Abstract
There is a need for a reliable and independent evaluation and confirmation of the postmortem interval (PMI) based on objective factors other than only postmortem changes or temperature measurements. Estimating the PMI by examining the concentration of potassium ions in the vitreous humor (VH) has a tradition in forensic toxicology dating back to the mid-20th century. So far, the methods for determining the presence of potassium ions have not been characterized in terms of the measurement uncertainty of types A and B, which directly affect the estimation of time of death uncertainty. The study evaluated the uncertainty of the determination of potassium ion concentrations using potentiometric ion-selective electrode (ISE) and microwave-induced plasma with optical emission spectrometry (MIP-OES) methods. In addition, the influence of the estimated measurement uncertainty on the results of the analysis of potassium ions in the VH was investigated. The estimated values of the expanded uncertainty determined by the type A experimental method indicate lower uncertainty in the determination of potassium ion concentration in the case of MIP-OES than ISE; that is, for concentrations of 2, 10, and 25 mg/L of potassium ions, the expanded uncertainties by MIP-OES were 1.2%, 2.2%, and 2.5% and the uncertainties by ISE were 12.2%, 6.1%, and 3.1%, respectively. Similarly, in the case of estimating the uncertainty of type B, the MIP-OES method compared to ISE was characterized by almost two times lower expanded uncertainty; that is, for MIP-OES, it was 2.53%, and for ISE, it was 4.75%. Both methods of uncertainty estimation, types A and B, can be used interchangeably, as they provide comparable results.
1. Introduction
Estimating the postmortem interval (PMI, also called “time of death” [TOD] or “time since death” [TSD]) is an important element in prosecution proceedings (e.g., in the case of searching for a person suspected of murder). Witness testimonies are often associated with a high risk of inaccuracy, manipulation, or even forgery. Witnesses may also have prejudices against the suspect. However, PMI determination can verify the course of events, including limiting the number of suspects or verifying their alibi [1,2].
There are many methods for estimating the PMI. Nowadays, more and more often attempts are made to use microRNAs as useful tool to estimate TSD [3]. The frequently used ones include temperature measurement (which is usually performed in the anus), evaluation of rigor mortis, and assessment of livor mortis. Among these, the temperature measurement method has the widest scope of application. Unfortunately, the time window for the PMI in which it can be used is 36 h [1]. Methods with longer time windows for the PMI include biochemical processes in which the determination of the TOD is based on the correlation of the concentration of substances (e.g., potassium ions) in biological material (most often in the vitreous humor [VH]). The method of assessing potassium concentration in the VH enables us to determine the TOD within 120 h [4]. The VH is a valuable postmortem material because it is composed primarily of water and the autolysis process takes place much slower in it than in peripheral blood. However, this type of material also has disadvantages, such as low volume (1–2 mL) and relatively high viscosity. The analysis of the concentration of potassium ions concerning the PMI dates back to the 1960s [5,6,7]. Since then, researchers have proposed more than a dozen formulas that can be used to estimate the PMI (the uncertainty of this time estimate was difficult to determine) (Table 1).

Table 1.
Formulas for estimating the PMI based on the concentration of potassium ions ([K+] in mmol/L).
The most frequently used method to measure the concentration of potassium ions in the VH is direct potentiometry in the form of an ion-selective electrode (ISE) and, less frequently, the method of flame photometry, for example, in the form of microwave-induced plasma with optical emission spectrometry (MIP-OES) (Table 2).

Table 2.
Methods applied to determine the postmortem concentration of potassium ions in the VH.
To the best of the authors’ knowledge, both ISE and MIP-OES methods have not yet been characterized in terms of measurement uncertainty of types A and B, which directly affect the estimation of PMI uncertainty. Measurement uncertainty is the numerically expressed dispersion of the measured quantity, in which it is expected, with a certain probability, to find the true measurement value. Measurement uncertainty can be determined using two methodologies: type A and type B. The type A method most often uses a series of measurements and determines the variation of the results (e.g., standard deviation), considering the coverage factor equal to 2 and the corresponding confidence interval. The type B method uses data contained in the specifications of measuring devices, safety data sheets for reagents, laboratory equipment, and previous tests described in publications and test reports. The feature that distinguishes the type B from the type A method is the lack of necessity to conduct experimental tests to estimate the uncertainty.
This study aimed to estimate the uncertainty of potassium ion determination using ISE and MIP-OES methods and to examine the impact of the estimated measurement uncertainty on the analysis of potassium ions in the VH and, consequently, on the PMI estimation.
2. Materials and Methods
2.1. Sample Collection
During the medicolegal autopsy in the Department of Forensic Medicine of the Medical University of Gdańsk in 2021–2022, VH samples were initially collected from 42 deceased individuals. The deceased with head injuries (risk of VH contamination by peripheral blood) and those who died more than 120 h before dissection were excluded from the study. After sampling, the material was transferred to 2 mL microcentrifuge tubes (Eppendorf-type vials) and clearly labeled. The collected VH samples were centrifuged shortly after dissection for 10 min at 3500 rpm and transferred to 2 mL Eppendorf vials, which were then stored at −20 °C until analysis.
Table 3 summarizes the data of the deceased: gender, age of death, blood alcohol content and other biological materials, cause of death, TOD or disclosure of the body, and time of postmortem examination.

Table 3.
Selected data of the deceased individuals autopsied in 2021–2022.
The results of the deceased who died due to hanging, who may also rupture the blood vessels of the eyes, should be interpreted with great care. Samples from 26 deceased individuals were taken for further analysis.
From the TOD to the autopsy examination, the time usually did not exceed a few days. During the time preceding the autopsy, the bodies were stored at +4 °C. In the case of analytical methods, the time from the moment of collecting the vitreous humor during autopsy to the moment of taking measurements is important. The storage time of the vitreous humor samples at −20 °C is also essential, as we assume no significant changes in the chemical composition during this time, which is not necessarily accurate.
2.2. VH Preparation
The potassium ion determination procedure was developed based on the research results of Mihailovic et al. [15] and Yang et al. [27] (Table 2). Below, we present the methodology optimized to our conditions. Samples with the centrifuged VH (supernatant) were left at room temperature for about 1 h after removal from the −20 °C freezer. Each sample was vortexed for 15 s. Then, 50 μL of the supernatant was collected twice into two 5 mL Eppendorf vials and refilled (completely) with deionized water. As a result, an 80-fold dilution was achieved. Each sample was vortexed for 15 s. Depending on the method, the remaining steps of the procedure differed from each other and are described in Table 4.

Table 4.
Procedure for determining potassium ion concentration in VH by ISE and MIP-OES methods.
2.3. Instrumentation
Measurements were carried out using two methods: direct potentiometry (ISE) and plasma-excited optical emission spectrometry (MIP-OES). Measurements by direct potentiometry were conducted using a combination of ISE (HI 4114, Hanna Instruments) and a pH meter (S220, Mettler Toledo SevenCompact). When performing measurements using the MIP-OES method with plasma excitation, the Agilent 4210 MP-AES (the older and trade name derived from “microwave plasma–atomic emission spectroscopy”) device was applied, and mineralization was performed with the Multiwave GO Plus by Anton Paar.
3. Results
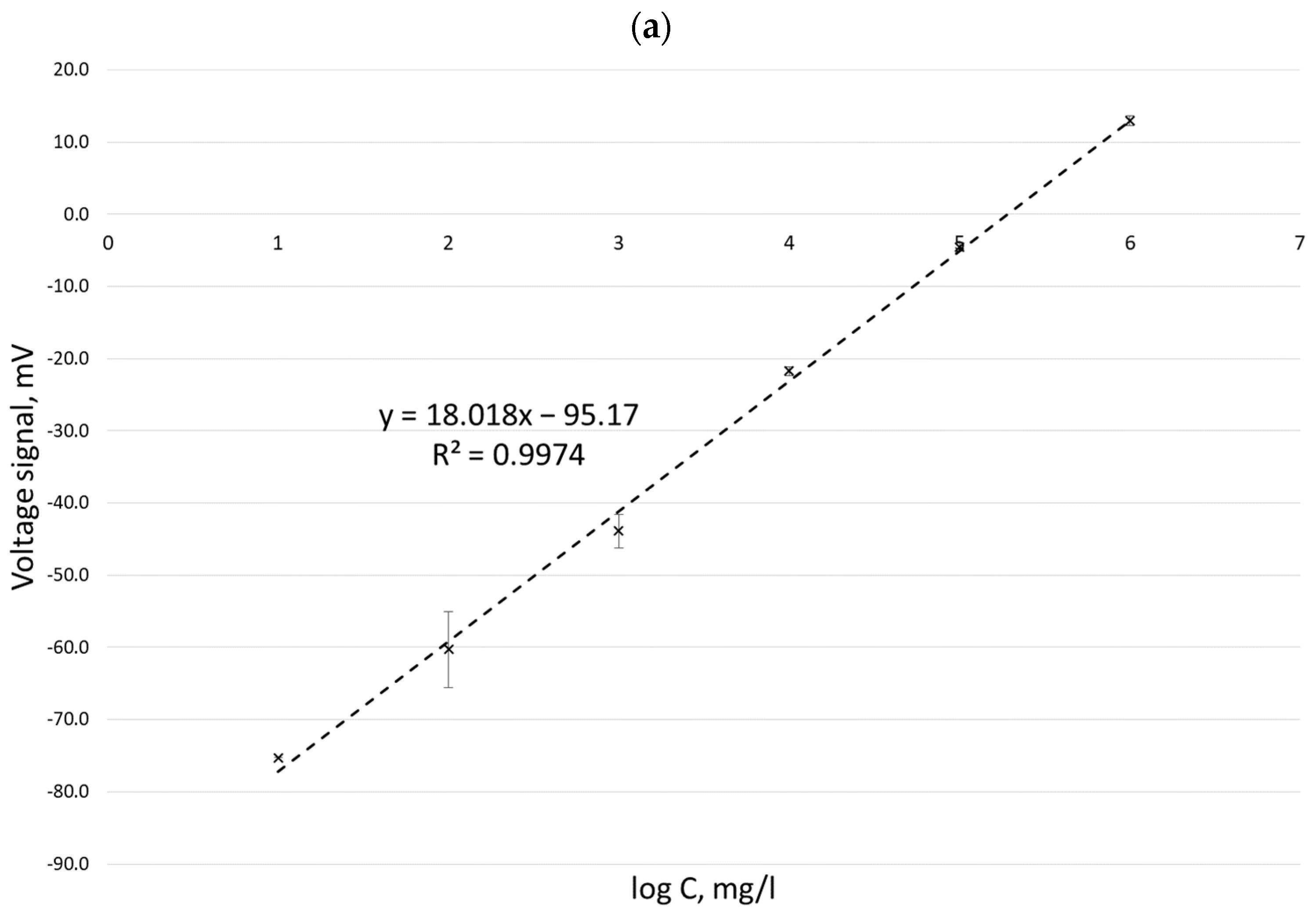
Calibration curves obtained for potassium ion concentrations ranging from 2 to 100 mg/L by the ISE method in three measurement series (performed on three different days) carried out with six replications for each concentration are presented in Figure 1.
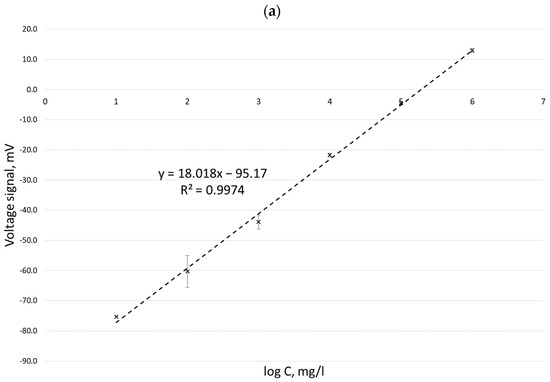
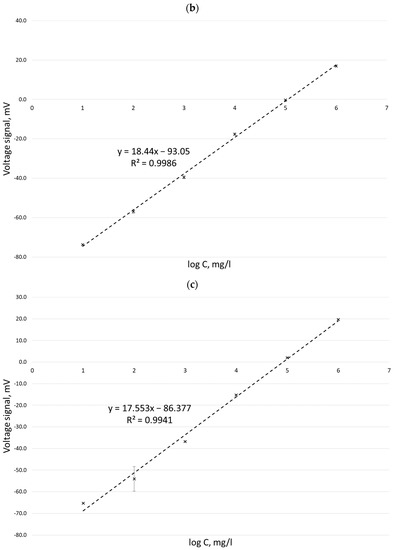
Figure 1.
Calibration curves for the determination of potassium ion concentration by the ISE method in three different measurement series: 10 March 2022 (a), 11 March 2022 (b), and 14 March 2022 (c).
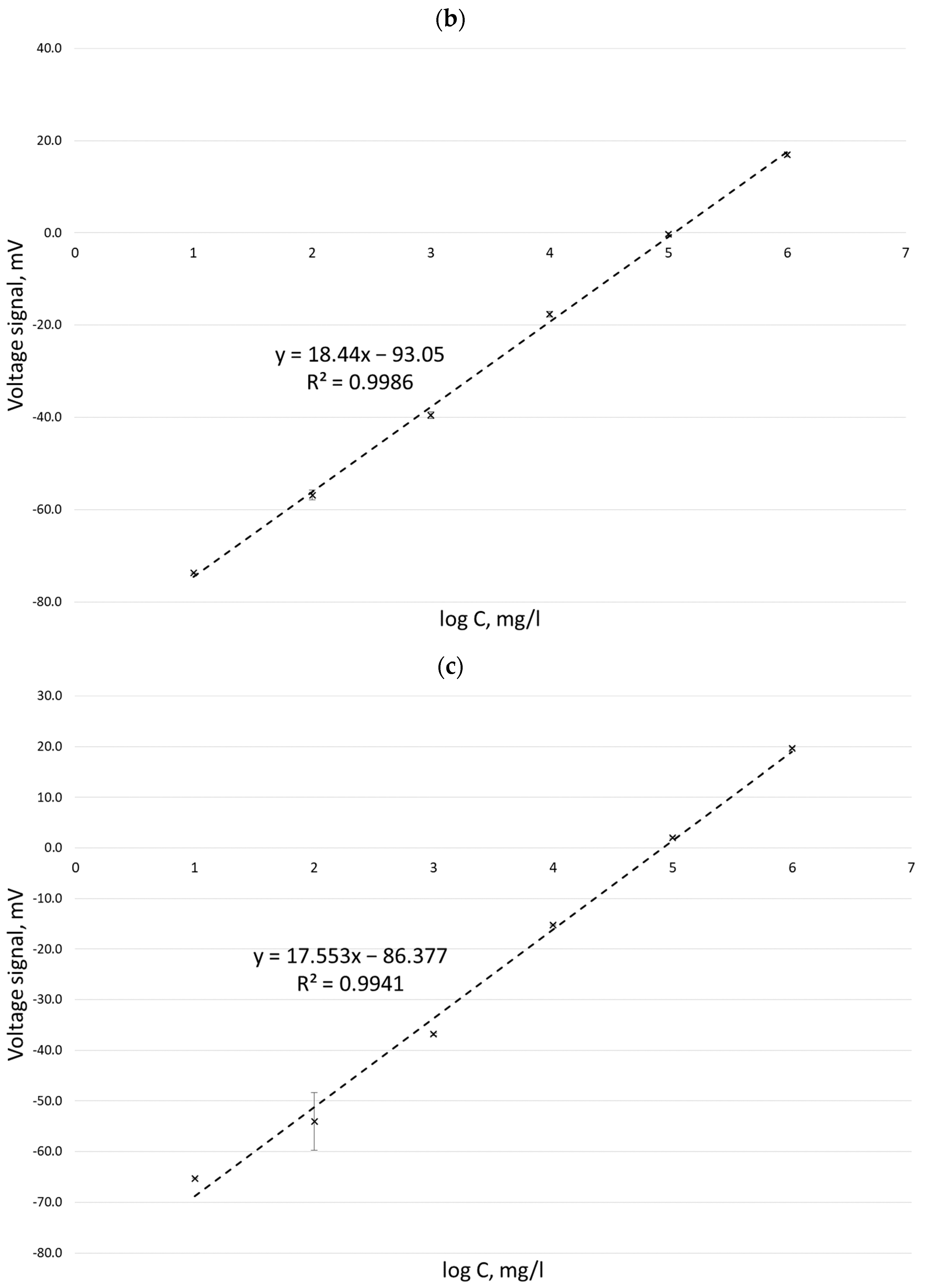
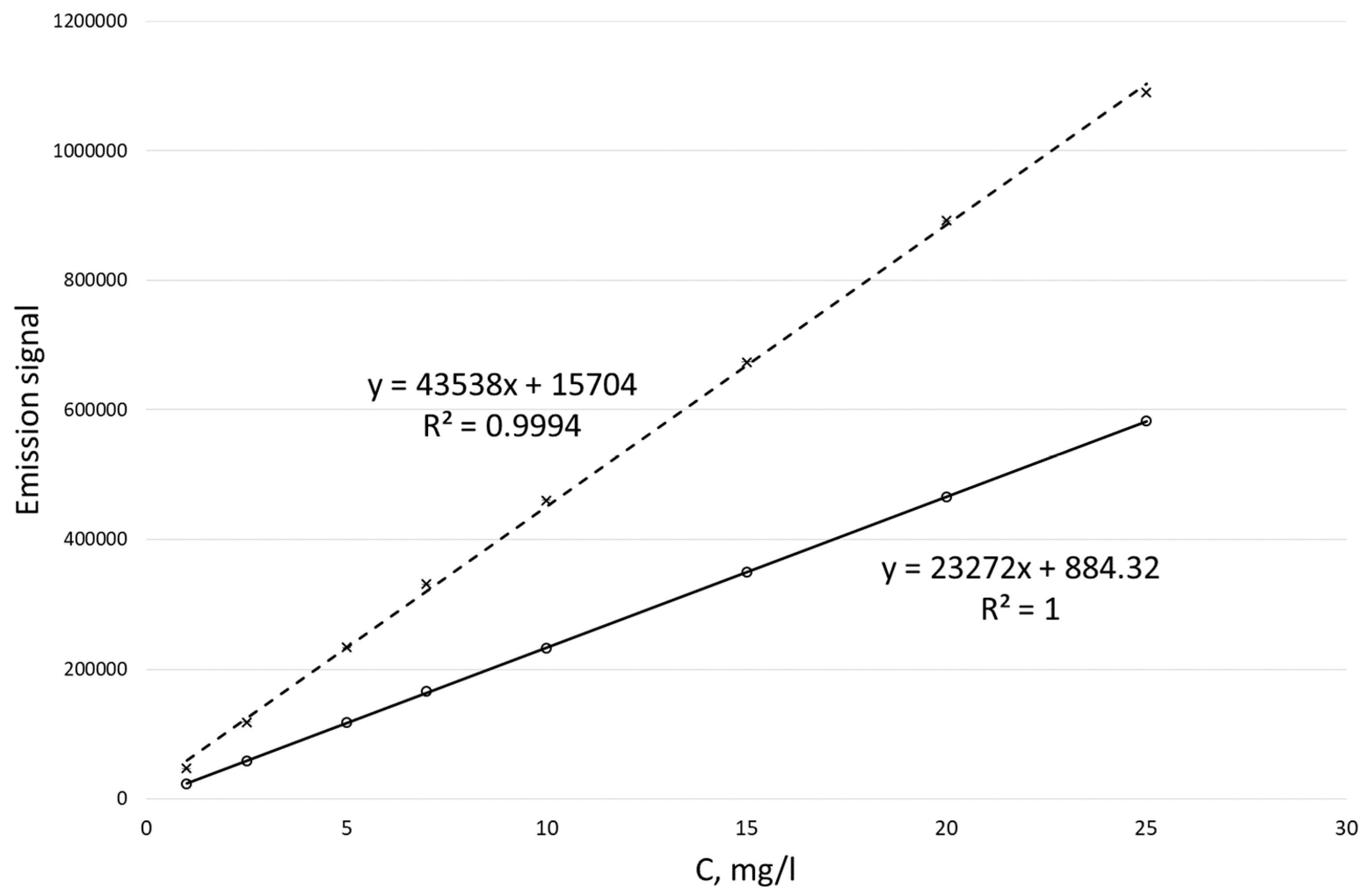
Calibration curves obtained for potassium ion concentrations ranging from 1 to 25 mg/L by the MIP-OES method for two different wavelengths are shown in Figure 2.
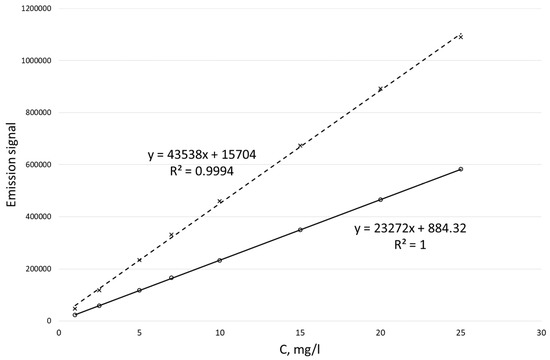
Figure 2.
Calibration curves for the determination of potassium ion concentration by the MIP-OES method for two wavelengths at 766.49 nm (dotted line) and 769.89 nm (solid line).
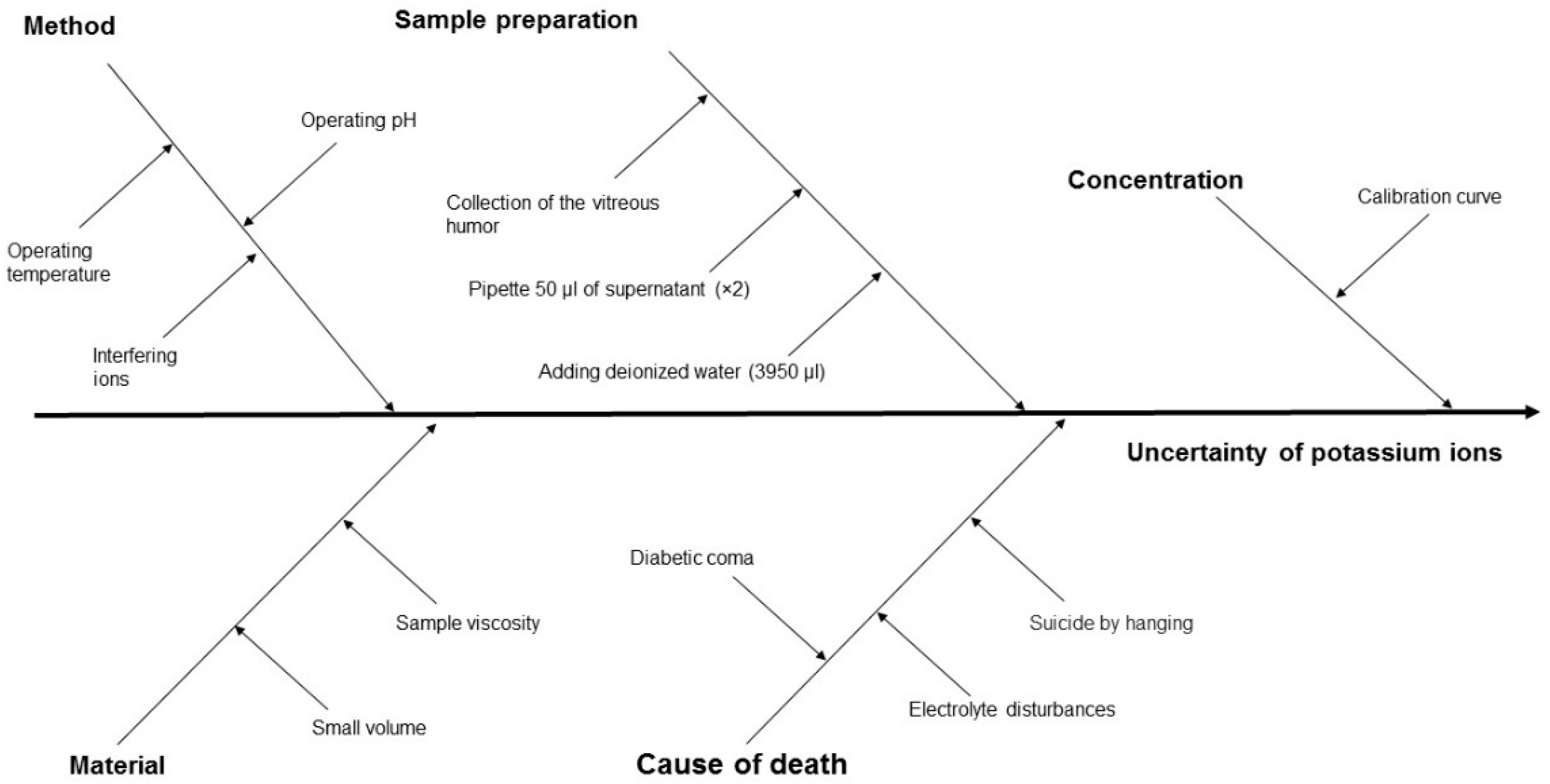
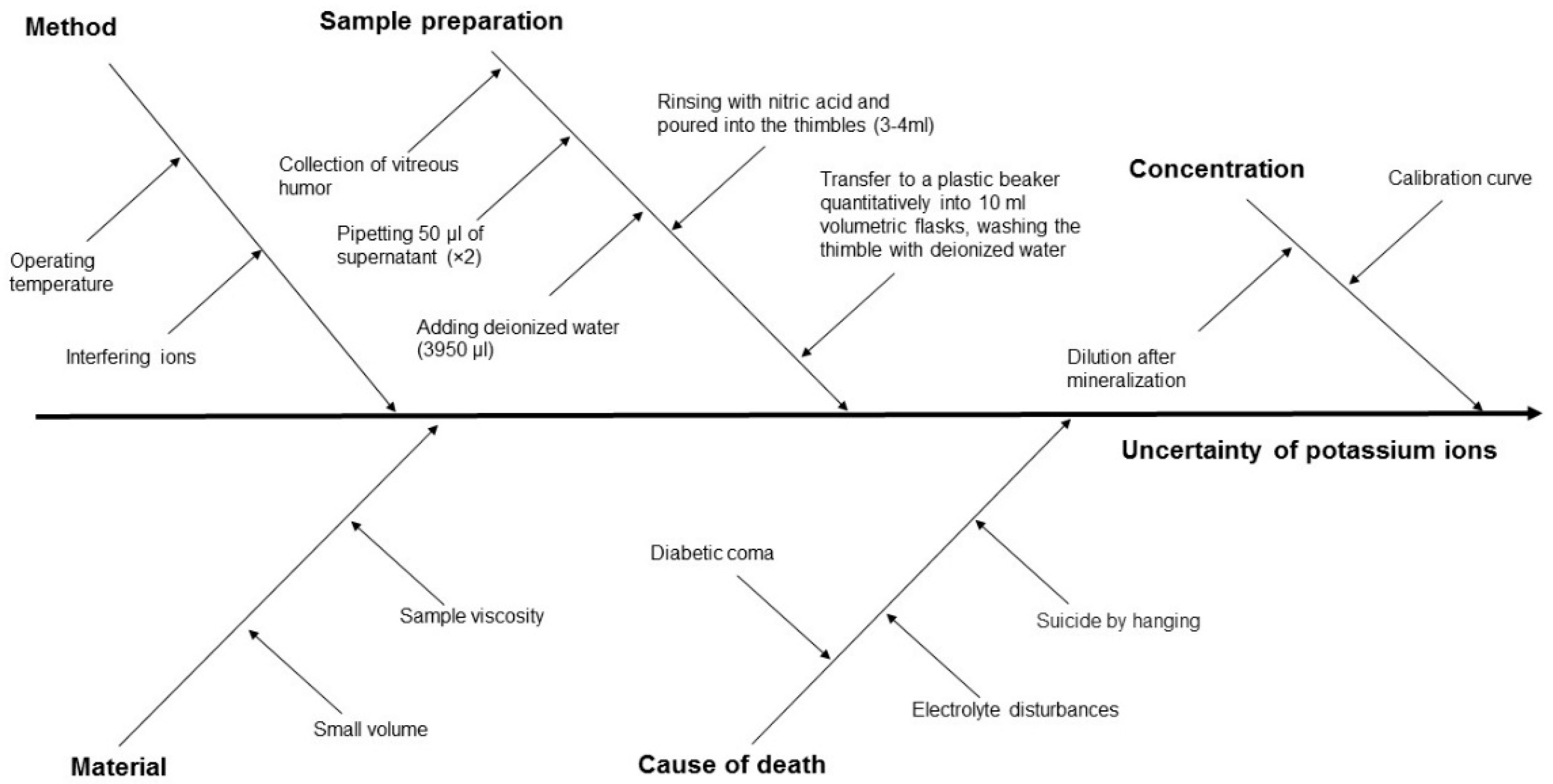
The measurement uncertainty, and hence the reliability of PMI estimation, is influenced by several factors, including the properties of the biological material, the measurement method applied, the process of sample preparation, the sample concentration, and the cause of death (Figure 3 and Figure 4).
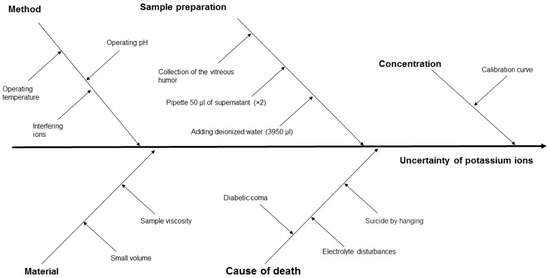
Figure 3.
Cause-and-effect diagram for ion potassium concentration in VH using the ISE method, showing the key components that contribute to measurement uncertainty.
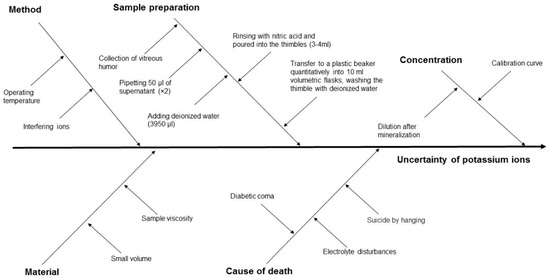
Figure 4.
Cause-and-effect diagram for ion potassium concentration in VH using the MIP-OES method, showing the key components that contribute to measurement uncertainty.
To obtain the A-type uncertainty, water samples with low (2 mg/L), medium (10 mg/L), and high (25 mg/L) potassium ion concentrations were tested using the ISE and MIP-OES methods, and the results with the estimated uncertainty are presented in Table 5.

Table 5.
Type A uncertainty estimation for the determination of potassium ion concentrations of 2 mg/L (low), 10 mg/L (medium), and 25 mg/L (high) using the ISE and MIP-OES methods.
The components of the uncertainty budget of ISE and MIP-OES methods to estimate type B uncertainty are presented in Table 6 and Table 7, respectively.

Table 6.
Components of the uncertainty budget of the ISE method [28,29].

Table 7.
Components of the uncertainty budget of the MIP-OES method [28,29].
Using the components of the uncertainty budget listed in Table 6 and Table 7, the expanded uncertainty using the ISE and MIP-OES methods was estimated (Table 8).

Table 8.
Results of the B-type expanded uncertainty estimation for the determination of potassium ion concentration using ISE and MIP-OES methods.
Table 9 presents 26 cases for which the PMI was determined (based on the data from Table 3), and the concentration of potassium ions was determined using the ISE and MIP-OES methods. The concentration units were converted from mg/L to mmol/L.

Table 9.
Results of the PMI assessment (based on the data from Table 3) and the determination of potassium ion concentration in the VH using ISE and MIP-OES methods in 26 deceased individuals.
4. Discussion
4.1. Properties of Biological Material
VH is characterized by a much lower rate of decomposition changes (or increased resistance to autolysis) than blood. However, the described type of material has limitations related to its relatively high viscosity and small volume (up to 2 mL). Because of the above aspects, the samples were diluted to reduce the matrix effect. The first factor influencing measurement uncertainty is interfering ions, which include NH4+, Li+, Na+, and Ca2+. To reduce the interference, the samples were diluted 80-fold.
It is important to keep the temperature constant during the entire test (in the range of 20–25 °C) because, depending on the temperature, the measurement results may fluctuate. According to the manufacturer, the temperature range of the ISE operation is equal to 0–40 °C. Maintaining the pH of the measuring environment also influences the measurement, and the environment is stable in the range of 1.5–12.00 pH.
VH samples were collected using a metal, pointed (35 × 2 mm) injection needle. The time the samples were collected varied according to the duration of the autopsy. The bodies of the deceased were stored in autopsy refrigerators (4 °C) at various times.
4.2. Measurement Uncertainty by ISE and MIP-OES
In this study, an attempt was made to estimate the uncertainty components (A and B types) related to sample preparation and instrumental analysis using ISE and MIP-OES (Table 10).

Table 10.
Summary of the uncertainty budget results of types A (experimental) and B (nonexperimental) for determining potassium ion concentration using ISE and MIP-OES methods.
The estimated values of the expanded uncertainty determined by the experimental method A (UA) indicate a few to even ten-times lower uncertainty in the determination of potassium ion concentration in the case of the MIP-OES method. Similarly, in the case of estimating the expanded uncertainty with the B (UB) method, the MIP-OES method is characterized by twice the lower uncertainty. Based on our experience in determining the concentration of potassium ions using the ISE and MIP-OES methods, it is worth considering their advantages and disadvantages before implementing them into diagnostic practice (Table 11).

Table 11.
Advantages and disadvantages of ion-selective electrode (ISE) and microwave-induced plasma with optical emission spectrometry (MIP-OES) methods.
4.3. Causes of Death
The causes of death that affect the postmortem concentration of potassium ions in VH are hanging, diabetic coma, and electrolyte disturbances. The increased concentration of potassium ions in hanging cases is due to the tightness of the loops in the neck, which causes a rupture of the jugular system and a significant increase in venous pressure. The cause of death strongly influences the change in the concentration of potassium ions after death, which is mainly related to the possibility of blood contamination of the VH. Venous circulation can lead to capillary hyperemia and thus increase vascular leakage from the capillaries into the retina [23,30]. In addition, if metabolic acidosis occurs, hyperkalemia is expected, which may make it impossible to reliably estimate the PMI from the concentration of potassium ions.
Differences were observed in lethal diabetic coma. It is known that, in a diabetic coma, there is a potassium liberation from the cell to the outside due to insulin deficiency and acidosis [21,31].
Any sudden mechanical movement during or after death can introduce the breaking of blood vessels in the eyes and changes in the concentration of potassium ions. The uncertainty of determination related strictly to the ISE and MIP-OES methods is relatively small concerning the other factors influencing the concentration of potassium ions (cause of death, ambient temperature, and cadaver storage conditions). However, based on our studies, the uncertainty of type A (experimental) for low concentrations (2 mg/L) of potassium ions can be as high as 12.2% using the ISE method. Reducing the uncertainty of PMI estimation can be performed by introducing additional indicators that determine the TOD zystkie surowe dane mogą być udostępnione na prośbę skierowaną do autora korespondencyjnego. (muscle degradation products by means of quantitative MRI).
5. Conclusions
The postmortem concentration of potassium ions in the VH has a limited PMI time range, which usually does not exceed four days, and may be influenced by interfering factors, including analytical factors (e.g., sample viscosity, the necessity of dilution, failures, and nonselectivity of the ISE) and nonanalytical factors (e.g., age, cause of death, and ambient temperature).
To determine the concentration of potassium ions in the VH, the analytical uncertainty of types A and B should be taken into account, as it generally has a significant effect on the estimation of PMI. Both A and B estimates give similar results, which may prove the usefulness of both methods in estimating the measurement uncertainty of the concentration of potassium ions, and thus PMI estimation.
The cause of death also influences the postmortem concentration of potassium ions in the VH. Caution should be exercised if death occurs due to hanging or electrolyte disturbances, as they may significantly affect the measurement results and thus the estimated TOD.
Author Contributions
Conceptualization, S.Z., M.W. and B.M.C.; methodology, S.Z., M.W., B.M.C. and M.K.; validation, S.Z., B.M.C. and M.W.; investigation, S.Z., M.W. and B.M.C.; resources, S.Z. and M.W.; writing—original draft preparation, S.Z., M.W., B.M.C., J.S.A. and M.K.; writing—review and editing, S.Z., M.W., B.M.C., J.S.A. and M.K.; supervision, M.W. and J.S.A. All authors have read and agreed to the published version of the manuscript.
Funding
The publication of the article was supported by the project POWR.03.02.00-00-I014/17-00 co-financed by the European Union through the European Social Fund under the Operational Programme Knowledge Education Development 2014–2020.
Informed Consent Statement
Patient consent was waived due to the consent of the bioethics committee of the Medical University of Gdańsk, dated 28 February 2020 (NKBBN/18/2020).
Data Availability Statement
All research data can be made available upon request send to the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Henßge, C.; Madea, B. Estimation of the time since death in the early post-mortem period. Forensic Sci. Int. 2004, 144, 167–175. [Google Scholar] [CrossRef]
- Mathur, A.; Agrawal, Y.K. An overview of methods used for estimation of time since death. Aust. J. Forensic Sci. 2011, 43, 275–285. [Google Scholar] [CrossRef]
- Maiese, A.; Scatena, A.; Costantino, A.; Di Paolo, M.; La Russa, R.; Turillazzi, E.; Frati, P.; Fineschi, V. MicroRNAs as useful tools to estimate time since death. A systematic review of current literature. Diagnostics 2021, 11, 64. [Google Scholar] [CrossRef] [PubMed]
- Madea, B. Methods for determining time of death. Forensic Sci. Med. Pathol. 2016, 12, 451–485. [Google Scholar] [CrossRef] [PubMed]
- Markowska, J.; Szopa, M.; Zawadzki, M.; Piekoszewski, W. Ciało szkliste oka—Rutynowy czy alternatywny materiał do badań w medycynie sądowej. Arch. Med. Sadowej Kryminol. 2017, 67, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Madea, B. Is there recent progress in the estimation of the postmortem interval by means of thanatochemistry? Forensic Sci. Int. 2005, 151, 139–149. [Google Scholar] [CrossRef]
- McCleskey, B.C.; Dye, D.W.; Davis, G.G. Review of postmortem interval estimation using vitreous humor: Past, present, and future. Acad. Forensic Pathol. 2016, 6, 12–18. [Google Scholar] [CrossRef]
- Adelson, L.; Sunshine, I.; Rushforth, N.B.; Mankoff, M. Vitreous potassium concentration as an indicator of the postmortem interval. J. Forensic Sci. 1963, 8, 503–514. [Google Scholar]
- Coe, J.I. Vitreous potassium as a measure of the postmortem interval: An historical review and critical evaluation. Forensic Sci. Int. 1989, 42, 201–213. [Google Scholar] [CrossRef]
- Ross, J.; Hoadley, P.; Sampson, B. Determination of postmortem interval by sampling vitreous humour. Am. J. Forensic Med. Pathol. 1997, 18, 158–162. [Google Scholar]
- Barús, J.I.M.; Suárez-Peñaranda, J.M.; Otero, X.L.; Rodríguez-Calvo, M.S.; Costas, E.; Miguéns, X.; Concheiro, L. Improved estimation of postmortem interval based on differential behaviour of vitreous potassium and hypoxantine in death by hanging. Forensic Sci. Int. 2002, 125, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Zhang, L.; Zhang, G.; Zhang, X.; Jiang, X. The determination of potassium concentration in vitreous humor by low pressure ion chromatography and its application in the estimation of postmortem interval. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2007, 852, 278–281. [Google Scholar] [CrossRef] [PubMed]
- Jashnani, K.D.; Kale, S.A.; Rupani, A.B. Vitreous humor: Biochemical constituents in estimation of postmortem interval. J. Forensic Sci. 2010, 55, 1523–1527. [Google Scholar] [CrossRef] [PubMed]
- Bortolotti, F.; Pascali, J.P.; Davis, G.G.; Smith, F.P.; Brissie, R.M.; Tagliaro, F. Study of vitreous potassium correlation with time since death in the postmortem range from 2 to 110 hours using capillary ion analysis. Med. Sci. Law 2011, 51 (Suppl. S1), 20–23. [Google Scholar] [CrossRef] [PubMed]
- Mihailovic, Z.; Atanasijevic, T.; Popovic, V.; Milosevic, M.B.; Sperhake, J.P. Estimation of the postmortem interval by analyzing potassium in the vitreous humor: Could repetitive sampling enhance accuracy? Am. J. Forensic Med. Pathol. 2012, 33, 400–403. [Google Scholar] [CrossRef]
- Siddhamsetty, A.K.; Verma, S.K.; Kohli, A.; Verma, A.; Puri, D.; Singh, A. Exploring time of death from potassium, sodium, chloride, glucose & calcium analysis of postmortem synovial fluid in semi arid climate. J. Forensic Leg. Med. 2014, 28, 11–14. [Google Scholar] [CrossRef] [PubMed]
- Bohra, B.; Verma, R.; Mathur, I.B.; Sharma, Y.; Khangwal, V.P. Estimation of postmortem interval by measuring potassium level in vitreous humor. J. Indian Acad. Forensic Med. 2014, 36, 374–378. [Google Scholar]
- Foster, S.N.; Smith, P.R.; Biggs, M.; Rutty, G.N.; Hollingbury, F.E.; Morley, S.R. Estimation of postmortem interval using vitreous potassium levels in cases of fatal road traffic collision. Arch. Med. Sadowej Kryminol. 2016, 66, 71–82. [Google Scholar] [CrossRef]
- Murthy, A.S.; Das, S.; Thazhath, H.K.; Chaudhari, V.A.; Adole, P.S. The effect of cold chamber temperature on the cadaver’s electrolyte changes in vitreous humor and plasma. J. Forensic Leg. Med. 2019, 62, 87–91. [Google Scholar] [CrossRef]
- Focardi, M.; Lanzilao, L.; Bonari, A.; Lazzeretti, M.; Lorubbio, M.; Ognibene, A.; Gualco, B.; Fanelli, A.; Pinchi, V. Improvement in estimation of time since death by albumin and potassium concentrations in vitreous humor. Forensic Sci. Int. 2020, 314, 110393. [Google Scholar] [CrossRef] [PubMed]
- Zilg, B.; Bernard, S.; Alkass, K.; Berg, S.; Druid, H. A new model for the estimation of time of death from vitreous potassium levels corrected for age and temperature. Forensic Sci. Int. 2015, 254, 158–166. [Google Scholar] [CrossRef]
- Rognum, T.O.; Hauge, S.; Øyasaeter, S.; Saugstad, O.D. A new biochemical method for estimation of postmortem time. Forensic Sci. Int. 1991, 51, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Chandrakanth, H.V.; Kanchan, T.; Balaraj, B.M.; Virupaksha, H.S.; Chandrashekar, T.N. Postmortem vitreous chemistry—An evaluation of sodium, potassium and chloride levels in estimation of time since death (during the first 36 h after death). J. Forensic Leg. Med. 2013, 20, 211–216. [Google Scholar] [CrossRef]
- Cordeiro, C.; Ordóñez-Mayán, L.; Lendoiro, E.; Febrero-Bande, M.; Vieira, D.N.; Muñoz-Barús, J.I. A reliable method for estimating the postmortem interval from the biochemistry of the vitreous humor, temperature and body weight. Forensic Sci. Int. 2019, 295, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Hansson, L.; Uotila, U.; Lindfors, R. Potassium content of the vitreous body as an aid in determining the time of death. J. Forensic Sci. 1966, 11, 390–394. [Google Scholar]
- Moriya, F.; Hashimoto, Y. Endogenous γ-hydroxybutyric acid levels in postmortem blood. Leg. Med. 2004, 6, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Li, H.; Yang, T.; Ding, Z.; Wu, S.; Qiu, X.; Liu, Q. A study on the estimation of postmortem interval based on environmental temperature and concentrations of substance in vitreous humor. J. Forensic Sci. 2018, 63, 745–751. [Google Scholar] [CrossRef]
- Stephen, R.J. Vitreous humor chemistry. The use of potassium concentration for the prediction of postmortem interval. J. Forensic Sci. 1987, 32, 503–509. [Google Scholar] [CrossRef]
- Eppendorf.com. Eppendorf Research Instruction Manual. 2016. Available online: http://www.sun-way.com.tw/Files/DownloadFile/Researchplus.pdf (accessed on 28 February 2016).
- Hanna Instruments. Instruction Manual Potassium Ion Selective Electrode HI 4114, MAN4114 07/06 REV3. 2007. Available online: https://www.hannainst.com/hi4114-potassium-combination-ion-selective-electrode.html (accessed on 23 November 2022).
- Kitabchi, A.E. Hyperglycemic crises in diabetes. Diabetes Care 2004, 27 (Suppl. S1), s94–s102. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).