Abstract
The efficient recovery of platinum (Pt) from spent catalysts is of critical strategic importance to alleviate resource scarcity and supply chain dependencies. This study developed an ultrasonic-assisted leaching process using a H2SO4-NaCl system for Pt extraction from spent petroleum catalysts. Single-factor experiments were first conducted to identify the preliminary effects of key parameters. Subsequently, Response Surface Methodology (RSM) based on a Box–Behnken design was employed to model and optimize the interactive effects of ultrasonic power, sulfuric acid concentration, leaching time and NaCl concentration. The results demonstrated that ultrasonic power had the most significant influence on Pt leaching efficiency. The optimized conditions were determined as ultrasonic power of 300 W, H2SO4 concentration of 60%, leaching time of 100 min, and NaCl concentration of 0.10 mol/L. Under these optimal parameters, the Pt leaching rate reached approximately 99.8%, validating the model’s high accuracy and reliability. This work provides an efficient and stable technical pathway for the sustainable recycling of platinum from secondary resources.
1. Introduction
Platinum group metals (PGMs), as key strategic scarce metals, are core strategic resources supporting strategic emerging industries such as high-tech energy, high-end manufacturing, catalytic engineering and national defense science and technology. However, the global platinum group metal mineral resources show an extremely concentrated geographical feature. The combined reserves of southern Africa, Eastern Europe and North America account for 99% of the global total. In contrast, China’s platinum group metal resources are severely scarce: the prospective reserves are less than 360 tons, accounting for only 0.76% of the global industrial total reserves and 0.45% of the global prospective reserves, respectively [1]. This gap between supply and demand of resources not only restricts the supply chain security of related industries in our country, but also highlights the strategic significance of developing efficient platinum group metal resource utilization technologies and innovative leaching processes to reduce the degree of dependence on foreign sources [2].
Platinum (Pt), as the most core consumer variety among platinum group metals (PGMs), accounts for approximately 90% of the global consumption [3]. In light of the current predicament in China where platinum resources are scarce and the degree of dependence on foreign imports remains high, developing and applying advanced platinum recovery technologies has become a key approach to alleviating this prominent imbalance between supply and demand. The catalyst industry, as the most core industrial application field of platinum (Pt), according to statistics, the global demand for Pt in the petrochemical catalyst sector will account for more than 50.6% in 2024 [4]. This data fully highlights the irreplaceability of Pt in key industrial processes such as catalytic conversion in petrochemicals, and also confirms the core impact of this field on the global balance of Pt resource supply and demand. With the continuous shrinkage of the exploitable amount of high-grade platinum group metal deposits and the increasing economic input in the mining and processing of minerals, the efficient recovery and resource utilization of related industrial products and terminal products with platinum as the core component at the end of their service life have become the main research content to ensure the supply and demand of platinum [5]. The industrial-scale prospects for the recycling of platinum group metals are broad. Benefiting from the continuous improvement of the strategic value of resources and the demand of the clean energy market, the recycling and utilization of Pt secondary resources have received attention. This not only accelerates the industrialization process but has also become an important source of supply for Pt components. However, this field still faces many technical bottlenecks: complex raw material composition, long separation and purification processes with high energy consumption, and difficulty in balancing recovery efficiency and product purity. In the future, green process innovation and system optimization are needed to achieve key breakthroughs in large-scale application and sustainable development. [6]. In conclusion, the recycling and reuse of waste resources have become a key means to fill the gap of precious platinum group metals. Failed petrochemical catalysts, rich in platinum components, have extremely strong recycling and utilization value [7].
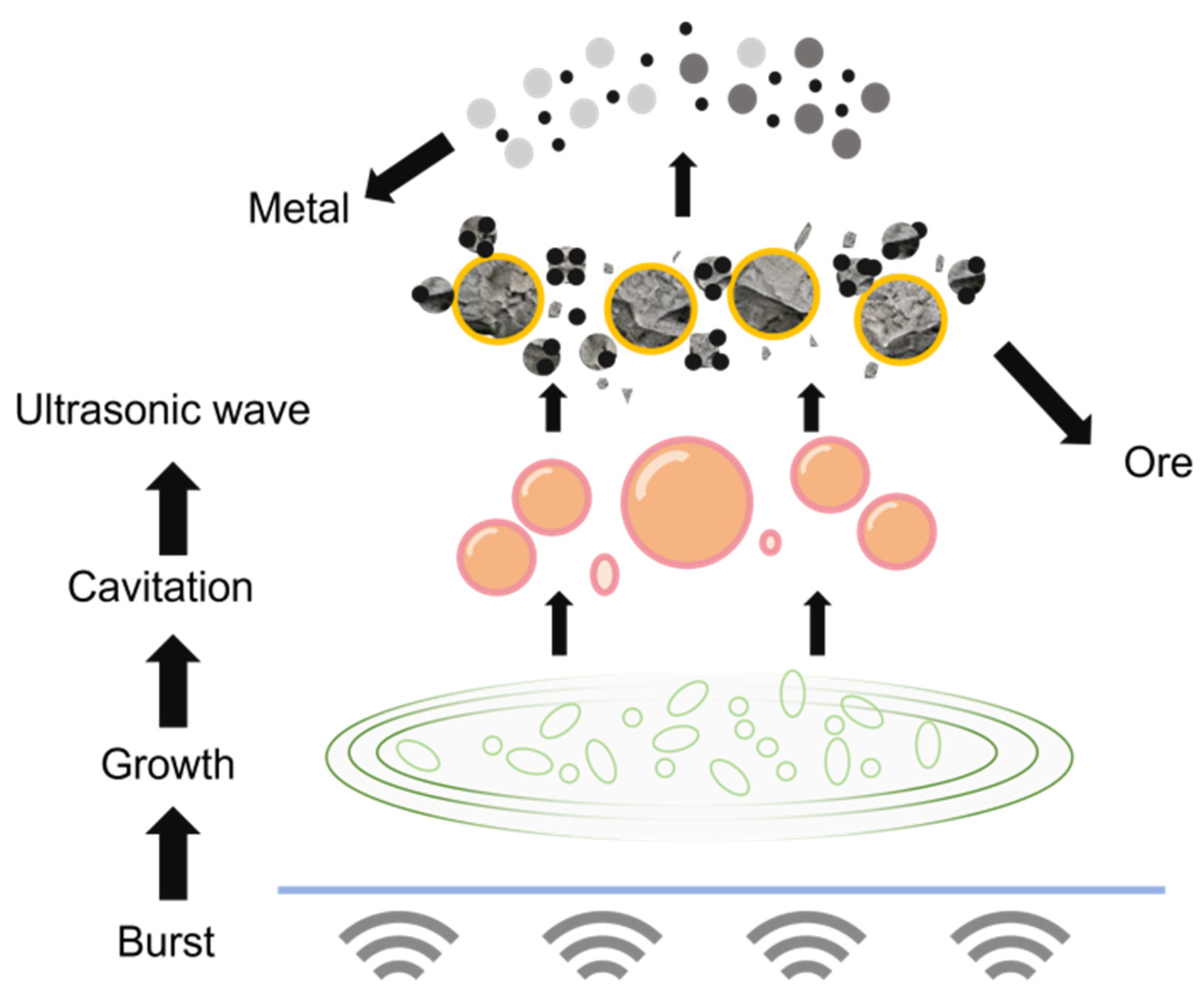
The application of efficient and cost-controllable platinum recovery methods can not only reduce the demand for raw ore resources but also effectively alleviate the environmental pollution caused by discarded catalysts to the ecological environment. In the traditional field of platinum extraction technology, the wet leaching process, with its advantages such as mild reaction conditions and controllable selectivity, has become the main technical process for platinum extraction under conventional circumstances [8]. The core mechanism of this type of process is the coordination dissolution effect in the acid medium. By using sulfuric acid, hydrochloric acid and corresponding chlorides, oxides, etc., as leaching agents, the dissolution of platinum is achieved through the coordination interaction between ligands and Pt ions, providing a prerequisite for subsequent separation, purification and recovery [9]. On this basis, introducing ultrasonic waves as the leaching enhancement technology can further optimize the leaching efficiency [10]. Its core function stems from the synergistic effect of cavitation, mechanical and thermal effects triggered by ultrasonic waves: When ultrasonic waves propagate in a liquid, they generate periodic pressure changes. During the negative pressure phase, tiny bubbles expand rapidly, while during the positive pressure phase, the bubbles collapse instantly. This is the cavitation effect. At the moment of collapse, local high temperatures ranging from 500 to 15,000 K and high pressures from 100 to 5000 mpa can be produced, accompanied by intense shock waves and micro-jets. It can not only destroy the passivation film on the surface of platinum particles, promote the micro-fragmentation of the alumina carrier to expose the hidden platinum sites, but also vigorously stir and wash the surrounding medium, enhancing the material transfer of [11]. Meanwhile, the high-frequency vibration of liquid molecules triggered by ultrasonic waves forms a mechanical effect, achieving liquid stirring and particle dispersion, and further enhancing the contact efficiency between the leaching agent and platinum. The thermal effect where the vibration energy is absorbed by the medium and converted into thermal energy can increase the system temperature, optimize the physical properties of the liquid, simultaneously enhance the efficiency of mass transfer and heat transfer, and create favorable conditions for the leaching reaction [12]. Ultrasonic vibration can significantly reduce the thickness of the diffusion layer at the solid–liquid interface, lower the mass transfer resistance between the leaching agent and platinum ions, accelerate particle breakage and induce the generation of oxidative free radicals [13]. The ultrasonic enhancement mechanism and the coordination dissolution effect of the acid medium form a synergy, which not only shortens the time for leaching to reach equilibrium, but also breaks through the mass transfer limitations of traditional wet leaching, achieving a significant improvement in leaching efficiency and providing technical support for the platinum extraction process [14]. The principle is shown in Figure 1.
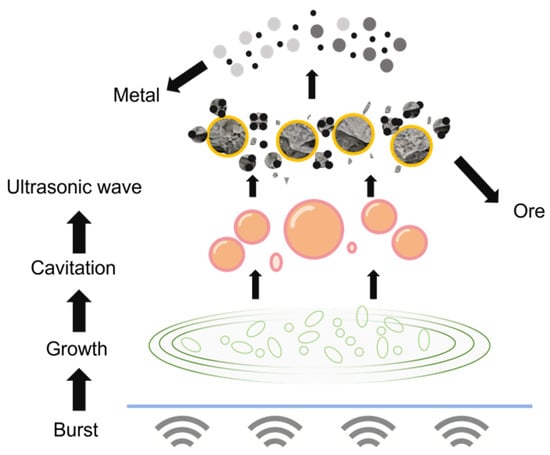
Figure 1.
Schematic diagram of ultrasonic cavitation principle.
Based on the above process logic and the technical advantages of ultrasonic-enhanced leaching, this study selected the ultrasonic-assisted H2SO4-NaCl system as the leaching medium to conduct leaching tests on platinum metals. On the basis of M.H.H. Mahmoud’s experiments, ultrasonic waves were introduced. This system can avoid the use of aqua or high-pressure conditions and is more operationally safe [15]. The reagents used in the experiment were concentrated sulfuric acid (H2SO4) and sodium chloride (NaCl), which played a synergistic role in the leaching system: the core function of H2SO4 was to create a stable acidic reaction environment, providing thermodynamic driving force for the dissociation of platinum from the Al2O3 carrier phase. NaCl acts as a chlorine source to release Cl−, and by forming a stable soluble chlorination complex with Pt, it enhances the dissolution kinetics process of platinum in acidic media [16].
The leaching efficiency of platinum is jointly influenced by many key factors, including sulfuric acid concentration, sodium chloride concentration, reaction temperature, liquid–solid ratio, reaction duration, ultrasonic power, etc. There exists an interaction mechanism among these factors. The traditional single-factor variable method can only investigate the influence law of a single factor in isolation and is difficult to accurately reveal the synergistic effect of the interaction of these factors on the leaching efficiency. This further restricts the optimization of the leaching process under multi-factor conditions [17].
The Response Surface Methodology (RSM), as a classic multivariable experimental design, process modeling and optimization technique, has the core advantage of being able to reveal the nonlinear interaction mechanism among multiple factors and accurately display the relationship among process parameters [18]. Therefore, in view of the complex characteristics of multiple factors in the platinum leaching process, this study adopts this method to conduct multi-factor optimization research, providing scientific support for achieving precise regulation of the leaching process. Compared with the traditional single-factor variable method, the response surface method (RSM) not only significantly reduces the experimental workload and shortens the experimental period, but also clearly clarifies the interaction among multiple factors, thereby effectively enhancing the scientificity and accuracy of experimental optimization [19]. In this study, by leveraging RSM technology, the extraction process conditions of platinum from spent catalysts under an ultrasonic-assisted H2SO4-NaCl leaching system were optimized. The regulation laws of key parameters such as sulfuric acid concentration, sodium chloride concentration, reaction temperature, liquid–solid ratio, reaction duration and ultrasonic power on the platinum leaching efficiency were deeply explored, and the optimal combination of process parameters was screened out. It provides theoretical support and technical references for the efficient recovery of platinum from spent catalysts, the recycling of mineral resources, and the research and development of green and environmentally friendly recovery processes.
2. Experimental Materials and Research Methods
2.1. Experimental Materials
The raw materials for this study were gray spherical platinum-containing spent catalysts supplied by a certain precious metal recycling enterprise in Xuzhou, China, as shown in Figure 2.

Figure 2.
Platinum-containing spent catalyst.
The elemental composition of the platinum-containing spent catalyst was quantitatively analyzed in both major and trace amounts by X-ray Fluorescence Spectrometry (XRF). The results showed that the quantitative detection value of platinum (Pt) in the sample was 660.016 g/t, and the main carrier component was alumina (Al2O3), as shown in Table 1.

Table 1.
Qualitative analysis of spent catalysts by XRF.
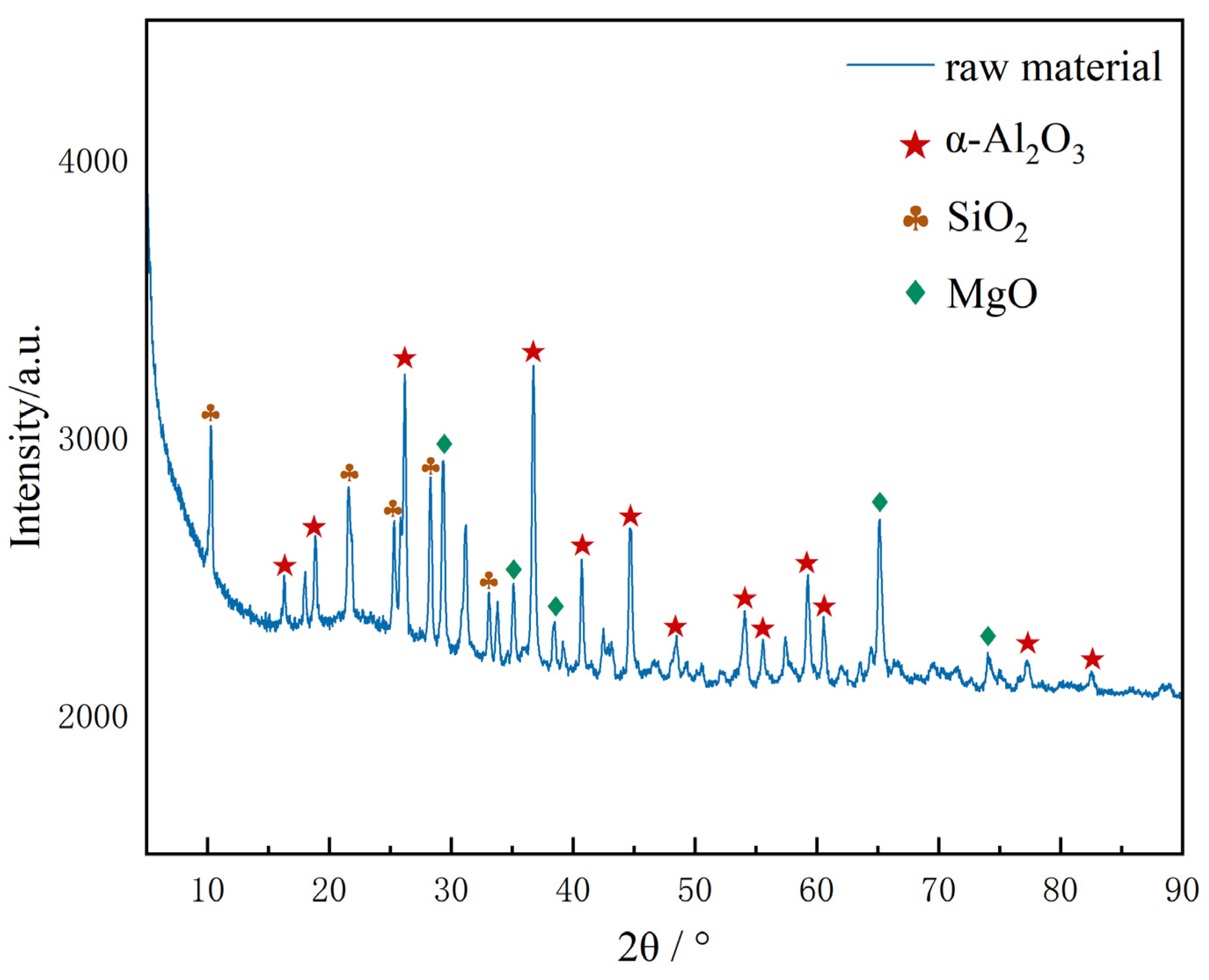
Figure 3 shows the X-ray diffraction (XRD) pattern of the platinum-containing spent catalyst raw materials used in the experiment. The pattern analysis indicates that the main components of the sample are Al2O3 and SiO2. Due to the low content of element Pt in the raw materials, its characteristic diffraction peak signal is masked by the diffraction peak of the strong carrier phase, and no obvious response is observed. Among the catalyst raw materials used, α-Al2O3 is the main phase. As a classic carrier widely applied in the field of precious metal catalysts, α-Al2O3 features excellent thermodynamic stability, mechanical strength and chemical inertness. Its crystal structure characteristics can provide a stable carrier for the Pt active component, ensuring the structural integrity during the catalytic reaction process [20]. To reduce the mass transfer resistance during the leaching process, the spent catalyst was ground to 80 mesh fine powder before the experiment to ensure full contact between platinum and the leaching agent.
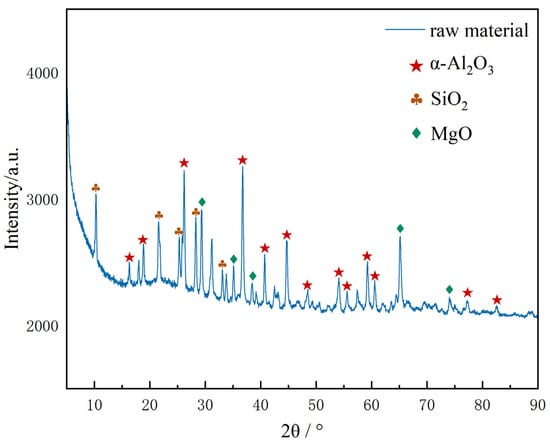
Figure 3.
XRD pattern of platinum-containing spent catalyst.
All experimental solutions were prepared with deionized water to effectively eliminate the interference of impurity ions, ensure the purity of the experimental system, and guarantee the reliability and repeatability of the data. The ultrasonic generator selected for the experiment is of the KC-300W specification model produced by Suzhou Kangchuan Electronics Co., Ltd., From Suzhou, China. First, the influence of each factor on the leaching rate was identified in the single-factor experiment. Then, ultrasonic waves were introduced on the basis of the traditional leaching experiment, and the ultrasonic frequency was set for the control group, with the parameter range being 60–360 W. Ultrasonic waves as leaching enhancement technology. The ultrasonic field, through the synergistic effect of cavitation, micro-jet and mechanical vibration, can thin the diffusion layer at the solid–liquid interface, break the residual passivation film on the platinum surface, and simultaneously accelerate the mass transfer rate of Cl− to the platinum active site. This, in turn, forms a gain with the core dissolution mechanism of the H2SO4-NaCl system, breaking through the leaching equilibrium bottleneck under the basic optimal conditions, achieving a secondary improvement in platinum leaching efficiency. The optimal process parameters based on H2SO4 concentration, temperature, liquid–solid ratio, NaCl concentration and ultrasonic power were determined through multi-factor collaborative optimization.
2.2. Basic Experimental Operation Procedures
The leaching solution used in the experiment was precisely prepared by compounding 98% (by mass fraction) concentrated sulfuric acid (corresponding to a molar concentration of 18.4 mol/L), analytical grade sodium chloride and ultrapure water. A chlorinated acidic system suitable for platinum selective leaching was constructed by adjusting the component parameters Among them, the volume fraction of concentrated sulfuric acid is controlled within the range of 30% to 70%, and the NaCl concentration is set at 0.02 to 0.14 mol/L to ensure that the system has both carrier dissolution capacity and platinum coordination dissolution activity [15].
The specific experimental operation process is as follows: ① based on the preset liquid–solid ratio (4–14 mL/g), precisely transfer the corresponding volume of H2SO4-NaCl complex leachate through a pipette and transfer it to a 50 mL beaker; ② place the beaker on the constant-temperature magnetic stirrer and the ultrasonic generation device, and use the water bath heating method to heat the leachate to the preset temperature (40–90 °C) and keep it at a constant temperature for 30 min to ensure the controllability of the experimental conditions. At the same time, adjust the ultrasonic equipment to the preset power (60–360 W) to ensure that the ultrasonic field evenly covers the reaction system. ③ Add 5 g of the pre-treated waste catalyst sample that has been ground and sieved (80 mesh) to the system, and simultaneously adjust the stirring speed to 450 rpm to enhance solid–liquid mass transfer. Then, immediately start the ultrasonic and leaching reaction. After the reaction reaches the preset duration of 2 h, simultaneously terminate the ultrasonic, stirring and heating programs, remove the beaker and let it cool naturally to room temperature. ⑤ Centrifuge the solid–liquid mixture at 8000 rpm for 10 min, repeating the operation three times to ensure thorough separation. Collect the supernatant filtrate and leaching residue, respectively, for subsequent detection and analysis. ⑥ The platinum concentration in the filtrate was precisely quantitatively analyzed by inductively coupled plasma atomic emission spectrometry (ICP-OES). Combined with the initial grade of platinum in the raw materials, the final leaching rate was calculated according to the leaching rate formula:
3. The Influence of Single-Variable Experimental Conditions on the Platinum Leaching Effect
3.1. The Influence of Sulfuric Acid Concentration on Platinum Leaching Efficiency
The concentration of H2SO4 serves as the core regulatory factor in the platinum leaching process. The H+ produced by the dissociation of H2SO4 in aqueous solution is the key thermodynamic condition driving the oxidative dissolution reaction of platinum. The presence of hydrogen ions can regulate the system potential and promote the transfer of platinum ions from the solid phase to the liquid phase through coordination. H+ can protonate the hydroxyl groups on the surface of the α-Al2O3 carrier, weaken the interaction between the carrier and the platinum active component, and promote the exposure of the encapsulated platinum particles. Meanwhile, high concentrations of H+ can optimize the coordination environment of Cl−, promote the formation of stable [PtCl6]2− soluble complexes between Pt4+ and Cl−, and enhance the mass transfer process of platinum from the solid phase to the liquid phase [21].
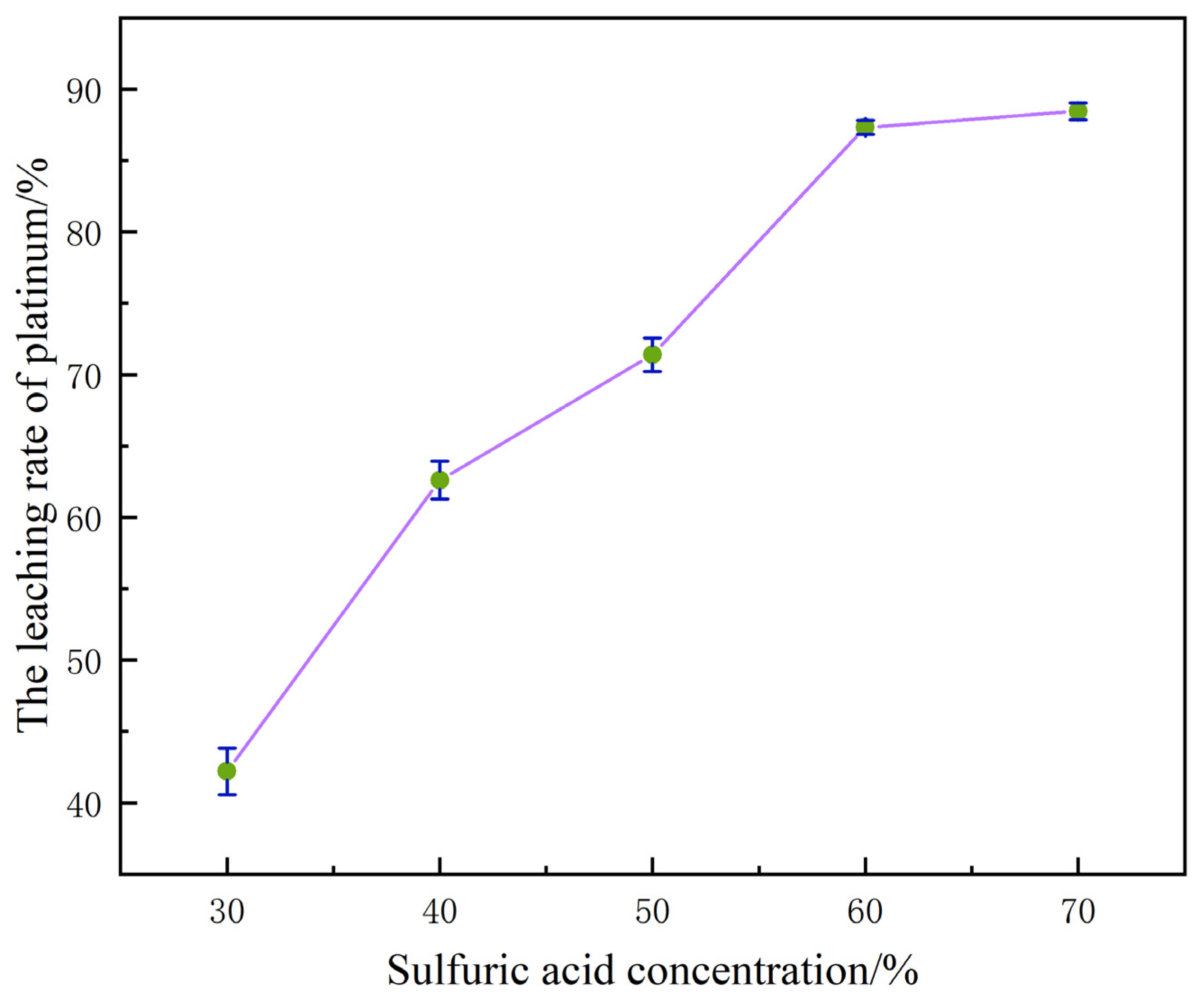
When the concentration of H2SO4 is within the range of 30% to 70%, the dynamic variation law of the platinum leaching rate is shown in Figure 4.
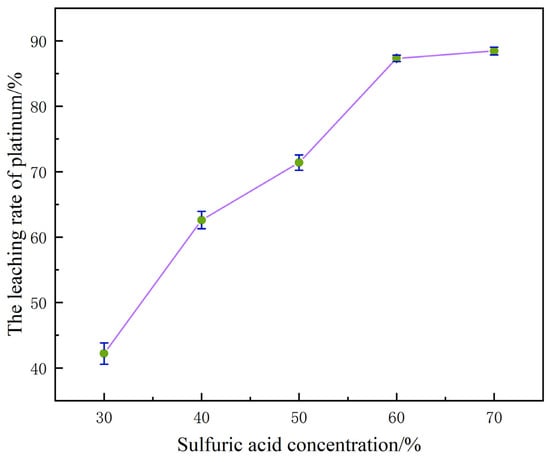
Figure 4.
The influence of sulfuric acid concentration on the platinum leaching rate.
When the concentration of H2SO4 increased from 30% to 70%, the leaching rate of platinum showed a significant upward trend, rapidly climbing from the initial 42.53% to 88.78%. The essence of this phenomenon is that the continuous increase in H+ concentration in the system not only accelerates the oxidation process of platinum but also promotes the transformation of metallic platinum into Pt2+. In a system where acidic media coexist with chlorides, Pt2+ can further form stable soluble compounds, thereby effectively inhibiting the precipitation of platinum in the form of precipitates or its retention in the solid phase. It can be seen from this that increasing the dosage of H2SO4 within the above-mentioned concentration range can promote the more efficient transition of platinum from the solid phase to the liquid phase system, thereby significantly optimizing the leaching efficiency of platinum [22].
However, the higher the concentration of H2SO4, the better the leaching effect is not necessarily. When the concentration of H2SO4 increased from 60% to 70%, the platinum leaching rate only achieved a weak increase of 1.3%. This phenomenon indicates that at this time, the H+ concentration in the system has approached the saturation threshold of the platinum oxidation dissolution and the proproton reaction of the α-Al2O3 carrier, and the destruction of the passivation film on the platinum surface and the weakening of the carrier interface effect by H+ have reached the optimal effect. Further increasing the acid concentration cannot further enhance the leaching rate of the reaction [23].
From this, it can be inferred that further increasing the concentration of sulfuric acid is no longer sufficient to substantially optimize the reaction kinetics rate. In addition, the use of high-concentration sulfuric acid can also trigger multiple negative effects: on the one hand, the increase in acid consumption will directly push up the operating costs of the process; a strongly acidic environment can cause intense corrosion to process equipment and materials in contact. This not only shortens the service life of experimental devices but may also undermine their operational stability.
3.2. The Influence of Chloride Ion Concentration on the Platinum Leaching Effect
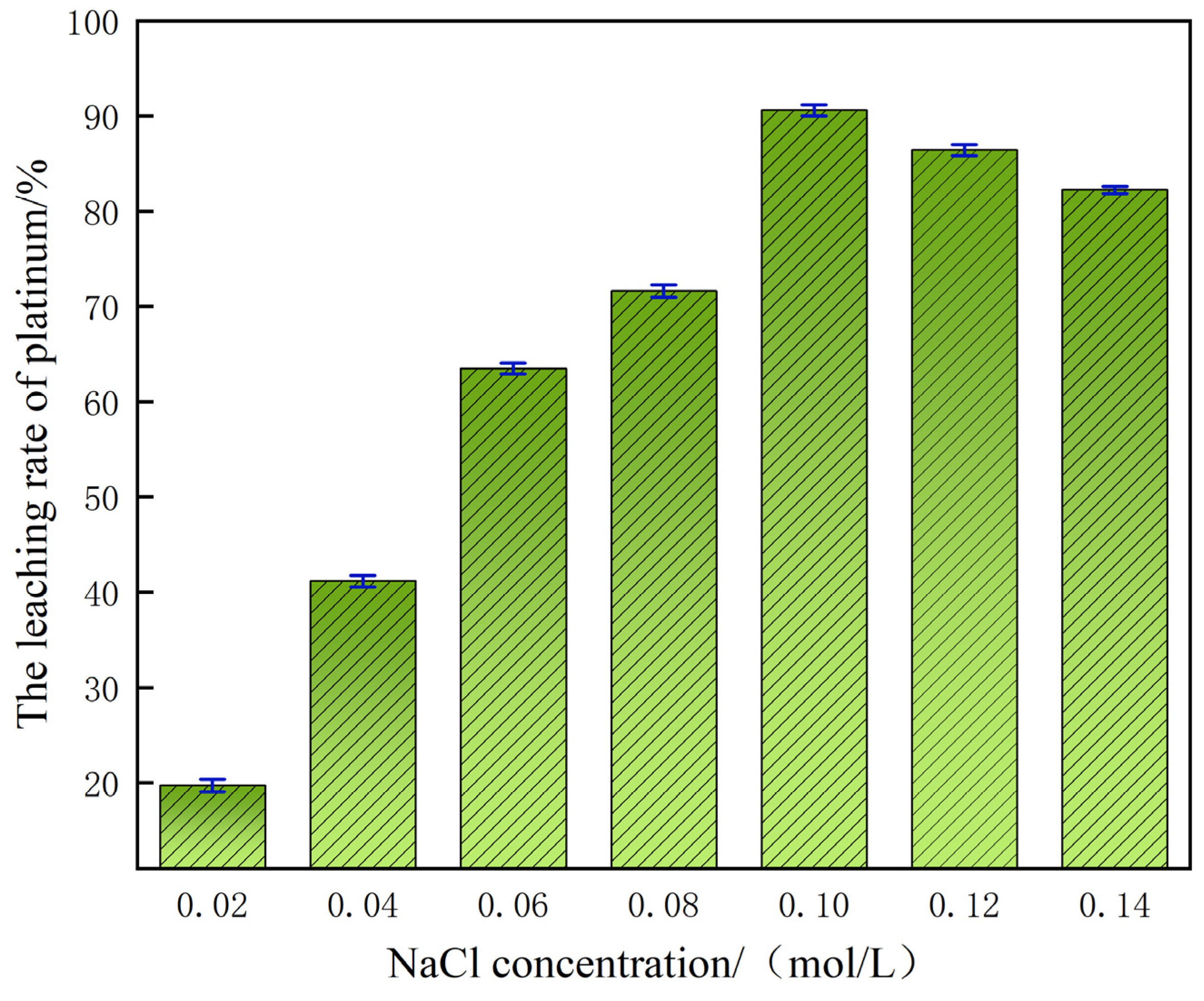
In a sulfuric acid system with a mass fraction of 60%, the concentration of chloride ions (Cl−) shows an extremely significant regulatory effect on the leaching rate of platinum. As shown in Figure 5, when the NaCl addition concentration gradually increased from 0.02 mol/L to 0.10 mol/L, the increase in Cl− concentration in the system accelerated the dissolution process of platinum, promoting the formation of a stable soluble hexachloroplatinate complex [(PtCl6)2−] between platinum and Cl−. The formation of this complex can not only effectively inhibit the precipitation of platinum in the solution, but also significantly enhance the leaching efficiency of platinum, ultimately achieving a high leaching rate of 90.70%. However, when the NaCl concentration reaches the critical value of 0.10 mol/L, the gain effect of further increasing the Cl− concentration on the platinum leaching rate shows a significant attenuation trend. The experimental results show that an excessively high concentration of Cl− will instead have an inhibitory effect on the leaching of platinum. The mechanism of this phenomenon is as follows: the presence of high concentrations of Cl− will cause the viscosity of the reaction system to increase, thereby reducing the efficiency of the mass transfer process, restricting the diffusion rate of reactants in the solution phase, and ultimately hindering the dissolution reaction of platinum [24].
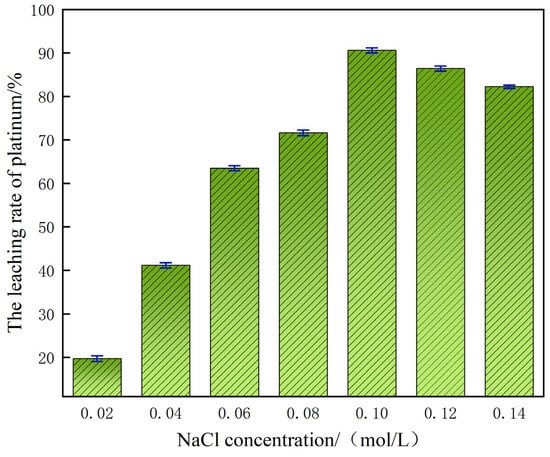
Figure 5.
Effect of NaCl concentration on platinum leaching efficiency.
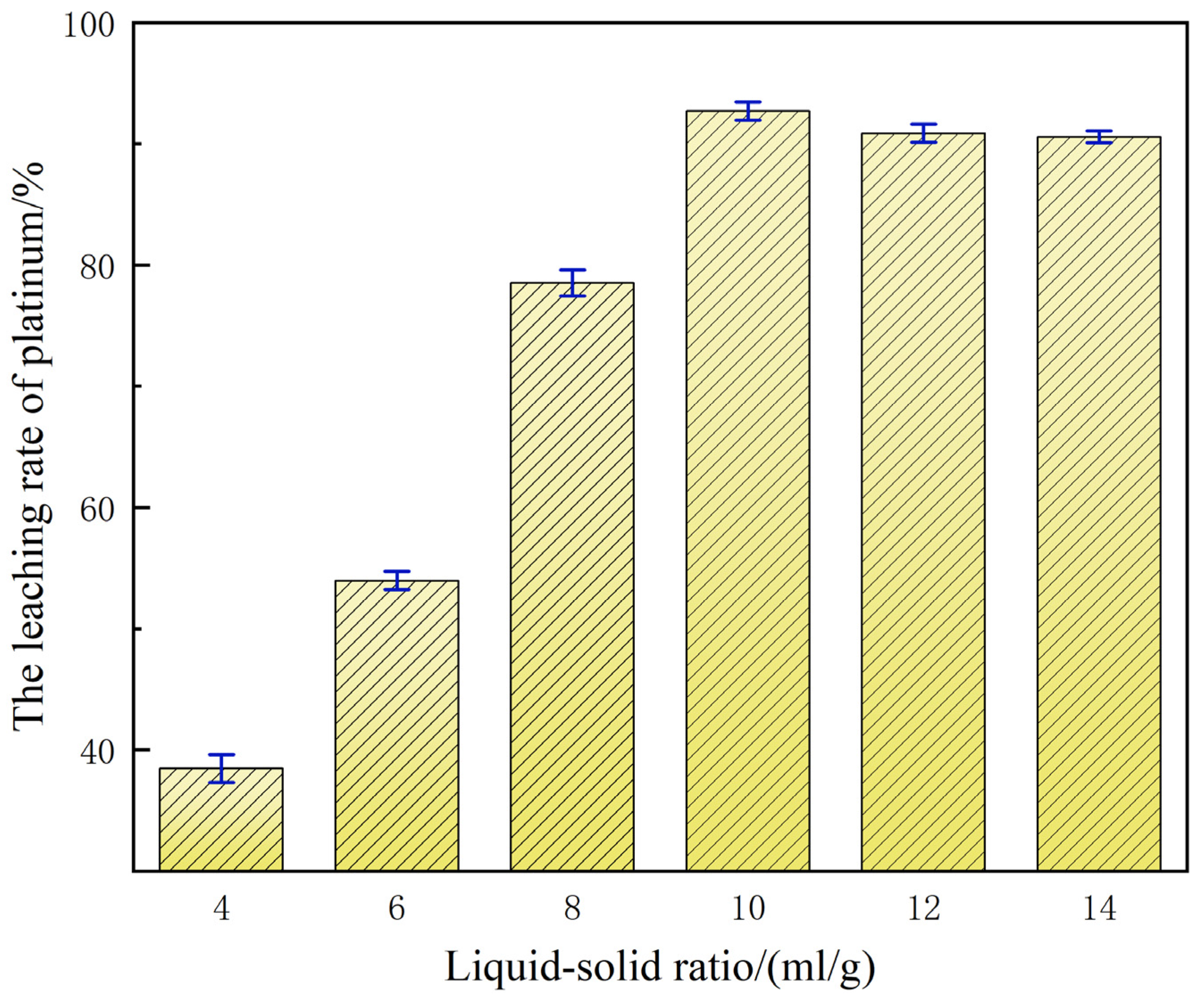
3.3. The Influence Law of Liquid–Solid Ratio on Platinum Leaching Efficiency
With 0.1 mol/L NaCl added as a leaching aid in a 60% H2SO4 system, the liquid-to-solid ratio demonstrated a significant regulatory effect on platinum leaching efficiency, as shown in Figure 6.
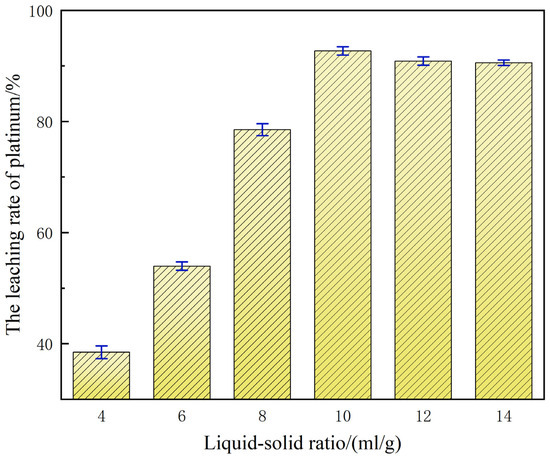
Figure 6.
Influence of liquid-to-solid ratio on platinum leaching efficiency.
When the liquid-to-solid ratio is 4:1, the leaching rate of platinum is merely 38.80%. This relatively low leaching efficiency is primarily ascribed to the insufficient effective concentrations of H+ and Cl− in the system under this liquid-to-solid ratio, which constrains the sufficient oxidation and dissolution reactions of platinum, ultimately leading to the leaching performance failing to meet the expected standards [25]. As the ratio gradually increased to 10:1, the reaction environment improved the sufficient liquid volume and maintained the acidic environment and stable formation of chloro-complexes, raising platinum leaching rate to 92.97%. Compared with the leaching efficiency at a liquid-to-solid ratio of 4:1, this represents a significant increase of 54%. Nevertheless, further elevation of the liquid-to-solid ratio beyond 10:1 (e.g., 12:1) engenders a progressive dilution of H+ and Cl− activities, thereby diminishing the driving force for both oxidative and complexation reactions. Concomitantly, the augmented volumetric excess escalates reagent usage and post-treatment expenditure, manifesting a pronounced downward trajectory in platinum leaching efficiency.
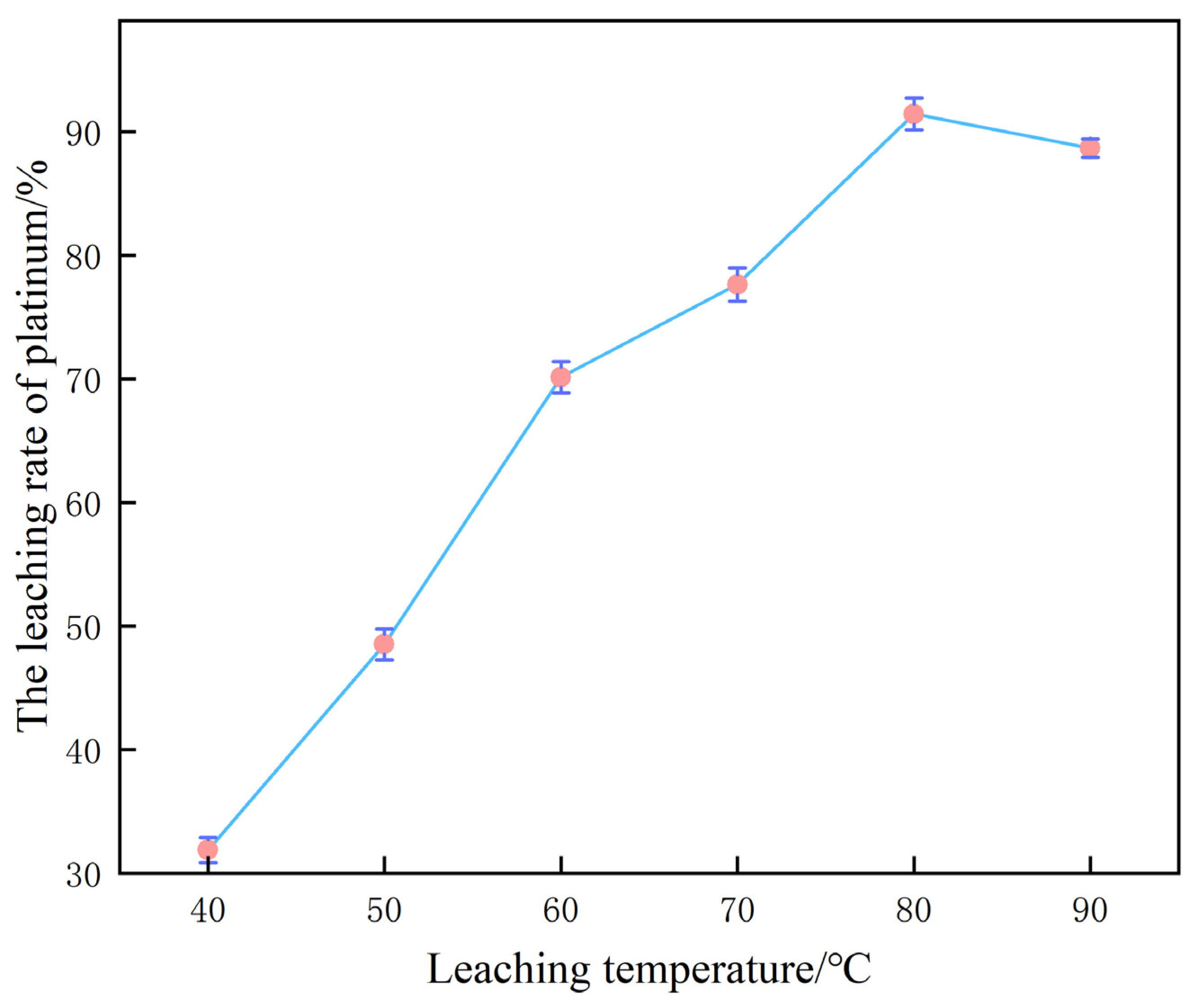
3.4. The Regulatory Effect of Temperature on the Platinum Leaching Rate
Under the baseline parameter matrix of 60% H2SO4, 0.10 mol/L NaCl and a liquid-to-solid ratio of 10 mL/g, reaction temperature is identified as the pivotal intensive variable governing platinum leaching efficiency; the corresponding experimental dataset is presented in Figure 7.
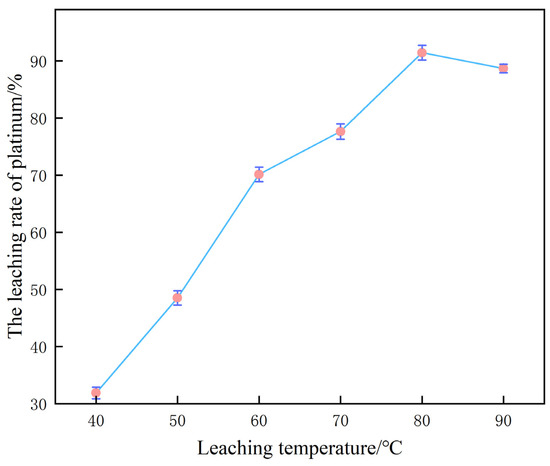
Figure 7.
Influence of temperature on platinum leaching efficiency.
When the reaction temperature was raised incrementally from 40 °C to 80 °C, the platinum leaching yield exhibited a pronounced ascending trajectory, ultimately attaining 91.5%. From a reaction kinetics perspective, the temperature elevation substantially increases the diffusion coefficients of solutes in the aqueous phase, thereby accelerating the migration of Pt-containing species, H+ and Cl−. This intensification facilitates both the formation of the (PtCl6)2− complex and the concurrent evolution of H2, resulting in a marked enhancement of mass transfer kinetic efficiency.
Owing to the decisive role of H+ and Cl− diffusion toward the reactive interface in the dissolution of platinum, elevated temperatures effectively attenuate mass transfer resistance and curtail the time required to attain equilibrium, thereby affording a pronounced increase in leaching efficiency [26].
Upon further elevation to 90 °C, a conspicuous retrogression in platinum leaching yield was observed. This deterioration is ascribed to the concurrent amplification of undesirable side phenomena: whereas the dissolution kinetics of the target metal are accelerated, the solubilities of ancillary elements are likewise increased, liberating additional impurity ions into the lixivium. These alien ions can perturb the platinum dissolution trajectory via competitive adsorption or by forming secondary precipitates, and may destabilize the (PtCl6)2− complex, collectively culminating in a diminished leaching efficiency [27].
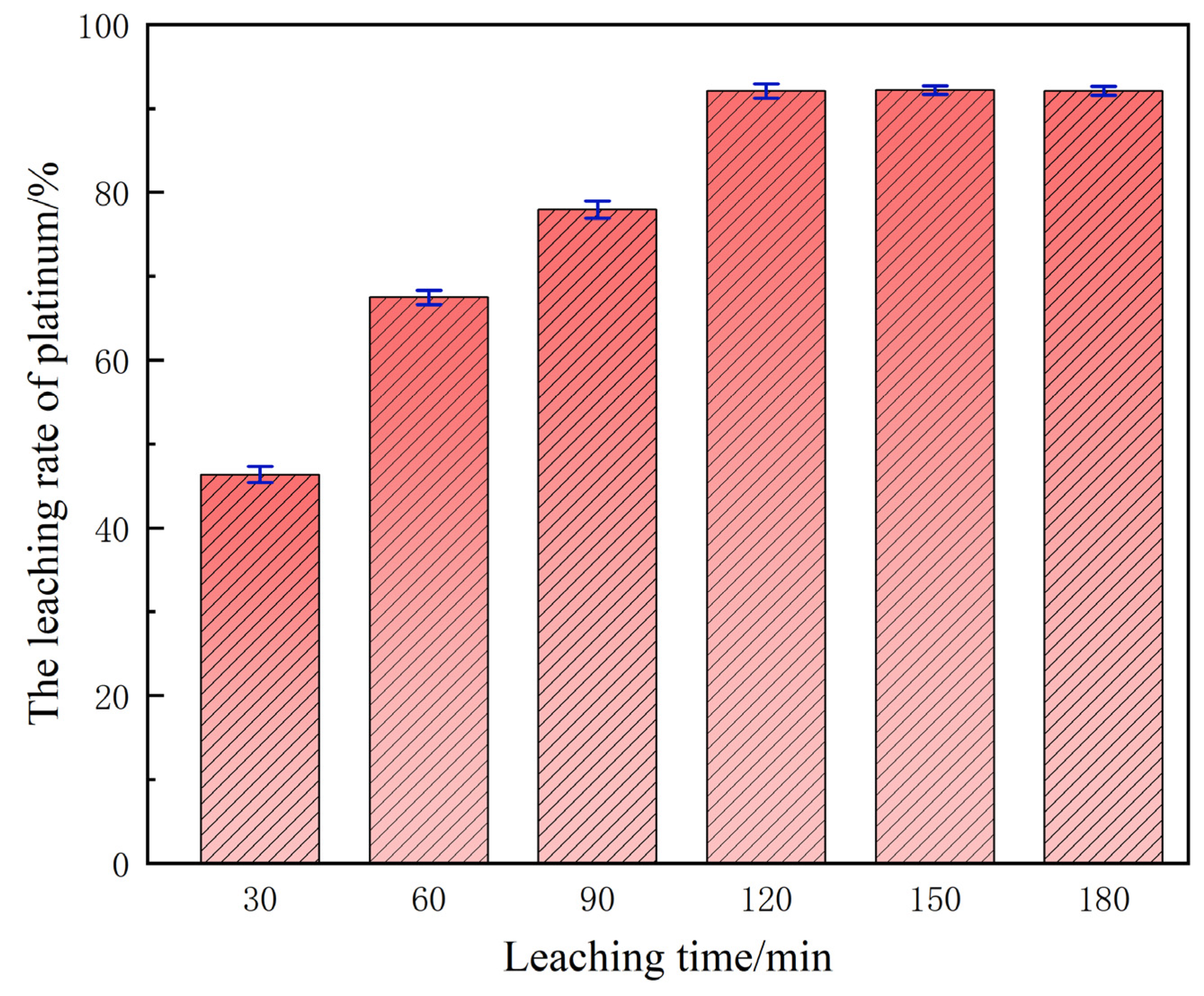
3.5. Influence of Leaching Duration on Platinum Extraction Efficiency
Under the aforesaid baseline optimum conditions, the dependence of platinum leaching efficiency on residence time is depicted in Figure 8. During the initial stage (30–60 min), the yield remains depressed because interfacial mass transfer is rate-limiting: the ingress of H+ and D toward the platinum active sites is incomplete and the superficial passivation layer is only partially disrupted, so the oxidative turnover frequency is low. Prolonging the leaching horizon to 120 min raises the platinum recovery to 92.01%.
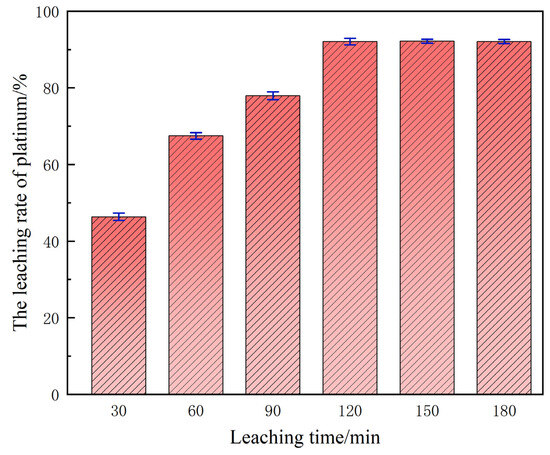
Figure 8.
The influence of time on the leaching rate.
This interval constitutes the fast-reaction regime: the passivation film has been dissociated, and H+/Cl− are afforded unhindered access to the platinum surface. Beyond 120 min the leaching plateau is essentially flat, evidencing that the system has attained thermodynamic equilibrium—the chemical potentials of H+ and Cl− in solution and the residual reactivity of solid-phase platinum are mutually saturated. Any further extension of residence time cannot displace this equilibrium constraint; it merely inflates energy expenditure and temporal cost, eroding the economic viability of the process.
Systematic single-factor investigations have established 120 min as the optimal leaching duration for the conventional system. Under this condition, both the oxidative dissolution of platinum and its subsequent chloro-complexation proceed to completion, affording a compromise between maximal extraction efficiency and process economy, thereby furnishing a baseline parameter set for the subsequent ultrasound-intensified optimization.
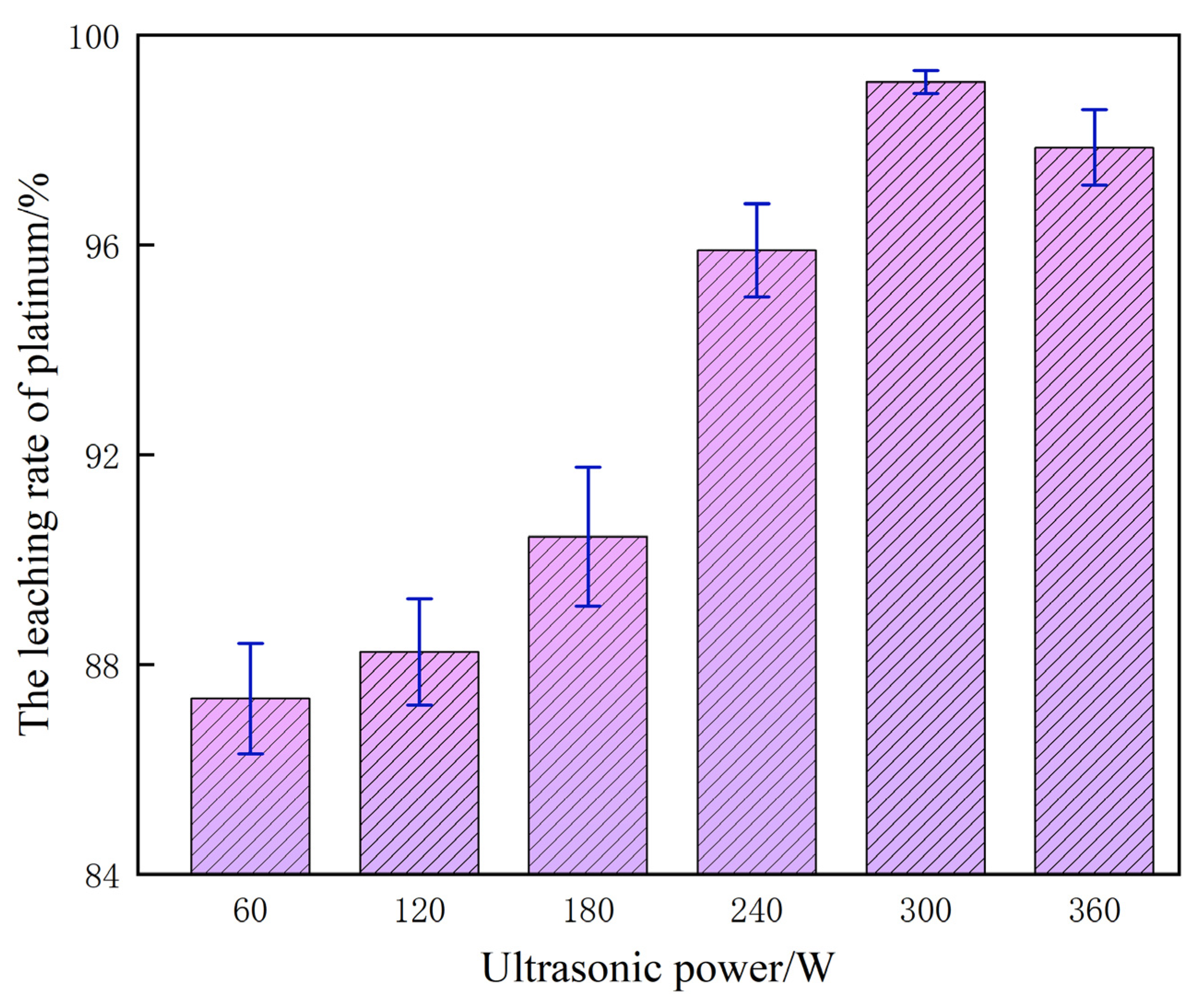
3.6. Effect of Ultrasonic Power on Platinum Leaching Efficiency
Under the reference parameters of 60% H2SO4, 0.10 mol/L NaCl, 80 °C, liquid–solid ratio of 10 mL/g, and reaction time of 120 min, ultrasonic power rather than temperature is the key strengthening variable controlling the kinetics of platinum leaching. The corresponding experimental dataset is shown in Figure 9.
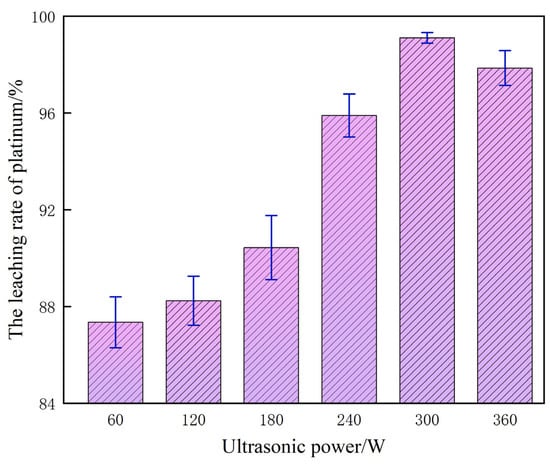
Figure 9.
The influence of ultrasonic waves on the platinum leaching rate.
Building on the preceding experiments, in the absence of ultrasonic assistance the platinum leaching process is constrained by the inherently low mass transfer efficiency across the solid–liquid interface; H+ and Cl− cannot adequately penetrate to the active sites embedded within the catalyst particles, resulting in a depressed leaching yield, a protracted reaction cycle and a forced reliance on elevated H2SO4 concentrations. Upon introducing ultrasound, the power regulation exerts a pronounced nonlinear influence on extraction performance: as the power increases from 60 W to 300 W, the Pt leaching yield rises markedly. This enhancement is primarily ascribed to the localized high-temperature and high-pressure environments generated by ultrasonic cavitation, which effectively disrupt the passivation layer on the catalyst surface and intensify interfacial contact among Pt, H+, and Cl−. Simultaneously, micro-jets induced by acoustic streaming create micro-scale turbulence, significantly promoting mass transfer kinetics and accelerating both the oxidative dissolution and complexation–diffusion of platinum.
At an acoustic power of 300 W, the synergistic interplay of cavitation, micro-jet perturbation and interfacial intensification reaches its optimum; H2SO4 and NaCl can fully access the Pt-active sites, driving the leaching efficiency to 99.12% and simultaneously maximizing both rate and recovery. Beyond this threshold, a further increase to 360 W yields only a marginal increment that rapidly plateaus. This attenuation originates from excessive cavitation that generates a bubble-shielding layer, diminishing the effective transmission of ultrasonic energy, and localized overheating that promotes H2SO4 volatilization, destabilizes the reaction medium and, in conjunction with vigorous mechanical vibration, induces catalyst particle agglomeration—collectively impairing solid–liquid interfacial contact.
Integrating experimental evidence, intensification mechanisms and process–economics considerations, 300 W is identified as the optimal ultrasonic power, affording peak Pt leaching performance while avoiding energy redundancy and system disturbances associated with higher power levels.
3.7. Conclusion of the Single-Factor Experiment on Ultrasonic-Enhanced Platinum Leaching
In summary, the single-factor variable study on the leaching of Pt from spent catalysts by ultrasonic enhancement ultimately determined the optimal process parameters as follows: 60% H2SO4, leaching temperature 80 °C, liquid–solid ratio 10 mL/g, NaCl addition 0.10 mol/L, and ultrasonic power 300 W.
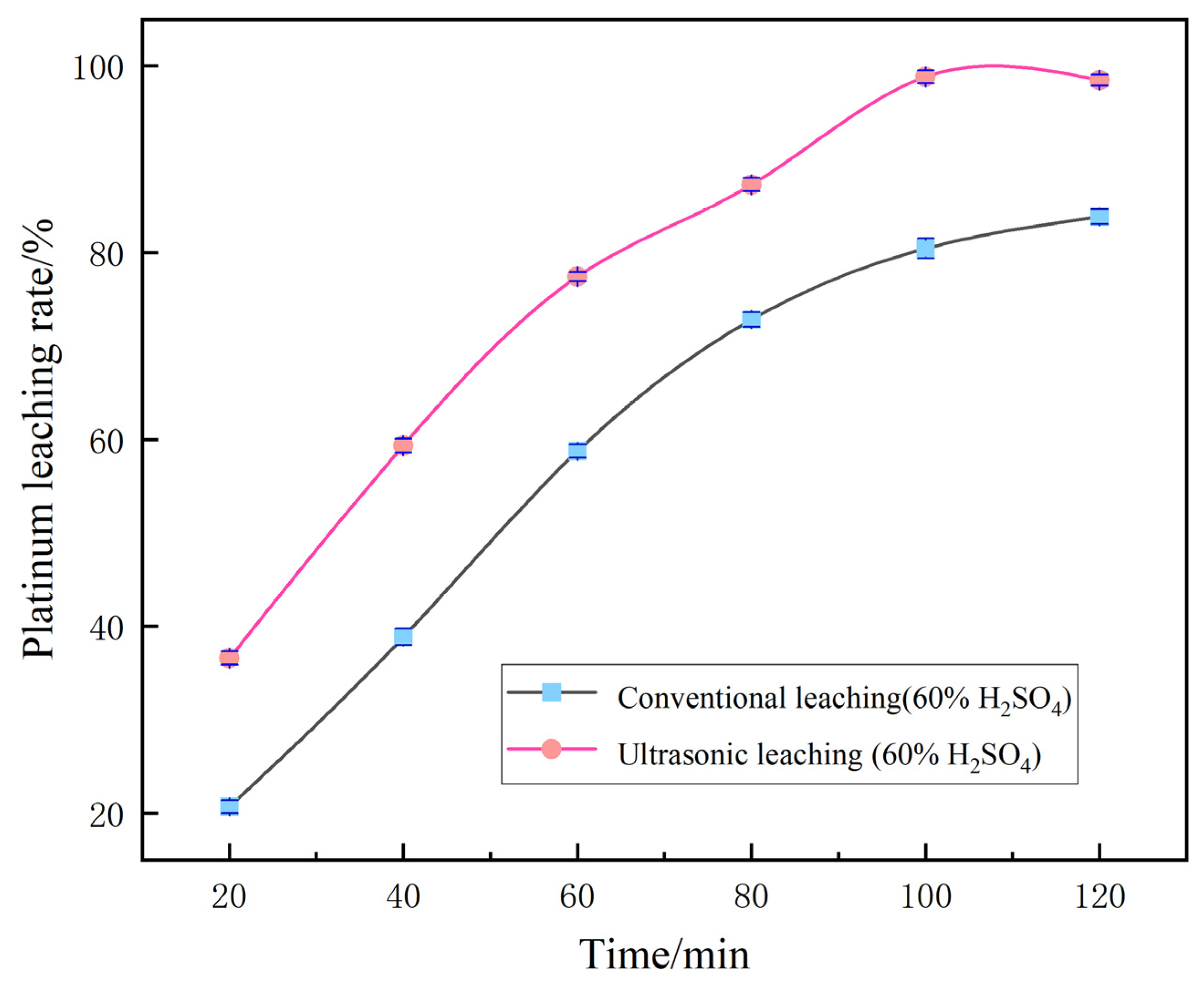
For the remaining single-factor variable “leaching time”, a parallel comparative experiment was further carried out between the ultrasound-assisted group and the conventional group without ultrasound. The results showed that the ultrasound-assisted group achieved the optimal Pt leaching rate of 99.12% at 100 min. However, when the residence time was extended to 120 min, determined by single-factor optimization, the Pt leaching rate of the ultrasound-assisted group decreased instead. The conventional non-ultrasound group showed a continuous and slow increase in the leaching rate over time. The specific data are shown in Figure 10.
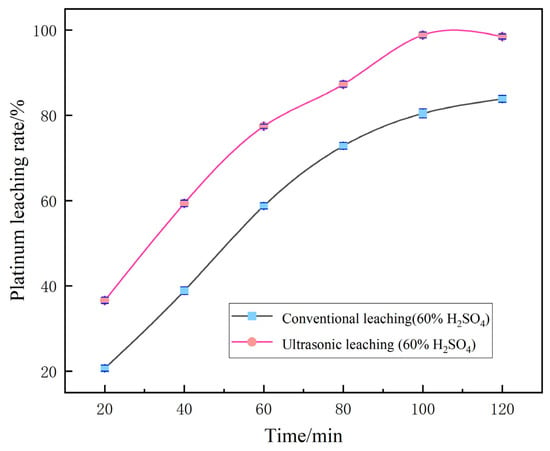
Figure 10.
Comparison between ultrasonic enhancement and conventional leaching.
This decline phenomenon is caused by the instantaneous high temperature and high pressure generated by excessive ultrasonic cavitation and the oxidation and decomposition of complex Pt species such as [PtCl6]2− in the leachate by free radicals, which leads to their redeposition. The cavitation stirring disrupts the mass transfer equilibrium at the solid–liquid interface, triggering the secondary adsorption of Pt ions by the tiny particles of the carrier. Moreover, the long-term combined effect of high temperature and ultrasonic waves causes the volatilization and loss of Cl−, which weakens the complexation and stability of the leaching agent. Therefore, the optimal retention time in the actual process is 100 min.
4. Response Surface Methodology (RSM) for Process Optimization
4.1. Experimental Design and Model Construction
Based on the optimal value range of platinum leaching rate for each factor clearly defined in the single-factor experiment, ultrasonic power (A), sulfuric acid concentration (B), time (C), and NaCl concentration (D) were selected as the independent variables for response surface optimization. With platinum leaching rate (Y, %) as the response value, the response surface method (RSM) was used for multi-factor collaborative optimization [28].
The experimental design was based on the Design-Expert 13.0.5 software and adopted the Box–Behnken design model. This model has the advantages of high experimental efficiency and precise characterization of interaction effects, and is suitable for the optimization system of four factors [29]. The coding levels and actual value ranges of each factor are shown in Table 2, where +1, 0 and −1, respectively, represent the maximum level, central level and minimum level of each factor. By using the encoded values to simplify the regression analysis, the interference caused by the unit differences in different variables is eliminated.

Table 2.
Coded and actual levels of the variables.
According to the BBD design plan, a total of 28 groups of experiments were constructed, and the experimental results are shown in Table 3. The leaching rate of the experimental data was analyzed by Design-Expert 13.0.5 software, and the regression equations of the platinum leaching rate and each factor were obtained as follows:

Table 3.
Box–Behnken experimental design matrix and observed responses.
4.2. Analysis of Response Surface Interaction Effects and Process Optimization
In the equation, Y denotes the response variable—platinum leaching yield (%); the independent variables A, B, C and D represent coded values of ultrasonic power (W), sulfuric acid concentration (%), leaching time (min) and NaCl concentration (mol/L), respectively. Analysis of variance (ANOVA) was employed to statistically evaluate the quadratic polynomial regression model, providing a systematic assessment of its goodness-of-fit and predictive reliability; the results are presented in Table 4.

Table 4.
Analysis of variance of the regression equation for platinum leaching.
In the ANOVA framework, the F value represents the ratio of the model explanatory power to random error, whereas the p-value quantifies the statistical significance of each term, p < 0.05 denotes significance, p < 0.001 indicates extreme significance [30]. The F value of this model is 22.63, with a corresponding p value < 0.0001, indicating that the model as a whole has extremely high statistical significance. Its prediction results are significantly better than random errors and can effectively represent the nonlinear relationship between various factors and the platinum leaching rate. The coefficient of multiple determination R2 = 0.9606, indicating that the model has excellent fitting accuracy and good consistency between the response value and the predicted value. The adjusted coefficient of multiple determination R2adj = 0.9181, suggesting that 91.81% of the variation in the platinum leaching rate can be explained by the main effects, interaction effects and quadratic effects of the selected independent variables, demonstrating significant practical application value of the model. The signal-to-noise ratio Adeq Precision is 17.664, far exceeding the critical value of 4.0, indicating that the model has sufficient effective signal strength and strong anti-interference ability, which can precisely guide the optimization of process parameters [31].
Further analysis of the significant influence of each factor is shown in Table 4: in the first-order terms, each factor has an extremely significant influence, and its influence intensity is ranked according to the F value as A > B > C > D. Among the interaction terms, AB and AC show obvious interaction effects, while the interaction effects of AD and CD are not significant. The quadratic terms A2, B2, C2 and D2 all exhibit extremely significant influences, indicating that the effects of each factor on the platinum leaching rate have significant nonlinear characteristics, that is, there exists an optimal value [32]. Based on the absolute values of the regression equation coefficients and the results of variance analysis, it can be known that the order of the influence weights of each factor on the platinum leaching rate is ultrasonic power > sulfuric acid concentration > NaCl concentration > time.
4.3. Response Surfaces and Analysis of Influencing Factors
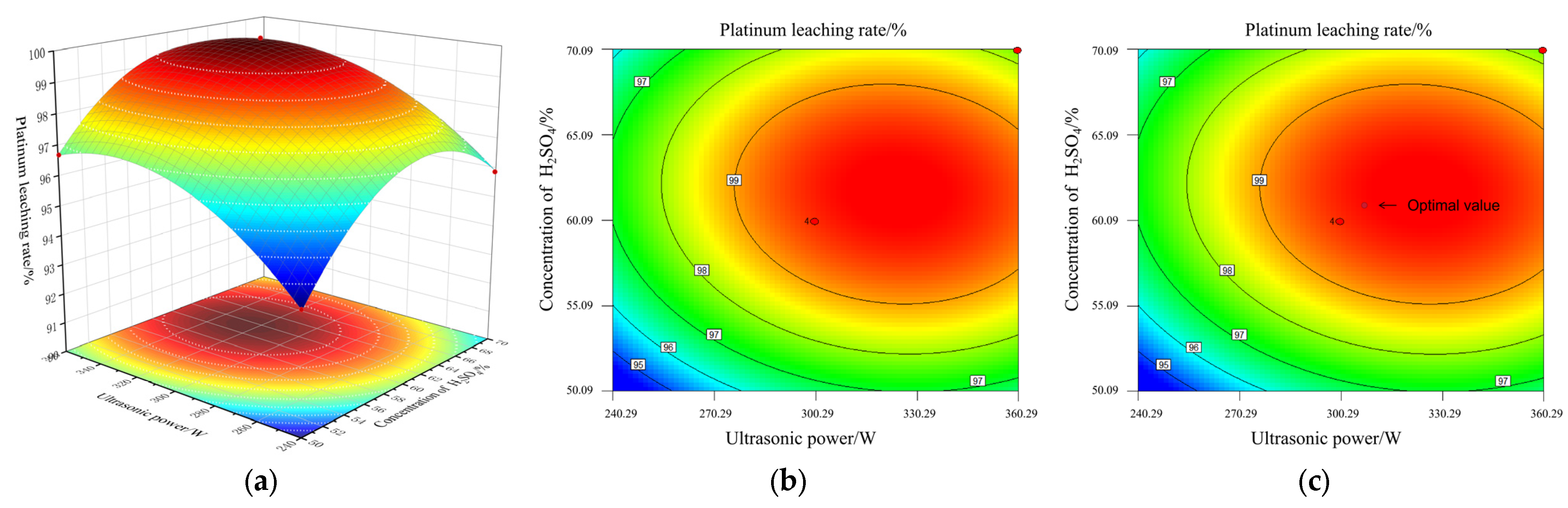
Figure 11a–c shows the interaction between AB and BC on variables. Figure 11a shows the response surface and contour plots generated by the combined effects of A and B on the Pt leaching rate under the conditions of fixed leaching time C = 100 min and NaCl concentration D = 0.1 mol/L. It can be seen from Figure 1 that the response surface shows a distinct convex arch shape, indicating that there is a significant nonlinear relationship between ultrasonic power and sulfuric acid concentration on the leaching rate. At a lower parameter level, synergistically enhancing these two factors can effectively increase the leaching rate. However, when either factor exceeds its optimal range, further increase will instead lead to a decrease in the leaching rate, which reveals a negative interaction effect between them. This phenomenon can be attributed to the following: a moderate ultrasonic cavitation effect combined with an appropriate sulfuric acid concentration can synergistically destroy the carrier structure and promote mass transfer and reaction; however, excessively high ultrasonic power may cause local overheating of the solution and generate an excessive bubble shielding effect, which instead inhibits mass transfer.

Figure 11.
The response surface and contour lines of the cross-influence between ultrasonic waves and concentrated sulfuric acid: (a) response surface AB; (b) contour lines AB; (c) the optimal value of AB.
The contour lines in Figure 11b are elliptical, and the red area in the figure indicates the optimal leaching rate range for effective platinum leaching.
According to the model prediction, at the marked points shown in Figure 11c, when the ultrasonic power is around 300 W as the experimental center point and the sulfuric acid concentration is around 60% as the experimental center point, the Pt leaching rate reaches its peak. This optimal parameter combination is highly consistent with the result of a leaching rate of ≥99.76% obtained by repeatedly testing the center point in the experimental design. This indicates that under the condition of moderate enhancement, the process efficiency and stability can reach the best balance rather than maximizing a single factor, providing a clear and reliable scientific basis for the adjustment of industrial parameters.
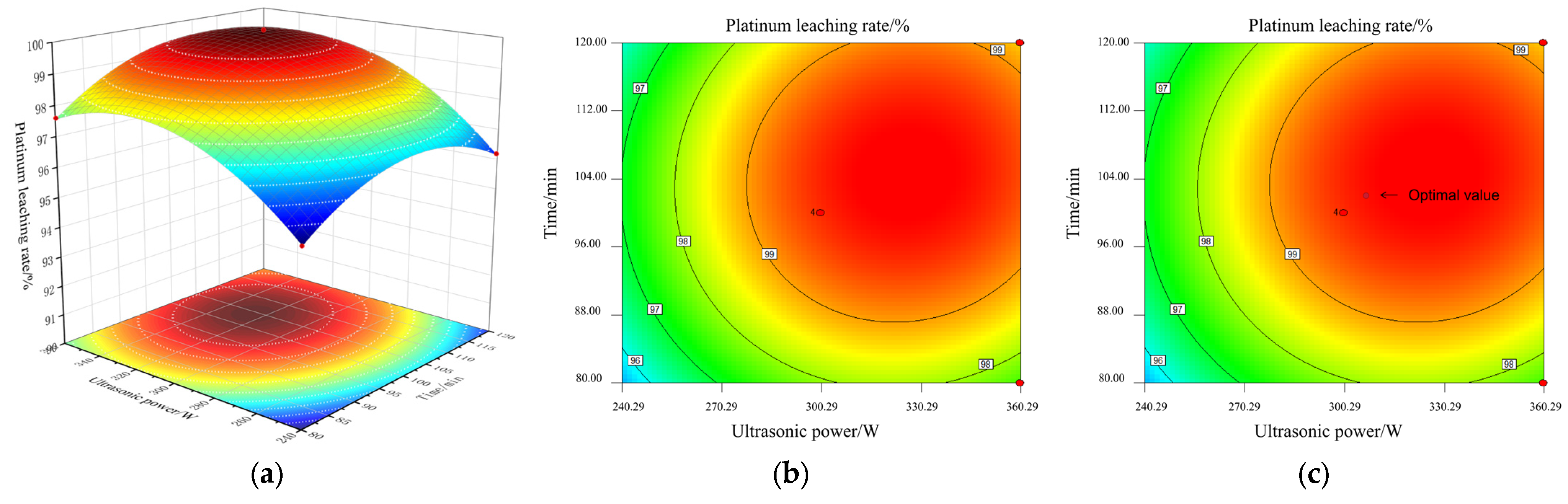
Figure 12a–c shows the response surface and contour lines of the interaction between A and C to the Pt leaching rate under the conditions of fixed concentrated sulfur concentration B = 60% and NaCl concentration D = 0.1 mol/L.
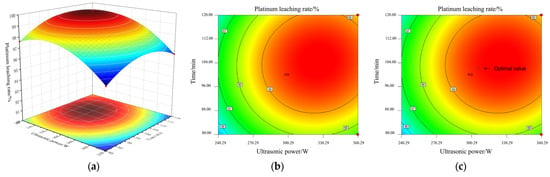
Figure 12.
Response surface and contour lines of the cross-influence of ultrasonic waves and time: (a) response surface AC; (b) contour lines AC; (c) the optimal value of AC.
As shown in (a), the response surface is in a typical convex arch shape. This indicates that there is a significant nonlinear relationship between ultrasonic power and leaching time on the leaching rate. Below their respective optimal values, increasing the ultrasonic power or extending the leaching time can effectively promote the leaching of platinum. However, when any factor exceeds its optimal threshold, further increasing the level of that factor will instead lead to a decrease in the leaching rate.
The mechanism lies in the kinetic balance regulation and side reaction competition effect during the ultrasonic-assisted leaching process: moderate ultrasonic power, through the instantaneous high temperature and high pressure generated by cavitation and the micro-jet effect, effectively breaks the passivation layer on the catalyst surface and enhances the mass transfer at the solid–liquid interface, providing sufficient kinetic driving force for the oxidation dissolution of Pt and the chloride complexation reaction. Sufficient and appropriate leaching time can ensure the efficient migration of Pt from the interior of the catalyst to the liquid phase and the stable formation of the [PtCl6]2− complex, ensuring that the leaching reaction approaches complete. However, excessive prolongation of the ultrasonic action time can trigger a series of negative effects: continuous cavitation causes the formed [PtCl6]2− complex to decompose and redeposit on the carrier surface; intense cavitation stirring can disrupt the mass transfer equilibrium at the solid–liquid interface, promoting the secondary adsorption of Pt ions in the leachate by the tiny particles of the carrier that are not completely dissolved. Meanwhile, the combined effect of ultrasonic waves and high temperatures will accelerate the volatilization and loss of Cl− in the form of HCl or Cl2, weakening the complexation and stabilization ability of the leachate for Pt. Ultimately, these factors jointly lead to a significant decrease in the Pt leaching rate.
It can be seen from the contour map in (b) that there is significant interaction between A and C. The red area in the figure indicates the optimal process window with the highest leaching rate.
According to the model prediction, at the marked points shown in (c), in the central point area where A is approximately 300 W and C is about 100 min, the Pt leaching rate can reach up to approximately 99.8%. This optimized combination provides a clear basis for the precise control of ultrasonic power and leaching time in actual processes.
Based on the prediction and optimization functions of the response surface model, the model provides the process parameters for maximizing the Pt leaching rate. Based on these parameters, the optimal values are near the experimental data and within the investigation range determined by the model, as shown in Table 4. In summary, the best combination of process conditions is as follows: ultrasonic power 300 W, sulfuric acid concentration 60%, time 100 min, NaCl concentration 0.10 mol/L. This combination is completely consistent with the center point conditions that have been repeatedly verified in the experimental design. The actual measured average leaching rate is as high as 99.8%, proving that this parameter combination is stable and reliable. The ultrasonic-enhanced leaching process can achieve the best results, providing a clear and repeatable optimization solution for the efficient recovery of platinum from spent catalysts. Table 5 lists the extraction rates obtained from the simulation for each parameter.

Table 5.
The optimal parameter model results within the process range.
5. Conclusions
Through single-factor pre-experiments, the effects of four factors, namely ultrasonic power, sulfuric acid concentration, time and NaCl concentration, on the platinum leaching rate were determined. When the ultrasonic power is 300 W, the sulfuric acid concentration is 60%, the time is 100 min and the NaCl concentration is 0.10 mol/L, the leaching effect of platinum reaches a relatively high level.
Experiments were conducted based on the Box–Behnken design of four factors and three levels, and a quadratic regression prediction model for platinum leaching rate was established. Analysis of variance indicated that the model was extremely significant (F value was 22.63, p value < 0.0001), and the fitting effect was excellent (R2 = 0.9606, R2adj = 0.9181), which could reliably interpret and predict the experimental data. Response surface analysis shows that the influence degree of each factor on the platinum leaching rate is in the following order: ultrasonic power > sulfuric acid concentration > NaCl concentration > time. The optimal process conditions obtained through model optimization are highly consistent with the experimental center point, namely, ultrasonic power of 300 W, sulfuric acid concentration of 60%, time of 100 min and NaCl concentration of 0.10 mol/L.
Under the above-mentioned optimized conditions, repeated experiments were conducted, and the platinum leaching rate remained stable at around 99.8%, which was highly consistent with the predicted values of the model, with extremely small relative errors. This fully validates the accuracy and reliability of the model, providing a solid process basis and theoretical guidance for the efficient and stable recovery of platinum from spent catalysts.
The process conditions determined in this study have good potential for transformation. Ultrasonic intensification can be applied in practical industrial applications, significantly shortening the leaching time and enhancing the leaching efficiency. It is suitable for precious metal resource recovery systems. The reagents used, such as sulfuric acid and sodium chloride, are all commonly used industrial raw materials with relatively low costs. This process is simple and easy to integrate with the existing hydrometallurgical production line. However, this process has limitations in practical industrial applications. Due to the high concentration of sulfuric acid, stricter requirements are placed on the corrosion resistance of the equipment, which may increase investment and maintenance costs. In addition, in actual spent petroleum catalysts, platinum (Pt) often coexists with other platinum group metals (PGMs) such as palladium (Pd) and rhodium (Rh). Their similar chemical properties may lead to mutual interference during the leaching process, making the process selectivity of Pt a key technical issue. In large-scale equipment, ultrasonic waves may have problems such as uneven energy distribution and intensified attenuation, which require further optimization of reactor design and mass transfer methods. Future research can further verify the stability of this process, overcome the technical problems related to selectivity, and promote its application in industry.
Author Contributions
X.Z.: Supervision, Validation, Writing—review and editing. X.S.: Conceptualization, Methodology, Writing—original draft. C.L.: Formal analysis, Funding acquisition, Writing—review and editing. Y.Y.: Investigation, Software, Data curation. B.R.: Resources, Visualization, Investigation. K.S.: Data curation, Methodology, Writing—review and editing. S.W.: Formal analysis, Validation, Writing—review and editing. H.P.: Supervision, Validation, Writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sponsored by Natural Science Foundation of Chongqing, China, Certificate Number: CSTB2024NSCQ-MSX0799; China Postdoctoral Science Foundation, Certificate Number: 2024M754220; Liaoning Provincial Science and Technology Program, Certificate Number: 2023-MSLH-256.
Data Availability Statement
The data will be made available upon request.
Conflicts of Interest
Author Chuan Liu was employed by the company Chongqing Sanhang Advanced Materials Research Institute Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Guo, J.M.; He, X.T.; Wang, H.; Wu, X.; Zhao, Y.; Li, Y.; Li, K.; Han, S. Reviews of metallurgical technology to recovery platinum group metals from secondary resource in China. Precious Met. 2012, 33, 18–23. [Google Scholar]
- Hao, H.; Geng, Y.; Tate, J.E.; Liu, F.; Sun, X.; Mu, Z.; Xun, D.; Liu, Z.; Zhao, F. Securing platinum-group metals for transport low-carbon transition. One Earth 2019, 1, 117–125. [Google Scholar] [CrossRef]
- Tong, X.; Dai, H.; Lu, P.; Zhang, A.; Ma, T. Saving global platinum demand while achieving carbon neutrality in the passenger transport sector: Linking material flow analysis with integrated assessment model. Resour. Conserv. Recycl. 2022, 179, 106110. [Google Scholar] [CrossRef]
- Hosseinzadeh, M.; Petersen, J. Recovery of Pt, Pd, and Rh from spent automotive catalysts through combined chloride leaching and ion exchange: A review. Hydrometallurgy 2024, 228, 106360. [Google Scholar] [CrossRef]
- Karim, S.; Ting, Y.P. Recycling pathways for platinum group metals from spent automotive catalyst: A review on conventional approaches and bio-processes. Resour. Conserv. Recycl. 2021, 170, 105588. [Google Scholar] [CrossRef]
- Dong, H.; Zhao, J.; Chen, J.; Wu, Y.; Li, B. Recovery of platinum group metals from spent catalysts: A review. Int. J. Miner. Process. 2015, 145, 108–113. [Google Scholar] [CrossRef]
- Zhao, Y.; Wu, X.; Wang, B. Recovery of platinum in spent Pt/TiO2 catalyst by carrier dissolution method. Precious Met. 2021, 42, 37–40. (In Chinese) [Google Scholar] [CrossRef]
- Benson, M.; Bennett, C.R.; Harry, J.E.; Patel, M.K.; Cross, M. The recovery mechanism of platinum group metals from catalytic converters in spent automotive exhaust systems. Resour. Conserv. Recycl. 2000, 31, 1–7. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, L.; Song, Q.; Xu, Z. Recovery of palladium as nanoparticles from waste multilayer ceramic capacitors by potential-controlled electrodeposition. J. Clean. Prod. 2020, 257, 120370. [Google Scholar] [CrossRef]
- Ma, S.; Xu, H.; Gao, T.; Hou, S.; Chen, M.; Zeng, Q.; Liu, Y.; Feng, W.; Gao, K.; Li, Y.; et al. Research Progress of Microwave Technology in Rare Earth Field. Hydrometall. China 2024, 43, 489–496. (In Chinese) [Google Scholar] [CrossRef]
- Radziuk, D.; Möhwald, H.; Shchukin, D. Ultrasonic activation of platinum catalysts. J. Phys. Chem. C 2008, 112, 19257–19262. [Google Scholar] [CrossRef]
- Han, Q. Ultrasonic processing of materials. Metall. Mater. Trans. B 2015, 46, 1603–1614. [Google Scholar] [CrossRef]
- Chen, L.; Lu, D.; Wang, Y.; Cheng, Z. Research and application of ultrasonic technology in mineral flotation and development trend. J. Nonferrous Met. 2021, 31, 1042–1056. (In Chinese) [Google Scholar] [CrossRef]
- Chen, X.; Ning, F.; Hou, J.; Le, Q.; Tang, Y. Dual-frequency ultrasonic treatment on microstructure and mechanical properties of ZK60 magnesium alloy. Ultrason. Sonochem. 2018, 40, 433–441. [Google Scholar] [CrossRef]
- Mahmoud, M.H.H. Leaching platinum-group metals in a sulfuric acid/chloride solution. JOM 2003, 55, 37–40. [Google Scholar] [CrossRef]
- Ding, Y. Mechanism and Application Research on Platinum Group Metal Enrichment from Spent Catalysts. Ph.D. Dissertation, University of Science and Technology Beijing, Beijing, China, 2019. (In Chinese). [Google Scholar]
- Zhang, C.; Yan, W.; Xiao, J.; Zhao, K.; Peng, S.; Wei, Y. Reductive leaching technology of manganese anode slag using tartaric acid as reducing agent optimized by RSM. Inorg. Chem. Ind. 2023, 55, 106–113. (In Chinese) [Google Scholar] [CrossRef]
- Cao, Y.; Peng, D.; Zhang, Q.; Liu, Y.; Yang, H. Optimization of recovery process for insoluble particles from waste platinum catalyst by HCl-DCEA system using response surface methodology. Chin. J. Nonferrous Met. Metall. 2024, 53, 37–45. (In Chinese) [Google Scholar] [CrossRef]
- Cornaja, S.; Zhizhkuna, S.; Vladiko, J. Kinetic Model of the 1, 2-Propanediol Oxidation Reaction in the Presence of 3wt% Pd/α-Al2O3 Catalyst. Key Eng. Mater. 2021, 903, 143–148. [Google Scholar] [CrossRef]
- Kim, M.-S.; Kim, E.-Y.; Jeong, J.; Lee, J.-C.; Kim, W. Recovery of platinum and palladium from the spent petroleum catalysts by substrate dissolution in sulfuric acid. Mater. Trans. 2010, 51, 1927–1933. [Google Scholar] [CrossRef]
- Kedari, S.; Coll, M.T.; Fortuny, A.; Goralska, E.; Sastre, A.M. Liquid-Liquid Extraction of Ir, Ru, and Rh from Chloride Solutions and Their Separation Using Different Commercially Available Solvent Extraction Reagents. Sep. Sci. Technol. 2005, 40, 1927–1946. [Google Scholar] [CrossRef]
- Nikoloski, A.N.; Ang, K.L. Review of the application of ion exchange resins for the recovery of platinum-group metals from hydrochloric acid solutions. Miner. Process. Extr. Metall. Rev. 2014, 35, 369–389. [Google Scholar] [CrossRef]
- Jia, M.; Jiang, G.; Chen, H.; Pang, Y.; Yuan, F.; Zhang, Z.; Miao, N.; Zheng, C.; Song, J.; Li, Y.; et al. Recent developments on processes for recovery of rhodium metal from spent catalysts. Catalysts 2022, 12, 1415. [Google Scholar] [CrossRef]
- Zhao, P.; Yi, W.; Cao, Q.; Zhang, B.; Chen, K.; Li, J.; Yao, C. Synthesis and Characterization of High-Purity Ultrafine Platinum Particles by Chemical Refining Method. J. Nanomater. 2019, 2019, 8524950. [Google Scholar] [CrossRef]
- Trinh, H.B.; Lee, J.C.; Suh, Y.J.; Lee, J. A review on the recycling processes of spent auto-catalysts: Towards the development of sustainable metallurgy. Waste Manag. 2020, 114, 148–165. [Google Scholar] [CrossRef]
- Zeng, L.; Yu, M.; Niu, H.; Cheng, M.; Zhao, W. Microwave-assisted Acid Leaching Test of Rare Earth Elements and Phosphorus in an Associated Rare Earth Phosphate Rock. Met. Mines 2024, 12, 217–222. (In Chinese) [Google Scholar] [CrossRef]
- Upadhyay, A.K.; Lee, J.C.; Kim, E.Y.; Kim, M.S.; Kim, B.S.; Kumar, V. Leaching of platinum group metals (PGMs) from spent automotive catalyst using electro-generated chlorine in HCl solution. J. Chem. Technol. Biotechnol. 2013, 88, 1991–1999. [Google Scholar] [CrossRef]
- Li, S.; Gao, G.; Liu, D.; Zhang, Y.; Hong, Y.; Zhao, Y.; Wu, L. Process Optimization of Li2CO3 Preparation from Lithium-Containing Wastewater Based on Box-Behnken Response Surface Method. Rare Met. Hard Alloys 2025, 53, 98–103+111. (In Chinese) [Google Scholar] [CrossRef]
- Dai, C.; Chen, P.; Sun, W.; Wang, H.; Yang, Y. Optimization of flotation process of ilmenite based on Box-Behnken response surface methodology. Iron Steel Vanadium Titan. 2023, 44, 1–7. (In Chinese) [Google Scholar] [CrossRef]
- Cui, G.; Yao, N.; Zhang, Y.; Su, D.; Song, L.; Song, T. Optimization of enzymatic hydrolysis of yak milk ghee based on response surface design. J. Food Saf. Qual. Insp. 2025, 16, 155–162. (In Chinese) [Google Scholar] [CrossRef]
- Chen, Y.; Lu, W.; Yang, Y. Optimization of Laser Paint Removal Process for Carbon Fiber Composite Matrix Based on Response Surface Analysis. Chin. J. Lasers 2023, 50, 1202208. [Google Scholar]
- Yakoumis, I.; Moschovi, A.; Panou, M.; Panias, D. Single-step hydrometallurgical method for the platinum group metals leaching from commercial spent automotive catalysts. J. Sustain. Metall. 2020, 6, 259–268. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.