Abstract
Calculations are presented for the encapsulation of two CO2 molecules in the most common C84 fullerenes, producing (CO2)2@(22)-C84 and (CO2)2@(23)-C84. The calculations are performed at the DFT M06-2X/6-31+G* level with the BSSE correction. The encapsulation energy for (CO2)2@(22)-C84 and (CO2)2@(23)-C84 is calculated as −4.9 and −5.6 kcal/mol, respectively. The encapsulation of two CO2 molecules is attractive, though the energy gain is, owing to a steric hindrance, smaller than previously found for the encapsulation of one CO2. The IR vibrational spectra are presented, too.
1. Introduction
The interplay of fullerenes with CO2 has recently been treated [1,2,3,4], even for boron-based fullerene-like cage B80 [1]. Actually, such species can even have interesting practical applications—additions of endohedral metallofullerenes [5,6] or CO2 [7] can improve properties of solar cells. In addition to metals, non-metallic species can also be incorporated inside fullerene polyhedrons. First such non-metal endohedrals are represented by N2@C60 and N2@C70 produced [8] at high temperatures and high pressures with a catalyst. Another approach first places molecules inside open fullerene cages, and the cage window is then closed synthetically, e.g., preparation [9] of (H2O)2@C70. Such fullerene endohedrals with encapsulated non-metals have also been studied by calculations, e.g., [10,11,12,13,14,15]. The first such calculations [10] treated diatomic molecules like H2 or HF trapped inside the C60 cage and predicted shifts in vibrational spectra. Encapsulations of small polyatomic non-metal species into fullerene cages have also been evaluated, including water, e.g., [11,14,15].
CO@C60 can be produced [7] by the high-pressure and high-temperature treatment, and there is thus a related problem concerning CO2 encapsulations in some larger fullerene polyhedrons. We have already calculated [4] possibilities of encapsulation of one CO2 molecule in the two most common C84 fullerenes as well as such encapsulations for one, two, and three water molecules [14,15]. The present report continues with such evaluations for encapsulation of two CO2 molecules in the two C84 cages, namely and C84, conventionally labeled as (22)-C84 and (23)-C84. It should be noted for completeness that the two C84 cages are commonly produced in the carbon-arc fullerene synthesis [16] in a ratio [16,17] of about 2:1. Interestingly, the pristine cage is in potential energy located [17,18] about 0.5 kcal/mol above the structure. However, the entropy contributions influence the / relative isomeric populations as well.
2. Calculations
The calculations are performed with the DFT (density-functional theory) approach, namely with the M06-2X functional thoroughly tested [19] for various applications including non-covalent interactions. The M06-2X functional is applied with the standard 6-31+G* basis set [20]; the approach is denoted by M06-2X/6-31+G*. The molecular-structure optimizations are carried out with the 1st derivatives of the potential energy constructed analytically (Figure 1 and Figure 2). The stationary points found are checked by the GF harmonic vibrational analysis using the analytical 2nd derivatives of the potential energy. The GF vibrational analysis can prove that the localized stationary points are indeed the local energy minima. Moreover, it can also simulate the vibrational spectra and supply input information for thermodynamic-stability evaluations. The wavefunction stability was tested throughout so that unstable SCF solutions without a physical applicability could be eliminated.
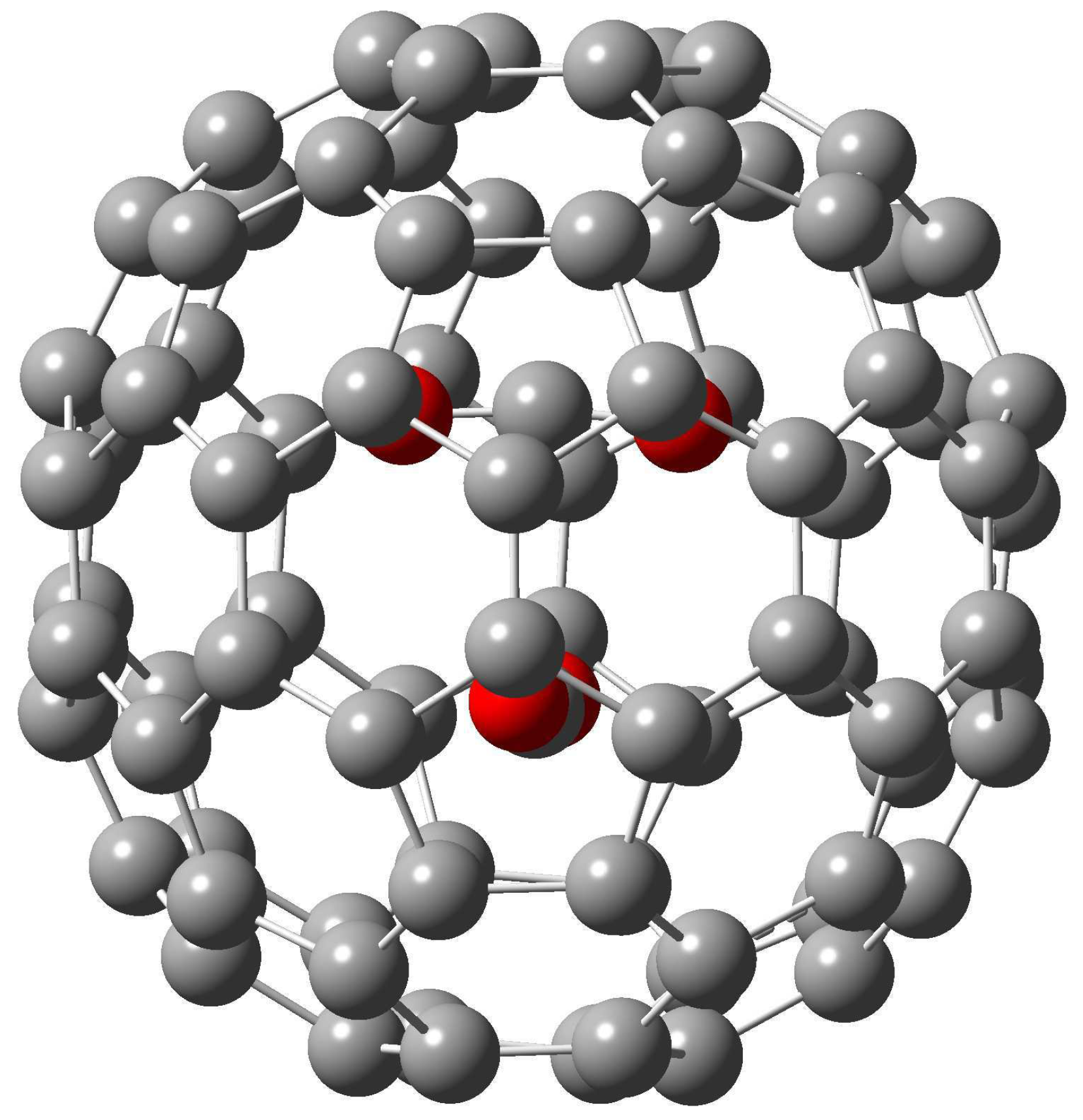
Figure 1.
The M06-2X/6-31+G* optimized structure of (CO2)2@(22)-C84.
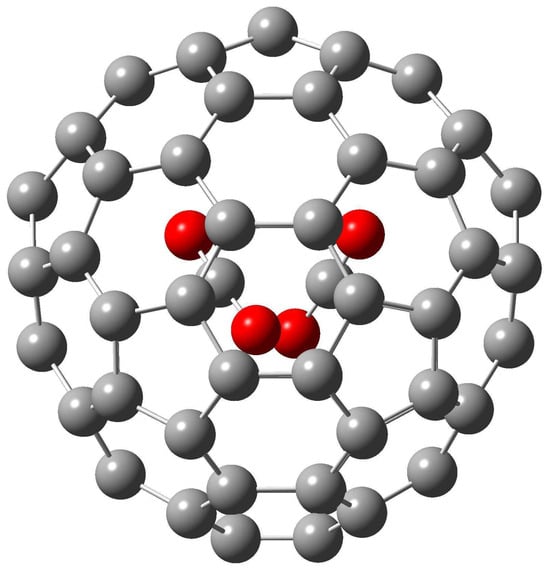
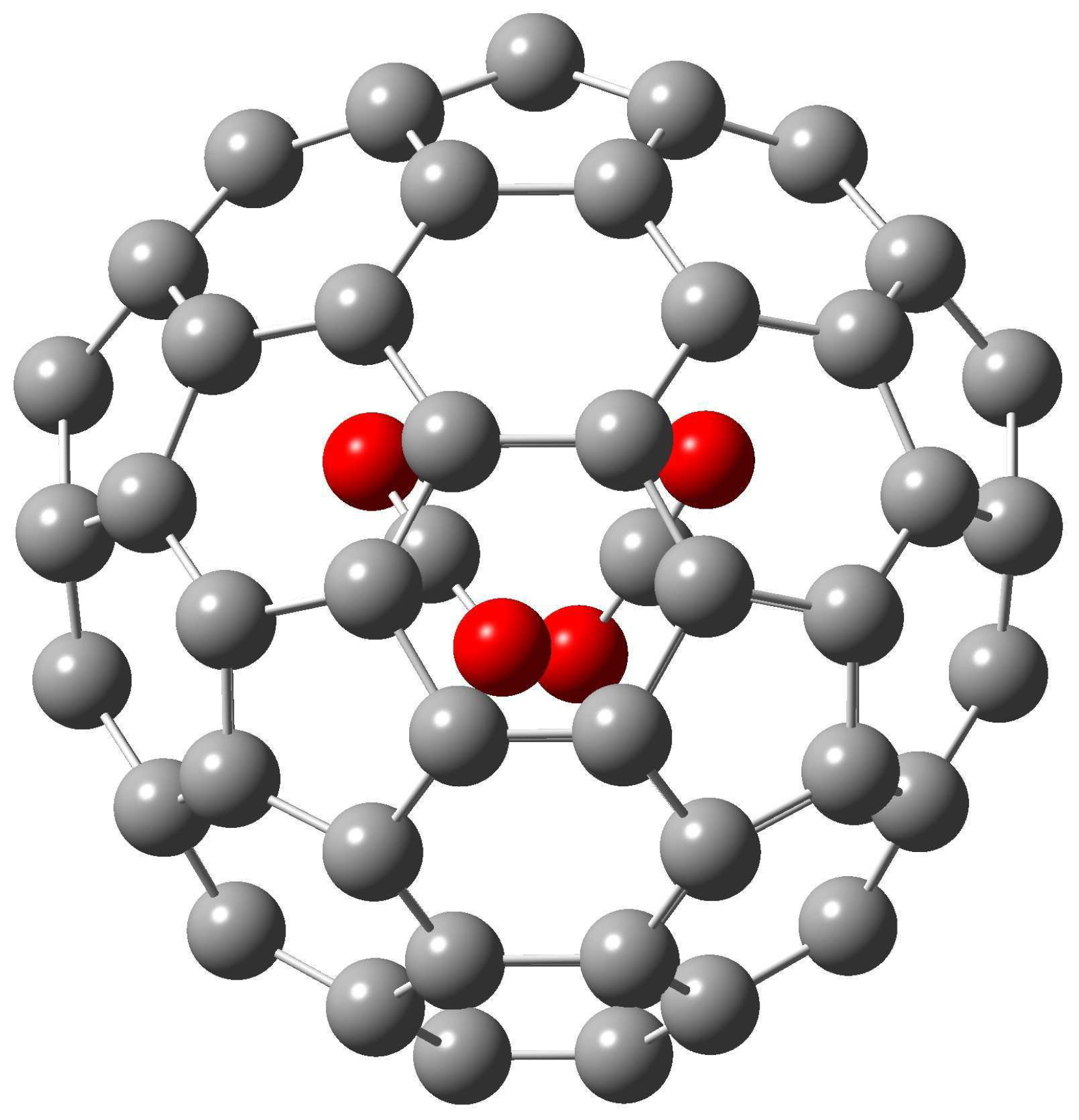
Figure 2.
The M06-2X/6-31+G* optimized structure of (CO2)2@(23)-C84.
The potential energy changes connected with encapsulations into the carbon cages are corrected [21] with respect to the basis set superposition error (BSSE). The BSSE correction term is evaluated by the counterpoise method introduced by Boys and Bernardi [22]—it is also coded [23,24] CP2 in the case of a dimerization. The counterpoise method ensures that every species participating in a studied chemical process is formally described by the same number of the atomic basis-set functions so that their potential energies can be compared. The BSSE correction term represents a crucial improvement of the encapsulation energy, as without the BSSE term, the potential energy gain yielded by the encapsulation process would be overvalued so that reaction products would be artificially over-stabilized.
The presented evaluations were carried out with the Gaussian program package [23,24]. The calculations were performed on computers operating in parallel regime, with 8–24 processors (computational frequency up to 3 GHz each, operational memory up to 60 GB).
3. Results and Discussion
This study deals with the encapsulation energy , representing the potential-energy change in the gas-phase process involving the (22)-C84 fullerene cage:
as well as with similar encapsulation in the (23)-C84 cage. Table 1 presents the M06-2X/6-31+G* encapsulation energetics with and also without the BSSE correction.

Table 1.
The calculated encapsulation energy for (CO2)2@(22)-C84 a and (CO2)2@(23)-C84 b.
The M06-2X/6-31+G* encapsulation energy corrected with the BSSE term is for (CO2)2@(22)-C84 and (CO2)2@(23)-C84 equal to −4.9 and −5.6 kcal/mol, respectively. The encapsulation of two CO2 molecules is attractive though the energy gain is owing to a steric hindrance smaller than found [4] for the encapsulation of just one CO2. The BSSE term decreases the energy gain significantly—by about 8 kcal/mol. Table 1 presents for methodological comparison also the values obtained with a simpler approach, viz. M06-2X/6-31G* in the M06-2X/6-31+G* optimized geometry. The encapsulation of two CO2 molecules is attractive at the simpler level as well, though the stabilization is reduced.
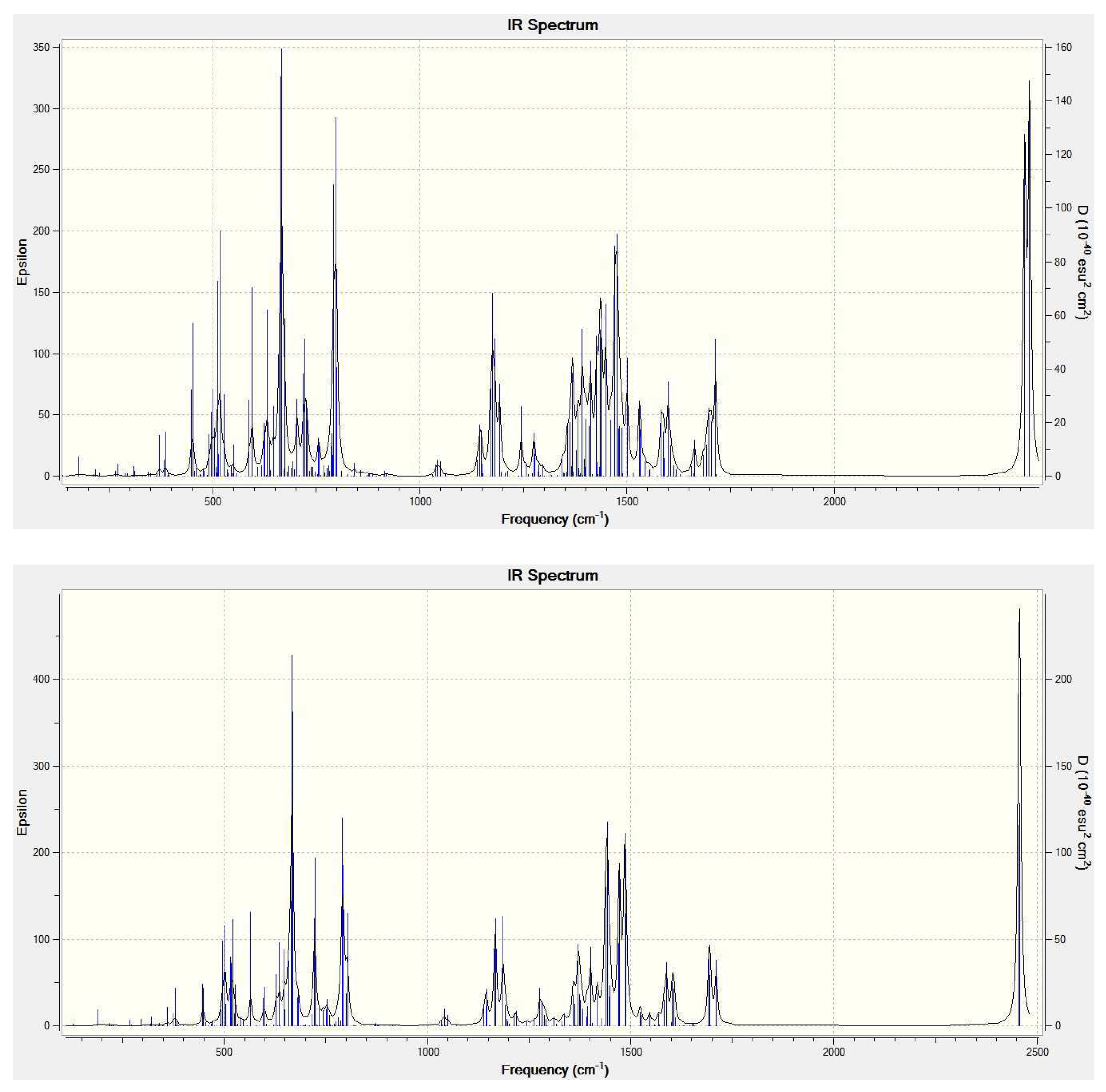
Table 2 reports some computed molecular parameters of the (CO2)2@C84 endohedrals—the closest contact of the carbon atom in CO2 with the cage carbons, the total charge on the C84 cage, and the lowest/highest vibrational frequency. The closest contact is in both species about 3.1 Å. The total charge on the cage evaluated using the M06-2X/3-21G Mulliken atomic charges is only about −0.08 in elementary charge units. Such a small charge transfer is considerably different from substantial charge transfers calculated for metallofullerenes [14]. It should be mentioned that the Mulliken charges on atoms should be evaluated owing to their definition [25,26] with smaller basis sets like the standard 3–21G basis set considered in this work. Such small basis sets are indeed known [14] to produce for metallofullerenes a good agreement with the atomic charges obtained from observations [27]. As vibrational spectra can be useful for identification of nanocarbon species [28,29], Figure 3 presents the M06-2X/6-31+G* simulated IR vibrational spectra of (CO2)2@(22)-C84 and (CO2)2@(23)-C84.

Table 2.
The selected characteristics of (CO2)2@(22)-C84 and (CO2)2@(23)-C84—the closest C-cage contact a , the total charge b on the cage , the lowest vibrational frequency a , the highest vibrational frequency a .
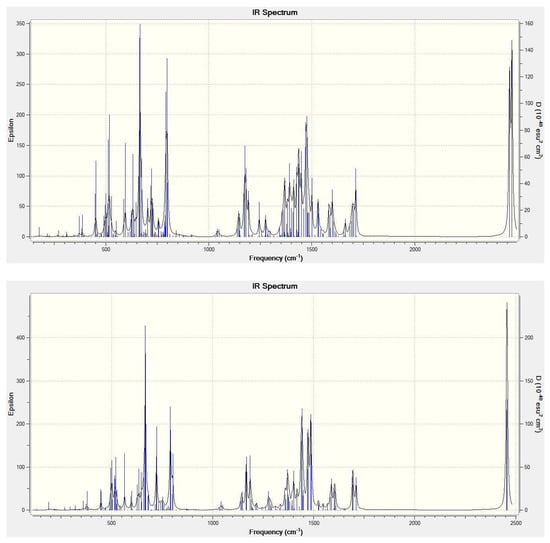
Figure 3.
M06-2X/6-31+G* calculated IR spectrum of (CO2)2@(22)-C84 (Top) and (CO2)2@(23)-C84 (Bottom).
However, as the energy gain after encapsulation of two CO2 molecules in C84 is smaller than in the case of just one CO2, and also smaller than for two water molecules [15], the C84-based endohedrals containing two CO2 molecules are less likely to be prepared. Still, an application of high CO2 pressures can improve [4] the yield. Let us consider (2) the equilibrium constant for process (1)— expressed using the partial pressures p of the reaction components is as follows:
and also (4) the equilibrium constant for the encapsulation (3) of just one carbon dioxide:
The two equilibrium constants yield [4] the ratio of the endohedrals in the following form:
which straightforwardly shows the influence of the CO2 pressure on the endohedral population ratio. The related detailed thermodynamic-stability evaluations are to be supplied later on. Incidentally, the finding of C60 in the cosmic space [30] suggests astrophysical searches [31,32,33] also for fullerene endohedrals—for example, the pressure of CO2 at Venus can reach [15] some 90 atm.
There is an interesting related issue, namely isomerism of the free, i.e., not encapsulated, gas-phase (CO2)2 species described by quantum-chemical calculations [34]. Two different local energy minima have been located and named [34] parallel P and T shape structures. Recently, we have studied [15] the water-dimer thermodynamics using a high-level energetics G3 & MP2 = FC/6-311+G* and anharmonic-thermodynamic treatment. As the combined treatment produces a very good agreement with observed data in the water-dimer case, it can also be applied to (CO2)2. At the advanced G3 & MP2 = FC/6-311+G* level, the P form is lower in energy, though only by some 0.14 kcal/mol. The rotational and vibrational terms needed for the anharmonic treatment originate [15] from the MP2 = FC/6-311+G* calculations. At very low temperatures the lower-energy isomer must be prevailing, i.e., the P form. However, with increasing temperature, the relative isomeric populations are mutually approaching; finally, the stability order is interchanged, and at high temperatures the T isomer becomes prevailing. The equal populations of both isomers are obtained at a temperature of about 120 K; at room temperature the T isomer then represents 77% of the equilibrium mixture. Obviously, with the exception of the lowest temperatures, one has to expect the presence of both isomeric forms, which should be taken into account with interpretations of observations, including planetary atmospheres.
4. Conclusions
The recent theoretical treatments of rather complex endohedral and other nanocarbon species [35,36,37,38,39,40,41,42,43,44,45,46] are extended here to encapsulation of two CO2 molecules in the most common C84 fullerenes, producing endohedrals (CO2)2@(22)-C84 and (CO2)2@(23)-C84. The calculations are performed at the DFT M06-2X/6-31+G* level with the BSSE correction. The encapsulation energy for (CO2)2@(22)-C84 and (CO2)2@(23)-C84 is calculated as −4.9 and −5.6 kcal/mol, respectively. The encapsulation of two CO2 molecules is attractive, though the energy gain is, owing to a steric hindrance, smaller than previously found for the encapsulation of one CO2 molecule.
Author Contributions
Conceptualization, Z.S., F.U., T.A., X.L. and L.A.; Hardware and software, Z.S., F.U. and L.A.; Validation, Z.S., F.U. and X.L.; Writing—original draft, Z.S. and F.U.; Visualization, Z.S. and F.U. All authors have read and agreed to the published version of the manuscript.
Funding
The reported research has been supported by the National Natural Science Foundation of China (21925104 and 92261204), the Hubei Provincial Natural Science Foundation of China (No. 2021CFA020), and the International Cooperation Key Project of Science and Technology Department of Shaanxi, and by the Charles University Centre of Advanced Materials/CUCAM (CZ.02.1.01/0.0/0.0/15_003/0000417) and the MetaCentrum (LM2010005) and CERIT-SC (CZ.1.05/3.2.00/08.0144) computing facilities. A very initial phase of the research line was supported by the Alexander von Humboldt-Stiftung and the Max-Planck-Institut für Chemie (Otto-Hahn-Institut).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sun, Q.; Wang, M.; Li, Z.; Du, A.; Searles, D.J. Carbon dioxide capture and gas separation on B80 fullerene. J. Phys. Chem. C 2014, 118, 2170–2177. [Google Scholar] [CrossRef]
- Meloni, G.; Giustini, A.; Park1, H. CO2 activation within a superalkali-doped fullerene. Front. Chem. 2021, 9, 712960-1–712960-8. [Google Scholar] [CrossRef]
- Palakkal, A.S.; Pillai, R.S. Unraveling the role of fullerene encapsulation driven CO2 capture in square pillared Bio-HOF under humid condition by advanced molecular simulation. Separ. Purif. Technol. 2023, 325, 124650-1–124650-12. [Google Scholar]
- Slanina, Z.; Uhlík, F.; Akasaka, T.; Lu, X.; Adamowicz, L. CO2@C84: DFT calculations of structure and energetics. Inorganics 2025, 13, 19-1–19-8. [Google Scholar] [CrossRef]
- Ross, R.B.; Cardona, C.M.; Guldi, D.M.; Sankaranarayanan, S.G.; Reese, M.O.; Kopidakis, N.; Peet, J.; Walker, B.; Bazan, G.C.; Van Keuren, E.; et al. Endohedral fullerenes for organic photovoltaic devices. Nat. Mater. 2009, 8, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Yu, P.; Shen, W.; Hu, S.; Akasaka, T.; Lu, X. Er@C82 as a bifunctional additive to the spiro-OMeTAD hole transport layer for improving performance and stability of perovskite solar cells. Sol. RRL 2021, 5, 2100463-1–2100463-9. [Google Scholar] [CrossRef]
- Kong, J.; Shin, Y.; Röhr, J.A.; Wang, H.; Meng, J.; Wu, Y.; Katzenberg, A.; Kim, G.; Kim, D.Y.; Li, T.-D.; et al. CO2 doping of organic interlayers for perovskite solar cells. Nature 2021, 594, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Peres, T.; Cao, B.P.; Cui, W.D.; Khong, A.; Cross, R.J.; Saunders, M.; Lifshitz, C. Some new diatomic molecule containing endohedral fullerenes. Int. J. Mass Spectr. 2001, 210–211, 241–247. [Google Scholar]
- Zhang, R.; Murata, M.; Aharen, T.; Wakamiya, A.; Shimoaka, T.; Hasegawa, T.; Murata, Y. Synthesis of a distinct water dimer inside fullerene C70. Nat. Chem. 2016, 8, 435–441. [Google Scholar] [CrossRef]
- Cioslowski, J. Endohedral chemistry: Electronic structures of molecules trapped inside the C60 cage. J. Am. Chem. Soc. 1991, 113, 4139–4141. [Google Scholar] [CrossRef]
- Ramachandran, C.N.; Sathyamurthy, N. Water clusters in a confined nonpolar environment. Chem. Phys. Lett. 2005, 410, 348–351. [Google Scholar] [CrossRef]
- Bloodworth, S.; Sitinova, G.; Alom, S.; Vidal, S.; Bacanu, G.R.; Elliott, S.J.; Light, M.E.; Herniman, J.M.; Langley, G.J.; Levitt, M.H.; et al. First synthesis and characterization of CH4@C60. Angew. Chem. Int. Ed. 2019, 58, 5038–5043. [Google Scholar] [CrossRef] [PubMed]
- Jaworski, A.; Hedin, N. Local energy decomposition analysis and molecular properties of encapsulated methane in fullerene (CH4@C60). Phys. Chem. Chem. Phys. 2021, 23, 21554–21567. [Google Scholar] [CrossRef]
- Slanina, Z.; Uhlík, F.; Adamowicz, L. Theoretical predictions of fullerene stabilities. In 2022 Handbook of Fullerene Science and Technology; Lu, X., Akasaka, T., Slanina, Z., Eds.; Springer: Singapore, 2022; pp. 111–179. [Google Scholar]
- Slanina, Z.; Uhlík, F.; Akasaka, T.; Lu, X.; Adamowicz, L. Theoretical studies of non-metal endohedral fullerenes. Nanomaterials 2025, 15, 1287-1–1287-17. [Google Scholar] [CrossRef]
- Kikuchi, K.; Nakahara, N.; Honda, M.; Suzuki, S.; Saito, K.; Shiromaru, H.; Yamauchi, K.; Ikemoto, I.; Kuramochi, T.; Hino, S.; et al. Separation, detection and UV/Visible absorption spectra of fullerenes; C76, C78 and C84. Chem. Lett. 1991, 20, 1607–1610. [Google Scholar] [CrossRef]
- Slanina, Z.; François, J.-P.; Kolb, M.; Bakowies, D.; Thiel, W. Calculated relative stabilities of C84. Fuller. Sci. Technol. 1993, 1, 221–230. [Google Scholar] [CrossRef]
- Bakowies, D.; Kolb, M.; Thiel, W.; Richard, S.; Ahlrichs, R.; Kappes, M.M. Quantum-chemical study of C84 fullerene isomers. Chem. Phys. Lett. 1992, 200, 411–417. [Google Scholar] [CrossRef]
- Zhao, Y.; Truhlar, D.G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 2008, 120, 215–241. [Google Scholar]
- Ditchfield, R.; Hehre, W.J.; Pople, J.A. Self-consistent molecular-orbital methods. IX. An extended Gaussian-type basis for molecular-orbital studies of organic molecules. J. Chem. Phys. 1971, 54, 724–728. [Google Scholar] [CrossRef]
- Slanina, Z.; Uhlík, F.; Lee, S.-L.; Adamowicz, L.; Akasaka, T.; Nagase, S. Computed stabilities in metallofullerene series: Al@C82, Sc@C82, Y@C82, and La@C82. Int. J. Quant. Chem. 2011, 111, 2712–2718. [Google Scholar] [CrossRef]
- Boys, S.F.; Bernardi, F. The calculation of small molecular interactions by the difference of separate total energies. Some procedures with reduced errors. Mol. Phys. 1970, 19, 553–566. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 16, Rev. B.01; Gaussian Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Rev. C.01; Gaussian Inc.: Wallingford, CT, USA, 2013. [Google Scholar]
- Hehre, W.J. A Guide to Molecular Mechanics and Quantum Chemical Calculations; Wavefunction: Irvine, CA, USA, 2003; p. 435. [Google Scholar]
- Jensen, F. Introduction to Computational Chemistry; Wiley: Chichester, UK, 2017; p. 319. [Google Scholar]
- Takata, M.; Nishibori, E.; Sakata, M.; Shinohara, H. Charge density level structures of endohedral metallofullerenes determined by synchrotron radiation powder method. New Diam. Front. Carb. Technol. 2002, 12, 271–286. [Google Scholar]
- Slanina, Z.; Rudziński, J.M.; Togasi, M.; Ōsawa, E. Quantum-chemically supported vibrational analysis of giant molecules: The C60 and C70 clusters. J. Mol. Struct. Theochem 1989, 202, 169–176. [Google Scholar] [CrossRef]
- Krätschmer, W.; Lamb, L.D.; Fostiropoulos, K.; Huffman, D.R. Solid C60: A new form of carbon. Nature 1990, 347, 354–358. [Google Scholar] [CrossRef]
- Campbell, E.K.; Holz, M.; Gerlich, D.; Maier, J.P. Laboratory confirmation of C60+ as the carrier of two diffuse interstellar bands. Nature 2015, 523, 322–323. [Google Scholar] [CrossRef] [PubMed]
- Berné, O.; Tielens, A.G. Formation of buckminsterfullerene (C60) in interstellar space. Proc. Natl. Acad. Sci. USA 2012, 109, 401–406. [Google Scholar] [CrossRef]
- Berné, O.; Montillaud, J.; Joblin, C. Top-down formation of fullerenes in the interstellar medium. Astronom. Astrophys. 2015, 577, A133-1–A133-9. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.S.; Peeters, E.; Cami, J.; Schmidt, T.W. Open questions on carbon-based molecules in space. Commun. Chem. 2022, 5, 94. [Google Scholar] [CrossRef] [PubMed]
- Brigot, N.; Odiot, S.; Walmsley, S.H. A priori and empirical calculations of external vibration frequencies of the dimer of carbon dioxide. Chem. Phys. Lett. 1982, 88, 543–546. [Google Scholar] [CrossRef]
- Rodríguez-Fortea, A.; Balch, A.L.; Poblet, J.M. Endohedral metallofullerenes: A unique host-guest association. Chem. Soc. Rev. 2011, 40, 3551–3563. [Google Scholar] [CrossRef]
- Weisman, R.B. New frontiers in nanocarbons. Electrochem. Soc. Interface 2013, 22, 49. [Google Scholar] [CrossRef][Green Version]
- Basiuk, V.A.; Basiuk, E.V. Noncovalent complexes of Ih-C80 fullerene with phthalocyanines. Fuller. Nanotub. Carbon Nanostruct. 2018, 26, 69–75. [Google Scholar] [CrossRef]
- Basiuk, V.A.; Tahuilan-Anguiano, D.E. Complexation of free-base and 3d transition metal(II) phthalocyanines with endohedral fullerene Sc3N@C80. Chem. Phys. Lett. 2019, 722, 146–152. [Google Scholar] [CrossRef]
- Li, M.; Zhao, R.; Dang, J.; Zhao, X. Theoretical study on the stabilities, electronic structures, and reaction and formation mechanisms of fullerenes and endohedral metallofullerenes. Coor. Chem. Rev. 2022, 471, 214762-1–214762-12. [Google Scholar] [CrossRef]
- Li, Y.B.; Biswas, R.; Kopcha, W.P.; Dubroca, T.; Abella, L.; Sun, Y.; Crichton, R.A.; Rathnam, C.; Yang, L.T.; Yeh, Y.W.; et al. Structurally defined water-soluble metallofullerene derivatives towards biomedical applications. Angew. Chem. Int. Ed. Engl. 2023, 62, e202211704-1–e202211704-10. [Google Scholar]
- Basiuk, V.A.; Wu, Y.F.; Prezhdo, O.V.; Basiuk, E.V. Lanthanide atoms induce strong graphene sheet distortion when adsorbed on Stone-Wales defects. J. Phys. Chem. Lett. 2024, 15, 9706–9713. [Google Scholar] [CrossRef] [PubMed]
- Soares, G.G.C.; Rigolo, K.; Dini, D.; Rosolen, J.M. Self-assembly graphene into fullerenes and hollow spherical graphene particles during anodic polarization of graphite. Diam. Relat. Mater. 2025, 156, 112379-1–112379-7. [Google Scholar] [CrossRef]
- Bacanu, G.R. Spectroscopy of non-metallic C60 endofullerenes. Chem. Phys. Rev. 2025, 6, 031307-1–031307-61. [Google Scholar] [CrossRef]
- Choi, C.; Yun, T.G.; Hwang, B. Dispersion stability of carbon nanotubes and their impact on energy storage devices. Inorganics 2023, 11, 383-1–383-14. [Google Scholar] [CrossRef]
- Markopoulou, E.; Nikolakis, P.; Savvakis, G.; Rissanou, A.N. Employing molecular dynamics simulations to explore the behavior of diphenylalanine dipeptides in graphene-based nanocomposite systems. Inorganics 2025, 13, 92-1–92-18. [Google Scholar] [CrossRef]
- McLean, B.; Page, A.J. Carbon nanomaterials for advanced technology. Inorganics 2025, 13, 129-1–129-4. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.