juice (SGJ) for 2 h and simulated intestinal juice (SIJ) for 4 h. Firstly, the formulation for SGJ was prepared according to a study by Kulkarni et al. [51]. Briefly, MRS broth were added with glucose (3.5 g/L), NaCl (2.05 g/L), KH2PO4 (0.60 g/L), CaCl2 (0.11 g/L), and KCl (0.37 g/L), and then adjusted to pH 2.0 using 1 M HCl. Then, pepsin (from porcine stomach mucosa, Sigma Chemical Co., St. Louis, MO, USA) was added to the sterile SGJ stock by 13.3 mg/L. Sterile 25-mL Erlenmeyer flasks were prepared and filled with 9 mL of sterile SGJ. Into each 9 mL sterile SGJ solution, 1 g of spray-dried samples or 1 mL of free cells (control) were added, producing suspension that was homogenized using a vortex for 2 min. Viable cell counting was performed to obtain initial cell concentration before GIT simulation. Next, the suspensions were incubated in a shaker incubator (TOU-50N Orbital Shaker Incubator, MRC, Holon, Israel) for 2 h at 37 °C and 150 rpm for gastric juice simulation. Once the incubation in SGJ was done, the samples were neutralised with 1 M NaOH to pH 7, quenching the pepsin enzymatic reaction. Viable cell counting was performed to obtain viable cell concentration after SGJ simulation. Then, porcine bile (0.7% w/v; Sigma Chemical Co., St. Louis, MO, USA) was added to each flask and incubated at 37 °C and 150 rpm for 4 h for SIJ simulation. Once the incubation was complete, the last viable cell counting was conducted to obtain viable cell concentration after sequential SGJ and SIJ simulation.
2.7. Storage Test
P. acidilactici viability during storage was investigated to observe the viable cell reduction during storage at room temperature for 4 months. During the 1st month, the viable cell count of spray-dried P. acidilactici was measured every week, and then the frequency was changed to once every month. Viable cell counting was done as described in Section 2.3.
2.8. Physicochemical Analysis
The production yield was expressed in percentage, calculated using Equation (2). The mass of encapsulation material used in all samples was 200 g. After weighing, the powder was stored in a sealed aluminium foil bag until further analysis.
The moisture content analysis was adapted from a previous study [52]. A gram of sample was heated at 105 °C for 5 min and the average moisture content was measured using a moisture analyser (Ohaus® MB-45 moisture balances, Parsippany, NJ, USA). The water activity (aw) was determined by Pawkit Water Activity Meter (Decagon, Pullman, WA, USA). Samples were poured into the water activity measurement cup, and the water activity meter analysed the moisture content for 5 min [52,53].
2.9. Data Analysis
All the experiments were performed in triplicates. Data from the experiment were analysed by GraphPad Prism version 8.0.0 for Windows, GraphPad Software, San Diego. Ca, USA, www.graphad.com, accessed on 19 November 2022. The effects were considered significant at p < 0.05. Results were presented as the mean ± standard deviation. Statistical analysis on encapsulation efficiency and physicochemical properties (excluding FT-IR and powder morphology) was performed by one-way analysis of variance (ANOVA). Meanwhile, statistical analysis on gastrointestinal simulation and storage test was performed using a two-way analysis of variance (ANOVA). Tukey’s honestly significant difference test (p < 0.05) was conducted as a posteriori contrast after rejecting the null hypothesis.
3. Results and Discussion
3.1. Survivability of Spray-Dried P. acidilactici
Spray drying is mainly utilized to transform liquid into powder for extended shelf-life and ease of storage and transportation, and, compared to freeze drying, it is considered a low-cost process [32,33,34]. However, when it is used for probiotic encapsulation, spray drying has been associated with reduced cell viability as the inlet air temperature increases [54,55]. Such a viability loss is related to high heat stress resulting from drying, which may damage the structural integrity of cellular components and membranes [54,55,56]. This study investigates P. acidilactici viability after spray drying in an equal ratio of WPI:GA at different inlet air temperatures (120 °C, 150 °C, and 170 °C).
This study found that there was no significant difference (p > 0.05) in viability loss when the inlet temperature was increased from 120 °C to 150 °C (Table 1). S120 and S150 had 0.59 and 0.62 log cycle reductions, respectively. Meanwhile, the viability decreased significantly (p < 0.05) by 1.36 log cycles, when the temperature was increased to 170 °C. Viable cell loss induced by high inlet air temperature has also been reported in other studies [28,29,31,57]. Behboudi-Jobbehdar et al. [57] tested different inlet air temperatures (120 °C, 140 °C and 160 °C) and reported that the viability of Lactobacillus acidophilus spray-dried in a combination of maltodextrin, whey protein, and glucose decreased from 9.02 to 7.37 log CFU/g as the inlet temperature increased. A similar pattern was reported for L. acidophilus encapsulated in WPI-GA spray-dried at inlet temperatures of 100 °C, 120 °C and 140 °C [44].

Table 1.
Survivability of spray-dried P. acidilactici produced by different inlet air temperatures.
In addition to inlet temperature, selecting the appropriate encapsulation material also plays a critical role in protecting the probiotics during spray drying, storage, and GIT transit [21,28,29]. GA is one of the most widely studied probiotic encapsulating agents. Its protective effect has been observed during spray drying of multiple probiotic species, including L. acidophilus [39,44], Enterococcus faecalis [58], Saccharomyces cerevisiae var. boulardii [47], and Lactobacillus plantarum [59]. WPI is another commonly used probiotic encapsulating material known for its high resistance and stability against pepsin digestion, providing better protection against stomach conditions. To enhance its protective effect, GA and WPI are usually used together [44,59,60]. When combined, WPI and GA can create an emulsion that protects P. acidilactici from cellular injury during spray drying [60,61]. The resulting emulsion demonstrates suitable physicochemical properties, such as molecular weight, glass transition, crystallinity, diffusibility, film-forming properties, and feed solution viscosity, that are suitable for spray drying encapsulation [43,46,62]. Since the success of encapsulation depends on the WPI-GA ratio [44,59,60], this study tested different WPI-GA ratios (1:3, 1:1, and 3:1) using fixed inlet air temperature (150 °C).
As shown in Table 2, all WPI:GA ratios generated P. acidilactici powder with high viable cell counts (7.49–7.78 log CFU/g), and no significant difference (p > 0.05) in viability loss (0.58–1.11 log cycles) was observed between samples. Past studies had shown different success when WPI and GA were used for encapsulating probiotics. For example, a viable cell reduction of 1.43 log CFU/g was achieved by Leylak et al. [44] using a 1:0 WPI:GA ratio and inlet temperature of 120 °C. Meanwhile, Sharifi et al. [59] reported a higher log reduction of approximately 3 log CFU/g in L. plantarum spray-dried using WPI-GA alone or in combination with phytosterol. Similarly, Eratte et al. [60] reported about 5.4 and 4.1 log cycle loss of Lactobacillus casei spray-dried using WPI-GA alone and in combination with omega 3, respectively.

Table 2.
Survivability of spray-dried P. acidilactici produced by different WPI-GA ratios.
3.2. Production Yield, Moisture Content, and Water Activity of Spray-Dried P. acidilactici
Increasing the inlet air temperature was found to increase the spray-dried P. acidilactici yield (Table 3), which was consistent with the previous studies [63,64,65]. The lowest production yield was obtained with inlet air temperature 120 °C (58.97%), followed by 150 °C (63.12%) and 170 °C (69.62%). Since higher inlet air temperature is associated with a greater heat transfer efficiency, the probability of carrier material sticking onto the drying chamber wall is reduced, therefore increasing the production yield [66,67]. Increasing inlet air temperature was also found to reduce the powder’s moisture content and water activity (aw) (Table 2), which are highly desirable for a longer shelf-life [31,68,69,70]. As shown in Table 3, all spray-dried P. acidilactici had moisture content within a range of 4.66–6.80%, which was comparable to other studies [63,64,65] and considered suitable for storage at room temperature (25 °C) [71,72,73]. Furthermore, the aw of all spray-dried P. acidilactici fell within the range of 0.16–0.29, which is sufficient for preventing bacteria from being metabolically active until consumption [69]. According to the literature, probiotic powders are recommended to have aw value of around 0.25 to preserve its viability and prevent undesirable microbial growth [68,69,74].

Table 3.
Physicochemical properties of spray-dried P. acidilactici produced by different inlet air temperatures.
The production yield of all WPI-GA ratios ranges from 63.12 to 67.10%, and no significant differences (p > 0.05) were observed between samples (Table 4). The moisture content (4.66–5.91%) also fell within the acceptable range for room temperature (25 °C) storage with minimum viability loss [71,72,73]. The moisture content was found to increase with increasing GA proportion, which could be attributed to the water holding capacity of proteins (1.5–2.6%) contained in the GA [75,76]. Regardless, the aw between all samples showed no significant difference (p > 0.05), ranging from 0.17 to 0.21.

Table 4.
Physicochemical properties of spray-dried P. acidilactici produced in different WPI-GA ratios.
3.3. Survival of P. acidilactici in Simulated GIT
Encapsulation is expected to retain the probiotic viability during gastrointestinal digestion [44,56,62]. In this study, P. acidilactici spray-dried with various inlet air temperatures and WPI:GA ratios was subjected to a survivability test in simulated GIT, comprising sequential exposure to SGJ and SIJ. As shown in Table 5, no viable cells were detected in the non-spray-dried P. acidilactici after 2 h of incubation in SGJ (reduction of 8 log CFU/g). By the end of the simulation, S150 exhibited the lowest log reduction (0.50 log CFU/g), followed by S120 (0.77 log CFU/g) and S170 (0.78 log CFU/g). According to the literature, to a certain extent, a higher inlet air temperature could improve the survivability of spray-dried probiotics under all pH conditions [47,56,62]. Studies by Arslan et al. and Baghwat et al. [47,62] suggested that higher inlet temperature could affect the powder’s physical form by forming an impermeable and solid structure, providing better protection to the probiotics. Meanwhile, increasing the inlet temperature further could generate incomplete coverage, as previously observed by Ng et al. [77], decreasing the probiotics’ survivability in adverse conditions.

Table 5.
Viability of P. acidilactici spray-dried at different inlet air temperatures during GIT simulation.
The viable cell concentration of P. acidilactici encapsulated in different WPI-GA ratios during GIT simulation is shown in Table 6. All WPI-GA ratios maintained 5.62–6.41 log CFU/g of viable cells after 2 h incubation in SGJ. The viable cell reduction of 0.3–0.75 log CFU/g observed might have been caused by the porous nature of the encapsulation wall and possible WPI digestion by pepsin, allowing some acid to penetrate the matrix and cause cell injuries [62,78]. Higher WPI content in WG31 might have been able to withstand this phenomenon, thus retaining a high number of viable cells within the encapsulation matrix. Another possible contributing factor is the surface topography of the encapsulating matrix, as suggested by Doherty et al. [79]. The disrupted surface topography (such as dents and wrinkled surface) could increase the surface area of the encapsulation wall [79], which corresponds to higher powder diffusion and active components (the probiotic cell) release rate [80,81]. The encapsulation wall with low WPI content (WG13) might have experienced such a disrupted surface topography, verified by performing scanning electron microscopy (SEM) as described in Section 3.6.

Table 6.
Viability of P. acidilactici spray-dried in different WPI-GA ratios during GIT simulation.
After 4 h of incubation in SIJ, WG31 maintained stable viability as no significant change (p > 0.05) was observed from the initial counts. On the other hand, viable cell counts in WG11 and WG13 decreased significantly (p < 0.05) by 0.5 and 0.75 log cycles, respectively, by the end of the simulation. Bile is detrimental to bacteria, possibly affecting cell permeability and interactions between the membrane and its environment [62,82]. It disrupts the cell membrane by altering the lipid matrix in the cell membrane due to its detergent and lipophilic properties [83]. The bile extract also contains pancreatin and lipase, which are potentially harmful to bacteria [84]. The results of this study suggest that reducing the WPI:GA ratio to 1:3 (WG13) could not provide sufficient protection against bile extract in simulated SIJ. The inability of WG13 to withstand acid and pepsin digestion during the SGJ simulation could have promoted the diffusion of bile extract through the encapsulation wall during simulated SIJ.
3.4. Survival of Spray-Dried P. acidilactici during Storage
Storage testing is an essential evaluation to determine the product shelf life and ensure overall product quality over time. The International Dairy Federation and previous reports [1,8,9,10] suggest a minimum probiotic cell concentration of 7 log CFU/g should be retained until the end of the shelf life to maintain its product efficacy. Since drying causes damage to the cell membrane, proteins, and nucleic acids, preserving probiotics viability in the powder form over a long period of storage at ambient temperature remains a challenge. In this study, the viable cell counts of spray-dried P. acidilactici were evaluated over four months of storage at room temperature (25 °C). During the 1st month, the viability was measured weekly, and then the measurement was performed monthly.
The survival of P. acidilactici spray-dried at different inlet air temperature over 4 months of storage is presented in Figure 1. As depicted in Figure 1, the viability of P. acidilactici gradually decreased over time from 7.98–7.25 log CFU/g to 4.5–4.75 log CFU/g by the end of storage. A similar declining pattern was also observed in the previous studies [29,44,85]. Viable cell counts reached below 7 log CFU/g more rapidly with the increasing inlet temperature, which correlates with lower initial counts in higher inlet temperature.
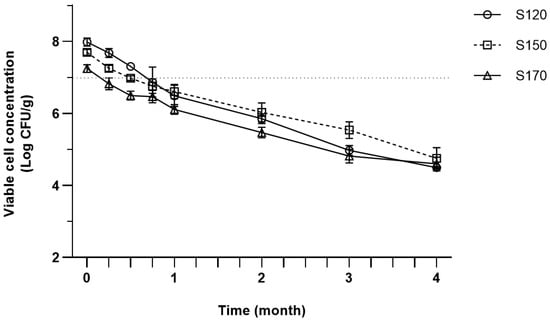
Figure 1.
Viable cell concentration of spray-dried P. acidilactici produced by different inlet air temperatures during storage at room temperature (25 °C). Error bars represent ± Standard Error of the Mean (SEM) of three replicates (n = 3). Dotted lines in y axis at 7 log CFU/g shows the suggested minimum concentration of viable probiotics that should be incorporated in the final product to exert its optimum health benefits.
All samples with different WPI:GA ratios also follow a similar declining trend during four months of storage (Figure 2). However, all samples maintained high viability above 7 log CFU/g until the 3rd week of storage, then continuously decreased to 4.75–5.24 log CFU/g by the end of the storage test.
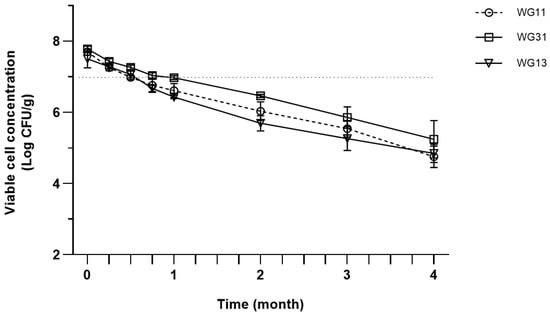
Figure 2.
Viable cell concentration of spray-dried P. acidilactici produced in different ratios of encapsulation matrices during storage at room temperature (25 °C). Error bars represent ± standard error of the mean (SEM) of three replicates (n = 3).
The survival of probiotics powder during storage is associated with the storage temperature, and aw. Abe et al. [86] found that the viability of B. longum powder increases with decreasing storage temperature and aw, while high survivability at 25 °C could be achieved with aw < 0.16. These findings suggest that the aw of the spray-dried P. acidilactici obtained in this study might not be low enough to prevent viability loss during storage at 25 °C. In addition, high survivability could be achieved by increasing the powder’s initial viable cell counts in the powder as well as keeping the powder at a lower storage temperature (4–10 °C) [29,44,85,87].
3.5. FT-IR Analysis of Spray-Dried P. acidilactici
The FT-IR spectra of raw WPI and GA powder are illustrated in Figure 3. The FT-IR spectra of raw WPI powder (WPI) showed two weak peaks, indicated by high transmittance at 80–90% in the functional group region. The peak around 3260–3280 cm−1 is attributable to the amine N-H stretching [59] and 2920–2930 cm−1 to the alkyl C-H stretch. The raw GA powder (GA) spectra have two peaks at the functional region with slightly lower transmittance and a broader peak than the WPI profile. The peak within 3500–3000 cm−1 may be attributed to the O-H stretching of alcohol/phenol, and the peak around 2892 cm−1 is closely related to carboxylic groups. The result of the WPI profile is consistent with the previous study by Bhagwat et al. [62], Sharifi et al. [59], and Rajam and Anandharamakrishnan [36], whereas GA is consistent with studies by Sharifi et al. [59], Santos et al. [88], and Daoub et al. [89].
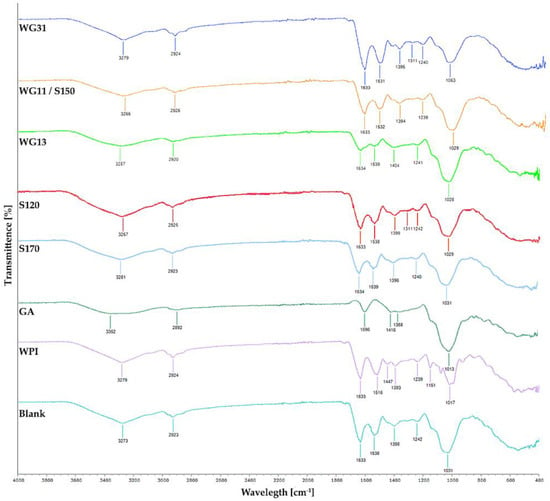
Figure 3.
FTIR spectra of encapsulation matrices and spray-dried P. acidilactici that were produced in the different inlet air temperature and ratio of encapsulation matrices. WPI: raw Whey Protein Isolate, GA: raw Gum Arabic, Blank: spray-dried WPI:GA at 1:1 ratio.
Apart from the raw encapsulation powder profile, raw WPI:GA (1:1) solution (blank) was spray-dried to identify any structural changes between the biopolymers and to check whether it is retained after spray drying. The blank spectra show WPI dominance in its functional region, while the fingerprint region indicates WPI dominance in 1700–1250 cm−1 and GA dominance in the 1250–900 cm−1 region. This result suggests no interaction between the wall material and the WPI-GA compound is retained after spray drying.
The FT-IR spectra of P. acidilactici spray-dried with different inlet air temperatures are also presented in Figure 3. It is shown that the FT-IR spectra follow a similar profile observed in blank and other WPI-GA combinations reported in the previous study [59], indicating no interaction between wall material and probiotics. Bonds mentioned previously could be seen in all samples which indicate that different inlet air temperature has no impact on the chemical structure of encapsulation materials as well. Similar profiles were also found in the FT-IR spectra of spray-dried P. acidilactici samples containing different WPI:GA ratios (Figure 3). A slight change in peak widths in the functional region (around 3500–3000 cm−1) corresponds to the increasing GA ratios. This result confirmed that WPI and GA structures were retained after spray drying. Additionally, there is no chemical interaction between the cells and the encapsulation material, comparing the blank and spray-dried P. acidilactici sample.
3.6. SEM Analysis
The morphology of spray-dried P. acidilactici was examined using SEM as shown in Figure 4. Mechanical defects such as fractures, cracks, or holes are unfavourable as they could expose the bacteria to oxidation and lead to viable loss [90]. Surface morphology is often found to be affected by the drying condition, the composition of the feed solution, and the total solid content used in spray drying [91,92]. In this study, the morphology was analysed for the P. acidilactici spray-dried at the inlet temperature of 150 °C varying in WPI:GA ratios, since 150 °C could produce a powder with high production yield and good physicochemical properties while retaining high viable cell count.

Figure 4.
SEM results of spray-dried P. acidilactici in different WPI:GA ratios.
As depicted in Figure 4, all samples were able to form separated microcapsules without any aggregation between particles. The spray-dried powder mainly contains larger particle sizes between 20–50 μm. WG31 shows a smooth spherical surface, which could be due to the difference in surface tension and viscosity of the final feed solution. In a previous study, WPI was reported to have a lower surface tension than GA [46]. In addition, when WPI and GA were mixed, a higher WPI ratio promoted lower surface tension in the final suspension. This surface tension is closely related to viscosity, a key factor affecting the particle morphology in spray drying [91]. Therefore, the lower viscosity in the WG31, resulting from its lower surface tension, may contribute to its smooth morphological feature.
On the contrary, more dents, wrinkled surfaces, and blow-holes were present in samples WG13 and WG11, potentially due to the increased viscosity of the feed solution with the increasing concentration of GA [93]. During drying, increased viscosity lowers the diffusion rate of water to the surface allowing solid crust formation while some moisture is still trapped inside. Upon further drying the vapor forced its way out by creating holes in the crust. These findings suggest that equal or higher GA ratios were not favourable as they produce wrinkled surface encapsulation, blow-holes, and shredded particles. This suggests that the addition of GA in a low ratio helps WPI withstand the mechanical stress during spray drying.
4. Conclusions
This study investigates the effect of inlet air temperature (120 °C, 150 °C and 170 °C) and WPI:GA ratio (1:1, 3:1, 1:3) on spray-dried P. acidilactici survivability and physicochemical properties. The results show that increasing the inlet air temperature was favourable in terms of production yield, moisture content, and aw. However, increasing the inlet temperature to 170 °C could lead to a significant viability loss. Unlike the inlet air temperature, the effect of WPI:GA ratio on the survival of spray-dried cells, yield, moisture content, and aw was not apparent. Furthermore, increasing the inlet air temperature to 150 °C resulted in the lowest viable cell reduction (0.5 log cycle) after the GIT simulation. For samples containing various WPI:GA concentrations, the viable cell reduction after simulated GIT was significantly lower when a higher WPI proportion (WG11 and WG31) was used. According to the SEM analysis, samples with higher WPI content exhibited a smoother spherical surface, while those containing a higher GA content demonstrated more dents, wrinkled surfaces, and blow-holes. During storage at 25 °C, viable cell counts decreased to below 7 log CFU/g more rapidly as the inlet temperature increased, which correlates with the encapsulation efficiency. Meanwhile, varying WPI:GA ratios did not influence the survivability of spray-dried P. acidilactici during storage. FT-IR spectra indicated no chemical interaction between wall materials and P. acidilactici. This study confirms that selecting the appropriate inlet air temperature and wall material ratio is vital in obtaining P. acidilactici powder with good physicochemical properties, high encapsulation efficiency, and survivability against gastrointestinal and storage conditions. Future investigations could focus on the effect of spray drying parameters on P. acidilactici functionalities.
Author Contributions
Conceptualization, K.K., I.T.P. and P.V.P.D.; methodology, G.D.T., L.M., K.K., I.T.P. and P.V.P.D.; validation, K.K., I.T.P., L.P.P., Y.H.H. and P.V.P.D.; formal analysis, G.D.T. and L.M.; investigation, G.D.T. and L.M.; resources, G.D.T. and P.V.P.D.; data curation, G.D.T. and L.M.; writing—original draft preparation, G.D.T., L.M. and M.D.B.; writing—review and editing, M.D.B., K.K., I.T.P., L.P.P., Y.H.H. and P.V.P.D.; visualization, G.D.T. and L.M.; supervision, M.D.B., P.V.P.D., K.K., I.T.P., L.P.P. and Y.H.H.; project administration, M.D.B., K.K., I.T.P., C.S.Y.L. and P.V.P.D.; funding acquisition, M.D.B., K.K., I.T.P., C.S.Y.L. and P.V.P.D. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by Indonesian Ministry of Research and Technology/National Research and Innovation Agency, grant number 698/LL3/AK.04/2022.
Acknowledgments
The authors acknowledge Ribath Faruqi and Jonathan for their assistance during the spray drying process.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kechagia, M.; Basoulis, D.; Konstantopoulou, S.; Dimitriadi, D.; Gyftopoulou, K.; Skarmoutsou, N.; Fakiri, E.M. Health Benefits of Probiotics: A Review. ISRN Nutr. 2013, 2013, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Huq, T.; Vu, K.D.; Riedl, B.; Bouchard, J.; Han, J.; Lacroix, M. Development of Probiotic Tablet Using Alginate, Pectin, and Cellulose Nanocrystals as Excipients. Cellulose 2016, 23, 1967–1978. [Google Scholar] [CrossRef]
- Lourens-Hattingh, A.; Viljoen, B.C. Yogurt as Probiotic Carrier Food. Int. Dairy J. 2001, 11, 1–17. [Google Scholar] [CrossRef]
- Rolim, F.R.L.; Freitas Neto, O.C.; Oliveira, M.E.G.; Oliveira, C.J.B.; Queiroga, R.C.R.E. Cheeses as Food Matrixes for Probiotics: In Vitro and in Vivo Tests. Trends Food Sci. Technol. 2020, 100, 138–154. [Google Scholar] [CrossRef]
- Ogunremi, O.R.; Agrawal, R.; Sanni, A.I. Development of Cereal-based Functional Food Using Cereal-mix Substrate Fermented with Probiotic Strain—Pichia Kudriavzevii OG32. Food Sci. Nutr. 2015, 3, 486–494. [Google Scholar] [CrossRef]
- Mirković, M.; Seratlić, S.; Kilcawley, K.; Mannion, D.; Mirković, N.; Radulović, Z. The Sensory Quality and Volatile Profile of Dark Chocolate Enriched with Encapsulated Probiotic Lactobacillus Plantarum Bacteria. Sensors 2018, 18, 2570. [Google Scholar] [CrossRef]
- Dias, C.O.; dos Santos Opuski de Almeida, J.; Pinto, S.S.; de Oliveira Santana, F.C.; Verruck, S.; Müller, C.M.O.; Prudêncio, E.S.; de Mello Castanho Amboni, R.D. Development and Physico-Chemical Characterization of Microencapsulated Bifidobacteria in Passion Fruit Juice: A Functional Non-Dairy Product for Probiotic Delivery. Food Biosci. 2018, 24, 26–36. [Google Scholar] [CrossRef]
- Tripathi, M.K.; Giri, S.K. Probiotic Functional Foods: Survival of Probiotics during Processing and Storage. J. Funct. Foods 2014, 9, 225–241. [Google Scholar] [CrossRef]
- Corona-Hernandez, R.I.; Álvarez-Parrilla, E.; Lizardi-Mendoza, J.; Islas-Rubio, A.R.; de la Rosa, L.A.; Wall-Medrano, A. Structural Stability and Viability of Microencapsulated Probiotic Bacteria: A Review. Compr. Rev. Food Sci. Food Safety 2013, 12, 614–628. [Google Scholar] [CrossRef]
- Chean, S.X.; Hoh, P.Y.; How, Y.H.; Nyam, K.L.; Pui, L.P. Microencapsulation of Lactiplantibacillus Plantarum with Inulin and Evaluation of Survival in Simulated Gastrointestinal Conditions and Roselle Juice. Braz. J. Food Technol. 2021, 24. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics Consensus Statement on the Scope and Appropriate Use of the Term Probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Villamil, L.; Figueras, A.; Planas, M.; Novoa, B. Pediococcus Acidilactici in the Culture of Turbot (Psetta Maxima) Larvae: Administration Pathways. Aquaculture 2010, 307, 83–88. [Google Scholar] [CrossRef]
- Yavorov-Dayliev, D.; Milagro, F.I.; Ayo, J.; Oneca, M.; Aranaz, P. Pediococcus Acidilactici CECT9879 (PA1c) Counteracts the Effect of a High-Glucose Exposure in C. Elegans by Affecting the Insulin Signaling Pathway (IIS). Int. J. Mol. Sci. 2022, 23, 2689. [Google Scholar] [CrossRef]
- Abbasiliasi, S.; Tan, J.S.; Bashokouh, F.; Ibrahim, T.A.T.; Mustafa, S.; Vakhshiteh, F.; Sivasamboo, S.; Ariff, A.B. In Vitro Assessment of Pediococcus Acidilactici Kp10 for Its Potential Use in the Food Industry. BMC Microbiol. 2017, 17, 121. [Google Scholar] [CrossRef]
- Hasan, M.T.; Je Jang, W.; Lee, J.M.; Lee, B.-J.; Hur, S.W.; Gu Lim, S.; Kim, K.W.; Han, H.-S.; Kong, I.-S. Effects of Immunostimulants, Prebiotics, Probiotics, Synbiotics, and Potentially Immunoreactive Feed Additives on Olive Flounder (Paralichthys Olivaceus): A Review. Rev. Fish. Sci. Aquac. 2019, 27, 417–437. [Google Scholar] [CrossRef]
- Porto, M.C.W.; Kuniyoshi, T.M.; Azevedo, P.O.S.; Vitolo, M.; Oliveira, R.P.S. Pediococcus Spp.: An Important Genus of Lactic Acid Bacteria and Pediocin Producers. Biotechnol. Adv. 2017, 35, 361–374. [Google Scholar] [CrossRef]
- Marlida, Y.; Wizna, W.; Jamsari, J.; Mirzah, M.; Anggraini, L. Optimization of Nutrient Medium for Pediococcus Acidilactici DS15 to Produce GABA. J. Worlds Poult Res. 2019, 9, 139–146. [Google Scholar] [CrossRef]
- Merrifield, D.L.; Bradley, G.; Harper, G.M.; Baker, R.T.M.; Munn, C.B.; Davies, S.J. Assessment of the Effects of Vegetative and Lyophilized Pediococcus Acidilactici on Growth, Feed Utilization, Intestinal Colonization and Health Parameters of Rainbow Trout (Oncorhynchus Mykiss Walbaum). Aquac. Nutr. 2011, 17, 73–79. [Google Scholar] [CrossRef]
- Standen, B.T.; Rawling, M.D.; Davies, S.J.; Castex, M.; Foey, A.; Gioacchini, G.; Carnevali, O.; Merrifield, D.L. Probiotic Pediococcus Acidilactici Modulates Both Localised Intestinal- and Peripheral-Immunity in Tilapia (Oreochromis Niloticus). Fish Shellfi. Immunol. 2013, 35, 1097–1104. [Google Scholar] [CrossRef]
- Ruiz-Moyano, S.; Martín, A.; Benito, M.J.; Hernández, A.; Casquete, R.; de Guia Córdoba, M. Application of Lactobacillus Fermentum HL57 and Pediococcus Acidilactici SP979 as Potential Probiotics in the Manufacture of Traditional Iberian Dry-Fermented Sausages. Food Microbiol. 2011, 28, 839–847. [Google Scholar] [CrossRef]
- Barbosa, J.; Borges, S.; Amorim, M.; Pereira, M.J.; Oliveira, A.; Pintado, M.E.; Teixeira, P. Comparison of Spray Drying, Freeze Drying and Convective Hot Air Drying for the Production of a Probiotic Orange Powder. J. Funct. Foods 2015, 17, 340–351. [Google Scholar] [CrossRef]
- Cristiny de Oliveira Vieira, K.; da Silva Ferreira, C.; Toso Bueno, E.B.; de Moraes, Y.A.; Campagnolo Gonçalves Toledo, A.C.; Nakagaki, W.R.; Pereira, V.C.; Winkelstroter, L.K. Development and Viability of Probiotic Orange Juice Supplemented by Pediococcus Acidilactici CE51. LWT 2020, 130, 109637. [Google Scholar] [CrossRef]
- İncili, G.K.; Karatepe, P.; İlhak, O.İ. Effect of Chitosan and Pediococcus Acidilactici on E. Coli O157:H7, Salmonella Typhimurium and Listeria Monocytogenes in Meatballs. LWT 2020, 117, 108706. [Google Scholar] [CrossRef]
- Sangami, R.; Radhai Sri, S. Emerging Trends in Improving Viability, Advanced Stability Techniques and Health Claims of Healthy Microbiome—The Probiotics. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 194–200. [Google Scholar] [CrossRef][Green Version]
- Fiocco, D.; Longo, A.; Arena, M.P.; Russo, P.; Spano, G.; Capozzi, V. How Probiotics Face Food Stress: They Get by with a Little Help. Crit. Rev. Food Sci. Nutr. 2020, 60, 1552–1580. [Google Scholar] [CrossRef]
- Fernandez, B.; Hammami, R.; Savard, P.; Jean, J.; Fliss, I. Pediococcus Acidilactici UL5 and Lactococcus Lactis ATCC 11454 Are Able to Survive and Express Their Bacteriocin Genes under Simulated Gastrointestinal Conditions. J. Appl. Microbiol. 2014, 116, 677–688. [Google Scholar] [CrossRef]
- Ranadheera, C.S.; Evans, C.A.; Baines, S.K.; Balthazar, C.F.; Cruz, A.G.; Esmerino, E.A.; Freitas, M.Q.; Pimentel, T.C.; Wittwer, A.E.; Naumovski, N.; et al. Probiotics in Goat Milk Products: Delivery Capacity and Ability to Improve Sensory Attributes. Compr. Rev. Food Sci. Food Saf. 2019, 18, 867–882. [Google Scholar] [CrossRef]
- Fu, N.; Huang, S.; Xiao, J.; Chen, X.D. Producing Powders Containing Active Dry Probiotics With the Aid of Spray Drying. Adv. Food Nutr. Res. 2018, 85, 211–262. [Google Scholar]
- Lipan, L.; Rusu, B.; Sendra, E.; Hernández, F.; Vázquez-Araújo, L.; Vodnar, D.C.; Carbonell-Barrachina, Á.A. Spray Drying and Storage of Probiotic-enriched Almond Milk: Probiotic Survival and Physicochemical Properties. J. Sci. Food Agric. 2020, 100, 3697–3708. [Google Scholar] [CrossRef]
- Santos Monteiro, S.; Albertina Silva Beserra, Y.; Miguel Lisboa Oliveira, H.; Pasquali, M. Production of Probiotic Passion Fruit (Passiflora Edulis Sims f. Flavicarpa Deg.) Drink Using Lactobacillus Reuteri and Microencapsulation via Spray Drying. Foods 2020, 9, 335. [Google Scholar] [CrossRef]
- Huang, S.; Vignolles, M.-L.; Chen, X.D.; le Loir, Y.; Jan, G.; Schuck, P.; Jeantet, R. Spray Drying of Probiotics and Other Food-Grade Bacteria: A Review. Trends Food Sci. Technol. 2017, 63, 1–17. [Google Scholar] [CrossRef]
- Tao, T.; Ding, Z.; Hou, D.; Prakash, S.; Zhao, Y.; Fan, Z.; Zhang, D.; Wang, Z.; Liu, M.; Han, J. Influence of Polysaccharide as Co-Encapsulant on Powder Characteristics, Survival and Viability of Microencapsulated Lactobacillus Paracasei Lpc-37 by Spray Drying. J. Food Eng. 2019, 252, 10–17. [Google Scholar] [CrossRef]
- Santos, D.; Mauricio, A.C.; Sencadas, V.; Santos, J.D.; Fernandes, M.H.; Gomes, P.S. Spray Drying: An Overview. Biomaterials-Physics and Chemistry-New Edition; Pignatello, R., Musumeci, T., Eds.; IntechOpen: London, UK, 2018; pp. 9–35. [Google Scholar]
- Selvamuthukumaran, M. Handbook on Spray Drying Applications for Food Industries, 1st ed.; Selvamuthukumaran, M., Ed.; CRC Press: Boca Raton, FL, USA, 2019; ISBN 9780429055133. [Google Scholar]
- Makinen, K.; Berger, B.; Bel-Rhlid, R.; Ananta, E. Science and Technology for the Mastership of Probiotic Applications in Food Products. J. Biotechnol. 2012, 162, 356–365. [Google Scholar] [CrossRef]
- Rajam, R.; Anandharamakrishnan, C. Microencapsulation of Lactobacillus Plantarum (MTCC 5422) with Fructooligosaccharide as Wall Material by Spray Drying. LWT Food Sci. Technol. 2015, 60, 773–780. [Google Scholar] [CrossRef]
- Fazilah, N.F.; Hamidon, N.H.; Ariff, A.B.; Khayat, M.E.; Wasoh, H.; Halim, M. Microencapsulation of Lactococcus Lactis Gh1 with Gum Arabic and Synsepalum Dulcificum via Spray Drying for Potential Inclusion in Functional Yogurt. Molecules 2019, 24, 1422. [Google Scholar] [CrossRef]
- Kandansamy, K.; Somasundaram, P.D. Microencapsulation of Colors by Spray Drying—A Review. Int. J. Food Eng. 2012, 8. [Google Scholar] [CrossRef]
- Flores-Belmont, I.A.; Palou, E.; López-Malo, A.; Jiménez-Munguía, M.T. Simple and Double Microencapsulation of Lactobacillus Acidophilus with Chitosan Using Spray Drying. Int. J. Food Stud. 2015, 4. [Google Scholar] [CrossRef]
- Ortega, R.; Vandeker, V. Comparative Viability of Spray Dried Lactobacilli Affected by Different Protective Agents and Storage Conditions. Master Thesis, Louisiana State University and Agricultural & Mechanical College, Baton Rouge, LA, USA, 2017. [Google Scholar]
- Chew, S.-C.; Tan, C.-H.; Pui, L.-P.; Chong, P.N.; Gunasekaran, B.; Lin, N.K. Encapsulation Technologies: A Tool for Functional Foods Development. Int. J. Innov. Technol. Explor. Eng. 2019, 8, 154–162. [Google Scholar]
- Perdana, J.; Bereschenko, L.; Fox, M.B.; Kuperus, J.H.; Kleerebezem, M.; Boom, R.M.; Schutyser, M.A.I. Dehydration and Thermal Inactivation of Lactobacillus Plantarum WCFS1: Comparing Single Droplet Drying to Spray and Freeze Drying. Food Res. Int. 2013, 54, 1351–1359. [Google Scholar] [CrossRef]
- Chandralekha, A.; Rani, A.; Tavanandi, H.A.; Amrutha, N.; Hebbar, U.; Raghavarao, K.S.M.S. Role of Carrier Material in Encapsulation of Yeast (Saccharomyces cerevisiae) by Spray Drying. Dry. Technol. 2017, 35, 1029–1042. [Google Scholar] [CrossRef]
- Leylak, C.; Özdemir, K.S.; Gurakan, G.C.; Ogel, Z.B. Optimisation of Spray Drying Parameters for Lactobacillus Acidophilus Encapsulation in Whey and Gum Arabic: Its Application in Yoghurt. Int. Dairy J. 2021, 112, 104865. [Google Scholar] [CrossRef]
- Krunić, T.Ž.; Obradović, N.S.; Rakin, M.B. Application of Whey Protein and Whey Protein Hydrolysate as Protein Based Carrier for Probiotic Starter Culture. Food Chem. 2019, 293, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.; Aserin, A.; Ben Ishai, P.; Garti, N. Interactions between Whey Protein Isolate and Gum Arabic. Colloids Surf. B Biointerfaces 2010, 79, 377–383. [Google Scholar] [CrossRef]
- Arslan, S.; Erbas, M.; Tontul, I.; Topuz, A. Microencapsulation of Probiotic Saccharomyces Cerevisiae Var. Boulardii with Different Wall Materials by Spray Drying. LWT Food Sci. Technol. 2015, 63, 685–690. [Google Scholar] [CrossRef]
- Miles, A.A.; Misra, S.S.; Irwin, J.O. The Estimation of the Bactericidal Power of the Blood. Epidemiol. Infect. 1938, 38, 732–749. [Google Scholar] [CrossRef] [PubMed]
- Eckert, C.; Serpa, V.G.; Felipe dos Santos, A.C.; Marinês da Costa, S.; Dalpubel, V.; Lehn, D.N.; Volken de Souza, C.F. Microencapsulation of Lactobacillus Plantarum ATCC 8014 through Spray Drying and Using Dairy Whey as Wall Materials. LWT Food Sci. Technol. 2017, 82, 176–183. [Google Scholar] [CrossRef]
- Aponte, M.; Troianiello, G.D.; di Capua, M.; Romano, R.; Blaiotta, G. Impact of Different Spray-Drying Conditions on the Viability of Wine Saccharomyces Cerevisiae Strains. World J. Microbiol. Biotechnol. 2016, 32, 13. [Google Scholar] [CrossRef][Green Version]
- Kulkarni, S.; Haq, S.F.; Samant, S.; Sukumaran, S. Adaptation of Lactobacillus Acidophilus to Thermal Stress Yields a Thermotolerant Variant Which Also Exhibits Improved Survival at PH 2. Probiotics Antimicrob. Proteins 2018, 10, 717–727. [Google Scholar] [CrossRef]
- Femat-Castañeda, C.; Chávez-Rodríguez, A.; Chávez-Rodríguez, A.M.; Flores-Martínez, H.; Farías-Cervantes, V.S.; Andrade-González, I. Effect of Agave Fructans and Maltodextrin on Zn 2+ Chlorophyll Microencapsulation by Spray Drying. J. Food Qual. 2019, 2019, 6312584. [Google Scholar] [CrossRef]
- Gallagher, E.; Kenny, S.; Arendt, E.K. Impact of Dairy Protein Powders on Biscuit Quality. Eur. Food Res. Technol. 2005, 221, 237–243. [Google Scholar] [CrossRef]
- Foerst, P.; Kulozik, U.; Schmitt, M.; Bauer, S.; Santivarangkna, C. Storage Stability of Vacuum-Dried Probiotic Bacterium Lactobacillus Paracasei F19. Food Bioprod. Process. 2012, 90, 295–300. [Google Scholar] [CrossRef]
- Lapsiri, W.; Bhandari, B.; Wanchaitanawong, P. Viability of Lactobacillus Plantarum TISTR 2075 in Different Protectants during Spray Drying and Storage. Dry. Technol. 2012, 30, 1407–1412. [Google Scholar] [CrossRef]
- Arslan-Tontul, S.; Erbas, M. Single and Double Layered Microencapsulation of Probiotics by Spray Drying and Spray Chilling. LWT Food Sci. Technol. 2017, 81, 160–169. [Google Scholar] [CrossRef]
- Behboudi-Jobbehdar, S.; Soukoulis, C.; Yonekura, L.; Fisk, I. Optimization of Spray-Drying Process Conditions for the Production of Maximally Viable Microencapsulated L. Acidophilus NCIMB 701748. Dry. Technol. 2013, 31, 1274–1283. [Google Scholar] [CrossRef]
- Rishabh, D.; Athira, A.; Preetha, R.; Nagamaniammai, G. Freeze Dried Probiotic Carrot Juice Powder for Better Storage Stability of Probiotic. J. Food Sci. Technol. 2021, 58, 1–9. [Google Scholar] [CrossRef]
- Sharifi, S.; Rezazad-Bari, M.; Alizadeh, M.; Almasi, H.; Amiri, S. Use of Whey Protein Isolate and Gum Arabic for the Co-Encapsulation of Probiotic Lactobacillus Plantarum and Phytosterols by Complex Coacervation: Enhanced Viability of Probiotic in Iranian White Cheese. Food Hydrocoll. 2021, 113, 106496. [Google Scholar] [CrossRef]
- Eratte, D.; McKnight, S.; Gengenbach, T.R.; Dowling, K.; Barrow, C.J.; Adhikari, B.P. Co-Encapsulation and Characterisation of Omega-3 Fatty Acids and Probiotic Bacteria in Whey Protein Isolate–Gum Arabic Complex Coacervates. J. Funct. Foods 2015, 19, 882–892. [Google Scholar] [CrossRef]
- Khem, S.; Bansal, V.; Small, D.M.; May, B.K. Comparative Influence of PH and Heat on Whey Protein Isolate in Protecting Lactobacillus Plantarum A17 during Spray Drying. Food Hydrocoll. 2016, 54, 162–169. [Google Scholar] [CrossRef]
- Bhagwat, A.; Bhushette, P.; Annapure, U.S. Spray Drying Studies of Probiotic Enterococcus Strains Encapsulated with Whey Protein and Maltodextrin. Beni-Suef Univ. J. Basic Appl. Sci. 2020, 9, 33. [Google Scholar] [CrossRef]
- Goula, A.M.; Adamopoulos, K.G. Spray Drying of Tomato Pulp in Dehumidified Air: II. The Effect on Powder Properties. J. Food Eng. 2005, 66, 35–42. [Google Scholar] [CrossRef]
- Tonon, R.V.; Brabet, C.; Hubinger, M.D. Influence of Process Conditions on the Physicochemical Properties of Açai (Euterpe Oleraceae Mart.) Powder Produced by Spray Drying. J. Food Eng. 2008, 88, 411–418. [Google Scholar] [CrossRef]
- Tontul, I.; Topuz, A. Spray-Drying of Fruit and Vegetable Juices: Effect of Drying Conditions on the Product Yield and Physical Properties. Trends Food Sci. Technol. 2017, 63, 91–102. [Google Scholar] [CrossRef]
- Fazaeli, M.; Emam-Djomeh, Z.; Kalbasi Ashtari, A.; Omid, M. Effect of Spray Drying Conditions and Feed Composition on the Physical Properties of Black Mulberry Juice Powder. Food Bioprod. Process. 2012, 90, 667–675. [Google Scholar] [CrossRef]
- Fazaeli, M.; Emam-Djomeh, Z.; Yarmand, M.S. Influence of Black Mulberry Juice Addition and Spray Drying Conditions on Some Physical Properties of Ice Cream Powder. Int. J. Food Eng. 2016, 12, 277–285. [Google Scholar] [CrossRef]
- Willaert, R.; Nedovic, V.A. Primary Beer Fermentation by Immobilised Yeast—A Review on Flavour Formation and Control Strategies. J. Chem. Technol. Biotechnol. 2006, 81, 1353–1367. [Google Scholar] [CrossRef]
- Vesterlund, S.; Salminen, K.; Salminen, S. Water Activity in Dry Foods Containing Live Probiotic Bacteria Should Be Carefully Considered: A Case Study with Lactobacillus Rhamnosus GG in Flaxseed. Int. J. Food Microbiol. 2012, 157, 319–321. [Google Scholar] [CrossRef]
- Poddar, D.; Das, S.; Jones, G.; Palmer, J.; Jameson, G.B.; Haverkamp, R.G.; Singh, H. Stability of Probiotic Lactobacillus Paracasei during Storage as Affected by the Drying Method. Int. Dairy J. 2014, 39, 1–7. [Google Scholar] [CrossRef]
- Jouppila, K.; Roos, Y.H. Glass Transitions and Crystallization in Milk Powders. J. Dairy Sci. 1994, 77, 2907–2915. [Google Scholar] [CrossRef]
- Heidebach, T.; Först, P.; Kulozik, U. Influence of Casein-Based Microencapsulation on Freeze-Drying and Storage of Probiotic Cells. J. Food Eng. 2010, 98, 309–316. [Google Scholar] [CrossRef]
- Shokri, Z.; Fazeli, M.R.; Ardjmand, M.; Mousavi, S.M.; Gilani, K. Factors Affecting Viability of Bifidobacterium Bifidum during Spray Drying. DARU J. Pharm. Sci. 2015, 23, 7. [Google Scholar] [CrossRef]
- Barbosa-Cánovas, G.V.; Fontana, A.J.J.R.; Schmidt, S.J.; Labuza, T.P. (Eds.) Water Activity in Foods; Wiley: Hoboken, NJ, USA, 2020; ISBN 9781118768310. [Google Scholar]
- Arepally, D.; Goswami, T.K. Effect of Inlet Air Temperature and Gum Arabic Concentration on Encapsulation of Probiotics by Spray Drying. LWT 2019, 99, 583–593. [Google Scholar] [CrossRef]
- Liu, H.; Gong, J.; Chabot, D.; Miller, S.S.; Cui, S.W.; Ma, J.; Zhong, F.; Wang, Q. Incorporation of Polysaccharides into Sodium Caseinate-Low Melting Point Fat Microparticles Improves Probiotic Bacterial Survival during Simulated Gastrointestinal Digestion and Storage. Food Hydrocoll. 2016, 54, 328–337. [Google Scholar] [CrossRef]
- Ng, S.; Wong, P.; Tan, C.; Long, K.; Nyam, K. Influence of the Inlet Air Temperature on the Microencapsulation of Kenaf (Hibiscus cannabinus L.) Seed Oil. Eur. J. Lipid Sci. Technol. 2013, 115, 1309–1318. [Google Scholar] [CrossRef]
- Suez, J.; Zmora, N.; Zilberman-Schapira, G.; Mor, U.; Dori-Bachash, M.; Bashiardes, S.; Zur, M.; Regev-Lehavi, D.; Ben-Zeev Brik, R.; Federici, S.; et al. Post-Antibiotic Gut Mucosal Microbiome Reconstitution Is Impaired by Probiotics and Improved by Autologous FMT. Cell 2018, 174, 1406–1423.e16. [Google Scholar] [CrossRef] [PubMed]
- Doherty, S.B.; Gee, V.L.; Ross, R.P.; Stanton, C.; Fitzgerald, G.F.; Brodkorb, A. Development and Characterisation of Whey Protein Micro-Beads as Potential Matrices for Probiotic Protection. Food Hydrocoll. 2011, 25, 1604–1617. [Google Scholar] [CrossRef]
- Zhang, K.; Zhang, H.; Hu, X.; Bao, S.; Huang, H. Synthesis and Release Studies of Microalgal Oil-Containing Microcapsules Prepared by Complex Coacervation. Colloids Surf. B Biointerfaces 2012, 89, 61–66. [Google Scholar] [CrossRef]
- Vasisht, N. Factors and Mechanisms in Microencapsulation. In Microencapsulation in the Food Industry; Elsevier: Amsterdam, The Netherlands, 2014; pp. 15–24. [Google Scholar]
- Taranto, M.P.; Fernandez Murga, M.L.; Lorca, G.; Valdez, G.F. Bile Salts and Cholesterol Induce Changes in the Lipid Cell Membrane of Lactobacillus Reuteri. J. Appl. Microbiol. 2003, 95, 86–91. [Google Scholar] [CrossRef]
- Ilango, S.; Pandey, R.; Antony, U. Functional Characterization and Microencapsulation of Probiotic Bacteria from Koozh. J. Food Sci. Technol. 2016, 53, 977–989. [Google Scholar] [CrossRef]
- Wendel, U. Assessing Viability and Stress Tolerance of Probiotics—A Review. Front. Microbiol. 2022, 12, 818468. [Google Scholar] [CrossRef]
- Golowczyc, M.A.; Silva, J.; Abraham, A.G.; de Antoni, G.L.; Teixeira, P. Preservation of Probiotic Strains Isolated from Kefir by Spray Drying. Lett. Appl. Microbiol. 2010, 50, 7–12. [Google Scholar] [CrossRef]
- Abe, F.; Miyauchi, H.; Uchijima, A.; Yaeshima, T.; Iwatsuki, K. Effects of Storage Temperature and Water Activity on the Survival of Bifidobacteria in Powder Form. Int. J. Dairy Technol. 2009, 62, 234–239. [Google Scholar] [CrossRef]
- Teixeira, P.; Castro, H.; Kirby, R. Evidence of Membrane Lipid Oxidation of Spray-Dried Lactobacillus Bulgaricus during Storage. Lett. Appl. Microbiol. 1996, 22, 34–38. [Google Scholar] [CrossRef]
- Santos, M.G.; Bozza, F.T.; Thomazini, M.; Favaro-Trindade, C.S. Microencapsulation of Xylitol by Double Emulsion Followed by Complex Coacervation. Food Chem. 2015, 171, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Daoub, R.M.A.; Elmubarak, A.H.; Misran, M.; Hassan, E.A.; Osman, M.E. Characterization and Functional Properties of Some Natural Acacia Gums. J. Saudi Soc. Agric. Sci. 2018, 17, 241–249. [Google Scholar] [CrossRef]
- Mahdi, A.A.; Mohammed, J.K.; Al-Ansi, W.; Ghaleb, A.D.S.; Al-Maqtari, Q.A.; Ma, M.; Ahmed, M.I.; Wang, H. Microencapsulation of Fingered Citron Extract with Gum Arabic, Modified Starch, Whey Protein, and Maltodextrin Using Spray Drying. Int. J. Biol. Macromol. 2020, 152, 1125–1134. [Google Scholar] [CrossRef]
- Kim, E.H.-J.; Chen, X.D.; Pearce, D. Surface Composition of Industrial Spray-Dried Milk Powders. 2. Effects of Spray Drying Conditions on the Surface Composition. J. Food Eng. 2009, 94, 169–181. [Google Scholar] [CrossRef]
- Nishanthi, M.; Chandrapala, J.; Vasiljevic, T. Properties of Whey Protein Concentrate Powders Obtained by Spray Drying of Sweet, Salty and Acid Whey under Varying Storage Conditions. J. Food Eng. 2017, 214, 137–146. [Google Scholar] [CrossRef]
- Rosland Abel, S.E.; Yusof, Y.A.; Chin, N.L.; Chang, L.S.; Mohd Ghazali, H.; Manaf, Y.N. Characterisation of Physicochemical Properties of Gum Arabic Powder at Various Particle Sizes. Food Res. 2020, 4, 107–115. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).