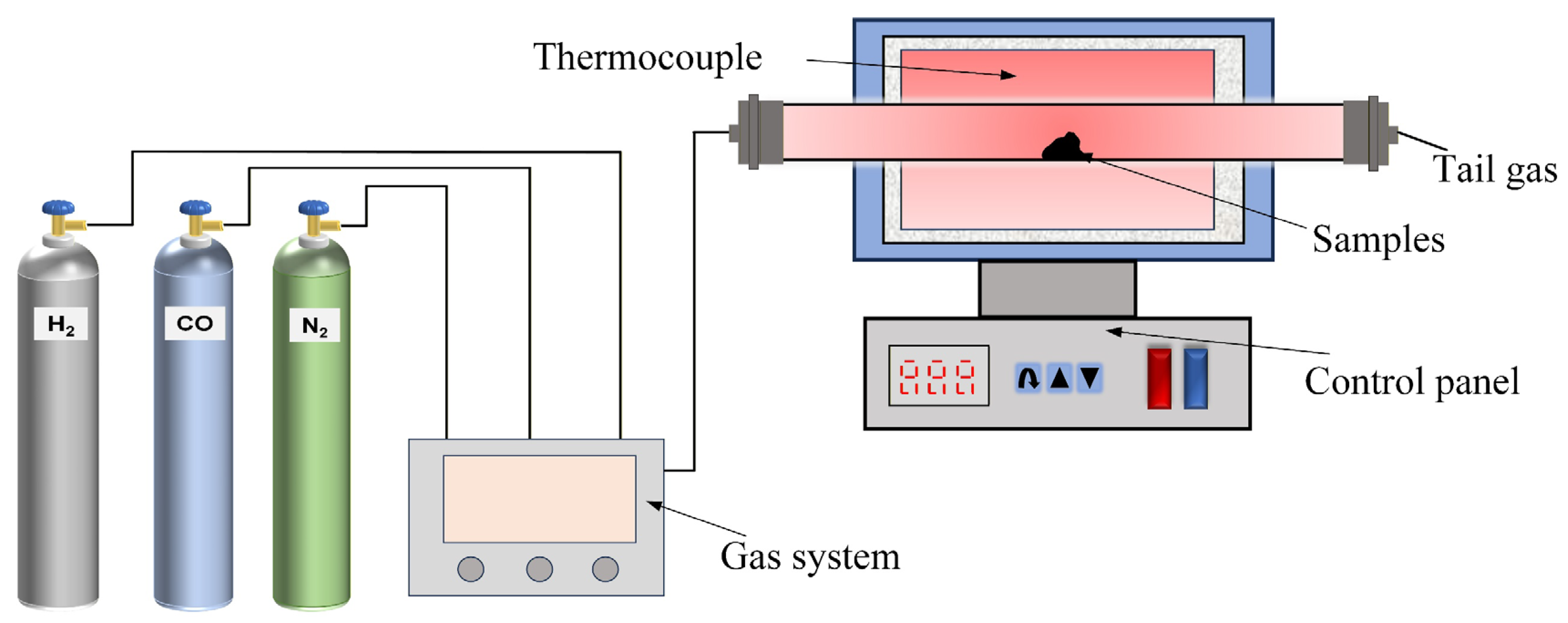
3.1. Effect of Cooling Methods on Relative Grindability
To visually analyze the grinding characteristics of HMPT products obtained through different cooling methods, grinding tests were conducted for 0, 2, 4, 6, 8, and 10 min, as specified by the grinding conditions in
Table 2. Overall, as the grinding process progresses, the content of the finest particle size (−0.30 mm) in the grinding product increases, while the content of the coarser particle size (+0.074 mm) decreases. The content of intermediate particle sizes increases initially and then decreases slightly. Comparative analysis showed that after 10 min of grinding, the +0.074 mm content in the four cooled grinding products decreases to 0.32%, 0.19%, 0.48% and 0.42%, respectively, while the −0.030 mm content increases to 89.20%, 90.11%, 88.41%, and 88.80%, respectively. It is observed that the second cooling method results in the lowest content of coarser particles and a significantly higher content of finer particles compared to the other cooling methods.
Specifically, as shown in
Figure 5a, for the hydrogen-based mineral phase transformation products obtained using the first cooling method, after 2 min of grinding, the content of +0.074 mm rapidly decreased from 41.10% to 10.12%. The content of −0.074 + 0.043 mm increased from 17.60% to 18.88%; the content of −0.043 + 0.038 mm increased from 6.22% to 7.90%; the content of −0.038 + 0.030 mm increased from 8.37% to 11.51%; and the content of the smallest particle size, −0.030 mm, increased from 26.71% to 52.61%. As shown in
Figure 5b, for the hydrogen-based mineral products from phase conversion obtained using the second cooling method, after 2 min of grinding, the content of +0.074 mm rapidly decreased from 40.46% to 9.85%; the content of −0.074 + 0.043 mm increased from 17.48% to 18.59%; the content of −0.043 + 0.038 mm increased from 6.17% to 7.52%; the content of −0.038 + 0.030 mm increased from 8.75% to 10.98%; and the content of the smallest particle size, −0.030 mm, increased from 27.14% to 53.05%. As shown in
Figure 5c, for the hydrogen-based mineral products from phase conversion obtained using the third cooling method, after 2 min of grinding, the content of +0.074 mm decreased from 41.98% to 10.99%; the content of −0.074 + 0.043 mm increased from 18.29% to 19.37%; the content of −0.043 + 0.038 mm increased from 6.71% to 8.38%; the content of −0.038 + 0.030 mm increased from 8.18% to 9.83%; and the content of the smallest particle size, −0.030 mm, increased from 24.84% to 51.43%. As shown in
Figure 5d, for the hydrogen-based mineral phase conversion products obtained using the fourth cooling method, after 2 min of grinding, the content of +0.074 mm decreased from 41.76% to 10.41%; the content of −0.074 + 0.043 mm increased from 18.08% to 18.99%; the content of −0.043 + 0.038 mm increased from 6.55% to 8.16%; the content of −0.038 + 0.030 mm increased from 8.22% to 10.20%; and the content of the smallest particle size, −0.030 mm, increased from 25.39% to 52.24%. In addition, as the grinding time increases from 2 min to 10 min, the rate of change slows down. Based on the reduction in coarse particle content and the increase in fine particle content, it can be concluded that the HMPT products treated by the second cooling method exhibit better relative grindability.
From the above analysis results, it can be concluded that the grinding performance of ore treated by HMPT directly depends on the physical and chemical properties of the ore, with the cooling method after the reaction having a significant regulatory effect on these properties. The results that among the four cooling methods, cooling mode II produced the lowest content of coarse particles and the highest content of fine particles. This enhanced grindability may be attributed to the microcracks induced by thermal stress. During the cooling process, the temperature gradient between the surface and the interior the ore generates thermal stress. In cooling mode II, the ore is first cooled to 200 °C in a reducing atmosphere and then rapidly quenched with water. The difference in thermal expansion coefficients between iron ore and gangue minerals amplifies this thermal stress during rapid cooling. As a result, a significant number of microcracks form, which reduces the energy required for particle fragmentation during the grinding process and promotes the formation of fine particles. In contrast, cooling mode I involves a slow cooling process, resulting in minimal thermal stress and smaller microcracks, which have a relatively minor impact on grindability. Cooling modes III and IV are adversely affected by insufficient oxidation of magnetite and inadequate formation of microcracks, respectively, leading to poor grindability of the ore.
3.2. Effect of Cooling Methods on Grinding Kinetics
To further analyze the influence of cooling methods on the grinding characteristics of materials, the particle size distribution of the grinding products at different grinding time was fitted using Equation (1). The fitting process is illustrated in
Figure 6. The grinding kinetic parameters for products processed with different cooling methods are presented in
Table 3,
Table 4,
Table 5 and
Table 6. The fitting optimization parameter
R2 is consistently greater than 0.95, indicating that the fitting quality meets the required standards.
Figure 6a and
Table 3 show that the parameters of the grinding particle size characteristic equation obtained using cooling mode I exhibit different trends as the grinding time increases. Specifically, the uniformity index
n increases and then decreases as the grinding process progresses, while the particle characteristic parameter
b increases with grinding time. After 2 min of grinding, the uniformity index
n reaches its maximum value of 1.3063, and the optimal uniformity of the grinding product is achieved. Based on the corresponding particle characteristic parameter
b value of 0.0088, the
R-
R particle size characteristic equation, as shown in Equation (2), is further derived.
Figure 6b and
Table 4 show that the uniformity index
n in the grinding particle size characteristic equation obtained using cooling mole II exhibits a trend of initially increasing and then decreasing as the grinding time increases. The particle characteristic parameter
b increases gradually. Moreover, the uniformity index
n reaches its maximum value of 1.3363 after 2 min of grinding, with the corresponding particle characteristic parameter
b value of 0.0080. The corresponding
R-
R particle size characteristic equation is given in Equation (3).
Figure 6c and
Table 5 show that the uniformity index
n in the grinding particle size characteristic equation obtained using cooling mode III exhibits a trend of initially increasing and then decreasing as the grinding time increases. The particle characteristic parameter
b increases gradually. Additionally, the uniformity index
n reaches its maximum value of 1.3190 after 2 min of grinding, with the corresponding particle characteristic parameter
b value of 0.0078. The corresponding
R-
R particle size characteristic equation is given in Equation (4).
Finally,
Figure 6d and
Table 6 show that the uniformity index
n in the grinding particle size characteristic equation obtained using cooling mode IV exhibits a trend of initially increasing and then decreasing as the grinding time increases. The particle characteristic parameter
b increases gradually. Additionally, the uniformity index
n reaches its maximum value of 1.3109 after 2 min of grinding, with the corresponding particle characteristic parameter
b value of 0.0083. The corresponding
R-
R particle size characteristic equation is given in Equation (5).
Overall, the comparison reveals that, for the same grinding time, the kinetic parameter n corresponding to cooling mode II is the largest, indicating superior uniformity.
The results from the grinding kinetics model indicate that the uniformity coefficient n for cooling mode II is highest, signifying the most uniform particle size distribution. The n value reflects the consistency of particle fragmentation during the grinding process, which is closely related to the mechanical properties and structural uniformity of the ore. In cooling mode II, the combination of a larger average pore size and abundant microcracks ensures that the ore is crushed more uniformly during grinding. The cracks expand uniformly under mechanical stress, thereby preventing the formation of excessively coarse or excessively fine particles. The grinding time exhibits a trend of initially increasing and then decreasing, with the cooling method influencing both the magnitude and rate of this trend. During the initial stage of grinding, the number of surface cracks and pores in the ore increases, resulting in a more uniform crushing speed and an increase in the value of n. As grinding progresses, the remaining particles possess greater structural integrity, causing the rate of crack expansion to slow down, which leads to a gradual decrease in the value of n. Cooling mode II maintains a relatively high value of n throughout the grinding process because its initial crack and pore structure is more conducive to continuous and uniform crushing.
3.3. Effect of Cooling Methods on Physical and Chemical Properties
The hydrogen-based mineral phase transformation is a complex physical and chemical process [
20]. Different cooling methods induce variations in the physical and chemical properties of the ore, leading to differences in the grinding characteristics of the processed ore. Therefore, analyzing the impact of cooling methods on the physical and chemical properties of hydrogen-based mineral phase products is crucial for further research into the mechanisms. This study focused on analyzing the pore size characteristics and microscopic morphology of the products using tests such as BET and SEM.
Table 7 presents the BET analysis results of the products following hydrogen-based mineral phase transformation under different cooling methods. According to
Table 7, the products obtained by different cooling methods, ranked from largest to smallest in specific surface area, are: cooling method IV > I > III > II. In terms of total pore volume, from largest to smallest, the ranking is: III > I > II > IV. For average pore diameter, from largest to smallest, the ranking is: II > III > IV > I.
Figure 7 presents the adsorption–desorption isotherms and pore size distribution diagrams of the products obtained after hydrogen-based mineral phase transformation under different cooling methods. As shown in
Figure 7a, the adsorption–desorption isotherms of the products obtained via different cooling methods exhibit distinct hysteresis loops, all classified as H3 type. The H3-type hysteresis loop primarily indicates the presence of mesopores or macropores.
Figure 7b shows that, by comparing micropore volume (pore diameter < 2 nm), the water-quenched–cooled samples subjected to water immersion cooling to 200 °C contain the highest number of micropores, while the water-quenched–cooled samples contain the fewest. In terms of mesopore volume (2 nm < pore diameter < 100 nm), the reducing atmosphere-cooled samples contain the largest number of mesopores, whereas the water-quenched–cooled samples contain the fewest. Comparing the total pore diameter reveals that the water-quenched–cooled samples, after cooling to 200 °C in a reducing atmosphere, exhibit a larger number of pores with the largest diameter, which is more conducive to improving subsequent grinding efficiency.
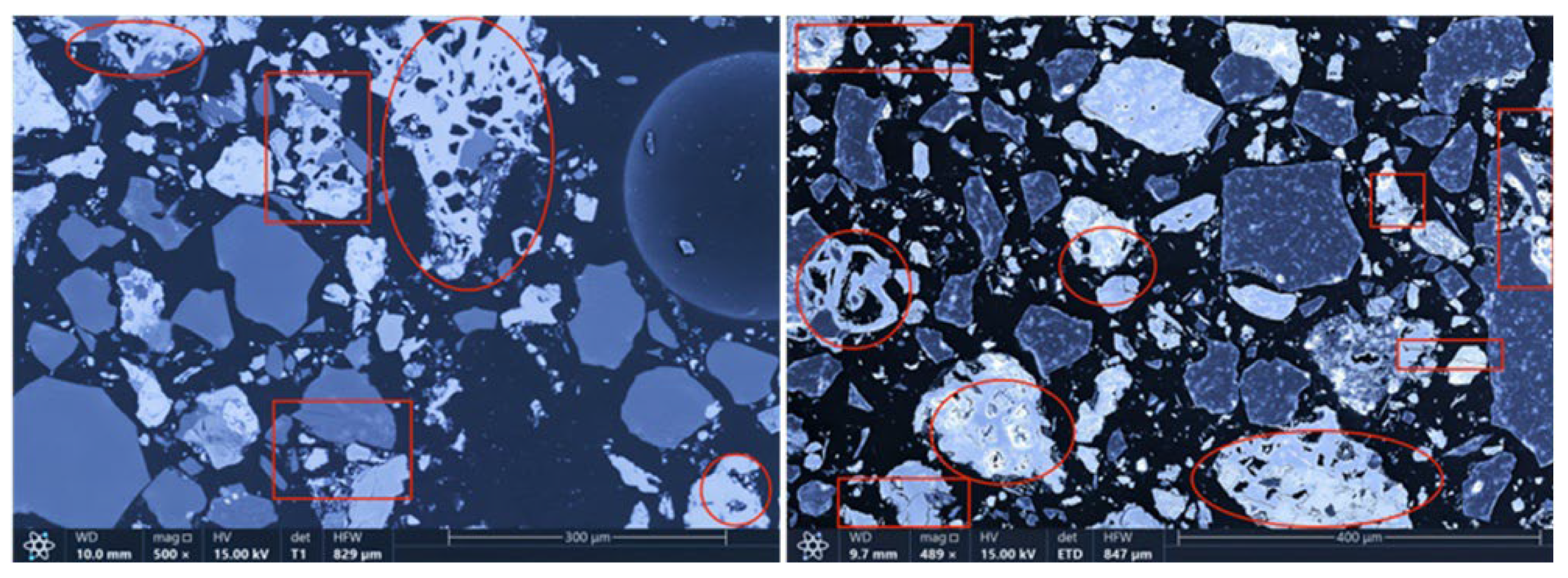
The microstructures of the products obtained via different cooling methods were examined using scanning electron microscopy (SEM), with the results presented in
Figure 8,
Figure 9,
Figure 10 and
Figure 11. As shown in the figures, the number of cracks and micropores on the surface of the ore particles varies with the cooling method. Ore particles treated with water quenching after cooling to 200 °C in a reducing atmosphere exhibit the highest number of cracks and pores. Studies have shown that during the rapid quenching and cooling process following hydrogen-based mineral phase transformation, differences in thermal properties among the minerals in the ore induce stress at mineral interfaces, leading to contraction and the formation of microcracks. The presence of microcracks increases the degree of monomer dissociation and the exposed surface area of iron minerals during subsequent grinding. Consequently, the water-quenched–cooled samples exhibit more cracks and pores than the reduced-atmosphere-cooled samples.
In addition, restoring the reducing atmosphere further reduces the mineral phase conversion products, whereas natural cooling leads to magnetite oxidation. The most intense phase transformations in the mineral phase conversion products occur within the 400–500 °C. At this temperature range, exposure to air causes Fe3O4 to oxidize to form α-Fe2O3. Hematite has significantly higher hardness and Young’s modulus than magnetite, so the oxidation of Fe3O4 increases the hardness and modulus of the mineral phase conversion products, reducing their grindability and impairing grinding characteristics. In the water quenching process, magnetite in the mineral phase conversion products experiences slight oxidation. However, if the mineral phase conversion products are sealed and cooled below 400 °C before exposure to air, Fe3O4 remains largely unoxidized. Therefore, water quenching after reducing the atmosphere to 200 °C effectively prevents the newly formed magnetite from oxidizing into harder, denser hematite, preserving the beneficial effects of hydrogen-based mineral phase conversion on the grinding characteristics of iron ore. Additionally, samples cooled by reducing the atmosphere to 200 °C and then water quenched exhibit the highest number of cracks and pores in the mineral particles. Therefore, this cooling method is more suitable for subsequent grinding, dissociation, and separation operations than the other three methods discussed in this paper.