Abstract
Utilization of bioenergy with carbon capture can realize carbon-negative syngas production. The quadruple fluidized bed gasifier (QFBG) integrates a chemical looping oxygen generation process and a dual fluidized bed gasifier with limestone as bed material. It is one promising device that can convert biomass to H2-rich syngas whilst capturing CO2 with little energy penalty. However, experimental or numerical simulation of QFBG is rarely reported on due to its complex structure, hindering the further commercialization and deployment of QFBG. In this work, a new computational fluid dynamics (CFD) solver is proposed to predict the complex physicochemical processes in QFBG based on the multi-phase particle in cell (MPPIC) methodology with the assistance of the open source software, OpenFOAM. The solver is first validated against experimental data in terms of hydrodynamics and reaction kinetics. Then, the solver is used to investigate the QFBG property. It is found that the QFBG can operate stably. The cold gas efficiency, H2 molar fraction, and CO2 capture rate of the QFBG are predicted to be 87.2%, 93.3%, and 90.5%, respectively, which is promising. It is believed that the solver can give reliable predictions for similar fluidized bed reactors.
1. Introduction
The whole world is fighting against global warming so as to keep the atmosphere’s CO2 concentration well below the critical value of 450 ppm [1]. To meet the requirement of the Paris Agreement [2], it has been reported that carbon-negative power generation technologies should be developed and deployed [3]. Bioenergy with carbon capture provides such a transformative route, not only because biomass is carbon-neutral during the thermal conversion process, but also because biomass is the fourth largest fuel source following coal, oil, and gas on the earth [4]. However, low calorific value is one essential drawback that hinders the direct utilization of biomass [5]. Converting biomass into high-quality syngas via gasification can well solve this issue. The syngas from biomass can then be used for power generation or for utilization in the steel industry [6,7]. Because biomass is hard to grind into fine powder but relatively easy to compress into pellets, a fluidized bed is then the proper reactor for biomass gasification. A dual fluidized bed gasifier (DFBG), which consists of a bubbling fluidized bed gasifier and a fast fluidized bed combustor, can generate high-quality syngas by avoiding direct contact between air and syngas [8]. However, DFBG cannot capture CO2 in situ, so carbon-negative gasification cannot be realized. Based on DFBG, a quadruple fluidized bed gasifier (QFBG) is conceptually designed by integrating the chemical looping oxygen generation technology and DFBG [9]. The chemical looping oxygen generation process is also realized in a dual fluidized bed with Mn2O3 and Mn3O4 as bed material. Thereby, there are four fluidized bed reactors including a bubbling fluidized bed gasifier for biomass gasification and CaO carbonation, a fast fluidized bed oxyfuel combustor for residual char combustion and CaCO3 calcination, a bubbling fluidized bed oxidation reactor for Mn3O4 oxidation, and a fast fluidized bed reduction for Mn2O3 reduction. With the four integrated fluidized bed reactors, biomass can be converted into H2-rich syngas. Since the bed materials are circulated inside the reactors, the heat absorbed and the heat released can be theoretically balanced. However, due to the formidable complexity of QFBG, related experiments or numerical simulations are seldom reported.
Liu [10] did the thermodynamic modeling of biomass sorption-enhanced chemical looping gasification, and the energy efficiency of this technology was found to be 64.6%. Pröll [11] studied the H2-rich syngas production by selective CO2 removal during biomass gasification in a 100 kW DFGB using the thermodynamic equilibrium approach and found that H2 volume fraction in the dry syngas could reach 65–75%, and CO2 volume fraction was in the range of 6–13%. Koppatz [12] experimentally studied the calcium-enhanced biomass gasification process in an 8-MW DFBG and found that the H2 volume fraction in the syngas was about 50%, and the CO2 volume fraction was about 12.3%. The low H2 fraction was probably caused by the low temperature (about 650 °C), and high steam partial pressure. Hejazi [13] once modeled biomass steam gasification in a DFBG with lime-based CO2 capture using the stoichiometric equilibrium method. It was found that the CO2 capture rate could be over 70%. Cormos [14] studied the chemical looping air separation cycle for decarbonized power generation based on oxyfuel combustion in terms of energy and cost. It was found that the manganese looping cycle could be more efficient than the cryogenic process, and could improve the net efficiency by 2–3.5 percentage points. The CO2 capture penalty was reduced to 5–7 net energy efficiency points. Mei [15] studied the reactivity and lifetime of an oxygen releasable manganese ore and found that repeatable O2 gas release was available, and the reactivity and lifetime of Mn ore were better than the often-used ilmenite. Yan [9] studied the biomass/coal co-gasification properties in a DFBG reactor using a one-dimensional model with the assistance of the commercial software, Aspen Plus. The cold gas efficiency was up to 78.9% under the proposed optimum condition. This model coupled the fluidized bed hydrodynamics and reaction kinetics. However, the model is one-dimensional and the research object is DFBG. Yan [16] studied the property of QFBG using the commercial software, Aspen Plus. It was found that the H2 mole fraction in the dry syngas is higher than 70%, the CO2 mole fraction in the dry flue gas is around 97%, and the net carbon discharge can be negative when the biomass blending ratio is over 0.5. The QFGB property was preliminarily studied. However the research simplified QFGB as one-dimensional, so the predictions cannot reflect the practical situation. Yan [17] studied the biomass steam gasification process in a DFBG reactor based on the granular kinetic theory (GKT) with the assistance of the commercial software, Fluent. The H2 mole fraction was predicted to be 46.62%, and the cold gas efficiency was 82.9%. Although this is a three-dimensional simulation, the object is DFBG, which is quite simple compared with QFBG. Moreover, GKT treated the bed material and solid fuel as a fluid phase, which requires that the particle dimension should be uniform rather than dispersed. Yan [18] also studied the property of the DFBG reactor based on the multiphase particle in cell (MPPIC) method with the assistance of the open-source software, OpenFOAM. The model predictions were compared with experimental data, and the operating characteristics of the DFBG were predicted. The MPPIC method considered all particles as computation parcels and track each parcel in Lagrangian coordinates, so it is more advanced than GKT for fluidized bed simulation. However, the research object was still DFBG rather than QFBG. Pissot [19] compared four DFBG configurations including heat supply by air combustion, oxyfuel combustion, chemical looping gasification, and electrical thermal, and found that the oxyfuel and the chemical looping gasification scheme exhibited the lowest energy demand for CO2 separation. This is also the reason why QFBG integrates chemical looping technology. Wang [20] predicted hydrogen production via chemical looping reforming in a DFBG reactor based on the granular kinetic theory. A bubble-structure-dependent drag coefficient model was proposed and the model predictions were validated against experimental data.
From a literature review, it can be concluded that calcium-enhanced biomass gasification can increase the H2 fraction and decrease the CO2 fraction in dry syngas. However, the stripped carbon cannot be captured if air is used as an oxidizer for the calcination process. To gasify biomass whilst capturing CO2, oxyfuel combustion should be introduced. According to literature reports, the more efficient manganese-based chemical looping air separation approach is chosen and is coupled with the calcium-based DFBG to form the QFBG in this work. Although the QFBG property has been studied with the Aspen Plus platform, the three-dimensional simulation of QFBG has not been carried out. This work aims to further study the physicochemical processes of QFBG with the computational fluid dynamics approach so that the QFBG characteristics can be known better. The processes in QFBG include the fluidization of different particles like limestone, manganese ore, and biomass, heterogeneous reactions like biomass pyrolysis and char gasification, homogeneous reactions, and the coupling of hydrodynamics with chemical reactions. There is currently no solver that can simulate such a complex process, so a new solver based on OpenFOAM is proposed and validated in this work.
2. Materials and Methods
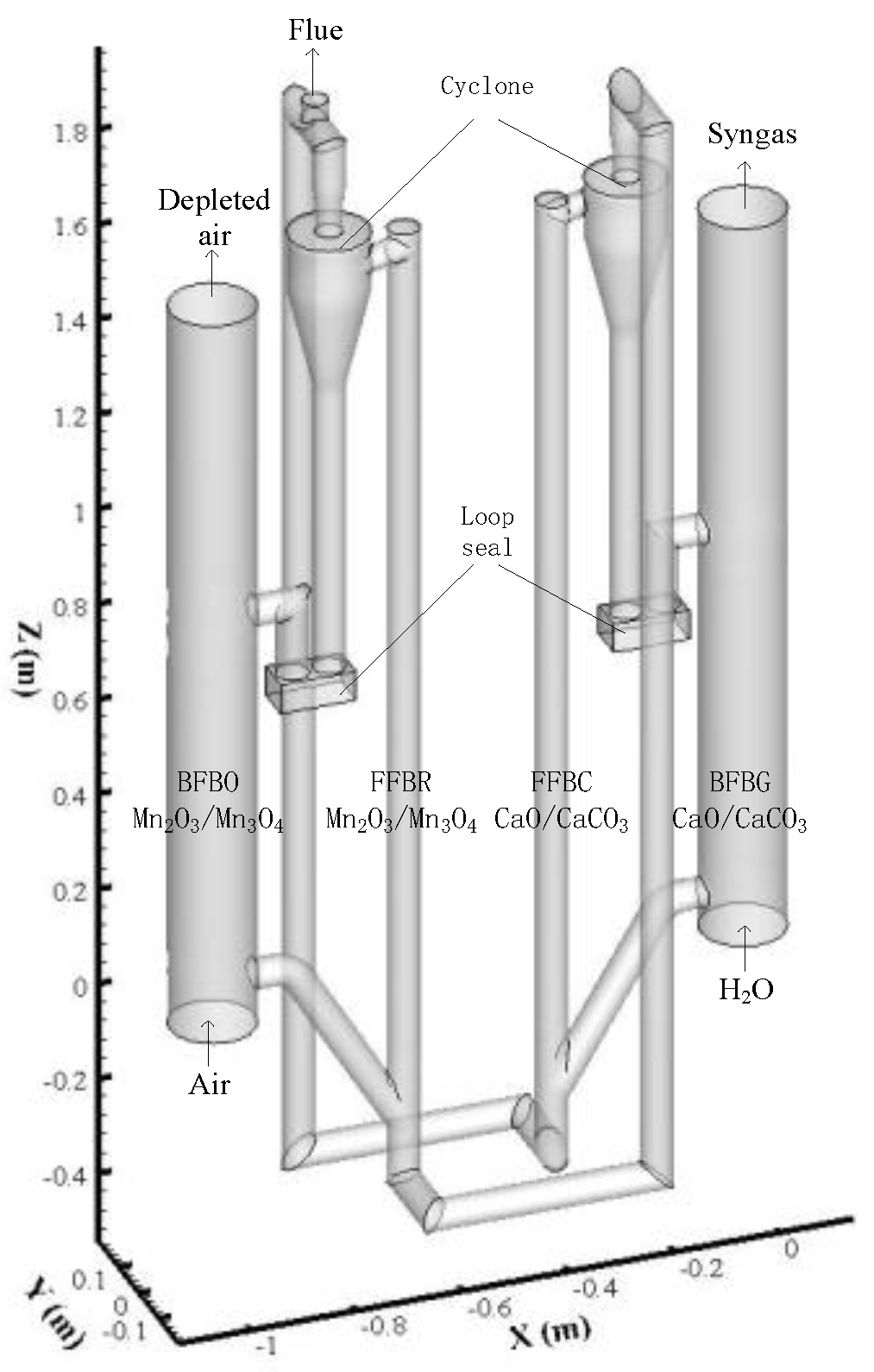
As shown in Figure 1, the QFBG reactor investigated in this work is composed of a cylinder-shaped bubbling fluidized bed gasifier (BFBG) with a height of 1.5 m and diameter of 0.16 m, a cylinder-shaped fast fluidized bed combustor (FFBC) with a height of 2.26 m and diameter of 0.06 m, a cylinder-shaped bubbling fluidized bed oxidation (BFBO) reactor with a height of 1.5 m and diameter of 0.16 m, and a cylinder-shaped fast fluidized bed reduction (FFBR) reactor with a height of 2.26 m and diameter of 0.06 m. The four fluidized beds are connected by pipes with a diameter of 0.06 m. The cyclone diameter is 0.151 m at the top and 0.06 m at the bottom. The total height of the cyclone is 0.32 m and the height of the cone section is 0.2 m. The height of the loop seal is 0.205 m. The bottoms of the bubbling fluidized bed reactors are 0.4 m higher than those of the fast fluidized bed reactors. The recirculated bed material enters the fluidized bed reactors at a height of 0.85 m from the bottom. The super-heated steam is used as the gasification agent for the gasifier. The O2/CO2 mixture gas is used as the oxidant for the oxidation reactor. The flue gas is discharged from the reactor outlet.

Figure 1.
Sketch of the quadruple fluidized bed gasifier.
To simulate the physicochemical processes in QFBG, a new solver based on the MPPIC method in OpenFOAM was compiled so that it could simultaneously consider the gas-solid flow hydrodynamics and the dominant chemical reactions. The PIMPLE algorithm [21] was chosen to solve the partial differential equation sets. The Euler scheme was chosen to implicitly discrete the transient term. The Gauss linear scheme was chosen to discrete the gradient term. The Gauss linear limited scheme was used to discrete the Laplacian term. The Gauss upwind unlimited scheme and the Gauss limited linear scheme were used to discrete the divergence terms. The linear scheme was used to interpolate values from the cell center to the face center. The limited scheme was used for the surface normal gradient terms. For the pressure correction equation, the relax factor was set to 0.5, while for other equations, the relax factors were set to 0.7. The final residuals of all these equations during iterations were controlled to be smaller than 10−6. For all the simulations, grid sensitivities were implemented to make sure that the simulation results were independent of the grid size.
2.1. The Governing Equations
To mathematically describe the physicochemical processes in the QFBG reactor, governing equations for both the gas phase and solid phase are listed in Table 1 and Table 2.

Table 1.
Governing equations for gas phase.

Table 2.
Governing equations for solid phase.
In Table 1, θg, ρg, ug, Sgm, and t denote the gas volume fraction, the gas density, the gas velocity, mass source, and the residence time, respectively; p, F, g, and τg denote the gas thermodynamic pressure, the interphase momentum transfer rate, the gravitational acceleration and the gas stress tensor; respectively, hg, Tg, and λg denote the gas enthalpy, the gas temperature and the gas mixture thermal conductivity, respectively; ϕ, , , Sh,p, and Sh,p denote the viscous dissipation, enthalpy diffusion, radiation source, homogeneous reaction enthalpy source and heterogeneous reaction enthalpy source, respectively; Yg,i, D, , and denote the mass fraction of species i, the effective mass diffusivity, the net production rate of species i due to homogeneous reactions and the net production rate of species i due to the heterogeneous reactions, respectively; k, ε, and μt are the turbulence kinetic energy, turbulence dissipation rate and the turbulent viscosity, respectively.
In Table 2, A, us, fD, and τD denote the particle acceleration, velocity, the probability distribution function of particle velocity, and the collision damping time, respectively; Ds, θs, ρs, τs, and Fs are the interphase momentum transfer coefficient calculated with Ergun, and Wen and Yu equations, the solids volume fraction, the solid density, the particle normal stress and the particle friction per unit mass; Ps, β, and γ are constants; θcp is the close-packed particle volume fraction; and δ is the Kronecker delta.
2.2. The Reaction Kinetics
The dominant reactions in QFB include the biomass gasification reactions, the CaO/CaCO3 carbonation/calcination reactions, and the Mn2O3/Mn3O4 reduction/oxidation reactions. The biomass gasification reactions include moisture evaporation, dry biomass pyrolysis, and char gasification. The moisture evaporation rate is calculated according to Equation (10). The pyrolysis kinetics is calculated with Equation (11). The other homogeneous and heterogeneous reaction kinetic correlations are listed in Table 3. The CaO carbonation kinetics is calculated by Equation (12) and the CaCO3 calcination kinetics is calculated with Equation (13). The Mn2O3 reduction kinetics is calculated with Equation (14), and the Mn3O4 oxidation kinetics is calculated with Equation (15).
where, ShAB is Sherwood number; dp is the particle diameter; Di,m denotes the vapor diffusion coefficient; psat is the saturation pressure at a specific particle temperature Tp; Xi is the local bulk mole fraction of species i; p is the local absolute pressure; T∞ is the local bulk temperature of the gas; As is the particle surface area; Mw is the H2O molar weight.
where, mdevol is the residual volatile mass; R is the ideal gas constant. Char is assumed to be carbon, and the volatile further decomposed into H2, CO, CO2, and CH4 based on element mass balance.
where, and denote the CO2 partial pressures (atm) at the equilibrium state and at any state, respectively; is the mass flow rate (kg/s) of CaO.
where, xcal denotes the conversion of CaCO3; and CCO2 denote the CO2 concentrations (kmol/m3) at the equilibrium state and at any state.
where, denotes the conversion of Mn2O3.
where, CO2,eq and CO2 denote the oxygen concentrations (mol/m3) at the equilibrium state and at any state.

Table 3.
Dominant reactions and corresponding kinetic correlations.
2.3. Model Validation
Solution Scheme
The new solver was built based on OpenFOAM 3.0.0 [26]. The ReactingMultiphase Cloud and the MPPIC Cloud were combined to form the User-defined ReactingMultiphase MPPIC Cloud so that the four-way interaction could be considered for the reacting parcels. The homogeneous reactions were determined in the constant package of each case. The pyrolysis reactions were determined in the DevolatilisationModel package in the src library. The heterogeneous reactions were determined in the surface ReactionModel package in the src library. Before the final simulation, many types of grids and grid numbers were tested for the simulation of the complex process. To obtain a satisfying solution convergence when solving the equation sets for dense particle flow with chemical reaction, the computational domains of the fluidized bed reactors were meshed with the stairstep scheme to generate the hexahedron cells so that perfect grid orthogonality can be reached. To balance the calculation accuracy and the calculation efficiency, the set of stairstep grids with about 18,000 cells was chosen for the QFBG simulation. The transient time step was set variable to maintain the Courant number not bigger than 0.3. The residuals of the energy equations were restricted below 1 × 10−6, and those of the other equations were restricted below 1 × 10−3 to ensure iteration accuracy.
3. Results and Discussions
3.1. Model Validation
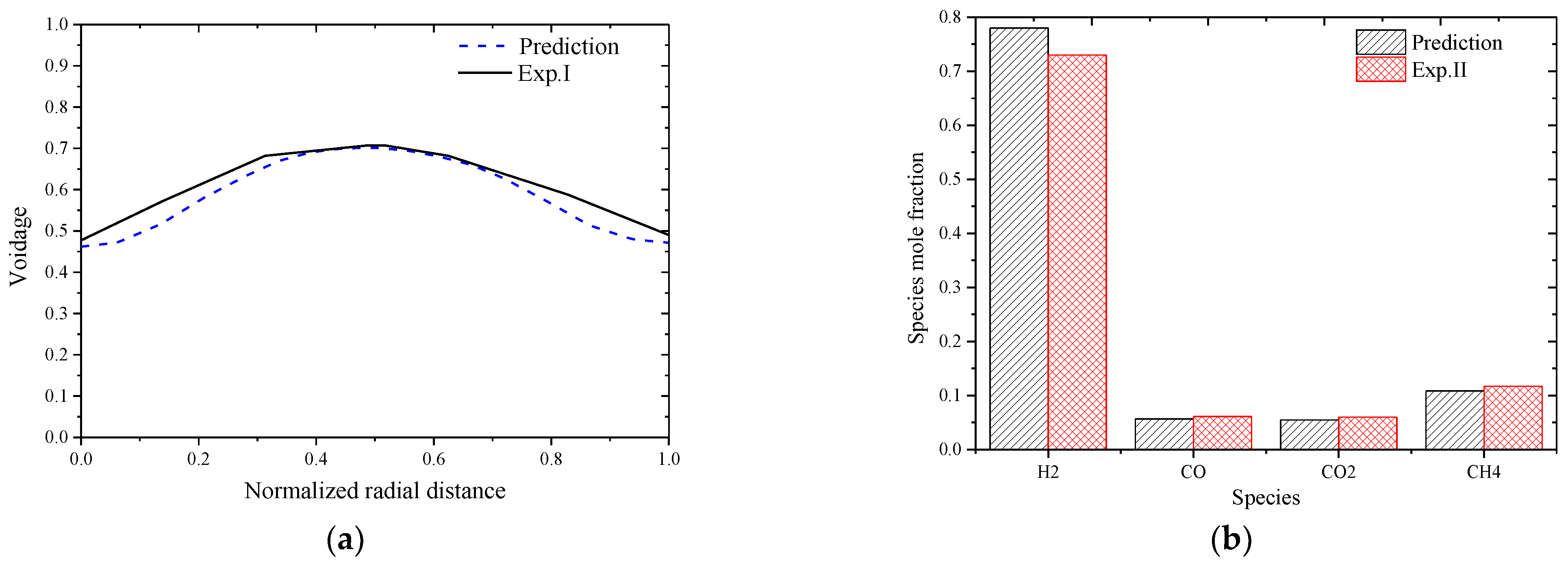
To ensure the rationality of the solver and solution methods, the simulation predictions were first compared with reported experiment data including the voidage distribution in a cold state traveling fluidized bed (Exp. I) [27] and the outlet species molar fractions from a hot state calcium enhanced fluidized bed gasifier (Exp. II) [28]. The operating conditions of the two experimental fluidized beds are listed in Table 4, and the comparison results are shown In Figure 2a,b. The biomass composition is given in Table 5.

Table 4.
Experimental parameters.
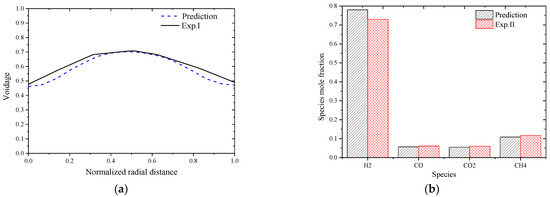
Figure 2.
Comparison between predictions and experimental data. (a) Voidage comparison between prediction and Exp. I; (b) Species molar fraction comparison between prediction and Exp. II.

Table 5.
Analysis of biomass.
From Figure 2, it is seen that good agreement between the model predictions and the reported experimental data can be obtained with the solver and the corresponding solution methods. The errors between prediction and Exp. I data are mainly caused by the simplification of the physical particles into parcels that can include thousands of real particles so as to reduce the computational cost. The errors between model predictions with Exp. II data are mainly caused by the simplification of the biomass steam gasification process into finite global reactions, and the kinetic parameters are general ones and are not specifically generated for the gasification process in this work.
3.2. Predictive Investigation of the QFBG
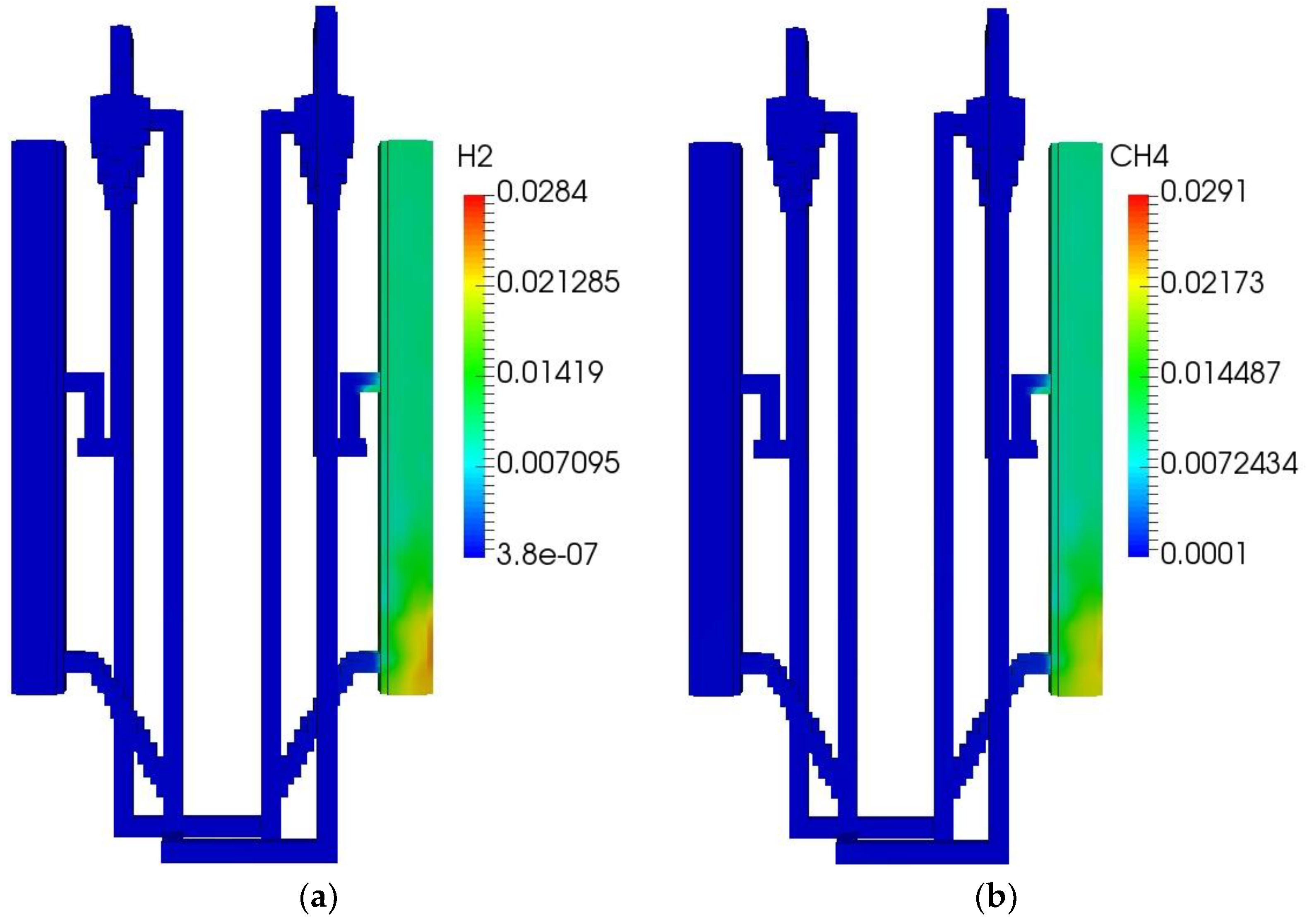
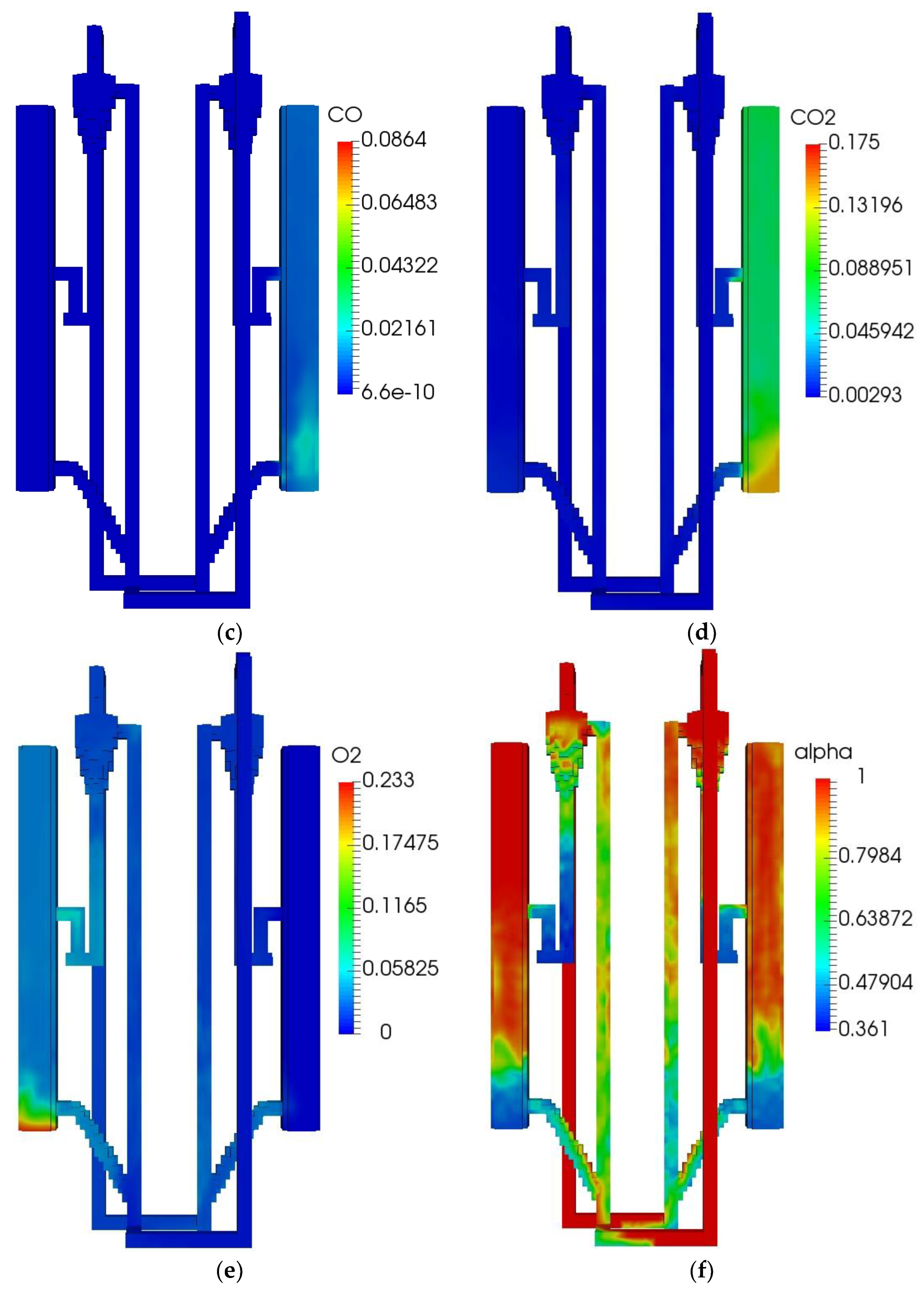
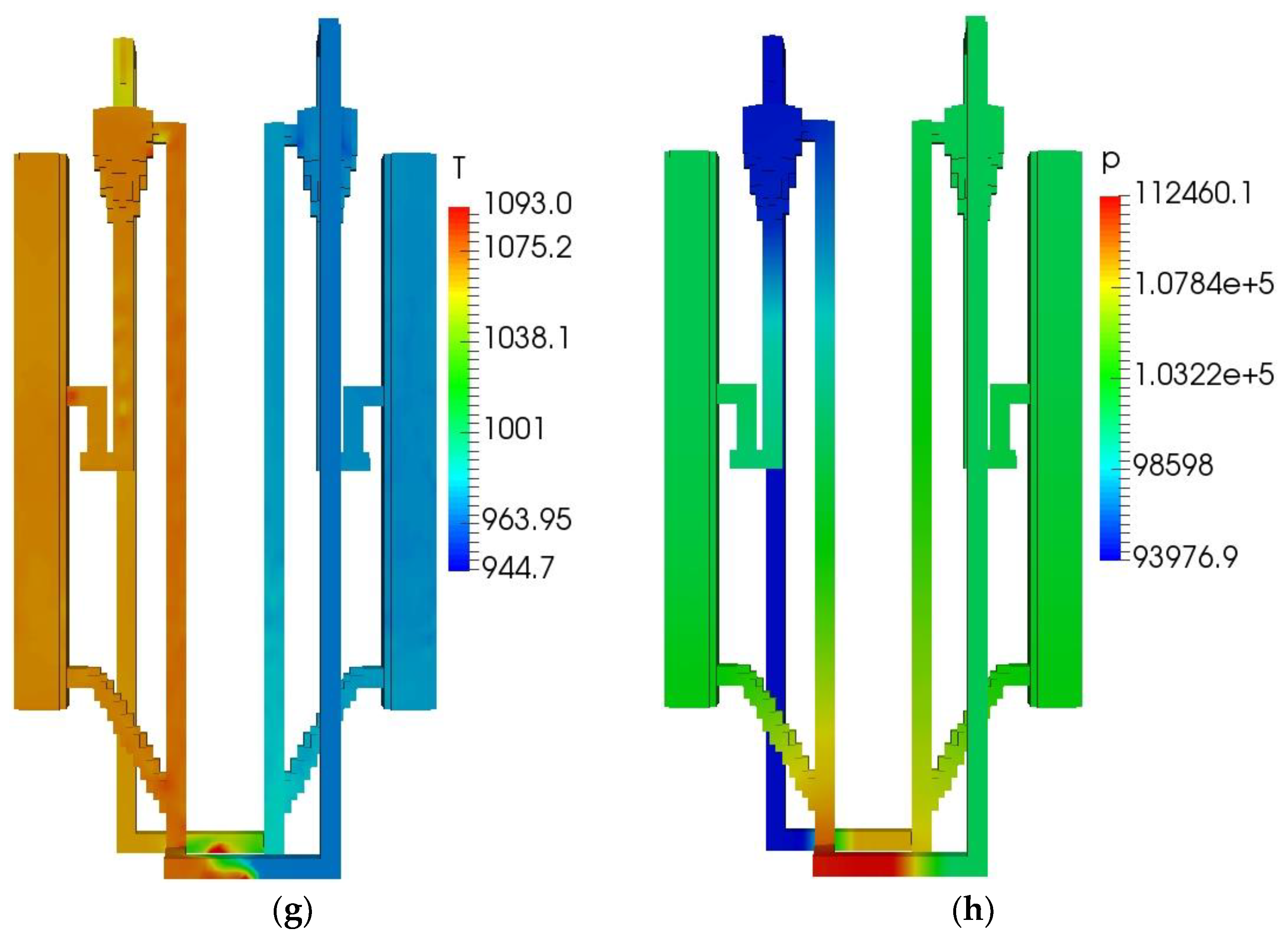
The initial and boundary operating parameters of the QFBG are listed in Table 6. The biomass feed rate is 4.5 kg/h, and its composition is given in Table 5. Under this condition, the operation characteristics of the QFBG are predicted and the key results are depicted in Figure 3a–h.

Table 6.
The initial and boundary operating parameters of the QFBG.
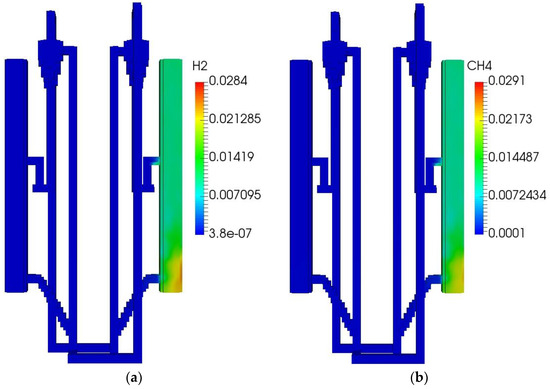
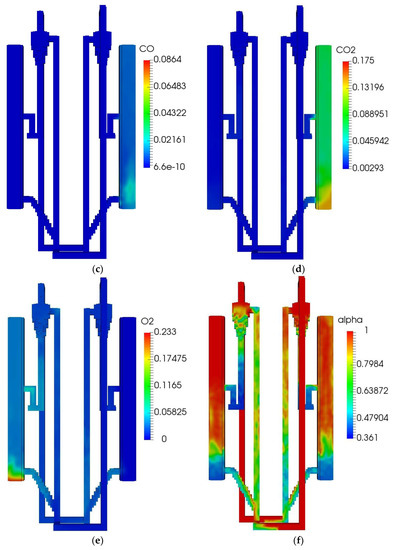
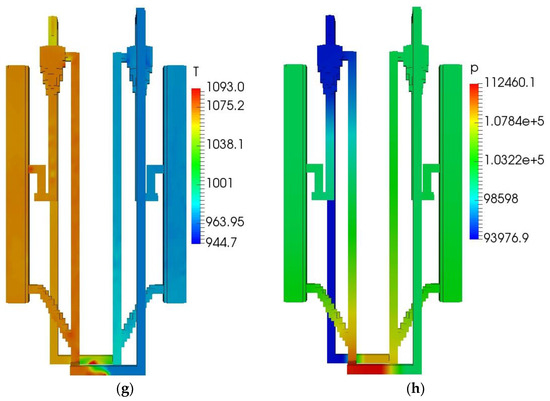
Figure 3.
QFBG operation results. (a) H2 mass fraction; (b) CH4 mass fraction; (c) CO mass fraction; (d) CO2 mass fraction; (e) O2 mass fraction; (f) Voidage distribution; (g) Temperature distribution; (h) Static pressure distribution.
From panels (a–d) of Figure 3, it is seen that the main gas species have reached the gasifier outlet. From the mass fractions of H2, CH4, CO, and CO2, it can be deduced that the mole fractions of these species in dry gas are around 71.6%, 8.3%,3.9%, and 16.2%, respectively. It is noted that the CO2 mole fraction in the produced syngas is still a bit high, this is mainly because the system operates at atmospheric pressure. When the total pressure is 1 atm, the CO2 partial pressure can be quite low because there can be a large amount of steam in the gasifier, leading to the CO2 can only be partially absorbed by CaO. To solve this issue, it is suggested that the system operates at higher pressures. From panel (e) of Figure 3, it is seen that O2 is generated and transported to the FFBC. This is caused by the reduction of Mn2O3 in the FFBR. However, the O2 concentration is still not high and can be increased by increasing the manganese ore circulating flux. From panel (f) of Figure 3, it is seen that the bed materials circulate well in the QFBG, indicating that the system can work reliably. From panel (g) of Figure 3, it is seen that the temperatures in BFBO and FFBR are higher because the chemical looping air separation process with manganese ore as bed material operates at around 1050 K, while the dual bed gasification process with limestone as bed material operates at around 923 K. From panel (h) of Figure 3, it is seen that the highest static pressure appears at the lowest point of QFBG and the pressure drop of chemical looping air separation section is higher. This is because the particles are all fluidized in the connected pipes, and the manganese bed material is heavier.
4. Conclusions
To study of operation characteristics of the quadruple fluidized bed gasifier (QFBG), a new solver based on the multiphase particle in cell scheme is built and accounts for the biomass pyrolysis, char gasification, gases species homogeneous reactions, CaO carbonation, CaCO3 calcination, Mn2O3 reduction, and Mn3O4 oxidation. The solver is then validated against reported experimental data and finally used to predict the QFBG operation property. From this research, the key conclusions can be drawn as follows:
- (1)
- The new solver can give reliable predictions for the complex physicochemical processes in QFBG.
- (2)
- The QFBG works stably and the mole fractions of H2, CH4, CO, and CO2 in dry gas are around 71.6%, 8.3%, 3.9%, and 16.2%, respectively, when the QFBG works at atmospheric pressure.
- (3)
- The QFBG performance can be further increased by increasing the operation pressure and the bed material circulating flux.
Author Contributions
Conceptualization, L.Y. and Z.J.; methodology, L.Y. and Z.J.; software, L.Y. and Z.J.; validation, L.Y. and Z.J.; formal analysis, L.Y. and Z.J.; investigation, Z.J.; resources, L.Y.; data curation, L.Y. and Z.J.; writing—original draft preparation, L.Y., Z.J., Z.W., B.H. and B.F.; writing—review and editing, L.Y. and B.F.; visualization, L.Y. and B.F.; supervision, L.Y.; project administration, L.Y.; funding acquisition, L.Y. All authors have read and agreed to the published version of the manuscript.
Funding
National Natural Science Foundation of China (NSFC, 51706012).
Data Availability Statement
Data will be available upon request from the corresponding authors.
Acknowledgments
The authors gratefully acknowledge financial support from the National Natural Science Foundation of China (NSFC, 51706012) for this work.
Conflicts of Interest
The authors declare no conflict of interest.
Nomenclature
| Parameters | |
| A | particle acceleration, m/s2 |
| Aad | ash mass fraction in air-dried basis |
| As | particle surface area, m2 |
| CO2 concentration, kmol/m3 | |
| CO2 concentration at the equilibrium state, kmol/m3 | |
| Cd | carbon mass fraction in dry basis |
| oxygen concentrations, mol/m3 | |
| oxygen concentration at the equilibrium state, mol/m3 | |
| dp | particle diameter, m |
| D | effective mass diffusivity, m2/s |
| Di,m | vapor diffusion coefficient, m2/s |
| Ds | momentum transfer coefficient, 1/s |
| F | momentum transfer rate, N/m3 |
| FCad | fixed carbon mass fraction in air-dried basis |
| Fs | particle friction per unit mass, N/kg |
| g | gravitational acceleration, m/s2 |
| hg | gas enthalpy, J/kg |
| Hd | hydrogen mass fraction in dry basis |
| k | turbulence kinetic energy, m2/s2 |
| mass flow rate of CaO, kg/s | |
| mdevol | residual volatile mass, kg |
| Mad | moisture mass fraction in air-dried basis |
| Mw | H2O molar weight, kg/mol |
| Nd | nitrogen mass fraction in dry basis |
| Od | oxygen mass fraction in dry basis |
| p | gas pressure, pa |
| CO2 partial pressure, atm | |
| CO2 partial pressure at the equilibrium state, atm | |
| Ps | particle stress coefficient, pa |
| psat | saturation pressure, pa |
| enthalpy diffusion source, W/m3 | |
| radiation source, W/m3 | |
| R | universal gas constant, 8.3145 J/K |
| Sd | sulfur mass fraction in dry basis |
| Sgm | mass source, kg/m3/s |
| Sh | homogeneous reaction enthalpy source, W/m3 |
| ShAB | Sherwood number |
| Sh,p | heterogeneous reaction enthalpy source, W/m3 |
| T | residence time, s |
| Tg | gas temperature, K |
| Tp | particle temperature, K |
| T∞ | bulk temperature of gas, K |
| ug | gas velocity, m/s |
| us | particle velocity, m/s |
| μt | turbulent viscosity, kg/m/s |
| Vad | volatile mass fraction in air-dried basis |
| xcal | conversion of CaCO3 |
| Xi | bulk mole fraction of species i |
| conversion of Mn2O3 | |
| Yg,i | mass fraction of species i |
| Greek symbols | |
| β | coefficient to calculate particle stress |
| γ | coefficient to calculate particle stress |
| δ | Kronecker delta |
| production rate of species i due to homogeneous reactions, kg/m3/s | |
| production rate of species i due to heterogeneous reactions, kg/m3/s | |
| ε | turbulence dissipation rate, m2/s3 |
| θcp | packed particle volume fraction |
| θg | gas volume fraction |
| θs | solids volume fraction |
| λg | thermal conductivity, W/m/K |
| ρg | gas density, kg/m3 |
| ρs | solid density, kg/m3 |
| τD | collision damping time, s |
| τg | gas stress tensor, pa |
| τs | particle normal stress, pa |
| ϕ | viscous dissipation, W/m3 |
| Abbreviations | |
| BFBG | bubbling fluidized bed gasifier |
| BFBO | bubbling fluidized bed oxidation |
| CFD | computational fluid dynamics |
| DFBG | dual fluidized bed gasifier |
| FFBC | fast fluidized bed combustor |
| FFBR | fast fluidized bed reduction |
| GKT | granular kinetic theory |
| LHV | lower heating value |
| MPPIC | multi-phase particle in cell |
| QFBG | quadruple fluidized bed gasifier |
References
- EIMekawy, A.; Hegab, H.M.; Mohanakrishna, G.; Elbaz, A.F.; Bulut, M.; Deepak, P. Technological advances in CO2 conversion electro-biorefinery: A step toward commercialization. Bioresour. Technol. 2016, 215, 357–370. [Google Scholar] [CrossRef] [PubMed]
- Outcomes of the, U.N. Climate Change Conference in Paris. In Proceedings of the 21st Session of the Conference of the Parties to the United Nations Framework Convention on Climate Change (COP 21), Paris, France, 30 November–12 December 2015. [Google Scholar]
- Marcucci, A.; Panos, E.; Kypreos, S.; Fragkos, P. Probabilistic assessment of realizing the 1.5 °C climate target. Appl. Energy 2019, 239, 239–251. [Google Scholar] [CrossRef]
- Tian, X.; Wang, Y.; Zeng, Z.; Dai, L.; Peng, Y.; Jiang, L.; Yang, X.; Yue, L.; Liu, Y.; Ruan, R. Study on the mechanism of co-catalyzed pyrolysis of biomass by potassium and calcium. Bioresour. Technol. 2021, 320, 124415. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.B.; Cao, Y.; Li, X.Z.; He, B.S. Characterization of a dual fluidized bed gasifier with blended biomass/coal. Bioresour. Technol. 2018, 254, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Gunarathne, D.S.; Mellin, P.; Yang, W.; Pettersson, M.; Ljunggren, R. Performance of an effectively integrated biomass multi-stage gasification system and a steel industry heat treatment furnace. Appl. Energy 2016, 170, 353–361. [Google Scholar] [CrossRef]
- Liu, H.; Saffaripour, M.; Mellin, P.; Grip, C.E.; Yang, W.; Blasiak, W. A thermodynamic study of hot syngas impurities in steel reheating furnaces—Corrosion and interaction with oxide scales. Energy 2014, 77, 352–361. [Google Scholar] [CrossRef]
- Yan, L.; Lim, C.J.; Yue, G.; He, B.; Grace, J.R. One-dimensional modeling of a dual fluidized bed for biomass steam gasification. Energy Convers. Manag. 2016, 127, 612–622. [Google Scholar] [CrossRef]
- Yan, L.; He, B. On a clean power generation system with the co-gasification of biomass and coal in a quadruple fluidized bed gasifier. Bioresour. Technol. 2017, 235, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Zhao, Y.; Heberlein, S.; Veksha, A.; Giannis, A.; Chan, W.P.; Lim, T.T.; Lisak, G. Hydrogen and power co-production from autothermal biomass sorption enhanced chemical looping gasification: Thermodynamic modeling and comparative study. Energy Convers. Manag. 2022, 269, 16087. [Google Scholar] [CrossRef]
- Pröll, T.; Hofbauer, H. H2 rich syngas by selective CO2 removal from biomass gasification in a dual fluidized bed system—Process modelling approach. Fuel Process. Technol. 2008, 89, 1207–1217. [Google Scholar] [CrossRef]
- Koppatz, S.; Pfeifer, C.; Rauch, R.; Hofbauer, H.; Marquard-Moellenstedt, T.; Specht, M. H2 rich product gas by steam gasification of biomass with in situ CO2 absorption in a dual fluidized bed system of 8 MW fuel input. Fuel Process. Technol. 2009, 90, 914–921. [Google Scholar] [CrossRef]
- Hejazi, B.; Grace, J.R.; Bi, X.; Mahecha-Botero, A. Steam gasification of biomass coupled with lime-based CO2 capture in a dual fluidized bed reactor: A modeling study. Fuel 2014, 117, 1256–1266. [Google Scholar] [CrossRef]
- Cormos, C. Energy and cost efficient manganese chemical looping air separation cycle for decarbonized power generation based on oxy-fuel combustion and gasification. Energy 2020, 191, 116579. [Google Scholar] [CrossRef]
- Mei, D.; Soleimanisalim, A.H.; Linderholm, C.; Lyngfelt, A.; Mattisson, T. Reactivity and lifetime assessment of an oxygen releasable manganese ore with biomass fuels in a 10 kWth pilot rig for chemical looping combustion. Fuel Process. Technol. 2021, 215, 106743. [Google Scholar] [CrossRef]
- Yan, L.B.; Cao, Y.; Li, X.Z.; He, B.S. On a carbon-negative energy production scheme via a quadruple fluidized bed gasifier. Energy Convers. Manag. 2018, 171, 326–338. [Google Scholar] [CrossRef]
- Yan, L.B.; Cao, Y.; He, B.S. On the kinetic modeling of biomass/coal char co-gasification with steam. Chem. Eng. J. 2018, 331, 435–442. [Google Scholar] [CrossRef]
- Yan, L.; Lim, C.J.; Yue, G.; He, B.; Grace, J.R. Simulation of biomass-steam gasification in fluidized bed reactors: Model setup, comparisons and preliminary predictions. Bioresour. Technol. 2016, 221, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Pissot, S.; Vilches, T.B.; Thunman, V.H.; Seemann, M. Dual Fluidized Bed Gasification Configurations for Carbon Recovery from Biomass. Energy Fuel 2020, 34, 16187–16200. [Google Scholar] [CrossRef]
- Wang, S.; Yan, L.; Zhao, F.; Lu, H.; Sun, L.; Zhang, Q. Numerical simulation of hydrogen production via chemical looping. reforming in interconnected fluidized bed reactor. Ind. Eng. Chem. Res. 2014, 53, 4182–4191. [Google Scholar] [CrossRef]
- Robertson, E.; Choudhury, V.; Bhushan, S.; Walters, D.K. Validation of OpenFOAM numerical methods and turbulence models for incompressible bluff body flows. Comput. Fluids 2015, 123, 122–145. [Google Scholar] [CrossRef]
- Wen, C.Y.; Chaung, T.Z. Entrainment coal gasification modeling. Ind. Eng. Chem. Proc. Des. Dev. 1979, 18, 684–695. [Google Scholar] [CrossRef]
- Ku, X.K.; Li, T.; Løvås, T. CFD–DEM simulation of biomass gasification with steam in a fluidized bed reactor. Chem. Eng. Sci. 2015, 122, 270–283. [Google Scholar] [CrossRef]
- Gómez-Barea, A.; Leckner, B. Modeling of biomass gasification in fluidized bed. Prog. Energy Combust. Sci. 2010, 36, 444–509. [Google Scholar] [CrossRef]
- Lanoye, L.; Vierendeels, J.; Segers, P.; Verdonck, P. Vascular fluid-structure-interaction using Fluent and Abaqus software. J. Biomech. 2006, 39, S440. [Google Scholar] [CrossRef]
- OpenFOAM. The Open Source CFD Toolbox, Ver. 3.0.0; ESI Group: Paris, France, 2015. [Google Scholar]
- Dubrawski, K.; Tebianian, S.; Bi, H.T.; Chaouki, J.; Ellis, N.; Gerspacher, R.; Jafari, R.; Kantzas, A.; Lim, C.; Patience, G.S.; et al. Traveling column for comparison of invasive and non-invasive fluidization voidage measurement techniques. Powder Technol. 2013, 235, 203–220. [Google Scholar] [CrossRef]
- Pfeifer, C.; Puchner, B.; Hofbauer, H. Comparison of dual fluidized bed steam gasification of biomass with and without selective transport of CO2. Chem. Eng. Sci. 2009, 64, 5073–5083. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).